Neurological Disorders and Risk of Arrhythmia
Abstract
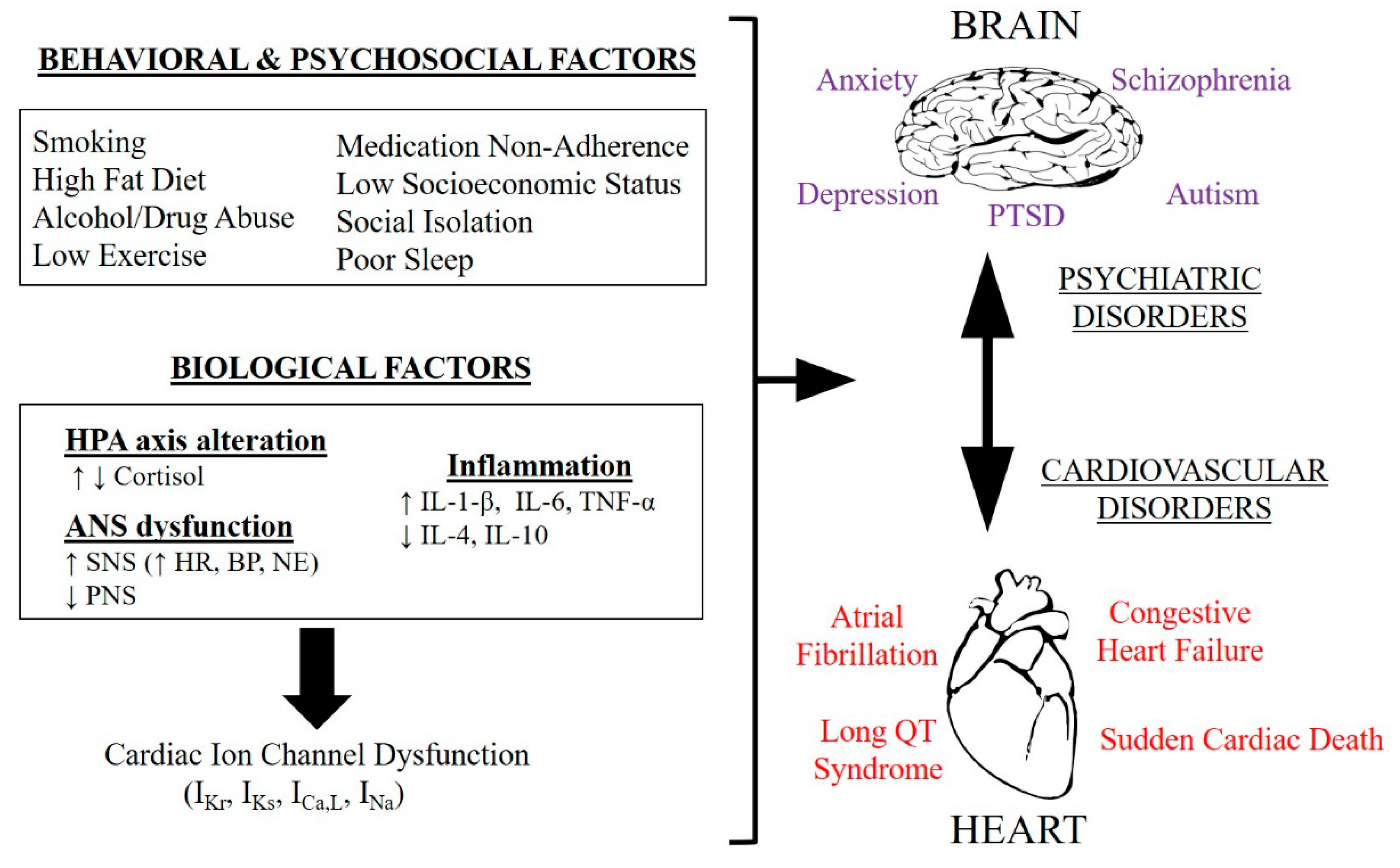
1. Introduction
2. Psychiatric Disorders and Cardiovascular Diseases
3. Common Molecular Mechanisms of Cardiovascular Disorders Acquired in Mental Disorders
4. Evidence for a Potential Role for Ion Channels Linking Neuropsychiatric Disorders and CVD
5. hERG and IKr Channel Modulation as an Important Pathological Link between Neuronal Disorders and Vulnerability to Arrhythmias
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Disclosures
Conflicts of Interest
Abbreviations
| ANS | Autonomic Nervous System |
| ASD | Autism Spectrum Disorder |
| CHD | Congenital Heart Diseases |
| CVD | Cardiovascular Disorders |
| hERG | Human Ether-A-Go-Go-Related Gene |
| HPA axis | Hypothalamic–Pituitary–Adrenal Axis |
| HRV | Heart Rate Variability |
| ICa,L | L-Type Calcium Current |
| IKr | Rapid Delayed Rectifier Potassium Current |
| Ito | Transient Outward Potassium Current |
| IL-6 | Interleukin 6 |
| IL-1β | Interleukin 1β |
| LQTS | Long QT Syndrome |
| PNS | Parasympathetic Nervous System |
| PTSD | Post-Traumatic Stress Disorder |
| QTc | Corrected QT interval |
| SCD | Sudden Cardiac Death |
| SNP | Single Nucleotide Polymorphisms |
| SNS | Sympathetic Nervous System |
| SQTS | Short QT Syndrome |
| SSRI | Selective Serotonin Reuptake Inhibitors |
| TNF-α | Tumor Necrosis Factor Alpha |
| TS | Timothy Syndrome |
References
- Sansone, R.A.; Sansone, L.A. Psychiatric disorders: A global look at facts and figures. Psychiatry 2010, 7, 16–19. [Google Scholar] [PubMed]
- Correll, C.U.; Solmi, M.; Veronese, N.; Bortolato, B.; Rosson, S.; Santonastaso, P.; Thapa-Chhetri, N.; Fornaro, M.; Gallicchio, D.; Collantoni, E.; et al. Prevalence, incidence and mortality from cardiovascular disease in patients with pooled and specific severe mental illness: A large-scale meta-analysis of 3,211,768 patients and 113,383,368 controls. World Psychiatry 2017, 16, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Rosengren, A.; Hawken, S.; Ôunpuu, S.; Sliwa, K.; Zubaid, M.; Almahmeed, W.; Blackett, K.N.; Sitthi-Amorn, C.; Sato, H.; Yusufali, A. Association of psychosocial risk factors with risk of acute myocardial infarction in 11 119 cases and 13 648 controls from 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 953–962. [Google Scholar] [CrossRef]
- Ratajczak, M.Z.; Pedziwiatr, D.; Cymer, M.; Kucia, M.; Kucharska-Mazur, J.; Samochowiec, J. Sterile Inflammation of Brain, due to Activation of Innate Immunity, as a Culprit in Psychiatric Disorders. Front Psychiatry 2018, 9, 60. [Google Scholar] [CrossRef]
- Hamer, M.; Molloy, G.J.; Stamatakis, E. Psychological distress as a risk factor for cardiovascular events: Pathophysiological and behavioral mechanisms. J. Am. Coll. Cardiol. 2008, 52, 2156–2162. [Google Scholar] [CrossRef]
- Whooley, M. Depressive Symptoms, Health Behaviors, and Risk of Cardiovascular Events in Patients with Coronary Heart Disease. J. Am. Med. Assoc. 2008, 300, 2379–2388. [Google Scholar] [CrossRef]
- Correll, C.U.; Detraux, J.; De Lepeleire, J.; De Hert, M. Effects of antipsychotics, antidepressants and mood stabilizers on risk for physical diseases in people with schizophrenia, depression and bipolar disorder. World Psychiatry 2015, 14, 119–136. [Google Scholar] [CrossRef]
- Yu, Z.-H.; Jiang, H.-Y.; Shao, L.; Zhou, Y.-Y.; Shi, H.-Y.; Ruan, B. Use of antipsychotics and risk of myocardial infarction: A systematic review and meta-analysis. Br. J. Clin. Pharmacol. 2016, 82, 624–632. [Google Scholar] [CrossRef]
- Murray-Thomas, T.; Jones, M.E.; Patel, D.; Brunner, E.; Shatapathy, C.C.; Motsko, S.; Van Staa, T.P. Risk of Mortality (Including Sudden Cardiac Death) and Major Cardiovascular Events in Atypical and Typical Antipsychotic Users: A Study with the General Practice Research Database. Cardiovasc. Psychiatry Neurol. 2013, 2013, 1–15. [Google Scholar] [CrossRef]
- Blalock, J.A.; Joiner, T.E. Interaction of Cognitive Avoidance Coping and Stress in Predicting Depres-sion/Anxiety. Cogn. Ther. Res. 2000, 24, 47–65. [Google Scholar] [CrossRef]
- Aromaa, A.; Raitasalo, R.; Reunanen, A.; Impivaara, O.; Heliovaara, M.; Knekt, P.; Lehtinet, V.; Joukamaa, M.; Maatela, J. Depression and cardiovascular diseases. Acta Psychiatr. Scand. 1994, 89, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, D.; Cohen, B.E. Posttraumatic stress disorder and cardiovascular disease. Prog. Cardiovasc. Dis. 2013, 55, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, I.; Sparrow, D.; Vokonas, P.S.; Weiss, S.T. Symptoms of anxiety and risk of coronary heart disease. The Normative Aging Study. Circulation 1994, 90, 2225–2229. [Google Scholar] [CrossRef] [PubMed]
- Westman, J.; Eriksson, S.V.; Gissler, M.; Hällgren, J.; Prieto, M.L.; Bobo, W.V.; Frye, M.A.; Erlinge, D.; Alfredsson, L.; Ösby, U. Increased cardiovascular mortality in people with schizophrenia: A 24-year national register study. Epidemiol. Psychiatr. Sci. 2017, 27, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Tyler, C.V.; Schramm, S.C.; Karafa, M.; Tang, A.S.; Jain, A.K. Chronic disease risks in young adults with autism spectrum disorder: Forewarned is forearmed. Am. J. Intellect. Dev. Disabil. 2011, 116, 371–380. [Google Scholar] [CrossRef]
- Lane, D.; Carroll, D.; Ring, C.; Beevers, D.G.; Lip, G.Y.H. The prevalence and persistence of depression and anxiety following myocardial infarction. Br. J. Health Psychol. 2002, 7, 11–21. [Google Scholar] [CrossRef]
- Lett, H.S.; Blumenthal, J.A.; Babyak, M.A.; Sherwood, A.; Strauman, T.; Robins, C.; Newman, M.F. Depression as a risk factor for coronary artery disease: Evidence, mechanisms, and treatment. Psychosom. Med. 2004, 66, 305–315. [Google Scholar]
- Rugulies, R. Depression as a predictor for coronary heart disease. Am. J. Prev. Med. 2002, 23, 51–61. [Google Scholar] [CrossRef]
- Moser, D.K.; Dracup, K. Is Anxiety Early after Myocardial Infarction Associated with Subsequent Ischemic and Arrhythmic Events? Psychosom. Med. 1996, 58, 395–401. [Google Scholar] [CrossRef]
- Watkins, L.L.; Blumenthal, J.A.; Davidson, J.R.T.; Babyak, M.A.; McCants, C.B.; Sketch, M.H. Phobic Anxiety, Depression, and Risk of Ventricular Arrhythmias in Patients with Coronary Heart Disease. Psychosom. Med. 2006, 68, 651–656. [Google Scholar] [CrossRef]
- Rasmussen, K.; Ravnsbaek, J.; Funch-Jensen, P.; Bagger, J.P. Oesophageal spasm in patients with coronary artery spasm. Lancet 1986, 1, 174–176. [Google Scholar] [CrossRef]
- Hendriks, M.L.; Allaart, C.P.; Bronzwaer, J.G.; Res, J.J.; De Cock, C.C. Recurrent ventricular fibrillation caused by coronary artery spasm leading to implantable cardioverter defibrillator implantation. Europace 2008, 10, 1456–1457. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, Y.; Tamura, Y.; Suzuki, K.; Tanabe, Y.; Yamaguchi, T.; Fujita, T.; Yamazoe, M.; Aizawa, Y.; Shibata, A. Coronary artery spasm is a major cause of sudden cardiac arrest in survivors without underly-ing heart disease. Coron. Artery Dis. 1993, 4, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Akosile, W.; Young, R.; Lawford, B.; Voisey, J.; Colquhoun, D. PTSD symptoms associated with myocardial infarction: Practical clinical implications. Australas. Psychiatry 2017, 26, 60–64. [Google Scholar] [CrossRef]
- Cohen, B.E.; Marmar, C.; Ren, L.; Bertenthal, D.; Seal, K.H. Association of cardiovascular risk factors with mental health diagnoses in Iraq and Afghani-stan war veterans using VA health care. J. Am. Med. Assoc. 2009, 302, 489–492. [Google Scholar] [CrossRef]
- Kubzansky, L.; Koenen, K.C.; Spiro, A.; Vokonas, P.S.; Sparrow, D. Prospective Study of Posttraumatic Stress Disorder Symptoms and Coronary Heart Disease in the Normative Aging Study. Arch. Gen. Psychiatry 2007, 64, 109–116. [Google Scholar] [CrossRef]
- Boscarino, J. A Prospective Study of PTSD and Early-Age Heart Disease Mortality Among Vietnam Veterans: Implications for Surveillance and Prevention. Psychosom. Med. 2008, 70, 668–676. [Google Scholar] [CrossRef]
- Edmondson, D.; Kronish, I.M.; Shaffer, J.A.; Falzon, L.; Burg, M.M. Posttraumatic stress disorder and risk for coronary heart disease: A meta-analytic review. Am. Heart J. 2013, 166, 806–814. [Google Scholar] [CrossRef]
- Edmondson, D.; Richardson, S.; Falzon, L.; Davidson, K.W.; Mills, M.A.; Neria, Y. Correction: Posttraumatic stress disorder prevalence and risk of recurrence in acute coronary syn-drome patients: A meta-analytic review. PLoS ONE 2012, 7, e38915. [Google Scholar] [CrossRef]
- Kronish, I.M.; Edmondson, D.; Goldfinger, J.Z.; Fei, K.; Horowitz, C.R. Posttraumatic Stress Disorder and Adherence to Medications in Survivors of Strokes and Transient Ischemic Attacks. Stroke 2012, 43, 2192–2197. [Google Scholar] [CrossRef]
- Roberge, M.-A.; Dupuis, G.; Marchand, A. Post-traumatic stress disorder following myocardial infarction: Prevalence and risk factors. Can. J. Cardiol. 2010, 26, e170–e175. [Google Scholar] [CrossRef]
- Fan, Z.; Wu, Y.; Shen, J.; Ji, T.; Zhan, R. Schizophrenia and the risk of cardiovascular diseases: A meta-analysis of thirteen cohort studies. J. Psychiatr. Res. 2013, 47, 1549–1556. [Google Scholar] [CrossRef] [PubMed]
- Laursen, T.M.; Wahlbeck, K.; Hällgren, J.; Westman, J.; Ösby, U.; Alinaghizadeh, H.; Gissler, M.; Nordentoft, M. Life Expectancy and Death by Diseases of the Circulatory System in Patients with Bipolar Disorder or Schizophrenia in the Nordic Countries. PLoS ONE 2013, 8, e67133. [Google Scholar] [CrossRef] [PubMed]
- Sigmon, E.R.; Kelleman, M.; Susi, A.; Nylund, C.M.; Oster, M.E. Congenital Heart Disease and Autism: A Case-Control Study. Pediatrics 2019, 144, e20184114. [Google Scholar] [CrossRef]
- Desnous, B.; Lenoir, M.; Doussau, A.; Marandyuk, B.; Beaulieu-Genest, L.; Poirier, N.; Carmant, L.; Birca, A. Epilepsy and seizures in children with congenital heart disease: A prospective study. Seizure 2019, 64, 50–53. [Google Scholar] [CrossRef]
- Ryan, A.K.; Goodship, J.A.; Wilson, D.I.; Philip, N.; Levy, A.; Seidel, H.; Schuffenhauer, S.; Oechsler, H.; Belohradsky, B.; Prieur, M.; et al. Spectrum of clinical features associated with interstitial chromosome 22q11 deletions: A European collaborative study. J. Med. Genet. 1997, 34, 798–804. [Google Scholar] [CrossRef]
- Wernovsky, G.; Licht, D.J. Neurodevelopmental Outcomes in Children with Congenital Heart Disease-What Can We Impact? Pediatr. Crit. Care Med. 2016, 17 (Suppl. 1), S232–S242. [Google Scholar] [CrossRef]
- Mahle, W.T.; Clancy, R.R.; Moss, E.M.; Gerdes, M.; Jobes, D.R.; Wernovsky, G. Neurodevelopmental outcome and lifestyle assessment in school-aged and adolescent chil-dren with hypoplastic left heart syndrome. Pediatrics 2000, 105, 1082–1089. [Google Scholar] [CrossRef]
- Haim, T.E.; Wang, W.; Flagg, T.P.; Tones, M.A.; Bahinski, A.; Numann, R.E.; Nichols, C.G.; Nerbonne, J.M. Palmitate attenuates myocardial contractility through augmentation of repolarizing Kv currents. J. Mol. Cell Cardiol. 2010, 48, 395–405. [Google Scholar] [CrossRef]
- O’Connell, R.P.; Musa, H.; Gomez, M.S.; Avula, U.M.; Herron, T.J.; Kalifa, J.; Anumonwo, J.M. Free Fatty Acid Effects on the Atrial Myocardium: Membrane Ionic Currents Are Remodeled by the Disruption of T-Tubular Architecture. PLoS ONE 2015, 10, e0133052. [Google Scholar] [CrossRef]
- Aromolaran, A.S.; Colecraft, H.M.; Boutjdir, M. High-fat diet-dependent modulation of the delayed rectifier K + current in adult guinea pig atrial myocytes. Biochem. Biophys. Res. Commun. 2016, 474, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Mateu, L.; Saiz, J.; Aromolaran, A.S. Differential Modulation of IK and ICa,L Channels in High-Fat Diet-Induced Obese Guinea Pig Atria. Front. Physiol. 2019, 10, 1212. [Google Scholar] [CrossRef] [PubMed]
- Jana, A.K.; Praharaj, S.K.; Roy, N. Olanzapine-induced Orthostatic Hypotension. Clin. Psychopharmacol. Neurosci. 2015, 13, 113–114. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Roh, S.; Ahn, D.H.; Nam, J.H.; Yang, B.H.; Lee, B.H.; Kim, Y.S. Cardiomyopathy associated with clozapine. Exp. Clin. Psychopharmacol. 2006, 14, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Chohan, P.S.; Mittal, R.; Javed, A. Antipsychotic Medication and QT Prolongation. Pak. J. Med. Sci. 2015, 31, 1269–1271. [Google Scholar] [PubMed]
- Stock, E.M.; Zeber, J.E.; McNeal, C.J.; Banchs, J.E.; Copeland, L.A. Psychotropic Pharmacotherapy Associated with QT Prolongation Among Veterans with Post-traumatic Stress Disorder. Ann. Pharmacother. 2018, 52, 838–848. [Google Scholar] [CrossRef]
- Aronow, W.S.; Shamliyan, T. Effects of antidepressants on QT interval in people with mental disorders. Arch. Med Sci. 2020, 16, 727–741. [Google Scholar] [CrossRef]
- Wybraniec, M.T.; Mizia-Stec, K.; Krzych, Ł. Neurocardiogenic injury in subarachnoid hemorrhage: A wide spectrum of catecholamin-mediated brain-heart interactions. Cardiol. J. 2014, 21, 220–228. [Google Scholar] [CrossRef]
- Ghadri, J.R.; Wittstein, I.S.; Prasad, A.; Sharkey, S.; Dote, K.; Akashi, Y.J.; Cammann, V.L.; Crea, F.; Galiuto, L.; Desmet, W. International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Character-istics, Diagnostic Criteria, and Pathophysiology. Eur. Heart J. 2018, 39, 2032–2046. [Google Scholar] [CrossRef]
- De Hert, M.; Detraux, J.; Vancampfort, D. The intriguing relationship between coronary heart disease and mental disorders. Dialog- Clin. Neurosci. 2018, 20, 31–40. [Google Scholar]
- Cohen, B.E.; Edmondson, D.; Kronish, I.M. State of the Art Review: Depression, Stress, Anxiety, and Cardiovascular Disease. Am. J. Hypertens. 2015, 28, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Zorn, J.V.; Schür, R.R.; Boks, M.P.; Kahn, R.S.; Joëls, M.; Vinkers, C.H. Cortisol stress reactivity across psychiatric disorders: A systematic review and meta-analysis. Psychoneuroendocrinology 2017, 77, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.L.; Corbett, B.A. A review of rhythm and responsiveness of cortisol in individuals with autism spectrum disorders. Psychoneuroendocrinology 2014, 49, 207–228. [Google Scholar] [CrossRef] [PubMed]
- Girod, J.P.; Brotman, D.J. Does altered glucocorticoid homeostasis increase cardiovascular risk? Cardiovasc. Res. 2004, 64, 217–226. [Google Scholar] [CrossRef]
- Etxabe, J.; Vazquez, J.A. Morbidity and mortality in Cushing’s disease: An epidemiological approach. Clin. Endocrinol. 1994, 40, 479–484. [Google Scholar] [CrossRef]
- Alvares, G.A.; Quintana, D.S.; Hickie, I.B.; Guastella, A.J. Autonomic nervous system dysfunction in psychiatric disorders and the impact of psycho-tropic medications: A systematic review and meta-analysis. J. Psychiatry Neurosci. 2016, 41, 89–104. [Google Scholar] [CrossRef]
- Neuhaus, E.; Bernier, R.A.; Beauchaine, T.P. Children with Autism Show Altered Autonomic Adaptation to Novel and Familiar Social Partners. Autism Res. 2016, 9, 579–591. [Google Scholar] [CrossRef]
- Carney, R.M.; Freedland, K.E.; Veith, R.C. Depression, the Autonomic Nervous System, and Coronary Heart Disease. Psychosom. Med. 2005, 67, S29–S33. [Google Scholar] [CrossRef]
- Park, J.; Marvar, P.J.; Liao, P.; Kankam, M.L.; Norrholm, S.D.; Downey, R.M.; McCullough, S.A.; Le, N.A.; Rothbaum, B.O. Baroreflex dysfunction and augmented sympathetic nerve responses during mental stress in veterans with post-traumatic stress disorder. J. Physiol. 2017, 595, 4893–4908. [Google Scholar] [CrossRef]
- Thapa, R.; Alvares, G.A.; Zaidi, T.A.; Thomas, E.E.; Hickie, I.B.; Park, S.H.; Guastella, A.J. Reduced heart rate variability in adults with autism spectrum disorder. Autism Res. 2019, 12, 922–930. [Google Scholar] [CrossRef]
- Willerson, J.T.; Ridker, P.M. Inflammation as a cardiovascular risk factor. Circulation 2004, 109 (Suppl. 1), II2–II10. [Google Scholar] [CrossRef] [PubMed]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctôt, K.L. A Meta-Analysis of Cytokines in Major Depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.J.; Buckley, P.; Seabolt, W.; Mellor, A.; Kirkpatrick, B. Meta-analysis of cytokine alterations in schizophrenia: Clinical status and antipsychotic effects. Biol. Psychiatry 2011, 70, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Passos, I.C.; Vasconcelos-Moreno, M.P.; Costa, L.G.; Kunz, M.; Brietzke, E.; Quevedo, J.; Salum, G.A.; Magalhaes, P.V.S.; Kapczinski, F.; Kauer-Sant’Anna, M. Inflammatory markers in post-traumatic stress disorder: A systematic review, meta-analysis, and meta-regression. Lancet Psychiatry 2015, 2, 1002–1012. [Google Scholar] [CrossRef]
- Hou, R.; Garner, M.; Holmes, C.; Osmond, C.; Teeling, J.L.; Lau, L.; Baldwin, D.S. Peripheral inflammatory cytokines and immune balance in Generalised Anxiety Disorder: Case-controlled study. Brain Behav. Immun. 2017, 62, 212–218. [Google Scholar] [CrossRef]
- Croonenberghs, J.; Bosmans, E.; Deboutte, D.; Kenis, G.; Maes, M. Activation of the Inflammatory Response System in Autism. Neuropsychobiology 2002, 45, 1–6. [Google Scholar] [CrossRef]
- Gullestad, L.; Ueland, T.; Vinge, L.E.; Finsen, A.; Yndestad, A.; Aukrust, P. Inflammatory Cytokines in Heart Failure: Mediators and Markers. Cardiology 2012, 122, 23–35. [Google Scholar] [CrossRef]
- Lazzerini, P.E.; Capecchi, P.L.; Laghi-Pasini, F. Long QT Syndrome: An Emerging Role for Inflammation and Immunity. Front. Cardiovasc. Med. 2015, 2, 26. [Google Scholar] [CrossRef]
- Kouvas, N.; Kontogiannis, C.; Georgiopoulos, G.; Spartalis, M.; Tsilimigras, D.I.; Spartalis, E.; Kapelouzou, A.; Kosmopoulos, M.; Chatzidou, S. The complex crosstalk between inflammatory cytokines and ventricular arrhythmias. Cytokine 2018, 111, 171–177. [Google Scholar] [CrossRef]
- Li, Y.H.; Rozanski, G.J. Effects of human recombinant interleukin-1 on electrical properties of guinea pig ventricular cells. Cardiovasc. Res. 1993, 27, 525–530. [Google Scholar] [CrossRef]
- Hagiwara, Y.; Miyoshi, S.; Fukuda, K.; Nishiyama, N.; Ikegami, Y.; Tanimoto, K.; Murata, M.; Takahashi, E.; Shimoda, K.; Hirano, T. SHP2-mediated signaling cascade through gp130 is essential for LIF-dependent I CaL, [Ca2+]i transient, and APD increase in cardiomyocytes. J. Mol. Cell Cardiol. 2007, 43, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Velasco, M.; Ruiz-Hurtado, G.; Hurtado, O.; Moro, M. Ángeles; Delgado, C. TNF-α downregulates transient outward potassium current in rat ventricular myocytes through iNOS overexpression and oxidant species generation. Am. J. Physiol. Circ. Physiol. 2007, 293, H238–H245. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, H.; Zhang, Y.; Gao, H.; Nattel, S.; Wang, Z. Impairment of HERG K(+) channel function by tumor necrosis factor-alpha: Role of reactive oxy-gen species as a mediator. J. Biol. Chem. 2004, 279, 13289–13292. [Google Scholar] [CrossRef] [PubMed]
- Lazzerini, P.E.; Laghi-Pasini, F.; Bertolozzi, I.; Morozzi, G.; Lorenzini, S.; Simpatico, A.; Selvi, E.; Bacarelli, M.R.; Finizola, F.; Vanni, F.; et al. Systemic inflammation as a novel QT-prolonging risk factor in patients with torsades de pointes. Heart 2017, 103, 1821–1829. [Google Scholar] [CrossRef]
- Medenwald, D.; Kors, J.A.; Loppnow, H.; Thiery, J.; Kluttig, A.; Nuding, S.; Tiller, D.; Greiser, K.H.; Werdan, K.; Haerting, J. Inflammation and Prolonged QT Time: Results from the Cardiovascular Disease, Living and Ageing in Halle (CARLA) Study. PLoS ONE 2014, 9, e95994. [Google Scholar] [CrossRef]
- Empana, J.-P.; Jouven, X.; Canouï-Poitrine, F.; Luc, G.; Tafflet, M.; Haas, B.; Arveiler, D.; Ferrieres, J.; Ruidavets, J.-B.; Montaye, M.; et al. C-Reactive Protein, Interleukin 6, Fibrinogen and Risk of Sudden Death in European Middle-Aged Men: The PRIME Study. Arter. Thromb. Vasc. Biol. 2010, 30, 2047–2052. [Google Scholar] [CrossRef]
- Hernesniemi, J.A.; Karhunen, P.J.; Oksala, N.; Kähönen, M.; Levula, M.; Rontu, R.; Ilveskoski, E.; Kajander, O.; Goebeler, S.; Viiri, L.E.; et al. Interleukin 18 gene promoter polymorphism: A link between hypertension and pre-hospital sudden cardiac death: The Helsinki Sudden Death Study. Eur. Heart J. 2009, 30, 2939–2946. [Google Scholar] [CrossRef][Green Version]
- Sgoifo, A.; Carnevali, L.; Grippo, A.J. The socially stressed heart. Insights from studies in rodents. Neurosci. Biobehav. Rev. 2014, 39, 51–60. [Google Scholar] [CrossRef]
- Hodes, G.E.; Pfau, M.L.; Leboeuf, M.; Golden, S.A.; Christoffel, D.J.; Bregman, D.; Rebusi, N.; Heshmati, M.; Aleyasin, H.; Warren, B.L.; et al. Individual differences in the peripheral immune system promote resilience versus susceptibil-ity to social stress. Proc. Natl. Acad. Sci. USA 2014, 111, 16136–16141. [Google Scholar] [CrossRef]
- Stewart, A.M.; Roy, S.; Wong, K.; Gaikwad, S.; Chung, K.M.; Kalueff, A.V. Cytokine and endocrine parameters in mouse chronic social defeat: Implications for trans-lational ’cross-domain’ modeling of stress-related brain disorders. Behav. Brain Res. 2015, 276, 84–91. [Google Scholar] [CrossRef]
- Wood, S.K.; Walker, H.E.; Valentino, R.J.; Bhatnagar, S. Individual Differences in Reactivity to Social Stress Predict Susceptibility and Resilience to a Depressive Phenotype: Role of Corticotropin-Releasing Factor. Endocrinology 2010, 151, 1795–1805. [Google Scholar] [CrossRef] [PubMed]
- Hiippala, A.; Tallila, J.; Myllykangas, S.; Koskenvuo, J.W.; Alastalo, T.P. Expanding the phenotype of Timothy syndrome type 2: An adolescent with ventricular fibrilla-tion but normal development. Am. J. Med. Genet. A 2015, 167A, 629–634. [Google Scholar] [CrossRef]
- Gillis, J.; Burashnikov, E.; Antzelevitch, C.; Blaser, S.; Gross, G.; Turner, L.; Babul-Hirji, R.; Chitayat, D. Long QT, syndactyly, joint contractures, stroke and novel CACNA1C mutation: Expanding the spectrum of Timothy syndrome. Am. J. Med Genet. Part A 2011, 158A, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Kosaki, R.; Ono, H.; Terashima, H.; Kosaki, K. Timothy syndrome-like condition with syndactyly but without prolongation of the QT interval. Am. J. Med Genet. Part A 2018, 176, 1657–1661. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, K.V.; Hennessey, J.A.; Barnett, A.S.; Yin, X.; Stadt, H.A.; Foster, E.; Shah, R.A.; Yazawa, M.; Dolmetsch, R.E.; Kirby, M.L.; et al. Calcium influx through L-type CaV1.2 Ca2+ channels regulates mandibular develop-ment. J. Clin. Investig. 2013, 123, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Raab-Graham, K.F.; Radeke, C.M.; Vandenberg, C.A. Molecular cloning and expression of a human heart inward rectifier potassium channel. NeuroReport 1994, 5, 2501–2505. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, M.R.; Yoon, G.; Fu, Y.-H.; Ptacek, L.J. Andersen-Tawil syndrome: A model of clinical variability, pleiotropy, and genetic heterogeneity. Ann. Med. 2004, 36, 92–97. [Google Scholar] [CrossRef]
- Ambrosini, E.; Sicca, F.; Brignone, M.S.; D’Adamo, M.C.; Napolitano, C.; Servettini, I.; Moro, F.; Ruan, Y.; Guglielmi, L.; Pieroni, S.; et al. Genetically induced dysfunctions of Kir2.1 channels: Implications for short QT3 syndrome and autism–epilepsy phenotype. Hum. Mol. Genet. 2014, 23, 4875–4886. [Google Scholar] [CrossRef]
- Binda, A.; Rivolta, I.; Villa, C.; Chisci, E.; Beghi, M.; Cornaggia, C.M.; Giovannoni, R.; Combi, R. A Novel KCNJ2 Mutation Identified in an Autistic Proband Affects the Single Channel Properties of Kir2.1. Front. Cell. Neurosci. 2018, 12, 76. [Google Scholar] [CrossRef]
- Lu, C.-J.; Hao, G.; Nikiforova, N.; Larsen, H.E.; Liu, K.; Crabtree, M.J.; Li, D.; Herring, N.; Paterson, D. CAPON Modulates Neuronal Calcium Handling and Cardiac Sympathetic Neurotransmission During Dysautonomia in Hypertension. Hypertension 2015, 65, 1288–1297. [Google Scholar] [CrossRef]
- Ronchi, C.; Bernardi, J.; Mura, M.; Stefanello, M.; Badone, B.; Rocchetti, M.; Crotti, L.; Brink, P.; Schwartz, P.J.; Gnecchi, M.; et al. NOS1AP polymorphisms reduce NOS1 activity and interact with prolonged repolarization in arrhythmogenesis. Cardiovasc. Res. 2020. [Google Scholar] [CrossRef]
- Crotti, L.; Monti, M.C.; Insolia, R.; Peljto, A.; Goosen, A.; Brink, P.A.; Greenberg, D.A.; Schwartz, P.J.; George, A.L., Jr. NOS1AP Is a Genetic Modifier of the Long-QT Syndrome. Circulation 2009, 120, 1657–1663. [Google Scholar] [CrossRef]
- Brzustowicz, L.M.; Simone, J.; Mohseni, P.; Hayter, J.E.; Hodgkinson, K.A.; Chow, E.W.C.; Bassett, A.S. Linkage Disequilibrium Mapping of Schizophrenia Susceptibility to the CAPON Region of Chromosome 1q22. Am. J. Hum. Genet. 2004, 74, 1057–1063. [Google Scholar] [CrossRef][Green Version]
- Tu, E.; Bagnall, R.D.; Duflou, J.; Semsarian, C. Post-Mortem Review and Genetic Analysis of Sudden Unexpected Death in Epilepsy (SUDEP) Cases. Brain Pathol. 2010, 21, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Glasscock, E. Genomic biomarkers of SUDEP in brain and heart. Epilepsy Behav. 2013, 38, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Tiron, C.; Campuzano, O.; Pérez-Serra, A.; Mademont, I.; Coll, M.; Allegue, C.; Iglesias, A.; Partemi, S.; Striano, P.; Oliva, A.; et al. Further evidence of the association between LQT syndrome and epilepsy in a family with KCNQ1 pathogenic variant. Seizure 2015, 25, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.N.; Hofman, N.; Haglund, C.M.; Cascino, G.D.; Wilde, A.A.; Ackerman, M.J. Identification of a possible pathogenic link between congenital long QT syndrome and epilepsy. Neurology 2009, 72, 224–231. [Google Scholar] [CrossRef]
- Parisi, P.; Oliva, A.; Vidal, M.C.; Partemi, S.; Campuzano, O.; Iglesias, A.; Pisani, D.; Pascali, V.L.; Paolino, M.C.; Villa, M.P.; et al. Coexistence of epilepsy and Brugada syndrome in a family with SCN5A mutation. Epilepsy Res. 2013, 105, 415–418. [Google Scholar] [CrossRef]
- Cross-Disorder Group of the Psychiatric Genomics Consortium. Identification of risk loci with shared effects on five ma-jor psychiatric disorders: A genome-wide analysis. Lancet 2013, 381, 1371–1379. [Google Scholar] [CrossRef]
- Green, E.K.; Wellcome Trust Case Control Consortium; Grozeva, D.; Jones, I.; Jones, L.; Kirov, G.; Caesar, S.; Gordon-Smith, K.; Fraser, C.; Forty, L.; et al. The bipolar disorder risk allele at CACNA1C also confers risk of recurrent major depression and of schizophrenia. Mol. Psychiatry 2009, 15, 1016–1022. [Google Scholar] [CrossRef]
- Bigos, K.L.; Mattay, V.S.; Callicott, J.H.; Straub, R.E.; Vakkalanka, R.; Kolachana, B.; Hyde, T.M.; Lipska, B.K.; Kleinman, J.E.; Weinberger, D.R. Genetic variation in CACNA1C affects brain circuitries related to mental illness. Arch. Gen. Psychiatry 2010, 67, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Meisler, M.H.; O’Brien, J.E.; Sharkey, L.M. Sodium channel gene family: Epilepsy mutations, gene interactions and modifier effects. J. Physiol. 2010, 588 Pt 11, 1841–1848. [Google Scholar] [CrossRef]
- Osaka, H.; Ogiwara, I.; Mazaki, E.; Okamura, N.; Yamashita, S.; Iai, M.; Yamada, M.; Kurosawa, K.; Iwamoto, H.; Yasui-Furukori, N.; et al. Patients with a sodium channel alpha 1 gene mutation show wide phenotypic variation. Epilepsy Res. 2007, 75, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.P.; Lin, S.P.; Chern, S.R.; Chen, Y.J.; Tsai, F.J.; Wu, P.C.; Wang, W. Array-CGH detection of a de novo 2.8 Mb deletion in 2q24.2-->q24.3 in a girl with autistic features and developmental delay. Eur. J. Med. Genet. 2010, 53, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Bartnik, M.; Chun-Hui Tsai, A.; Xia, Z.; Cheung, S.W.; Stankiewicz, P. Disruption of the SCN2A and SCN3A genes in a patient with mental retardation, neurobehavioral and psychiatric abnormalities, and a history of infantile seizures. Clin. Genet. 2011, 80, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, I.E.; Nabbout, R. SCN1A-related phenotypes: Epilepsy and beyond. Epilepsia 2019, 60 (Suppl. 3), S17–S24. [Google Scholar] [CrossRef]
- Imbrici, P.; Camerino, D.C.; Tricarico, D. Major channels involved in neuropsychiatric disorders and therapeutic perspectives. Front. Genet. 2013, 4, 76. [Google Scholar] [CrossRef]
- Judy, J.T.; Seifuddin, F.; Pirooznia, M.; Mahon, P.B.; Bipolar Genome Study, C.; Jancic, D.; Goes, F.S.; Schulze, T.; Cichon, S.; Noethen, M.; et al. Converging Evidence for Epistasis between ANK3 and Potassium Channel Gene KCNQ2 in Bipolar Disorder. Front. Genet. 2013, 4, 87. [Google Scholar] [CrossRef][Green Version]
- Kaminsky, Z.; Jones, I.; Verma, R.; Saleh, L.; Trivedi, H.; Guintivano, J.; Akman, R.; Zandi, P.; Lee, R.S.; Potash, J.B. DNA methylation and expression of KCNQ3 in bipolar disorder. Bipolar Disord. 2015, 17, 150–159. [Google Scholar] [CrossRef]
- Borsotto, M.; Cavarec, L.; Bouillot, M.; Romey, G.; Macciardi, F.; Delaye, A.; Nasroune, M.; Bastucci, M.; Sambucy, J.L.; Luan, J.J.; et al. PP2A-Bgamma subunit and KCNQ2 K+ channels in bipolar disorder. Pharm. J. 2007, 7, 123–132. [Google Scholar]
- Heide, J.; Mann, S.A.; Vandenberg, J.I. The schizophrenia-associated Kv11.1-3.1 isoform results in reduced current accumulation during repetitive brief depolarizations. PLoS ONE 2012, 7, e45624. [Google Scholar] [CrossRef] [PubMed]
- Puckerin, A.; Aromolaran, K.A.; Chang, D.D.; Zukin, R.S.; Colecraft, H.M.; Boutjdir, M.; Aromolaran, A.S. hERG 1a LQT2 C-terminus truncation mutants display hERG 1b-dependent dominant negative mechanisms. Heart Rhythm 2016, 13, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Aldana, B.I.; Sitges, M. Sertraline inhibits pre-synaptic Na(+) channel-mediated responses in hippocampus-isolated nerve endings. J. Neurochem. 2012, 121, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Shaw, D.M. Mineral metabolism, mania, and melancholia. Br. Med. J. 1966, 2, 262–267. [Google Scholar] [CrossRef]
- Yanagita, T.; Maruta, T.; Uezono, Y.; Satoh, S.; Yoshikawa, N.; Nemoto, T.; Kobayashi, H.; Wada, A. Lithium inhibits function of voltage-dependent sodium channels and catecholamine secretion independent of glycogen synthase kinase-3 in adrenal chromaffin cells. Neuropharmacology 2007, 53, 881–889. [Google Scholar] [CrossRef]
- Darbar, D.; Yang, T.; Churchwell, K.; Wilde, A.A.; Roden, D.M. Unmasking of brugada syndrome by lithium. Circulation 2005, 112, 1527–1531. [Google Scholar] [CrossRef]
- Dubovsky, S.L.; Buzan, R.; Thomas, M.; Kassner, C.; Cullum, C.M. Nicardipine improves the antidepressant action of ECT but does not improve cognition. J. ECT 2001, 17, 3–10. [Google Scholar] [CrossRef]
- Large, C.H.; Di Daniel, E.; Li, X.; George, M.S. Neural network dysfunction in bipolar depression: Clues from the efficacy of lamotrigine. Biochem. Soc. Trans. 2009, 37 Pt 5, 1080–1084. [Google Scholar] [CrossRef]
- Kristensen, L.V.; Sandager-Nielsen, K.; Hansen, H.H. K(v) 7 (KCNQ) channel openers normalize central 2-deoxyglucose uptake in a mouse model of mania and increase prefrontal cortical and hippocampal serine-9 phosphorylation levels of GSK3beta. J. Neurochem. 2012, 121, 373–382. [Google Scholar] [CrossRef]
- Wickenden, A.D.; Yu, W.; Zou, A.; Jegla, T.; Wagoner, P.K. Retigabine, a novel anti-convulsant, enhances activation of KCNQ2/Q3 potassium channels. Mol. Pharmacol. 2000, 58, 591–600. [Google Scholar] [CrossRef]
- Rogawski, M.A.; Loscher, W. The neurobiology of antiepileptic drugs for the treatment of nonepileptic conditions. Nat. Med. 2004, 10, 685–692. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, J.R.; Bauer, C.K. Functions of erg K+ channels in excitable cells. J. Cell Mol. Med. 2004, 8, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.A. Clinical and therapeutic aspects of congenital and acquired long QT syndrome. Am. J. Med. 2002, 112, 58–66. [Google Scholar] [CrossRef]
- El-Sherif, N.; Turitto, G. Torsade de pointes. Curr. Opin. Cardiol. 2003, 18, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Guasti, L.; Cilia, E.; Crociani, O.; Hofmann, G.; Polvani, S.; Becchetti, A.; Wanke, E.; Tempia, F.; Arcangeli, A. Expression pattern of the ether-a-go-go-related (ERG) family proteins in the adult mouse central nervous system: Evidence for coassembly of different subunits. J. Comp. Neurol. 2005, 491, 157–174. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Tucker, K.R.; Putzier, I.; Huertas, M.A.; Horn, J.P.; Canavier, C.C.; Levitan, E.S.; Shepard, P.D. Functional characterization of ether-a-go-go-related gene potassium channels in midbrain dopamine neurons—Implications for a role in depolarization block. Eur. J. Neurosci. 2012, 36, 2906–2916. [Google Scholar] [CrossRef]
- Chiesa, N.; Rosati, B.; Arcangeli, A.; Olivotto, M.; Wanke, E. A novel role for HERG K+ channels: Spike-frequency adaptation. J. Physiol. 1997, 501 Pt 2, 313–318. [Google Scholar] [CrossRef]
- Kongsamut, S.; Kang, J.; Chen, X.L.; Roehr, J.; Rampe, D. A comparison of the receptor binding and HERG channel affinities for a series of antipsychotic drugs. Eur. J. Pharmacol. 2002, 450, 37–41. [Google Scholar] [CrossRef]
- Sale, H.; Wang, J.; O’Hara, T.J.; Tester, D.J.; Phartiyal, P.; He, J.Q.; Rudy, Y.; Ackerman, M.J.; Robertson, G.A. Physiological properties of hERG 1a/1b heteromeric currents and a hERG 1b-specific mutation associated with Long-QT syndrome. Circ. Res. 2008, 103, e81–e95. [Google Scholar] [CrossRef]
- Aromolaran, A.S.; Srivastava, U.; Ali, A.; Chahine, M.; Lazaro, D.; El-Sherif, N.; Capecchi, P.L.; Laghi-Pasini, F.; Lazzerini, P.E.; Boutjdir, M. Interleukin-6 inhibition of hERG underlies risk for acquired long QT in cardiac and systemic inflammation. PLoS ONE 2018, 13, e0208321. [Google Scholar] [CrossRef]
- Huffaker, S.J.; Chen, J.; Nicodemus, K.K.; Sambataro, F.; Yang, F.; Mattay, V.; Lipska, B.K.; Hyde, T.M.; Song, J.; Rujescu, D.; et al. A primate-specific, brain isoform of KCNH2 affects cortical physiology, cognition, neuronal repolarization and risk of schizophrenia. Nat. Med. 2009, 15, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Apud, J.A.; Zhang, F.; Decot, H.; Bigos, K.L.; Weinberger, D.R. Genetic variation in KCNH2 associated with expression in the brain of a unique hERG isoform modulates treatment response in patients with schizophrenia. Am. J. Psychiatry 2012, 169, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Uher, R.; Tansey, K.E.; Dew, T.; Maier, W.; Mors, O.; Hauser, J.; Dernovsek, M.Z.; Henigsberg, N.; Souery, D.; Farmer, A.; et al. An inflammatory biomarker as a differential predictor of outcome of depression treatment with escitalopram and nortriptyline. Am. J. Psychiatry 2014, 171, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Raison, C.L.; Rutherford, R.E.; Woolwine, B.J.; Shuo, C.; Schettler, P.; Drake, D.F.; Haroon, E.; Miller, A.H. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: The role of baseline inflammatory biomarkers. JAMA Psychiatry 2013, 70, 31–41. [Google Scholar] [CrossRef]
- Cheng, Y.F.; Leu, H.B.; Su, C.C.; Huang, C.C.; Chiang, C.H.; Huang, P.H.; Chung, C.M.; Lin, S.J.; Chen, J.W.; Chan, W.L. Association between panic disorder and risk of atrial fibrillation: A nationwide study. Psychosom. Med. 2013, 75, 30–35. [Google Scholar] [CrossRef]
- Eaker, E.D.; Sullivan, L.M.; Kelly-Hayes, M.; D’Agostino, R.B., Sr.; Benjamin, E.J. Tension and anxiety and the prediction of the 10-year incidence of coronary heart disease, atrial fibrillation, and total mortality: The Framingham Offspring Study. Psychosom. Med. 2005, 67, 692–696. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernardi, J.; Aromolaran, K.A.; Aromolaran, A.S. Neurological Disorders and Risk of Arrhythmia. Int. J. Mol. Sci. 2021, 22, 188. https://doi.org/10.3390/ijms22010188
Bernardi J, Aromolaran KA, Aromolaran AS. Neurological Disorders and Risk of Arrhythmia. International Journal of Molecular Sciences. 2021; 22(1):188. https://doi.org/10.3390/ijms22010188
Chicago/Turabian StyleBernardi, Joyce, Kelly A. Aromolaran, and Ademuyiwa S. Aromolaran. 2021. "Neurological Disorders and Risk of Arrhythmia" International Journal of Molecular Sciences 22, no. 1: 188. https://doi.org/10.3390/ijms22010188
APA StyleBernardi, J., Aromolaran, K. A., & Aromolaran, A. S. (2021). Neurological Disorders and Risk of Arrhythmia. International Journal of Molecular Sciences, 22(1), 188. https://doi.org/10.3390/ijms22010188




