The Impact of MNRI Therapy on the Levels of Neurotransmitters Associated with Inflammatory Processes
Abstract
1. Introduction
- A.
- Reliability and quality assurance are achieved using the CLIA certified labs.
- B.
- Since the 1960s, several studies depended upon biomarkers of neurotransmitters and metabolites [14].
- C.
2. Results
3. Discussion
3.1. Developmental Disorders
3.2. Anxiety/OCD (Obsessive Compulsive Disorder)/PTSD (Post-Traumatic Stress Disorder)
3.3. Palsy and Seizures
3.4. Attention Deficit Disorder/Attention Deficit Disorder Hyperactive Disorder (ADD/ADHD)
3.5. Autism Spectrum Disorders
4. Materials and Methods
4.1. MNRI Method
4.2. Study Populatio
Description of Study Groups
4.3. Laboratory Methods: Analysis of Urinary Catecholamine’s and Neurotransmitters
4.4. Statistical Analysis
4.5. The MNRI Reflex Integration Therapy Modality
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| MNRI | Masgutova Neurosensorimotor Reflex Neuromodulation Intervention |
| ADHD | Attention-Deficit/Hyperactivity Disorder |
| OCD | Obsessive-Compulsive Disorder |
| TAAR1 | The Trace Amine-Associated Receptor |
| MAO | Monoamine Oxidase A |
| PMNT | Phenylethanolamine N-Methyltransferase |
| CNS | Central Nervous System |
| SNS | Sympathetic Nervous System |
| PNS | Parasympathetic Nervous System |
| PEA | Palmitoylethanolamide |
| ASD | Autism Spectrum Disorder |
| HPA | Hypothalamus-Pituitary-Adrenal |
| ADD | Attention Deficit Disorder |
| NMDA | N-Methyl-D-Aspartate Receptor |
References
- Tripathy, K. Epigenetic and Therapeutic Analysis of various Neurological Disorders. J. Genet. Syndr. Gene Ther. 2011, 2. [Google Scholar] [CrossRef]
- Fischer, R.; Maier, O. Interrelation of Oxidative Stress and Inflammation in Neurodegenerative Disease: Role of TNF. Oxidative Med. Cell. Longev. 2015, 2015, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Z.; Wang, Y.-X.; Jiang, C.-L. Inflammation: The Common Pathway of Stress-Related Diseases. Front. Hum. Neurosci. 2017, 11, 316. [Google Scholar] [CrossRef] [PubMed]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How Do Glucocorticoids Influence Stress Responses? Integrating Permissive, Suppressive, Stimulatory, and Preparative Actions*. Endocr. Rev. 2000, 21, 55–89. [Google Scholar] [PubMed]
- Ziegler, C.; Wolf, C.; Schiele, M.A.; Bojic, E.F.; Kucukalic, S.; Dzananovic, E.S.; Uka, A.G.; Hoxha, B.; Haxhibeqiri, V.; Haxhibeqiri, S.; et al. Monoamine Oxidase A Gene Methylation and Its Role in Posttraumatic Stress Disorder: First Evidence from the South Eastern Europe (SEE)-PTSD Study. Int. J. Neuropsychopharmacol. 2017, 21, 423–432. [Google Scholar] [CrossRef]
- Nordahl, T.E.; Salo, R.; Natsuaki, Y.; Galloway, G.P.; Waters, C.; Moore, C.D.; Buonocore, M.H. Methamphetamine users in sustained abstinence: A proton magnetic resonance spectroscopy study. Arch. Gen. Psychiatry 2005, 62, 444–452. [Google Scholar] [CrossRef]
- Gold, M.S.; Blum, K.; Berman, M.O.; Braverman, E.R. Low dopamine function in attention deficit/hyperactivity disorder: Should genotyping signify early diagnosis in children? Postgrad. Med. 2014, 126, 153–177. [Google Scholar] [CrossRef]
- Novelli, A.; Reilly, J.; Lysko, P.; Henneberry, R. Glutamate becomes neurotoxic via the N-methyl-d-aspartate receptor when intracellular energy levels are reduced. Brain Res. 1988, 451, 205–212. [Google Scholar] [CrossRef]
- Schuch, V.; Utsumi, D.A.; Costa, T.V.M.M.; Kulikowski, L.D.; Muszkat, M. Attention Deficit Hyperactivity Disorder in the Light of the Epigenetic Paradigm. Front. Psychol. 2015, 6, 716. [Google Scholar] [CrossRef]
- Pinna, A. Novel investigational adenosine A2Areceptor antagonists for Parkinson’s disease. Expert Opin. Investig. Drugs 2009, 18, 1619–1631. [Google Scholar] [CrossRef]
- Buowari, O.Y. Complications of venepuncture. Adv. Biosci. Biotechnol. 2013, 4, 126–128. [Google Scholar] [CrossRef]
- Niemantsverdriet, E.; Struyfs, H.; Duits, F.; Teunissen, C.E.; Engelborghs, S. Techniques, Contraindications, and Complications of CSF Collection Procedures. In Cerebrospinal Fluid in Clinical Neurology; Springer Science and Business Media LLC: Berlin, Germany, 2015; pp. 35–57. [Google Scholar]
- Cavaleri, J.; Perez, J.L.; Ozpinar, A.; Alan, N.; Monaco, E., III. Epidural cerebrospinal fluid collection following lumbar puncture in an adult patient: A case report and literature review. Surg. Neurol. Int. 2018, 9, 169. [Google Scholar] [CrossRef]
- Giustarini, D.; Dalle-Donne, I.; Tsikas, D.; Rossi, R. Oxidative stress and human diseases: Origin, link, measurement, mechanisms, and biomarkers. Crit. Rev. Clin. Lab. Sci. 2009, 46, 241–281. [Google Scholar] [CrossRef] [PubMed]
- Gedal, K.B.; Goldstein, D.S. Catecholamines and their metabolites. J. Chromatogr. B: Biomed. Sci. Appl. 1988, 429, 177–233. [Google Scholar]
- Marc, D.T.; Ailts, J.W.; Campeau, D.C.; Bull, M.J.; Olson, K.L. Neurotransmitters excreted in the urine as biomarkers of nervous system activity: Validity and clinical applicability. Neurosci. Biobehav. Rev. 2010. [Google Scholar] [CrossRef]
- Panholzer, T.J.; Beyer, J.; Lichtwald, K. Coupled-Column Liquid Chromatographic Analysis of Catecholamines, Serotonin, and Metabolites in Human Urine. Clin. Chem. 1999, 45, 262–268. [Google Scholar] [CrossRef]
- Lynn-Bullock, C.P.; Welshhans, K.; Pallas, S.L.; Katz, P.S. The effect of oral 5-HTP administration on 5-HTP and 5-HT immunoreactivity in monoaminergic brain regions of rats. J. Chem. Neuroanat. 2004, 27, 129–138. [Google Scholar] [CrossRef]
- Westermann, B. Merging mitochondria matters: Cellular role and molecular machinery of mitochondrial fusion. EMBO Rep. 2002, 3. [Google Scholar] [CrossRef]
- Selye, H. Stress without Distress; J. B. Lippincott Co.: Philadelphia, PA, USA, 1974. [Google Scholar]
- Haines, D.E. Fundamental Neuroscience, 2nd ed.; The Curtis Center: Philadelphia, PA, USA, 2002. [Google Scholar]
- Virella, G.; Goust, J.M.; Fudenberg, H.H. Introduction to Medical Immunology, 2nd ed.; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Ziemssen, F.; Kern, S. Psychoneuroimmunology Cross-talk between the immune and nervous systems. J. Neurol. 2007, 254, II8–II11. [Google Scholar] [CrossRef]
- Olguin, H.J.; Guzmán, D.C.; García, E.H.; Mejía, G.B. The Role of Dopamine and Its Dysfunction as a Consequence of Oxidative Stress. Oxidative Med. Cell. Longev. 2016, 2016, 1–13. [Google Scholar] [CrossRef]
- Tank, A.W.; Wong, N.L. Peripheral and Central Effects of Circulating Catecholamines. Comprehensive Physiology 2014, 5, 1–15. [Google Scholar]
- Strac, D.S.; Pivac, N.; Smolders, I.; Fogel, W.A.; De Deurwaerdere, P.; Di Giovanni, G. Monoaminergic Mechanisms in Epilepsy May Offer Innovative Therapeutic Opportunity for Monoaminergic Multi-Target Drugs. Front. Mol. Neurosci. 2016, 10, 324. [Google Scholar] [CrossRef] [PubMed]
- Delahanty, D.; Nugent, N.R.; Christopher, N.C.; Walsh, M. Initial urinary epinephrine and cortisol levels predict acute PTSD symptoms in child trauma victims. Psychoneuroendocrinology 2005, 30, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Irsfeld, M.; Spadafore, M.; Prüß, B.M. β-phenylethylamine, a small molecule with a large impact. webmedcentral 2013, 4, 4409. [Google Scholar]
- Baker, G.; Bornstein, R.; Rouget, A.; Ashton, S.; Van Muyden, J.; Coutts, R. Phenylethylaminergic mechanisms in attention-deficit disorder. Boil. Psychiatry 1991, 29, 15–22. [Google Scholar] [CrossRef]
- Schaumann, T.; Kraus, D.; Winter, J.; Wolf, M.; Deschner, J.; Jäger, A. Potential Immune Modularly Role of Glycine in Oral Gingival Inflammation. Clin. Dev. Immunol. 2013, 2013, 1–9. [Google Scholar] [CrossRef]
- Knigge, U.; Warberg, J. The role of histamine in the neuroendocrine regulation of pituitary hormone secretion. Eur. J. Endocrinol. 1991, 124, 609–619. [Google Scholar] [CrossRef]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Kusaga, A. Decreased beta-phenylethylamine in urine of children with attention deficit hyperactivity disorder and autistic disorder. No Hattatsu Brain Dev. 2002, 34, 243–248. [Google Scholar] [CrossRef]
- Capuron, L.; Miller, A.H. Immune system to brain signaling: Neuropsychopharmacological implications. Pharmacol. Ther. 2011, 130, 226–238. [Google Scholar] [CrossRef]
- Cerebral Palsy. Available online: https://www.mayoclinic.org/diseases-conditions/cerebral-palsy/symptoms-causes/syc-20353999 (accessed on 17 August 2019).
- Autism Spectrum Disorder Fact Sheet. (8-29-19). Available online: https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Autism-Spectrum-Disorder-Fact-Sheet (accessed on 17 August 2019).
- Hall, J.E.; Guyton, A.C. Textbook of Medical Physiology, 12th ed.; Saunders Elsevier: Philadelphia, PA, USA, 2011. [Google Scholar]
- Myhrer, T. Neurotransmitter systems involved in learning and memory in the rat: A meta-analysis based on studies of four behavioral tasks. Brain Res. Rev. 2003, 41, 268–287. [Google Scholar] [CrossRef]
- Campbell, N.; Reece, J.; Mitchell, L. Biology, 5th ed.; Benjamin Cummings, Inc.: Menlo Park, CA, USA, 2000; p. 8. [Google Scholar]
- Frick, A.; Åhs, F.; Engman, J.; Jonasson, M.; Alaie, I.; Björkstrand, J.; Wahlstedt, K. Serotonin Synthesis and Reuptake in Social Anxiety Disorder: A Positron Emission Tomography Study. JAMA Psychiatry 2015, 72, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Jayamohananan, H.; Kumar, M.K.M. 5-HIAA as a Potential Biological Marker for Neurological and Psychiatric Disorders. Adv. Pharm. Bull. 2019, 9, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Bell, C.; Whitney, J.; Deiss, T.; Tatarinova, T.; Franckle, L.; Beaven, S.; Davis, J. The Effect of the MNRI Method on Neurotransmitter Biomarkers of Individuals with Neurodevelopmental Disorders. Neurosci. Med. 2019, 10, 292–321. [Google Scholar] [CrossRef][Green Version]
- Ortego, L.; Pelican, E.; Callaba, L.; Marks, T. An investigation of the effects of MNRI techniques on the educational performance of kindergarten students. In Reflexes: Portal to Neurodevelopment and Learning, A Collective Work; Svetlana Masgutova Educational Institute: Orlando, FL, USA, 2015. [Google Scholar]
- Zhong, Z.; Wheeler, M.D.; Li, X.; Froh, M.; Schemmer, P.; Yin, M.; Bunzendaul, H.; Bradford, B.; Lemasters, J.J. L-Glycine: A novel antiinflammatory, immunomodulatory, and cytoprotective agent. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 229–240. [Google Scholar] [CrossRef]
- Reyes-Garcia, M.G.; Hernandez-Hernandez, F.; Hernandez-Tellez, B.; Garcia-Tamayo, F. GABA (A) receptor subunits RNA expression in mice peritoneal macrophages modulate their IL-6/IL-12 production. J. Neuroimmunol. 2007, 188, 64–68. [Google Scholar] [CrossRef]
- Garlick, P.J. The nature of human hazards associated with excessive intake of amino acids. J. Nutr. 2004, 134, 1633S–1639S. [Google Scholar] [CrossRef]
- Cowan, L.D. The epidemiology of the epilepsies in children. Ment. Retard. Dev. Disabil. Res. Rev. 2002, 8, 171–181. [Google Scholar] [CrossRef]
- Bonhaus, D.W.; Yeh, G.C.; Skaryak, L.; McNamara, J.O. Glycine regulation of the N-methyl-D-aspartate receptor-gated ion channel in hippocampal membranes. Mol. Pharmacol. 1989, 36, 273–279. [Google Scholar]
- Wu, C.; Sun, D. GABA receptors in brain development, function, and injury. Metab. Brain Dis. 2015, 30, 367–379. [Google Scholar] [CrossRef]
- GABA and Glycine. Available online: https://www.acnp.org/g4/GN401000008/Default.htm (accessed on 9 October 2019).
- Anand, D.; Colpo, G.D.; Zeni, G.; Zeni, C.P.; Teixeira, A.L. Attention-Deficit/Hyperactivity Disorder and Inflammation: What Does Current Knowledge Tell Us? A Systematic Review. Front. Psychiatry 2017, 8, 228. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Lee, S.-K.; Park, M.; Cho, I.H.; Hyun, S.H.; Lee, J.C.; Yang, S.Y.; Kim, S.A. Family- and population-based association studies of monoamine oxidase A and autism spectrum disorders in Korean. Neurosci. Res. 2009, 63, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Gu, F.; Chauhan, V.; Chauhan, A. Monoamine oxidase-A and B activities in the cerebellum and frontal cortex of children and young adults with autism. J. Neurosci. Res. 2017, 95, 1965–1972. [Google Scholar] [CrossRef] [PubMed]
- Antonucci, N.; Cirillo, A.; Siniscalco, D. Beneficial Effects of Palmitoylethanolamide on Expressive Language, Cognition, and Behaviors in Autism: A Report of Two Cases. Case Rep. Psychiatry 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Masgutova, S. Use of a Neurosensorimotor Reflex Integration Program to Improve Reflex Patterns of Children with Down Syndrome. J. Neurol. Neurosci. 2015, 6, 59. [Google Scholar] [CrossRef]
- Masgutova, S.; Akhmatova, N.; Sadowska, L.; Shackleford, P.; Akhmatov, E. Progress with neurosensorimotor reflex integration for children with Autism Spectrum Disorder. J. Neurol. Psychol. 2016, 4, 14. [Google Scholar]
- Akhmatova, N.; Akhmatova, E. Influence of MNRI on the immune status of children with Down syndrome. J. Clin. Cell. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Pilecki, W.; Kipiński, L.; Szawrowicz-Pełka, T.; Kałka, D.; Masgutova, S. Spectral brain mapping in children with cerebral palsy treated by the Masgutova Neurosensorimotor Reflex Integration method. J. Neurol. Sci. 2013, 333, 550. [Google Scholar] [CrossRef]
- Pilecki, W.; Masgutova, S.; Kowalewska, J.; Masgutov, D.; Akhmatova, N.; Poreba, M.; Sobieszczańska, M.; Koleda, P.; Pilecka, A.; Kałka, D. The impact of rehabilitation carried out using the Masgutova Neurosensorimotor Reflex Integration method in children with cerebral palsy on the results of brain stem auditory potential examinations. Adv. Clin. Exp. Med. 2012, 21, 363–371. [Google Scholar]
- Koberda, J.L.; Akhmatova, N. Masgutova Neurosensorimotor Reflex Integration (MNRI) as a new form of manual neuromodulation technique. J. Neurol. Neurobiol. 2016, 2. [Google Scholar] [CrossRef]
- Koberda, J.L.; Akhmatova, N.; Akhmatova, E.; Bienkiewicz, A.; Nowak, K.; Nawrocka, H. Masgutova Neurosensorimotor Reflex Integration (MNRI) neuromodulation technique induces positive brain maps (QEEG) changes. J. Neurol. Neurobiol. 2016, 2. [Google Scholar] [CrossRef]
- Rentschler, M.; Averkamp, S. (Eds.) Reflexes: Portal to Neurodevelopment and Learning; Svetlana Masgutova Educational Inst.: Orlando, FL, USA, 2015. [Google Scholar]
- Yuan, R.; Di, X.; Taylor, P.A.; Gohel, S.; Tsai, Y.H.; Biswal, B.B. Functional topography of the thalamocortical system in human. Brain Struct. Funct. 2016, 221, 1971. [Google Scholar] [CrossRef] [PubMed]
- Sechenov, I.M. Reflexes of the Brain; The M.I.T. Press: Cambridge, MA, USA, 1965. [Google Scholar]
- Sechenov, I.M. Physiology of Behavior. Scientific Works; Yaroshevsky, M.G., Ed.; Publisher Izd-vo “In-t Prakticheskoĭ Psikhologii”: Moscow, Russia, 1995. [Google Scholar]
- Pavlov, I.P. Conditioned Reflexes: An Investigation of the Physiological Activity of the Cerebral Cortex; Dover Publications Inc.: New York, NY, USA, 1925. [Google Scholar]
- Anokhin, P.K. Biology and Neurophysiology of the Conditioned Reflex and Its Role in Adaptive Behavior; Corson, S.A., Ed.; Pergamon Press: Oxford, UK, 1974. [Google Scholar]
- Akhmatova, N.; Akhmatova, E.; Lebedinskaya, O. MNRI: An immunomodulating effect on lymphocyte subpopulation structure of children with Down syndrome. Russian J. Immun. 2016, 10, 485–487. [Google Scholar]
- Akhmatova, E.; Akhmatova, N.; Masgutova, S. MNRI: Immunocorrective effect on the immune status of children with Down syndrome. In Proceedings of the 17th Biennial Meeting of the European Society for Immunodeficiencies, Barcelona, Spain, 21–24 September 2016. [Google Scholar]
- Hoover-Fong, J.E.; Shah, S.; Van Hove JL, K.; Applegarth, D.; Toone, J.; Hamosh, A. Natural history of nonketotic hyperglycinemia in 65 patients. Neurology 2004, 63, 1847–1853. [Google Scholar] [CrossRef]
- Masgutova, S. Post-Trauma Recovery in Children of Newtown, CT using MNRI Reflex Integration. J. Trauma. Stress Disord. Treat. 2016, 5. [Google Scholar] [CrossRef]
- Masgutova, S.; Regner, A. Language Development Using Sensory-Motor Integration Approach; Continuo MISM: Wroclaw, Poland, 2008; p. 167. [Google Scholar]
- Akhmatova, N.K.; Masgutova, S.; Shubina, I.Z.; Akhmatov, E.A.; Khomenkov, V.V.; Sorokina, E.V.; Korovkina, E.S.; Kostinov, M.P. Immunological Effects of Masgutova Neurosensorimotor Reflex Integration in Children with Recurrent Obstructive Bronchitis. Int. J. Neurorehabilit. 2015, 2, 1–9. [Google Scholar]
- Renard-Fontaine, I. Effect of Reflex Neuromodulation on an Infant with Severe Amniotic Band Syndrome: A Case Report on the Use of MNRI Techniques for Physical Therapy. Int. J. Neurorehabilit. 2017, 4, 248. [Google Scholar]
- Peacock, B.N.; Scheiderer, D.J.; Kellermann, G.H. Biomolecular aspects of depression: A retrospective analysis. Compr. Psychiatry 2017, 73, 168–180. [Google Scholar] [CrossRef]
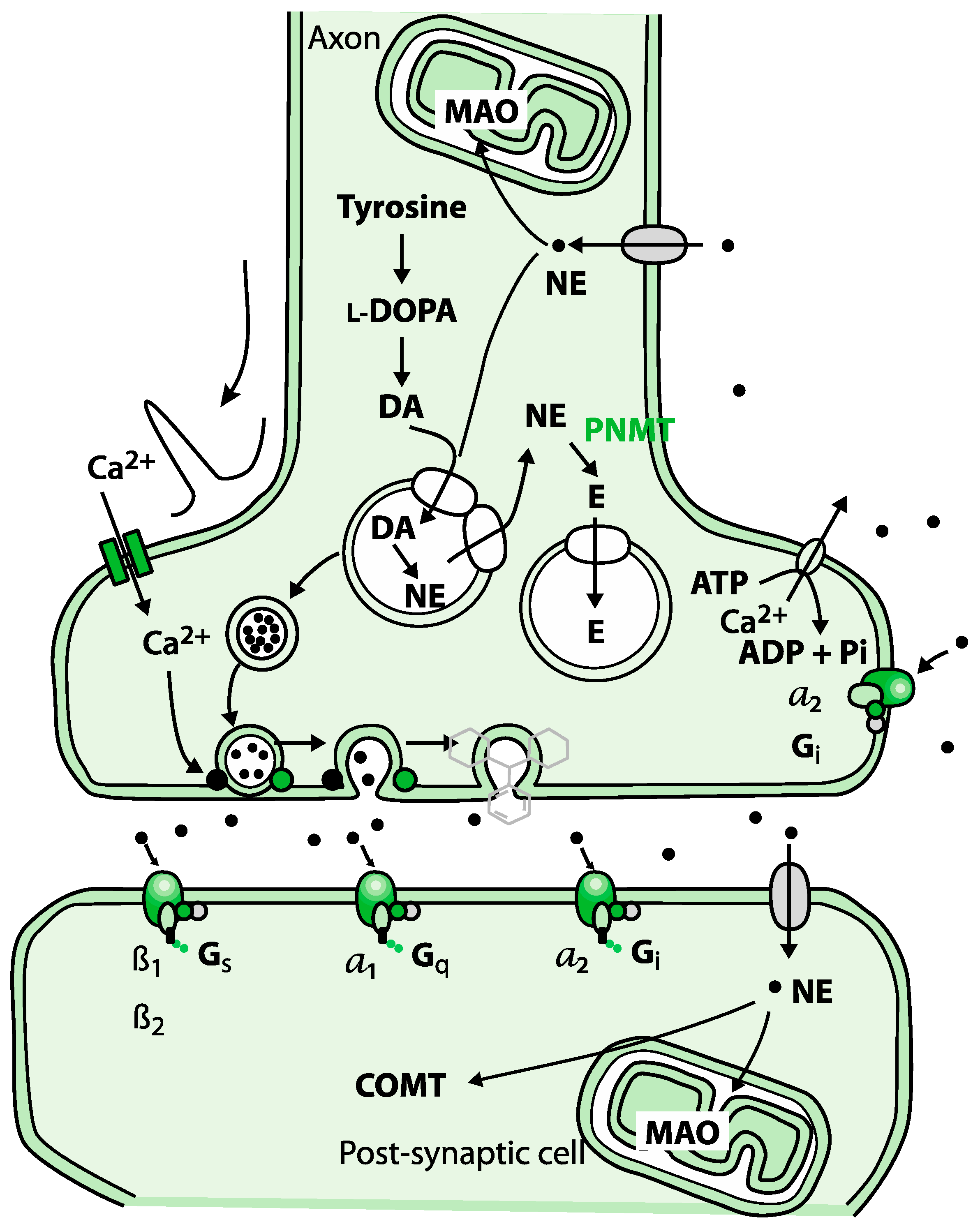
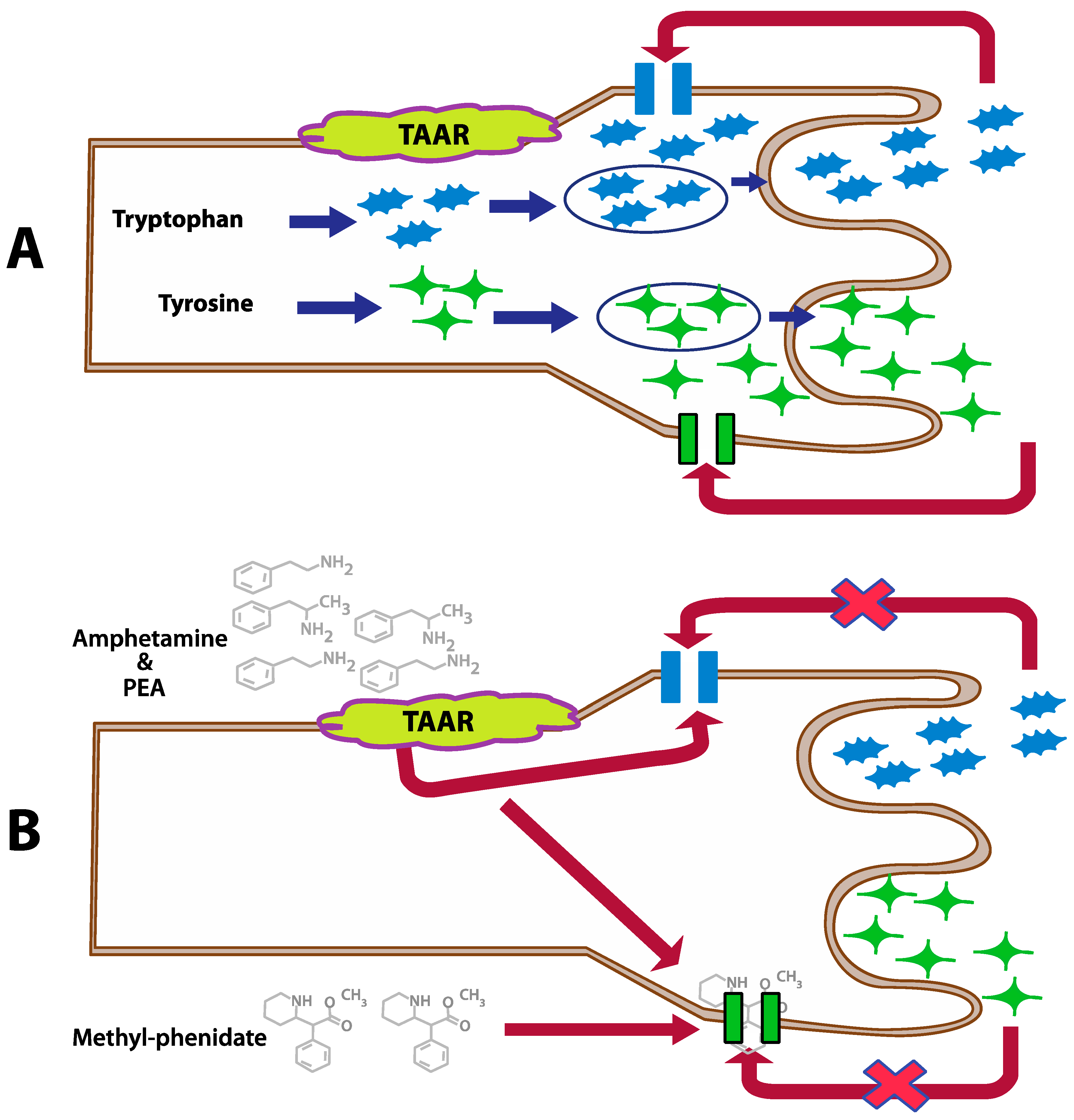
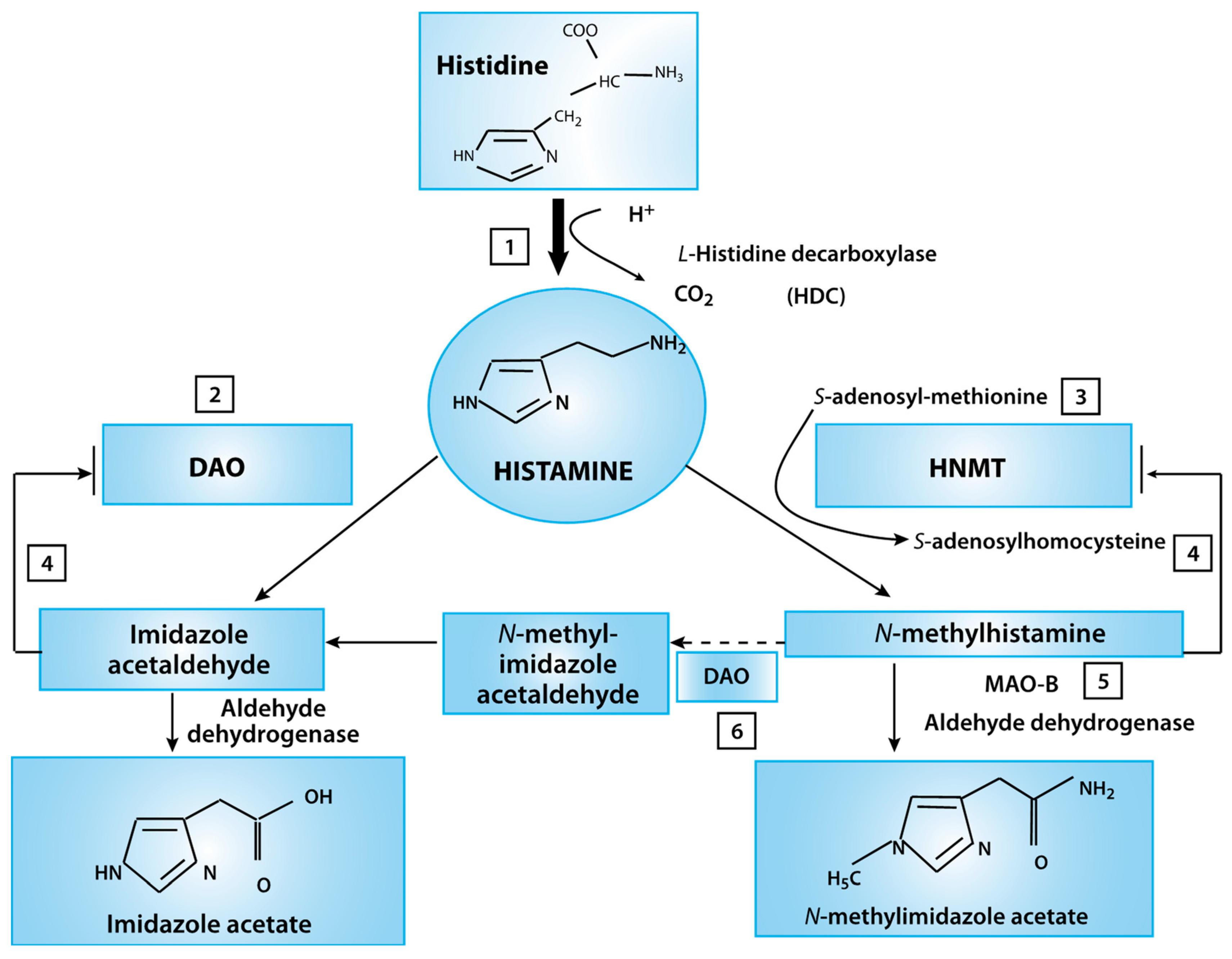
| Developmental Disorders (n = 34) | Epinephrine (ug/g Cr) | Norepinephrine (ug/g Cr) | Glutamate (umol/g Cr) | Glycine (umol/g Cr) | bPEA (nmol/g Cr) | Histamine (ug/g Cr) | GABA (umol/g Cr) | DOPAC (umol/g Cr) | 5-HIAA (ug/g Cr) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Controls | Mean (μ) | 1.80 | 31.8 | 21.5 | 1004.8 | 40.7 | 24.7 | 4.21 | 468.9 | 2460.1 |
| SD | 0.75 | 13.6 | 10.1 | 552.1 | 19.1 | 9.7 | 1.15 | 131.1 | 1434.8 | |
| Pre-Tx | Mean (μ) | 4.64 | 62.3 | 70.9 | 1536.2 | 57.4 | 44.1 | 9.40 | 997.7 | 6271.0 |
| SD | 2.91 | 27.7 | 49.9 | 558.7 | 18.3 | 20.1 | 3.43 | 744.7 | 3274.9 | |
| Post-Tx | Mean (μ) | 3.46 | 62.2 | 65.7 | 1398.6 | 51.7 | 41.9 | 8.95 | 1003.5 | 6181.5 |
| SD | 1.76 | 29.2 | 43.5 | 600.9 | 22.4 | 23.0 | 3.41 | 491.6 | 2788.6 | |
| Diffe-rence (%) | Controls vs. PreTx | 158% | 96% | 229% | 53% | 41% | 79% | 123% | 113% | 155% |
| Controls vs. PostTx | 92% | 95% | 205% | 39% | 27% | 70% | 113% | 114% | 151% | |
| PreTx vs. PostTx | −25% | 0% | −7% | −9% | −10% | −5% | −5% | 1% | −1% | |
| Statis-tical Signi-ficance | Controls vs. PreTx | *** | *** | *** | ** | ** | *** | *** | *** | *** |
| Controls vs. PostTx | *** | *** | *** | * | T | *** | *** | *** | *** | |
| PreTx vs. PostTx | * | ns | ns | ns | ns | ns | ns | T | ns | |
| Effect Size | Controls vs. PreTx | VL | VL | VL | Lg | Lg | VL | VL | Lg | VL |
| Controls vs. PostTx | VL | VL | VL | Md | Md | Lg | VL | VL | VL | |
| PreTx vs. PostTx | Sm | NoEf | NoEf | Sm | Sm | NoEf | NoEf | NoEf | NoEf | |
| Anxiety/OCD/ PTSD (n = 20) | Epinephrine (ug/g Cr) | Norepinephrine (ug/g Cr) | Glutamate (umol/g Cr) | Glycine (umol/g Cr) | bPEA (nmol/g Cr) | Histamine (ug/g Cr) | GABA (umol/g Cr) | DOPAC (umol/g Cr) | 5-HIAA (ug/g Cr) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Controls | Mean (μ) | 1.80 | 31.8 | 21.5 | 1004.8 | 40.7 | 24.7 | 4.21 | 468.9 | 2460.1 |
| SD | 0.75 | 13.6 | 10.1 | 552.1 | 19.1 | 9.7 | 1.15 | 131.1 | 1434.8 | |
| Pre-Tx | Mean (μ) | 3.05 | 50.5 | 65.3 | 1474.3 | 62.2 | 42.4 | 9.32 | 714.8 | 6385.2 |
| SD | 1.41 | 20.5 | 42.8 | 404.6 | 33.1 | 21.4 | 4.01 | 233.6 | 3368.1 | |
| Post-Tx | Mean (μ) | 2.98 | 50.3 | 53.2 | 1367.1 | 55.8 | 41.0 | 8.40 | 796.5 | 6195.0 |
| SD | 1.29 | 27.5 | 30.3 | 442.9 | 26.0 | 21.0 | 3.63 | 242.2 | 2886.2 | |
| Difference (%) | Controls vs. PreTx | 70% | 59% | 203% | 47% | 53% | 72% | 121% | 52% | 160% |
| Controls vs. PostTx | 66% | 58% | 147% | 36% | 37% | 66% | 99% | 70% | 152% | |
| PreTx vs. PostTx | −2% | 0% | −19% | −7% | −10% | −3% | −10% | 11% | −3% | |
| Statistical Signi-ficance | Controls vs. PreTx | *** | *** | *** | * | * | *** | *** | *** | *** |
| Controls vs. PostTx | *** | ** | *** | T | * | *** | *** | *** | *** | |
| PreTx vs. PostTx | ns | ns | ns | ns | ns | ns | ns | ns | ns | |
| Effect Size | Controls vs. PreTx | Lg | Lg | VL | Lg | Md | Lg | VL | VL | VL |
| Controls vs. PostTx | Lg | Lg | VL | Md | Md | Lg | VL | VL | VL | |
| PreTx vs. PostTx | NoEf | NoEf | Sm | Sm | Sm | NoEf | Sm | Sm | NoEf | |
| Palsy & Seizures (n = 16) | Epinephrine (ug/g Cr) | Norepinephrine (ug/g Cr) | Glutamate (umol/g Cr) | Glycine (umol/g Cr) | bPEA (nmol/g Cr) | Histamine (ug/g Cr) | GABA (umol/g Cr) | DOPAC (umol/g Cr) | 5-HIAA (ug/g Cr) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Controls | Mean (μ) | 1.80 | 31.8 | 21.5 | 1004.8 | 40.7 | 24.7 | 4.21 | 468.9 | 2460.1 |
| SD | 0.75 | 13.6 | 10.1 | 552.1 | 19.1 | 9.7 | 1.15 | 131.1 | 1434.8 | |
| Pre-Tx | Mean (μ) | 4.21 | 59.0 | 101.9 | 3136.9 | 76.2 | 52.9 | 15.01 | 921.9 | 10119.4 |
| SD | 2.61 | 20.6 | 49.2 | 1681.4 | 36.2 | 20.7 | 7.29 | 544.5 | 5422.2 | |
| Post-Tx | Mean (μ) | 3.45 | 66.6 | 103.9 | 2734.5 | 80.5 | 52.8 | 13.48 | 878.0 | 10331.0 |
| SD | 1.70 | 24.7 | 41.8 | 1312.0 | 39.3 | 25.8 | 6.96 | 379.7 | 6384.1 | |
| Difference (%) | Controls vs. PreTx | 134% | 85% | 373% | 212% | 87% | 114% | 257% | 97% | 311% |
| Controls vs. PostTx | 92% | 109% | 382% | 172% | 98% | 114% | 220% | 87% | 320% | |
| PreTx vs. PostTx | −18% | 13% | 2% | −13% | 6% | 0% | −10% | −5% | 2% | |
| Statis-tical Signi-ficance | Controls vs. PreTx | *** | *** | *** | *** | *** | *** | *** | *** | *** |
| Controls vs. PostTx | *** | *** | *** | *** | *** | *** | *** | *** | *** | |
| PreTx vs. PostTx | ns | ns | ns | ns | ns | ns | ns | ns | ns | |
| Effect Size | Controls vs. PreTx | VL | VL | Hg | VL | VL | VL | Hg | Lg | VL |
| Controls vs. PostTx | VL | VL | Hg | VL | VL | VL | VL | VL | VL | |
| PreTx vs. PostTx | Sm | Sm | NoEf | Sm | NoEf | NoEf | Sm | NoEf | NoEf | |
| ADD/ADHD (n = 24) | Epinephrine (ug/g Cr) | Norepinephrine (ug/g Cr) | Glutamate (umol/g Cr) | Glycine (umol/g Cr) | bPEA (nmol/g Cr) | Histamine (ug/g Cr) | GABA (umol/g Cr) | DOPAC (umol/g Cr) | 5-HIAA (ug/g Cr) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Controls | Mean (μ) | 1.80 | 31.8 | 21.5 | 1004.8 | 40.7 | 24.7 | 4.21 | 468.9 | 2460.1 |
| SD | 0.75 | 13.6 | 10.1 | 552.1 | 19.1 | 9.7 | 1.15 | 131.1 | 1434.8 | |
| Pre-Tx | Mean (μ) | 3.04 | 60.6 | 74.6 | 1541.7 | 68.7 | 49.5 | 8.88 | 678.1 | 5527.5 |
| SD | 1.47 | 28.4 | 50.2 | 582.0 | 32.9 | 22.5 | 3.53 | 211.5 | 2569.4 | |
| Post-Tx | Mean (μ) | 2.87 | 57.7 | 55.3 | 1332.4 | 54.6 | 39.3 | 7.71 | 845.0 | 5327.7 |
| SD | 1.28 | 30.8 | 33.4 | 433.0 | 25.0 | 19.6 | 3.12 | 307.6 | 2020.1 | |
| Diffe-rence (%) | Controls vs. PreTx | 69% | 90% | 246% | 53% | 69% | 101% | 111% | 45% | 125% |
| Controls vs. PostTx | 59% | 81% | 157% | 33% | 34% | 59% | 83% | 80% | 117% | |
| PreTx vs. PostTx | −6% | −5% | −26% | −14% | −21% | −21% | −13% | 25% | −4% | |
| Statis-tical Significance | Controls vs. PreTx | *** | *** | *** | ** | *** | *** | *** | *** | *** |
| Controls vs. PostTx | *** | *** | *** | T | * | *** | *** | *** | *** | |
| PreTx vs. PostTx | ns | ns | * | ns | * | * | T | ** | ns | |
| Effect Size | Controls vs. PreTx | Lg | VL | VL | Lg | Lg | VL | VL | Lg | VL |
| Controls vs. PostTx | Lg | Lg | VL | Md | Md | Lg | VL | VL | VL | |
| PreTx vs. PostTx | NoEf | NoEf | Sm | Sm | Sm | Sm | Sm | Md | NoEf | |
| Autism Spectrum Disorder (n = 22) | Epinephrine (ug/g Cr) | Norepinephrine (ug/g Cr) | Glutamate (umol/g Cr) | Glycine (umol/g Cr) | bPEA (nmol/g Cr) | Histamine (ug/g Cr) | GABA (umol/g Cr) | DOPAC (umol/g Cr) | 5-HIAA (ug/g Cr) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Controls | Mean (μ) | 1.80 | 31.8 | 21.5 | 1004.8 | 40.7 | 24.7 | 4.21 | 468.9 | 2460.1 |
| SD | 0.75 | 13.6 | 10.1 | 552.1 | 19.1 | 9.7 | 1.15 | 131.1 | 1434.8 | |
| Pre-Tx | Mean (μ) | 6.64 | 55.6 | 55.5 | 1640.1 | 71.7 | 55.5 | 9.13 | 730.5 | 7142.1 |
| SD | 6.08 | 26.4 | 34.2 | 585.4 | 34.1 | 32.3 | 3.03 | 339.7 | 3173.6 | |
| Post-Tx | Mean (μ) | 6.57 | 46.2 | 54.7 | 1584.6 | 60.6 | 48.4 | 7.69 | 750.6 | 7240.0 |
| SD | 6.20 | 24.0 | 35.8 | 741.9 | 35.1 | 32.4 | 2.29 | 296.7 | 2763.0 | |
| Diffe-rence (%) | Controls vs. PreTx | 269% | 75% | 158% | 63% | 76% | 125% | 117% | 56% | 190% |
| Controls vs. PostTx | 265% | 45% | 154% | 58% | 49% | 96% | 83% | 60% | 194% | |
| PreTx vs. PostTx | −1% | −17% | −2% | −3% | −16% | −13% | −16% | 3% | 1% | |
| Statis-tical Signi-ficance | Controls vs. PreTx | *** | *** | *** | *** | *** | *** | *** | *** | *** |
| Controls vs. PostTx | *** | ** | *** | ** | ** | *** | *** | *** | *** | |
| PreTx vs. PostTx | ns | * | ns | ns | T | ns | * | ns | ns | |
| Effect Size | Controls vs. PreTx | Lg | Lg | VL | Lg | Lg | VL | Hg | Lg | VL |
| Controls vs. PostTx | Lg | Md | VL | Lg | Md | Lg | VL | VL | Hg | |
| PreTx vs. PostTx | NoEf | Sm | NoEf | NoEf | Sm | Sm | Md | NoEf | NoEf | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tatarinova, T.V.; Deiss, T.; Franckle, L.; Beaven, S.; Davis, J. The Impact of MNRI Therapy on the Levels of Neurotransmitters Associated with Inflammatory Processes. Int. J. Mol. Sci. 2020, 21, 1358. https://doi.org/10.3390/ijms21041358
Tatarinova TV, Deiss T, Franckle L, Beaven S, Davis J. The Impact of MNRI Therapy on the Levels of Neurotransmitters Associated with Inflammatory Processes. International Journal of Molecular Sciences. 2020; 21(4):1358. https://doi.org/10.3390/ijms21041358
Chicago/Turabian StyleTatarinova, Tatiana V., Trina Deiss, Lorri Franckle, Susan Beaven, and Jeffrey Davis. 2020. "The Impact of MNRI Therapy on the Levels of Neurotransmitters Associated with Inflammatory Processes" International Journal of Molecular Sciences 21, no. 4: 1358. https://doi.org/10.3390/ijms21041358
APA StyleTatarinova, T. V., Deiss, T., Franckle, L., Beaven, S., & Davis, J. (2020). The Impact of MNRI Therapy on the Levels of Neurotransmitters Associated with Inflammatory Processes. International Journal of Molecular Sciences, 21(4), 1358. https://doi.org/10.3390/ijms21041358






