A Review of the Evidence for and against a Role for Mast Cells in Cutaneous Scarring and Fibrosis
Abstract
1. Introduction
2. Mast Cells in Scarless and Fibrotic Fetal Skin Wounds
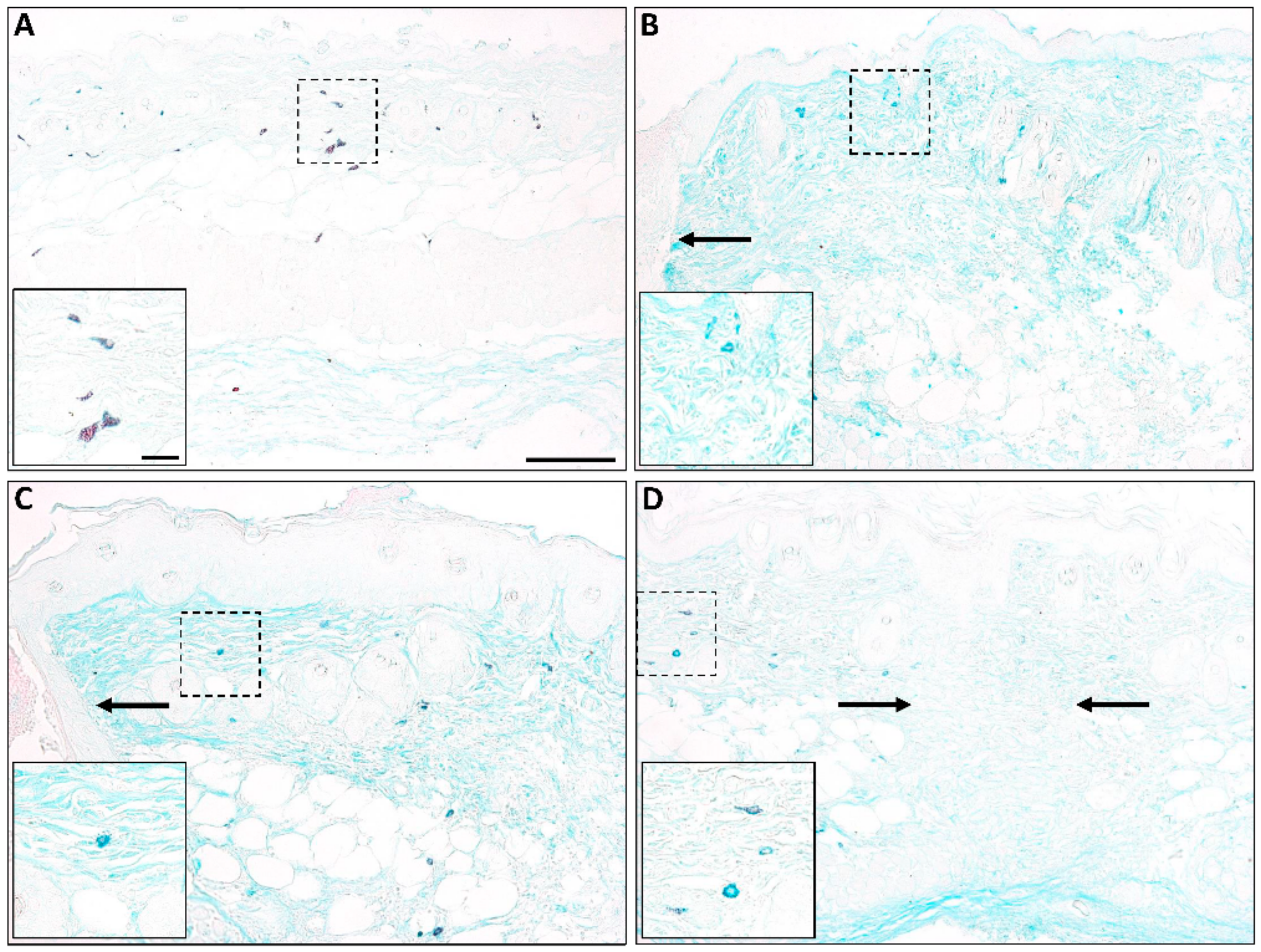
3. Mast Cells in Normal Scarring
4. Mast Cells in Abnormal Scarring
4.1. Hypertrophic Scars
4.2. Keloids
5. Mast Cells in Cutaneous Fibrosis
5.1. Systemic Sclerosis
5.2. Mouse Models of Scleroderma
5.3. Graft-Versus-Host Disease
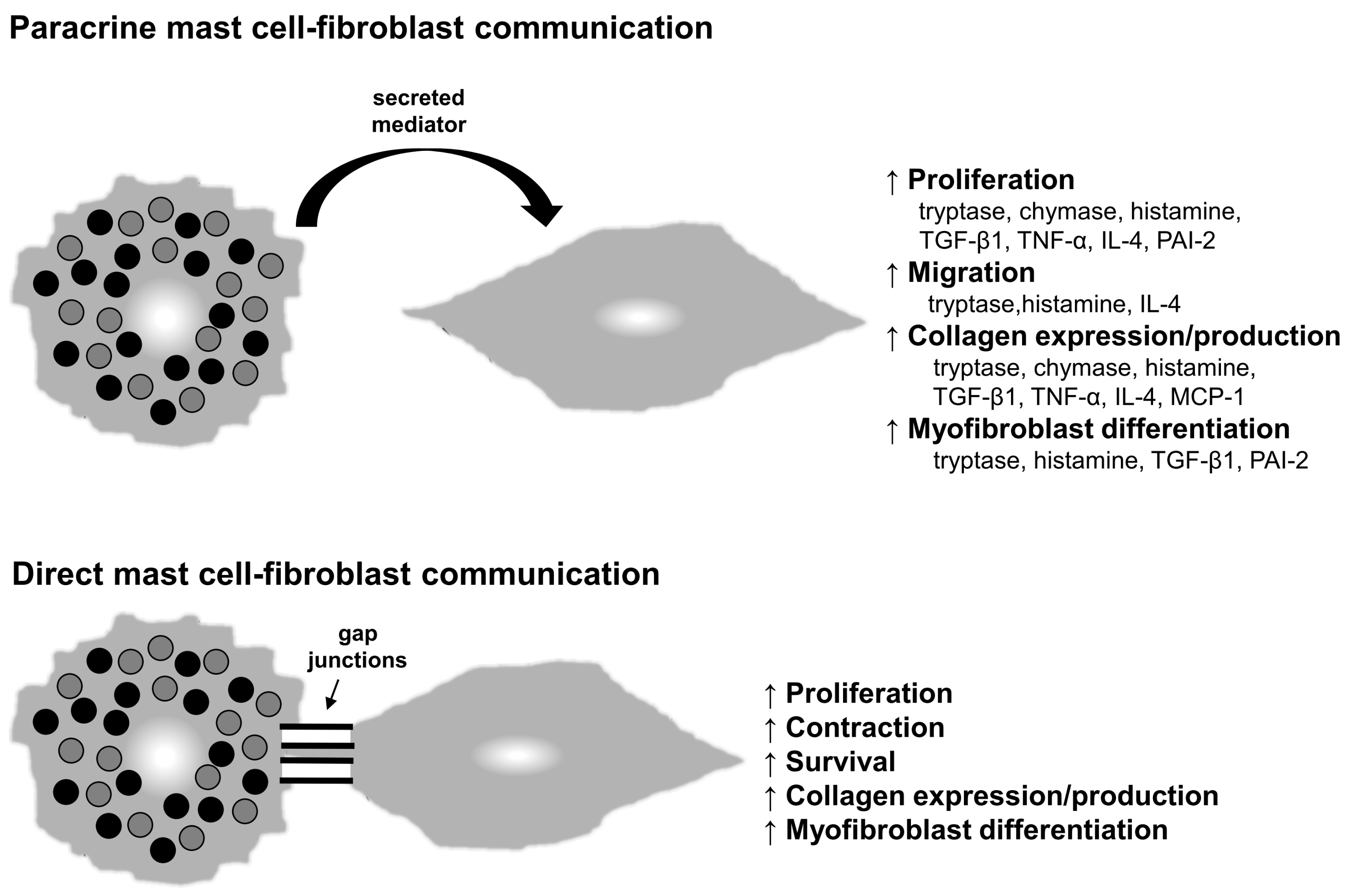
6. Mechanistic Insights: Mast Cell–Fibroblast Interactions
6.1. Paracrine Communication
6.2. Direct Cell–Cell Communication
7. Mast Cells in Fibrosis: Unanswered Questions and Future Directions
7.1. Experimental Limitations—Mast Cell Stabilizers
7.2. Experimental Limitations—Mouse Models
7.3. Mast Cell Heterogeneity
7.4. Mast Cells in Human Skin
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CCL | CC chemokine ligand |
| Cpa3 | carboxypeptidase 3 |
| CXCL | CXC chemokine ligand |
| EGCG | epigallocatechin-3-gallate |
| FGF | fibroblast growth factor |
| GVHD | graft-versus-host disease |
| HTS | hypertrophic scar |
| iDTR | inducible diphtheria toxin receptor |
| IL | interleukin |
| LT | leukotriene |
| Mcl | myeloid leukemia cell differentiation protein |
| MCP-1 | monocyte chemoattractant protein-1 (also known as CCL2) |
| Mcpt | mast cell protease |
| mMCP | murine mast cell protease |
| PAF | platelet activating factor |
| PAI | plasminogen activator inhibitor |
| PDGF | platelet derived growth factor |
| PG | prostaglandin |
| SCF | stem cell factor |
| SMA | smooth muscle actin |
| SSc | systemic sclerosis |
| TGF | transforming growth factor |
| TNF | tumor necrosis factor |
| Tsk | tight skin |
| VEGF | vascular endothelial growth factor |
References
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef]
- Martin, P. Wound healing—Aiming for perfect skin regeneration. Science 1997, 276, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Corr, D.T.; Gallant-Behm, C.L.; Shrive, N.G.; Hart, D.A. Biomechanical behavior of scar tissue and uninjured skin in a porcine model. Wound Repair Regen. 2009, 17, 250–259. [Google Scholar] [CrossRef]
- Dunn, M.G.; Silver, F.H.; Swann, D.A. Mechanical analysis of hypertrophic scar tissue: Structural basis for apparent increased rigidity. J. Investig. Dermatol. 1985, 84, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.C.; McKenna, S.P.; Siddhi, K.; McGrouther, D.A.; Bayat, A. The hidden cost of skin scars: Quality of life after skin scarring. J. Plast. Reconstr. Aesthet. Surg. 2008, 61, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Wilgus, T.A. Inflammation as an orchestrator of cutaneous scar formation: A review of the literature. Plast. Aesthet. Res. 2020, 7, 54. [Google Scholar] [CrossRef]
- Dong, J.; Chen, L.; Zhang, Y.; Jayaswal, N.; Mezghani, I.; Zhang, W.; Veves, A. Mast Cells in Diabetes and Diabetic Wound Healing. Adv. Ther. 2020, 37, 4519–4537. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Metz, M.; Starkl, P.; Marichal, T.; Tsai, M. Mast cells and IgE in defense against lethality of venoms: Possible “benefit” of allergy. Allergo J. Int. 2020, 29, 46–62. [Google Scholar] [CrossRef] [PubMed]
- Dudeck, A.; Koberle, M.; Goldmann, O.; Meyer, N.; Dudeck, J.; Lemmens, S.; Rohde, M.; Roldan, N.G.; Dietze-Schwonberg, K.; Orinska, Z.; et al. Mast cells as protectors of health. J. Allergy Clin. Immunol. 2019, 144, S4–S18. [Google Scholar] [CrossRef]
- Varricchi, G.; de Paulis, A.; Marone, G.; Galli, S.J. Future Needs in Mast Cell Biology. Int. J. Mol. Sci. 2019, 20, 4397. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, A.M.; Kissell, S. Granule changes of human skin mast cells characteristic of piecemeal degranulation and associated with recovery during wound healing in situ. J. Leukoc. Biol. 1991, 49, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Lateef, Z.; Stuart, G.; Jones, N.; Mercer, A.; Fleming, S.; Wise, L. The Cutaneous Inflammatory Response to Thermal Burn Injury in a Murine Model. Int. J. Mol. Sci. 2019, 20, 538. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Kitamura, Y. Migration of stromal cells supporting mast-cell differentiation into open wound produced in the skin of mice. Exp. Hematol. 1981, 9, 38–43. [Google Scholar]
- Trautmann, A.; Toksoy, A.; Engelhardt, E.; Brocker, E.B.; Gillitzer, R. Mast cell involvement in normal human skin wound healing: Expression of monocyte chemoattractant protein-1 is correlated with recruitment of mast cells which synthesize interleukin-4 in vivo. J. Pathol. 2000, 190, 100–106. [Google Scholar] [CrossRef]
- Nishikori, Y.; Kakizoe, E.; Kobayashi, Y.; Shimoura, K.; Okunishi, H.; Dekio, S. Skin mast cell promotion of matrix remodeling in burn wound healing in mice: Relevance of chymase. Arch. Dermatol. Res. 1998, 290, 553–560. [Google Scholar] [CrossRef]
- Ud-Din, S.; Foden, P.; Mazhari, M.; Al-Habba, S.; Baguneid, M.; Bulfone-Paus, S.; McGeorge, D.; Bayat, A. A Double-Blind, Randomized Trial Shows the Role of Zonal Priming and Direct Topical Application of Epigallocatechin-3-Gallate in the Modulation of Cutaneous Scarring in Human Skin. J. Investig. Dermatol. 2019, 139, 1680–1690. [Google Scholar] [CrossRef]
- Komi, D.E.A.; Khomtchouk, K.; Santa Maria, P.L. A Review of the Contribution of Mast Cells in Wound Healing: Involved Molecular and Cellular Mechanisms. Clin. Rev. Allergy Immunol. 2020, 58, 298–312. [Google Scholar] [CrossRef]
- Ud-Din, S.; Wilgus, T.A.; Bayat, A. Mast Cells in Skin Scarring: A Review of Animal and Human Research. Front. Immunol. 2020, 11, 552205. [Google Scholar] [CrossRef]
- Zimmermann, C.; Troeltzsch, D.; Gimenez-Rivera, V.A.; Galli, S.J.; Metz, M.; Maurer, M.; Siebenhaar, F. Mast cells are critical for controlling the bacterial burden and the healing of infected wounds. Proc. Natl. Acad. Sci. USA 2019, 116, 20500–20504. [Google Scholar] [CrossRef]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 2018, 282, 121–150. [Google Scholar] [CrossRef] [PubMed]
- Moore, A.L.; Marshall, C.D.; Barnes, L.A.; Murphy, M.P.; Ransom, R.C.; Longaker, M.T. Scarless wound healing: Transitioning from fetal research to regenerative healing. Wiley Interdiscip. Rev. Dev. Biol. 2018, 7, e309. [Google Scholar] [CrossRef] [PubMed]
- Wilgus, T.A. Regenerative healing in fetal skin: A review of the literature. Ostomy Wound Manag. 2007, 53, 16–31. [Google Scholar]
- Wilgus, T.A. Fetal wound healing. In Wound Healing, Tissue Repair, and Regeneration in Diabetes; Bagchi, D., Das, A., Roy, S., Eds.; Academic Press: Cambridge, MA, USA, 2020. [Google Scholar]
- Armstrong, J.R.; Ferguson, M.W. Ontogeny of the skin and the transition from scar-free to scarring phenotype during wound healing in the pouch young of a marsupial, Monodelphis domestica. Dev. Biol. 1995, 169, 242–260. [Google Scholar] [CrossRef] [PubMed]
- Cowin, A.J.; Brosnan, M.P.; Holmes, T.M.; Ferguson, M.W. Endogenous inflammatory response to dermal wound healing in the fetal and adult mouse. Dev. Dyn. 1998, 212, 385–393. [Google Scholar] [CrossRef]
- Kumta, S.; Ritz, M.; Hurley, J.V.; Crowe, D.; Romeo, R.; O’Brien, B.M. Acute inflammation in foetal and adult sheep: The response to subcutaneous injection of turpentine and carrageenan. Br. J. Plast. Surg. 1994, 47, 360–368. [Google Scholar] [CrossRef]
- Wilgus, T.A.; Bergdall, V.K.; Tober, K.L.; Hill, K.J.; Mitra, S.; Flavahan, N.A.; Oberyszyn, T.M. The impact of cyclooxygenase-2 mediated inflammation on scarless fetal wound healing. Am. J. Pathol. 2004, 165, 753–761. [Google Scholar] [CrossRef]
- Savall, R.; Ferrer, I. Mast cells in the skin of rats during development. Med. Cutanea Ibero Lat. Am. 1981, 9, 345–350. [Google Scholar]
- Wulff, B.C.; Parent, A.E.; Meleski, M.A.; DiPietro, L.A.; Schrementi, M.E.; Wilgus, T.A. Mast cells contribute to scar formation during fetal wound healing. J. Investig. Dermatol. 2012, 132, 458–465. [Google Scholar] [CrossRef]
- Kasugai, T.; Oguri, K.; Jippo-Kanemoto, T.; Morimoto, M.; Yamatodani, A.; Yoshida, K.; Ebi, Y.; Isozaki, K.; Tei, H.; Tsujimura, T.; et al. Deficient differentiation of mast cells in the skin of mi/mi mice. Usefulness of in situ hybridization for evaluation of mast cell phenotype. Am. J. Pathol. 1993, 143, 1337–1347. [Google Scholar]
- Walraven, M.; Talhout, W.; Beelen, R.H.; van Egmond, M.; Ulrich, M.M. Healthy human second-trimester fetal skin is deficient in leukocytes and associated homing chemokines. Wound Repair Regen. 2016, 24, 533–541. [Google Scholar] [CrossRef]
- Combs, J.W.; Lagunoff, D.; Benditt, E.P. Differentiation and proliferation of embryonic mast cells of the rat. J. Cell Biol. 1965, 25, 577–592. [Google Scholar] [CrossRef]
- Wulff, B.C.; Wilgus, T.A. Examining the role of mast cells in fetal wound healing using cultured cells in vitro. Methods Mol. Biol. 2013, 1037, 495–506. [Google Scholar] [PubMed]
- Bagabir, R.; Byers, R.J.; Chaudhry, I.H.; Muller, W.; Paus, R.; Bayat, A. Site-specific immunophenotyping of keloid disease demonstrates immune upregulation and the presence of lymphoid aggregates. Br. J. Dermatol. 2012, 167, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Hermes, B.; Feldmann-Boddeker, I.; Welker, P.; Algermissen, B.; Steckelings, M.U.; Grabbe, J.; Henz, B.M. Altered expression of mast cell chymase and tryptase and of c-Kit in human cutaneous scar tissue. J. Investig. Dermatol. 2000, 114, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Hermes, B.; Welker, P.; Feldmann-Boddeker, I.; Kruger-Krasagakis, S.; Hartmann, K.; Zuberbier, T.; Henz, B.M. Expression of mast cell growth modulating and chemotactic factors and their receptors in human cutaneous scars. J. Investig. Dermatol. 2001, 116, 387–393. [Google Scholar] [CrossRef]
- Beer, T.W.; Baldwin, H.; West, L.; Gallagher, P.J.; Wright, D.H. Mast cells in pathological and surgical scars. Br. J. Ophthalmol. 1998, 82, 691–694. [Google Scholar] [CrossRef][Green Version]
- Dabrowski, R.; Drobnik, J. The effect of disodium cromoglycate on the skin wound healing and collagen content in the wounds of rats. Acta Physiol. Pol. 1990, 41, 195–198. [Google Scholar]
- Chen, L.; Schrementi, M.E.; Ranzer, M.J.; Wilgus, T.A.; DiPietro, L.A. Blockade of mast cell activation reduces cutaneous scar formation. PLoS ONE 2014, 9, e85226. [Google Scholar] [CrossRef]
- Shiota, N.; Nishikori, Y.; Kakizoe, E.; Shimoura, K.; Niibayashi, T.; Shimbori, C.; Tanaka, T.; Okunishi, H. Pathophysiological role of skin mast cells in wound healing after scald injury: Study with mast cell-deficient W/W(V) mice. Int. Arch. Allergy Immunol. 2010, 151, 80–88. [Google Scholar] [CrossRef]
- Younan, G.; Suber, F.; Xing, W.; Shi, T.; Kunori, Y.; Abrink, M.; Pejler, G.; Schlenner, S.M.; Rodewald, H.R.; Moore, F.D., Jr.; et al. The inflammatory response after an epidermal burn depends on the activities of mouse mast cell proteases 4 and 5. J. Immunol. 2010, 185, 7681–7690. [Google Scholar] [CrossRef] [PubMed]
- Iba, Y.; Shibata, A.; Kato, M.; Masukawa, T. Possible involvement of mast cells in collagen remodeling in the late phase of cutaneous wound healing in mice. Int. Immunopharmacol. 2004, 4, 1873–1880. [Google Scholar] [CrossRef] [PubMed]
- Younan, G.J.; Heit, Y.I.; Dastouri, P.; Kekhia, H.; Xing, W.; Gurish, M.F.; Orgill, D.P. Mast cells are required in the proliferation and remodeling phases of microdeformational wound therapy. Plast. Reconstr. Surg. 2011, 128, 649e–658e. [Google Scholar] [CrossRef] [PubMed]
- Nauta, A.C.; Grova, M.; Montoro, D.T.; Zimmermann, A.; Tsai, M.; Gurtner, G.C.; Galli, S.J.; Longaker, M.T. Evidence that mast cells are not required for healing of splinted cutaneous excisional wounds in mice. PLoS ONE 2013, 8, e59167. [Google Scholar] [CrossRef]
- Lilla, J.N.; Chen, C.C.; Mukai, K.; BenBarak, M.J.; Franco, C.B.; Kalesnikoff, J.; Yu, M.; Tsai, M.; Piliponsky, A.M.; Galli, S.J. Reduced mast cell and basophil numbers and function in Cpa3-Cre; Mcl-1fl/fl mice. Blood 2011, 118, 6930–6938. [Google Scholar] [CrossRef]
- Feyerabend, T.B.; Weiser, A.; Tietz, A.; Stassen, M.; Harris, N.; Kopf, M.; Radermacher, P.; Moller, P.; Benoist, C.; Mathis, D.; et al. Cre-mediated cell ablation contests mast cell contribution in models of antibody- and T cell-mediated autoimmunity. Immunity 2011, 35, 832–844. [Google Scholar] [CrossRef]
- Antsiferova, M.; Martin, C.; Huber, M.; Feyerabend, T.B.; Forster, A.; Hartmann, K.; Rodewald, H.R.; Hohl, D.; Werner, S. Mast cells are dispensable for normal and activin-promoted wound healing and skin carcinogenesis. J. Immunol. 2013, 191, 6147–6155. [Google Scholar] [CrossRef]
- Dudeck, A.; Dudeck, J.; Scholten, J.; Petzold, A.; Surianarayanan, S.; Kohler, A.; Peschke, K.; Vohringer, D.; Waskow, C.; Krieg, T.; et al. Mast cells are key promoters of contact allergy that mediate the adjuvant effects of haptens. Immunity 2011, 34, 973–984. [Google Scholar] [CrossRef]
- Willenborg, S.; Eckes, B.; Brinckmann, J.; Krieg, T.; Waisman, A.; Hartmann, K.; Roers, A.; Eming, S.A. Genetic ablation of mast cells redefines the role of mast cells in skin wound healing and bleomycin-induced fibrosis. J. Investig. Dermatol. 2014, 134, 2005–2015. [Google Scholar] [CrossRef]
- Galli, S.J. Rethinking the potential roles of mast cells in skin wound healing and bleomycin-induced skin fibrosis. J. Investig. Dermatol. 2014, 134, 1802–1804. [Google Scholar] [CrossRef]
- Cohen, I.K.; Beaven, M.A.; Horakova, Z.; Keiser, H.R. Histamine and collagen synthesis in keloid and hypertrophic scar. Surg. Forum 1972, 23, 509–510. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Smith, J.C.; Finn, M.C. The possible role of mast cells (allergy) in the production of keloid and hypertrophic scarring. J. Burn Care Rehabil. 1987, 8, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Kwak, I.S.; Park, S.Y.; Choi, Y.H.; Cho, S.I.; Yang, Y.S.; Cho, Y.S.; Choi, M.G.; Seo, C.H.; Park, C.W.; Kim, H.O. Clinical and Histopathological Features of Post Burn Pruritus. J. Burn Care Res. 2016, 37, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Harunari, N.; Zhu, K.Q.; Armendariz, R.T.; Deubner, H.; Muangman, P.; Carrougher, G.J.; Isik, F.F.; Gibran, N.S.; Engrav, L.H. Histology of the thick scar on the female, red Duroc pig: Final similarities to human hypertrophic scar. Burns 2006, 32, 669–677. [Google Scholar] [CrossRef]
- Tredget, E.E.; Shankowsky, H.A.; Pannu, R.; Nedelec, B.; Iwashina, T.; Ghahary, A.; Taerum, T.V.; Scott, P.G. Transforming growth factor-beta in thermally injured patients with hypertrophic scars: Effects of interferon alpha-2b. Plast. Reconstr. Surg. 1998, 102, 1317–1328, discussion 1329–1330. [Google Scholar] [CrossRef]
- Choi, Y.H.; Kim, K.M.; Kim, H.O.; Jang, Y.C.; Kwak, I.S. Clinical and histological correlation in post-burn hypertrophic scar for pain and itching sensation. Ann. Dermatol. 2013, 25, 428–433. [Google Scholar] [CrossRef]
- Chen, H.; Xu, Y.; Yang, G.; Zhang, Q.; Huang, X.; Yu, L.; Dong, X. Mast cell chymase promotes hypertrophic scar fibroblast proliferation and collagen synthesis by activating TGF-beta1/Smads signaling pathway. Exp. Ther. Med. 2017, 14, 4438–4442. [Google Scholar]
- Kischer, C.W.; Bailey, J.F. The mast cell in hypertrophic scars. Tex. Rep. Biol. Med. 1972, 30, 327–338. [Google Scholar]
- Kischer, C.W.; Bunce, H., 3rd; Shetlah, M.R. Mast cell analyses in hypertrophic scars, hypertrophic scars treated with pressure and mature scars. J. Investig. Dermatol. 1978, 70, 355–357. [Google Scholar] [CrossRef]
- Gaber, M.A.; Seliet, I.A.; Ehsan, N.A.; Megahed, M.A. Mast cells and angiogenesis in wound healing. Anal. Quant. Cytopathol. Histopathol. 2014, 36, 32–40. [Google Scholar]
- Weiss, D.S.; Eaglstein, W.H.; Falanga, V. Exogenous electric current can reduce the formation of hypertrophic scars. J. Dermatol. Surg. Oncol. 1989, 15, 1272–1275. [Google Scholar] [CrossRef] [PubMed]
- Niessen, F.B.; Schalkwijk, J.; Vos, H.; Timens, W. Hypertrophic scar formation is associated with an increased number of epidermal Langerhans cells. J. Pathol. 2004, 202, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Algermissen, B.; Hermes, B.; Feldmann-Boeddeker, I.; Bauer, F.; Henz, B.M. Mast cell chymase and tryptase during tissue turnover: Analysis on in vitro mitogenesis of fibroblasts and keratinocytes and alterations in cutaneous scars. Exp. Dermatol. 1999, 8, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Hellstrom, M.; Hellstrom, S.; Engstrom-Laurent, A.; Bertheim, U. The structure of the basement membrane zone differs between keloids, hypertrophic scars and normal skin: A possible background to an impaired function. J. Plast. Reconstr. Aesthetic Surg. 2014, 67, 1564–1572. [Google Scholar] [CrossRef]
- Gallant-Behm, C.L.; Hildebrand, K.A.; Hart, D.A. The mast cell stabilizer ketotifen prevents development of excessive skin wound contraction and fibrosis in red Duroc pigs. Wound Repair Regen. 2008, 16, 226–233. [Google Scholar] [CrossRef]
- Aarabi, S.; Bhatt, K.A.; Shi, Y.; Paterno, J.; Chang, E.I.; Loh, S.A.; Holmes, J.W.; Longaker, M.T.; Yee, H.; Gurtner, G.C. Mechanical load initiates hypertrophic scar formation through decreased cellular apoptosis. FASEB J. 2007, 21, 3250–3261. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Bond, J.; Bergeron, A.; Miller, K.J.; Ehanire, T.; Quiles, C.; Lorden, E.R.; Medina, M.A.; Fisher, M.; Klitzman, B.; et al. A novel immune competent murine hypertrophic scar contracture model: A tool to elucidate disease mechanism and develop new therapies. Wound Repair Regen. 2014, 22, 755–764. [Google Scholar] [CrossRef]
- Wang, J.; Ding, J.; Jiao, H.; Honardoust, D.; Momtazi, M.; Shankowsky, H.A.; Tredget, E.E. Human hypertrophic scar-like nude mouse model: Characterization of the molecular and cellular biology of the scar process. Wound Repair Regen. 2011, 19, 274–285. [Google Scholar] [CrossRef]
- Hakanson, R.; Owman, C.; Sjoberg, N.O.; Sporrong, B. Direct histochemical demonstration of histamine in cutaneous mast cells: Urticaria pigmentosa and keloids. Experientia 1969, 25, 854–855. [Google Scholar] [CrossRef]
- Mantel, A.; Newsome, A.; Thekkudan, T.; Frazier, R.; Katdare, M. The role of aldo-keto reductase 1C3 (AKR1C3)-mediated prostaglandin D2 (PGD2) metabolism in keloids. Exp. Dermatol. 2016, 25, 38–43. [Google Scholar] [CrossRef]
- Ammendola, M.; Zuccala, V.; Patruno, R.; Russo, E.; Luposella, M.; Amorosi, A.; Vescio, G.; Sammarco, G.; Montemurro, S.; De Sarro, G.; et al. Tryptase-positive mast cells and angiogenesis in keloids: A new possible post-surgical target for prevention. Updates Surg. 2013, 65, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Zhang, C.; Ma, S.; Wen, H. Mast cell chymase in keloid induces profibrotic response via transforming growth factor-beta1/Smad activation in keloid fibroblasts. Int. J. Clin. Exp. Pathol. 2014, 7, 3596–3607. [Google Scholar] [PubMed]
- Eishi, K.; Bae, S.J.; Ogawa, F.; Hamasaki, Y.; Shimizu, K.; Katayama, I. Silicone gel sheets relieve pain and pruritus with clinical improvement of keloid: Possible target of mast cells. J. Dermatol. Treat. 2003, 14, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Yoon, B.S.; Moon, J.H.; Kim, B.; Jun, E.K.; Oh, S.; Kim, H.; Song, H.J.; Noh, J.Y.; Oh, C.; et al. Green tea polyphenol epigallocatechin-3-gallate suppresses collagen production and proliferation in keloid fibroblasts via inhibition of the STAT3-signaling pathway. J. Investig. Dermatol. 2008, 128, 2429–2441. [Google Scholar] [CrossRef]
- Zhang, Q.; Kelly, A.P.; Wang, L.; French, S.W.; Tang, X.; Duong, H.S.; Messadi, D.V.; Le, A.D. Green tea extract and (-)-epigallocatechin-3-gallate inhibit mast cell-stimulated type I collagen expression in keloid fibroblasts via blocking PI-3K/AkT signaling pathways. J. Investig. Dermatol. 2006, 126, 2607–2613. [Google Scholar] [CrossRef]
- Syed, F.; Bagabir, R.A.; Paus, R.; Bayat, A. Ex vivo evaluation of antifibrotic compounds in skin scarring: EGCG and silencing of PAI-1 independently inhibit growth and induce keloid shrinkage. Lab. Investig. 2013, 93, 946–960. [Google Scholar] [CrossRef]
- Theoret, C.L.; Olutoye, O.O.; Parnell, L.K.; Hicks, J. Equine exuberant granulation tissue and human keloids: A comparative histopathologic study. Vet. Surg. 2013, 42, 783–789. [Google Scholar] [CrossRef]
- Craig, S.S.; DeBlois, G.; Schwartz, L.B. Mast cells in human keloid, small intestine, and lung by an immunoperoxidase technique using a murine monoclonal antibody against tryptase. Am. J. Pathol. 1986, 124, 427–435. [Google Scholar]
- Allanore, Y.; Simms, R.; Distler, O.; Trojanowska, M.; Pope, J.; Denton, C.P.; Varga, J. Systemic sclerosis. Nat. Rev. Dis. Primers 2015, 1, 15002. [Google Scholar] [CrossRef]
- Ozbilgin, M.K.; Inan, S. The roles of transforming growth factor type beta3 (TGF-beta3) and mast cells in the pathogenesis of scleroderma. Clin. Rheumatol. 2003, 22, 189–195. [Google Scholar] [CrossRef]
- Takeda, K.; Hatamochi, A.; Ueki, H. Increased number of mast cells accompany enhanced collagen synthesis in linear localized scleroderma. Arch. Dermatol. Res. 1989, 281, 288–290. [Google Scholar] [CrossRef] [PubMed]
- Akimoto, S.; Ishikawa, O.; Igarashi, Y.; Kurosawa, M.; Miyachi, Y. Dermal mast cells in scleroderma: Their skin density, tryptase/chymase phenotypes and degranulation. Br. J. Dermatol. 1998, 138, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Prescott, R.J.; Freemont, A.J.; Jones, C.J.; Hoyland, J.; Fielding, P. Sequential dermal microvascular and perivascular changes in the development of scleroderma. J. Pathol. 1992, 166, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Seibold, J.R.; Giorno, R.C.; Claman, H.N. Dermal mast cell degranulation in systemic sclerosis. Arthritis Rheumatol. 1990, 33, 1702–1709. [Google Scholar] [CrossRef]
- Nishioka, K.; Kobayashi, Y.; Katayama, I.; Takijiri, C. Mast cell numbers in diffuse scleroderma. Arch. Dermatol. 1987, 123, 205–208. [Google Scholar] [CrossRef]
- Hawkins, R.A.; Claman, H.N.; Clark, R.A.; Steigerwald, J.C. Increased dermal mast cell populations in progressive systemic sclerosis: A link in chronic fibrosis? Ann. Intern. Med. 1985, 102, 182–186. [Google Scholar] [CrossRef]
- Hugle, T.; Hogan, V.; White, K.E.; van Laar, J.M. Mast cells are a source of transforming growth factor beta in systemic sclerosis. Arthritis Rheumatol. 2011, 63, 795–799. [Google Scholar] [CrossRef]
- Kihira, C.; Mizutani, H.; Asahi, K.; Hamanaka, H.; Shimizu, M. Increased cutaneous immunoreactive stem cell factor expression and serum stem cell factor level in systemic scleroderma. J. Dermatol. Sci. 1998, 20, 72–78. [Google Scholar] [CrossRef]
- Irani, A.M.; Gruber, B.L.; Kaufman, L.D.; Kahaleh, M.B.; Schwartz, L.B. Mast cell changes in scleroderma. Presence of MCT cells in the skin and evidence of mast cell activation. Arthritis Rheumatol. 1992, 35, 933–939. [Google Scholar] [CrossRef]
- Truchetet, M.E.; Brembilla, N.C.; Montanari, E.; Lonati, P.; Raschi, E.; Zeni, S.; Fontao, L.; Meroni, P.L.; Chizzolini, C. Interleukin-17A+ cell counts are increased in systemic sclerosis skin and their number is inversely correlated with the extent of skin involvement. Arthritis Rheumatol. 2013, 65, 1347–1356. [Google Scholar] [CrossRef]
- Claman, H.N. Mast cell changes in a case of rapidly progressive scleroderma-ultrastructural analysis. J. Investig. Dermatol. 1989, 92, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Falanga, V.; Soter, N.A.; Altman, R.D.; Kerdel, F.A. Elevated plasma histamine levels in systemic sclerosis (scleroderma). Arch. Dermatol. 1990, 126, 336–338. [Google Scholar] [CrossRef] [PubMed]
- Pearson, M.E.; Huff, J.C.; Giorno, R.C.; Panicheewa, S.; Claman, H.N.; Steigerwald, J.C. Immunologic dysfunction in scleroderma: Evidence for increased mast cell releasability and HLA-DR positivity in the dermis. Arthritis Rheumatol. 1988, 31, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, R.G.; Varga, J.; Tourtellotte, W.G. Animal models of scleroderma: Recent progress. Curr. Opin. Rheumatol. 2016, 28, 561–570. [Google Scholar] [CrossRef]
- Yamamoto, T.; Takagawa, S.; Katayama, I.; Yamazaki, K.; Hamazaki, Y.; Shinkai, H.; Nishioka, K. Animal model of sclerotic skin. I: Local injections of bleomycin induce sclerotic skin mimicking scleroderma. J. Investig. Dermatol. 1999, 112, 456–462. [Google Scholar] [CrossRef]
- Kitaba, S.; Murota, H.; Terao, M.; Azukizawa, H.; Terabe, F.; Shima, Y.; Fujimoto, M.; Tanaka, T.; Naka, T.; Kishimoto, T.; et al. Blockade of interleukin-6 receptor alleviates disease in mouse model of scleroderma. Am. J. Pathol. 2012, 180, 165–176. [Google Scholar] [CrossRef]
- Kohno, S.; Endo, H.; Hashimoto, A.; Hayashi, I.; Murakami, Y.; Kitasato, H.; Kojima, F.; Kawai, S.; Kondo, H. Inhibition of skin sclerosis by 15deoxy delta12,14-prostaglandin J2 and retrovirally transfected prostaglandin D synthase in a mouse model of bleomycin-induced scleroderma. Biomed. Pharmacother. 2006, 60, 18–25. [Google Scholar] [CrossRef]
- Kawai, M.; Masuda, A.; Kuwana, M. A CD40-CD154 interaction in tissue fibrosis. Arthritis Rheumatol. 2008, 58, 3562–3573. [Google Scholar] [CrossRef]
- Yamamoto, T.; Takagawa, S.; Katayama, I.; Mizushima, Y.; Nishioka, K. Effect of superoxide dismutase on bleomycin-induced dermal sclerosis: Implications for the treatment of systemic sclerosis. J. Investig. Dermatol. 1999, 113, 843–847. [Google Scholar] [CrossRef][Green Version]
- Yamamoto, T.; Takagawa, S.; Katayama, I.; Nishioka, K. Anti-sclerotic effect of transforming growth factor-beta antibody in a mouse model of bleomycin-induced scleroderma. Clin. Immunol. 1999, 92, 6–13. [Google Scholar] [CrossRef]
- Lakos, G.; Melichian, D.; Wu, M.; Varga, J. Increased bleomycin-induced skin fibrosis in mice lacking the Th1-specific transcription factor T-bet. Pathobiology 2006, 73, 224–237. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, M.; Yamamoto, T. Antifibrogenic effects of C-C chemokine receptor type 2 antagonist in a bleomycin-induced scleroderma model. Exp. Dermatol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, C.; Cantarero, I.; Palomares, B.; Gomez-Canas, M.; Fernandez-Ruiz, J.; Pavicic, C.; Garcia-Martin, A.; Luz Bellido, M.; Ortega-Castro, R.; Perez-Sanchez, C.; et al. VCE-004.3, a cannabidiol aminoquinone derivative, prevents bleomycin-induced skin fibrosis and inflammation through PPARgamma- and CB2 receptor-dependent pathways. Br. J. Pharmacol. 2018, 175, 3813–3831. [Google Scholar] [CrossRef] [PubMed]
- Pamuk, O.N.; Can, G.; Ayvaz, S.; Karaca, T.; Pamuk, G.E.; Demirtas, S.; Tsokos, G.C. Spleen tyrosine kinase (Syk) inhibitor fostamatinib limits tissue damage and fibrosis in a bleomycin-induced scleroderma mouse model. Clin. Exp. Rheumatol. 2015, 33 (Suppl. 91), S15–S22. [Google Scholar] [PubMed]
- Yamamoto, T.; Takahashi, Y.; Takagawa, S.; Katayama, I.; Nishioka, K. Animal model of sclerotic skin. II. Bleomycin induced scleroderma in genetically mast cell deficient WBB6F1-W/W(V) mice. J. Rheumatol. 1999, 26, 2628–2634. [Google Scholar]
- Siracusa, L.D.; McGrath, R.; Ma, Q.; Moskow, J.J.; Manne, J.; Christner, P.J.; Buchberg, A.M.; Jimenez, S.A. A tandem duplication within the fibrillin 1 gene is associated with the mouse tight skin mutation. Genome Res. 1996, 6, 300–313. [Google Scholar] [CrossRef][Green Version]
- Walker, M.; Harley, R.; Maize, J.; DeLustro, F.; LeRoy, E.C. Mast cells and their degranulation in the Tsk mouse model of scleroderma. Proc. Soc. Exp. Biol. Med. 1985, 180, 323–328. [Google Scholar] [CrossRef]
- Wang, H.W.; Tedla, N.; Hunt, J.E.; Wakefield, D.; McNeil, H.P. Mast cell accumulation and cytokine expression in the tight skin mouse model of scleroderma. Exp. Dermatol. 2005, 14, 295–302. [Google Scholar] [CrossRef]
- Kakizoe, E.; Shiota, N.; Tanabe, Y.; Shimoura, K.; Kobayashi, Y.; Okunishi, H. Isoform-selective upregulation of mast cell chymase in the development of skin fibrosis in scleroderma model mice. J. Investig. Dermatol. 2001, 116, 118–123. [Google Scholar] [CrossRef]
- Walker, M.; Harley, R.; LeRoy, E.C. Ketotifen prevents skin fibrosis in the tight skin mouse. J. Rheumatol. 1990, 17, 57–59. [Google Scholar]
- Walker, M.A.; Harley, R.A.; LeRoy, E.C. Inhibition of fibrosis in TSK mice by blocking mast cell degranulation. J. Rheumatol. 1987, 14, 299–301. [Google Scholar] [PubMed]
- Shiota, N.; Kakizoe, E.; Shimoura, K.; Tanaka, T.; Okunishi, H. Effect of mast cell chymase inhibitor on the development of scleroderma in tight-skin mice. Br. J. Pharmacol. 2005, 145, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.A.; Harley, R.A.; DeLustro, F.A.; LeRoy, E.C. Adoptive transfer of tsk skin fibrosis to +/+ recipients by tsk bone marrow and spleen cells. Proc. Soc. Exp. Biol. Med. 1989, 192, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Everett, E.T.; Pablos, J.L.; Harley, R.A.; LeRoy, E.C.; Norris, J.S. The role of mast cells in the development of skin fibrosis in tight-skin mutant mice. Comp. Biochem. Physiol. A Physiol. 1995, 110, 159–165. [Google Scholar] [CrossRef]
- Pincha, N.; Hajam, E.Y.; Badarinath, K.; Batta, S.P.R.; Masudi, T.; Dey, R.; Andreasen, P.; Kawakami, T.; Samuel, R.; George, R.; et al. PAI1 mediates fibroblast-mast cell interactions in skin fibrosis. J. Clin. Investig. 2018, 128, 1807–1819. [Google Scholar] [CrossRef]
- Spielvogel, R.L.; Goltz, R.W.; Kersey, J.H. Scleroderma-like changes in chronic graft vs host disease. Arch. Dermatol. 1977, 113, 1424–1428. [Google Scholar] [CrossRef]
- Strattan, E.; Palaniyandi, S.; Kumari, R.; Du, J.; Hakim, N.; Huang, T.; Kesler, M.V.; Jennings, C.D.; Sturgill, J.L.; Hildebrandt, G.C. Mast Cells Are Mediators of Fibrosis and Effector Cell Recruitment in Dermal Chronic Graft-vs.-Host Disease. Front. Immunol. 2019, 10, 2470. [Google Scholar] [CrossRef]
- Janin-Mercier, A.; Devergie, A.; Van Cauwenberge, D.; Saurat, J.H.; Bourges, M.; Lapiere, C.M.; Gluckman, E. Immunohistologic and ultrastructural study of the sclerotic skin in chronic graft-versus-host disease in man. Am. J. Pathol. 1984, 115, 296–306. [Google Scholar]
- Charley, M.R.; Bangert, J.L.; Hamilton, B.L.; Gilliam, J.N.; Sontheimer, R.D. Murine graft-versus-host skin disease: A chronologic and quantitative analysis of two histologic patterns. J. Investig. Dermatol. 1983, 81, 412–417. [Google Scholar] [CrossRef]
- Claman, H.N. Mast cell depletion in murine chronic graft-versus-host disease. J. Investig. Dermatol. 1985, 84, 246–248. [Google Scholar] [CrossRef]
- Claman, H.N.; Jaffee, B.D.; Huff, J.C.; Clark, R.A. Chronic graft-versus-host disease as a model for scleroderma. II. Mast cell depletion with deposition of immunoglobulins in the skin and fibrosis. Cell. Immunol. 1985, 94, 73–84. [Google Scholar] [CrossRef]
- Claman, H.N.; Choi, K.L.; Sujansky, W.; Vatter, A.E. Mast cell “disappearance” in chronic murine graft-vs-host disease (GVHD)-ultrastructural demonstration of “phantom mast cells”. J. Immunol. 1986, 137, 2009–2013. [Google Scholar] [PubMed]
- Choi, K.L.; Giorno, R.; Claman, H.N. Cutaneous mast cell depletion and recovery in murine graft-vs-host disease. J. Immunol. 1987, 138, 4093–4101. [Google Scholar] [PubMed]
- Jaffee, B.D.; Claman, H.N. Chronic graft-versus-host disease (GVHD) as a model for scleroderma. I. Description of model systems. Cell. Immunol. 1983, 77, 1–12. [Google Scholar] [CrossRef]
- Levi-Schaffer, F.; Goldenhersh, M.A.; Segal, V.; Nagler, A. Nedocromil sodium ameliorates skin manifestations in a murine model of chronic graft-versus-host disease. Bone Marrow Transpl. 1997, 19, 823–828. [Google Scholar] [CrossRef][Green Version]
- Abe, M.; Yokoyama, Y.; Amano, H.; Matsushima, Y.; Kan, C.; Ishikawa, O. Effect of activated human mast cells and mast cell-derived mediators on proliferation, type I collagen production and glycosaminoglycans synthesis by human dermal fibroblasts. Eur. J. Dermatol. 2002, 12, 340–346. [Google Scholar]
- Levi-Schaffer, F.; Kupietzky, A. Mast cells enhance migration and proliferation of fibroblasts into an in vitro wound. Exp. Cell Res. 1990, 188, 42–49. [Google Scholar] [CrossRef]
- Gordon, J.R.; Galli, S.J. Promotion of mouse fibroblast collagen gene expression by mast cells stimulated via the Fc epsilon RI. Role for mast cell-derived transforming growth factor beta and tumor necrosis factor alpha. J. Exp. Med. 1994, 180, 2027–2037. [Google Scholar] [CrossRef]
- Kendall, J.C.; Li, X.H.; Galli, S.J.; Gordon, J.R. Promotion of mouse fibroblast proliferation by IgE-dependent activation of mouse mast cells: Role for mast cell tumor necrosis factor-alpha and transforming growth factor-beta 1. J. Allergy Clin. Immunol. 1997, 99 Pt 1, 113–123. [Google Scholar] [CrossRef]
- Albrecht, M.; Frungieri, M.B.; Kunz, L.; Ramsch, R.; Meineke, V.; Kohn, F.M.; Mayerhofer, A. Divergent effects of the major mast cell products histamine, tryptase and TNF-alpha on human fibroblast behaviour. Cell. Mol. Life Sci. 2005, 62, 2867–2876. [Google Scholar] [CrossRef]
- Abe, M.; Kurosawa, M.; Ishikawa, O.; Miyachi, Y.; Kido, H. Mast cell tryptase stimulates both human dermal fibroblast proliferation and type I collagen production. Clin. Exp. Allergy 1998, 28, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Garbuzenko, E.; Nagler, A.; Pickholtz, D.; Gillery, P.; Reich, R.; Maquart, F.X.; Levi-Schaffer, F. Human mast cells stimulate fibroblast proliferation, collagen synthesis and lattice contraction: A direct role for mast cells in skin fibrosis. Clin. Exp. Allergy 2002, 32, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Gruber, B.L.; Kew, R.R.; Jelaska, A.; Marchese, M.J.; Garlick, J.; Ren, S.; Schwartz, L.B.; Korn, J.H. Human mast cells activate fibroblasts: Tryptase is a fibrogenic factor stimulating collagen messenger ribonucleic acid synthesis and fibroblast chemotaxis. J. Immunol. 1997, 158, 2310–2317. [Google Scholar] [PubMed]
- Gailit, J.; Marchese, M.J.; Kew, R.R.; Gruber, B.L. The differentiation and function of myofibroblasts is regulated by mast cell mediators. J. Investig. Dermatol. 2001, 117, 1113–1119. [Google Scholar] [CrossRef]
- Abe, M.; Kurosawa, M.; Ishikawa, O.; Miyachi, Y. Effect of mast cell-derived mediators and mast cell-related neutral proteases on human dermal fibroblast proliferation and type I collagen production. J. Allergy Clin. Immunol. 2000, 106 Pt 2, S78–S84. [Google Scholar] [CrossRef]
- Dong, X.; Chen, J.; Zhang, Y.; Cen, Y. Mast cell chymase promotes cell proliferation and expression of certain cytokines in a dose-dependent manner. Mol. Med. Rep. 2012, 5, 1487–1490. [Google Scholar]
- Dong, X.; Zhang, C.; Ma, S.; Wen, H. High concentrations of mast cell chymase facilitate the transduction of the transforming growth factor-beta1/Smads signaling pathway in skin fibroblasts. Exp. Ther. Med. 2015, 9, 955–960. [Google Scholar] [CrossRef]
- Kofford, M.W.; Schwartz, L.B.; Schechter, N.M.; Yager, D.R.; Diegelmann, R.F.; Graham, M.F. Cleavage of type I procollagen by human mast cell chymase initiates collagen fibril formation and generates a unique carboxyl-terminal propeptide. J. Biol. Chem. 1997, 272, 7127–7131. [Google Scholar] [CrossRef]
- Murota, H.; Bae, S.; Hamasaki, Y.; Maruyama, R.; Katayama, I. Emedastine difumarate inhibits histamine-induced collagen synthesis in dermal fibroblasts. J. Investig. Allergol. Clin. Immunol. 2008, 18, 245–252. [Google Scholar]
- Abe, M.; Kurosawa, M.; Igarashi, Y.; Ishikawa, O.; Miyachi, Y. Influence of IgE-mediated activation of cultured human mast cells on proliferation and type I collagen production by human dermal fibroblasts. J. Allergy Clin. Immunol. 2000, 106 Pt 2, S72–S77. [Google Scholar] [CrossRef]
- Kupietzky, A.; Levi-Schaffer, F. The role of mast cell-derived histamine in the closure of an in vitro wound. Inflamm. Res. 1996, 45, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Trautmann, A.; Krohne, G.; Brocker, E.B.; Klein, C.E. Human mast cells augment fibroblast proliferation by heterotypic cell-cell adhesion and action of IL-4. J. Immunol. 1998, 160, 5053–5057. [Google Scholar] [PubMed]
- Postlethwaite, A.E.; Seyer, J.M. Fibroblast chemotaxis induction by human recombinant interleukin-4. Identification by synthetic peptide analysis of two chemotactic domains residing in amino acid sequences 70–88 and 89–122. J. Clin. Investig. 1991, 87, 2147–2152. [Google Scholar] [CrossRef] [PubMed]
- Postlethwaite, A.E.; Holness, M.A.; Katai, H.; Raghow, R. Human fibroblasts synthesize elevated levels of extracellular matrix proteins in response to interleukin 4. J. Clin. Investig. 1992, 90, 1479–1485. [Google Scholar] [CrossRef]
- Fertin, C.; Nicolas, J.F.; Gillery, P.; Kalis, B.; Banchereau, J.; Maquart, F.X. Interleukin-4 stimulates collagen synthesis by normal and scleroderma fibroblasts in dermal equivalents. Cell. Mol. Biol. 1991, 37, 823–829. [Google Scholar]
- Kim, C.D.; Sohn, K.C.; Lee, S.S.; Lee, J.H.; Kim, S.; Lee, Y.H.; Ryu, E.K.; Park, Y.S. Plasminogen activator inhibitor-2 (PAI-2) secreted from activated mast cells induces alpha-smooth muscle actin (alpha-SMA) expression in dermal fibroblasts. J. Dermatol. Sci. 2011, 62, 204–206. [Google Scholar] [CrossRef]
- Gordon, J.R. Monocyte chemoattractant peptide-1 expression during cutaneous allergic reactions in mice is mast cell dependent and largely mediates the monocyte recruitment response. J. Allergy Clin. Immunol. 2000, 106 Pt 1, 110–116. [Google Scholar] [CrossRef]
- Yamamoto, T.; Hartmann, K.; Eckes, B.; Krieg, T. Role of stem cell factor and monocyte chemoattractant protein-1 in the interaction between fibroblasts and mast cells in fibrosis. J. Dermatol. Sci. 2001, 26, 106–111. [Google Scholar] [CrossRef]
- Nabeshima, Y.; Hiragun, T.; Morita, E.; Mihara, S.; Kameyoshi, Y.; Hide, M. IL-4 modulates the histamine content of mast cells in a mast cell/fibroblast co-culture through a Stat6 signaling pathway in fibroblasts. FEBS Lett. 2005, 579, 6653–6658. [Google Scholar] [CrossRef]
- Gordon, J.R. TGFbeta1 and TNFalpha secreted by mast cells stimulated via the FcepsilonRI activate fibroblasts for high-level production of monocyte chemoattractant protein-1 (MCP-1). Cell. Immunol. 2000, 201, 42–49. [Google Scholar] [CrossRef]
- Ferreira, A.M.; Takagawa, S.; Fresco, R.; Zhu, X.; Varga, J.; DiPietro, L.A. Diminished induction of skin fibrosis in mice with MCP-1 deficiency. J. Investig. Dermatol. 2006, 126, 1900–1908. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.W.; Rustad, K.C.; Akaishi, S.; Sorkin, M.; Glotzbach, J.P.; Januszyk, M.; Nelson, E.R.; Levi, K.; Paterno, J.; Vial, I.N.; et al. Focal adhesion kinase links mechanical force to skin fibrosis via inflammatory signaling. Nat. Med. 2011, 18, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Foley, T.T.; Ehrlich, H.P. Through gap junction communications, co-cultured mast cells and fibroblasts generate fibroblast activities allied with hypertrophic scarring. Plast. Reconstr. Surg. 2013, 131, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Au, S.R.; Au, K.; Saggers, G.C.; Karne, N.; Ehrlich, H.P. Rat mast cells communicate with fibroblasts via gap junction intercellular communications. J. Cell. Biochem. 2007, 100, 1170–1177. [Google Scholar] [CrossRef]
- Yamamoto, T.; Hartmann, K.; Eckes, B.; Krieg, T. Mast cells enhance contraction of three-dimensional collagen lattices by fibroblasts by cell-cell interaction: Role of stem cell factor/c-kit. Immunology 2000, 99, 435–439. [Google Scholar] [CrossRef]
- Foley, T.T.; Ehrlich, H.P. Mast cells prevent dexamethasone-induced cell death of cultured fibroblasts: Relationship to gap junctional intercellular communications. Plast. Reconstr. Surg. 2014, 133, 638e–644e. [Google Scholar] [CrossRef]
- Pistorio, A.L.; Ehrlich, H.P. Modulatory effects of connexin-43 expression on gap junction intercellular communications with mast cells and fibroblasts. J. Cell. Biochem. 2011, 112, 1441–1449. [Google Scholar] [CrossRef]
- Arbi, S.; Eksteen, E.C.; Oberholzer, H.M.; Taute, H.; Bester, M.J. Premature collagen fibril formation, fibroblast-mast cell interactions and mast cell-mediated phagocytosis of collagen in keloids. Ultrastruct. Pathol. 2015, 39, 95–103. [Google Scholar] [CrossRef][Green Version]
- Shaker, S.A.; Ayuob, N.N.; Hajrah, N.H. Cell talk: A phenomenon observed in the keloid scar by immunohistochemical study. Appl. Immunohistochem. Mol. Morphol. 2011, 19, 153–159. [Google Scholar] [CrossRef]
- Tellechea, A.; Bai, S.; Dangwal, S.; Theocharidis, G.; Nagai, M.; Koerner, S.; Cheong, J.E.; Bhasin, S.; Shih, T.Y.; Zheng, Y.; et al. Topical Application of a Mast Cell Stabilizer Improves Impaired Diabetic Wound Healing. J. Investig. Dermatol. 2020, 140, 901–911. [Google Scholar] [CrossRef]
- Bai, S.; Nagai, M.; Koerner, S.K.; Veves, A.; Sun, L. Structure-activity relationship study and discovery of indazole 3-carboxamides as calcium-release activated calcium channel blockers. Bioorg. Med. Chem. Lett. 2017, 27, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Reber, L.L.; Marichal, T.; Galli, S.J. New models for analyzing mast cell functions in vivo. Trends Immunol. 2012, 33, 613–625. [Google Scholar] [CrossRef] [PubMed]
- Grimbaldeston, M.A.; Chen, C.C.; Piliponsky, A.M.; Tsai, M.; Tam, S.Y.; Galli, S.J. Mast cell-deficient W-sash c-kit mutant Kit W-sh/W-sh mice as a model for investigating mast cell biology in vivo. Am. J. Pathol. 2005, 167, 835–848. [Google Scholar] [CrossRef]
- Katz, H.R.; Austen, K.F. Mast cell deficiency, a game of kit and mouse. Immunity 2011, 35, 668–670. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Gaudenzio, N.; Tsai, M. Mast Cells in Inflammation and Disease: Recent Progress and Ongoing Concerns. Annu. Rev. Immunol. 2020, 38, 49–77. [Google Scholar] [CrossRef] [PubMed]
- Wulff, B.C.; Pappa, N.K.; Wilgus, T.A. Interleukin-33 encourages scar formation in murine fetal skin wounds. Wound Repair Regen. 2019, 27, 19–28. [Google Scholar] [CrossRef]
- Gaudenzio, N.; Marichal, T.; Galli, S.J.; Reber, L.L. Genetic and Imaging Approaches Reveal Pro-Inflammatory and Immunoregulatory Roles of Mast Cells in Contact Hypersensitivity. Front. Immunol. 2018, 9, 1275. [Google Scholar] [CrossRef]
- Tsai, M.; Grimbaldeston, M.; Galli, S.J. Mast cells and immunoregulation/immunomodulation. Adv. Exp. Med. Biol. 2011, 716, 186–211. [Google Scholar]
- Amiot, L.; Vu, N.; Drenou, B.; Scrofani, M.; Chalin, A.; Devisme, C.; Samson, M. The anti-fibrotic role of mast cells in the liver is mediated by HLA-G and interaction with hepatic stellate cells. Cytokine 2019, 117, 50–58. [Google Scholar] [CrossRef]
- Beghdadi, W.; Madjene, L.C.; Claver, J.; Pejler, G.; Beaudoin, L.; Lehuen, A.; Daugas, E.; Blank, U. Mast cell chymase protects against renal fibrosis in murine unilateral ureteral obstruction. Kidney Int. 2013, 84, 317–326. [Google Scholar] [CrossRef]
- Legere, S.A.; Haidl, I.D.; Legare, J.F.; Marshall, J.S. Mast Cells in Cardiac Fibrosis: New Insights Suggest Opportunities for Intervention. Front. Immunol. 2019, 10, 580. [Google Scholar] [CrossRef] [PubMed]
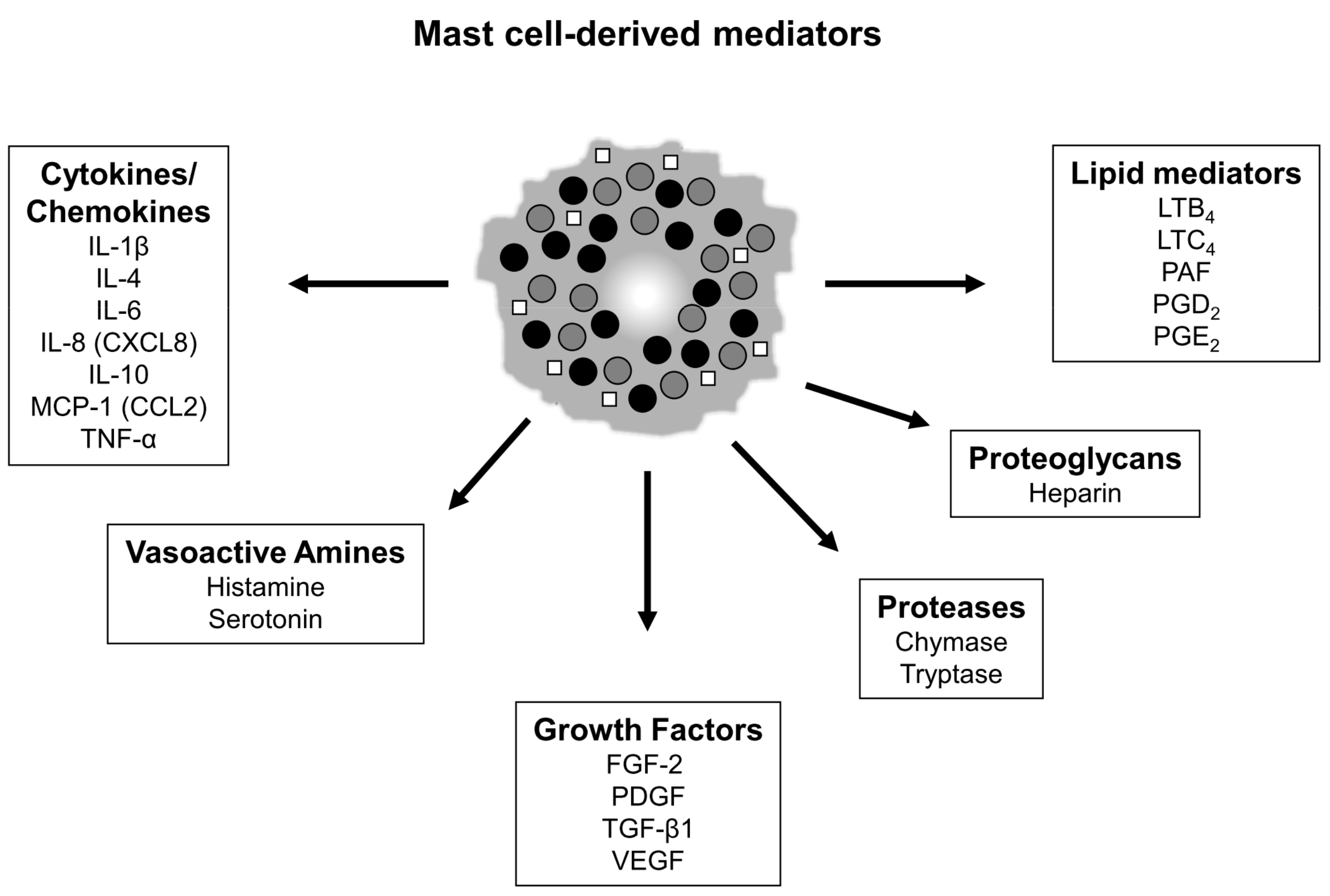

| Early- to Mid-Gestation Skin (Scarless) | Late-Gestation/ Postnatal Skin (Fibrotic) | ||
|---|---|---|---|
| Unwounded skin | Mast cell number | lower | higher |
| Mast cell maturity | less mature | more mature | |
| Mast cell size | smaller | larger | |
| Mast cell granularity | less dense | more dense | |
| Wounded skin | Mast cell degranulation | infrequent | pronounced |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wilgus, T.A.; Ud-Din, S.; Bayat, A. A Review of the Evidence for and against a Role for Mast Cells in Cutaneous Scarring and Fibrosis. Int. J. Mol. Sci. 2020, 21, 9673. https://doi.org/10.3390/ijms21249673
Wilgus TA, Ud-Din S, Bayat A. A Review of the Evidence for and against a Role for Mast Cells in Cutaneous Scarring and Fibrosis. International Journal of Molecular Sciences. 2020; 21(24):9673. https://doi.org/10.3390/ijms21249673
Chicago/Turabian StyleWilgus, Traci A., Sara Ud-Din, and Ardeshir Bayat. 2020. "A Review of the Evidence for and against a Role for Mast Cells in Cutaneous Scarring and Fibrosis" International Journal of Molecular Sciences 21, no. 24: 9673. https://doi.org/10.3390/ijms21249673
APA StyleWilgus, T. A., Ud-Din, S., & Bayat, A. (2020). A Review of the Evidence for and against a Role for Mast Cells in Cutaneous Scarring and Fibrosis. International Journal of Molecular Sciences, 21(24), 9673. https://doi.org/10.3390/ijms21249673





