Whole Lotta Lipids—From HCV RNA Replication to the Mature Viral Particle
Abstract
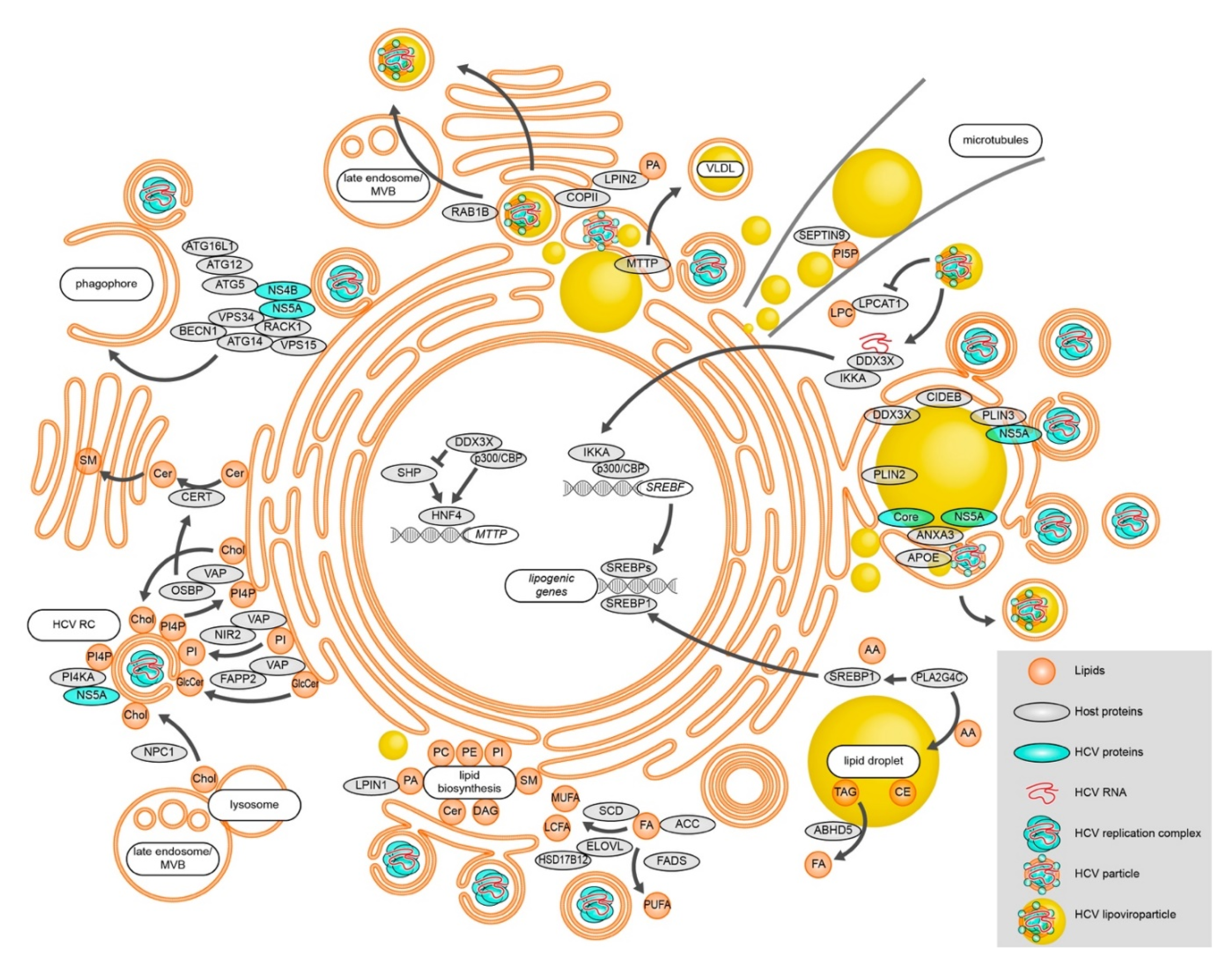
1. Fatty Acids in HCV Replication
2. HCV-Induced Membrane Remodeling
3. Another Brick in the Wall—Cholesterol in HCV Replication
4. Lipid Transfer Proteins
5. Autophagosomes, Membranes, and HCV RNA Replication
6. Come Together—LDs Provide a Stage for HCV Assembly
7. Dress for Success—HCV Lipoviroparticle Formation and Egress
8. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AA | Arachidonic acid |
| ABHD5 | α/β hydrolase domain containing protein 5 |
| ACC | Acetyl-CoA carboxylase |
| ADFP | Adipophilin |
| ADRP | Adipose differentiation-related protein |
| AMPK | AMP-activated protein kinase |
| ANXA3 | Annexin A3 |
| APO | Apoplipoprotein |
| ATG | Autophagy-related protein |
| ATG12 | Ubiquitin-like protein ATG12 |
| ATG14 | Beclin 1-associated autophagy-related key regulator |
| ATG16L1 | Autophagy-related protein 16-1 |
| ATG5 | Autophagy protein 5 |
| ATG7 | Ubiquitin-like modifier-activating enzyme ATG7 |
| ATGL | Adipose triglyceride lipase |
| BECN1 | Beclin-1 |
| BMV | Brome mosaic virus |
| CBP | CREB-binding protein-histone acetyltransferase |
| CE | Cholesterol ester |
| Cer | Ceramide |
| CERT | Ceramide transfer protein |
| Chol | Cholesterol |
| CIDEB | Cell-death-inducing DFFA-like effector B |
| DAG | Diacylgylcerol |
| DDX3X | ATP-dependent RNA helicase |
| DENV | Dengue Virus |
| DGAT1 | Diacylglycerol acyltransferase 1 |
| DMV | Double membrane vesicle |
| ELOVL | Elongation of very long chain fatty acids protein |
| ER | Endoplasmic reticulum |
| FA | Fatty acid |
| FADS2 | Acyl-CoA 6-desaturase |
| FAPP2 | Phosphatidylinositol four-phosphate adaptor protein 2 |
| FIG4 | PI(3,5)P2 5-phosphatase |
| GlcCer | Glucosylceramide |
| HCV | Hepatitis C virus |
| HNF4 | Hepatocyte nuclear factor 4 |
| HSD17B12 | 17-betahydroxysteroid dehydrogenase type 12 |
| IKKA | Inhibitor of nuclear factor kappa-B kinase subunit alpha |
| KLHL12 | Kelch-like protein 12 |
| LC3 | Microtubule-associated proteins 1A/1B light chain 3B |
| LD | Lipid droplet |
| LPC | Lysophosphatidylcholine |
| LPCAT1 | Lyso-PC acyltransferase 1 |
| LPIN | Lipin |
| LTP | Lipid transfer protein |
| LVP | Lipoviroparticle |
| LysoPL | Lysophospholipid |
| MCS | Membrane contact sites |
| MTTP | Microsomal triglyceride transfer protein |
| MUFA | Monounsaturated fatty acid |
| MVB | Multivesicular bodies |
| NIR2 | Pyk2 N-terminal domain-interacting receptor 2 |
| NPC1 | Niemann-Pick type C1 |
| NS | Non-structural |
| OSBP | Oxysterol-binding protein |
| PA | Phosphatidic acid |
| PC | Phosphatidylcholine |
| PE | Phosphatidylethanolamine |
| PI | Phosphatidylinositol |
| PI4K | Phosphatidylinositol 4-kinase |
| PI4P | Phosphatidylinositol 4-phosphate |
| PI5P | Phosphatidylinositol 5-phosphate |
| PITPNM1 | Membrane-associated phosphatidylinositol transfer protein 1 |
| PLA2G4C | Cytosolic phospholipase A2 gamma |
| PLIN | Perilipin |
| PPARα | Peroxisome proliferator-activated receptor α |
| PUFA | Polyunsaturated fatty acid |
| RAB1B | Ras-related protein Rab-1B |
| RACK1 | Receptor for activated protein C kinase 1 |
| SCD | Stearoyl-CoA desaturase |
| SHP | Small heterodimer partner |
| SM | Sphingomyelin |
| SREPB1 | Sterol regulatory element-binding protein |
| STX7 | Syntaxin 7 |
| TAG | Triglycerides |
| TGN | Trans-Golgi-network |
| TIP47 | Tail interacting protein of 47 kDa |
| UFA | Unsaturated fatty acid |
| VAP | Vesicle-associated membrane protein-associated protein |
| (V)LCFA | (Very)-long chain fatty acid |
| VLDL | Very-low-density-lipoprotein |
| VPS15 | Phosphoinositide 3-kinase regulatory subunit 4 |
| VPS34 | Phosphatidylinositol 3-kinase catalytic subunit type 3 |
| ZIKV | Zika Virus |
References
- Zhang, Z.; He, G.; Filipowicz, N.A.; Randall, G.; Belov, G.A.; Kopek, B.G.; Wang, X. Host Lipids in Positive-Strand RNA Virus Genome Replication. Front. Microbiol. 2019, 10, 286. [Google Scholar] [CrossRef]
- Lupberger, J.; Croonenborghs, T.; Roca Suarez, A.A.; Van Renne, N.; Juhling, F.; Oudot, M.A.; Virzi, A.; Bandiera, S.; Jamey, C.; Meszaros, G.; et al. Combined Analysis of Metabolomes, Proteomes, and Transcriptomes of Hepatitis C Virus-Infected Cells and Liver to Identify Pathways Associated With Disease Development. Gastroenterology 2019, 157, 537–551.e9. [Google Scholar] [CrossRef]
- Hofmann, S.; Krajewski, M.; Scherer, C.; Scholz, V.; Mordhorst, V.; Truschow, P.; Schobel, A.; Reimer, R.; Schwudke, D.; Herker, E. Complex lipid metabolic remodeling is required for efficient hepatitis C virus replication. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2018, 1863, 1041–1056. [Google Scholar] [CrossRef] [PubMed]
- Douglas, D.N.; Pu, C.H.; Lewis, J.T.; Bhat, R.; Anwar-Mohamed, A.; Logan, M.; Lund, G.; Addison, W.R.; Lehner, R.; Kneteman, N.M. Oxidative Stress Attenuates Lipid Synthesis and Increases Mitochondrial Fatty Acid Oxidation in Hepatoma Cells Infected with Hepatitis C Virus. J. Biol. Chem. 2016, 291, 1974–1990. [Google Scholar] [CrossRef] [PubMed]
- Popescu, C.I.; Riva, L.; Vlaicu, O.; Farhat, R.; Rouille, Y.; Dubuisson, J. Hepatitis C virus life cycle and lipid metabolism. Biology (Basel) 2014, 3, 892–921. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Nieh, A.H.; Kenwood, B.M.; Davis, C.A.; Tosello-Trampont, A.C.; Elich, T.D.; Breazeale, S.D.; Ward, E.; Anderson, R.J.; Caldwell, S.H.; et al. Distinct Roles for Intracellular and Extracellular Lipids in Hepatitis C Virus Infection. PLoS ONE 2016, 11, e0156996. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, B.; Mazeaud, C.; Baril, M.; Poirier, D.; Sow, A.A.; Chatel-Chaix, L.; Titorenko, V.; Lamarre, D. Very-long-chain fatty acid metabolic capacity of 17-beta-hydroxysteroid dehydrogenase type 12 (HSD17B12) promotes replication of hepatitis C virus and related flaviviruses. Sci. Rep. 2020, 10, 4040. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Jiang, H.; Qu, L.; Yao, W.; Cai, H.; Chen, L.; Peng, T. Hepatocyte nuclear factor 4alpha and downstream secreted phospholipase A2 GXIIB regulate production of infectious hepatitis C virus. J. Virol. 2014, 88, 612–627. [Google Scholar] [CrossRef]
- Guo, M.; Pei, R.; Yang, Q.; Cao, H.; Wang, Y.; Wu, C.; Chen, J.; Zhou, Y.; Hu, X.; Lu, M.; et al. Phosphatidylserine-specific phospholipase A1 involved in hepatitis C virus assembly through NS2 complex formation. J. Virol. 2015, 89, 2367–2377. [Google Scholar] [CrossRef]
- Xu, S.; Pei, R.; Guo, M.; Han, Q.; Lai, J.; Wang, Y.; Wu, C.; Zhou, Y.; Lu, M.; Chen, X. Cytosolic phospholipase A2 gamma is involved in hepatitis C virus replication and assembly. J. Virol. 2012, 86, 13025–13037. [Google Scholar] [CrossRef]
- Menzel, N.; Fischl, W.; Hueging, K.; Bankwitz, D.; Frentzen, A.; Haid, S.; Gentzsch, J.; Kaderali, L.; Bartenschlager, R.; Pietschmann, T. MAP-kinase regulated cytosolic phospholipase A2 activity is essential for production of infectious hepatitis C virus particles. PLoS Pathog. 2012, 8, e1002829. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Liu, S.; Zhang, X.; Lam, S.M.; Hu, X.; Zhou, Y.; Chen, J.; Wang, Y.; Wu, C.; Shui, G.; et al. Requirement of cytosolic phospholipase A2 gamma in lipid droplet formation. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2017, 1862, 692–705. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, Z.; Chukkapalli, V.; Nchoutmboube, J.A.; Li, J.; Randall, G.; Belov, G.A.; Wang, X. Positive-strand RNA viruses stimulate host phosphatidylcholine synthesis at viral replication sites. Proc. Natl. Acad. Sci. USA 2016, 113, E1064–E1073. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.R.; Donkor, J.; Zhang, P.; Csaki, L.S.; Vergnes, L.; Lee, J.M.; Dewald, J.; Brindley, D.N.; Atti, E.; Tetradis, S.; et al. Mouse lipin-1 and lipin-2 cooperate to maintain glycerolipid homeostasis in liver and aging cerebellum. Proc. Natl. Acad. Sci. USA 2012, 109, E2486–E2495. [Google Scholar] [CrossRef]
- Castro, V.; Calvo, G.; Avila-Perez, G.; Dreux, M.; Gastaminza, P. Differential Roles of Lipin1 and Lipin2 in the Hepatitis C Virus Replication Cycle. Cells 2019, 8, 1456. [Google Scholar] [CrossRef]
- Mingorance, L.; Castro, V.; Avila-Perez, G.; Calvo, G.; Rodriguez, M.J.; Carrascosa, J.L.; Perez-Del-Pulgar, S.; Forns, X.; Gastaminza, P. Host phosphatidic acid phosphatase lipin1 is rate limiting for functional hepatitis C virus replicase complex formation. PLoS Pathog. 2018, 14, e1007284. [Google Scholar] [CrossRef]
- Felmlee, D.J.; Hafirassou, M.L.; Lefevre, M.; Baumert, T.F.; Schuster, C. Hepatitis C virus, cholesterol and lipoproteins--impact for the viral life cycle and pathogenesis of liver disease. Viruses 2013, 5, 1292–1324. [Google Scholar] [CrossRef]
- Paul, D.; Bartenschlager, R. Flaviviridae Replication Organelles: Oh, What a Tangled Web We Weave. Annu Rev. Virol 2015, 2, 289–310. [Google Scholar] [CrossRef]
- Aligeti, M.; Roder, A.; Horner, S.M. Cooperation between the Hepatitis C Virus p7 and NS5B Proteins Enhances Virion Infectivity. J. Virol. 2015, 89, 11523–11533. [Google Scholar] [CrossRef]
- Wang, H.; Tai, A.W. Continuous de novo generation of spatially segregated hepatitis C virus replication organelles revealed by pulse-chase imaging. J. Hepatol. 2017, 66, 55–66. [Google Scholar] [CrossRef]
- Vieyres, G.; Pietschmann, T. HCV Pit Stop at the Lipid Droplet: Refuel Lipids and Put on a Lipoprotein Coat before Exit. Cells 2019, 8, 233. [Google Scholar] [CrossRef] [PubMed]
- Neufeldt, C.J.; Cortese, M.; Acosta, E.G. Bartenschlager, R., Rewiring cellular networks by members of the Flaviviridae family. Nat. Rev. Microbiol. 2018, 16, 125–142. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tai, A.W. Nir2 Is an Effector of VAPs Necessary for Efficient Hepatitis C Virus Replication and Phosphatidylinositol 4-Phosphate Enrichment at the Viral Replication Organelle. J. Virol. 2019, 93, e00742-19. [Google Scholar] [CrossRef] [PubMed]
- Stoeck, I.K.; Lee, J.Y.; Tabata, K.; Romero-Brey, I.; Paul, D.; Schult, P.; Lohmann, V.; Kaderali, L.; Bartenschlager, R. Hepatitis C Virus Replication Depends on Endosomal Cholesterol Homeostasis. J. Virol. 2018, 92, e01196-17. [Google Scholar] [CrossRef]
- Elgner, F.; Ren, H.; Medvedev, R.; Ploen, D.; Himmelsbach, K.; Boller, K.; Hildt, E. The Intracellular Cholesterol Transport Inhibitor U18666A Inhibits the Exosome-Dependent Release of Mature Hepatitis C Virus. J. Virol. 2016, 90, 11181–11196. [Google Scholar] [CrossRef]
- Wang, L.; Ou, J.H. Hepatitis C virus and autophagy. Biol. Chem. 2015, 396, 1215–1222. [Google Scholar] [CrossRef]
- Wang, L.; Kim, J.Y.; Liu, H.M.; Lai, M.M.C.; Ou, J.J. HCV-induced autophagosomes are generated via homotypic fusion of phagophores that mediate HCV RNA replication. PLoS Pathog. 2017, 13, e1006609. [Google Scholar] [CrossRef]
- Kim, J.Y.; Wang, L.; Lee, J.; Ou, J.J. Hepatitis C Virus Induces the Localization of Lipid Rafts to Autophagosomes for Its RNA Replication. J. Virol. 2017, 91, e00541-17. [Google Scholar] [CrossRef]
- Mizushima, N.; Komatsu, M. Autophagy: Renovation of cells and tissues. Cell 2011, 147, 728–741. [Google Scholar] [CrossRef]
- Fahmy, A.M.; Labonte, P. The autophagy elongation complex (ATG5-12/16L1) positively regulates HCV replication and is required for wild-type membranous web formation. Sci. Rep. 2017, 7, 40351. [Google Scholar] [CrossRef]
- Ploen, D.; Hildt, E. Hepatitis C virus comes for dinner: How the hepatitis C virus interferes with autophagy. World J. Gastroenterol. 2015, 21, 8492–8507. [Google Scholar] [CrossRef]
- Lee, J.S.; Tabata, K.; Twu, W.I.; Rahman, M.S.; Kim, H.S.; Yu, J.B.; Jee, M.H.; Bartenschlager, R.; Jang, S.K. RACK1 mediates rewiring of intracellular networks induced by hepatitis C virus infection. PLoS Pathog. 2019, 15, e1008021. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Q.; Qiu, G.; Zhou, S.; Jing, Z.; Wang, J.; Wang, W.; Cao, J.; Han, K.; Cheng, Q.; et al. RACK1 Promotes Autophagy by Enhancing the Atg14L-Beclin 1-Vps34-Vps15 Complex Formation upon Phosphorylation by AMPK. Cell Rep. 2015, 13, 1407–1417. [Google Scholar] [CrossRef] [PubMed]
- Roingeard, P.; Melo, R.C. Lipid droplet hijacking by intracellular pathogens. Cell. Microbiol. 2017, 19, e12688. [Google Scholar] [CrossRef] [PubMed]
- Walther, T.C.; Farese, R.V., Jr. Lipid droplets and cellular lipid metabolism. Annu. Rev. Biochem 2012, 81, 687–714. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, Y.; Itabe, H.; Sakai, J.; Makita, M.; Noda, J.; Mori, M.; Higashi, Y.; Kojima, S.; Takano, T. Identification of major proteins in the lipid droplet-enriched fraction isolated from the human hepatocyte cell line HuH7. Biochim. Biophys. Acta 2004, 1644, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Brasaemle, D.L.; Barber, T.; Wolins, N.E.; Serrero, G.; Blanchette-Mackie, E.J.; Londos, C. Adipose differentiation-related protein is an ubiquitously expressed lipid storage droplet-associated protein. J. Lipid Res. 1997, 38, 2249–2263. [Google Scholar] [PubMed]
- Lassen, S.; Gruttner, C.; Nguyen-Dinh, V.; Herker, E. Perilipin-2 is critical for efficient lipoprotein and hepatitis C virus particle production. J. Cell Sci. 2019, 132, jcs.217042. [Google Scholar] [CrossRef]
- Zhang, N.; Yin, P.; Zhou, L.; Li, H.; Zhang, L. ARF1 activation dissociates ADRP from lipid droplets to promote HCV assembly. Biochem. Biophys. Res. Commun. 2016, 475, 31–36. [Google Scholar] [CrossRef]
- Branche, E.; Conzelmann, S.; Parisot, C.; Bedert, L.; Levy, P.L.; Bartosch, B.; Clement, S.; Negro, F. Hepatitis C Virus Increases Occludin Expression via the Upregulation of Adipose Differentiation-Related Protein. PLoS ONE 2016, 11, e0146000. [Google Scholar] [CrossRef]
- Lee, J.Y.; Cortese, M.; Haselmann, U.; Tabata, K.; Romero-Brey, I.; Funaya, C.; Schieber, N.L.; Qiang, Y.; Bartenschlager, M.; Kallis, S.; et al. Spatiotemporal Coupling of the Hepatitis C Virus Replication Cycle by Creating a Lipid Droplet- Proximal Membranous Replication Compartment. Cell Rep. 2019, 27, 3602–3617.e5. [Google Scholar] [CrossRef] [PubMed]
- Akil, A.; Peng, J.; Omrane, M.; Gondeau, C.; Desterke, C.; Marin, M.; Tronchere, H.; Taveneau, C.; Sar, S.; Briolotti, P.; et al. Septin 9 induces lipid droplets growth by a phosphatidylinositol-5-phosphate and microtubule-dependent mechanism hijacked by HCV. Nat. Commun 2016, 7, 12203. [Google Scholar] [CrossRef] [PubMed]
- Heaton, N.S.; Randall, G. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microbe. 2010, 8, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Vieyres, G.; Welsch, K.; Gerold, G.; Gentzsch, J.; Kahl, S.; Vondran, F.W.; Kaderali, L.; Pietschmann, T. ABHD5/CGI-58, the Chanarin-Dorfman Syndrome Protein, Mobilises Lipid Stores for Hepatitis C Virus Production. PLoS Pathog. 2016, 12, e1005568. [Google Scholar] [CrossRef]
- Schweiger, M.; Lass, A.; Zimmermann, R.; Eichmann, T.O.; Zechner, R. Neutral lipid storage disease: Genetic disorders caused by mutations in adipose triglyceride lipase/PNPLA2 or CGI-58/ABHD5. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E289–E296. [Google Scholar] [CrossRef]
- Lavie, M.; Dubuisson, J. Interplay between hepatitis C virus and lipid metabolism during virus entry and assembly. Biochimie 2017, 141, 62–69. [Google Scholar] [CrossRef]
- Rosch, K.; Kwiatkowski, M.; Hofmann, S.; Schobel, A.; Gruttner, C.; Wurlitzer, M.; Schluter, H.; Herker, E. Quantitative Lipid Droplet Proteome Analysis Identifies Annexin A3 as a Cofactor for HCV Particle Production. Cell Rep. 2016, 16, 3219–3231. [Google Scholar] [CrossRef]
- Ye, J.; Li, J.Z.; Liu, Y.; Li, X.; Yang, T.; Ma, X.; Li, Q.; Yao, Z.; Li, P. Cideb, an ER- and lipid droplet-associated protein, mediates VLDL lipidation and maturation by interacting with apolipoprotein B. Cell Metab 2009, 9, 177–190. [Google Scholar] [CrossRef]
- Tiwari, S.; Siddiqi, S.; Siddiqi, S.A. CideB protein is required for the biogenesis of very low density lipoprotein (VLDL) transport vesicle. J. Biol. Chem. 2013, 288, 5157–5165. [Google Scholar] [CrossRef]
- Cai, H.; Yao, W.; Li, L.; Li, X.; Hu, L.; Mai, R.; Peng, T. Cell-death-inducing DFFA-like Effector B Contributes to the Assembly of Hepatitis C Virus (HCV) Particles and Interacts with HCV NS5A. Sci. Rep. 2016, 6, 27778. [Google Scholar] [CrossRef]
- Weller, R.; Hueging, K.; Brown, R.J.P.; Todt, D.; Joecks, S.; Vondran, F.W.R.; Pietschmann, T. Hepatitis C Virus Strain-Dependent Usage of Apolipoprotein E Modulates Assembly Efficiency and Specific Infectivity of Secreted Virions. J. Virol. 2017, 91, e00422-17. [Google Scholar] [CrossRef] [PubMed]
- Cottarel, J.; Plissonnier, M.L.; Kullolli, M.; Pitteri, S.; Clement, S.; Millarte, V.; Si-Ahmed, S.N.; Farhan, H.; Zoulim, F.; Parent, R. FIG4 is a hepatitis C virus particle-bound protein implicated in virion morphogenesis and infectivity with cholesteryl ester modulation potential. J. Gen. Virol. 2016, 97, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.Y.; Wang, W.T.; Li, H.K.; Chen, W.J.; Tsai, Y.H.; Chao, C.H.; Wu Lee, Y.H. RNA helicase DDX3 maintains lipid homeostasis through upregulation of the microsomal triglyceride transfer protein by interacting with HNF4 and SHP. Sci. Rep. 2017, 7, 41452. [Google Scholar] [CrossRef] [PubMed]
- Syed, G.H.; Khan, M.; Yang, S.; Siddiqui, A. Hepatitis C Virus Lipoviroparticles Assemble in the Endoplasmic Reticulum (ER) and Bud off from the ER to the Golgi Compartment in COPII Vesicles. J. Virol. 2017, 91, e00499-17. [Google Scholar] [CrossRef] [PubMed]
- Takacs, C.N.; Andreo, U.; Dao Thi, V.L.; Wu, X.; Gleason, C.E.; Itano, M.S.; Spitz-Becker, G.S.; Belote, R.L.; Hedin, B.R.; Scull, M.A.; et al. Differential Regulation of Lipoprotein and Hepatitis C Virus Secretion by Rab1b. Cell Rep. 2017, 21, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Moessinger, C.; Kuerschner, L.; Spandl, J.; Shevchenko, A.; Thiele, C. Human lysophosphatidylcholine acyltransferases 1 and 2 are located in lipid droplets where they catalyze the formation of phosphatidylcholine. J. Biol. Chem. 2011, 286, 21330–21339. [Google Scholar] [CrossRef]
- Beilstein, F.; Lemasson, M.; Pene, V.; Rainteau, D.; Demignot, S.; Rosenberg, A.R. Lysophosphatidylcholine acyltransferase 1 is downregulated by hepatitis C virus: Impact on production of lipo-viro-particles. Gut 2017, 66, 2160–2169. [Google Scholar] [CrossRef]
- Mankouri, J.; Walter, C.; Stewart, H.; Bentham, M.; Park, W.S.; Heo, W.D.; Fukuda, M.; Griffin, S.; Harris, M. Release of Infectious Hepatitis C Virus from Huh7 Cells Occurs via a trans-Golgi Network-to-Endosome Pathway Independent of Very-Low-Density Lipoprotein Secretion. J. Virol. 2016, 90, 7159–7170. [Google Scholar] [CrossRef]
- Bayer, K.; Banning, C.; Bruss, V.; Wiltzer-Bach, L.; Schindler, M. Hepatitis C Virus Is Released via a Noncanonical Secretory Route. J. Virol. 2016, 90, 10558–10573. [Google Scholar] [CrossRef]
- Elgner, F.; Donnerhak, C.; Ren, H.; Medvedev, R.; Schreiber, A.; Weber, L.; Heilmann, M.; Ploen, D.; Himmelsbach, K.; Finkernagel, M.; et al. Characterization of alpha-taxilin as a novel factor controlling the release of hepatitis C virus. Biochem. J. 2016, 473, 145–155. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bley, H.; Schöbel, A.; Herker, E. Whole Lotta Lipids—From HCV RNA Replication to the Mature Viral Particle. Int. J. Mol. Sci. 2020, 21, 2888. https://doi.org/10.3390/ijms21082888
Bley H, Schöbel A, Herker E. Whole Lotta Lipids—From HCV RNA Replication to the Mature Viral Particle. International Journal of Molecular Sciences. 2020; 21(8):2888. https://doi.org/10.3390/ijms21082888
Chicago/Turabian StyleBley, Hanna, Anja Schöbel, and Eva Herker. 2020. "Whole Lotta Lipids—From HCV RNA Replication to the Mature Viral Particle" International Journal of Molecular Sciences 21, no. 8: 2888. https://doi.org/10.3390/ijms21082888
APA StyleBley, H., Schöbel, A., & Herker, E. (2020). Whole Lotta Lipids—From HCV RNA Replication to the Mature Viral Particle. International Journal of Molecular Sciences, 21(8), 2888. https://doi.org/10.3390/ijms21082888




