Temperature-Dependent Structural Variability of Prion Protein Amyloid Fibrils
Abstract
1. Introduction
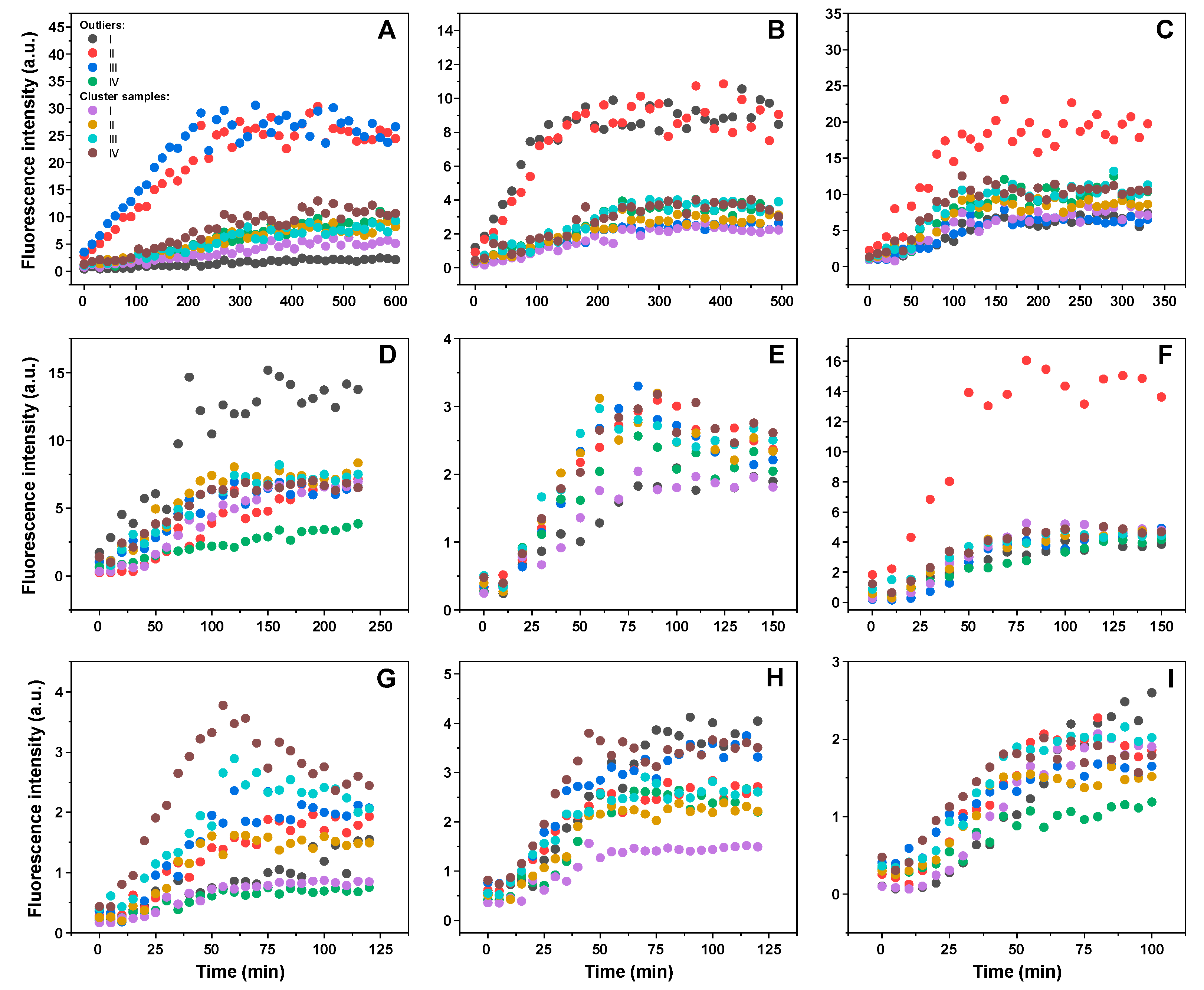
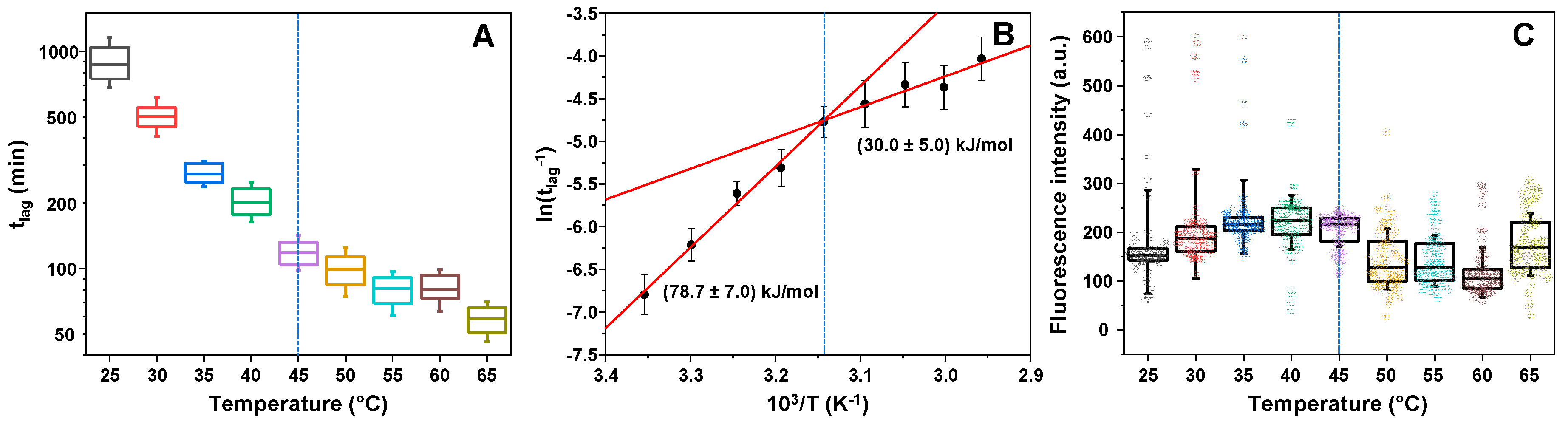
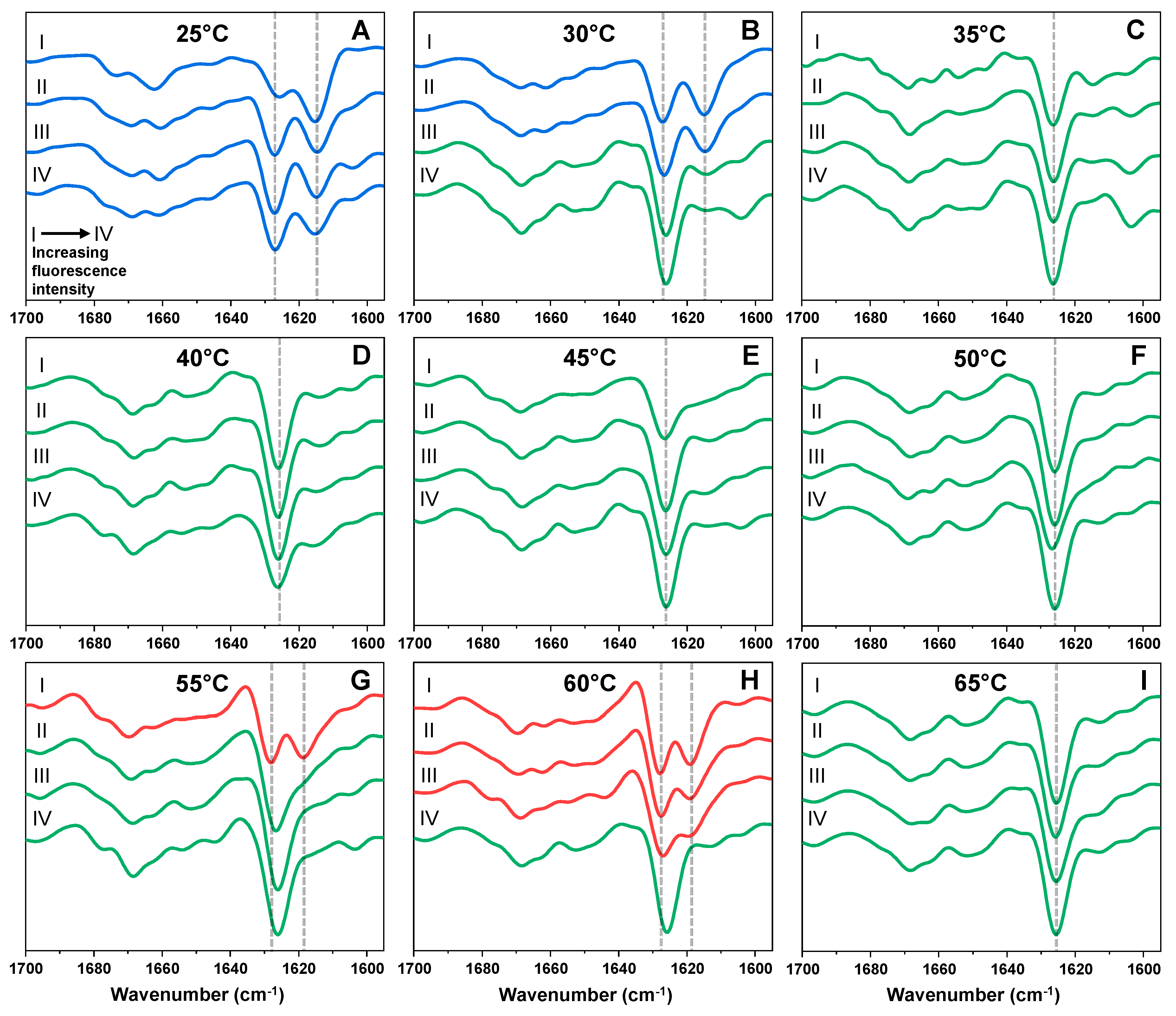
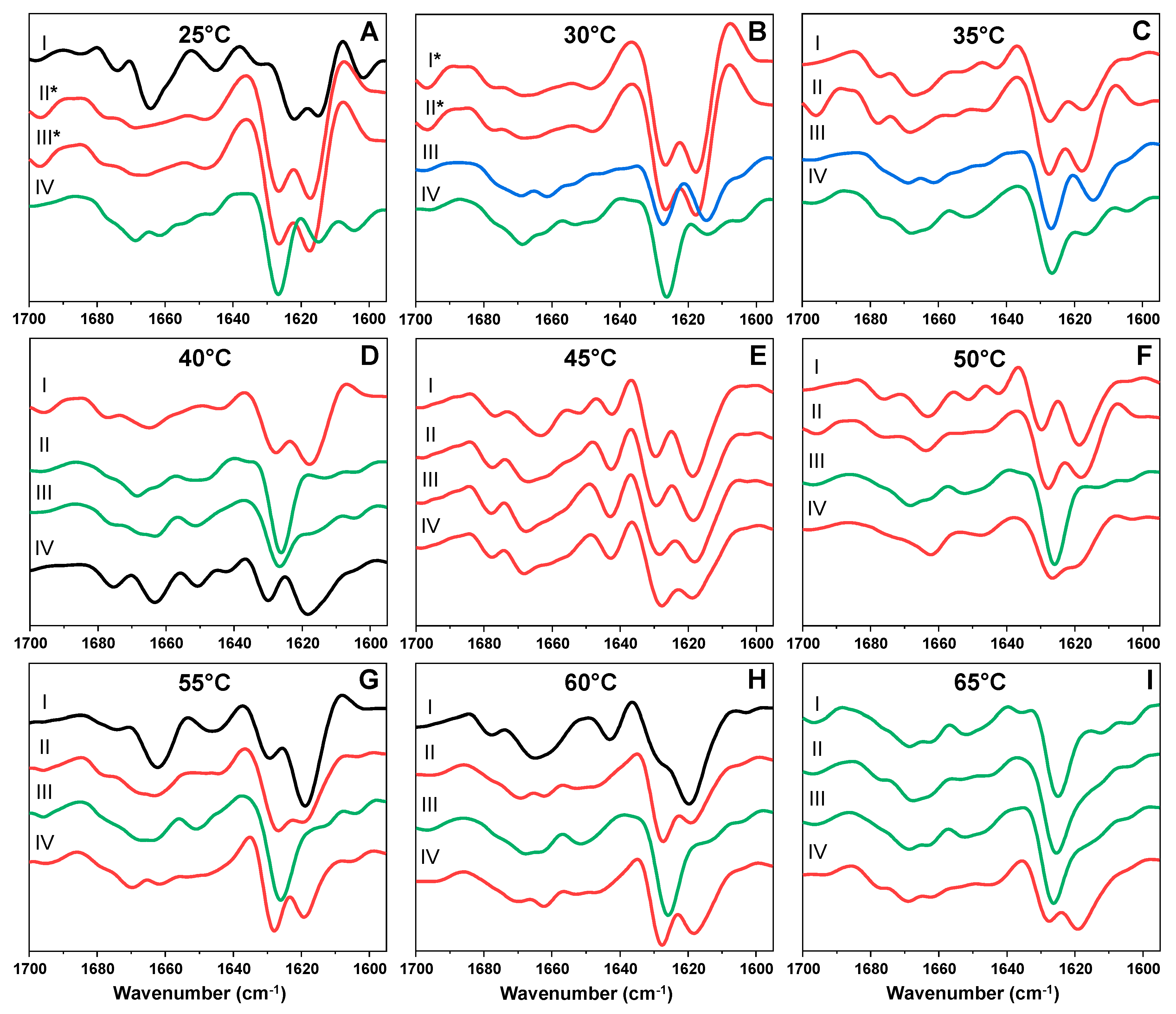
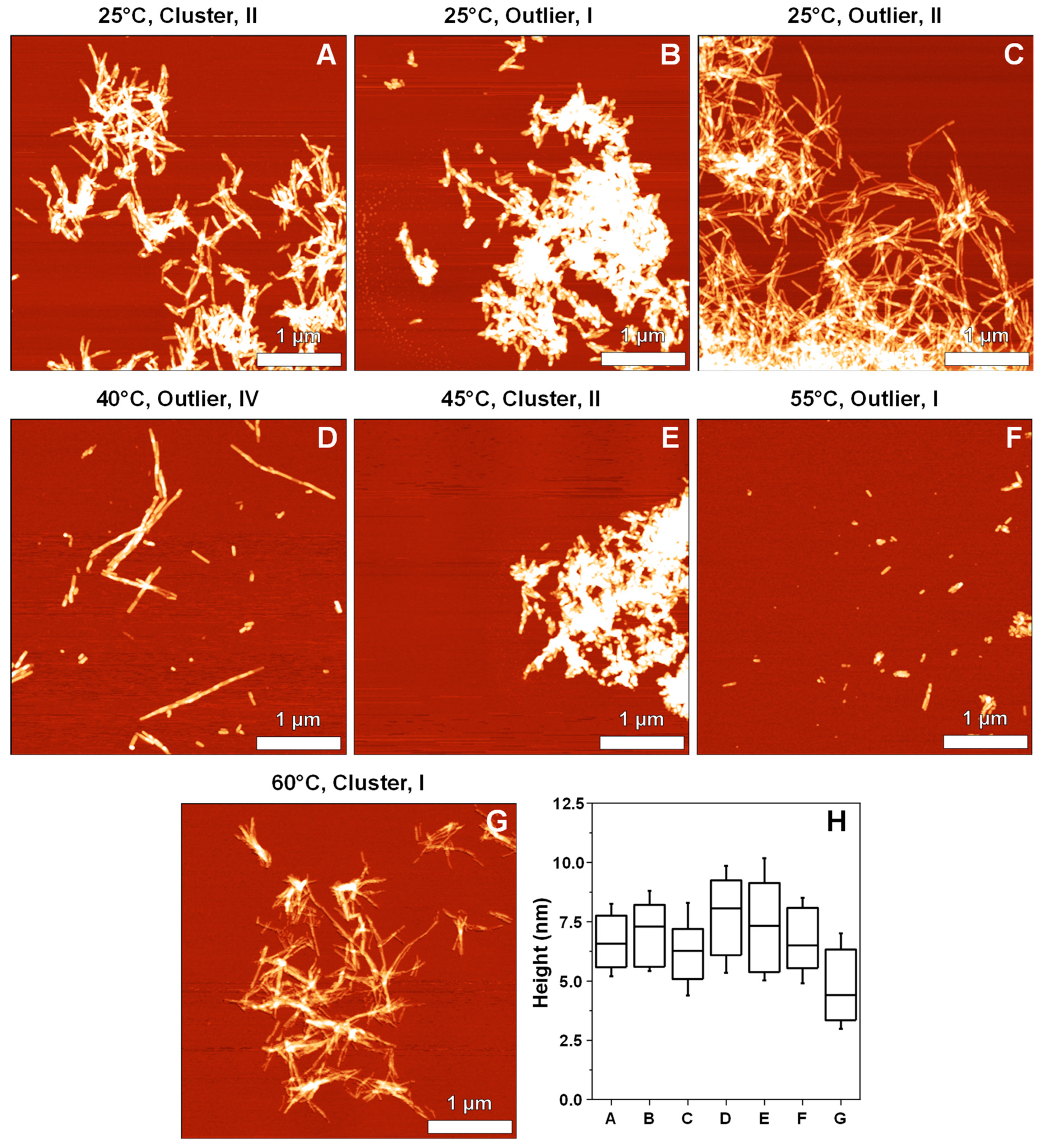
2. Results
3. Discussion
4. Materials and Methods
4.1. Prion Protein Aggregation
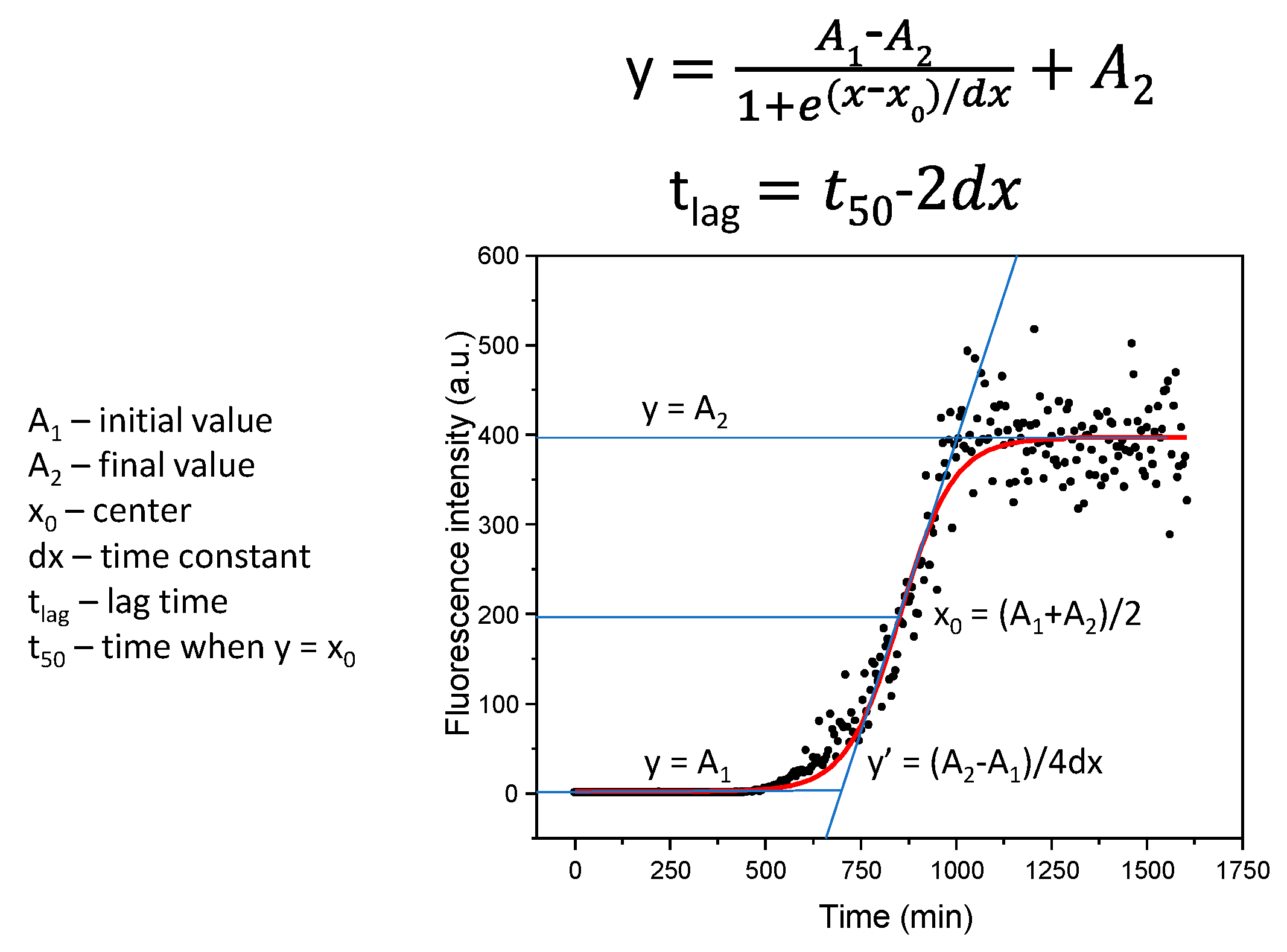
4.2. ThT-Assay
4.3. Fourier-Transform Infrared Spectroscopy
4.4. Atomic Force Microscopy
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| T | Cluster | Excitation | Emission | Intensity | Outliers | Excitation | Emission | Intensity |
|---|---|---|---|---|---|---|---|---|
| 25 °C | I | 444.1 | 483.7 | 98.0 | I | 445.0 | 482.3 | 61.1 |
| 25 °C | II | 444.3 | 484.1 | 145.6 | II | 446.4 | 486.1 | 440.6 |
| 25 °C | III | 443.0 | 483.9 | 154.7 | III | 447.4 | 485.3 | 594.5 |
| 25 °C | IV | 445.9 | 484.3 | 200.6 | IV | 446.8 | 483.6 | 187.6 |
| 30 °C | I | 443.5 | 484.3 | 113.7 | I | 446.8 | 486.3 | 587.1 |
| 30 °C | II | 444.2 | 483.9 | 164.8 | II | 447.2 | 485.4 | 590.8 |
| 30 °C | III | 446.2 | 483.1 | 198.6 | III | 441.8 | 484.1 | 144.4 |
| 30 °C | IV | 448.7 | 483.1 | 251.7 | IV | 444.9 | 482.8 | 201.4 |
| 35 °C | I | 445.7 | 483.7 | 180.5 | I | 451.5 | 482.6 | 183.9 |
| 35 °C | II | 446.2 | 483.4 | 210.9 | II | 446.8 | 485.9 | 465.7 |
| 35 °C | III | 446.3 | 483.8 | 220.9 | III | 443.7 | 483.8 | 154.6 |
| 35 °C | IV | 446.8 | 483.8 | 271.4 | IV | 450.1 | 483.4 | 236.4 |
| 40 °C | I | 446.1 | 483.3 | 71.4 | I | 446.2 | 485.4 | 293.8 |
| 40 °C | II | 448.0 | 483.3 | 207.0 | II | 442.8 | 483.5 | 38.8 |
| 40 °C | III | 447.8 | 482.9 | 242.1 | III | 452.0 | 482.4 | 175.3 |
| 40 °C | IV | 446.3 | 483.4 | 274.4 | IV | 457.8 | 482.9 | 141.3 |
| 45 °C | I | 445.9 | 483.0 | 117.1 | I | 454.1 | 482.3 | 132.6 |
| 45 °C | II | 447.1 | 483.0 | 212.0 | II | 450.8 | 482.9 | 142.8 |
| 45 °C | III | 445.1 | 483.2 | 224.2 | III | 442.4 | 485.1 | 142.4 |
| 45 °C | IV | 447.5 | 483.7 | 250.1 | IV | 442.7 | 484.3 | 111.8 |
| 50 °C | I | 443.2 | 483.4 | 49.9 | I | 455.6 | 482.6 | 88.9 |
| 50 °C | II | 444.6 | 483.3 | 107.6 | II | 447.1 | 485.1 | 404.2 |
| 50 °C | III | 446.0 | 483.2 | 175.8 | III | 439.7 | 484.6 | 26.1 |
| 50 °C | IV | 447.1 | 482.9 | 273.2 | IV | 442.5 | 486.3 | 79.3 |
| 55 °C | I | 442.8 | 483.8 | 70.3 | I | 441.8 | 486.8 | 104.7 |
| 55 °C | II | 443.7 | 482.9 | 112.2 | II | 443.0 | 485.5 | 119.2 |
| 55 °C | III | 445.6 | 482.7 | 170.5 | III | 449.7 | 482.5 | 158.8 |
| 55 °C | IV | 446.4 | 483.6 | 277.7 | IV | 441.6 | 483.7 | 62.8 |
| 60 °C | I | 442.8 | 482.9 | 57.9 | I | 442.2 | 485.8 | 122.7 |
| 60 °C | II | 444.7 | 483.2 | 92.2 | II | 448.9 | 484.6 | 108.3 |
| 60 °C | III | 443.9 | 483.8 | 117.2 | III | 450.1 | 482.4 | 166.6 |
| 60 °C | IV | 448.9 | 482.8 | 295.2 | IV | 442.1 | 483.9 | 106.6 |
| 65 °C | I | 444.9 | 483.4 | 30.0 | I | 439.4 | 483.9 | 29.7 |
| 65 °C | II | 446.6 | 482.8 | 146.3 | II | 443.9 | 484.5 | 112.0 |
| 65 °C | III | 447.3 | 482.7 | 201.9 | III | 447.8 | 481.9 | 219.1 |
| 65 °C | IV | 448.6 | 482.7 | 305.8 | IV | 447.9 | 483.9 | 127.6 |


References
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress Over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef] [PubMed]
- Knowles, T.P.J.; Vendruscolo, M.; Dobson, C.M. The amyloid state and its association with protein misfolding diseases. Nat. Rev. Mol. Cell Biol. 2014, 15, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Meisl, G.; Kirkegaard, J.B.; Arosio, P.; Michaels, T.C.T.; Vendruscolo, M.; Dobson, C.M.; Linse, S.; Knowles, T.P.J. Molecular mechanisms of protein aggregation from global fitting of kinetic models. Nat. Protoc. 2016, 11, 252–272. [Google Scholar] [CrossRef]
- Taguchi, Y.; Otaki, H.; Nishida, N. Mechanisms of Strain Diversity of Disease-Associated in-Register Parallel β-Sheet Amyloids and Implications About Prion Strains. Viruses 2019, 11, 110. [Google Scholar] [CrossRef]
- Linse, S. Mechanism of amyloid protein aggregation and the role of inhibitors. Pure Appl. Chem. 2019, 91, 211–229. [Google Scholar] [CrossRef]
- Mehta, D.; Jackson, R.; Paul, G.; Shi, J.; Sabbagh, M. Why do trials for Alzheimer’s disease drugs keep failing? A discontinued drug perspective for 2010–2015. Expert Opin. Investig. Drugs 2017, 26, 735–739. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Lee, G.; Ritter, A.; Sabbagh, M.; Zhong, K. Alzheimer’s disease drug development pipeline: 2020. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2020, 6, 1–29. [Google Scholar] [CrossRef]
- Arthur, K.C.; Calvo, A.; Price, T.R.; Geiger, J.T.; Chiò, A.; Traynor, B.J. Projected increase in amyotrophic lateral sclerosis from 2015 to 2040. Nat. Commun. 2016, 7, 12408. [Google Scholar] [CrossRef]
- Hebert, L.E.; Weuve, J.; Scherr, P.A.; Evans, D.A. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology 2013, 80, 1778–1783. [Google Scholar] [CrossRef]
- Tycko, R. Physical and structural basis for polymorphism in amyloid fibrils. Protein Sci. 2014, 23, 1528–1539. [Google Scholar] [CrossRef] [PubMed]
- Collinge, J.; Clarke, A.R. A General Model of Prion Strains and Their Pathogenicity. Science 2007, 318, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Thackray, A.M.; Hopkins, L.; Monie, T.P.; Burke, D.F.; Bujdoso, R. Polymorphisms at amino acid residues 141 and 154 influence conformational variation in ovine PrP. Biomed Res. Int. 2014, 2014, 372491. [Google Scholar] [CrossRef]
- Fraser, H.; Dickinson, A.G. Scrapie in mice. J. Comp. Pathol. 1973, 83, 29–40. [Google Scholar] [CrossRef]
- Cobb, N.J.; Apostol, M.I.; Chen, S.; Smirnovas, V.; Surewicz, W.K. Conformational Stability of Mammalian Prion Protein Amyloid Fibrils Is Dictated by a Packing Polymorphism within the Core Region. J. Biol. Chem. 2014, 289, 2643–2650. [Google Scholar] [CrossRef]
- Petkova, A.T.; Leapman, R.D.; Guo, Z.; Yau, W.M.; Mattson, M.P.; Tycko, R. Self-propagating, molecular-level polymorphism in Alzheimer’s β-amyloid fibrils. Science 2005, 307, 262–265. [Google Scholar] [CrossRef]
- Heise, H.; Hoyer, W.; Becker, S.; Andronesi, O.C.; Riedel, D.; Baldus, M. Molecular-level secondary structure, polymorphism, and dynamics of full-length -synuclein fibrils studied by solid-state NMR. Proc. Natl. Acad. Sci. USA 2005, 102, 15871–15876. [Google Scholar] [CrossRef]
- Bousset, L.; Pieri, L.; Ruiz-Arlandis, G.; Gath, J.; Jensen, P.H.; Habenstein, B.; Madiona, K.; Olieric, V.; Böckmann, A.; Meier, B.H.; et al. Structural and functional characterization of two alpha-synuclein strains. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Sneideris, T.; Darguzis, D.; Botyriute, A.; Grigaliunas, M.; Winter, R.; Smirnovas, V. pH-Driven Polymorphism of Insulin Amyloid-Like Fibrils. PLoS ONE 2015, 10, e0136602. [Google Scholar] [CrossRef]
- Surmacz-Chwedoruk, W.; Nieznańska, H.; Wójcik, S.; Dzwolak, W. Cross-seeding of fibrils from two types of insulin induces new amyloid strains. Biochemistry 2012, 51, 9460–9469. [Google Scholar] [CrossRef]
- Zampieri, M.; Legname, G.; Altafini, C. Investigating the Conformational Stability of Prion Strains through a Kinetic Replication. Model. PLoS Comput. Biol. 2009, 5, e1000420. [Google Scholar] [CrossRef] [PubMed]
- Tixador, P.; Herzog, L.; Reine, F.; Jaumain, E.; Chapuis, J.; Le Dur, A.; Laude, H.; Béringue, V. The Physical Relationship between Infectivity and Prion Protein Aggregates Is Strain-Dependent. PLoS Pathog. 2010, 6, e1000859. [Google Scholar] [CrossRef]
- Sidhu, A.; Segers-Nolten, I.; Raussens, V.; Claessens, M.M.A.E.; Subramaniam, V. Distinct Mechanisms Determine α-Synuclein Fibril Morphology during Growth and Maturation. ACS Chem. Neurosci. 2017, 8, 538–547. [Google Scholar] [CrossRef]
- Ziaunys, M.; Sneideris, T.; Smirnovas, V. Formation of distinct prion protein amyloid fibrils under identical experimental conditions. Sci. Rep. 2020, 10, 4572. [Google Scholar] [CrossRef]
- Sneideris, T.; Milto, K.; Smirnovas, V. Polymorphism of amyloid-like fibrils can be defined by the concentration of seeds. PeerJ 2015, 3, e1207. [Google Scholar] [CrossRef] [PubMed]
- Poma, A.B.; Chwastyk, M.; Cieplak, M. Elastic moduli of biological fibers in a coarse-grained model: Crystalline cellulose and β-amyloids. Phys. Chem. Chem. Phys. 2017, 19, 28195–28206. [Google Scholar] [CrossRef] [PubMed]
- Poma, A.B.; Guzman, H.V.; Li, M.S.; Theodorakis, P.E. Mechanical and thermodynamic properties of Aβ42, Aβ40, and α-synuclein fibrils: A coarse-grained method to complement experimental studies. Beilstein J. Nanotechnol. 2019, 10, 500–513. [Google Scholar] [CrossRef]
- Ruggeri, F.S.; Adamcik, J.; Jeong, J.S.; Lashuel, H.A.; Mezzenga, R.; Dietler, G. Influence of the β-sheet content on the mechanical properties of aggregates during amyloid fibrillization. Angew. Chemie Int. Ed. 2015, 54, 2462–2466. [Google Scholar] [CrossRef] [PubMed]
- Sneideris, T.; Sakalauskas, A.; Sternke-Hoffmann, R.; Peduzzo, A.; Ziaunys, M.; Buell, A.K.; Smirnovas, V. The Environment Is a Key Factor in Determining the Anti-Amyloid Efficacy of EGCG. Biomolecules 2019, 9, 855. [Google Scholar] [CrossRef] [PubMed]
- Tycko, R. Amyloid Polymorphism: Structural Basis and Neurobiological Relevance. Neuron 2015, 86, 632–645. [Google Scholar] [CrossRef]
- Close, W.; Neumann, M.; Schmidt, A.; Hora, M.; Annamalai, K.; Schmidt, M.; Reif, B.; Schmidt, V.; Grigorieff, N.; Fändrich, M. Physical basis of amyloid fibril polymorphism. Nat. Commun. 2018, 9, 699. [Google Scholar] [CrossRef]
- Tanaka, M.; Chien, P.; Yonekura, K.; Weissman, J.S. Mechanism of cross-species prion transmission: An infectious conformation compatible with two highly divergent yeast prion proteins. Cell 2005, 121, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Makarava, N.; Ostapchenko, V.G.; Savtchenko, R.; Baskakov, I.V. Conformational switching within individual amyloid fibrils. J. Biol. Chem. 2009, 284, 14386–14395. [Google Scholar] [CrossRef]
- Flynn, J.D.; McGlinchey, R.P.; Walker, R.L.; Lee, J.C. Structural features of α-synuclein amyloid fibrils revealed by Raman spectroscopy. J. Biol. Chem. 2018, 293, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Sakalauskas, A.; Ziaunys, M.; Smirnovas, V. Concentration-dependent polymorphism of insulin amyloid fibrils. PeerJ 2019, 7, e8208. [Google Scholar] [CrossRef]
- Adamcik, J.; Mezzenga, R. Study of amyloid fibrils via atomic force microscopy. Curr. Opin. Colloid Interface Sci. 2012, 17, 369–376. [Google Scholar] [CrossRef]
- Iannuzzi, C.; Borriello, M.; Portaccio, M.; Irace, G.; Sirangelo, I. Insights into insulin fibril assembly at physiological and acidic ph and related amyloid intrinsic fluorescence. Int. J. Mol. Sci. 2017, 18, 2551. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, M.; Zurdo, J.; Nettleton, E.J.; Dobson, C.M.; Robinson, C.V. Formation of insulin amyloid fibrils followed by FTIR simultaneously with CD and electron microscopy. Protein Sci. 2000, 9, 1960–1967. [Google Scholar] [CrossRef]
- Aldewachi, H.; Al-Zidan, R.N.; Conner, M.T.; Salman, M.M. High-throughput screening platforms in the discovery of novel drugs for neurodegenerative diseases. Bioengineering 2021, 8, 30. [Google Scholar] [CrossRef]
- Salman, M.M.; Al-Obaidi, Z.; Kitchen, P.; Loreto, A.; Bill, R.M.; Wade-Martins, R. Advances in Applying Computer-Aided Drug Design for Neurodegenerative Diseases. Int. J. Mol. Sci. 2021, 22, 4688. [Google Scholar] [CrossRef]
- Ziaunys, M.; Sakalauskas, A.; Smirnovas, V. Identifying Insulin Fibril Conformational Differences by Thioflavin-T Binding Characteristics. Biomacromolecules 2020, 21, 4989–4997. [Google Scholar] [CrossRef]
- Sidhu, A.; Vaneyck, J.; Blum, C.; Segers-Nolten, I.; Subramaniam, V. Polymorph-specific distribution of binding sites determines thioflavin-T fluorescence intensity in α-synuclein fibrils. Amyloid 2018, 25, 189–196. [Google Scholar] [CrossRef]
- Kovachev, P.S.; Gomes, M.P.B.; Cordeiro, Y.; Ferreira, N.C.; Valadão, L.P.F.; Ascari, L.M.; Rangel, L.P.; Silva, J.L.; Sanyal, S. RNA modulates aggregation of the recombinant mammalian prion protein by direct interaction. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Milto, K.; Michailova, K.; Smirnovas, V. Elongation of Mouse Prion Protein Amyloid-Like Fibrils: Effect of Temperature and Denaturant Concentration. PLoS ONE 2014, 9, e94469. [Google Scholar] [CrossRef] [PubMed]
- Rodina, N.P.; Sulatsky, M.I.; Sulatskaya, A.I.; Kuznetsova, I.M.; Uversky, V.N.; Turoverov, K.K. Photophysical Properties of Fluorescent Probe Thioflavin T in Crowded Milieu. J. Spectrosc. 2017, 2017. [Google Scholar] [CrossRef]
- Foderà, V.; Groenning, M.; Vetri, V.; Librizzi, F.; Spagnolo, S.; Cornett, C.; Olsen, L.; van de Weert, M.; Leone, M. Thioflavin T Hydroxylation at Basic pH and Its Effect on Amyloid Fibril Detection. J. Phys. Chem. B 2008, 112, 15174–15181. [Google Scholar] [CrossRef] [PubMed]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef]
- Ma, B.; Nussinov, R. Simulations as analytical tools to understand protein aggregation and predict amyloid conformation. Curr. Opin. Chem. Biol. 2006, 10, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Ziaunys, M.; Smirnovas, V. Additional Thioflavin-T Binding Mode in Insulin Fibril Inner Core Region. J. Phys. Chem. B 2019, 123, 8727–8732. [Google Scholar] [CrossRef]
- Mikalauskaite, K.; Ziaunys, M.; Sneideris, T.; Smirnovas, V. Effect of Ionic Strength on Thioflavin-T Affinity to Amyloid Fibrils and Its Fluorescence Intensity. Int. J. Mol. Sci. 2020, 21, 8916. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziaunys, M.; Sakalauskas, A.; Mikalauskaite, K.; Snieckute, R.; Smirnovas, V. Temperature-Dependent Structural Variability of Prion Protein Amyloid Fibrils. Int. J. Mol. Sci. 2021, 22, 5075. https://doi.org/10.3390/ijms22105075
Ziaunys M, Sakalauskas A, Mikalauskaite K, Snieckute R, Smirnovas V. Temperature-Dependent Structural Variability of Prion Protein Amyloid Fibrils. International Journal of Molecular Sciences. 2021; 22(10):5075. https://doi.org/10.3390/ijms22105075
Chicago/Turabian StyleZiaunys, Mantas, Andrius Sakalauskas, Kamile Mikalauskaite, Ruta Snieckute, and Vytautas Smirnovas. 2021. "Temperature-Dependent Structural Variability of Prion Protein Amyloid Fibrils" International Journal of Molecular Sciences 22, no. 10: 5075. https://doi.org/10.3390/ijms22105075
APA StyleZiaunys, M., Sakalauskas, A., Mikalauskaite, K., Snieckute, R., & Smirnovas, V. (2021). Temperature-Dependent Structural Variability of Prion Protein Amyloid Fibrils. International Journal of Molecular Sciences, 22(10), 5075. https://doi.org/10.3390/ijms22105075







