Natural Phytochemicals Derived from Gymnosperms in the Prevention and Treatment of Cancers
Abstract
:1. Introduction
2. Gymnosperms in Cancer Therapy
3. Clinically Available Natural Products
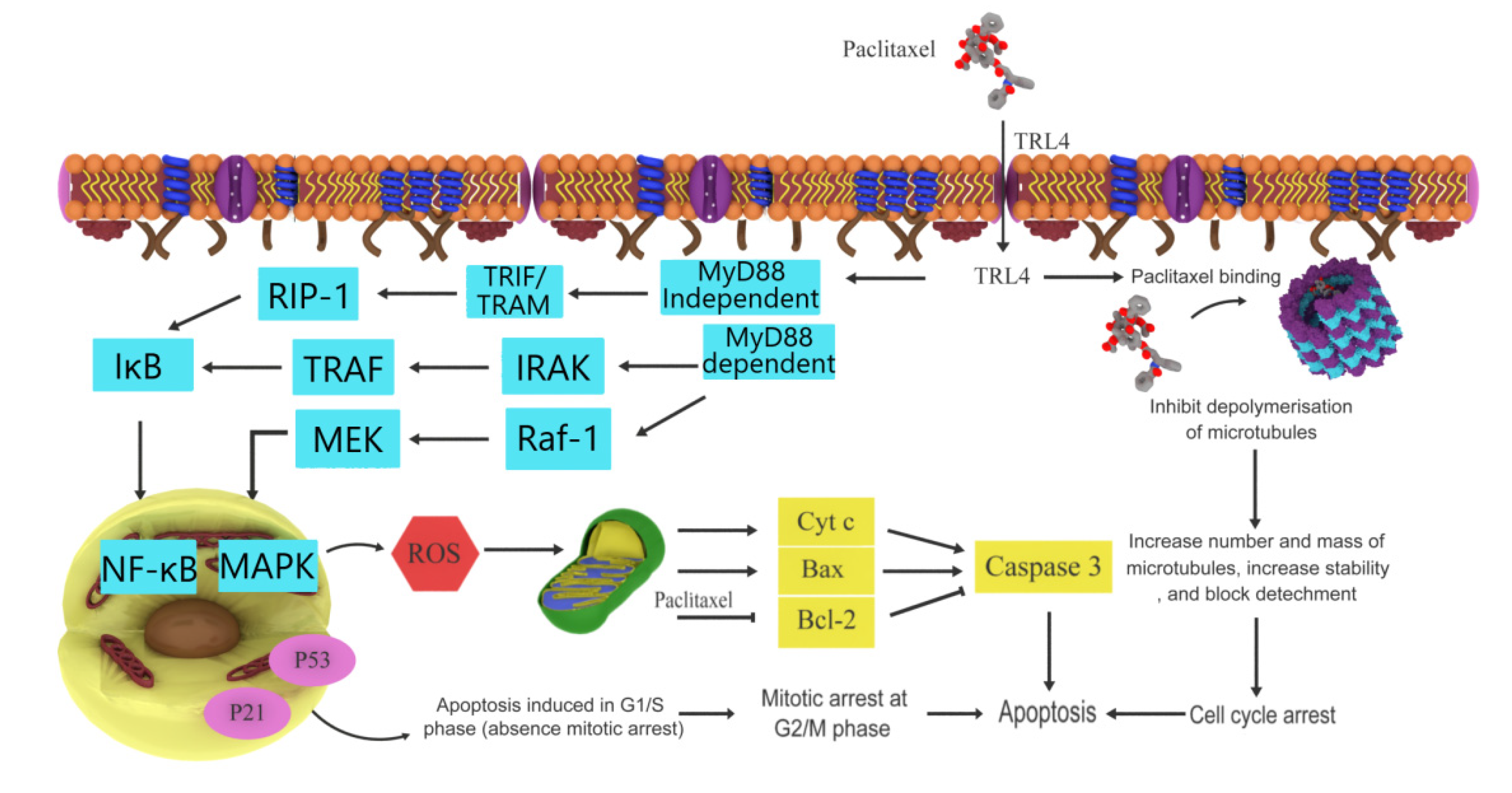
3.1. Paclitaxel
3.2. Pinostrobin
3.3. Pycnogenol
3.4. Enzogenol
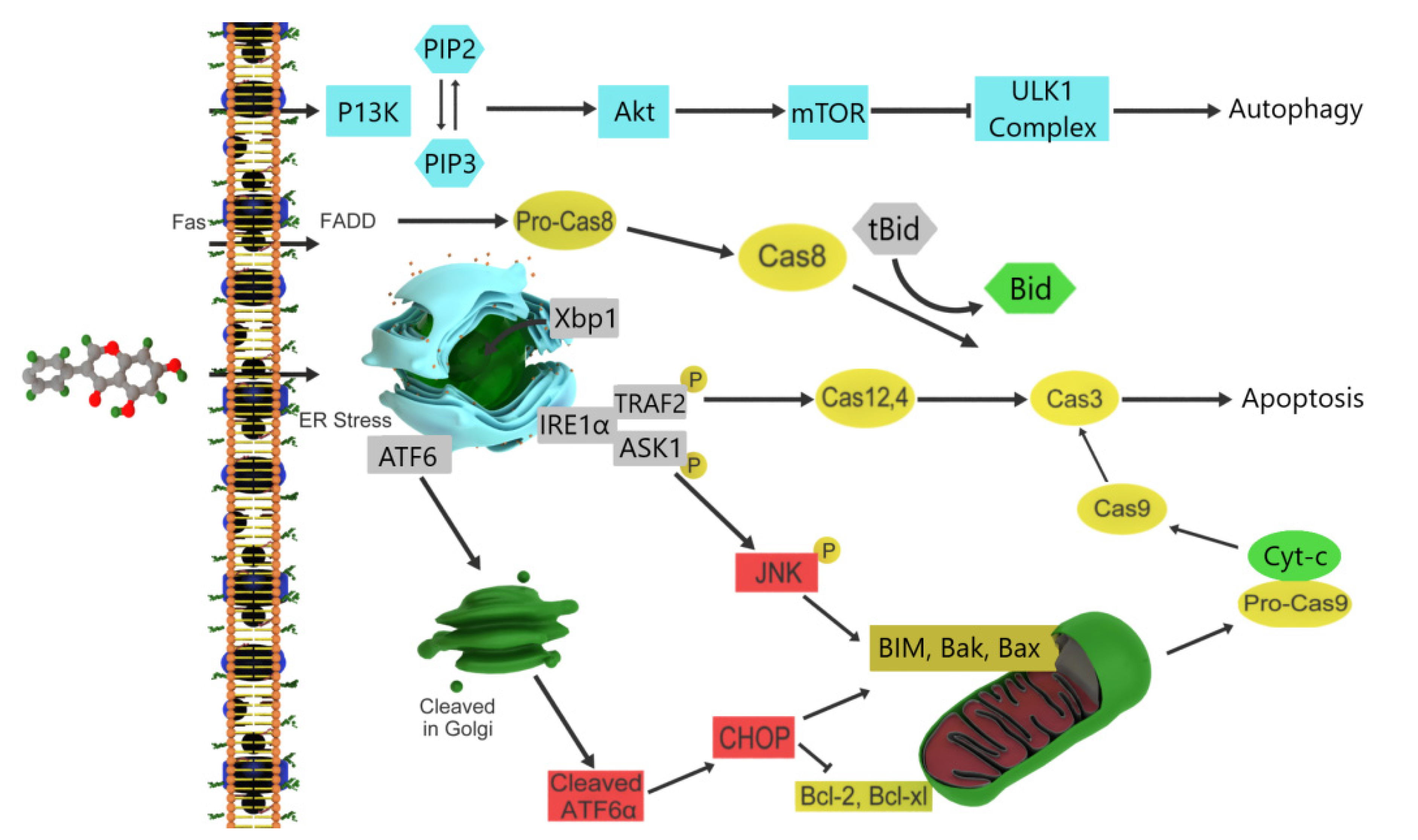
3.5. Pinocembrin
4. The Future Role of Gymnosperms in Cancer Therapy
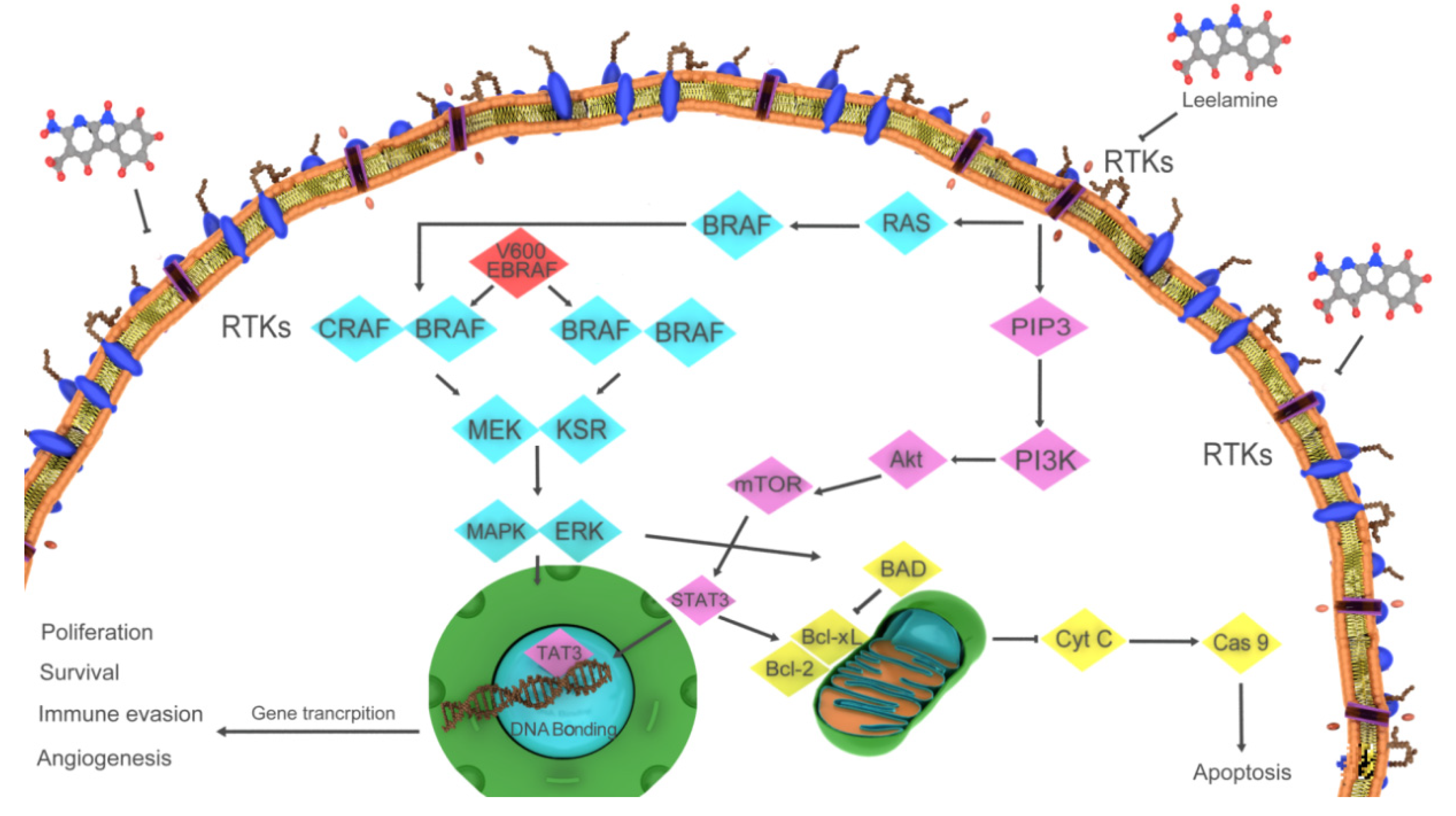
4.1. Leelamine
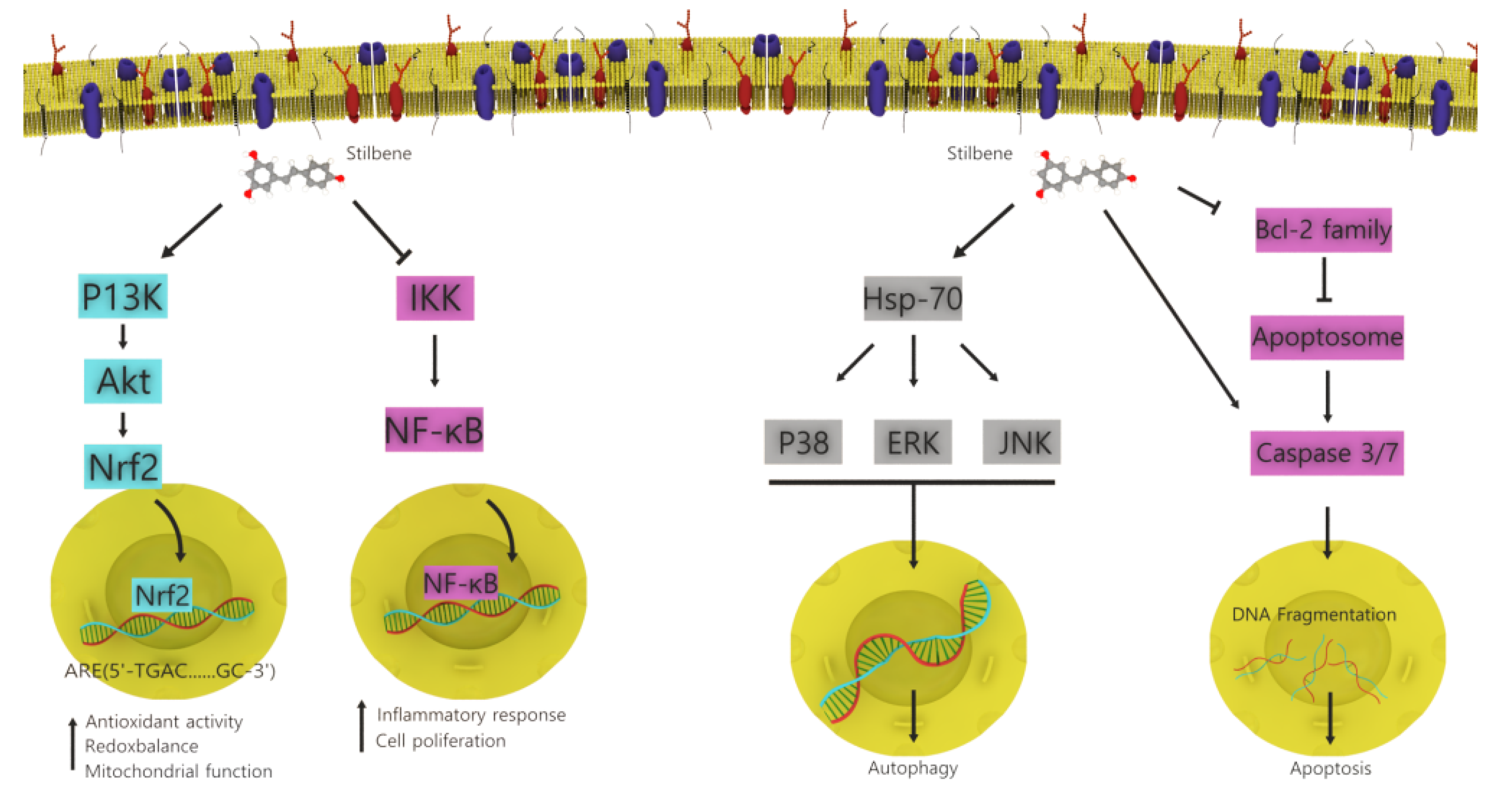
4.2. Stilbenoids
4.3. Pinenes
4.4. Caryophyllenes
5. Conclusions and Future Development
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Akt | protein kinase B |
| ASK1 | apoptosis signal-regulating kinase 1 |
| ATF6 | activating transcription factor 6 |
| Bak | B-cell lymphoma-2 homologous antagonist/killer |
| Bax | Bcl-2 associated X protein |
| Bcl-2 | B cell lymphoma 2 |
| Bcl-xL | B-cell lymphoma-extra-large |
| BIM | B-cell lymphoma-2-like protein 11 |
| Cas | cellular apoptosis susceptibility protein |
| caspase | cysteine-aspartic proteases |
| CDKs | Cyclin-dependent kinases |
| CHOP | C/EBP-homologous protein |
| Cyt c | cytochrome c |
| ER | endoplasmic reticulum |
| ERK | extracellular signal-regulated kinase |
| FADD | Fas-associated protein death domain |
| G1 | Gap 1 phase for cell enlargement |
| G2 | Gap 2 phase |
| Hsp 70 | heat shock protein 70 |
| ICAM-1 | intercellular adhesion molecule 1 |
| IKK | NF-κB kinase |
| IRAK | interleukin-1 receptor associated kinase |
| IRE1 | inositol-requiring enzyme 1 |
| JNK | c-Jun-N terminal kinase |
| KSR | kinase suppressor of Ras |
| MAPK | mitogen-activated protein kinase |
| MEK | mitogen-activated protein kinase kinase |
| M phase | mitosis phase |
| mTOR | mammalian target of rapamycin |
| MYD88 | myeloid differentiation primary response 88 |
| NFκB | Nuclear factor-κB pathway |
| Nrf2 | Nuclear factor erythroid 2-related factor 2 |
| PI3K | phosphoinositide 3-kinase |
| PIP2 | phosphatidylinositol-4, 5-bisphosphate |
| PIP3 | phosphatidylinositol-3, 4, 5-triphosphate |
| Raf-1 | Raf kinase family |
| ROS | reactive oxygen species |
| RTK | receptor tyrosine kinase |
| S phase | synthesis phase |
| STAT3 | signal transducer and activator of transcription 3 |
| TAT3 | tyrosine aminotransferase 3 |
| tBid | truncated Bid |
| TLR4 | Toll-like receptor 4 |
| TRAF | TNF receptor associated factor |
| TRIF | TIR-domain-containing adapter-inducing interferon-β |
| TRAM | TRIF-related adapter molecule |
| ULK1 | unc-51-like kinase 1 |
| VCAM-1 | vascular cell adhesion molecule 1 |
| Xbp1 | X-box binding protein 1 |
References
- Sheikh, I.; Sharma, V.; Tuli, H.S.; Aggarwal, D.; Sankhyan, A.; Vyas, P.; Sharma, A.K.; Bishayee, A. Cancer chemoprevention by flavonoids, dietary polyphenols and terpenoids. Biointerface Res. Appl. Chem. 2020, 11, 8502–8537. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [Green Version]
- Loizzo, M.R.; Tundis, R.; Menichini, F.; Saab, A.M.; Statti, G.A.; Menichini, F. Antiproliferative effects of essential oils and their major constituents in human renal adenocarcinoma and amelanotic melanoma cells. Cell Prolif. 2008, 41, 1002–1012. [Google Scholar] [CrossRef]
- Aoki, H.; Takada, Y.; Kondo, S.; Sawaya, R.; Aggarwal, B.B.; Kondo, Y. Evidence that curcumin suppresses the growth of malignant gliomas in vitro and in vivo through induction of autophagy: Role of akt and extracellular signal-regulated kinase signaling pathways. Mol. Pharmacol. 2007, 72, 29–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhuang, W.; Long, L.; Zheng, B.; Ji, W.; Yang, N.; Zhang, Q.; Liang, Z. Curcumin promotes differentiation of glioma-initiating cells by inducing autophagy. Cancer Sci. 2012, 103, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yu, T.; Zhu, C.; Sun, H.; Qiu, Y.; Zhu, X.; Li, J. Resveratrol triggers protective autophagy through the ceramide/Akt/mTOR pathway in melanoma B16 cells. Nutr. Cancer 2014, 66, 435–440. [Google Scholar] [CrossRef]
- Ling, H.; Yang, H.; Tan, S.H.; Chui, W.K.; Chew, E.H. 6-Shogaol, an active constituent of ginger, inhibits breast cancer cell invasion by reducing matrix metalloproteinase-9 expression via blockade of nuclear factor-κB activation. Br. J. Pharmacol. 2010, 161, 1763–1777. [Google Scholar] [CrossRef] [Green Version]
- Zi, X.; Feyes, D.K.; Agarwal, R. Anticarcinogenic effect of a flavonoid antioxidant, silymarin, in human breast cancer cells MDA-MB 468: Induction of G1 arrest through an increase in Cipl/p21 concomitant with a decrease in kinase activity of cyclin- dependent kinases and associated cyclins. Clin. Cancer Res. 1998, 4, 1055–1064. [Google Scholar]
- Choi, J.A.; Kim, J.Y.; Lee, J.Y.; Kang, C.M.; Kwon, H.J.; Yoo, Y.D.; Kim, T.W.; Lee, Y.S.; Lee, S.J. Induction of cell cycle arrest and apoptosis in human breast cancer cells by quercetin. Int. J. Oncol. 2001, 19, 837–844. [Google Scholar] [CrossRef]
- Casagrande, F.; Darbon, J.M. Effects of structurally related flavonoids on cell cycle progression of human melanoma cells: Regulation of cyclin-dependent kinases CDK2 and CDK1. Biochem. Pharmacol. 2001, 61, 1205–1215. [Google Scholar] [CrossRef]
- Manohar, M.; Fatima, I.; Saxena, R.; Chandra, V.; Sankhwar, P.L.; Dwivedi, A. (-)-Epigallocatechin-3-gallate induces apoptosis in human endometrial adenocarcinoma cells via ROS generation and p38 MAP kinase activation. J. Nutr. Biochem. 2013, 24, 940–947. [Google Scholar] [CrossRef]
- Kampan, N.C.; Madondo, M.T.; McNally, O.M.; Quinn, M.; Plebanski, M. Paclitaxel and its evolving role in the management of ovarian cancer. BioMed Res. Int. 2015, 2015, 413076. [Google Scholar] [CrossRef] [PubMed]
- Kuzu, O.F.; Gowda, R.; Sharma, A.; Robertson, G.P. Leelamine mediates cancer cell death through inhibition of intracellular cholesterol transport. Mol. Cancer Ther. 2014, 13, 1690–1703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, W.; Liu, Y.; Li, M.; Mao, J.; Zhang, L.; Huang, R.; Jin, X.; Ye, L. Anti-tumor effect of α-pinene on human hepatoma cell lines through inducing G2/M cell cycle arrest. J. Pharmacol. Sci. 2015, 127, 332–338. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.L.; Yeung, C.M.; Chiu, L.C.M.; Cen, Y.Z.; Ooi, V.E.C. Chemical composition and antiproliferative activity of essential oil from the leaves of a medicinal herb, schefflera Heptaphylla. Phytother. Res. 2009, 23, 140–142. [Google Scholar] [CrossRef]
- Lin, J.J.; Lu, K.W.; Ma, Y.S.; Tang, N.Y.; Wu, P.P.; Wu, C.C.; Lu, H.F.; Lin, J.G.; Chung, J.G. Alpha-phellandrene, a natural active monoterpene, influences a murine WEHI-3 leukemia model in vivo by enhancing macrophague phagocytosis and natural killer cell activity. In Vivo 2014, 28, 583–588. [Google Scholar]
- Shapira, S.; Pleban, S.; Kazanov, D.; Tirosh, P.; Arber, N. Terpinen-4-ol: A novel and promising therapeutic agent for human gastrointestinal cancers. PLoS ONE 2016, 11, e0156540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sajid, A.; Manzoor, Q.; Iqbal, M.; Tyagi, A.K.; Sarfraz, R.A.; Sajid, A. Pinus roxburghii essential oil anticancer activity and chemical composition evaluation. EXCLI J. 2018, 17, 233–245. [Google Scholar]
- Tilaoui, M.; Mouse, H.A.; Jaafari, A.; Aboufatima, R.; Chait, A.; Zyad, A. Chemical composition and antiproliferative activity of essential oil from aerial parts of a medicinal herb Artemisia herba-alba. Braz. J. Pharmacogn. 2011, 21, 781–785. [Google Scholar] [CrossRef] [Green Version]
- Ghaffari, T.; Kafil, H.S.; Asnaashari, S.; Farajnia, S.; Delazar, A.; Baek, S.C.; Hamishehkar, H.; Kim, K.H. Chemical Composition and Antimicrobial Activity of Essential Oils from the Aerial Parts of Pinus eldarica Grown in Northwestern Iran. Molecules 2019, 24, 3203. [Google Scholar] [CrossRef] [Green Version]
- Simpson, M. Plant Systematics, 2nd ed.; Academic Press: Cambridge, MA, USA, 2010; pp. 1–741. [Google Scholar]
- Christenhusz, M.J.M.; Reveal, J.L.; Farjon, A.; Gardner, M.F.; Mill, R.R.; Chase, M.W. A new classification and linear sequence of extant gymnosperms. Phytotaxa 2011, 19, 55–70. [Google Scholar] [CrossRef]
- Meher, J.G.; Dixit, S.; Pathan, D.K.; Singh, Y.; Chandasana, H.; Pawar, V.K.; Sharma, M.; Bhatta, R.S.; Konwar, R.; Kesharwani, P.; et al. Paclitaxel-loaded TPGS enriched self-emulsifying carrier causes apoptosis by modulating survivin expression and inhibits tumour growth in syngeneic mammary tumours. Artif. Cells Nanomed. Biotechnol. 2018, 46 (Suppl. S3), S344–S358. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feroz, S.R.; Mohamad, S.B.; Bakri, Z.S.D.; Malek, S.N.A.; Tayyab, S. Probing the Interaction of a Therapeutic Flavonoid, Pinostrobin with Human Serum Albumin: Multiple Spectroscopic and Molecular Modeling Investigations. PLoS ONE 2013, 8, e76067. [Google Scholar] [CrossRef] [Green Version]
- Le Bail, J.C.; Aubourg, L.; Habrioux, G. Effects of pinostrobin on estrogen metabolism and estrogen receptor transactivation. Cancer Lett. 2000, 156, 37–44. [Google Scholar] [CrossRef]
- Wu, N.; Kong, Y.; Zu, Y.; Fu, Y.; Liu, Z.; Meng, R.; Liu, X.; Efferth, T. Activity investigation of pinostrobin towards herpes simplex virus-1 as determined by atomic force microscopy. Phytomedicine 2011, 18, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Jaudan, A.; Sharma, S.; Abd Malek, S.N.; Dixit, A. Induction of apoptosis by pinostrobin in human cervical cancer cells: Possible mechanism of action. PLoS ONE 2018, 13, e0191523. [Google Scholar] [CrossRef] [Green Version]
- Rasul, A.; Millimouno, F.M.; Ali Eltayb, W.; Ali, M.; Li, J.; Li, X. Pinocembrin: A novel natural compound with versatile pharmacological and biological activities. BioMed Res. Int. 2013, 2013, 379850. [Google Scholar] [CrossRef]
- Enseleit, F.; Sudano, I.; Périat, D.; Winnik, S.; Wolfrum, M.; Flammer, A.J.; Fröhlich, G.M.; Kaiser, P.; Hirt, A.; Haile, S.R.; et al. Effects of Pycnogenol on endothelial function in patients with stable coronary artery disease: A double-blind, randomized, placebo-controlled, cross-over study. Eur. Heart J. 2012, 33, 1589–1597. [Google Scholar] [CrossRef] [Green Version]
- Virgili, F.; Kobuchi, H.; Packer, L. Procyanidins extracted from pinus Maritima (Pycnogenol®): Scavengers of free radical species and modulators of nitrogen monoxide metabolism in activated murine raw 264.7 macrophages. Free Radic. Biol. Med. 1998, 24, 1120–1129. [Google Scholar] [CrossRef]
- Nelson, A.B.; Lau, B.H.S.; Ide, N.; Rong, Y. Pycnogenol inhibits macrophage oxidative burst, lipoprotein oxidation, and hydroxyl radical-induced DNA damage. Drug Dev. Ind. Pharm. 1998, 24, 139–144. [Google Scholar] [CrossRef]
- Wei, Z.H.; Peng, Q.L.; Lau, B.H.S. Pycnogenol enhances endothelial cell antioxidant defenses. Redox Rep. 1997, 3, 219–224. [Google Scholar] [CrossRef]
- Li, Y.Y.; Feng, J.; Zhang, X.L.; Cui, Y.Y. Pine bark extracts: Nutraceutical, pharmacological, and toxicological evaluation. J. Pharmacol. Exp. Ther. 2015, 353, 9–16. [Google Scholar] [CrossRef] [Green Version]
- Iliya, I.; Akao, Y.; Matsumoto, K.; Nakagawa, Y.; Zulfiqar, A.; Ito, T.; Oyama, M.; Murata, H.; Tanaka, T.; Nozawa, Y.; et al. Growth inhibition of stilbenoids in Welwitschiaceae and Gnetaceae through induction of apoptosis in human leukemia HL60 cells. Biol. Pharm. Bull. 2006, 29, 1490–1492. [Google Scholar] [CrossRef] [Green Version]
- Chidambara Murthy, K.N.; Jayaprakasha, G.K.; Patil, B.S. D-limonene rich volatile oil from blood oranges inhibits angiogenesis, metastasis and cell death in human colon cancer cells. Life Sci. 2012, 91, 429–439. [Google Scholar] [CrossRef]
- Kim, C.; Cho, S.K.; Kapoor, S.; Kumar, A.; Vali, S.; Abbasi, T.; Kim, S.H.; Sethi, G.; Ahn, K.S. β-Caryophyllene oxide inhibits constitutive and inducible STAT3 signaling pathway through induction of the SHP-1 protein tyrosine phosphatase. Mol. Carcinog. 2014, 53, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Dash, A.K. Paclitaxel in cancer treatment: Perspectives and prospects of its delivery challenges. Crit. Rev. Ther. Drug Carr. Syst. 2009, 26, 333–372. [Google Scholar] [CrossRef] [PubMed]
- Barbuti, A.M.; Chen, Z.S. Paclitaxel through the ages of anticancer therapy: Exploring its role in chemoresistance and radiation therapy. Cancers 2015, 7, 2360–2371. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, S.B.; Cohen, D.; Rao, S.; Ringel, I.; Shen, H.J.; Yang, C.P. Taxol: Mechanisms of action and resistance. J. Natl. Cancer Inst. Monogr. 1993, 55–61. [Google Scholar] [CrossRef]
- Guénard, D.; Guéritte-Voegelein, F.; Dubois, J.; Potier, P. Structure-activity relationships of Taxol and Taxotere analogues. J. Natl. Cancer Inst. Monogr. 1993, 79–82. [Google Scholar]
- Kingston, D.G.I. Taxol: The chemistry and structure-activity relationships of a novel anticancer agent. Trends Biotechnol. 1994, 12, 222–227. [Google Scholar] [CrossRef]
- Li, Z.; Tang, X.; Luo, Y.; Chen, B.; Zhou, C.; Wu, X.; Tang, Z.; Qi, X.; Cao, G.; Hao, J.; et al. NK007 helps in mitigating paclitaxel resistance through p38MAPK activation and HK2 degradation in ovarian cancer. J. Cell. Physiol. 2019, 234, 16178–16190. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Yoon, E.K.; Chung, H.J.; Park, S.Y.; Hong, K.M.; Lee, C.H.; Lee, Y.S.; Choi, K.; Yang, Y.; Kim, K.; et al. P53 acetylation enhances Taxol-induced apoptosis in human cancer cells. Apoptosis 2013, 18, 110–120. [Google Scholar] [CrossRef]
- Sun, N.K.; Huang, S.L.; Chang, T.C.; Chao, C.C.K. TLR4 and NFκB signaling is critical for taxol resistance in ovarian carcinoma cells. J. Cell. Physiol. 2018, 233, 2489–2501. [Google Scholar] [CrossRef] [PubMed]
- Panno, M.L.; Giordano, F.; Mastroianni, F.; Morelli, C.; Brunelli, E.; Palma, M.G.; Pellegrino, M.; Aquila, S.; Miglietta, A.; Mauro, L.; et al. Evidence that low doses of Taxol enhance the functional transactivatory properties of p53 on p21 waf promoter in MCF-7 breast cancer cells. FEBS Lett. 2006, 580, 2371–2380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roman Junior, W.A.; Gomes, D.B.; Zanchet, B.; Schönell, A.P.; Diel, K.A.P.; Banzato, T.P.; Ruiz, A.L.T.G.; Carvalho, J.E.; Neppel, A.; Barison, A.; et al. Antiproliferative effects of pinostrobin and 5,6-dehydrokavain isolated from leaves of alpinia zerumbet. Braz. J. Pharmacogn. 2017, 27, 592–598. [Google Scholar] [CrossRef]
- Bankova, V.; Galabov, A.S.; Antonova, D.; Vilhelmova, N.; Di Perri, B. Chemical composition of Propolis Extract ACF® and activity against herpes simplex virus. Phytomedicine 2014, 21, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Jadaun, A.; Sharma, S.; Verma, R.; Dixit, A. Pinostrobin inhibits proliferation and induces apoptosis in cancer stem-like cells through a reactive oxygen species-dependent mechanism. RSC Adv. 2019, 9, 12097–12109. [Google Scholar] [CrossRef] [Green Version]
- Abd El-Hady, F.K.; Shaker, K.H.; Imhoff, J.F.; Zinecker, H.; Salah, N.M.; Ibrahim, A.M. Bioactive metabolites from propolis inhibit superoxide anion radical, acetylcholinesterase and phosphodiesterase (PDE4). Int. J. Pharm. Sci. Rev. Res. 2013, 21, 338–344. [Google Scholar]
- Ashidi, J.S.; Houghton, P.J.; Hylands, P.J.; Efferth, T. Ethnobotanical survey and cytotoxicity testing of plants of South-western Nigeria used to treat cancer, with isolation of cytotoxic constituents from Cajanus cajan Millsp. leaves. J. Ethnopharmacol. 2010, 128, 501–512. [Google Scholar] [CrossRef]
- Cheah, K.Y.; Howarth, G.S.; Bindon, K.A.; Kennedy, J.A.; Bastian, S.E.P. Low molecular weight procyanidins from grape seeds enhance the impact of 5-Fluorouracil chemotherapy on Caco-2 human colon cancer cells. PLoS ONE 2014, 9, e98921. [Google Scholar] [CrossRef]
- Minker, C.; Duban, L.; Karas, D.; Järvinen, P.; Lobstein, A.; Muller, C.D. Impact of procyanidins from different berries on caspase 8 activation in colon cancer. Oxidative Med. Cell. Longev. 2015, 2015, 154164. [Google Scholar] [CrossRef]
- Silva, C.; Correia-Branco, A.; Andrade, N.; Ferreira, A.C.; Soares, M.L.; Sonveaux, P.; Stephenne, J.; Martel, F. Selective pro-apoptotic and antimigratory effects of polyphenol complex catechin:lysine 1:2 in breast, pancreatic and colorectal cancer cell lines. Eur. J. Pharmacol. 2019, 859, 172533. [Google Scholar] [CrossRef]
- Naasani, I.; Seimiya, H.; Tsuruo, T. Telomerase inhibition, telomere shortening, and senescence of cancer cells by tea catechins. Biochem. Biophys. Res. Commun. 1998, 249, 391–396. [Google Scholar] [CrossRef]
- Fusi, J.; Bianchi, S.; Daniele, S.; Pellegrini, S.; Martini, C.; Galetta, F.; Giovannini, L.; Franzoni, F. An in vitro comparative study of the antioxidant activity and SIRT1 modulation of natural compounds. Biomed. Pharmacother. 2018, 101, 805–819. [Google Scholar] [CrossRef] [PubMed]
- David, I.M.B.; de Souza Fernandes, F.; dos Santos Silva Ferreira, J.B.; Lüdtke, D.D.; Martins, D.F.; Bobinski, F.; da Silva, T.B.G.C.; Buffon, L.D.; Kopper, M.B.R.; da Silva, G.S.; et al. Dietary supplementation with procyanidin-rich Pinus pinaster extract is associated with attenuated Ehrlich tumor development in mice. Nutr. Res. 2019, 62, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Packer, L.; Rimbach, G.; Virgili, F. Antioxidant activity and biologic properties of a procyanidin-rich extract from pine (pinus maritima) bark, pycnogenol. Free Radic. Biol. Med. 1999, 27, 704–724. [Google Scholar] [CrossRef]
- Kim, Y.G.; Park, H.Y. The effects of pycnogenol on DNA damage in vitro and expression of superoxide dismutase and HP1 in Escherichia coli SOD and catalase deficient mutant cells. Phytother. Res. 2004, 18, 900–905. [Google Scholar] [CrossRef]
- Aydın, S.; Bacanlı, M.; Anlar, H.G.; Çal, T.; Arı, N.; Ündeğer Bucurgat, Ü.; Başaran, A.A.; Başaran, N. Preventive role of Pycnogenol® against the hyperglycemia-induced oxidative stress and DNA damage in diabetic rats. Food Chem. Toxicol. 2019, 124, 54–63. [Google Scholar] [CrossRef]
- Tokaç, M.; Bacanli, M.; Dumlu, E.G.; Aydin, S.; Engin, M.; Bozkurt, B.; Yalçin, A.; Erel, Ö.; Kiliç, M.; Başaran, N. The ameliorative effects of pycnogenol® on liver ischemia-reperfusion injury in rats. Turk. J. Pharm. Sci. 2017, 14, 257–263. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Xia, R.; Ji, C.; Zhang, L. Neuroprotective Effects of Pycnogenol Against Oxygen-Glucose Deprivation/Reoxygenation-Induced Injury in Primary Rat Astrocytes via NF-κB and ERK1/2 MAPK Pathways. Cell. Physiol. Biochem. 2017, 42, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Peng, Q.; Wei, Z.; Lau, B.H.S. Pycnogenol inhibits tumor necrosis factor-α-induced nuclear factor kappa B activation and adhesion molecule expression in human vascular endothelial cells. Cell. Mol. Life Sci. 2000, 57, 834–841. [Google Scholar] [CrossRef]
- Ramos, V.; Bocalandro, C.; Riquelme, S.; Sanhueza, V.; Aspé, E.; Roeckel, M.; Fernández, K. Effect of the bench scale extraction conditions on Pinus radiata bark extract yield, antioxidant properties and composition. Maderas Cienc. Tecnol. 2013, 15, 31–44. [Google Scholar] [CrossRef] [Green Version]
- Walter, A.; Finelli, K.; Bai, X.; Arnett, P.; Bream, T.; Seidenberg, P.; Lynch, S.; Johnson, B.; Slobounov, S. Effect of Enzogenol® Supplementation on Cognitive, Executive, and Vestibular/Balance Functioning in Chronic Phase of Concussion. Dev. Neuropsychol. 2017, 42, 93–103. [Google Scholar] [CrossRef]
- Yazaki, Y. Utilization of flavonoid compounds from bark and wood: A review. Nat. Prod. Commun. 2015, 10, 513–520. [Google Scholar] [CrossRef] [Green Version]
- Shen, Q.; Hiebert, J.B.; Hartwell, J.; Thimmesch, A.R.; Pierce, J.D. Systematic Review of Traumatic Brain Injury and the Impact of Antioxidant Therapy on Clinical Outcomes. Worldviews Evid. Based Nurs. 2016, 13, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Kim, M.S.; Kang, S.W.; Sung, H.Y.; Kang, Y.H. Pine bark extract enzogenol attenuated tumor necrosis factor-α- induced endothelial cell adhesion and monocyte transmigration. J. Agric. Food Chem. 2010, 58, 7088–7095. [Google Scholar] [CrossRef] [PubMed]
- Bahramsoltani, R.; Ebrahimi, F.; Farzaei, M.H.; Baratpourmoghaddam, A.; Ahmadi, P.; Rostamiasrabadi, P.; Rasouli Amirabadi, A.H.; Rahimi, R. Dietary polyphenols for atherosclerosis: A comprehensive review and future perspectives. Crit. Rev. Food Sci. Nutr. 2019, 59, 114–132. [Google Scholar] [CrossRef] [PubMed]
- Wood, J.E.; Senthilmohan, S.T.; Peskin, A.V. Antioxidant activity of procyanidin-containing plant extracts at different pHs. Food Chem. 2002, 77, 155–161. [Google Scholar] [CrossRef]
- O’Callaghan, Y.C.; Drummond, E.; O’Gorman, D.M.; Obrien, N.M. Antioxidant and pro-Apoptotic effects of marine-derived, multi-mineral Aquamin supplemented with a pine bark extract, Enzogenol, and a green tea extract, Sunphenon. J. Med. Food 2013, 16, 920–926. [Google Scholar] [CrossRef]
- Sato, M.; Yamada, Y.; Matsuoka, H.; Nakashima, S.; Kamiya, T.; Ikeguchi, M.; Imaizumi, K. Dietary pine bark extract reduces atherosclerotic lesion development in male apoE-deficient mice by lowering the serum cholesterol level. Biosci. Biotechnol. Biochem. 2009, 73, 1314–1317. [Google Scholar] [CrossRef]
- Diaz Napal, G.N.; Carpinella, M.C.; Palacios, S.M. Antifeedant activity of ethanolic extract from Flourensia oolepis and isolation of pinocembrin as its active principle compound. Bioresour. Technol. 2009, 100, 3669–3673. [Google Scholar] [CrossRef]
- Mandal, M.; Jaganathan, S.K. Antiproliferative effects of honey and of its polyphenols: A review. J. Biomed. Biotechnol. 2009, 2009, 830616. [Google Scholar]
- García Forero, A.; Villamizar Mantilla, D.A.; Núñez, L.A.; Ocazionez, R.E.; Stashenko, E.E.; Fuentes, J.L. Photoprotective and Antigenotoxic Effects of the Flavonoids Apigenin, Naringenin and Pinocembrin. Photochem. Photobiol. 2019, 95, 1010–1018. [Google Scholar] [CrossRef]
- Aryappalli, P.; Shabbiri, K.; Masad, R.J.; Al-Marri, R.H.; Haneefa, S.M.; Mohamed, Y.A.; Arafat, K.; Attoub, S.; Cabral-Marques, O.; Ramadi, K.B.; et al. Inhibition of Tyrosine-Phosphorylated STAT3 in Human Breast and Lung Cancer Cells by Manuka Honey is Mediated by Selective Antagonism of the IL-6 Receptor. Int. J. Mol. Sci. 2019, 20, 4340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbarić, M.; Mišković, K.; Bojić, M.; Lončar, M.B.; Smolčić-Bubalo, A.; Debeljak, Z.; Medić-Šarić, M. Chemical composition of the ethanolic propolis extracts and its effect on HeLa cells. J. Ethnopharmacol. 2011, 135, 772–778. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.A.S.; Nair, M.; Hema, P.S.; Mohan, J.; Santhoshkumar, T.R. Pinocembrin triggers Bax-dependent mitochondrial apoptosis in colon cancer cells. Mol. Carcinog. 2007, 46, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Punvittayagul, C.; Wongpoomchai, R.; Taya, S.; Pompimon, W. Effect of pinocembrin isolated from Boesenbergia pandurata on xenobiotic-metabolizing enzymes in rat liver. Drug Metab. Lett. 2011, 5, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Žižić, J.B.; Vuković, N.L.; Jadranin, M.B.; Andelković, B.D.; Tešević, V.V.; Kacaniova, M.M.; Sukdolak, S.B.; Marković, S.D. Chemical composition, cytotoxic and antioxidative activities of ethanolic extracts of propolis on HCT-116 cell line. J. Sci. Food Agric. 2013, 93, 3001–3009. [Google Scholar] [CrossRef] [PubMed]
- Ricci, M.S.; Zong, W.X. Chemotherapeutic approaches for targeting cell death pathways. Oncologist 2006, 11, 342–357. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Li, X.; Chen, Z.; Rasul, A.; Zhao, C.; Millimouno, F.M.; Tsuji, I.; Yamamura, T.; Iqbal, R.; Malhi, M. Antiproliferative and apoptotic effects of pinocembrin in human prostate cancer cells. Bangladesh J. Pharmacol. 2013, 8, 255–262. [Google Scholar]
- Zheng, Y.; Wang, K.; Wu, Y.; Chen, Y.; Chen, X.; Hu, C.W.; Hu, F. Pinocembrin induces ER stress mediated apoptosis and suppresses autophagy in melanoma cells. Cancer Lett. 2018, 431, 31–42. [Google Scholar] [CrossRef]
- Zachari, M.; Ganley, I.G. The mammalian ULK1 complex and autophagy initiation. Essays Biochem. 2017, 61, 585–596. [Google Scholar]
- Yu, C.; Minemoto, Y.; Zhang, J.; Liu, J.; Tang, F.; Bui, T.N.; Xiang, J.; Lin, A. JNK Suppresses Apoptosis via Phosphorylation of the Proapoptotic Bcl-2 Family Protein BAD. Mol. Cell 2004, 13, 329–340. [Google Scholar] [CrossRef]
- Fulda, S.; Debatin, K.M. Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene 2006, 25, 4798–4811. [Google Scholar] [CrossRef] [Green Version]
- Merarchi, M.; Jung, Y.Y.; Fan, L.; Sethi, G.; Ahn, K.S. A Brief Overview of the Antitumoral Actions of Leelamine. Biomedicines 2019, 7, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martins, A.P.; Salgueiro, L.R.; Gonçalves, M.J.; Proença Da Cunha, A.; Vila, R.; Cañigueral, S. Essential oil composition and antimicrobial activity of Santiria trimera bark. Planta Med. 2003, 69, 77–79. [Google Scholar] [CrossRef]
- Zhang, Y.; Xin, C.; Qiu, J.; Wang, Z. Essential oil from Pinus koraiensis pinecones inhibits gastric cancer cells via the Hippo/YAP signaling pathway. Molecules 2019, 24, 3851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, K.R.; Nam, D.; Yun, H.M.; Lee, S.G.; Jang, H.J.; Sethi, G.; Cho, S.K.; Ahn, K.S. β-Caryophyllene oxide inhibits growth and induces apoptosis through the suppression of PI3K/AKT/mTOR/S6K1 pathways and ROS-mediated MAPKs activation. Cancer Lett. 2011, 312, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Gowda, R.; Madhunapantula, S.V.; Kuzu, O.F.; Sharma, A.; Robertson, G.P. Targeting multiple key signaling pathways in melanoma using leelamine. Mol. Cancer Ther. 2014, 13, 1679–1689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lars Bastiaanse, E.M.; Höld, K.M.; Van Der Laarse, A. The effect of membrane cholesterol content on ion transport processes in plasma membranes. Cardiovasc. Res. 1997, 33, 272–283. [Google Scholar] [CrossRef] [Green Version]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The role of resveratrol in cancer therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kallunki, T.; Olsen, O.D.; Jäättelä, M. Cancer-associated lysosomal changes: Friends or foes? Oncogene 2013, 32, 1995–2004. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.; Carr, M.J.; Khushalani, N.I. Principles of Targeted Therapy for Melanoma. Surg. Clin. N. Am. 2020, 100, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; et al. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef]
- Bae, J.S.; Kim, Y.; Jeon, S.; Kim, S.H.; Kim, T.J.; Lee, S.; Kim, M.H.; Lim, D.J.; Lee, Y.S.; Jung, C.K. Clinical utility of TERT promoter mutations and ALK rearrangement in thyroid cancer patients with a high prevalence of the BRAF V600E mutation. Diagn. Pathol. 2016, 11, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, T.; Wang, X.N.; Lou, H.X. Natural stilbenes: An overview. Nat. Prod. Rep. 2009, 26, 916–935. [Google Scholar] [CrossRef]
- Rivière, C.; Pawlus, A.D.; Mérillon, J.M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef]
- Akinwumi, B.C.; Bordun, K.-A.M.; Anderson, H.D. Biological activities of stilbenoids. Int. J. Mol. Sci. 2018, 19, 792. [Google Scholar] [CrossRef] [Green Version]
- Wahedi, H.M.; Ahmad, S.; Abbasi, S.W. Stilbene-based natural compounds as promising drug candidates against COVID-19. J. Biomol. Struct. Dyn. 2020, 39, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lambert, C.; Lemaire, J.; Auger, H.; Guilleret, A.; Reynaud, R.; Clément, C.; Courot, E.; Taidi, B. Optimize, modulate, and scale-up resveratrol and resveratrol dimers bioproduction in Vitis Labrusca L. Cell suspension from flasks to 20 L bioreactor. Plants 2019, 8, 567. [Google Scholar] [CrossRef] [Green Version]
- Colin, D.; Lancon, A.; Delmas, D.; Lizard, G.; Abrossinow, J.; Kahn, E.; Jannin, B.; Latruffe, N. Antiproliferative activities of resveratrol and related compounds in human hepatocyte derived HepG2 cells are associated with biochemical cell disturbance revealed by fluorescence analyses. Biochimie 2008, 90, 1674–1684. [Google Scholar] [CrossRef] [Green Version]
- Hyo, J.K.; Eun, J.C.; Song, J.B.; Sun, M.S.; Heui, D.P.; Chang, H.R.; Jun, H.P.; Sang, W.C. Cytotoxic and antimutagenic stilbenes from seeds of Paeonia lactiflora. Arch. Pharmacal Res. 2002, 25, 293–299. [Google Scholar]
- Xu, L.; Botchway, B.O.; Zhang, S.; Zhou, J.; Liu, X. Inhibition of NF-κB signaling pathway by resveratrol improves spinal cord injury. Front. Neurosci. 2018, 12, 690. [Google Scholar] [CrossRef]
- Hui, Y.; Chengyong, T.; Cheng, L.; Haixia, H.; Yuanda, Z.; Weihua, Y. Resveratrol attenuates the cytotoxicity induced by amyloid-β 1–42 in PC12 cells by upregulating heme oxygenase-1 via the PI3K/Akt/Nrf2 pathway. Neurochem. Res. 2018, 43, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Sirerol, J.A.; Rodríguez, M.L.; Mena, S.; Asensi, M.A.; Estrela, J.M.; Ortega, A.L. Role of natural stilbenes in the prevention of cancer. Oxid. Med. Cell. Longev. 2016, 2016, 3128951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Marques, A.P.S.; Bonfim, F.P.G.; Dantas, W.F.C.; Puppi, R.J.; Marques, M.O.M. Chemical composition of essential oil from Varronia curassavica Jacq. accessions in different seasons of the year. Ind. Crop. Prod. 2019, 140, 11656. [Google Scholar] [CrossRef]
- Hennia, A.; Nemmiche, S.; Dandlen, S.; Miguel, M.G. Myrtus communis essential oils: Insecticidal, antioxidant and antimicrobial activities: A review. J. Essent. Oil Res. 2019, 31, 487–545. [Google Scholar] [CrossRef]
- El-Abid, H.; Amaral, C.; Cunha, S.C.; Augusto, T.V.; Fernandes, J.O.; Correia-da-Silva, G.; Teixeira, N.; Moumni, M. Chemical composition and anti-cancer properties of Juniperus oxycedrus L. essential oils on estrogen receptor-positive breast cancer cells. J. Funct. Foods 2019, 59, 261–271. [Google Scholar] [CrossRef]
- Azad, T.; Nouri, K.; Janse van Rensburg, H.J.; Maritan, S.M.; Wu, L.; Hao, Y.; Montminy, T.; Yu, J.; Khanal, P.; Mulligan, L.M.; et al. A gain-of-functional screen identifies the Hippo pathway as a central mediator of receptor tyrosine kinases during tumorigenesis. Oncogene 2020, 39, 334–355. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Ma, F.; Tian, R.; Zhou, Y.; Lan, W.; Song, Q.; Cheng, X. AJUBA increases the cisplatin resistance through hippo pathway in cervical cancer. Gene 2018, 644, 148–154. [Google Scholar] [CrossRef]
- Sarvmeili, N.; Jafarian-Dehkordi, A.; Zolfaghari, B. Cytotoxic effects of Pinus eldarica essential oil and extracts on HeLa and MCF-7 cell lines. Res. Pharm. Sci. 2016, 11, 476–483. [Google Scholar] [PubMed] [Green Version]
- Zheng, G.Q.; Kenney, P.M.; Lam, L.K.T. Sesquiterpenes from clove (eugenia caryophyllata) as potential anticarcinogenic agents. J. Nat. Prod. 1992, 55, 999–1003. [Google Scholar] [CrossRef]
- Xiao, J.B.; Chen, X.Q.; Zhang, Y.W.; Jiang, X.Y.; Xu, M. Cytotoxicity of Marchantia convoluta leaf extracts to human liver and lung cancer cells. Braz. J. Med. Biol. Res. 2006, 39, 731–738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagoa, R.; Silva, J.; Rodrigues, J.R.; Bishayee, A. Advances in phytochemical delivery systems for improved anticancer activity. Biotechnol. Adv. 2020, 38, 107382. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef] [PubMed]
- Scheinfeld, N. Sinecatechins: Treatment of genital warts. Drugs Future 2008, 33, 27–30. [Google Scholar] [CrossRef]
- Tradtrantip, L.; Namkung, W.; Verkman, A.S. Crofelemer, an antisecretory antidiarrheal proanthocyanidin oligomer extracted from Croton lechleri, targets two distinct intestinal chloride channels. Mol. Pharmacol. 2010, 77, 69–78. [Google Scholar] [CrossRef] [Green Version]
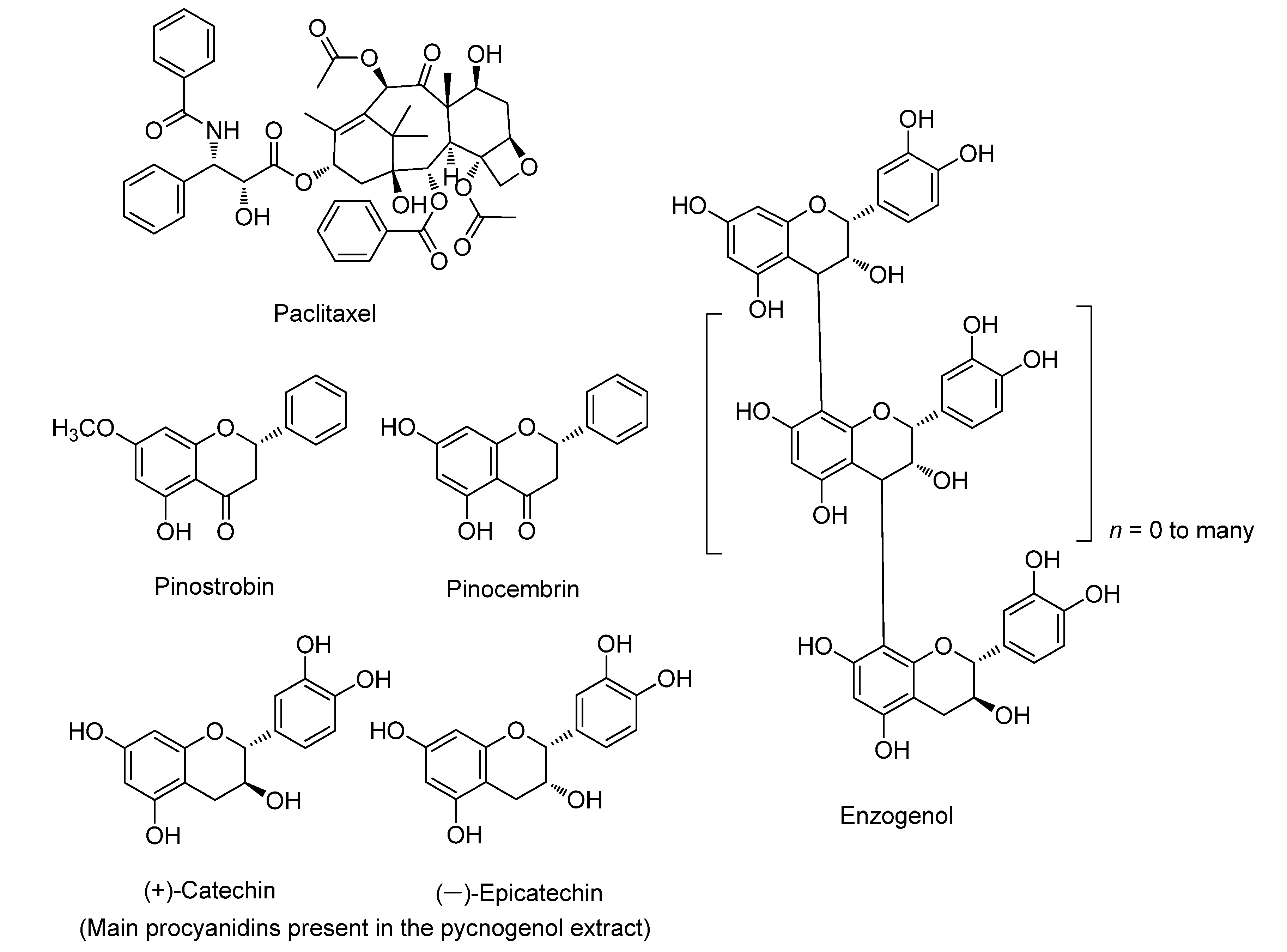

| Commercially Available Product a | Parental Plants | Common Name | Biological Activity | Mechanism of Anticancer Activity | References |
|---|---|---|---|---|---|
| Paclitaxel | Taxus brevifolia | Pacific yew | Anticancer effects | Microtubule inhibitor, induces apoptosis, Bcl-2 inhibitor | [12,23] |
| Pinostrobin | Pinus strobus | Eastern white pine | Anti-oxidative, anticancer, anti-inflammatory, and antimicrobial effects | Induces apoptosis, ROS generation in cancer cells, DNA fragmentation | [24,25,26,27] |
| Pinocembrin | Pinus pinaster | French maritime pine | Anticancer, antimicrobial, anti-inflammatory, and antioxidant effects | Increases the activity of heme oxygenase, caspase-3 and 9, and Bax | [28] |
| Pycnogenol | Pinus pinaster | French maritime pine | Anticancer, anti-inflammatory, antioxidant, blood clotting reduction, and LDL cholesterol-lowering effects | Increases nitric oxide levels in serum, inhibits NF-kB activity | [29,30,31,32] |
| Enzogenol | Pinus radiata | Monterey pine | Anticancer, anti-inflammatory, antioxidant, cardioprotective, and neuroprotective effects | Induces apoptosis, increases the activity of caspase-3, Bcl-2 inhibitor | [33] |
| Chemical Compound * | Parental Plants | Biological Activity | Mechanism of Anticancer Activity | References |
|---|---|---|---|---|
| Leelamine | Pinus species | Anticancer | Disruption of cholesterol homeostasis and autophagic flux inhibitor | [13,87] |
| Stilbenoids | Welwitschia mirabilis | Anticancer, antibacterial, anti-inflammatory, and antioxidant | Apoptosis and growth inhibition | [34] |
| Pinenes | Pinus species | Anticancer, antibacterial, and anti-fungal | Cell-cycle arrest in the G2/M phase and induction of apoptosis | [14,88,89] |
| Caryophyllenes | Pinus species | Anticancer, antibacterial, and anti-inflammatory | Suppression of STAT3 activation and suppression of the PI3K/AKT signaling pathway | [36,90] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghaffari, T.; Hong, J.-H.; Asnaashari, S.; Farajnia, S.; Delazar, A.; Hamishehkar, H.; Kim, K.H. Natural Phytochemicals Derived from Gymnosperms in the Prevention and Treatment of Cancers. Int. J. Mol. Sci. 2021, 22, 6636. https://doi.org/10.3390/ijms22126636
Ghaffari T, Hong J-H, Asnaashari S, Farajnia S, Delazar A, Hamishehkar H, Kim KH. Natural Phytochemicals Derived from Gymnosperms in the Prevention and Treatment of Cancers. International Journal of Molecular Sciences. 2021; 22(12):6636. https://doi.org/10.3390/ijms22126636
Chicago/Turabian StyleGhaffari, Tayyebeh, Joo-Hyun Hong, Solmaz Asnaashari, Safar Farajnia, Abbas Delazar, Hamed Hamishehkar, and Ki Hyun Kim. 2021. "Natural Phytochemicals Derived from Gymnosperms in the Prevention and Treatment of Cancers" International Journal of Molecular Sciences 22, no. 12: 6636. https://doi.org/10.3390/ijms22126636
APA StyleGhaffari, T., Hong, J.-H., Asnaashari, S., Farajnia, S., Delazar, A., Hamishehkar, H., & Kim, K. H. (2021). Natural Phytochemicals Derived from Gymnosperms in the Prevention and Treatment of Cancers. International Journal of Molecular Sciences, 22(12), 6636. https://doi.org/10.3390/ijms22126636







