Generation of Hepatobiliary Cell Lineages from Human Induced Pluripotent Stem Cells: Applications in Disease Modeling and Drug Screening
Abstract
1. Introduction
2. Generation of Hepatocytes and Cholangiocytes from Human Induced Pluripotent Stem Cells and Their Applications
2.1. Introduction to iPSC Technology and Lessons from Development for iPSC Differentiation into the Hepatoblast
2.2. Generation of Hepatocyte-Like Cells and Applications in Disease Modeling and Drug Testing
2.3. Generation of Cholangiocyte-Like Cells and Applications in Disease Modeling and Drug Testing
3. Toward Improved Human iPSC-Based Hepatobiliary Models
4. Concluding Remarks and Open Questions
Author Contributions
Funding
Conflicts of Interest
References
- Chiang, J. Liver Physiology: Metabolism and Detoxification; Linda, M., McManus, R.N.M., Eds.; Elsevier Inc.: San Diego, CA, USA, 2014. [Google Scholar]
- Yoo, K.S.; Lim, W.T.; Choi, H.S. Biology of Cholangiocytes: From Bench to Bedside. Gut Liver 2016, 10, 687. [Google Scholar] [CrossRef]
- Friedman, J.R.; Kaestner, K.H. On the origin of the liver. J. Clin. Investig. 2011, 121, 4630–4633. [Google Scholar] [CrossRef]
- Tanimizu, N.; Miyajima, A. Notch signaling controls hepatoblast differentiation by altering the expression of liver-enriched transcription factors. J. Cell Sci. 2004, 117, 3165–3174. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.R.; Lange, A.W.; Lin, S.C.J.; Kaestner, K.H.; Lowy, A.M.; Kim, I.; Whitsett, J.A.; Wells, J.M. Sox17 regulates organ lineage segregation of ventral foregut progenitor cells. Dev. Cell 2009, 17, 62–74. [Google Scholar] [CrossRef]
- Si-Tayeb, K.; Lemaigre, F.P.; Duncan, S.A. Organogenesis and development of the liver. Dev. Cell 2010, 18, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Michalopoulos, G.K. The regenerative altruism of hepatocytes and cholangiocytes. Stem Cell 2018, 23, 11–12. [Google Scholar] [CrossRef]
- Haruna, Y.; Saito, K.; Spaulding, S.; Nalesnik, M.A.; Gerber, M.A. Identification of bipotential progenitor cells in human liver development. Hepatology 1996, 23, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Crosby, H.A.; Hubscher, S.; Fabris, L.; Joplin, R.; Sell, S.; Kelly, D.; Strain, A.J. Immunolocalization of putative human liver progenitor cells in livers from patients with end-stage primary biliary cirrhosis and sclerosing cholangitis using the monoclonal antibody OV-6. Am. J. Pathol. 1998, 152, 771–779. [Google Scholar]
- Theise, N.D.; Saxena, R.; Portmann, B.C.; Thung, S.N.; Yee, H.; Chiriboga, L.; Ashok, A.; Crawford, J.M. The canals of Hering and hepatic stem cells in humans. Hepatology 1999, 30, 1425–1433. [Google Scholar] [CrossRef]
- Limaye, P.B.; Alarcón, G.; Walls, A.L.; Nalesnik, M.A.; Michalopoulos, G.K.; Demetris, A.J.; Ochoa, E.R. Expression of specific hepatocyte and cholangiocyte transcription factors in human liver disease and embryonic development. Lab. Investig. 2009, 88, 865–872. [Google Scholar] [CrossRef]
- Fausto, N.; Campbell, J.S. The role of hepatocytes and oval cells in liver regeneration and repopulation. Mech. Dev. 2003, 120, 117–130. [Google Scholar] [CrossRef]
- Fausto, N. Liver regeneration and repair: Hepatocytes, progenitor cells, and stem cells. Hepatology 2004, 39, 1477–1487. [Google Scholar] [CrossRef]
- Lu, W.Y.; Bird, T.G.; Boulter, L.; Tsuchiya, A.; Cole, A.M.; Hay, T.; Guest, R.V.; Wojtacha, D.; Man, T.Y.; Mackinnon, A.; et al. Hepatic progenitor cells of biliary origin with liver repopulation capacity. Nat. Cell Biol. 2015, 17, 971–983. [Google Scholar] [CrossRef] [PubMed]
- Lukacs-Kornek, V.; Lammert, F. The progenitor cell dilemma: Cellular and functional heterogeneity in assistance or escalation of liver injury. J. Hepatol. 2017, 66, 619–630. [Google Scholar] [CrossRef]
- Falkowski, O.; An, H.J.; Ianus, I.A.; Chiriboga, L.; Yee, H.; West, A.B.; Theise, N.D. Regeneration of hepatocyte ‘buds’ in cirrhosis from intrabiliary stem cells. J. Hepatol. 2003, 39, 357–364. [Google Scholar] [CrossRef]
- Fukuda, K.; Sugihara, A.; Nakasho, K.; Tsujimura, T.; Yamada, N.; Okaya, A.; Sakagami, M.; Terada, N. The origin of biliary ductular cells that appear in the spleen after transplantation of hepatocytes. Cell Transplant. 2004, 13, 27–33. [Google Scholar] [CrossRef]
- Michalopoulos, G.K.; Barua, L.; Bowen, W.C. Transdifferentiation of rat hepatocytes into biliary cells after bile duct ligation and toxic biliary injury. Hepatology 2005, 41, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Hata, M.; Terada, N.; Ueda, H.; Yamada, N.; Yamanegi, K.; Ohyama, H.; Kakihana, M.; Okamura, H.; Nakasho, K. Transdifferentiation into biliary ductular cells of hepatocytes transplanted into the spleen. Pathology 2008, 40, 272–276. [Google Scholar] [CrossRef]
- Yanger, K.; Zong, Y.; Maggs, L.R.; Shapira, S.N.; Maddipati, R.; Aiello, N.M.; Thung, S.N.; Wells, R.G.; Greenbaum, L.E.; Stanger, B.Z. Robust cellular reprogramming occurs spontaneously during liver regeneration. Genes Dev. 2013, 27, 719–724. [Google Scholar] [CrossRef]
- Schaub, J.R.; Huppert, K.A.; Kurial, S.N.; Hsu, B.Y.; Cast, A.E.; Donnelly, B.; Karns, R.A.; Chen, F.; Rezvani, M.; Luu, H.Y.; et al. De novo formation of the biliary system by TGFβ-mediated hepatocyte transdifferentiation. Nature 2018, 557, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Sekiya, S.; Suzuki, A. Hepatocytes, rather than cholangiocytes, can be the major source of primitive ductules in the chronically injured mouse liver. Am. J. Pathol. 2014, 184, 1468–1478. [Google Scholar] [CrossRef]
- Yanger, K.; Knigin, D.; Zong, Y.; Maggs, L.; Gu, G.; Akiyama, H.; Pikarsky, E.; Stanger, B.Z. Adult hepatocytes are generated by self-duplication rather than stem cell differentiation. Cell Stem Cell 2014, 15, 340–349. [Google Scholar] [CrossRef]
- Raven, A.; Lu, W.Y.; Man, T.Y.; Ferreira-Gonzalez, S.; O’Duibhir, E.; Dwyer, B.J.; Thomson, J.P.; Meehan, R.R.; Bogorad, R.; Koteliansky, V.; et al. Cholangiocytes act as facultative liver stem cells during impaired hepatocyte regeneration. Nature 2017, 547, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Zhang, X.; Li, W.; Feng, R.X.; Li, L.; Yi, G.R.; Zhang, X.N.; Yin, C.; Yu, H.Y.; Zhang, J.P.; et al. Chronic liver injury induces conversion of biliary epithelial cells into hepatocytes. Cell Stem Cell 2018, 23, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Alison, M.R. Cholangiocytes: No longer cinderellas to the hepatic regenerative response. Cell Stem Cell 2017, 21, 159–160. [Google Scholar] [CrossRef]
- Lanzoni, G.; Cardinale, V.; Carpino, G. The hepatic, biliary, and pancreatic network of stem/progenitor cell niches in humans: A new reference frame for disease and regeneration. Hepatology 2016, 64, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Cardinale, V.; Wang, Y.; Carpino, G.; Alvaro, D.; Reid, L.; Gaudio, E. Multipotent stem cells in the biliary tree. Ital. J. Anat. Embryol. 2010, 115, 85. [Google Scholar] [PubMed]
- Cardinale, V.; Wang, Y.; Carpino, G.; Mendel, G.; Alpini, G.; Gaudio, E.; Reid, L.M.; Alvaro, D. The biliary tree—a reservoir of multipotent stem cells. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 231. [Google Scholar] [CrossRef] [PubMed]
- Kochhar, G.; Parungao, J.M.; Hanouneh, I.A.; Parsi, M.A. Biliary complications following liver transplantation. World J. Gastroenterol. 2013, 19, 2841. [Google Scholar] [CrossRef] [PubMed]
- Chamuleau, R.A.; Poyck, P.P.; Van De Kerkhove, M.P. Bioartificial liver: Its pros and cons. Ther. Apher. Dial. 2006, 10, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Secunda, R.; Vennila, R.; Mohanashankar, A.M.; Rajasundari, M.; Jeswanth, S.; Surendran, R. Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: A comparative study. Cytotechnology 2015, 67, 793–807. [Google Scholar] [CrossRef] [PubMed]
- Ullah, I.; Subbarao, R.B.; Rho, G.J. Human mesenchymal stem cells-current trends and future prospective. Biosci. Rep. 2015, 35, e00191. [Google Scholar] [CrossRef]
- Francipane, M.G.; Cervello, M.; Vizzini, G.B.; Pietrosi, G.; Montalto, G. Management of liver failure: From transplantation to cell-based therapy. Cell Med. 2011, 2, 9–26. [Google Scholar] [CrossRef][Green Version]
- Liang, X.; Ding, Y.; Zhang, Y.; Tse, H.F.; Lian, Q. Paracrine mechanisms of mesenchymal stem cell-based therapy: Current status and perspectives. Cell Transplant. 2014, 23, 1045–1059. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Fiorotto, R.; Amenduni, M.; Mariotti, V.; Fabris, L.; Spirli, C.; Strazzabosco, M. Liver diseases in the dish: iPSC and organoids as a new approach to modeling liver diseases. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2019, 1865, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Huch, M.; Koo, B.K. Modeling mouse and human development using organoid cultures. Development 2015, 142, 3113–3125. [Google Scholar] [CrossRef] [PubMed]
- Francipane, M.G.; Lagasse, E. Toward organs on demand: Breakthroughs and challenges in models of organogenesis. Curr. Pathobiol. Rep. 2016, 4, 77–85. [Google Scholar] [CrossRef][Green Version]
- Zhou, Y.Y.; Zeng, F. Integration-free methods for generating induced pluripotent stem cells. Genomics. Proteom. Bioinform. 2013, 11, 284–287. [Google Scholar] [CrossRef]
- Liu, G.; David, B.T.; Trawczynski, M.; Fessler, R.G. Advances in pluripotent stem cells: History, mechanisms, technologies, and applications. Stem Cell Rev. Rep. 2020, 16, 3–32. [Google Scholar] [CrossRef]
- Ghaedi, M.; Niklason, L.E. Human pluripotent stem cells (iPSC) generation, culture, and differentiation to lung progenitor cells. Methods Mol. Biol. 2019, 1576, 55–92. [Google Scholar] [PubMed]
- Lin, T.; Wu, S. Reprogramming with small molecules instead of exogenous transcription factors. Stem Cells Int. 2015, 2015, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Siller, R.; Greenhough, S.; Naumovska, E.; Sullivan, G.J. Small-molecule-driven hepatocyte differentiation of human pluripotent stem cells. Stem Cell Rep. 2015, 4, 939–952. [Google Scholar] [CrossRef]
- Du, C.; Feng, Y.; Qiu, D.; Xu, Y.; Pang, M.; Cai, N.; Xiang, A.P.; Zhang, Q. Highly efficient and expedited hepatic differentiation from human pluripotent stem cells by pure small-molecule cocktails. Stem Cell Res. Ther. 2018, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ben-David, U.; Benvenisty, N. The tumorigenicity of human embryonic and induced pluripotent stem cells. Nat. Rev. Cancer 2011, 11, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Hannoun, Z.; Steichen, C.; Dianat, N.; Weber, A.; Dubart-Kupperschmitt, A. The potential of induced pluripotent stem cell derived hepatocytes. J. Hepatol. 2016, 65, 182–199. [Google Scholar] [CrossRef]
- Song, Z.; Cai, J.; Liu, Y.; Zhao, D.; Yong, J.; Duo, S.; Song, S.; Guo, Y.; Zhao, Y.; Qin, H.; et al. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 2009, 19, 1233–1242. [Google Scholar] [CrossRef] [PubMed]
- Dianat, N.; Dubois-Pot-Schneider, H.; Steichen, C.; Desterke, C.; Leclerc, P.; Raveux, A.; Combettes, L.; Weber, A.; Corlu, A.; Dubart-Kupperschmitt, A. Generation of functional cholangiocyte-like cells from human pluripotent stem cells and HepaRG cells. Hepatology 2014, 60, 700–714. [Google Scholar] [CrossRef]
- Zhou, X.; Sasaki, H.; Lowe, L.; Hogan, B.L.; Kuehn, M.R. Nodal is a novel TGF-β-like gene expressed in the mouse node during gastrulation. Nature 1993, 361, 543–547. [Google Scholar] [CrossRef]
- Conlon, F.L.; Lyons, K.M.; Takaesu, N.; Barth, K.S.; Kispert, A.; Herrmann, B.; Robertson, E.J. A primary requirement for nodal in the formation and maintenance of the primitive streak in the mouse. Development 1994, 120, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Whitman, M. Nodal signaling in early vertebrate embryos: Themes and variations. Dev. Cell 2001, 1, 605–617. [Google Scholar] [CrossRef]
- D’Amour, K.A.; Agulnick, A.D.; Eliazer, S.; Kelly, O.G.; Kroon, E.; Baetge, E.E. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat. Biotechnol. 2005, 23, 1534–1541. [Google Scholar] [CrossRef]
- McLean, A.B.; D’Amour, K.A.; Jones, K.L.; Krishnamoorthy, M.; Kulik, M.J.; Reynolds, D.M.; Sheppard, A.M.; Liu, H.; Xu, Y.; Baetge, E.E.; et al. Activin a efficiently specifies definitive endoderm from human embryonic stem cells only when phosphatidylinositol 3-kinase signaling is suppressed. Stem Cells 2007, 25, 29–38. [Google Scholar] [CrossRef]
- Pauklin, S.; Vallier, L. Activin/Nodal signalling in stem cells. Development 2015, 142, 607–619. [Google Scholar] [CrossRef]
- Phillips, B.W.; Hentze, H.; Rust, W.L.; Chen, Q.P.; Chipperfield, H.; Tan, E.K.; Abraham, S.; Sadasivam, A.; Soong, P.L.; Wang, S.T.; et al. Directed differentiation of human embryonic stem cells into the pancreatic endocrine lineage. Stem Cells Dev. 2007, 16, 561–578. [Google Scholar] [CrossRef]
- Touboul, T.; Hannan, N.R.; Corbineau, S.; Martinez, A.; Martinet, C.; Branchereau, S.; Mainot, S.; Strick-Marchand, H.; Pedersen, R.; Di Santo, J.; et al. Generation of functional hepatocytes from human embryonic stem cells under chemically defined conditions that recapitulate liver development. Hepatology 2010, 51, 1754–1765. [Google Scholar] [CrossRef]
- Teo, A.K.K.; Tsuneyoshi, N.; Hoon, S.; Tan, E.K.; Stanton, L.W.; Wright, C.V.; Dunn, N.R. PDX1 binds and represses hepatic genes to ensure robust pancreatic commitment in differentiating human embryonic stem cells. Stem Cell Rep. 2015, 4, 578–590. [Google Scholar] [CrossRef]
- Soto-Gutierrez, A.; Navarro-Alvarez, N.; Rivas-Carrillo, J.D.; Chen, Y.; Yamatsuji, T.; Tanaka, N.; Kobayashi, N. Differentiation of human embryonic stem cells to hepatocytes using deleted variant of HGF and poly-amino-urethane-coated nonwoven polytetrafluoroethylene fabric. Cell Transplant. 2006, 15, 335–342. [Google Scholar] [CrossRef]
- Pan, T.; Tao, J.; Chen, Y.; Zhang, J.; Getachew, A.; Zhuang, Y.; Wang, N.; Xu, Y.; Tan, S.; Fang, J.; et al. Robust expansion and functional maturation of human hepatoblasts by chemical strategy. Stem Cell Res. Ther. 2021, 12, 1–13. [Google Scholar] [CrossRef]
- Geisler, F.; Nagl, F.; Mazur, P.K.; Lee, M.; Zimber-Strobl, U.; Strobl, L.J.; Radtke, F.; Schmid, R.M.; Siveke, J.T. Liver-specific inactivation of Notch2, but not Notch1, compromises intrahepatic bile duct development in mice. Hepatology 2008, 48, 607–616. [Google Scholar] [CrossRef]
- Piccoli, D.A.; Spinner, N.B. Alagille syndrome and the Jagged1 gene. Semin. Liver Dis. 2001, 21, 525–534. [Google Scholar] [CrossRef]
- Sumazaki, R.; Shiojiri, N.; Isoyama, S.; Masu, M.; Keino-Masu, K.; Osawa, M.; Nakauchi, H.; Kageyama, R.; Matsui, A. Conversion of biliary system to pancreatic tissue in Hes1-deficient mice. Nat. Genet. 2004, 36, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Sampaziotis, F.; De Brito, M.C.; Madrigal, P.; Bertero, A.; Saeb-Parsy, K.; Soares, F.A.C.; Schrumpf, E.; Melum, E.; Karlsen, T.H.; Bradley, J.A.; et al. Cholangiocytes derived from human induced pluripotent stem cells for disease modeling and drug validation. Nat. Biotechnol. 2015, 33, 845–852. [Google Scholar] [CrossRef]
- Si-Tayeb, K.; Noto, F.K.; Nagaoka, M.; Li, J.; Battle, M.A.; Duris, C.; North, P.E.; Dalton, S.; Duncan, S.A. Highly efficient generation of human hepatocyte–like cells from induced pluripotent stem cells. Hepatology 2010, 51, 297–305. [Google Scholar] [CrossRef]
- Ao, Y.; Mich-Basso, J.D.; Lin, B.; Yang, L. High efficient differentiation of functional hepatocytes from porcine induced pluripotent stem cells. PLoS ONE 2014, 9, e100417. [Google Scholar] [CrossRef]
- Li, S.; Huang, S.Q.; Zhao, Y.X.; Ding, Y.J.; Ma, D.J.; Ding, Q.R. Derivation and applications of human hepatocyte-like cells. World J. Stem Cells 2019, 11, 535. [Google Scholar] [CrossRef]
- Lu, J.; Einhorn, S.; Venkatarangan, L.; Miller, M.; Mann, D.A.; Watkins, P.B.; LeCluyse, E. Morphological and functional characterization and assessment of iPSC-derived hepatocytes for in vitro toxicity testing. Toxicol. Sci. 2015, 147, 39–54. [Google Scholar] [CrossRef]
- Guengerich, F.P. Cytochrome P-450 3A4: Regulation and role in drug metabolism. Annu. Rev. Pharmacol. Toxicol. 1999, 39, 1–17. [Google Scholar] [CrossRef]
- Ulvestad, M.; Nordell, P.; Asplund, A.; Rehnström, M.; Jacobsson, S.; Holmgren, G.; Davidson, L.; Brolén, G.; Edsbagge, J.; Björquist, P.; et al. Drug metabolizing enzyme and transporter protein profiles of hepatocytes derived from human embryonic and induced pluripotent stem cells. Biochem. Pharmacol. 2013, 86, 691–702. [Google Scholar] [CrossRef]
- Medine, C.N.; Lucendo-Villarin, B.; Storck, C.; Wang, F.; Szkolnicka, D.; Khan, F.; Pernagallo, S.; Black, J.R.; Marriage, H.M.; Ross, J.A.; et al. Developing high-fidelity hepatotoxicity models from pluripotent stem cells. Stem Cells Transl. Med. 2013, 2, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Holmgren, G.; Sjögren, A.K.; Barragan, I.; Sabirsh, A.; Sartipy, P.; Synnergren, J.; Björquist, P.; Ingelman-Sundberg, M.; Andersson, T.B.; Edsbagge, J. Long-term chronic toxicity testing using human pluripotent stem cell–derived hepatocytes. Drug Metab. Dispos. 2014, 42, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Ware, B.R.; Berger, D.R.; Khetani, S.R. Prediction of drug-induced liver injury in micropatterned co-cultures containing iPSC-derived human hepatocytes. Toxicol. Sci. 2015, 145, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Berger, D.R.; Ware, B.R.; Davidson, M.D.; Allsup, S.R.; Khetani, S.R. Enhancing the functional maturity of induced pluripotent stem cell–derived human hepatocytes by controlled presentation of cell–cell interactions in vitro. Hepatology 2014, 61, 1370–1381. [Google Scholar] [CrossRef] [PubMed]
- Baxter, M.; Withey, S.; Harrison, S.; Segeritz, C.P.; Zhang, F.; Atkinson-Dell, R.; Rowe, C.; Gerrard, D.; Sison-Young, R.; Jenkins, R.; et al. Phenotypic and functional analyses show stem cell-derived hepatocyte-like cells better mimic fetal rather than adult hepatocytes. J. Hepatol. 2015, 62, 581–589. [Google Scholar] [CrossRef]
- Gao, X.; Liu, Y. A transcriptomic study suggesting human iPSC-derived hepatocytes potentially offer a better in vitro model of hepatotoxicity than most hepatoma cell lines. Cell Biol. Toxicol. 2017, 33, 407–421. [Google Scholar] [CrossRef]
- Cayo, M.A.; Cai, J.; DeLaForest, A.; Noto, F.K.; Nagaoka, M.; Clark, B.S.; Collery, R.F.; Si-Tayeb, K.; Duncan, S.A. JD induced pluripotent stem cell–derived hepatocytes faithfully recapitulate the pathophysiology of familial hypercholesterolemia. Hepatology 2012, 56, 2163–2171. [Google Scholar] [CrossRef]
- Gurevich, I.; Burton, S.A.; Munn, C.; Ohshima, M.; Goedland, M.E.; Czysz, K.; Rajesh, D. iPSC-derived hepatocytes generated from NASH donors provide a valuable platform for disease modeling and drug discovery. Biol. Open 2020, 9, 055087. [Google Scholar]
- Ramli, M.N.B.; Lim, Y.S.; Koe, C.T.; Demircioglu, D.; Tng, W.; Gonzales, K.A.U.; Tan, C.P.; Szczerbinska, I.; Liang, H.; Soe, E.L.; et al. Human pluripotent stem cell-derived organoids as models of liver disease. Gastroenterology 2020, 159, 1471–1486.e12. [Google Scholar] [CrossRef]
- Overeem, A.W.; Klappe, K.; Parisi, S.; Klöters-Planchy, P.; Mataković, L.; du Teil Espina, M.; Drouin, C.A.; Weiss, K.H.; van IJzendoorn, S.C. Pluripotent stem cell-derived bile canaliculi-forming hepatocytes to study genetic liver diseases involving hepatocyte polarity. J. Hepatol. 2019, 71, 344–356. [Google Scholar] [CrossRef]
- Yusa, K.; Rashid, S.T.; Strick-Marchand, H.; Varela, I.; Liu, P.Q.; Paschon, D.E.; Miranda, E.; Ordóñez, A.; Hannan, N.; Rouhani, F.J.; et al. Targeted gene correction of α 1-antitrypsin deficiency in induced pluripotent stem cells. Nature 2011, 478, 391–394. [Google Scholar] [CrossRef]
- Choi, S.M.; Kim, Y.; Shim, J.S.; Park, J.T.; Wang, R.H.; Leach, S.D.; Liu, J.O.; Deng, C.X.; Ye, Z.; Jang, Y.Y. Efficient drug screening and gene correction for treating liver disease using patient-specific stem cells. Hepatology 2013, 57, 2458–2468. [Google Scholar] [CrossRef]
- Omer, L.; Hudson, E.A.; Zheng, S.; Hoying, J.B.; Shan, Y.; Boyd, N.L. CRISPR correction of a homozygous low-density lipoprotein receptor mutation in familial hypercholesterolemia induced pluripotent stem cells. Hepatol. Commun. 2017, 1, 886–898. [Google Scholar] [CrossRef]
- Wu, X.; Robotham, J.M.; Lee, E.; Dalton, S.; Kneteman, N.M.; Gilbert, D.M.; Tang, H. Productive hepatitis C virus infection of stem cell-derived hepatocytes reveals a critical transition to viral permissiveness during differentiation. PLoS Pathog. 2012, 8, e1002617. [Google Scholar] [CrossRef]
- Schwartz, R.E.; Trehan, K.; Andrus, L.; Sheahan, T.P.; Ploss, A.; Duncan, S.A.; Rice, C.M.; Bhatia, S.N. Modeling hepatitis C virus infection using human induced pluripotent stem cells. Proc. Natl. Acad. Sci. USA 2011, 109, 2544–2548. [Google Scholar] [CrossRef]
- Shlomai, A.; Schwartz, R.E.; Ramanan, V.; Bhatta, A.; de Jong, Y.P.; Bhatia, S.N.; Rice, C.M. Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc. Natl. Acad. Sci. USA 2014, 111, 12193–12198. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, A.; Tesfaye, A.; Chu, V.; Nimgaonkar, I.; Zhang, F.; Lee, S.B.; Thorgeirsson, S.S.; Feinstone, S.M.; Liang, T.J. Engrafted human stem cell–derived hepatocytes establish an infectious HCV murine model. J. Clin. Investig. 2014, 124, 4953–4964. [Google Scholar] [CrossRef]
- Chen, S.W.; Himeno, M.; Koui, Y.; Sugiyama, M.; Nishitsuji, H.; Mizokami, M.; Shimotohno, K.; Miyajima, A.; Kido, T. Modulation of hepatitis B virus infection by epidermal growth factor secreted from liver sinusoidal endothelial cells. Sci. Rep. 2020, 10, 1–10. [Google Scholar]
- Raynaud, P.; Carpentier, R.; Antoniou, A.; Lemaigre, F.P. Biliary differentiation and bile duct morphogenesis in development and disease. Int. J. Biochem. Cell Biol. 2011, 43, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.; Kawabata, K.; Nagamoto, Y.; Inamura, M.; Ohashi, K.; Okuno, H.; Yamaguchi, T.; Tashiro, K.; Sakurai, F.; Hayakawa, T.; et al. CCAAT/enhancer binding protein-mediated regulation of TGFβ receptor 2 expression determines the hepatoblast fate decision. Development 2014, 141, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Takeishi, K.; de l’Hortet, A.C.; Wang, Y.; Handa, K.; Guzman-Lepe, J.; Matsubara, K.; Morita, K.; Jang, S.; Haep, N.; Florentino, R.M.; et al. Assembly and function of a bioengineered human liver for transplantation generated solely from induced pluripotent stem cells. Cell Rep. 2020, 31, 107711. [Google Scholar] [CrossRef]
- De Assuncao, T.M.; Sun, Y.; Jalan-Sakrikar, N.; Drinane, M.C.; Huang, B.Q.; Li, Y.; Davila, J.I.; Wang, R.; O’Hara, S.P.; Lomberk, G.A.; et al. Development and characterization of human-induced pluripotent stem cell-derived cholangiocytes. Lab. Investig. 2015, 95, 684–696. [Google Scholar] [CrossRef]
- Ogawa, M.; Ogawa, S.; Bear, C.E.; Ahmadi, S.; Chin, S.; Li, B.; Grompe, M.; Keller, G.; Kamath, B.M.; Ghanekar, A. Directed differentiation of cholangiocytes from human pluripotent stem cells. Nat. Biotechnol. 2015, 33, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.M.; Chen, C.S. Deconstructing the third dimension–how 3D culture microenvironments alter cellular cues. J. Cell Sci. 2012, 125, 3015–3024. [Google Scholar] [CrossRef]
- Birgersdotter, A.; Sandberg, R.; Ernberg, I. Gene expression perturbation in vitro—a growing case for three-dimensional (3D) culture systems. Semin. Cancer Biol. 2005, 15, 405–412. [Google Scholar] [CrossRef]
- Lee, K.H.; Lee, J.; Lee, S.H. 3D liver models on a microplatform: Well-defined culture, engineering of liver tissue and liver-on-a-chip. Lab Chip 2015, 15, 3822–3837. [Google Scholar]
- Caliari, S.R.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Di Gesù, R.; Amato, G.; Gottardi, R. Electrospun scaffolds in tendons regeneration: A review. Muscles. Ligaments Tendons J. 2019, 9, 478–493. [Google Scholar] [CrossRef]
- Miranda, J.P.; Rodrigues, A.; Tostoes, R.M.; Leite, S.; Zimmerman, H.; Carrondo, M.J.; Alves, P.M. Extending hepatocyte functionality for drug-testing applications using high-viscosity alginate–encapsulated three-dimensional cultures in bioreactors. Tissue Eng. Part C Methods 2010, 16, 1223–1232. [Google Scholar] [CrossRef]
- Pasqua, M.; Pereira, U.; Messina, A.; de Lartigue, C.; Vigneron, P.; Dubart-Kupperschmitt, A.; Legallais, C. HepaRG self-assembled spheroids in alginate beads meet the clinical needs for bioartificial liver. Tissue Eng. Part A 2020, 26, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Rebelo, S.P.; Costa, R.; Estrada, M.; Shevchenko, V.; Brito, C.; Alves, P.M. HepaRG microencapsulated spheroids in DMSO-free culture: Novel culturing approaches for enhanced xenobiotic and biosynthetic metabolism. Arch. Toxicol. 2015, 89, 1347–1358. [Google Scholar] [CrossRef]
- Ye, S.; Boeter, J.W.; Mihajlovic, M.; van Steenbeek, F.G.; van Wolferen, M.E.; Oosterhoff, L.A.; Marsee, A.; Caiazzo, M.; van der Laan, L.J.W.; Penning, L.C.; et al. Chemically Defined Hydrogel for Human Liver Organoid Culture. Adv. Funct. Mater. 2020, 30, 2000893. [Google Scholar] [CrossRef]
- MacPherson, D.; Bram, Y.; Park, J.; Schwartz, R.E. Peptide-based scaffolds for the culture and maintenance of primary human hepatocytes. Sci. Rep. 2021, 11, 1–9. [Google Scholar]
- Gottardi, R.; Moeller, K.; Di Gesù, R.; Tuan, R.S.; van Griensven, M.; Balmayor, E.R. Application of a Hyperelastic 3D Printed Scaffold for Mesenchymal Stem Cell-Based Fabrication of a Bizonal Tendon Enthesis-like Construct. Front. Mater. 2021, 8, 1–9. [Google Scholar] [CrossRef]
- Mei, Y.; Saha, K.; Bogatyrev, S.R.; Yang, J.; Hook, A.L.; Kalcioglu, Z.I.; Cho, S.; Mitalipova, M.; Pyzocha, N.; Rojas, F.; et al. Combinatorial development of biomaterials for clonal growth of human pluripotent stem cells. Nat. Mater. 2010, 9, 768–778. [Google Scholar] [CrossRef]
- Nandivada, H.; Villa-Diaz, L.G.; O’shea, K.S.; Smith, G.D.; Krebsbach, P.H.; Lahann, J. Fabrication of synthetic polymer coatings and their use in feeder-free culture of human embryonic stem cells. Nat. Protoc. 2011, 6, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Tao, R.; Wu, W.; Cao, H.; Xin, J.; Guo, J.; Jiang, L.; Gao, C.; Demetriou, A.A.; Farkas, D.L.; et al. 3D PLGA scaffolds improve differentiation and function of bone marrow mesenchymal stem cell-derived hepatocytes. Stem Cells Dev. 2010, 19, 1427–1436. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Lee, J.H.; Shirahama, H.; Seo, J.; Glenn, J.S.; Cho, N.J. Extracellular matrix functionalization and Huh-7.5 cell coculture promote the hepatic differentiation of human adipose-derived mesenchymal stem cells in a 3D ICC hydrogel scaffold. ACS Biomater. Sci. Eng. 2016, 2, 2255–2265. [Google Scholar] [CrossRef] [PubMed]
- Isomursu, A.; Lerche, M.; Taskinen, M.E.; Ivaska, J.; Peuhu, E. Integrin signaling and mechanotransduction in regulation of somatic stem cells. Exp. Cell Res. 2019, 378, 217–225. [Google Scholar] [CrossRef]
- Wang, Y.; Kim, M.H.; Shirahama, H.; Lee, J.H.; Ng, S.S.; Glenn, J.S.; Cho, N.J. ECM proteins in a microporous scaffold influence hepatocyte morphology, function, and gene expression. Sci. Rep. 2016, 6, 1–13. [Google Scholar] [CrossRef]
- Hwang, Y.; Goh, M.; Kim, M.; Tae, G. Injectable and detachable heparin-based hydrogel micropatches for hepatic differentiation of hADSCs and their liver targeted delivery. Biomaterials 2018, 165, 94–104. [Google Scholar] [CrossRef]
- Mittal, N.; Tasnim, F.; Yue, C.; Qu, Y.; Phan, D.; Choudhury, Y.; Tan, M.-H.; Yu, H. Substrate stiffness modulates the maturation of human pluripotent stem-cell-derived hepatocytes. ACS Biomater. Sci. Eng. 2016, 2, 1649–1657. [Google Scholar] [CrossRef]
- Cozzolino, A.M.; Noce, V.; Battistelli, C.; Marchetti, A.; Grassi, G.; Cicchini, C.; Tripodi, M.; Amicone, L. Modulating the substrate stiffness to manipulate differentiation of resident liver stem cells and to improve the differentiation state of hepatocytes. Stem Cells Int. 2016, 2016, 12. [Google Scholar] [CrossRef] [PubMed]
- Nemir, S.; West, J.L. Synthetic materials in the study of cell response to substrate rigidity. Ann. Biomed. Eng. 2010, 38, 2–20. [Google Scholar] [CrossRef]
- Lee, H.J.; Son, M.J.; Ahn, J.; Oh, S.J.; Lee, M.; Kim, A.; Jeung, Y.J.; Kim, H.G.; Won, M.; Lim, J.H.; et al. Elasticity-based development of functionally enhanced multicellular 3D liver encapsulated in hybrid hydrogel. Acta Biomater. 2017, 64, 67–79. [Google Scholar] [CrossRef]
- Badylak, S.F.; Taylor, D.; Uygun, K. Whole-organ tissue engineering: Decellularization and recellularization of three-dimensional matrix scaffolds. Annu. Rev. Biomed. Eng. 2011, 13, 27–53. [Google Scholar] [CrossRef]
- Jaramillo, M.; Yeh, H.; Yarmush, M.L.; Uygun, B.E. Decellularized human liver extracellular matrix (hDLM)-mediated hepatic differentiation of human induced pluripotent stem cells (hIPSCs). J. Tissue Eng. Regen. Med. 2018, 12, e1962–e1973. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; Hussein, K.H.; Hong, S.H.; Ahn, C.; Yang, S.R.; Park, S.M.; Kweon, O.K.; Kim, B.M.; Woo, H.M. Decellularized liver extracellular matrix as promising tools for transplantable bioengineered liver promotes hepatic lineage commitments of induced pluripotent stem cells. Tissue Eng.-Part A 2016, 22, 449–460. [Google Scholar] [CrossRef]
- Wang, B.; Jakus, A.E.; Baptista, P.M.; Soker, S.; Soto-Gutierrez, A.; Abecassis, M.M.; Shah, R.N.; Wertheim, J.A. Functional maturation of induced pluripotent stem cell hepatocytes in extracellular matrix—A comparative analysis of bioartificial liver microenvironments. Stem Cells Transl. Med. 2016, 5, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Coronado, R.E.; Somaraki-Cormier, M.; Natesan, S.; Christy, R.J.; Ong, J.L.; Halff, G.A. Decellularization and solubilization of porcine liver for use as a substrate for porcine hepatocyte culture: Method optimization and comparison. Cell Transplant. 2017, 26, 1840–1854. [Google Scholar] [CrossRef]
- Hirata, M.; Yamaoka, T. Hepatocytic differentiation of iPS cells on decellularized liver tissue. J. Artif. Organs 2017, 20, 318–325. [Google Scholar] [CrossRef]
- Vyas, D.; Baptista, P.M.; Brovold, M.; Moran, E.; Gaston, B.; Booth, C.; Samuel, M.; Atala, A.; Soker, S. Self-assembled liver organoids recapitulate hepatobiliary organogenesis In Vitro. Hepatology 2018, 67, 750–761. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Zheng, Y.W.; Lin, H.L.; Lan, Q.H.; Huang, Z.W.; Wang, L.F.; Chen, R.; Xiao, J.; Kou, L.; Xu, H.; et al. Exploiting crosslinked decellularized matrix to achieve uterus regeneration and construction. Artif. Cells Nanomed. Biotechnol. 2020, 48, 218–229. [Google Scholar] [CrossRef]
- Chiesa, I.; De Maria, C.; Lapomarda, A.; Fortunato, G.M.; Montemurro, F.; Di Gesù, R.; Tuan, R.S.; Vozzi, G.; Gottardi, R. Endothelial cells support osteogenesis in an in vitro vascularized bone model developed by 3D bioprinting. Biofabrication 2020, 12, 025013. [Google Scholar] [CrossRef] [PubMed]
- Di Gesù, R.; Acharya, A.P.; Jacobs, I.; Gottardi, R. 3D printing for tissue engineering in otolaryngology. Connect. Tissue Res. 2020, 61, 117–136. [Google Scholar] [CrossRef]
- Di Gesù, R.; Merlettini, A.; Gualandi, C.; Focarete, M.L. Advances in multidrug delivery from electrospun nanomaterials. In Core-Shell Nanostructures for Drug Delivery and Theranostics; Maria, L., Focarete, A.T., Eds.; Woodhead Publishing: Sawston, UK, 2018; pp. 405–430. [Google Scholar]
- Yu, C.; Ma, X.; Zhu, W.; Wang, P.; Miller, K.L.; Stupin, J.; Koroleva-Maharajh, A.; Hairabedian, A.; Chen, S. Scanningless and continuous 3D bioprinting of human tissues with decellularized extracellular matrix. Biomaterials 2019, 194, 1–13. [Google Scholar] [CrossRef]
- Ruoß, M.; Vosough, M.; Königsrainer, A.; Nadalin, S.; Wagner, S.; Sajadian, S.; Huber, D.; Heydari, Z.; Ehnert, S.; Hengstler, J.G.; et al. Towards improved hepatocyte cultures: Progress and limitations. Food Chem. Toxicol. 2020, 138, 111188. [Google Scholar] [CrossRef]
- Zeilinger, K.; Freyer, N.; Damm, G.; Seehofer, D.; Knöspel, F. Cell sources for in vitro human liver cell culture models. Exp. Biol. Med. 2016, 241, 1684–1698. [Google Scholar] [CrossRef]
- Corbett, J.L.; Duncan, S.A. iPSC-derived hepatocytes as a platform for disease modeling and drug discovery. Front. Med. 2019, 6, 265. [Google Scholar] [CrossRef]
- Miranda, J.P.; Serras, A.S.; Rodrigues, J.S.; Cipriano, M.; Rodrigues, A.V.; Oliveira, N.G. A Critical Perspective on 3D Liver Models for Drug Metabolism and Toxicology Studies. Front. Cell Dev. Biol. 2021, 9, 203. [Google Scholar]
- Gripon, P.; Rumin, S.; Urban, S.; Le Seyec, J.; Glaise, D.; Cannie, I.; Guyomard, C.; Lucas, J.; Trepo, C.; Guguen-Guillouzo, C. Infection of a human hepatoma cell line by hepatitis B virus. Proc. Natl. Acad. Sci. USA 2002, 99, 15655–15660. [Google Scholar] [CrossRef]
- Andersson, T.B.; Kanebratt, K.P.; Kenna, J.G. The HepaRG cell line: A unique in vitro tool for understanding drug metabolism and toxicology in human. Expert Opin. Drug Metab. Toxicol. 2012, 8, 909–920. [Google Scholar] [CrossRef]
- Luce, E.; Dubart-Kupperschmitt, A. Pluripotent stem cell-derived cholangiocytes and cholangiocyte organoids. Hum. Pluripotent Stem Cell Deriv. Organoid Model. 2020, 159, 69–93. [Google Scholar]
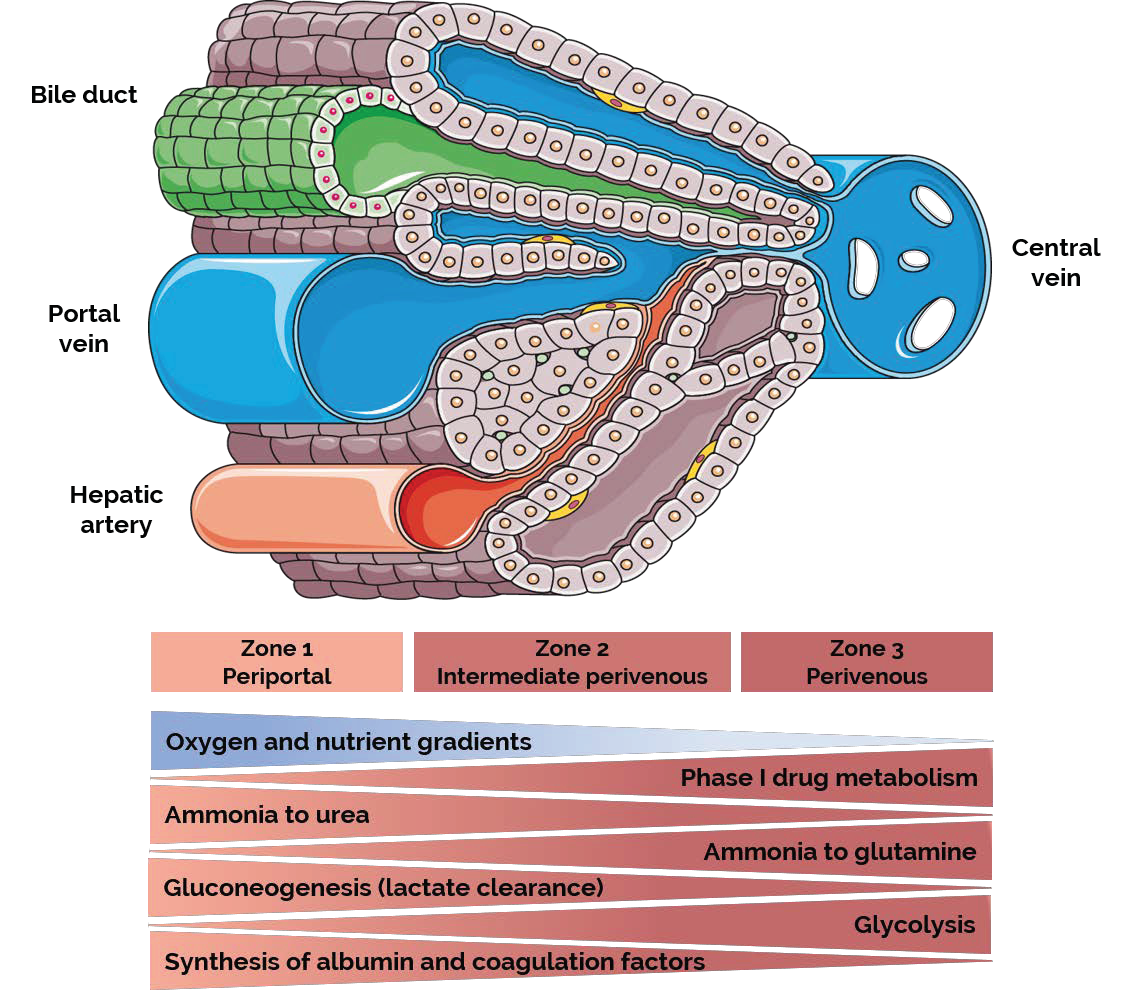
- Jungermann, K.; Sasse, D. Heterogeneity of liver parenchymal cells. Trends Biochem. Sci. 1978, 3, 198–202. [Google Scholar] [CrossRef]
- Jungermann, K.; Katz, N. Functional specialization of different hepatocyte populations. Physiol. Rev. 1989, 69, 708–764. [Google Scholar] [CrossRef]
- Ma, R.; Martínez-Ramírez, A.S.; Borders, T.L.; Gao, F.; Sosa-Pineda, B. Metabolic and non-metabolic liver zonation is established non-synchronously and requires sinusoidal Wnts. Elife 2020, 9, e46206. [Google Scholar] [CrossRef]
- Mitani, S.; Takayama, K.; Nagamoto, Y.; Imagawa, K.; Sakurai, F.; Tachibana, M.; Sumazaki, R.; Mizuguchi, H. Human ESC/iPSC-derived hepatocyte-like cells achieve zone-specific hepatic properties by modulation of WNT signaling. Mol. Ther. 2017, 25, 1420–1433. [Google Scholar] [CrossRef]
- Wu, F.; Wu, D.; Ren, Y.; Huang, Y.; Feng, B.; Zhao, N.; Zhang, T.; Chen, X.; Chen, S.; Xu, A. Generation of hepatobiliary organoids from human induced pluripotent stem cells. J. Hepatol. 2019, 70, 1145–1158. [Google Scholar] [CrossRef]
- Marzioni, M.; Glaser, S.S.; Francis, H.; Phinizy, J.L.; LeSage, G.; Alpini, G. Functional heterogeneity of cholangiocytes. Semin. Liver Dis. 2002, 22, 227–240. [Google Scholar] [CrossRef]
- Tabibian, J.H.; Masyuk, A.I.; Masyuk, T.V.; O’Hara, S.P.; LaRusso, N.F. Physiology of cholangiocytes. Compr. Physiol. 2013, 3, 541–565. [Google Scholar]
- Sampaziotis, F.; Justin, A.W.; Tysoe, O.C.; Sawiak, S.; Godfrey, E.M.; Upponi, S.S.; Gieseck, R.L.; De Brito, M.C.; Berntsen, N.L.; Gómez-Vázquez, M.J.; et al. Reconstruction of the mouse extrahepatic biliary tree using primary human extrahepatic cholangiocyte organoids. Nat. Med. 2017, 23, 954. [Google Scholar] [CrossRef]
- Kuo, Y.C.; Rajesh, R. Guided differentiation and tissue regeneration of induced pluripotent stem cells using biomaterials. J. Taiwan Inst. Chem. Eng. 2017, 77, 41–53. [Google Scholar] [CrossRef]
- Smith, A.S.; Macadangdang, J.; Leung, W.; Laflamme, M.A.; Kim, D.H. Human iPSC-derived cardiomyocytes and tissue engineering strategies for disease modeling and drug screening. Biotechnol. Adv. 2017, 35, 77–94. [Google Scholar] [CrossRef]
- Qian, T.; Shusta, E.V.; Palecek, S.P. Advances in microfluidic platforms for analyzing and regulating human pluripotent stem cells. Curr. Opin. Genet. Dev. 2015, 34, 54–60. [Google Scholar] [CrossRef]
- Mathur, A.; Loskill, P.; Hong, S.; Lee, J.Y.; Marcus, S.G.; Dumont, L.; Conklin, B.R.; Willenbring, H.; Lee, L.P.; Healy, K.E. Human induced pluripotent stem cell-based microphysiological tissue models of myocardium and liver for drug development. Stem Cell Res. Ther. 2013, 4, 1–5. [Google Scholar] [CrossRef]
- Tonon, F.; Giobbe, G.G.; Zambon, A.; Luni, C.; Gagliano, O.; Floreani, A.; Grassi, G.; Elvassore, N. In Vitro metabolic zonation through oxygen gradient on a chip. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
| Differentiation Stage | Small Molecule for Induction | Pathway Involved | Effect |
|---|---|---|---|
| DE | Activin A | TGF-β | Activation |
| LY29002 | IRS/PI3K | Activation | |
| CHIR99021 | Wnt/β-catenin | Activation | |
| Hepatoblast | A83-01 | TGF-β | Inhibition |
| CHIR99021 | Wnt/β-catenin | Inhibition | |
| Dihexa | HGF/Met | Activation | |
| Foskolin | PKA | Activation | |
| SAG | Sonic Hedgehog | Activation | |
| Vitamin C | Non-specific | Modulation of cell senescence | |
| Hepatocyte-like cells | DMSO | Non-specific | Increase in cell differentiation |
| DEX | Glucocorticoid | Activation | |
| Dihexa | HGF/Met | Activation | |
| A83-01 | TGF-β | Inhibition | |
| Sodium butyrate | p21Waf1/Cip1 | Activation (cell cycle arrest) | |
| Cholangiocyte progenitors | Activin A | TGF-β | Activation |
| Retinoic acid | Wnt/β-catenin | Inhibition of canonical Wnt and activation of non-canonical Wnt pathway | |
| Cholangiocyte-like cells | Activin A | TGF-β | Activation |
| Retinoic acid | Wnt/β-catenin | Inhibition of canonical Wnt and activation of non-canonical Wnt pathway | |
| DEX | Glucocorticoid | Activation | |
| Sodium pyruvate | Energy metabolism | Increase in cell survival |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasqua, M.; Di Gesù, R.; Chinnici, C.M.; Conaldi, P.G.; Francipane, M.G. Generation of Hepatobiliary Cell Lineages from Human Induced Pluripotent Stem Cells: Applications in Disease Modeling and Drug Screening. Int. J. Mol. Sci. 2021, 22, 8227. https://doi.org/10.3390/ijms22158227
Pasqua M, Di Gesù R, Chinnici CM, Conaldi PG, Francipane MG. Generation of Hepatobiliary Cell Lineages from Human Induced Pluripotent Stem Cells: Applications in Disease Modeling and Drug Screening. International Journal of Molecular Sciences. 2021; 22(15):8227. https://doi.org/10.3390/ijms22158227
Chicago/Turabian StylePasqua, Mattia, Roberto Di Gesù, Cinzia Maria Chinnici, Pier Giulio Conaldi, and Maria Giovanna Francipane. 2021. "Generation of Hepatobiliary Cell Lineages from Human Induced Pluripotent Stem Cells: Applications in Disease Modeling and Drug Screening" International Journal of Molecular Sciences 22, no. 15: 8227. https://doi.org/10.3390/ijms22158227
APA StylePasqua, M., Di Gesù, R., Chinnici, C. M., Conaldi, P. G., & Francipane, M. G. (2021). Generation of Hepatobiliary Cell Lineages from Human Induced Pluripotent Stem Cells: Applications in Disease Modeling and Drug Screening. International Journal of Molecular Sciences, 22(15), 8227. https://doi.org/10.3390/ijms22158227







