Interactions via α2β1 Cell Integrin May Protect against the Progression of Airway Structural Changes in Asthma
Abstract
1. Introduction
2. Results
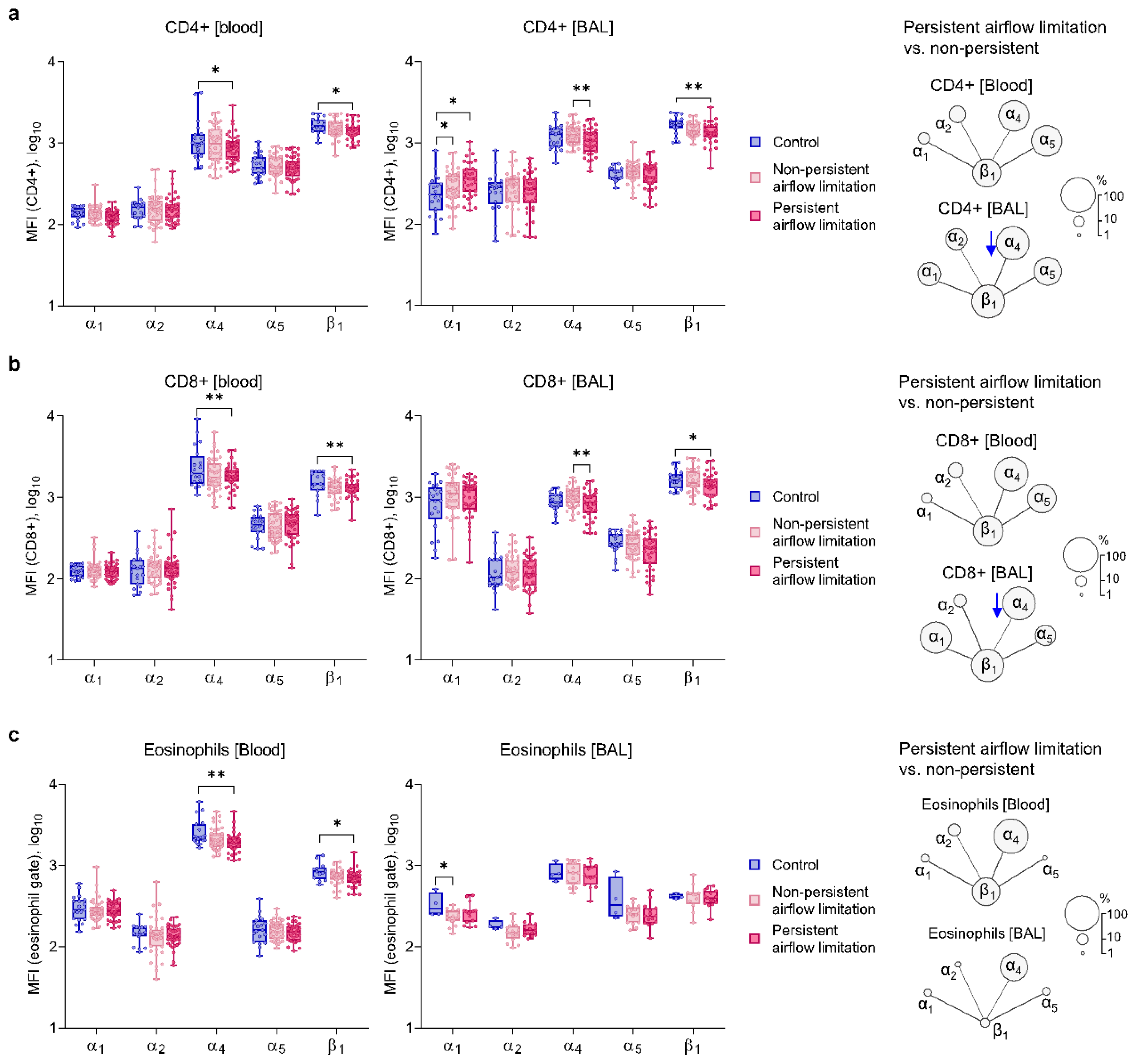
2.1. Clinical Characteristics and Airway Inflammatory Signature in Asthma Patients with Persistent Airflow Limitation
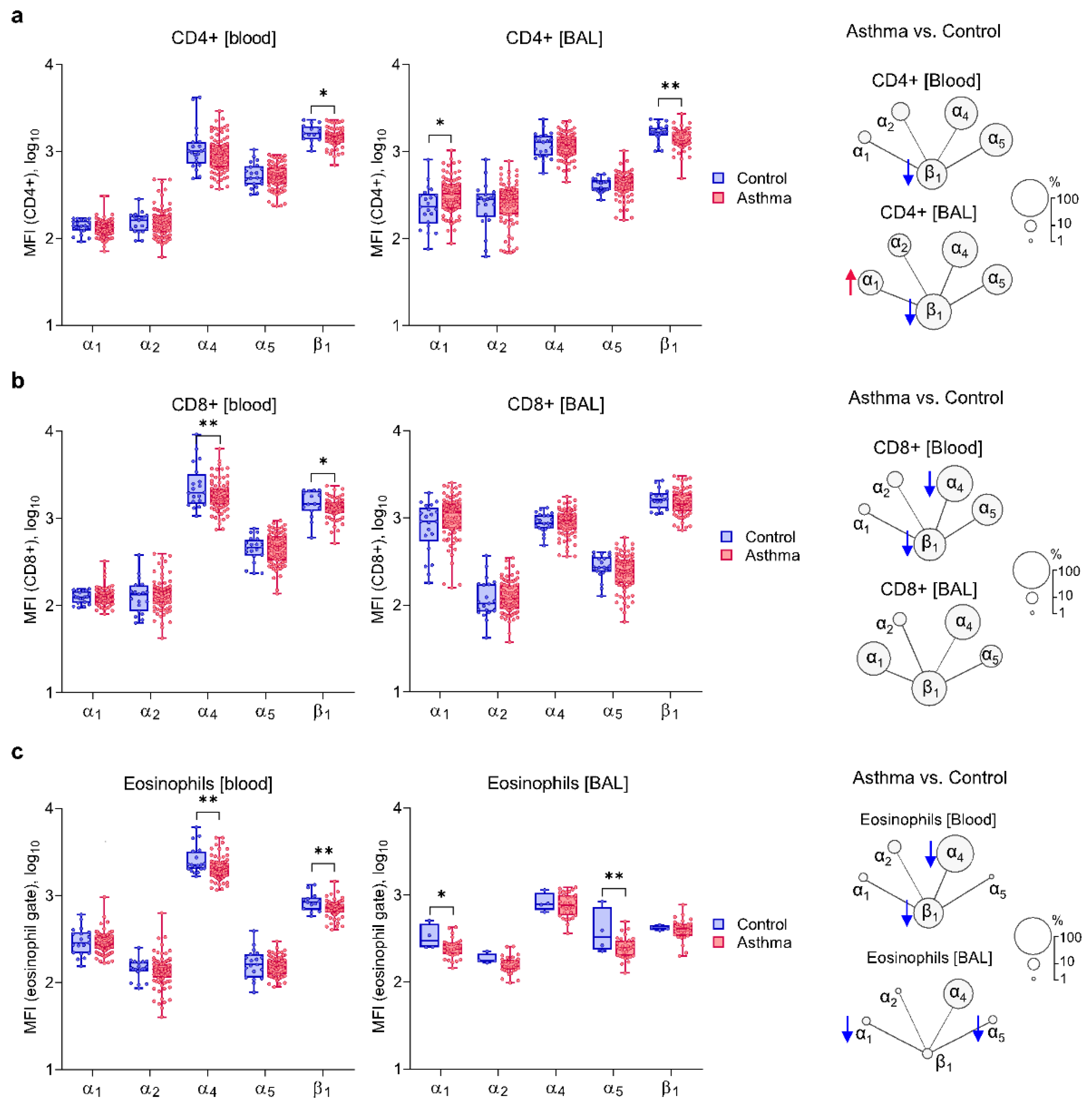
2.2. Asthma Is Characterized by Decreased Expression of α4 and β1 on Circulating Inflammatory Cells and Increased Expression of α1 Integrin Chain
2.3. Similar Expressions of α1 and α2 Integrin Chains on Blood and BAL Inflammatory Cells of Both Asthma Subsets
2.4. Increased Systemic Levels of Soluble α2 Chain in Asthma Patients with Persistent Airflow Limitation
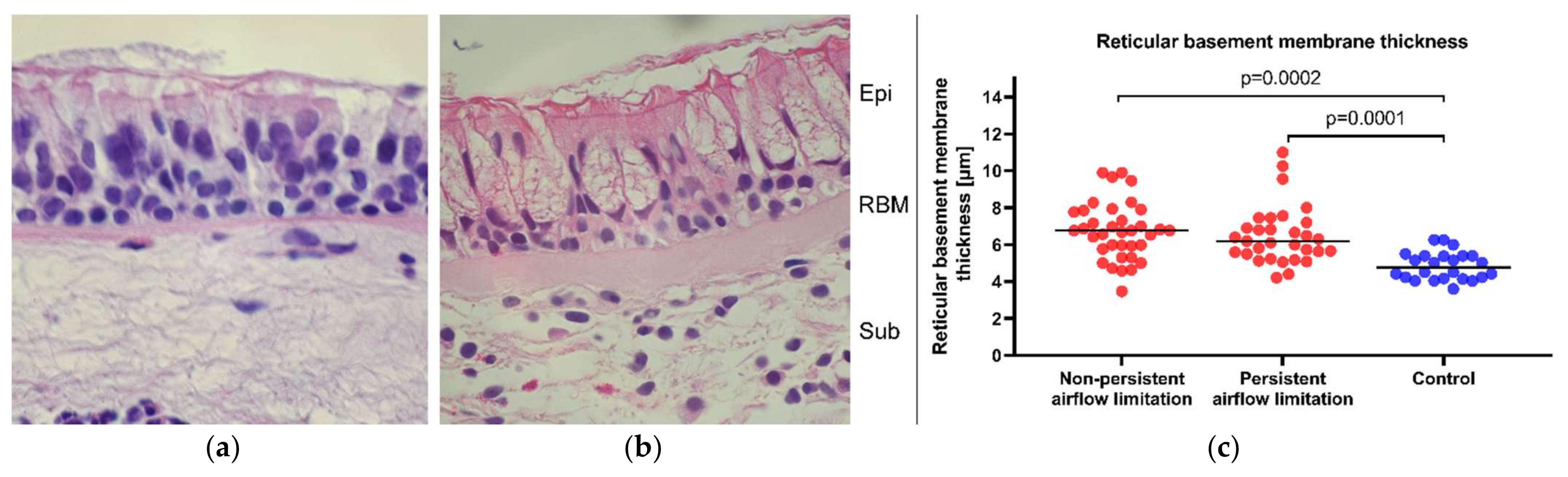
2.5. Similar Bronchial Mucosa Expression of α1 and α2 Integrin Subunits and RBM Thickness in Asthma Patients with Persistent and Non-Persistent Airflow Limitation
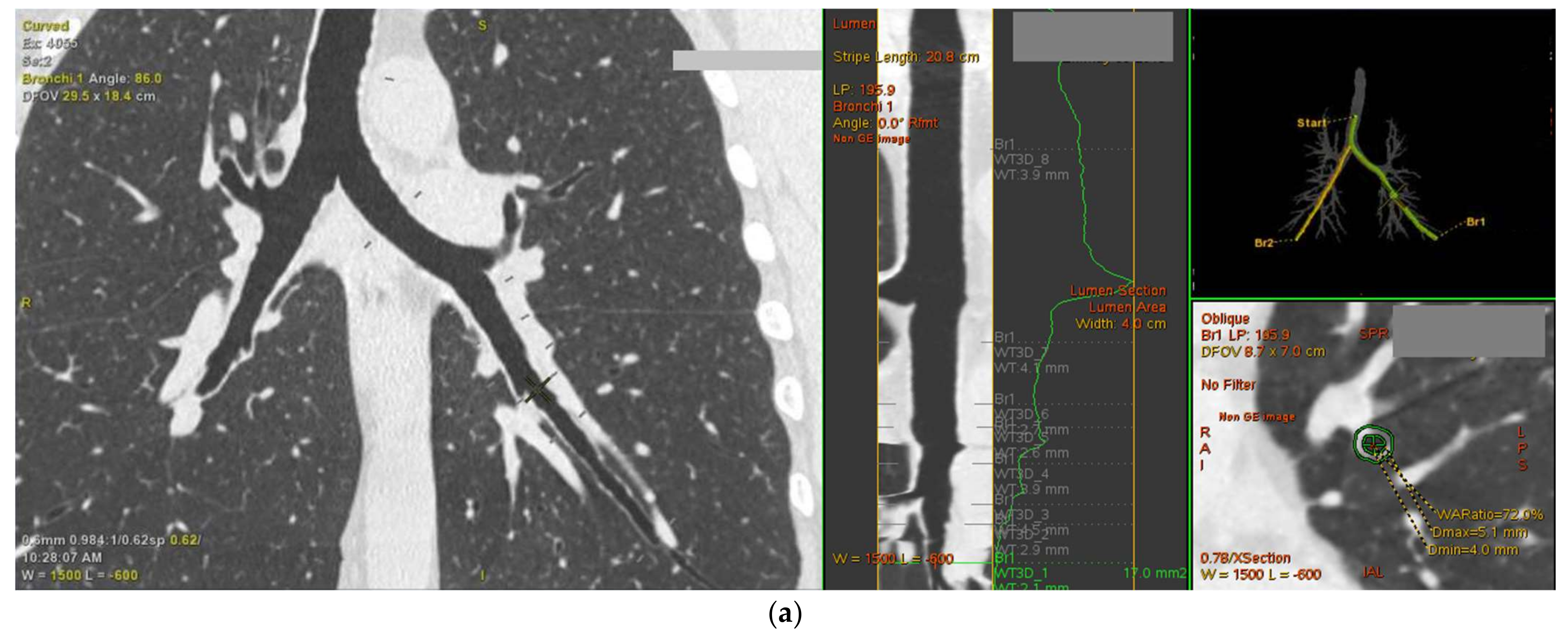
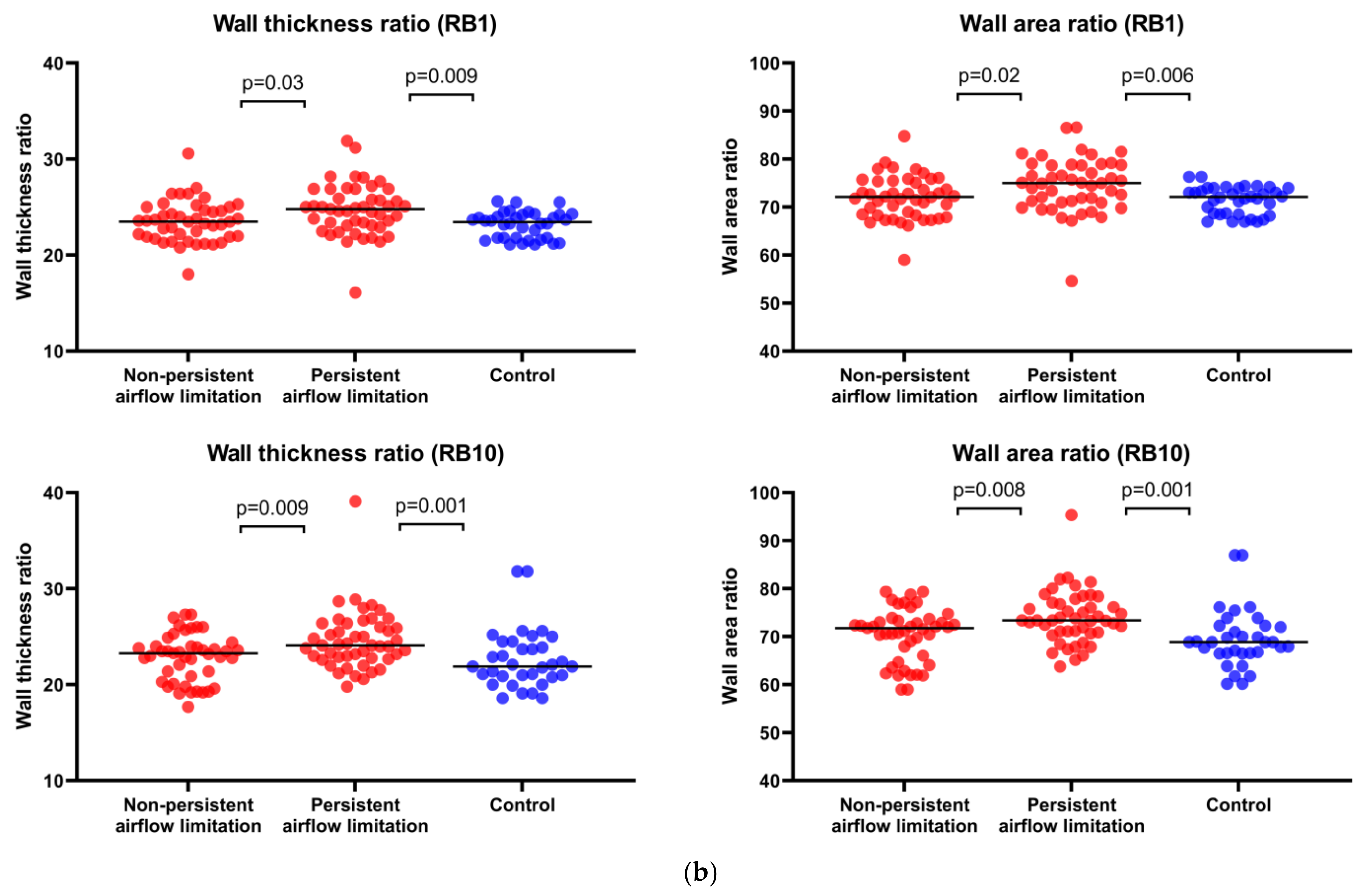
2.6. Persistent Airflow Limitation in Asthma Is Linked with Computed Tomography Parameters of the Bronchial Tree, Suggesting More Advanced Airway Wall Remodeling
3. Discussion
Limitations of the Study
4. Materials and Methods
4.1. Patients
4.2. Laboratory Investigations
4.2.1. Basic Laboratory Tests
4.2.2. Asthma and Inflammatory Biomarkers in Peripheral Blood and Bronchoalveolar Lavage Fluid
4.2.3. Flow Cytometry of Blood and Bronchoalveolar Lavage Fluid Inflammatory Cells
4.3. Bronchofiberoscopy, Endobronchial Biopsy, and Bronchoalveolar Lavage
4.4. Bronchoalveolar Lavage Fluid Examination
4.5. Histologic Examination
4.6. Lung Computed Tomography and Airway Cross-Sectional Geometry
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Busse, W.W.; Lemanske, R.F. Asthma. N. Engl. J. Med. 2001, 344, 350–362. [Google Scholar] [CrossRef]
- Trejo Bittar, H.E.; Yousem, S.A.; Wenzel, S.E. Pathobiology of Severe Asthma. Annu. Rev. Pathol. Mech. Dis. 2015, 10, 511–545. [Google Scholar] [CrossRef]
- Ito, J.T.; Lourenço, J.D.; Righetti, R.F.; Tibério, I.F.L.C.; Prado, C.M.; Lopes, F.D.T.Q.S. Extracellular Matrix Component Remodeling in Respiratory Diseases: What Has Been Found in Clinical and Experimental Studies? Cells 2019, 8, 342. [Google Scholar] [CrossRef]
- Mostaço-Guidolin, L.B.; Osei, E.T.; Ullah, J.; Hajimohammadi, S.; Fouadi, M.; Li, X.; Li, V.; Shaheen, F.; Yang, C.X.; Chu, F.; et al. Defective Fibrillar Collagen Organization by Fibroblasts Contributes to Airway Remodeling in Asthma. Am. J. Respir. Crit. Care Med. 2019, 200, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Halfter, W.; Oertle, P.; Monnier, C.A.; Camenzind, L.; Reyes-Lua, M.; Hu, H.; Candiello, J.; Labilloy, A.; Balasubramani, M.; Henrich, P.B.; et al. New Concepts in Basement Membrane Biology. FEBS J. 2015, 282, 4466–4479. [Google Scholar] [CrossRef]
- Hirota, N.; Martin, J.G. Mechanisms of Airway Remodeling. Chest 2013, 144, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Fehrenbach, H.; Wagner, C.; Wegmann, M. Airway Remodeling in Asthma: What Really Matters. In Cell and Tissue Research; Springer: Cham, Switzerland, 2017; pp. 551–569. [Google Scholar] [CrossRef]
- James, A.L.; Wenzel, S. Clinical Relevance of Airway Remodelling in Airway Diseases. Eur. Respir. J. 2007, 30, 134–155. [Google Scholar] [CrossRef] [PubMed]
- James, A.L.; Bai, T.R.; Mauad, T.; Abramson, M.J.; Dolhnikoff, M.; McKay, K.O.; Maxwell, P.S.; Elliot, J.G.; Green, F.H. Airway Smooth Muscle Thickness in Asthma Is Related to Severity but Not Duration of Asthma. Eur. Respir. J. 2009, 34, 1040–1045. [Google Scholar] [CrossRef]
- Grainge, C.L.; Lau, L.C.K.; Ward, J.A.; Dulay, V.; Lahiff, G.; Wilson, S.; Holgate, S.; Davies, D.E.; Howarth, P.H. Effect of Bronchoconstriction on Airway Remodeling in Asthma. N. Engl. J. Med. 2011, 364, 2006–2015. [Google Scholar] [CrossRef]
- Bazan-Socha, S.; Buregwa-Czuma, S.; Jakiela, B.; Zareba, L.; Zawlik, I.; Myszka, A.; Soja, J.; Okon, K.; Zarychta, J.; Kozlik, P.; et al. Reticular Basement Membrane Thickness Is Associated with Growth-and Fibrosis-Promoting Airway Transcriptome Profile-Study in Asthma Patients. Int. J. Mol. Sci. 2021, 22, 998. [Google Scholar] [CrossRef] [PubMed]
- Kozlik, P.; Zuk, J.; Bartyzel, S.; Zarychta, J.; Okon, K.; Zareba, L.; Bazan, J.G.; Kosalka, J.; Soja, J.; Musial, J.; et al. The Relationship of Airway Structural Changes to Blood and Bronchoalveolar Lavage Biomarkers, and Lung Function Abnormalities in Asthma. Clin. Exp. Allergy 2020, 50. [Google Scholar] [CrossRef]
- Choi, S.; Hoffman, E.A.; Wenzel, S.E.; Castro, M.; Fain, S.; Jarjour, N.; Schiebler, M.L.; Chen, K.; Lin, C.-L.; National Heart, Lung and Blood Institute’s Severe Asthma Research Program. Quantitative Computed Tomographic Imaging-Based Clustering Differentiates Asthmatic Subgroups with Distinctive Clinical Phenotypes. J. Allergy Clin. Immunol. 2017, 140, 690–700.e8. [Google Scholar] [CrossRef]
- Bazan-Socha, S.; Bukiej, A.; Marcinkiewicz, C.; Musial, J. Integrins in Pulmonary Inflammatory Diseases. Curr. Pharm. Des. 2005, 11, 893–901. [Google Scholar] [CrossRef]
- Zeltz, C.; Gullberg, D. The Integrin-Collagen Connection—A Glue for Tissue Repair? J. Cell Sci. 2016, 129, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Bertoni, A.; Alabiso, O.; Galetto, A.; Baldanzi, G. Integrins in T Cell Physiology. Int. J. Mol. Sci. 2018, 19, 485. [Google Scholar] [CrossRef] [PubMed]
- Schuliga, M.; Harris, T.; Stewart, A.G. Plasminogen Activation by Airway Smooth Muscle Is Regulated by Type I Collagen. Am. J. Respir. Cell Mol. Biol. 2011, 44, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Seeman, J.; Hong, J.; Hergert, P.; Bodem, V.; Jessurun, J.; Smith, K.; Nho, R.; Kahm, J.; Gaillard, P.; et al. Low α2β1 Integrin Function Enhances the Proliferation of Fibroblasts from Patients with Idiopathic Pulmonary Fibrosis by Activation of the β-Catenin Pathway. Am. J. Pathol. 2012, 181, 222–233. [Google Scholar] [CrossRef]
- Boisvert, M.; Chetoui, N.; Gendron, S.; Aoudjit, F. Alpha2beta1 Integrin Is the Major Collagen-Binding Integrin Expressed on Human Th17 Cells. Eur. J. Immunol. 2010, 40, 2710–2719. [Google Scholar] [CrossRef] [PubMed]
- Rao, W.H.; Hales, J.M.; Camp, R.D.R. Potent Costimulation of Effector T Lymphocytes by Human Collagen Type I. J. Immunol. 2000, 165, 4935–4940. [Google Scholar] [CrossRef]
- Gendron, S.; Couture, J.; Aoudjit, F. Integrin Α2β1 Inhibits Fas-Mediated Apoptosis in T Lymphocytes by Protein Phosphatase 2A-Dependent Activation of the MAPK/ERK Pathway. J. Biol. Chem. 2003, 278, 48633–48643. [Google Scholar] [CrossRef]
- Bazan-Socha, S.; Mastalerz, L.; Cybulska, A.; Zareba, L.; Kremers, R.; Zabczyk, M.; Pulka, G.; Iwaniec, T.; Hemker, C.; Undas, A. Prothrombotic State in Asthma Is Related to Increased Levels of Inflammatory Cytokines, IL-6 and TNFα, in Peripheral Blood. Inflammation 2017, 40, 1125–1235. [Google Scholar] [CrossRef]
- Kuczia, P.; Zuk, J.; Iwaniec, T.; Soja, J.; Dropinski, J.; Malesa-Wlodzik, M.; Zareba, L.; Bazan, J.G.; Undas, A.; Bazan-Socha, S. Citrullinated Histone H3, a Marker of Extracellular Trap Formation, Is Increased in Blood of Stable Asthma Patients. Clin. Transl. Allergy 2020, 10, 31. [Google Scholar] [CrossRef]
- Wright, D.B.; Meurs, H.; Dekkers, B.G.J. Integrins: Therapeutic Targets in Airway Hyperresponsiveness and Remodelling? In Trends in Pharmacological Sciences; Elsevier Ltd.: London, UK, 2014; pp. 567–574. [Google Scholar] [CrossRef]
- Global Initiative for Asthma—Global Initiative for Asthma—GINA. Available online: https://ginasthma.org/ (accessed on 26 September 2018).
- Meyer, K.C.; Raghu, G.; Baughman, R.P.; Brown, K.K.; Costabel, U.; Du Bois, R.M.; Drent, M.; Haslam, P.L.; Kim, D.S.; Nagai, S.; et al. An Official American Thoracic Society Clinical Practice Guideline: The Clinical Utility of Bronchoalveolar Lavage Cellular Analysis in Interstitial Lung Disease. Am. J. Respir. Crit. Care Med. 2012, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Hartley, R.; Khan, U.T.; Singapuri, A.; Hargadon, B.; Monteiro, W.; Pavord, I.D.; Sousa, A.R.; Marshall, R.P.; Subramanian, D.; et al. Quantitative Computed Tomography–Derived Clusters: Redefining Airway Remodeling in Asthmatic Patients. J. Allergy Clin. Immunol. 2014, 133, 729–738.e18. [Google Scholar] [CrossRef]
- Lambrecht, B.N.; Hammad, H. The Airway Epithelium in Asthma. Nat. Med. 2012, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Van Rijt, L.S.; Vos, N.; Willart, M.; Muskens, F.; Tak, P.P.; Van Der Horst, C.; Hoogsteden, H.C.; Lambrecht, B.N. Persistent Activation of Dendritic Cells after Resolution of Allergic Airway Inflammation Breaks Tolerance to Inhaled Allergens in Mice. Am. J. Respir. Crit. Care Med. 2011, 184, 303–311. [Google Scholar] [CrossRef]
- Zastrzeżyńska, W.; Przybyszowski, M.; Bazan-Socha, S.; Gawlewicz-Mroczka, A.; Sadowski, P.; Okoń, K.; Jakieła, B.; Plutecka, H.; Ćmiel, A.; Sładek, K.; et al. Omalizumab May Decrease the Thickness of the Reticular Basement Membrane and Fibronectin Deposit in the Bronchial Mucosa of Severe Allergic Asthmatics. J. Asthma 2020, 57. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.N.R.; Rogers, A.V.; Ädelroth, E.; Bandi, V.; Guntupalli, K.K.; Bush, A.; Jeffery, P.K. Early Thickening of the Reticular Basement Membrane in Children with Difficult Asthma. Am. J. Respir. Crit. Care Med. 2003, 167, 78–82. [Google Scholar] [CrossRef]
- Saglani, S.; Payne, D.N.; Zhu, J.; Wang, Z.; Nicholson, A.G.; Bush, A.; Jeffery, P.K. Early Detection of Airway Wall Remodeling and Eosinophilic Inflammation in Preschool Wheezers. Am. J. Respir. Crit. Care Med. 2007, 176, 858–864. [Google Scholar] [CrossRef]
- Tripathi, P.; Awasthi, S.; Gao, P. ADAM Metallopeptidase Domain 33 (ADAM33): A Promising Target for Asthma. Mediat. Inflamm. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Bazan-Socha, S.; Zuk, J.; Jakieła, B.; Pulka, G.; Pełka, K.; Musiał, J. Increased Expression of Α2 (CD49b), Α4 (CD49d) and Β1 (CD29) Integrin Subunits on Peripheral Blood T Lymphocytes in Clinically Stable Mild-to-Moderate Persistent Asthma. Pol. Arch. Med. Wewn. 2012, 122, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Bazan-Socha, S.; Bukiej, A.; Pulka, G.; Marcinkiewicz, C.; Musial, J. Increased Expression of Collagen Receptors: Α1β1 and Α2β1 Integrins on Blood Eosinophils in Bronchial Asthma. Clin. Exp. Allergy 2006, 36. [Google Scholar] [CrossRef]
- Betts, R.J.; Kemeny, D.M. CD8+ T Cells in Asthma: Friend or Foe? Pharmacol. Therapeutics. 2009, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Culver, B.H.; Graham, B.L.; Coates, A.L.; Wanger, J.; Berry, C.E.; Clarke, P.K.; Hallstrand, T.S.; Hankinson, J.L.; Kaminsky, D.A.; MacIntyre, N.R.; et al. ATS Committee on Proficiency Standards for Pulmonary Function Laboratories. Recommendations for a Standardized Pulmonary Function Report. An Official American Thoracic Society Technical Statement. Am. J. Respir. Crit. Care Med. 2017, 196, 1463–1472. [Google Scholar] [CrossRef] [PubMed]
- Sokolowski, J.W.; Burgher, L.W.; Jones, F.L.; Patterson, J.R.; Selecky, P.A. Position Paper on Guidelines for Fiberoptic Bronchoscopy in Adults. Am. Rev. Respir. Dis. 1987, 136, 1066. [Google Scholar] [CrossRef] [PubMed]
- Little, S.A.; Sproule, M.W.; Cowan, M.D.; Macleod, K.J.; Robertson, M.; Love, J.G.; Chalmers, G.W.; McSharry, C.P.; Thomson, N.C. High Resolution Computed Tomographic Assessment of Airway Wall Thickness in Chronic Asthma: Reproducibility and Relationship with Lung Function and Severity. Thorax 2002, 57, 247–253. [Google Scholar] [CrossRef]
| Non-Persistent Airflow Limitation n = 45 | Persistent Airflow Limitation n = 47 | Control n = 36 | p-Value Non-Persistent vs. Persistent Limitation | p-Value Non-Persistent Limitation vs. Control | p-Value Persistent Limitation vs. Control | |
|---|---|---|---|---|---|---|
| Demographic variables | ||||||
| Age, years | 52 (41–59) | 58 (52–65) | 55 (45–65) | 0.004 | 0.07 | 0.27 |
| Male gender, n (%) | 10 (22) | 16 (34) | 5 (14) | 0.15 | 0.5 | 0.07 |
| Body mass index, kg/m2 | 27.8 (24.8–30.8) | 26.4 (23.5–31.6) | 27.3 (23.0–27.9) | 0.53 | 0.13 | 0.95 |
| Smoking history | ||||||
| Past smoking, n (%) | 13 (29) | 15 (32) | 12 (33) | 0.93 | 0.85 | 0.92 |
| Pack-years of smoking | 0 (0–7) | 0 (0–8) | 0 (0–4) | 0.85 | 0.84 | 0.9 |
| Comorbidities | ||||||
| Atopy, n (%) | 27 (60) | 23 (49) | 6 (17) | 0.39 | 0.0002 | 0.005 |
| GERD, n (%) | 16 (36) | 22 (47) | 23 (64) | 0.38 | 0.02 | 0.19 |
| Arterial hypertension, n (%) | 18 (40) | 28 (60) | 15 (42) | 0.09 | 0.94 | 0.16 |
| Diabetes mellitus, n (%) | 6 (13) | 12 (26) | 3 (8) | 0.23 | 0.72 | 0.08 |
| Hypercholesterolemia, n (%) | 9 (20) | 16 (34) | 6 (17) | 0.2 | 0.92 | 0.13 |
| Coronary heart disease, n (%) | 2 (4) | 5 (11) | 2 (6) | 0.47 | 0.77 | 0.67 |
| Asthma-related variables | ||||||
| Asthma duration, years | 11.5 (5–19.5) | 10 (7–20) | 0.86 | |||
| Asthma severity (GINA): persistent mild, n (%) persistent moderate, n (%) persistent severe, n (%) | 8 (18) 22 (49) 15 (33) | 7 (15) 15 (32) 25 (53) | 0.14 | |||
| Asthma treatment: Inhaled corticosteroids, n (%) Long-acting β2-agonists, n (%) Montelukast, n (%) Theophylline, n (%) Oral corticosteroids, n (%) | 45 (100) 31 (69) 9 (20) 4 (9) 8 (18) | 47 (100) 42 (89) 4 (9) 10 (21) 15 (32) | 0.17 | |||
| Spirometry results | ||||||
| FEV1 before bronchodilator, L | 2.79 ± 0.76 | 1.79 ± 0.8 | 2.71 ± 0.75 | <0.001 | 0.65 | <0.001 |
| FEV1 before bronchodilator, % of the predicted value | 100.3 (89.5–111.1) | 66.7 (54.1–80.6) | 110.9 (106.8–114.7) | <0.001 | <0.001 | <0.001 |
| FEV1 after bronchodilator, L | 2.92 ± 0.73 | 2.07 ± 0.95 | 2.84 ± 0.79 | <0.001 | 0.66 | <0.001 |
| FEV1 after bronchodilator, % of the predicted value | 103.8 (96.4–116.5) | 79.2 (62.8–87.2) | 116 (112.1–122.3) | <0.001 | <0.001 | <0.001 |
| FEV1/VC (before bronchodilator) | 73.3 (67.8–78.18) | 59.1 (51.7–63.8) | 74.84 (73.23–78.38) | <0.001 | 0.16 | <0.001 |
| FEV1/VC (after bronchodilator) | 76.99 (73.05–81.88) | 65.4 (54.5–68.6) | 79.33 (77.25–80.38) | <0.001 | 0.25 | <0.001 |
| Reference Range | Non-Persistent Airflow Limitation n = 45 | Persistent Airflow Limitation n = 47 | Control n = 36 | p-Value Non-Persistent vs. Persistent Limitation | p-Value Non-Persistent Limitation vs. Control | p-Value Persistent Limitation vs. Control | |
|---|---|---|---|---|---|---|---|
| Basic laboratory tests | |||||||
| Red blood cells, 106/μL | 4–5 | 4.65 ± 0.4 | 4.7 ± 0.5 | 4.48 ± 0.4 | 0.68 | 0.048 | 0.03 |
| White blood cells, 103/μL | 4–10 | 6.26 (5.43–7.33) | 7.44 (6.39–9.25) | 5.44 (5.16–7.08) | <0.001 | 0.07 | <0.001 |
| Neutrophils, 103/μL | 1.8–7.7 | 3.1 (2.7–4.1) | 3.7 (2.9–4.8) | 3.3 (2.9–3.6) | 0.049 | 0.76 | 0.04 |
| Lymphocytes, 103/μL | 1–4.5 | 1.94 (1.58–2.43) | 2.2 (1.58–2.61) | 1.65 (1.44–2.08) | 0.5 | 0.03 | 0.03 |
| Monocytes, 103/μL | 0.1–0.8 | 0.57 (0.49–0.74) | 0.71 (0.53–0.9) | 0.49 (0.41–0.62) | 0.02 | 0.006 | <0.001 |
| Blood platelets, 103/μL | 140–400 | 223 (193–247) | 225 (191–265) | 228 (189–246) | 0.78 | 0.98 | 0.85 |
| Asthma and inflammatory biomarkers (blood) | |||||||
| Eosinophilia/μL | 40–450 | 230 (130–310) | 400 (180–680) | 110 (70–170) | 0.009 | <0.001 | <0.001 |
| Immunoglobulin E, IU/mL | 0–100 | 90 (26–400) | 88 (43–511) | 23 (18–48) | 0.6 | <0.001 | <0.001 |
| C-reactive protein, mg/L | 0–5 | 1.64 (0.53–8) | 4.53 (0.58–9.38) | 1.78 (0.89–2.29) | 0.39 | 0.28 | 0.008 |
| Fibrinogen, g/L | 1.8–3.5 | 3.1 (2.8–3.5) | 3.5 (3.2–4.2) | 2.9 (2.3–3.7) | 0.03 | 0.11 | 0.002 |
| Periostin, ng/mL | 0.29–0.61 § | 0.28 (0.24–0.33) | 0.38 (0.31–0.51) | 0.37 (0.36–0.45) | 0.01 | 0.001 | 0.85 |
| Interleukin 6, pg/mL | 0.005–1.432 § | 0.72 (0.43–1.19) | 1.09 (0.47–2.38) | 0.57 (0.43–0.97) | 0.14 | 0.29 | 0.03 |
| Interleukin 10, pg/mL | 0.163–1.022 § | 0.6 (0.22–1.06) | 0.55 (0.35–0.89) | 0.43 (0.2–0.76) | 0.95 | 0.17 | 0.1 |
| Interleukin 12 (p70), pg/mL | 0.005–2.618 § | 0.005 (0.005–1.2) | 0.005 (0.005–1.25) | 0.005 (0.005–0.33) | 0.7 | 0.13 | 0.26 |
| ADAM-33, ng/mL | 0.083–2.257 § | 0.73 (0.2–1.29) | 1.32 (0.33–2.37) | 0.41 (0.13–1.5) | 0.01 | 0.65 | 0.007 |
| Circulating integrin subunits | |||||||
| α1 integrin, ng/mL | 6.45–103.67 § | 17.32 (6.88–52.4) | 32.7 (14.7–55.7) | 24.1 (8.90–76.5) | 0.14 | 0.21 | 0.83 |
| α2 integrin, ng/mL | 7.79–36.19 § | 15.5 (9.7–25.5) | 22.9 (15–39) | 20.5 (11.7–26.5) | 0.03 | 0.25 | 0.21 |
| Asthma and inflammatory biomarkers (bronchoalveolar lavage fluid) | |||||||
| Periostin, ng/mL | 0.1–1.15 § | 0.86 (0.8–0.99) | 0.81 (0.72–0.95) | 0.8 (0.51–0.88) | 0.34 | 0.17 | 0.49 |
| Eosinophils, % | 0–1 # | 0.5 (0–1) | 1 (0.1–3) | 0.1 (0–1) | 0.02 | 0.62 | 0.006 |
| Non-Persistent Airflow Limitation n = 45 | Persistent Airflow Limitation n = 47 | Control n = 36 | p-Value Non-Persistent vs. Persistent Limitation | p-Value Non-Persistent Limitation vs. Control | p-Value Persistent Limitation vs. Control | |
|---|---|---|---|---|---|---|
| Collagen I, % of the stroma showing reactivity in bronchial mucosa section | 30 (20–60) | 30 (20–60) | 30 (20–70) | 0.77 | 0.67 | 0.89 |
| Collagen I, strength of the staining | 1 (0–1) | 1 (0–1) | 2 (1–2) | 0.53 | 0.86 | 0.59 |
| Collagen IV, strength of the staining | 1 (1–2) | 1 (1–2) | 2 (1–2) | 0.99 | 0.27 | 0.3 |
| α1 integrin, strength of the staining | 1 (1–2) | 1 (1–2) | 2 (1–2) | 0.31 | 0.13 | 0.61 |
| α2 integrin, strength of the staining | 1 (0–1) | 1 (0–2) | 1 (0–2) | 0.66 | 0.24 | 0.12 |
| Thickness of the reticular basement membrane of bronchial mucosa, μm | 6.77 (5.85–7.81) | 6.2 (5.5–7.3) | 4.77 (4.3–5.41) | 0.66 | 0.0002 | 0.0001 |
| Non-Persistent Airflow Limitation n = 45 | Persistent Airflow Limitation n = 47 | Control n = 36 | p *-Value Non-Persistent vs. Persistent Limitation | p *-Value Non-Persistent Limitation vs. Control | p *-Value Persistent Limitation vs. Control | |
|---|---|---|---|---|---|---|
| Total lung volume, L | 5.07 ± 1.05 | 5.49 ± 1.4 | 4.74 ± 1.09 | 0.3 | 0.18 | 0.07 |
| Low-attenuation lung area (threshold level of −950 Hounsfield units), % | 7.53 (2.3–18.44) | 18.26 (4.1–30.56) | 11.14 (2.3–23.28) | 0.26 | 0.85 | 0.48 |
| The right upper lobe apical segmental bronchus (RB1) | ||||||
| Lumen diameter, mm | 4.24 ± 0.95 | 4.12 ± 1.06 | 4.29 ± 0.85 | 0.07 | 0.73 | 0.07 |
| Airway diameter, mm | 8.02 ± 1.47 | 8.18 ± 1.9 | 7.98 ± 1.45 | 0.34 | 0.46 | 0.54 |
| Wall thickness, mm | 1.8 (1.6–2.1) | 1.9 (1.7–2.3) | 1.8 (1.7–2.1) | 0.74 | 0.69 | 0.6 |
| Wall thickness ratio (WTR) | 23.5 (21.7–25) | 24.8(22.9–26.9) | 23.6 (21.8–24.2) | 0.03 | 0.94 | 0.009 |
| Lumen area, mm2 | 12.2 (10–17.4) | 12.5 (9.5–16) | 14.5 (9.1–16.3) | 0.06 | 0.22 | 0.32 |
| Wall area, mm2 | 34.9 (27.2–48.3) | 35 (27.4–45.4) | 33 (28.6–43.4) | 0.63 | 0.49 | 0.86 |
| Wall area ratio (WAR) | 72 (68.3–75.3) | 75 (71.3–78.7) | 72.2 (68.5–74) | 0.02 | 0.98 | 0.006 |
| The right lower lobe basal posterior bronchus (RB10) | ||||||
| Lumen diameter, mm | 4.3 (3.6–4.9) | 3.7 (3.3–4.5) | 4.4 (4.3–4.8) | 0.003 | 0.67 | 0.001 |
| Airway diameter, mm | 7.9 (6.8–9.3) | 7.7 (6.6–8.5) | 8 (7.4–8.5) | 0.02 | 0.63 | 0.22 |
| Wall thickness, mm | 1.8 (1.6–1.9) | 1.8 (1.6–2.1) | 1.7 (1.5–1.8) | 0.73 | 0.25 | 0.18 |
| Wall thickness ratio (WTR) | 23.3 (20.1–24.5) | 24.1 (22.9–26.4) | 21.9 (20–24.5) | 0.009 | 0.17 | 0.001 |
| Lumen area, mm2 | 13.9 (8.9–18.9) | 10.8 (8.7–15.8) | 14.9 (14.2–18) | 0.005 | 0.53 | 0.002 |
| Wall area, mm2 | 34.4 (27–45.8) | 33.7 (24.9–42) | 32.3 (21.4–36.3) | 0.09 | 0.31 | 0.8 |
| Wall area ratio (WAR) | 71.8 (64.1–73.9) | 73.4 (70.7–77.9) | 68.9 (63.9–73.9) | 0.008 | 0.18 | 0.0009 |
| The left apicoposterior bronchus (LB1 + 2) | ||||||
| Lumen diameter, mm | 5.09 ± 1.09 | 5.04 ± 1.15 | 5 ± 1.15 | 0.79 | 0.69 | 0.86 |
| Airway diameter, mm | 9.3 (8–10.3) | 9.8 (8.3–10.4) | 8.6 (7.4–10.7) | 0.81 | 0.52 | 0.53 |
| Wall thickness, mm | 2 (1.7–2.2) | 2.2 (1.9–2.4) | 1.8 (1.6–2.3) | 0.09 | 0.16 | 0.03 |
| Wall thickness ratio (WTR) | 22.4 ± 2.7 | 23.5 ± 2.9 | 22.8 ± 3.7 | 0.17 | 0.61 | 0.67 |
| Lumen area, mm2 | 20.2 (13.4–28.3) | 22.1 (14.2–27.8) | 15.2 (13.2–26.3) | 0.76 | 0.69 | 0.88 |
| Wall area, mm2 | 46.6 ± 13.7 | 50.9 ± 16.2 | 45.7 ± 15 | 0.42 | 0.77 | 0.36 |
| Wall area ratio (WAR) | 69.3 ± 6 | 71.5 ± 6.2 | 69.7 ± 8.1 | 0.18 | 0.7 | 0.62 |
| The left lower posterior basal bronchus (LB10) | ||||||
| Lumen diameter, mm | 4.3 (3.9–4.7) | 4.2 (3.5–5) | 5.1 (4–5.6) | 0.04 | 0.41 | 0.0003 |
| Airway diameter, mm | 8.33 ± 1.3 | 8.1 ± 1.4 | 8.9 ± 1.7 | 0.13 | 0.13 | 0.003 |
| Wall thickness, mm | 1.9 (1.7–2.1) | 1.8 (1.7–2.2) | 2 (1.7–2.1) | 0.55 | 0.69 | 0.44 |
| Wall thickness ratio (WTR) | 23.2 (21.8–25) | 24.2 (21.8–26.1) | 22.4 (21.7–24.3) | 0.16 | 0.15 | 0.009 |
| Lumen area, mm2 | 14.3 (11.8–17.2) | 13.8 (9.9–19.5) | 19.8 (10.4–24.8) | 0.14 | 0.25 | 0.01 |
| Wall area, mm2 | 36.4 (31.1–46.6) | 36.7 (29.6–45.9) | 43.3 (28.2–52.8) | 0.24 | 0.48 | 0.13 |
| Wall area ratio (WAR) | 71.3 (68.3–75.3) | 73.6 (68.1–77.2) | 69.3 (65.3–73.7) | 0.06 | 0.07 | 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bazan-Socha, S.; Jakiela, B.; Zuk, J.; Zarychta, J.; Soja, J.; Okon, K.; Dziedzina, S.; Zareba, L.; Dropinski, J.; Wojcik, K.; et al. Interactions via α2β1 Cell Integrin May Protect against the Progression of Airway Structural Changes in Asthma. Int. J. Mol. Sci. 2021, 22, 6315. https://doi.org/10.3390/ijms22126315
Bazan-Socha S, Jakiela B, Zuk J, Zarychta J, Soja J, Okon K, Dziedzina S, Zareba L, Dropinski J, Wojcik K, et al. Interactions via α2β1 Cell Integrin May Protect against the Progression of Airway Structural Changes in Asthma. International Journal of Molecular Sciences. 2021; 22(12):6315. https://doi.org/10.3390/ijms22126315
Chicago/Turabian StyleBazan-Socha, Stanislawa, Bogdan Jakiela, Joanna Zuk, Jacek Zarychta, Jerzy Soja, Krzysztof Okon, Sylwia Dziedzina, Lech Zareba, Jerzy Dropinski, Krzysztof Wojcik, and et al. 2021. "Interactions via α2β1 Cell Integrin May Protect against the Progression of Airway Structural Changes in Asthma" International Journal of Molecular Sciences 22, no. 12: 6315. https://doi.org/10.3390/ijms22126315
APA StyleBazan-Socha, S., Jakiela, B., Zuk, J., Zarychta, J., Soja, J., Okon, K., Dziedzina, S., Zareba, L., Dropinski, J., Wojcik, K., Padjas, A., Marcinkiewicz, C., & Bazan, J. G. (2021). Interactions via α2β1 Cell Integrin May Protect against the Progression of Airway Structural Changes in Asthma. International Journal of Molecular Sciences, 22(12), 6315. https://doi.org/10.3390/ijms22126315







