Role and Regulation of Mechanotransductive HIF-1α Stabilisation in Periodontal Ligament Fibroblasts
Abstract
1. Introduction
2. Results
2.1. Strain-Dependent Stabilisation of HIF-1α
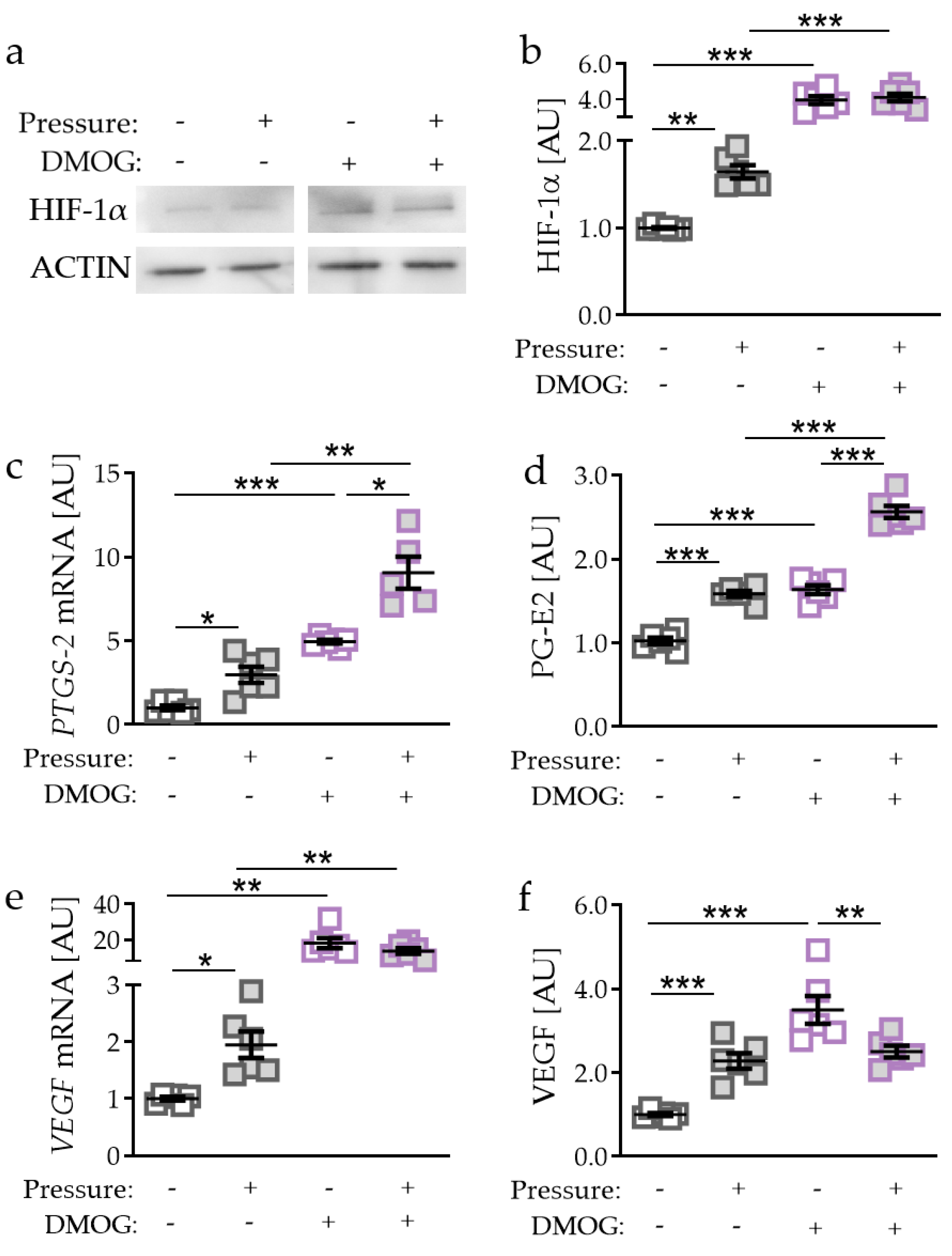
2.2. Effects of HIF-1α Stabilisation via DMOG in PDLF during Compressive Strain
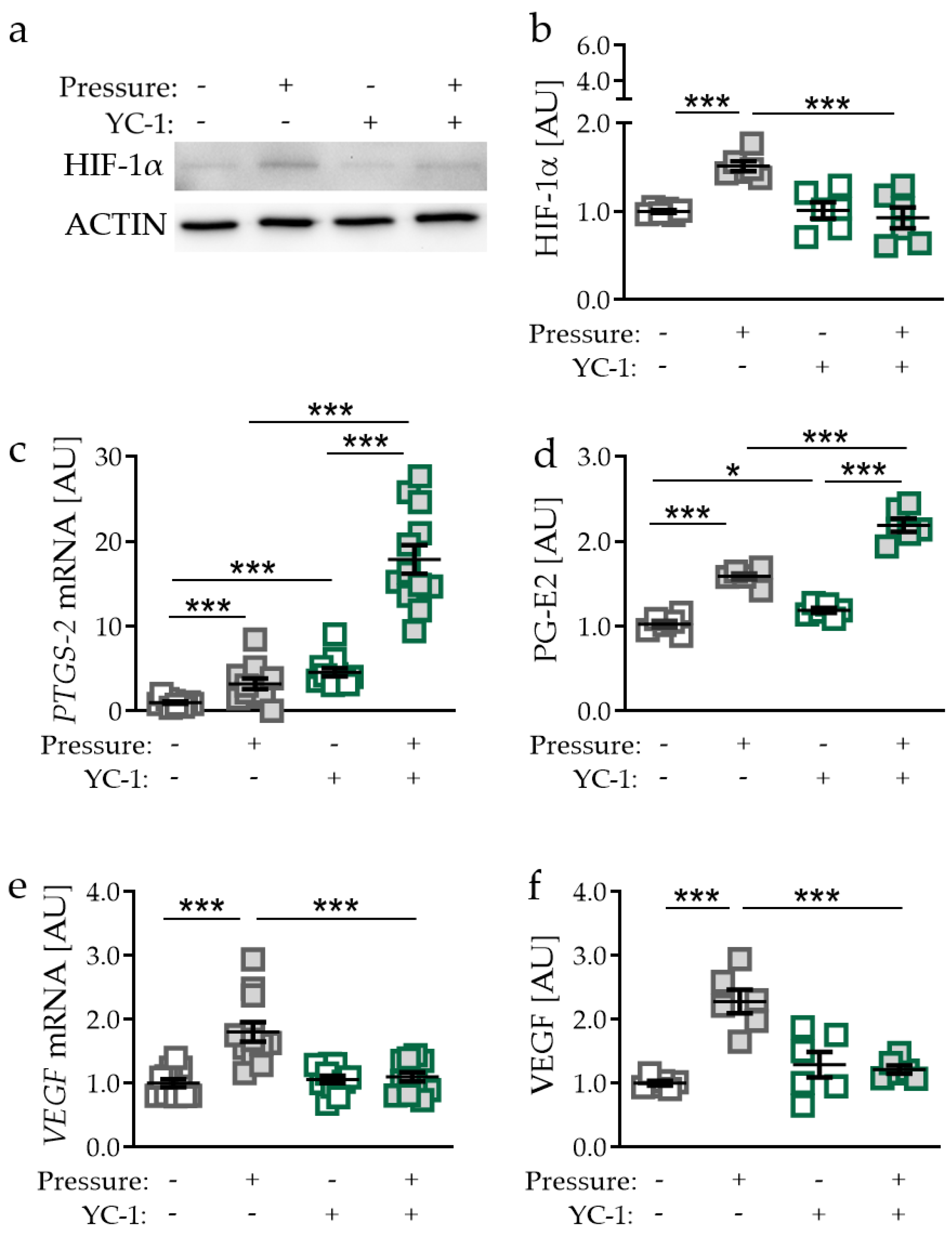
2.3. Effects of Inhibited HIF-1α Stabilisation via YC-1 in PDLF during Compressive Strain
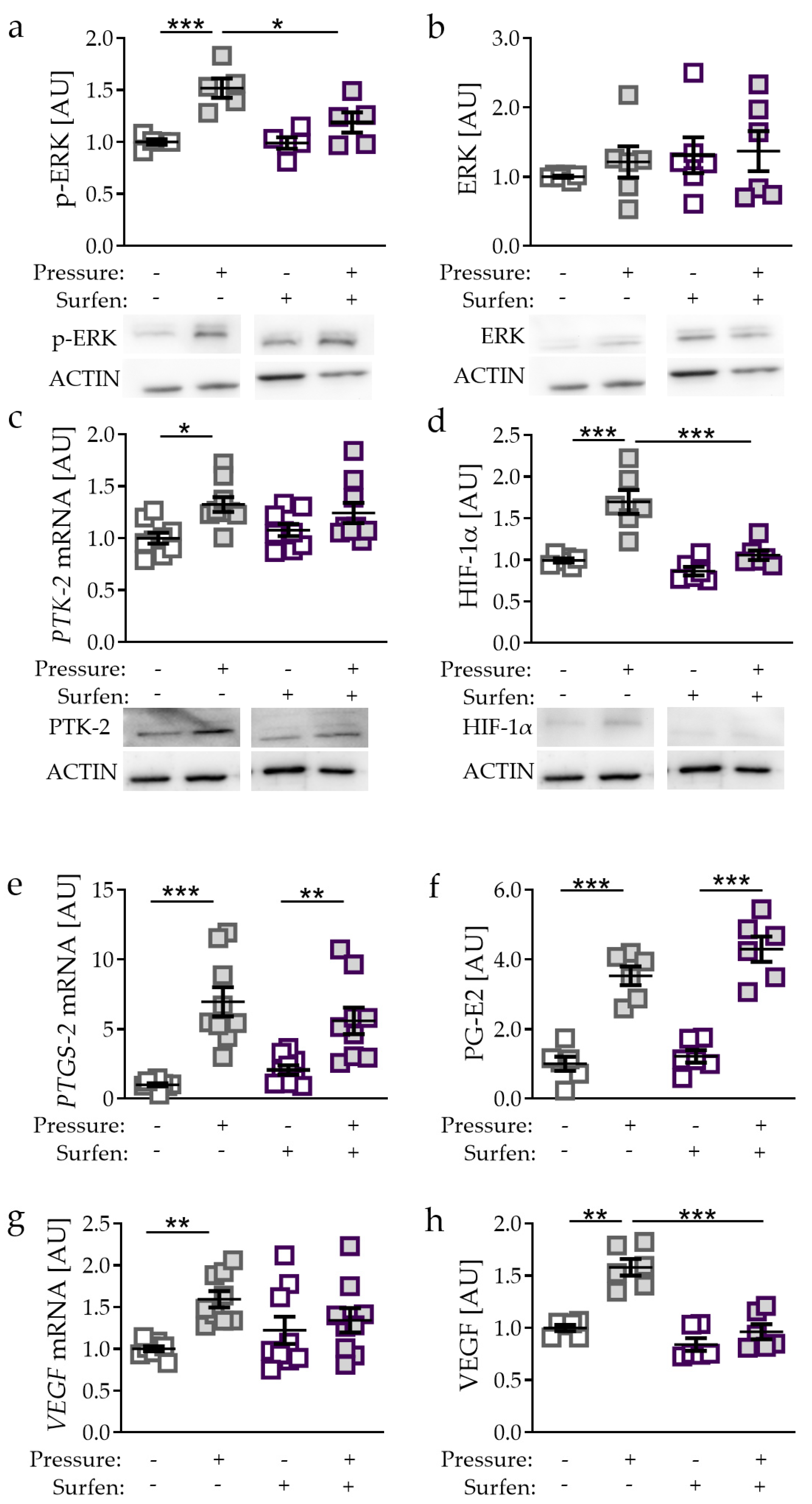
2.4. Effects of Glycosaminoglycan Antagonist Surfen on Pressure-Induced HIF-1α Stabilisation
2.5. Effects of PTK-2 Inhibitor Genistein on Pressure-Induced HIF-1α Stabilisation
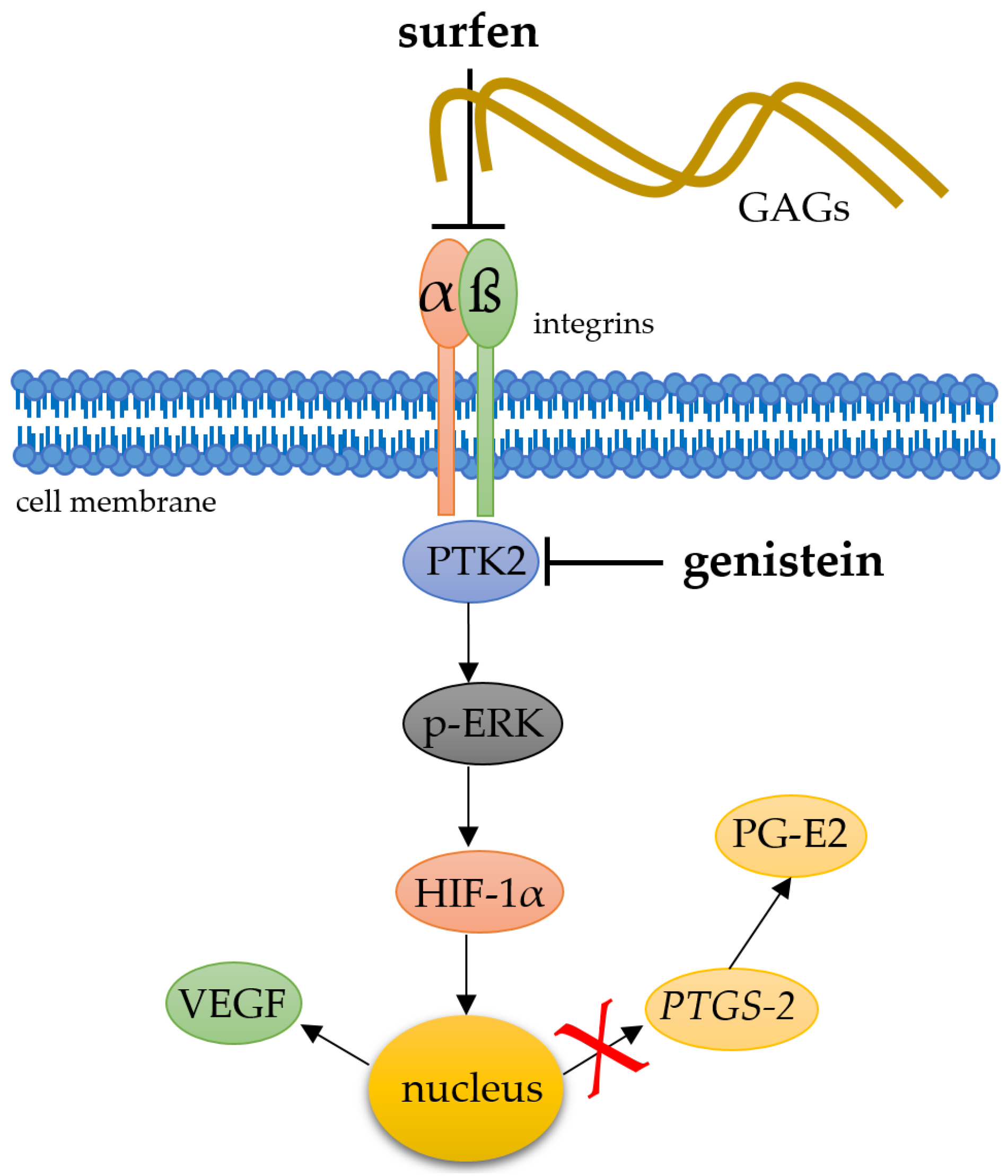
3. Discussion
4. Materials and Methods
4.1. General Cell Culture Conditions
4.2. Reagents
4.3. Experimental Setup
4.4. RNA Extraction
4.5. cDNA Synthesis
4.6. Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR)
4.7. Western Blot Analysis
4.8. Enzyme-Linked Immunosorbent Assays (ELISA)
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Asiry, M.A. Biological aspects of orthodontic tooth movement: A review of literature. Saudi J. Biol. Sci. 2018, 25, 1027–1032. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jacox, L.A.; Little, S.H.; Ko, C.-C. Orthodontic tooth movement: The biology and clinical implications. Kaohsiung J. Med. Sci. 2018, 34, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Meikle, M.C. The tissue, cellular, and molecular regulation of orthodontic tooth movement: 100 years after Carl Sandstedt. Eur. J. Orthod. 2006, 28, 221–240. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.; Lossdörfer, S.; Marciniak, J.; Römer, P.; Kirschneck, C.; Craveiro, R.; Deschner, J.; Jäger, A. CD8+ T cells mediate the regenerative PTH effect in hPDL cells via Wnt10b signaling. Innate. Immun. 2016, 22, 674–681. [Google Scholar] [CrossRef]
- Wolf, M.; Lossdörfer, S.; Römer, P.; Kirschneck, C.; Küpper, K.; Deschner, J.; Jäger, A. Short-term heat pre-treatment modulates the release of HMGB1 and pro-inflammatory cytokines in hPDL cells following mechanical loading and affects monocyte behavior. Clin. Oral Investig. 2016, 20, 923–931. [Google Scholar] [CrossRef]
- Schröder, A.; Bauer, K.; Spanier, G.; Proff, P.; Wolf, M.; Kirschneck, C. Expression kinetics of human periodontal ligament fibroblasts in the early phases of orthodontic tooth movement. J. Orofac. Orthop. 2018, 79, 337–351. [Google Scholar] [CrossRef]
- Choudhary, S.; Blackwell, K.; Voznesensky, O.; Deb Roy, A.; Pilbeam, C. Prostaglandin E2 acts via bone marrow macrophages to block PTH-stimulated osteoblast differentiation in vitro. Bone 2013, 56, 31–41. [Google Scholar] [CrossRef]
- Kanzaki, H.; Chiba, M.; Shimizu, Y.; Mitani, H. Periodontal ligament cells under mechanical stress induce osteoclastogenesis by receptor activator of nuclear factor kappaB ligand up-regulation via prostaglandin E2 synthesis. J. Bone Miner. Res. 2002, 17, 210–220. [Google Scholar] [CrossRef]
- Ullrich, N.; Schröder, A.; Jantsch, J.; Spanier, G.; Proff, P.; Kirschneck, C. The role of mechanotransduction versus hypoxia during simulated orthodontic compressive strain-an in vitro study of human periodontal ligament fibroblasts. Int. J. Oral Sci. 2019, 11, 33. [Google Scholar] [CrossRef]
- Tirpe, A.A.; Gulei, D.; Ciortea, S.M.; Crivii, C.; Berindan-Neagoe, I. Hypoxia: Overview on Hypoxia-Mediated Mechanisms with a Focus on the Role of HIF Genes. Int. J. Mol. Sci. 2019, 20, 6140. [Google Scholar] [CrossRef]
- Hirota, K. Basic Biology of Hypoxic Responses Mediated by the Transcription Factor HIFs and its Implication for Medicine. Biomedicines 2020, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Schröder, A.; Nazet, U.; Neubert, P.; Jantsch, J.; Spanier, G.; Proff, P.; Kirschneck, C. Sodium-chloride-induced effects on the expression profile of human periodontal ligament fibroblasts with focus on simulated orthodontic tooth movement. Eur. J. Oral Sci. 2019, 127, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Regulation of hypoxia-induced angiogenesis: A chaperone escorts VEGF to the dance. J. Clin. Investig. 2001, 108, 39–40. [Google Scholar] [CrossRef] [PubMed]
- Miyagawa, A.; Chiba, M.; Hayashi, H.; Igarashi, K. Compressive force induces VEGF production in periodontal tissues. J. Dent. Res. 2009, 88, 752–756. [Google Scholar] [CrossRef]
- Sastry, S.K.; Burridge, K. Focal adhesions: A nexus for intracellular signaling and cytoskeletal dynamics. Exp. Cell Res. 2000, 261, 25–36. [Google Scholar] [CrossRef]
- Clark, E.A.; Brugge, J.S. Integrins and signal transduction pathways: The road taken. Science 1995, 268, 233–239. [Google Scholar] [CrossRef]
- Basdra, E.K.; Papavassiliou, A.G.; Huber, L.A. Rab and rho GTPases are involved in specific response of periodontal ligament fibroblasts to mechanical stretching. Biochim. Biophys. Acta 1995, 1268, 209–213. [Google Scholar] [CrossRef][Green Version]
- Peverali, F.A.; Basdra, E.K.; Papavassiliou, A.G. Stretch-mediated activation of selective MAPK subtypes and potentiation of AP-1 binding in human osteoblastic cells. Mol. Med. 2001, 7, 68–78. [Google Scholar] [CrossRef]
- Wang, W.-S.; Liang, H.-Y.; Cai, Y.-J.; Yang, H. DMOG ameliorates IFN-γ-induced intestinal barrier dysfunction by suppressing PHD2-dependent HIF-1α degradation. J. Interferon. Cytokine Res. 2014, 34, 60–69. [Google Scholar] [CrossRef]
- Yeo, E.-J.; Chun, Y.-S.; Cho, Y.-S.; Kim, J.; Lee, J.-C.; Kim, M.-S.; Park, J.-W. YC-1: A potential anticancer drug targeting hypoxia-inducible factor 1. J. Natl. Cancer Inst. 2003, 95, 516–525. [Google Scholar] [CrossRef]
- Surve, C.R.; To, J.Y.; Malik, S.; Kim, M.; Smrcka, A.V. Dynamic regulation of neutrophil polarity and migration by the heterotrimeric G protein subunits Gαi-GTP and Gβγ. Sci. Signal 2016, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Wang, J.; Wu, Q.; Qian, J.; Yang, C.; Bo, P. Genistein inhibits the growth and regulates the migration and invasion abilities of melanoma cells via the FAK/paxillin and MAPK pathways. Oncotarget 2017, 8, 21674–21691. [Google Scholar] [CrossRef] [PubMed]
- Sawada, Y.; Sheetz, M.P. Force transduction by Triton cytoskeletons. J. Cell. Biol. 2002, 156, 609–615. [Google Scholar] [CrossRef]
- Mendes, R.T.; Nguyen, D.; Stephens, D.; Pamuk, F.; Fernandes, D.; Hasturk, H.; van Dyke, T.E.; Kantarci, A. Hypoxia-induced endothelial cell responses-possible roles during periodontal disease. Clin. Exp. Dent. Res. 2018, 4, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Tamura, R.; Tanaka, T.; Akasaki, Y.; Murayama, Y.; Yoshida, K.; Sasaki, H. The role of vascular endothelial growth factor in the hypoxic and immunosuppressive tumor microenvironment: Perspectives for therapeutic implications. Med. Oncol. 2019, 37, 2. [Google Scholar] [CrossRef]
- Kaidi, A.; Qualtrough, D.; Williams, A.C.; Paraskeva, C. Direct transcriptional up-regulation of cyclooxygenase-2 by hypoxia-inducible factor (HIF)-1 promotes colorectal tumor cell survival and enhances HIF-1 transcriptional activity during hypoxia. Cancer Res. 2006, 66, 6683–6691. [Google Scholar] [CrossRef]
- Nazet, U.; Schröder, A.; Spanier, G.; Wolf, M.; Proff, P.; Kirschneck, C. Simplified method for applying static isotropic tensile strain in cell culture experiments with identification of valid RT-qPCR reference genes for PDL fibroblasts. Eur. J. Orthod. 2019, 42, 359–370. [Google Scholar] [CrossRef]
- Di Domenico, M.; D’apuzzo, F.; Feola, A.; Cito, L.; Monsurrò, A.; Pierantoni, G.M.; Berrino, L.; de Rosa, A.; Polimeni, A.; Perillo, L. Cytokines and VEGF induction in orthodontic movement in animal models. J. Biomed. Biotechnol. 2012, 2012, 201689. [Google Scholar] [CrossRef]
- Mousavizadeh, R.; Khosravi, S.; Behzad, H.; McCormack, R.G.; Duronio, V.; Scott, A. Cyclic strain alters the expression and release of angiogenic factors by human tendon cells. PLoS ONE 2014, 9, e97356. [Google Scholar] [CrossRef]
- Kirschneck, C.; Küchler, E.C.; Wolf, M.; Spanier, G.; Proff, P.; Schröder, A. Effects of the Highly COX-2-Selective Analgesic NSAID Etoricoxib on Human Periodontal Ligament Fibroblasts during Compressive Orthodontic Mechanical Strain. Mediat. Inflamm. 2019, 2019, 2514956. [Google Scholar] [CrossRef]
- Yue, Y.; Chen, Z.; Xie, B.; Yao, H.-L. Expression of vascular endothelial growth factor in periodontal tissues during orthodontic tooth movement and its role in bone remodeling. Shanghai J. Stomatol. 2018, 27, 18–21. [Google Scholar]
- Zachariae, C.O. Chemotactic cytokines and inflammation. Biological properties of the lymphocyte and monocyte chemotactic factors ELCF, MCAF and IL-8. Acta Derm. Venereol. Suppl. 1993, 181, 1–37. [Google Scholar]
- Brunetti, G.; Colucci, S.; Pignataro, P.; Coricciati, M.; Mori, G.; Cirulli, N.; Zallone, A.; Grassi, F.R.; Grano, M. T cells support osteoclastogenesis in an in vitro model derived from human periodontitis patients. J. Periodontol. 2005, 76, 1675–1680. [Google Scholar] [CrossRef]
- Schröder, A.; Neubert, P.; Titze, J.; Bozec, A.; Neuhofer, W.; Proff, P.; Kirschneck, C.; Jantsch, J. Osteoprotective action of low-salt diet requires myeloid cell-derived NFAT5. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.L.; Urade, Y.; Jakobsson, P.-J. Enzymes of the cyclooxygenase pathways of prostanoid biosynthesis. Chem. Rev. 2011, 111, 5821–5865. [Google Scholar] [CrossRef] [PubMed]
- Pilbeam, C. Prostaglandins and Bone. In Handbook of Experimental Pharmacology; Springer: Berlin, Germany, 2019. [Google Scholar] [CrossRef]
- Ueda, K.; Saito, A.; Nakano, H.; Aoshima, M.; Yokota, M.; Muraoka, R.; Iwaya, T. Cortical hyperostosis following long-term administration of prostaglandin E1 in infants with cyanotic congenital heart disease. J. Pediatr. 1980, 97, 834–836. [Google Scholar] [CrossRef]
- Jee, W.S.; Ma, Y.F. The in vivo anabolic actions of prostaglandins in bone. Bone 1997, 21, 297–304. [Google Scholar] [CrossRef]
- Yoshida, K.; Oida, H.; Kobayashi, T.; Maruyama, T.; Tanaka, M.; Katayama, T.; Yamaguchi, K.; Segi, E.; Tsuboyama, T.; Matsushita, M.; et al. Stimulation of bone formation and prevention of bone loss by prostaglandin E EP4 receptor activation. Proc. Natl. Acad. Sci. USA 2002, 99, 4580–4585. [Google Scholar] [CrossRef]
- Klein, D.C.; Raisz, L.G. Prostaglandins: Stimulation of bone resorption in tissue culture. Endocrinology 1970, 86, 1436–1440. [Google Scholar] [CrossRef]
- Lader, C.S.; Flanagan, A.M. Prostaglandin E2, interleukin 1alpha, and tumor necrosis factor-alpha increase human osteoclast formation and bone resorption in vitro. Endocrinology 1998, 139, 3157–3164. [Google Scholar] [CrossRef] [PubMed]
- Tai, H.; Miyaura, C.; Pilbeam, C.C.; Tamura, T.; Ohsugi, Y.; Koishihara, Y.; Kubodera, N.; Kawaguchi, H.; Raisz, L.G.; Suda, T. Transcriptional induction of cyclooxygenase-2 in osteoblasts is involved in interleukin-6-induced osteoclast formation. Endocrinology 1997, 138, 2372–2379. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sarosi, I.; Yan, X.Q.; Morony, S.; Capparelli, C.; Tan, H.L.; McCabe, S.; Elliott, R.; Scully, S.; Van, G.; et al. RANK is the intrinsic hematopoietic cell surface receptor that controls osteoclastogenesis and regulation of bone mass and calcium metabolism. Proc. Natl. Acad. Sci. USA 2000, 97, 1566–1571. [Google Scholar] [CrossRef] [PubMed]
- Favale, N.O.; Casali, C.I.; Lepera, L.G.; Pescio, L.G.; Fernández-Tome, M.C. Hypertonic induction of COX2 expression requires TonEBP/NFAT5 in renal epithelial cells. Biochem. Biophys. Res. Commun. 2009, 381, 301–305. [Google Scholar] [CrossRef]
- Messerschmidt, L.; Fischer, S.; Wiedemann, P.; Bringmann, A.; Hollborn, M. Osmotic induction of cyclooxygenase-2 in RPE cells: Stimulation of inflammasome activation. Mol. Vis. 2019, 25, 329–344. [Google Scholar]
- Wu, K.K.; Liou, J.-Y.; Cieslik, K. Transcriptional Control of COX-2 via C/EBPbeta. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 679–685. [Google Scholar] [CrossRef]
- Milkiewicz, M.; Doyle, J.L.; Fudalewski, T.; Ispanovic, E.; Aghasi, M.; Haas, T.L. HIF-1alpha and HIF-2alpha play a central role in stretch-induced but not shear-stress-induced angiogenesis in rat skeletal muscle. J. Physiol. 2007, 583, 753–766. [Google Scholar] [CrossRef]
- Neubert, P.; Weichselbaum, A.; Reitinger, C.; Schatz, V.; Schröder, A.; Ferdinand, J.R.; Simon, M.; Bär, A.-L.; Brochhausen, C.; Gerlach, R.G.; et al. HIF1A and NFAT5 coordinate Na+-boosted antibacterial defense via enhanced autophagy and autolysosomal targeting. Autophagy 2019, 15, 1899–1916. [Google Scholar] [CrossRef]
- Vogel, J.; Thiel, C.S.; Tauber, S.; Stockmann, C.; Gassmann, M.; Ullrich, O. Expression of Hypoxia-Inducible Factor 1α (HIF-1α) and Genes of Related Pathways in Altered Gravity. Int. J. Mol. Sci. 2019, 20, 436. [Google Scholar] [CrossRef]
- Brower, J.B.; Targovnik, J.H.; Caplan, M.R.; Massia, S.P. High glucose-mediated loss of cell surface heparan sulfate proteoglycan impairs the endothelial shear stress response. Cytoskeleton 2010, 67, 135–141. [Google Scholar] [CrossRef]
- Shi, Z.-D.; Tarbell, J.M. Fluid flow mechanotransduction in vascular smooth muscle cells and fibroblasts. Ann. Biomed. Eng. 2011, 39, 1608–1619. [Google Scholar] [CrossRef] [PubMed]
- Nikmanesh, M.; Cancel, L.M.; Shi, Z.-D.; Tarbell, J.M. Heparan sulfate proteoglycan, integrin, and syndecan-4 are mechanosensors mediating cyclic strain-modulated endothelial gene expression in mouse embryonic stem cell-derived endothelial cells. Biotechnol. Bioeng. 2019, 116, 2730–2741. [Google Scholar] [CrossRef] [PubMed]
- Saoncella, S.; Echtermeyer, F.; Denhez, F.; Nowlen, J.K.; Mosher, D.F.; Robinson, S.D.; Hynes, R.O.; Goetinck, P.F. Syndecan-4 signals cooperatively with integrins in a Rho-dependent manner in the assembly of focal adhesions and actin stress fibers. Proc. Natl. Acad. Sci. USA 1999, 96, 2805–2810. [Google Scholar] [CrossRef] [PubMed]
- Baeyens, N.; Bandyopadhyay, C.; Coon, B.G.; Yun, S.; Schwartz, M.A. Endothelial fluid shear stress sensing in vascular health and disease. J. Clin. Investig. 2016, 126, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Warford, J.R.; Lamport, A.-C.; Clements, D.R.; Malone, A.; Kennedy, B.E.; Kim, Y.; Gujar, S.A.; Hoskin, D.W.; Easton, A.S. Surfen, a proteoglycan binding agent, reduces inflammation but inhibits remyelination in murine models of Multiple Sclerosis. Acta Neuropathol. Commun. 2018, 6, 4. [Google Scholar] [CrossRef]
- Feller, L.; Khammissa, R.A.G.; Schechter, I.; Moodley, A.; Thomadakis, G.; Lemmer, J. Periodontal Biological Events Associated with Orthodontic Tooth Movement: The Biomechanics of the Cytoskeleton and the Extracellular Matrix. Sci. World J. 2015, 2015, 894123. [Google Scholar] [CrossRef]
- Kirschneck, C.; Batschkus, S.; Proff, P.; Köstler, J.; Spanier, G.; Schröder, A. Valid gene expression normalization by RT-qPCR in studies on hPDL fibroblasts with focus on orthodontic tooth movement and periodontitis. Sci. Rep. 2017, 7, 14751. [Google Scholar] [CrossRef]
- Kirschneck, C.; Meier, M.; Bauer, K.; Proff, P.; Fanghänel, J. Meloxicam medication reduces orthodontically induced dental root resorption and tooth movement velocity: A combined in vivo and in vitro study of dental-periodontal cells and tissue. Cell Tissue Res. 2017, 368, 61–78. [Google Scholar] [CrossRef]
- Schröder, A.; Küchler, E.C.; Omori, M.; Spanier, G.; Proff, P.; Kirschneck, C. Effects of ethanol on human periodontal ligament fibroblasts subjected to static compressive force. Alcohol 2019, 77, 59–70. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Koretsi, V.; Kirschneck, C.; Proff, P.; Römer, P. Expression of glutathione peroxidase 1 in the spheno-occipital synchondrosis and its role in ROS-induced apoptosis. Eur. J. Orthod. 2015, 37, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Kirschneck, C.; Proff, P.; Fanghänel, J.; Wolf, M.; Roldán, J.C.; Römer, P. Reference genes for valid gene expression studies on rat dental, periodontal and alveolar bone tissue by means of RT-qPCR with a focus on orthodontic tooth movement and periodontitis. Ann. Anat. 2016, 204, 93–105. [Google Scholar] [CrossRef] [PubMed]


| Gene Symbol | Gene Name | Accession Number | 5′-Forward Primer-3′ | 5′-Reverse Primer-3′ |
|---|---|---|---|---|
| PPIB | peptidylprolyl isomerase A | NM_000942.4 | TTCCATCGTGTAATCAAGGACTTC | GCTCACCGTAGATGCTCTTTC |
| RPL22 | ribosomal protein L22 | NM_000983.3 | TGATTGCACCCACCCTGTAG | GGTTCCCAGCTTTTCCGTTC |
| TBP | TATA-box-binding protein | NM_003194.4 | CGGCTGTTTAACTTCGCTTCC | TGGGTTATCTTCACACGCCAAG |
| PTGS-2 | prostaglandin endoperoxidase synthase-2 | NM_000963.3 | GAGCAGGCAGATGAAATACCAGTC | TGTCACCATAGAGTGCTTCCAAC |
| PTK-2 | protein tyrosine kinase-2 | NM_001352694.1 | AGCTACAACGAGGGTGTCAAG | TGGGGCTGGCTGGATTTTAC |
| VEGF | vascular endothelial growth factor A | NM_001171623.1 | TGCAGACCAAAGAAAGATAGAGC | ACGCTCCAGGACTTATACCG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirschneck, C.; Thuy, M.; Leikam, A.; Memmert, S.; Deschner, J.; Damanaki, A.; Spanier, G.; Proff, P.; Jantsch, J.; Schröder, A. Role and Regulation of Mechanotransductive HIF-1α Stabilisation in Periodontal Ligament Fibroblasts. Int. J. Mol. Sci. 2020, 21, 9530. https://doi.org/10.3390/ijms21249530
Kirschneck C, Thuy M, Leikam A, Memmert S, Deschner J, Damanaki A, Spanier G, Proff P, Jantsch J, Schröder A. Role and Regulation of Mechanotransductive HIF-1α Stabilisation in Periodontal Ligament Fibroblasts. International Journal of Molecular Sciences. 2020; 21(24):9530. https://doi.org/10.3390/ijms21249530
Chicago/Turabian StyleKirschneck, Christian, Magdalena Thuy, Alexandra Leikam, Svenja Memmert, James Deschner, Anna Damanaki, Gerrit Spanier, Peter Proff, Jonathan Jantsch, and Agnes Schröder. 2020. "Role and Regulation of Mechanotransductive HIF-1α Stabilisation in Periodontal Ligament Fibroblasts" International Journal of Molecular Sciences 21, no. 24: 9530. https://doi.org/10.3390/ijms21249530
APA StyleKirschneck, C., Thuy, M., Leikam, A., Memmert, S., Deschner, J., Damanaki, A., Spanier, G., Proff, P., Jantsch, J., & Schröder, A. (2020). Role and Regulation of Mechanotransductive HIF-1α Stabilisation in Periodontal Ligament Fibroblasts. International Journal of Molecular Sciences, 21(24), 9530. https://doi.org/10.3390/ijms21249530






