Curcumin Attenuates Both Acute and Chronic Immune Nephritis
Abstract
1. Introduction
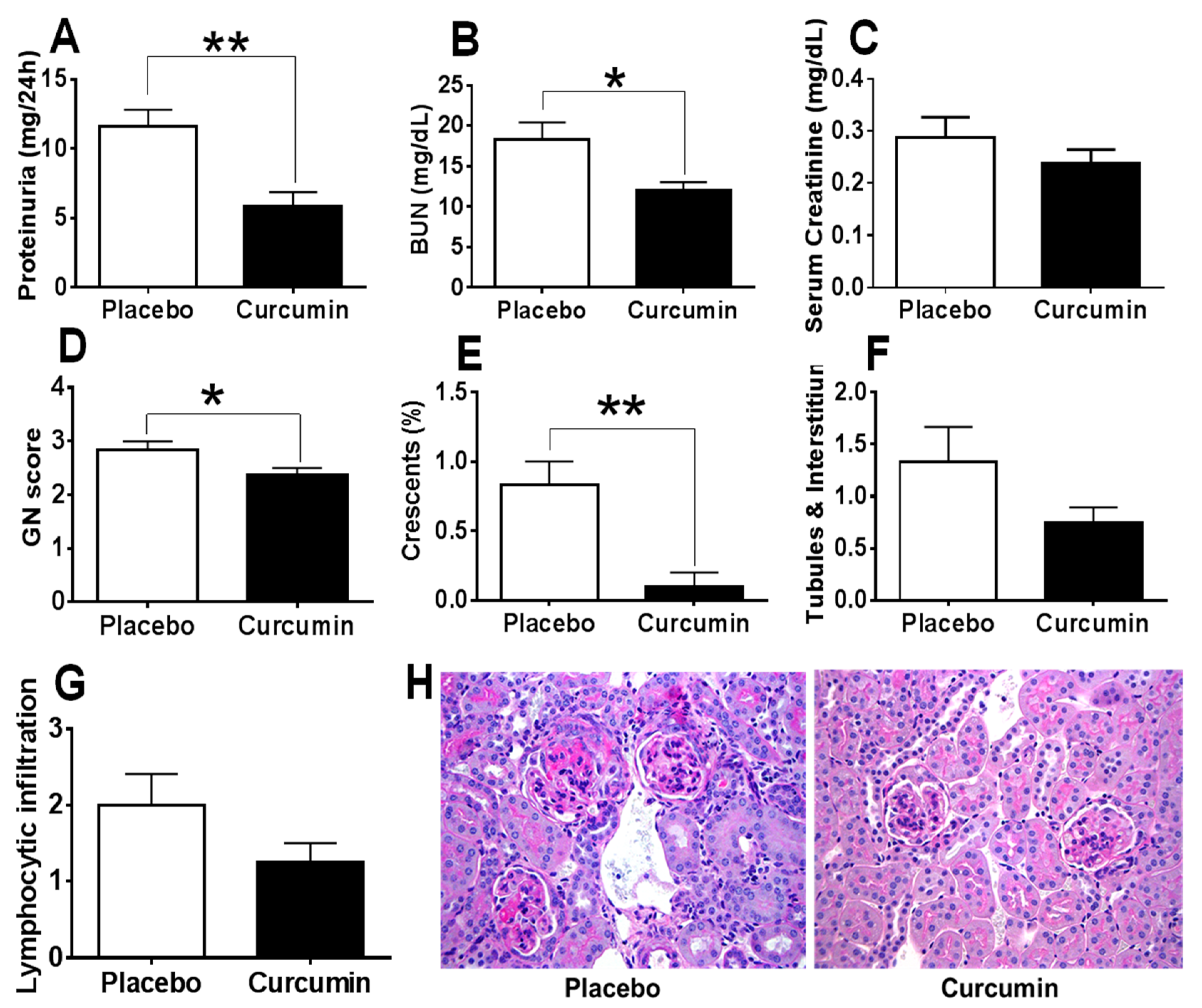
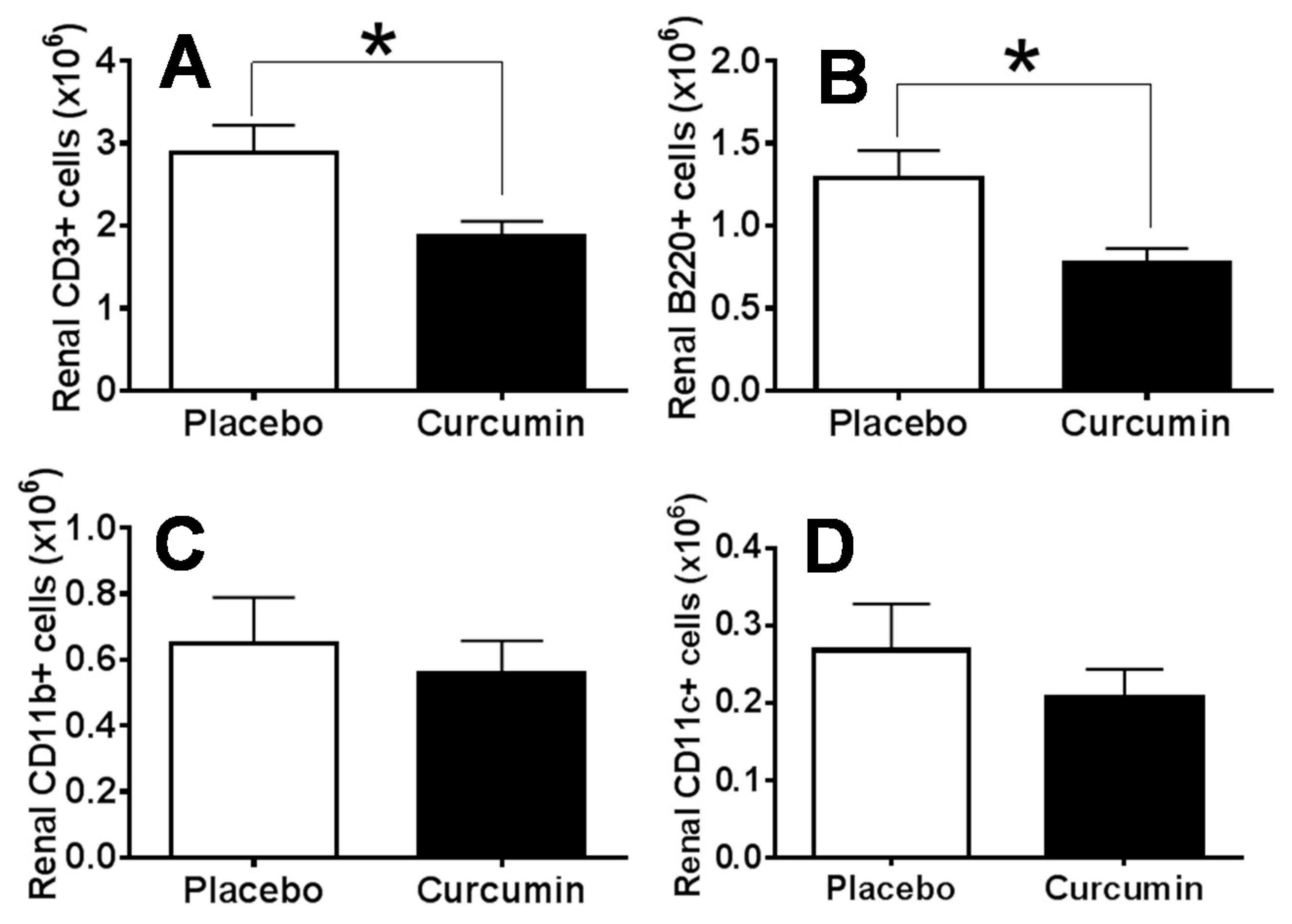
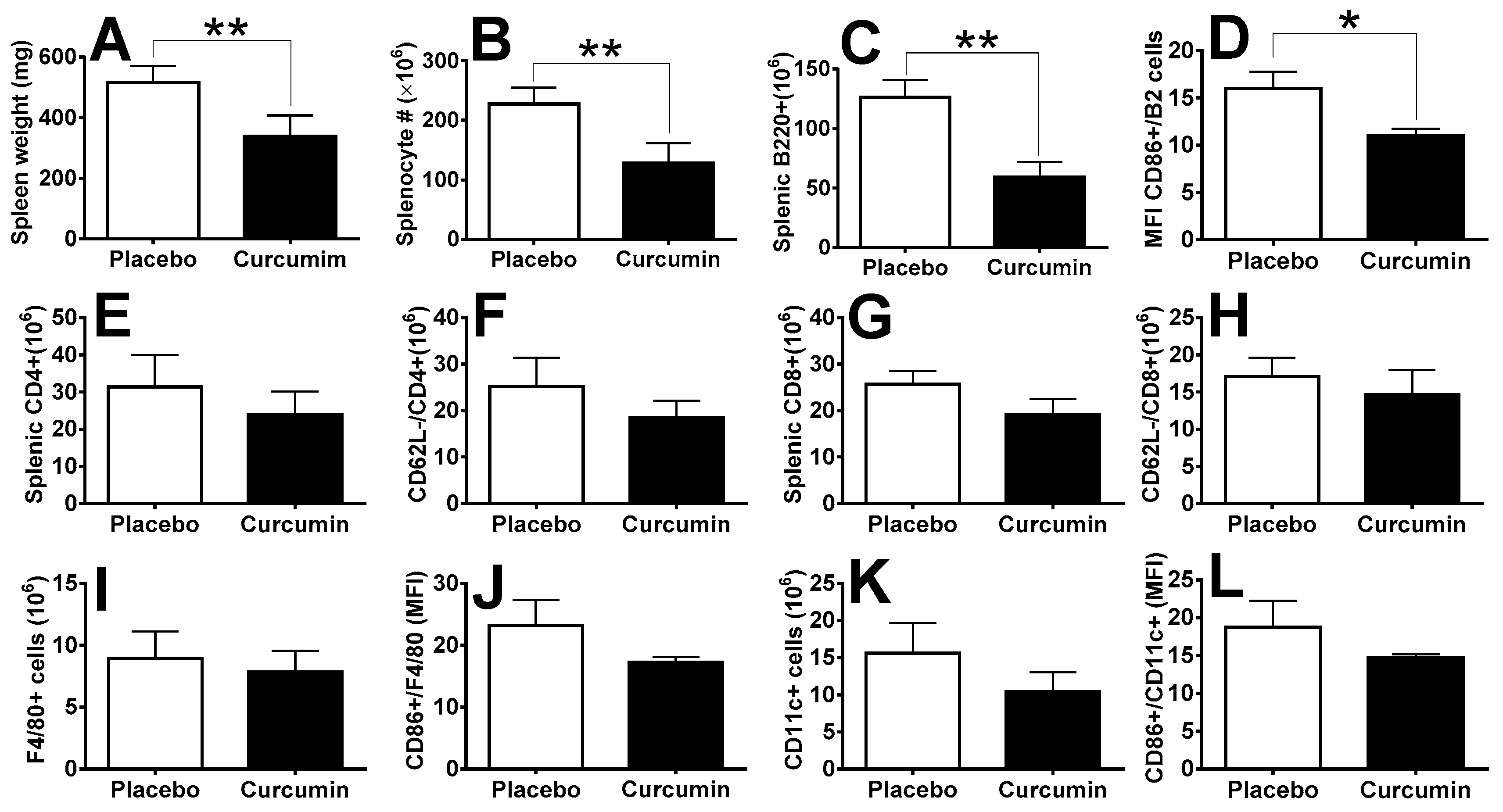
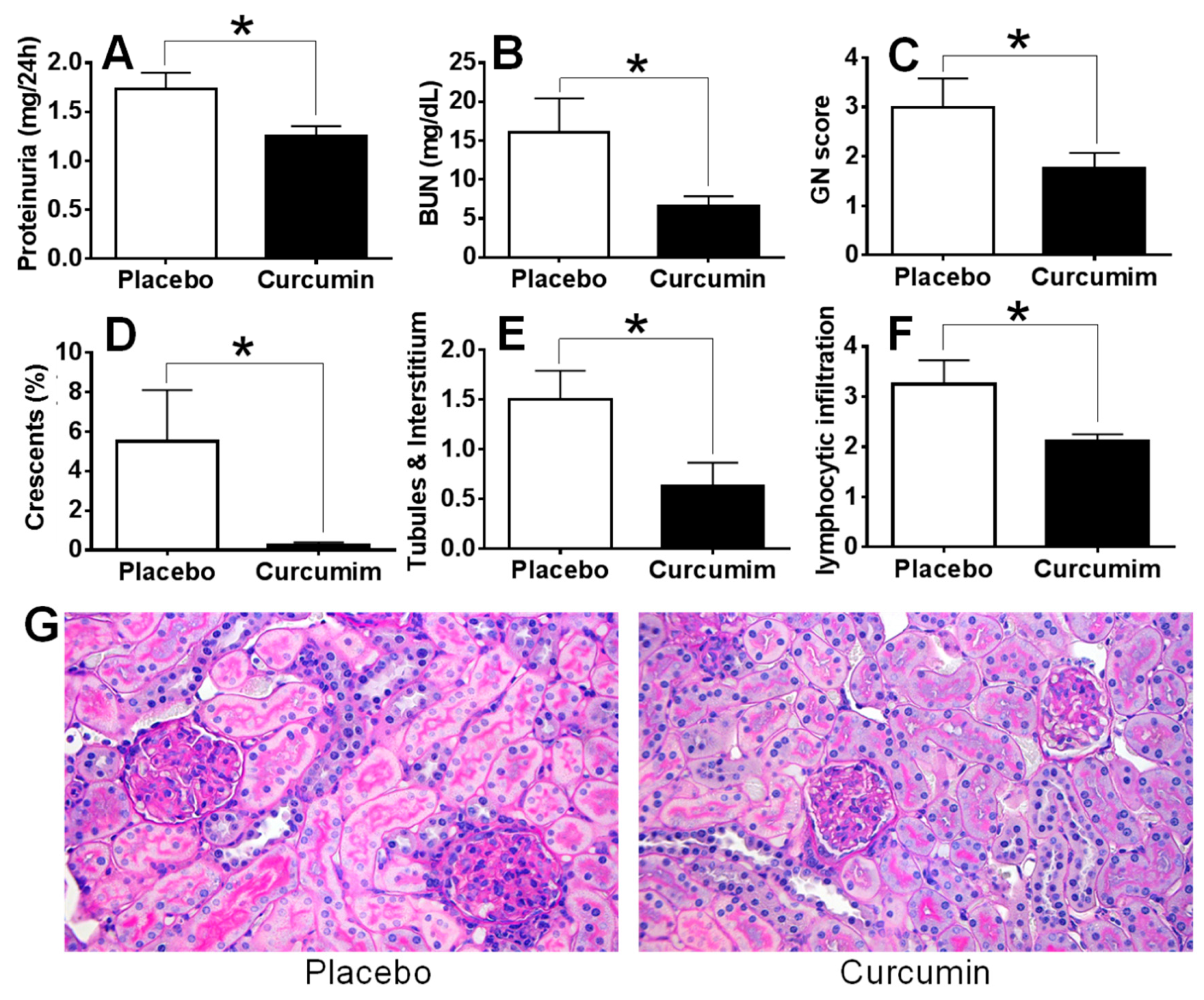
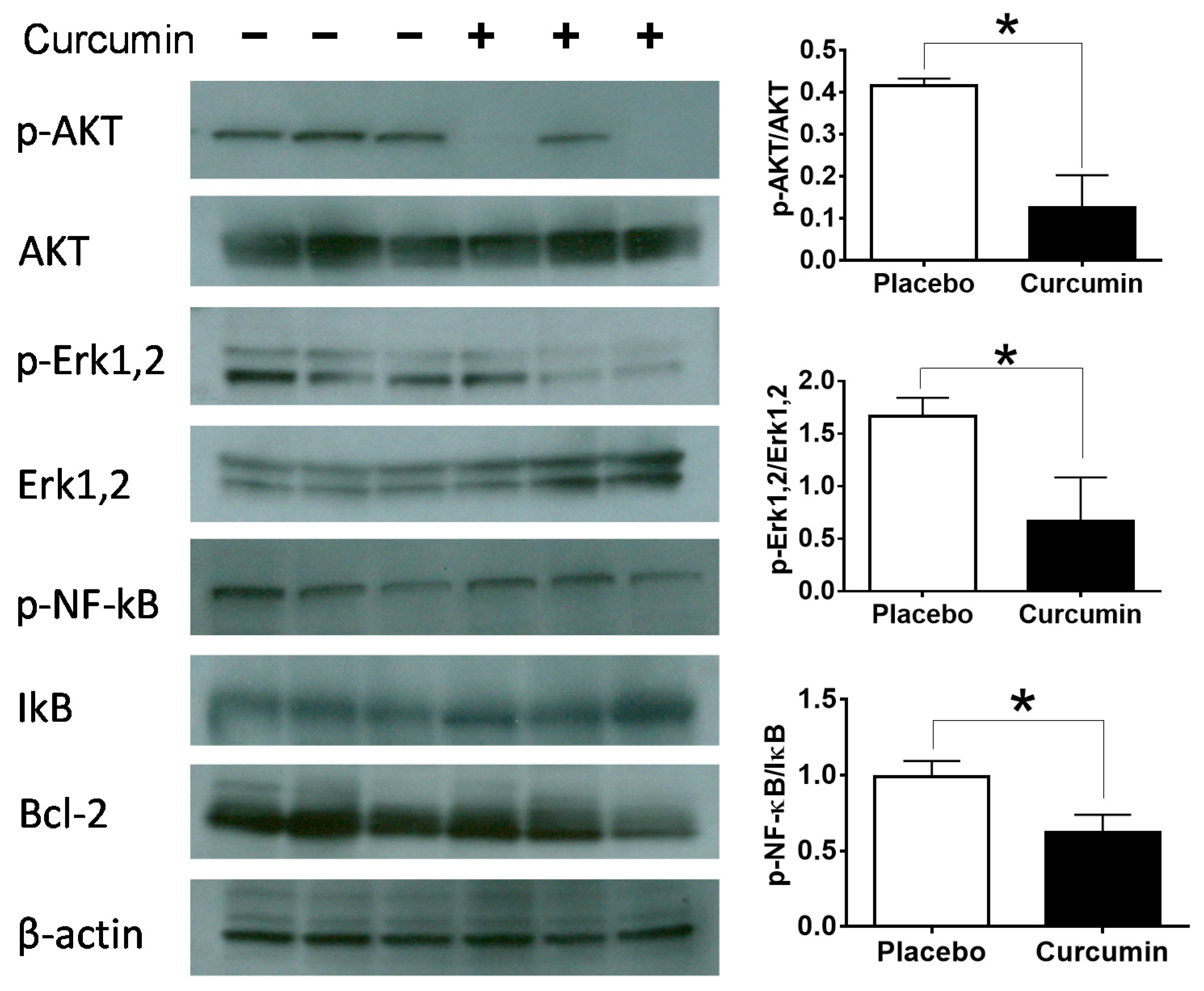
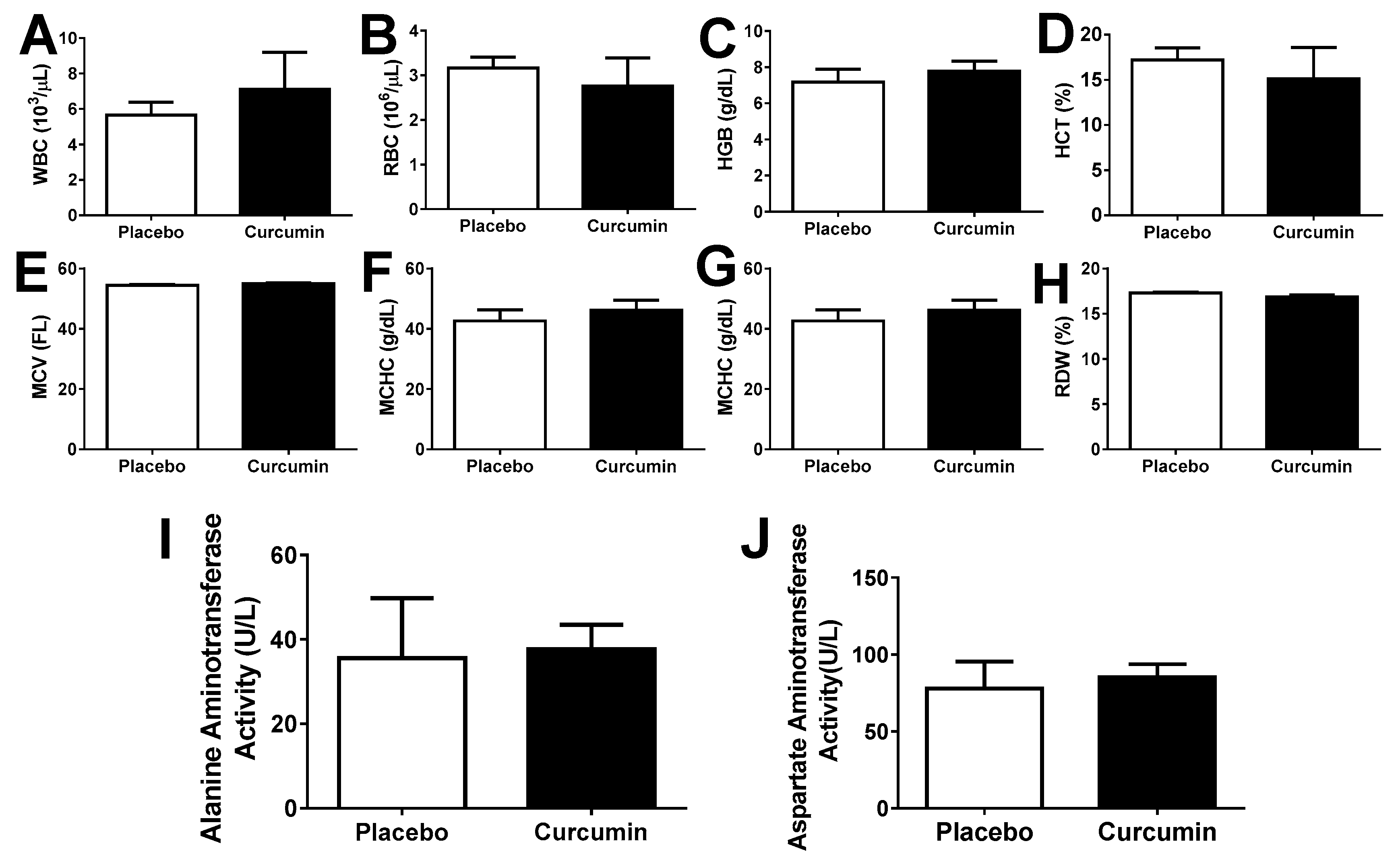
2. Results
3. Discussion
4. Materials and Methods
4.1. Mice and Treatment
4.2. Renal Disease
4.3. Flow Cytometric Analysis
4.4. ELISA for Autoantibodies
4.5. Western Blot Analysis
4.6. Statistics
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Nasr, S.H.; D’Agati, V.D.; Park, H.-R.; Sterman, P.L.; Goyzueta, J.D.; Dressler, R.M.; Hazlett, S.M.; Pursell, R.N.; Caputo, C.; Markowitz, G.S. Necrotizing and crescentic lupus nephritis with antineutrophil cytoplasmic antibody seropositivity. Clin. J. Am. Soc. Nephrol. 2008, 3, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Couser, W.G. Basic and translational concepts of immune-mediated glomerular diseases. J. Am. Soc. Nephrol. 2012, 23, 381–399. [Google Scholar] [CrossRef] [PubMed]
- Weening, J.J.; D’agati, V.D.; Schwartz, M.M.; Seshan, S.V.; Alpers, C.E.; Appel, G.B.; Balow, J.E.; Bruijn, J.; Cook, T.; Ferrario, F. The classification of glomerulonephritis in systemic lupus erythematosus revisited. Kidney Int. 2004, 65, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Jennette, J.C.; Wilkman, A.S.; Falk, R.J. Anti-neutrophil cytoplasmic autoantibody-associated glomerulonephritis and vasculitis. Am. J. Pathol. 1989, 135, 921. [Google Scholar]
- Donadio, J.V.; Grande, J.P. IgA nephropathy. New Engl. J. Med. 2002, 347, 738–748. [Google Scholar] [CrossRef] [PubMed]
- Mathieson, P.W. Immune dysregulation in minimal change nephropathy. Nephrol. Dial. Transplant. 2003, 18, vi26–vi29. [Google Scholar] [CrossRef]
- Cameron, J.S. Lupus nephritis. J. Am. Soc. Nephrol. 1999, 10, 413–424. [Google Scholar]
- Tian, S.Y.; Feldman, B.M.; Beyene, J.; Brown, P.E.; Uleryk, E.M.; Silverman, E.D. Immunosuppressive therapies for the induction treatment of proliferative lupus nephritis: A systematic review and network metaanalysis. J. Rheumatol. 2014, 41, 1998–2007. [Google Scholar] [CrossRef]
- Van Vollenhoven, R.F.; Mosca, M.; Bertsias, G.; Isenberg, D.; Kuhn, A.; Lerstrøm, K.; Aringer, M.; Bootsma, H.; Boumpas, D.; Bruce, I.N. Treat-to-target in systemic lupus erythematosus: Recommendations from an international task force. Ann. Rheum. Dis. 2014. [Google Scholar] [CrossRef]
- Ammon, H.; Anazodo, M.I.; Safayhi, H.; Dhawan, B.N.; Srimal, R.C. Curcumin: A potent inhibitor of leukotriene B4 formation in rat peritoneal polymorphonuclear neutrophils (PMNL). Planta Medica. 1992, 58, 226. [Google Scholar] [CrossRef]
- Jain, S.K.; Rains, J.; Jones, K. Effect of curcumin on protein glycosylation, lipid peroxidation, and oxygen radical generation in human red blood cells exposed to high glucose levels. Free Radic. Biol. Med. 2006, 41, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Kanwar, M. Effects of curcumin on retinal oxidative stress and inflammation in diabetes. Nutr. Metab. 2007, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Chainani-Wu, N. Safety and anti-inflammatory activity of curcumin: A component of tumeric (Curcuma longa). J. Altern. Complementary Med. 2003, 9, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Cole, G.M.; Lim, G.P.; Yang, F.; Teter, B.; Begum, A.; Ma, Q.; Harris-White, M.E.; Frautschy, S.A. Prevention of Alzheimer’s disease: Omega-3 fatty acid and phenolic anti-oxidant interventions. Neurobiol. Aging 2005, 26, 133–136. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Aggarwal, B.B. “Spicing up” of the immune system by curcumin. J. Clin. Immunol. 2007, 27, 19–35. [Google Scholar] [CrossRef]
- Gao, X.; Kuo, J.; Jiang, H.; Deeb, D.; Liu, Y.; Divine, G.; Chapman, R.A.; Dulchavsky, S.A.; Gautam, S.C. Immunomodulatory activity of curcumin: Suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production in vitro. Biochem. Pharmacol. 2004, 68, 51–61. [Google Scholar] [CrossRef]
- Ammon, H.P.; Wahl, M.A. Pharmacology of Curcuma longa. Planta Med. 1991, 57, 1–7. [Google Scholar] [CrossRef]
- Mandal, M.N.; Patlolla, J.M.; Zheng, L.; Agbaga, M.P.; Tran, J.T.; Wicker, L.; Kasus-Jacobi, A.; Elliott, M.H.; Rao, C.V.; Anderson, R.E. Curcumin protects retinal cells from light-and oxidant stress-induced cell death. Free Radic. Biol. Med. 2009, 46, 672–679. [Google Scholar] [CrossRef]
- Jung, K.A.; Kwak, M.K. The Nrf2 system as a potential target for the development of indirect antioxidants. Molecules 2010, 15, 7266–7291. [Google Scholar] [CrossRef]
- Wong, S.H.; Barlow, J.L.; Nabarro, S.; Fallon, P.G.; McKenzie, A.N. Tim-1 is induced on germinal centre B cells through B-cell receptor signalling but is not essential for the germinal centre response. Immunology 2010, 131, 77–88. [Google Scholar] [CrossRef]
- Ichimura, T.; Brooks, C.R.; Bonventre, J.V. Kim-1/Tim-1 and immune cells: Shifting sands. Kidney Int. 2012, 81, 809–811. [Google Scholar] [CrossRef] [PubMed]
- Binion, D.G.; Heidemann, J.; Li, M.S.; Nelson, V.M.; Otterson, M.F.; Rafiee, P. Vascular cell adhesion molecule-1 expression in human intestinal microvascular endothelial cells is regulated by PI 3-kinase/Akt/MAPK/NF-kappaB: Inhibitory role of curcumin. Am. J. Physiol. 2009, 297, G259–G268. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M.; Tripathi, L.M.; Srivastava, V.M.; Puri, A.; Shukla, R. Modulation of inflammatory mediators by ibuprofen and curcumin treatment during chronic inflammation in rat. Immunopharmacol. Immunotoxicol. 2003, 25, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Deeb, D.; Jiang, H.; Gao, X.; Al-Holou, S.; Danyluk, A.L.; Dulchavsky, S.A.; Gautam, S.C. Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1-6-heptadine-3,5-dione; C21H20O6] sensitizes human prostate cancer cells to tumor necrosis factor-related apoptosis-inducing ligand/Apo2L-induced apoptosis by suppressing nuclear factor-kappaB via inhibition of the prosurvival Akt signaling pathway. J. Pharmacol. Exp. Ther. 2007, 321, 616–625. [Google Scholar]
- Guimarães, M.R.; Leite, F.R.M.; Spolidorio, L.C.; Kirkwood, K.L.; Rossa, C. Curcumin abrogates LPS-induced pro-inflammatory cytokines in RAW 264.7 macrophages. Evidence for novel mechanisms involving SOCS-1,-3 and p38 MAPK. Arch. Oral Biol. 2013, 58, 1309–1317. [Google Scholar]
- Hanai, H.; Iida, T.; Takeuchi, K.; Watanabe, F.; Maruyama, Y.; Andoh, A.; Tsujikawa, T.; Fujiyama, Y.; Mitsuyama, K.; Sata, M.; et al. Curcumin maintenance therapy for ulcerative colitis: Randomized, multicenter, double-blind, placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2006, 4, 1502–1506. [Google Scholar] [CrossRef]
- Hong, J.; Bose, M.; Ju, J.; Ryu, J.H.; Chen, X.; Sang, S.; Lee, M.J.; Yang, C.S. Modulation of arachidonic acid metabolism by curcumin and related beta-diketone derivatives: Effects on cytosolic phospholipase A(2), cyclooxygenases and 5-lipoxygenase. Carcinogenesis 2004, 25, 1671–1679. [Google Scholar] [CrossRef]
- Lin, J.K.; Shih, C.A. Inhibitory effect of curcumin on xanthine dehydrogenase/oxidase induced by phorbol-12-myristate-13-acetate in NIH3T3 cells. Carcinogenesis 1994, 15, 1717–1721. [Google Scholar] [CrossRef]
- Ghoneim, A.I.; Abdel-Naim, A.B.; Khalifa, A.E.; El-Denshary, E.S. Protective effects of curcumin against ischaemia/reperfusion insult in rat forebrain. Pharmacol. Res. 2002, 46, 273–279. [Google Scholar] [CrossRef]
- Manikandan, R.; Thiagarajan, R.; Beulaja, S.; Sudhandiran, G.; Arumugam, M. Curcumin protects against hepatic and renal injuries mediated by inducible nitric oxide synthase during selenium-induced toxicity in Wistar rats. Microsc. Res. Tech. 2010, 73, 631–637. [Google Scholar] [CrossRef]
- Chainani-Wu, N.; Silverman, S., Jr.; Reingold, A.; Bostrom, A.; Mc Culloch, C.; Lozada-Nur, F.; Weintraub, J. A randomized, placebo-controlled, double-blind clinical trial of curcuminoids in oral lichen planus. Phytomedicine 2007, 14, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Baum, L.; Lam, C.W.; Cheung, S.K.; Kwok, T.; Lui, V.; Tsoh, J.; Lam, L.; Leung, V.; Hui, E.; Ng, C.; et al. Six-month randomized, placebo-controlled, double-blind, pilot clinical trial of curcumin in patients with Alzheimer disease. J. Clin. Psychopharmacol. 2008, 28, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Usharani, P.; Mateen, A.A.; Naidu, M.U.; Raju, Y.S.; Chandra, N. Effect of NCB-02, atorvastatin and placebo on endothelial function, oxidative stress and inflammatory markers in patients with type 2 diabetes mellitus: A randomized, parallel-group, placebo-controlled, 8-week study. Drugs R D 2008, 9, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Khajehdehi, P.; Pakfetrat, M.; Javidnia, K.; Azad, F.; Malekmakan, L.; Nasab, M.H.; Dehghanzadeh, G. Oral supplementation of turmeric attenuates proteinuria, transforming growth factor-beta and interleukin-8 levels in patients with overt type 2 diabetic nephropathy: A randomized, double-blind and placebo-controlled study. Scand. J. Urol. Nephrol. 2011, 45, 365–370. [Google Scholar] [CrossRef]
- Agarwal, K.A.; Tripathi, C.D.; Agarwal, B.B.; Saluja, S. Efficacy of turmeric (curcumin) in pain and postoperative fatigue after laparoscopic cholecystectomy: A double-blind, randomized placebo-controlled study. Surg. Endosc. 2011, 25, 3805–3810. [Google Scholar] [CrossRef]
- Golombick, T.; Diamond, T.H.; Manoharan, A.; Ramakrishna, R. Monoclonal gammopathy of undetermined significance, smoldering multiple myeloma, and curcumin: A randomized, double-blind placebo-controlled cross-over 4g study and an open-label 8g extension study. Am. J. Hematol. 2012, 87, 455–460. [Google Scholar] [CrossRef]
- Wu, T.; Qin, X.; Kurepa, Z.; Kumar, K.R.; Liu, K.; Kanta, H.; Zhou, X.J.; Satterthwaite, A.B.; Davis, L.S.; Mohan, C. Shared signaling networks active in B cells isolated from genetically distinct mouse models of lupus. J. Clin. Investigig. 2007, 117, 2186–2196. [Google Scholar] [CrossRef]
- Wu, T.; Fu, Y.; Brekken, D.; Yan, M.; Zhou, X.J.; Vanarsa, K.; Deljavan, N.; Ahn, C.; Putterman, C.; Mohan, C. Urine proteome scans uncover total urinary protease, prostaglandin D synthase, serum amyloid P, and superoxide dismutase as potential markers of lupus nephritis. J. Immunol. 2010, 184, 2183–2193. [Google Scholar] [CrossRef]
- Xie, C.; Sharma, R.; Wang, H.; Zhou, X.J.; Mohan, C. Strain distribution pattern of susceptibility to immune-mediated nephritis. J. Immunol. 2004, 172, 5047–5055. [Google Scholar] [CrossRef]
- Wu, T.; Ye, Y.; Min, S.Y.; Zhu, J.; Khobahy, E.; Zhou, J.; Yan, M.; Hemachandran, S.; Pathak, S.; Zhou, X.J.; et al. Prevention of murine lupus nephritis by targeting multiple signaling axes and oxidative stress using a synthetic triterpenoid. Arthritis Rheumatol. 2014, 66, 3129–3139. [Google Scholar] [CrossRef]
- Carlsson, A.; Wuttge, D.M.; Ingvarsson, J.; Bengtsson, A.A.; Sturfelt, G.; Borrebaeck, C.A.; Wingren, C. Serum protein profiling of systemic lupus erythematosus and systemic sclerosis using recombinant antibody microarrays. Mol. Cell. Proteom. 2011, 10, M110. 005033. [Google Scholar] [CrossRef]
- Chandran, B.; Goel, A. A randomized, pilot study to assess the efficacy and safety of curcumin in patients with active rheumatoid arthritis. Phytother. Res. 2012, 26, 1719–1725. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.T.A.; El-Asmar, M.F.; El-Ibrashy, I.N.; Rezq, A.M.; Al-Malki, A.L.; Wassef, M.A.; Fouad, H.H.; Ahmed, H.H.; Taha, F.M.; Hassouna, A.A. Effect of novel water soluble curcumin derivative on experimental type-1 diabetes mellitus (short term study). Diabetol. Metab. Syndr. 2012, 4, 1. [Google Scholar]
- Sun, J.; Zhao, Y.; Hu, J. Curcumin inhibits imiquimod-induced psoriasis-like inflammation by inhibiting IL-1beta and IL-6 production in mice. PloS ONE 2013, 8, e67078. [Google Scholar] [CrossRef] [PubMed]
- Ung, V.Y.; Foshaug, R.R.; MacFarlane, S.M.; Churchill, T.A.; Doyle, J.S.; Sydora, B.C.; Fedorak, R.N. Oral administration of curcumin emulsified in carboxymethyl cellulose has a potent anti-inflammatory effect in the IL-10 gene-deficient mouse model of IBD. Dig. Dis. Sci. 2010, 55, 1272–1277. [Google Scholar] [CrossRef] [PubMed]
- Ireson, C.; Orr, S.; Jones, D.J.; Verschoyle, R.; Lim, C.-K.; Luo, J.-L.; Howells, L.; Plummer, S.; Jukes, R.; Williams, M. Characterization of metabolites of the chemopreventive agent curcumin in human and rat hepatocytes and in the rat in vivo, and evaluation of their ability to inhibit phorbol ester-induced prostaglandin E2 production. Cancer Res. 2001, 61, 1058–1064. [Google Scholar]
- Forward, N.A.; Conrad, D.M.; Coombs, M.R.P.; Doucette, C.D.; Furlong, S.J.; Lin, T.-J.; Hoskin, D.W. Curcumin blocks interleukin (IL)-2 signaling in T-lymphocytes by inhibiting IL-2 synthesis, CD25 expression, and IL-2 receptor signaling. Biochem. Biophys. Res. Commun. 2011, 407, 801–806. [Google Scholar] [CrossRef]
- Kliem, C.; Merling, A.; Giaisi, M.; Köhler, R.; Krammer, P.H.; Li-Weber, M. Curcumin suppresses T cell activation by blocking Ca2+ mobilization and nuclear factor of activated T cells (NFAT) activation. J. Biol. Chem. 2012, 287, 10200–10209. [Google Scholar] [CrossRef]
- Han, S.-S.; Chung, S.-T.; Robertson, D.A.; Ranjan, D.; Bondada, S. Curcumin causes the growth arrest and apoptosis of B cell lymphoma by downregulation of egr-1, c-myc, bcl-X L, NF-κB, and p53. Clin. Immunol. 1999, 93, 152–161. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Kay, N.E.; Secreto, C.R.; Shanafelt, T.D. Curcumin inhibits prosurvival pathways in chronic lymphocytic leukemia B cells and may overcome their stromal protection in combination with EGCG. Clin. Cancer Res. 2009, 15, 1250–1258. [Google Scholar] [CrossRef]
- Brouet, I.; Ohshima, H. Curcumin, an anti-tumor promoter and anti-inflammatory agent, inhibits induction of nitric oxide synthase in activated macrophages. Biochem. Biophys. Res. Commun. 1995, 206, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Young, N.A.; Bruss, M.S.; Gardner, M.; Willis, W.L.; Mo, X.; Valiente, G.R.; Cao, Y.; Liu, Z.; Jarjour, W.N.; Wu, L.-C. Oral administration of nano-emulsion curcumin in mice suppresses inflammatory-induced NFκB signaling and macrophage migration. PloS ONE 2014, 9, e111559. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Li, C.; Sun, H.; Luo, T.; Tan, Y.; Tian, D.; Guo, Z. Curcumin inhibits monocyte chemoattractant protein-1 expression and enhances cholesterol efflux by suppressing the c-Jun N-terminal kinase pathway in macrophage. Inflamm. Res. 2014, 63, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Castro, C.; Barcala Tabarrozzi, A.; Winnewisser, J.; Gimeno, M.; Antunica Noguerol, M.; Liberman, A.; Paz, D.; Dewey, R.; Perone, M. Curcumin ameliorates autoimmune diabetes. Evidence in accelerated murine models of type 1 diabetes. Clin. Exp. Immunol. 2014, 177, 149–160. [Google Scholar] [CrossRef]
- Rogers, N.; Kireta, S.; Coates, P. Curcumin induces maturation-arrested dendritic cells that expand regulatory T cells in vitro and in vivo. Clin. Exp. Immunol. 2010, 162, 460–473. [Google Scholar] [CrossRef]
- Bright, J.J. Curcumin and autoimmune disease. In The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease; Springer: Berlin/Heidelberg, Germany, 2007; pp. 425–451. [Google Scholar]
- Kang, D.; Li, B.; Luo, L.; Jiang, W.; Lu, Q.; Rong, M.; Lai, R. Curcumin shows excellent therapeutic effect on psoriasis in mouse model. Biochimie 2016, 123, 73–80. [Google Scholar] [CrossRef]
- Khajehdehi, P.; Zanjaninejad, B.; Aflaki, E.; Nazarinia, M.; Azad, F.; Malekmakan, L.; Dehghanzadeh, G.-R. Oral supplementation of turmeric decreases proteinuria, hematuria, and systolic blood pressure in patients suffering from relapsing or refractory lupus nephritis: A randomized and placebo-controlled study. J. Ren. Nutr. 2012, 22, 50–57. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, T.; Marakkath, B.; Ye, Y.; Khobahy, E.; Yan, M.; Hutcheson, J.; Zhu, J.; Zhou, X.; Mohan, C. Curcumin Attenuates Both Acute and Chronic Immune Nephritis. Int. J. Mol. Sci. 2020, 21, 1745. https://doi.org/10.3390/ijms21051745
Wu T, Marakkath B, Ye Y, Khobahy E, Yan M, Hutcheson J, Zhu J, Zhou X, Mohan C. Curcumin Attenuates Both Acute and Chronic Immune Nephritis. International Journal of Molecular Sciences. 2020; 21(5):1745. https://doi.org/10.3390/ijms21051745
Chicago/Turabian StyleWu, Tianfu, Bindiya Marakkath, Yujin Ye, Elhaum Khobahy, Mei Yan, Jack Hutcheson, Jiankun Zhu, Xinjin Zhou, and Chandra Mohan. 2020. "Curcumin Attenuates Both Acute and Chronic Immune Nephritis" International Journal of Molecular Sciences 21, no. 5: 1745. https://doi.org/10.3390/ijms21051745
APA StyleWu, T., Marakkath, B., Ye, Y., Khobahy, E., Yan, M., Hutcheson, J., Zhu, J., Zhou, X., & Mohan, C. (2020). Curcumin Attenuates Both Acute and Chronic Immune Nephritis. International Journal of Molecular Sciences, 21(5), 1745. https://doi.org/10.3390/ijms21051745






