Diabetes Mellitus and Its Implications in Aortic Stenosis Patients
Abstract
1. Introduction
2. Diabetes Mellitus as an Important Risk Factor of Aortic Stenosis
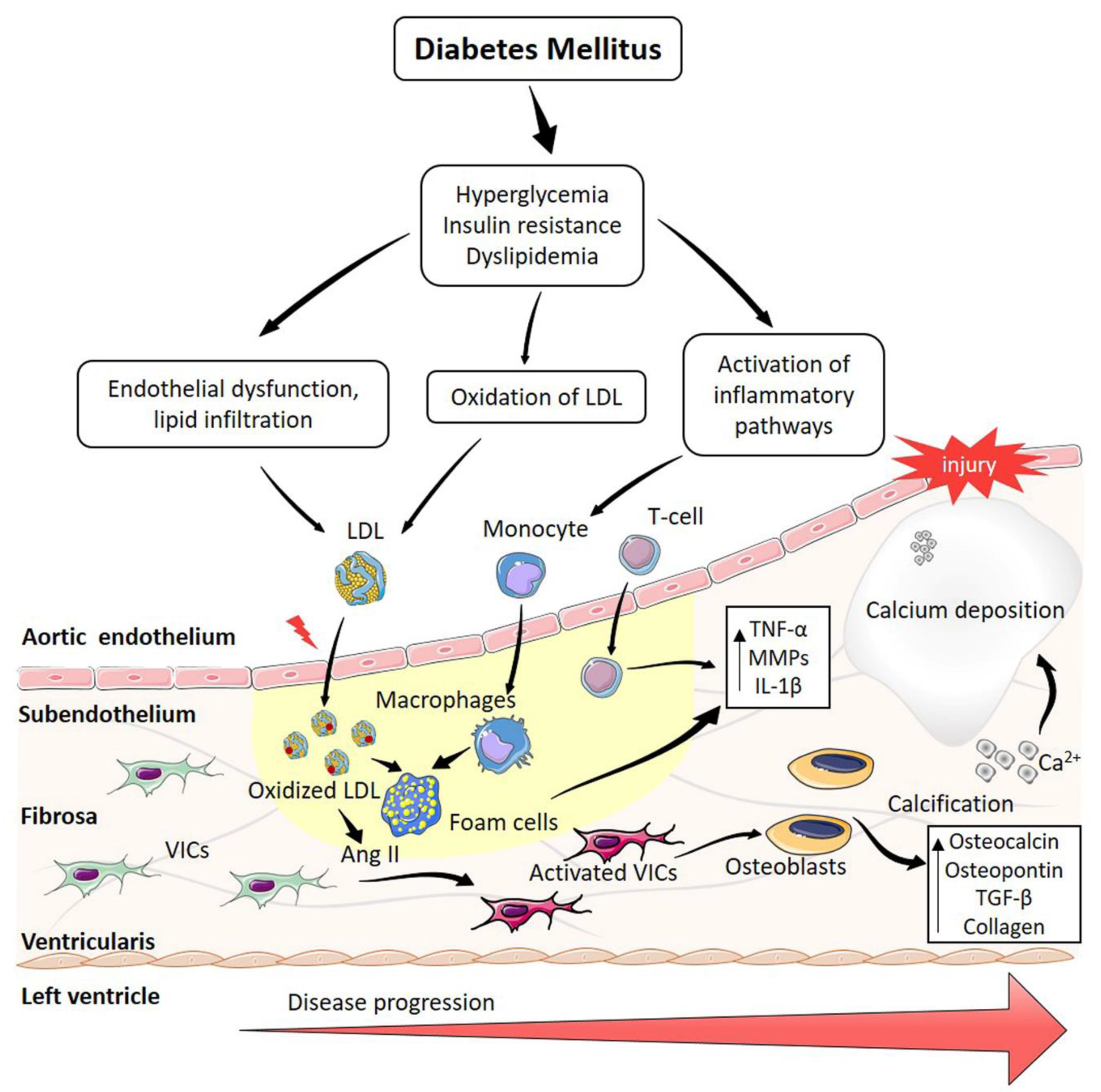
3. Role of Diabetes Mellitus in the Calcification of Aortic Stenosis
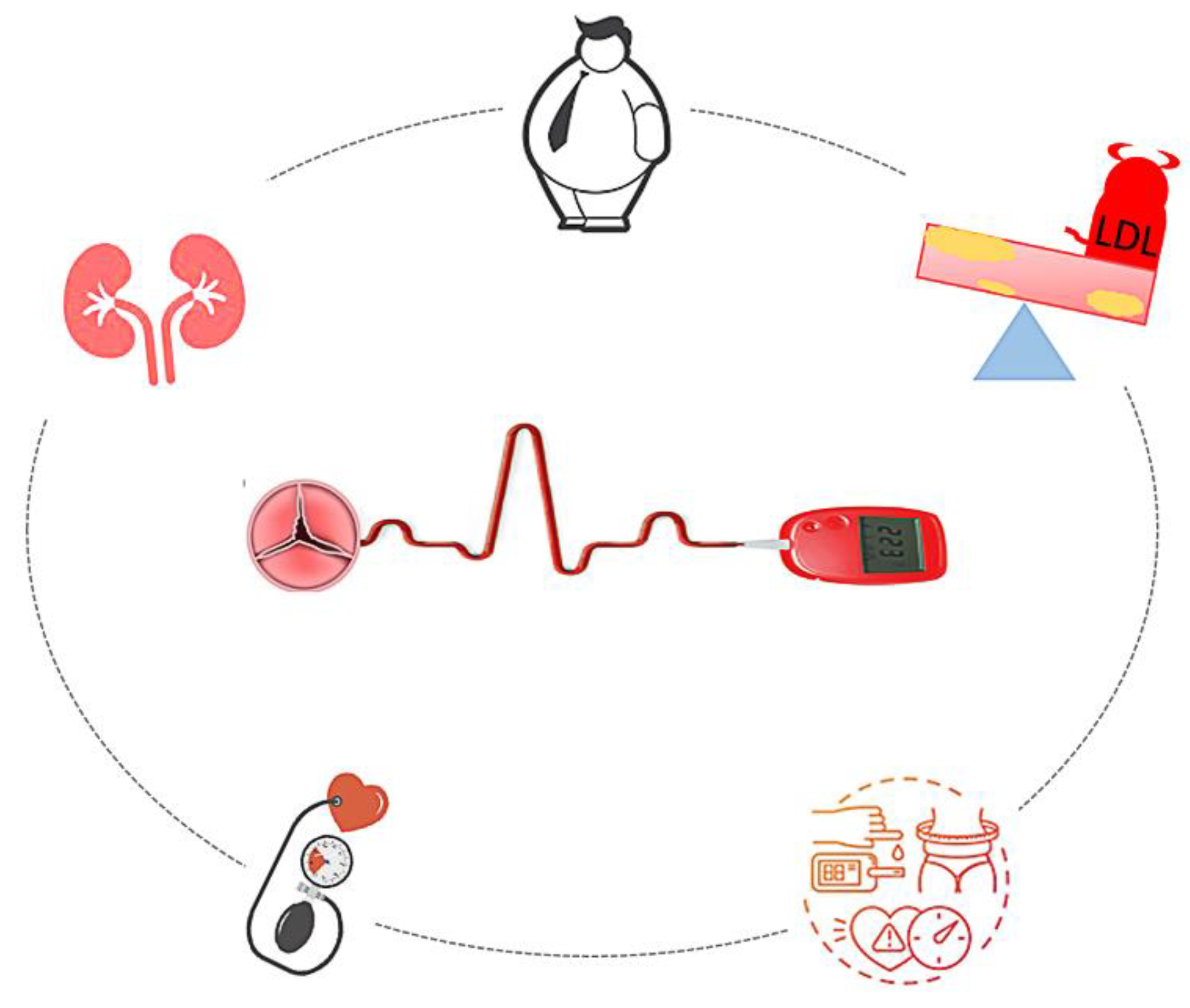
4. Comorbidities Common to Type2 Diabetes Mellitus and Degenerative Aortic Stenosis
4.1. Hypertension
4.2. Chronic Kidney Disease
4.3. Dyslipidemia and Atherosclerosis
4.4. Obesity
4.5. Metabolic Syndrome
5. The Importance of Risk Prediction in AS Patients
6. The Management of Patients with DAS and DM
6.1. Treatment/Therapies: Cardiac Effects during Glycaemic Control
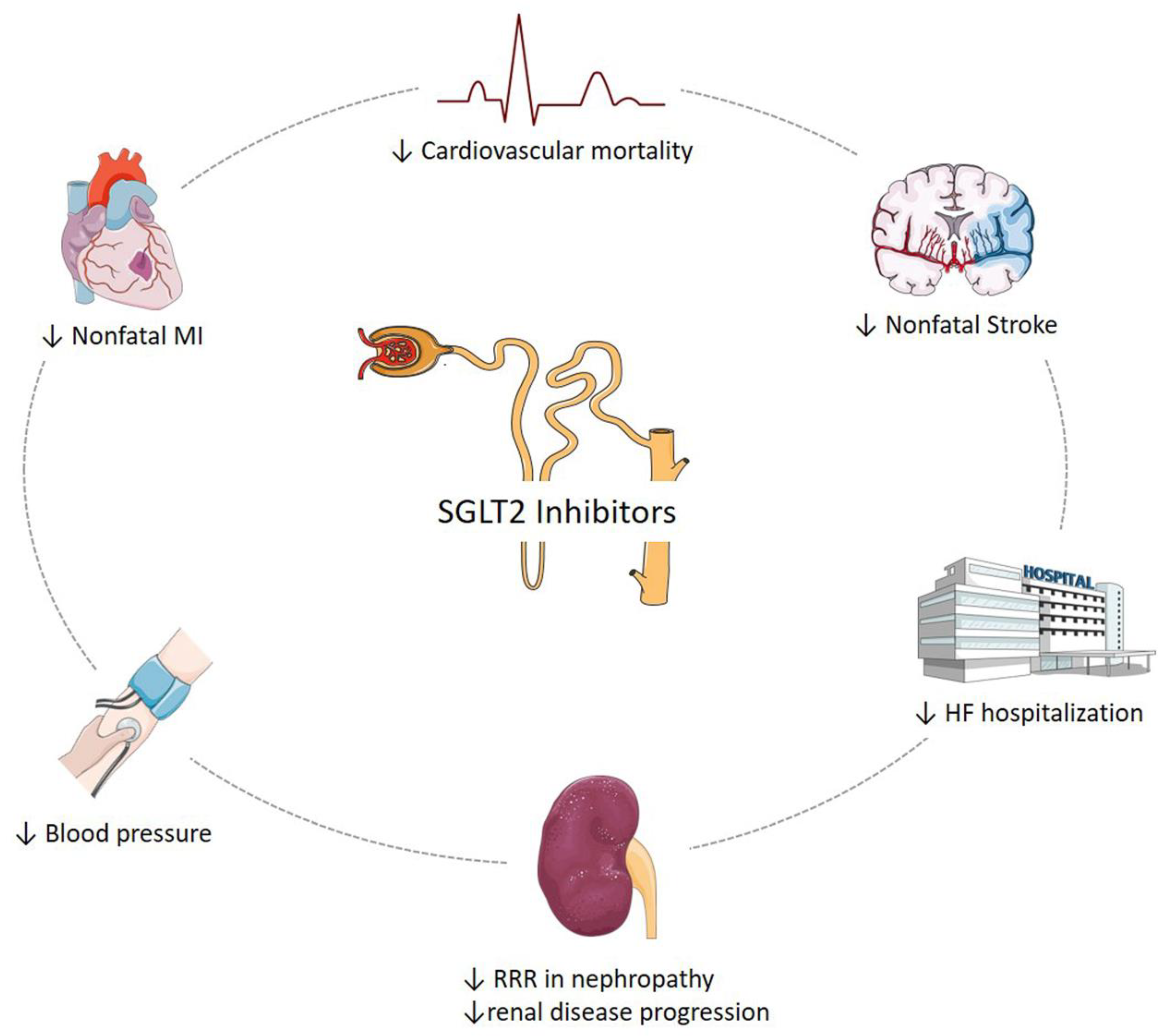
6.2. SGLT2 Inhibitors and Cardiovascular Outcomes
6.3. Treatment and Prognosis of AS in Patients with DM: The Impact of DM after Trans-Catheter AV Implantation
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lindman, B.; Clavel, M.-A.; Mathieu, P.; Iung, B.; Lancellotti, P.; Otto, C.M.; Pibarot, P. Calcific aortic stenosis. Nat. Rev. Dis. Prim. 2016, 2, 1–28. [Google Scholar] [CrossRef]
- Freeman, R.V.; Otto, C. Spectrum of Calcific Aortic Valve Disease. Circulation 2005, 111, 3316–3326. [Google Scholar] [CrossRef]
- Rajamannan, N.M.; Bonow, R.O.; Rahimtoola, S.H. Calcific aortic stenosis: An update. Nat. Clin. Pr. Neurol. 2007, 4, 254–262. [Google Scholar] [CrossRef]
- Falcão-Pires, I.; Hamdani, N.; Borbély, A.; Gavina, C.; Schalkwijk, C.G.; van der Velden, J.; van Heerebeek, L.; Stienen, G.; Niessen, H.W.; Leite-Moreira, A.F.; et al. Diabetes Mellitus Worsens Diastolic Left Ventricular Dysfunction in Aortic Stenosis Through Altered Myocardial Structure and Cardiomyocyte Stiffness. Circulation 2011, 124, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, R.A.; Otto, C.M.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P.; Guyton, R.A.; O’Gara, P.T.; Ruiz, C.E.; Skubas, N.J.; Sorajja, P.; et al. 2014 AHA/ACC Guideline for the Management of Patients with Valvular Heart Disease: Executive Summary. J. Am. Coll. Cardiol. 2014, 63, 2438–2488. [Google Scholar] [CrossRef]
- Kang, D.-H.; Park, S.-J.; Lee, S.-A.; Lee, S.; Kim, D.-H.; Kim, H.-K.; Yun, S.-C.; Hong, G.-R.; Song, J.-M.; Chung, C.-H.; et al. Early Surgery or Conservative Care for Asymptomatic Aortic Stenosis. N. Engl. J. Med. 2020, 382, 111–119. [Google Scholar] [CrossRef]
- Nashef, S.A.M.; Roques, F.; Michel, P.; Gauducheau, E.; Lemeshow, S.; Salamon, R. European system for cardiac operative risk evaluation (EuroSCORE). Eur. J. Cardio-Thorac. Surg. 1999, 16, 9–13. [Google Scholar] [CrossRef]
- Mathieu, P.; Boulanger, M.-C.; Bouchareb, R. Molecular biology of calcific aortic valve disease: Towards new pharmacological therapies. Expert Rev. Cardiovasc. Ther. 2014, 12, 851–862. [Google Scholar] [CrossRef]
- Banovic, M.; Athithan, L.; McCann, G.P. Aortic stenosis and diabetes mellitus: An ominous combination. Diabetes Vasc. Dis. Res. 2019, 16, 310–323. [Google Scholar] [CrossRef] [PubMed]
- Katz, R.; Wong, N.D.; Kronmal, R.; Takasu, J.; Shavelle, D.M.; Probstfield, J.L.; Bertoni, A.; Budoff, M.J.; O’Brien, K. Features of the Metabolic Syndrome and Diabetes Mellitus as Predictors of Aortic Valve Calcification in the Multi-Ethnic Study of Atherosclerosis. Circulation 2006, 113, 2113–2119. [Google Scholar] [CrossRef] [PubMed]
- Stewart, B.; Siscovick, D.; Lind, B.K.; Gardin, J.M.; Gottdiener, J.S.; Smith, V.E.; Kitzman, D.W.; Otto, C.M. Clinical Factors Associated with Calcific Aortic Valve Disease. J. Am. Coll. Cardiol. 1997, 29, 630–634. [Google Scholar] [CrossRef]
- Jawień, J. New insights into immunological aspects of atherosclerosis. Pol. Arch. Intern. Med. 2008, 118, 127–131. [Google Scholar] [CrossRef]
- Peacock, J.D.; Levay, A.K.; Gillaspie, D.B.; Tao, G.; Lincoln, J. Reduced Sox9 Function Promotes Heart Valve Calcification Phenotypes in Vivo. Circ. Res. 2010, 106, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Sathyamurthy, I.; Alex, S.; Kirubakaran, K.; Sengottuvelu, G.; Srinivasan, K. Risk factor profile of calcific aortic stenosis. Indian Heart J. 2016, 68, 828–831. [Google Scholar] [CrossRef][Green Version]
- Moura, L.M.; Ramos, S.F.; Zamorano, J.L.; Barros, I.M.; Azevedo, L.F.; Rocha-Gonçalves, F.; Rajamannan, N.M. Rosuvastatin Affecting Aortic Valve Endothelium to Slow the Progression of Aortic Stenosis. J. Am. Coll. Cardiol. 2007, 49, 554–561. [Google Scholar] [CrossRef]
- Hachicha, Z.; Dumesnil, J.G.; Bogaty, P.; Pibarot, P. Paradoxical Low-Flow, Low-Gradient Severe Aortic Stenosis Despite Preserved Ejection Fraction Is Associated with Higher Afterload and Reduced Survival. Circulation 2007, 115, 2856–2864. [Google Scholar] [CrossRef]
- Cowell, S.J.; Newby, D.E.; Prescott, R.J.; Bloomfield, P.; Reid, J.; Northridge, D.B.; Boon, N.A. A Randomized Trial of Intensive Lipid-Lowering Therapy in Calcific Aortic Stenosis. N. Engl. J. Med. 2005, 352, 2389–2397. [Google Scholar] [CrossRef]
- Ortlepp, J.R.; Schmitz, F.; Bozoglu, T.; Hanrath, P.; Hoffmann, R.G. Cardiovascular risk factors in patients with aortic stenosis predict prevalence of coronary artery disease but not of aortic stenosis: An angiographic pair matched case-control study. Heart 2003, 89, 1019–1022. [Google Scholar] [CrossRef] [PubMed]
- Stritzke, J.; Linsel-Nitschke, P.; Markus, M.R.P.; Mayer, B.; Lieb, W.; Luchner, A.; Döring, A.; Koenig, W.; Keil, U.; Hense, H.-W.; et al. Association between degenerative aortic valve disease and long-term exposure to cardiovascular risk factors: Results of the longitudinal population-based KORA/MONICA survey. Eur. Heart J. 2009, 30, 2044–2053. [Google Scholar] [CrossRef]
- Katz, R.; Budoff, M.J.; Takasu, J.; Shavelle, D.M.; Bertoni, A.; Blumenthal, R.S.; Ouyang, P.; Wong, N.D.; O’Brien, K. Relationship of Metabolic Syndrome with Incident Aortic Valve Calcium and Aortic Valve Calcium Progression: The Multi-Ethnic Study of Atherosclerosis (MESA). Diabetes 2009, 58, 813–819. [Google Scholar] [CrossRef]
- Yan, A.T.; Koh, M.; Chan, K.K.; Guo, H.; Alter, D.A.; Austin, P.C.; Tu, J.V.; Wijeysundera, H.C.; Ko, D.T. Association Between Cardiovascular Risk Factors and Aortic Stenosis. J. Am. Coll. Cardiol. 2017, 69, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Rosenhek, R.; Zilberszac, R.; Schemper, M.; Czerny, M.; Mundigler, G.; Graf, S.; Bergler-Klein, J.; Grimm, M.; Gabriel, H.; Maurer, G. Natural History of Very Severe Aortic Stenosis. Circulation 2010, 121, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Lancellotti, P.; Magne, J.; Dulgheru, R.; Clavel, M.-A.; Donal, E.; Vannan, M.A.; Chambers, J.; Rosenhek, R.; Habib, G.; Lloyd, G.; et al. Outcomes of Patients with Asymptomatic Aortic Stenosis Followed Up in Heart Valve Clinics. JAMA Cardiol. 2018, 3, 1060–1068. [Google Scholar] [CrossRef] [PubMed]
- Minamino-Muta, E.; Kato, T.; Morimoto, T.; Taniguchi, T.; Ando, K.; Kanamori, N.; Murata, K.; Kitai, T.; Kawase, Y.; Miyake, M.; et al. A risk prediction model in asymptomatic patients with severe aortic stenosis: CURRENT-AS risk score. Eur. Heart J.-Qual. Care Clin. Outcomes 2020, 6, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Xu, Y.; Pan, X.; Xu, J.; Ding, Y.; Sun, X.; Song, X.; Ren, Y.; Shan, P.-F. Global, regional, and national burden and trend of diabetes in 195 countries and territories: An analysis from 1990 to 2025. Sci. Rep. 2020, 10, 14790. [Google Scholar] [CrossRef]
- Capoulade, R.; Clavel, M.-A.; Dumesnil, J.G.; Chan, K.L.; Teo, K.K.; Tam, J.W.; Côté, N.; Mathieu, P.; Després, J.-P.; Pibarot, P. Insulin Resistance and LVH Progression in Patients with Calcific Aortic Stenosis: A Substudy of the ASTRONOMER Trial. JACC Cardiovasc. Imaging 2013, 6, 165–174. [Google Scholar] [CrossRef]
- Heath, J.M.; Sun, Y.; Yuan, K.; Bradley, W.E.; Litovsky, S.; Dell’Italia, L.J.; Chatham, J.C.; Wu, H.; Chen, Y. Activation of AKT by O-Linked N-Acetylglucosamine Induces Vascular Calcification in Diabetes Mellitus. Circ. Res. 2014, 114, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- McDonald, T.O.; Gerrity, R.G.; Jen, C.; Chen, H.-J.; Wark, K.; Wight, T.N.; Chait, A.; O’Brien, K. Diabetes and Arterial Extracellular Matrix Changes in a Porcine Model of Atherosclerosis. J. Histochem. Cytochem. 2007, 55, 1149–1157. [Google Scholar] [CrossRef]
- Mangat, R.; Warnakula, S.; Borthwick, F.; Hassanali, Z.; Uwiera, R.R.; Russell, J.C.; Cheeseman, C.I.; Vine, D.F.; Proctor, S.D. Arterial Retention of Remnant Lipoproteins Ex Vivo Is Increased in Insulin Resistance Because of Increased Arterial Biglycan and Production of Cholesterol-Rich Atherogenic Particles That Can Be Improved by Ezetimibe in the JCR:LA- cp Rat. J. Am. Heart Assoc. 2012, 1, e003434. [Google Scholar] [CrossRef]
- Scatena, M.; Jackson, M.F.; Speer, M.Y.; Leaf, E.M.; Wallingford, M.C.; Giachelli, C.M. Increased Calcific Aortic Valve Disease in response to a diabetogenic, procalcific diet in the LDLr −/− ApoB 100/100 mouse model. Cardiovasc. Pathol. 2018, 34, 28–37. [Google Scholar] [CrossRef]
- Le Quang, K.; Bouchareb, R.; Lachance, D.; Laplante, M.-A.; Husseini, D.E.; Boulanger, M.-C.; Fournier, D.; Fang, X.P.; Avramoglu, R.K.; Pibarot, P.; et al. Early Development of Calcific Aortic Valve Disease and Left Ventricular Hypertrophy in a Mouse Model of Combined Dyslipidemia and Type 2 Diabetes Mellitus. Arter. Thromb. Vasc. Biol. 2014, 34, 2283–2291. [Google Scholar] [CrossRef]
- Natorska, J.; Wypasek, E.; Grudzień, G.; Sobczyk, D.; Marek, G.; Filip, G.; Sadowski, J.; Undas, A. Does Diabetes Accelerate the Progression of Aortic Stenosis through Enhanced Inflammatory Response within Aortic valves? Inflammation 2011, 35, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Barzilay, J.I.; Abraham, L.; Heckbert, S.R.; Cushman, M.; Kuller, L.H.; Resnick, H.E.; Tracy, R.P. The Relation of Markers of Inflammation to the Development of Glucose Disorders in the Elderly: The Cardiovascular Health Study. Diabetes 2001, 50, 2384–2389. [Google Scholar] [CrossRef] [PubMed]
- Sjöholm, Å.; Nyström, T. Inflammation and the etiology of type 2 diabetes. Diabetes/Metab. Res. Rev. 2005, 22, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Gleissner, C.A.; Galkina, E.; Nadler, J.L.; Ley, K. Mechanisms by which diabetes increases cardiovascular disease. Drug Discov. Today: Dis. Mech. 2007, 4, 131–140. [Google Scholar] [CrossRef]
- Gleißner, C.A. The vulnerable vessel. Hämostaseologie 2015, 35, 267–271. [Google Scholar] [CrossRef]
- Wilhelmsen, L.; Welin, L.; Svärdsudd, K.; Wedel, H.; Eriksson, H.; Rosengren, A.; Hansson, P.-O. Secular changes in cardiovascular risk factors and attack rate of myocardial infarction among men aged 50 in Gothenburg, Sweden. Accurate prediction using risk models. J. Intern. Med. 2008, 263, 636–643. [Google Scholar] [CrossRef] [PubMed]
- The Emerging Risk Factors Collaboration. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010, 375, 2215–2222. [Google Scholar] [CrossRef]
- Mosch, J.; Gleissner, C.A.; Body, S.; Aikawa, E. Histopathological assessment of calcification and inflammation of calcific aortic valves from patients with and without diabetes mellitus. Histol. Histopathol. 2016, 32, 293–306. [Google Scholar]
- Hernandez, B.; Reilly, R.B.; Kenny, R.A. Investigation of multimorbidity and prevalent disease combinations in older Irish adults using network analysis and association rules. Sci. Rep. 2019, 9, 14567. [Google Scholar] [CrossRef]
- Johnston, M.C.; Crilly, M.; Black, C.; Prescott, G.; Mercer, S.W. Defining and measuring multimorbidity: A systematic review of systematic reviews. Eur. J. Public Health 2019, 29, 182–189. [Google Scholar] [CrossRef] [PubMed]
- De Souza, D.L.B.; Oliveras-Fabregas, A.; Espelt, A.; Bosque-Prous, M.; Cancela, M.D.C.; Teixidó-Compañó, E.; Jerez-Roig, J. Multimorbidity and its associated factors among adults aged 50 and over: A cross-sectional study in 17 European countries. PLoS ONE 2021, 16, e0246623. [Google Scholar] [CrossRef]
- Rudolph, T.K.; Messika-Zeitoun, D.; Frey, N.; Thambyrajah, J.; Serra, A.; Schulz, E.; Maly, J.; Aiello, M.; Lloyd, G.; Bortone, A.S.; et al. Impact of selected comorbidities on the presentation and management of aortic stenosis. Open Heart 2020, 7, e001271. [Google Scholar] [CrossRef]
- Nowakowska, M.; Zghebi, S.S.; Ashcroft, D.M.; Buchan, I.; Chew-Graham, C.; Holt, T.; Mallen, C.; Van Marwijk, H.; Peek, N.; Perera-Salazar, R.; et al. The comorbidity burden of type 2 diabetes mellitus: Patterns, clusters and predictions from a large English primary care cohort. BMC Med. 2019, 17, 145. [Google Scholar] [CrossRef] [PubMed]
- Leon, B.M. Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J. Diabetes 2015, 6, 1246–1258. [Google Scholar] [CrossRef]
- Montezano, A.C.; Touyz, R.M. Molecular Mechanisms of Hypertension—Reactive Oxygen Species and Antioxidants: A Basic Science Update for the Clinician. Can. J. Cardiol. 2012, 28, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Engert, J.C.; Thanassoulis, G. Risk factors for valvular calcification. Curr. Opin. Endocrinol. Diabetes Obes. 2019, 26, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Do, A.N.L.; Dagogo-Jack, S. Comorbidities of Diabetes and Hypertension: Mechanisms and Approach to Target Organ Protection. J. Clin. Hypertens. 2011, 13, 244–251. [Google Scholar] [CrossRef]
- Saeed, S.; Scalise, F.; Chambers, J.B.; Mancia, G. Hypertension in aortic stenosis: A focused review and recommendations for clinical practice. J. Hypertens. 2020, 38, 1211–1219. [Google Scholar] [CrossRef]
- Rossebø, A.B.; Pedersen, T.R.; Boman, K.; Brudi, P.; Chambers, J.B.; Egstrup, K.; Gerdts, E.; Gohlke-Bärwolf, C.; Holme, I.; Kesäniemi, Y.A.; et al. Intensive Lipid Lowering with Simvastatin and Ezetimibe in Aortic Stenosis. N. Engl. J. Med. 2008, 359, 1343–1356. [Google Scholar] [CrossRef]
- Otto, C.M.; Nishimura, R.A.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P.; Gentile, F.; Jneid, H.; Krieger, E.V.; Mack, M.; McLeod, C.; et al. 2020 ACC/AHA Guideline for the Management of Patients with Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 2021, 143, e72–e227. [Google Scholar] [CrossRef]
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- Vavilis, G.; Bäck, M.; Occhino, G.; Trevisan, M.; Bellocco, R.; Evans, M.; Lindholm, B.; Szummer, K.; Carrero, J.J. Kidney Dysfunction and the Risk of Developing Aortic Stenosis. J. Am. Coll. Cardiol. 2019, 73, 305–314. [Google Scholar] [CrossRef]
- Bohbot, Y.; Candellier, A.; Diouf, M.; Rusinaru, D.; Altes, A.; Pasquet, A.; Maréchaux, S.; Vanoverschelde, J.; Tribouilloy, C. Severe Aortic Stenosis and Chronic Kidney Disease: Outcomes and Impact of Aortic Valve Replacement. J. Am. Heart Assoc. 2020, 9, e017190. [Google Scholar] [CrossRef] [PubMed]
- Hahr, A.J.; Molitch, M.E. Management of diabetes mellitus in patients with chronic kidney disease. Clin. Diabetes Endocrinol. 2015, 1, 2. [Google Scholar] [CrossRef]
- De Boer, I.H.; Bakris, G.L. Diabetic Kidney Disease: A Determinant of Cardiovascular Risk in Type 1 Diabetes. Diabetes Care 2018, 41, 662–663. [Google Scholar] [CrossRef]
- Hedayatnia, M.; Asadi, Z.; Zare-Feyzabadi, R.; Yaghooti-Khorasani, M.; Ghazizadeh, H.; Ghaffarian-Zirak, R.; Nosrati-Tirkani, A.; Mohammadi-Bajgiran, M.; Rohban, M.; Sadabadi, F.; et al. Dyslipidemia and cardiovascular disease risk among the MASHAD study population. Lipids Health Dis. 2020, 19, 42. [Google Scholar] [CrossRef] [PubMed]
- Garg, R.; Aggarwal, S.; Kumar, R.; Sharma, G. Association of atherosclerosis with dyslipidemia and co-morbid conditions: A descriptive study. J. Nat. Sci. Biol. Med. 2015, 6, 163–168. [Google Scholar] [CrossRef]
- Arsenault, B.J.; Boekholdt, S.M.; Dubé, M.-P.; Rhéaume, E.; Wareham, N.J.; Khaw, K.-T.; Sandhu, M.S.; Tardif, J.-C. Lipoprotein(a) Levels, Genotype, and Incident Aortic Valve Stenosis. Circ. Cardiovasc. Genet. 2014, 7, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Hafiane, A.; Thanassoulis, G.; Ott, L.; Filwood, N.; Cerruti, M.; Gourgas, O.; Shum-Tim, D.; Al Kindi, H.; de Varennes, B.; et al. Lipoprotein(a) Induces Human Aortic Valve Interstitial Cell Calcification. JACC Basic Transl. Sci. 2017, 2, 358–371. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Khan, K.; Hamid, Q.; Mardini, A.; Siddique, A.; Aguilar-Gonzalez, L.P.; Makhoul, G.; Alaws, H.; Genest, J.; Thanassoulis, G.; et al. Pathological significance of lipoprotein(a) in aortic valve stenosis. Atherosclerosis 2018, 272, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.L.; Hess, C.N.; Hiatt, W.R.; Goldfine, A.B. Clinical Update: Cardiovascular Disease in Diabetes Mellitus. Circulation 2016, 133, 2459–2502. [Google Scholar] [CrossRef] [PubMed]
- Katakami, N. Mechanism of Development of Atherosclerosis and Cardiovascular Disease in Diabetes Mellitus. J. Atheroscler. Thromb. 2018, 25, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Dumesnil, J.G.; Pibarot, P. The Obesity Paradox in Aortic Stenosis. J. Am. Coll. Cardiol. 2013, 62, 1691–1693. [Google Scholar] [CrossRef]
- Thanassoulis, G.; Campbell, C.Y.; Owens, D.S.; Smith, J.G.; Smith, A.V.; Peloso, G.M.; Kerr, K.F.; Pechlivanis, S.; Budoff, M.J.; Harris, T.B.; et al. Genetic Associations with Valvular Calcification and Aortic Stenosis. N. Engl. J. Med. 2013, 368, 503–512. [Google Scholar] [CrossRef]
- LlNDROOS, M.; Kupari, M.; Valvanne, J.; Strandberg, T.; Heikkilä, J.; TlLVIS, R. Factors associated with calcific aortic valve degeneration in the elderly. Eur. Heart J. 1994, 15, 865–870. [Google Scholar] [CrossRef]
- Larsson, S.C.; Wolk, A.; Håkansson, N.; Bäck, M. Overall and abdominal obesity and incident aortic valve stenosis: Two prospective cohort studies. Eur. Heart J. 2017, 38, 2192–2197. [Google Scholar] [CrossRef] [PubMed]
- Kaltoft, M.; Langsted, A.; Nordestgaard, B.G. Obesity as a Causal Risk Factor for Aortic Valve Stenosis. J. Am. Coll. Cardiol. 2020, 75, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Bramante, C.T.; Lee, C.J.; Gudzune, K.A. Treatment of Obesity in Patients with Diabetes. Diabetes Spectr. 2017, 30, 237–243. [Google Scholar] [CrossRef]
- Chobot, A.; Górowska-Kowolik, K.; Sokołowska, M.; Jarosz-Chobot, P. Obesity and diabetes-Not only a simple link between two epidemics. Diabetes/Metab. Res. Rev. 2018, 34, e3042. [Google Scholar] [CrossRef]
- Aguilar-Salinas, C.A.; Viveros-Ruiz, T. Recent advances in managing/understanding the metabolic syndrome. F1000Research 2019, 8, 370. [Google Scholar] [CrossRef]
- Ezquerra, E.A.; Vázquez, J.M.C.; Alegria-Barrero, A. (Ana) Obesity, Metabolic Syndrome, and Diabetes: Cardiovascular Implications and Therapy. Rev. Esp. Cardiol. 2008, 61, 752–764. [Google Scholar] [CrossRef]
- Capoulade, R.; Clavel, M.-A.; Dumesnil, J.G.; Chan, K.L.; Teo, K.K.; Tam, J.W.; Côté, N.; Mathieu, P.; Després, J.-P.; Pibarot, P. Impact of Metabolic Syndrome on Progression of Aortic Stenosis. J. Am. Coll. Cardiol. 2012, 60, 216–223. [Google Scholar] [CrossRef]
- Go, J.L.; Prem, K.; Al-Hijji, M.A.; Qin, Q.; Noble, C.; Young, M.D.; Lerman, L.O.; Lerman, A. Experimental Metabolic Syndrome Model Associated with Mechanical and Structural Degenerative Changes of the Aortic Valve. Sci. Rep. 2018, 8, 17835. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-K.; Han, K.; Kim, M.K.; Koh, E.S.; Kim, E.S.; Nam, G.E.; Kwon, H.-S. Changes in metabolic syndrome and its components and the risk of type 2 diabetes: A nationwide cohort study. Sci. Rep. 2020, 10, 2313. [Google Scholar] [CrossRef]
- Baumgartner, H.; Falk, V.; Bax, J.J.; De Bonis, M.; Hamm, C.; Holm, P.J.; Iung, B.; Lancellotti, P.; Lansac, E.; Muñoz, D.R.; et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur. Heart J. 2017, 38, 2739–2791. [Google Scholar] [CrossRef]
- Taniguchi, T.; Morimoto, T.; Shiomi, H.; Ando, K.; Kanamori, N.; Murata, K.; Kitai, T.; Kawase, Y.; Izumi, C.; Miyake, M.; et al. Initial Surgical Versus Conservative Strategies in Patients with Asymptomatic Severe Aortic Stenosis. J. Am. Coll. Cardiol. 2015, 66, 2827–2838. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.-H.; Park, S.-J.; Rim, J.H.; Yun, S.-C.; Kim, D.-H.; Song, J.-M.; Choo, S.J.; Park, S.W.; Song, J.-K.; Lee, J.-W.; et al. Early Surgery Versus Conventional Treatment in Asymptomatic Very Severe Aortic Stenosis. Circulation 2010, 121, 1502–1509. [Google Scholar] [CrossRef]
- Taniguchi, T.; Morimoto, T.; Shiomi, H.; Ando, K.; Kanamori, N.; Murata, K.; Kitai, T.; Kawase, Y.; Izumi, C.; Kato, T.; et al. Sudden Death in Patients with Severe Aortic Stenosis: Observations From the CURRENT AS Registry. J. Am. Heart Assoc. 2018, 7, e008397. [Google Scholar] [CrossRef]
- January, C.T.; Wann, L.S.; Calkins, H.; Chen, L.Y.; Cigarroa, J.E.; Cleveland, J.C.; Ellinor, P.T.; Ezekowitz, M.D.; Field, M.E.; Furie, K.L.; et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients with Atrial Fibrillation. J. Am. Coll. Cardiol. 2019, 74, 104–132. [Google Scholar] [CrossRef]
- Douglas, P.S.; Garcia, M.J.; Haines, D.E.; Lai, W.W.; Manning, W.J.; Patel, A.R.; Picard, M.H.; Polk, D.M.; Ragosta, M.; Ward, R.P.; et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 appropriate use criteria for echocardiography. A report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, American Society of Echocardiography, American Heart Association, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Critical Care Medicine, Society of Cardiovascular Computed Tomography, and Society for Cardiovascular Magnetic Resonance Endorsed by the American College of Chest Physicians. J. Am. Coll. Cardiol. 2011, 57, 1126–1166. [Google Scholar] [CrossRef] [PubMed]
- Rosen, A.B.; Fowler, V.G.; Corey, G.R.; Downs, S.M.; Biddle, A.K.; Li, J.; Jollis, J.G. Cost-effectiveness of transesophageal echocardiography to determine the duration of therapy for intravascular catheter-associated Staphylococcus aureus bacteremia. Ann. Intern. Med. 1999, 130, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Zuern, C.S.; Eick, C.; Rizas, K.; Stoleriu, C.; Barthel, P.; Scherer, C.; Müller, K.A.L.; Gawaz, M.; Bauer, A. Severe autonomic failure in moderate to severe aortic stenosis: Prevalence and association with hemodynamics and biomarkers. Clin. Res. Cardiol. 2012, 101, 565–572. [Google Scholar] [CrossRef]
- Chace, D.H.; Kalas, T.A. A biochemical perspective on the use of tandem mass spectrometry for newborn screening and clinical testing. Clin. Biochem. 2005, 38, 296–309. [Google Scholar] [CrossRef] [PubMed]
- Belczacka, I.; Latosinska, A.; Metzger, J.; Marx, D.; Vlahou, A.; Mischak, H.; Frantzi, M. Proteomics biomarkers for solid tumors: Current status and future prospects. Mass Spectrom. Rev. 2019, 38, 49–78. [Google Scholar] [CrossRef] [PubMed]
- Mischak, H.; Delles, C.; Vlahou, A.; Vanholder, R. Proteomic biomarkers in kidney disease: Issues in development and implementation. Nat. Rev. Nephrol. 2015, 11, 221–232. [Google Scholar] [CrossRef]
- Maisel, A.S.; Krishnaswamy, P.; Nowak, R.M.; Mccord, J.; Hollander, J.E.; Duc, P.; Omland, T.; Storrow, A.B.; Abraham, W.T.; Wu, A.H.; et al. Rapid Measurement of B-Type Natriuretic Peptide in the Emergency Diagnosis of Heart Failure. N. Engl. J. Med. 2002, 347, 161–167. [Google Scholar] [CrossRef]
- Newby, L.K.; Christenson, R.H.; Ohman, E.M.; Armstrong, P.; Thompson, T.D.; Lee, K.L.; Hamm, C.W.; Katus, H.A.; Cianciolo, C.; Granger, C.B.; et al. Value of Serial Troponin T Measures for Early and Late Risk Stratification in Patients with Acute Coronary Syndromes. Circulation 1998, 98, 1853–1859. [Google Scholar] [CrossRef]
- Mourino-Alvarez, L.; Martin-Rojas, T.; Corros-Vicente, C.; Corbacho-Alonso, N.; Padial, L.R.; Solis, J.; Barderas, M.G. Patient Management in Aortic Stenosis: Towards Precision Medicine Through Protein Analysis, Imaging and Diagnostic Tests. J. Clin. Med. 2020, 9, 2421. [Google Scholar] [CrossRef]
- Olkowicz, M.; Debski, J.; Jablonska, P.; Dadlez, M.; Smolenski, R.T. Application of a new procedure for liquid chromatography/mass spectrometry profiling of plasma amino acid-related metabolites and untargeted shotgun proteomics to identify mechanisms and biomarkers of calcific aortic stenosis. J. Chromatogr. A 2017, 1517, 66–78. [Google Scholar] [CrossRef]
- Mourino-Alvarez, L.; Baldan-Martin, M.; Gonzalez-Calero, L.; Martinez-Laborde, C.; Sastre-Oliva, T.; Moreno-Luna, R.; Lopez-Almodovar, L.F.; Sanchez, P.L.; Fernandez-Aviles, F.; Vivanco, F.; et al. Patients with calcific aortic stenosis exhibit systemic molecular evidence of ischemia, enhanced coagulation, oxidative stress and impaired cholesterol transport. Int. J. Cardiol. 2016, 225, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Van Driel, B.; Schuldt, M.; Algül, S.; Levin, E.; Güclü, A.; Germans, T.; Rossum, A.; Pei, J.; Harakalova, M.; Baas, A.; et al. Metabolomics in Severe Aortic Stenosis Reveals Intermediates of Nitric Oxide Synthesis as Most Distinctive Markers. Int. J. Mol. Sci. 2021, 22, 3569. [Google Scholar] [CrossRef]
- Al Hageh, C.; Rahy, R.; Khazen, G.; Brial, F.; Khnayzer, R.S.; Gauguier, D.; Zalloua, P.A. Plasma and urine metabolomic analyses in aortic valve stenosis reveal shared and biofluid-specific changes in metabolite levels. PLoS ONE 2020, 15, e0242019. [Google Scholar] [CrossRef] [PubMed]
- Haase, D.; Bäz, L.; Bekfani, T.; Neugebauer, S.; Kiehntopf, M.; Möbius-Winkler, S.; Franz, M.; Schulze, P.C. Metabolomic profiling of patients with high gradient aortic stenosis undergoing transcatheter aortic valve replacement. Clin. Res. Cardiol. 2021, 110, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Surendran, A.; Edel, A.; Chandran, M.; Bogaert, P.; Hassan-Tash, P.; Asokan, A.K.; Hiebert, B.; Solati, Z.; Sandhawalia, S.; Raabe, M.; et al. Metabolomic Signature of Human Aortic Valve Stenosis. JACC Basic Transl. Sci. 2020, 5, 1163–1177. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-Z.; Gerszten, R.E. Metabolomics and Proteomics in Type 2 Diabetes. Circ. Res. 2020, 126, 1613–1627. [Google Scholar] [CrossRef]
- Flores, E.; Santos-Gallego, C.G.; Diaz-Mejía, N.; Badimon, J.J. Do the SGLT-2 Inhibitors Offer More than Hypoglycemic Activity? Cardiovasc. Drugs Ther. 2018, 32, 213–222. [Google Scholar] [CrossRef]
- Scheen, A.J. Pharmacodynamics, Efficacy and Safety of Sodium–Glucose Co-Transporter Type 2 (SGLT2) Inhibitors for the Treatment of Type 2 Diabetes Mellitus. Drugs 2015, 75, 33–59. [Google Scholar] [CrossRef]
- Shubrook, J.H.; Bokaie, B.B.; Adkins, S.E. Empagliflozin in the treatment of type 2 diabetes: Evidence to date. Drug Des. Dev. Ther. 2015, 9, 5793–5803. [Google Scholar] [CrossRef] [PubMed]
- Triplitt, C.; Cornell, S. Canagliflozin Treatment in Patients with Type 2 Diabetes Mellitus. Clin. Med. Insights Endocrinol. Diabetes 2015, 8, 73–81. [Google Scholar] [CrossRef]
- Fioretto, P.; Giaccari, A.; Sesti, G. Efficacy and safety of dapagliflozin, a sodium glucose cotransporter 2 (SGLT2) inhibitor, in diabetes mellitus. Cardiovasc. Diabetol. 2015, 14, 142. [Google Scholar] [CrossRef]
- Marx, N.; McGuire, D.K. Sodium-glucose cotransporter-2 inhibition for the reduction of cardiovascular events in high-risk patients with diabetes mellitus. Eur. Heart J. 2016, 37, 3192–3200. [Google Scholar] [CrossRef] [PubMed]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.H.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef] [PubMed]
- Wanner, C.; Inzucchi, S.E.; Lachin, J.M.; Fitchett, D.; Von Eynatten, M.; Mattheus, M.; Johansen, O.E.; Woerle, H.J.; Broedl, U.C.; Zinman, B. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 323–334. [Google Scholar] [CrossRef]
- Abdul-Ghani, M.; Del Prato, S.; Chilton, R.; De Fronzo, R.A. SGLT2 Inhibitors and Cardiovascular Risk: Lessons Learned From the EMPA-REG OUTCOME Study. Diabetes Care 2016, 39, 717–725. [Google Scholar] [CrossRef]
- Santos-Gallego, C.G.; Requena-Ibanez, J.A.; Antonio, R.S.; Garcia-Ropero, A.; Ishikawa, K.; Watanabe, S.; Picatoste, B.; Vargas-Delgado, A.P.; Flores-Umanzor, E.J.; Sanz, J.; et al. Empagliflozin Ameliorates Diastolic Dysfunction and Left Ventricular Fibrosis/Stiffness in Nondiabetic Heart Failure. JACC Cardiovasc. Imaging 2021, 14, 393–407. [Google Scholar] [CrossRef]
- Santos-Gallego, C.G.; Garcia-Ropero, A.; Mancini, D.; Pinney, S.P.; Contreras, J.P.; Fergus, I.; Abascal, V.; Moreno, P.; Atallah-Lajam, F.; Tamler, R.; et al. Rationale and Design of the EMPA-TROPISM Trial (ATRU-4): Are the “Cardiac Benefits” of Empagliflozin Independent of its Hypoglycemic Activity? Cardiovasc. Drugs Ther. 2019, 33, 87–95. [Google Scholar] [CrossRef]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Trimarco, B.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Neal, B.; Perkovic, V.; Mahaffey, K.W.; De Zeeuw, D.; Fulcher, G.; Erondu, N.; Shaw, W.; Law, G.; Desai, M.; Matthews, D.R. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 644–657. [Google Scholar] [CrossRef]
- Ryan, P.B.; Buse, J.B.; Schuemie, M.J.; DeFalco, F.; Yuan, Z.; Stang, P.E.; Berlin, J.A.; Rosenthal, N. Comparative effectiveness of canagliflozin, SGLT2 inhibitors and non-SGLT2 inhibitors on the risk of hospitalization for heart failure and amputation in patients with type 2 diabetes mellitus: A real-world meta-analysis of 4 observational databases (OBSERVE-4D). Diabetes Obes. Metab. 2018, 20, 2585–2597. [Google Scholar] [CrossRef] [PubMed]
- Quattrocchi, E.; Goldberg, T.; Marzella, N. Management of type 2 diabetes: Consensus of diabetes organizations. Drugs Context 2020, 9, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Halkos, M.E.; Kilgo, P.; Lattouf, O.M.; Puskas, J.D.; Cooper, W.A.; Guyton, R.A.; Thourani, V.H. The Effect of Diabetes Mellitus on In-Hospital and Long-Term Outcomes after Heart Valve Operations. Ann. Thorac. Surg. 2010, 90, 124–130. [Google Scholar] [CrossRef]
- Nakamura, T.; Toda, K.; Kuratani, T.; Miyagawa, S.; Yoshikawa, Y.; Fukushima, S.; Saito, S.; Yoshioka, D.; Kashiyama, N.; Daimon, T.; et al. Diabetes Mellitus Impairs Left Ventricular Mass Regression after Surgical or Transcatheter Aortic Valve Replacement for Severe Aortic Stenosis. Heart Lung Circ. 2016, 25, 68–74. [Google Scholar] [CrossRef] [PubMed]
- López-De-Andrés, A.; Perez-Farinos, N.; De Miguel-Díez, J.; Hernández-Barrera, V.; Méndez-Bailón, M.; De Miguel-Yanes, J.M.; Garcia, R.J. Impact of type 2 diabetes mellitus in the utilization and in-hospital outcomes of surgical aortic valve replacement in Spain (2001–2015). Cardiovasc. Diabetol. 2018, 17, 135. [Google Scholar] [CrossRef]
- Lindman, B.R.; Pibarot, P.; Arnold, S.V.; Suri, R.M.; McAndrew, T.C.; Maniar, H.S.; Zajarias, A.; Kodali, S.; Kirtane, A.J.; Thourani, V.H.; et al. Transcatheter Versus Surgical Aortic Valve Replacement in Patients with Diabetes and Severe Aortic Stenosis at High Risk for Surgery. J. Am. Coll. Cardiol. 2014, 63, 1090–1099. [Google Scholar] [CrossRef] [PubMed]
- Tzamalis, P.; Herzberger, V.; Bergmann, J.; Wuerth, A.; Bramlage, P.; Schroefel, H.; Schmitt, C.; Schymik, G. The association of diabetes mellitus treated with oral antidiabetic drugs and insulin with mortality after transcatheter valve implantation: A 3-year follow-up of the TAVIK registry. Cardiovasc. Diabetol. 2019, 18, 63. [Google Scholar] [CrossRef]
- Tokarek, T.; Dziewierz, A.; Wiktorowicz, A.; Bagienski, M.; Rzeszutko, L.; Sorysz, D.; Kleczynski, P.; Dudek, D. Effect of diabetes mellitus on clinical outcomes and quality of life after transcatheter aortic valve implantation for severe aortic valve stenosis. Hell. J. Cardiol. 2018, 59, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Mendez-Bailon, M.; Lorenzo-Villalba, N.; Muñoz-Rivas, N.; De Miguel-Yanes, J.M.; De Miguel-Diez, J.; Comín-Colet, J.; Hernandez-Barrera, V.; Garcia, R.J.; Lopez-De-Andres, A. Transcatheter aortic valve implantation and surgical aortic valve replacement among hospitalized patients with and without type 2 diabetes mellitus in Spain (2014-2015). Cardiovasc. Diabetol. 2017, 16, 144. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, X.; He, Y.; Tang, M.; Zhu, Q.; Xu, X.; Guo, D.; Shi, K.; Hu, P.; Wang, L.; et al. Meta-Analysis of Impact of Diabetes Mellitus on Outcomes After Transcatheter Aortic Valve Implantation. Am. J. Cardiol. 2017, 119, 623–629. [Google Scholar] [CrossRef]
- Abramowitz, Y.; Vemulapalli, S.; Chakravarty, T.; Li, Z.; Kapadia, S.; Holmes, D.; Matsouaka, R.A.; Wang, A.; Cheng, W.; Forrester, J.S.; et al. Clinical Impact of Diabetes Mellitus on Outcomes After Transcatheter Aortic Valve Replacement. Circ. Cardiovasc. Interv. 2017, 10, e005417. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mourino-Alvarez, L.; Corbacho-Alonso, N.; Sastre-Oliva, T.; Corros-Vicente, C.; Solis, J.; Tejerina, T.; Padial, L.R.; Barderas, M.G. Diabetes Mellitus and Its Implications in Aortic Stenosis Patients. Int. J. Mol. Sci. 2021, 22, 6212. https://doi.org/10.3390/ijms22126212
Mourino-Alvarez L, Corbacho-Alonso N, Sastre-Oliva T, Corros-Vicente C, Solis J, Tejerina T, Padial LR, Barderas MG. Diabetes Mellitus and Its Implications in Aortic Stenosis Patients. International Journal of Molecular Sciences. 2021; 22(12):6212. https://doi.org/10.3390/ijms22126212
Chicago/Turabian StyleMourino-Alvarez, Laura, Nerea Corbacho-Alonso, Tamara Sastre-Oliva, Cecilia Corros-Vicente, Jorge Solis, Teresa Tejerina, Luis R. Padial, and Maria G. Barderas. 2021. "Diabetes Mellitus and Its Implications in Aortic Stenosis Patients" International Journal of Molecular Sciences 22, no. 12: 6212. https://doi.org/10.3390/ijms22126212
APA StyleMourino-Alvarez, L., Corbacho-Alonso, N., Sastre-Oliva, T., Corros-Vicente, C., Solis, J., Tejerina, T., Padial, L. R., & Barderas, M. G. (2021). Diabetes Mellitus and Its Implications in Aortic Stenosis Patients. International Journal of Molecular Sciences, 22(12), 6212. https://doi.org/10.3390/ijms22126212






