Progress and Challenges of Amniotic Fluid Derived Stem Cells in Therapy of Ischemic Heart Disease
Abstract
1. Introduction
2. Advantages and Limitations of Different Types of Stem Cells in Cardiac Regeneration
3. Therapeutic Effect of Human Pluripotent Stem Cell-Derived Cardiomyocytes (PSC-CMs) on Myocardial Infarction
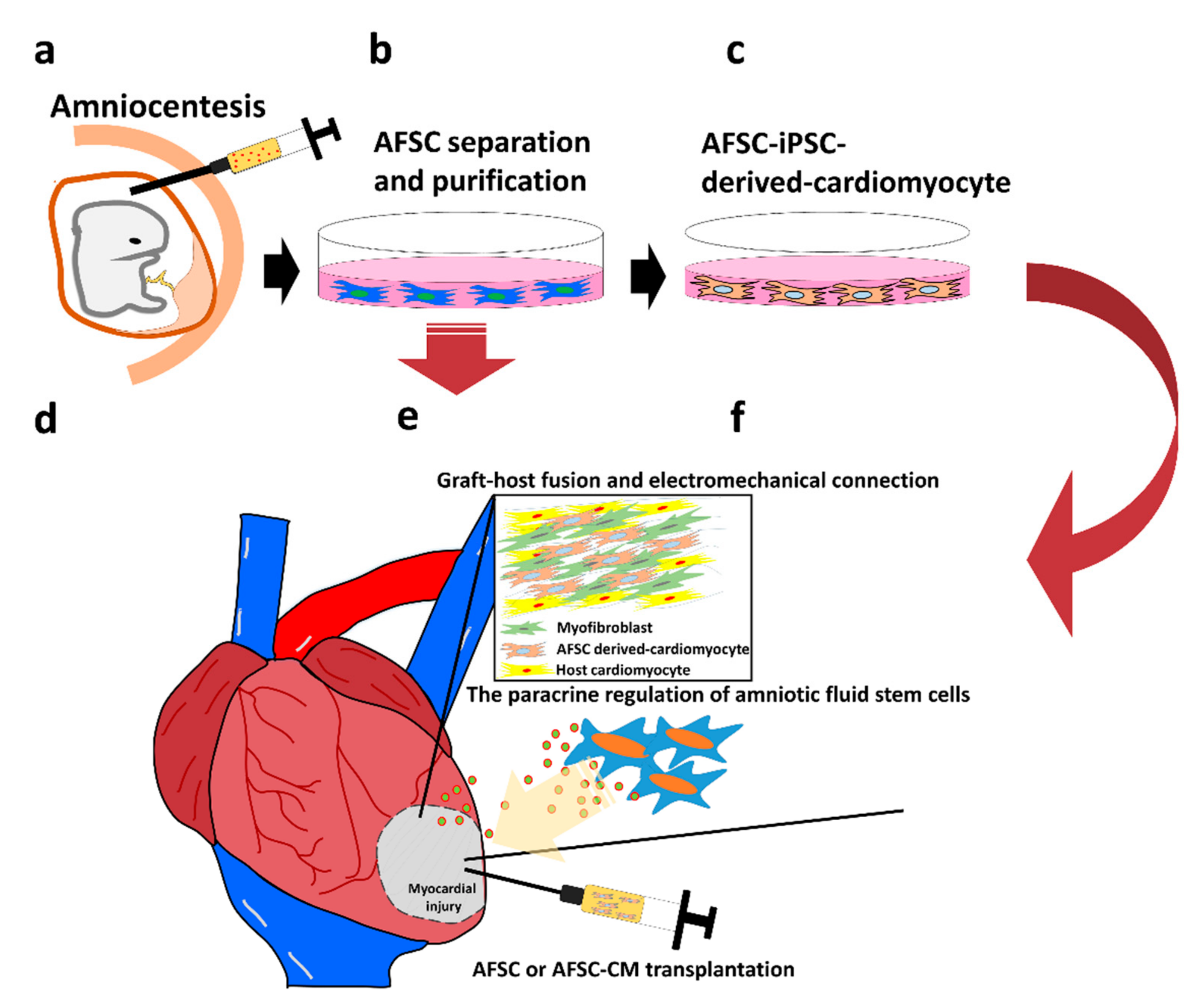
4. Characteristics and Application of Amniotic Fluid Derived Stem Cells (AFSCs)
5. Possibility of Developing Cardiomyocytes from AFSCs
6. Paracrine Regulation of Amniotic Fluid Stem Cells
7. Unmet Needs before Clinical Trial of Amniotic Fluid Stem Cell-iPSC-CMs in Myocardial Regeneration
7.1. Genetic Inheritance of Donor
7.2. Tumorigenesis
7.3. Arrhythmia
8. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFSCs | amniotic fluid stem cells |
| ESCs | embryonic stem cells |
| iPSCs | induced pluripotent stem cells |
| FGF2 | fibroblast growth factor 2 |
| TGF-β | transforming growth factor β |
| BMP4 | bone morphogenetic protein 4 |
| ESC-CMs | cardiomyocytes differentiated from embryonic stem cells |
| hESCs | human embryonic stem cells |
| hESC-CM | hESC-derived cardiomyocytes |
| AFSC-CM | AFSC-derived cardiomyocytes |
References
- World Health Organization. Cardiovascular disease risk charts: Revised models to estimate risk in 21 global regions. Lancet Glob. Health 2019, 7, e1332–e1345. [Google Scholar] [CrossRef]
- Leone, M.; Magadum, A.; Engel, F.B. Cardiomyocyte proliferation in cardiac development and regeneration: A guide to methodologies and interpretations. Am. J. Physiol. Circ. Physiol. 2015, 309, H1237–H1250. [Google Scholar] [CrossRef] [PubMed]
- Tzahor, E.; Poss, K.D. Cardiac regeneration strategies: Staying young at heart. Science 2017, 356, 1035–1039. [Google Scholar] [CrossRef] [PubMed]
- Bektik, E.; Fu, J.D. Ameliorating the Fibrotic Remodeling of the Heart through Direct Cardiac Reprogramming. Cells 2019, 8, 679. [Google Scholar] [CrossRef]
- Wang, M.; Yuan, Q.; Xie, L. Mesenchymal Stem Cell-Based Immunomodulation: Properties and Clinical Application. Stem Cells Int. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Chinnadurai, R.; Copland, I.B.; Patel, S.R.; Galipeau, J. IDO-Independent Suppression of T Cell Effector Function by IFN-γ–Licensed Human Mesenchymal Stromal Cells. J. Immunol. 2014, 192, 1491–1501. [Google Scholar] [CrossRef]
- Lim, M.; Wang, W.; Liang, L.; Han, Z.-B.; Li, Z.; Geng, J.; Zhao, M.; Jia, H.; Feng, J.; Wei, Z.; et al. Intravenous injection of allogeneic umbilical cord-derived multipotent mesenchymal stromal cells reduces the infarct area and ameliorates cardiac function in a porcine model of acute myocardial infarction. Stem Cell Res. Ther. 2018, 9, 1–17. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Breckwoldt, K.; Letuffe-Brenière, D.; Mannhardt, I.; Schulze, T.; Ulmer, B.; Werner, T.; Benzin, A.; Klampe, B.; Reinsch, M.C.; Laufer, S.; et al. Differentiation of cardiomyocytes and generation of human engineered heart tissue. Nat. Protoc. 2017, 12, 1177–1197. [Google Scholar] [CrossRef]
- Lian, X.; Zhang, J.; Azarin, S.M.; Zhu, K.; Hazeltine, L.B.; Bao, X.; Hsiao, C.; Kamp, T.J.; Palecek, S.P. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nat. Protoc. 2013, 8, 162–175. [Google Scholar] [CrossRef]
- Nichols, J.; Smith, A. The origin and identity of embryonic stem cells. Development (Cambridge, UK) 2011, 138, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Sensébé, L.; Bourin, P. Mesenchymal Stem Cells for Therapeutic Purposes. Transplant 2009, 87, S49–S53. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Discher, D.E.; Péault, B.M.; Phinney, D.G.; Hare, J.M.; Caplan, A.I. Mesenchymal stem cell perspective: Cell biology to clinical progress. NPJ Regen. Med. 2019, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Przyborski, S. Differentiation of Human Embryonic Stem Cells After Transplantation in Immune-Deficient Mice. Stem Cells 2005, 23, 1242–1250. [Google Scholar] [CrossRef]
- Gidekel, S.; Pizov, G.; Bergman, Y.; Pikarsky, E. Oct-3/4 is a dose-dependent oncogenic fate determinant. Cancer Cell 2003, 4, 361–370. [Google Scholar] [CrossRef]
- Chen, J.; Ji, T.; Wu, D.; Jiang, S.; Zhao, J.; Lin, H.; Cai, X. Human mesenchymal stem cells promote tumor growth via MAPK pathway and metastasis by epithelial mesenchymal transition and integrin α5 in hepatocellular carcinoma. Cell Death Dis. 2019, 10, 425. [Google Scholar] [CrossRef]
- Cuiffo, B.G.; Karnoub, A.E. Mesenchymal stem cells in tumor development: Emerging roles and concepts. Cell Adhes. Migr. 2012, 6, 220–230. [Google Scholar] [CrossRef]
- Rodini, C.O.; Da Silva, P.B.G.; Assoni, A.F.; Carvalho, V.M.; Okamoto, O.K. Mesenchymal stem cells enhance tumorigenic properties of human glioblastoma through independent cell-cell communication mechanisms. Oncotarget 2018, 9, 24766–24777. [Google Scholar] [CrossRef]
- Abad, M.; Mosteiro, L.; Pantoja, C.; Cañamero, M.; Rayon, T.; Ors, I.; Graña, O.; Megías, D.; Domínguez, O.; Martínez, D.; et al. Reprogramming in vivo produces teratomas and iPS cells with totipotency features. Nat. Cell Biol. 2013, 502, 340–345. [Google Scholar] [CrossRef]
- Liu, Y.W.; Chen, B.; Yang, X.; Fugate, J.A.; Kalucki, F.A.; Futakuchi-Tsuchida, A.; Couture, L.; Vogel, K.W.; Astley, C.A.; Baldessari, A.; et al. Human embryonic stem cell–derived cardiomyocytes restore function in infarcted hearts of non-human primates. Nat. Biotechnol. 2018, 36, 597–605. [Google Scholar] [CrossRef]
- Fang, Y.H.; Wang, S.P.; Gao, Z.H.; Wu, S.N.; Chang, H.Y.; Yang, P.J.; Liu, P.Y.; Liu, Y.W. Efficient Cardiac Differentiation of Human Amniotic Fluid-Derived Stem Cells into Induced Pluripotent Stem Cells and Their Potential Immune Privilege. Int. J. Mol. Sci. 2020, 21, 2359. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.J.H.; Yang, X.; Don, C.W.; Minami, E.; Liu, Y.-W.; Weyers, J.J.; Mahoney, W.M.; Van Biber, B.; Cook, S.M.; Palpant, N.J.; et al. Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts. Nature 2014, 510, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Gregorich, Z.R.; Zhu, W.; Mattapally, S.; Oduk, Y.; Lou, X.; Kannappan, R.; Borovjagin, A.V.; Walcott, G.P.; Pollard, A.E.; et al. Large Cardiac Muscle Patches Engineered From Human Induced-Pluripotent Stem Cell-Derived Cardiac Cells Improve Recovery From Myocardial Infarction in Swine. Circulation 2018, 137, 1712–1730. [Google Scholar] [CrossRef]
- Ishida, M.; Miyagawa, S.; Saito, A.; Fukushima, S.; Harada, A.; Ito, E.; Ohashi, F.; Watabe, T.; Hatazawa, J.; Matsuura, K.; et al. Transplantation of Human-induced Pluripotent Stem Cell-derived Cardiomyocytes Is Superior to Somatic Stem Cell Therapy for Restoring Cardiac Function and Oxygen Consumption in a Porcine Model of Myocardial Infarction. Transplantation 2019, 103, 291–298. [Google Scholar] [CrossRef]
- Robinson, J.; Halliwell, J.A.; Hayhurst, J.D.; Flicek, P.; Parham, P.; Marsh, S.G.E. The IPD and IMGT/HLA database: Allele variant databases. Nucleic Acids Res. 2015, 43, D423–D431. [Google Scholar] [CrossRef]
- Morizane, A.; Kikuchi, T.; Hayashi, T.; Mizuma, H.; Takara, S.; Doi, H.; Mawatari, A.; Glasser, M.F.; Shiina, T.; Ishigaki, H.; et al. MHC matching improves engraftment of iPSC-derived neurons in non-human primates. Nat. Commun. 2017, 8, 385. [Google Scholar] [CrossRef]
- Deuse, T.; Hu, X.; Gravina, A.; Wang, D.; Tediashvili, G.; De, C.; Thayer, W.O.; Wahl, A.; Garcia, J.V.; Reichenspurner, H.; et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat. Biotechnol. 2019, 37, 252–258. [Google Scholar] [CrossRef]
- De Coppi, P.; Bartsch, G.; Atala, A. Amniotic fluid and placental stem cells as a source for urological regenerative medicine. Biomater. Tissue Eng. Urol. 2009, 18, 378–394. [Google Scholar] [CrossRef]
- In’t Anker, P.S.; Scherjon, S.A.; Der Keur, C.K.V.; Noort, W.A.; Claas, F.H.; Willemze, R.; Fibbe, W.E.; Kanhai, H.H. Amniotic fluid as a novel source of mesenchymal stem cells for therapeutic transplantation. Blood 2003, 102, 1548–1549. [Google Scholar] [CrossRef]
- Loukogeorgakis, S.P.; De Coppi, P. Concise Review: Amniotic Fluid Stem Cells: The Known, the Unknown, and Potential Regenerative Medicine Applications. Stem Cells 2017, 35, 1663–1673. [Google Scholar] [CrossRef]
- Bollini, S.; Pozzobon, M.; Nobles, M.; Riegler, J.; Dong, X.; Piccoli, M.; Chiavegato, A.; Price, A.N.; Ghionzoli, M.; Cheung, K.K.; et al. In Vitro and In Vivo Cardiomyogenic Differentiation of Amniotic Fluid Stem Cells. Stem Cell Rev. Rep. 2011, 7, 364–380. [Google Scholar] [CrossRef] [PubMed]
- De Coppi, P.; Bartsch, G., Jr.; Siddiqui, M.M.; Xu, T.; Santos, C.C.; Perin, L.; Mostoslavsky, G.; Serre, A.C.; Snyder, E.Y.; Yoo, J.J.; et al. Isolation of amniotic stem cell lines with potential for therapy. Nat. Biotechnol. 2007, 25, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Bajek, A.; Olkowska, J.; Walentowicz-Sadłecka, M.; Sadłecki, P.; Grabiec, M.; Porowinska, D.; Drewa, T.; Roszkowski, K. Human Adipose-Derived and Amniotic Fluid-Derived Stem Cells: A Preliminary In Vitro Study Comparing Myogenic Differentiation Capability. Med. Sci. Monit. 2018, 24, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Murry, C.E.; Wiseman, R.W.; Schwartz, S.M.; Hauschka, S.D. Skeletal myoblast transplantation for repair of myocardial necrosis. J. Clin. Investig. 1996, 98, 2512–2523. [Google Scholar] [CrossRef] [PubMed]
- Hattan, N.; Kawaguchi, H.; Ando, K.; Kuwabara, E.; Fujita, J.; Murata, M.; Suematsu, M.; Mori, H.; Fukuda, K. Purified cardiomyocytes from bone marrow mesenchymal stem cells produce stable intracardiac grafts in mice. Cardiovasc. Res. 2005, 65, 334–344. [Google Scholar] [CrossRef]
- Bollini, S.; Cheung, K.; Riegler, J.; Dong, X.; Smart, N.; Ghionzoli, M.; Loukogeorgakis, S.P.; Maghsoudlou, P.; Dubé, K.N.; Riley, P.R.; et al. Amniotic Fluid Stem Cells Are Cardioprotective Following Acute Myocardial Infarction. Stem Cells Dev. 2011, 20, 1985–1994. [Google Scholar] [CrossRef]
- Balbi, C.; Piccoli, M.; Barile, L.; Papait, A.; Armirotti, A.; Principi, E.; Reverberi, D.; Pascucci, L.; Becherini, P.; Varesio, L.; et al. First Characterization of Human Amniotic Fluid Stem Cell Extracellular Vesicles as a Powerful Paracrine Tool Endowed with Regenerative Potential. Stem Cells Transl. Med. 2017, 6, 1340–1355. [Google Scholar] [CrossRef]
- Shen, X.; Pan, B.; Zhou, H.; Liu, L.; Lv, T.; Zhu, J.; Huang, X.; Tian, J. Differentiation of mesenchymal stem cells into cardiomyocytes is regulated by miRNA-1-2 via WNT signaling pathway. J. Biomed. Sci. 2017, 24, 1–8. [Google Scholar] [CrossRef]
- Gwak, J.; Hwang, S.G.; Park, H.-S.; Choi, S.R.; Park, S.-H.; Kim, H.; Ha, N.-C.; Bae, S.J.; Han, J.-K.; Kim, D.-E.; et al. Small molecule-based disruption of the Axin/β-catenin protein complex regulates mesenchymal stem cell differentiation. Cell Res. 2011, 22, 237–247. [Google Scholar] [CrossRef]
- Han, L.; Li, Y.; Tchao, J.; Kaplan, A.D.; Lin, B.; Li, Y.; Mich-Basso, J.; Lis, A.; Hassan, N.; London, B.; et al. Study familial hypertrophic cardiomyopathy using patient-specific induced pluripotent stem cells. Cardiovasc. Res. 2014, 104, 258–269. [Google Scholar] [CrossRef]
- Liu, Y.W.; Fang, Y.H.; Su, C.T.; Hwang, S.M.; Liu, P.Y.; Wu, S.N. The biochemical and electrophysiological profiles of amniotic fluid-derived stem cells following Wnt signaling modulation cardiac differentiation. Cell Death Discov. 2019, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wilson, G.F.; Soerens, A.G.; Koonce, C.H.; Yu, J.; Palecek, S.P.; Thomson, J.A.; Kamp, T.J. Functional Cardiomyocytes Derived From Human Induced Pluripotent Stem Cells. Circ. Res. 2009, 104, e30–e41. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, A.; Sen, D. Mesenchymal stem cells in cardiac regeneration: A detailed progress report of the last 6 years (2010–2015). Stem Cell Res. Ther. 2016, 7, 1–25. [Google Scholar] [CrossRef]
- Guan, X.; Xu, W.; Zhang, H.; Wang, Q.; Yu, J.; Zhang, R.; Chen, Y.; Xia, Y.; Wang, J.; Wang, D. Transplantation of human induced pluripotent stem cell-derived cardiomyocytes improves myocardial function and reverses ventricular remodeling in infarcted rat hearts. Stem Cell Res. 2020, 11, 73. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Delo, D.M.; Atala, A.; Soker, S. In vitro cardiomyogenic potential of human amniotic fluid stem cells. J. Tissue Eng. Regen. Med. 2011, 5, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Dolnikov, K.; Shilkrut, M.; Zeevi-Levin, N.; Gerecht-Nir, S.; Amit, M.; Danon, A.; Itskovitz-Eldor, J.; Binah, O.; Itskovitz-Eldor, J. Functional Properties of Human Embryonic Stem Cell-Derived Cardiomyocytes: Intracellular Ca2+Handling and the Role of Sarcoplasmic Reticulum in the Contraction. Stem Cells 2006, 24, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Satin, J.; Itzhaki, I.; Rapoport, S.; Schroder, E.A.; Izu, L.; Arbel, G.; Beyar, R.; Balke, C.W.; Schiller, J.; Gepstein, L. Calcium Handling in Human Embryonic Stem Cell-Derived Cardiomyocytes. Stem Cells 2008, 26, 1961–1972. [Google Scholar] [CrossRef]
- Shadrin, I.Y.; Yoon, W.; Li, L.; Shepherd, N.; Bursac, N. Rapid fusion between mesenchymal stem cells and cardiomyocytes yields electrically active, non-contractile hybrid cells. Sci. Rep. 2015, 5, 12043. [Google Scholar] [CrossRef]
- Fatima, A.; Kaifeng, S.; Dittmann, S.; Xu, G.; Gupta, M.K.; Linke, M.; Zechner, U.; Nguemo, F.; Milting, H.; Farr, M.; et al. The Disease-Specific Phenotype in Cardiomyocytes Derived from Induced Pluripotent Stem Cells of Two Long QT Syndrome Type 3 Patients. PLoS ONE 2013, 8, e83005. [Google Scholar] [CrossRef]
- Di Baldassarre, A.; D’Amico, M.A.; Izzicupo, P.; Gaggi, G.; Guarnieri, S.; Mariggiò, M.A.; Antonucci, I.; Corneo, B.; Sirabella, D.; Stuppia, L.; et al. Cardiomyocytes Derived from Human (Cardiopoietic) Amniotic Fluids. Sci. Rep. 2018, 8, 12028. [Google Scholar] [CrossRef]
- Wang, K.; Terrenoire, C.; Sampson, K.J.; Iyer, V.; Osteen, J.D.; Lu, J.; Keller, G.M.; Kotton, D.N.; Kass, R.S. Biophysical properties of slow potassium channels in human embryonic stem cell derived cardiomyocytes implicate subunit stoichiometry. J. Physiol. 2011, 589, 6093–6104. [Google Scholar] [CrossRef] [PubMed]
- Florin, I.; Constantinescu, A.; Andrei, E.; Amuzescu, B.; Halitzchi, F.; Savu, L.; Maniu, H. Electrophysiology, immunophenotype, and gene expression characterization of senescent and cryopreserved human amniotic fluid stem cells. J. Physiol. Sci. 2016, 66, 463–476. [Google Scholar] [CrossRef]
- Mellows, B.; Mitchell, R.; Antonioli, M.; Kretz, O.; Chambers, D.; Zeuner, M.-T.; Denecke, B.; Musante, L.; Ramachandra, D.L.; Debacq-Chainiaux, F.; et al. Protein and Molecular Characterization of a Clinically Compliant Amniotic Fluid Stem Cell-Derived Extracellular Vesicle Fraction Capable of Accelerating Muscle Regeneration Through Enhancement of Angiogenesis. Stem Cells Dev. 2017, 26, 1316–1333. [Google Scholar] [CrossRef] [PubMed]
- Romani, R.; Pirisinu, I.; Calvitti, M.; Pallotta, M.T.; Gargaro, M.; Bistoni, G.; Vacca, C.; Di Michele, A.; Orabona, C.; Rosati, J.; et al. Stem cells from human amniotic fluid exert immunoregulatory function via secreted indoleamine 2,3-dioxygenase1. J. Cell. Mol. Med. 2015, 19, 1593–1605. [Google Scholar] [CrossRef]
- Takov, K.; He, Z.; Johnston, H.E.; Timms, J.F.; Guillot, P.V.; Yellon, D.M.; Davidson, S.M. Small extracellular vesicles secreted from human amniotic fluid mesenchymal stromal cells possess cardioprotective and promigratory potential. Basic Res. In Cardiol. 2020, 115, 26. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Hoseini, S.M.; Sheikhha, M.H.; Kalantar, S.M.; Matin, M.M.; Aflatoonian, B.; Bahrami, A.R.; Zarein, F.; Rahmani, S.; Montazeri, F. A comparative analysis of immunomodulatory genes in two clonal subpopulations of CD90+ amniocytes isolated from human amniotic fluid. Placenta 2020, 101, 234–241. [Google Scholar] [CrossRef]
- Hoseini, S.M.; Montazeri, F.; Bahrami, A.R.; Kalantar, S.M.; Rahmani, S.; Zarein, F.; Matin, M.M. Investigating the expression of pluripotency-related genes in human amniotic fluid cells: A semi-quantitative comparison between different subpopulations, from primary to cultured amniocytes. Reprod. Biol. 2020, 20, 338–347. [Google Scholar] [CrossRef]
- Gore, A.; Li, Z.; Fung, H.L.; Young, J.E.; Agarwal, S.; Antosiewicz-Bourget, J.; Canto, I.; Giorgetti, A.; Israel, M.A.; Kiskinis, E.; et al. Somatic coding mutations in human induced pluripotent stem cells. Nat. Cell Biol. 2011, 471, 63–67. [Google Scholar] [CrossRef]
- Nazor, K.L.; Altun, G.; Lynch, C.; Tran, H.; Harness, J.V.; Slavin, I.; Garitaonandia, I.; Müller, F.J.; Wang, Y.C.; Boscolo, F.S.; et al. Recurrent variations in DNA methylation in human pluripotent stem cells and their differentiated derivatives. Cell Stem Cell 2012, 10, 620–634. [Google Scholar] [CrossRef]
- Gao, S.; Zheng, C.; Chang, G.; Liu, W.; Kou, X.; Tan, K.; Tao, L.; Xu, K.; Wang, H.; Cai, J.; et al. Unique features of mutations revealed by sequentially reprogrammed induced pluripotent stem cells. Nat. Commun. 2015, 6, 6318. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, M.; Takizawa, N.; Narita, M.; Ichisaka, T.; Yamanaka, S. Promotion of direct reprogramming by transformation-deficient Myc. Proc. Natl. Acad. Sci. 2010, 107, 14152–14157. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.L.; Ying, S.Y. Mechanism and Method for Generating Tumor-Free iPS Cells Using Intronic MicroRNA miR-302 Induction. In Bioinformatics in MicroRNA Research; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2018; Volume 1733, pp. 265–282. [Google Scholar]
- Araki, R.; Hoki, Y.; Suga, T.; Obara, C.; Sunayama, M.; Imadome, K.; Fujita, M.; Kamimura, S.; Nakamura, M.; Wakayama, S.; et al. Genetic aberrations in iPSCs are introduced by a transient G1/S cell cycle checkpoint deficiency. Nat. Commun. 2020, 11, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Kusakawa, S.; Kuroda, T.; Miura, T.; Tano, K.; Takada, N.; Matsuyama, S.; Matsuyama, A.; Nasu, M.; Umezawa, A.; et al. Tumorigenicity-associated characteristics of human iPS cell lines. PLoS ONE 2018, 13, e0205022. [Google Scholar] [CrossRef] [PubMed]
- Shiba, Y.; Fernandes, S.; Zhu, W.-Z.; Filice, D.; Muskheli, V.; Kim, J.; Palpant, N.J.A.; Gantz, J.; Moyes, K.W.; Reinecke, H.; et al. Human ES-cell-derived cardiomyocytes electrically couple and suppress arrhythmias in injured hearts. Nat. Cell Biol. 2012, 489, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Shiba, Y.; Gomibuchi, T.; Seto, T.; Wada, Y.; Ichimura, H.; Tanaka, Y.; Ogasawara, T.; Okada, K.; Shiba, N.; Sakamoto, K.; et al. Allogeneic transplantation of iPS cell-derived cardiomyocytes regenerates primate hearts. Nat. Cell Biol. 2016, 538, 388–391. [Google Scholar] [CrossRef]
- Horváth, A.; Lemoine, M.D.; Löser, A.; Mannhardt, I.; Flenner, F.; Uzun, A.U.; Neuber, C.; Breckwoldt, K.; Hansen, A.; Girdauskas, E.; et al. Low Resting Membrane Potential and Low Inward Rectifier Potassium Currents Are Not Inherent Features of hiPSC-Derived Cardiomyocytes. Stem Cell Rep. 2018, 10, 822–833. [Google Scholar] [CrossRef]
| Type | Embryonic Stem Cells (ESC) | Mesenchymal Stem Cells (MSC) | Induced Pluripotent Stem Cells (iPSC) |
|---|---|---|---|
| Cell source | Inner cell mass of mulberry embryo [11] | Skin, fat, cord blood, amniotic membrane, bone marrow [12] | Somatic cell [8] |
| Plasticity | Pluripotent [11] | Multipotent [13] | Pluripotent [8] |
| Tumorization | Yes [14,15] | No [16,17,18] | Yes [19] |
| Ethical restriction | Yes | No | No |
| Functional cardiomyocyte differentiation ability | Yes [20] | No [7] | Yes [21] |
| Type | ESC-CM | MSC-CM | iPSC-CM | AFSC-CM | AFSC-iPSC-CM |
|---|---|---|---|---|---|
| cTnT expression | Yes [22] | Yes [38,39] | Yes [40] | Yes [41] | Yes [41] |
| Contractility | Yes [22] | No [7] | Yes [42] | No [41] | Yes [41] |
| Therapeutic effect of myocardial infarction in animal | Yes [20] | Restrictive [43] | Yes [44] | Restrictive [45] | Yes [21] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, Y.-H.; Wang, S.P.H.; Chang, H.-Y.; Yang, P.-J.; Liu, P.-Y.; Liu, Y.-W. Progress and Challenges of Amniotic Fluid Derived Stem Cells in Therapy of Ischemic Heart Disease. Int. J. Mol. Sci. 2021, 22, 102. https://doi.org/10.3390/ijms22010102
Fang Y-H, Wang SPH, Chang H-Y, Yang P-J, Liu P-Y, Liu Y-W. Progress and Challenges of Amniotic Fluid Derived Stem Cells in Therapy of Ischemic Heart Disease. International Journal of Molecular Sciences. 2021; 22(1):102. https://doi.org/10.3390/ijms22010102
Chicago/Turabian StyleFang, Yi-Hsien, Saprina P. H. Wang, Hsien-Yuan Chang, Pei-Jung Yang, Ping-Yen Liu, and Yen-Wen Liu. 2021. "Progress and Challenges of Amniotic Fluid Derived Stem Cells in Therapy of Ischemic Heart Disease" International Journal of Molecular Sciences 22, no. 1: 102. https://doi.org/10.3390/ijms22010102
APA StyleFang, Y.-H., Wang, S. P. H., Chang, H.-Y., Yang, P.-J., Liu, P.-Y., & Liu, Y.-W. (2021). Progress and Challenges of Amniotic Fluid Derived Stem Cells in Therapy of Ischemic Heart Disease. International Journal of Molecular Sciences, 22(1), 102. https://doi.org/10.3390/ijms22010102





