Ionic Liquids—A Review of Their Toxicity to Living Organisms
Abstract
1. Introduction
2. Effect of Ionic Liquids on Microorganisms
2.1. Bacteria
2.2. Fungi
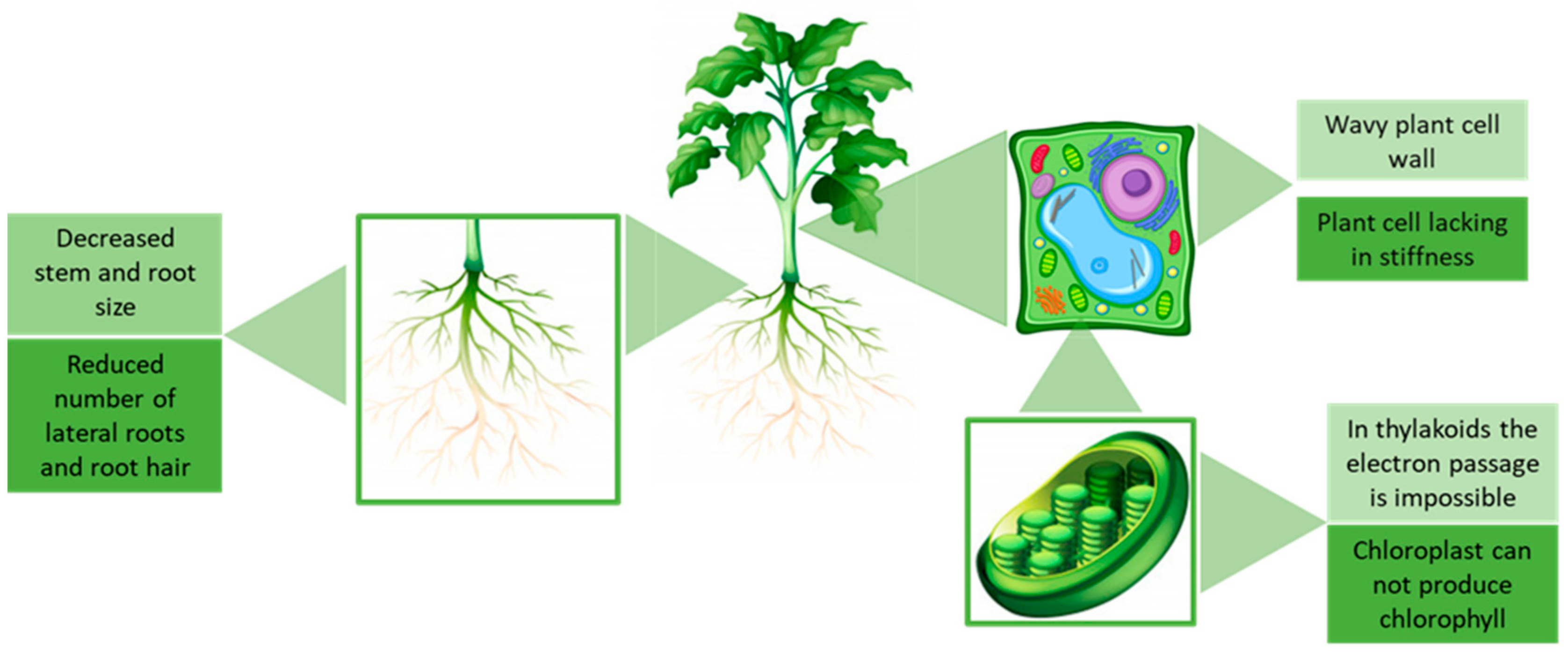
3. Effect of Ionic Liquids on Plants
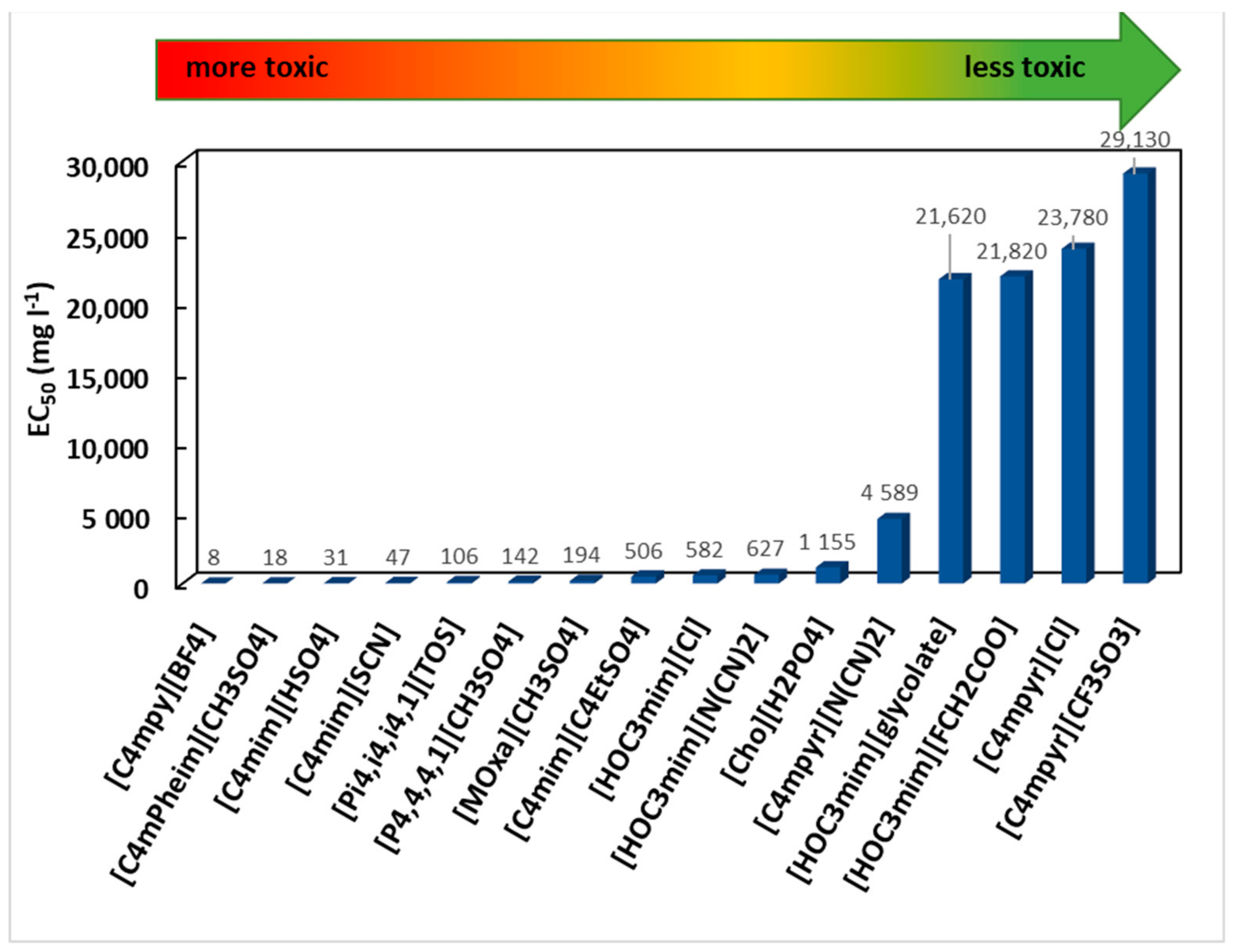
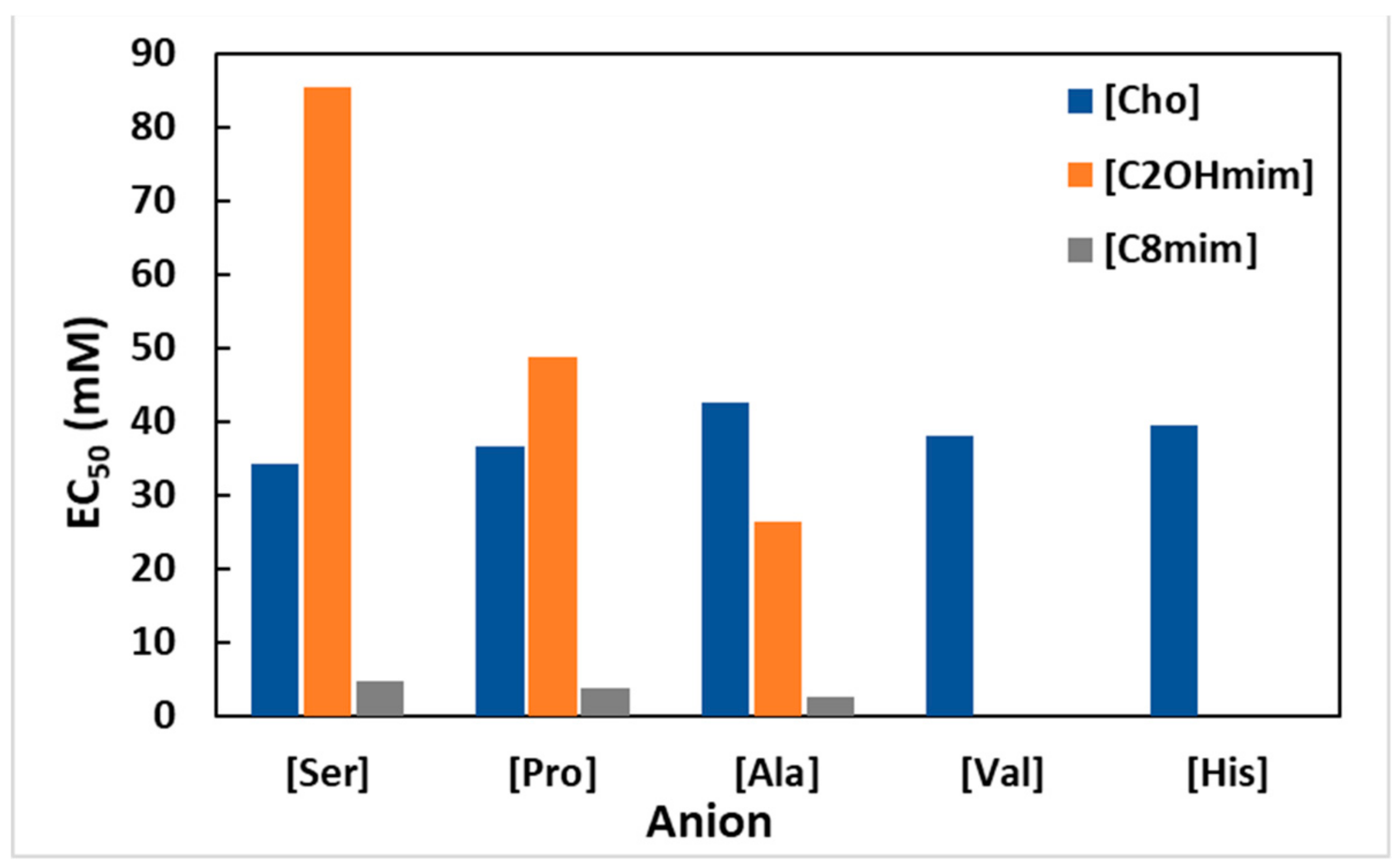
4. Effect of Ionic Liquids on Animals
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| A. corymbifera | Absidia corymbifera (fungus) |
| A. | Alcaligenes (Gram-negative bacteria) |
| A. fischeri | Aliivibrio fischeri (Gram-negative bacteria) |
| A. fumigatus | Aspergillus fumigatus (fungus) |
| A. hydrophila | Aeromonas hydrophila (Gram-negative bacteria) |
| A. niger | Aspergillus niger (fungus) |
| ACY I | Acylase I |
| AILs | Aprotic ionic liquids |
| amoA | Ammonia monooxygenase subunit A gene |
| AOA | Ammonia-oxidizing archaea |
| AOB | Ammonia-oxidizing bacteria |
| B. cereus | Bacillus cereus (Gram-positive bacteria) |
| B. japonicum | Bradyrhizobium japonicum (Gram-negative bacteria) |
| B. licheniformis | Bacillus licheniforms (Gram-positive bacteria) |
| B. subtilis | Bacillus subtilis (Gram-positive bacteria) |
| BOD | Biochemical oxygen demand |
| BS-C-1 | African green monkey kidney cells |
| BSLT | Brine shrimp lethality test |
| BTFIP | 3-bis(2,2,2-trifluoroethoxy)propan-2-ol |
| C. albicans | Candida albicans (yeast) |
| C. elegans | Caenorhabditis elegans |
| C. freundii | Citrobacter freundii (Gram-negative bacteria) |
| C. glabrata | Candida glabrata (yeast) |
| C. krusei | Candida krusei (yeast) |
| C. lusitaniae | Candida lusitaniae (yeast) |
| C. parapsilosis | Candida parapsilosis (yeast) |
| C. pyrenoidosa | Chlorella pyrenoidosa |
| C. tropicalis | Candida tropicalis (yeast) |
| CAT | Catalase |
| CCO | Channel Catfish Ovary cell line |
| CHO | Chinese hamster ovary cells |
| CMC | Critical micellar concentration |
| COD | Chemical oxygen demand |
| D. japonica | Dugesia japonica |
| D. magna | Daphnia magna |
| DESI-MSI | Desorption electrospray ionization–mass spectrometry imaging |
| DMSO | Dimethyl sulfoxide |
| E. coli | Escherichia coli (Gram-negative bacteria) |
| E. faecalis | Enterococcus faecalis (Gram-positive bacteria) |
| E. fetida | Eisenia fetida |
| EC50 | Half-maximal effective concentration |
| ED100 | Preservative concentrations retarding the fungal growth by 100% than the reference |
| ELM | Extreme learning machine |
| ESH | Environmental, safety and health impact |
| G. mellonella | Galleria mellonella |
| G. stearothermophilus | Geobacillus stearothermophilus (Gram-positive bacteria) |
| G3TFSA | Triglyme |
| G4TFSA | Tetraglyme |
| GHS | United Nations globally harmonized system |
| GPx | Glutathione peroxidase |
| GSH | Glutathione |
| GST | Glutathione S-transferase |
| H2O | Water |
| H2O2 | Hydrogen peroxide |
| HaCaT | Human immortal keratinocyte cells |
| HeLa | Human cervical carcinoma cells |
| HepG2 | Human hepatocellular carcinoma cells |
| Hs68 | Human fibroblast cell line |
| IC50 | 50% inhibitory concentration |
| IC80 | 80% inhibitory concentration |
| IC95 | 95% inhibitory concentration |
| ILs | Ionic liquids |
| IPC-81 | Rat leukemia cell line |
| K. pneumoniae | Klebsiella pneumoniae (Gram-negative bacteria) |
| L. lactis | Lactobacillus lactis (Gram-positive bacteria) |
| L. monocytogenes | Listeria monocytogenes (Gram-positive bacteria) |
| L. sakei | Lactobacillus sakei (Gram-positive bacteria) |
| LC50 | 50% lethal concentration |
| LD50 | Median lethal dose |
| LDH | Lactic dehydrogenase |
| LFER | Linear free energy relationship descriptors |
| LPS | Lipopolysaccharide |
| M. catarrahalis | Moraxella catarrhalis (Gram-negative bacteria) |
| MBC | Minimum bactericidal concentration |
| MDA | Malondialdehyde |
| MDA-MB-231 cells | Human breast cancerous cells |
| MIC | Minimum inhibitory concentration |
| NRRT | Neutral red retention assay |
| O2•− | Superoxide radicals |
| OECD | Organization for Economic Cooperation and Development |
| OH•− | Hydroxyl radicals |
| P. aeruginosa | Pseudomonas aeruginosa (Gram-negative bacteria) |
| P. fluorescens | Pseudomonas fluorescens (Gram-negative bacteria) |
| P. mirabilis | Proteus mirabilis (Gram-negative bacteria) |
| P. putida | Pseudomonas putida (Gram-negative bacteria) |
| P. vulgaris | Proteus vulgaris (Gram-negative bacteria) |
| PANC-1 cells | Human pancreatic cancerous cells |
| PILs | Protic ionic liquids |
| POD | Peroxidase |
| PSI | Photosynthetic system I |
| PSII | Photosynthetic system II |
| QSAR | Quantitative structure–activity relationship model |
| R. rubra | Rhodoturula rubra (yeast) |
| RAPD | Randomly amplified polymorphic DNA assay |
| RCOO | Peroxide radicals |
| ROS | Reactive oxygen species |
| S. aureus | Staphylococcus aureus (Gram-positive bacteria) |
| S. cerevisiae | Saccharomyces cerevisiae (yeast) |
| S. enterica | Salmonella enterica (Gram-negative bacteria) |
| S. epidermidis | Staphylococcus epidermidis (Gram-positive bacteria) |
| S. marcescens | Serrata marcescens (Gram-negative bacteria) |
| S. obliquus | Scenedesmus obliquus |
| S. typhimurium | Salmonella typhimurium (Gram-negative bacteria) |
| SEM | Scanning electron microscopy |
| SIA | Sequential injection analysis |
| SOD | Superoxide dismutase |
| T. asahii | Trichosporon asahii (fungus) |
| T. mentagrophytes | Trichophyton mentagrophytes (fungus) |
| T. novellus | Thiobacillus novellus (Gram-negative bacteria) |
| ThOD | Theoretical oxygen demand |
| [BA] | Alkyl-benzyldimethylammonium |
| [BTDA] | Benzyldimethyltetradecylammonium |
| [C10-Men-Am] | [(1R,2S,5R)-(−)-Menthoxymethyl] dimethyldecylammonium |
| [C10mim] | 1-Decyl-3-methylimidazolium |
| [C11-Men-Am] | [(1R,2S,5R)-(−)-Menthoxymethyl] dimethylundecylammonium |
| [N1,1,12,2(OH)] | Dodecyl(2-hydroxyethyl)dimethylammonium |
| [C12-Men-Am] | [(1R,2S,5R)-(−)-menthoxymethyl] dimethyldodecylammonium |
| [C12mim] | 1-Dodecyl-3-methylimidazolium |
| [C14mim] | 1-Tetradecyl-3-methylimidazolium |
| [C16mim] | 1-Hexadecyl-3-methylimidazolium |
| [C1mim] | 1,3-Dimethylimidazolium |
| [C1OC2mim] | 1-Methoxyethyl-3-methylimidazolium |
| [C1pi] | 1-Methylpiperazinium |
| [(C2)2(C1)2(C1)23gu] | 2-Ethyl-1,1,3,3-tetramethylguanidinium |
| [(C2)22(C1)2(C1)23gu] | 2,2-Diethyl-1,1,3,3-tetramethylguanidinium |
| [C2mim] | 1-Ethyl-3-methylimidazolium |
| [C2mpy] | 1-Ethyl-3-methylpyridinium |
| [OHC2C1NH2] | N-Methyl-2-hydroxyethylammonium |
| [OHC2mim] | 1-(2-Hydroxyethyl)-3-methylimidazolium |
| [C2pi] | 1-Ethylpiperazinium |
| [C3mpyr] | 1-Methyl-1-propylpyrrolidinium |
| [C4mim] | 1-Butyl-3-methylimidazolium |
| [C4mPheim] | 1-Butyl-3-methyl-2-phenylimidazolium |
| [C4mpy] | 1-Butyl-4-methylpyridinium |
| [C4mpyr] | 1-Butyl-1-methylpyrrolidinium |
| [C4py] | N-Butylpyridinium |
| [C6mim] | 1-Hexyl-3-methylimidazolium |
| [C6mpy] | 1-Hexyl-1-methylpyrrolidinium |
| [C8mim] | 1-Octyl-3-methylimidazolium |
| [C8OC1mim] | 1-Octyloximethyl-3-methylimidazolium |
| [Cho] | Choline |
| [DBNH] | 1,5-Diazabicyclo(4.3.0)non-5-enium |
| [DMEEEAH] | N,N-Dimethyl-2-(2-ethoxyethoxy)ethylammonium |
| [DMEtAH] | N,N-Dimethylethanolammonium |
| [DMHEEAH] | N,N-Dimethyl-2-(2-hydroxyethoxy)ethylammonium |
| [HOC2mim] | 1-Hydroxyethyl-3-methylimidazolium |
| [HOC3mim] | Hydroxypropylmethylimidazolium |
| [MOxa] | Methyloxazolinium |
| [N1,1,1,1] | Tetramethylammonium |
| [N1,1,1,16] | Trimethylhexadecylammonium |
| [N1,1,1,2(OH)] | (2-Hydroxyethyl)trimethylammonium |
| [N1,1,1,8] | Trimethyloctylammonium |
| [N1,1,12,2(OH)] | Dodecyl(2-hydroxyethyl)dimethylammonium |
| [N1,1,18,18] | Dimethyldioctadecylammonium |
| [N1,1,2,2(OH)] | (2-Hydroxyethyl)ethyldimethylammonium |
| [N1,1,6,2(OH)] | Hexyl(2-hydroxyethyl)dimethylammonium |
| [N1,1,8,2(OH)] | Octyl(2-hydroxyethyl)dimethylammonium |
| [N1,4,2(OH),2(OH)] | Butyldi(2-hydroxyethyl)methylammonium |
| [N1,6,2(OH),2(OH)] | Hexyldi(2-hydroxyethyl)methylammonium |
| [N10,10,1,1] | Didecyldimethylammonium |
| [N4,4,4,4] | Tetrabutylammonium |
| [N6,2(OH),2(OH),2(OH)] | Hexyltri(2-hydroxyethyl)ammonium |
| [N8,8,1,1] | Dioctyldimethylammonium |
| [N8,8,8,1] | Trioctylmethylammonium |
| [NH2C2mim] | 1-aminoethyl-3-methylimidazolium |
| [OHC3mim] | Hydroxypropylmethylimidazolium |
| [P14,4,4,4] | Tributyl(tetradecyl)phosphonium |
| [P4,4,4,1] | Tributylmethylphosphonium |
| [P4,4,4,4] | Tetrabutylphosphonium |
| [P6,6,6,14] | Tri(hexyl)tetradecylphosphonium |
| [P8,4,4,4] | Tri(butyl)octylphosphonium |
| [P8,8,8,1] | Trioctylmethylphosphonium |
| [Phpi] | 1-phenylpiperazinium |
| [Pi4,i4,i4,1] | Triisobutyl(methyl)phosphonium |
| [TMEEEA] | N,N,N-Trimethyl-2-(2-ethoxyethoxy)ethylammonium |
| [TMHEEA] | N,N,N-Trimethyl-2-(2-hydroxyethoxy)ethylammonium |
| [(C2F5)3PF3] | Tris(pentafluoroethyl)trifluorophosphate |
| [(CF3SO2)2N] | Bis(trifluoromethylsulfonyl)imide |
| [3AT] | 3-Amino-1,2,4-triazolate |
| [Ala] | Alaninate |
| [Arg] | Argininate |
| [Asc] | Ascorbate |
| [Asp] | Asparaginate |
| [BF4] | Tetrafluoroborate |
| [Biot] | Biotinate |
| [Br] | Bromide |
| [Bt] | Benzotrizolate |
| [C(CN)3] | Tricyanomethanide |
| [C2H5COO] | Propionate |
| [C2H5OSO3] | Ethylsulfate |
| [C3H7COO] | Butyrate |
| [C4H9COO] | Pentanoate |
| [C5H11COO] | Hexanoate |
| [C7H5O2] | Benzoate |
| [C9H19COO] | Decanoate |
| [CF3SO3] | Trifluoromethanesulfonate |
| [CH3COO] | Acetate |
| [CH3OSO3] | Methylsulfate |
| [CH3SO3] | Methanesulfonate |
| [CH3SO4] | Methylsulfate |
| [Cit] | Citrate |
| [Cl] | Chloride |
| [ClO4] | Perchlorate |
| [CoCl4] | Cobalt tetrachloride |
| [DEP] | Diethylphosphate |
| [DMP] | Dimethylphosphate |
| [DMSIP] | Dimethyl-5-sulfoisophthalate |
| [docusate] | Docusate |
| [EtOPr] | 3-Ethoxypropionate |
| [FCH2COO] | Fluoroacetate |
| [FeCl4] | Iron tetrachloride |
| [GdCl6] | Gadolinium hexachloride |
| [Gly] | Glycinate |
| [glycolate] | Glycolate |
| [H2PO4] | Dihydrogen phosphate |
| [HSO4] | Hydrogen sulfate |
| [I] | Iodide |
| [Lac] | Lactate |
| [Lau] | Laurate |
| [Mal] | Malate |
| [Md] | Mandelate |
| [MDEGSO4] | 2-(2-Methoxyethoxy)ethylsulfate |
| [MnCl4] | Manganese tetrachloride |
| [MnCl4] | Manganese tetrachloride |
| [N(CN)2] | Dicyanamide |
| [Nicot] | Nicotinate |
| [NO3] | Nitrate |
| [O2C2O2] | Oxalate |
| [OH] | Hydroxide |
| [Ol] | Oleate |
| [PF6] | Hexafluorophosphate |
| [Phe] | Phenylalanine |
| [Pro] | Prolinate |
| [Pyr] | Pyrithione |
| [SCN] | Thiocyanate |
| [Ser] | Serine |
| [Tau] | Taurine |
| [TMPP] | Bis(2,4,4-trimethyl)phosphate |
| [TOS] | Tosylate |
References
- Plechkova, N.V.; Seddon, K.R. Applications of Ionic Liquids in the Chemical Industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef]
- Greer, A.J.; Jacquemin, J.; Hardacre, C. Industrial Applications of Ionic Liquids. Molecules 2020, 25, 5207. [Google Scholar] [CrossRef]
- Seddon, K.R. Ionic Liquids for Clean Technology. J. Chem. Technol. Biotechnol. 1997, 68, 351–356. [Google Scholar] [CrossRef]
- Feng, R.; Zhao, D.; Guo, Y. Revisiting Characteristics of Ionic Liquids: A Review for Further Application Development. J. Environ. Prot. 2010, 1, 95–104. [Google Scholar] [CrossRef]
- Martins, V.L.; Torresi, R.M. Ionic Liquids in Electrochemical Energy Storage. Curr. Opin. Electrochem. 2018, 26–32. [Google Scholar] [CrossRef]
- Rogers, R.D.; Seddon, K.R. Ionic Liquids-Solvents of the Future? Science 2003, 302, 792–793. [Google Scholar] [CrossRef]
- Aparicio, S.; Atilhan, M.; Karadas, F. Thermophysical Properties of Pure Ionic Liquids: Review of Present Situation. Ind. Eng. Chem. Res. 2010, 49, 9580–9595. [Google Scholar] [CrossRef]
- Gaune-Escard, M.; Seddon, K.R. (Eds.) Molten Salts and Ionic Liquids: Never the Twain? John Wiley: Hoboken, NJ, USA, 2010; ISBN 978-0-471-77392-4. [Google Scholar]
- Armand, M.; Endres, F.; MacFarlane, D.R.; Ohno, H.; Scrosati, B. Ionic-Liquid Materials for the Electrochemical Challenges of the Future. Nat. Mater. 2009, 8, 621–629. [Google Scholar] [CrossRef]
- Welton, T.; Endres, F.; Abedin, S.Z.E.; Antonietti, M.; Smarsly, B.; Zhou, Y. Inorganic Synthesis. In Ionic Liquids in Synthesis; Wasserscheid, P., Welton, T., Eds.; John Wiley: Hoboken, NJ, USA, 2007; pp. 569–617. ISBN 978-3-527-62119-4. [Google Scholar]
- Malla, A.M.; Parveen, M.; Ahmad, F.; Azaz, S.; Alam, M. [Et3NH][HSO4]-Catalyzed Eco-Friendly and Expeditious Synthesis of Thiazolidine and Oxazolidine Derivatives. RSC Adv. 2015, 5, 19552–19569. [Google Scholar] [CrossRef]
- Zhang, Q.; Shreeve, J.M. Energetic Ionic Liquids as Explosives and Propellant Fuels: A New Journey of Ionic Liquid Chemistry. Chem. Rev. 2014, 114, 10527–10574. [Google Scholar] [CrossRef]
- Ribeiro, A.P.C.; Vieira, S.I.C.; França, J.M.; Queirós, C.S.; Langa, E.; Lourenço, M.J.V.; Murshed, S.M.S.; de Castro, C.A.N. Thermal Properties of Ionic Liquids and Ionanofluids. In Ionic Liquids: Theory, Properties, New Approaches; IntechOpen: Rijeka, Croatia, 2011. [Google Scholar] [CrossRef]
- Prado, R.; Weber, C.C. Chapter 1—Applications of Ionic Liquids. In Application, Purification, and Recovery of Ionic Liquids; Kuzmina, O., Hallett, J.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–58. ISBN 978-0-444-63713-0. [Google Scholar]
- Zhang, Z.; Song, J.; Han, B. Catalytic Transformation of Lignocellulose into Chemicals and Fuel Products in Ionic Liquids. Chem. Rev. 2017, 117, 6834–6880. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.; Gräsvik, J.; Hallett, J.P.; Welton, T. Deconstruction of Lignocellulosic Biomass with Ionic Liquids. Green Chem. 2013, 15, 550–583. [Google Scholar] [CrossRef]
- Marrucho, I.M.; Branco, L.C.; Rebelo, L.P.N. Ionic Liquids in Pharmaceutical Applications. Annu. Rev. Chem. Biomol. Eng. 2014, 5, 527–546. [Google Scholar] [CrossRef]
- Earle, M.J.; Seddon, K.R. Ionic Liquids. Green Solvents for the Future. Pure Appl. Chem. 2000, 72, 1391–1398. [Google Scholar] [CrossRef]
- Cvjetko Bubalo, M.; Hanousek, K.; Radošević, K.; Gaurina Srček, V.; Jakovljević, T.; Radojčić Redovniković, I. Imidiazolium Based Ionic Liquids: Effects of Different Anions and Alkyl Chains Lengths on the Barley Seedlings. Ecotoxicol. Environ. Saf. 2014, 101, 116–123. [Google Scholar] [CrossRef]
- Neumann, J.; Steudte, S.; Cho, C.-W.; Thöming, J.; Stolte, S. Biodegradability of 27 Pyrrolidinium, Morpholinium, Piperidinium, Imidazolium and Pyridinium Ionic Liquid Cations under Aerobic Conditions. Green Chem. 2014, 16, 2174–2184. [Google Scholar] [CrossRef]
- Pawłowska, B.; Telesiński, A.; Biczak, R. Phytotoxicity of Ionic Liquids. Chemosphere 2019, 237, 124436. [Google Scholar] [CrossRef]
- Tang, S.L.Y.; Smith, R.L.; Poliakoff, M. Principles of Green Chemistry: PRODUCTIVELY. Green Chem. 2005, 7, 761–762. [Google Scholar] [CrossRef]
- Anastas, P.; Eghbali, N. Green Chemistry: Principles and Practice. Chem. Soc. Rev. 2009, 39, 301–312. [Google Scholar] [CrossRef]
- Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemicals Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as Well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC. Volume 396. 2006. Available online: http://data.europa.eu/eli/reg/2006/1907/2014-04-10 (accessed on 12 February 2021).
- Pernak, J.; Rogoża, J.; Mirska, I. Synthesis and Antimicrobial Activities of New Pyridinium and Benzimidazolium Chlorides. Eur. J. Med. Chem. 2001, 36, 313–320. [Google Scholar] [CrossRef]
- Pernak, J.; Kalewska, J.; Ksycińska, H.; Cybulski, J. Synthesis and Anti-Microbial Activities of Some Pyridinium Salts with Alkoxymethyl Hydrophobic Group. Eur. J. Med. Chem. 2001, 36, 899–907. [Google Scholar] [CrossRef]
- Amde, M.; Liu, J.-F.; Pang, L. Environmental Application, Fate, Effects, and Concerns of Ionic Liquids: A Review. Environ. Sci. Technol. 2015, 49, 12611–12627. [Google Scholar] [CrossRef]
- Heckenbach, M.E.; Romero, F.N.; Green, M.D.; Halden, R.U. Meta-Analysis of Ionic Liquid Literature and Toxicology. Chemosphere 2016, 150, 266–274. [Google Scholar] [CrossRef]
- Egorova, K.S.; Gordeev, E.G.; Ananikov, V.P. Biological Activity of Ionic Liquids and Their Application in Pharmaceutics and Medicine. Chem. Rev. 2017, 117, 7132–7189. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, M.; Moosavi, F. The Effects of Temperature, Alkyl Chain Length, and Anion Type on Thermophysical Properties of the Imidazolium Based Amino Acid Ionic Liquids. J. Mol. Liq. 2018, 250, 121–130. [Google Scholar] [CrossRef]
- Tian, T.; Hu, X.; Guan, P.; Ding, X. Research on Solubility and Bio-Solubility of Amino Acids Ionic Liquids. J. Mol. Liq. 2017, 225, 224–230. [Google Scholar] [CrossRef]
- Costa, S.P.F.; Pinto, P.C.A.G.; Lapa, R.A.S.; Saraiva, M.L.M.F.S. Toxicity Assessment of Ionic Liquids with Vibrio Fischeri: An Alternative Fully Automated Methodology. J. Hazard. Mater. 2015, 284, 136–142. [Google Scholar] [CrossRef]
- Montalbán, M.G.; Hidalgo, J.M.; Collado-González, M.; Díaz Baños, F.G.; Víllora, G. Assessing Chemical Toxicity of Ionic Liquids on Vibrio Fischeri: Correlation with Structure and Composition. Chemosphere 2016, 155, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Mellado, N.; Ayuso, M.; Villar-Chavero, M.M.; García, J.; Rodríguez, F. Ecotoxicity Evaluation towards Vibrio Fischeri of Imidazolium- and Pyridinium-Based Ionic Liquids for Their Use in Separation Processes. SN Appl. Sci. 2019, 1, 896. [Google Scholar] [CrossRef]
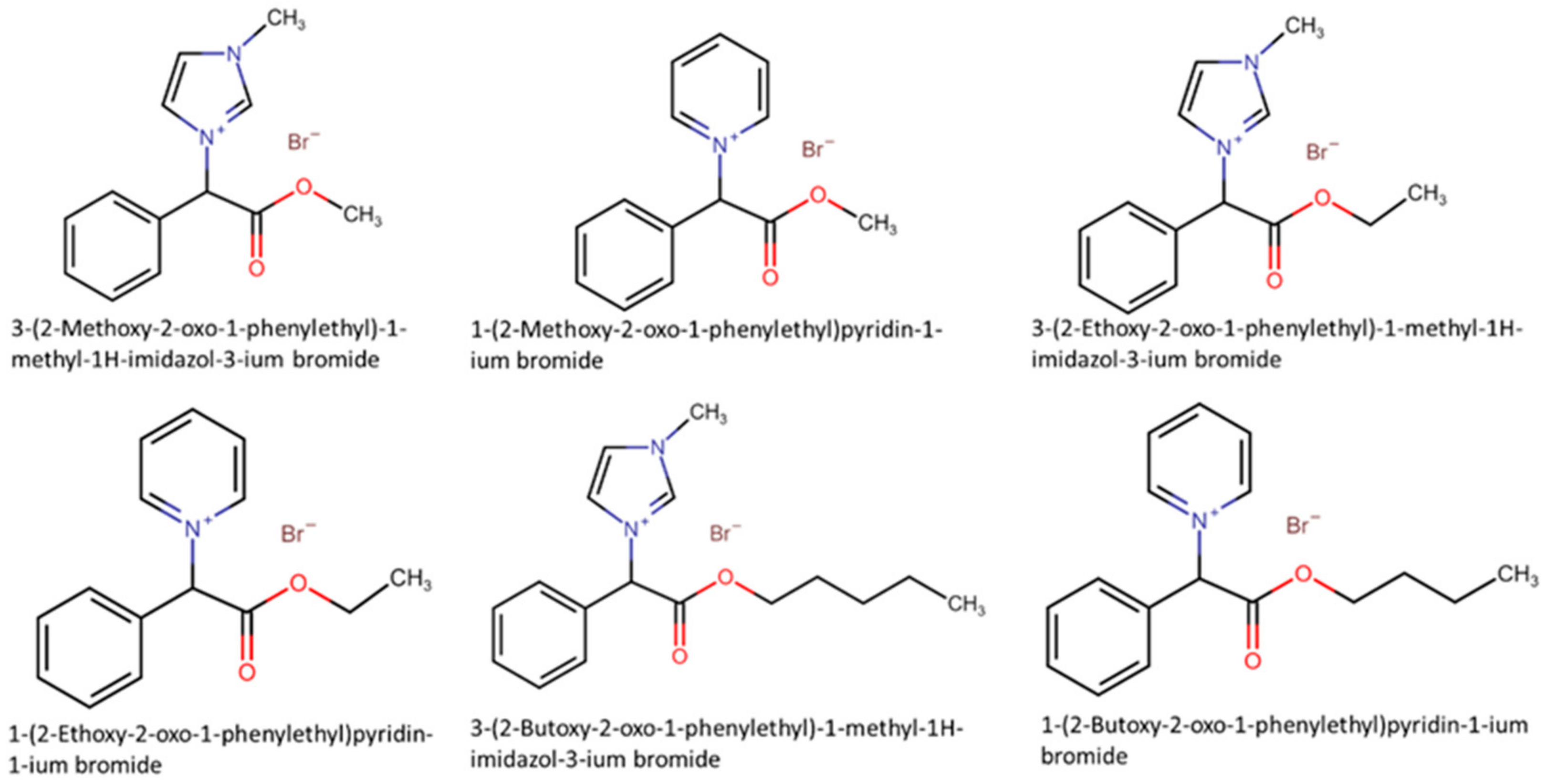
- Kusumahastuti, D.K.A.; Sihtmäe, M.; Kapitanov, I.V.; Karpichev, Y.; Gathergood, N.; Kahru, A. Toxicity Profiling of 24 L-Phenylalanine Derived Ionic Liquids Based on Pyridinium, Imidazolium and Cholinium Cations and Varying Alkyl Chains Using Rapid Screening Vibrio Fischeri Bioassay. Ecotoxicol. Environ. Saf. 2019, 172, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Fernández, F.J.; Bayo, J.; Pérez de los Ríos, A.; Vicente, M.A.; Bernal, F.J.; Quesada-Medina, J. Discovering Less Toxic Ionic Liquids by Using the Microtox® Toxicity Test. Ecotoxicol. Environ. Saf. 2015, 116, 29–33. [Google Scholar] [CrossRef]
- Ghanem, O.B.; Papaiconomou, N.; Abdul Mutalib, M.I.; Viboud, S.; El-Harbawi, M.; Uemura, Y.; Gonfa, G.; Azmi Bustam, M.; Lévêque, J.-M. Thermophysical Properties and Acute Toxicity towards Green Algae and Vibrio Fischeri of Amino Acid-Based Ionic Liquids. J. Mol. Liq. 2015, 212, 352–359. [Google Scholar] [CrossRef]
- Sintra, T.E.; Nasirpour, M.; Siopa, F.; Rosatella, A.A.; Gonçalves, F.; Coutinho, J.A.P.; Afonso, C.A.M.; Ventura, S.P.M. Ecotoxicological Evaluation of Magnetic Ionic Liquids. Ecotoxicol. Environ. Saf. 2017, 143, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Das, R.N.; Sintra, T.E.; Coutinho, J.A.P.; Ventura, S.P.M.; Roy, K.; Popelier, P.L.A. Development of Predictive QSAR Models for Vibrio Fischeri Toxicity of Ionic Liquids and Their True External and Experimental Validation Tests. Toxicol. Res. 2016, 5, 1388–1399. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Shang, Q.; Xia, S.; Wang, Q.; Ma, P. Topological Study on the Toxicity of Ionic Liquids on Vibrio Fischeri by the Quantitative Structure–Activity Relationship Method. J. Hazard. Mater. 2015, 286, 410–415. [Google Scholar] [CrossRef]
- Wang, C.; Wei, Z.; Wang, L.; Sun, P.; Wang, Z. Assessment of Bromide-Based Ionic Liquid Toxicity toward Aquatic Organisms and QSAR Analysis. Ecotoxicol. Environ. Saf. 2015, 115, 112–118. [Google Scholar] [CrossRef]
- Mester, P.; Wagner, M.; Rossmanith, P. Antimicrobial Effects of Short Chained Imidazolium-Based Ionic Liquids—Influence of Anion Chaotropicity. Ecotoxicol. Environ. Saf. 2015, 111, 96–101. [Google Scholar] [CrossRef]
- Mester, P.; Gundolf, T.; Kalb, R.; Wagner, M.; Rossmanith, P. Molecular Mechanisms Mediating Tolerance to Ionic Liquids in Listeria Monocytogenes. Sep. Purif. Technol. 2015, 155, 32–37. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, S.; Dai, Y.; Lu, X.; Lei, Q.; Fang, W. Antimicrobial Activity and Cytotoxicity of Piperazinium- and Guanidinium-Based Ionic Liquids. J. Hazard. Mater. 2016, 307, 73–81. [Google Scholar] [CrossRef]
- Borkowski, A.; Ławniczak, Ł.; Cłapa, T.; Narożna, D.; Selwet, M.; Pęziak, D.; Markiewicz, B.; Chrzanowski, Ł. Different Antibacterial Activity of Novel Theophylline-Based Ionic Liquids—Growth Kinetic and Cytotoxicity Studies. Ecotoxicol. Environ. Saf. 2016, 130, 54–64. [Google Scholar] [CrossRef]
- Borkowski, A.; Kowalczyk, P.; Czerwonka, G.; Cieśla, J.; Cłapa, T.; Misiewicz, A.; Szala, M.; Drabik, M. Interaction of Quaternary Ammonium Ionic Liquids with Bacterial Membranes—Studies with Escherichia Coli R1–R4-Type Lipopolysaccharides. J. Mol. Liq. 2017, 246, 282–289. [Google Scholar] [CrossRef]
- Kowalczyk, P.; Borkowski, A.; Czerwonka, G.; Cłapa, T.; Cieśla, J.; Misiewicz, A.; Borowiec, M.; Szala, M. The Microbial Toxicity of Quaternary Ammonium Ionic Liquids Is Dependent on the Type of Lipopolysaccharide. J. Mol. Liq. 2018, 266, 540–547. [Google Scholar] [CrossRef]
- Borkowski, A.; Gutowski, Ł.; Syczewski, M.; Cłapa, T.; Czerwonka, G. Adaptation of Bacteria Escherichia Coli in Presence of Quaternary Ammonium Ionic Liquids. Ecotoxicol. Environ. Saf. 2018, 164, 370–378. [Google Scholar] [CrossRef]
- Nancharaiah, Y.V.; Francis, A.J. Hormetic Effect of Ionic Liquid 1-Ethyl-3-Methylimidazolium Acetate on Bacteria. Chemosphere 2015, 128, 178–183. [Google Scholar] [CrossRef]
- Foulet, A.; Ghanem, O.B.; El-Harbawi, M.; Lévêque, J.-M.; Mutalib, M.I.A.; Yin, C.-Y. Understanding the Physical Properties, Toxicities and Anti-Microbial Activities of Choline-Amino Acid-Based Salts: Low-Toxic Variants of Ionic Liquids. J. Mol. Liq. 2016, 221, 133–138. [Google Scholar] [CrossRef]
- Shah, M.U.H.; Sivapragasam, M.; Moniruzzaman, M.; Talukder, M.M.R.; Yusup, S.B.; Goto, M. Aggregation Behavior and Antimicrobial Activity of a Micellar System of Binary Ionic Liquids. J. Mol. Liq. 2018, 266, 568–576. [Google Scholar] [CrossRef]
- Sivapragasam, M.; Jaganathan, J.R.; Levêque, J.-M.; Moniruzzaman, M.; Abdul Mutalib, M.I. Microbial Biocompatibility of Phosphonium- and Ammonium-Based Ionic Liquids. J. Mol. Liq. 2019, 273, 107–115. [Google Scholar] [CrossRef]
- Weyhing-Zerrer, N.; Gundolf, T.; Kalb, R.; Oßmer, R.; Rossmanith, P.; Mester, P. Predictability of Ionic Liquid Toxicity from a SAR Study on Different Systematic Levels of Pathogenic Bacteria. Ecotoxicol. Environ. Saf. 2017, 139, 394–403. [Google Scholar] [CrossRef]
- Weyhing-Zerrer, N.; Kalb, R.; Oßmer, R.; Rossmanith, P.; Mester, P. Evidence of a Reverse Side-Chain Effect of Tris(Pentafluoroethyl)Trifluorophosphate [FAP]-Based Ionic Liquids against Pathogenic Bacteria. Ecotoxicol. Environ. Saf. 2018, 148, 467–472. [Google Scholar] [CrossRef]
- Bhattacharya, G.; Giri, R.P.; Dubey, A.; Mitra, S.; Priyadarshini, R.; Gupta, A.; Mukhopadhyay, M.K.; Ghosh, S.K. Structural Changes in Cellular Membranes Induced by Ionic Liquids_ From Model to Bacterial Membranes. Chem. Phys. Lipids 2018, 215, 1–10. [Google Scholar] [CrossRef]
- Clarizia, G.; Bernardo, P.; Carroccio, S.C.; Ussia, M.; Restuccia, C.; Parafati, L.; Calarco, A.; Zampino, D. Heterogenized Imidazolium-Based Ionic Liquids in Pebax®Rnew. Thermal, Gas Transport and Antimicrobial Properties. Polymers 2020, 12, 1419. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, O.B.; Mutalib, M.I.A.; El-Harbawi, M.; Gonfa, G.; Kait, C.F.; Alitheen, N.B.M.; Leveque, J.-M. Effect of Imidazolium-Based Ionic Liquids on Bacterial Growth Inhibition Investigated via Experimental and QSAR Modelling Studies. J. Hazard. Mater. 2015, 297, 198–206. [Google Scholar] [CrossRef]
- Ghanem, O.B.; Shah, S.N.; Lévêque, J.-M.; Mutalib, M.I.A.; El-Harbawi, M.; Khan, A.S.; Alnarabiji, M.S.; Al-Absi, H.R.H.; Ullah, Z. Study of the Antimicrobial Activity of Cyclic Cation-Based Ionic Liquids via Experimental and Group Contribution QSAR Model. Chemosphere 2018, 195, 21–28. [Google Scholar] [CrossRef]
- Hossain, M.I.; El-Harbawi, M.; Alitheen, N.B.M.; Noaman, Y.A.; Lévêque, J.-M.; Yin, C.-Y. Synthesis and Anti-Microbial Potencies of 1-(2-Hydroxyethyl)-3-Alkylimidazolium Chloride Ionic Liquids: Microbial Viabilities at Different Ionic Liquids Concentrations. Ecotoxicol. Environ. Saf. 2013, 87, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Abramenko, N.; Kustov, L.; Metelytsia, L.; Kovalishyn, V.; Tetko, I.; Peijnenburg, W. A Review of Recent Advances towards the Development of QSAR Models for Toxicity Assessment of Ionic Liquids. J. Hazard. Mater. 2020, 384, 121429. [Google Scholar] [CrossRef]
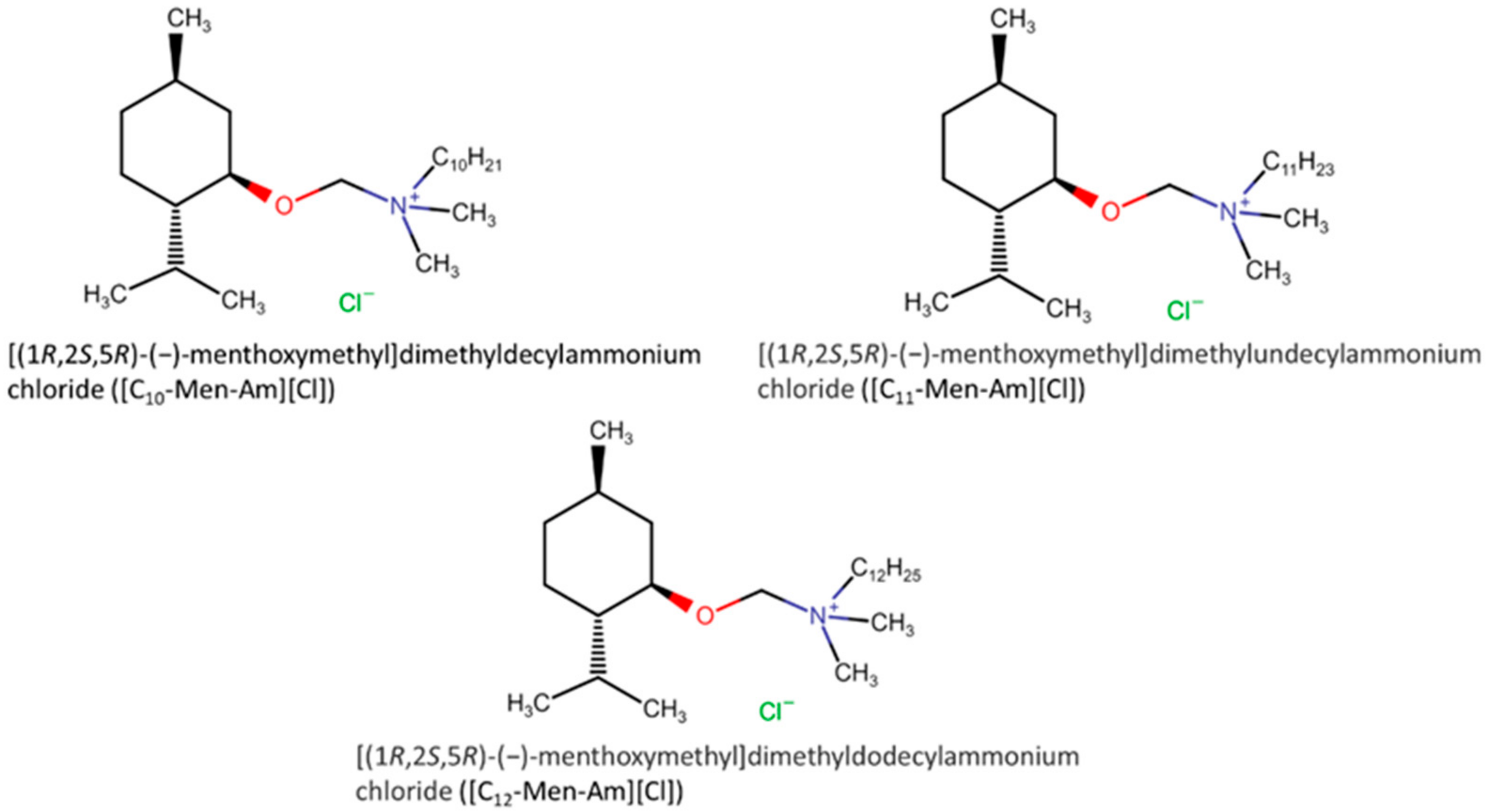
- Suchodolski, J.; Feder-Kubis, J.; Krasowska, A. Antifungal Activity of Ionic Liquids Based on (−)-Menthol: A Mechanism Study. Microbiol. Res. 2017, 197, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Bromberger, B.; Sommer, J.; Robben, C.; Trautner, C.; Kalb, R.; Rossmanith, P.; Mester, P.-J. Evaluation of the Antimicrobial Activity of Pyrithione-Based Ionic Liquids. Sep. Purif. Technol. 2020, 251, 117309. [Google Scholar] [CrossRef]
- Kapitanov, I.V.; Jordan, A.; Karpichev, Y.; Spulak, M.; Perez, L.; Kellett, A.; Kümmerer, K.; Gathergood, N. Synthesis, Self-Assembly, Bacterial and Fungal Toxicity, and Preliminary Biodegradation Studies of a Series of L-Phenylalanine-Derived Surface-Active Ionic Liquids. Green Chem. 2019, 21, 1777–1794. [Google Scholar] [CrossRef]
- Jordan, A.; Haiß, A.; Spulak, M.; Karpichev, Y.; Kümmerer, K.; Gathergood, N. Synthesis of a Series of Amino Acid Derived Ionic Liquids and Tertiary Amines: Green Chemistry Metrics Including Microbial Toxicity and Preliminary Biodegradation Data Analysis. Green Chem. 2016, 18, 4374–4392. [Google Scholar] [CrossRef]
- OECD. Test No. 301: Ready Biodegradability; OECD Guidelines for the Testing of Chemicals, Section 3; OECD: Paris, France, 1992; ISBN 978-92-64-07034-9. [Google Scholar]
- Prydderch, H.; Haiβ, A.; Spulak, M.; Quilty, B.; Kümmerer, K.; Heise, A.; Gathergood, N. Mandelic Acid Derived Ionic Liquids: Synthesis, Toxicity and Biodegradability. RSC Adv. 2017, 7, 2115–2126. [Google Scholar] [CrossRef]
- Thamke, V.R.; Tapase, S.R.; Kodam, K.M. Evaluation of Risk Assessment of New Industrial Pollutant, Ionic Liquids on Environmental Living Systems. Water Res. 2017, 125, 237–248. [Google Scholar] [CrossRef]
- Greaves, T.L.; Weerawardena, A.; Fong, C.; Krodkiewska, I.; Drummond, C.J. Protic Ionic Liquids: Solvents with Tunable Phase Behavior and Physicochemical Properties. J. Phys. Chem. B 2006, 110, 22479–22487. [Google Scholar] [CrossRef] [PubMed]
- Peric, B.; Sierra, J.; Martí, E.; Cruañas, R.; Garau, M.A.; Arning, J.; Bottin-Weber, U.; Stolte, S. (Eco)Toxicity and Biodegradability of Selected Protic and Aprotic Ionic Liquids. J. Hazard. Mater. 2013, 261, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Peric, B.; Sierra, J.; Martí, E.; Cruañas, R.; Garau, M.A. A Comparative Study of the Terrestrial Ecotoxicity of Selected Protic and Aprotic Ionic Liquids. Chemosphere 2014, 108, 418–425. [Google Scholar] [CrossRef]
- Oliveira, M.V.S.; Vidal, B.T.; Melo, C.M.; de Miranda, R.d.C.M.; Soares, C.M.F.; Coutinho, J.A.P.; Ventura, S.P.M.; Mattedi, S.; Lima, Á.S. (Eco)Toxicity and Biodegradability of Protic Ionic Liquids. Chemosphere 2016, 147, 460–466. [Google Scholar] [CrossRef]
- Reid, J.E.S.J.; Prydderch, H.; Spulak, M.; Shimizu, S.; Walker, A.J.; Gathergood, N. Green Profiling of Aprotic versus Protic Ionic Liquids: Synthesis and Microbial Toxicity of Analogous Structures. Sustain. Chem. Pharm. 2018, 7, 17–26. [Google Scholar] [CrossRef]
- Sydow, M.; Owsianiak, M.; Framski, G.; Woźniak-Karczewska, M.; Piotrowska-Cyplik, A.; Ławniczak, Ł.; Szulc, A.; Zgoła-Grześkowiak, A.; Heipieper, H.J.; Chrzanowski, Ł. Biodiversity of Soil Bacteria Exposed to Sub-Lethal Concentrations of Phosphonium-Based Ionic Liquids: Effects of Toxicity and Biodegradation. Ecotoxicol. Environ. Saf. 2018, 147, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Sydow, M.; Szczepaniak, Z.; Framski, G.; Staninska, J.; Owsianiak, M.; Szulc, A.; Piotrowska-Cyplik, A.; Zgoła-Grześkowiak, A.; Wyrwas, B.; Chrzanowski, Ł. Persistence of Selected Ammonium- and Phosphonium-Based Ionic Liquids in Urban Park Soil Microcosms. Int. Biodeterior. Biodegrad. 2015, 103, 91–96. [Google Scholar] [CrossRef]
- Kaczmarek, D.K.; Rzemieniecki, T.; Gwiazdowska, D.; Kleiber, T.; Praczyk, T.; Pernak, J. Choline-Based Ionic Liquids as Adjuvants in Pesticide Formulation. J. Mol. Liq. 2020, 114792. [Google Scholar] [CrossRef]
- Zabielska-Matejuk, J.; Pernak, J.; Fojutowski, A.; Kot, M.; Stangierska, A.; Przybylska, W. Preservatives for Protection against Fungi and Algae and Methods of Wood Protection against Fungi and Algae Using Those Preservatives. 2014. Available online: https://worldwide.espacenet.com/patent/search/family/049117799/publication/EP2700310A1?q=13460046.9 (accessed on 12 February 2021).
- Dimitrić, N.; Spremo, N.; Vraneš, M.; Belić, S.; Karaman, M.; Kovačević, S.; Karadžić, M.; Podunavac-Kuzmanović, S.; Korolija-Crkvenjakov, D.; Gadžurić, S. New Protic Ionic Liquids for Fungi and Bacteria Removal from Paper Heritage Artefacts. RSC Adv. 2019, 9, 17905–17912. [Google Scholar] [CrossRef]
- Zabielska-Matejuk, J.; Stangierska, A.; Kot, M. New Ammonium- and 1,2,4-Triazolium-Based Ionic Liquids for Wood Preservation. J. Wood Chem. Technol. 2015, 35, 178–192. [Google Scholar] [CrossRef]
- Schmitz, K.; Wagner, S.; Reppke, M.; Maier, C.L.; Windeisen-Holzhauser, E.; Benz, J.P. Preserving Cultural Heritage: Analyzing the Antifungal Potential of Ionic Liquids Tested in Paper Restoration. PLoS ONE 2019, 14, e0219650. [Google Scholar] [CrossRef]
- Croitoru, C.; Roata, I.C. Ionic Liquids as Antifungal Agents for Wood Preservation. Molecules 2020, 25, 4289. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-W.; Park, J.-S.; Stolte, S.; Yun, Y.-S. Modelling for Antimicrobial Activities of Ionic Liquids towards Escherichia Coli, Staphylococcus Aureus and Candida Albicans Using Linear Free Energy Relationship Descriptors. J. Hazard. Mater. 2016, 311, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-W.; Stolte, S.; Yun, Y.-S. Comprehensive Approach for Predicting Toxicological Effects of Ionic Liquids on Several Biological Systems Using Unified Descriptors. Sci. Rep. 2016, 6, 33403. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; He, W.; Jia, Q.; Xia, S.; Wang, Q. QSAR Models for Describing the Toxicological Effects of ILs against Candida Albicans Based on Norm Indexes. Chemosphere 2018, 201, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, S.; Zhang, X.; Chen, C. Growth Inhibition and Effect on Photosystem by Three Imidazolium Chloride Ionic Liquids in Rice Seedlings. J. Hazard. Mater. 2015, 286, 440–448. [Google Scholar] [CrossRef]
- Liu, T.; Zhu, L.; Wang, J.; Wang, J.; Xie, H. The Genotoxic and Cytotoxic Effects of 1-Butyl-3-Methylimidazolium Chloride in Soil on Vicia Faba Seedlings. J. Hazard. Mater. 2015, 285, 27–36. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, X.; Chen, C.; Du, S.; Dong, Y. Effects of Imidazolium Chloride Ionic Liquids and Their Toxicity to Scenedesmus Obliquus. Ecotoxicol. Environ. Saf. 2015, 122, 83–90. [Google Scholar] [CrossRef]
- Liu, D.; Liu, H.; Wang, S.; Chen, J.; Xia, Y. The Toxicity of Ionic Liquid 1-Decylpyridinium Bromide to the Algae Scenedesmus Obliquus: Growth Inhibition, Phototoxicity, and Oxidative Stress. Sci. Total Environ. 2018, 622–623, 1572–1580. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, X.; Dong, Y.; Chen, C.; Zhu, S.; Ma, X. Enantioselective Toxicities of Chiral Ionic Liquids 1-Alkyl-3-Methyl Imidazolium Tartrate on Scenedesmus Obliquus. Aquat. Toxicol. 2015, 169, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Zhou, Y.; Cao, K.; Gao, W.; Gao, B.; Sun, L.; Liu, S.; Wang, L.; Ding, Y. Phytotoxicity of Ionic Liquids with Different Structures on Wheat Seedlings and Evaluation of Their Toxicity Attenuation at the Presence of Modified Biochar by Adsorption Effect. Chemosphere 2018, 196, 331–338. [Google Scholar] [CrossRef]
- Xia, Y.; Liu, D.; Dong, Y.; Chen, J.; Liu, H. Effect of Ionic Liquids with Different Cations and Anions on Photosystem and Cell Structure of Scenedesmus Obliquus. Chemosphere 2018, 195, 437–447. [Google Scholar] [CrossRef]
- Li, Y.; Yang, M.; Liu, L.; Zhang, R.; Cui, Y.; Dang, P.; Ge, X.; Chen, X. Effects of 1-Butyl-3-Methylimidazolium Chloride on the Photosynthetic System and Metabolism of Maize (Zea Mays L.) Seedlings. Ecotoxicol. Environ. Saf. 2018, 161, 648–654. [Google Scholar] [CrossRef]
- Jin, M.; Wang, H.; Li, Z.; Fu, L.; Chu, L.; Wu, J.; Du, S.; Liu, H. Physiological Responses of Chlorella Pyrenoidosa to 1-Hexyl-3-Methyl Chloride Ionic Liquids with Different Cations. Sci. Total Environ. 2019, 685, 315–323. [Google Scholar] [CrossRef]
- Liu, T.; Zhu, L.-S.; Wang, J.; Wang, J.; Tan, M. Phytotoxicity of Imidazolium-Based ILs with Different Anions in Soil on Vicia Faba Seedlings and the Influence of Anions on Toxicity. Chemosphere 2016, 145, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, J.; Zhu, L.; Du, Z.; Wang, J.; Wei, K. Physiological and Biochemical Responses of Wheat (Triticum Aestivum L.) Seedlings to Three Imidazolium-Based Ionic Liquids in Soil. Chemosphere 2018, 191, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Wang, H.; Liu, H.; Xia, Y.; Ruan, S.; Huang, Y.; Qiu, J.; Du, S.; Xu, L. Oxidative Stress Response and Proteomic Analysis Reveal the Mechanisms of Toxicity of Imidazolium-Based Ionic Liquids against Arabidopsis Thaliana. Environ. Pollut. 2020, 260, 114013. [Google Scholar] [CrossRef]
- Habibul, N.; Chen, J.-J.; Hu, Y.-Y.; Hu, Y.; Yin, H.; Sheng, G.-P.; Yu, H.-Q. Uptake, Accumulation and Metabolization of 1-Butyl-3-Methylimidazolium Bromide by Ryegrass from Water: Prospects for Phytoremediation. Water Res. 2019, 156, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Habibul, N.; Hu, Y.-Y.; Hu, Y.; Sheng, G.-P. Alkyl Chain Length Affecting Uptake of Imidazolium Based Ionic Liquids by Ryegrass (Lolium Perenne L.). J. Hazard. Mater. 2021, 401, 123376. [Google Scholar] [CrossRef]
- Habibul, N.; Ilmurat, M.; Habibul, Z.; Hu, Y.; Ma, X. Uptake and Accumulation of Imidazolium Ionic Liquids in Rice Seedlings: Impacts of Alkyl Chain Length. Chemosphere 2020, 242, 125228. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, J.; Du, Z.; Li, B.; Juhasz, A.; Tan, M.; Zhu, L.; Wang, J. Toxicity Evaluation of Three Imidazolium-Based Ionic Liquids ([C6mim]R) on Vicia Faba Seedlings Using an Integrated Biomarker Response (IBR) Index. Chemosphere 2020, 240, 124919. [Google Scholar] [CrossRef]
- Peric, B.; Sierra, J.; Martí, E.; Cruañas, R.; Garau, M.A. Quantitative Structure–Activity Relationship (QSAR) Prediction of (Eco)Toxicity of Short Aliphatic Protic Ionic Liquids. Ecotoxicol. Environ. Saf. 2015, 115, 257–262. [Google Scholar] [CrossRef]
- Couling, D.J.; Bernot, R.J.; Docherty, K.M.; Dixon, J.K.; Maginn, E.J. Assessing the Factors Responsible for Ionic Liquid Toxicity to Aquatic Organisms via Quantitative Structure–Property Relationship Modeling. Green Chem. 2006, 8, 82–90. [Google Scholar] [CrossRef]
- Zhang, B.; Li, X.; Chen, D.; Wang, J. Effects of 1-Octyl-3-Methylimidazolium Bromide on the Antioxidant System of Lemna Minor. Protoplasma 2013, 250, 103–110. [Google Scholar] [CrossRef]
- Du, Z.; Zhu, L.; Dong, M.; Wang, J.; Wang, J.; Xie, H.; Zhu, S. Effects of the Ionic Liquid [Omim]PF6 on Antioxidant Enzyme Systems, ROS and DNA Damage in Zebrafish (Danio Rerio). Aquat. Toxicol. 2012, 124–125, 91–93. [Google Scholar] [CrossRef]
- Dong, M.; Zhu, L.; Zhu, S.; Wang, J.; Wang, J.; Xie, H.; Du, Z. Toxic Effects of 1-Decyl-3-Methylimidazolium Bromide Ionic Liquid on the Antioxidant Enzyme System and DNA in Zebrafish (Danio Rerio) Livers. Chemosphere 2013, 91, 1107–1112. [Google Scholar] [CrossRef]
- Kumar, M.; Trivedi, N.; Reddy, C.R.K.; Jha, B. Toxic Effects of Imidazolium Ionic Liquids on the Green Seaweed Ulva Lactuca: Oxidative Stress and DNA Damage. Chem. Res. Toxicol. 2011, 24, 1882–1890. [Google Scholar] [CrossRef]
- Wu, X.; Tong, Z.-H.; Li, L.-L.; Yu, H.-Q. Toxic Effects of Imidazolium-Based Ionic Liquids on Caenorhabditis Elegans: The Role of Reactive Oxygen Species. Chemosphere 2013, 93, 2399–2404. [Google Scholar] [CrossRef]
- Luo, Y.-R.; Wang, S.-H.; Yun, M.-X.; Li, X.-Y.; Wang, J.-J.; Sun, Z.-J. The Toxic Effects of Ionic Liquids on the Activities of Acetylcholinesterase and Cellulase in Earthworms. Chemosphere 2009, 77, 313–318. [Google Scholar] [CrossRef]
- Passino, D.R.M.; Smith, S.B. Acute Bioassays and Hazard Evaluation of Representative Contaminants Detected in Great Lakes Fish. Environ. Toxicol. Chem. 1987, 6, 901–907. [Google Scholar] [CrossRef]
- Liu, T.; Guo, Y.; Wang, J.; Wang, J.; Zhu, L.; Zhang, J.; Zhang, C. Assessing Toxic Effects of [Omim]Cl and [Omim]BF4 in Zebrafish Adults Using a Biomarker Approach. Environ. Sci. Pollut. Res. 2016, 23, 7360–7368. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhu, L.; Wang, J.; Wang, J.; Zhou, T.; Xu, Y.; Cheng, C. The Acute Toxic Effects of Imidazolium-Based Ionic Liquids with Different Alkyl-Chain Lengths and Anions on Zebrafish (Danio Rerio). Ecotoxicol. Environ. Saf. 2017, 140, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, J.; Dong, M.; Wang, J.; Du, Z.; Li, B.; Zhu, L. Effect of 1-Methyl-3-Hexylimidazolium Bromide on Zebrafish (Danio Rerio). Chemosphere 2018, 192, 348–353. [Google Scholar] [CrossRef]
- Zhang, C.; Shao, Y.; Zhu, L.; Wang, J.; Wang, J.; Guo, Y. Acute Toxicity, Biochemical Toxicity and Genotoxicity Caused by 1-Butyl-3-Methylimidazolium Chloride and 1-Butyl-3-Methylimidazolium Tetrafluoroborate in Zebrafish (Danio Rerio) Livers. Environ. Toxicol. Pharmacol. 2017, 51, 131–137. [Google Scholar] [CrossRef]
- Zhang, C.; Du, Z.; Wang, J.; Wang, J.; Zhou, T.; Li, B.; Zhu, L.; Li, W.; Hou, K. Exposed Zebrafish (Danio Rerio) to Imidazolium-Based Ionic Liquids with Different Anions and Alkyl-Chain Lengths. Chemosphere 2018, 203, 381–386. [Google Scholar] [CrossRef]
- Liu, T.; Zhu, L.; Wang, J.; Wang, J.; Zhang, J.; Sun, X.; Zhang, C. Biochemical Toxicity and DNA Damage of Imidazolium-Based Ionic Liquid with Different Anions in Soil on Vicia Faba Seedlings. Sci. Rep. 2015, 5, 18444. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Zhu, L.; Du, Z.; Li, B.; Wang, J.; Wang, J.; Zhang, C.; Zhu, L. Acute Toxicity, Oxidative Stress and DNA Damage of Three Task-Specific Ionic Liquids ([C2NH2MIm]BF4, [MOEMIm]BF4, and [HOEMIm]BF4) to Zebrafish (Danio Rerio). Chemosphere 2020, 249, 126119. [Google Scholar] [CrossRef] [PubMed]
- Perales, E.; García, C.B.; Lomba, L.; Aldea, L.; García, J.I.; Giner, B. Comparative Ecotoxicology Study of Two Neoteric Solvents: Imidazolium Ionic Liquid vs. Glycerol Derivative. Ecotoxicol. Environ. Saf. 2016, 132, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Ruokonen, S.-K.; Sanwald, C.; Sundvik, M.; Polnick, S.; Vyavaharkar, K.; Duša, F.; Holding, A.J.; King, A.W.T.; Kilpeläinen, I.; Lämmerhofer, M.; et al. Effect of Ionic Liquids on Zebrafish (Danio Rerio) Viability, Behavior, and Histology; Correlation between Toxicity and Ionic Liquid Aggregation. Environ. Sci. Technol. 2016, 50, 7116–7125. [Google Scholar] [CrossRef]
- Baharuddin, S.H.; Mustahil, N.A.; Abdullah, A.A.; Sivapragasam, M.; Moniruzzaman, M. Ecotoxicity Study of Amino Acid Ionic Liquids Towards Danio Rerio Fish: Effect of Cations. Procedia Eng. 2016, 148, 401–408. [Google Scholar] [CrossRef]
- Tang, G.; Zhang, W.; Tang, J.; Niu, J.; Yang, J.; Tang, R.; Dong, H.; Liang, Y.; Luo, L.; Cao, Y. Development of Triflumizole Ionic Liquids Containing Anions of Natural Origin for Improving the Utilization and Minimizing the Adverse Impacts on Aquatic Ecosystems. Sci. Total Environ. 2019, 670, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Yoganantharajah, P.; Eyckens, D.J.; Pedrina, J.L.; Henderson, L.C.; Gibert, Y. A Study on Acute Toxicity and Solvent Capacity of Solvate Ionic Liquids in Vivo Using a Zebrafish Model (Danio Rerio). New J. Chem. 2016, 40, 6599–6603. [Google Scholar] [CrossRef]
- Younes, N.; Salem, R.; Al-Asmakh, M.; Altamash, T.; Pintus, G.; Khraisheh, M.; Nasrallah, G.K. Toxicity Evaluation of Selected Ionic Liquid Compounds on Embryonic Development of Zebrafish. Ecotoxicol. Environ. Saf. 2018, 161, 17–24. [Google Scholar] [CrossRef]
- Perez, C.J.; Tata, A.; de Campos, M.L.; Peng, C.; Ifa, D.R. Monitoring Toxic Ionic Liquids in Zebrafish (Danio Rerio) with Desorption Electrospray Ionization Mass Spectrometry Imaging (DESI-MSI). J. Am. Soc. Mass Spectrom. 2017, 28, 1136–1148. [Google Scholar] [CrossRef]
- Thamke, V.R.; Kodam, K.M. Toxicity Study of Ionic Liquid, 1-Butyl-3-Methylimidazolium Bromide on Guppy Fish, Poecilia Reticulata and Its Biodegradation by Soil Bacterium Rhodococcus Hoagii VRT1. J. Hazard. Mater. 2016, 320, 408–416. [Google Scholar] [CrossRef]
- Ma, J.; Li, X.; Cui, M.; Li, W.; Li, X. Negative Impact of the Imidazolium-Based Ionic Liquid [C8mim]Br on Silver Carp (Hypophthalmichthys Molitrix): Long-Term and Low-Level Exposure. Chemosphere 2018, 213, 358–367. [Google Scholar] [CrossRef]
- Tsarpali, V.; Belavgeni, A.; Dailianis, S. Investigation of Toxic Effects of Imidazolium Ionic Liquids, [Bmim][BF4] and [Omim][BF4], on Marine Mussel Mytilus Galloprovincialis with or without the Presence of Conventional Solvents, Such as Acetone. Aquat. Toxicol. 2015, 164, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Tsarpali, V.; Dailianis, S. [Omim][BF4]-Mediated Toxicity in Mussel Hemocytes Includes Its Interaction with Cellular Membrane Proteins. Aquat. Toxicol. 2018, 203, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A.; Russo, D.; Spasiano, D.; Marotta, R.; Race, M.; Fabbricino, M.; Galdiero, E.; Guida, M. Chronic Toxicity of Treated and Untreated Aqueous Solutions Containing Imidazole-Based Ionic Liquids and Their Oxydized by-Products. Ecotoxicol. Environ. Saf. 2019, 180, 466–472. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, S.; Zhu, L.; Wang, J.; Wang, J.; Zhou, T. The Acute Toxic Effects of 1-Alkyl-3-Methylimidazolium Nitrate Ionic Liquids on Chlorella Vulgaris and Daphnia Magna. Environ. Pollut. 2017, 229, 887–895. [Google Scholar] [CrossRef]
- Bado-Nilles, A.; Diallo, A.-O.; Marlair, G.; Pandard, P.; Chabot, L.; Geffard, A.; Len, C.; Porcher, J.-M.; Sanchez, W. Coupling of OECD Standardized Test and Immunomarkers to Select the Most Environmentally Benign Ionic Liquids Option--towards an Innovative “Safety by Design” Approach. J. Hazard. Mater. 2015, 283, 202–210. [Google Scholar] [CrossRef]
- Bioucas, F.; Vieira, S.; Lourenco, M.; Santos, F.; Nieto de Castro, C.; Massonne, K. [C2mim][CH3SO3]—A Suitable New Heat Transfer Fluid? Part 1. Thermophysical and Toxicological Properties. Ind. Eng. Chem. Res. 2018, 57. [Google Scholar] [CrossRef]
- Sakamoto, M.; Ohama, Y.; Aoki, S.; Fukushi, K.; Mori, T.; Yoshimura, Y.; Shimizu, A. Effect of Ionic Liquids on the Hatching of Artemia Salina Cysts. Aust. J. Chem. 2018, 71. [Google Scholar] [CrossRef]
- Tzani, A.; Elmaloglou, M.; Kyriazis, C.; Aravopoulou, D.; Kleidas, I.; Papadopoulos, A.; Ioannou, E.; Kyritsis, A.; Voutsas, E.; Detsi, A. Synthesis and Structure-Properties Relationship Studies of Biodegradable Hydroxylammonium-Based Protic Ionic Liquids. J. Mol. Liq. 2016, 224, 366–376. [Google Scholar] [CrossRef]
- Shi, C.-Y.; Zhang, H.-C.; Chen, G.-W.; Sun, L.-Q.; Liu, D.-Z. Toxicity of Ionic Liquid 1-Octyl-3-Methylimidazolium Bromide on Freshwater Planarian, Dugesia Japonica. Fresenius Environ. Bull. 2013, 22, 1609–1615. [Google Scholar]
- Zhang, H.-C.; Shi, C.-Y.; Yang, H.-H.; Chen, G.-W.; Liu, D.-Z. Genotoxicity Evaluation of Ionic Liquid 1-Octyl-3-Methylimidazolium Bromide in Freshwater Planarian Dugesia Japonica Using RAPD Assay. Ecotoxicol. Environ. Saf. 2016, 134P1, 17–22. [Google Scholar] [CrossRef]
- Zhang, H.-C.; Shi, C.-Y.; Sun, L.-Q.; Wang, F.; Chen, G.-W. Toxic Effects of Ionic Liquid 1-Octyl-3-Methylimidazolium Bromide on the Antioxidant Defense System of Freshwater Planarian, Dugesia Japonica. Toxicol. Ind. Health 2016, 32, 1675–1683. [Google Scholar] [CrossRef]
- Zhang, H.-C.; Shi, C.-Y.; Wang, F.; Chen, G.-W.; Liu, D.-Z. Imidazolium-Based Ionic Liquids: Effects of Different Alkyl-Chain Lengths on Planarian Dugesia Japonica Based on Ingestion, Regeneration and Antioxidant Response. Fresenius Environ. Bull. 2016, 25, 3205–3212. [Google Scholar]
- Tsarpali, V.; Dailianis, S. Toxicity of Two Imidazolium Ionic Liquids, [Bmim][BF4] and [Omim][BF4], to Standard Aquatic Test Organisms: Role of Acetone in the Induced Toxicity. Ecotoxicol. Environ. Saf. 2015, 117, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on Classification, Labelling and Packaging of Substances and Mixtures, Amending and Repealing Directives 67/548/EEC and 1999/45/EC, and Amending Regulation (EC) No 1907/2006 (Text with EEA Relevance); Volume 353. 2008. Available online: http://data.europa.eu/eli/reg/2008/1272/oj (accessed on 11 February 2021).
- Liu, T.; Yan, F.; Jia, Q.; Wang, Q. Norm Index-Based QSAR Models for Acute Toxicity of Organic Compounds toward Zebrafish Embryo. Ecotoxicol. Environ. Saf. 2020, 203, 110946. [Google Scholar] [CrossRef]
- Cho, C.-W.; Yun, Y.-S. Application of General Toxic Effects of Ionic Liquids to Predict Toxicities of Ionic Liquids to Spodoptera Frugiperda 9, Eisenia Fetida, Caenorhabditis Elegans, and Danio Rerio. Environ. Pollut. 2019, 255, 113185. [Google Scholar] [CrossRef]
- Cho, C.-W.; Yun, Y.-S. Correlating Toxicological Effects of Ionic Liquids on Daphnia Magna with in Silico Calculated Linear Free Energy Relationship Descriptors. Chemosphere 2016, 152, 207–213. [Google Scholar] [CrossRef]
- Lu, L.; Zhang, Y.; Chen, J.; Tong, Z. Toxicity of Selected Imidazolium-Based Ionic Liquids on Caenorhabditis Elegans: A Quantitative Structure-Activity Relationship Study. Chin. J. Chem. Phys. 2017, 30, 423–428. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, J.; Du, Z.; Li, B.; Zhu, L.; Wang, J.; Zhang, S. Toxic Effect of [Omim]BF4 and [Omim]Br on Antioxidant Stress and Oxidative Damage in Earthworms (Eisenia Fetida). Environ. Toxicol. Pharmacol. 2018, 60, 37–44. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, T.; Zhang, J.; Wang, J.; Wang, J.; Zhu, L.; Yang, J. Biochemical and Genetic Toxicity of the Ionic Liquid 1-Octyl-3-Methylimidazolium Chloride on Earthworms (Eisenia Fetida). Environ. Toxicol. Chem. 2016, 35, 411–418. [Google Scholar] [CrossRef]
- Shao, Y.; Hou, K.; Du, Z.; Li, B.; Wang, J.; Juhasz, A.; Wang, J.; Zhu, L. Evaluation of the Toxicity of 1-Butyl-3-Methyl Imidazolium Tetrafluoroborate Using Earthworms (Eisenia Fetida) in Two Soils. Sci. Total Environ. 2019, 686, 946–958. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, J.; Du, Z.; Li, B.; Zhu, L.; Wang, J. Toxicity of 1-Alkyl-3-Methyl Imidazolium Nitrate Ionic Liquids to Earthworms: The Effects of Carbon Chains of Different Lengths. Chemosphere 2018, 206, 302–309. [Google Scholar] [CrossRef]
- Shao, Y.; Du, Z.; Zhang, C.; Zhu, L.; Wang, J.; Wang, J. Acute Toxicity of Imidazole Nitrate Ionic Liquids with Varying Chain Lengths to Earthworms (Eisenia Foetida). Bull. Environ. Contam. Toxicol. 2017, 99, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Wang, J.; Wang, J.; Du, Z.; Li, B.; Zhu, L.; Juhasz, A.; Liu, X.; Xu, Y.; Li, W. Oxidative Stress and Genotoxic Effects in Earthworms Induced by Five Imidazolium Bromide Ionic Liquids with Different Alkyl Chains. Chemosphere 2019, 227, 570–579. [Google Scholar] [CrossRef]
- Liu, H.; Mo, L.; Hou, M.; Zhang, J. Life Stage-Dependent Toxicities of 1-Ethyl-3-Methylimidazolium Bromide on Caenorhabditis Elegans. Environ. Pollut. 2020, 265, 114848. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Tong, Z.-H.; Chong, H.-J.; Shao, X.-Y. Toxic Effects of Prolonged Exposure to [C14mim]Br on Caenorhabditis Elegans. Chemosphere 2018, 208, 226–232. [Google Scholar] [CrossRef]
- Megaw, J.; Thompson, T.P.; Lafferty, R.A.; Gilmore, B.F. Galleria Mellonella as a Novel in Vivo Model for Assessment of the Toxicity of 1-Alkyl-3-Methylimidazolium Chloride Ionic Liquids. Chemosphere 2015, 139, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Wan, R.; Wang, P.; Huo, W.; Dong, H.; Du, Q. Toxicity of Imidazoles Ionic Liquid [C16mim]Cl to Hela Cells. Ecotoxicol. Environ. Saf. 2018, 162, 408–414. [Google Scholar] [CrossRef]
- Wan, R.; Xia, X.; Wang, P.; Huo, W.; Dong, H.; Chang, Z. Toxicity of Imidazoles Ionic Liquid [C16mim]Cl to HepG2 Cells. Toxicology Vitro 2018, 52, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Radošević, K.; Železnjak, J.; Cvjetko Bubalo, M.; Radojčić Redovniković, I.; Slivac, I.; Gaurina Srček, V. Comparative in Vitro Study of Cholinium-Based Ionic Liquids and Deep Eutectic Solvents toward Fish Cell Line. Ecotoxicol. Environ. Saf. 2016, 131, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Cvjetko Bubalo, M.; Radošević, K.; Gaurina Srček, V.; Das, R.N.; Popelier, P.; Roy, K. Cytotoxicity towards CCO Cells of Imidazolium Ionic Liquids with Functionalized Side Chains: Preliminary QSTR Modeling Using Regression and Classification Based Approaches. Ecotoxicol. Environ. Saf. 2015, 112, 22–28. [Google Scholar] [CrossRef]
- Li, X.; Ma, J.; Wang, J. Cytotoxicity, Oxidative Stress, and Apoptosis in HepG2 Cells Induced by Ionic Liquid 1-Methyl-3-Octylimidazolium Bromide. Ecotoxicol. Environ. Saf. 2015, 120, 342–348. [Google Scholar] [CrossRef]
- Liu, H.; Dong, Y.; Wu, J.; Chen, C.; Liu, D.; Zhang, Q.; Du, S. Evaluation of Interaction between Imidazolium-Based Chloride Ionic Liquids and Calf Thymus DNA. Sci. Total Environ. 2016, 566–567, 1–7. [Google Scholar] [CrossRef]
- Thamke, V.R.; Chaudhari, A.U.; Tapase, S.R.; Paul, D.; Kodam, K.M. In Vitro Toxicological Evaluation of Ionic Liquids and Development of Effective Bioremediation Process for Their Removal. Environ. Pollut. 2019, 250, 567–577. [Google Scholar] [CrossRef]
- Hwang, J.; Park, H.; Choi, D.W.; Nam, K.T.; Lim, K.-M. Investigation of Dermal Toxicity of Ionic Liquids in Monolayer-Cultured Skin Cells and 3D Reconstructed Human Skin Models. Toxicol. Vitro 2018, 46, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Vraneš, M.; Tot, A.; Papović, S.; Četojević-Simin, D.; Markov, S.; Velićanski, A.; Popsavin, M.; Gadžurić, S. Physicochemical Features and Toxicity of Some Vitamin Based Ionic Liquids. J. Mol. Liq. 2017, 247, 411–424. [Google Scholar] [CrossRef]
- Leitch, A.C.; Abdelghany, T.M.; Probert, P.M.; Dunn, M.P.; Meyer, S.K.; Palmer, J.M.; Cooke, M.P.; Blake, L.I.; Morse, K.; Rosenmai, A.K.; et al. The Toxicity of the Methylimidazolium Ionic Liquids, with a Focus on M8OI and Hepatic Effects. Food Chem. Toxicol. 2020, 136, 111069. [Google Scholar] [CrossRef] [PubMed]
- Leitch, A.C.; Abdelghany, T.M.; Charlton, A.; Grigalyte, J.; Oakley, F.; Borthwick, L.A.; Reed, L.; Knox, A.; Reilly, W.J.; Agius, L.; et al. Renal Injury and Hepatic Effects from the Methylimidazolium Ionic Liquid M8OI in Mouse. Ecotoxicol. Environ. Saf. 2020, 202, 110902. [Google Scholar] [CrossRef] [PubMed]
- Abdelghany, T.M.; Leitch, A.C.; Nevjestić, I.; Ibrahim, I.; Miwa, S.; Wilson, C.; Heutz, S.; Wright, M.C. Emerging Risk from “Environmentally-Friendly” Solvents: Interaction of Methylimidazolium Ionic Liquids with the Mitochondrial Electron Transport Chain Is a Key Initiation Event in Their Mammalian Toxicity. Food Chem. Toxicol. 2020, 111593. [Google Scholar] [CrossRef]
- Ma, J.; Chen, X.; Xin, G.; Li, X. Chronic Exposure to the Ionic Liquid [C8mim]Br Induces Inflammation in Silver Carp Spleen: Involvement of Oxidative Stress-Mediated P38MAPK/NF-ΚB Signalling and MicroRNAs. Fish Shellfish Immunol. 2019, 84, 627–638. [Google Scholar] [CrossRef]
- Zhang, C.; Du, Z.; Li, B.; Sun, X.; Wang, J.; Wang, J.; Zhu, L. Evaluating Toxicity of 1-Octyl-3-Methylimidazolium Hexafluorophosphate to Microorganisms in Soil. Chemosphere 2018, 210, 762–768. [Google Scholar] [CrossRef]
- Fan, Y.; Dong, X.; Yan, L.; Li, D.; Hua, S.; Hu, C.; Pan, C. Evaluation of the Toxicity of Ionic Liquids on Trypsin: A Mechanism Study. Chemosphere 2016, 148, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Fan, Y.; Zhang, H.; Zhong, Y.; Yang, Y.; Miao, J.; Hua, S. Inhibitory Effects of Ionic Liquids on the Lactic Dehydrogenase Activity. Int. J. Biol. Macromol. 2016, 86, 155–161. [Google Scholar] [CrossRef]
- Fan, Y.; Dong, X.; Li, X.; Zhong, Y.; Kong, J.; Hua, S.; Miao, J.; Li, Y. Spectroscopic Studies on the Inhibitory Effects of Ionic Liquids on Lipase Activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2016, 159, 128–133. [Google Scholar] [CrossRef]
- Fan, Y.; Dong, X.; Zhong, Y.; Li, J.; Miao, J.; Hua, S.; Li, Y.; Cheng, B.; Chen, W. Effects of Ionic Liquids on the Hydrolysis of Casein by Lumbrokinase. Biochem. Eng. J. 2016, 109, 35–42. [Google Scholar] [CrossRef]
- Azevedo, A.M.O.; Pereira, S.A.P.; Passos, M.L.C.; Costa, S.P.F.; Pinto, P.C.A.G.; Araujo, A.R.T.S.; Saraiva, M.L.M.F.S. Assessment of Ionic Liquids’ Toxicity through the Inhibition of Acylase I Activity on a Microflow System. Chemosphere 2017, 173, 351–358. [Google Scholar] [CrossRef]
- Costa, S.P.F.; Martins, B.S.F.; Pinto, P.C.A.G.; Saraiva, M.L.M.F.S. Automated Cytochrome c Oxidase Bioassay Developed for Ionic Liquids’ Toxicity Assessment. J. Hazard. Mater. 2016, 309, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Cunha, E.; Pinto, P.C.A.G.; Saraiva, M.L.M.F.S. Evaluation of Ionic Liquids as Alternative Solvents for Aldolase Activity: Use of a New Automated SIA Methodology—ScienceDirect. Talanta 2015, 141, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Das, R.N.; Roy, K.; Popelier, P.L.A. Exploring Simple, Transparent, Interpretable and Predictive QSAR Models for Classification and Quantitative Prediction of Rat Toxicity of Ionic Liquids Using OECD Recommended Guidelines. Chemosphere 2015, 139, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Sosnowska, A.; Grzonkowska, M.; Puzyn, T. Global versus Local QSAR Models for Predicting Ionic Liquids Toxicity against IPC-81 Leukemia Rat Cell Line: The Predictive Ability. J. Mol. Liq. 2017, 231, 333–340. [Google Scholar] [CrossRef]
- Yan, F.; Lan, T.; Yan, X.; Jia, Q.; Wang, Q. Norm Index-Based QSTR Model to Predict the Eco-Toxicity of Ionic Liquids towards Leukemia Rat Cell Line. Chemosphere 2019, 234, 116–122. [Google Scholar] [CrossRef]
- Zhu, P.; Kang, X.; Zhao, Y.; Latif, U.; Zhang, H. Predicting the Toxicity of Ionic Liquids toward Acetylcholinesterase Enzymes Using Novel QSAR Models. Int. J. Mol. Sci. 2019, 20, 2186. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-W.; Yun, Y.-S. Interpretation of Toxicological Activity of Ionic Liquids to Acetylcholinesterase Inhibition via in Silico Modelling. Chemosphere 2016, 159, 178–183. [Google Scholar] [CrossRef]
- Zhu, J.; Kong, Q.; Zheng, S.; Wang, Y.; Jiao, Z.; Nie, Y.; Liu, T.; Song, K. Toxicological Evaluation of Ionic Liquid in a Biological Functional Tissue Construct Model Based on Nano-Hydroxyapatite/Chitosan/Gelatin Hybrid Scaffolds. Int. J. Biol. Macromol. 2020, 158, 800–810. [Google Scholar] [CrossRef]

| Name | Acronym | EC50 (mg L−1) | Classification |
|---|---|---|---|
| (2-Hydroxyethyl) trimethyl ammonium cobalt tetrachloride | [N1,1,1,2(OH)]2[CoCl4] | 8.9 | Toxic |
| (2-Hydroxyethyl) trimethyl ammonium iron tetrachloride | [N1,1,1,2(OH)][FeCl4] | 14.17 | Moderately toxic |
| (2-Hydroxyethyl) trimethyl ammonium manganese tetrachloride | [N1,1,1,2(OH)]2[MnCl4] | 48.18 | Moderately toxic |
| (2-Hydroxyethyl) trimethyl ammonium gadolinium hexachloride | [N1,1,1,2(OH)]3[GdCl6] | 26.12 | Moderately toxic |
| (2-Hydroxyethyl) ethyldimethyl ammonium iron tetrachloride | [N1,1,2,2(OH)][FeCl4] | 15.49 | Moderately toxic |
| Hexyl(2-hydroxyethyl)dimethylammonium iron tetrachloride | [N1,1,6,2(OH)][FeCl4] | 17.19 | Moderately toxic |
| Hexyl (2-hydroxyethyl) dimethylammonium manganese tetrachloride | [N1,1,6,2(OH)]2[MnCl4] | 37.51 | Moderately toxic |
| Octyl (2-hydroxyethyl) dimethylammonium cobalt tetrachloride | [N1,1,8,2(OH)]2[CoCl4] | 10.75 | Moderately toxic |
| Octyl (2-hydroxyethyl) dimethyl ammonium iron tetrachloride | [N1,1,8,2(OH)][FeCl4] | 17.49 | Moderately toxic |
| Octyl (2-hydroxyethyl) dimethylammonium manganese tetrachloride | [N1,1,8,2(OH)]2[MnCl4] | 27.96 | Moderately toxic |
| Dodecyl (2-hydroxyethyl) dimethylammonium cobalt tetrachloride | [N1,1,12,2(OH)]2[CoCl4] | 7.84 | Toxic |
| Dodecyl (2-hydroxyethyl) dimethylammonium iron tetrachloride | [N1,1,12,2(OH)][FeCl4] | 1.04 | Toxic |
| Dodecyl (2-hydroxyethyl) dimethylammonium manganese tetrachloride | [N1,1,12,2(OH)]2[MnCl4] | 0.76 | Toxic |
| Butyl di(2-hydroxyethyl) methylammonium cobalt tetrachloride | [N1,4,2(OH),2(OH)]2[CoCl4] | 10.41 | Moderately toxic |
| Butyl di(2-hydroxyethyl) methylammonium manganese tetrachloride | [N1,4,2(OH),2(OH)]2[MnCl4] | 32.26 | Moderately toxic |
| Butyl di(2-hydroxyethyl) methylammonium gadolinium hexachloride | [N1,4,2(OH),2(OH)]3[GdCl6] | 24.1 | Moderately toxic |
| Hexyl di(2-hydroxyethyl) methylammonium cobalt tetrachloride | [N1,6,2(OH),2(OH)]2[CoCl4] | 18.08 | Moderately toxic |
| Hexyl di(2-hydroxyethyl) methylammonium iron tetrachloride | [N1,6,2(OH),2(OH)][FeCl4] | 16.81 | Moderately toxic |
| Hexyl di(2-hydroxyethyl) methylammonium manganese tetrachloride | [N1,6,2(OH),2(OH)]2[MnCl4] | 69.54 | Moderately toxic |
| Hexyl di(2-hydroxyethyl) methylammonium gadolinium hexachloride | [N1,6,2(OH),2(OH)]3[GdCl6] | 34.17 | Moderately toxic |
| Hexyl tri(2-hydroxyethyl) ammonium cobalt tetrachloride | [N6,2(OH),2(OH),2(OH)]2[CoCl4] | 13.11 | Moderately toxic |
| Hexyl tri(2-hydroxyethyl) ammonium iron tetrachloride | [N6,2(OH),2(OH),2(OH)][FeCl4] | 5.99 | Toxic |
| Hexyl tri(2-hydroxyethyl) ammonium manganese tetrachloride | [N6,2(OH),2(OH),2(OH)]2[MnCl4] | 10.19 | Moderately toxic |
| Hexyl tri(2-hydroxyethyl) ammonium gadolinium hexachloride | [N6,2(OH),2(OH),2(OH)]3[GdCl6] | 17.52 | Moderately toxic |
| Ionic Liquid | Bacteria | Yeasts | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Staphylococcus aureus ATCC 33862 | Staphylococcus epidermidis ATCC 12228 | Bacillus subtilis ATCC 11774 | Enterococcus faecalis ATCC 19433 | Micrococcus luteus ATCC 4698 | Pseudomonas aeruginosa ATCC 9027 | Serratia marcescens ATCC 8100 | Proteus vulgaris ATCC 49132 | Moraxella catarrhalis ATCC 25238 | Escherichia coli ATCC 8739 | Rhodotorula rubra | Candida albicans ATCC 10231 | |
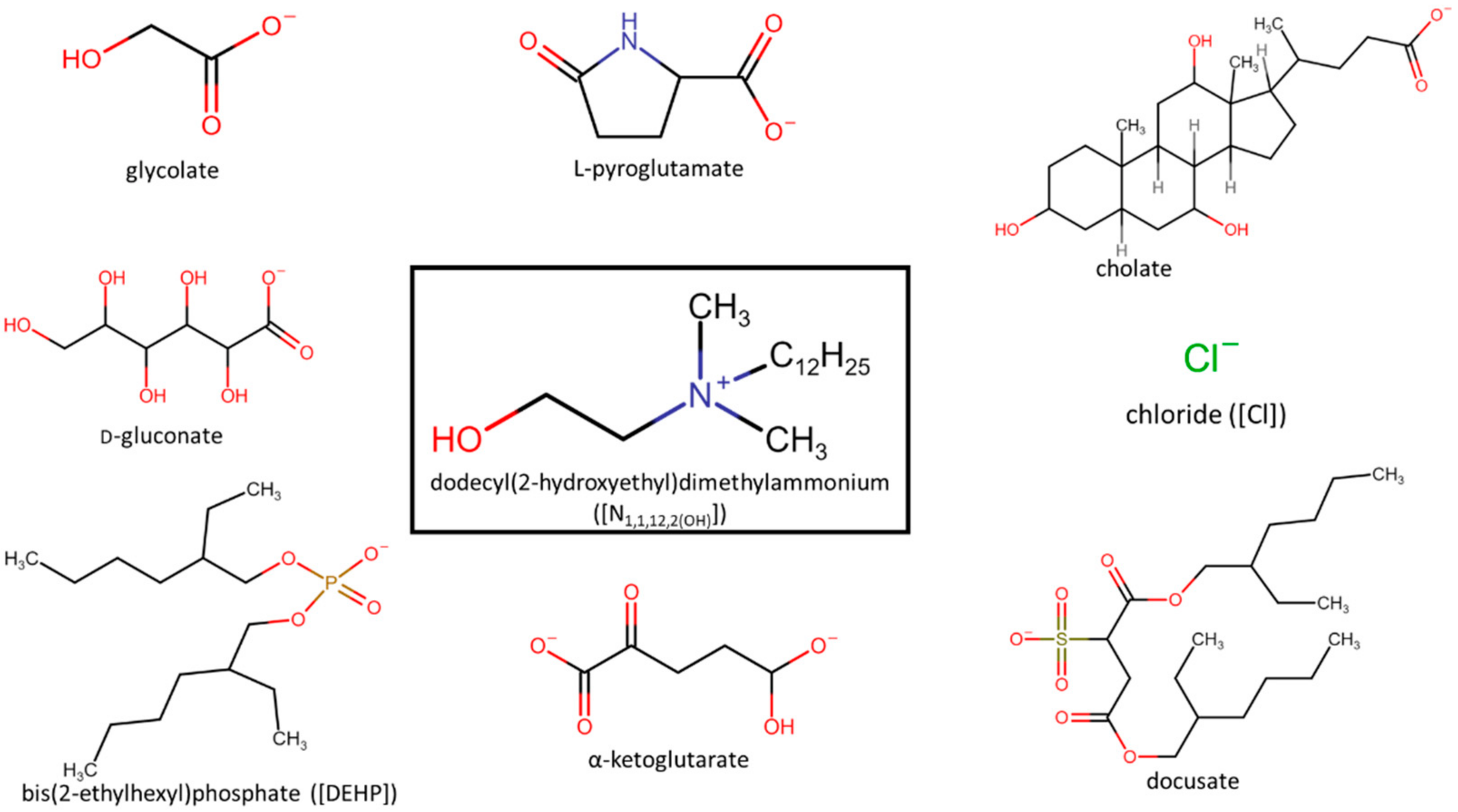
| [N1,1,12,2(OH)][glycolate] | 8 | 31 | 16 | 8 | <0.5 | 250 | 62 | 125 | 125 | 125 | 125 | 16 |
| [N1,1,12,2(OH)][D-gluconate] | 16 | 31 | 16 | 16 | 1 | 500 | 125 | 250 | 250 | 125 | 125 | 31 |
| [N1,1,12,2(OH)][α-ketoglutarate] | 8 | 31 | 16 | 8 | <0.5 | 250 | 62 | 125 | 125 | 125 | 125 | 16 |
| [N1,1,12,2(OH)][L-pyroglutamate] | 16 | 31 | 16 | 8 | 1 | 250 | 62 | 125 | 62 | 125 | 125 | 16 |
| [N1,1,12,2(OH)][cholate] | 8 | 31 | 16 | 16 | 1 | 250 | 125 | 250 | 125 | 250 | 125 | 31 |
| [N1,1,12,2(OH)][docusate] | 125 | 250 | 125 | 125 | 250 | >1000 | >1000 | >1000 | >1000 | >1000 | 500 | 125 |
| [N1,1,12,2(OH)][DEHP] | 16 | 31 | 31 | 16 | 31 | 250 | 62 | 250 | 62 | 125 | 125 | 8 |
| [N1,1,12,2(OH)][Cl] | 8 | 2 | 125 | 16 | 31 | 250 | 500 | 125 | 8 | 125 | 32 | 125 |
| [N10,10,1,1][Cl] | <0.5 | <0.5 | 0.5 | <0.5 | <0.5 | 8 | 4 | 4 | < 0.5 | 1 | 1 | 2 |
| [BA][Cl] | <0.5 | <0.5 | 0.5 | 0.5 | <0.5 | 16 | 4 | 8 | 0.5 | 4 | 4 | 4 |
| Relative Toxicity | EC50 (μg·mL−1) |
|---|---|
| Super toxic | <0.01 |
| Extremely toxic | 0.01–0.1 |
| Highly toxic | 0.1–1 |
| Moderate toxic | 1–10 |
| Slightly toxic | 10–100 |
| Practically harmless | 100–1000 |
| Relatively harmless | >1000 |
| Ionic Liquid | 96 h LC50 (µg·mL−1) | Hazard Ranking | Reference | |
|---|---|---|---|---|
| Cation | Anion | |||
| [C2mim] | [Cl] | 2620 (2360–2830) | Relatively harmless | [93,96] |
| [Br] | 2970 (2760–3160) | |||
| [BF4] | 2170 (1690–2540) | |||
| [NO3] | 3193.5 (2912.7–3472.2) | |||
| [C4mim] | [Cl] | 632.8 ± 67.4 | Practically harmless | [93,95,96,99] |
| [Br] | 897 (618–2220) | |||
| [BF4] | 604.6 ± 56.2 | |||
| [NO3] | 867.1 (805.1–928.8) | |||
| [PF6] | 550 ± 6.348 | |||
| [C6mim] | [Cl] | 128.5 (116.1–140.0) | Practically harmless | [93] |
| [Br] | 164.4 (154.7–174.5) | |||
| [BF4] | 120.1 (109.8–129.1) | |||
| [NO3] | 144.4 (113.1–188.0) | |||
| [PF6] | 146.6 (130.1–162.4) | |||
| [C8mim] | [Cl] | 152.3 ± 12.1 | Practically harmless | [92,93,96] |
| [Br] | 47.8 (39.5–55.8) | Slightly toxic | ||
| [BF4] | 144.0 ± 11.4 | Practically harmless | ||
| [NO3] | 23.1 (18.5–28.7) | Slightly toxic | ||
| [C10mim] | [Br] | 3.80 (2.80–5.10) | Moderate toxic | [93,96] |
| [NO3] | 4.5 (3.2–6.2) | |||
| [C12mim] | [Br] | 3.60 (3.00–4.20) | Moderate toxic | [93,96] |
| [NO3] | 3.7 (3.0–4.6) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, A.R.P.; Paredes, X.; Cristino, A.F.; Santos, F.J.V.; Queirós, C.S.G.P. Ionic Liquids—A Review of Their Toxicity to Living Organisms. Int. J. Mol. Sci. 2021, 22, 5612. https://doi.org/10.3390/ijms22115612
Gonçalves ARP, Paredes X, Cristino AF, Santos FJV, Queirós CSGP. Ionic Liquids—A Review of Their Toxicity to Living Organisms. International Journal of Molecular Sciences. 2021; 22(11):5612. https://doi.org/10.3390/ijms22115612
Chicago/Turabian StyleGonçalves, Ana R.P., Xavier Paredes, A. F. Cristino, F. J.V. Santos, and Carla S.G.P. Queirós. 2021. "Ionic Liquids—A Review of Their Toxicity to Living Organisms" International Journal of Molecular Sciences 22, no. 11: 5612. https://doi.org/10.3390/ijms22115612
APA StyleGonçalves, A. R. P., Paredes, X., Cristino, A. F., Santos, F. J. V., & Queirós, C. S. G. P. (2021). Ionic Liquids—A Review of Their Toxicity to Living Organisms. International Journal of Molecular Sciences, 22(11), 5612. https://doi.org/10.3390/ijms22115612