Ozone Induced Stomatal Regulations, MAPK and Phytohormone Signaling in Plants
Abstract
1. Introduction
2. Ozone Sources, Formation, Emission, and Transport
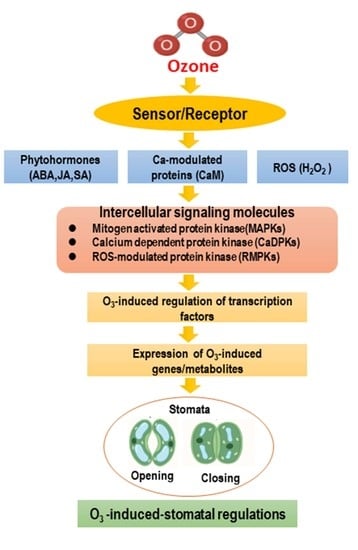
3. Ozone-Induced Stomatal Regulation in Plants
3.1. Stomata Control the Influx of Ozone into Leaves
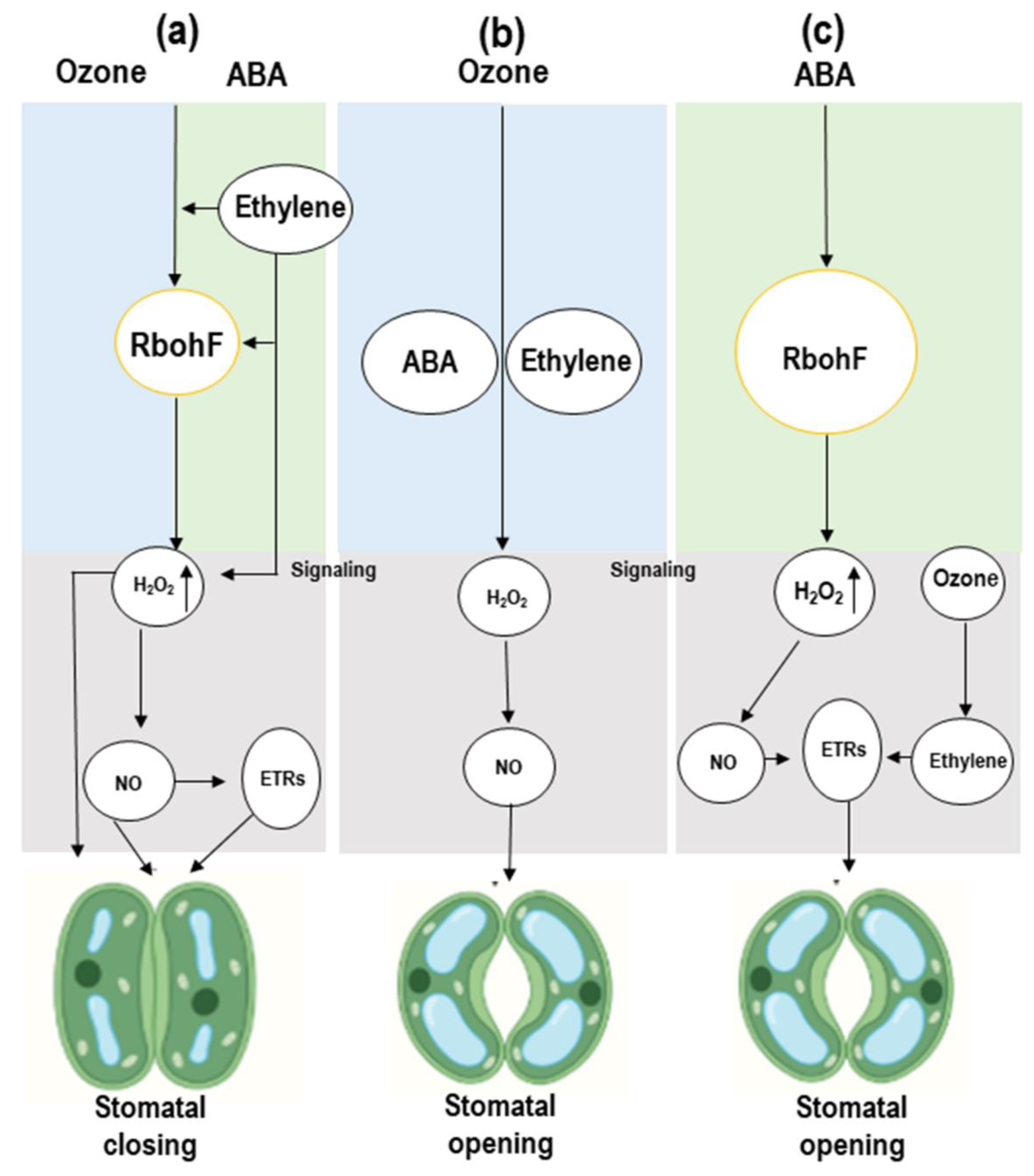
3.2. Stomatal Closure and Opening Is Linked to ABA and Ethylene and Shows Cross-Talk in Response to Ozone
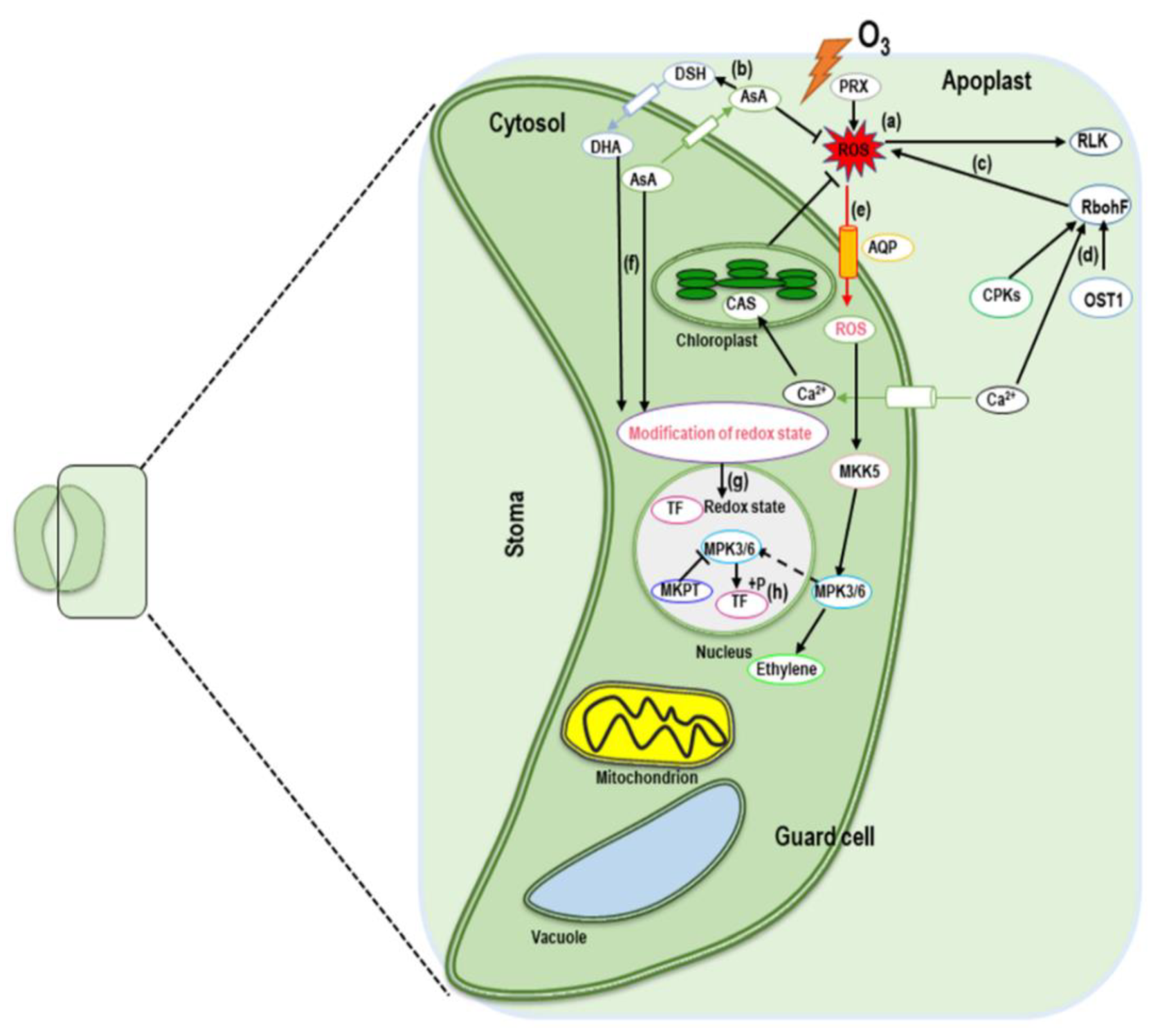
4. MAPK Cascade Signaling in Response to Ozone Stress
5. Alterations of Phytohormone Signaling in Response to Ozone Stress
5.1. Ozone-Induced Regulation of Signaling Molecules (ROS, H2O2, and NO)
5.2. Phytohormones Involved in Signaling and Gene Expression during Ozone-Induced Oxidative Stress and Cellular Injury
5.2.1. Salicylic Acid
5.2.2. Ethylene
5.2.3. Jasmonic Acid
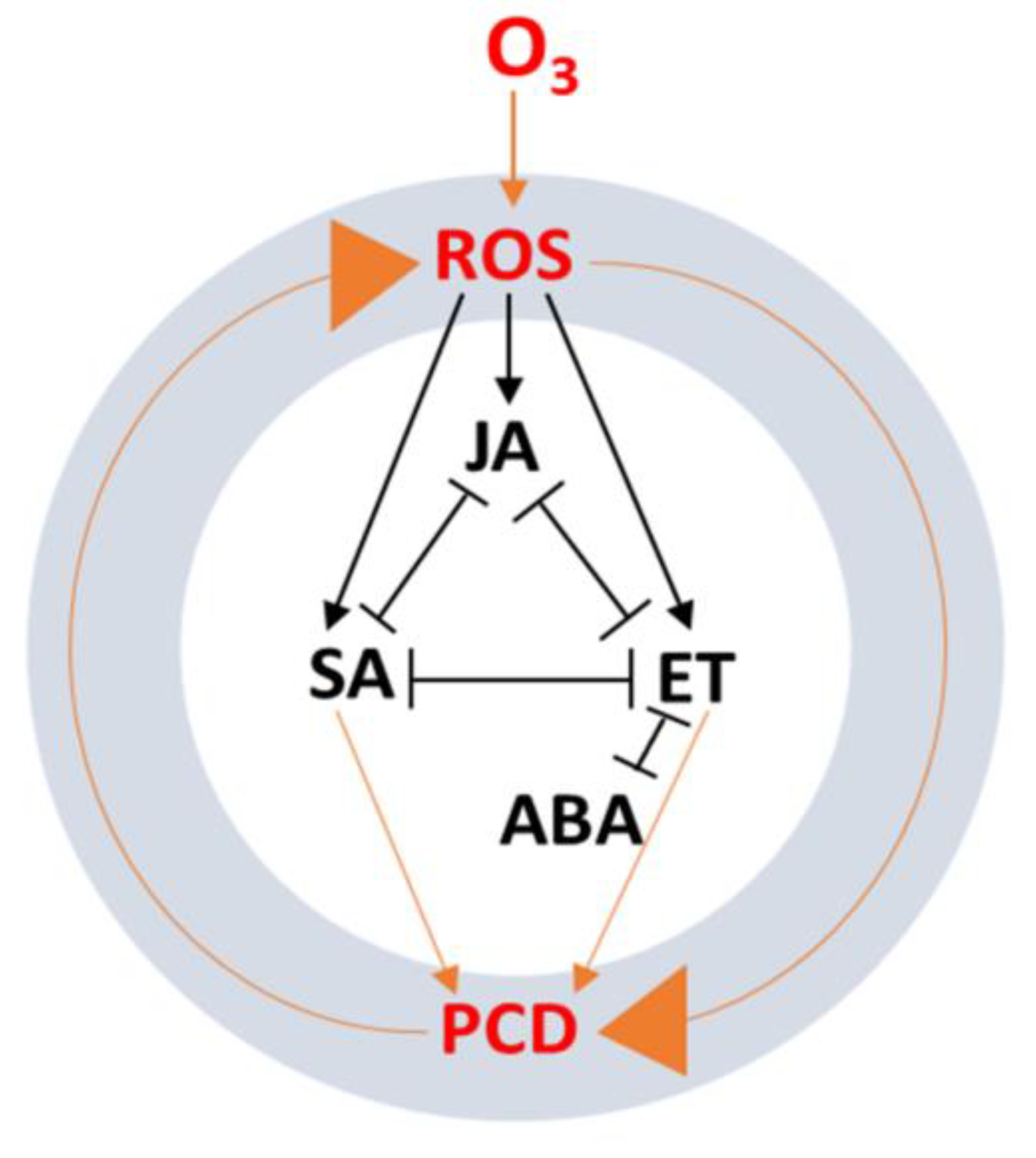
6. Phytohormone Signaling Cross-Talk in Response to Ozone
7. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mills, G.; Pleijel, H.; Malley, C.S.; Sinha, B.; Cooper, O.R.; Schultz, M.G.; Neufeld, H.S.; Simpson, D.; Sharps, K.; Feng, Z.; et al. Tropospheric ozone assessment report: Present-day tropospheric ozone distribution and trends relevant to vegetation. Elem. Sci. Anthrop. 2018, 6. [Google Scholar] [CrossRef]
- Morales, L.O.; Shapiguzov, A.; Safronov, O.; Leppälä, J.; Vaahtera, L.; Yarmolinsky, D.; Kollist, H.; Brosché, M. Ozone responses in Arabidopsis: Beyond stomatal conductance. Plant Physiol. 2021, 186, 180–192. [Google Scholar] [CrossRef]
- Chen, Z.; Cao, J.; Yu, H.; Shang, H. Effects of elevated ozone levels on photosynthesis, biomass and non-structural carbohydrates of Phoebe bournei and Phoebe zhennan in subtropical china. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef]
- Feng, Z.; De Marco, A.; Anav, A.; Gualtieri, M.; Sicard, P.; Tian, H.; Fornasier, F.; Tao, F.; Guo, A.; Paoletti, E. Economic losses due to ozone impacts on human health, forest productivity and crop yield across China. Environ. Int. 2019, 131, 104966. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.; Aucamp, P.J.; Austin, A.T.; Bais, A.F.; Ballare, C.L.; Barnes, P.W.; Bernhard, G.H.; Bjoern, L.O.; Bornman, J.F.; Congdon, N.; et al. Environmental effects of ozone depletion and its interactions with climate change: Progress report, 2016. Photochem. Photobiol. Sci. 2017, 16, 107–145. [Google Scholar] [CrossRef]
- Ahlfors, R.; Brosché, M.; Kangasjärvi, J. Ozone and nitric oxide interaction in Arabidopsis thaliana: A role for ethylene? Plant Signal. Behav. 2009, 4, 878–879. [Google Scholar] [CrossRef]
- Gandin, A.; Dizengremel, P.; Jolivet, Y. Integrative role of plant mitochondria facing oxidative stress: The case of ozone. Plant Physiol. Biochem. 2021, 159, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Caregnato, F.F.; Clebsch, C.C.; Rocha, R.F.; Feistauer, L.B.H.; Oliveira, P.L.; Divan Junior, A.D.; Moreira, J.C.F. Ozone exposure differentially affects oxidative stress parameters in distinct Phaseolus vulgaris L. varieties. J. Plant Interact. 2010, 5, 111–115. [Google Scholar] [CrossRef]
- Pellegrini, E.; Trivellini, A.; Cotrozzi, L.; Vernieri, P.; Nali, C. Involvement of phytohormones in plant responses to ozone. In Plant Hormones under Challenging Environmental Factors; Ahammed, G.J., Yu, J.-Q., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 215–245. [Google Scholar]
- Zhang, J.; Wei, Y.; Fang, Z. Ozone pollution: A major health hazard worldwide. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Jolivet, Y.; Bagard, M.; Cabané, M.; Vaultier, M.-N.; Gandin, A.; Afif, D.; Dizengremel, P.; Le Thiec, D. Deciphering the ozone-induced changes in cellular processes: A prerequisite for ozone risk assessment at the tree and forest levels. Ann. For. Sci. 2016, 73, 923–943. [Google Scholar] [CrossRef]
- Monks, P.S.; Archibald, A.T.; Colette, A.; Cooper, O.; Coyle, M.; Derwent, R.; Fowler, D.; Granier, C.; Law, K.S.; Mills, G.E.; et al. Tropospheric ozone and its precursors from the urban to the global scale from air quality to short-lived climate forcer. Atmos. Chem. Phys. 2015, 15, 8889–8973. [Google Scholar] [CrossRef]
- Lin, M.; Fiore, A.M.; Horowitz, L.W.; Cooper, O.R.; Naik, V.; Holloway, J.; Johnson, B.J.; Middlebrook, A.M.; Oltmans, S.J.; Pollack, I.B.; et al. Transport of Asian ozone pollution into surface air over the western United States in spring. J. Geophys. Res. Atmos. 2012, 117. [Google Scholar] [CrossRef]
- Super, I.; Vilà-Guerau de Arellano, J.; Krol, M.C. Cumulative ozone effect on canopy stomatal resistance and the impact on boundary layer dynamics and CO2 assimilation at the diurnal scale: A case study for grassland in the Netherlands. J. Geophys. Res. Biogeosci. 2015, 120, 1348–1365. [Google Scholar] [CrossRef]
- Li, S.; Harley, P.C.; Niinemets, Ü. Ozone-induced foliar damage and release of stress volatiles is highly dependent on stomatal openness and priming by low-level ozone exposure in Phaseolus vulgaris. Plant Cell Environ. 2017, 40, 1984–2003. [Google Scholar] [CrossRef]
- Horaruang, W.; Hills, A.; Blatt, M.R. Communication between the Plasma Membrane and Tonoplast Is an Emergent Property of Ion Transport. Plant Physiol. 2020, 182, 1833–1835. [Google Scholar] [CrossRef] [PubMed]
- Kollist, H.; Nuhkat, M.; Roelfsema, M.R.G. Closing gaps: Linking elements that control stomatal movement. New Phytol. 2014, 203, 44–62. [Google Scholar] [CrossRef]
- Negi, J.; Matsuda, O.; Nagasawa, T.; Oba, Y.; Takahashi, H.; Kawai-Yamada, M.; Uchimiya, H.; Hashimoto, M.; Iba, K. CO2 regulator SLAC1 and its homologues are essential for anion homeostasis in plant cells. Nature 2008, 452, 483–486. [Google Scholar] [CrossRef]
- Vahisalu, T.; Kollist, H.; Wang, Y.-F.; Nishimura, N.; Chan, W.-Y.; Valerio, G.; Lamminmäki, A.; Brosché, M.; Moldau, H.; Desikan, R.; et al. SLAC1 is required for plant guard cell S-type anion channel function in stomatal signalling. Nature 2008, 452, 487–491. [Google Scholar] [CrossRef]
- Ma, Y.; Szostkiewicz, I.; Korte, A.; Moes, D.; Yang, Y.; Christmann, A.; Grill, E. Regulators of PP2C phosphatase activity function as abscisic acid sensors. Science 2009, 324, 1064–1068. [Google Scholar] [CrossRef]
- Hasan, M.M.; Gong, L.; Nie, Z.-F.; Li, F.-P.; Ahammed, G.J.; Fang, X.-W. ABA-induced stomatal movements in vascular plants during dehydration and rehydration. Environ. Exp. Bot. 2021, 186, 104436. [Google Scholar] [CrossRef]
- Postiglione, A.E.; Muday, G.K. The Role of ROS Homeostasis in ABA-Induced guard cell signaling. Front. Plant Sci. 2020, 11, 968. [Google Scholar] [CrossRef]
- Kollist, H.; Zandalinas, S.I.; Sengupta, S.; Nuhkat, M.; Kangasjärvi, J.; Mittler, R. Rapid Responses to Abiotic Stress: Priming the Landscape for the Signal Transduction Network. Trends Plant Sci. 2019, 24, 25–37. [Google Scholar] [CrossRef]
- Gong, L.; Liu, X.D.; Zeng, Y.Y.; Tian, X.Q.; Li, Y.L.; Turner, N.C.; Fang, X.W. Differences in stomatal morphology and physiology explain differences in stomatal sensitivity to abscisic acid across vascular plant lineages. Plant Physiol. 2021, 186, 782–797. [Google Scholar] [CrossRef] [PubMed]
- Kurusu, T.; Saito, K.; Horikoshi, S.; Hanamata, S.; Negi, J.; Yagi, C.; Kitahata, N.; Iba, K.; Kuchitsu, K. An S-type anion channel SLAC1 is involved in cryptogein-induced ion fluxes and modulates hypersensitive responses in tobacco BY-2 cells. PLoS ONE 2013, 8, e70623. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.; El-Maarouf-Bouteau, H.; Rossi, M.; Biligui, B.; Briand, J.; Kawano, T.; Mancuso, S.; Bouteau, F. Post-transcriptional regulation of GORK channels by superoxide anion contributes to increases in outward-rectifying K+ currents. New Phytol. 2013, 198, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Daszkowska-Golec, A.; Szarejko, I. Open or close the gate—Stomata action under the control of phytohormones in drought stress conditions. Front. Plant Sci. 2013, 4, 138. [Google Scholar] [CrossRef]
- Yang, Y.J.; Bi, M.H.; Nie, Z.F.; Jiang, H.; Liu, X.D.; Fang, X.W.; Brodribb, T.J. Evolution of stomatal closure to optimise water use efficiency in response to dehydration in ferns and seed plants. New Phytol. 2021, 230, 2001–2010. [Google Scholar] [CrossRef]
- Yao, G.Q.; Nie, Z.F.; Turner, N.C.; Li, F.M.; Gao, T.P.; Fang, X.W.; Scoffoni, C. Combined high leaf hydraulic safety and efficiency provides drought tolerance in Caragana species adapted to low mean annual precipitation. New Phytol. 2021, 229, 230–244. [Google Scholar] [CrossRef]
- McAdam, E.L.; Brodribb, T.J.; McAdam, S.A.M. Does ozone increase ABA levels by non-enzymatic synthesis causing stomata to close? Plant Cell Environ. 2017, 40, 741–747. [Google Scholar] [CrossRef]
- Wilkinson, S.; Davies, W.J. Drought, ozone, ABA and ethylene: New insights from cell to plant to community. Plant Cell Environ. 2010, 33, 510–525. [Google Scholar] [CrossRef]
- Saxena, I.; Srikanth, S.; Chen, Z. Cross talk between H2O2 and interacting signal molecules under plant stress response. Front. Plant Sci. 2016, 7, 570. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Qi, C.; Ren, H.; Huang, A.; Hei, S.; She, X. Ethylene mediates brassinosteroid-induced stomatal closure via Gα protein-activated hydrogen peroxide and nitric oxide production in Arabidopsis. Plant J. 2015, 82, 280–301. [Google Scholar] [CrossRef] [PubMed]
- Gomi, K.; Ogawa, D.; Katou, S.; Kamada, H.; Nakajima, N.; Saji, H.; Soyano, T.; Sasabe, M.; Machida, Y.; Mitsuhara, I.; et al. A mitogen-activated protein kinase NtMPK4 activated by SIPKK is required for jasmonic acid signaling and involved in ozone tolerance via stomatal movement in Tobacco. Plant Cell Physiol. 2005, 46, 1902–1914. [Google Scholar] [CrossRef]
- Sinha, A.K.; Jaggi, M.; Raghuram, B.; Tuteja, N. Mitogen-activated protein kinase signaling in plants under abiotic stress. Plant Signal. Behav. 2011, 6, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Pitzschke, A.; Schikora, A.; Hirt, H. MAPK cascade signalling networks in plant defence. Curr. Opin. Plant Biol. 2009, 12, 421–426. [Google Scholar] [CrossRef]
- Samuel, M.A.; Ellis, B.E. Double jeopardy: Both overexpression and suppression of a redox-activated plant mitogen-activated protein kinase render tobacco plants ozone sensitive. Plant Cell 2002, 14, 2059–2069. [Google Scholar] [CrossRef]
- Miles, G.P.; Samuel, M.A.; Zhang, Y.; Ellis, B.E. RNA interference-based (RNAi) suppression of AtMPK6, an Arabidopsis mitogen-activated protein kinase, results in hypersensitivity to ozone and misregulation of AtMPK3. Environ. Pollut. 2005, 138, 230–237. [Google Scholar] [CrossRef]
- Rasmussen, M.; Roux, M.; Petersen, M.; Mundy, J. MAP kinase cascades in Arabidopsis innate immunity. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef]
- Vainonen, J.P.; Kangasjärvi, J. Plant signalling in acute ozone exposure. Plant Cell Environ. 2015, 38, 240–252. [Google Scholar] [CrossRef]
- Overmyer, K.; Brosché, M.; Pellinen, R.; Kuittinen, T.; Tuominen, H.; Ahlfors, R.; Keinänen, M.; Saarma, M.; Scheel, D.; Kangasjärvi, J. Ozone-induced programmed cell death in the Arabidopsis radical-induced cell death1 mutant. Plant Physiol. 2005, 137, 1092–1104. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, S. Regulation of ethylene biosynthesis and signaling by protein kinases and phosphatases. Mol. Plant 2014, 7, 939–942. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ngwenyama, N.; Liu, Y.; Walker, J.C.; Zhang, S. Stomatal development and patterning are regulated by environmentally responsive mitogen-activated protein kinases in Arabidopsis. Plant Cell 2007, 19, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Astier, J.; Gross, I.; Durner, J. Nitric oxide production in plants: An update. J. Exp. Bot. 2017, 69, 3401–3411. [Google Scholar] [CrossRef]
- Pasqualini, S.; Meier, S.; Gehring, C.; Madeo, L.; Fornaciari, M.; Romano, B.; Ederli, L. Ozone and nitric oxide induce cGMP-dependent and -independent transcription of defence genes in tobacco. Plant Signal. Behav. 2009, 181, 860–870. [Google Scholar] [CrossRef]
- Niu, L.; Liao, W. Hydrogen peroxide signaling in plant development and abiotic responses: Crosstalk with nitric oxide and calcium. Front. Plant Sci. 2016, 7, 230. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.R.; Fatma, M.; Per, T.S.; Anjum, N.A.; Khan, N.A. Salicylic acid-induced abiotic stress tolerance and underlying mechanisms in plants. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Prodhan, M.Y.; Munemasa, S.; Nahar, M.N.-E.-N.; Nakamura, Y.; Murata, Y. Guard cell salicylic acid signaling is integrated into abscisic acid signaling via the Ca2+/CPK-dependent pathway. Plant Physiol. 2018, 178, 441–450. [Google Scholar] [CrossRef]
- Melotto, M.; Zhang, L.; Oblessuc, P.R.; He, S.Y. Stomatal defense a decade later. Plant Physiol. 2017, 174, 561–571. [Google Scholar] [CrossRef]
- Fragnière, C.; Serrano, M.; Abou-Mansour, E.; Métraux, J.-P.; L’Haridon, F. Salicylic acid and its location in response to biotic and abiotic stress. FEBS Lett. 2011, 585, 1847–1852. [Google Scholar] [CrossRef]
- Shine, M.B.; Yang, J.-W.; El-Habbak, M.; Nagyabhyru, P.; Fu, D.-Q.; Navarre, D.; Ghabrial, S.; Kachroo, P.; Kachroo, A. Cooperative functioning between phenylalanine ammonia lyase and isochorismate synthase activities contributes to salicylic acid biosynthesis in soybean. New Phytol. 2016, 212, 627–636. [Google Scholar] [CrossRef]
- Ogawa, D.; Nakajima, N.; Sano, T.; Tamaoki, M.; Aono, M.; Kubo, A.; Kanna, M.; Ioki, M.; Kamada, H.; Saji, H. Salicylic acid accumulation under O3 exposure is regulated by ethylene in tobacco plants. Plant Cell Physiol. 2005, 46, 1062–1072. [Google Scholar] [CrossRef]
- Li, P.; Mane, S.P.; Sioson, A.A.; Robinet, C.V.; Heath, L.S.; Bohnert, H.J.; Grene, R. Effects of chronic ozone exposure on gene expression in Arabidopsis thaliana ecotypes and in Thellungiella halophila. Plant Cell Environ. 2006, 29, 854–868. [Google Scholar] [CrossRef]
- Rao, M.V.; Davis, K.R. Ozone-induced cell death occurs via two distinct mechanisms in Arabidopsis: The role of salicylic acid. Plant J. 1999, 17, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Xu, E.; Vaahtera, L.; Brosché, M. Roles of defense hormones in the regulation of ozone-induced changes in gene expression and cell death. Mol. Plant 2015, 8, 1776–1794. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kumar, V.; Sidhu, G.P.S.; Kumar, R.; Kohli, S.K.; Yadav, P.; Kapoor, D.; Bali, A.S.; Shahzad, B.; Khanna, K.; et al. Abiotic stress management in plants: Role of ethylene. In Molecular Plant Abiotic Stress; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 185–208. [Google Scholar]
- Mirica, L.M.; Klinman, J.P. The nature of O2 activation by the ethylene-forming enzyme 1-aminocyclopropane-1-carboxylic acid oxidase. Proc. Natl. Acad. Sci. USA 2008, 105, 1814. [Google Scholar] [CrossRef] [PubMed]
- Houben, M.; Van de Poel, B. 1-Aminocyclopropane-1-carboxylic acid oxidase (ACO): The enzyme that makes the plant hormone ethylene. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Sinn, J.P.; Schlagnhaufer, C.D.; Arteca, R.N.; Pell, E.J. Ozone-induced ethylene and foliar injury responses are altered in 1-aminocyclopropane-1-carboxylate synthase antisense potato plants. New Phytol. 2004, 164, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Tsuchisaka, A.; Theologis, A. Unique and overlapping expression patterns among the Arabidopsis 1-amino-cyclopropane-1-carboxylate synthase gene family members. Plant Physiol. 2004, 136, 2982–3000. [Google Scholar] [CrossRef]
- Bandurska, H.; Borowiak, K.; Miara, M.J.A.B.C.S.B. Effect of two different ambient ozone concentrations on antioxidative enzymes in leaves of two tobacco cultivars with contrasting ozone sensitivity. Acta Biol. Cracov. Ser. Bot. 2009, 51, 37–44. [Google Scholar]
- Gupta, S.K.; Sharma, M.; Majumder, B.; Maurya, V.K.; Lohani, M.; Deeba, F.; Pandey, V. Impact of Ethylene diurea (EDU) on growth, yield and proteome of two winter wheat varieties under high ambient ozone phytotoxicity. Chemosphere 2018, 196, 161–173. [Google Scholar] [CrossRef]
- Ruan, J.; Zhou, Y.; Zhou, M.; Yan, J.; Khurshid, M.; Weng, W.; Cheng, J.; Zhang, K. Jasmonic acid signaling pathway in plants. Int. J. Mol. Sci. 2019, 20, 2479. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yuan, J.; Yang, W.; Zhu, L.; Su, C.; Wang, X.; Wu, H.; Sun, Z.; Li, X. Genome wide identification and expression profiling of ethylene receptor genes during soybean nodulation. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Sasaki-Sekimoto, Y.; Taki, N.; Obayashi, T.; Aono, M.; Matsumoto, F.; Sakurai, N.; Suzuki, H.; Hirai, M.Y.; Noji, M.; Saito, K.; et al. Coordinated activation of metabolic pathways for antioxidants and defence compounds by jasmonates and their roles in stress tolerance in Arabidopsis. Plant J. 2005, 44, 653–668. [Google Scholar] [CrossRef]
- Tamaoki, M. The role of phytohormone signaling in ozone-induced cell death in plants. Plant Signal. Behav. 2008, 3, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Kangasjärvi, J.; Jaspers, P.; Kollist, H. Signalling and cell death in ozone-exposed plants. Plant Cell Environ. 2005, 28, 1021–1036. [Google Scholar] [CrossRef]
- Pellegrini, E.; Trivellini, A.; Campanella, A.; Francini, A.; Lorenzini, G.; Nali, C.; Vernieri, P. Signaling molecules and cell death in Melissa officinalis plants exposed to ozone. Plant Cell Rep. 2013, 32, 1965–1980. [Google Scholar] [CrossRef]
- Nunn, A.J.; Kozovits, A.R.; Reiter, I.M.; Heerdt, C.; Leuchner, M.; Lütz, C.; Liu, X.; Lo¨w, M.; Winkler, J.B.; Grams, T.E.E.; et al. Comparison of ozone uptake and sensitivity between a phytotron study with young beech and a field experiment with adult beech (Fagus sylvatica). Environ. Pollut. 2005, 137, 494–506. [Google Scholar] [CrossRef] [PubMed]
- Betz, G.; Gerstner, E.; Olbrich, M.; Winkler, J.; Langebartels, C.; Heller, W.; Sandermann, H.; Ernst, D. Forestry Effects of abiotic stress on gene transcription in European beech: Ozone affects ethylene biosynthesis in saplings of Fagus sylvatica L. IForest 2009, 2, 114. [Google Scholar] [CrossRef]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Gene networks involved in drought stress response and tolerance. J. Exp. Bot. 2006, 58, 221–227. [Google Scholar] [CrossRef]
- Yao, G.Q.; Li, F.P.; Nie, Z.F.; Bi, M.H.; Jiang, H.; Liu, X.D.; Wei, Y.; Fang, X.W. Ethylene, not ABA, is closely linked to the recovery of gas exchange after drought in four Caragana species. Plant Cell Environ. 2020, 44, 399–411. [Google Scholar] [CrossRef]
- Blomster, T.; Salojärvi, J.; Sipari, N.; Brosché, M.; Ahlfors, R.; Keinänen, M.; Overmyer, K.; Kangasjärvi, J. Apoplastic reactive oxygen species transiently decrease auxin signaling and cause stress-induced morphogenic response in Arabidopsis. Plant Physiol. 2011, 157, 1866–1883. [Google Scholar] [CrossRef] [PubMed]
| Gene Accession | Gene Family | Response Marker Gene/Metabolites | Col-0 (Control vs. Stress) | Cvi-0 (Control vs. Stress) | Col 0 and Cvi-0 (Control vs. Stress) |
|---|---|---|---|---|---|
| SA induced: | |||||
| AT2G14610 | PR-1-like protein | induced | 0 | + | − |
| AT1G19320 | pathogenesis-related protein 5 precursors, | induced | + | 0 | + |
| AT2G22300 | ET-induced calmodulin-binding | NFkB2-F | 0 | + | 0 |
| ET induced: | |||||
| AT1G49830 | ET-responsive protein ET-responsive element binding | Not responded | 0 | + | − |
| AT1G55150 | box RNA helicase protein, putative ET-responsive element binding | Not responded | 0 | + | 0 |
| AT2G22300 | binding plant defensin protein, putative | NGkB2-F | 0 | + | 0 |
| AT5G44420 | (PDF1.2a) plant defensin protein, putative | induced | − | − | 0 |
| AT2G26020 | (PDF1.2b) | induced | − | − | 0 |
| JA induced: | |||||
| AT2G24850 | putative tyrosine aminotransferase | induced | − | − | + |
| AT5G24770 | vegetative storage protein Vsp2 | induced | 0 | 0 | + |
| Gene Accession | Functional Group | Col 0 (Control vs. Stress) | Cvi-0 (Control vs. Stress) | Col 0 and Cvi- 0 (Control vs. Stress) |
|---|---|---|---|---|
| AT3G09640 | ascorbate peroxidase | repressed | stable | induced |
| AT2G28190 | thylakoid bound ascorbate peroxidase | repressed | stable | repressed |
| AT3G10920 | manganese superoxide dismutase | stable | induced | repressed |
| AT1G03850 | glutaredoxin protein family glutaredoxin | stable | induced | repressed |
| AT2G41680 | thioredoxin | stable | stable | repressed |
| Gene Accession | Gene Name | Response (WT O3/WT Control) | Response (coi1 ein2 sid2 O3/coi1 ein2 sid2 O3 Control) |
|---|---|---|---|
| AT1G15520 | PDR12 | Up-regulated | Stable |
| AT1G26380 | FAD-binding Berberine family protein | Up-regulated | Stable |
| AT1G21120 | IGMT2 | Up-regulated | Stable |
| AT2G26560 | PLA2A | Up-regulated | Stable |
| AT4G08770 | PRX37 | Up-regulated | Up-regulated |
| AT4G30280 | XTH18 | Up-regulated | Up-regulated |
| AT5G65730 | XTH6 | Down-regulated | Down-regulated |
| AT2G42380 | BZIP34 | Down-regulated | Down-regulated |
| AT1G60590 | Pectin lyase-like | Down-regulated | Down-regulated |
| AT5G15310 | MYB16 | Down-regulated | Down-regulated |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasan, M.M.; Rahman, M.A.; Skalicky, M.; Alabdallah, N.M.; Waseem, M.; Jahan, M.S.; Ahammed, G.J.; El-Mogy, M.M.; El-Yazied, A.A.; Ibrahim, M.F.M.; et al. Ozone Induced Stomatal Regulations, MAPK and Phytohormone Signaling in Plants. Int. J. Mol. Sci. 2021, 22, 6304. https://doi.org/10.3390/ijms22126304
Hasan MM, Rahman MA, Skalicky M, Alabdallah NM, Waseem M, Jahan MS, Ahammed GJ, El-Mogy MM, El-Yazied AA, Ibrahim MFM, et al. Ozone Induced Stomatal Regulations, MAPK and Phytohormone Signaling in Plants. International Journal of Molecular Sciences. 2021; 22(12):6304. https://doi.org/10.3390/ijms22126304
Chicago/Turabian StyleHasan, Md. Mahadi, Md. Atikur Rahman, Milan Skalicky, Nadiyah M. Alabdallah, Muhammad Waseem, Mohammad Shah Jahan, Golam Jalal Ahammed, Mohamed M. El-Mogy, Ahmed Abou El-Yazied, Mohamed F. M. Ibrahim, and et al. 2021. "Ozone Induced Stomatal Regulations, MAPK and Phytohormone Signaling in Plants" International Journal of Molecular Sciences 22, no. 12: 6304. https://doi.org/10.3390/ijms22126304
APA StyleHasan, M. M., Rahman, M. A., Skalicky, M., Alabdallah, N. M., Waseem, M., Jahan, M. S., Ahammed, G. J., El-Mogy, M. M., El-Yazied, A. A., Ibrahim, M. F. M., & Fang, X.-W. (2021). Ozone Induced Stomatal Regulations, MAPK and Phytohormone Signaling in Plants. International Journal of Molecular Sciences, 22(12), 6304. https://doi.org/10.3390/ijms22126304