Recent Findings Unravel Genes and Genetic Factors Underlying Leptosphaeria maculans Resistance in Brassica napus and Its Relatives
Abstract
1. Introduction
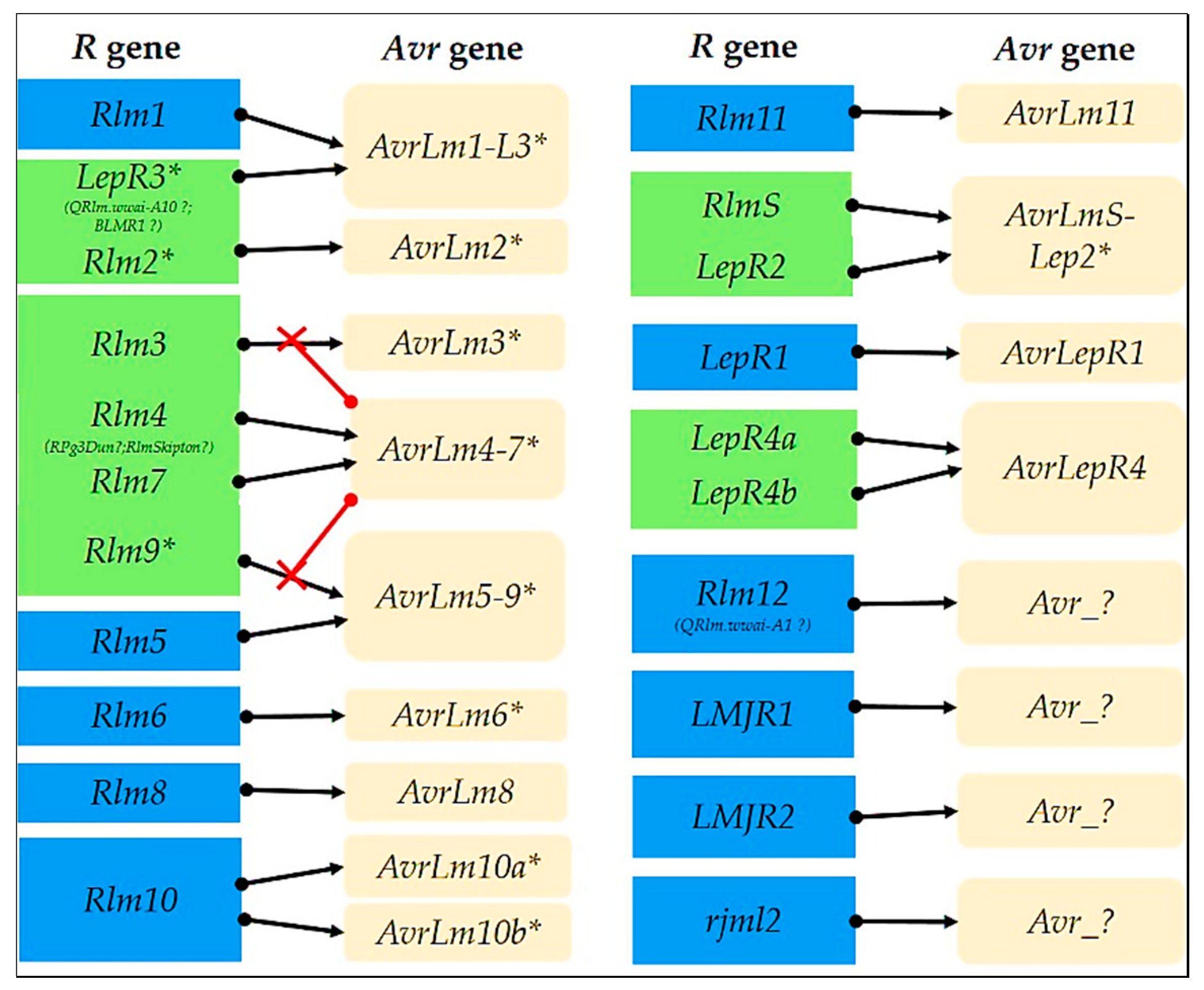
2. The Current Resistance Genes Go Beyond Simple Allelism
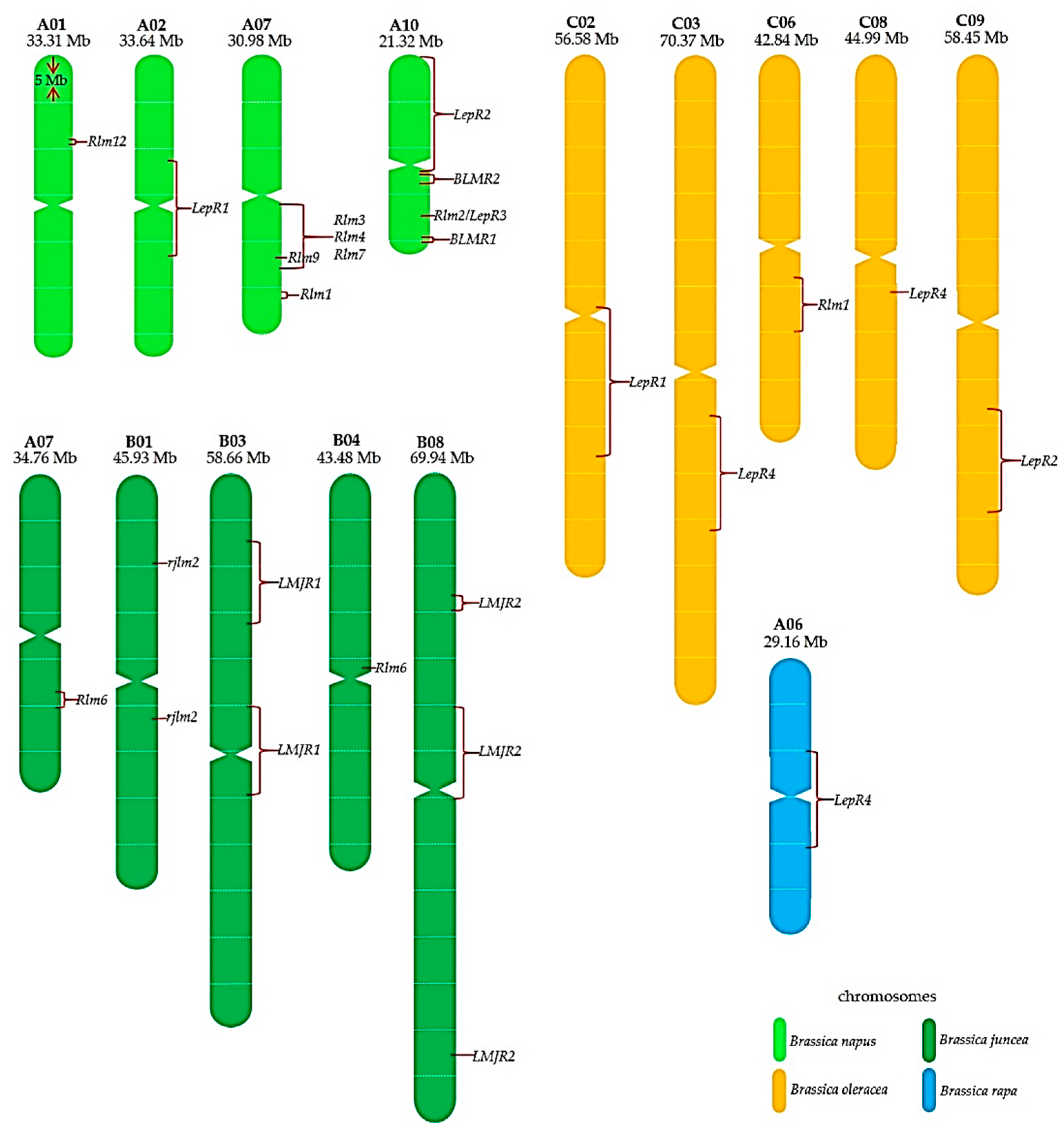
3. Exploring Resistance Genes in Brassica napus and Its Relatives
4. Genome Sequencing in Brassica Species Hastened the Identification of Resistance Genes
5. Genetic Factors Involving the Pathogenicity and Resistance in Leptosphaeria maculans
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tamokou, J.D.D.; Mbaveng, A.T.; Kuete, V. Chapter 8—Antimicrobial Activities of African Medicinal Spices and Vegetables. In Medicinal Spices and Vegetables from Africa; Kuete, V., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 207–237. [Google Scholar] [CrossRef]
- Nagaharu, U. Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn. J. Bot. 1935, 7, 389–452. [Google Scholar]
- Avato, P.; Argentieri, M.P. Brassicaceae: A rich source of health improving phytochemicals. Phytochem. Rev. 2015, 14, 1019–1033. [Google Scholar] [CrossRef]
- Koornneef, M.; Meinke, D. The development of Arabidopsis as a model plant. Plant J. 2010, 61, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Khatun, A.; Liu, L.; Barkla, B.J. Brassicaceae Mustards: Traditional and Agronomic Uses in Australia and New Zealand. Molecules 2018, 23, 231. [Google Scholar] [CrossRef]
- Schmidt, R.; Bancroft, I. Genetics and Genomics of the Brassicaceae, 1st ed.; Springer Science & Business Media: New York, NY, USA, 2010; p. 680. [Google Scholar] [CrossRef]
- Simpson, M.G. 8—Diversity and Classification of Flowering Plants: Eudicots. In Plant Systematics, 2nd ed.; Simpson, M.G., Ed.; Academic Press: San Diego, CA, USA, 2010; pp. 275–448. [Google Scholar] [CrossRef]
- Weeks, D.P. Chapter Four—Gene Editing in Polyploid Crops: Wheat, Camelina, Canola, Potato, Cotton, Peanut, Sugar Cane, and Citrus. In Progress in Molecular Biology and Translational Science; Weeks, D.P., Yang, B., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 149, pp. 65–80. [Google Scholar]
- CODEX ALIMENTARIUS. International Food Standards: Standard for the Named Vegetable Oils CX-S 210—1999; Food and Agriculture Organization of the United Nations: Rome, Italy, 2019. [Google Scholar]
- FAO. FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 20 February 2020).
- AOF. Canola Oil and Cancer the Facts; Australian Oilseeds Federation: Australia Square, NSW, Australia, 2012. [Google Scholar]
- Lin, L.; Allemekinders, H.; Dansby, A.; Campbell, L.; Durance-Tod, S.; Berger, A.; Jones, P.J.H. Evidence of health benefits of canola oil. Nutr. Rev. 2013, 71, 370–385. [Google Scholar] [CrossRef]
- Gaber, M.; Tujillo, F.; Mansour, M.; Juliano, P. Improving Oil Extraction from Canola Seeds by Conventional and Advanced Methods. Food Eng. Rev. 2018, 10, 198–210. [Google Scholar]
- Chambers, K. Pathogenicity Genes of Leptosphaeria maculans, the Fungus that Causes Blackleg Disease of Canola (Brassica napus). Ph.D. Thesis, University of Melbourne, Melbourne, Australia, 2017. [Google Scholar]
- Howlett, B.J. Current knowledge of the interaction between Brassica napus and Leptosphaeria maculans. Can. J. Plant Pathol. 2004, 26, 245–252. [Google Scholar] [CrossRef]
- McVetty, P.B.E.; Duncan, R.W. Canola/Rapeseed: Genetics and Breeding☆. In Reference Module in Food Science; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- CCC. Canola Council of Canada “Canola Encyclopedia: About Blackleg”. Available online: https://www.canolacouncil.org/canola-encyclopedia/diseases/blackleg/about-blackleg/ (accessed on 10 April 2020).
- Li, H.; Sivasithamparam, K.; Barbetti, M.J. Soilborne ascospores and pycnidiospores of Leptosphaeria maculans can contribute significantly to blackleg disease epidemiology in oilseed rape (Brassica napus) in Western Australia. Australas Plant Pathol. 2007, 36, 439–444. [Google Scholar] [CrossRef]
- West, J.S.; Kharbanda, P.D.; Barbetti, M.J.; Fitt, B.D.L. Epidemiology and management of Leptosphaeria maculans (phoma stem canker) on oilseed rape in Australia, Canada and Europe. Plant Pathol. 2001, 50, 10–27. [Google Scholar] [CrossRef]
- Bokor, A.; Barbetti, M.J.; Brown, A.G.P.; Mac Nish, G.C.A.; Wood, P.M. Blackleg of rapeseed. J. Dep. Agric. West. Aust. Ser. 4 1975, 16, 7–10. [Google Scholar]
- Chen, C.Y.; Howlett, B.J. Rapid necrosis of guard cells is associated with the arrest of fungal growth in leaves of Indian mustard (Brassica juncea) inoculated with avirulent isolates of Leptosphaeria maculans. Physiol. Mol. Plant Pathol. 1996, 48, 73–81. [Google Scholar] [CrossRef]
- Guo, M.; Chen, Y.; Du, Y.; Dong, Y.; Guo, W.; Zhai, S.; Zhang, H.; Dong, S.; Zhang, Z.; Wang, Y.; et al. The bZIP transcription factor MoAP1 mediates the oxidative stress response and is critical for pathogenicity of the rice blast fungus Magnaporthe oryzae. PLoS Pathog. 2011, 7, e1001302. [Google Scholar] [CrossRef]
- Hammond, K.E.; Lewis, B.G.; Musa, T.M. A systemic pathway in the infection of oilseed rape plants by Leptosphaeria maculans. Plant Pathol. 1985, 34, 557–565. [Google Scholar] [CrossRef]
- Howlett, B.J.; Idnurm, A.; Pedras, M.S.C. Leptosphaeria maculans, the Causal Agent of Blackleg Disease of Brassicas. Fungal Genet. Biol. 2001, 33, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rimmer, S.R.; Buchwaldt, L. Diseases. In Brassica Oilseeds: Production and Utilization; Kimber, D.M.D.I., Ed.; CABI: Wallingford, UK, 1995; pp. 114–118. [Google Scholar]
- Wang, D. Transferring Blackleg Resistance from Brassica carinata and Synthetic Hexaploid Brassica accessions into Brassica napus. Master’s Thesis, The University of Manitoba, Winnipeg, MB, Canada, 2016. [Google Scholar]
- Williams, P.H. Biology of Leptosphaeria maculans. Can. J. Plant Pathol. 1992, 14, 30–35. [Google Scholar] [CrossRef]
- Henderson, M.P. The Black-leg Disease of Cabbage Caused by Phoma lingam (Tode) Desmaz. Univ. Wis. Madison 1918, 8, 379–431. [Google Scholar]
- Gugel, R.K.; Petrie, G.A. History, occurrence, impact, and control of blackleg of rapeseed. Can. J. Plant Pathol. 1992, 14, 36–45. [Google Scholar] [CrossRef]
- McGee, D.; Emmett, R. Blackleg (Leptosphaeria maculans (Desm.) Ces. et de Not.) of rapeseed in Victoria: Crop losses and factors which affect disease severity. Aust. J. Agric. Res. 1977, 28, 47–51. [Google Scholar] [CrossRef]
- Vanterpool, T.C. Rape diseases in Saskatchewan in 1961. Can. Plant Dis. Surv. 1961, 41, 372–373. [Google Scholar]
- Fitt, B.; Brun, H.; Barbetti, M.J.; Rimmer, S.R. World-Wide Importance of Phoma Stem Canker (Leptosphaeria maculans and L. biglobosa) on Oilseed Rape (Brassica napus). Eur. J. Plant Pathol. 2006, 114, 3–15. [Google Scholar] [CrossRef]
- Toscano-Underwood, C.; Huang, Y.; Fitt, B.; Hall, A. Effects of temperature on maturation of pseudothecia of Leptosphaeria maculans and L. biglobosa on oilseed rape stem debris. Plant Pathol. 2003, 52, 726–736. [Google Scholar] [CrossRef]
- Van de Wouw, A.P.; Marcroft, S.J.; Howlett, B.J. Blackleg disease of canola in Australia. Crop Pasture Sci. 2016, 67, 273–283. [Google Scholar] [CrossRef]
- Zhang, X.; Fernando, W.G.D. Insights into fighting against blackleg disease of Brassica napus in Canada. Crop Pasture Sci. 2018, 69, 40–47. [Google Scholar] [CrossRef]
- GRDC. Grains Research & Development Corporation “Plan Ahead to Fight Blackleg in Canola This Season”. Available online: https://grdc.com.au/news-and-media/news-and-media-releases/west/2019/4/plan-ahead-to-fight-blackleg-in-canola-this-season (accessed on 10 April 2020).
- Sprague, S.; Marcroft, S.; van De Wouw, A.P.; Lindbeck, K.; Brill, R.; McMaster, C. Blackleg in Canola—Outcomes from 2016 and Update for 2017. Available online: https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2017/08/blackleg-in-canola-outcomes-from-2016-and-update-for-2017 (accessed on 10 April 2020).
- Sprague, S.J.; Balesdent, M.-H.; Brun, H.; Hayden, H.L.; Marcroft, S.J.; Pinochet, X.; Rouxel, T.; Howlett, B.J. Major gene resistance in Brassica napus (oilseed rape) is overcome by changes in virulence of populations of Leptosphaeria maculans in France and Australia. Eur. J. Plant Pathol. 2006, 114, 33–40. [Google Scholar] [CrossRef]
- Van de Wouw, A.P.; Cozijnsen, A.J.; Hane, J.K.; Brunner, P.C.; McDonald, B.A.; Oliver, R.P.; Howlett, B.J. Evolution of Linked Avirulence Effectors in Leptosphaeria maculans Is Affected by Genomic Environment and Exposure to Resistance Genes in Host Plants. PLoS Pathog. 2010, 6, e1001180. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.Q.; Dong, X. Systemic acquired resistance: Turning local infection into global defense. Annu. Rev. Plant Biol. 2013, 64, 839–863. [Google Scholar] [CrossRef] [PubMed]
- Malinovsky, F.G.; Batoux, M.; Schwessinger, B.; Youn, J.H.; Stransfeld, L.; Win, J.; Kim, S.K.; Zipfel, C. Antagonistic regulation of growth and immunity by the Arabidopsis basic helix-loop-helix transcription factor homolog of brassinosteroid enhanced expression2 interacting with increased leaf inclination1 binding bHLH1. Plant Physiol. 2014, 164, 1443–1455. [Google Scholar] [CrossRef]
- Boyd, L.A.; Ridout, C.; O’Sullivan, D.M.; Leach, J.E.; Leung, H. Plant-pathogen interactions: Disease resistance in modern agriculture. Trends Genet. TIG 2013, 29, 233–240. [Google Scholar] [CrossRef]
- Freeman, B.C.; Beattie, G.A. An Overview of Plant Defenses against Pathogens and Herbivores. Plant Health Instr. 2008. [Google Scholar] [CrossRef]
- Jones, J.D.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, J.-M. Plant Immunity Triggered by Microbial Molecular Signatures. Mol. Plant 2010, 3, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Tsuda, K.; Parker, J.E. Effector-triggered immunity: From pathogen perception to robust defense. Annu. Rev. Plant Biol. 2015, 66, 487–511. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Feng, B.; He, P.; Shan, L. From Chaos to Harmony: Responses and Signaling upon Microbial Pattern Recognition. Annu. Rev. Phytopathol. 2017, 55, 109–137. [Google Scholar] [CrossRef] [PubMed]
- Dangl, J.L.; Jones, J.D.G. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826–833. [Google Scholar] [CrossRef]
- Sekhwal, M.K.; Li, P.; Lam, I.; Wang, X.; Cloutier, S.; You, F.M. Disease Resistance Gene Analogs (RGAs) in Plants. Int. J. Mol. Sci. 2015, 16, 19248–19290. [Google Scholar] [CrossRef]
- Gururani, M.A.; Venkatesh, J.; Upadhyaya, C.P.; Nookaraju, A.; Pandey, S.K.; Park, S.W. Plant disease resistance genes: Current status and future directions. Physiol. Mol. Plant Pathol. 2012, 78, 51–65. [Google Scholar] [CrossRef]
- McHale, L.; Tan, X.; Koehl, P.; Michelmore, R.W. Plant NBS-LRR proteins: Adaptable guards. Genome Biol. 2006, 7, 212. [Google Scholar] [CrossRef]
- Meyers, B.C.; Dickerman, A.W.; Michelmore, R.W.; Sivaramakrishnan, S.; Sobral, B.W.; Young, N.D. Plant disease resistance genes encode members of an ancient and diverse protein family within the nucleotide-binding superfamily. Plant J. 1999, 20, 317–332. [Google Scholar] [CrossRef]
- Bohm, H.; Albert, I.; Fan, L.; Reinhard, A.; Nurnberger, T. Immune receptor complexes at the plant cell surface. Curr. Opin. Plant Biol. 2014, 20, 47–54. [Google Scholar] [CrossRef]
- Zipfel, C. Plant pattern-recognition receptors. Trends Immunol. 2014, 35, 345–351. [Google Scholar] [CrossRef]
- Yang, X.; Deng, F.; Ramonell, K. Receptor-like kinases and receptor-like proteins: Keys to pathogen recognition and defense signaling in plant innate immunity. Front. Biol. 2012, 7, 155–166. [Google Scholar] [CrossRef]
- Balesdent, M.H.; Barbetti, M.J.; Li, H.; Sivasithamparam, K.; Gout, L.; Rouxel, T. Analysis of Leptosphaeria maculans Race Structure in a Worldwide Collection of Isolates. Phytopathology 2005, 95, 1061. [Google Scholar] [CrossRef] [PubMed]
- Delourme, R.; Chèvre, A.M.; Brun, H.; Rouxel, T.; Balesdent, M.H.; Dias, J.S.; Salisbury, P.; Renard, M.; Rimmer, S.R. Major Gene and Polygenic Resistance to Leptosphaeria maculans in Oilseed Rape (Brassica napus). Eur. J. Plant Pathol. 2006, 114, 41–52. [Google Scholar] [CrossRef]
- Elliott, V.L.; Marcroft, S.J.; Howlett, B.J.; Van de Wouw, A.P. Gene-for-gene resistance is expressed in cotyledons, leaves and pods, but not during late stages of stem colonization in the Leptosphaeria maculans–Brassica napus pathosystem. Plant Breed 2016, 135, 200–207. [Google Scholar] [CrossRef]
- Rimmer, S.R.; van den Berg, C.G.J. Resistance of oilseed Brassica spp. to blackleg caused by Leptosphaeria maculans. Can. J. Plant Pathol. 1992, 14, 56–66. [Google Scholar] [CrossRef]
- Huang, Y.J.; Pirie, E.J.; Evans, N.; Delourme, R.; King, G.J.; Fitt, B.D.L. Quantitative resistance to symptomless growth of Leptosphaeria maculans (phoma stem canker) in Brassica napus (oilseed rape). Plant Pathol. 2009, 58, 314–323. [Google Scholar] [CrossRef]
- Brun, H.; Chèvre, A.-M.; Fitt, B.D.; Powers, S.; Besnard, A.-L.; Ermel, M.; Huteau, V.; Marquer, B.; Eber, F.; Renard, M.; et al. Quantitative resistance increases the durability of qualitative resistance to Leptosphaeria maculans in Brassica napus. New Phytol. 2010, 185, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Rouxel, T.; Penaud, A.; Pinochet, X.; Brun, H.; Gout, L.; Delourme, R.; Schmit, J.; Balesdent, M.-H. A 10-year Survey of Populations of Leptosphaeria maculans in France Indicates a Rapid Adaptation towards the Rlm1 Resistance Gene of Oilseed Rape. Eur. J. Plant Pathol. 2003, 109, 871–881. [Google Scholar] [CrossRef]
- Sprague, S.J.; Marcroft, S.J.; Hayden, H.L.; Howlett, B.J. Major Gene Resistance to Blackleg in Brassica napus Overcome Within Three Years of Commercial Production in Southeastern Australia. Plant Dis. 2006, 90, 190–198. [Google Scholar] [CrossRef]
- Van de Wouw, A.P.; Howlett, B.J.; Idnurm, A. Changes in allele frequencies of avirulence genes in the blackleg fungus, Leptosphaeria maculans, over two decades in Australia. Crop Pasture Sci. 2017, 69, 20–29. [Google Scholar] [CrossRef]
- Winter, M.; Koopmann, B. Race spectra of Leptosphaeria maculans, the causal agent of blackleg disease of oilseed rape, in different geographic regions in northern Germany. Eur. J. Plant Pathol. 2016, 145, 629–641. [Google Scholar] [CrossRef]
- Zhang, X.; Peng, G.; Kutcher, H.; Balesdent, M.-H.; Delourme, R.; Fernando, W. Breakdown of Rlm3 resistance in the Brassica napus—Leptosphaeria maculans pathosystem in western Canada. Eur. J. Plant Pathol. 2016, 145, 659–674. [Google Scholar] [CrossRef]
- Raman, H.; Raman, R.; Larkan, N. Genetic Dissection of Blackleg Resistance Loci in Rapeseed (Brassica napus L.). In Plant Breed from Laboratories to Fields; Andersen, S.V., Ed.; IntechOpen Limited: London, UK, 2013; pp. 85–120. [Google Scholar] [CrossRef]
- Salisbury, P.A.; Cowling, W.A.; Potter, T.D. Continuing innovation in Australian canola breeding. Crop Pasture Sci. 2016, 67, 266–272. [Google Scholar] [CrossRef]
- Bayer, P.E.; Hurgobin, B.; Golicz, A.A.; Chan, C.K.; Yuan, Y.; Lee, H.; Renton, M.; Meng, J.; Li, R.; Long, Y.; et al. Assembly and comparison of two closely related Brassica napus genomes. Plant Biotechnol. J. 2017, 15, 1602–1610. [Google Scholar] [CrossRef] [PubMed]
- Dolatabadian, A.; Bayer, P.E.; Tirnaz, S.; Hurgobin, B.; Edwards, D.; Batley, J. Characterization of disease resistance genes in the Brassica napus pangenome reveals significant structural variation. Plant Biotechnol. J. 2019, 18, 969–982. [Google Scholar] [CrossRef] [PubMed]
- Hurgobin, B.; Golicz, A.A.; Bayer, P.E.; Chan, C.K.; Tirnaz, S.; Dolatabadian, A.; Schiessl, S.V.; Samans, B.; Montenegro, J.D.; Parkin, I.A.P.; et al. Homoeologous exchange is a major cause of gene presence/absence variation in the amphidiploid Brassica napus. Plant Biotechnol. J. 2018, 16, 1265–1274. [Google Scholar] [CrossRef]
- Bayer, P.; Golicz, A.; Tirnaz, S.; Chan, C.K.K.; Edwards, D.; Batley, J. Variation in abundance of predicted resistance genes in the Brassica oleracea pangenome. Plant Biotechnol. J. 2019, 17. [Google Scholar] [CrossRef]
- Golicz, A.A.; Bayer, P.E.; Barker, G.C.; Edger, P.P.; Kim, H.; Martinez, P.A.; Chan, C.K.K.; Severn-Ellis, A.; McCombie, W.R.; Parkin, I.A.P.; et al. The pangenome of an agronomically important crop plant Brassica oleracea. Nat. Commun. 2016, 7, 13390. [Google Scholar] [CrossRef]
- Yang, J.; Liu, D.; Wang, X.; Ji, C.; Cheng, F.; Liu, B.; Hu, Z.; Chen, S.; Pental, D.; Ju, Y.; et al. The genome sequence of allopolyploid Brassica juncea and analysis of differential homoeolog gene expression influencing selection. Nat. Genet. 2016, 48, 1225. [Google Scholar] [CrossRef]
- Zhang, L.; Cai, X.; Wu, J.; Liu, M.; Grob, S.; Cheng, F.; Liang, J.; Cai, C.; Liu, Z.; Liu, B.; et al. Improved Brassica rapa reference genome by single-molecule sequencing and chromosome conformation capture technologies. Hortic. Res. 2018, 5, 50. [Google Scholar] [CrossRef]
- Larkan, N.J.; Yu, F.; Lydiate, D.J.; Rimmer, S.R.; Borhan, M.H. Single R Gene Introgression Lines for Accurate Dissection of the Brassica—Leptosphaeria Pathosystem. Front. Plant Sci. 2016, 7, 1771. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Yu, F. Understanding the Mechanisms for Race-Specific and Non-Specific Resistance for Effective Use of Cultivar Resistance against Blackleg of Canola in Western Canada; Agriculture and Agri-Food Canada: Saskatoon, SK, Canada, 2018; p. 15. [Google Scholar]
- Fu, F.; Liu, X.; Wang, R.; Zhai, C.; Peng, G.; Yu, F.; Fernando, W.G.D. Fine mapping of Brassica napus blackleg resistance gene Rlm1 through bulked segregant RNA sequencing. Sci. Rep. 2019, 9, 14600. [Google Scholar] [CrossRef] [PubMed]
- Neik, T.X. Identification of a Candidate Blackleg Resistance Gene in Brassica napus and a Candidate Avirulence Gene in Leptosphaeria maculans in the B. napus—L. maculans Pathosystem. Ph.D. Thesis, The University of Western Australia, Perth, WA, Australia, 2019. [Google Scholar]
- Raman, R.; Diffey, S.; Barbulescu, D.M.; Coombes, N.; Luckett, D.; Salisbury, P.; Cowley, R.; Marcroft, S.; Raman, H. Genetic and physical mapping of loci for resistance to blackleg disease in canola (Brassica napus L.). Sci. Rep. 2020, 10, 4416. [Google Scholar] [CrossRef] [PubMed]
- Raman, H.; Raman, R.; Coombes, N.; Song, J.; Diffey, S.; Kilian, A.; Lindbeck, K.; Barbulescu, D.M.; Batley, J.; Edwards, D.; et al. Genome-wide Association Study Identifies New Loci for Resistance to Leptosphaeria maculans in Canola. Front. Plant Sci. 2016, 7, 1513. [Google Scholar] [CrossRef]
- Stotz, H.U.; Harvey, P.J.; Haddadi, P.; Mashanova, A.; Kukol, A.; Larkan, N.J.; Borhan, M.H.; Fitt, B.D.L. Genomic evidence for genes encoding leucine-rich repeat receptors linked to resistance against the eukaryotic extra- and intracellular Brassica napus pathogens Leptosphaeria maculans and Plasmodiophora brassicae. PLoS ONE 2018, 13, e0198201. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Lydiate, D.J.; Gugel, R.K.; Sharpe, A.G.; Rimmer, S.R. Introgression of Brassica rapa subsp. sylvestris blackleg resistance into B. napus. Mol. Breed 2012, 30, 1495–1506. [Google Scholar] [CrossRef]
- Becker, M.G.; Zhang, X.; Walker, P.L.; Wan, J.C.; Millar, J.L.; Khan, D.; Granger, M.J.; Cavers, J.D.; Chan, A.C.; Fernando, D.W.G.; et al. Transcriptome analysis of the Brassica napus–Leptosphaeria maculans pathosystem identifies receptor, signaling and structural genes underlying plant resistance. Plant J. 2017, 90, 573–586. [Google Scholar] [CrossRef]
- Long, Y.; Wang, Z.; Sun, Z.; Fernando, D.; McVetty, P.; Li, G. Identification of two blackleg resistance genes and fine mapping of one of these two genes in a Brassica napus canola cultivar ‘Surpass 400’. Theor. Appl. Genet. 2011, 122, 1223–1231. [Google Scholar] [CrossRef]
- Yu, F.; Gugel, R.K.; Kutcher, H.R.; Peng, G.; Rimmer, S.R. Identification and mapping of a novel blackleg resistance locus LepR4 in the progenies from Brassica napus × B. rapa subsp. sylvestris. Theor. Appl. Genet. 2012, 126, 307–315. [Google Scholar] [CrossRef]
- Ferdous, M.J.; Hossain, M.R.; Park, J.-I.; Robin, A.H.K.; Jesse, D.M.I.; Jung, H.-J.; Kim, H.-T.; Nou, I.-S. Inheritance Pattern and Molecular Markers for Resistance to Blackleg Disease in Cabbage. Plants (Basel) 2019, 8, 583. [Google Scholar] [CrossRef]
- Ferdous, M.J.; Hossain, M.R.; Park, J.-I.; Kim, H.-T.; Robin, A.H.K.; Natarajan, S.; Biswas, M.K.; Jung, H.-J.; Nou, I.-S. In silico characterization and expression of disease-resistance-related genes within the collinear region of Brassica napus blackleg resistant locus LepR1′ in B. oleracea. J. Gen. Plant Pathol. 2020, 86, 442–456. [Google Scholar] [CrossRef]
- Hossain, M.R.; Ferdous, M.J.; Park, J.-I.; Robin, A.H.K.; Natarajan, S.; Jung, H.-J.; Kim, H.-T.; Nou, I.-S. In-silico identification and differential expression of putative disease resistance-related genes within the collinear region of Brassica napus blackleg resistance locus LepR2′ in Brassica oleracea. Hortic. Environ. Biotechnol. 2020, 61, 879–890. [Google Scholar] [CrossRef]
- Ferdous, M.J.; Hossain, M.R.; Park, J.-I.; Robin, A.H.K.; Natarajan, S.; Jesse, D.M.I.; Jung, H.-J.; Kim, H.-T.; Nou, I.-S. In-silico identification and differential expressions of LepR4-syntenic disease resistance related domain containing genes against blackleg causal fungus Leptosphaeria maculans in Brassica oleracea. Gene Rep. 2020, 19, 100598. [Google Scholar] [CrossRef]
- Yang, H. Identification of Candidate Genes for Blackleg Resistance in the New Brassica juncea Canola. Ph.D. Thesis, The University of Queensland, Brisbane, Australia, 2018. [Google Scholar]
- Inturrisi, F.C. Genome-Wide Analysis of NBS-LRR Genes in Indian Mustard (Brassica juncea) and Prediction of Candidate Disease Resistance Genes. Ph.D. Thesis, The University of Western Australia, Perth, Australia, 2018. [Google Scholar]
- Flor, H.H. Current Status of the Gene-For-Gene Concept. Annu. Rev. Phytopathol. 1971, 9, 275–296. [Google Scholar] [CrossRef]
- Stakman, E. Relation between Puccinia graminis and plants highly resistant to its attack. J. Agric. Res. 1915, 4, 193–200. [Google Scholar]
- Freeman, S. Chapter 37: Plant Defense Systems. In Biological Science; Prentice Hall: Upper Saddle River, NJ, USA, 2003. [Google Scholar]
- Ansan-Melayah, D.; Balesdent, M.; Rouxel, T.; Delourme, R.; Pilet, M.; Tanguy, X.; Renard, M. Genes for race-specific resistance against blackleg disease in Brassica napus L. Plant Breed. 1998, 117, 373–378. [Google Scholar] [CrossRef]
- Larkan, N.J.; Lydiate, D.J.; Parkin, I.A.; Nelson, M.N.; Epp, D.J.; Cowling, W.A.; Rimmer, S.R.; Borhan, M.H. The Brassica napus blackleg resistance gene LepR3 encodes a receptor-like protein triggered by the Leptosphaeria maculans effector AVRLM1. New Phytol. 2013, 197, 595–605. [Google Scholar] [CrossRef]
- Chalhoub, B.; Denoeud, F.; Liu, S.; Parkin, I.A.; Tang, H.; Wang, X.; Chiquet, J.; Belcram, H.; Tong, C.; Samans, B.; et al. Plant genetics. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science 2014, 345, 950–953. [Google Scholar] [CrossRef]
- Johnson, R.D.; Lewis, B.G. Variation in host range, systemic infection and epidemiology of Leptosphaeria maculans. Plant Pathol. 1994, 43, 269–277. [Google Scholar] [CrossRef]
- Larkan, N.J.; Ma, L.; Borhan, M.H. The Brassica napus receptor-like protein RLM2 is encoded by a second allele of the LepR3/Rlm2 blackleg resistance locus. Plant Biotechnol. J. 2015, 13, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Borhan, M.H. The receptor-like kinase SOBIR1 interacts with Brassica napus LepR3 and is required for Leptosphaeria maculans AvrLm1-triggered immunity. Front. Plant Sci. 2015, 6, 933. [Google Scholar] [CrossRef] [PubMed]
- Neik, T.X.; Ghanbarnia, K.; Ollivier, B.; Scheben, A.; Severn-Ellis, A.; Larkan, N.J.; Haddadi, P.; Fernando, W.G.D.; Rouxel, T.; Batley, J.; et al. Two independent approaches converge to the cloning of a new Leptosphaeria maculans avirulence effector gene AvrLmS-Lep2. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ghanbarnia, K.; Ma, L.; Larkan, N.J.; Haddadi, P.; Fernando, W.G.D.; Borhan, M.H. Leptosphaeria maculans AvrLm9: A new player in the game of hide and seek with AvrLm4-7. Mol. Plant Pathol. 2018, 19, 1754–1764. [Google Scholar] [CrossRef] [PubMed]
- Balesdent, M.H.; Attard, A.; Kuhn, M.L.; Rouxel, T. New Avirulence Genes in the Phytopathogenic Fungus Leptosphaeria maculans. Phytopathology 2002, 92, 1122–1133. [Google Scholar] [CrossRef] [PubMed]
- Rimmer, S. Resistance genes to Leptosphaeria maculans in Brassica napus. Can. J. Plant Pathol. 2006, 28, S288–S297. [Google Scholar] [CrossRef]
- Larkan, N.J.; Ma, L.; Haddadi, P.; Buchwaldt, M.; Parkin, I.A.P.; Djavaheri, M.; Borhan, M.H. The Brassica napus Wall-Associated Kinase-Like (WAKL) gene Rlm9 provides race-specific blackleg resistance. Plant J. 2020, n/a. [Google Scholar] [CrossRef]
- Parlange, F.; Daverdin, G.; Fudal, I.; Kuhn, M.L.; Balesdent, M.H.; Blaise, F.; Grezes-Besset, B.; Rouxel, T. Leptosphaeria maculans avirulence gene AvrLm4-7 confers a dual recognition specificity by the Rlm4 and Rlm7 resistance genes of oilseed rape, and circumvents Rlm4-mediated recognition through a single amino acid change. Mol. Microbiol. 2009, 71, 851–863. [Google Scholar] [CrossRef]
- Delourme, R.; Pilet-Nayel, M.L.; Archipiano, M.; Horvais, R.; Tanguy, X.; Rouxel, T.; Brun, H.; Renard, M.; Balesdent, M.H. A Cluster of Major Specific Resistance Genes to Leptosphaeria maculans in Brassica napus. Phytopathology 2004, 94, 578–583. [Google Scholar] [CrossRef]
- Van de Wouw, A.P.; Howlett, B.J. Advances in understanding the Leptosphaeria maculans—Brassica pathosystem and their impact on disease management. Can. J. Plant Pathol. 2019, 1–15. [Google Scholar] [CrossRef]
- Wang, X.; Wang, H.; Wang, J.; Sun, R.; Wu, J.; Liu, S.; Bai, Y.; Mun, J.H.; Bancroft, I.; Cheng, F.; et al. The genome of the mesopolyploid crop species Brassica rapa. Nat. Genet. 2011, 43, 1035–1039. [Google Scholar] [CrossRef]
- Robin, A.; Larkan, N.; Laila, R.; Park, J.-I.; Ahmed, N.; Borhan, H.; Parkin, I.; Nou, I.-S. Korean Brassica oleracea germplasm offers a novel source of qualitative resistance to blackleg disease. Eur. J. Plant Pathol. 2017, 149, 611–623. [Google Scholar] [CrossRef]
- Eber, F.; Lourgant, K.; Brun, H.; Lode, M.; Huteau, V.; Coriton, O.; Alix, K.; Balesdent, M.; Chevre, A.M. Analysis of Brassica nigra Chromosomes Allows Identification of a New Effective Leptosphaeria maculans resistance Gene Introgressed in Brassica napus. In Proceedings of the 13th International Rapeseed Congress, Prague, Czech Republic, 5–9 June 2011. [Google Scholar]
- Petit-Houdenot, Y.; Degrave, A.; Meyer, M.; Blaise, F.; Ollivier, B.; Marais, C.-L.; Jauneau, A.; Audran, C.; Rivas, S.; Veneault-Fourrey, C.; et al. A two genes—for—one gene interaction between Leptosphaeria maculans and Brassica napus. New Phytol. 2019, 223, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Balesdent, M.H.; Fudal, I.; Ollivier, B.; Bally, P.; Grandaubert, J.; Eber, F.; Chevre, A.M.; Leflon, M.; Rouxel, T. The dispensable chromosome of Leptosphaeria maculans shelters an effector gene conferring avirulence towards Brassica rapa. New Phytol. 2013, 198, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Lydiate, D.J.; Rimmer, S.R. Identification of two novel genes for blackleg resistance in Brassica napus. Theor. Appl. Genet. 2005, 110, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Xu, W.; Hirani, A.H.; Liu, Z.; Tuan, P.A.; Ayele, B.T.; Daayf, F.; McVetty, P.B.E.; Duncan, R.W.; Li, G. Transcriptional Insight Into Brassica napus Resistance Genes LepR3 and Rlm2-Mediated Defense Response Against the Leptosphaeria maculans Infection. Front. Plant Sci. 2019, 10, 823. [Google Scholar] [CrossRef]
- Dusabenyagasani, M.; Fernando, D. Development of a SCAR Marker to Track Canola Resistance against Blackleg Caused by Leptosphaeria maculans Pathogenicity Group 3. Plant Dis. 2008, 92, 903–908. [Google Scholar] [CrossRef]
- Larkan, N.; Lydiate, D.; Yu, F.; Rimmer, S.; Borhan, H. Co-localisation of the blackleg resistance genes Rlm2 and LepR3 on Brassica napus chromosome A10. BMC Plant Biol. 2014, 14, 1595. [Google Scholar] [CrossRef]
- Raman, R.; Taylor, B.; Marcroft, S.; Stiller, J.; Eckermann, P.; Coombes, N.; Rehman, A.; Lindbeck, K.; Luckett, D.; Wratten, N.; et al. Molecular mapping of qualitative and quantitative loci for resistance to Leptosphaeria maculans causing blackleg disease in canola (Brassica napus L.). Theor. Appl. Genet. 2012, 125, 405–418. [Google Scholar] [CrossRef]
- Rahman, H. Review: Breeding spring canola (Brassica napus L.) by the use of exotic germplasm. Can. J. Plant Sci. 2013, 93, 363–373. [Google Scholar] [CrossRef]
- Li, C.X.; Cowling, W.A. Identification of a single dominant allele for resistance to blackleg in Brassica napus ’Surpass 400’. Plant Breed 2003, 122, 485–488. [Google Scholar] [CrossRef]
- Chen, C.Y.; Séguin-Swartz, G. A comparative study of the response of wild crucifers to the blackleg fungus, Phoma lingam. Can. J. Plant Pathol. 1997, 19, 107. [Google Scholar]
- Chen, C.Y.; Séguin-Swartz, G. Reaction of wild crucifers to Leptosphaeria maculans, the causal agent of blackleg of crucifers. Can. J. Plant Pathol. 1999, 21, 361–367. [Google Scholar] [CrossRef]
- Gugel, R.; Séguin-Swartz, G.; Warwick, S.I. Transfer of blackleg resistance from Erucastrum gallicum to Brassica rapa. In Proceedings of the 67th Annual Meetings of Canadian Phytopathological Society, Saskatoon, SK, Canada, 23–26 June 1996. [Google Scholar]
- Gugel, R.K.; Séguin-Swartz, G. Introgression of blackleg resistance from Sinapis alba into Brassica napus. In Proceedings of the Brassica 97, Int Soc Hortic Sci Symp Brassicas/10th Crucifer Genetics Workshop, Rennes, France, 23–27 September 1997; p. 222. [Google Scholar]
- Li, H.; Barbetti, M.J.; Sivasithamparam, K. Hazard from reliance on cruciferous hosts as sources of major gene-based resistance for managing blackleg (Leptosphaeria maculans) disease. Field Crops Res. 2005, 91, 185–198. [Google Scholar] [CrossRef]
- Mithen, R.F.; Lewis, B.G.; Heaney, R.K.; Fenwick, G.R. Resistance of leaves of Brassica species to Leptosphaeria maculans. Trans. Br. Mycol. Soc. 1987, 88, 525–531. [Google Scholar] [CrossRef]
- Mithen, R.F.; Magrath, R. Glucosinolates and Resistance to Leptosphaeria maculans in Wild and Cultivated Brassica Species. Plant Breed 1992, 108, 60–68. [Google Scholar] [CrossRef]
- Pedras, M.S.; Chumala, P.B.; Suchy, M. Phytoalexins from Thlaspi arvense, a wild crucifer resistant to virulent Leptosphaeria maculans: Structures, syntheses and antifungal activity. Phytochemistry 2003, 64, 949–956. [Google Scholar] [CrossRef]
- Plümper, B. Somatische und sexuelle Hybridisierung für den Transfer von Krankheitsresistenzen auf Brassica napus L. Ph.D. Thesis, Free University of Berlin, Berlin, Germany, 1995. [Google Scholar]
- Tewari, J.P.; Bansal, V.K.; Tewari, I.; Gómez-Campo, C.; Stringam, G.R.; Thiagarajah, M.R. Reactions of some wild and cultivated accessions of Eruca against Leptosphaeria maculans. Crucif. Newsl. Eucarpia 1996, 18, 130–131. [Google Scholar]
- Winter, H. Untersuchungen zur Introgression von Resistenzen gegen die Wurzelhals- und Stengelfäule [Leptosphaeria maculans (Desm.) Ces. et De Not.] aus Verwandten Arten in den Raps (Brassica napus L.): Examinations on the Introgression of Resistances to Blackleg [Leptosphaeria maculans (Desm.) Ces. et De Not.] into oilseed rape (Brassica napus L.) from Related Species. Ph.D. Thesis, Freie Universität Berlin Universitätsbibliothek, Berlin, Germany, 2004. [Google Scholar]
- Winter, H.; Diestel, A.; Gärtig, S.; Krone, N.; Sterenberg, K.; Sacristán, M.D. Transfer of new blackleg resistances into oilseed rape. In Proceedings of the GCIRC 11th Int. Rapeseed Congress, Copenhagen, Denmark, 6–10 July 2003; pp. 19–21. [Google Scholar]
- Winter, H.; Gaertig, S.; Diestel, A.; Sacristán, M.D. Blackleg resistance of different origin transferred into Brassica napus. In Proceedings of the GCIRC 10th Int Rapeseed Congress, Canberra, Australia, 26–29 September 1999. [Google Scholar]
- Alamery, S.; Tirnaz, S.; Bayer, P.; Tollenaere, R.; Chaloub, B.; Edwards, D.; Batley, J. Genome-wide identification and comparative analysis of NBS-LRR resistance genes in Brassica napus. Crop Pasture Sci. 2017, 69, 79–93. [Google Scholar] [CrossRef]
- Fu, Y.; Zhang, Y.; Mason, A.S.; Lin, B.; Zhang, D.; Yu, H.; Fu, D. NBS-Encoding Genes in Brassica napus Evolved Rapidly After Allopolyploidization and Co-localize With Known Disease Resistance Loci. Front. Plant Sci. 2019, 10, 26. [Google Scholar] [CrossRef]
- Hofberger, J.A.; Zhou, B.; Tang, H.; Jones, J.D.G.; Schranz, M.E. A novel approach for multi-domain and multi-gene family identification provides insights into evolutionary dynamics of disease resistance genes in core eudicot plants. BMC Genom. 2014, 15, 966. [Google Scholar] [CrossRef]
- Li, P.; Quan, X.; Jia, G.; Xiao, J.; Cloutier, S.; You, F.M. RGAugury: A pipeline for genome-wide prediction of resistance gene analogs (RGAs) in plants. BMC Genom. 2016, 17, 852. [Google Scholar] [CrossRef]
- Liu, Z.; Xie, J.; Wang, H.; Zhong, X.; Li, H.; Yu, J.; Kang, J. Identification and expression profiling analysis of NBS–LRR genes involved in Fusarium oxysporum f.sp. conglutinans resistance in cabbage. 3 Biotech 2019, 9, 202. [Google Scholar] [CrossRef]
- Tirnaz, S.; Bayer, P.; Inturrisi, F.; Neik, T.; Yang, H.; Dolatabadian, A.; Zhang, F.; Severn-Ellis, A.; Patel, D.; Pradhan, A.; et al. Resistance gene analogs in the Brassicaceae: Identification, characterization, distribution and evolution. Plant Physiol. 2020, 184, 909–922. [Google Scholar] [CrossRef]
- Toda, N.; Rustenholz, C.; Baud, A.; Le Paslier, M.-C.; Amselem, J.; Merdinoglu, D.; Faivre-Rampant, P. NLGenomeSweeper: A Tool for Genome-Wide NBS-LRR Resistance Gene Identification. Genes 2020, 11, 333. [Google Scholar] [CrossRef]
- Yu, J.; Tehrim, S.; Zhang, F.; Tong, C.; Huang, J.; Cheng, X.; Dong, C.; Zhou, Y.; Qin, R.; Hua, W.; et al. Genome-wide comparative analysis of NBS-encoding genes between Brassica species and Arabidopsis thaliana. BMC Genom. 2014, 15, 3. [Google Scholar] [CrossRef]
- Zhang, Y.-M.; Shao, Z.-Q.; Wang, Q.; Hang, Y.-Y.; Xue, J.-Y.; Wang, B.; Chen, J.-Q. Uncovering the dynamic evolution of nucleotide-binding site-leucine-rich repeat (NBS-LRR) genes in Brassicaceae. J. Integr. Plant Biol. 2015. [Google Scholar] [CrossRef]
- Chëvre, A.M.; dePonce Leon, A.; Jenczewski, E.; Eber, F.; Delourme, R.; Renard, M.; Brun, H. Introduction of blackleg resistance from Brassica rapa into Brassica napus. In Proceedings of the 11th International Rapeseed Congress, Copenhagen, Denmark, 6–10 July 2003; pp. 32–35. [Google Scholar]
- Fredua-Agyeman, R.; Coriton, O.; Huteau, V.; Parkin, I.A.P.; Chèvre, A.-M.; Rahman, H. Molecular cytogenetic identification of B genome chromosomes linked to blackleg disease resistance in Brassica napus × B. carinata interspecific hybrids. Theor. Appl. Genet. 2014, 127, 1305–1318. [Google Scholar] [CrossRef]
- Navabi, Z.K.; Parkin, I.A.; Pires, J.C.; Xiong, Z.; Thiagarajah, M.R.; Good, A.G.; Rahman, M.H. Introgression of B-genome chromosomes in a doubled haploid population of Brassica napus × B-carinata. Genome 2010, 53, 619–629. [Google Scholar] [CrossRef]
- Navabi, Z.K.; Stead, K.E.; Pires, J.C.; Xiong, Z.; Sharpe, A.G.; Parkin, I.A.P.; Rahman, M.H.; Good, A.G. Analysis of B-genome chromosome introgression in interspecific hybrids of Brassica napus × B. carinata. Genetics 2011, 187, 659–673. [Google Scholar] [CrossRef][Green Version]
- Chèvre, A.M.; Brun, H.; Eber, F.; Letanneur, J.C.; Vallee, P.; Ermel, M.; Glais, I.; Hua, L.; Sivasithamparam, K.; Barbetti, M.J. Stabilization of Resistance to Leptosphaeria maculans in Brassica napus—B. juncea Recombinant Lines and Its Introgression into Spring-Type Brassica napus. Plant Dis. 2008, 92, 1208–1214. [Google Scholar] [CrossRef]
- Eber, F.; Delourme, R.; Barret, P.; Lourgant, K.; Brun, H.; Renard, M.; Chevre, A.M. Characterisation and efficiency of mustard blackleg resistance genes introgressed into oilseed rape. In Proceedings of the 10th International Rapeseed Congress, Canberra, Australia, 26–29 September 1999. [Google Scholar]
- Saal, B.; Brun, H.; Glais, I.; Struss, D. Identification of a Brassica juncea-derived recessive gene conferring resistance to Leptosphaeria maculans in oilseed rape. Plant Breed 2004, 123, 505–511. [Google Scholar] [CrossRef]
- Gaebelein, R.; Alnajar, D.; Koopmann, B.; Mason, A.S. Hybrids between Brassica napus and B. nigra show frequent pairing between the B and A/C genomes and resistance to blackleg. Chromosome Res. 2019, 27, 221–236. [Google Scholar] [CrossRef] [PubMed]
- Gaebelein, R.; Alnajar, D.; Mason, A. Brassica napus × Brassica nigra hybrids for blackleg resistance introgression in rapeseed breeding. In Proceedings of the German Plant Breeding Conference, Wernigerode, Deutschland, 28 February–2 March 2018. [Google Scholar]
- Hu, Q.; Andersen, S.; Dixelius, C.; Hansen, L. Production of fertile intergeneric somatic hybrids between Brassica napus and Sinapis arvensis for the enrichment of the rapeseed gene pool. Plant Cell Rep. 2002, 21, 147–152. [Google Scholar] [CrossRef]
- Snowdon, R.; Winter, H.; Diestel, A.; Sacristán, M. Development and characterisation of Brassica napus-Sinapis arvensis addition lines exhibiting resistance to Leptosphaeria maculans. Theor. Appl. Genet. 2000, 101, 1008–1014. [Google Scholar] [CrossRef]
- Liu, J.-H.; Landgren, M.; Glimelius, K. Transfer of the Brassica tournefortii cytoplasm to B. napus for the production of cytoplasmic male sterile B. napus. Physiol. Plant 1996, 96, 123–129. [Google Scholar] [CrossRef]
- Goff, K.E.; Ramonell, K.M. The role and regulation of receptor-like kinases in plant defense. Gene Regul. Syst. Biol. 2007, 1, 167–175. [Google Scholar] [CrossRef]
- Belkhadir, Y.; Chory, J. Brassinosteroid Signaling: A Paradigm for Steroid Hormone Signaling from the Cell Surface. Science 2006, 314, 1410. [Google Scholar] [CrossRef]
- Ogawa, M.; Shinohara, H.; Sakagami, Y.; Matsubayashi, Y. Arabidopsis CLV3 peptide directly binds CLV1 ectodomain. Science 2008, 319, 294. [Google Scholar] [CrossRef]
- Torii, K.U. Mix-and-match: Ligand–receptor pairs in stomatal development and beyond. Trends Plant Sci. 2012, 17, 711–719. [Google Scholar] [CrossRef]
- Ade, J.; DeYoung, B.J.; Golstein, C.; Innes, R.W. Indirect activation of a plant nucleotide binding site–leucine-rich repeat protein by a bacterial protease. Proc. Natl. Acad. Sci. USA 2007, 104, 2531. [Google Scholar] [CrossRef]
- Albert, I.; Böhm, H.; Albert, M.; Feiler, C.E.; Imkampe, J.; Wallmeroth, N.; Brancato, C.; Raaymakers, T.M.; Oome, S.; Zhang, H.; et al. An RLP23–SOBIR1–BAK1 complex mediates NLP-triggered immunity. Nat. Plants 2015, 1, 15140. [Google Scholar] [CrossRef]
- Albert, I.; Zhang, L.; Bemm, H.; Nürnberger, T. Structure-Function Analysis of Immune Receptor AtRLP23 with Its Ligand nlp20 and Coreceptors AtSOBIR1 and AtBAK1. Mol. Plant Microbe Interact. 2019, 32, 1038–1046. [Google Scholar] [CrossRef]
- Liu, J.; Elmore, J.M.; Lin, Z.-J.D.; Coaker, G. A Receptor-like Cytoplasmic Kinase Phosphorylates the Host Target RIN4, Leading to the Activation of a Plant Innate Immune Receptor. Cell Host Microbe 2011, 9, 137–146. [Google Scholar] [CrossRef]
- Mackey, D.; Holt, B.F.; Wiig, A.; Dangl, J.L. RIN4 Interacts with Pseudomonas syringae Type III Effector Molecules and Is Required for RPM1-Mediated Resistance in Arabidopsis. Cell 2002, 108, 743–754. [Google Scholar] [CrossRef]
- Shao, F.; Golstein, C.; Ade, J.; Stoutemyer, M.; Dixon, J.E.; Innes, R.W. Cleavage of Arabidopsis PBS1 by a Bacterial Type III Effector. Science 2003, 301, 1230. [Google Scholar] [CrossRef]
- Swiderski, M.R.; Innes, R.W. The Arabidopsis PBS1 resistance gene encodes a member of a novel protein kinase subfamily. Plant J. 2001, 26, 101–112. [Google Scholar] [CrossRef]
- van der Burgh, A.M.; Postma, J.; Robatzek, S.; Joosten, M.H.A.J. Kinase activity of SOBIR1 and BAK1 is required for immune signalling. Mol. Plant Pathol. 2019, 20, 410–422. [Google Scholar] [CrossRef]
- Shiu, S.-H.; Bleecker, A.B. Receptor-like kinases from Arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc. Natl. Acad. Sci. USA 2001, 98, 10763. [Google Scholar] [CrossRef]
- Kourelis, J.; van der Hoorn, R.A.L. Defended to the Nines: 25 Years of Resistance Gene Cloning Identifies Nine Mechanisms for R Protein Function. Plant Cell 2018, 30, 285. [Google Scholar] [CrossRef]
- Caplan, J.L.; Mamillapalli, P.; Burch-Smith, T.M.; Czymmek, K.; Dinesh-Kumar, S.P. Chloroplastic protein NRIP1 mediates innate immune receptor recognition of a viral effector. Cell 2008, 132, 449–462. [Google Scholar] [CrossRef]
- Lewis, J.D.; Lee, A.H.; Hassan, J.A.; Wan, J.; Hurley, B.; Jhingree, J.R.; Wang, P.W.; Lo, T.; Youn, J.Y.; Guttman, D.S.; et al. The Arabidopsis ZED1 pseudokinase is required for ZAR1-mediated immunity induced by the Pseudomonas syringae type III effector HopZ1a. Proc. Natl. Acad. Sci. USA 2013, 110, 18722–18727. [Google Scholar] [CrossRef]
- Bernoux, M.; Ellis, J.G.; Dodds, P.N. New insights in plant immunity signaling activation. Curr. Opin. Plant Biol. 2011, 14, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, K.; Suwabe, K.; Tomita, R.N.; Kato, T.; Nunome, T.; Fukuoka, H.; Matsumoto, S. Identification and Characterization of Crr1a, a Gene for Resistance to Clubroot Disease (Plasmodiophora brassicae Woronin) in Brassica rapa L. PLoS ONE 2013, 8, e54745. [Google Scholar] [CrossRef] [PubMed]
- Ueno, H.; Matsumoto, E.; Aruga, D.; Kitagawa, S.; Matsumura, H.; Hayashida, N. Molecular characterization of the CRa gene conferring clubroot resistance in Brassica rapa. Plant Mol. Biol. 2012, 80, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, C.; Oldroyd, G.E. Plant signalling in symbiosis and immunity. Nature 2017, 543, 328–336. [Google Scholar] [CrossRef]
- Roy, N.N. Interspecific transfer of Brassica juncea-type high blackleg resistance to Brassica napus. Euphytica 1984, 33, 295–303. [Google Scholar] [CrossRef]
- Schelfhout, C.J.; Snowdon, R.; Cowling, W.A.; Wroth, J.M. Tracing B-genome chromatin in Brassica napus × B. juncea interspecific progeny. Genome 2006, 49, 1490–1497. [Google Scholar] [CrossRef]
- Perumal, S.; Koh, C.S.; Jin, L.; Buchwaldt, M.; Higgins, E.E.; Zheng, C.; Sankoff, D.; Robinson, S.J.; Kagale, S.; Navabi, Z.-K.; et al. A high-contiguity Brassica nigra genome localizes active centromeres and defines the ancestral Brassica genome. Nat. Plants 2020, 6, 929–941. [Google Scholar] [CrossRef]
- Khedikar, Y.; Clarke, W.E.; Chen, L.; Higgins, E.E.; Kagale, S.; Koh, C.S.; Bennett, R.; Parkin, I.A.P. Narrow genetic base shapes population structure and linkage disequilibrium in an industrial oilseed crop, Brassica carinata A. Braun. Sci. Rep. 2020, 10, 12629. [Google Scholar] [CrossRef]
- Cheng, C.Y.; Krishnakumar, V.; Chan, A.P.; Thibaud-Nissen, F.; Schobel, S.; Town, C.D. Araport11: A complete reannotation of the Arabidopsis thaliana reference genome. Plant J. 2017, 89, 789–804. [Google Scholar] [CrossRef]
- Peele, H.M.; Guan, N.; Fogelqvist, J.; Dixelius, C. Loss and retention of resistance genes in five species of the Brassicaceae family. BMC Plant Biol. 2014, 14, 298. [Google Scholar] [CrossRef] [PubMed]
- Staal, J.; Kaliff, M.; Bohman, S.; Dixelius, C. Transgressive segregation reveals two Arabidopsis TIR-NB-LRR resistance genes effective against Leptosphaeria maculans, causal agent of blackleg disease. Plant J. 2006, 46, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Bohman, S.; Staal, J.; Thomma, B.P.; Wang, M.; Dixelius, C. Characterisation of an Arabidopsis-Leptosphaeria maculans pathosystem: Resistance partially requires camalexin biosynthesis and is independent of salicylic acid, ethylene and jasmonic acid signalling. Plant J. 2004, 37, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Persson, M.; Staal, J.; Oide, S.; Dixelius, C. Layers of Defense Responses to Leptosphaeria maculans below the RLM1- and Camalexin-Dependent Resistances. New Phytol. 2009, 182, 470–482. [Google Scholar] [CrossRef]
- Staal, J.; Kaliff, M.; Dewaele, E.; Persson, M.; Dixelius, C. RLM3, a TIR domain encoding gene involved in broad-range immunity of Arabidopsis to necrotrophic fungal pathogens. Plant J. 2008, 55, 188–200. [Google Scholar] [CrossRef]
- Peele, H.M. Defense Gene Responses toward Necrotrophic Fungi in Arabidopsis Thaliana; Swedish University of Agricultural Sciences: Uppsala, Sweden, 2015. [Google Scholar]
- Cavaiuolo, M.; Cocetta, G.; Spadafora, N.D.; Müller, C.T.; Rogers, H.J.; Ferrante, A. Gene expression analysis of rocket salad under pre-harvest and postharvest stresses: A transcriptomic resource for Diplotaxis tenuifolia. PLoS ONE 2017, 12, e0178119. [Google Scholar] [CrossRef]
- Kumari, P.; Singh, K.P.; Rai, P.K. Draft genome of multiple resistance donor plant Sinapis alba: An insight into SSRs, annotations and phylogenetics. PLoS ONE 2020, 15, e0231002. [Google Scholar] [CrossRef]
- Liu, J.; Mei, D.; Li, Y.; Huang, S.; Hu, Q. Deep RNA-Seq to unlock the gene bank of floral development in Sinapis arvensis. PLoS ONE 2014, 9, e105775. [Google Scholar] [CrossRef]
- Cai, C.; Wang, X.; Liu, B.; Wu, J.; Liang, J.; Cui, Y.; Cheng, F.; Wang, X. Brassica rapa Genome 2.0: A Reference Upgrade through Sequence Re-assembly and Gene Re-annotation. Mol. Plant 2017, 10, 649–651. [Google Scholar] [CrossRef]
- Parkin, I.A.P.; Koh, C.; Tang, H.; Robinson, S.J.; Kagale, S.; Clarke, W.E.; Town, C.D.; Nixon, J.; Krishnakumar, V.; Bidwell, S.L.; et al. Transcriptome and methylome profiling reveals relics of genome dominance in the mesopolyploid Brassica oleracea. Genome Biol. 2014, 15, R77. [Google Scholar] [CrossRef]
- Bayer, P.E.; Golicz, A.A.; Scheben, A.; Batley, J.; Edwards, D. Plant pan-genomes are the new reference. Nat. Plants 2020, 6, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Danilevicz, M.F.; Tay Fernandez, C.G.; Marsh, J.I.; Bayer, P.E.; Edwards, D. Plant pangenomics: Approaches, applications and advancements. Curr. Opin. Plant Biol. 2020, 54, 18–25. [Google Scholar] [CrossRef]
- Golicz, A.A.; Bayer, P.E.; Bhalla, P.L.; Batley, J.; Edwards, D. Pangenomics Comes of Age: From Bacteria to Plant and Animal Applications. Trends Genet. 2020, 36, 132–145. [Google Scholar] [CrossRef]
- Montenegro, J.D.; Golicz, A.A.; Bayer, P.E.; Hurgobin, B.; Lee, H.; Chan, C.-K.K.; Visendi, P.; Lai, K.; Doležel, J.; Batley, J.; et al. The pangenome of hexaploid bread wheat. Plant J. 2017, 90, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Golicz, A.A.; Lu, K.; Dossa, K.; Zhang, Y.; Chen, J.; Wang, L.; You, J.; Fan, D.; Edwards, D.; et al. Insight into the evolution and functional characteristics of the pan-genome assembly from sesame landraces and modern cultivars. Plant Biotechnol. J. 2019, 17, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-M.; Guan, Z.; Hu, J.; Guo, C.; Yang, Z.; Wang, S.; Liu, D.; Wang, B.; Lu, S.; Zhou, R.; et al. Eight high-quality genomes reveal pan-genome architecture and ecotype differentiation of Brassica napus. Nat. Plants 2020, 6, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Aylward, J.; Steenkamp, E.T.; Dreyer, L.L.; Roets, F.; Wingfield, B.D.; Wingfield, M.J. A plant pathology perspective of fungal genome sequencing. IMA Fungus 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Fudal, I.; Ross, S.; Gout, L.; Blaise, F.; Kuhn, M.L.; Eckert, M.R.; Cattolico, L.; Bernard-Samain, S.; Balesdent, M.H.; Rouxel, T. Heterochromatin-like regions as ecological niches for avirulence genes in the Leptosphaeria maculans genome: Map-based cloning of AvrLm6. Mol. Plant Microbe Interact. 2007, 20, 459–470. [Google Scholar] [CrossRef]
- Ghanbarnia, K.; Fudal, I.; Larkan, N.J.; Links, M.G.; Balesdent, M.H.; Profotova, B.; Fernando, W.G.; Rouxel, T.; Borhan, M.H. Rapid identification of the Leptosphaeria maculans avirulence gene AvrLm2 using an intraspecific comparative genomics approach. Mol. Plant Pathol. 2015, 16, 699–709. [Google Scholar] [CrossRef]
- Plissonneau, C.; Daverdin, G.; Ollivier, B.; Blaise, F.; Degrave, A.; Fudal, I.; Rouxel, T.; Balesdent, M.H. A game of hide and seek between avirulence genes AvrLm4-7 and AvrLm3 in Leptosphaeria maculans. New Phytol. 2016, 209, 1613–1624. [Google Scholar] [CrossRef]
- Van de Wouw, A.P.; Lowe, R.G.; Elliott, C.E.; Dubois, D.J.; Howlett, B.J. An avirulence gene, AvrLmJ1, from the blackleg fungus, Leptosphaeria maculans, confers avirulence to Brassica juncea cultivars. Mol. Plant Pathol. 2014, 15, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Plissonneau, C.; Rouxel, T.; Chevre, A.M.; Van De Wouw, A.P.; Balesdent, M.H. One gene-one name: The AvrLmJ1 avirulence gene of Leptosphaeria maculans is AvrLm5. Mol. Plant Pathol. 2018, 19, 1012–1016. [Google Scholar] [CrossRef] [PubMed]
- Gout, L.; Fudal, I.; Kuhn, M.L.; Blaise, F.; Eckert, M.; Cattolico, L.; Balesdent, M.H.; Rouxel, T. Lost in the middle of nowhere: The AvrLm1 avirulence gene of the Dothideomycete Leptosphaeria maculans. Mol. Microbiol. 2006, 60, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Rouxel, T.; Grandaubert, J.; James, K.H.; Claire, H.; Angela, P.V.D.W.; Arnaud, C.; Victoria, D.; Véronique, A.; Pascal, B.; Salim, B.; et al. Effector diversification within compartments of the Leptosphaeria maculans genome affected by Repeat-Induced Point mutations. Nat. Commun. 2011, 2, 202. [Google Scholar] [CrossRef] [PubMed]
- Daverdin, G.; Rouxel, T.; Gout, L.; Aubertot, J.-N.; Fudal, I.; Meyer, M.; Parlange, F.; Carpezat, J.; Balesdent, M.-H. Genome Structure and Reproductive Behaviour Influence the Evolutionary Potential of a Fungal Phytopathogen. PLoS Pathog. 2012, 8, e1003020. [Google Scholar] [CrossRef]
- Fudal, I.; Ross, S.; Brun, H.; Besnard, A.L.; Ermel, M.; Kuhn, M.L.; Balesdent, M.H.; Rouxel, T. Repeat-induced point mutation (RIP) as an alternative mechanism of evolution toward virulence in Leptosphaeria maculans. Mol. Plant Microbe Interact. 2009, 22, 932–941. [Google Scholar] [CrossRef]
- Gout, L.; Kuhn, M.L.; Vincenot, L.; Bernard-Samain, S.; Cattolico, L.; Barbetti, M.; Moreno-Rico, O.; Balesdent, M.-H.; Rouxel, T. Genome structure impacts molecular evolution at the AvrLm1 avirulence locus of the plant pathogen Leptosphaeria maculans. Environ. Microbiol. 2007, 9, 2978–2992. [Google Scholar] [CrossRef]
- Novakova, M.; Sasek, V.; Trda, L.; Krutinova, H.; Mongin, T.; Valentova, O.; Balesdent, M.H.; Rouxel, T.; Burketova, L. Leptosphaeria maculans effector AvrLm4-7 affects salicylic acid (SA) and ethylene (ET) signalling and hydrogen peroxide (H2O2) accumulation in Brassica napus. Mol. Plant Pathol. 2016, 17, 818–831. [Google Scholar] [CrossRef]
- Ma, L.; Djavaheri, M.; Wang, H.; Larkan, N.J.; Haddadi, P.; Beynon, E.; Gropp, G.; Borhan, M.H. Leptosphaeria maculans Effector Protein AvrLm1 Modulates Plant Immunity by Enhancing MAP Kinase 9 Phosphorylation. iScience 2018, 3, 177–191. [Google Scholar] [CrossRef]
- Becker, M.G.; Haddadi, P.; Wan, J.; Adam, L.; Walker, P.; Larkan, N.J.; Daayf, F.; Borhan, M.H.; Belmonte, M.F. Transcriptome Analysis of Rlm2-Mediated Host Immunity in the Brassica napus-Leptosphaeria maculans Pathosystem. Mol. Plant Microbe Interact. 2019, 32, 1001–1012. [Google Scholar] [CrossRef]
- Schweighofer, A.; Kazanaviciute, V.; Scheikl, E.; Teige, M.; Doczi, R.; Hirt, H.; Schwanninger, M.; Kant, M.; Schuurink, R.; Mauch, F.; et al. The PP2C-type phosphatase AP2C1, which negatively regulates MPK4 and MPK6, modulates innate immunity, jasmonic acid, and ethylene levels in Arabidopsis. Plant Cell 2007, 19, 2213–2224. [Google Scholar] [CrossRef] [PubMed]
- Teper, D.; Sunitha, S.; Martin, G.B.; Sessa, G. Five Xanthomonas type III effectors suppress cell death induced by components of immunity-associated MAP kinase cascades. Plant Signal. Behav. 2015, 10, e1064573. [Google Scholar] [CrossRef] [PubMed]
- Trdá, L.; Barešová, M.; Šašek, V.; Nováková, M.; Zahajská, L.; Dobrev, P.I.; Motyka, V.; Burketová, L. Cytokinin Metabolism of Pathogenic Fungus Leptosphaeria maculans Involves Isopentenyltransferase, Adenosine Kinase and Cytokinin Oxidase/Dehydrogenase. Front. Microbiol. 2017, 8, 1374. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Zhang, H.; Strelkov, S.E.; Hwang, S.-F. The LmSNF1 Gene Is Required for Pathogenicity in the Canola Blackleg Pathogen Leptosphaeria maculans. PLoS ONE 2014, 9, e92503. [Google Scholar] [CrossRef] [PubMed]
- Haddadi, P.; Ma, L.; Wang, H.; Borhan, M.H. Genome-wide transcriptomic analyses provide insights into the lifestyle transition and effector repertoire of Leptosphaeria maculans during the colonization of Brassica napus seedlings. Mol. Plant Pathol. 2016, 17, 1196–1210. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.G.; Cassin, A.; Grandaubert, J.; Clark, B.L.; Van de Wouw, A.P.; Rouxel, T.; Howlett, B.J. Genomes and transcriptomes of partners in plant-fungal-interactions between canola (Brassica napus) and two Leptosphaeria species. PLoS ONE 2014, 9, e103098. [Google Scholar] [CrossRef]
- Sonah, H.; Zhang, X.; Deshmukh, R.K.; Borhan, M.H.; Fernando, W.G.D.; Bélanger, R.R. Comparative Transcriptomic Analysis of Virulence Factors in Leptosphaeria maculans during Compatible and Incompatible Interactions with Canola. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Soyer, J.L.; Hamiot, A.; Ollivier, B.; Balesdent, M.H.; Rouxel, T.; Fudal, I. The APSES transcription factor LmStuA is required for sporulation, pathogenic development and effector gene expression in Leptosphaeria maculans. Mol. Plant Pathol. 2015, 16, 1000–1005. [Google Scholar] [CrossRef]
- Idnurm, A.; Howlett, B.J. Isocitrate lyase is essential for pathogenicity of the fungus Leptosphaeria maculans to canola (Brassica napus). Eukaryot. Cell 2002, 1, 719–724. [Google Scholar] [CrossRef]
- Kaczmarek, J.; Latunde-Dada, A.O.; Irzykowski, W.; Cools, H.J.; Stonard, J.F.; Brachaczek, A.; Jedryczka, M. Molecular screening for avirulence alleles AvrLm1 and AvrLm6 in airborne inoculum of Leptosphaeria maculans and winter oilseed rape (Brassica napus) plants from Poland and the UK. J. Appl. Genet. 2014, 55, 529–539. [Google Scholar] [CrossRef]
- Singh, K.; Zouhar, M.; Mazakova, J.; Rysanek, P. Genome wide identification of the immunophilin gene family in Leptosphaeria maculans: A causal agent of Blackleg disease in Oilseed Rape (Brassica napus). OMICS 2014, 18, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Gervais, J.; Plissonneau, C.; Linglin, J.; Meyer, M.; Labadie, K.; Cruaud, C.; Fudal, I.; Rouxel, T.; Balesdent, M.H. Different waves of effector genes with contrasted genomic location are expressed by Leptosphaeria maculans during cotyledon and stem colonization of oilseed rape. Mol. Plant Pathol. 2017, 18, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Sasek, V.; Novakova, M.; Jindrichova, B.; Boka, K.; Valentova, O.; Burketova, L. Recognition of avirulence gene AvrLm1 from hemibiotrophic ascomycete Leptosphaeria maculans triggers salicylic acid and ethylene signaling in Brassica napus. Mol. Plant Microbe Interact. 2012, 25, 1238–1250. [Google Scholar] [CrossRef]
- Dubiella, U.; Seybold, H.; Durian, G.; Komander, E.; Lassig, R.; Witte, C.P.; Schulze, W.X.; Romeis, T. Calcium-dependent protein kinase/NADPH oxidase activation circuit is required for rapid defense signal propagation. Proc. Natl. Acad. Sci. USA 2013, 110, 8744–8749. [Google Scholar] [CrossRef]
- Gravino, M.; Savatin, D.V.; Macone, A.; De Lorenzo, G. Ethylene production in Botrytis cinerea- and oligogalacturonide-induced immunity requires calcium-dependent protein kinases. Plant J. 2015, 84, 1073–1086. [Google Scholar] [CrossRef]
- Noman, A.; Liu, Z.; Aqeel, M.; Zainab, M.; Khan, M.I.; Hussain, A.; Ashraf, M.F.; Li, X.; Weng, Y.; He, S. Basic leucine zipper domain transcription factors: The vanguards in plant immunity. Biotechnol. Lett. 2017, 39, 1779–1791. [Google Scholar] [CrossRef]
- Xu, F.; Kapos, P.; Cheng, Y.T.; Li, M.; Zhang, Y.; Li, X. NLR-associating transcription factor bHLH84 and its paralogs function redundantly in plant immunity. PLoS Pathog. 2014, 10, e1004312. [Google Scholar] [CrossRef]
- Zhou, Y.; Sun, L.; Wassan, G.M.; He, X.; Shaban, M.; Zhang, L.; Zhu, L.; Zhang, X. GbSOBIR1 confers Verticillium wilt resistance by phosphorylating the transcriptional factor GbbHLH171 in Gossypium barbadense. Plant Biotechnol. J. 2019, 17, 152–163. [Google Scholar] [CrossRef]
- Pedras, M.S.; Yaya, E.E.; Glawischnig, E. The phytoalexins from cultivated and wild crucifers: Chemistry and biology. Nat. Prod. Rep. 2011, 28, 1381–1405. [Google Scholar] [CrossRef]
- Ebeed, H.T. Bioinformatics Studies on the Identification of New Players and Candidate Genes to Improve Brassica Response to Abiotic Stress. In The Plant Family Brassicaceae: Biology and Physiological Responses to Environmental Stresses; Hasanuzzaman, M., Ed.; Springer: Singapore, 2020; pp. 483–496. [Google Scholar] [CrossRef]
- Khan, A.W.; Garg, V.; Roorkiwal, M.; Golicz, A.A.; Edwards, D.; Varshney, R.K. Super-Pangenome by Integrating the Wild Side of a Species for Accelerated Crop Improvement. Trends Plant Sci. 2020, 25, 148–158. [Google Scholar] [CrossRef]
- Scheben, A.; Edwards, D. Genome editors take on crops. Science 2017, 355, 1122–1123. [Google Scholar] [CrossRef]
- Scheben, A.; Edwards, D. Bottlenecks for genome-edited crops on the road from lab to farm. Genome Biol. 2018, 19, 178. [Google Scholar] [CrossRef]
- Scheben, A.; Edwards, D. Towards a more predictable plant breeding pipeline with CRISPR/Cas-induced allelic series to optimize quantitative and qualitative traits. Curr. Opin. Plant Biol. 2018, 45, 218–225. [Google Scholar] [CrossRef]
- Scheben, A.; Wolter, F.; Batley, J.; Puchta, H.; Edwards, D. Towards CRISPR/Cas crops—bringing together genomics and genome editing. New Phytol. 2017, 216, 682–698. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M. The Plant Family Brassicaceae, 1st ed.; Springer: Singapore, 2020. [Google Scholar] [CrossRef]
| Country | Production (Tonnes, 106) | Area Harvested (Ha, 106) | Yield (Tonnes Per Ha) |
|---|---|---|---|
| 1. Canada | 20.34 | 9.12 | 2.23 |
| 2. China | 13.28 | 6.55 | 2.03 |
| 3. India | 8.43 | 6.70 | 1.26 |
| 4. France | 4.95 | 1.62 | 3.06 |
| 5. Australia | 3.89 | 3.17 | 1.23 |
| 6. Germany | 3.67 | 1.22 | 3.00 |
| 7. Ukraine | 2.75 | 1.04 | 2.65 |
| 8. Poland | 2.20 | 0.84 | 2.64 |
| 9. USA | 2.01 | 0.79 | 2.55 |
| 10. Russia | 1.99 | 1.50 | 1.33 |
| Gene (Position) | Candidate Genes | RGA Type | Gene Ortholog (TAIR10) | Molecular Function | References |
|---|---|---|---|---|---|
| Rlm1 (A07 in Bna) | BnaA07g28760D | RLP | AT1G56140 | LRR TM prot_k | [76] |
| BnaA07g29310D | RLK | AT1G71390 | RLP 11 | [76] | |
| BnaA07g27720D | NLR | AT1G69160 | BIG GRAIN LIKE 1 supressor | [76] | |
| BnaA07g28550D | - | AT1G33612 | Receptor for the Plant Natriuretic Peptide | [76] | |
| BnaA07g28840D | RLK | AT1G70740 | Prot_k superfam_prot | [77] | |
| BnaA07g27460D | RLK | AT1G68830 | STN7 prot_k | [78] | |
| Rlm3, Rlm4 & Rlm7 (A07 in Bna) | BnaA07g20490D- | AT1G79090 | Protein PAT1 homolog | [76] | |
| BnaA07g20910D | NLR | AT1G77610 | UDP-galactose transporter 1 | [76] | |
| BnaA07g17000D | NLR | AT1G12220 | DRP RPS5/nucleotide binding | [79] | |
| BnaA07g17760D | RLK | AT1G56145 | LRR TM prot_k | [79] | |
| BnaA07g18000D | RLK | AT3G58690 | Prot_k superfam_prot | [79] | |
| BnaA07g18480D | RLK | AT3G59700 | L-type lectin-domain containing receptor kinase V.5 | [79] | |
| BnaA07g20630D | RLK | AT1G78290 | SRK2C/ST_k | [79] | |
| BnaA07g18680D | TM-CC | AT3G60470 | LRR TM prot_k | [79] | |
| BnaA07g18770D | TM-CC | AT3G60600 | VAP 1-1/protein binding | [79] | |
| BnaA07g18880D | TM-CC | AT3G61050 | NTMC2T4/lipid binding | [79] | |
| BnaA07g19680D | TM-CC | AT1G79830 | GC5 (golgin candidate 5)/protein binding | [79] | |
| BnaA07g20240D | RLK | AT1G79640 | Prot_k superfam_prot/ST_k tyrosine | [80] | |
| Rlm12 (A01 in Bna) | BnaA01g12900D | RLP | AT4G23100 | Glutamate-cysteine ligase, chloroplastic | [81] |
| BnaA01g12800D | RLP | AT4G22990 | Major Facilitator Superfamily with SPX | [81] | |
| BnaA01g12940D | RLP | AT4G23240 | Putative cysteine-rich RLP kinase 16 | [81] | |
| LepR1 (A02 in Bna) | BnaA02g15610D | RLK | AT1G71870 | Protein DETOXIFICATION 54/MATE efflux fam_prot | [70,76] |
| BnaA02g15810D | RLK | AT1G72140 | Protein NRT1/PTR FAMILY 5.12/proton-dependent oligopeptide transport (POT) fam_prot | [70,76] | |
| BnaA02g15820D | RLK | AT1G72150 | Patellin-1/transporter | [70,76] | |
| BnaA02g15890D | RLK | AT1G72290.1 (CDS) | Cysteine protease inhibitor WSCP | [70,76] | |
| BnaA02g16700D | RLK | AT2G18910 | Expressed protein/hydroxyproline-rich glycoprotein fam_prot | [70,76] | |
| BnaA02g16770D | RLK | AT1G74190 | RLP 15 | [70,76] | |
| BnaA02g16960D | NLR | AT1G30490.1 (CDS) | Homeobox-leucine zipper protein ATHB-9 | [70,76] | |
| BnaA02g18160D | TM-CC | AT1G76570 | Chlorophyll a-b binding protein 7, chloroplastic | [70,76] | |
| BnaA02g20380D | RLK | AT4G01440 | WAT1-related protein | [70,76] | |
| BnaA02g20440D | RLK | AT4G01590 | DNA-directed RNA polymerase III subunit | [70,76] | |
| BnaA02g20610D | RLK | AT4G02510 | Translocase of chloroplast 159, chloroplastic/TM receptor | [70,76] | |
| BnaA02g21110D | RLK | AT5G19010 | MAP kinase 16 | [70,76] | |
| BnaA02g21890D | RLK | AT4G11010 | Nucleoside diphosphate kinase/ATP binding | [70,76] | |
| BnaA02g22210D | RLK | AT5G43370 | Probable inorganic phosphate transporter 1-2 | [70,76] | |
| BnaA02g22280D | RLK | AT5G43710 | Alpha-mannosidase/glycoside hydrolase family 47 protein | [70,76] | |
| BnaA02g22610D | NLR | AT5G40910 | DRP (TNL class) | [70,76] | |
| BnaA02g23050D | TM-CC | AT5G42570 | Intracellular protein transport | [82,83] | |
| BnaA02g24000D | NLR | AT5G45490 | Probable DRP | [82,83] | |
| BnaA02g24440D | RLP | AT5G46330 | LRR RLP kinase/TM ST_k | [82,83] | |
| BnaA02g24500D | NLR | AT5G46510 | DRP (TNL class) | [82,83] | |
| BnaA02g24510D | NLR | AT5G46450 | DRP (TNL class) | [82,83] | |
| BnaA02g24530D | NLR | AT5G46450 | DRP (TNL class) | [82,83] | |
| BnaA02g24540D | NLR | AT5G46450 | DRP (TNL class) | [82,83] | |
| BnaA02g24560D | NLR | AT5G46451 | DRP (TNL class) | [82,83] | |
| BnaA02g25110D | NLR | AT5G47220 | Ethylene responsive element binding factor 2 | [84] | |
| LepR2 (A10 in Bna) | BnaA10g03460D | RLK | AT1G05300 | Zinc transporter 5 | [70,76] |
| BnaA10g06440D | RLK | AT5G53070 | Ribosomal protein L9/RNase H1 | [70,76] | |
| BnaA10g07140D | RLK | AT3G15240 | ST_k WNK (With No Lysine)-like protein | [70,76] | |
| BnaA10g09460D | NLR | AT5G55220 | Trigger factor-like protein TIG, chloroplastic | [70,76] | |
| BnaA10g09870D | RLK | AT5G55670 | RNA-binding (RRM/RBD/RNP motifs) fam_prot | [70,76] | |
| BnaA10g10000D | NLR | AT5G55910 | ST_k D6PK | [70,76] | |
| BnaA10g12510D | RLK | AT5G59200 | Putative pentatricopeptide repeat-containing protein, chloroplastic | [70,76] | |
| BnaA10g13610D | NLR | AT5G60000 | TM protein | [70,76] | |
| BnaA10g14660D | RLK | AT5G20900 | TIFY 3B/JAZ12 (JASMONATE-ZIM-DOMAIN PROTEIN 12) | [70,76] | |
| BnaA10g14840D | RLK | AT5G20670 | Unknown protein | [70,76] | |
| BnaA10g06390D | RLK | AT5G53000 | PP2A regulatory subunit TAP46 | [70,76] | |
| BnaA10g07390D | RLK | AT5G52520 | Proline--tRNA ligase, chloroplastic/mitochondrial | [70,76] | |
| BnaA10g07400D | RLK | AT5G52510 | SCL8 | [70,76] | |
| BnaA10g07410D | RLK | AT5G52510 | SCL8 | [70,76] | |
| BnaA10g07650D | RLK | AT5G51970 | Sorbitol dehydrogenase | [70,76] | |
| BnaA10g09120D | RLK | AT5G54850 | Unknown protein | [70,76] | |
| BnaA10g09500D | RLK | AT5G55280 | Cell division protein FtsZ homolog 1, chloroplastic | [70,76] | |
| BnaA10g10380D | RLK | AT5G56220 | P-loop containing nucleoside triphosphate hydrolases superfam_prot/nucleotide binding | [70,76] | |
| BnaA10g10430D | RLK | AT5G56210 | WPP domain-interacting protein 2 | [70,76] | |
| BnaA10g11120D | RLK | AT5G57110 | Calcium-transporting ATPase | [70,76] | |
| BnaA10g11930D | RLK | AT5G58410 | E3 ubiquitin-protein ligase listerin/zinc ion binding | [70,76] | |
| BnaA10g12560D | RLK | AT5G59610 | Chaperone DnaJ-domain superfam_prot/DNAJ heat shock N-terminal domain-containing protein | [70,76] | |
| BnaA10g12830D | RLK | AT4G34110 | Polyadenylate-binding/RNA binding/translation initiation factor | [70,76] | |
| BnaA10g12860D | RLK | AT5G59900 | Putative pentatricopeptide repeat-containing protein | [70,76] | |
| BnaA10g12870D | RLK | AT5G22880 | Histone H2B/DNA binding | [70,76] | |
| BnaA10g12880D | RLK | AT5G59950 | RNA-binding fam_prot/RNA and export factor-binding protein | [70,76] | |
| BnaA10g12890D | RLK | AT5G59990 | CCT motif fam_prot | [70,76] | |
| BnaA10g12900D | RLK | AT5G60020 | Laccase-17 | [70,76] | |
| BnaA10g12950D | RLK | AT5G60120 | Target of early activation tagged (EAT) 2/TF | [70,76] | |
| BnaA10g14170D | RLK | AT5G22170 | TM protein | [70,76] | |
| BnaA10g14640D | RLK | AT2G24080 | F-box protein (DUF295) | [70,76] | |
| BnaA10g15480D | RLK | AT5G19690 | Dolichyl-diphosphooligosaccharide-protein glycosyltransferase subunit STT3A | [70,76] | |
| BnaA10g18330D | RLK | AT5G16000 | Protein NSP-INTERACTING KINASE 1 | [70,76] | |
| BnaA10g19700D | RLK | AT5G13870 | Xyloglucan endotransglucosylase/hydrolase | [70,76] | |
| BnaA10g20110D | RLK | AT5G13180 | NAC domain-containing protein 83/TF | [70,76] | |
| BnaA10g23030D | RLK | AT5G08450 | Zinc finger CCCH domain protein | [70,76] | |
| BnaA10g23040D | RLK | AT5G08440 | Unknown protein | [70,76] | |
| BnaA10g26650D | RLK | AT5G03290 | Isocitrate dehydrogenase (NAD) catalytic subunit 5, mitochondrial | [70,76] | |
| BLMR1 (A10 in Bna) | BnaA10g21910D | - | AT5G10360 | 40S ribosomal protein S6 (RPS6B) | [85] |
| BnaA10g19660D | - | AT3G17620 | Putative F-box domain protein | [85] | |
| BLMR2 (A10 in Bna) | BnaA10g11390D | - | AT5G57340 | Ras guanine nucleotide exchange factor Q-like protein | [85] |
| BnaA10g11500D | TM | AT5G57560 | Xyloglucan endotransglucosylase/hydrolase | [85] | |
| LepR4 (A06 in Bra) | Bra018037 | NLR | AT5G17680 | DRP (TNL class) | [86] |
| Bra018057 | NLR | AT5G66900 | DRP (CNL class) | [86] | |
| Bra018198 | NLR | AT3G46710 | DRP (CNL class) | [86] | |
| Bra019483 | NLR | AT2G15530 | RING/U-box superfam_prot | [86] | |
| Rlm1 (C06 in Bol) | Bo6g077080 | NLR | AT3G60490 | Ethylene-responsive TF ERF035 APETALA2 | [87] |
| Bo6g088090 | RLK | AT1G73080 | RLP kinase LRR-RLK, STKc | [87] | |
| Bo6g080150 | RLK | AT1G80080 | Protein TOO MANY MOUTHS_TMM LRR | [87] | |
| Bo6g093010 | RLK | AT1G71830 | Somatic embryogenesis receptor kinase 1 LRR-RLK, STKc | [87] | |
| Bo6g089160 | NLR | AT1G72890 | DRP (TIR-NBS class) | [87] | |
| Bo6g089290 | NLR | AT1G72850 | DRP (TIR-NBS class) | [88] | |
| LepR1 (C02 in Bol) | Bo2g093170 | NLR | AT1G57850 | TIR domain protein family | [88] |
| Bo2g095430 | LRR | AT1G22000 | Putative F-box/LRR protein | [88] | |
| Bo2g095460 | RLK | AT1G79620 | LRR RLP kinase | [88] | |
| Bo2g103360 | NLR | AT5G36930 | DRP (TNL class) | [88] | |
| Bo2g103380 | LRR | AT4G03220 | Putative F-box/LRR protein | [88] | |
| Bo2g104830 | LRR | AT3G47580 | LRR RLP kinase | [88] | |
| Bo2g118150 | RLK | AT1G56120 | LRR TM prot_k | [88] | |
| Bo2g118200 | RLK | AT1G56130 | Probable LRR RLK ST_k | [88] | |
| Bo2g124490 | NLR | AT1G63730 | DRP (TNL class) | [88] | |
| Bo2g124590 | RLK | AT5G44700 | LRR RLK ST_k GSO2 | [88] | |
| Bo2g125680 | RLK | AT3G47570 | Probable LRR RLK ST_k | [88] | |
| Bo2g125700 | RLK | AT5G20480 | LRR RLK ST_k | [88] | |
| Bo2g126850 | NLR | AT5G45220 | DRP (TNL class) | [88] | |
| Bo2g126860 | NLR | AT2G17050 | DRP (TNL class) | [88] | |
| Bo2g126870 | NLR | AT5G45210 | DRP (TNL class) | [88] | |
| Bo2g126880 | NLR | AT5G17880 | Disease resistance-like protein CSA1 | [88] | |
| Bo2g126900 | NLR | AT5G45220 | DRP (TNL class) | [88] | |
| Bo2g126920 | NLR | AT5G45230 | DRP (TNL class) | [88] | |
| Bo2g126980 | NLR | AT5G45240 | DRP (TNL class) | [88] | |
| Bo2g127270 | NLR | AT5G45490 | Probable DRP | [88] | |
| Bo2g127290 | NLR | AT5G45490 | Probable DRP | [88] | |
| Bo2g127320 | NLR | AT5G45510 | Probable DRP | [88] | |
| Bo2g129990 | RLK | AT5G46330 | LRR RLP kinase | [88] | |
| Bo2g130040 | NLR | AT5G46470 | DRP RPS6 | [88] | |
| Bo2g130050 | LRR | AT5G40060 | DRP (NLR class) | [88] | |
| Bo2g130080 | NLR | AT5G46270 | DRP (TNL class) | [88] | |
| Bo2g130090 | NLR | AT5G46450 | DRP (TNL class) | [88] | |
| Bo2g130100 | NLR | AT5G46450 | DRP (TNL class) | [88] | |
| Bo2g130110 | NLR | AT4G08450 | DRP (TNL class) | [88] | |
| Bo2g130150 | NLR | AT4G08450 | DRP (TNL class) | [88] | |
| Bo2g130180 | NLR | AT5G46450 | DRP (TNL class) | [88] | |
| Bo2g131530 | NLR | AT4G16920 | DRP (TNL class) | [88] | |
| Bo2g131540 | NLR | AT5G46270 | DRP (TNL class) | [88] | |
| Bo2g131590 | NLR | AT5G46450 | DRP (TNL class) | [88] | |
| Bo2g131610 | NLR | AT5G46260 | DRP (TNL class) | [88] | |
| Bo2g131620 | NLR | AT5G40060 | DRP (NLR class) | [88] | |
| LepR2 (C09 in Bol) | Bo9g111490 | LRR | AT1G51370 | F-box domain/LRR protein | [89] |
| Bo9g111500 | LRR | AT5G25850 | Putative F-box domain/LRR protein | [89] | |
| Bo9g111510 | LRR | AT5G53840 | F-box domain/LRR protein | [89] | |
| Bo9g113780 | RLK | AT5G53890 | LRR RLP kinase | [89] | |
| Bo9g117290 | RLK | AT5G54380 | RLP kinase THESEUS 1 | [89] | |
| Bo9g119130 | RLK | AT5G55090 | MAP kinase 15 | [89] | |
| Bo9g120720 | LRR | AT5G66330 | LRR fam_prot | [89] | |
| Bo9g122300 | RLK | AT5G56040 | LRR RLP kinase | [89] | |
| Bo9g125930 | LRR | AT3G56780 | F-box domain/LRR protein | [89] | |
| Bo9g126120 | LRR | AT5G56560 | F-box domain/LRR protein | [89] | |
| Bo9g126140 | LRR | AT5G56560 | F-box domain/LRR protein | [89] | |
| Bo9g126150 | RLK | AT5G56580 | MAP kinase 6 | [89] | |
| Bo9g135700 | CC | AT2G42480 | MATH & CC domain-containing protein | [89] | |
| LepR4 (C03 in Bol) | Bo3g099380 | RLK | AT5G65240 | LRR prot_k | [90] |
| Bo3g102880 | NLR | AT4G36150 | DRP (TNL class) | [90] | |
| Bo3g103150 | RLK | AT5G63710 | LRR prot_k | [90] | |
| Bo3g107230 | RLK | AT5G62710 | LRR prot_k | [90] | |
| Bo3g107530 | RLK | AT1G53510 | MAP Pkinase 18 | [90] | |
| Bo3g110840 | RLK | AT3G47090 | LRR prot_k | [90] | |
| Bo3g114980 | RLK | AT3G47090 | MAP Pkinase_Tyr, ST_k | [90] | |
| Bo3g130040 | RLK | AT3G45640 | MAP Pkinase | [90] | |
| Bo3g134690 | RLK | AT3G47580 | LRR protein Pkinase | [90] | |
| LepR4 (C08 in Bol) | Bo8g077170 | RLK | AT1G53510 | MAP Pkinase 18 | [90] |
| Bo8g077270 | NLR | AT5G17680 | DRP (TNL class) | [90] | |
| Bo8g077320 | CC | AT3G48860 | CC domain containing protein SCD2 | [90] | |
| Rlm6 (A07 in Bju) | BjuA027357 | RLK | AT1G66830 | Probable inactive LRR RLP kinase | [91] |
| BjuA043308 | RLK | AT1G67510 | LRR prot_k fam_prot | [91] | |
| Rlm6 (B04 in Bju) | BjuB042709 | RLK | AT1G10850 | LRR prot_k fam_prot/ST_k | [91] |
| BjuB042726 | RLK | AT1G11130 | LRR prot_k fam_prot/receptor signalling protein ST_k | [91] | |
| LMJR1 (B03 in Bju) | BjuB043144 | RLK | AT1G61360 | ST_k | [92] |
| BjuB032936 | RLK | AT1G29720 | Probable LRR RLK ST_k | [92] | |
| BjuB043487 | RLK | AT1G21209 | Wall associated kinase 4 | [92] | |
| BjuB043117 | RLP | AT1G10520 | DNA polymerase lambda | [92] | |
| BjuB047419 | RLP | AT1G71400 | RLP 12 | [92] | |
| LMJR2 (B08 in Bju) | BjuB015599 | RLK | AT5G59680 | Probable LRR RLK ST_k | [92] |
| BjuB041327 | RLK | AT4G08850 | MDIS1-interacting RLK 2 | [92] | |
| BjuB040922 | RLK | AT5G07620 | Prot_k superfam_prot | [92] | |
| BjuB041021 | RLK | AT5G10530 | L-type lectin-domain containing receptor kinase IX.1 | [92] | |
| BjuB019224 | RLK | AT3G56050 | Probable inactive RLP kinase | [92] | |
| BjuB045981 | RLP | AT5G56810 | Putative F-box domain/LRR protein | [92] | |
| rjlm2 (B01 in Bju) | BjuB026698 | RLK | AT2G35620 | LRR RLK ST_k FEI 2 | [92] |
| BjuB025797 | RLK | AT5G38210 | Prot_k fam_prot | [92] | |
| Species | Types of Progenies | References |
|---|---|---|
| Brassica carinata and Brassica rapa | Double haploid (DH) lines | [86,115,144,145,146,147] |
| Brassica juncea | Recombinant and backcrossed (BC) lines | [148,149,150] |
| Brassica nigra | Hybrid and recombinant lines | [149,151,152] |
| Brassica elongata, Brassica fruticulosa, Brassica souliei and Diplotaxis tenuifolia | Hybrid | [130] |
| Coincya monensis and Hirschfeldia incana | Hybrid and BC lines | [130,132] |
| Sinapsis arvensis | Somatic hybrids and BC lines | [130,132,153,154] |
| Brassica tournefortii | Somatic hybrids | [155] |
| Species (Common Name) | NLR | RLK | RLP | Software Used | References |
|---|---|---|---|---|---|
| Arabidopsis lyrata (Lyre-leaved rock-cress) | 243 | 514 | 73 | RGAugury | [140] |
| 506 | 495 | 56 | RGAugury | [138] | |
| 198 | - | - | HMM/MEME | [143] | |
| 200 | - | - | HMM/LRRfinder | [137] | |
| Arabidopsis thaliana (Mouse-ear cress) | 205 | 516 | 73 | RGAugury | [140] |
| 410 | 517 | 75 | RGAugury | [138] | |
| 152 | - | - | NLGenome Sweeper | [141] | |
| 213 | - | - | HMMER | [142] | |
| 165 | - | - | HMM/MEME | [143] | |
| 167 | - | - | HMM/LRRfinder | [137] | |
| Brassica juncea (Indian mustard) | 315 | 1085 | 191 | RGAugury | [140] |
| - | 493 | 228 | RGAugury | [91] | |
| Brassica napus (Oilseed rape) | 286 1 | 989 1 | 77 1 | RGAugury | [140] |
| 208 2 | 680 2 | 223 2 | RGAugury | [140] | |
| 621 3 | 1497 3 | 273 3 | RGAugury | [140] | |
| 566 4 | 1517 4 | 260 4 | RGAugury | [140] | |
| 464 | - | - | HMMER | [136] | |
| 641 | - | - | MEME/MAST | [135] | |
| B. napus pangenome | 16430 | 51611 | 5229 | RGAugury | [70] |
| Brassica nigra (Black mustard) | 372 | 776 | 176 | RGAugury | [140] |
| - | 317 | 176 | RGAugury | [91] | |
| Brassica oleracea (Cabbage) | 493 | 822 | 159 | RGAugury | [72] |
| 438 | 796 | 155 | RGAugury | [140] | |
| 146 | - | - | HMMER | [136] | |
| 443 | - | - | MEME/MAST | [135] | |
| 157 | - | - | HMMER | [142] | |
| 408 | - | - | HMMER | [139] | |
| B. oleracea pangenome | 616 | 932 | 223 | RGAugury | [72] |
| Brassica rapa (Field mustard) | 263 | 670 | 106 | RGAugury | [140] |
| 488 | 747 | 118 | RGAugury | [138] | |
| - | 300 | 65 | RGAugury | [91] | |
| 202 | - | - | HMMER | [136] | |
| 249 | - | - | MEME/MAST | [135] | |
| 206 | - | - | HMMER | [142] | |
| 204 | - | - | HMM/MEME | [143] | |
| 201 | - | - | HMM/LRRfinder | [137] | |
| Brassica macrocarpa(‘Egadi‘ cabbage) | 447 | 862 | 186 | RGAugury | [72] |
| Camelina sativa (False flax) | 504 | 1469 | 280 | RGAugury | [140] |
| Capsella rubella (pink shepherd’s-purse) | 180 | 539 | 87 | RGAugury | [140] |
| 200 | 536 | 97 | RGAugury | [138] | |
| 127 | - | - | HMM/MEME | [143] | |
| Eutrema salsugineum (Saltwater cress) | 165 | 509 | 77 | RGAugury | [140] |
| 348 | 483 | 83 | RGAugury | [138] | |
| 88 | - | - | HMM/MEME | [143] | |
| 87 | - | - | HMM/LRRfinder | [137] | |
| Raphanus raphanistrum (Wild radish) | 206 | 585 | 142 | RGAugury | [140] |
| Thlaspi arvense (Field penny-cress) | 183 | 474 | 120 | RGAugury | [140] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cantila, A.Y.; Saad, N.S.M.; Amas, J.C.; Edwards, D.; Batley, J. Recent Findings Unravel Genes and Genetic Factors Underlying Leptosphaeria maculans Resistance in Brassica napus and Its Relatives. Int. J. Mol. Sci. 2021, 22, 313. https://doi.org/10.3390/ijms22010313
Cantila AY, Saad NSM, Amas JC, Edwards D, Batley J. Recent Findings Unravel Genes and Genetic Factors Underlying Leptosphaeria maculans Resistance in Brassica napus and Its Relatives. International Journal of Molecular Sciences. 2021; 22(1):313. https://doi.org/10.3390/ijms22010313
Chicago/Turabian StyleCantila, Aldrin Y., Nur Shuhadah Mohd Saad, Junrey C. Amas, David Edwards, and Jacqueline Batley. 2021. "Recent Findings Unravel Genes and Genetic Factors Underlying Leptosphaeria maculans Resistance in Brassica napus and Its Relatives" International Journal of Molecular Sciences 22, no. 1: 313. https://doi.org/10.3390/ijms22010313
APA StyleCantila, A. Y., Saad, N. S. M., Amas, J. C., Edwards, D., & Batley, J. (2021). Recent Findings Unravel Genes and Genetic Factors Underlying Leptosphaeria maculans Resistance in Brassica napus and Its Relatives. International Journal of Molecular Sciences, 22(1), 313. https://doi.org/10.3390/ijms22010313








