Characterization of FcXTH2, a Novel Xyloglucan Endotransglycosylase/Hydrolase Enzyme of Chilean Strawberry with Hydrolase Activity
Abstract
1. Introduction
2. Results
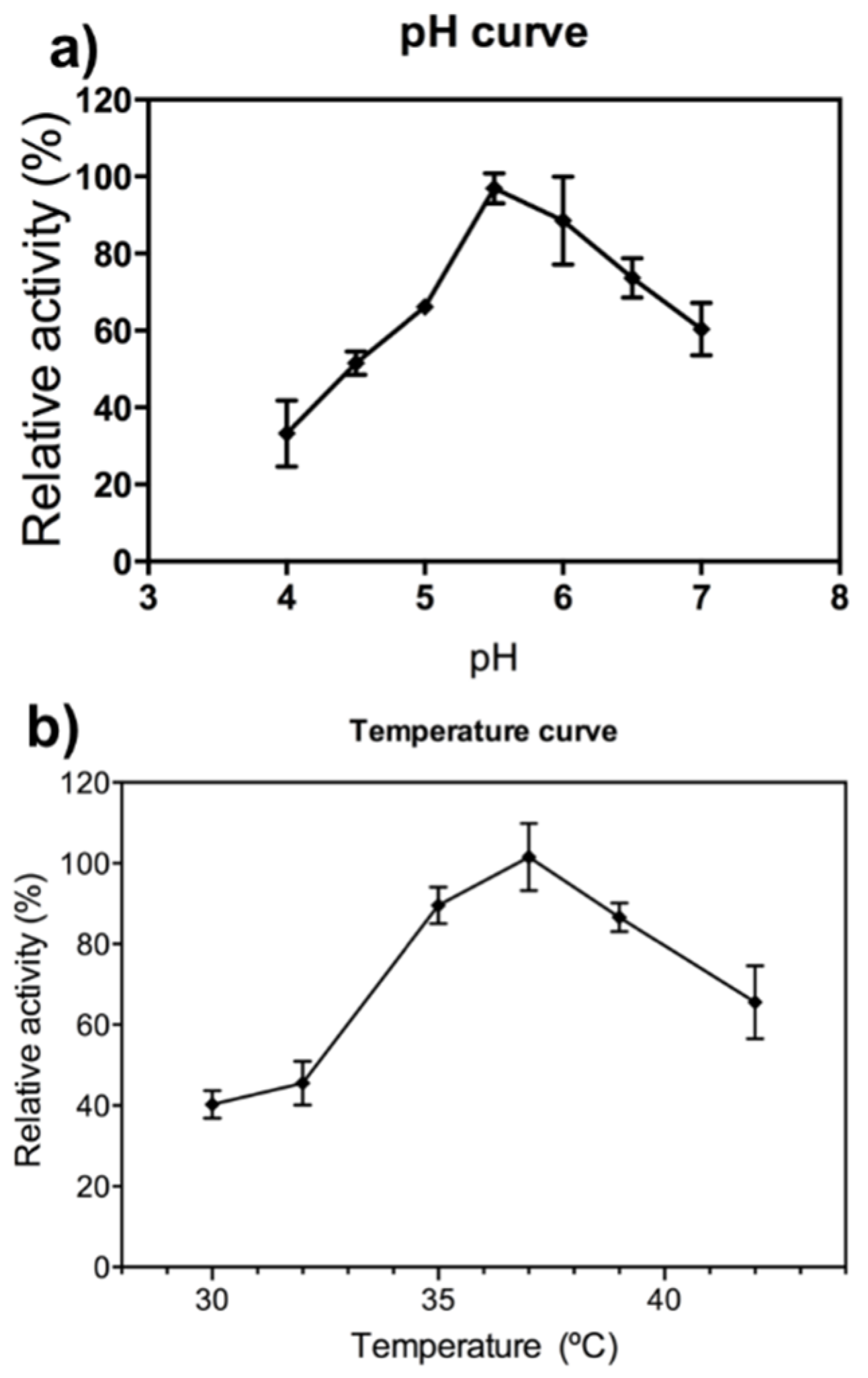
2.1. FcXTH2 Recombinant Protein Characterization
2.2. FcXTH1 Structural Model
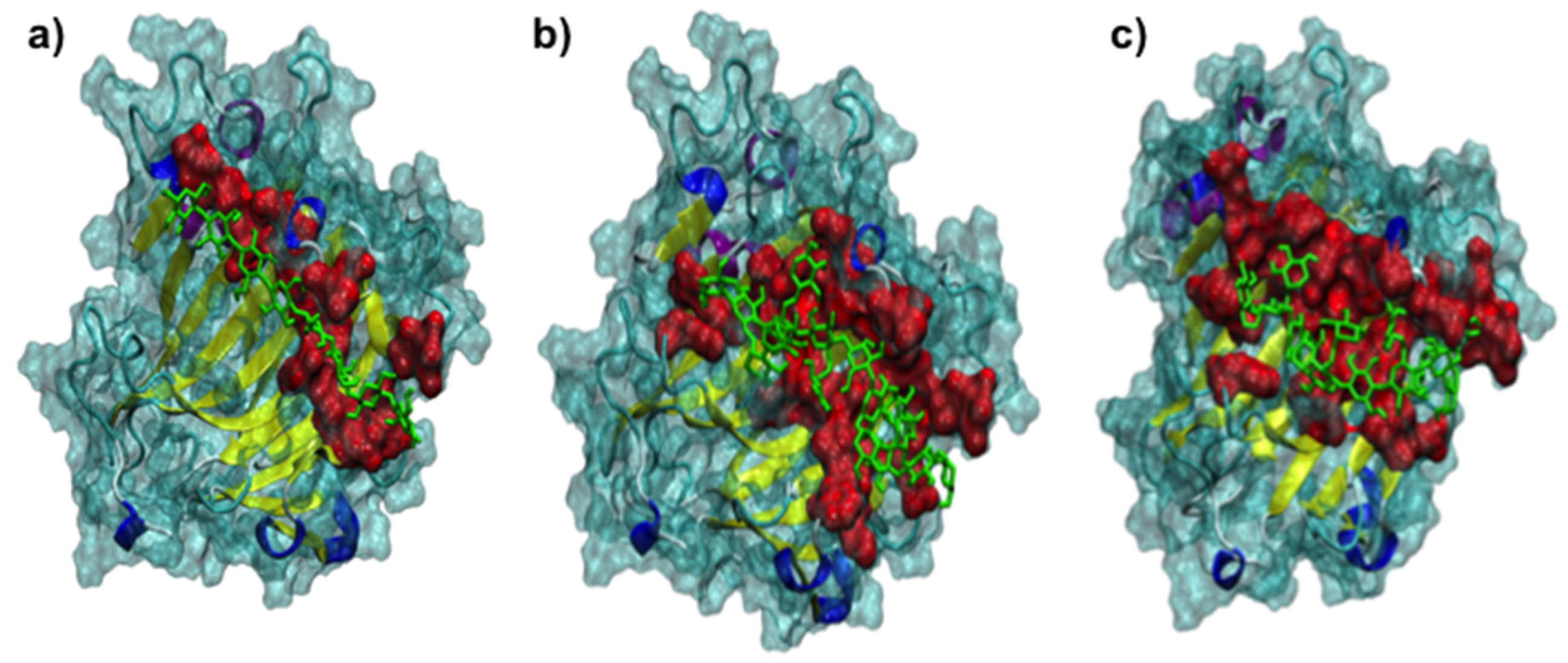
2.3. Protein-Ligand Interaction Analysis
3. Discussion
3.1. FcXTH2 Mainly Has XEH Activity
3.2. The 3D Structure of FcXTH2 and Protein-Ligand Interaction Based on Comparative Modeling and Docking Studies
4. Methods
4.1. Plant Material
4.2. Extraction of Proteins
4.3. Protein Production and Purification
4.4. XET and XEH Assays
4.5. pH and Temperature Dependence and Kinetic Characterization
4.6. Obtaining the 3D Structure of FcXTH2 by Homology Modeling
4.7. Protein-Ligand Interactions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| XET | Xyloglucan endotransglycosylase activity |
| XEH | Xyloglucan hydrolase activity |
| FaXTH2 | Xyloglucan endotransglycosylase/hydrolases from Fragaria chiloensis isoform 2. |
| XG | Xyloglucan |
| BCA | Disodium 2,20-bicinchoninic acid |
References
- Marchessault, R.H.; Sundararajan, P.R. Cellulose. In The Polysaccharides; Aspinall, G., Ed.; Academic Press Inc.: New York, NY, USA, 1983; Volume 2, pp. 11–95. [Google Scholar]
- Purves, C.B. Chain structure. In Cellulose and Cellulose Derivatives Part I; Ott, E., Spurlin, H.M., Grafflin, M.W., Eds.; Wiley-Interscience: New York, NY, USA, 1954; pp. 54–98. [Google Scholar]
- Pauly, M.; Gille, S.; Liu, L.; Mansoori, N.; De Souza, A.; Schultink, A.; Xiong, G. Hemicellulose biosynthesis. Planta 2013, 238, 627–642. [Google Scholar] [CrossRef]
- Brummell, D.A.; Harpster, M.H. Cell wall metabolism in fruit softening and quality and its manipulation in transgenic plants. Plant Mol. Biol. 2001, 47, 311–339. [Google Scholar] [CrossRef] [PubMed]
- Tuomivaara, S.T.; Yaoi, K.; O’Neill, M.A.; York, W.S. Generation and structural validation of a library of diverse xyloglucan-derived oligosaccharides, including an update on xyloglucan nomenclature. Carbohydr. Res. 2015, 402, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Kato, Y. A study of the structures of xyloglucans from the fruit cell walls of strawberry, persimmon, prune and banana. J. Appl. Glycosci. 2002, 49, 501–504. [Google Scholar] [CrossRef]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Eklof, J.M.; Brumer, H. The XTH gene family: An update on enzyme structure, function, and phylogeny in xyloglucan remodeling. Plant Physiol. 2010, 153, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Franková, L.; Fry, S.C. Biochemistry and physiological roles of enzymes that ‘cut and paste’ plant cell-wall polysaccharides. J. Exp. Bot. 2013, 64, 3519–3550. [Google Scholar] [CrossRef] [PubMed]
- Moya-León, M.A.; Mattus, E.; Herrera, R. Molecular events occurring during softening of strawberry fruit. Front. Plant Sci. 2019, 10, 615. [Google Scholar] [CrossRef]
- Iqbal, A.; Tang, W.; Wang, T.; Wang, H. Identification and functional characterization of the promoter driving “xyloglucan endotransglucosylase/hydrolase gene (XET)” gene for root growth in the desert Populus euphratica. S. Afr. J. Bot. 2017, 112, 437–446. [Google Scholar] [CrossRef]
- Wolf, S.; Hematy, K.; Hofte, H. Growth control and cell wall signaling in plants. Annu. Rev. Plant Biol. 2012, 63, 381–407. [Google Scholar] [CrossRef]
- Johansson, P.; Brumer, H.; Baumann, M.J.; Kallas, A.M.; Henriksson, H.; Denman, S.E.; Teeri, T.T.; Jones, T.A. Crystal structures of a poplar xyloglucan endotransglycosylase reveal details of transglycosylation acceptor binding. Plant Cell 2004, 16, 874–886. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Denman, S.; Brumer, H.; Kallas, A.M.; Henriksson, H.; Bergfors, T.; Teeri, T.T.; Jones, T.A. Crystallization and preliminary X-ray analysis of a xyloglucan endotransglycosylase from Populus tremula x tremuloides. Acta Crystallogr. D Biol. Crystallogr. 2003, 59, 535–537. [Google Scholar] [CrossRef] [PubMed]
- Minic, Z.; Jouanin, L. Plant glycoside hydrolases involved in cell wall polysaccharide degradation. Plant Physiol. Biochem. 2006, 44, 435–449. [Google Scholar] [CrossRef] [PubMed]
- Michailidis, G.; Argiriou, A.; Darzentas, D.; Tsaftaris, A. Analysis of xyloglucan endotransglycosylase/hydrolase (XTH) genes from allotetraploid (Gossypium hirsutum) cotton and its diploid progenitors expressed during fiber elongation. J. Plant Physiol. 2009, 166, 403–416. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, N.; Sunagawa, N.; Tamura, S.; Yokoyama, R.; Ueda, M.; Igarashi, K.; Nishitani, K. The plant cell-wall enzyme AtXTH3 catalyses covalent cross-linking between cellulose and cello-oligosaccharide. Sci. Rep. 2017, 7, 46099. [Google Scholar] [CrossRef] [PubMed]
- Rubianes, D.; Valdivia, E.R.; Revilla, G.; Zarra, I.; Sampedro, J. Xyloglucan exoglycosidases in the monocot model Brachypodium distachyon and the conservation of xyloglucan disassembly in angiosperms. Plant Mol. Biol. 2019, 100, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Campbell, P.; Braam, J. Xyloglucan endotransglycosylases: Diversity of genes, enzymes and potential wall-modifying functions. Trends Plant Sci. 1999, 4, 361–366. [Google Scholar] [CrossRef]
- Rose, J.K.C.; Braam, J.; Fry, S.C.; Nishitani, K. The XTH family of enzymes involved in xyloglucan endotransglucosylation and endohydrolysis: Current perspectives and a new unifying nomenclature. Plant Cell Physiol. 2002, 43, 1421–1435. [Google Scholar] [CrossRef] [PubMed]
- Fry, S.C. Primary cell wall metabolism: Tracking the careers of wall polymers in living plant cells. New Phytol. 2004, 161, 641–675. [Google Scholar] [CrossRef]
- Nishitani, K.; Vissenberg, K. Roles of the XTH protein family in the expanding cell. In Plant Cell Monographs; Verbelen, J.K., Vissenberg, K., Eds.; Springer: Berlin, Germany, 2006. [Google Scholar]
- Méndez-Yañez, Á.; Beltrán, D.; Campano-Romero, C.; Molinett, S.; Herrera, R.; Moya-León, M.A.; Morales-Quintana, L. Glycosylation is important for FcXTH1 activity as judged by its structural and biochemical characterization. Plant Physiol. Biochem. 2017, 119, 200–210. [Google Scholar] [CrossRef]
- Morales-Quintana, L.; Carrasco-Orellana, C.; Beltrán, D.; Moya-León, M.A.; Herrera, R. Molecular insights of a xyloglucan endo-transglycosylase/hydrolase of radiata pine (PrXTH1) expressed in response to inclination: Kinetics and computational study. Plant Physiol. Biochem. 2019, 136, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.Z.; Zhu, X.F.; Miller, J.G.; Gregson, T.; Zheng, S.J.; Fry, S.C. Distinct catalytic capacities of two aluminium-repressed Arabidopsis thaliana xyloglucan endotransglucosylase/hydrolases, XTH15 and XTH31, heterologously produced in Pichia. Phytochemistry 2015, 112, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, C.; Ramos, P.; Carrasco, C.; Moya-León, M.A.; Herrera, R. Cloning and characterization of a xyloglucan endo-transglycosylase/hydrolase gene expressed in response to inclination in radiata pine seedlings. Tree Genet. Genomes 2014, 10, 1305–1315. [Google Scholar] [CrossRef]
- Kushwah, S.; Banasiak, A.; Nishikubo, N.; Derba-Maceluch, M.; Majda, M.; Endo, S.; Kumar, V.; Gomez, L.; Gorzsás, A.; McQueen-Mason, S.; et al. Arabidopsis XTH4 and XTH9 contribute to wood cell expansion and secondary wall formation. Plant Physiol. 2020, 01529. [Google Scholar] [CrossRef] [PubMed]
- Miedes, E.; Herbers, K.; Sonnewald, U.; Lorences, E.P. Overexpression of a cell wall enzyme reduces xyloglucan depolymerization and softening of transgenic tomato fruits. J. Agric. Food Chem. 2010, 12, 5708–5713. [Google Scholar] [CrossRef]
- Han, Y.; Zhu, Q.; Zhang, Z.; Meng, K.; Hou, Y.; Ban, Q.; Suo, J.; Rao, J. Analysis of xyloglucan endotransglycosylase/hydrolase (XTH) genes and diverse roles of isoenzymes during persimmon fruit development and postharvest softening. PLoS ONE 2015, 10, 4. [Google Scholar] [CrossRef]
- Han, Y.; Ban, Q.; Hou, Y.; Meng, K.; Suo, J.; Rao, J. Isolation and characterization of two Persimmon xyloglucan endotransglycosylase/hydrolase (XTH) genes that have divergent functions in cell wall modification and fruit postharvest softening. Front. Plant Sci. 2016, 7, 624. [Google Scholar] [CrossRef]
- Han, Y.; Ban, Q.; Li, H.; Hou, Y.; Jin, M.; Han, S.; Rao, J. DkXTH8, a novel xyloglucan endotransglucosylase/hydrolase in persimmon, alters cell wall structure and promotes leaf senescence and fruit postharvest softening. Sci. Rep. 2017, 6, 39155. [Google Scholar] [CrossRef]
- Nardi, C.; Villarreal, N.; Opazo, M.C.; Martínez, G.; Moya-León, M.A.; Civello, P.M. Expression of FaXTH1 and FaXTH2 genes in strawberry fruit. Cloning of promoter regions and effect of plant growth regulators. Sci. Hortic. 2014, 165, 111–122. [Google Scholar] [CrossRef]
- Witasari, L.D.; Huang, F.-C.; Hoffmann, T.; Rozhon, W.; Fry, S.C.; Wilfried, S. Higher expression of the strawberry xyloglucan endotransglucosylase/hydrolase genes FvXTH9 and FvXTH6 accelerates fruit ripening. Plant J. 2019, 100, 1237–1253. [Google Scholar] [CrossRef]
- Li, Q.; Li, H.; Yin, C.; Wang, X.; Jiang, Q.; Zhang, R.; Ge, F.; Chen, Y.; Yang, L. Genome-wide identification and characterization of xyloglucan endotransglycosylase/hydrolase in Ananas comosus during development. Genes 2019, 10, 537. [Google Scholar] [CrossRef] [PubMed]
- Morales-Quintana, L.; Ramos, P. Chilean strawberry (Fragaria chiloensis): An integrative and comprehensive review. Food Res. Int. 2019, 119, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, C.R.; Pimentel, P.; Gaete-Eastman, C.; Moya, M.; Herrera, R.; Caligari, P.D.S.; Moya-Leóon, M.A. Softening rate of the Chilean strawberry (Fragaria chiloensis) fruit reflects the expression of polygalacturonase and pectate lyase genes. Postharvest Biol. Technol. 2008, 49, 210–220. [Google Scholar] [CrossRef]
- Opazo, M.C.; Figueroa, C.R.; Henriquez, J.; Herrera, R.; Bruno, C.; Valenzuela, P.D.T.; Moya-León, M.A. Characterization of two divergent cDNAs encoding xyloglucan endotransglucosylase/ hydrolase (XTH) expressed in Fragaria chiloensis fruit. Plant Sci. 2010, 179, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Riffo, F.; Gaete-Eastman, C.; Stappung, Y.; Lizana, R.; Herrera, R.; Moya-León, M.A.; Morales-Quintana, L. Comparative in silico study of the differences in the structure and ligand interaction properties of three alpha-expansin proteins from Fragaria chiloensis fruit. J. Biomol. Struct. Dyn. 2019, 37, 3245–3258. [Google Scholar] [CrossRef] [PubMed]
- Ramos, P.; Parra-Palma, C.; Figueroa, C.R.; Zuñiga, P.E.; Valenzuela-Riffo, F.; Gonzalez, J.; Gaete-Eastman, C.; Morales-Quintana, L. Cell wall-related enzymatic activities and transcriptional profiles in four Strawberry (Fragaria x ananassa) cultivars during fruit development and ripening. Sci. Hortic. 2018, 238, 325–332. [Google Scholar] [CrossRef]
- Opazo, M.C.; Lizana, R.; Stappung, Y.; Davis, T.M.; Herrera, R.; Moya-León, M.A. XTHs from Fragaria vesca: Genomic structure and transcriptomic analysis in ripening fruit and other tissues. BMC Genom. 2017, 18, 852. [Google Scholar] [CrossRef]
- Sulová, Z.; Lednická, M.; Farkâs, V. A colorimetric assay for xyloglucan endotransglycosylase from germinating seeds. Anal. Biochem. 1995, 229, 80–85. [Google Scholar] [CrossRef]
- Arnal, G.; Attia, M.A.; Asohan, J.; Brumer, H. A low-volume, parallel copper-bicinchoninic acid (BCA) assay for glycoside hydrolases. Methods Mol. Biol. 2017, 1588, 3–14. [Google Scholar] [CrossRef]
- Baumann, M.J.; Eklof, J.M.; Michel, G.; Kallas, A.M.; Teeri, T.T.; Czjzek, M.; Brumer, H., 3rd. Structural evidence for the evolution of xyloglucanase activity from xyloglucan endo-transglycosylases: Biological implications for cell wall metabolism. Plant Cell 2007, 19, 1947–1963. [Google Scholar] [CrossRef]
- Giovannoni, J. Molecular biology of fruit maturation and ripening. Annu. Rev. Plant Biol. 2001, 52, 725–749. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.K.C.; Bennett, A.B. Cooperative disassembly of the cellulose-xyloglucan network of plant cell walls: Parallels between cell expansion and fruit ripening. Trends Plant Sci. 1999, 4, 176–183. [Google Scholar] [CrossRef]
- Perkins-Veazie, P. Growth and ripening of strawberry fruit. Hortic. Rev. 1995, 17, 267–297. [Google Scholar]
- Manning, K. Changes in gene expression during strawberry fruit ripening and their regulation by auxin. Planta 1994, 194, 62–68. [Google Scholar] [CrossRef]
- Manning, K. Isolation of a set of ripening-related genes from strawberry: Their identification and possible relationship to fruit quality traits. Planta 1998, 205, 622–631. [Google Scholar] [CrossRef] [PubMed]
- Medina-Escobar, N.; Cardenas, J.; Moyano, E.; Caballero, J.L.; Muñoz-Blanco, J. Cloning, molecular characterization and expression pattern of a strawberry ripening-specific cDNA with sequence homology to pectate lyase from higher plants. Plant Mol. Biol. 1997, 34, 867–877. [Google Scholar] [CrossRef]
- Atkinson, R.G.; Johnston, S.L.; Yauk, Y.-K.; Sharma, N.N.; Schröder, R. Analysis of xyloglucan endotransglucosylase/hydrolase (XTH) gene families in kiwifruit and apple. Postharvest Biol. Technol. 2009, 51, 149–157. [Google Scholar] [CrossRef]
- Miedes, E.; Zarra, I.; Hoson, T.; Herbers, K.; Sonnewald, U.; Lorences, E.P. Xyloglucan endotransglucosylase and cell wall extensibility. J. Plant Physiol. 2011, 168, 196–203. [Google Scholar] [CrossRef]
- Saladié, M.; Rose, J.K.C.; Cosgrove, D.J.; Catalá, C. Characterization of a new xyloglucan endotransglucosylase/hydrolase (XTH) from ripening tomato fruit and implications for the diverse modes of enzymic action. Plant J. 2006, 47, 282–295. [Google Scholar] [CrossRef]
- Tabuchi, A.; Mori, H.; Kamisaka, S.; Hoson, T. A new type of endo xyloglucan transferase devoted to xyloglucan hydrolysis in the cell wall of azuki vean epicotyls. Plant Cell Physiol. 2001, 42, 154–161. [Google Scholar] [CrossRef]
- Fanutti, C.; Gidley, M.J.; Reid, J.S.G. Action of a pure xyloglucan endotransglycosylase (formerly called xyloglucan-specific endo-(1-4)-β-D-glucanase) from the cotyledons of germinated nasturtium seeds. Plant J. 1993, 3, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.F.; Shi, Y.Z.; Lei, G.J.; Fry, S.C.; Zhang, B.C.; Zhou, Y.H.; Braam, J.; Jiang, T.; Xu, X.Y.; Mao, Y.Z.; et al. XTH31, encoding an in vitro XEH/XET-active enzyme, regulates aluminum sensitivity by modulating in vivo XET action, cell wall xyloglucan content, and aluminum binding capacity in Arabidopsis. Plant Cell 2012, 24, 4731–4747. [Google Scholar] [CrossRef] [PubMed]
- Maris, A.; Suslov, D.; Fry, S.C.; Verbelen, J.-P.; Vissenberg, K. Enzymic characterization of two recombinant xyloglucan endotransglucosylase/hydrolase (XTHs) proteins of Arabidopsis and their effect on root growth and cell wall extension. J. Exp. Bot. 2009, 60, 3959–3972. [Google Scholar] [CrossRef] [PubMed]
- De Silva, J.; Jarman, C.D.; Arrowsmith, D.A.; Stronach, M.S.; Chengappa, S.; Sidebottom, C.; Reid, J.S.G. Molecular characterization of a xyloglucanspecific endo-(1-4)-beta-D-glucanase (xyloglucan endotransglycosylase) from nasturtium seeds. Plant J. 1993, 3, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y.; Yokoyama, R.; Osakabe, K.; Toki, S.; Nishitani, K. Function of xyloglucan endotransglucosylase/hydrolases in rice. Ann. Bot. 2014, 114, 1309–1318. [Google Scholar] [CrossRef]
- McGregor, N.; Yin, V.; Tung, C.-C.; Van Petegem, F.; Brumer, H. Crystallographic insight into the evolutionary origins of xyloglucan endotransglycosylases and endohydrolases. Plant J. 2017, 89, 651–670. [Google Scholar] [CrossRef]
- Mark, P.; Baumann, J.M.; Eklöf, J.M.; Gullfot, F.; Michel, D.; Kallas, A.M.; Teeri, T.T.; Brumer, H.; Czjzek, M. Analysis of nasturtium TmNXG1 complexes by crystallography and molecular dynamics provides detailed insight into substrate recognition by family GH16 xyloglucan endo-transglycosylases and endohydrolases. Proteins 2009, 75, 820–836. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–252. [Google Scholar] [CrossRef]
- Morales-Quintana, L.; Fuentes, L.; Gaete-Eastman, C.; Herrera, R.; Moya-León, M.A. Structural characterization and substrate specificity of VpAAT1 protein related to ester biosynthesis in mountain papaya fruit. J. Mol. Graph. Model. 2011, 29, 635–642. [Google Scholar] [CrossRef]
- Šali, A.; Blundell, T.L. Comparative protein modelling by satisfaction of spatial restraints. J. Mol. Biol. 1993, 234, 779–815. [Google Scholar] [CrossRef]
- Brooks, B.R.; Bruccoleri, R.E.; Olafson, B.D.; States, D.J.; Swaminathan, S.; Karplus, M. CHARMM: A program for macromolecular energy minimization and dynamics calculation. J. Comp. Chem. 1983, 4, 187–217. [Google Scholar] [CrossRef]
- Phillips, J.C.; Braun, R.; Wang, W.; Gumbart, J.; Tajkhorshid, E.; Villa, E.; Chipot, C.; Skeel, R.D.; Kale, L.; Schulten, K. Scalable molecular dynamics with NAMD. J. Comput. Chem. 2005, 26, 1781–1802. [Google Scholar] [CrossRef] [PubMed]
- Sippl, M.J. Recognition of errors in three-dimensional structures of proteins. Proteins 1993, 17, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Wiederstein, M.; Sippl, M. ProSA-web: Interactive web service for the recognition of errors in three-dimensional structures of proteins. Nucleic Acids Res. 2007, 35, W407–W410. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, R.A.; MacArthur, M.W.; Moss, D.S.; Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Cryst. 1993, 26, 283–291. [Google Scholar] [CrossRef]
- Gaete-Eastman, C.; Morales-Quintana, L.; Herrera, R.; Moya-León, M.A. In silico analysis of the structure and binding site features of an α-expansin protein from mountain papaya fruit (VpEXPA2), through molecular modeling, docking and dynamics simulation studies. J. Mol. Model. 2015, 21, 5. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDockVina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]

| Fraction | Volume (mL) | Protein (mg/mL) | XET Activity (a.u. μg−1 h−1) | XEH Activity (a.u. μg−1 h−1) | Purification Fold (XEH Act) |
|---|---|---|---|---|---|
| Concentrated crude extract | 10 | 6.82 | 6.47 ± 1.6 | 1.23 ± 2.3 | 1 |
| Anion exchange chromatography Eluted protein (0.5 M NaCl) | 10 | 5.70 | 2.56 ± 2.0 | 5.67 ± 2.0 | 4.61 |
| Affinity chromatography Eluted purified protein (25 mM imidazole) | 2 | 3.91 | 2.71 ± 1.1 | 15.30 ± 1.5 | 12.43 |
| Cellulase from A. niger 1 | - | - | 0.0 ± 1.1 | 65.7 ± 4.0 | - |
| Recombinant FcXTH1 protein from F. chiloensis 2 | - | - | 63.45 ± 4.0 | 0.0 | - |
| Fruit extract of F. chiloensis 3 | - | - | 75.0 ± 3.2 | 13.82 ± 0.7 | - |
| Empty vector pPICZαA 4 | - | - | 4.24 ± 2.6 | 7.4 ± 2.7 | - |
| Protein Name | KM # (mg mL−1) | Vmax (μg mL−1 s−1) | kcat # (s−1) | kcat * KM−1 ((mg/mL)−1 s−1) |
|---|---|---|---|---|
| FcXTH2 | 0.029 ± 0.003 | 4.1 × 105 | 8.21 × 103 | 2.83 × 102 |
| Substrates | Affinity Energy ( kcal mol−1) |
|---|---|
| XXXGXXXG | −9.2 ± 0.9 |
| XXFGXXFG | −7.1 ± 0.8 |
| cellodextrin 8-mer | −7.2 ± 1.1 |
| FcXTH2 in Complex with | ||
|---|---|---|
| Cellodextrin 8-mer | XXFGXXFG | XXXGXXXG |
| Asn13 | Asn13 | Asn13 |
| Val43 | Val43 | Val43 |
| Gln45 | Gln45 | Ala70 |
| Ser74 | Leu47 | Met78 |
| Asp77 | Tyr73 | Pro80 |
| His83 | His83 | His83 |
| Gly109 | Asn104 | Glu85 |
| Thr111 | Tyr106 | Asp87 |
| Thr113 | Gly109 | Gln102 |
| Ser169 | Arg118 | Asn104 |
| Tyr171 | Trp180 | Tyr106 |
| Ile213 | Thr111 | |
| Gly114 | ||
| Glu116 | ||
| Arg118 | ||
| Ser169 | ||
| Thr173 | ||
| Trp180 | ||
| Arg259 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morales-Quintana, L.; Beltrán, D.; Mendez-Yañez, Á.; Valenzuela-Riffo, F.; Herrera, R.; Moya-León, M.A. Characterization of FcXTH2, a Novel Xyloglucan Endotransglycosylase/Hydrolase Enzyme of Chilean Strawberry with Hydrolase Activity. Int. J. Mol. Sci. 2020, 21, 3380. https://doi.org/10.3390/ijms21093380
Morales-Quintana L, Beltrán D, Mendez-Yañez Á, Valenzuela-Riffo F, Herrera R, Moya-León MA. Characterization of FcXTH2, a Novel Xyloglucan Endotransglycosylase/Hydrolase Enzyme of Chilean Strawberry with Hydrolase Activity. International Journal of Molecular Sciences. 2020; 21(9):3380. https://doi.org/10.3390/ijms21093380
Chicago/Turabian StyleMorales-Quintana, Luis, Dina Beltrán, Ángela Mendez-Yañez, Felipe Valenzuela-Riffo, Raúl Herrera, and María Alejandra Moya-León. 2020. "Characterization of FcXTH2, a Novel Xyloglucan Endotransglycosylase/Hydrolase Enzyme of Chilean Strawberry with Hydrolase Activity" International Journal of Molecular Sciences 21, no. 9: 3380. https://doi.org/10.3390/ijms21093380
APA StyleMorales-Quintana, L., Beltrán, D., Mendez-Yañez, Á., Valenzuela-Riffo, F., Herrera, R., & Moya-León, M. A. (2020). Characterization of FcXTH2, a Novel Xyloglucan Endotransglycosylase/Hydrolase Enzyme of Chilean Strawberry with Hydrolase Activity. International Journal of Molecular Sciences, 21(9), 3380. https://doi.org/10.3390/ijms21093380








