Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers
Abstract
1. Introduction
2. Overview of Gas Transport in Machine Perfusion
2.1. Physiological Gas Transport
2.1.1. The Impact of Temperature on Gas Transport and Oxygen Requirements
2.1.2. The Impact of Temperature on Gas Solubility and Pressure in Fluids
2.1.3. Gas Transport in Machine Perfusion
3. Overview of Oxygen Carriers
3.1. Hemoglobin in Red Blood Cells
3.2. Hemoglobin-Based Oxygen Carrier
3.2.1. Hemoglobin-Vesicles
3.2.2. Hemoglobin-Based Oxygen Carrier-201
3.3. Natural Extracellular Oxygen Carrier Hemarina M101
3.4. Perfluorocarbons
4. Future Perspectives
5. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AOC | artificial oxygen carrier |
| aPCO2 | arterial partial pressure of carbon dioxide |
| aPO2 | arterial partial pressure of oxygen |
| ATP | adenosine triphosphate |
| asO2 | arterial percentage of Hb saturated with oxygen |
| CO2 | carbon dioxide |
| Fe++ | iron ion |
| FiO2 | fraction of inspired oxygen |
| Hb | hemoglobin |
| HBOC | hemoglobin-based oxygen carrier |
| HBOC-201 | hemoglobin-based oxygen carrier-201 |
| Hb-Vs | hemoglobin-vesicles |
| HMP | hypothermic machine perfusion |
| IRI | Ischemia reperfusion injury |
| metHb | methemoglobin |
| MP | machine perfusion |
| NMP | normothermic machine perfusion |
| NO | nitric oxide |
| O2 | oxygen |
| OC | oxygen carrier |
| PCO2 | partial pressure of carbon dioxide |
| PFCs | perfluorocarbons |
| PO2 | partial pressure of oxygen |
| RBC | red blood cell |
| ROS | reactive oxygen species |
| RQ | respiratory quotient |
| SCS | Static cold storage |
| SNMP | subnormothermic machine perfusion |
| SO2 | oxygen saturation |
| VCO2 | production of CO2 |
| VO2 | consumption of O2 |
| vPCO2 | venous partial pressure of carbon dioxide |
| vPO2 | venous partial pressure of oxygen |
| vsO2 | venous percentage of Hb saturated with oxygen |
References
- Adam, R.; Karam, V.; Cailliez, V.; O Grady, J.G.; Mirza, D.; Cherqui, D.; Klempnauer, J.; Salizzoni, M.; Pratschke, J.; Jamieson, N.; et al. 2018 Annual Report of the European Liver Transplant Registry (ELTR)-50-year evolution of liver transplantation. Transpl. Int. 2018, 31, 1293–1317. [Google Scholar] [CrossRef]
- Monbaliu, D.; Brassil, J. Machine Perfusion of the Liver: Past, Present and Future. Curr. Opin. Organ Transplant. 2010, 15, 160–166. [Google Scholar] [CrossRef]
- Weeder, P.D.; van Rijn, R.; Porte, R.J. Machine Perfusion in Liver Transplantation as a Tool to Prevent Non-Anastomotic Biliary Strictures: Rationale, Current Evidence and Future Directions. J. Hepatol. 2015, 63, 265–275. [Google Scholar] [CrossRef]
- De Meijer, V.E.; Fujiyoshi, M.; Porte, R.J. Ex Situ Machine Perfusion Strategies in Liver Transplantation. J. Hepatol. 2019, 70, 203–205. [Google Scholar] [CrossRef]
- Salehi, S.; Tran, K.; Grayson, W.L. Focus: Medical Technology: Advances in Perfusion Systems for Solid Organ Preservation. Yale J. Biol. Med. 2018, 91, 301. [Google Scholar]
- Karangwa, S.; Dutkowski, P.; Fontes, P.; Friend, P.; Guarrera, J.; Markmann, J.; Mergental, H.; Minor, T.; Quintini, C.; Selzner, M. Machine Perfusion of Donor Livers for Transplantation: A Proposal for Standardized Nomenclature and Reporting Guidelines. Am. J. Transplant. 2016, 16, 2932–2942. [Google Scholar] [CrossRef]
- Dutkowski, P.; Polak, W.G.; Muiesan, P.; Schlegel, A.; Verhoeven, C.J.; Scalera, I.; De Oliveira, M.L.; Kron, P.; Clavien, P. First Comparison of Hypothermic Oxygenated Perfusion versus Static Cold Storage of Human Donation After Cardiac Death Liver Transplants. Ann. Surg. 2015, 262, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Dutkowski, P.; Schlegel, A.; de Oliveira, M.; Müllhaupt, B.; Neff, F.; Clavien, P. HOPE for Human Liver Grafts obtained from Donors After Cardiac Death. J. Hepatol. 2014, 60, 765–772. [Google Scholar] [CrossRef]
- Westerkamp, A.C.; Karimian, N.; Matton, A.P.; Mahboub, P.; van Rijn, R.; Wiersema-Buist, J.; de Boer, M.T.; Leuvenink, H.G.; Gouw, A.S.; Lisman, T.; et al. Oxygenated Hypothermic Machine Perfusion After Static Cold Storage Improves Hepatobiliary Function of Extended Criteria Donor Livers. Transplantation 2016, 100, 825–835. [Google Scholar] [CrossRef]
- Schlegel, A.; Kron, P.; Dutkowski, P. Hypothermic Machine Perfusion in Liver Transplantation. Curr. Opin. Organ Transplant. 2016, 21, 308–314. [Google Scholar] [CrossRef]
- Brüggenwirth, I.M.; van Leeuwen, O.B.; de Vries, Y.; Bodewes, S.B.; Adelmeijer, J.; Wiersema-Buist, J.; Lisman, T.; Martins, P.N.; de Meijer, V.E.; Porte, R.J. Extended Hypothermic Oxygenated Machine Perfusion Enables Ex Situ Preservation of Porcine Livers for Up to 24 Hours. JHep Rep. 2020, 2, 100092. [Google Scholar] [CrossRef] [PubMed]
- Spetzler, V.N.; Goldaracena, N.; Echiverri, J.; Kaths, J.M.; Louis, K.S.; Adeyi, O.A.; Yip, P.M.; Grant, D.R.; Selzner, N.; Selzner, M. Subnormothermic Ex Vivo Liver Perfusion is a Safe Alternative to Cold Static Storage for Preserving Standard Criteria Grafts. Liver Transplant. 2016, 22, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Bruinsma, B.G.; Yeh, H.; Özer, S.; Martins, P.N.; Farmer, A.; Wu, W.; Saeidi, N.; Op den Dries, S.; Berendsen, T.A.; Smith, R.N. Subnormothermic Machine Perfusion for Ex Vivo Preservation and Recovery of the Human Liver for Transplantation. Am. J. Transplant. 2014, 14, 1400–1409. [Google Scholar] [CrossRef] [PubMed]
- Matton, A.P.M.; de Vries, Y.; Burlage, L.C.; van Rijn, R.; Fujiyoshi, M.; de Meijer, V.E.; de Boer, M.T.; de Kleine, R.H.J.; Verkade, H.J.; Gouw, A.S.H.; et al. Biliary Bicarbonate, pH, and Glucose are Suitable Biomarkers of Biliary Viability during Ex Situ Normothermic Machine Perfusion of Human Donor Livers. Transplantation 2019, 103, 1405–1413. [Google Scholar] [CrossRef]
- Op den Dries, S.; Karimian, N.; Sutton, M.; Westerkamp, A.; Nijsten, M.; Gouw, A.; Wiersema-Buist, J.; Lisman, T.; Leuvenink, H.; Porte, R. Ex Vivo Normothermic Machine Perfusion and Viability Testing of Discarded Human Donor Livers. Am. J. Transplant. 2013, 13, 1327–1335. [Google Scholar] [CrossRef]
- Gilbo, N.; Monbaliu, D. Temperature and Oxygenation during Organ Preservation: Friends or Foes? Curr. Opin. Organ. Transplant. 2017, 22, 290–299. [Google Scholar] [CrossRef]
- Boteon, Y.L.; Attard, J.; Boteon, A.P.; Wallace, L.; Reynolds, G.; Hubscher, S.; Mirza, D.F.; Mergental, H.; Bhogal, R.H.; Afford, S.C. Manipulation of Lipid Metabolism during Normothermic Machine Perfusion: Effect of Defatting Therapies on Donor Liver Functional Recovery. Liver Transplant. 2019, 25, 1007–1022. [Google Scholar] [CrossRef]
- De Vries, R.J.; Tessier, S.N.; Banik, P.D.; Nagpal, S.; Cronin, S.E.; Ozer, S.; Hafiz, E.O.; van Gulik, T.M.; Yarmush, M.L.; Markmann, J.F. Subzero Non-Frozen Preservation of Human Livers in the Supercooled State. Nat. Protoc. 2020, 15, 1–17. [Google Scholar] [CrossRef]
- Thorne, A.M.; Ubbink, R.; Brüggenwirth, I.M.; Nijsten, M.W.; Porte, R.J.; de Meijer, V.E. Hyperthermia Induced Changes in Liver Physiology and Metabolism: A Rationale for Hyperthermic Machine Perfusion. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G43–G50. [Google Scholar] [CrossRef]
- Plaats, A.V.D. The Groningen Hypothermic Liver Perfusion System for Improved Preservation in Organ Transplantation. Diss. Print 2005, 34, 1924–1934. [Google Scholar]
- Karimian, N.; Matton, A.P.; Westerkamp, A.C.; Burlage, L.C.; Op den Dries, S.; Leuvenink, H.G.; Lisman, T.; Uygun, K.; Markmann, J.F.; Porte, R.J. Ex Situ Normothermic Machine Perfusion of Donor Livers. JoVE (J. Vis. Exp.) 2015, 99, e52688. [Google Scholar] [CrossRef] [PubMed]
- Eshmuminov, D.; Leoni, F.; Schneider, M.A.; Becker, D.; Muller, X.; Onder, C.; Hefti, M.; Schuler, M.J.; Dutkowski, P.; Graf, R. Perfusion Settings and Additives in Liver Normothermic Machine Perfusion with Red Blood Cells as Oxygen Carrier. A Systematic Review of Human and Porcine Perfusion Protocols. Transplant. Int. 2018, 31, 956–969. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.E. Guyton and Hall Textbook of Medical Physiology, Jordanian Edition E-Book; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Berg, J. Tymoczko, J.L.; Stryer, L. Biochemistry, Chapter 14. In Metabolism: Basic Concepts and Design, 5th ed.; W H Freeman: New York, NY, USA, 2002. [Google Scholar]
- Pittman, R.N. Regulation of Tissue Oxygenation. In Colloquium Series on Integrated Systems Physiology: From Molecule to Function; Morgan & Claypool Life Sciences: San Rafael, CA, USA, 2011; pp. 1–100. [Google Scholar]
- Nelson, D.L.; Lehninger, A.L.; Cox, M.M. Citric acid cycle. In Lehninger Principles of Biochemistry; W.H. Freeman: New York, NY, USA, 2005; p. 615. [Google Scholar]
- Blaxter, K. Energy Metabolism in Animals and Man; CUP Archive: Cambridge, UK, 1989. [Google Scholar]
- Gillooly, J.F.; Brown, J.H.; West, G.B.; Savage, V.M.; Charnov, E.L. Effects of Size and Temperature on Metabolic Rate. Science 2001, 293, 2248–2251. [Google Scholar] [CrossRef] [PubMed]
- Schulte, P.M. The Effects of Temperature on Aerobic Metabolism: Towards a Mechanistic Understanding of the Responses of Ectotherms to a Changing Environment. J. Exp. Biol. 2015, 218, 1856–1866. [Google Scholar] [CrossRef]
- Siggaard-Andersen, O.; Wimberley, P.D.; Gothgen, I.; Siggaard-Andersen, M. A Mathematical Model of the Hemoglobin-Oxygen Dissociation Curve of Human Blood and of the Oxygen Partial Pressure as a Function of Temperature. Clin. Chem. 1984, 30, 1646–1651. [Google Scholar] [CrossRef]
- Lutz, J.; Henrich, H.; Bauereisen, E. Oxygen Supply and Uptake in the Liver and the Intestine. Pflügers Arch. 1975, 360, 7–15. [Google Scholar] [CrossRef]
- Rettich, T.R.; Battino, R.; Wilhelm, E. Solubility of Gases in Liquids. 22. High-Precision Determination of Henry’s Law Constants of Oxygen in Liquid Water from T = 274 K to T = 328 K. J. Chem. Thermodyn. 2000, 32, 1145–1156. [Google Scholar] [CrossRef]
- Avishay, D.M.; Tenny, K.M. Henry’s Law. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2019. [Google Scholar]
- Schlegel, A.; De Rougemont, O.; Graf, R.; Clavien, P.; Dutkowski, P. Protective Mechanisms of End-Ischemic Cold Machine Perfusion in DCD Liver Grafts. J. Hepatol. 2013, 58, 278–286. [Google Scholar] [CrossRef]
- Westerkamp, A.C.; Mahboub, P.; Meyer, S.L.; Hottenrott, M.; Ottens, P.J.; Wiersema-Buist, J.; Gouw, A.S.H.; Lisman, T.; Leuvenink, H.G.D.; Porte, R.J. End-Ischemic Machine Perfusion Reduces Bile Duct Injury in Donation After Circulatory Death Rat Donor Livers Independent of the Machine Perfusion Temperature. Liver Transpl. 2015, 21, 1300–1311. [Google Scholar] [CrossRef]
- Monbaliu, D.; Vekemans, K.; De Vos, R.; Brassil, J.; Heedfeld, V.; Qiang, L.; D’ hollander, M.; Roskams, T.; Pirenne, J. Hemodynamic, Biochemical, and Morphological Characteristics during Preservation of Normal Porcine Livers by Hypothermic Machine Perfusion. In Transplantation Proceedings; Elsevier: Amsterdam, The Netherlands, 2007; Volume 39, pp. 2652–2658. [Google Scholar]
- Bral, M.; Gala-Lopez, B.; Thiesen, A.; Hatami, S.; Bigam, D.L.; Freed, D.M.; James Shapiro, A.M. Determination of Minimal Hemoglobin Level Necessary for Normothermic Porcine Ex Situ Liver Perfusion. Transplantation 2018, 102, 1284–1292. [Google Scholar] [CrossRef]
- Liu, Q.; Nassar, A.; Farias, K.; Buccini, L.; Mangino, M.J.; Baldwin, W.; Bennett, A.; O’Rourke, C.; Iuppa, G.; Soliman, B.G.; et al. Comparing Normothermic Machine Perfusion Preservation with Different Perfusates on Porcine Livers from Donors After Circulatory Death. Am. J. Transplant. 2016, 16, 794–807. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; Moallempour, M.; Lim, J.C. HBOC-201, Hemoglobin Glutamer-250 (Bovine), Hemopure® (Biopure Corporation). Expert Opin. Biol. Ther. 2008, 8, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Urata, Y.; Anraku, M.; Maruyama, T.; Watanabe, H.; Sakai, H.; Horinouchi, H.; Kobayashi, K.; Tsuchida, E.; Kai, T.; et al. Pharmacokinetic Study of Enclosed Hemoglobin and Outer Lipid Component After the Administration of Hemoglobin Vesicles as an Artificial Oxygen Carrier. Drug Metab. Dispos. 2009, 37, 1456–1463. [Google Scholar] [CrossRef] [PubMed]
- Sakai, H.; Hamada, K.; Takeoka, S.; Nishide, H.; Tsuchida, E. Physical Properties of Hemoglobin Vesicles as Red Cell Substitutes. Biotechnol. Prog. 1996, 12, 119–125. [Google Scholar] [CrossRef]
- Alix, P.; Val-Laillet, D.; Turlin, B.; Mosbah, I.B.; Burel, A.; Bobillier, E.; Bendavid, C.; Delpy, E.; Zal, F.; Corlu, A. Adding the Oxygen Carrier M101 to a Cold-Storage Solution could be an Alternative to HOPE for Liver Graft Preservation. JHEP Rep. 2020, 2, 100119. [Google Scholar]
- Kaminski, J.; Hannaert, P.; Kasil, A.; Thuillier, R.; Leize, E.; Delpy, E.; Steichen, C.; Goujon, J.M.; Zal, F.; Hauet, T. Efficacy of the Natural Oxygen Transporter HEMO2 Life® in Cold Preservation in a Preclinical Porcine Model of Donation After Cardiac Death. Transpl. Int. 2019, 32, 985–996. [Google Scholar] [CrossRef]
- Menz, D.; Feltgen, N.; Menz, H.; Müller, B.; Lechner, T.; Dresp, J.; Hoerauf, H. How to Ward off Retinal Toxicity of Perfluorooctane and Other Perfluorocarbon Liquids? Invest. Ophthalmol. Vis. Sci. 2018, 59, 4841–4846. [Google Scholar] [CrossRef]
- Riess, J.G. Oxygen Carriers (“blood Substitutes”) Raison D’Être, Chemistry, and some Physiology Blut Ist Ein Ganz Besondrer Saft. Chem. Rev. 2001, 101, 2797–2920. [Google Scholar] [CrossRef]
- Marengo-Rowe, A.J. Structure-Function Relations of Human Hemoglobins. Bayl Univ. Med. Cent. Proc. 2006, 19, 239–245. [Google Scholar] [CrossRef]
- Lodemann, P.; Schorer, G.; Frey, B.M. Wrong Molar Hemoglobin Reference Values—a Longstanding Error that should be Corrected. Ann. Hematol. 2010, 89, 209. [Google Scholar] [CrossRef]
- Banan, B.; Watson, R.; Xu, M.; Lin, Y.; Chapman, W. Development of a Normothermic Extracorporeal Liver Perfusion System Toward Improving Viability and Function of Human Extended Criteria Donor Livers. Liver Transplant. 2016, 22, 979–993. [Google Scholar] [CrossRef] [PubMed]
- Mergental, H.; Perera, M.; Laing, R.; Muiesan, P.; Isaac, J.; Smith, A.; Stephenson, B.; Cilliers, H.; Neil, D.; Hübscher, S. Transplantation of Declined Liver Allografts Following Normothermic Ex-situ Evaluation. Am. J. Transplant. 2016, 16, 3235–3245. [Google Scholar] [CrossRef] [PubMed]
- Vogel, T.; Brockmann, J.G.; Quaglia, A.; Morovat, A.; Jassem, W.; Heaton, N.D.; Coussios, C.C.; Friend, P.J. The 24-hour Normothermic Machine Perfusion of Discarded Human Liver Grafts. Liver Transplant. 2017, 23, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Laing, R.W.; Bhogal, R.H.; Wallace, L.; Boteon, Y.; Neil, D.A.H.; Smith, A.; Stephenson, B.T.F.; Schlegel, A.; Hubscher, S.G.; Mirza, D.F.; et al. The use of an Acellular Oxygen Carrier in a Human Liver Model of Normothermic Machine Perfusion. Transplantation 2017, 101, 2746–2756. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Scerbo, M.; Kramer, G. A Review of Blood Substitutes: Examining the History, Clinical Trial Results, and Ethics of Hemoglobin-Based Oxygen Carriers. Clinics 2009, 64, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Fontes, P.; Lopez, R.; van der Plaats, A.; Vodovotz, Y.; Minervini, M.; Scott, V.; Soltys, K.; Shiva, S.; Paranjpe, S.; Sadowsky, D.; et al. Liver Preservation with Machine Perfusion and a Newly Developed Cell-Free Oxygen Carrier Solution Under Subnormothermic Conditions. Am. J. Transplant. 2015, 15, 381–394. [Google Scholar] [CrossRef]
- Sadowsky, D.; Zamora, R.; Barclay, D.; Yin, J.; Fontes, P.; Vodovotz, Y. Machine Perfusion of Porcine Livers with Oxygen-Carrying Solution Results in Reprogramming of Dynamic Inflammation Networks. Front. Pharmacol. 2016, 7, 413. [Google Scholar] [CrossRef]
- Matton, A.P.; Burlage, L.C.; van Rijn, R.; de Vries, Y.; Karangwa, S.A.; Nijsten, M.W.; Gouw, A.S.; Wiersema-Buist, J.; Adelmeijer, J.; Westerkamp, A.C. Normothermic Machine Perfusion of Donor Livers without the Need for Human Blood Products. Liver Transplant. 2018, 24, 528–538. [Google Scholar] [CrossRef]
- Boteon, Y.L.; Boteon, A.P.; Attard, J.; Mergental, H.; Mirza, D.F.; Bhogal, R.H.; Afford, S.C. Ex Situ Machine Perfusion as a Tool to Recondition Steatotic Donor Livers: Troublesome Features of Fatty Livers and the Role of Defatting Therapies. A Systematic Review. Am. J. Transplant. 2018, 18, 2384–2399. [Google Scholar] [CrossRef]
- De Vries, Y.; van Leeuwen, O.B.; Matton, A.P.M.; Fujiyoshi, M.; de Meijer, V.E.; Porte, R.J. Ex Situ Normothermic Machine Perfusion of Donor Livers using a Haemoglobin-Based Oxygen Carrier: A Viable Alternative to Red Blood Cells. Transpl. Int. 2018, 31, 1281–1282. [Google Scholar] [CrossRef]
- Aburawi, M.M.; Fontan, F.M.; Karimian, N.; Eymard, C.; Cronin, S.; Pendexter, C.; Nagpal, S.; Banik, P.; Ozer, S.; Mahboub, P. Synthetic Hemoglobin-based Oxygen Carriers are an Acceptable Alternative for Packed Red Blood Cells in Normothermic Kidney Perfusion. Am. J. Transplant. 2019, 19, 2814–2824. [Google Scholar] [CrossRef] [PubMed]
- Vrselja, Z.; Daniele, S.G.; Silbereis, J.; Talpo, F.; Morozov, Y.M.; Sousa, A.M.; Tanaka, B.S.; Skarica, M.; Pletikos, M.; Kaur, N. Restoration of Brain Circulation and Cellular Functions Hours Post-Mortem. Nature 2019, 568, 336. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, O.B.; de Vries, Y.; Fujiyoshi, M.; Nijsten, M.W.N.; Ubbink, R.; Pelgrim, G.J.; Werner, M.J.M.; Reyntjens, K.M.E.M.; van den Berg, A.P.; de Boer, M.T.; et al. Transplantation of High-Risk Donor Livers After Ex Situ Resuscitation and Assessment using Combined Hypo- and Normothermic Machine Perfusion: A Prospective Clinical Trial. Ann. Surg. 2019, 270, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R.N.; Patel, S.V.B.; Sun, Q.; Jiang, L.; Richard-Mohamed, M.; Ruthirakanthan, A.; Aquil, S.; Al-Ogaili, R.; Juriasingani, S.; Sener, A.; et al. Renal Protection Against Ischemia Reperfusion Injury: Hemoglobin-Based Oxygen Carrier-201 Versus Blood as an Oxygen Carrier in Ex Vivo Subnormothermic Machine Perfusion. Transplantation 2020, 104, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Shonaka, T.; Matsuno, N.; Obara, H.; Yoshikawa, R.; Nishikawa, Y.; Gouchi, M.; Otani, M.; Takahashi, H.; Azuma, H.; Sakai, H.; et al. Application of Perfusate with Human-Derived Oxygen Carrier Solution Under Subnormothermic Machine Perfusion for Donation After Cardiac Death Liver Grafts in Pigs. Transplant. Proc. 2018, 50, 2821–2825. [Google Scholar] [CrossRef] [PubMed]
- Shonaka, T.; Matsuno, N.; Obara, H.; Yoshikawa, R.; Nishikawa, Y.; Ishihara, Y.; Bochimoto, H.; Gochi, M.; Otani, M.; Kanazawa, H. Impact of Human-Derived Hemoglobin Based Oxygen Vesicles as a Machine Perfusion Solution for Liver Donation After Cardiac Death in a Pig Model. PLoS ONE 2019, 14, e0226183. [Google Scholar] [CrossRef]
- Brasile, L.; DelVecchio, P.; Amyot, K.; Haisch, C.; Clarke, J. Organ Preservation without Extreme Hypothermia using an Oxygent™ Supplemented Perfusate. Artif. Cells Blood Substit. Biotechnol. 1994, 22, 1463–1468. [Google Scholar] [CrossRef]
- Inci, I.; Arni, S.; Iskender, I.; Citak, N.; Rodriguez, J.M.; Weisskopf, M.; Opitz, I.; Weder, W.; Frauenfelder, T.; Krafft, M.P. Functional, Metabolic and Morphologic Results of Ex Vivo Donor Lung Perfusion with a Perfluorocarbon-Based Oxygen Carrier Nanoemulsion in a Large Animal Transplantation Model. Cells 2020, 9, 2501. [Google Scholar] [CrossRef]
- Kohno, M.; Ikeda, T.; Hashimoto, R.; Izumi, Y.; Watanabe, M.; Horinouchi, H.; Sakai, H.; Kobayashi, K.; Iwazaki, M. Acute 40% Exchange-Transfusion with Hemoglobin-Vesicles in a Mouse Pneumonectomy Model. PLoS ONE 2017, 12, e0178724. [Google Scholar] [CrossRef]
- Sakai, H. Overview of Potential Clinical Applications of Hemoglobin Vesicles (HbV) as Artificial Red Cells, Evidenced by Preclinical Studies of the Academic Research Consortium. J. Funct. Biomater. 2017, 8, 10. [Google Scholar] [CrossRef]
- Montagne, K.; Huang, H.; Ohara, K.; Matsumoto, K.; Mizuno, A.; Ohta, K.; Sakai, Y. Use of Liposome Encapsulated Hemoglobin as an Oxygen Carrier for Fetal and Adult Rat Liver Cell Culture. J. Biosci. Bioeng. 2011, 112, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Araki, J.; Sakai, H.; Takeuchi, D.; Kagaya, Y.; Tashiro, K.; Naito, M.; Mihara, M.; Narushima, M.; Iida, T.; Koshima, I. Normothermic Preservation of the Rat Hind Limb with Artificial Oxygen-Carrying Hemoglobin Vesicles. Transplantation 2015, 99, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Jahr, J.S.; Mackenzie, C.; Pearce, L.B.; Pitman, A.; Greenburg, A.G. HBOC-201 as an Alternative to Blood Transfusion: Efficacy and Safety Evaluation in a Multicenter Phase III Trial in Elective Orthopedic Surgery. J. Trauma Acute Care Surg. 2008, 64, 1484–1497. [Google Scholar] [CrossRef] [PubMed]
- Natanson, C.; Kern, S.J.; Lurie, P.; Banks, S.M.; Wolfe, S.M. Cell-Free Hemoglobin-Based Blood Substitutes and Risk of Myocardial Infarction and Death: A Meta-Analysis. JAMA 2008, 299, 2304–2312. [Google Scholar] [CrossRef] [PubMed]
- Taverne, Y.J.; de Wijs-Meijler, D.; te Lintel Hekkert, M.; Moon-Massat, P.F.; Dubé, G.P.; Duncker, D.J.; Merkus, D. Normalization of Hemoglobin-Based Oxygen Carrier-201 Induced Vasoconstriction: Targeting Nitric Oxide and Endothelin. J. Appl. Physiol. 2017, 122, 1227–1237. [Google Scholar] [CrossRef]
- Cabrales, P.; Friedman, J.M. HBOC Vasoactivity: Interplay between Nitric Oxide Scavenging and Capacity to Generate Bioactive Nitric Oxide Species. Antioxid. Redox Signal. 2013, 18, 2284–2297. [Google Scholar] [CrossRef]
- Dubé, G.P.; Pitman, A.N.; Mackenzie, C.F. Relative Efficacies of HBOC-201 and Polyheme to Increase Oxygen Transport Compared to Blood and Crystalloids. Shock 2019, 52, 100–107. [Google Scholar] [CrossRef]
- Mackenzie, C.F.; Dubé, G.P.; Pitman, A.; Zafirelis, M. Users Guide to Pitfalls and Lessons Learned about HBOC-201 during Clinical Trials, Expanded Access, and Clinical use in 1701 Patients. Shock 2019, 52, 92–99. [Google Scholar] [CrossRef]
- Mackenzie, C.F.; Pitman, A.N.; Hodgson, R.E.; Sussman, M.J.; Levien, L.J.; Jahr, J.S.; Greenburg, A.G. Are Hemoglobin-Based Oxygen Carriers being withheld because of Regulatory Requirement for Equivalence to Packed Red Blood Cells? Am. J. Ther. 2015, 22, e115–e121. [Google Scholar] [CrossRef]
- Mackenzie, C.F.; Moon-Massat, P.F.; Shander, A.; Javidroozi, M.; Greenburg, A.G. When Blood is Not an Option: Factors Affecting Survival After the use of a Hemoglobin-Based Oxygen Carrier in 54 Patients with Life-Threatening Anemia. Anesth. Analg. 2010, 110, 685–693. [Google Scholar] [CrossRef]
- Posluszny, J.A.; Napolitano, L.M. Hemoglobin-Based Oxygen Carrier for Traumatic Hemorrhagic Shock Treatment in a Jehovah’s Witness. Arch. Trauma. Res. 2016, 5, e30610. [Google Scholar] [CrossRef] [PubMed]
- De Vries, Y.; Matton, A.P.; Nijsten, M.W.; Werner, M.J.; van den Berg, A.P.; de Boer, M.T.; Buis, C.I.; Fujiyoshi, M.; de Kleine, R.H.; van Leeuwen, O.B. Pretransplant Sequential Hypo-and Normothermic Machine Perfusion of Suboptimal Livers Donated After Circulatory Death using a Hemoglobin-based Oxygen Carrier Perfusion Solution. Am. J. Transplant. 2019, 19, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Linberg, R.; Conover, C.D.; Shum, K.L. Hemoglobin Based Oxygen Carriers: How Much Methemoglobin is Too Much? Artif. Cells Blood Substit. Biotechnol. 1998, 26, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Rousselot, M.; Delpy, E.; Drieu La Rochelle, C.; Lagente, V.; Pirow, R.; Rees, J.; Hagege, A.; Le Guen, D.; Hourdez, S.; Zal, F. Arenicola Marina Extracellular Hemoglobin: A New Promising Blood Substitute. Biotechnol. J. Healthc. Nutr. Technol. 2006, 1, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Thuillier, R.; Dutheil, D.; Trieu, M.; Mallet, V.; Allain, G.; Rousselot, M.; Denizot, M.; Goujon, J.; Zal, F.; Hauet, T. Supplementation with a New Therapeutic Oxygen Carrier Reduces Chronic Fibrosis and Organ Dysfunction in Kidney Static Preservation. Am. J. Transplant. 2011, 11, 1845–1860. [Google Scholar] [CrossRef] [PubMed]
- Brüggenwirth, I.M.; van Leeuwen, O.B.; Müller, M.; Dutkowski, P.; Monbaliu, D.; Martins, P.N.; Porte, R.J.; de Meijer, V.E. The Importance of Adequate Oxygenation during Hypothermic Machine Perfusion. JHEP Rep. 2020, 3, 100194. [Google Scholar] [CrossRef]
- Kasil, A.; Giraud, S.; Couturier, P.; Amiri, A.; Danion, J.; Donatini, G.; Matillon, X.; Hauet, T.; Badet, L. Individual and Combined Impact of Oxygen and Oxygen Transporter Supplementation during Kidney Machine Preservation in a Porcine Preclinical Kidney Transplantation Model. Int. J. Mol. Sci. 2019, 20, 1992. [Google Scholar] [CrossRef]
- Hosgood, S.A.; Nicholson, M.L. The Role of Perfluorocarbon in Organ Preservation. Transplantation 2010, 89, 1169–1175. [Google Scholar] [CrossRef]
- Martins, P.N.; Berendsen, T.A.; Yeh, H.; Bruinsma, B.G.; Izamis, M.; Den Dries, S.O.; Gillooly, A.R.; Porte, R.; Yarmush, M.L.; Uygun, K. Oxygenated UW Solution Decreases ATP Decay and Improves Survival After Transplantation of DCD Liver Grafts. Transplantation 2019, 103, 363–370. [Google Scholar] [CrossRef]
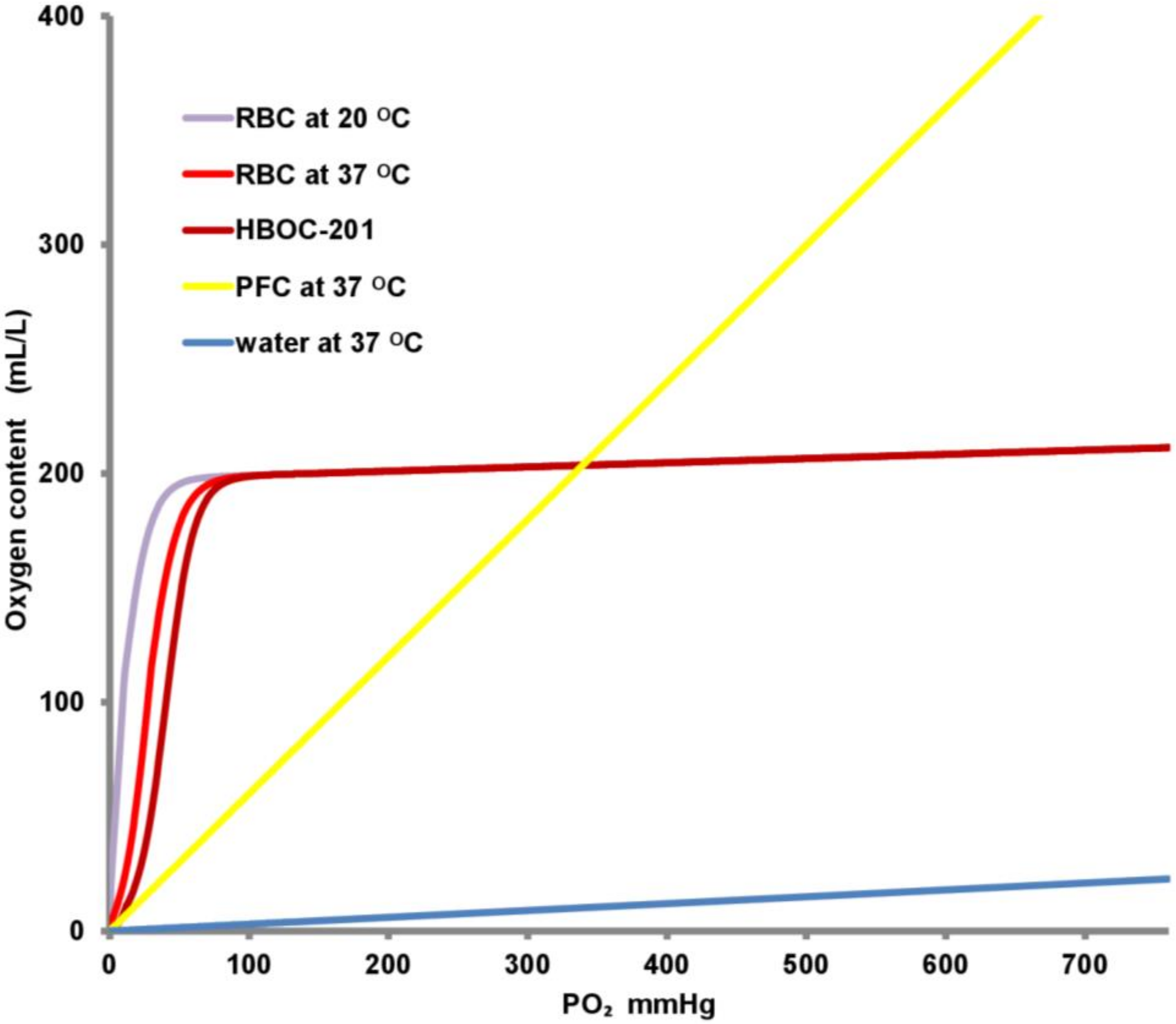
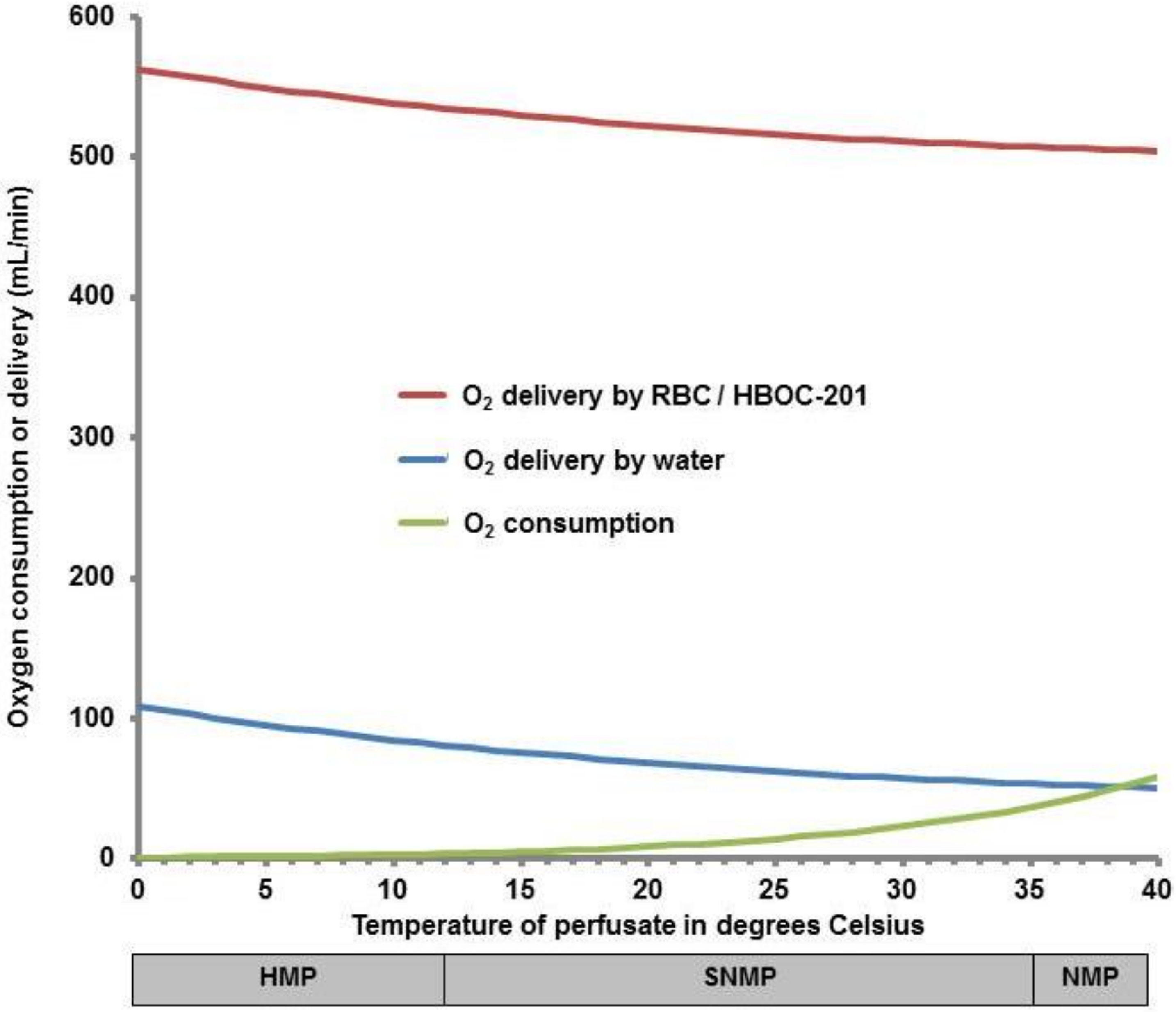

| Oxygen Carrier | Advantages | Disadvantages | Pharmacokinetics | Possible Toxicity in Humans |
|---|---|---|---|---|
| Hemoglobin in red blood cells (RBC) | Within its physiological microenvironment Human blood product Low methemoglobin production Dynamic shift of O2-hemoglobin dissociation curve | Immune-mediated phenomena Blood-borne infection transmission RBC hemolysis during hypothermic machine perfusion (HMP) Cross- matching difficulties Precious resource | T ½ = 115 days MW = 64 kDa [Hb] = 12–15 g/dL P50 = 27 mm Hg | ABO incompatibility |
| Hemoglobin-based oxygen carrier-201 [39] | Easy O2 release to tissue Sterile and pyrogen-free Large temperature range (4 °C–37 °C) Less viscous than RBC Long shelf life: three years Compatible with all blood types | Formation of methemoglobin Short half-life Systemic vasoconstriction Lower O2 affinity than RBC | T ½ = 20 h MW = ~250 kDa [Hb] = 13 g/dL P50 = 38–40 mm Hg | Systemic vasoconstriction |
| Hemoglobin vesicles [40,41] | Absence of RBC antigens Smaller than RBC sDo not generate colloid osmotic pressure Do not rupture Long shelf life: two years | Only used in animal models | T ½ = 2–3 days [Hb] 10 g/dL P50 = 9 mm Hg | Release of free Hb can cause renal toxicity |
| Hemarina M101 [42,43] | Preliminary evidence in static cold storage Large temperature range (4 °C–37 °C) Simple gradient release O2 High O2 affinity Non-immunogenic | Only used in preclinical HMP and clinically in static cold storage | MW = 3600 kDa P50 = 7 mm Hg | None reported |
| Perfluorocarbons [44,45] | High O2 solubility Inexpensive Obey Henry’s law O2 uptake and release insensitive to environment | Formulated as emulsion, which reduces O2 content Needs high PO2 to maximize O2 content | T ½ = 8–24 h Emulsion dependent | Visual loss |
| Water | Delivers sufficient O2 below 20 °C Inexpensive | O2 content decreases with higher temperature leading to a mismatch above 20 °C | T ½ = ∞ MW = 18 Da | None |
| Author and Year | Oxygen Carrier | Temperature Range | Machine | Sample Size n | Transplanted n | Species and Organ |
|---|---|---|---|---|---|---|
| Fontes et al. [53] AJT 2015 | HBOC-201 | SNMP | Liver Assist | 6 | 6 | Porcine livers |
| Sadowsky et al. [54] Front Pharmacol 2016 | HBOC-201 | SNMP | Liver Assist | 6 | 6 | Porcine livers |
| Matton et al. [55] Liver Transpl 2018 | HBOC-201 | NMP | Liver Assist | 24 | - | Human livers |
| Laing et al. [51] Transplantation 2017 | HBOC-201 | NMP | Liver Assist | 5 | - | Human livers |
| Boteon et al. [56] AJT 2018 | HBOC-201 | NMP | Liver Assist | 10 | - | Human livers |
| De Vries et al. [57] AJT 2019 | HBOC-201 | HMP to NMP | Liver Assist | 7 | 5 | Human livers |
| Aburawi et al. [58] AJT 2019 | HBOC-201 | NMP | Kidney Assist | 7 | - | Human kidneys |
| Vrselja et al. [59] Nature 2019 | HBOC-201 | NMP | - | 32 | - | Porcine brains |
| Van Leeuwen et al. [60] Ann Surg 2019 | HBOC-201 | HMP to NMP | Liver Assist | 16 | 11 | Human livers |
| Bhattacharjee et al. [61] Transplantation 2020 | HBOC-201 | SNMP | - | 5 | - | Porcine kidneys |
| Shonaka et al. [62] Transplantation 2018 | Hb-Vs | SNMP | - | 3 | - | Porcine livers |
| Shonaka et al. [63] Plos one 2019 | Hb-Vs | SNMP | - | 5 | - | Porcine livers |
| Brasile et al. [64] Biotechnol. 1994 | PFC | SNMP | - | 4 | - | Canine kidney |
| Inci et al. [65] Cells 2020 | PFC | SNMP | - | 11 | 4 | Porcine lungs |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bodewes, S.B.; van Leeuwen, O.B.; Thorne, A.M.; Lascaris, B.; Ubbink, R.; Lisman, T.; Monbaliu, D.; De Meijer, V.E.; Nijsten, M.W.N.; Porte, R.J. Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers. Int. J. Mol. Sci. 2021, 22, 235. https://doi.org/10.3390/ijms22010235
Bodewes SB, van Leeuwen OB, Thorne AM, Lascaris B, Ubbink R, Lisman T, Monbaliu D, De Meijer VE, Nijsten MWN, Porte RJ. Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers. International Journal of Molecular Sciences. 2021; 22(1):235. https://doi.org/10.3390/ijms22010235
Chicago/Turabian StyleBodewes, Silke B., Otto B. van Leeuwen, Adam M. Thorne, Bianca Lascaris, Rinse Ubbink, Ton Lisman, Diethard Monbaliu, Vincent E. De Meijer, Maarten W. N. Nijsten, and Robert J. Porte. 2021. "Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers" International Journal of Molecular Sciences 22, no. 1: 235. https://doi.org/10.3390/ijms22010235
APA StyleBodewes, S. B., van Leeuwen, O. B., Thorne, A. M., Lascaris, B., Ubbink, R., Lisman, T., Monbaliu, D., De Meijer, V. E., Nijsten, M. W. N., & Porte, R. J. (2021). Oxygen Transport during Ex Situ Machine Perfusion of Donor Livers Using Red Blood Cells or Artificial Oxygen Carriers. International Journal of Molecular Sciences, 22(1), 235. https://doi.org/10.3390/ijms22010235





