Angiotensin-(1-7) Prevents Lipopolysaccharide-Induced Autophagy via the Mas Receptor in Skeletal Muscle
Abstract
1. Introduction
2. Results
2.1. Angiotensin-(1-7) Prevented the Decline of Muscle Function and Tetanic Force via the Mas Receptor in LPS-Treated Mice
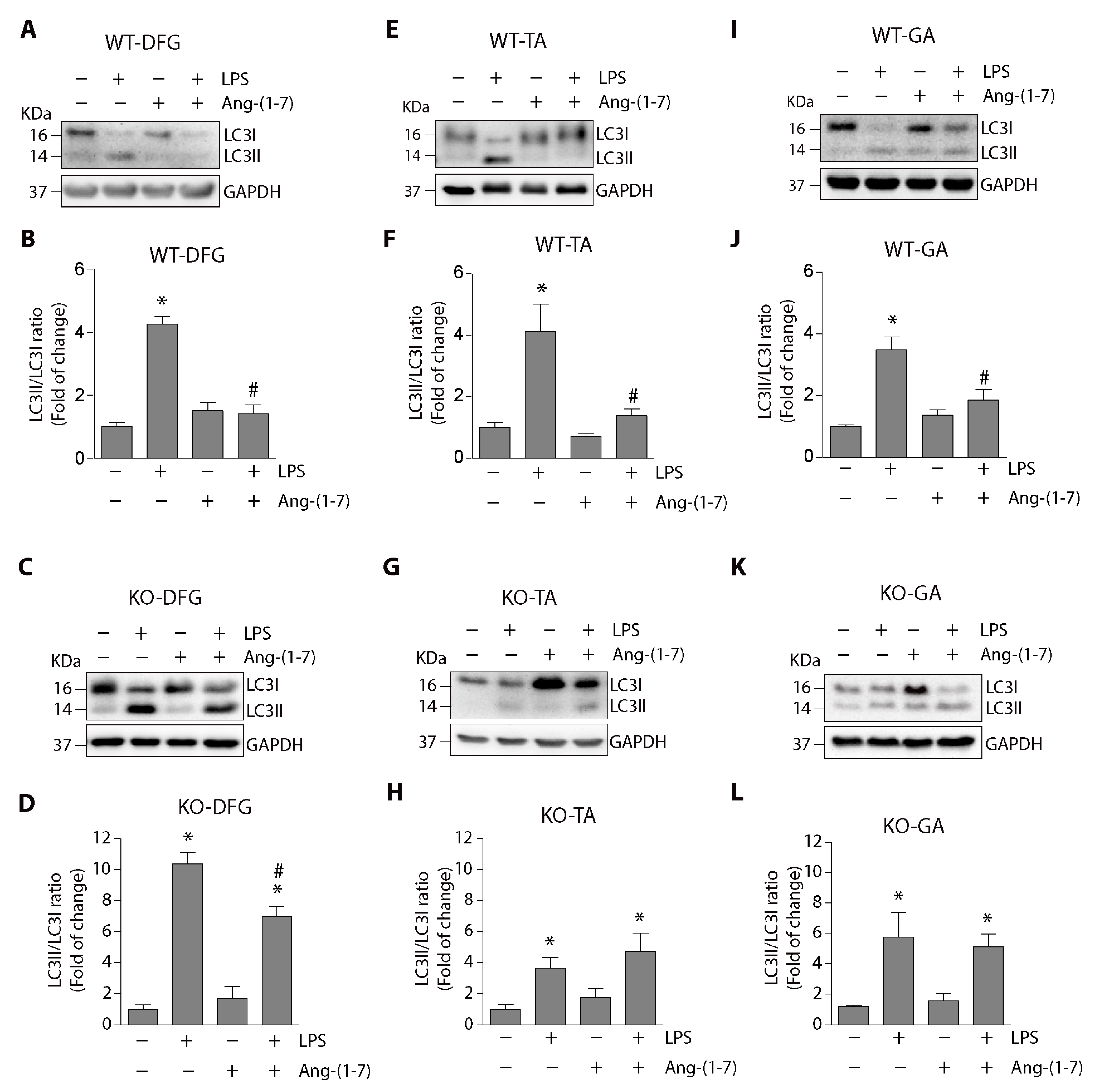
2.2. Angiotensin-(1-7) Decreased LPS-Induced Autophagy in the Diaphragm, Tibialis Anterior, and Gastrocnemius Muscles through a Mas Receptor-Dependent Mechanism
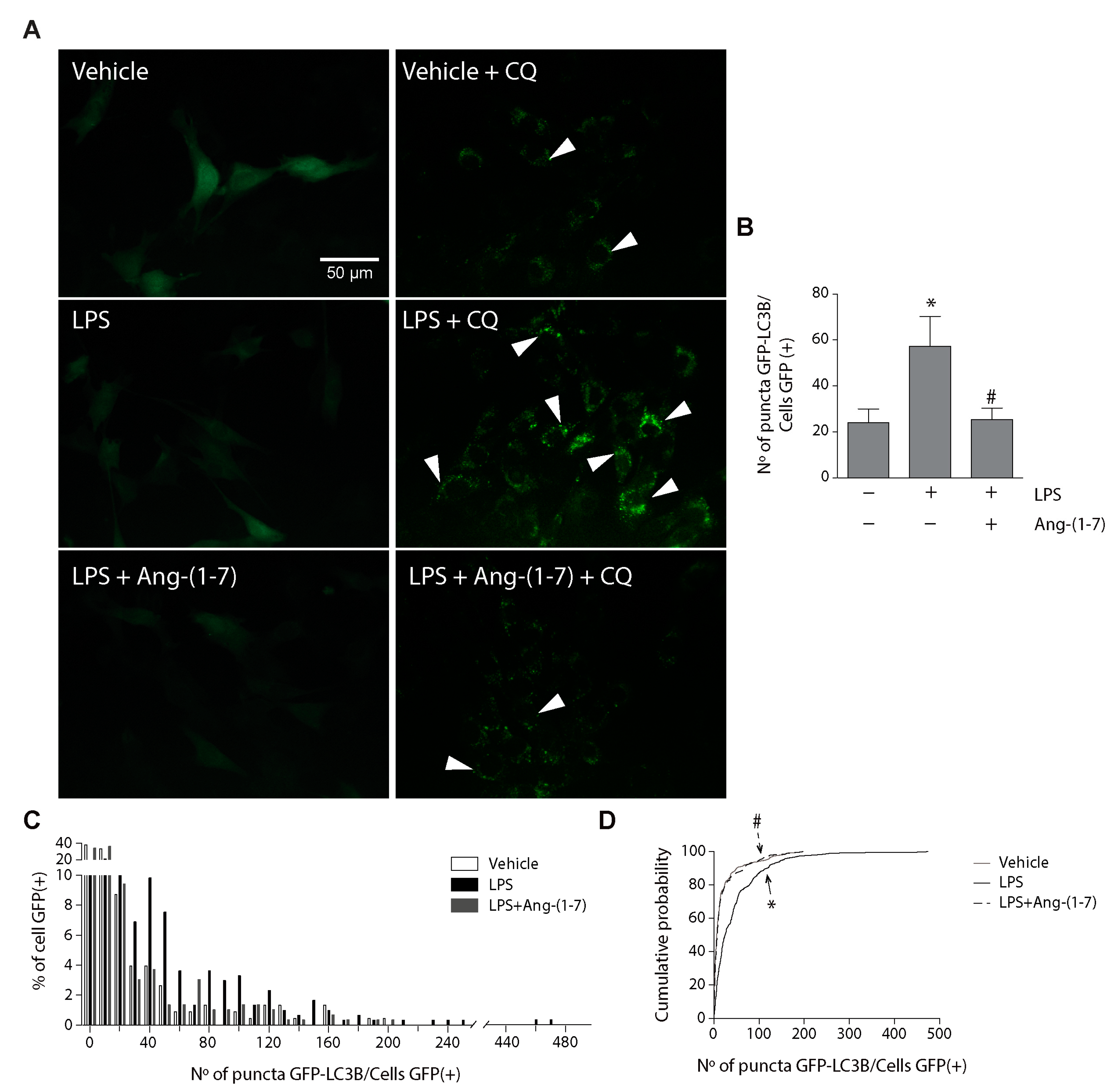
2.3. Angiotensin-(1-7) Decreased LPS-Induced Autophagy in Skeletal Muscle Cells
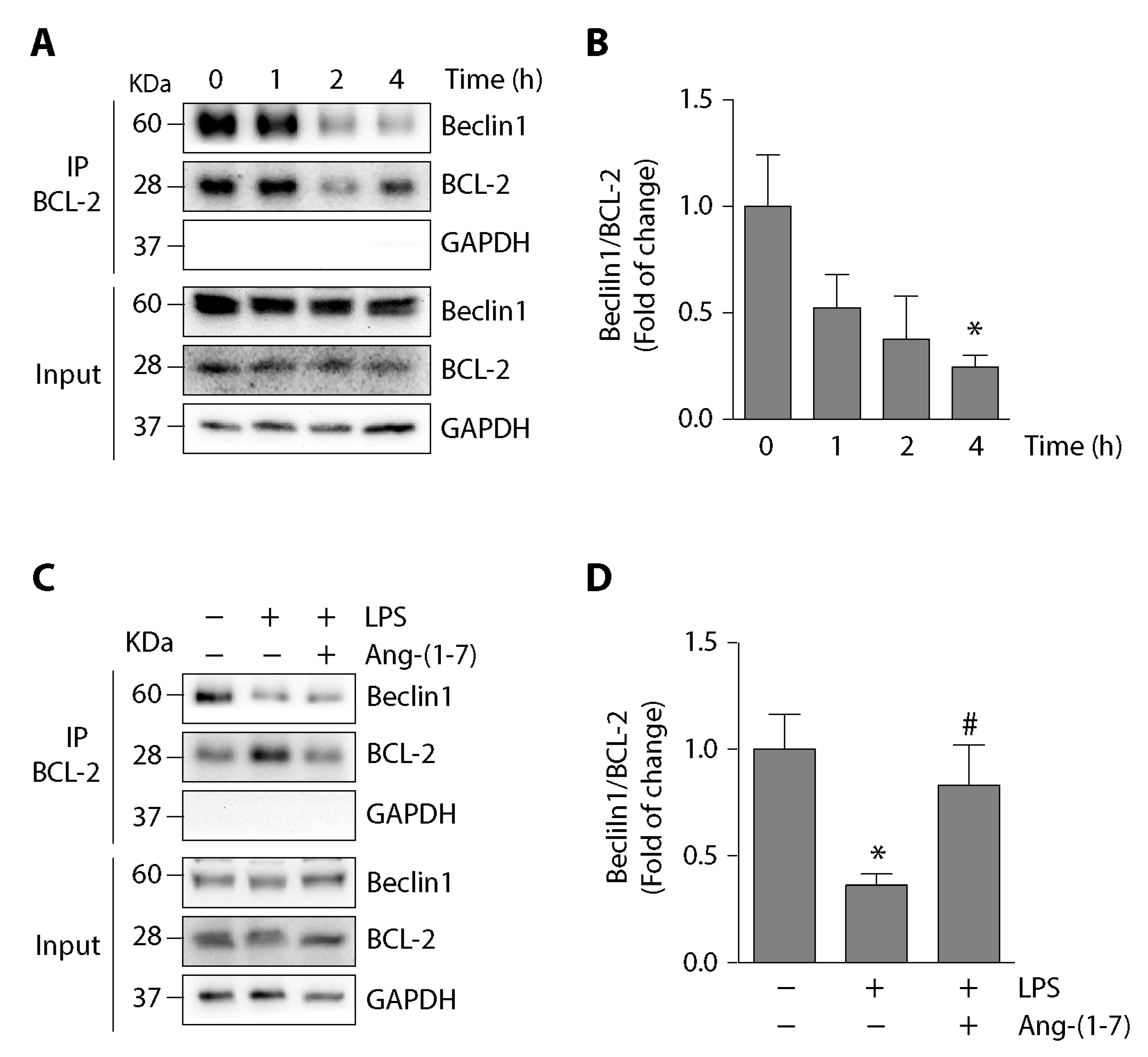
2.4. Angiotensin-(1-7) Prevented the Phosphorylation of p38, JNK, and BCL-2 and the Disassembly of the Beclin1/BCL-2 Complex
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Protocols
4.2. Strength Test
4.3. Contractile Measurements
4.4. Cell Cultures
4.5. Immunoblot Analysis
4.6. Reverse Transcription and Quantitative Real-Time PCR
4.7. Co-Immunoprecipitation Analysis
4.8. Indirect Immunofluorescence
4.9. Adenovirus Transduction and Analysis of GFP-LC3B Puncta
4.10. Autophagy Assay
4.11. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AdV-GFP-LC3 | Adenovirus for GFP-LC3 |
| AMPK | AMP-activated protein kinase |
| Ang-(1-7) | Angiotensin (1-7) |
| Atrogin-1 | atrogin-1/MAFbx |
| AT1 | Angiotensin II receptor type 1 |
| BCL-2 | B-cell lymphoma-2 |
| CQ | Chloroquine |
| DFG | Diaphragm |
| EDTA | Ethylenediaminetetraacetic acid |
| GA | Gastrocnemius |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| GFP | Green fluorescent protein |
| JNK | c-Jun N-terminal kinase |
| LC3B | Microtubule-associated proteins 1A/1B light chain 3B |
| LPS | Lipopolysaccharide |
| kDa | Kilodalton |
| KO | Knock out |
| MAPK | Mitogen-activated protein kinase |
| Mas KO | Mas knockout (deficient in Mas receptor) |
| MOI | Multiplicity of infection |
| MuRF-1 | Muscle RING-finger protein-1 |
| NBR1 | Neighbor of BRCA1 gene 1 |
| OPTN | Optineurin |
| PE | Phosphatidylethanolamine |
| p62/SQSTM1 | Sequestosome 1 |
| PI3P | Phosphatidylinositol 3-phosphate |
| PMSF | Phenylmethylsulfonyl fluoride |
| PVDF | Polyvinylidene fluoride |
| qPCR | Quantitative polymerase chain reaction |
| RAS | Renin-angiotensin system |
| RIPA | Radioimmunoprecipitation assay |
| ROI | Region of interest |
| ROS | Reactive oxygen species |
| RT | Reverse transcription |
| RAS | Renin angiotensin system |
| SHP-1 | Src homology 2 domain-containing protein tyrosine phosphatase 1 |
| TA | Tibialis anterior |
| UPS | Ubiquitin proteasome system |
| WT | Wild-type |
References
- Jackman, R.W.; Kandarian, S.C. The molecular basis of skeletal muscle atrophy. Am. J. Physiol. Cell Physiol. 2004, 287, C834–C843. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.J. Skeletal muscle loss: Cachexia, sarcopenia, and inactivity. Am. J. Clin. Nutr. 2010, 91, 1123S–1127S. [Google Scholar] [CrossRef] [PubMed]
- Sandri, M. Protein breakdown in muscle wasting: Role of autophagy-lysosome and ubiquitin-proteasome. Int. J. Biochem. Cell Biol. 2013, 45, 2121–2129. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Nathan, J.A.; Goldberg, A.L. Muscle wasting in disease: Molecular mechanisms and promising therapies. Nat. Rev. Drug Discov. 2015, 14, 58–74. [Google Scholar] [CrossRef] [PubMed]
- Raetz, C.R.; Whitfield, C. Lipopolysaccharide endotoxins. Annu. Rev. Biochem. 2002, 71, 635–700. [Google Scholar] [CrossRef]
- Fong, Y.; Moldawer, L.L.; Marano, M.; Wei, H.; Barber, A.; Manogue, K.; Tracey, K.J.; Kuo, G.; Fischman, D.A.; Cerami, A.; et al. Cachectin/TNF or IL-1 alpha induces cachexia with redistribution of body proteins. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1989, 256, R659–R665. [Google Scholar] [CrossRef]
- Lang, C.H.; Frost, R.A.; Jefferson, L.S.; Kimball, S.R.; Vary, T.C. Endotoxin-induced decrease in muscle protein synthesis is associated with changes in eIF2B, eIF4E, and IGF-I. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E1133–E1143. [Google Scholar] [CrossRef]
- Kim, H.C.; Cho, H.-Y.; Hah, Y.-S. Role of IL-15 in Sepsis-Induced Skeletal Muscle Atrophy and Proteolysis. Tuberc. Respir. Dis. 2012, 73, 312–319. [Google Scholar] [CrossRef][Green Version]
- Morales, M.G.; Olguin, H.; Di Capua, G.; Brandan, E.; Simon, F.; Cabello-Verrugio, C. Endotoxin-induced skeletal muscle wasting is prevented by angiotensin-(1-7) through a p38 MAPK-dependent mechanism. Clin. Sci. (Lond.) 2015, 129, 461–476. [Google Scholar] [CrossRef]
- Unroe, M.; Kahn, J.M.; Carson, S.S.; Govert, J.A.; Martinu, T.; Sathy, S.J.; Clay, A.S.; Chia, J.; Gray, A.; Tulsky, J.A.; et al. One-year trajectories of care and resource utilization for recipients of prolonged mechanical ventilation: A cohort study. Ann. Intern. Med. 2010, 153, 167–175. [Google Scholar] [CrossRef]
- Doyle, A.; Zhang, G.; Abdel Fattah, E.A.; Eissa, N.T.; Li, Y.P. Toll-like receptor 4 mediates lipopolysaccharide-induced muscle catabolism via coordinate activation of ubiquitin-proteasome and autophagy-lysosome pathways. FASEB J. 2011, 25, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Mofarrahi, M.; Sigala, I.; Guo, Y.; Godin, R.; Davis, E.C.; Petrof, B.; Sandri, M.; Burelle, Y.; Hussain, S.N. Autophagy and skeletal muscles in sepsis. PLoS ONE 2012, 7, e47265. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N.; Komatsu, M. Autophagy: Renovation of Cells and Tissues. Cell 2011, 147, 728–741. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Klionsky, D.J. Autophagosome formation: Core machinery and adaptations. Nat. Cell Biol. 2007, 9, 1102–1109. [Google Scholar] [CrossRef]
- Klionsky, D.J.; Abdelmohsen, K.; Abe, A.; Abedin, M.J.; Abeliovich, H.; Acevedo Arozena, A.; Adachi, H.; Adams, C.M.; Adams, P.D.; Adeli, K.; et al. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 2016, 12, 1–222. [Google Scholar] [CrossRef]
- Mizushima, N.; Yoshimori, T.; Levine, B. Methods in Mammalian Autophagy Research. Cell 2010, 140, 313–326. [Google Scholar] [CrossRef]
- Wei, Y.; Sinha, S.; Levine, B. Dual role of JNK1-mediated phosphorylation of Bcl-2 in autophagy and apoptosis regulation. Autophagy 2008, 4, 949–951. [Google Scholar] [CrossRef]
- Zhou, Y.Y.; Li, Y.; Jiang, W.Q.; Zhou, L.F. MAPK/JNK signalling: A potential autophagy regulation pathway. Biosci. Rep. 2015, 35, e00199. [Google Scholar] [CrossRef]
- Wei, Y.; An, Z.; Zou, Z.; Sumpter, R.; Su, M.; Zang, X.; Sinha, S.; Gaestel, M.; Levine, B. The stress-responsive kinases MAPKAPK2/MAPKAPK3 activate starvation-induced autophagy through Beclin 1 phosphorylation. eLife 2015, 4, e05289. [Google Scholar] [CrossRef]
- Liang, H.; Hussey, S.E.; Sanchez-Avila, A.; Tantiwong, P.; Musi, N. Effect of Lipopolysaccharide on Inflammation and Insulin Action in Human Muscle. PLoS ONE 2013, 8, e63983. [Google Scholar] [CrossRef]
- Cabello-Verrugio, C.; Rivera, J.C.; Garcia, D. Skeletal muscle wasting: New role of nonclassical renin-angiotensin system. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.G.; Abrigo, J.; Meneses, C.; Cisternas, F.; Simon, F.; Cabello-Verrugio, C. Expression of the Mas receptor is upregulated in skeletal muscle wasting. Histochem. Cell Biol. 2015, 143, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.A.S.; e Silva, A.C.S.; Maric, C.; Silva, D.M.R.; Machado, R.P.; de Buhr, I.; Heringer-Walther, S.; Pinheiro, S.V.B.; Lopes, M.T.; Bader, M.; et al. Angiotensin-(1–7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc. Natl. Acad. Sci. USA 2003, 100, 8258–8263. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.G.; Abrigo, J.; Acuna, M.J.; Santos, R.A.; Bader, M.; Brandan, E.; Simon, F.; Olguin, H.; Cabrera, D.; Cabello-Verrugio, C. Angiotensin-(1-7) attenuates disuse skeletal muscle atrophy in mice via its receptor, Mas. Dis. Models Mech. 2016, 9, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Cisternas, F.; Morales, M.G.; Meneses, C.; Simon, F.; Brandan, E.; Abrigo, J.; Vazquez, Y.; Cabello-Verrugio, C. Angiotensin-(1-7) decreases skeletal muscle atrophy induced by angiotensin II through a Mas receptor-dependent mechanism. Clin. Sci. (Lond.) 2015, 128, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Kabeya, Y.; Mizushima, N.; Ueno, T.; Yamamoto, A.; Kirisako, T.; Noda, T.; Kominami, E.; Ohsumi, Y.; Yoshimori, T. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000, 19, 5720–5728. [Google Scholar] [CrossRef]
- Wei, Y.; Pattingre, S.; Sinha, S.; Bassik, M.; Levine, B. JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol. Cell 2008, 30, 678–688. [Google Scholar] [CrossRef]
- Cabello-Verrugio, C.; Morales, M.G.; Rivera, J.C.; Cabrera, D.; Simon, F. Renin-angiotensin system: An old player with novel functions in skeletal muscle. Med. Res. Rev. 2015, 35, 437–463. [Google Scholar] [CrossRef]
- Meneses, C.; Morales, M.G.; Abrigo, J.; Simon, F.; Brandan, E.; Cabello-Verrugio, C. The angiotensin-(1-7)/Mas axis reduces myonuclear apoptosis during recovery from angiotensin II-induced skeletal muscle atrophy in mice. Pflug. Arch. 2015, 467, 1975–1984. [Google Scholar] [CrossRef]
- Pan, M.; Zheng, Z.; Chen, Y.; Sun, N.; Zheng, B.; Yang, Q.; Zhang, Y.; Li, X.; Meng, Y. Angiotensin-(1-7) Attenuated Cigarette Smoking–related Pulmonary Fibrosis via Improving the Impaired Autophagy Caused by Nicotinamide Adenine Dinucleotide Phosphate Reduced Oxidase 4–Dependent Reactive Oxygen Species. Am. J. Respir. Cell Mol. Biol. 2018, 59, 306–319. [Google Scholar] [CrossRef]
- Lin, L.; Liu, X.; Xu, J.; Weng, L.; Ren, J.; Ge, J.; Zou, Y. Mas receptor mediates cardioprotection of angiotensin-(1-7) against Angiotensin II-induced cardiomyocyte autophagy and cardiac remodelling through inhibition of oxidative stress. J. Cell. Mol. Med. 2016, 20, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Gao, L.; Zhu, X.-C.; Yu, J.-T.; Shi, J.-Q.; Tan, M.-S.; Lu, J.; Tan, L.; Zhang, Y.-D. Angiotensin-(1–7) inhibits autophagy in the brain of spontaneously hypertensive rats. Pharmacol. Res. 2013, 71, 61–68. [Google Scholar] [CrossRef]
- Silva, K.A.S.; Ghiarone, T.; Schreiber, K.; Grant, D.; White, T.; Frisard, M.I.; Sukhanov, S.; Chandrasekar, B.; Delafontaine, P.; Yoshida, T. Angiotensin II suppresses autophagy and disrupts ultrastructural morphology and function of mitochondria in mouse skeletal muscle. J. Appl. Physiol. (1985) 2019, 126, 1550–1562. [Google Scholar] [CrossRef] [PubMed]
- Masiero, E.; Agatea, L.; Mammucari, C.; Blaauw, B.; Loro, E.; Komatsu, M.; Metzger, D.; Reggiani, C.; Schiaffino, S.; Sandri, M. Autophagy is required to maintain muscle mass. Cell Metab. 2009, 10, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Neel, B.A.; Lin, Y.; Pessin, J.E. Skeletal muscle autophagy: A new metabolic regulator. Trends Endocrinol. Metab. 2013, 24, 635–643. [Google Scholar] [CrossRef]
- Xu, Y.; Jagannath, C.; Liu, X.-D.; Sharafkhaneh, A.; Kolodziejska, K.E.; Eissa, N.T. Toll-like Receptor 4 Is a Sensor for Autophagy Associated with Innate Immunity. Immunity 2007, 27, 135–144. [Google Scholar] [CrossRef]
- Shi, C.-S.; Kehrl, J.H. MyD88 and Trif Target Beclin 1 to Trigger Autophagy in Macrophages. J. Biol. Chem. 2008, 283, 33175–33182. [Google Scholar] [CrossRef]
- Shiota, C.; Abe, T.; Kawai, N.; Ohno, A.; Teshima-Kondo, S.; Mori, H.; Terao, J.; Tanaka, E.; Nikawa, T. Flavones Inhibit LPS-Induced Atrogin-1/MAFbx Expression in Mouse C2C12 Skeletal Myotubes. J. Nutr. Sci. Vitaminol. 2015, 61, 188–194. [Google Scholar] [CrossRef]
- Gava, E.; Samad-Zadeh, A.; Zimpelmann, J.; Bahramifarid, N.; Kitten, G.T.; Santos, R.A.; Touyz, R.M.; Burns, K.D. Angiotensin-(1-7) activates a tyrosine phosphatase and inhibits glucose-induced signalling in proximal tubular cells. Nephrol. Dial. Transplant. 2009, 24, 1766–1773. [Google Scholar] [CrossRef]
- Gopallawa, I.; Uhal, B.D. Angiotensin-(1-7)/mas inhibits apoptosis in alveolar epithelial cells through upregulation of MAP kinase phosphatase-2. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 310, L240–L248. [Google Scholar] [CrossRef]
- Pattingre, S.; Tassa, A.; Qu, X.; Garuti, R.; Liang, X.H.; Mizushima, N.; Packer, M.; Schneider, M.D.; Levine, B. Bcl-2 Antiapoptotic Proteins Inhibit Beclin 1-Dependent Autophagy. Cell 2005, 122, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Nikoletopoulou, V.; Markaki, M.; Palikaras, K.; Tavernarakis, N. Crosstalk between apoptosis, necrosis and autophagy. Biochim. Biophys. Acta Mol. Cell Res. 2013, 1833, 3448–3459. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Liu, R.; Dong, X.; Zhong, Q. Beclin orthologs: Integrative hubs of cell signaling, membrane trafficking, and physiology. Trends Cell Biol. 2015, 25, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Inata, Y.; Kikuchi, S.; Samraj, R.S.; Hake, P.W.; O’Connor, M.; Ledford, J.R.; O’Connor, J.; Lahni, P.; Wolfe, V.; Piraino, G.; et al. Autophagy and mitochondrial biogenesis impairment contribute to age-dependent liver injury in experimental sepsis: Dysregulation of AMP-activated protein kinase pathway. FASEB J. 2018, 32, 728–741. [Google Scholar] [CrossRef]
- Kim, J.; Kundu, M.; Viollet, B.; Guan, K.-L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat. Cell Biol. 2011, 13, 132–141. [Google Scholar] [CrossRef]
- Puente, C.; Hendrickson, R.C.; Jiang, X. Nutrient-regulated Phosphorylation of ATG13 Inhibits Starvation-induced Autophagy. J. Biol. Chem. 2016, 291, 6026–6035. [Google Scholar] [CrossRef]
- Gatica, D.; Lahiri, V.; Klionsky, D.J. Cargo recognition and degradation by selective autophagy. Nat. Cell Biol. 2018, 20, 233–242. [Google Scholar] [CrossRef]
- Xu, Y.; Shen, J.; Ran, Z. Emerging views of mitophagy in immunity and autoimmune diseases. Autophagy 2020, 16, 3–17. [Google Scholar] [CrossRef]
- Yao, R.Q.; Ren, C.; Xia, Z.F.; Yao, Y.M. Organelle-specific autophagy in inflammatory diseases: A potential therapeutic target underlying the quality control of multiple organelles. Autophagy 2020, 1–17. [Google Scholar] [CrossRef]
- Nalbandian, A.; Llewellyn, K.J.; Nguyen, C.; Yazdi, P.G.; Kimonis, V.E. Rapamycin and Chloroquine: The In Vitro and In Vivo Effects of Autophagy-Modifying Drugs Show Promising Results in Valosin Containing Protein Multisystem Proteinopathy. PLoS ONE 2015, 10, e0122888. [Google Scholar] [CrossRef]
- Nicot, A.-S.; Lo Verso, F.; Ratti, F.; Pilot-Storck, F.; Streichenberger, N.; Sandri, M.; Schaeffer, L.; Goillot, E. Phosphorylation of NBR1 by GSK3 modulates protein aggregation. Autophagy 2014, 10, 1036–1053. [Google Scholar] [CrossRef]
- Fuqua, J.D.; Mere, C.P.; Kronemberger, A.; Blomme, J.; Bae, D.; Turner, K.D.; Harris, M.P.; Scudese, E.; Edwards, M.; Ebert, S.M.; et al. ULK2 is essential for degradation of ubiquitinated protein aggregates and homeostasis in skeletal muscle. FASEB J. 2019, 33, 11735–11745. [Google Scholar] [CrossRef] [PubMed]
- Walther, T.; Balschun, D.; Voigt, J.-P.; Fink, H.; Zuschratter, W.; Birchmeier, C.; Ganten, D.; Bader, M. Sustained Long Term Potentiation and Anxiety in Mice Lacking the Mas Protooncogene. J. Biol. Chem. 1998, 273, 11867–11873. [Google Scholar] [CrossRef] [PubMed]
- Boczkowski, J.; Lanone, S.; Ungureanu-Longrois, D.; Danialou, G.; Fournier, T.; Aubier, M. Induction of diaphragmatic nitric oxide synthase after endotoxin administration in rats: Role on diaphragmatic contractile dysfunction. J. Clin. Investig. 1996, 98, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Campos, F.; Abrigo, J.; Aguirre, F.; Garces, B.; Arrese, M.; Karpen, S.; Cabrera, D.; Andia, M.E.; Simon, F.; Cabello-Verrugio, C. Sarcopenia in a mice model of chronic liver disease: Role of the ubiquitin-proteasome system and oxidative stress. Pflug. Arch. 2018, 470, 1503–1519. [Google Scholar] [CrossRef]
- Fajardo, V.A.; Smith, I.C.; Bombardier, E.; Chambers, P.J.; Quadrilatero, J.; Tupling, A.R. Diaphragm assessment in mice overexpressing phospholamban in slow-twitch type I muscle fibers. Brain Behav. 2016, 6, e00470. [Google Scholar] [CrossRef]
- Acuña, M.; Pessina, P.; Olguin, H.; Cabrera, D.; Vio, C.; Bader, M.; Muñoz-Canoves, P.; Santos, R.; Cabello-Verrugio, C.; Brandan, E. Restoration of muscle strength in dystrophic muscle by angiotensin-1-7 through inhibition of TGF-β signalling. Hum. Mol. Genet. 2014, 23, 1237–1249. [Google Scholar] [CrossRef]
- Yaffe, D.; Saxel, O. Serial passaging and differentiation of myogenic cells isolated from dystrophic mouse muscle. Nature 1977, 270, 725–727. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Abrigo, J.; Simon, F.; Cabrera, D.; Cabello-Verrugio, C. Angiotensin-(1-7) Prevents Skeletal Muscle Atrophy Induced by Transforming Growth Factor Type Beta (TGF-beta) via Mas Receptor Activation. Cell Physiol. Biochem. 2016, 40, 27–38. [Google Scholar] [CrossRef]
- Köchl, R.; Hu, X.W.; Chan, E.Y.W.; Tooze, S.A. Microtubules Facilitate Autophagosome Formation and Fusion of Autophagosomes with Endosomes. Traffic 2006, 7, 129–145. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivera, J.C.; Abrigo, J.; Tacchi, F.; Simon, F.; Brandan, E.; Santos, R.A.; Bader, M.; Chiong, M.; Cabello-Verrugio, C. Angiotensin-(1-7) Prevents Lipopolysaccharide-Induced Autophagy via the Mas Receptor in Skeletal Muscle. Int. J. Mol. Sci. 2020, 21, 9344. https://doi.org/10.3390/ijms21249344
Rivera JC, Abrigo J, Tacchi F, Simon F, Brandan E, Santos RA, Bader M, Chiong M, Cabello-Verrugio C. Angiotensin-(1-7) Prevents Lipopolysaccharide-Induced Autophagy via the Mas Receptor in Skeletal Muscle. International Journal of Molecular Sciences. 2020; 21(24):9344. https://doi.org/10.3390/ijms21249344
Chicago/Turabian StyleRivera, Juan Carlos, Johanna Abrigo, Franco Tacchi, Felipe Simon, Enrique Brandan, Robson A. Santos, Michael Bader, Mario Chiong, and Claudio Cabello-Verrugio. 2020. "Angiotensin-(1-7) Prevents Lipopolysaccharide-Induced Autophagy via the Mas Receptor in Skeletal Muscle" International Journal of Molecular Sciences 21, no. 24: 9344. https://doi.org/10.3390/ijms21249344
APA StyleRivera, J. C., Abrigo, J., Tacchi, F., Simon, F., Brandan, E., Santos, R. A., Bader, M., Chiong, M., & Cabello-Verrugio, C. (2020). Angiotensin-(1-7) Prevents Lipopolysaccharide-Induced Autophagy via the Mas Receptor in Skeletal Muscle. International Journal of Molecular Sciences, 21(24), 9344. https://doi.org/10.3390/ijms21249344





