Dimethyl Sulfoxide-Free Cryopreservation of Human Umbilical Cord Mesenchymal Stem Cells Based on Zwitterionic Betaine and Electroporation
Abstract
1. Introduction
2. Results
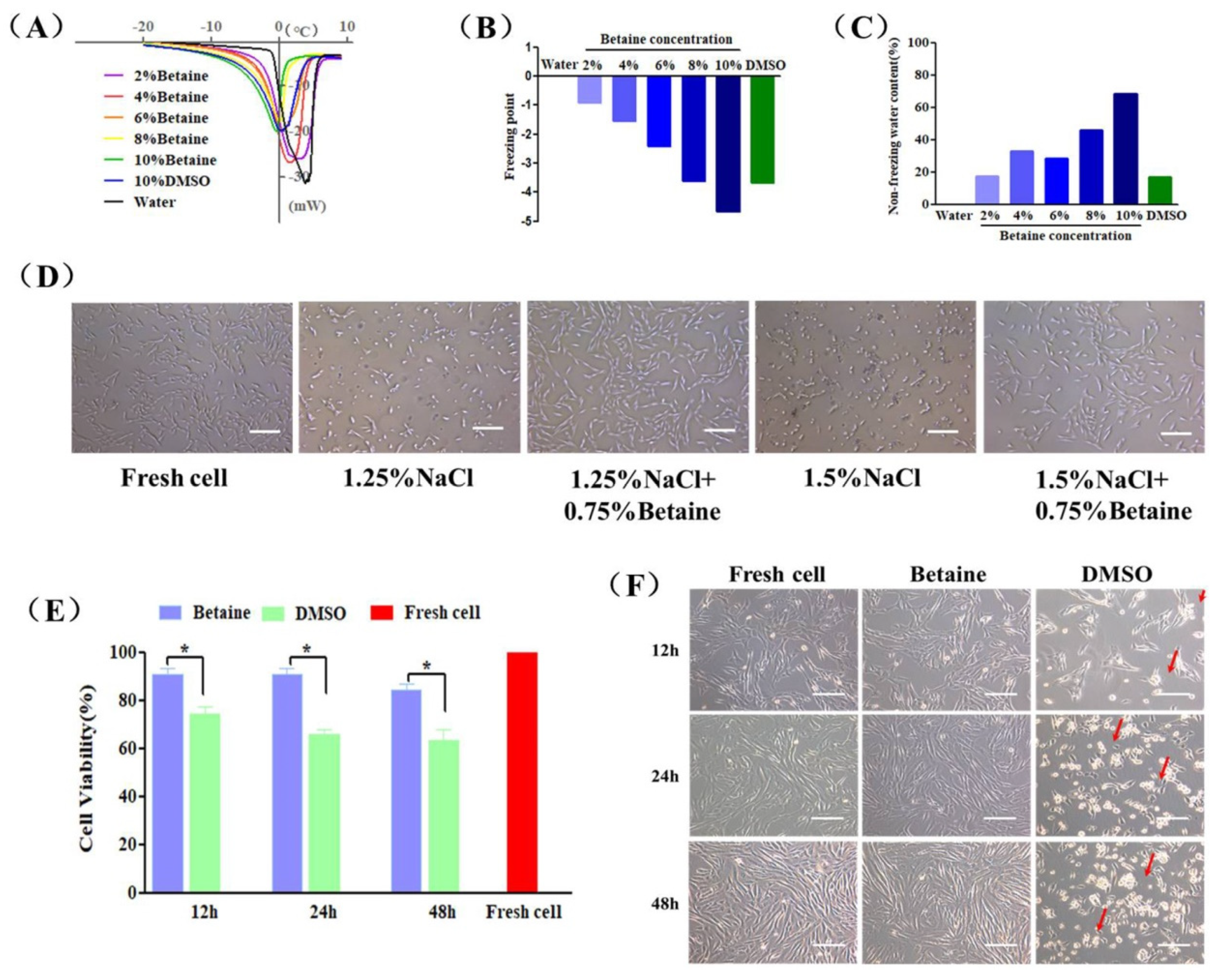
2.1. Cryoprotective Properties and Cytotoxicity of Betaine
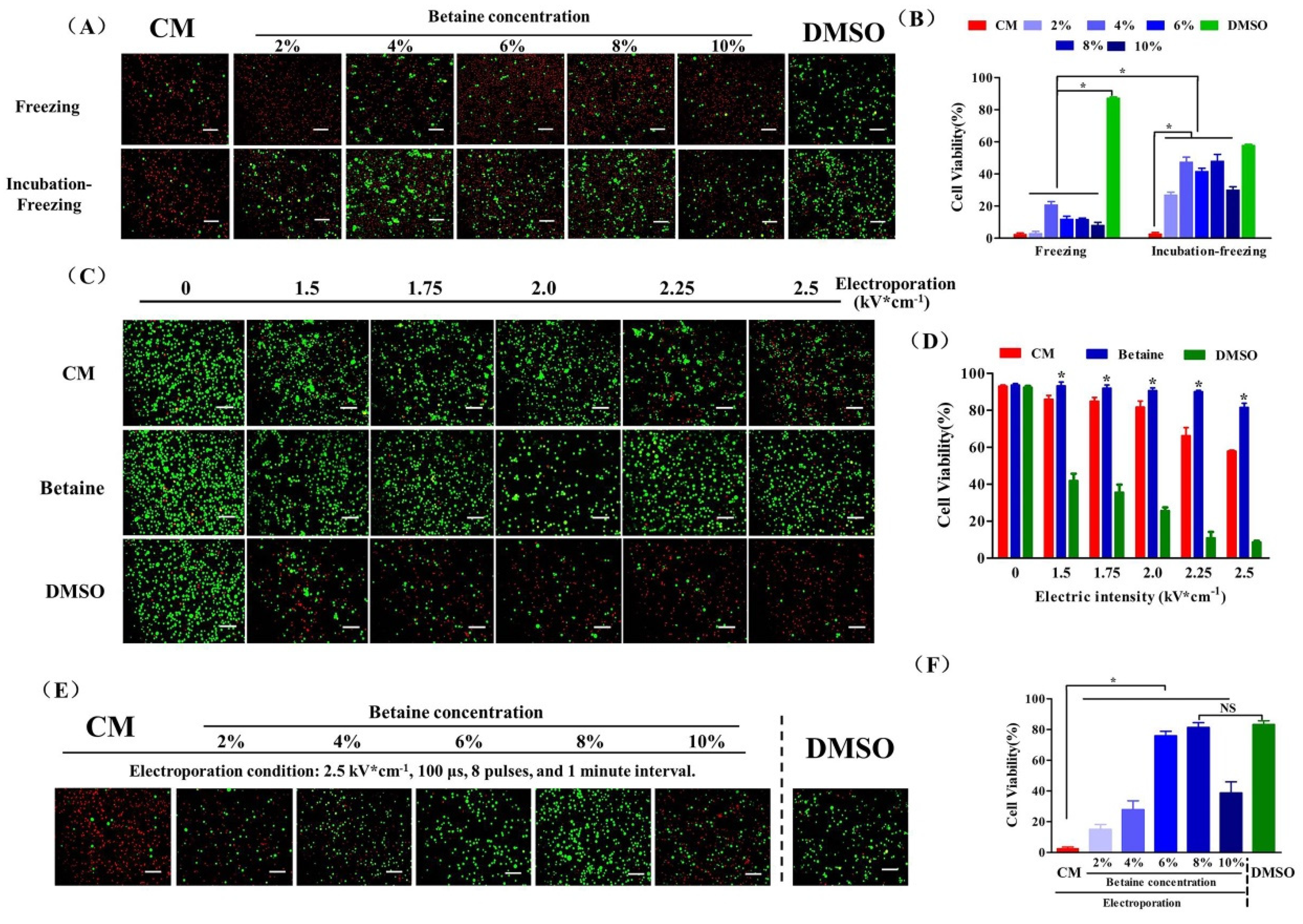
2.2. UCMSC Cryopreservation
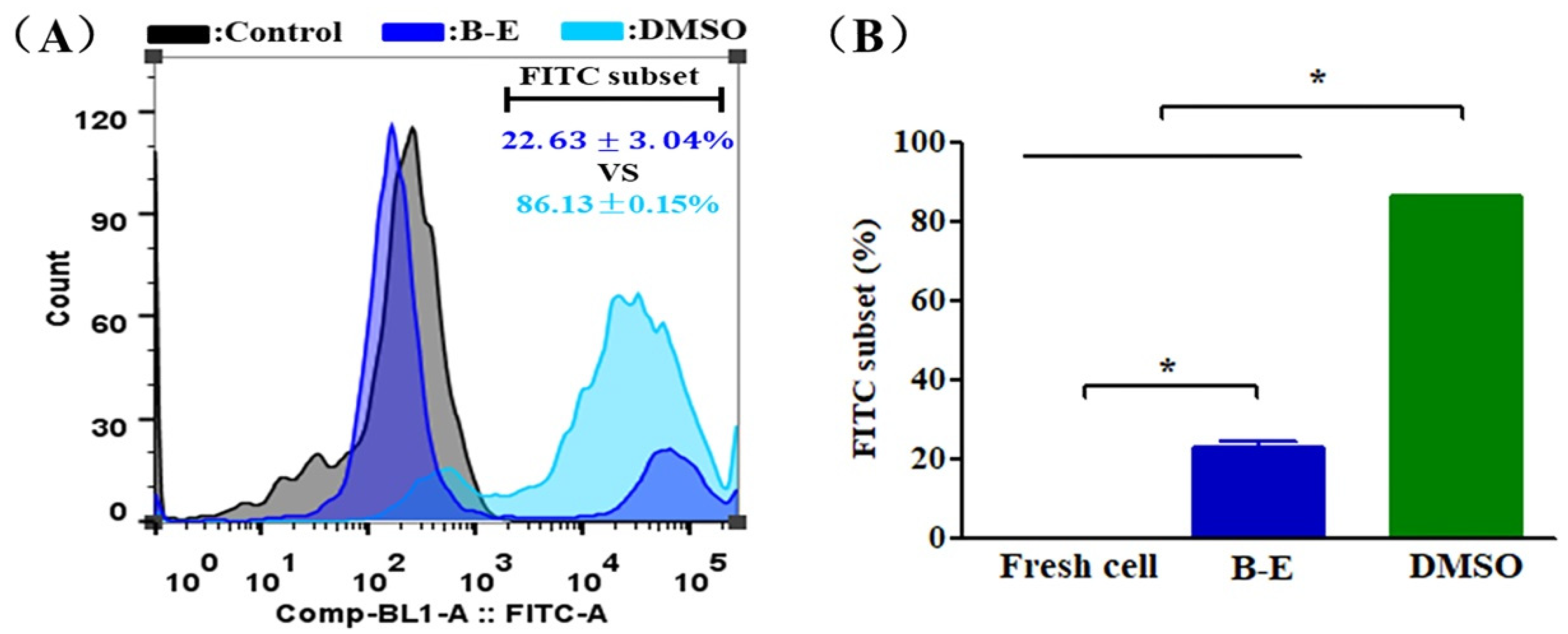
2.3. Evaluation of UCMSCs Functions
2.4. ROS Level of UCMSCs
2.5. Construction and Cryopreservation of GFP-Fluc-UCMSCs
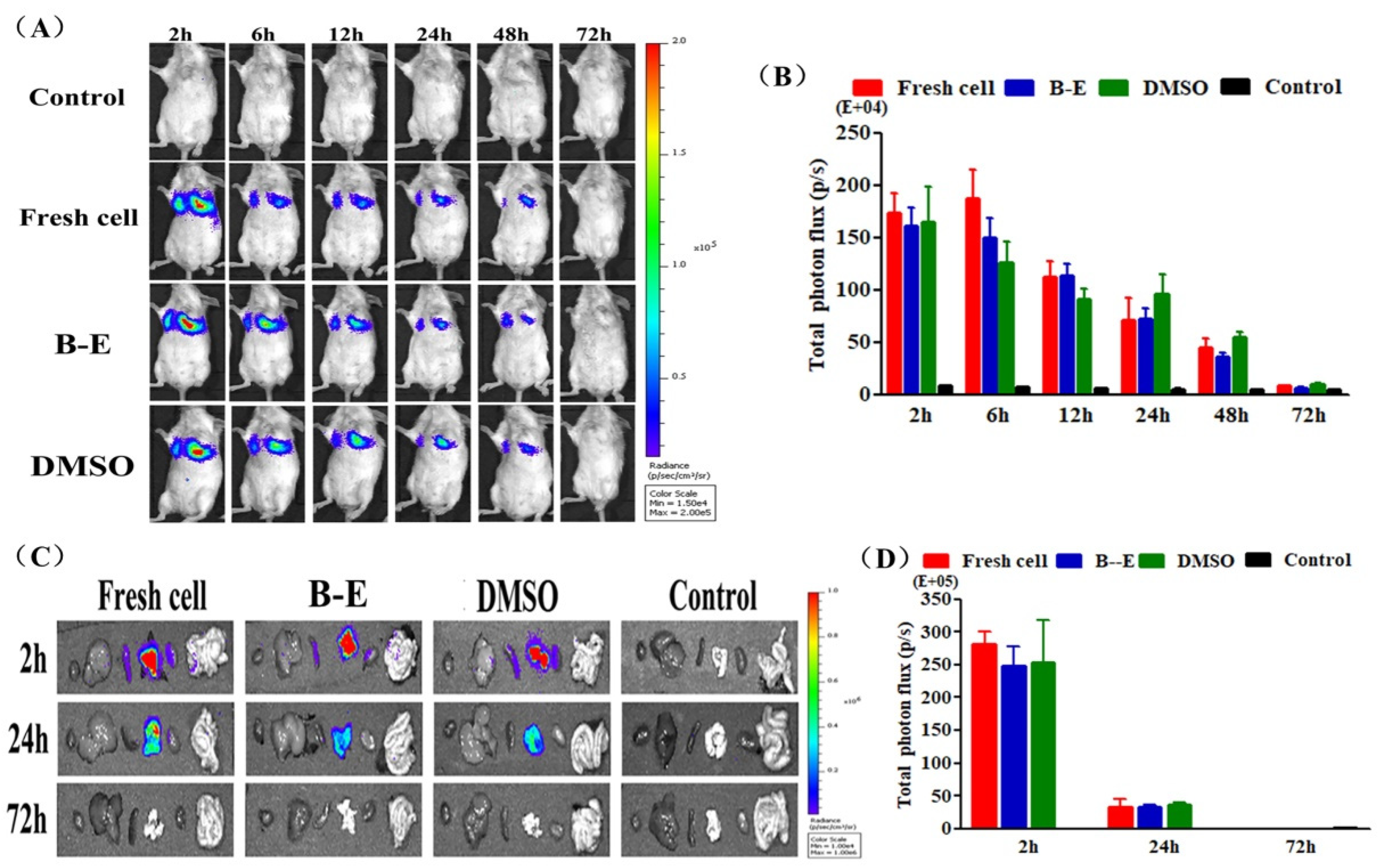
2.6. The In Vivo Distribution of GFP-Fluc-UCMSCs
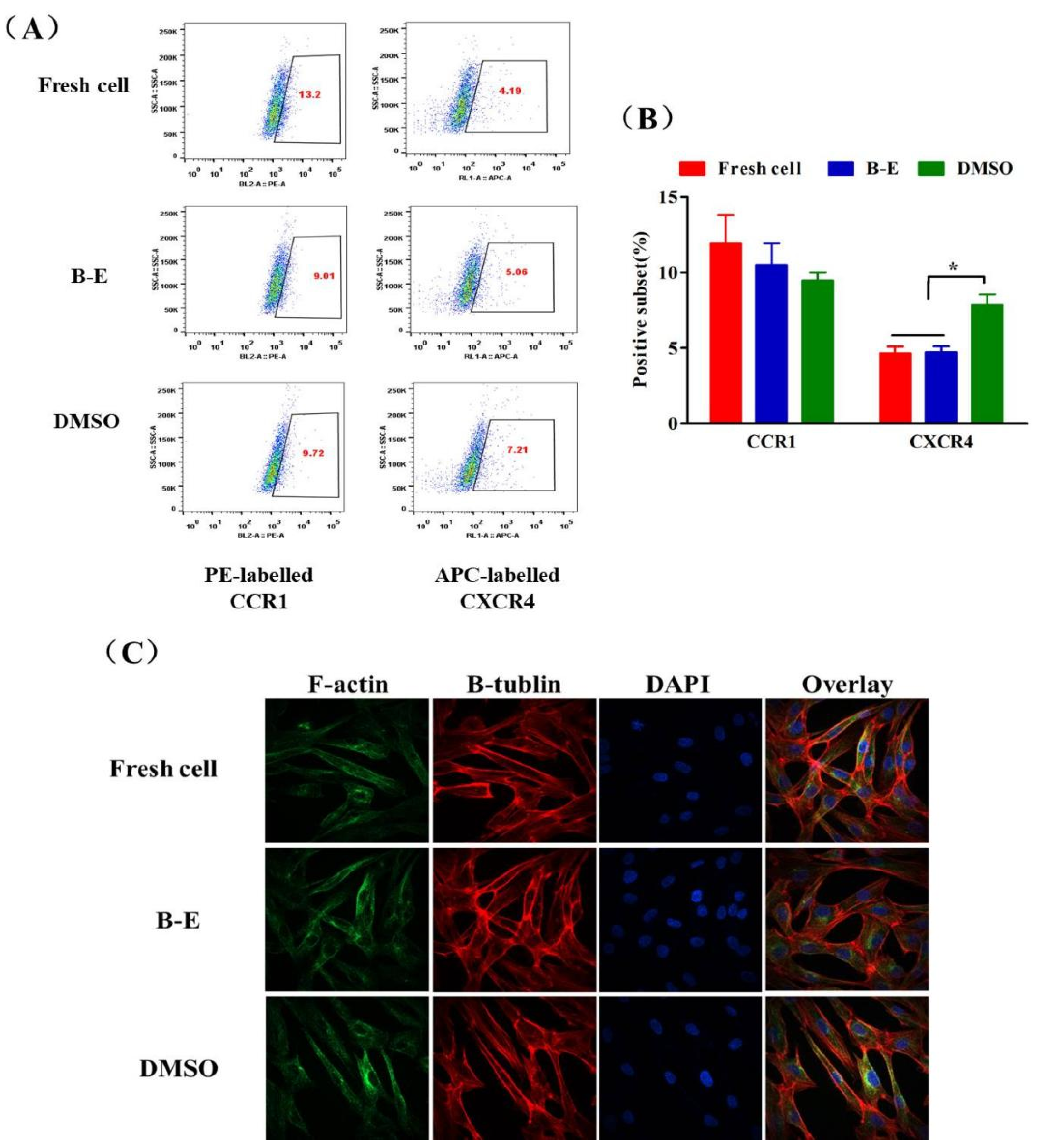
2.7. Chemokine Receptor Expression and Cytoskeleton Integrity of UCMSCs
3. Discussion
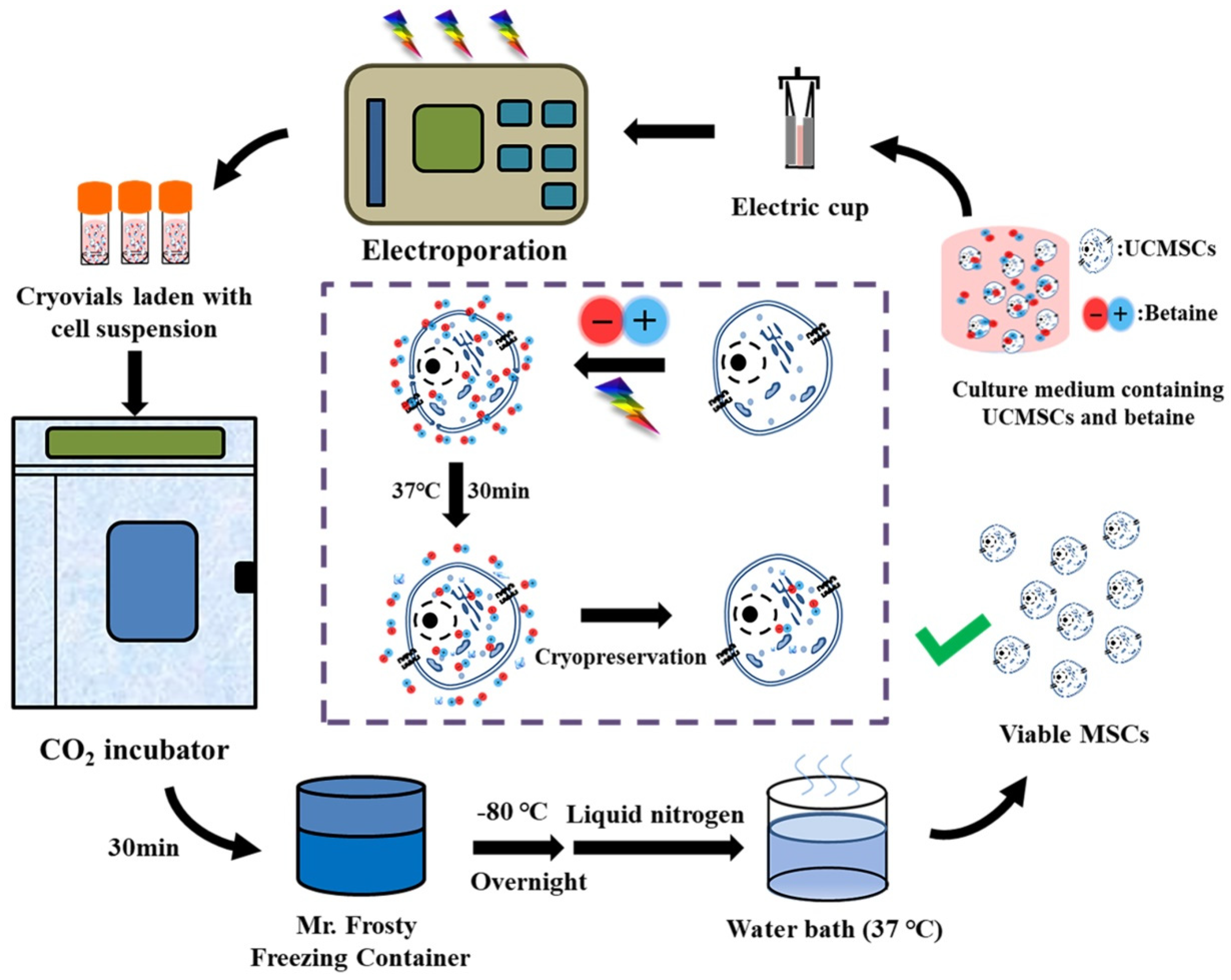
4. Materials and Method
4.1. Animals
4.2. Cell Preparation
4.3. DSC Test
4.4. Osmotic Regulation Test
4.5. Cytotoxicity
4.6. Influence of Electroporation on Cell Viability
4.7. Cell Cryopreservation
4.8. Cell Viability Assay
4.9. Functional Characterization
4.10. ROS Detection
4.11. BLI of GFP-Fluc-UCMSC Distribution In Vivo
4.12. Immunostaining of Cell Cytoskeleton
4.13. Chemokine Receptor Expression
4.14. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Castro, L.L.; Lopes-Pacheco, M.; Weiss, D.J.; Cruz, F.F.; Rocco, P.R.M. Current understanding of the immunosuppressive properties of mesenchymal stromal cells. J. Mol. Med. 2019, 97, 605–618. [Google Scholar] [CrossRef]
- Lo, W.C.; Chen, W.H.; Lin, T.C.; Hwang, S.M.; Zeng, R.; Hsu, W.C.; Chiang, Y.M.; Liu, M.C.; Williams, D.F.; Deng, W.P. Preferential therapy for osteoarthritis by cord blood MSCs through regulation of chondrogenic cytokines. Biomaterials 2013, 34, 4739–4748. [Google Scholar] [CrossRef] [PubMed]
- Pacini, S. Deterministic and stochastic approaches in the clinical application of mesenchymal stromal cells (MSCs). Front. Cell Dev. Biol. 2014, 2, 50. [Google Scholar] [CrossRef]
- Hunt, C.J. Cryopreservation of Human Stem Cells for Clinical Application: A Review. Transfus. Med. Hemotherapy 2011, 38, 107–123. [Google Scholar] [CrossRef] [PubMed]
- Gurruchaga, H.; del Burgo, L.S.; Hernandez, R.; Orive, G.; Selden, C.; Fuller, B.; Ciriza, J.; Pedraz, J. Advances in the slow freezing cryopreservation of microencapsulated cells. J. Control. Release Soc. 2018, 281, 119–138. [Google Scholar] [CrossRef]
- Zhao, G.; Fu, J. Microfluidics for cryopreservation. Biotechnol. Adv. 2017, 35, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Weng, L.; Beauchesne, P.R. Dimethyl sulfoxide-free cryopreservation for cell therapy: A review. Cryobiology 2020, 94, 9–17. [Google Scholar] [CrossRef]
- Verheijen, M.; Lienhard, M.; Schrooders, Y.; Clayton, O.; Nudischer, R.; Boerno, S.; Timmermann, B.; Selevsek, N.; Schlapbach, R.; Gmuender, H.; et al. DMSO induces drastic changes in human cellular processes and epigenetic landscape in vitro. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Windrum, P.; Subcommittee, O.B.O.T.E.C.L.W.P.C.; Morris, T.C.M.; Drake, M.B.; Niederwieser, D.; Ruutu, T. Variation in dimethyl sulfoxide use in stem cell transplantation: A survey of EBMT centres. Bone Marrow Transplant. 2005, 36, 601–603. [Google Scholar] [CrossRef] [PubMed]
- Woodbury, D.; Schwarz, E.J.; Prockop, D.J.; Black, I.B. Adult rat and human bone marrow stromal cells differentiate into neurons. J. Neurosci. Res. 2000, 61, 364–370. [Google Scholar] [CrossRef]
- Ntai, A.; La Spada, A.; De Blasio, P.; Biunno, I. Trehalose to cryopreserve human pluripotent stem cells. Stem Cell Res. 2018, 31, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Jha, A.; Bit, A.; Kiassov, A.P.; Rizvanov, A.A.; Ojha, A.; Bhoi, P.; Patra, P.K.; Kumar, A.; Bissoyi, A. Selaginella bryopteris Aqueous Extract Improves Stability and Function of Cryopreserved Human Mesenchymal Stem Cells. Oxidative Med. Cell. Longev. 2017, 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Biggs, C.I.; Bailey, T.L.; Graham, B.; Stubbs, C.; Fayter, A.; Gibson, M.I. Polymer mimics of biomacromolecular antifreezes. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- He, Z.; Liu, K.; Wang, J. Bioinspired Materials for Controlling Ice Nucleation, Growth, and Recrystallization. Acc. Chem. Res. 2018, 51, 1082–1091. [Google Scholar] [CrossRef]
- Matsumura, K.; Hyon, S.-H. Polyampholytes as low toxic efficient cryoprotective agents with antifreeze protein properties. Biomaterials 2009, 30, 4842–4849. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K.; Hayashi, F.; Nagashima, T.; Hyon, S.H. Long-term cryopreservation of human mesenchymal stem cells using carboxylated poly-l-lysine without the addition of proteins or dimethyl sulfoxide. Polymer 2013, 24, 1484–1497. [Google Scholar] [CrossRef]
- Jing, Y.; Lei, G.; Lei, Z. Advanced Biotechnology for Cell Cryopreservation. Trans. Tianjin Univ. 2020, 26, 409–423. [Google Scholar]
- Zhao, G.; Liu, X.; Zhu, K.; He, X. Hydrogel Encapsulation Facilitates Rapid-Cooling Cryopreservation of Stem Cell-Laden Core-Shell Microcapsules as Cell-Biomaterial Constructs. Adv. Healthc. Mater. 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- De Vries, R.J.; Banik, P.D.; Nagpal, S.; Weng, L.; Ozer, S.; Van Gulik, T.M.; Toner, M.; Tessier, S.N.; Uygun, K. Bulk Droplet Vitrification: An Approach to Improve Large-Scale Hepatocyte Cryopreservation Outcome. Langmuir 2018, 35, 7354–7363. [Google Scholar] [CrossRef]
- El Assal, R.; Guven, S.; Gurkan, U.A.; Gozen, I.; Shafiee, H.; Dalbeyler, S.; Abdalla, N.; Thomas, G.; Fuld, W.; Illigens, B.M.; et al. Bio-inspired cryoink preserves red blood cell phenotype and function during nanoliter vitrification. Adv. Mater. 2014, 26, 5815–5822. [Google Scholar] [CrossRef]
- Czajka, C. Nanowarming improves cryopreservation. Science 2017, 355, 920–921. [Google Scholar] [CrossRef]
- Bischof, J.C.; Diller, K.R. From Nanowarming to Thermoregulation: New Multiscale Applications of Bioheat Transfer. Annu. Rev. Biomed. Eng. 2018, 20, 301–327. [Google Scholar] [CrossRef] [PubMed]
- Assis, A.C.M.; Carvalho, J.; Jacoby, B.A.; Ferreira, R.L.B.; Castanheira, P.; Diniz, S.O.F.; Cardoso, V.N.; Goes, A.M.; Ferreira, A.J. Time-Dependent Migration of Systemically Delivered Bone Marrow Mesenchymal Stem Cells to the Infarcted Heart. Cell Transplant. 2010, 19, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wu, S.; Liu, Z.; Zhuo, Z.; Tan, K.; Xia, H.; Zhuo, L.; Deng, X.; Gao, Y.; Xu, Y. Ultrasound-Targeted Microbubble Destruction Improves the Migration and Homing of Mesenchymal Stem Cells after Myocardial Infarction by Upregulating SDF-1/CXCR4: A Pilot Study. Stem Cells Int. 2015, 2015, 691310. [Google Scholar] [CrossRef] [PubMed]
- Chinnadurai, R.; Garcia, M.A.; Sakurai, Y.; Lam, W.A.; Kirk, A.D.; Galipeau, J.; Copland, I.B. Actin cytoskeletal disruption following cryopreservation alters the biodistribution of human mesenchymal stromal cells in vivo. Stem Cell Rep. 2014, 3, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Castelo-Branco, M.T.L.; Soares, I.D.P.; Lopes, D.V.; Buongusto, F.; Martinusso, C.A.; Rosario, A.D.; Souza, S.; Gutfilen, B.; Fonseca, L.M.B.; Elia, C.; et al. Intraperitoneal but Not Intravenous Cryopreserved Mesenchymal Stromal Cells Home to the Inflamed Colon and Ameliorate Experimental Colitis. PLoS ONE 2012, 7, e33360. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Yang, J.; Zhang, J.; Pan, C.; Cai, N.; Zhu, Y.; Zhang, L. Natural zwitterionic l -Carnitine as efficient cryoprotectant for solvent-free cell cryopreservation. Biochem. Biophys. Res. Commun. 2017, 489, 76–82. [Google Scholar] [CrossRef]
- Yang, J.; Cai, N.; Zhai, H.; Zhang, J.; Zhu, Y.; Zhang, L. Natural zwitterionic betaine enables cells to survive ultrarapid cryopreservation. Sci. Rep. 2016, 6, 37458. [Google Scholar] [CrossRef]
- Shao, Q.; Jiang, S. Molecular understanding and design of zwitterionic materials. Adv. Mater. 2015, 27, 15–26. [Google Scholar] [CrossRef]
- Ratriyanto, A.; Mosenthin, R. Osmoregulatory function of betaine in alleviating heat stress in poultry. J. Anim. Physiol. Anim. Nutr. 2018, 102, 1634–1650. [Google Scholar] [CrossRef]
- Day, C.; Kempson, S.A. Betaine chemistry, roles, and potential use in liver disease. Biochim. Biophys. Acta (BBA) 2016, 1860, 1098–1106. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Zhang, X.; Guo, H.; Zhu, Y.; Wen, C.; Sui, X.; Yang, J. Dimethyl Sulfoxide-Free Cryopreservation of Chondrocytes Based on Zwitterionic Molecule and Polymers. Biomacromolecules 2019, 20, 3980–3988. [Google Scholar]
- Napotnik, T.B.; Miklavčič, D. In vitro electroporation detection methods—An overview. Bioelectrochemistry 2018, 120, 166–182. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Ma, E.; Cestellos-Blanco, S.; Zhang, B.; Qiu, R.; Su, Y.; Doudna, J.A.; Yang, P. Nontoxic nanopore electroporation for effective intracellular delivery of biological macromolecules. Proc. Natl. Acad. Sci. USA 2019, 116, 7899–7904. [Google Scholar] [CrossRef] [PubMed]
- Saulis, G.; Saule, R. Size of the pores created by an electric pulse: Microsecond vs millisecond pulses. Biochim. Biophys. Acta (BBA) 2012, 1818, 3032–3039. [Google Scholar] [CrossRef] [PubMed]
- Hatley, R.H.M.; Berg, C.v.d.; Franks, F. The unfrozen water content of maximally freeze concentrated solutions: Validity of the methods used for its determination. Cryo-Letters 1991, 12, 113–124. [Google Scholar]
- Li, C.; Li, X.; Tao, C.; Ren, L.; Zhao, Y.; Bai, S.; Yuan, X. Amphiphilic Antifogging/Anti-Icing Coatings Containing POSS-PDMAEMA-b-PSBMA. Appl. Mater. Interfaces 2017, 9, 22959–22969. [Google Scholar] [CrossRef] [PubMed]
- Weise, U.; Maloney, T.; Paulapuro, H. Quantification of water in different states of interaction with wood pulp fibres. Cellulose 1996, 3, 189–202. [Google Scholar] [CrossRef]
- Xiaojie, S.; Chiyu, W.; Jing, Y. Betaine combined with membrane stabilizers enables solvent-free whole blood cryopreservation and one-step cryoprotectant removal. ACS Biomater. Eng. 2018, 5, 125–142. [Google Scholar]
- Zhou, Q.; Zhang, Y.; Zhao, M.; Wang, X.; Ma, C.; Jiang, X.; Wu, T.; Wang, D.; Zhan, L. Mature dendritic cell derived from cryopreserved immature dendritic cell shows impaired homing ability and reduced anti-viral therapeutic effects. Sci. Rep. 2016, 6, 39071. [Google Scholar] [CrossRef]
- Ma, T.; Luan, S.-L.; Huang, H.; Sun, X.-K.; Yang, Y.-M.; Zhang, H.; Han, W.-D.; Li, H.; Han, Y. Upregulation of CC Chemokine Receptor 7 (CCR7) Enables Migration of Xenogeneic Human Adipose-Derived Mesenchymal Stem Cells to Rat Secondary Lymphoid Organs. Med. Sci. Monit. 2016, 22, 5206–5217. [Google Scholar] [CrossRef][Green Version]
- Huang, J.; Zhang, Z.; Guo, J.; Ni, A.; Deb, A.; Zhang, L.; Mirotsou, M.; Pratt, R.E.; Dzau, V.J. Genetic modification of mesenchymal stem cells overexpressing CCR1 increases cell viability, migration, engraftment, and capillary density in the injured myocardium. Circ. Res. 2010, 106, 1753–1762. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Song, X.Y.; He, M.; Yu, S.B. Effect of TGF-β1/SDF-1/CXCR4 signal on BM-MSCs homing in rat heart of ischemia/perfusion injury. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 899–905. [Google Scholar] [PubMed]
- Son, B.R.; Marquez-Curtis, L.A.; Kucia, M.; Wysoczynski, M.; Turner, A.R.; Ratajczak, J.; Ratajczak, M.Z.; Janowska-Wieczorek, A. Migration of bone marrow and cord blood mesenchymal stem cells in vitro is regulated by stromal-derived factor-1-CXCR4 and hepatocyte growth factor-c-met axes and involves matrix metalloproteinases. Stem Cells 2006, 24, 1254–1264. [Google Scholar] [CrossRef]
- Mazur, P.; Klein, D.C.; Berg, G.R.; Weller, J. Cryobiology: The Freezing of Biological Systems. Science 1970, 168, 939–949. [Google Scholar] [CrossRef]
- Mazur, P. Freezing of living cells: Mechanisms and implications. Am. J. Physiol. Physiol. 1984, 247, C125–C142. [Google Scholar] [CrossRef]
- Slade, L.; Levine, H. Beyond water activity: Recent advances based on an alternative approach to the assessment of food quality and safety. Crit. Rev. Food Sci. Nutr. 1991, 30, 115–360. [Google Scholar] [CrossRef]
- Zhao, G.; He, F.; Wu, C.; Li, P.; Li, N.; Deng, J.; Zhu, G.; Ren, W.; Peng, Y. Betaine in Inflammation: Mechanistic Aspects and Applications. Front. Immunol. 2018, 9, 1070. [Google Scholar] [CrossRef]
- Nakanishi, T.; Turner, R.J.; Burg, M.B. Osmoregulation of betaine transport in mammalian renal medullary cells. Am. J. Physiol. Physiol. 1990, 258, F1061–F1067. [Google Scholar] [CrossRef]
- Elliott, G.D.; Wang, S.; Fuller, B.J. Cryoprotectants: A review of the actions and applications of cryoprotective solutes that modulate cell recovery from ultra-low temperatures. Cryobiology 2017, 76, 74–91. [Google Scholar] [CrossRef] [PubMed]
- Stefanovich, P.; Ezzell, R.; Sheehan, S.; Tompkins, R.; Yarmush, M.; Toner, M. Effects of Hypothermia on the Function, Membrane Integrity, and Cytoskeletal Structure of Hepatocytes. Cryobiology 1995, 32, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Fuller, B.J.; Petrenko, A.; Rodriguez, J.V.; Somov, A.Y.; Balaban, C.L.; Guibert, E.E. Biopreservation of hepatocytes: Current concepts on hypothermic preservation, cryopreservation, and vitrification. CryoLetters 2013, 34, 432–452. [Google Scholar]
- Amidi, F.; Pazhohan, A.; Nashtaei, M.S.; Khodarahmian, M.; Nekoonam, S. The role of antioxidants in sperm freezing: A review. Cell Tissue Bank. 2016, 17, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Huang, L.; Gao, J.; Wen, S.; Tai, Y.; Chen, M.; Huang, Z.; Liu, R.; Tang, C.; Li, J. Betaine attenuates chronic alcohol-induced fatty liver by broadly regulating hepatic lipid metabolism. Mol. Med. Rep. 2017, 16, 5225–5234. [Google Scholar] [CrossRef]
- Bruździak, P.; Panuszko, A.; Stangret, J. Influence of osmolytes on protein and water structure: A step to understanding the mechanism of protein stabilization. J. Phys. Chem. B 2013, 117, 11502–11508. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.J.; Lee, H.W.; Kalimuthu, S.; Kim, T.J.; Kim, H.M.; Baek, S.H.; Zhu, L.; Oh, J.M.; Son, S.H.; Chung, H.Y.; et al. In vivo migration of mesenchymal stem cells to burn injury sites and their therapeutic effects in a living mouse model. J. Control. Release 2018, 279, 79–88. [Google Scholar] [CrossRef]
- Kalimuthu, S.; Zhu, L.; Oh, J.M.; Gangadaran, P.; Lee, H.W.; Baek, S.H.; Rajendran, R.L.; Gopal, A.; Jeong, S.Y.; Lee, S.-W.; et al. Migration of mesenchymal stem cells to tumor xenograft models and in vitro drug delivery by doxorubicin. Int. J. Med. Sci. 2018, 15, 1051–1061. [Google Scholar] [CrossRef]
- Roerig, D.L.; Kotrly, K.J.; Vucins, E.J.; Ahlf, S.B.; Dawson, C.A.; Kampine, J.P. First Pass Uptake of Fentanyl, Meperidine, and Morphine in the Human Lung. Anesthesiology 1987, 67, 466–472. [Google Scholar] [CrossRef]
- Müllers, Y.; Meiser, I.; Stracke, F.; Riemann, I.; Lautenschläger, F.; Neubauer, J.C.; Zimmermann, H. Quantitative analysis of F-actin alterations in adherent human mesenchymal stem cells: Influence of slow-freezing and vitrification-based cryopreservation. PLoS ONE 2019, 14, e0211382. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, L.; Zhou, Q.; Zhang, Y.; Sun, S.; Lv, L.; Ma, P.; Yang, J.; Liu, M.; Zhang, L.; Wang, X.; et al. Dimethyl Sulfoxide-Free Cryopreservation of Human Umbilical Cord Mesenchymal Stem Cells Based on Zwitterionic Betaine and Electroporation. Int. J. Mol. Sci. 2021, 22, 7445. https://doi.org/10.3390/ijms22147445
Gao L, Zhou Q, Zhang Y, Sun S, Lv L, Ma P, Yang J, Liu M, Zhang L, Wang X, et al. Dimethyl Sulfoxide-Free Cryopreservation of Human Umbilical Cord Mesenchymal Stem Cells Based on Zwitterionic Betaine and Electroporation. International Journal of Molecular Sciences. 2021; 22(14):7445. https://doi.org/10.3390/ijms22147445
Chicago/Turabian StyleGao, Lei, Qianqian Zhou, Yulong Zhang, Sujing Sun, Liping Lv, Ping Ma, Jing Yang, Min Liu, Lei Zhang, Xiaohui Wang, and et al. 2021. "Dimethyl Sulfoxide-Free Cryopreservation of Human Umbilical Cord Mesenchymal Stem Cells Based on Zwitterionic Betaine and Electroporation" International Journal of Molecular Sciences 22, no. 14: 7445. https://doi.org/10.3390/ijms22147445
APA StyleGao, L., Zhou, Q., Zhang, Y., Sun, S., Lv, L., Ma, P., Yang, J., Liu, M., Zhang, L., Wang, X., & Zhan, L. (2021). Dimethyl Sulfoxide-Free Cryopreservation of Human Umbilical Cord Mesenchymal Stem Cells Based on Zwitterionic Betaine and Electroporation. International Journal of Molecular Sciences, 22(14), 7445. https://doi.org/10.3390/ijms22147445





