1. Introduction
The bark, the total of all tissues outside of the vascular cambium, represents the outermost layer of stems of woody plants [
1]. It may fulfil a multitude of different physiological and ecological functions such as photosynthesis [
2], transport plus storage of various substances [
3,
4] and stemflow [
5]. Additionally, owing to its peripheral position in a plant’s stem, it fulfills several protective functions. These protective functions include, among others, herbivore and pathogen resistance and can be subdivided into chemical and anatomical defense mechanisms. The chemical defense mechanisms are based on the release of chemicals such as phenols, terpenoids or alkaloids that are harmful to the potential invaders [
6]. The anatomical defense, on the other hand, is based on the mere mechanical resistance of the more or less thick bark, thus representing a protective barrier. This resistance may be enhanced by the accumulation of lignified cells [
7] like in the bark of the tree of heaven (
Ailanthus altissima (Mill.) Swingle). Apart from herbivores and pathogens, the anatomical defense also involves protection from abiotic factors such as high temperatures during forest fires [
8,
9,
10,
11] and mechanical influences. One example of a tree species possessing a bark with an outstanding insulation capability during forest fires is the giant sequoia (
Sequoiadendron giganteum Lindl.). As previously indicated, the high bark thickness and its loose structure with air inclusions are the reasons for this high heat insulation capability [
12,
13]. Typically, for trees, a lethal situation during forest fires occurs when the temperature of the vascular cambium increases above 60 °C [
13,
14]. The vascular cambium is located subjacent to the bark and is only a few cell layers thick. As this sensitive tissue is crucial for the formation of secondary xylem (wood) and phloem (bast)—and thus the bark—it is essential for the survival of the whole tree. This highlights the protective function of the tree bark, since it represents the only layer surrounding the cambium. At the same time, the inner and outer structure of the bark differs from species to species, resulting in differences concerning the protective potential.
The bark as a protective layer surrounding the cambium becomes important not only during forest fires, but also when the cambium needs to be protected from mechanical influences. Such mechanical influences can be represented, for example, by rockfall events, which occur regularly on the slopes of the Sierra Nevada [
15], where the giant sequoia is native to. The capability of the giant sequoia bark to protect the cambium during such high-velocity impacts by dissipating high amounts of impact energy has been proven before [
16,
17]. This property becomes even more evident when being compared to tree species being native to regions with no regular rockfall events, like the tree of heaven. The differences in bark thickness and composition are most likely the reasons for the divergent energy dissipation capabilities of these two tree species. Thereby, the bark of the giant sequoia is prominent in comparison to other tree barks concerning, e.g., its high fiber content, pronounced inner and outer structuring of the bark and its high proportion of air-filled spaces resulting in a low bark density. Determining the importance of these aspects of the giant sequoia bark composition in the context of impact protection is one aim of the study presented here.
In the context of impact protection, technical foams are often mentioned and discussed in the literature. As will be demonstrated later, the deformation behavior of the giant sequoia bark when being compressed resembles in many ways that of elastomeric open-pore (also termed open-cell) foams. Without anticipating the results presented in this paper, the authors will address several aspects of technical foams and biological structures with comparable properties in the introduction, as this will become important in the following chapters.
Concerning technical open-pore elastomeric foams, three phases of deformation when being compressed can be distinguished and also identified in stress–strain diagrams [
18]. In a first step, the deformation is linear elastic, until a strain of typically around 5%. Here, the pore/cell walls bend when the load is applied. In a second step, characterized by nonlinear elasticity, the cells buckle elastically. Typically, the stress remains at a nearly constant level (termed elastic collapse stress) during this step, thus forming a long flat plateau in the stress–strain diagram. Finally, in a third step, the cells collapse completely and the stress rises rapidly due to cell faces and edges being forced together (densification). Especially during the second phase, high amounts of energy are dissipated. This is exploited in foams for crash protection and energy-absorbing systems. However, also when not dealing with elastomeric foams, such a plateau can be present. In this case, the cells collapse plastically. Generally, in foams, energy can be dissipated by (a) plastic bending of the cell walls, (b) buckling of the cell walls, (c) fracture or (d) compression or flow of fluid, if a fluid is present. Depending on the material and the composition of a foam as well as on the strain and strain rate applied, the foam’s deformation behavior upon compression can be plastic, elastic, viscoelastic or a combination of those.
A comparison of biological structures with technical foams can already be found in the textbook on the mechanical behavior of plants, [
19], in which apart from many of the general considerations being relevant to the present study, comparisons of plant tissues to technical foams can also be found. As a more specific example, the authors of [
20] reported the mechanically similar behavior of certain closed-pore living plant tissues (in their case, the inner primary cortex of stems of the liana
Aristolochia macrophylla) and closed-pore technical foam rubbers. Gibson and Ashby [
18] compared the compaction behavior of cork with that of polystyrene foam. Both show a low collapse stress of the cells, so that the peak stress during impact is limited. By a progressive collapse of cell walls, large compressive strains are possible, and thus large amounts of energy are dissipated. Unlike these closed-pore systems, the authors of [
21,
22] examined the compaction behavior and impact-damping capability of the pomelo peel, an open-pore parenchymatous foam layer with liquid-filled struts. Driven by its capability to dissipate high amounts of kinetic energy during impact, the structure of the pomelo peel was replicated as silica [
23]. Furthermore, it was adapted and transferred to bio-inspired metal–metal composites [
24] and fiber-reinforced open-pore metal-foam-based components [
25]. For the pomelo peel, it could be proven that all the seven hierarchical levels examined were markedly involved in energy dissipation, and the structures on several of these hierarchical levels were transferred to the metal-foam-based components. First, a gradient in intercellular space from the outside towards the inside in the thickest part of the peel (the mesocarp) was adapted and transferred into the metal foam. Secondly, lignified and branched vascular bundles present in the mesocarp inspired the implementation of directed fiber bundles into the technical aluminum alloy foam structures. Thirdly, the two-material structure of the biological cells of the pomelo peel (i.e., the cell wall and membranes as a semipermeable solid hull filled with a liquid phase) was transferred to the metallic foam as a dual-layered strut. A beneficial influence on the mechanical behavior, namely in a higher plateau stress and a more homogeneous deformation behavior, was found as a result of adapting the foam structure on the abovementioned hierarchical levels.
In addition to the parenchymatous foam-like pomelo peel, the macadamia seed shell was investigated in [
25,
26]. However, in this case, the main focus was placed on crack deflection within the shell as a mechanism of impact protection. Energy dissipation by crack propagation has been proven as being relevant also in case of the quasibrittle inner layer (endocarp) of the fruit wall of the coconut [
17,
27,
28]. For these structures, the relevance of several hierarchical levels has also been pointed out when regarding the impact-damping capability. Like all the above-mentioned biological examples, the bark of the giant sequoia can also be seen as a material compound consisting of multiple biological tissues. However, in this case, none of the tissues relevant for energy dissipation are parenchymatous (like the pomelo peel) or of high density, consisting of a hard and tough compound of cells (like the macadamia seed shell and the coconut endocarp).
In the following sections, the composition of the giant sequoia bark from the cellular level to the integral level (whole bark) is elucidated, giving an overview of the test object. Following this, the energy dissipation capability and diverse mechanical properties of the bark are presented, starting with the whole bark and ranging down to single bark fibers. By this, the underlying principles of the bark’s high energy dissipation capability are presented and discussed step by step. Finally, we outline a potential transfer of the insights gained during the present study into energy-dissipating bio-inspired fibrous concrete and ceramic components for building construction.
3. Discussion
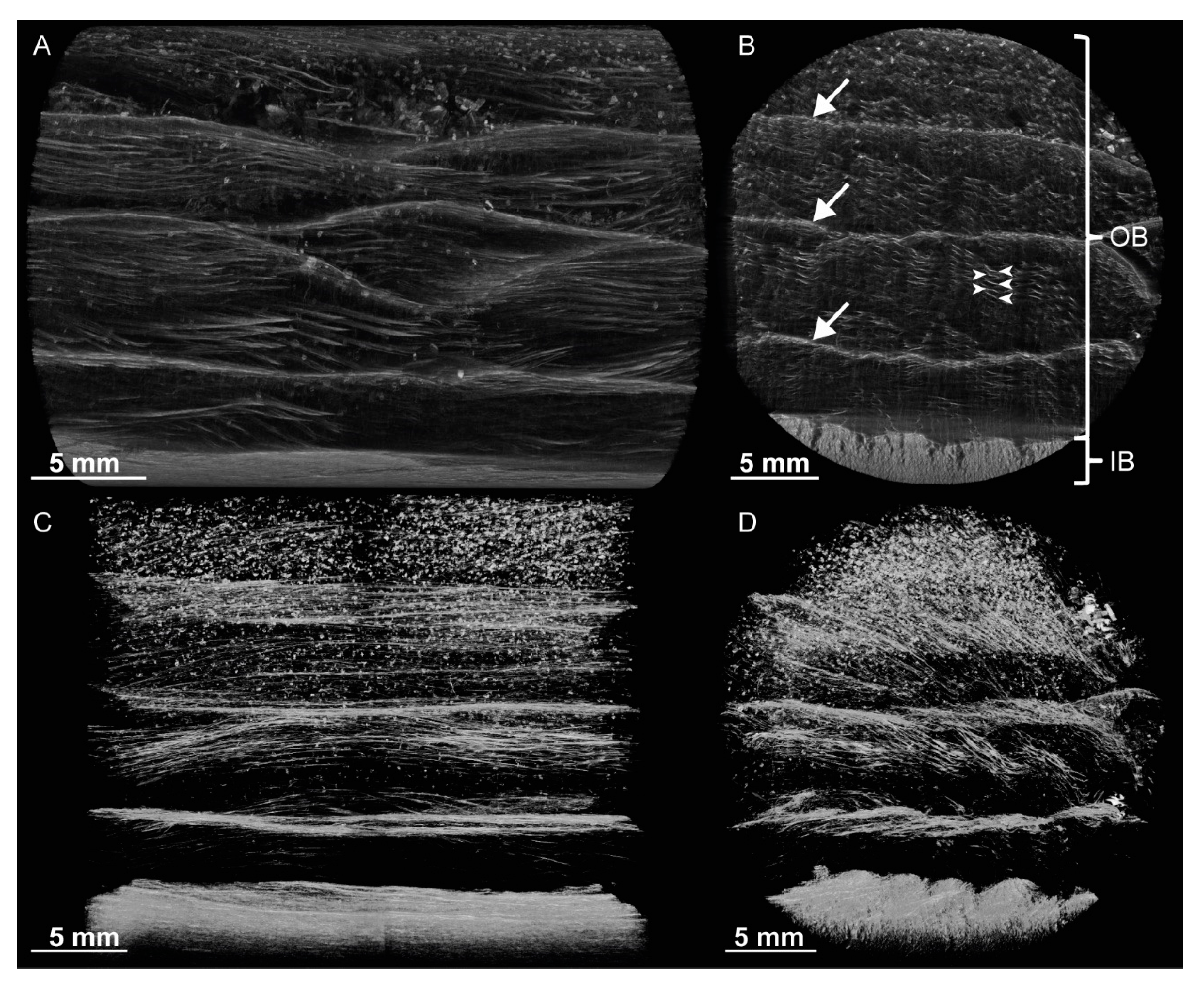
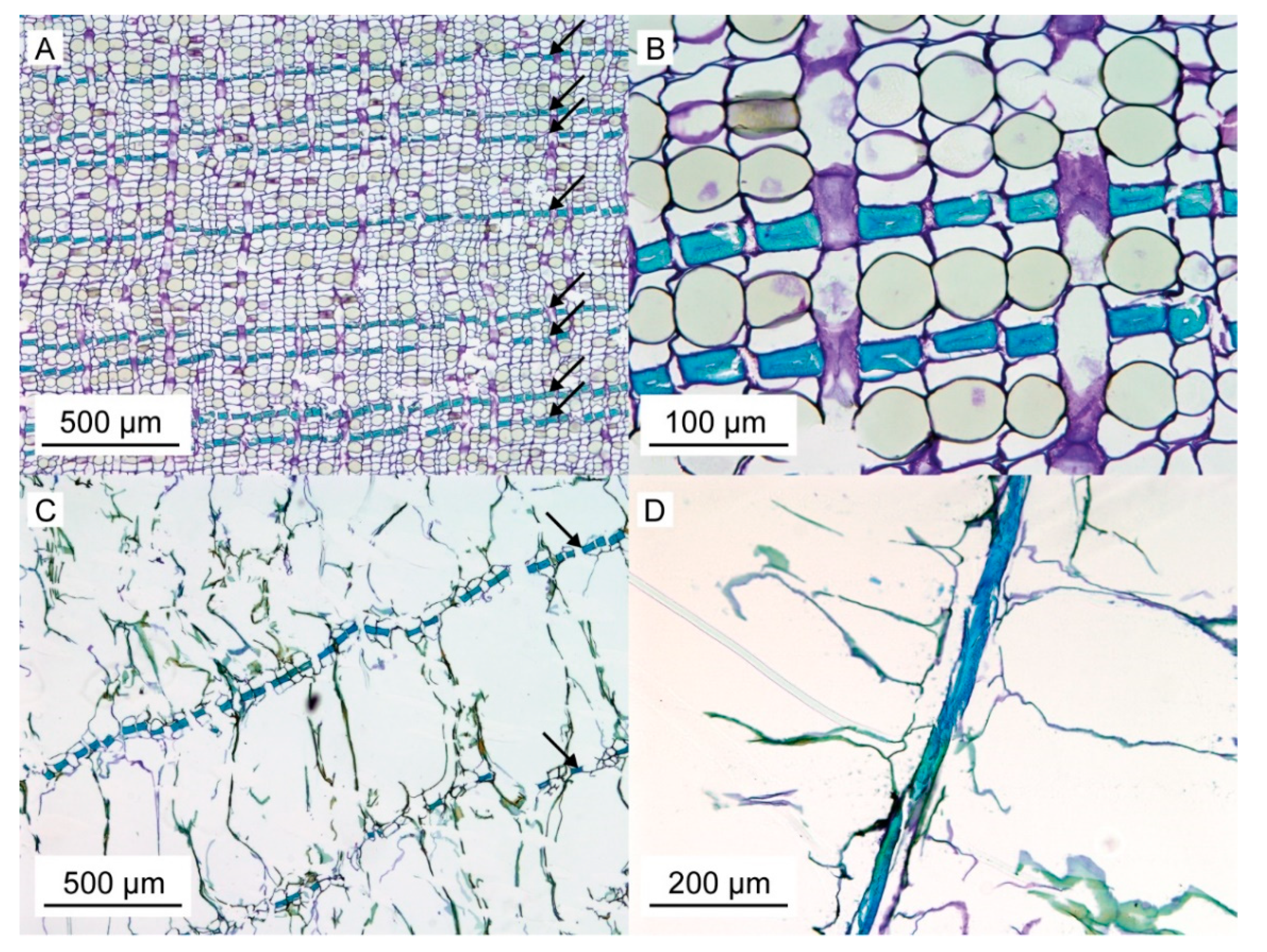
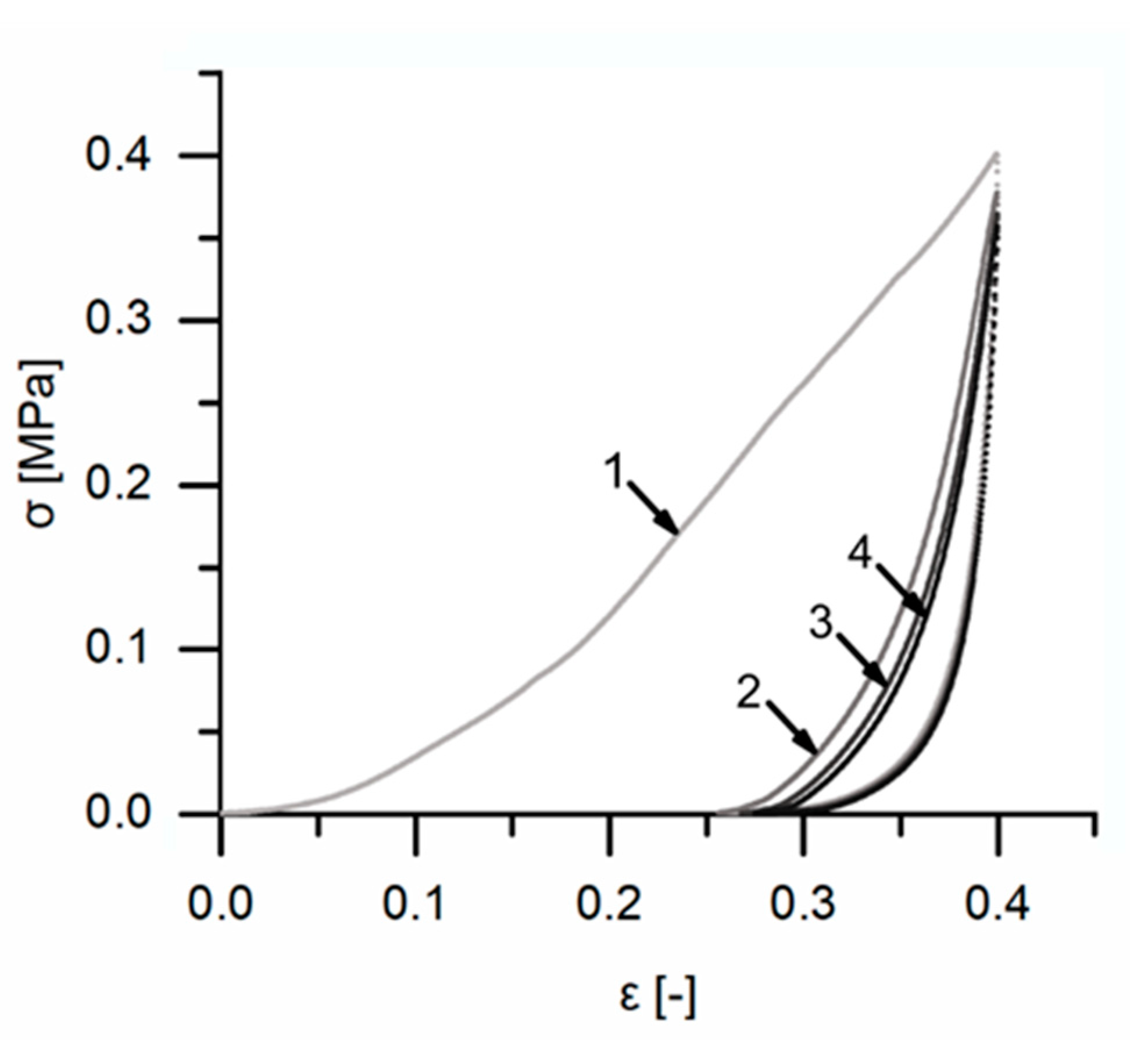
For a better understanding of the very specific composition and compaction behavior of the bark of the giant sequoia, the most relevant aspects will be compared to other selected biological and technical structures and materials. Concerning the stress–strain curves during quasistatic compression tests, the bark rather resembles an elastomeric open-pore foam, although the characteristic compression regimes of a typical elastomeric open-pore foam cannot clearly be distinguished for the bark. Instead, a smooth transition between the regimes can be observed, and no distinct plateau region can be found. During the high-speed videos of the dynamic drop-weight tests, macroscopically, a nonuniform compaction behavior can be observed. This also holds true for other biological structures, such as the pomelo peel [
21,
22], where the nonuniform compaction behavior is mainly attributed to the presence of lignified and branched vascular bundles, which can be regarded as fiber network in an open-pore parenchymatous foam layer. However, the bark of the giant sequoia (mainly) consists of fibers without any matrix in between; thus, its nonuniform compaction behavior can be mainly attributed to its layered structure on two orders. In contrast to these two biological structures, most technical elastomeric foams show a uniform compaction behavior. Concerning the absence of a transversal expansion during impact or compression (
Figure 6), the bark of the giant sequoia again can be compared to many polymer foams, which might even exhibit a negative Poisson’s ratio [
31,
32,
33]. From a structural point of view, the bark resembles a foam, especially when compared to the barks of most other tree species. It consists of a high amount of fibers and a considerable proportion of air-filled space, and the fibers form a network due to the interwoven fiber layers (
Figure 2 and
Figure 3). Thereby, the bark can be described as an open-pore foam-like structure with the bark fibers representing the ‘foam struts’. A similar comparison has also been made for the pomelo peel [
21,
22], where a considerable proportion of the peel’s impact-damping capabilities and viscoelastic behavior was attributed to the liquid cell-sap in the living parenchymatous cells, which represent the ‘foam struts’ of the peel. In contrast, in the giant sequoia bark, the thick cell walls of the dead fibers occupy almost the whole cell volume, leaving no or almost no cell lumen. Thus—besides water chemically bound in the cell wall—only small quantities of “free” water can be stored in this cell lumen, which is even further reduced upon drying of the bark prior to testing. Therefore, only a minor influence of liquid stored within the fibers on the deformation behavior is to be expected for the giant sequoia bark. The viscoelastic component occurring during bark compression seems to be a material and/or structure immanent property rather than being based on liquid displacement or squeezing.
Concerning wood and wood composites, the authors of [
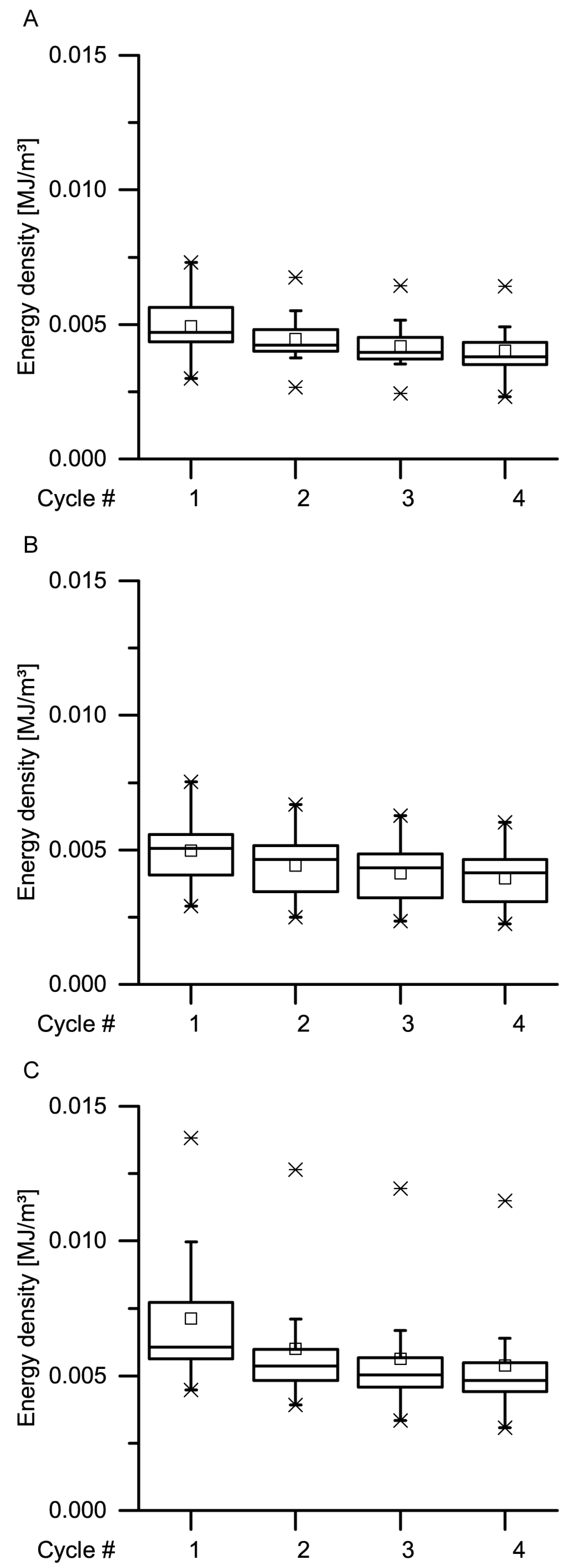
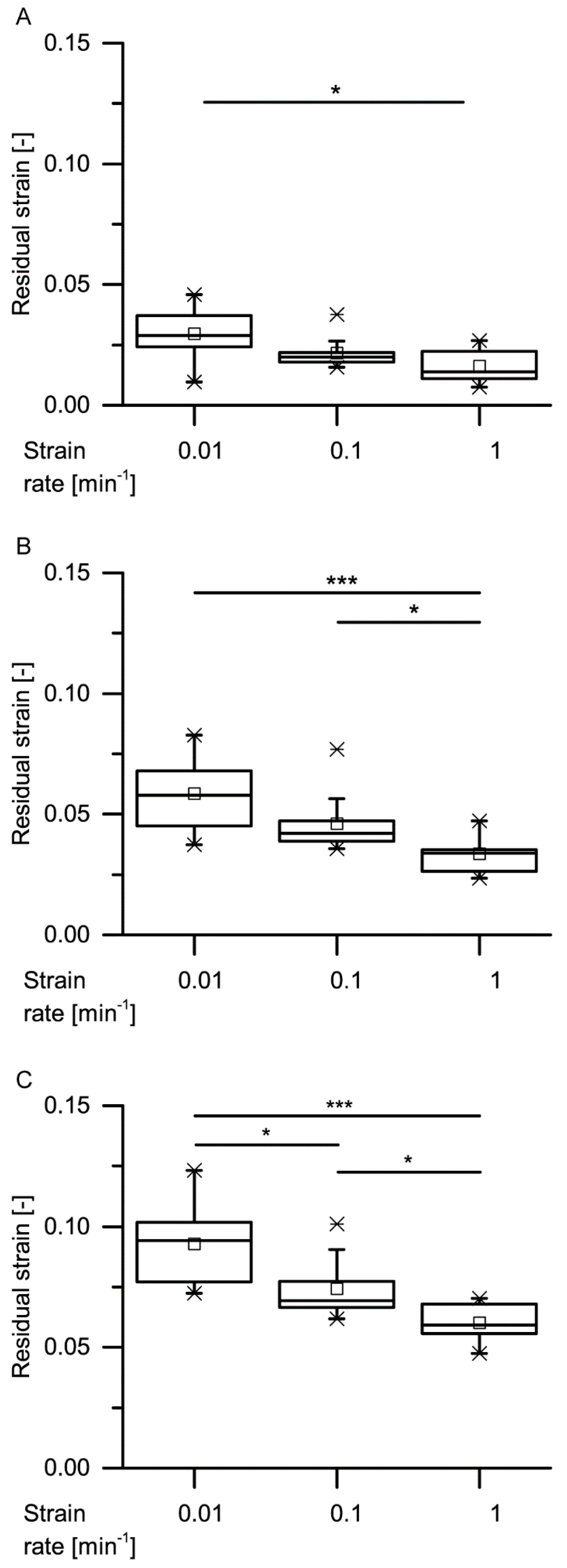
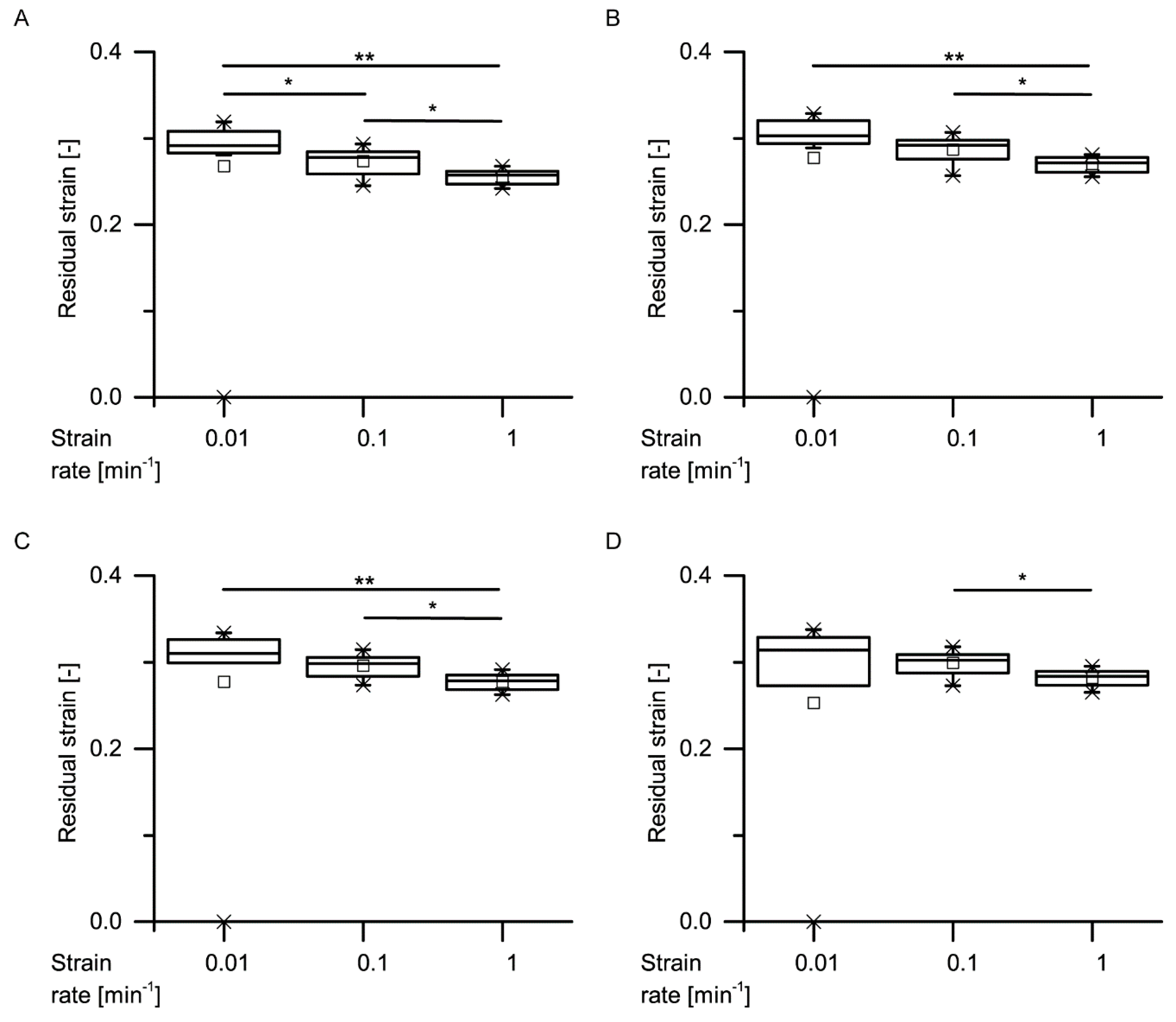
34] stated that during constant loading, initially, all the deformation is elastic. With elapsing time, the viscous (i.e., nonelastic) component responds, resulting in a decrease of the elastic component. In general, this holds true also for the bark samples under both quasistatic stepwise cyclic compression and cyclic compression with unchanged maximum compression. Even though the residual strains (representing the nonelastic components) significantly decrease with increasing strain rate for almost all cycles, a considerable amount of nonelastic components is still present during all these tests (
Figure 9,
Figure 10,
Figure 11 and
Figure 12).
When comparing the energy dissipation mechanisms of the giant sequoia bark with other biological protection structures such as pomelo peel [
21,
22], macadamia seed shell [
25,
26] and the coconut fruit wall [
17,
28], it could be shown that all these structures protect the inlying organs or tissues during impact events mainly by the reduction of force peaks. Hard and tough structures such as the coconut endocarp and macadamia seed shell dissipate energy by crack path deviation and elongation—energy dissipation mechanisms which are not present in the pomelo peel or giant sequoia bark, whose energy dissipation mechanisms are based on pronounced viscoelastic deformation.
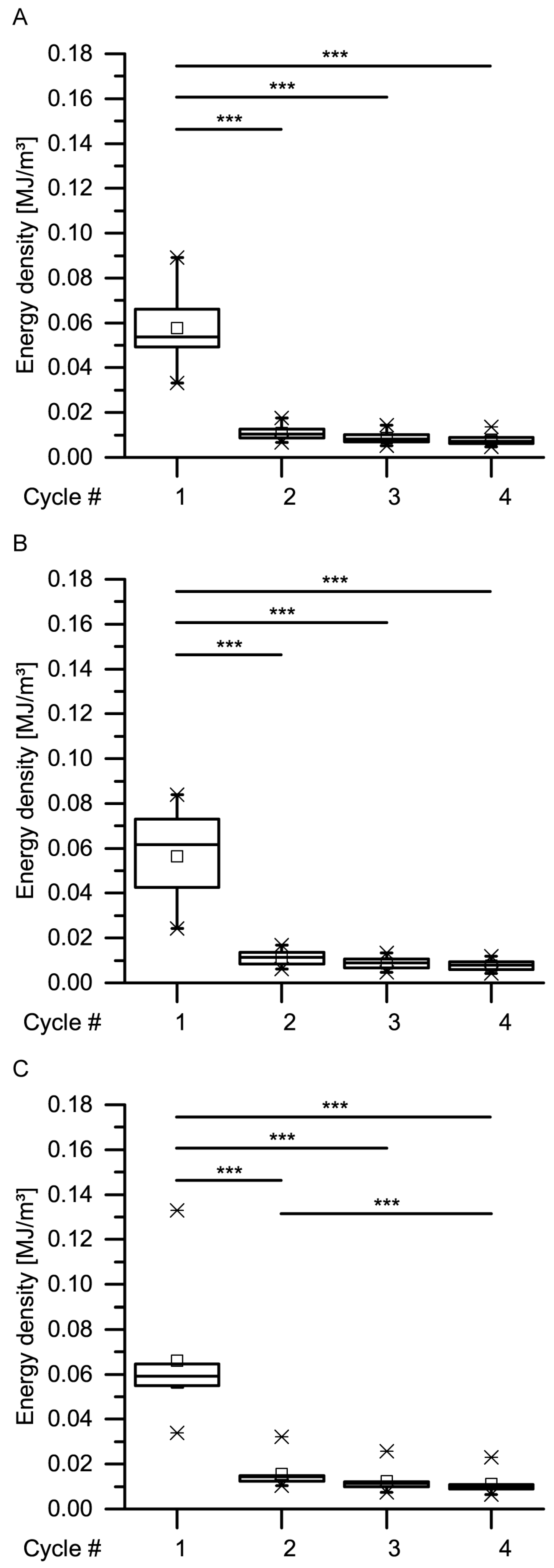
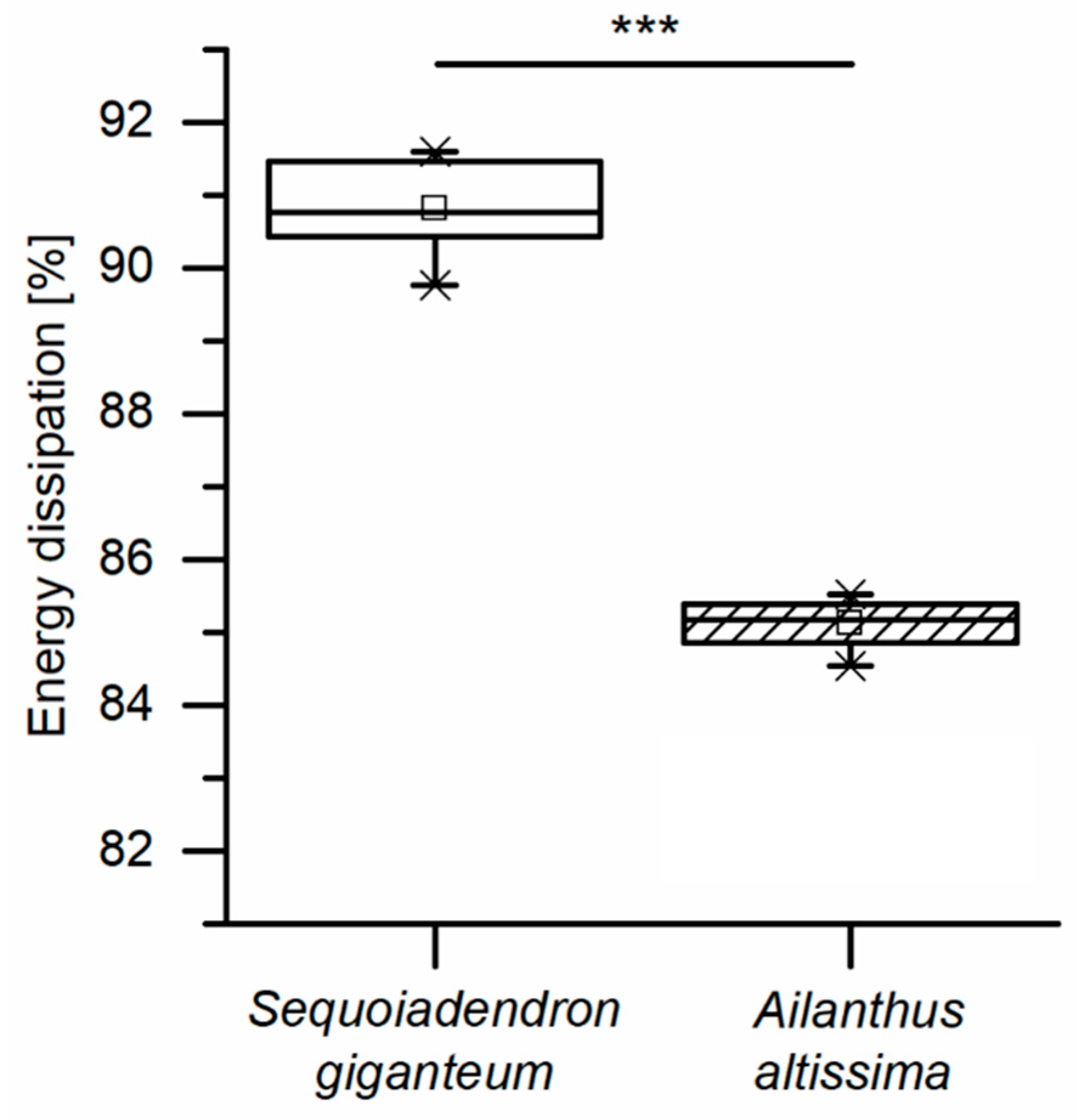
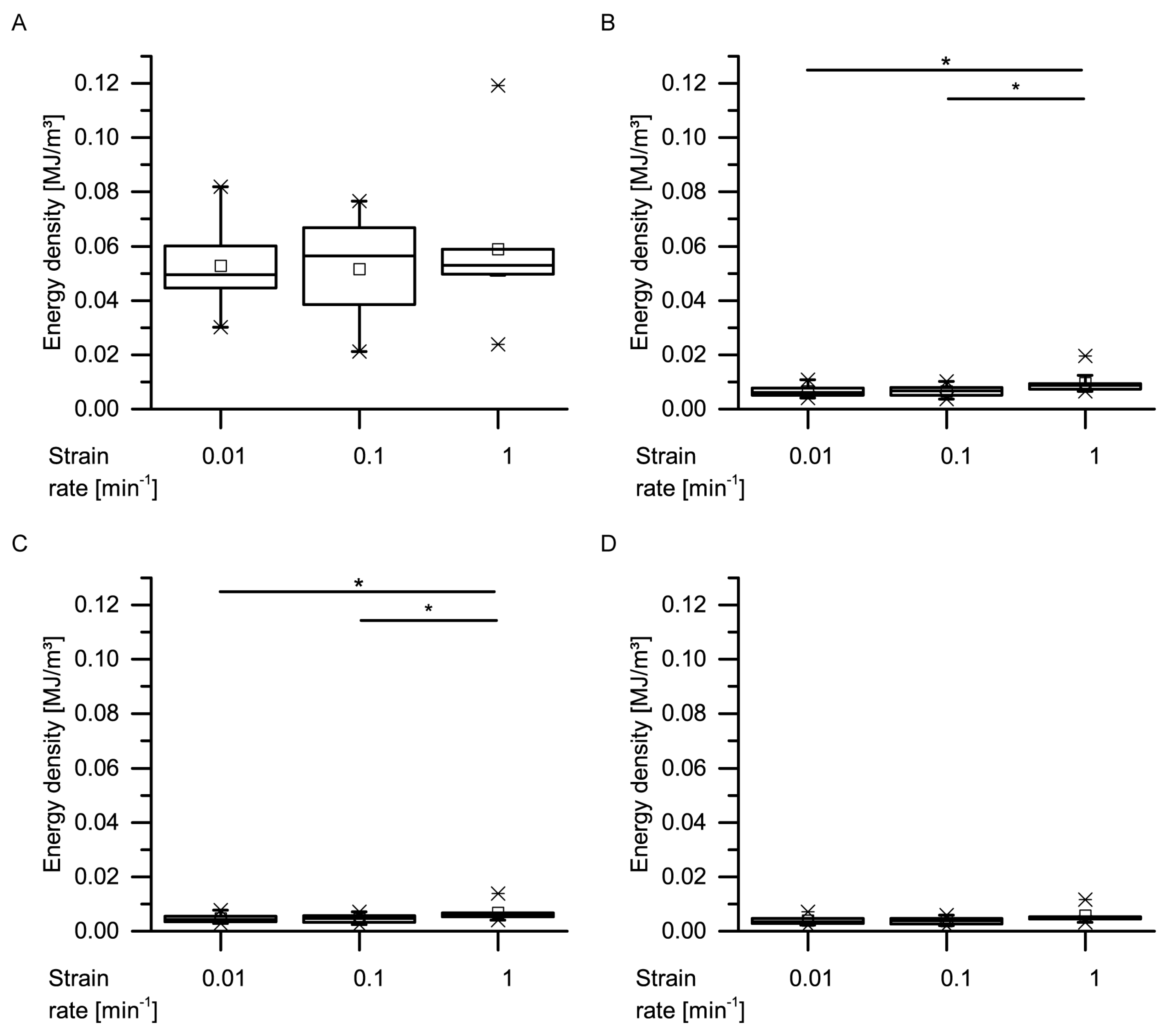
With regard to the energy densities dissipated during both dynamic drop-weight tests and quasistatic cyclic compression test, it is worthwhile to consider whether the dissipated energy density of the giant sequoia bark depends on the strain rate or not. When considering only the (most relevant) first cycle of the quasistatic cyclic compression tests with unchanged maximum compression, no strain rate dependency can be observed (
Figure 13). However, as these strain rates only range from 0.01 to 1 min
−1, they do not cover a very wide range. When thus comparing the dissipated energy density between the slowest quasistatic compression tests (a very slow strain rate of 0.01 min
−1) and the dynamic drop-weight tests (a comparably high strain rate of 7.5 × 10
3 min
−1), an increase of about 3.2 times is found. The dissipated energy density thus markedly depends on the strain rate, if a possible influence of the different methods is not taken into consideration. In general, this is not surprising, as many materials and structures exhibit a strain rate dependency of their mechanical properties. For example, the authors of [
35] examined the strain rate dependency of several mechanical properties of several general-purpose plastics, namely acrylonitrile butadiene styrene (ABS), high-density polyethylene (HDPE), polypropylene (PP) and polyvinylchloride (PVC). The loading scenarios of their compression experiments (the strain rate of the slowest quasistatic tests amounted 7.2 × 10
−2 min
−1, while the strain rate of the dynamic split Hopkins tests amounted between about 3.6 × 10
4 min
−1 and 4.7 × 10
4 min
−1) were approximately comparable to those of the present study. When comparing the dissipated energy densities between the quasistatic and the dynamic tests, they found an increase of about 1.19 times for PVC, 1.34 times for HDPE, 1.49 times for ABS and 2.47 times for PP. However, the authors of [
36] stated a low strain-rate dependency of biocomposites compared to technical composites and referred this difference, among others, to the natural fibers present in the biocomposites, a finding that would also indicate a low strain-rate dependency for the fibrous giant sequoia bark. However, the strain rate dependency of the giant sequoia bark is, with an increase of a factor of up to 3.2, considerably higher than for the tested plastics. This high strain-rate dependency for the dissipated energy densities of the giant sequoia bark becomes relevant during rockfall events, representing high-velocity impacts. The bark of the giant sequoia can be considered as adapted to the naturally occurring conditions during high-energy, high-velocity impact events.
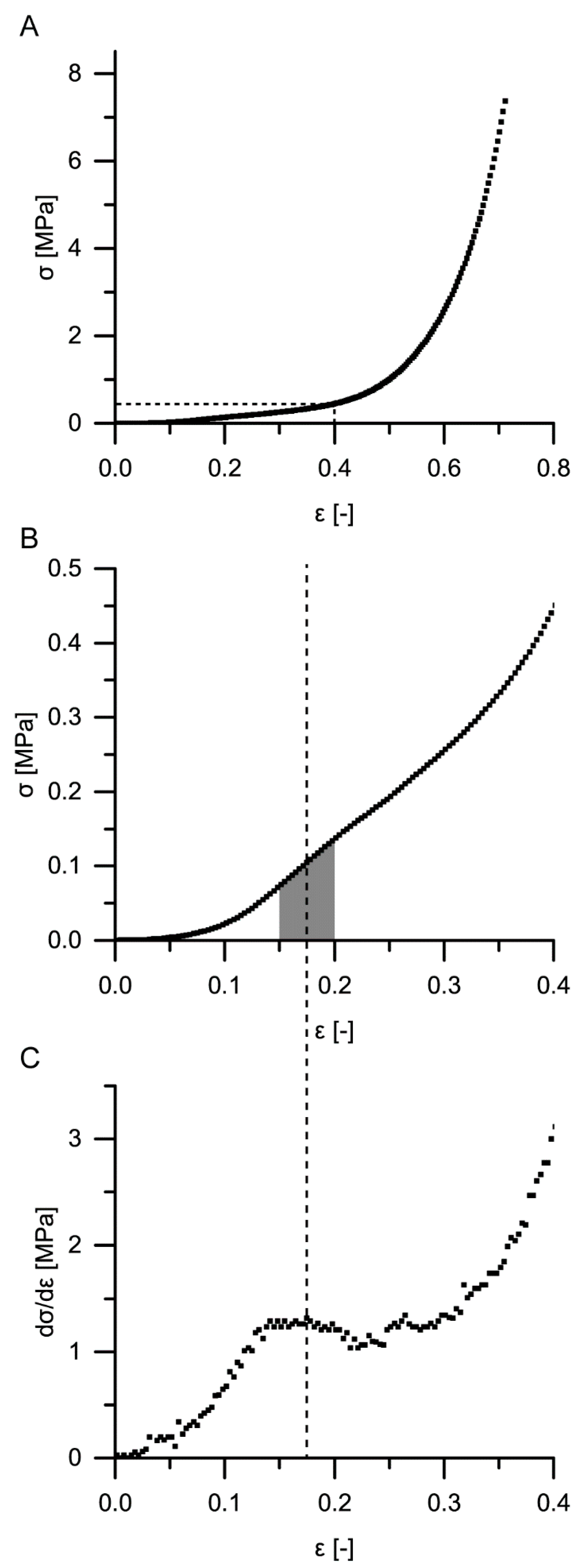
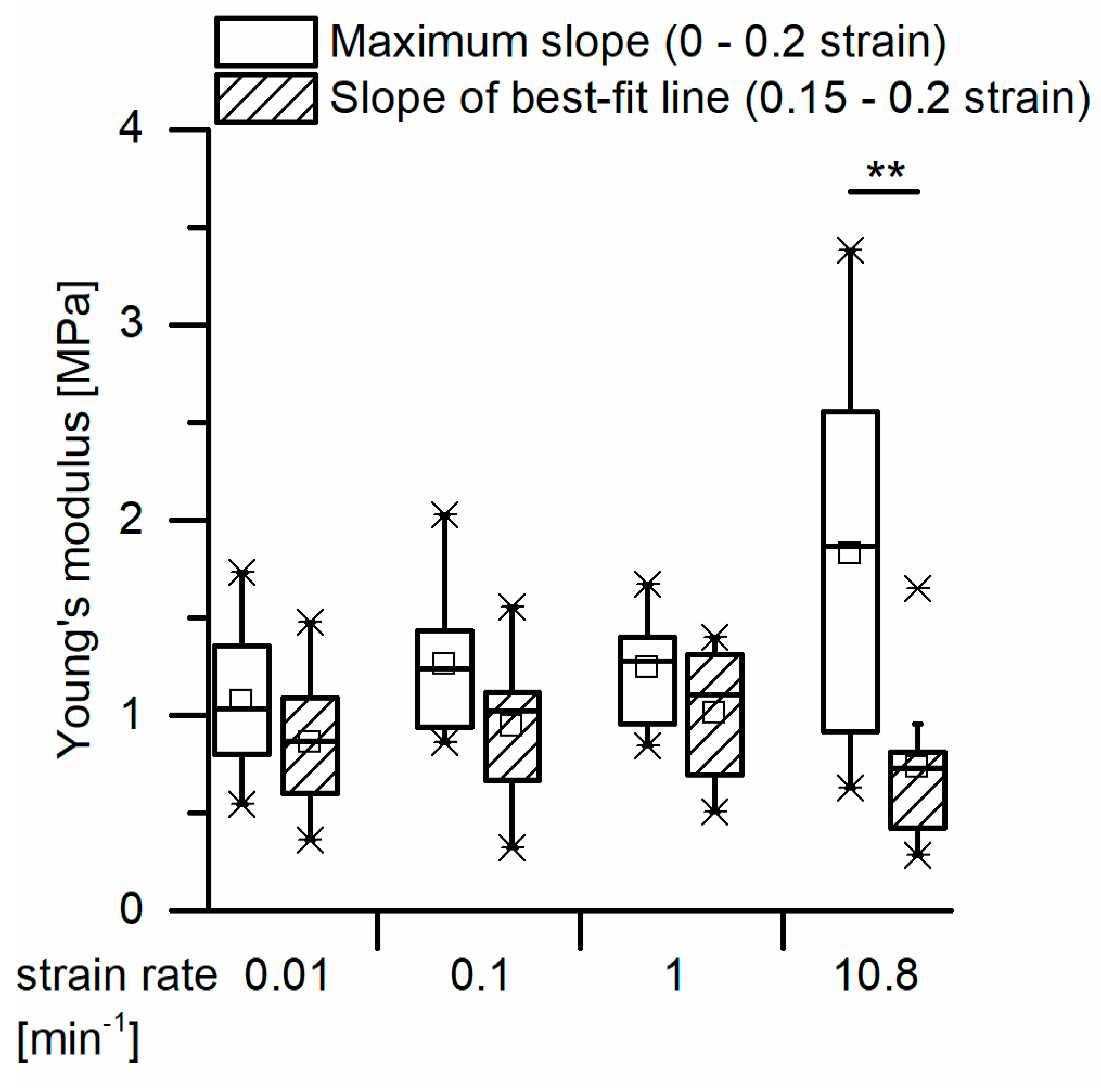
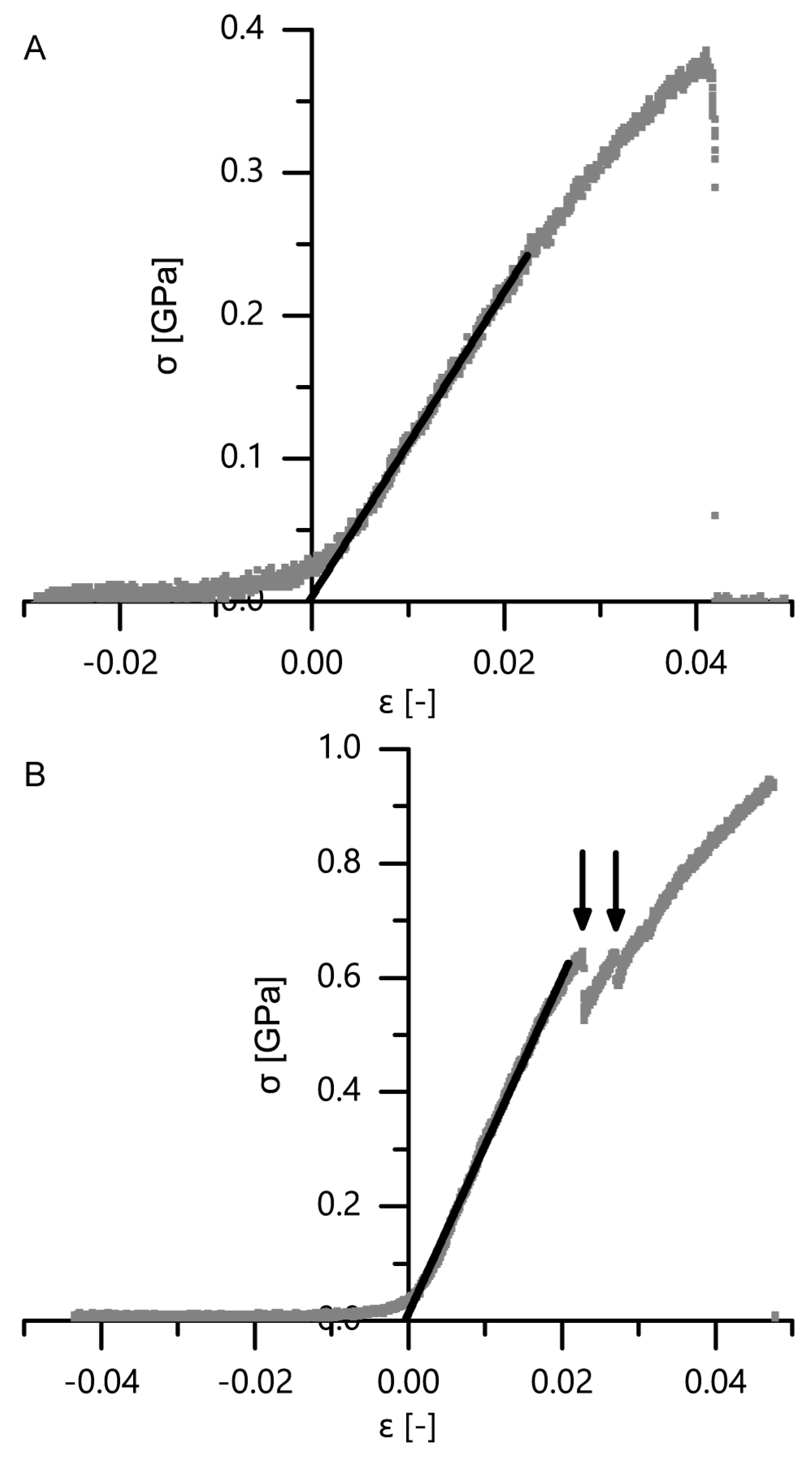
For a calculation of the structural Young’s modulus under compression for the bark samples, it is essential to identify the boundaries of the linear elastic region. However, in the case of the quasistatic compression tests, the identification of the linear elastic region is not straightforward. For cellular solids (e.g., an elastomeric foam), the linear elastic region is often referred to as occurring until ca. 0.05 strain (e.g., [
18,
19]), or the boundaries of this region are calculated involving physical quantities such as the density of the cellular material and/or the density of the cell wall material [
18]. However, in the case of the giant sequoia bark, calculating the density of the cellular material is difficult, as several cell types and layers are involved, and a mean density of the cell wall material cannot simply be calculated or assumed. Furthermore, the aforementioned assumed boundary of 0.05 strain seems to not apply with regard to the stress–strain curves found for the giant sequoia bark (
Figure 7). Thus, different approaches were used for calculating the Young’s modulus. The Young’s moduli of the two selected methods (i.e., calculation of the maximum slope between 0 and 0.2 strain as suggested by [
30] and the slope of the best-fit line between 0.15 and 0.2 strain) do not differ significantly for the strain rates between 0.01 and 1 min
−1. Amounting between 0.87 and 1.28 MPa, they are low when comparing them to values found for, e.g., technical foams or other biological “foam-like” materials such as cork. When considering the density of the giant sequoia bark (about 0.23 g/cm
2, as measured by [
13]), the abovementioned materials of comparable density are typically characterized by a Young’s modulus of 10 MPa or more [
37], being at least ca. 8 times higher than those measured for the giant sequoia bark. The high energy dissipation capability of the giant sequoia bark is remarkable, despite its comparatively low Young’s modulus. However, this goes along with the high energy dissipation capability found in the microtensile tests on single bark fibers and fiber pairs.
A comparison of mechanical properties of single fibers and fiber pairs as determined in microtensile tests proves that combining single fibers to fiber pairs does not worsen their extensibility (no significant differences in maximum strain) but significantly improves the other mechanical properties, such as tensile strength, Young’s modulus and dissipated energy (
Figure 14 and
Figure 15). When transferring this mechanical behavior to the whole bark structure, it most probably results in an advantageous response during and after an impact. During an impact, the bark is capable of protecting the cambium due to its beneficial mechanical properties. After an impact, the relaxation of the compressed bark towards its original, nondeformed, configuration is facilitated due to the high extensibility of the connected and unconnected fibers (especially in the thick outer bark), which is beneficial for a quick recovery of the protective functions of the bark after an impact.
The development of mechanical properties of fibers during the ontogenesis of the bark favors the bark’s protective potential during impact events. Although no significant differences can be found between outer and inner bark concerning tensile strength, maximum strain, Young’s modulus or dissipated energy of single fibers and fiber pairs, a general trend goes towards higher tensile strengths and Young’s moduli for fibers and fiber pairs in the inner bark, whereas the dissipated energy is highest for fiber pairs of the outer bark. Only the inner bark contains living cells, whereas all cells in the outer bark are dead and thus not metabolically active [
1]. As a consequence, the entire dead outer bark cannot be physiologically regenerated after injury or other forms of degradation, which goes along with the general trend of slightly worsening most mechanical properties in the outer bark, with exception of the dissipated energy being highest for fiber pairs in the outer bark. This means that the protective role of the outer bark can at least be preserved, if not tendentially improved, compared to the inner bark. The finding that the dimensions of the fiber cross-section do not differ between dead outer and living inner bark indicates that all examined fibers were already fully mature. Furthermore, it can be concluded that the fibers in the outer bark are not visibly altered despite being exposed to diverse environmental and growth-related influences during the naturally occurring lateral expansion of the bark. However, the authors did not examine the composition of the cell wall, where ageing processes might bring about an effect. Thus, besides the above-mentioned mechanical wear or abrasion, potential differences in the cell wall composition between outer and inner bark are most likely the cause for the differing mechanical properties of fibers between the outer and inner bark (
Figure 15).
The mechanical properties of single bark fibers and fiber pairs are in the range of other plant and technical fibers and fiber bundles tested in tension (
Table 1). The tensile strength of single giant sequoia fibers from the inner and outer bark are in the middle region compared to other single plant or technical fibers, whereas the tensile strength of fiber pairs is in the upper region compared to bundles of natural fibers. The maximum strain of single fibers and fiber pairs falls in the upper region compared to other natural plant fibers, except for single cotton fibers that can reach much higher maximum strains. The Young’s modulus of single fibers of the inner bark falls in the middle region, while that of single fibers of the outer bark fibers falls in the lower region. The Young’s modulus of fiber pairs of the inner bark falls in the upper region, while that of fiber pairs of the outer bark falls in the middle region. The energy dissipation capacity of the fibers of the outer bark is at least equal to the inner bark, despite Young’s modulus and tensile strength being smaller (
Figure 15); literature values for comparison of energy dissipation with other plant fibers were not found. Furthermore, only single fibers and fiber pairs were considered in this comparison to other plant and technical fibers; thus, the energy dissipation will most likely stand out even more when considering the whole bark as a highly sophisticated layered structure.
As already mentioned in the introduction, an implementation of several hierarchical levels of a biological role model has already been proven beneficial for the optimization of energy dissipation of technical products other than concrete- and ceramic-based components [
24,
25]. The findings of the present paper are relevant for the application in bio-inspired fibrous concrete and ceramic components for building construction [
16,
27]). However, a potential application is not limited to concrete or ceramic components but includes all classes of fiber-reinforced materials. Hereby, the structuring of the giant sequoia bark on several hierarchical levels including the high cell wall content of the bark fibers, the almost rectangular fiber cross-section, the mode of connection between adjacent bark fibers and the arrangement of fibers in two orders of partially interwoven layers serve as potential inspirations. A transfer into concrete- and ceramic-based components will potentially lead to an increased impact resistance and energy dissipation behavior of the bio-inspired structures and might help to increase earthquake security by an improved energy dissipation and a benign failure behavior.
4. Materials and Methods
4.1. Plant Material
Two giant sequoia (Sequoiadendron giganteum Lindl.) trees and one tree of heaven (Ailanthus altissima (Mill.) Swingle) were felled, and bark samples were used for the various analysis and testing methods. After felling of the trees, stem segments including both intact bark (defined here as comprising all tissues outside the vascular cambium) and xylem were stored at room temperature and a relative humidity of typically about 60% until further processing for the experiments. One S. giganteum tree (samples thereof were used for the dynamic drop-weight tests and the stained thin sections, see below) was about 90 years old when felled at the Botanic Garden Freiburg, the second S. giganteum tree (samples thereof were used for µCT, SEM, quasistatic compression tests and microtensile tests) was about 32 years old when felled in the city area of Freiburg i. Br., Germany. The A. altissima tree was about 70 years old when felled at the Botanic Garden Freiburg. All samples used in the studies presented here were taken approximately from breast height, except the samples used for the dynamic drop-weight tests (see below) that were taken from heights of 17.9 m (A. altissima) and 19 m (S. giganteum) above ground. The sampling height for the dynamic drop-weight tests deviates from that of all other studies because approximately matching sampling heights are required for a reasonable comparison between the two tree species, which was only the case for the sampling heights of 17.9 and 19 m.
4.2. µCT Scans, SEM and Thin Sections
Samples of the whole bark of S. giganteum were used for the µCT scans. µCT scans were performed using a Skyscan 1272, Bruker microCT, Kontich, Belgium. The µCT data were processed and volume-rendered (in the following, the term ‘segmented’ will be used) using the CT-Analyzer (V. 1.16.4.1, Bruker microCT, Kontich, Belgium) and CTVox (V. 3.2.0 r1294, Bruker microCT, Kontich, Belgium) software.
SEM images of dehydrated and gold-sputtered single bark fibers and fiber pairs of
S. giganteum were obtained from a Leo 435VP (LEO Electron Microscopy Ltd., Cambridge, England) at 15 kV. The single bark fibers and fiber pairs were extracted in the same way as described later on in
Section 4.5 ‘Microtensile Tests’.
Bark samples of S. giganteum used to prepare thin sections were dehydrated, embedded in Technovit 7100 and stained with toluidine blue after cutting with a microtome.
4.3. Dynamic Drop-Weight Tests
Dynamic drop-weight tests were performed on ten dried bark samples (DIN Norm EN 13183-1) of
S. giganteum and eight dried bark samples of
A. altissima. Prior to testing, bark samples were cut to a quadratic basal area of 3 × 3 cm. The sample height was left unaltered and was determined by the bark thickness. The drop-weight test rig was constituted of an instrumented anvil equipped with a 20 kN force sensor (model 8402–6020, Burster Präzisionsmesstechnik GmbH & Co KG, Gernsbach, Germany). After placing a bark sample on top of the instrumented anvil, a drop weight with a mass of 200 g was dropped on to the bark sample from a height of 1.99 m through a guided tube. Regardless of the sample size, the same drop weight and drop height were always used. Drop height and drop weight were determined in preliminary tests, so that a suitable impact and compaction were achieved. The drop-weight tests were recorded with a high-speed camera (model MotionPro Y4, Integrated Design Tools, Inc., Tallahassee, FL, USA). For further details on the test setup, we refer to [
16,
22].
With the help of the high-speed videos, the velocity of the impactor at any time of the experiment could be determined (by tracking marks that were painted onto the impactor, with the software Motion Studio, IDT Motion Studio, version 2.12.09.04). Converting the velocity of the impactor into its kinetic energy immediately before and after impact allowed the calculation of the energy dissipated during each test. However, it cannot be excluded that a (very) small percentage of this dissipated energy is lost elsewhere in the system (i.e., not by the bark samples), causing a systematic error. Nevertheless, the data derived from the dynamic drop-weight tests allow for a good first-order approximation of the energy dissipated by the tree bark.
Furthermore, the dissipated energy of each sample was converted to an energy density by dividing it by the volume of that sample. This way, the dissipated energy density can be compared with the dissipated energy densities obtained during the quasistatic cyclic compression tests with unchanged maximum strain (see
Section 4.4 and
Section 2.3.2 Subheading: Cyclic Test with Unchanged Maximum Strain).
4.4. Quasistatic Compression Tests
Uniaxial quasistatic compression tests comprising bark samples of S. giganteum only were performed using a universal testing machine (Instron 4466-10kN, with a retrofit kit to inspect-DC standard, Hegewald & Peschke Mess- und Prüftechnik GmbH, Nossen, Germany), equipped with a 1 or 10 kN load cell, depending on the maximum force to be expected. LabMaster 2.1.11.4 software (Hegewald & Peschke Mess- und Prüftechnik GmbH, Nossen, Germany) was used to control the universal testing machine. All data were recorded at a sampling rate of 50 Hz.
The sample dimensions were similar to those of the dynamic drop-weight tests, with a basal area of 3 × 3 cm, and the sample height was determined by bark thickness. Depending on which test was performed, several of four predetermined strain rates were applied: 0.01, 0.1, 1 and 10.8 min−1. Fixed strain rates were chosen in order to cope with the different sample heights—however, as a consequence, test velocities differ from sample to sample.
Quasistatic compression tests comprised tests including exclusively a loading stage (in the following termed as ‘quasistatic compression tests (one loading stage)’) as well as tests including several cycles, each involving a loading and an unloading stage (in the following termed as ‘cyclic compression tests’).
Quasistatic compression tests (one loading stage) were performed on ten bark samples for each of the four strain rates. These tests were conducted far beyond the yield point, when the densification regime was already reached (until 70% or 80% strain, depending on the strain rate). These tests were carried out to give an overall impression on the bark samples’ behavior during quasistatic compression. Young’s moduli of the samples were calculated using two different approaches: one in which the Young’s modulus was calculated by the maximum slope of each stress–strain curve until 20% strain and one in which the Young’s modulus was calculated with the slope of the best-fit line between 15% and 20% strain (see also
Section 2.3,
Figure 7 and
Figure 8). It needs to be denoted that the term ‘Young’s modulus’ is used in this paper to describe the mechanical property of the bark, which represents a structure and not a homogeneous material. It can thus rather be characterized as a ‘structural’ or ‘apparent’ Young’s modulus. All stresses given for both the quasistatic compression tests and the microtensile tests are nominal (engineering) stresses, and all strains are nominal (engineering) strains.
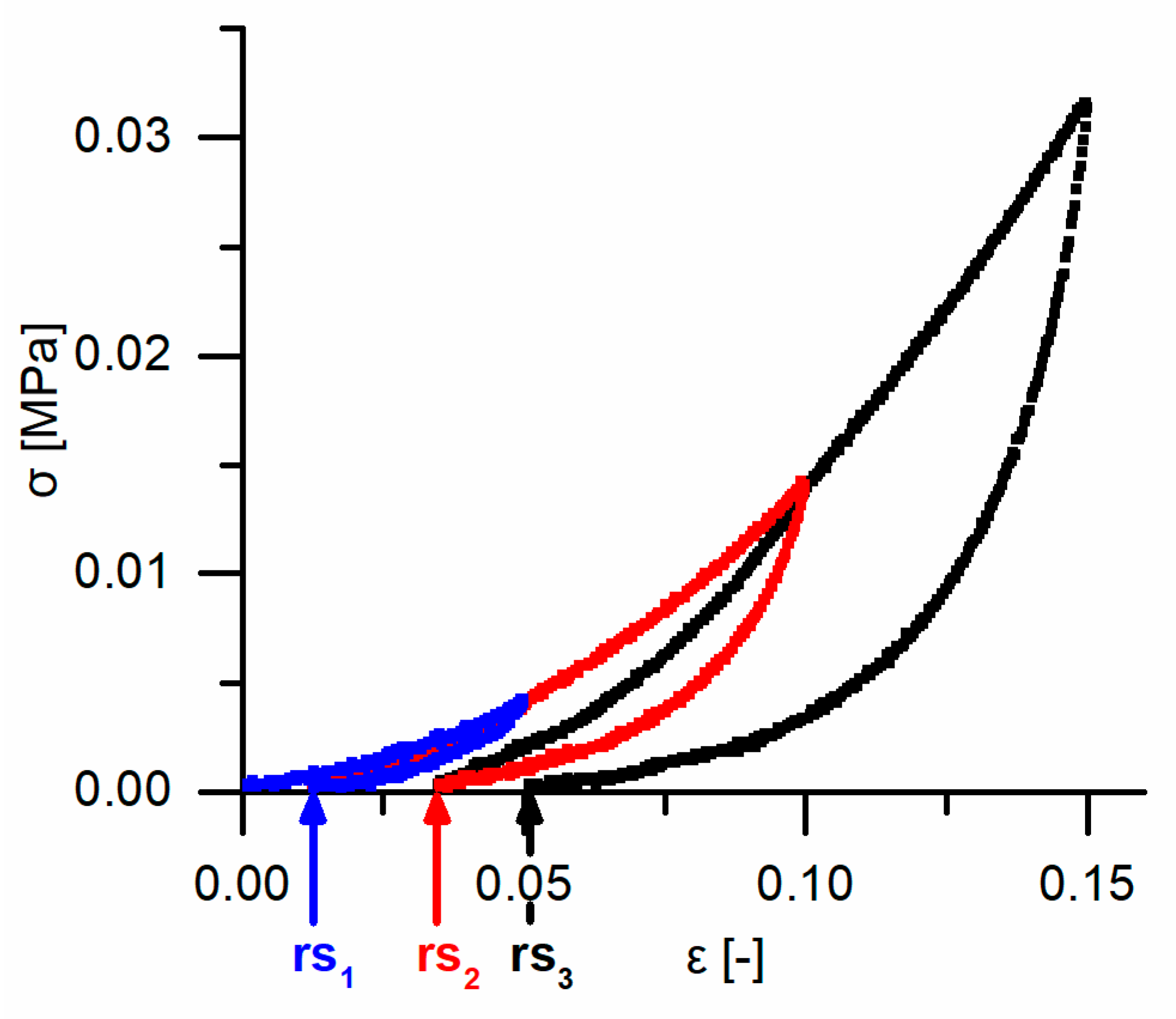
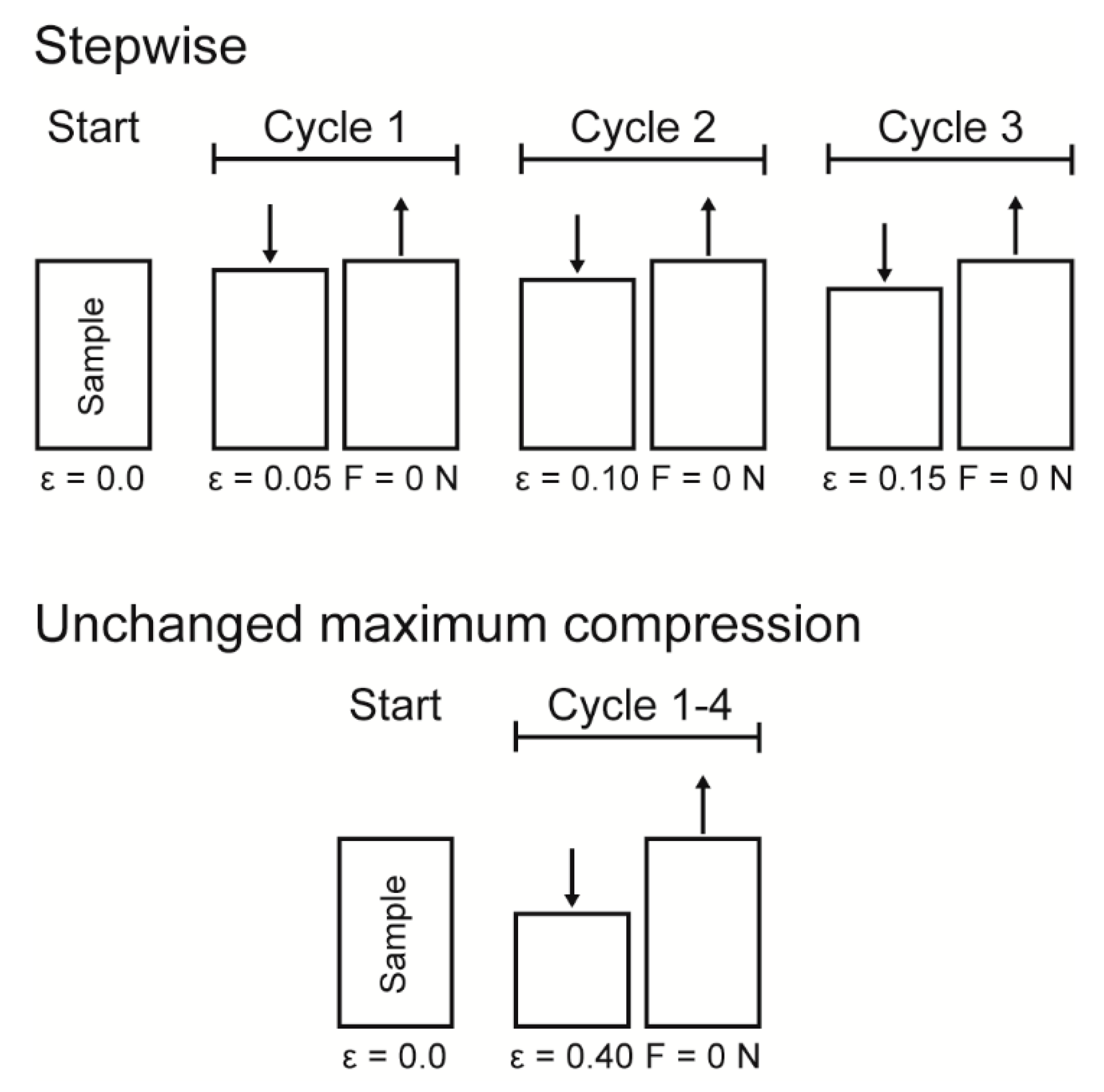
Cyclic compression tests were performed to gain insights into the potential presence of an elastic, plastic or even viscoelastic deformation behavior and for understanding the energy dissipation. Thereby, two different experimental procedures were carried out for the cyclic tests (see
Figure 16 for a sketch of both experimental procedures): (1) a cyclic test with stepwise increasing maximum strain at the end of each cycle and (2) a cyclic test with an unchanged maximum strain at the end of each cycle. The cyclic tests with stepwise increasing maximum strain were conducted in order to characterize the bark samples step by step during rather gentle loading, whereas the cyclic tests with an unchanged maximum strain characterize the durability of the bark under higher loading. For both experimental procedures, ten bark samples for each strain rate (0.01, 0.1 and 1 min
−1) were tested.
Data obtained from the cyclic compression tests were the residual strains after each cycle and—only for the cyclic test with unchanged maximum strain—energies absorbed/released/dissipated (absorbed energy Ea, released energy Er and dissipated energy Ed, in form of energy densities ED). The absorbed energy Ea characterizes the energy absorbed in the bark during loading, the released energy Er characterizes the energy released during unloading, and the dissipated energy Ed is represented by the difference between these two. Normalizing these energies by the volume of each sample yields the energy density ED, facilitating the comparability to other materials and testing methods (e.g., the dynamic drop-weight tests). The energy density can directly be derived from the stress–strain curves by calculating the area under the respective part of stress–strain curves. The residual strain is calculated for each cycle and is defined as the strain at which a force of 0 N is reached at the end of each cycle.
4.5. Microtensile Tests
Microtensile tests were performed on single fibers and fiber pairs extracted from indoor-dried inner and outer bark of S. giganteum. These experiments give quantitative insights into the mechanical properties of the basic ‘structural building blocks’ that the bark of S. giganteum is composed of. Thus, they provide the basis for the discussion and interpretation of the overall mechanical properties of the bark related to impact damping.
Single fibers and fiber pairs were extracted from the outer and inner bark using tweezers, as suggested by [
64] for wood fiber isolation. The cross-sectional dimensions of the fibers and fiber pairs were measured under a microscope (Olympus BX61) at several positions along the fiber(s). Each end of the fiber or fiber pair was glued onto a metal plate with instant glue (Uhu Sekundenkleber blitzschnell mini), followed by mounting the metal plates to the microtensile tester. During this procedure, an adhesive tape fixed the two metal plates in a given position, avoiding loading of the fibers prior to testing. Immediately before the testing procedure, the supporting adhesive tape was cut through. The tensile tests were performed using a microtensile tester (Guangzhou Lok Shun CNC Equipment Ltd., Guanghzou, China) equipped with a 10 N force transducer and controlled with the software LabVIEW (A3M Ingenieursgesellschaft mbH, Braunschweig, Germany). The test velocity was set to 5 µm/s, resulting in strain rates between 0.03 and 0.26 min
−1.
Due to possible pre-stresses that might occur despite the abovementioned gentle sample mounting procedure involving adhesive tape, the point of zero strain needed to be re-evaluated. Therefore, the best-fit line of the initial linear range (as used also for the calculation of the Young’s modulus) was determined. The intersection of this best-fit line with the abscissa corresponds to the strain at which the fiber is scarcely tensed and was thus defined as zero strain. Two examples of this re-evaluation of the zero strain can be seen in the exemplary stress–strain curves for single fibers and fiber pairs (
Figure 14). The tensile strength, maximum strain (i.e., strain of the last data point before sample failure), Young’s modulus and dissipated energy (area under the stress–strain curve from zero strain until maximum strain) were calculated from the stress–strain curves. The sample numbers for the analysis of the tensile strength and the Young’s modulus were nine samples for single fibers of the outer bark, ten for single fibers of the inner bark, eight for fiber pairs of the inner bark and eight for fiber pairs of the outer bark. For the calculation of the maximum strain and the dissipated energy, eight samples for single fibers of the outer bark, seven for single fibers of the inner bark, five for fiber pairs of the inner bark and six for fiber pairs of the outer bark were analyzed.
4.6. Statistics
Statistical tests were performed using the R 3.4.0 software (R Core Team 2017 [
65]). These tests comprised Mann–Whitney U-tests or Kruskal–Wallis tests (kruskal.test function) followed by post hoc tests by pairwise comparison using the Wilcoxon test (pairwise.wilcox.test function) with Holm adjustment. The statistical tests that were performed are indicated in the caption of each figure. Significant differences—if present—are marked with asterisks in the graphs, with the significance levels indicated as follows: *
p < 0.05, **
p < 0.01, ***
p < 0.001.
5. Conclusions
The bark of the giant sequoia, especially the thick outer bark, is characterized by a high fiber content. These fibers almost entirely consist of cell wall material, have a nearly rectangular cross-section and are laterally connected with adjacent fiber-to-fiber pairs, thus forming a first order of layers. On a second, higher, hierarchical order, layers are not exclusively parallel to the bark surface, but intersect each other, thus forming a three-dimensional network. The compaction behavior of the bark resembles in many ways that of elastomeric open-pore foams. Viscoelastic (most probably material and/or structure immanent, not based on liquid dislocation/squeezing) and elastic deformation components were shown. Furthermore, at least a small plastic component is present, as the bark does not entirely restore its initial shape after compression.
When compared to other tree species, such as Ailanthus altissima, the bark of the giant sequoia is capable of dissipating high amounts of impact energy and may thus protect the underlying vital tissues during rockfall events occurring in the natural habitat of the species. Thereby the energy dissipation capability of the bark is even underestimated in the present studies, since the bark structure (interwoven layers of long fibers) is harmed due to cutting of the test samples. The energy dissipation capability becomes even more impressive when keeping in mind that—apart from impact protection—the bark has to fulfill a multitude of different functions and needs to be optimized in terms of multiple criteria towards all of them. The amount of dissipated energy is considerably higher during the dynamic tests compared to quasistatic tests. Thus, due to the marked high strain-rate dependency, the bark of the giant sequoia can be considered as adapted to the naturally occurring conditions during high-energy, high-velocity impact events.
The composition of the bark, especially the subdivision into outer and inner bark, as well as the high fiber content and the arrangement of laterally connected fibers in layers within the outer bark are advantageous during compression. No weakening effect due to degradation, wear or abrasion of fibers was observed for the outer bark compared to the inner bark. Instead, the protective role of the thick and loose outer bark is at least equal to, if not larger than, that of the more compact inner bark. Thus, the outer bark dissipates high amounts of impact energy and reduces force peaks, whereas the inner bark serves as a padding layer. The fibrous and layered bark composition with a huge amount of air-filled spaces between the fibers is also advantageous after an impact event. It facilitates a quick recovery of the bark towards its original structure after compression and thus promotes a quick recovery of the protective function of the bark. In summary, at least on all hierarchical levels examined in this paper (i.e., from the cellular level to the integral level), the bark composition is optimized for energy dissipation under high-velocity impacts. It consists of strong but flexibly interconnected fibers that are arranged in partially interwoven layers on two orders.
All these properties of the giant sequoia bark may serve as a basis for a transfer into bio-inspired fibrous concrete and ceramic components with an increased impact resistance and energy dissipation behavior.