Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies
Abstract
1. Introduction
2. Background
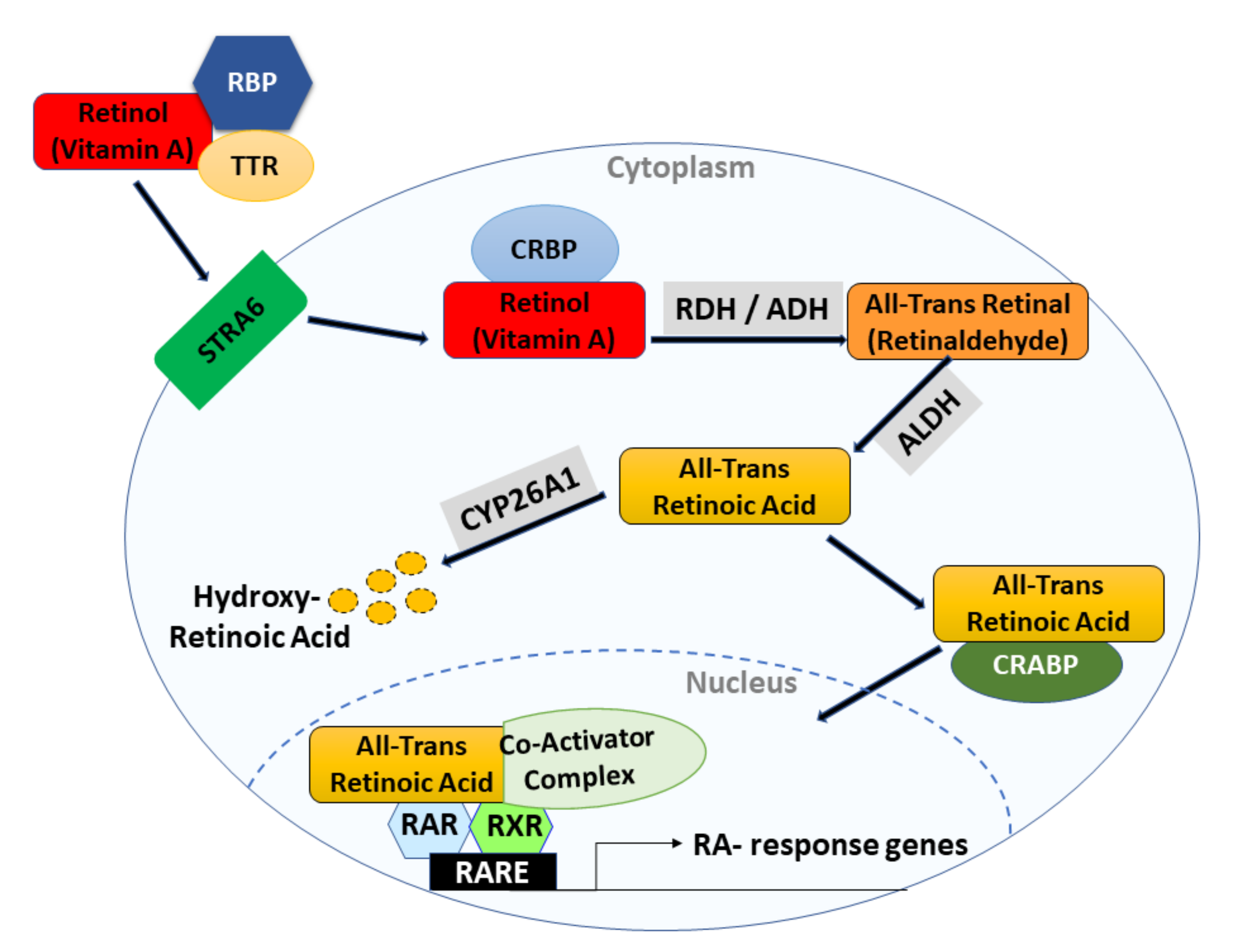
3. Retinoic Acid Signaling in Normal Cells
3.1. Retinoic Acid Receptors
3.2. Other Retinoid Signaling Components
4. Retinoid Agents
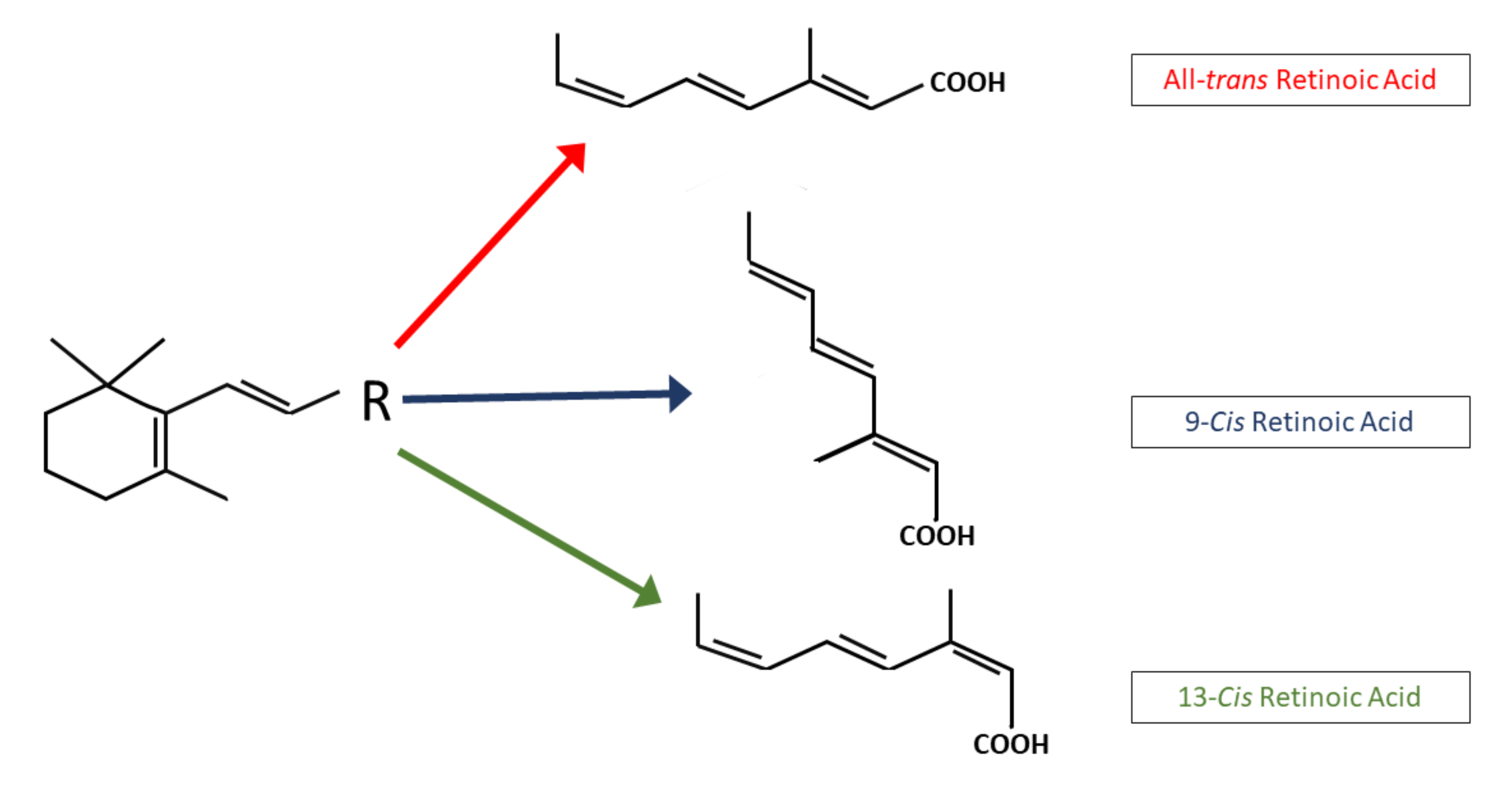
4.1. All-Trans Retinoic Acid (ATRA or Tretinoin)
4.1.1. Mechanism of Action
4.1.2. Pharmacological Uses
4.2. 13-cis Retinoic Acid (Isotretinoin)
4.2.1. Mechanism of Action
4.2.2. Pharmacological Uses
4.3. 9-cis Retinoic Acid (Alitretinoin)
4.3.1. Mechanism of Action
4.3.2. Pharmacological Uses
4.4. Liarozole (R75251)
4.4.1. Mechanism of Action
4.4.2. Pharmacological Uses
4.5. Talarozole (R115866)
4.5.1. Mechanism of Action
4.5.2. Pharmacological Uses
5. Effect of Retinoids in the Chemoprevention and Treatment of Various Cancers
5.1. Retinoids and Acute Promyelocytic Leukemia (APL)
5.2. Retinoids and Breast Cancer
5.3. Retinoids and Prostate Cancer
5.4. Retinoids and Lung Cancer
5.5. Retinoids and Colorectal Cancer (CRC)
5.6. Chemoprevention of Cancer Using Retinoids
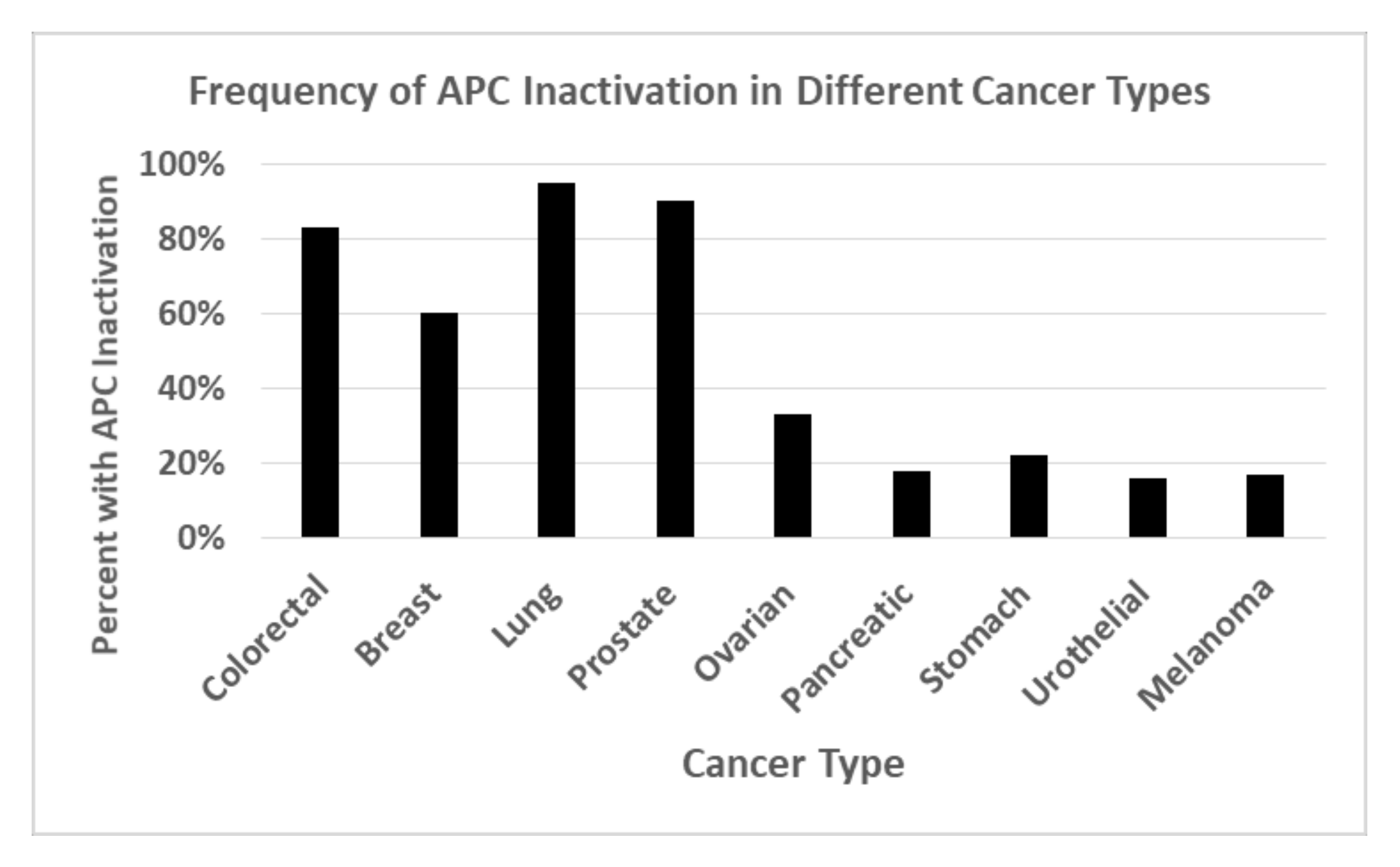
6. Retinoid Drugs Are Not Efficacious Against Solid Tumors—Why Not?
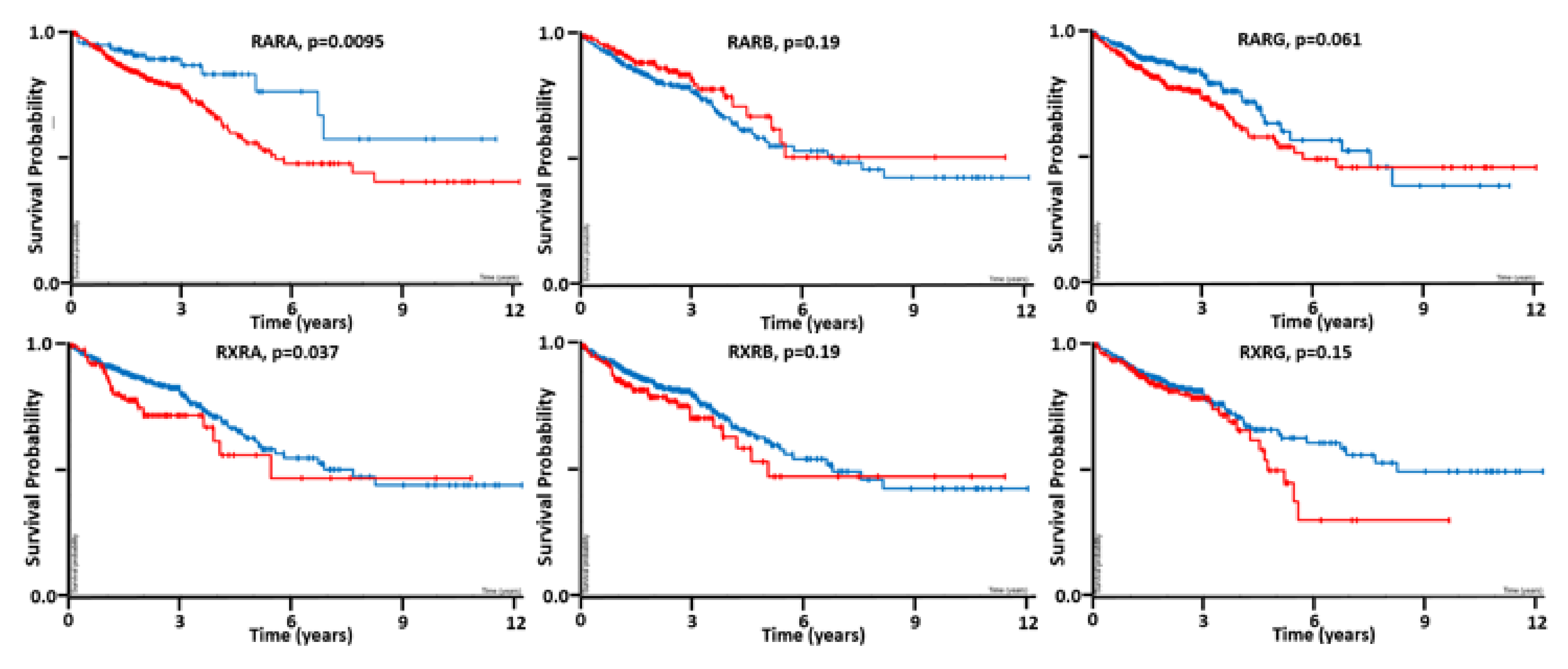
7. Pilot Study on the Effects of Retinoid Agents on APC-Mutant CRC Cells That Also Carry RA Receptor Mutations
8. The CYP26A1 Inhibitor Liarozole Decreases CRC Cell Proliferation, Sphere Formation, and Number of ALDH+ Cancer Stem Cells (CSCs)
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bushue, N.; Wan, Y.-J.Y. Retinoid pathway and cancer therapeutics. Adv. Drug Deliv. Rev. 2010, 62. [Google Scholar] [CrossRef]
- Blaner, W.S.; O’Byrne, S.M.; Wongsiriroj, N.; Kluwe, J.; D’Ambrosio, D.M.; Jiang, H.; Schwabe, R.F.; Hillman, E.M.C.; Piantedosi, R.; Libien, J. Hepatic stellate cell lipid droplets: A specialized lipid droplet for retinoid storage. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2009, 1791. [Google Scholar] [CrossRef]
- Mark, M.; Ghyselinck, N.B.; Chambon, P. Function of retinoic acid receptors during embryonic development. Nucl. Recept. Signal. 2009, 7. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Tang, X. Bexarotene: A promising anticancer agent. Cancer Chemother. Pharmacol. 2010, 65. [Google Scholar] [CrossRef] [PubMed]
- Hail, N.; Kim, H.J.; Lotan, R. Mechanisms of fenretinide-induced apoptosis. Apoptosis 2006, 11. [Google Scholar] [CrossRef] [PubMed]
- Gudas, L.J.; Wagner, J.A. Retinoids regulate stem cell differentiation. J. Cell. Physiol. 2011, 226. [Google Scholar] [CrossRef] [PubMed]
- Christov, K.T.; Moon, R.C.; Lantvit, D.D.; Boone, C.W.; Steele, V.E.; Lubet, R.A.; Kelloff, G.J.; Pezzuto, J.M. 9-cis-Retinoic Acid but not 4-(Hydroxyphenyl)retinamide inhibits prostate intraepithelial neoplasia in noble rats. Cancer Res. 2002, 62, 5178–5182. [Google Scholar]
- Wu, K.; Kim, H.-T.; Rodriquez, J.L.; Munoz-Medellin, D.; Mohsin, S.K.; Hilsenbeck, S.G.; Lamph, W.W.; Gottardis, M.M.; Shirley, M.A.; Kuhn, J.G.; et al. 9-cis-Retinoic acid suppresses mammary tumorigenesis in C3(1)-Simian Virus 40 T antigen-transgenic mice. Clin. Cancer Res. 2000, 6, 3696–3704. [Google Scholar]
- Matthay, K.K.; Reynolds, C.P.; Seeger, R.C.; Shimada, H.; Adkins, E.S.; Haas-Kogan, D.; Gerbing, R.B.; London, W.B.; Villablanca, J.G. Long-term results for children with high-risk neuroblastoma treated on a randomized trial of myeloablative therapy followed by 13- cis -retinoic acid: A Children’s Oncology Group Study. J. Clin. Oncol. 2009, 27. [Google Scholar] [CrossRef]
- Lefebvre, P.; Martin, P.J.; Flajollet, S.; Dedieu, S.; Billaut, X.; Lefebvre, B. Transcriptional activities of retinoic acid receptors. Vitam. Horm. 2005, 70, 199–264. [Google Scholar] [PubMed]
- Mongan, N.P.; Gudas, L.J. Diverse actions of retinoid receptors in cancer prevention and treatment. Differentiation 2007, 75, 853–870. [Google Scholar] [CrossRef] [PubMed]
- Gudas, L.J. Retinoids and vertebrate development. J. Biol. Chem. 1994, 269. [Google Scholar] [CrossRef]
- Di Masi, A.; Leboffe, L.; De Marinis, E.; Pagano, F.; Cicconi, L.; Rochette-Egly, C.; Lo-Coco, F.; Ascenzi, P.; Nervi, C. Retinoic acid receptors: From molecular mechanisms to cancer therapy. Mol. Asp. Med. 2015, 41. [Google Scholar] [CrossRef]
- Chambon, P. A decade of molecular biology of retinoic acid receptors. FASEB J. 1996, 10, 940–954. [Google Scholar] [CrossRef]
- Das, B.C.; Thapa, P.; Verma, A.; Ray, S.K.; Evans, T.; Karki, R.; Das, S.; Mahapatra, S.; Liu, T.-C.; Torregroza, I.; et al. Retinoic acid signaling pathways in development and diseases. Bioorg. Med. Chem. 2014, 22, 673–683. [Google Scholar] [CrossRef]
- Swift, M.E.; Wallden, B.; Wayner, E.A.; Swisshelm, K. Truncated RAR beta isoform enhances proliferation and retinoid resistance. J. Cell. Physiol. 2006, 209. [Google Scholar] [CrossRef]
- Facey, C.O.B.; Boman, B.M. Retinoids in Treatment of Colorectal Cancer [Online First]. IntechOpen. Available online: https://www.intechopen.com/online-first/retinoids-in-treatment-of-colorectal-cancer (accessed on 16 July 2021).
- Retinoic Acid Receptor RXR-alpha (Human)|Protein Target—PubChem. Available online: https://pubchem.ncbi.nlm.nih.gov/protein/P19793 (accessed on 18 June 2021).
- Zhang, R.; Wang, Y.; Li, R.; Chen, G. Transcriptional factors mediating retinoic acid signals in the control of energy metabolism. Int. J. Mol. Sci. 2015, 16, 14210–14244. [Google Scholar] [CrossRef]
- Perissi, V.; Rosenfeld, M.G. Controlling nuclear receptors: The circular logic of cofactor cycles. Nat. Rev. Mol. Cell Biol. 2005, 6. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, O.; Welch, J.S. Retinoic acid receptors in acute myeloid leukemia therapy. Cancers 2019, 11, 1915. [Google Scholar] [CrossRef]
- Rochette-Egly, C.; Chambon, P. F9 embryocarcinoma cells: A cell autonomous model to study the functional selectivity of RARs and RXRs in retinoid signaling. Histol. Histopathol. 2001, 16, 909–922. [Google Scholar] [PubMed]
- Donato, L.J.; Suh, J.H.; Noy, N. Suppression of mammary carcinoma cell growth by retinoic acid: The cell cycle control gene Btg2 is a direct target for retinoic acid receptor signaling. Cancer Res. 2007, 67. [Google Scholar] [CrossRef]
- Schug, T.T.; Berry, D.C.; Shaw, N.S.; Travis, S.N.; Noy, N. Opposing effects of retinoic acid on cell growth result from alternate activation of two different nuclear receptors. Cell 2007, 129. [Google Scholar] [CrossRef]
- Li, J.; Wang, J.; Wang, J.; Nawaz, Z.; Liu, J.M.; Qin, J.; Wong, J. Both corepressor proteins SMRT and N-CoR exist in large protein complexes containing HDAC3. EMBO J. 2000, 19. [Google Scholar] [CrossRef]
- Perissi, V.; Jepsen, K.; Glass, C.K.; Rosenfeld, M.G. Deconstructing repression: Evolving models of co-repressor action. Nat. Rev. Genet. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, B.W.; Kumar, R. Nuclear Receptor Coregulators in Cancer Biology: Figure 1. Cancer Res. 2009, 69. [Google Scholar] [CrossRef]
- Glass, C.K.; Rosenfeld, M.G. The coregulator exchange in transcriptional functions of nuclear receptors. Genes Dev. 2000, 14, 121–141. [Google Scholar]
- Yao, T.P.; Ku, G.; Zhou, N.; Scully, R.; Livingston, D.M. The nuclear hormone receptor coactivator SRC-1 is a specific target of p300. Proc. Natl. Acad. Sci. USA 1996, 93. [Google Scholar] [CrossRef]
- Gillespie, R.F.; Gudas, L.J. Retinoid regulated association of transcriptional co-regulators and the polycomb group protein SUZ12 with the retinoic acid response elements of Hoxa1, RARβ2, and Cyp26A1 in F9 embryonal carcinoma cells. J. Mol. Biol. 2007, 372. [Google Scholar] [CrossRef]
- Lara-Ramírez, R.; Zieger, E.; Schubert, M. Retinoic acid signaling in spinal cord development. Int. J. Biochem. Cell Biol. 2013, 45. [Google Scholar] [CrossRef]
- Bolis, M.; Paroni, G.; Fratelli, M.; Vallerga, A.; Guarrera, L.; Zanetti, A.; Kurosaki, M.; Garattini, S.K.; Gianni’, M.; Lupi, M.; et al. All-trans retinoic acid stimulates viral mimicry, interferon responses and antigen presentation in breast-cancer cells. Cancers 2020, 12, 1169. [Google Scholar] [CrossRef] [PubMed]
- Gocek, E.; Marcinkowska, E. Differentiation Therapy of Acute Myeloid Leukemia. Cancers 2011, 3, 2402–2420. [Google Scholar] [CrossRef] [PubMed]
- Ni, X.; Hu, G.; Cai, X. The success and the challenge of all-trans retinoic acid in the treatment of cancer. Crit. Rev. Food Sci. Nutr. 2019, 59. [Google Scholar] [CrossRef]
- Urbach, J.; Rando, R.R. Isomerization of all-trans-retinoic acid to 9-cis-retinoic acid. Biochem. J. 1994, 299. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-Y. Mechanism of action of all-trans retinoic acid and arsenic trioxide in the treatment of acute promyelocytic leukemia. Gan To Kagaku Ryoho 2002, 29, 214–218. [Google Scholar]
- Yoshida, H.; Kitamura, K.; Tanaka, K.; Omura, S.; Miyazaki, T.; Hachiya, T.; Ohno, R.; Naoe, T. Accelerated degradation of PML-retinoic acid receptor α (PML-RARA) oncoprotein by all-trans-retinoic acid in acute promyelocytic leukemia: Possible role of the proteasome pathway. Cancer Res. 1996, 56, 2945–2948. [Google Scholar]
- Chen, X.; He, M.; Zhou, G. All-trans retinoic acid induces anti-tumor effects via STAT3 signaling inhibition in oral squamous cell carcinoma and oral dysplasia. J. Oral Pathol. Med. 2019, 48. [Google Scholar] [CrossRef]
- Arisi, M.F.; Starker, R.A.; Addya, S.; Huang, Y.; Fernandez, S.V. All trans-retinoic acid (ATRA) induces re-differentiation of early transformed breast epithelial cells. Int. J. Oncol. 2014, 44. [Google Scholar] [CrossRef] [PubMed]
- Bhatlekar, S.; Viswanathan, V.; Fields, J.Z.; Boman, B.M. Overexpression of HOXA4 and HOXA9 genes promotes self-renewal and contributes to colon cancer stem cell overpopulation. J. Cell Physiol. 2018, 233, 727–735. [Google Scholar] [CrossRef]
- Young, M.-J.; Wu, Y.-H.; Chiu, W.-T.; Weng, T.-Y.; Huang, Y.-F.; Chou, C.-Y. All-trans retinoic acid downregulates ALDH1-mediated stemness and inhibits tumour formation in ovarian cancer cells. Carcinogenesis 2015, 36. [Google Scholar] [CrossRef]
- Layton, A. The use of isotretinoin in acne. Dermatoendocrinology 2009, 1. [Google Scholar] [CrossRef]
- Leyden, J.J.; Del Rosso, J.Q.; Baum, E.W.; Mcguigan, K.A. The use of isotretinoin in the treatment of acne vulgaris clinical considerations and future directions. J. Clin. Aesthet. Dermatol. 2014, 7, S3. [Google Scholar] [PubMed]
- Veal, G.J.; Errington, J.; Rowbotham, S.E.; Illingworth, N.A.; Malik, G.; Cole, M.; Daly, A.K.; Pearson, A.D.J.; Boddy, A.V. Adaptive dosing approaches to the individualization of 13-Cis-retinoic acid (Isotretinoin) treatment for children with high-risk neuroblastoma. Clin. Cancer Res. 2013, 19. [Google Scholar] [CrossRef]
- Lucek, R.W.; Colburn, W.A. Clinical Pharmacokinetics of the Retinoids. Clin. Pharmacokinet. 1985, 10. [Google Scholar] [CrossRef]
- Reynolds, C.P.; Matthay, K.K.; Villablanca, J.G.; Maurer, B.J. Retinoid therapy of high-risk neuroblastoma. Cancer Lett. 2003, 197. [Google Scholar] [CrossRef]
- Takahashi, Y.; Moiseyev, G.; Chen, Y.; Farjo, K.; Nikolaeva, O.; Ma, J.-X. An enzymatic mechanism for generating the precursor of endogenous 13-cis retinoic acid in the brain. FEBS J. 2011, 278. [Google Scholar] [CrossRef]
- Armstrong, J.L.; Redfern, C.P.F.; Veal, G.J. 13-cis Retinoic acid and isomerisation in paediatric oncology—Is changing shape the key to success? Biochem. Pharmacol. 2005, 69. [Google Scholar] [CrossRef]
- Allenby, G.; Bocquel, M.T.; Saunders, M.; Kazmer, S.; Speck, J.; Rosenberger, M.; Lovey, A.; Kastner, P.; Grippo, J.F.; Chambon, P. Retinoic acid receptors and retinoid X receptors: Interactions with endogenous retinoic acids. Proc. Natl. Acad. Sci. USA 1993, 90. [Google Scholar] [CrossRef]
- Blaner, W.S. Cellular metabolism and actions of 13-cis-retinoic acid. J. Am. Acad. Dermatol. 2001, 45. [Google Scholar] [CrossRef]
- Chen, H.; Juchau, R.M. Glutathione S-transferases act as isomerases in isomerization of 13-cis-retinoic acid to all-trans-retinoic acid in vitro. Biochem. J. 1997, 327. [Google Scholar] [CrossRef]
- Tsukada, M.; Schröder, M.; Orfanos, C.E.; Zouboulis, C.C.; Roos, T.C.; Chandraratna, R.A.S.; Reichert, U.; Merk, H.F. 13-cis retinoic acid exerts its specific activity on human sebocytes through selective intracellular isomerization to All-trans retinoic acid and binding to retinoid acid receptors. J. Investig. Dermatol. 2000, 115. [Google Scholar] [CrossRef]
- Orfanos, C.E.; Zouboulis, C.C.; Almond-Roesler, B.; Geilen, C.C. Current use and future potential role of retinoids in dermatology. Drugs 1997, 53. [Google Scholar] [CrossRef]
- O’Reilly, K.C.; Trent, S.; Bailey, S.J.; Lane, M.A. 13-cis-Retinoic acid alters intracellular serotonin, increases 5-HT1A receptor, and serotonin reuptake transporter levels in vitro. Exp. Biol. Med. 2007, 232. [Google Scholar] [CrossRef]
- Matthay, K.K.; Villablanca, J.G.; Seeger, R.C.; Stram, D.O.; Harris, R.E.; Ramsay, N.K.; Swift, P.; Shimada, H.; Black, C.T.; Brodeur, G.M.; et al. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. N. Engl. J. Med. 1999, 341. [Google Scholar] [CrossRef] [PubMed]
- Crandall, J.; Sakai, Y.; Zhang, J.; Koul, O.; Mineur, Y.; Crusio, W.E.; McCaffery, P. 13-cis-retinoic acid suppresses hippocampal cell division and hippocampal-dependent learning in mice. Proc. Natl. Acad. Sci. USA 2004, 101. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Crandall, J.E.; Brodsky, J.; Mccaffery, P. 13-cis Retinoic acid (Accutane) suppresses hippocampal cell survival in mice. Ann. N. Y. Acad. Sci. 2004, 1021. [Google Scholar] [CrossRef]
- Bremner, J.D.; McCaffery, P. The neurobiology of retinoic acid in affective disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2008, 32. [Google Scholar] [CrossRef]
- Nelson, A.M.; Gilliland, K.L.; Cong, Z.; Thiboutot, D.M. 13-cis Retinoic acid induces apoptosis and cell cycle arrest in human SEB-1 sebocytes. J. Investig. Dermatol. 2006, 126. [Google Scholar] [CrossRef] [PubMed]
- Melnik, B. Apoptosis May Explain the pharmacological mode of action and adverse effects of Isotretinoin, including teratogenicity. Acta Derm. Venereol. 2017, 97. [Google Scholar] [CrossRef]
- Germain, P.; Chambon, P.; Eichele, G.; Evans, R.M.; Lazar, M.A.; Leid, M.; De Lera, A.R.; Lotan, R.; Mangelsdorf, D.J.; Gronemeyer, H. International union of pharmacology. LXIII. Retinoid X receptors. Pharmacol. Rev. 2006, 58, 760–772. [Google Scholar] [CrossRef]
- Lin, Y.-W.; Lien, L.-M.; Yeh, T.-S.; Wu, H.-M.; Liu, Y.-L.; Hsieh, R.-H. 9-cis retinoic acid induces retinoid X receptor localized to the mitochondria for mediation of mitochondrial transcription. Biochem. Biophys. Res. Commun. 2008, 377. [Google Scholar] [CrossRef]
- Bubna, A. Alitretinoin in dermatology-An update. Indian J. Dermatol. 2015, 60. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Tao, Y.; Zhang, M.; Ma, P.; Li, L.; Diao, Q. Effects of 9-cis-retinoic acid on the proliferation and apoptosis of cutaneous T-cell lymphoma cells. Anticancer Drugs 2019, 30. [Google Scholar] [CrossRef]
- Yu, S.-J.; Airavaara, M.; Wu, K.-J.; Harvey, B.K.; Liu, H.S.; Yang, Y.; Zacharek, A.; Chen, J.; Wang, Y. 9-cis retinoic acid induces neurorepair in stroke brain. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Wauwe, J.V.; Nyen, G.V.; Coene, M.; Stoppie, P.; Cools, W.; Goossens, J.; Borghgraef, P.; Janssen, P. Liarozole, an inhibitor of retinoic acid metabolism, exerts retinoid-mimetic effects in vivo. J. Pharmacol. Exp. Ther. 1992, 261, 773–779. [Google Scholar] [PubMed]
- Miller, W.H. The emerging role of retinoids and retinoic acid metabolism blocking agents in the treatment of cancer. Cancer 1998, 83. [Google Scholar] [CrossRef]
- Bruynseels, J.; De Coster, R.; Van Rooy, P.; Wouters, W.; Coene, M.-C.; Snoeck, E.; Raeymaekers, A.; Freyne, E.; Sanz, G.; Bussche, G.V.; et al. R 75251, a new inhibitor of steroid biosynthesis. Prostate 1990, 16. [Google Scholar] [CrossRef] [PubMed]
- Nelson, C.; Buttrick, B.; Isoherranen, N. Therapeutic potential of the inhibition of the retinoic acid hydroxylases CYP26A1 and CYP26B1 by xenobiotics. Curr. Top. Med. Chem. 2013, 13. [Google Scholar] [CrossRef]
- Gilden, M.; Malik, M.; Britten, J.; Delgado, T.; Levy, G.; Catherino, W.H. Leiomyoma fibrosis inhibited by liarozole, a retinoic acid metabolic blocking agent. Fertil. Steril. 2012, 98. [Google Scholar] [CrossRef] [PubMed]
- Berth-Jones, J.; Todd, G.; Hutchinson, P.E.; Thestrup-Pedersen, K.; Vanhoutte, F.P. Treatment of psoriasis with oral liarozole: A dose-ranging study. Br. J. Dermatol. 2000, 143. [Google Scholar] [CrossRef]
- Goss, P.E.; Strasser, K.; Marques, R.; Clemons, M.; Oza, A.; Goel, R.; Blackstein, M.; Kaizer, L.; Sterns, E.E.; Nabholtz, J.-M.; et al. Liarozole Fumarate (R85246): In the treatment of ER negative, Tamoxifen refractory or chemotherapy resistant postmenopausal metastatic breast cancer. Breast Cancer Res. Treat. 2000, 64. [Google Scholar] [CrossRef]
- Verfaille, C.J.; Vanhoutte, F.P.; Blanchet-Bardon, C.; Van Steensel, M.A.; Steijlen, P.M. Oral liarozole vs. acitretin in the treatment of ichthyosis: A phase II/III multicentre, double-blind, randomized, active-controlled study. Br. J. Dermatol. 2007, 156, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Stoppie, P.; Borgers, M.; Borghgraef, P.; Dillen, L.; Goossens, J.; Sanz, G.; Szel, H.; Hove, C.V.; Nyen, G.V.; Nobels, G.; et al. R115866 Inhibits All-trans-retinoic acid metabolism and exerts retinoidal effects in rodents. J. Pharmacol. Exp. Ther. 2000, 293, 304–312. [Google Scholar]
- Giltaire, S.; Herphelin, F.; Frankart, A.; Hérin, M.; Stoppie, P.; Poumay, Y. The CYP26 inhibitor R115866 potentiates the effects of all- trans retinoic acid on cultured human epidermal keratinocytes. Br. J. Dermatol. 2009, 160. [Google Scholar] [CrossRef]
- Verfaille, C.; Thissen, C.; Bovenschen, H.; Mertens, J.; Steijlen, P.; Van De Kerkhof, P. Oral R115866 in the treatment of moderate to severe plaque-type psoriasis. J. Eur. Acad. Dermatol. Venereol. 2007, 21. [Google Scholar] [CrossRef] [PubMed]
- Verfaille, C.J.; Coel, M.; Boersma, I.H.; Mertens, J.; Borgers, M.; Roseeuw, D. Oral R115866 in the treatment of moderate to severe facial acne vulgaris: An exploratory study. Br. J. Dermatol. 2007, 157. [Google Scholar] [CrossRef] [PubMed]
- Breitman, T.R.; Selonick, S.E.; Collins, S.J. Induction of differentiation of the human promyelocytic leukemia cell line (HL-60) by retinoic acid. Proc. Natl. Acad. Sci. USA 1980, 77. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.A.; Fenaux, P.; Tallman, M.S.; Estey, E.H.; Löwenberg, B.; Naoe, T.; Lengfelder, E.; Döhner, H.; Burnett, A.K.; Chen, S.-J.; et al. Management of acute promyelocytic leukemia: Updated recommendations from an expert panel of the European LeukemiaNet. Blood 2019, 133. [Google Scholar] [CrossRef]
- Minucci, S.; Maccarana, M.; Cioce, M.; De Luca, P.; Gelmetti, V.; Segalla, S.; Di Croce, L.; Giavara, S.; Matteucci, C.; Gobbi, A.; et al. Oligomerization of RAR and AML1 transcription factors as a novel mechanism of oncogenic activation. Mol. Cell 2000, 5. [Google Scholar] [CrossRef]
- Chen, S.-J.; Zhou, G.-B. Targeted therapy: The new lease on life for acute promyelocytic leukemia, and beyond. IUBMB Life 2012, 64. [Google Scholar] [CrossRef]
- Martens, J.H.A.; Brinkman, A.B.; Simmer, F.; Francoijs, K.-J.; Nebbioso, A.; Ferrara, F.; Altucci, L.; Stunnenberg, H.G. PML-RARα/RXR alters the epigenetic landscape in Acute Promyelocytic Leukemia. Cancer Cell 2010, 17. [Google Scholar] [CrossRef]
- Ghavamzadeh, A.; Alimoghaddam, K.; Rostami, S.; Ghaffari, S.H.; Jahani, M.; Iravani, M.; Mousavi, S.A.; Bahar, B.; Jalili, M. Phase II study of single-agent arsenic trioxide for the front-line therapy of Acute Promyelocytic Leukemia. J. Clin. Oncol. 2011, 29. [Google Scholar] [CrossRef]
- Zhou, G.-B.; Zhang, J.; Wang, Z.-Y.; Chen, S.-J.; Chen, Z. Treatment of acute promyelocytic leukaemia with all-trans retinoic acid and arsenic trioxide: A paradigm of synergistic molecular targeting therapy. Philos. Trans. R. Soc. B Biol. Sci. 2007, 362. [Google Scholar] [CrossRef] [PubMed]
- Lotan, R. Different susceptibilities of human melanoma and breast carcinoma cell lines to retinoic acid-induced growth inhibition. Cancer Res. 1979, 39, 1014–1019. [Google Scholar] [PubMed]
- Veronesi, U.; De Palo, G.; Marubini, E.; Costa, A.; Formelli, F.; Mariani, L.; Decensi, A.; Camerini, T.; Del Turco, M.R.; Di Mauro, M.G.; et al. Randomized trial of Fenretinide to prevent second breast malignancy in women with early breast cancer. JNCI J. Natl. Cancer Inst. 1999, 91. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; Kittler, R.; White, K.P. Genomic antagonism between retinoic acid and estrogen signaling in breast cancer. Cell 2009, 137. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, M.D.; Passalacqua, R.; Michiara, M.; Franciosi, V.; Di Costanzo, F.; Bisagni, G.; Camisa, R.; Buti, S.; Tomasello, G.; Cocconi, G. Tamoxifen vs Tamoxifen plus 13-cis-retinoic acid vs Tamoxifen plus Interferon α-2a as first-line endocrine treatments in advanced breast cancer: Updated results of a phase II, prospective, randomised multicentre trial. Acta Biomed. Ateneo Parm. 2007, 78, 204–209. [Google Scholar]
- Bryan, M.; Pulte, E.D.; Toomey, K.C.; Pliner, L.; Pavlick, A.C.; Saunders, T.; Wieder, R. A pilot phase II trial of all-trans retinoic acid (Vesanoid) and paclitaxel (Taxol) in patients with recurrent or metastatic breast cancer. Investig. New Drugs 2011, 29, 1482–1487. [Google Scholar] [CrossRef]
- Abdollah, F.; Schmitges, J.; Sun, M.; Jeldres, C.; Tian, Z.; Briganti, A.; Shariat, S.F.; Perrotte, P.; Montorsi, F.; Karakiewicz, P.I. Comparison of mortality outcomes after radical prostatectomy versus radiotherapy in patients with localized prostate cancer: A population-based analysis. Int. J. Urol. 2012, 19. [Google Scholar] [CrossRef]
- Moore, M.M.; Stockler, M.; Lim, R.; Mok, T.S.K.; Millward, M.; Boyer, M.J. A phase II study of fenretinide in patients with hormone refractory prostate cancer: A trial of the Cancer Therapeutics Research Group. Cancer Chemother. Pharmacol. 2010, 66. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Stone, N.; Stock, R.; Bednar, M.; Esseesse, I.; Singh, H.; Baldwin, Y.; Mandeli, J. 13-cis Retinoic acid and complete androgen blockade in advanced hormone-naive prostate cancer patients: Report of a phase II randomized study. J. Clin. Oncol. 2002, 20. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Yang, S.; Huang, J.; Cohen, M.B.; Roy-Burman, P. Aberration in the expression of the retinoid receptor, RXRα, in prostate cancer. Cancer Biol. Ther. 2003, 2. [Google Scholar] [CrossRef] [PubMed]
- Geradts, J.; Chen, J.Y.; Russell, E.; Yankaskas, J.; Nieves, L.; Minna, J. Human lung cancer cell lines exhibit resistance to retinoic acid treatment. Cell Growth Differ. 1993, 4, 799–809. [Google Scholar] [PubMed]
- Arrieta, O.; la Rosa, C.H.G.-D.; Aréchaga-Ocampo, E.; Villanueva-Rodríguez, G.; Cerón-Lizárraga, T.L.; Martínez-Barrera, L.; Vázquez-Manríquez, M.E.; Ríos-Trejo, M.Á.; Álvarez-Avitia, M.Á.; Hernández-Pedro, N.; et al. Randomized phase II trial of All- trans -retinoic acid with chemotherapy based on Paclitaxel and Cisplatin as first-line treatment in patients with advanced non–small-cell lung cancer. J. Clin. Oncol. 2010, 28. [Google Scholar] [CrossRef]
- Blumenschein, G.R.; Khuri, F.R.; von Pawel, J.; Gatzemeier, U.; Miller, W.H.; Jotte, R.M.; Le Treut, J.; Sun, S.-L.; Zhang, J.K.; Dziewanowska, Z.E.; et al. Phase III trial comparing Carboplatin, Paclitaxel, and Bexarotene with Carboplatin and Paclitaxel in chemotherapy-naïve patients with advanced or metastatic non–small-cell lung cancer: SPIRIT II. J. Clin. Oncol. 2008, 26. [Google Scholar] [CrossRef] [PubMed]
- Fritz, H.; Kennedy, D.; Fergusson, D.; Fernandes, R.; Doucette, S.; Cooley, K.; Seely, A.; Sagar, S.; Wong, R.; Seely, D. Vitamin A and retinoid derivatives for lung cancer: A systematic review and meta analysis. PLoS ONE 2011, 6. [Google Scholar] [CrossRef]
- Tratnjek, L.; Jeruc, J.; Romih, R.; Zupančič, D. Vitamin A and retinoids in bladder cancer chemoprevention and treatment: A narrative review of current evidence, challenges and future prospects. Int J. Mol. Sci. 2021, 22, 3510. [Google Scholar] [CrossRef]
- Freemantle, S.J.; Spinella, M.J.; Dmitrovsky, E. Retinoids in cancer therapy and chemoprevention: Promise meets resistance. Oncogene 2003, 22, 7305–7315. [Google Scholar] [CrossRef]
- Okuno, M.; Kojima, S.; Matsushima-Nishiwaki, R.; Tsurumi, H.; Muto, Y.; Friedman, S.; Moriwaki, H. Retinoids in cancer chemoprevention. Curr. Cancer Drug Targets 2004, 4, 285–298. [Google Scholar] [CrossRef]
- Bunaciu, R.P.; Yen, A. Retinoid chemoprevention: Who can benefit? Curr. Pharmacol. Rep. 2015, 1, 391–400. [Google Scholar] [CrossRef]
- Hansen, L.A.; Sigman, C.C.; Andreola, F.; Ross, S.A.; Kelloff, G.J.; De Luca, L.M. Retinoids in chemoprevention and differentiation therapy. Carcinogenesis 2000, 21, 1271–1279. [Google Scholar] [CrossRef]
- Sporn, M.B.; Newton, D.L. Chemoprevention of cancer with retinoids. Fed. Proc. 1979, 38, 2528–2534. [Google Scholar]
- Idres, N.; Benoît, G.; Flexor, M.A.; Lanotte, M.; Chabot, G.G. Granulocytic differentiation of human NB4 promyelocytic leukemia cells induced by All-trans retinoic acid metabolites. Cancer Res. 2001, 61, 700–705. [Google Scholar] [PubMed]
- Zhu, J.; Shi, X.G.; Chu, H.Y.; Tong, J.H.; Wang, Z.Y.; Naoe, T.; Waxman, S.; Chen, S.J.; Chen, Z. Effect of retinoic acid isomers on proliferation, differentiation and PML relocalization in the APL cell line NB4. Leukemia 1995, 9, 302–309. [Google Scholar]
- Fang, J.; Chen, S.-J.; Tong, J.-H.; Wang, Z.-G.; Chen, G.-Q. Treatment of acute promyelocytic leukemia with ATRA and As2O3: A model of molecular. Cancer Biol. Ther. 2002, 1, 614–620. [Google Scholar] [CrossRef]
- Volate, S.R.; Muga, S.J.; Issa, A.Y.; Nitcheva, D.; Smith, T.; Wargovich, M.J. Epigenetic modulation of the retinoid X receptor α by green tea in the azoxymethane- Apc Min/+ mouse model of intestinal cancer. Mol. Carcinog. 2009, 48, 920–933. [Google Scholar] [CrossRef] [PubMed]
- Nicke, B.; Riecken, E.-O.; Rosewicz, S. Induction of retinoic acid receptor beta mediates growth inhibition in retinoid resistant human colon carcinoma cells. Gut 1999, 45. [Google Scholar] [CrossRef]
- Zheng, Y. Effect of retinoids on AOM-induced colon cancer in rats: Modulation of cell proliferation, apoptosis and aberrant crypt foci. Carcinogenesis 1999, 20, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Penny, H.L.; Prestwood, T.R.; Bhattacharya, N.; Sun, F.; Kenkel, J.A.; Davidson, M.G.; Shen, L.; Zuniga, L.A.; Seeley, E.S.; Pai, R.; et al. Restoring retinoic acid attenuates intestinal inflammation and tumorigenesis in APCMin/Mice. Cancer Immunol. Res. 2016, 4, 917–926. [Google Scholar] [CrossRef]
- Nadauld, L.D.; Phelps, R.; Moore, B.C.; Eisinger, A.; Sandoval, I.T.; Chidester, S.; Peterson, P.W.; Manos, E.J.; Sklow, B.; Burt, R.W.; et al. Adenomatous Polyposis Coli control of C-terminal Binding Protein-1 stability regulates expression of intestinal retinol dehydrogenases. J. Biol. Chem. 2006, 281. [Google Scholar] [CrossRef]
- The Human Protein Atlas. Available online: https://www.proteinatlas.org (accessed on 15 July 2021).
- Mollersen, L. Dietary retinoic acid supplementation stimulates intestinal tumour formation and growth in multiple intestinal neoplasia (Min)/+ mice. Carcinogenesis 2003, 25, 149–153. [Google Scholar] [CrossRef]
- Shelton, D.N.; Sandoval, I.T.; Eisinger, A.; Chidester, S.; Ratnayake, A.; Ireland, C.M.; Jones, D.A. Up-regulation of CYP26A1 in Adenomatous Polyposis Coli–deficient vertebrates via a WNT-dependent mechanism: Implications for intestinal cell differentiation and colon tumor development. Cancer Res. 2006, 66, 7571–7577. [Google Scholar] [CrossRef]
- Modarai, S.R.; Gupta, A.; Opdenaker, L.M.; Kowash, R.; Masters, G.; Viswanathan, V.; Zhang, T.; Fields, J.Z.; Boman, B.M. The anti-cancer effect of retinoic acid signaling in CRC occurs via decreased growth of ALDH+ colon cancer stem cells and increased differentiation of stem cells. Oncotarget 2018, 9. [Google Scholar] [CrossRef]
- Prosperi, J.R.; Luu, H.H.; Goss, K.H. Dysregulation of the Wnt pathway in solid tumors. In Targeting the Wnt Pathway in Cancer; Springer: New York, NY, USA, 2011. [Google Scholar]
- Lesko, A.; Goss, K.; Prosperi, J. Exploiting APC function as a novel cancer therapy. Curr. Drug Targets 2014, 15. [Google Scholar] [CrossRef]
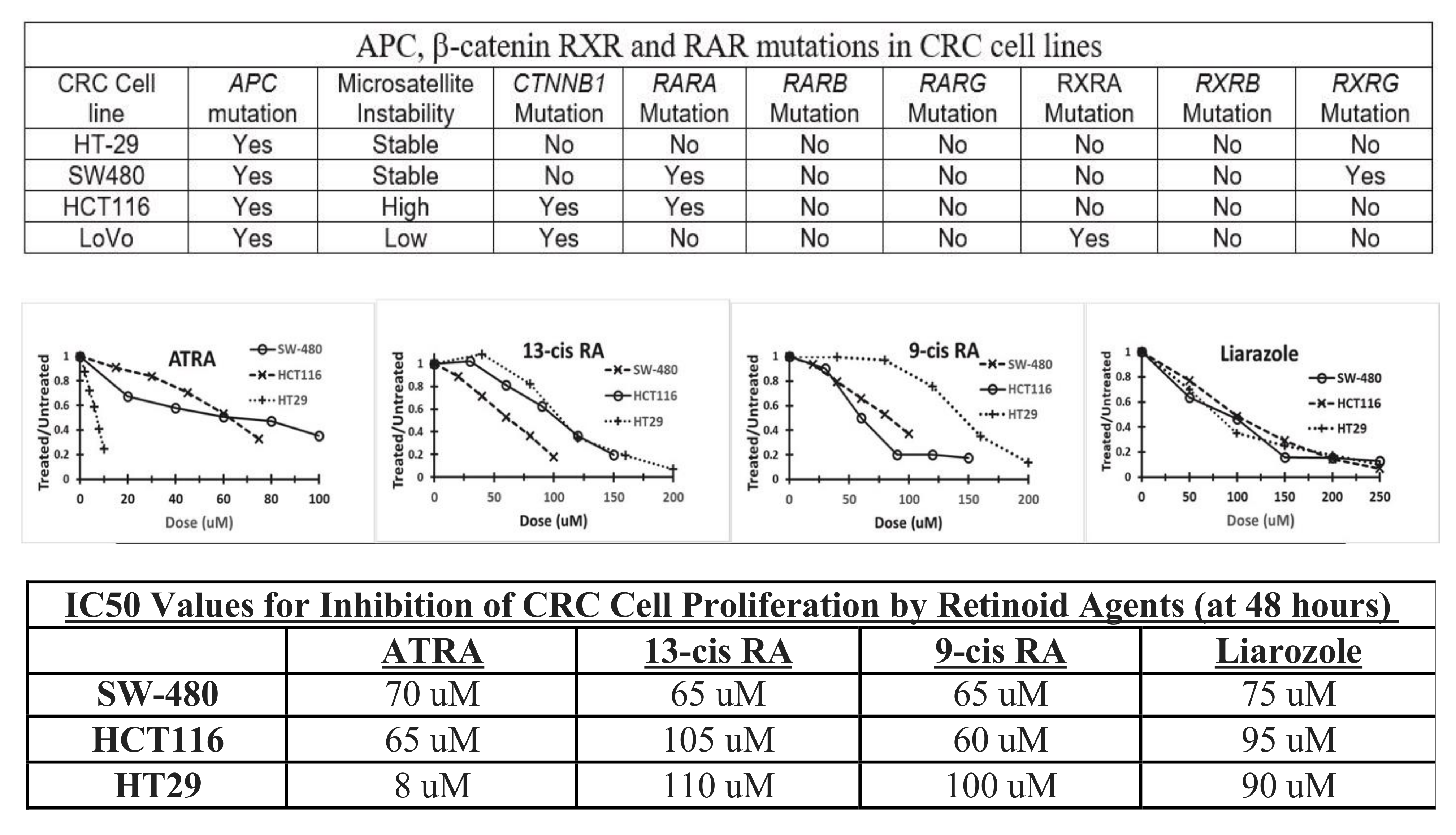
- Hunsu, V.O.; Facey, C.O.B.; Opdenaker, L.M.; Boman, B.M. Studying the ability of retinoids to inhibit growth of CRC cells based on their retinoid pathway genotype. In Proceedings of the AACR Annual Meeting, Philadelphia, PA, USA, 10–15 April 2021. [Google Scholar]
- Zhang, T.; Ahn, K.; Emerick, B.; Modarai, S.R.; Opdenaker, L.M.; Palazzo, J.; Schleiniger, G.; Fields, J.Z.; Boman, B.M. APC mutations in human colon lead to decreased neuroendocrine maturation of ALDH+ stem cells that alters GLP-2 and SST feedback signaling: Clue to a link between WNT and retinoic acid signalling in colon cancer development. PLoS ONE 2020, 15, e0239601. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, G.; Adida, C.; Altieri, D.C. A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat. Med. 1997, 3, 917–921. [Google Scholar] [CrossRef]
- Stobiecka, M.; Ratajczak, K.; Jakiela, S. Toward early cancer detection: Focus on biosensing systems and biosensors for an anti-apoptotic protein survivin and survivin mRNA. Biosens. Bioelectron. 2019, 137, 58–71. [Google Scholar] [CrossRef]
- Chen, X.; Duan, N.; Zhang, C.; Zhang, W. Survivin and Tumorigenesis: Molecular Mechanisms and Therapeutic Strategies. J. Cancer 2016, 7, 314–323. [Google Scholar] [CrossRef] [PubMed]
- Ratajczak, K.; Krazinski, B.E.; Kowalczyk, A.E.; Dworakowska, B.; Jakiela, S.; Stobiecka, M. Hairpin–hairpin molecular beacon interactions for detection of survivin mRNA in malignant SW480 cells. ACS Appl. Mater. Interfaces 2018, 10, 17028–17039. [Google Scholar] [CrossRef]
- Boman, B.M.; Walters, R.; Fields, J.Z.; Kovatich, A.J.; Zhang, T.; Isenberg, G.A.; Goldstein, S.D.; Palazzo, J.P. Colonic crypt changes during adenoma development in familial adenomatous polyposis: Immunohistochemical evidence for expansion of the crypt base cell population. Am. J. Pathol. 2004, 165, 1489–1498. [Google Scholar] [CrossRef]
- Boman, B.M.; Kopelovich, L.; Siracusa, L.D.; Zhang, T.; Henderson, K.; Cofer, Z.; Buchberg, A.M.; Fields, J.Z.; Otevrel, T. A Tcf4-GFP reporter mouse model for monitoring effects of Apc mutations during intestinal tumorigenesis. Mol. Carcinog. 2009, 48, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Otevrel, T.; Gao, Z.; Ehrlich, S.M.; Fields, J.Z.; Boman, B.M. Evidence that APC regulates survivin expression: A possible mechanism contributing to the stem cell origin of colon cancer. Cancer Res. 2001, 61, 8664–8667. [Google Scholar] [PubMed]
- Zhang, T.; Fields, J.Z.; Otevrel, T.; Masuda, E.; Palazzo, J.P.; Isenberg, G.A.; Goldstein, S.D.; Brand, M.; Boman, B.M. Survivin-induced Aurora-B kinase activation—A mechanism by which APC mutations contribute to increased mitoses during colon cancer development. Am. J. Pathol. 2010, 177, 2816–2826. [Google Scholar] [CrossRef]
- Kim, P.J.; Plescia, J.; Clevers, H.; Fearon, E.R.; Altieri, D.C. Survivin and molecular pathogenesis of colorectal cancer. Lancet 2003, 362, 205–209. [Google Scholar] [CrossRef]
- Zhang, T.; Fields, J.Z.; Ehrlich, S.M.; Boman, B.M. The chemopreventive agent sulindac attenuates expression of the antiapoptotic protein survivin in colorectal carcinoma cells. J. Pharmacol. Exp. Ther. 2004, 308, 434–437. [Google Scholar] [CrossRef] [PubMed]
- McEntee, M.F.; Chiu, C.H.; Whelan, J. Relationship of beta-catenin and Bcl-2 expression to sulindac induced regression of intestinal tumors in Min mice. Carcinogenesis 1999, 20, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, N.N.; Boolbol, S.K.; Bilinski, R.T.; Martucci, C.; Chadburn, A.; Bertagnolli, M.M. Apc gene mutation is associated with a dominant-negative effect upon intestinal cell migration. Cancer Res. 1997, 57, 5045–5050. [Google Scholar] [PubMed]
- Thompson, W.J.; Piazza, G.A.; Li, H.; Liu, L.; Fetter, J.; Zhu, B.; Sprel, G.; Ahnen, D.; Pamukcu, R. Exisulind induction of apoptosis involves guanosine 3,5-cyclic monophosphate phosphodiesterase inhibition, protein kinase G activation and attenuated beta-catenin. Cancer Res. 2000, 60, 3338–3342. [Google Scholar] [PubMed]
- Brown, W.A.; Skinner, S.A.; Vogiagis, D.; O’Brien, P.E. Inhibition of beta-catenin translocation in rodent colorectal tumors: A novel explanation for the protective effect of nonsteroidal anti-inflammatory drugs in colorectal cancer. Dig. Dis. Sci. 2001, 46, 2314–2321. [Google Scholar] [CrossRef]
- Rice, P.L.; Kelloff, J.; Sullivan, H.; Driggers, L.J.; Beard, K.S.; Kuwada, S.; Piazza, G.; Ahnen, D.J. Sulindac metabolites induce caspase- and proteasome-dependent degradation of beta-catenin protein in human colon cancer cells. Mol. Cancer Ther. 2003, 2, 885–892. [Google Scholar]
- Lee, J.J.; Wu, X.; Hildebrandt, M.A.; Yang, H.; Khuri, F.R.; Kim, E.; Gu, J.; Ye, Y.; Lotan, R.; Spitz, M.R.; et al. Global assessment of genetic variation influencing response to retinoid chemoprevention in head and neck cancer patients. Cancer Prev. Res. 2011, 4, 185–193. [Google Scholar] [CrossRef][Green Version]
| Drug | Completed Clinical Trials | Current/Recruiting Clinical Trials | CT.Gov Identifier/Ref |
|---|---|---|---|
| All-Trans retinoic acid | Effect of all-trans retinoic acid with chemotherapy-based paclitaxel and cisplatin as first-line treatment of patients with advanced non-small-cell lung cancer | NCT01048645 | |
| All-trans retinoic acid, and arsenic +/− idarubicin | NCT00413166 | ||
| A trial of all-trans retinoic acid (ATRA) in advanced adenoid cystic carcinoma | NCT03999684 | ||
| All-trans retinoic acid (ATRA) in the treatment of recurrent/metastatic adenoid cystic carcinoma of the head and neck (Aplus) | NCT04433169 | ||
| All-trans retinoic acid in combination with standard induction and consolidation therapy in older patients with newly diagnosed acute myeloid leukemia | NCT00151255 | ||
| To immunize patients with extensive stage SCLC combined with chemotherapy with or without all-trans retinoic acid | NCT00617409 | ||
| ATRA Trial—activity of ATRA in combination with anastrozole in pre-operative phase of operable HR-positive/HER2-negative early breast cancer eBC ATRA trial | NCT04113863 | ||
| Effect of all-trans retinoic acid with chemotherapy based on paclitaxel and cisplatin as first line treatment of patients with advanced non-small-cell lung cancer | NCT01048645 | ||
| All-trans retinoic acid in combination with standard induction and consolidation therapy in older patients with newly diagnosed acute myeloid leukemia | NCT00151255 | ||
| Study of dasatinib and all-trans retinoic acid for relapsed/refractory and/or elderly patients with acute myelogenous leukemia (AML) or myelodysplastic syndrome | NCT00892190 | ||
| Oral arsenic trioxide for newly diagnosed acute promyelocytic leukemia | NCT03624270 | ||
| 9-Cis retinoic acid | The pharmacokinetics of a single dose of 9-cis retinoic acid (alitretinoin, tocino) in patients with moderate-to-severe hepatic insufficiency | NCT01261923 | |
| Alitretinoin in the treatment of chronic hand eczema | NCT00519675 | ||
| Single dose of 9-cis retinoic acid in hepatic patients | NCT01891526 | ||
| Efficacy of alitretinoin treatment in patients with pustular form of psoriasis | NCT01245140 | ||
| A phase I trial of tamoxifen and 9-cis retinoic acid in breast cancer patients | NCT00001504 | ||
| A study of ALRT1057 in patients with AIDS-related Kaposi’s sarcoma | NCT00002188 | ||
| 13-Cis retinoic acid | Identifying the genetic predictors of severe acne vulgaris and the outcome of oral isotretinoin treatment (SA) | NCT01727440 | |
| 13-cis retinoic acid with or without Vitamin E for prevention of lung cancer (13-Cis) | NCT00002586 | ||
| RA-4: 13-cis retinoic acid for treatment of men with azoospermia | NCT03323801 | ||
| Oral liquid 13-cis retinoic acid (13-CRA) | NCT03291080 | ||
| Double-blind phase III trial of effects of low-dose 13-cis retinoic acid on prevention of second primaries in stages I–II head and neck cancer | NCT03370367 | ||
| Belinostat and isotretinoin in treating patients with solid tumors that are metastatic or that cannot be removed by surgery | NCT00334789 | ||
| Isotretinoin in preventing skin cancer | NCT00025012 | ||
| Safety and effectiveness of giving isotretinoin to HIV-infected women to treat cervical tumors | NCT00001073 | ||
| Interferon alfa, isotretinoin, and paclitaxel in treating patients with recurrent small-cell lung cancer | NCT00062010 | ||
| Cabozantinib in combination with 13-cis retinoic acid in children with relapsed or refractory solid tumors. | NCT03611595 | ||
| High-dose 3F8/GM immunotherapy plus 13-cis retinoic acid for consolidation of first remission after myeloablative therapy and autologous stem cell transplantation | NCT01183416 | ||
| Isotretinoin plus interferon in treating patients with recurrent cancer | NCT00002506 | ||
| 3F8/GM-CSF immunotherapy plus 13-cis retinoic acid for consolidation of first remission after non-myeloablative therapy in patients with high-risk neuroblastoma | NCT01183429 | ||
| Vorinostat and isotretinoin in treating patients with high-risk refractory or recurrent neuroblastoma | NCT01208454 | ||
| 3F8/GM-CSF immunotherapy plus 13-cis retinoic acid for primary refractory neuroblastoma in bone marrow | NCT01183897 | ||
| Liarozole | A randomized, double-blind, placebo-controlled phase II/III trial to evaluate the efficacy and safety of 2 doses of oral liarozole (75 mg od, and 150 mg od) given during 12 weeks in lamellar ichthyosis | NCT00282724 | |
| Talarozole | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hunsu, V.O.; Facey, C.O.B.; Fields, J.Z.; Boman, B.M. Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies. Int. J. Mol. Sci. 2021, 22, 7731. https://doi.org/10.3390/ijms22147731
Hunsu VO, Facey COB, Fields JZ, Boman BM. Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies. International Journal of Molecular Sciences. 2021; 22(14):7731. https://doi.org/10.3390/ijms22147731
Chicago/Turabian StyleHunsu, Victoria O., Caroline O. B. Facey, Jeremy Z. Fields, and Bruce M. Boman. 2021. "Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies" International Journal of Molecular Sciences 22, no. 14: 7731. https://doi.org/10.3390/ijms22147731
APA StyleHunsu, V. O., Facey, C. O. B., Fields, J. Z., & Boman, B. M. (2021). Retinoids as Chemo-Preventive and Molecular-Targeted Anti-Cancer Therapies. International Journal of Molecular Sciences, 22(14), 7731. https://doi.org/10.3390/ijms22147731





