GABAergic Input Affects Intracellular Calcium Levels in Developing Granule Cells of Adult Rat Hippocampus
Abstract
1. Introduction
2. Results
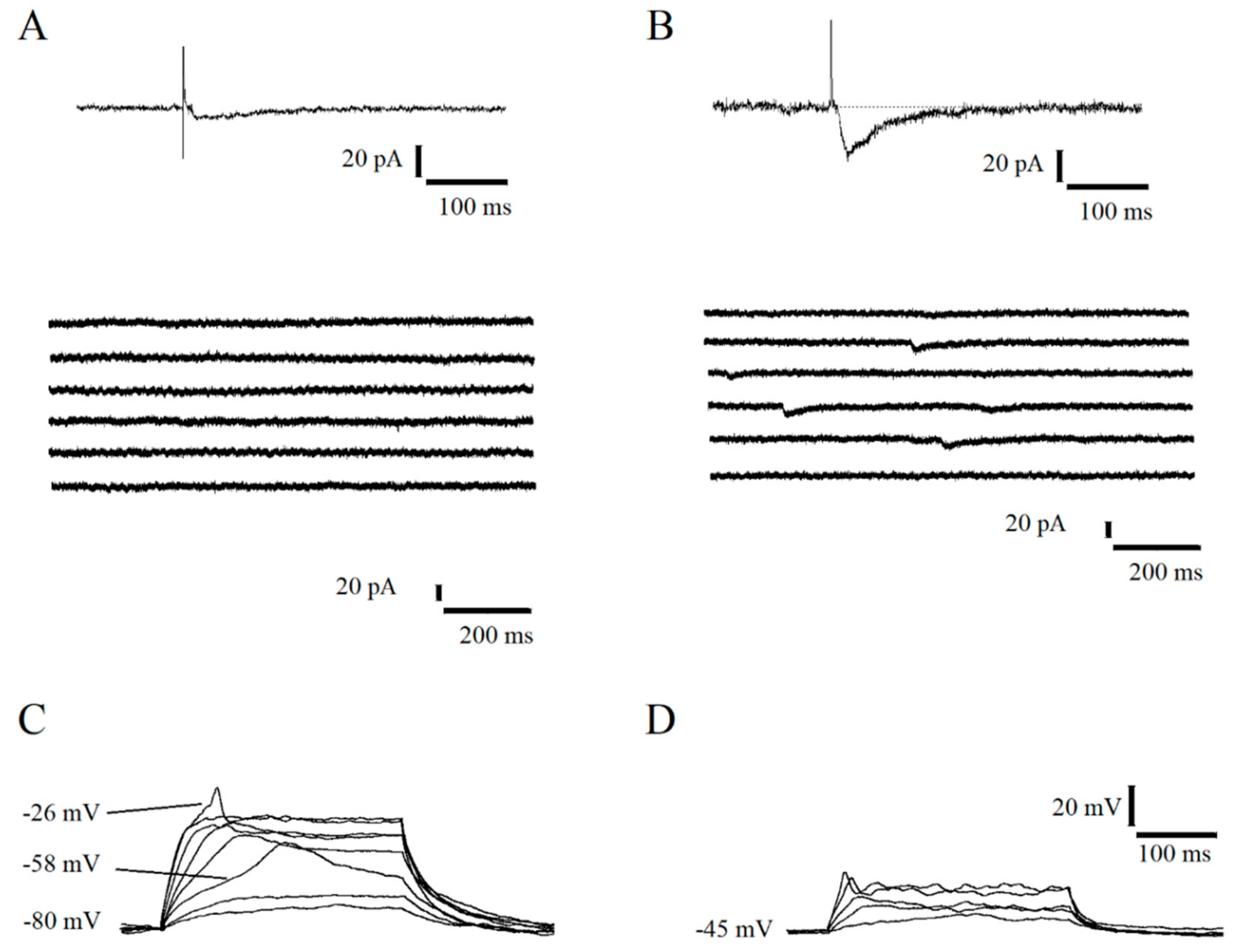
2.1. Functional Features of Immature Dentate Granule Cells with GABAergic Evoked Response
2.2. Resting Membrane Potential Oscillations and Intracellular Ca2+ Levels
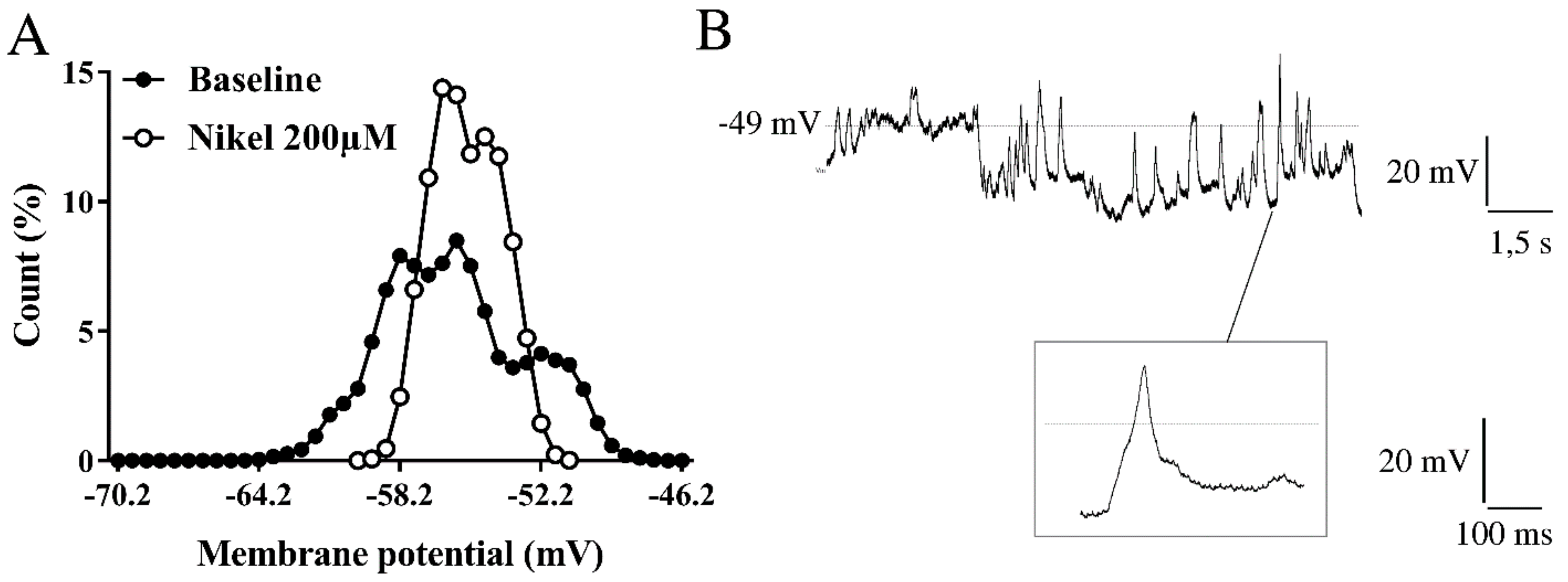
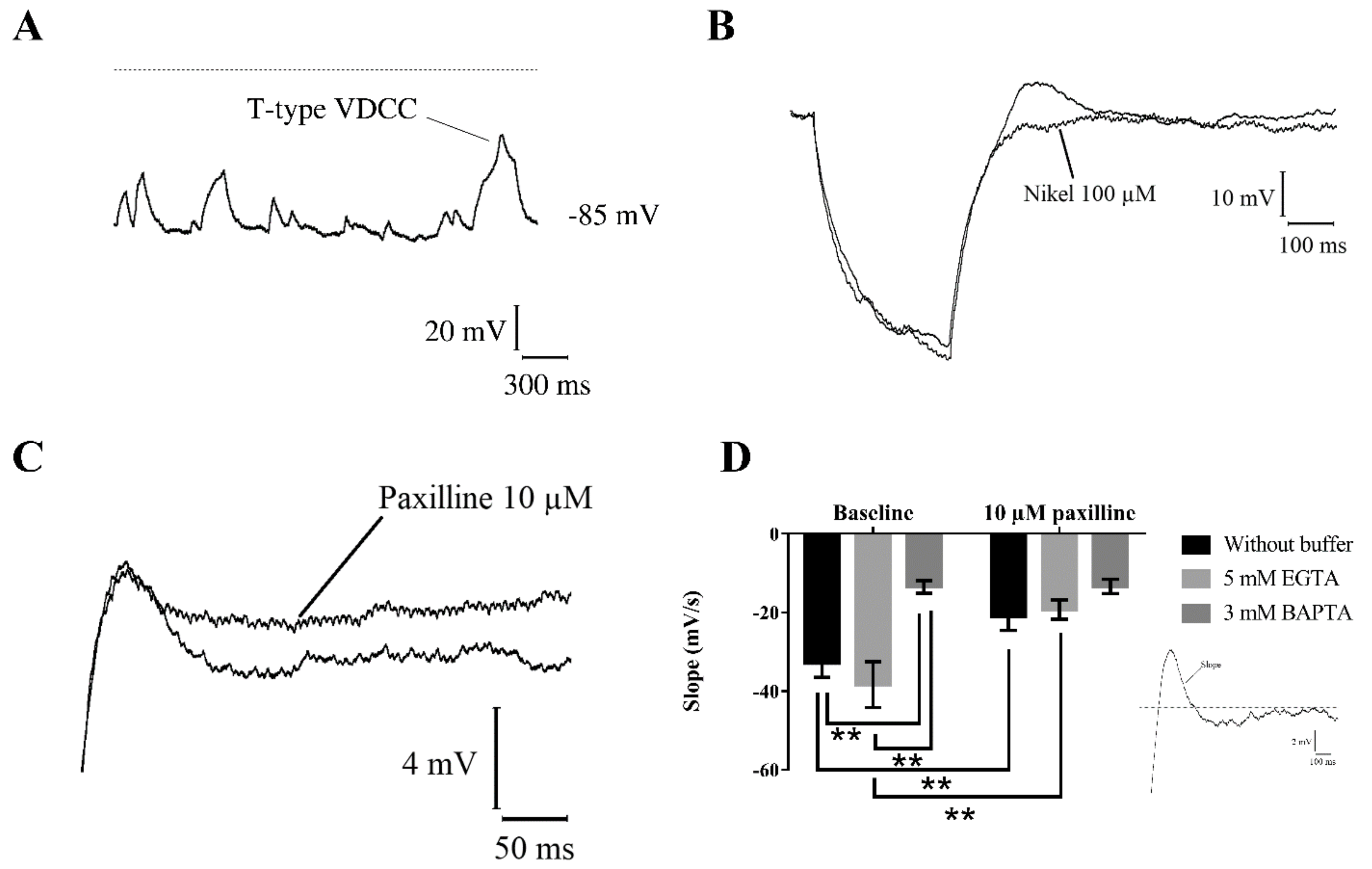
2.3. Membrane Channels Involved in Resting Membrane Potential Oscillations
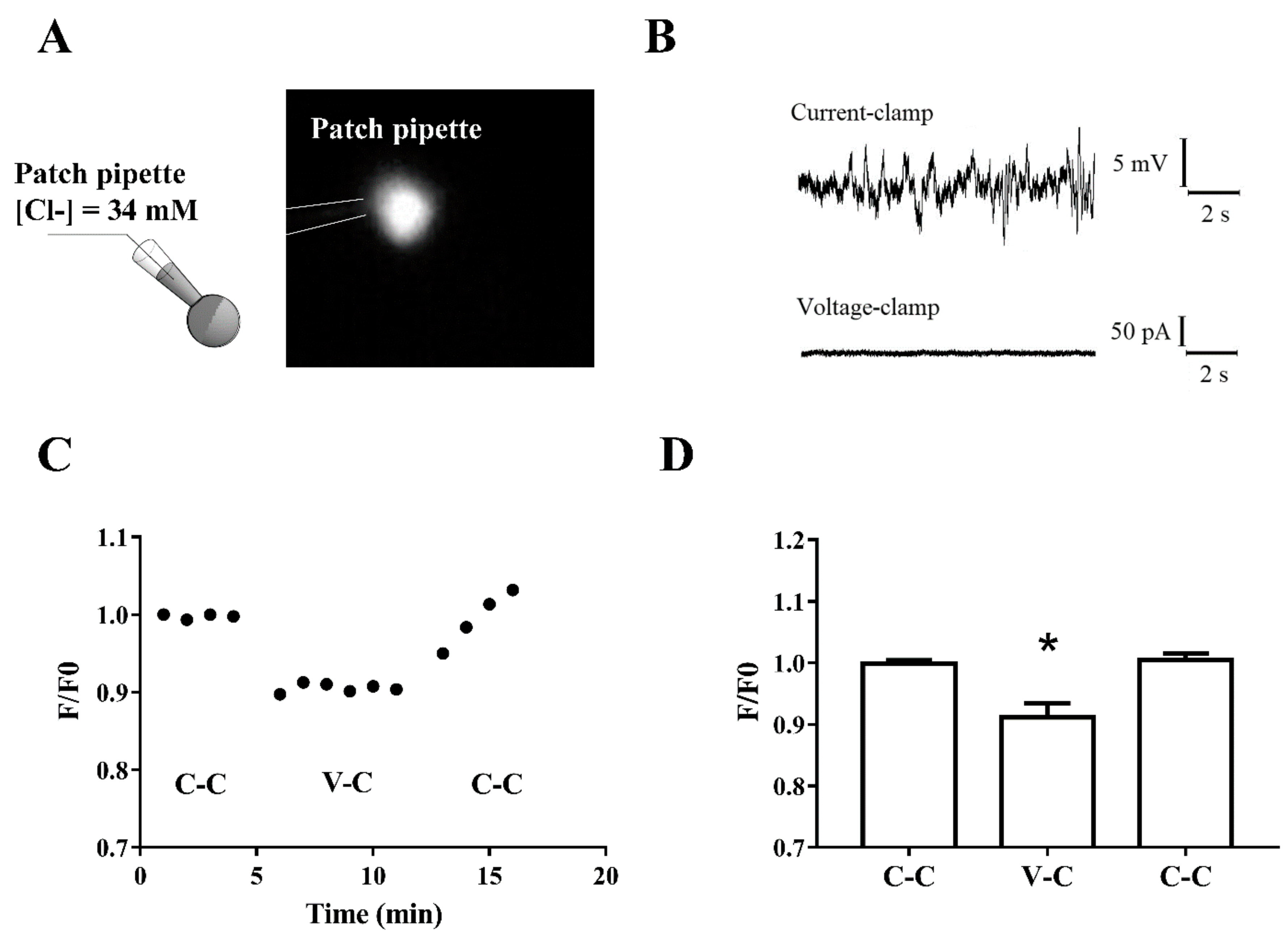
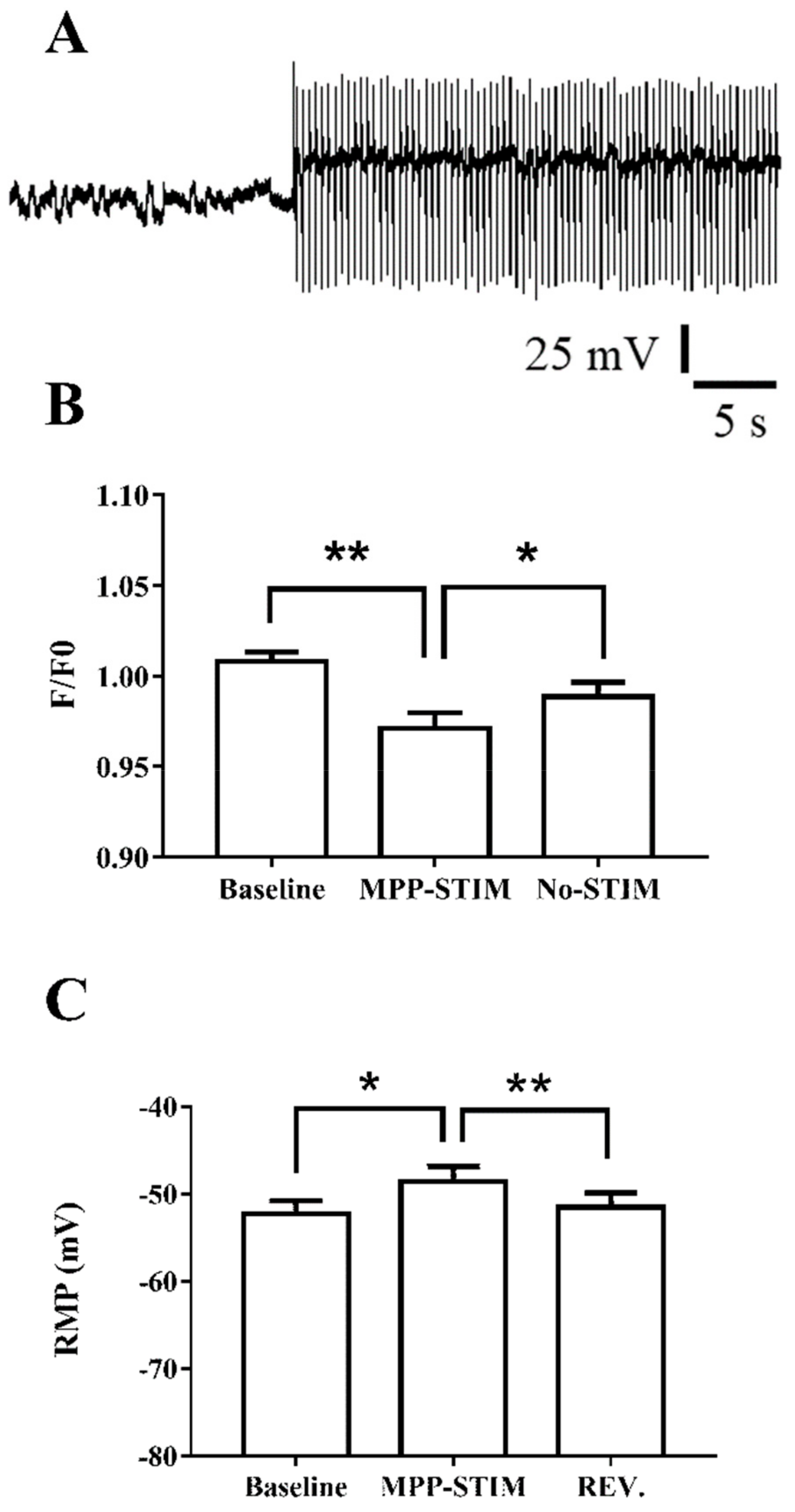
2.4. Hippocampal Circuit Activity and Intracellular Calcium Level in Immature Neurons
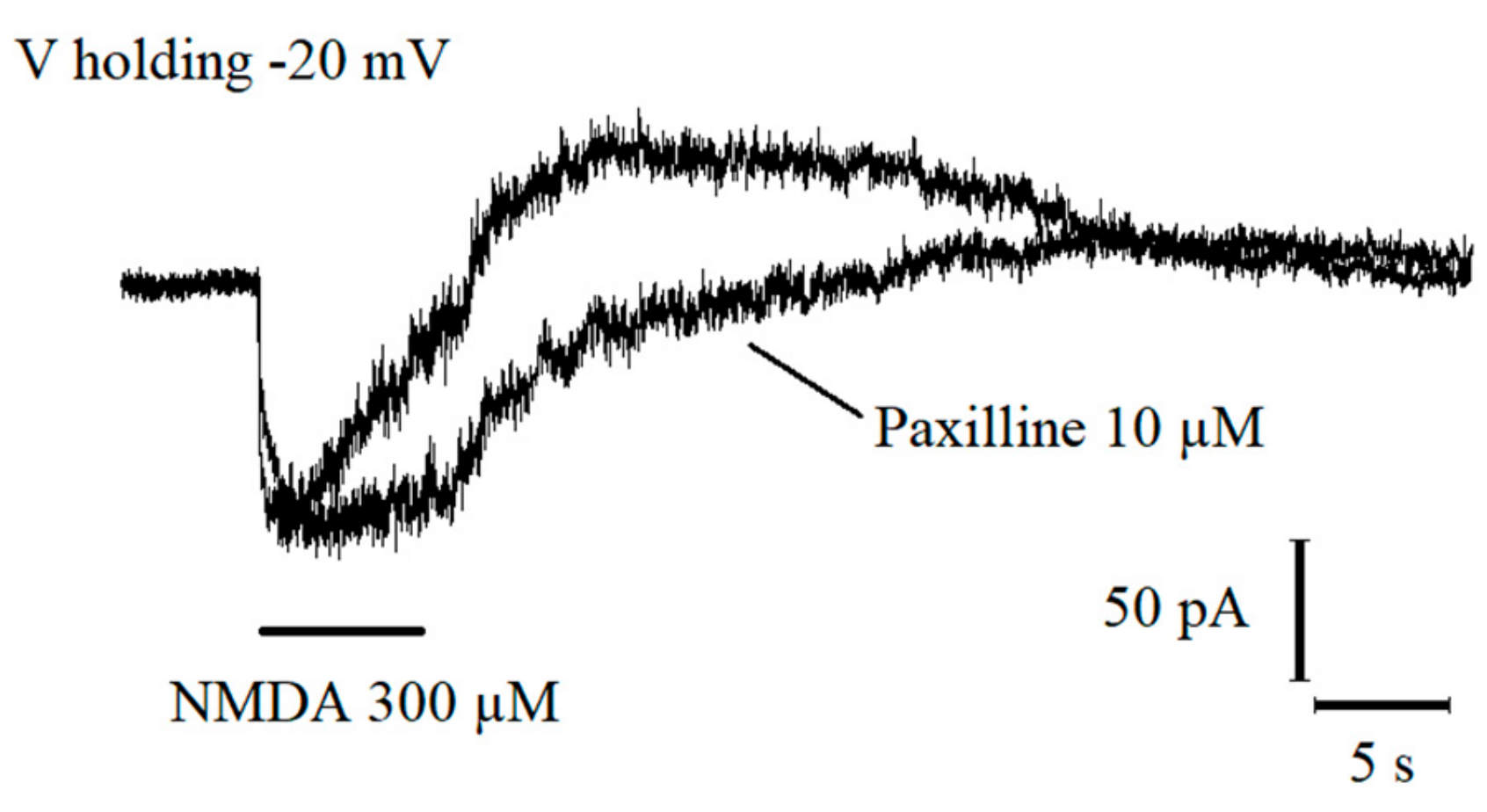
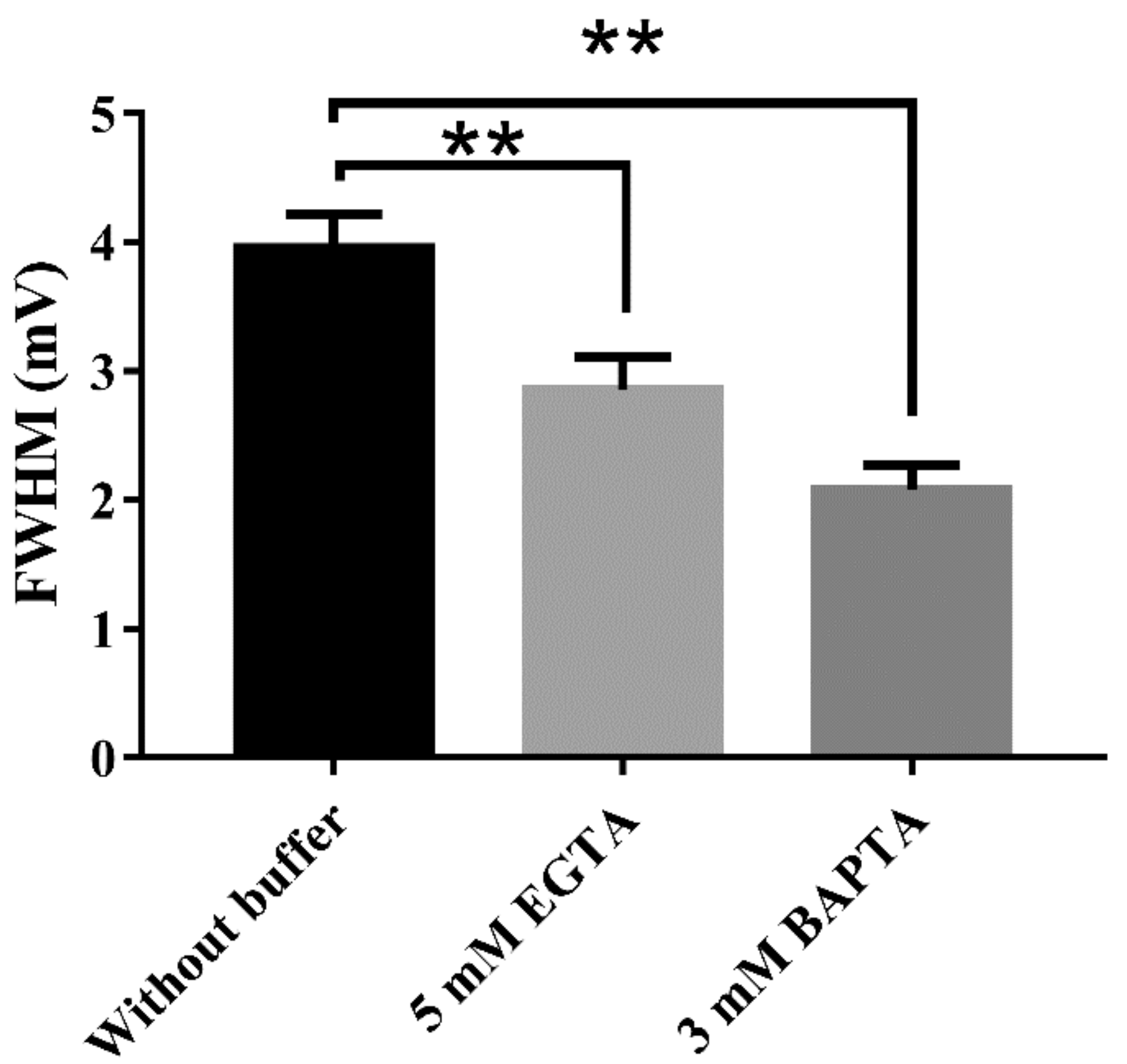
2.5. Interaction between BK Channels and T-Type Voltage-Dependent Ca2+ Channels
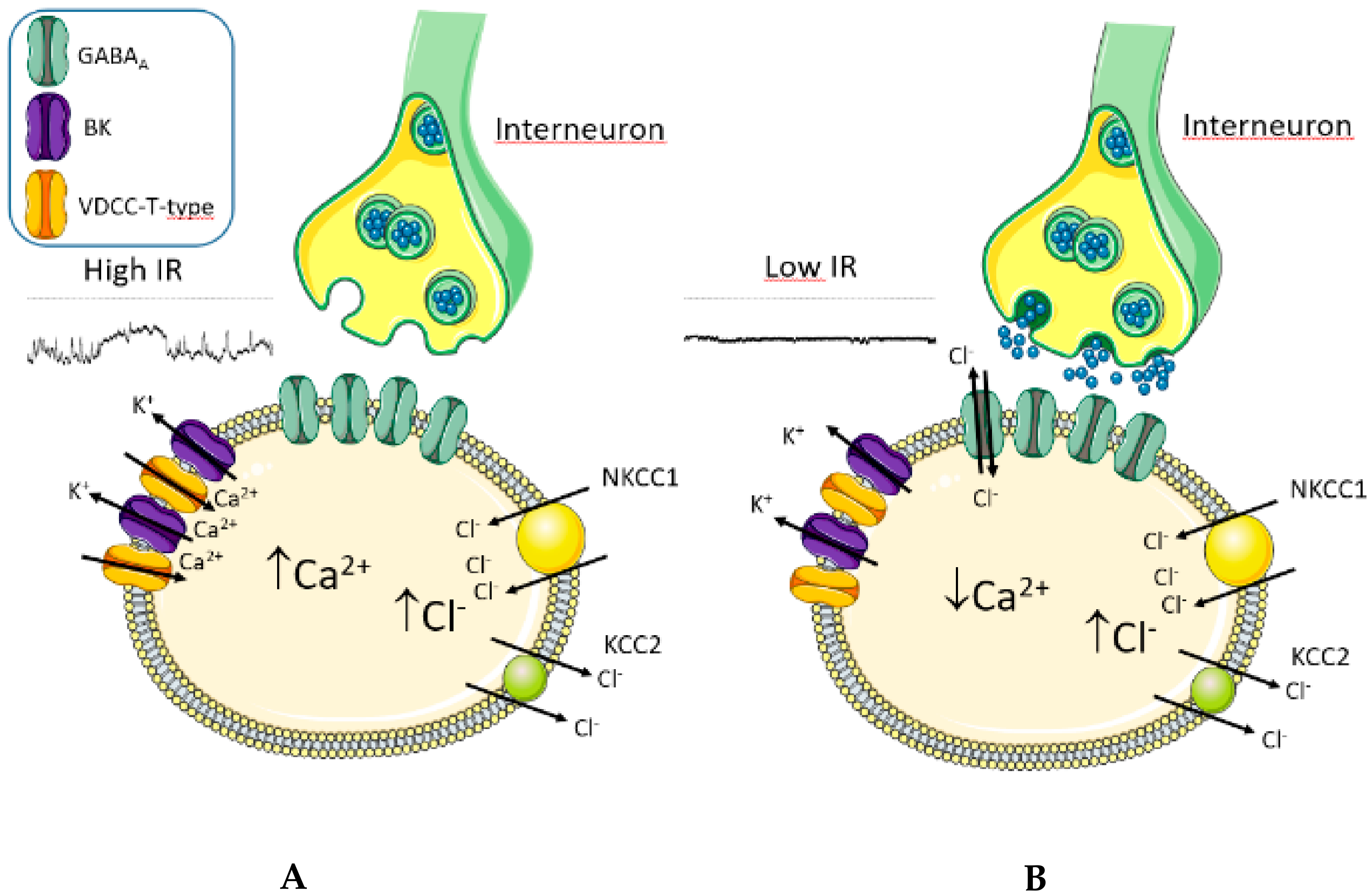
3. Discussion
4. Materials and Methods
4.1. Animals and Slices Preparation
4.2. Patch-Clamp Recordings
4.3. Calcium Imaging
4.4. Drugs Application
4.5. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Gonçalves, J.T.; Schafer, S.T.; Gage, F.H. Adult Neurogenesis in the Hippocampus: From Stem Cells to Behavior. Cell 2016, 167, 897–914. [Google Scholar] [CrossRef]
- Ambrogini, P.; Lattanzi, D.; Ciuffoli, S.; Agostini, D.; Bertini, L.; Stocchi, V.; Santi, S.; Cuppini, R. Morpho-functional characterization of neuronal cells at different stages of maturation in granule cell layer of adult rat dentate gyrus. Brain Res. 2004, 1017, 21–31. [Google Scholar] [CrossRef]
- Dieni, C.V.; Chancey, J.H.; Overstreet-Wadiche, L.S. Dynamic functions of GABA signaling during granule cell maturation. Front. Neural Circuits 2013, 6, 11. [Google Scholar] [CrossRef]
- Bao, H.; Asrican, B.; Li, W.; Gu, B.; Wen, Z.; Lim, S.A.; Haniff, I.; Ramakrishnan, C.; Deisseroth, K.; Philpot, B.; et al. Long-Range GABAergic Inputs Regulate Neural Stem Cell Quiescence and Control Adult Hippocampal Neurogenesis. Cell Stem Cell 2017, 21, 604–617. [Google Scholar] [CrossRef]
- Tozuka, Y.; Fukuda, S.; Namba, T.; Seki, T.; Hisatsune, T. GABAergic excitation promotes neuronal differentiation in adult hippocampal progenitor cells. Neuron 2005, 47, 803–815. [Google Scholar] [CrossRef]
- Ge, S.; Goh, E.L.; Sailor, K.A.; Kitabatake, Y.; Ming, G.L.; Song, H. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 2006, 439, 589–593. [Google Scholar] [CrossRef]
- Deshpande, A.; Bergami, M.; Ghanem, A.; Conzelmann, K.K.; Lepier, A.; Götz, M.; Berninger, B. Retrograde monosynaptic tracing reveals the temporal evolution of inputs onto new neurons in the adult dentate gyrus and olfactory bulb. Proc. Natl. Acad. Sci. USA 2013, 110, E1152–E1161. [Google Scholar] [CrossRef]
- Ben-Ari, Y. The GABA excitatory/inhibitory developmental sequence: A personal journey. Neuroscience 2014, 279, 187–219. [Google Scholar] [CrossRef]
- Ambrogini, P.; Cuppini, R.; Lattanzi, D.; Ciuffoli, S.; Frontini, A.; Fanelli, M. Synaptogenesis in adult-generated hippocampal granule cells is affected by behavioral experiences. Hippocampus 2010, 20, 799–810. [Google Scholar] [CrossRef]
- Ge, S.; Pradhan, D.A.; Ming, G.L.; Song, H. GABA sets the tempo for activity-dependent adult neurogenesis. Trends Neurosci. 2007, 30, 1–8. [Google Scholar] [CrossRef]
- Verheugen, J.A.H.; Vijverberg, H.P.M.; Oortgiesen, M.; Cahalan, M.D. Voltage-gated and Ca2+-activated K+ channels in intact human T lymphocytes: Noninvasive measurements of membrane currents, membrane potential, and intracellular calcium. J. Gen. Physiol. 1995, 105, 765–794. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Luhmann, H.J.; Kilb, W. Resonance properties of different neuronal populations in the immature mouse neocortex. Eur. J. Neurosci. 2012, 36, 2753–2762. [Google Scholar] [CrossRef] [PubMed]
- Nakaoka, Y.; Imaji, T.; Hara, M.; Hashimoto, N. Spontaneous fluctuation of the resting membrane potential in Paramecium: Amplification caused by intracellular Ca2+. J. Exp. Biol. 2009, 212, 270–276. [Google Scholar] [CrossRef][Green Version]
- Ambrogini, P.; Lattanzi, D.; Ciuffoli, S.; Betti, M.; Fanelli, M.; Cuppini, R. Physical exercise and environment exploration affect synaptogenesis in adult-generated neurons in the rat dentate gyrus: Possible role of BDNF. Brain Res. 2013, 1534, 1–12. [Google Scholar] [CrossRef]
- Ambrogini, P.; Minelli, A.; Lattanzi, D.; Ciuffoli, S.; Fanelli, M.; Cuppini, R. Synaptically-silent immature neurons show gaba and glutamate receptor-mediated currents in adult rat dentate gyrus. Arch. Ital. Biol. 2006, 144, 115–126. [Google Scholar]
- Wang, B.; Jaffe, D.B.; Brenner, R. Current understanding of iberiotoxin-resistant BK channels in the nervous system. Front. Physiol. 2014, 5, 382. [Google Scholar] [CrossRef]
- Vivas, O.; Moreno, C.M.; Santana, L.F.; Hille, B. Proximal clustering between BK and CaV1.3 channels promotes functional coupling and BK channel activation at low voltage. Elife 2017, 6, e20829. [Google Scholar] [CrossRef]
- LoTurco, J.J.; Owens, D.F.; Heath, M.J.S.; Davis, M.B.E.; Kriegstein, A.R. GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron 1995, 15, 1287–1298. [Google Scholar] [CrossRef]
- Deisseroth, K.; Singla, S.; Toda, H.; Monje, M.; Palmer, T.D.; Malenka, R.C. Excitation-neurogenesis coupling in adult neural stem/progenitor cells. Neuron 2004, 42, 535–552. [Google Scholar] [CrossRef]
- Heigele, S.; Sultan, S.; Toni, N.; Bischofberger, J. Bidirectional GABAergic control of action potential firing in newborn hippocampal granule cells. Nat. Neurosci. 2016, 19, 263–270. [Google Scholar] [CrossRef]
- Schmidt-Hieber, C.; Jonas, P.; Bischofberger, J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature 2004, 429, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sultan, S.; Heigele, S.; Schmidt-Salzmann, C.; Toni, N.; Bischofberger, J. Silent synapses generate sparse and orthogonal action potential firing in adult-born hippocampal granule cells. Elife 2017, 6, e23612. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Rothberg, B.S.; Brenner, R. Mechanism of β4 subunit modulation of BK channels. J. Gen. Physiol. 2006, 127, 449–465. [Google Scholar] [CrossRef] [PubMed]
- Contreras, G.F.; Neely, A.; Alvarez, O.; Gonzalez, C.; Latorre, R. Modulation of BK channel voltage gating by different auxiliary β subunits. Proc. Natl. Acad. Sci. USA 2012, 109, 18991–18996. [Google Scholar] [CrossRef] [PubMed]
- Brenner, R.; Chen, Q.H.; Vilaythong, A.; Toney, G.M.; Noebels, J.L.; Aldrich, R.W. BK channel β4 subunit reduces dentate gyrus excitability and protects against temporal lobe seizures. Nat. Neurosci. 2005, 8, 1752–1759. [Google Scholar] [CrossRef] [PubMed]
- Stocca, G.; Schmidt-Hieber, C.; Bischofberger, J. Differential dendritic Ca2+ signalling in young and mature hippocampal granule cells. J. Physiol. 2008, 586, 3795–3811. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guan, X.; Li, Q.; Meredith, A.L.; Pan, H.L.; Yan, J. Glutamate-activated BK channel complexes formed with NMDA receptors. Proc. Natl. Acad. Sci. USA 2018, 115, E9006–E9014. [Google Scholar] [CrossRef]
- Lohmann, C.; Wong, R.O. Regulation of dendritic growth and plasticity by local and global calcium dynamics. Cell Calcium 2005, 37, 403–409. [Google Scholar] [CrossRef]
- Bando, Y.; Irie, K.; Shimomura, T.; Umeshima, H.; Kushida, Y.; Kengaku, M.; Fujiyoshi, Y.; Hirano, T.; Tagawa, Y. Control of Spontaneous Ca2+ Transients Is Critical for Neuronal Maturation in the Developing Neocortex. Cereb. Cortex 2016, 26, 106–117. [Google Scholar] [CrossRef]
- Horigane, S.I.; Ozawa, Y.; Yamada, H.; Takemoto-Kimura, S. Calcium signalling: A key regulator of neuronal migration. J. Biochem. 2019, 165, 401–409. [Google Scholar] [CrossRef]
- Ambrogini, P.; Cuppini, R.; Cuppini, C.; Ciaroni, S.; Cecchini, T.; Ferri, P.; Sartini, S.; Del Grande, P. Spatial learning affects immature granule cell survival in adult rat dentate gyrus. Neurosci. Lett. 2000, 286, 21–24. [Google Scholar] [CrossRef]
- Pedroni, A.; Minh, D.D.; Mallamaci, A.; Cherubini, E. Electrophysiological characterization of granule cells in the dentate gyrus immediately after birth. Front. Cell. Neurosci. 2014, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Lattanzi, D.; Savelli, D.; Di Palma, M.; Sartini, S.; Eusebi, S.; Borroto-Escuela, D.O.; Cuppini, R.; Fuxe, K.; Ambrogini, P. Electrophysiological approach to GPCR–RTK interaction study in hippocampus of adult rats. Neuromethods 2018, 140, 71–90. [Google Scholar]
- Sartini, S.; Lattanzi, D.; Di Palma, M.; Savelli, D.; Eusebi, S.; Sestili, P.; Cuppini, R.; Ambrogini, P. Maternal Creatine Supplementation Positively Affects Male Rat Hippocampal Synaptic Plasticity in Adult Offspring. Nutrients 2019, 11, 2014. [Google Scholar] [CrossRef]
| FWHM (mv) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | Descriptive Statistics | RM-One-Way Anova | Tukey HSD | ||||||||||||
| n | BL | TREAT | REV | F(df) | p | BL vs. TREAT | BL vs. REV | TREAT vs. REV | |||||||
| Mean | SEM | Mean | SEM | Mean | SEM | Mean Diff. | p | Mean Diff. | p | Mean Diff. | p | ||||
| Nikel 200 µM | 7 | 3.8 | 0.5 | 2.4 | 0.4 | 3.6 | 0.5 | F(2,6) = 10.98 | 0.005 | 1.71 | 0.03 | 0.20 | ns | −1.51 | 0.02 |
| AP5 50 µM | 6 | 4.9 | 0.7 | 4.8 | 0.7 | 4.2 | 0.6 | F(2,5) = 0.79 | ns | 0.11 | ns | 0.66 | ns | 0.55 | ns |
| CNQX 10 µM | 5 | 4.8 | 0.9 | 4.7 | 0.9 | 4.2 | 0.8 | F(2,4) = 0.56 | ns | 0.08 | ns | 0.64 | ns | 0.56 | ns |
| Paxilline 10 µM | 8 | 2.5 | 0.1 | 1.8 | 0.1 | 2.7 | 0.2 | F(2,7) = 12.71 | 0.004 | 0.77 | 0.001 | −0.12 | ns | −0.9 | 0.01 |
| GABA 10 µM | 10 | 5.2 | 0.5 | 3.5 | 0.3 | 4.6 | 0.5 | F(2,9) = 4.01 | 0.049 | 1.68 | 0.02 | 0.54 | ns | −1.14 | ns |
| Muscimol 10 µM | 8 | 5.6 | 0.6 | 3.2 | 0.3 | 5.6 | 1.0 | F(2,7) = 4.97 | 0.04 | 2.36 | 0.007 | −0.03 | ns | −2.4 | ns |
| F/F0 | |||||||||||||||
| Nikel 200 µM | 10 | 1.01 | 0.01 | 0.91 | 0.02 | 0.94 | 0.03 | F(2,9) = 9.63 | 0.002 | 0.09 | 0.004 | 0.06 | ns | −0.03 | ns |
| AP5 50 µM | 7 | 1.01 | 0.01 | 1.01 | 0.02 | 1.03 | 0.03 | F(2,6) = 0.17 | ns | 0.00 | ns | −0.01 | ns | −0.01 | ns |
| CNQX 10 µM | 5 | 1.00 | 0.01 | 1.02 | 0.02 | 1.07 | 0.05 | F(2,4) = 0.83 | ns | −0.02 | ns | −0.06 | ns | −0.04 | ns |
| Paxilline 10 µM | 8 | 1.02 | 0.01 | 0.77 | 0.05 | 0.7 | 0.07 | F(2,7) = 17.02 | 0.001 | 0.24 | 0.008 | 0.31 | .007 | 0.07 | ns |
| GABA 10 µM | 6 | 0.99 | 0.00 | 0.92 | 0.02 | 1.00 | 0.00 | F(2,5) = 15.86 | 0.009 | 0.07 | 0.02 | −0.01 | ns | −0.07 | .02 |
| Muscimol 10 µM | 9 | 1.00 | 0.01 | 0.88 | 0.03 | 0.93 | 0.06 | F(2,8) = 4.42 | 0.04 | 0.14 | 0.01 | 0.06 | ns | −0.07 | ns |
| RMP (mv) | |||||||||||||||
| Nikel 200 µM | 8 | −55.8 | 4.9 | −55.4 | 5.3 | −55.8 | 5.3 | F(2,7) = 0.03 | ns | −0.31 | ns | 0.06 | ns | 0.37 | ns |
| AP5 50 µM | 6 | −52.3 | 2.6 | −52.0 | 2.5 | −55.2 | 3.2 | F(2,5) = 2.31 | ns | −0.33 | ns | 2.83 | ns | 3.16 | ns |
| CNQX 10 µM | 5 | −52.8 | 2.0 | −52.6 | 2.2 | −51.8 | 2.1 | F(2,4) = 2.31 | ns | −0.20 | ns | −1.00 | ns | −0.8 | ns |
| Paxilline 10 µM | 6 | −49.7 | 2.1 | −41.8 | 4.8 | −42.8 | 5.7 | F(2,5) = 4.20 | ns | −7.83 | ns | −6.83 | ns | 1.00 | ns |
| GABA 10 µM | 8 | −49.3 | 2.2 | −41.1 | 1.4 | −53.3 | 1.8 | F(2,7) = 19.56 | 0.0004 | −8.12 | 0.02 | 4.00 | ns | 12.13 | 0.0001 |
| Muscimol 10 µM | 8 | −50.1 | 2.5 | −41.0 | 1.3 | −52.5 | 1.7 | F(2,7) = 14.77 | 0.001 | −9.12 | 0.015 | 2.37 | ns | 11.5 | 0.0003 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lattanzi, D.; Di Palma, M.; Cuppini, R.; Ambrogini, P. GABAergic Input Affects Intracellular Calcium Levels in Developing Granule Cells of Adult Rat Hippocampus. Int. J. Mol. Sci. 2020, 21, 1715. https://doi.org/10.3390/ijms21051715
Lattanzi D, Di Palma M, Cuppini R, Ambrogini P. GABAergic Input Affects Intracellular Calcium Levels in Developing Granule Cells of Adult Rat Hippocampus. International Journal of Molecular Sciences. 2020; 21(5):1715. https://doi.org/10.3390/ijms21051715
Chicago/Turabian StyleLattanzi, Davide, Michael Di Palma, Riccardo Cuppini, and Patrizia Ambrogini. 2020. "GABAergic Input Affects Intracellular Calcium Levels in Developing Granule Cells of Adult Rat Hippocampus" International Journal of Molecular Sciences 21, no. 5: 1715. https://doi.org/10.3390/ijms21051715
APA StyleLattanzi, D., Di Palma, M., Cuppini, R., & Ambrogini, P. (2020). GABAergic Input Affects Intracellular Calcium Levels in Developing Granule Cells of Adult Rat Hippocampus. International Journal of Molecular Sciences, 21(5), 1715. https://doi.org/10.3390/ijms21051715





