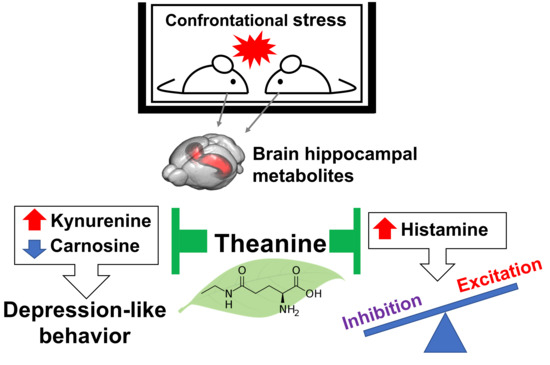
Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice
Abstract
1. Introduction
2. Results
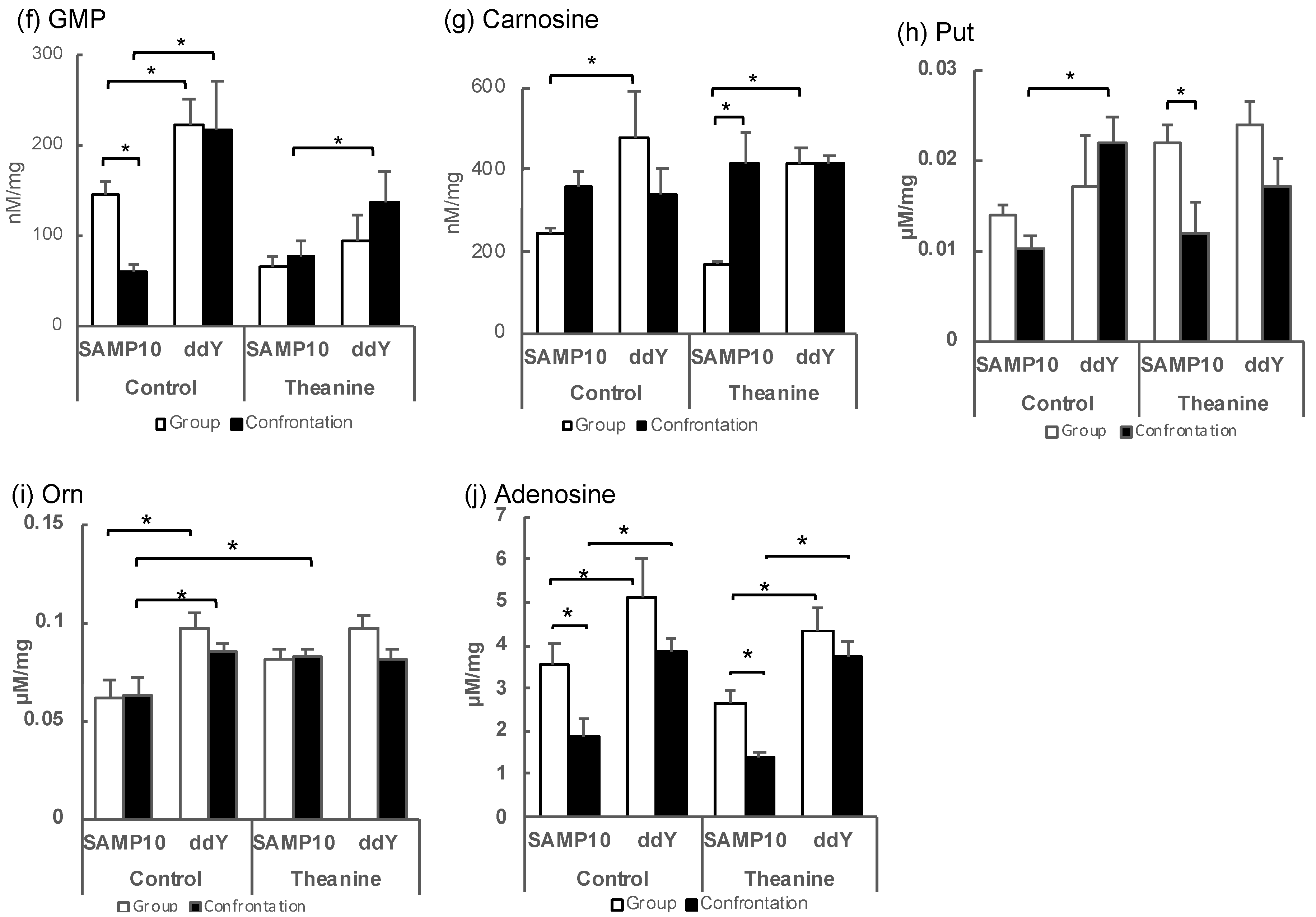
2.1. Effects of Theanine Ingestion on Brain Metabolites in Senescence-Accelerated Mouse Prone 10 (SAMP10) and ddY Mice Stressed by Confrontational Housing
2.2. Effect of Theanine Ingestion on Depression-Like Behavior
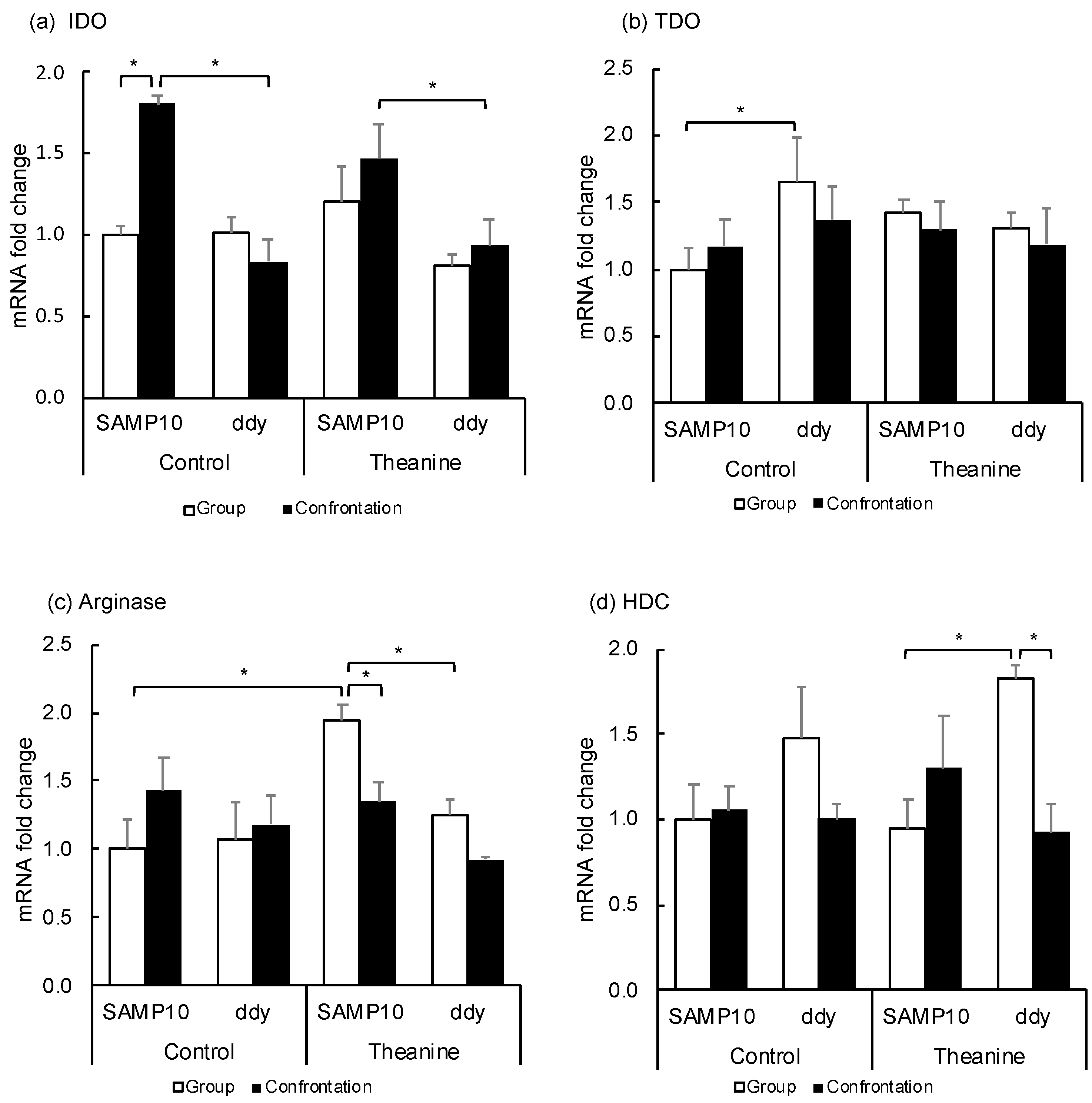
2.3. Effect of Theanine Ingestion on the Levels of Indoleamine/Tryptophan-2,3-dioxygenase, Arginase and Histidine Decarboxylase
3. Discussion
4. Materials and Methods
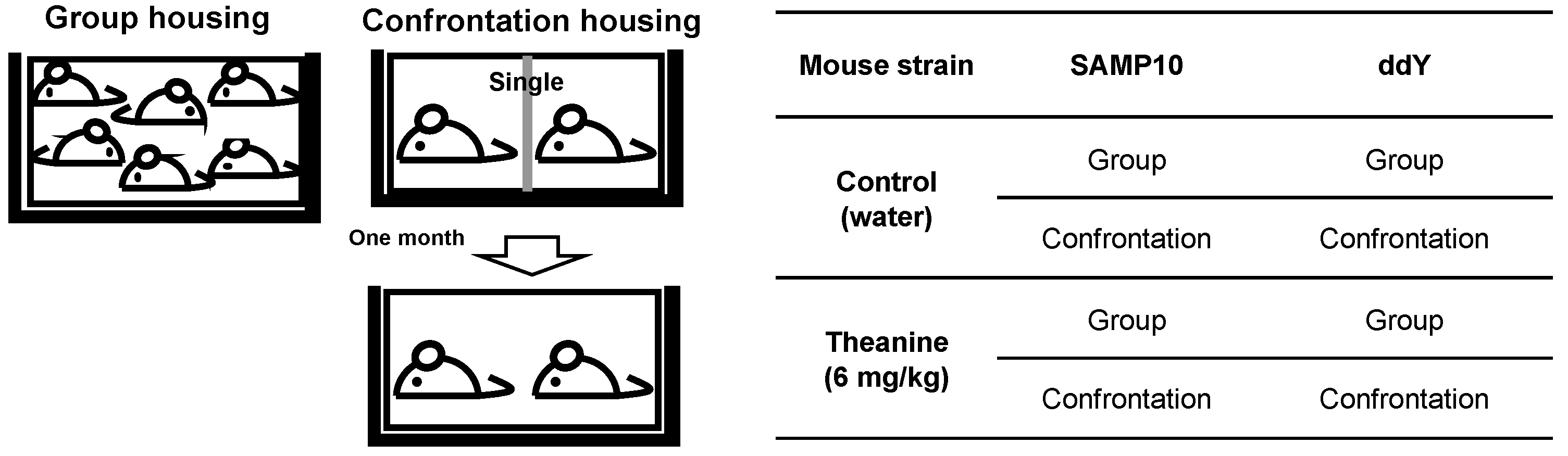
4.1. Animals and Theanine Preparation
4.2. Housing Conditions for Stress Experiments
4.3. Measurement of Metabolites by Ultrahigh Liquid Chromatography-Tandem Mass Spectrometry (UHPLC-MS/MS)
4.4. Principal Component Analysis
4.5. Tail-Suspension Test
4.6. Quantitative Real-Time Reverse Transcription PCR (qRT-PCR)
4.7. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Arg | Arginine |
| CMP | Cytidine monophosphate |
| GABA | γ-Aminobutyric acid |
| GMP | Guanosine monophosphate |
| HDC | Histidine decarboxylase |
| IDO | Indoleamine-2,3-dioxygenase |
| Kyn | Kynurenine |
| Lcn2 | Lipocalin 2 |
| NO | Nitric oxide |
| Npas4 | Neuronal PAS domain protein 4 |
| Orn | Ornithine |
| PC | Principal component |
| PCA | Principal component analysis |
| Put | Putrescine |
| SAMP10 | Senescence-accelerated mice prone 10 |
| Trp | Tryptophan |
| TDO | Tryptophan-2,3-dioxygenase |
| UHPLC-MS/MS | Ultra-high performance liquid chromatography-tandem mass spectrometry |
References
- Shimada, A.; Tsuzuki, M.; Keino, H.; Satoh, M.; Chiba, Y.; Saitoh, Y.; Hosokawa, M. Apical Vulnerability to Dendritic Retraction in Prefrontal Neurones of Ageing SAMP10 Mouse: A Model of Cerebral Degeneration. Neuropathol. Appl. Neurobiol. 2006, 32, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, M. Characteristics of age-related behavioral changes in senescence-accelerated mouse SAMP8 and SAMP10. Exp. Gerontol. 1997, 32, 139–148. [Google Scholar] [CrossRef]
- Unno, K.; Fujitani, K.; Takamori, N.; Takabayashi, F.; Maeda, K.; Miyazaki, H.; Tanida, N.; Iguchi, K.; Shimoi, K.; Hoshino, M. Theanine intake improves the shortened lifespan, cognitive dysfunction and behavioural depression that are induced by chronic psychosocial stress in mice. Free Radic. Res. 2011, 45, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Sumiyoshi, A.; Konishi, T.; Hayashi, M.; Taguchi, K.; Muguruma, Y.; Inoue, K.; Iguchi, K.; Nonaka, H.; Kawashima, R.; et al. Theanine, the Main Amino Acid in Tea, Prevents Stress-Induced Brain Atrophy by Modifying Early Stress Responses. Nutrients 2020, 12, 174. [Google Scholar] [CrossRef]
- Unno, K.; Iguchi, K.; Tanida, N.; Fujitani, K.; Takamori, N.; Yamamoto, H.; Ishii, N.; Nagano, H.; Nagashima, T.; Hara, A.; et al. Ingestion of theanine, an amino acid in tea, suppresses psychosocial stress in mice. Exp. Physiol. 2013, 98, 290–303. [Google Scholar] [CrossRef] [PubMed]
- Lopes Sakamoto, F.; Metzker Pereira Ribeiro, R.; Amador Bueno, A.; Oliveira Santos, H. Psychotropic effects of (L)-theanine and its clinical properties: From the management of anxiety and stress to a potential use in schizophrenia. Pharmacol. Res. 2019, 147, 104395. [Google Scholar] [CrossRef]
- Coutellier, L.; Beraki, S.; Ardestani, P.M.; Saw, N.L.; Shamloo, M. Npas4: A neuronal transcription factor with a key role in social and cognitive functions relevant to developmental disorders. PLoS ONE 2012, 7, e46604. [Google Scholar] [CrossRef]
- Suk, K. Lipocalin-2 as a therapeutic target for brain injury: An astrocentric perspective. Prog. Neurobiol. 2016, 144, 158–172. [Google Scholar] [CrossRef]
- Inoue, K.; Tsutsui, H.; Akatsu, H.; Hashizume, Y.; Matsukawa, N.; Yamamoto, T.; Toyo’oka, T. Metabolic profiling of Alzheimer’s disease brains. Sci. Rep. 2013, 3, 2364. [Google Scholar] [CrossRef]
- Anacker, C.; Hen, R. Adult hippocampal neurogenesis and cognitive flexibility—Linking memory and mood. Nat. Rev. Neurosci. 2017, 18, 335–346. [Google Scholar] [CrossRef]
- Gonçalves, J.T.; Schafer, S.T.; Gage, F.H. Adult neurogenesis in the hippocampus: From stem cells to behavior. Cell 2016, 167, 897–914. [Google Scholar] [CrossRef] [PubMed]
- Lieberwirth, C.; Pan, Y.; Liu, Y.; Zhang, Z.; Wang, Z. Hippocampal adult neurogenesis: Its regulation and potential role in spatial learning and memory. Brain Res. 2016, 1644, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.; Koike, H.; Ibi, D.; Toth, E.; Mizoguchi, H.; Nitta, A.; Yoneyama, M.; Ogita, K.; Yoneda, Y.; Nabeshima, T.; et al. Chronic restraint stress impairs neurogenesis and hippocampus-dependent fear memory in mice: Possible involvement of a brain-specific transcription factor Npas4. J. Neurochem. 2010, 114, 1840–1851. [Google Scholar] [CrossRef] [PubMed]
- Hollands, C.; Tobin, M.K.; Hsu, M.; Musaraca, K.; Yu, T.S.; Mishra, R.; Kernie, S.G.; Lazarov, O. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer’s disease by compromising hippocampal inhibition. Mol. Neurodegener. 2017, 12, 64. [Google Scholar] [CrossRef]
- Erickson, J.D.; De Gois, S.; Varoqui, H.; Schafer, M.K.; Weihe, E. Activity-dependent regulation of vesicular glutamate and GABA transporters: A means to scale quantal size. Neurochem. Int. 2006, 48, 643–649. [Google Scholar] [CrossRef]
- Purkayastha, P.; Malapati, A.; Yogeeswari, P.; Sriram, D. A review on GABA/glutamate pathway for therapeutic intervention of ASD and ADHD. Curr. Med. Chem. 2015, 22, 1850–1859. [Google Scholar] [CrossRef]
- Martisova, E.; Solas, M.; Horrillo, I.; Ortega, J.E.; Meana, J.J.; Tordera, R.M.; Ramírez, M.J. Long lasting effects of early-life stress on glutamatergic/GABAergic circuitry in the rat hippocampus. Neuropharmacology 2012, 62, 1944–1953. [Google Scholar] [CrossRef]
- Pérez, M.Á.; Peñaloza-Sancho, V.; Ahumada, J.; Fuenzalida, M.; Dagnino-Subiabre, A. n-3 Polyunsaturated fatty acid supplementation restored impaired memory and GABAergic synaptic efficacy in the hippocampus of stressed rats. Nutr. Neurosci. 2017, 8, 1–14. [Google Scholar] [CrossRef]
- Sibbe, M.; Kulik, A. GABAergic regulation of adult hippocampal neurogenesis. Mol. Neurobiol. 2017, 54, 5497–5510. [Google Scholar] [CrossRef]
- Yun, J.; Nagai, T.; Furukawa-Hibi, Y.; Kuroda, K.; Kaibuchi, K.; Greenberg, M.E.; Yamada, K. Neuronal Per Arnt Sim (PAS) domain protein 4 (NPAS4) regulates neurite outgrowth and phosphorylation of synapsin I. J. Biol. Chem. 2013, 288, 2655–2664. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, I.T. Principal Component Analysis; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Li, C.C.; Jiang, N.; Gan, L.; Zhao, M.J.; Chang, Q.; Liu, X.M.; Pan, R.L. Peripheral and cerebral abnormalities of the tryptophan metabolism in the depression-like rats induced by chronic unpredicted mild stress. Neurochem. Int. 2020, 138, 104771. [Google Scholar] [CrossRef] [PubMed]
- Cattelan Souza, L.; de Brito, M.L.O.; Jesse, C.R.; Boeira, S.P.; de Gomes, M.G.; Goes, A.T.R.; Fabbro, L.D.; Machado, F.R.; Prigol, M.; Nogueira, C.W. Involvement of kynurenine pathway in depressive-like behaviour induced by nandrolone decanoate in mice. Steroids 2020, 164, 108727. [Google Scholar] [CrossRef]
- Zhang, Q.; Sun, Y.; He, Z.; Xu, Y.; Li, X.; Ding, J.; Lu, M.; Hu, G. Kynurenine regulates NLRP2 inflammasome in astrocytes and its implications in depression. Brain Behav. Immun. 2020, 88, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, D.; Song, P.; Zou, M.H. Tryptophan-kynurenine pathway is dysregulated in inflammation, and immune activation. Front. Biosci. 2015, 20, 1116–1143. [Google Scholar] [CrossRef] [PubMed]
- Hipkiss, A.R. Possible Benefit of Dietary Carnosine towards Depressive Disorders. Aging Dis. 2015, 6, 300–303. [Google Scholar] [CrossRef]
- Tomonaga, S.; Yamane, H.; Onitsuka, E.; Yamada, S.; Sato, M.; Takahata, Y.; Morimatsu, F.; Furuse, M. Carnosine-induced antidepressant-like activity in rats. Pharmacol. Biochem. Behav. 2008, 89, 627–632. [Google Scholar] [CrossRef]
- Yin, C.; Gou, L.; Liu, Y.; Yin, X.; Zhang, L.; Jia, G.; Zhuang, X. Antidepressant-like effects of L-theanine in the forced swim and tail suspension tests in mice. Phytother. Res. 2011, 25, 1636–1639. [Google Scholar] [CrossRef]
- Ogawa, S.; Ota, M.; Ogura, J.; Kato, K.; Kunugi, H. Effects of L-theanine on anxiety-like behavior, cerebrospinal fluid amino acid profile, and hippocampal activity in Wistar Kyoto rats. Psychopharmacology 2018, 235, 37–45. [Google Scholar] [CrossRef]
- Shen, M.; Yang, Y.; Wu, Y.; Zhang, B.; Wu, H.; Wang, L.; Tang, H.; Chen, J. L-theanine ameliorate depressive-like behavior in a chronic unpredictable mild stress rat model via modulating the monoamine levels in limbic-cortical-striatal-pallidal-thalamic-circuit related brain regions. Phytother. Res. 2019, 33, 412–421. [Google Scholar] [CrossRef]
- Spiegel, I.; Mardinly, A.R.; Gabel, H.W.; Bazinet, J.E.; Couch, C.H.; Tzeng, C.P.; Harmin, D.A.; Greenberg, M.E. Npas4 regulates excitatory-inhibitory balance within neural circuits through cell-type-specific gene programs. Cell 2014, 157, 1216–1229. [Google Scholar] [CrossRef]
- Brown, R.E.; Stevens, D.R.; Haas, H.L. The physiology of brain histamine. Prog. Neurobiol. 2001, 63, 637–672. [Google Scholar] [CrossRef]
- Thakkar, M.M. Histamine in the regulation of wakefulness. Sleep Med. Rev. 2011, 15, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Rao, T.P.; Ozeki, M.; Juneja, L.R. In Search of a Safe Natural Sleep Aid. J. Am. Coll. Nutr. 2015, 34, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Hidese, S.; Ota, M.; Wakabayashi, C.; Noda, T.; Ozawa, H.; Okubo, T.; Kunugi, H. Effects of chronic l-theanine administration in patients with major depressive disorder: An open-label study. Acta Neuropsychiatr. 2017, 29, 72–79. [Google Scholar] [CrossRef]
- Pow, D.V.; Baldridge, W.; Crook, D.K. Activity-dependent transport of GABA analogues into specific cell types demonstrated at high resolution using a novel immunocytochemical strategy. Neuroscience 1996, 73, 1129–1143. [Google Scholar] [CrossRef]
- Zullo, J.M.; Drake, D.; Aron, L.; O’Hern, P.; Dhamne, S.C.; Davidsohn, N.; Mao, C.A.; Klein, W.H.; Rotenberg, A.; Bennett, D.A.; et al. Regulation of lifespan by neural excitation and REST. Nature 2019, 574, 359–364. [Google Scholar] [CrossRef]
- Cannizzaro, C.; Plescia, F.; Gagliano, M.; Cannizzaro, G.; Mantia, G.; La Barbera, M.; Provenzano, G.; Cannizzaro, E. Perinatal exposure to 5-methoxytryptamine, behavioural-stress reactivity and functional response of 5-HT1A receptors in the adolescent rat. Behav. Brain Res. 2008, 186, 98–106. [Google Scholar] [CrossRef]
- Vemula, P.; Jing, Y.; Zhang, H.; Hunt, J.B., Jr.; Sandusky-Beltran, L.A.; Lee, D.C.; Liu, P. Altered brain arginine metabolism in a mouse model of tauopathy. Amino Acids 2019, 51, 513–528. [Google Scholar] [CrossRef]
- Rushaidhi, M.; Jing, Y.; Kennard, J.T.; Collie, N.D.; Williams, J.M.; Zhang, H.; Liu, P. Aging affects L-arginine and its metabolites in memory-associated brain structures at the tissue and synaptoneurosome levels. Neuroscience 2012, 209, 21–31. [Google Scholar] [CrossRef]
- Suenaga, R.; Yamane, H.; Tomonaga, S.; Asechi, M.; Adachi, N.; Tsuneyoshi, Y.; Kurauchi, I.; Sato, H.; Denbow, D.M.; Furuse, M. Central L-arginine reduced stress responses are mediated by L-ornithine in neonatal chicks. Amino Acids 2008, 35, 107–113. [Google Scholar] [CrossRef]
- Unno, K.; Hara, A.; Nakagawa, A.; Iguchi, K.; Ohshio, M.; Morita, A.; Nakamura, Y. Anti-stress effects of drinking green tea with lowered caffeine and enriched theanine, epigallocatechin and arginine on psychosocial stress induced adrenal hypertrophy in mice. Phytomedicine 2016, 23, 1365–1374. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, J.A.; Sebastião, A.M. Modulation and metamodulation of synapses by adenosine. Acta Physiol. (Oxf) 2010, 199, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Sebastião, A.M.; Ribeiro, J.A. Neuromodulation and metamodulation by adenosine: Impact and subtleties upon synaptic plasticity regulation. Brain Res. 2015, 1621, 102–113. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, F.F.; Altenhofen, S.; Kist, L.W.; Leite, C.E.; Bogo, M.R.; Cognato, G.P.; Bonan, C.D. Unpredictable Chronic Stress Alters Adenosine Metabolism in Zebrafish Brain. Mol. Neurobiol. 2016, 53, 2518–2528. [Google Scholar] [CrossRef]
- Hidese, S.; Ogawa, S.; Ota, M.; Ishida, I.; Yasukawa, Z.; Ozeki, M.; Kunugi, H. Effects of L-Theanine Administration on Stress-Related Symptoms and Cognitive Functions in Healthy Adults: A Randomized Controlled Trial. Nutrients 2019, 11, 2362. [Google Scholar] [CrossRef]
- Muguruma, Y.; Tsutsui, H.; Noda, T.; Akatsu, H.; Inoue, K. Widely targeted metabolomics of Alzheimer’s disease postmortem cerebrospinal fluid based on 9-fluorenylmethyl chloroformate derivatized ultra-high performance liquid chromatography tandem mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2018, 1091, 53–66. [Google Scholar] [CrossRef]
- Konishi, T. Microarray test results should not be compensated for multiplicity of gene contents. BMC Syst. Biol. 2011, 5, S6. [Google Scholar] [CrossRef]
- Miyamoto, M.; Takahashi, H.; Ohta, H.; Sakamoto, J. Animal model of brain aging: Senescence-accelerated mouse (SAM). CNS Drug Rev. 1998, 4, 361–375. [Google Scholar] [CrossRef]
- Larkin, P.B.; Sathyasaikumar, K.V.; Notarangelo, F.M.; Funakoshi, H.; Nakamura, T.; Schwarcz, R.; Muchowski, P.J. Tryptophan 2,3-dioxygenase and indoleamine 2,3-dioxygenase 1 make separate, tissue-specific contributions to basal and inflammation-induced kynurenine pathway metabolism in mice. Biochim. Biophys. Acta 2016, 1860, 2345–2354. [Google Scholar] [CrossRef]
- Sakai, K.; Sanders, K.M.; Youssef, M.R.; Yanushefski, K.M.; Jensen, L.; Yosipovitch, G.; Akiyama, T. Mouse model of imiquimod-induced psoriatic itch. Pain 2016, 157, 2536–2543. [Google Scholar] [CrossRef]
- Perego, C.; Fumagalli, S.; Zanier, E.R.; Carlino, E.; Panini, N.; Erba, E.; De Simoni, M.G. Macrophages are essential for maintaining a M2 protective response early after ischemic brain injury. Neurobiol. Dis. 2016, 96, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Konishi, T. A thermodynamic model of transcriptome formation. Nucleic Acids Res. 2005, 33, 6587–6592. [Google Scholar] [CrossRef] [PubMed]
- Konishi, T. Principal component analysis for designed experiments. BMC Bioinform. 2015, 16 (Suppl. 18), S7. [Google Scholar] [CrossRef] [PubMed]


| Mouse | Condition | Treatment | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|---|---|
| SAMP10 | Group | Control | –0.07338 | 0.03785 | –0.00872 | –0.00107 |
| –0.03254 | –0.00421 | –0.02051 | –0.00247 | |||
| –0.07107 | 0.03476 | –0.00459 | –0.00835 | |||
| –0.03904 | –0.00196 | –0.00954 | –0.01278 | |||
| Theanine | –0.07666 | 0.02389 | 0.00652 | 0.01317 | ||
| –0.06840 | 0.02850 | –0.00088 | 0.00442 | |||
| –0.07974 | 0.02936 | 0.01287 | 0.00789 | |||
| Confrontation | Control | –0.04038 | 0.00882 | 0.01114 | –0.02924 | |
| –0.04848 | –0.01130 | 0.00611 | –0.01154 | |||
| –0.06143 | 0.00294 | 0.01720 | 0.00465 | |||
| –0.00513 | –0.06122 | 0.00394 | 0.00318 | |||
| Theanine | 0.00952 | –0.10804 | –0.01076 | 0.00681 | ||
| –0.00350 | –0.05210 | 0.01452 | 0.02649 | |||
| 0.01278 | –0.11432 | –0.00996 | –0.01817 | |||
| –0.02355 | –0.03912 | 0.00415 | –0.00177 | |||
| ddY | Group | Control | 0.01670 | 0.00225 | –0.01376 | –0.02015 |
| 0.01147 | 0.00520 | –0.01819 | –0.00116 | |||
| 0.02528 | 0.00516 | –0.01773 | 0.01430 | |||
| Theanine | –0.00682 | 0.00349 | 0.00519 | 0.01326 | ||
| –0.04108 | –0.01144 | –0.00352 | 0.02319 | |||
| 0.02797 | 0.00704 | 0.00463 | 0.00567 | |||
| 0.17347 | 0.04502 | 0.02486 | 0.01496 | |||
| Confrontation | Control | 0.09426 | 0.02753 | –0.00925 | 0.01045 | |
| 0.04019 | 0.00548 | –0.02618 | –0.01805 | |||
| 0.09584 | 0.02586 | 0.01472 | 0.00620 | |||
| –0.01268 | –0.03017 | –0.02188 | 0.02948 | |||
| Theanine | 0.07307 | 0.01096 | 0.02558 | –0.03502 | ||
| 0.06694 | 0.01848 | 0.00251 | 0.00074 | |||
| 0.04413 | 0.00230 | –0.02052 | –0.02141 | |||
| 0.14107 | 0.03713 | 0.02662 | 0.01047 |
| Metaborites | PC1 | PC2 |
|---|---|---|
| Kynurenine | −0.12390 | 0.02227 |
| Histamine | −0.06985 | 0.00491 |
| 5-Methoxytryptamine | −0.03622 | −0.08937 |
| 2.4-Diaminobutyric.acid | −0.03085 | −0.00003 |
| Histidinol | −0.02561 | 0.00316 |
| 5-Aminovaleric.acid | −0.02370 | −0.00610 |
| Cadaverine | −0.02067 | −0.00384 |
| Diacetyl spermidine | −0.01859 | 0.00364 |
| 3-Methoxyanthranilate | −0.01750 | −0.00783 |
| Leucine | −0.00513 | 0.00335 |
| Methionine | −0.00364 | 0.00280 |
| Proline | −0.00277 | 0.00327 |
| Creatinine | −0.00269 | −0.00184 |
| 2-Aminoadipate | −0.00263 | −0.00433 |
| Aspartic acid | −0.00229 | −0.00076 |
| Arginine | −0.00213 | 0.00345 |
| Cystine | −0.00201 | 0.00122 |
| 2-Aminobutyric.acid | −0.00160 | 0.00033 |
| Valine | −0.00124 | 0.00227 |
| Phenylalanine | −0.00119 | 0.00302 |
| Alanine | −0.00084 | 0.00084 |
| β-Alanine | −0.00064 | 0.00208 |
| NG-monomethyl-arginine | −0.00028 | −0.00849 |
| Isoleucine | −0.00019 | 0.00310 |
| Glutamine | −0.00008 | −0.00027 |
| Histidine | 0.00037 | 0.00187 |
| Hypoxanthine | 0.00103 | 0.00440 |
| Glutamic acid | 0.00132 | −0.00123 |
| Spermidine | 0.00143 | −0.00015 |
| Glutathione reduced | 0.00146 | −0.00238 |
| Serine | 0.00162 | −0.00050 |
| CMP | 0.00262 | 0.00253 |
| Adenine | 0.00368 | −0.01675 |
| Adenosine | 0.00482 | −0.00904 |
| Ornithine | 0.00487 | −0.00004 |
| Putrescine | 0.00668 | −0.01895 |
| Carnosine | 0.00901 | 0.00522 |
| GMP | 0.01038 | −0.00718 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unno, K.; Muguruma, Y.; Inoue, K.; Konishi, T.; Taguchi, K.; Hasegawa-Ishii, S.; Shimada, A.; Nakamura, Y. Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice. Int. J. Mol. Sci. 2021, 22, 193. https://doi.org/10.3390/ijms22010193
Unno K, Muguruma Y, Inoue K, Konishi T, Taguchi K, Hasegawa-Ishii S, Shimada A, Nakamura Y. Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice. International Journal of Molecular Sciences. 2021; 22(1):193. https://doi.org/10.3390/ijms22010193
Chicago/Turabian StyleUnno, Keiko, Yoshio Muguruma, Koichi Inoue, Tomokazu Konishi, Kyoko Taguchi, Sanae Hasegawa-Ishii, Atsuyoshi Shimada, and Yoriyuki Nakamura. 2021. "Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice" International Journal of Molecular Sciences 22, no. 1: 193. https://doi.org/10.3390/ijms22010193
APA StyleUnno, K., Muguruma, Y., Inoue, K., Konishi, T., Taguchi, K., Hasegawa-Ishii, S., Shimada, A., & Nakamura, Y. (2021). Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice. International Journal of Molecular Sciences, 22(1), 193. https://doi.org/10.3390/ijms22010193