Effects of Sphingosine-1-Phosphate on Cell Viability, Differentiation, and Gene Expression of Adipocytes
Abstract
1. Introduction
2. Results
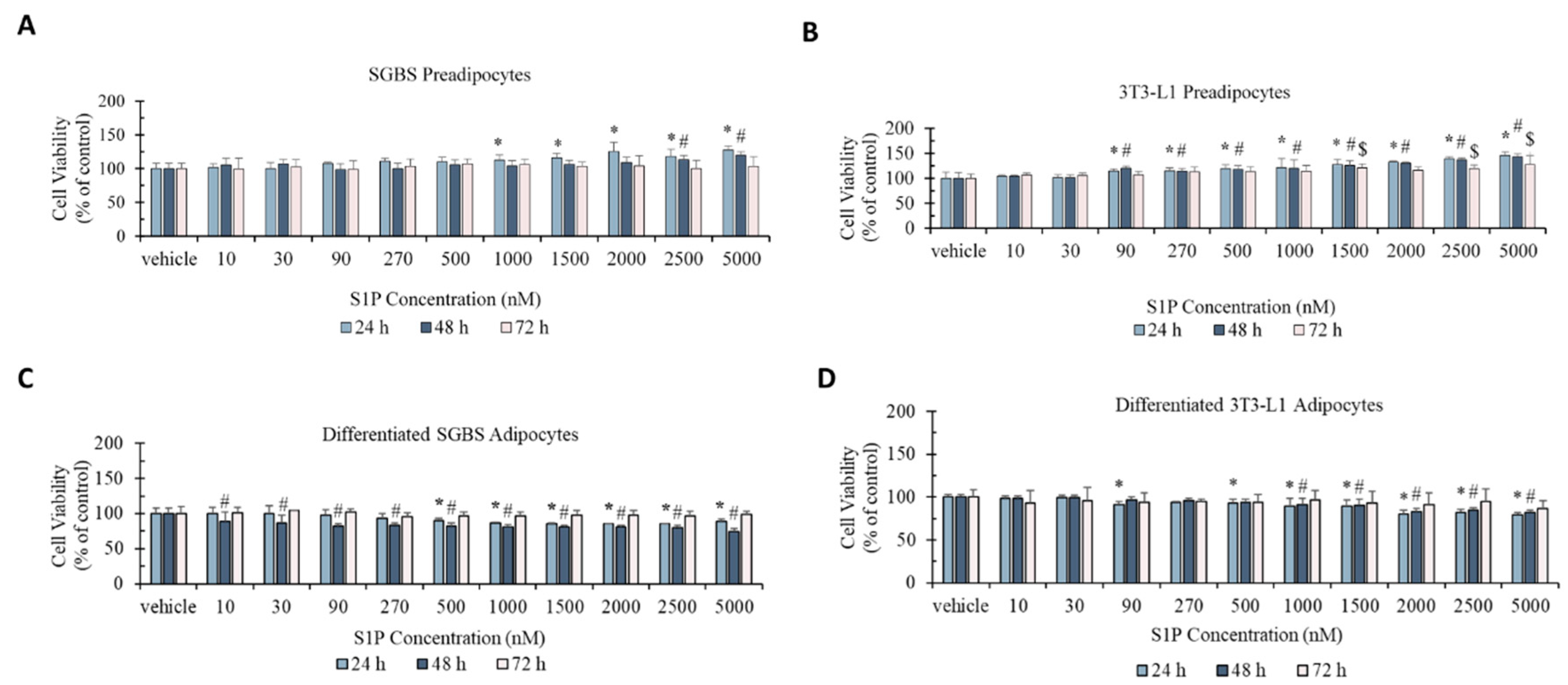
2.1. Effect of S1P on Cell Viability of Pre- and Differentiated Adipocytes
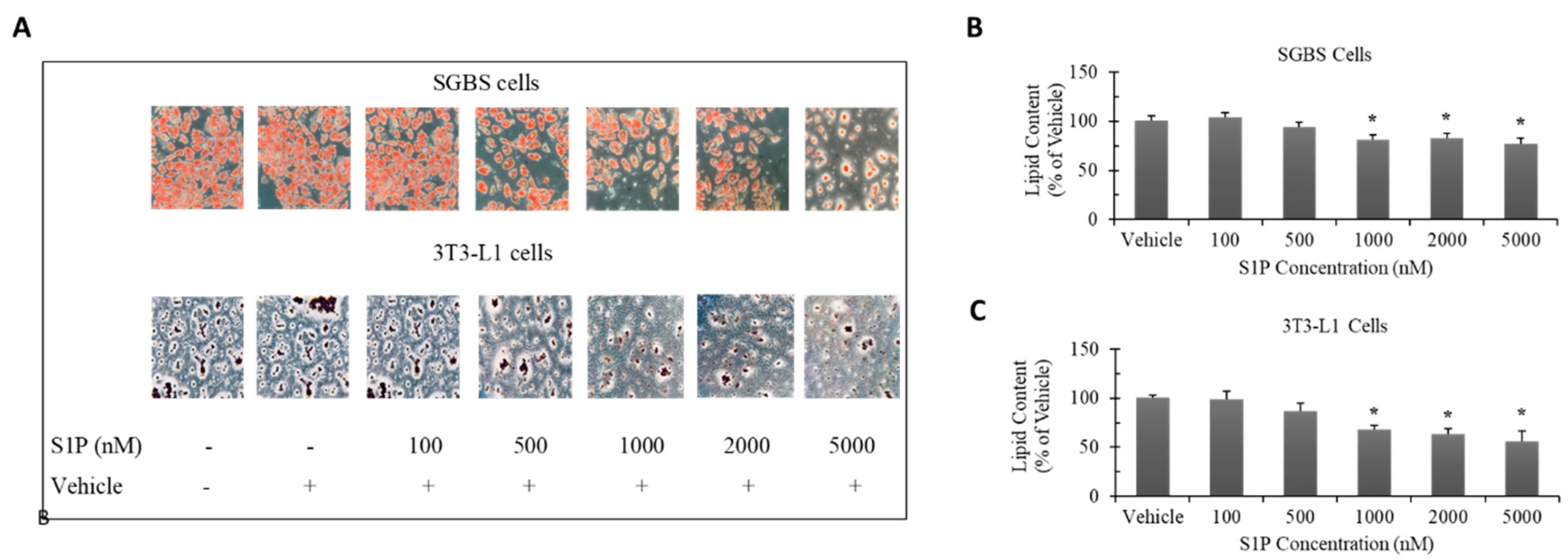
2.2. Effect of S1P on Adipocyte Differentiation
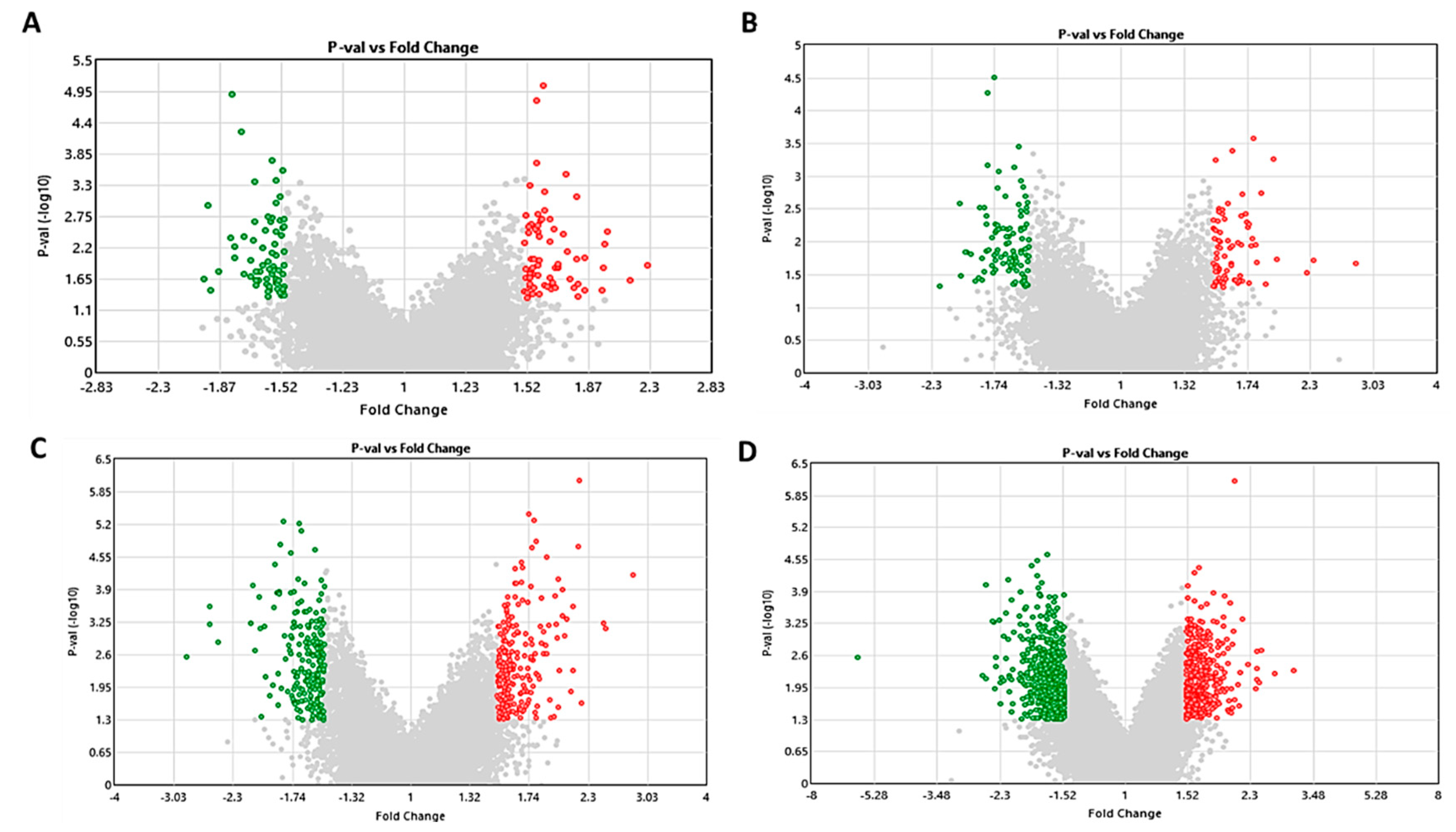
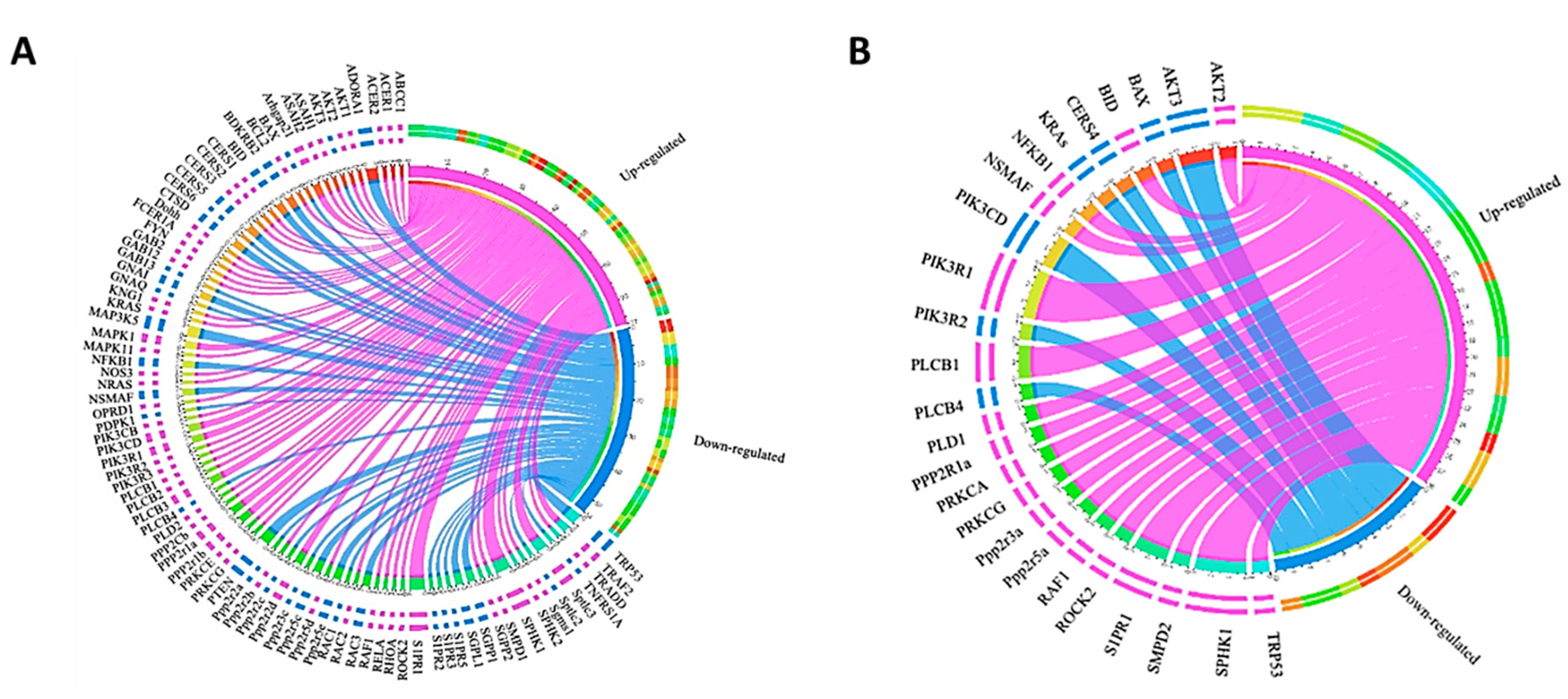
2.3. Effect of S1P on Gene Expression in Pre- and Differentiated Adipocytes
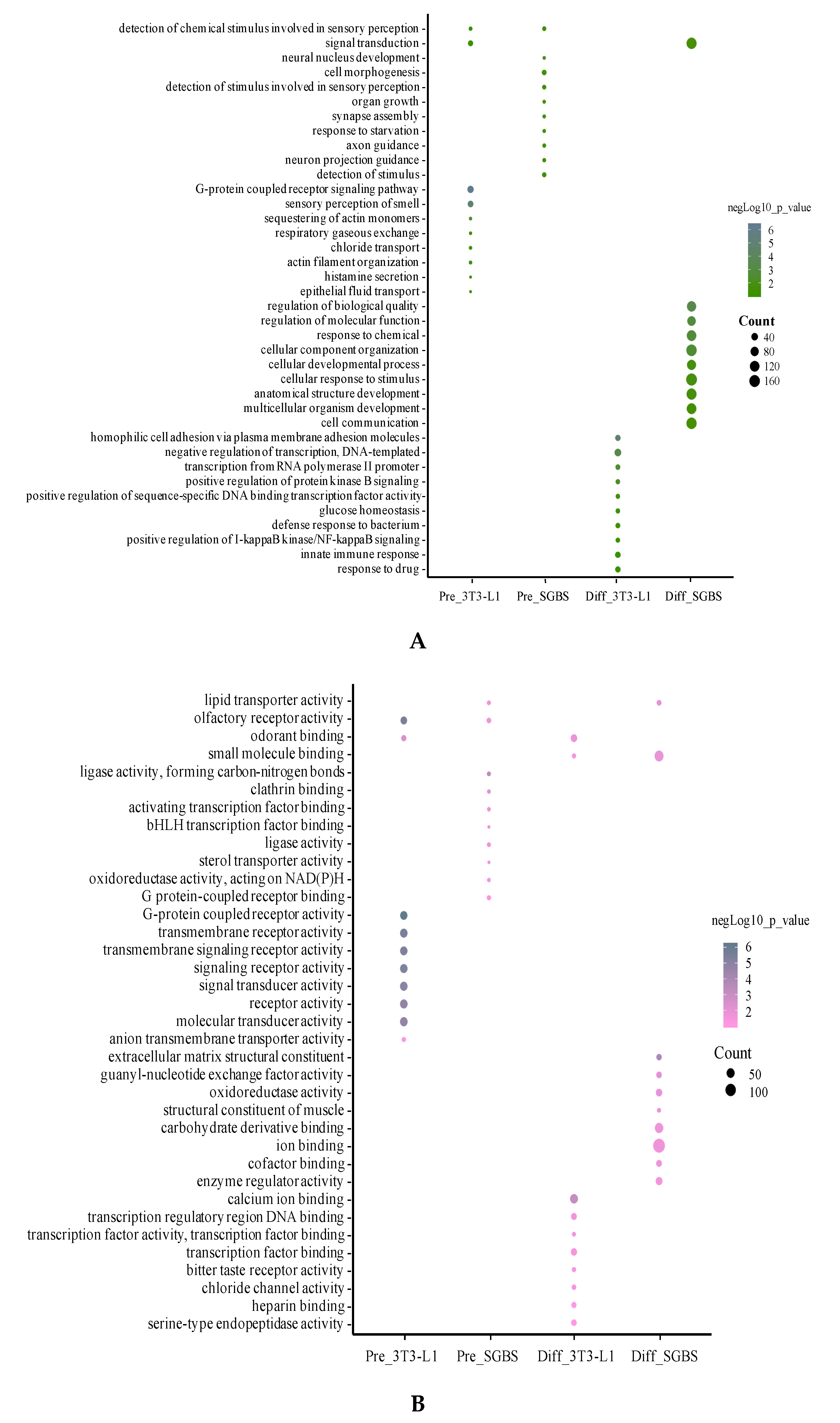
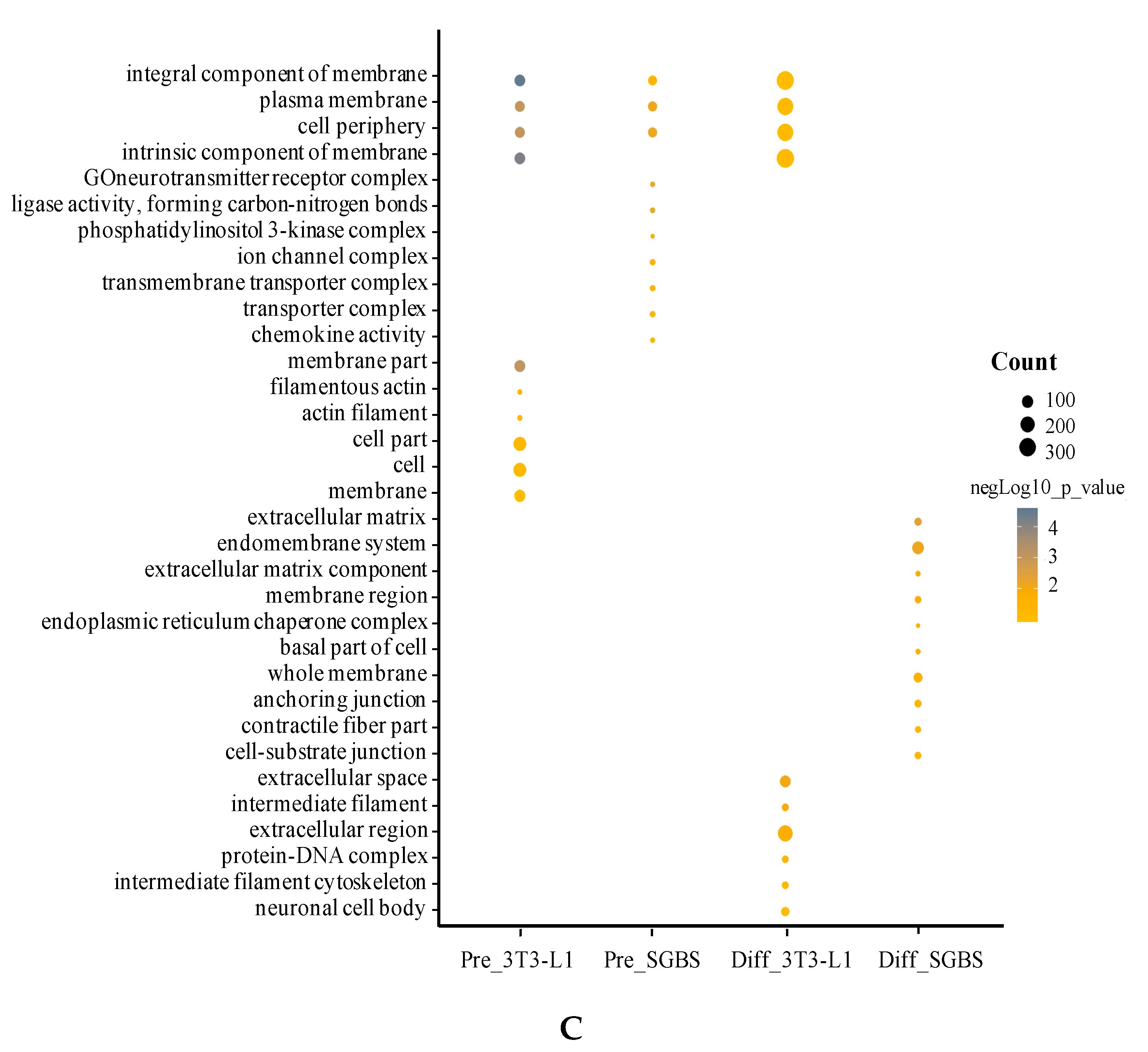
2.4. Gene Ontology (GO) Analysis
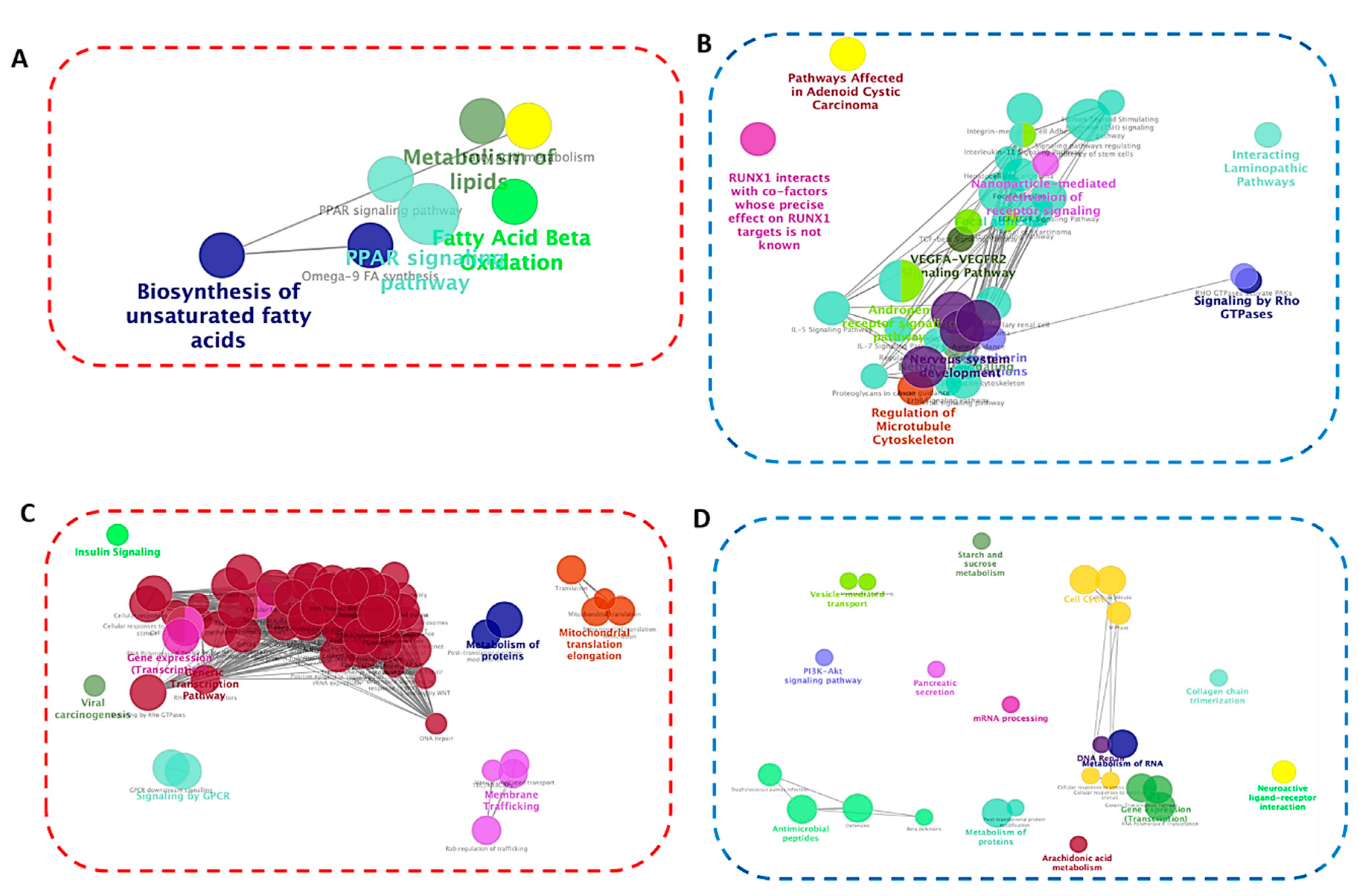
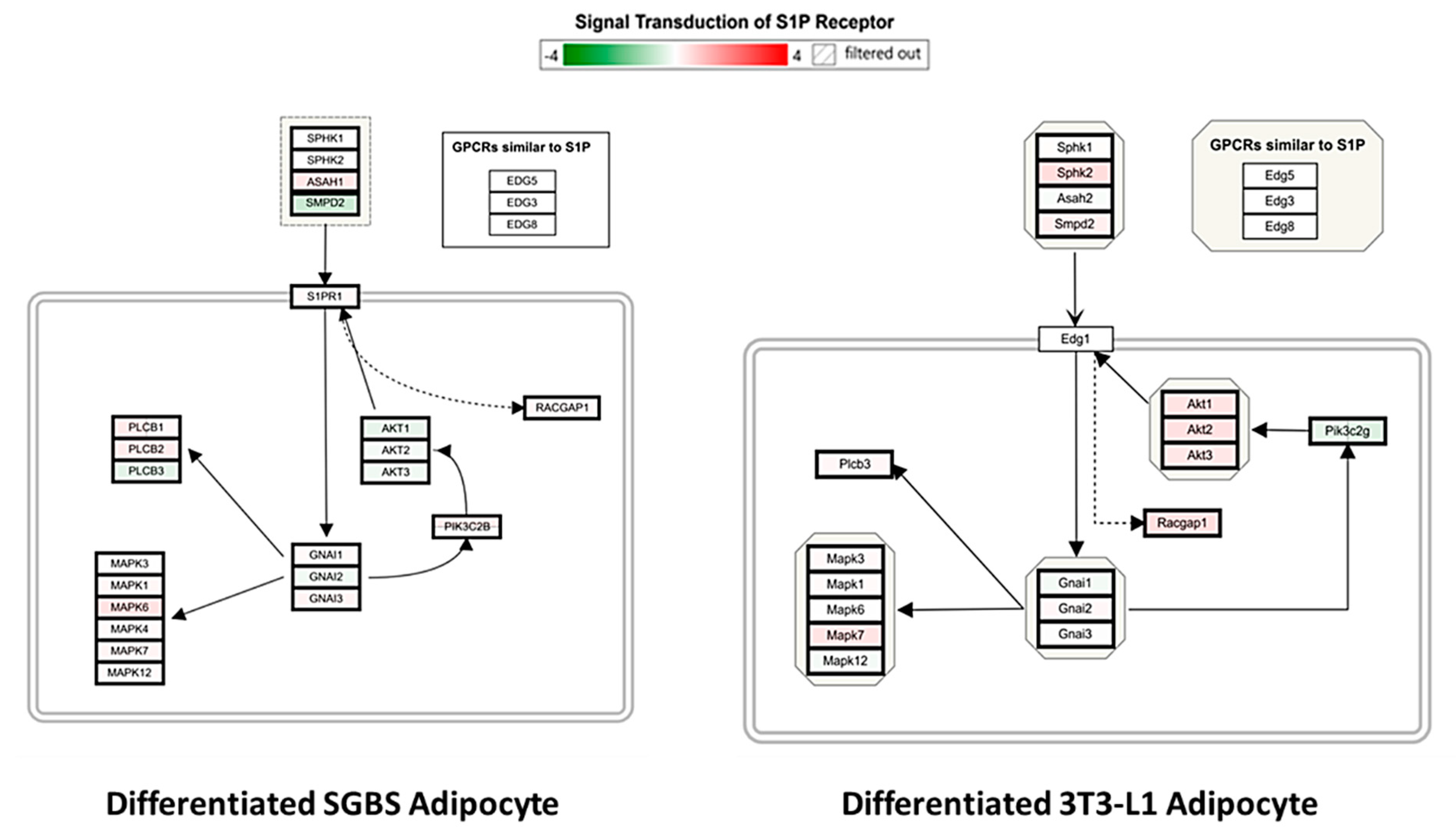
2.5. Pathway Entichment Analysis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture of Preadipocytes
4.3. Adipocyte Differentiation
4.4. Cell Viability Assay (MTT Assay)
4.5. Oil Red O Staining
4.6. Transcriptome Analysis
4.7. DEG Analysis
4.8. Gene Ontology (GO) and Pathway Enrichment Analyses
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cartier, A.; Hla, T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy. Science 2019, 366, eaar5551. [Google Scholar] [CrossRef]
- Pyne, N.J.; Adams, D.R.; Pyne, N.J. Sphingosine 1-phosphate and sphingosine kinases in health and disease: Recent advances. Prog. Lipid Res. 2016, 62, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Takabe, K.; Paugh, S.W.; Milstien, S.; Spiegel, S. “Inside-out” signaling of sphingosine-1-phosphate: Therapeutic targets. Pharmacol. Rev. 2008, 60, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.-C.; Wang, E.Y.; Yi, Y.; Thakur, A.; Tsai, S.-H.; Hoodless, P.A. S1P Stimulates Proliferation by Upregulating CTGF Expression through S1PR2-Mediated YAP Activation. Mol. Cancer Res. 2018, 16, 1543–1555. [Google Scholar] [CrossRef]
- Baeyens, A.A.; Schwab, S.R. Finding a Way Out: S1P Signaling and Immune Cell Migration. Annu. Rev. Immunol. 2020, 38, 759–784. [Google Scholar] [CrossRef] [PubMed]
- Rohrbach, T.; Maceyka, M.; Spiegel, S. Sphingosine kinase and sphingosine-1-phosphate in liver pathobiology. Crit. Rev. Biochem. Mol. Biol. 2017, 52, 543–553. [Google Scholar] [CrossRef]
- Dai, L.; Liu, Y.; Xie, L.; Wu, X.; Qiu, L.; Di, W. Sphingosine kinase 1/sphingosine-1-phosphate (S1P)/S1P receptor axis is involved in ovarian cancer angiogenesis. Oncotarget 2017, 8, 74947–74961. [Google Scholar] [CrossRef]
- Hait, N.C.; Allegood, J.; Maceyka, M.; Strub, G.M.; Harikumar, K.B.; Singh, S.K.; Luo, C.; Marmorstein, R.; Kordula, T.; Milstien, S.; et al. Regulation of Histone Acetylation in the Nucleus by Sphingosine-1-Phosphate. Science 2009, 325, 1254–1257. [Google Scholar] [CrossRef]
- Maceyka, M.; Harikumar, K.B.; Milstien, S.; Spiegel, S. Sphingosine-1-phosphate signaling and its role in disease. Trends Cell Biol. 2012, 22, 50–60. [Google Scholar] [CrossRef]
- Ebenezer, D.L.; Fu, P.; Suryadevara, V.; Zhao, Y.; Natarajan, V. Epigenetic regulation of pro-inflammatory cytokine secretion by sphingosine 1-phosphate (S1P) in acute lung injury: Role of S1P lyase. Adv. Biol. Regul. 2016, 63, 156–166. [Google Scholar] [CrossRef]
- Sankala, H.M.; Hait, N.C.; Paugh, S.W.; Shida, D.; Elmore, L.W.; Dent, P.; Milstien, S.; Spiegel, S.; Lepine, S. Involvement of Sphingosine Kinase 2 in p53-Independent Induction of p21 by the Chemotherapeutic Drug Doxorubicin. Cancer Res. 2007, 67, 10466–10474. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, N.; Okada, T.; Hayashi, S.; Fujita, T.; Jahangeer, S.; Nakamura, S.-I. Sphingosine Kinase 2 Is a Nuclear Protein and Inhibits DNA Synthesis. J. Biol. Chem. 2003, 278, 46832–46839. [Google Scholar] [CrossRef] [PubMed]
- Hovey, R.C.; Aimo, L. Diverse and Active Roles for Adipocytes During Mammary Gland Growth and Function. J. Mammary Gland. Biol. Neoplasia 2010, 15, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Cristea, S.; Polyak, K. Dissecting the mammary gland one cell at a time. Nat. Commun. 2018, 9, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Zwick, R.K.; Rudolph, M.C.; Shook, B.A.; Holtrup, B.; Roth, E.; Lei, V.; Van Keymeulen, A.; Seewaldt, V.; Kwei, S.; Wysolmerski, J.; et al. Adipocyte hypertrophy and lipid dynamics underlie mammary gland remodeling after lactation. Nat. Commun. 2018, 9, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Landskroner-Eiger, S.; Park, J.; Israel, D.; Pollard, J.W.; Scherer, P.E. Morphogenesis of the developing mammary gland: Stage-dependent impact of adipocytes. Dev. Biol. 2010, 344, 968–978. [Google Scholar] [CrossRef]
- Singh, S.K.; Spiegel, S. Sphingosine-1-phosphate signaling: A novel target for simultaneous adjuvant treatment of triple negative breast cancer and chemotherapy-induced neuropathic pain. Adv. Biol. Regul. 2020, 75, 100670. [Google Scholar] [CrossRef]
- Tsuchida, J.; Nagahashi, M.; Takabe, K.; Wakai, T. Clinical Impact of Sphingosine-1-Phosphate in Breast Cancer. Mediat. Inflamm. 2017, 2017, 1–9. [Google Scholar] [CrossRef]
- Wang, W.; Hind, T.; Lam, B.W.S.; Herr, D.R. Sphingosine 1–phosphate signaling induces SNAI2 expression to promote cell invasion in breast cancer cells. FASEB J. 2019, 33, 7180–7191. [Google Scholar] [CrossRef]
- Li, S.; Zhou, Y.; Zheng, X.; Wu, X.; Liang, Y.; Wang, S.; Zhang, Y. Sphk1 promotes breast epithelial cell proliferation via NF-κB-p65-mediated cyclin D1 expression. Oncotarget 2016, 7, 80579–80585. [Google Scholar] [CrossRef][Green Version]
- Moon, M.-H.; Jeong, J.-K.; Lee, Y.; Seol, J.-W.; Park, S. Sphingosine-1-phosphate inhibits the adipogenic differentiation of 3T3-L1 preadipocytes. Int. J. Mol. Med. 2014, 34, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.K.; Lambert, J.M.; Montefusco, D.J.; Tran, B.N.; Roddy, P.; Holland, W.L.; Cowart, L.A. Depletion of adipocyte sphingosine kinase 1 leads to cell hypertrophy, impaired lipolysis, and nonalcoholic fatty liver disease. J. Lipid Res. 2020, 61, 1328–1340. [Google Scholar] [CrossRef] [PubMed]
- Kitada, Y.; Kajita, K.; Taguchi, K.; Mori, I.; Yamauchi, M.; Ikeda, T.; Kawashima, M.; Asano, M.; Kajita, T.; Ishizuka, T.; et al. Blockade of Sphingosine 1-Phosphate Receptor 2 Signaling Attenuates High-Fat Diet-Induced Adipocyte Hypertrophy and Systemic Glucose Intolerance in Mice. Endocrinology 2016, 157, 1839–1851. [Google Scholar] [CrossRef] [PubMed]
- Moon, M.-H.; Jeong, J.-K.; Lee, J.-H.; Park, Y.-G.; Lee, Y.-J.; Seol, J.-W.; Park, S. Antiobesity activity of a sphingosine 1-phosphate analogue FTY720 observed in adipocytes and obese mouse model. Exp. Mol. Med. 2012, 44, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Badeanlou, L.; Bielawski, J.; Ciaraldi, T.P.; Samad, F. Sphingosine kinase 1 regulates adipose proinflammatory responses and insulin resistance. Am. J. Physiol. Metab. 2014, 306, E756–E768. [Google Scholar] [CrossRef] [PubMed]
- Rybinska, I.; Agresti, R.; Trapani, A.; Tagliabue, E.; Triulzi, T. Adipocytes in Breast Cancer, the Thick and the Thin. Cells 2020, 9, 560. [Google Scholar] [CrossRef]
- Boudreau, N.; Bissell, M.J. Extracellular matrix signaling: Integration of form and function in normal and malignant cells. Curr. Opin. Cell Biol. 1998, 10, 640–646. [Google Scholar] [CrossRef]
- Todd, J.R.; Ryall, K.A.; Vyse, S.; Wong, J.P.; Natrajan, R.C.; Yuan, Y.; Tan, A.-C.; Huang, P.H. Systematic analysis of tumour cell-extracellular matrix adhesion identifies independent prognostic factors in breast cancer. Oncotarget 2016, 7, 62939–62953. [Google Scholar] [CrossRef]
- Sonbol, H.S. Extracellular matrix remodeling in human disease. J. Microsc. Ultrastruct. 2018, 6, 123–128. [Google Scholar] [CrossRef]
- Hung, R.-J.; Hsu, I.-W.J.; Dreiling, J.L.; Lee, M.-J.; Williams, C.A.; Oberst, M.D.; Dickson, R.B.; Lin, C.-Y. Assembly of adherens junctions is required for sphingosine 1-phosphate-induced matriptase accumulation and activation at mammary epithelial cell-cell contacts. Am. J. Physiol. Physiol. 2004, 286, C1159–C1169. [Google Scholar] [CrossRef]
- Nagahashi, M.; Yamada, A.; Miyazaki, H.; Allegood, J.C.; Tsuchida, J.; Aoyagi, T.; Huang, W.-C.; Terracina, K.P.; Adams, B.J.; Rashid, O.M.; et al. Interstitial Fluid Sphingosine-1-Phosphate in Murine Mammary Gland and Cancer and Human Breast Tissue and Cancer Determined by Novel Methods. J. Mammary Gland. Biol. Neoplasia 2016, 21, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Kamburov, A.; Wierling, C.; Lehrach, H.; Herwig, R. ConsensusPathDB-a database for integrating human functional interaction networks. Nucleic Acids Res. 2009, 37, D623–D628. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.E.; Lee, E.J.; Chae, M.K.; Yoon, J.S. The Role of Sphingosine-1-Phosphate in Adipogenesis of Graves’ Orbitopathy. Investig. Opthalmology Vis. Sci. 2016, 57, 301. [Google Scholar] [CrossRef] [PubMed]
- Jun, D.-J.; Lee, J.-H.; Choi, B.-H.; Koh, T.-K.; Ha, D.-C.; Jeong, M.-W.; Kim, K.-T. Sphingosine-1-Phosphate Modulates Both Lipolysis and Leptin Production in Differentiated Rat White Adipocytes. Endocrinology 2006, 147, 5835–5844. [Google Scholar] [CrossRef] [PubMed]
- Moon, M.-H.; Jeong, J.-K.; Park, S. Activation of S1P2 receptor, a possible mechanism of inhibition of adipogenic differentiation by sphingosine 1-phosphate. Mol. Med. Rep. 2014, 11, 1031–1036. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A Software Environment for Integrated Models of Biomolecular Interaction Networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Alzahrani, A.S. PI3K/Akt/mTOR inhibitors in cancer: At the bench and bedside. Semin. Cancer Biol. 2019, 59, 125–132. [Google Scholar] [CrossRef]
- Verret, B.; Cortes, J.; Bachelot, T.; Andre, F.; Arnedos, M. Efficacy of PI3K inhibitors in advanced breast cancer. Ann. Oncol. 2019, 30, x12–x20. [Google Scholar] [CrossRef]
- Hoxhaj, G.; Manning, B.D. The PI3K–AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer 2019, 20, 74–88. [Google Scholar] [CrossRef]
- Crown, S.B.; Marze, N.; Antoniewicz, M.R. Catabolism of Branched Chain Amino Acids Contributes Significantly to Synthesis of Odd-Chain and Even-Chain Fatty Acids in 3T3-L1 Adipocytes. PLoS ONE 2015, 10, e0145850. [Google Scholar] [CrossRef]
- Popovich, D.G.; Lee, Y.; Li, L.; Zhang, W. Momordica charantia Seed Extract Reduces Pre-Adipocyte Viability, Affects Lactate Dehydrogenase Release, and Lipid Accumulation in 3T3-L1 Cells. J. Med. Food 2011, 14, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Thul, P.J.; Lindskog, C. The human protein atlas: A spatial map of the human proteome. Protein Sci. 2018, 27, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Lehr, S.; Hartwig, S.; Lamers, D.; Famulla, S.; Müller, S.; Hanisch, F.-G.; Cuvelier, C.; Ruige, J.; Eckardt, K.; Ouwens, D.M.; et al. Identification and Validation of Novel Adipokines Released from Primary Human Adipocytes. Mol. Cell. Proteom. 2012, 11, M111.010504. [Google Scholar] [CrossRef]
- Bauer-Mehren, A.; I Furlong, L.; Sanz, F. Pathway databases and tools for their exploitation: Benefits, current limitations and challenges. Mol. Syst. Biol. 2009, 5, 290. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Chu, C.; Lu, J.; Kong, X.; Huang, T.; Cai, Y.-D. Gene Ontology and KEGG Pathway Enrichment Analysis of a Drug Target-Based Classification System. PLoS ONE 2015, 10, e0126492. [Google Scholar] [CrossRef]
- Fabregat, A.; Sidiropoulos, K.; Viteri, G.; Forner-Martinez, O.; Marin-Garcia, P.; Arnau, V.; D’Eustachio, P.; Stein, L.; Hermjakob, H. Reactome pathway analysis: A high-performance in-memory approach. BMC Bioinform. 2017, 18, 1–9. [Google Scholar] [CrossRef]
- Slenter, D.N.; Kutmon, M.; Hanspers, K.; Riutta, A.; Windsor, J.; Nunes, N.; Mélius, J.; Cirillo, E.; Coort, S.L.; Digles, D.; et al. WikiPathways: A multifaceted pathway database bridging metabolomics to other omics research. Nucleic Acids Res. 2018, 46, D661–D667. [Google Scholar] [CrossRef]

| Upregulated DEGs | Downregulated DEGs | ||||||
|---|---|---|---|---|---|---|---|
| Pre- SGBS | Diff- SGBS | Pre- 3T3-L1 | Diff- 3T3-L1 | Pre- SGBS | Diff- SGBS | Pre- 3T3-L1 | Diff- 3T3-L1 |
| PIK3C2B | HAS1 | Gm17332 | Fstl1 | FGF20 | EMX2 | Tmsb15b2 | Stard9 |
| CTPS2 | CIDEC | Ces2c | Gm8281 | OLFM3 | ACAN | Gm14092 | Krtap16-3 |
| ZNF506 | TNFRSF19 | Olfr1477 | Mier2 | FCER1A | IFIT1 | Gm13298 | Folr1 |
| UBE2D3 | ZNF141 | Sftpc | Gm2237 | MRAP | SETD3 | Wdr17 | Usp26 |
| TRABD2A | GPRC5A | Vmn1r46 | Atg7 | PHYHIPL | IRAK1 | Slc25a31 | Mcoln2 |
| OSBPL1A | KLHL13 | Tcp11l1 | Lacc1 | CEMIP | FYCO1 | Gm20823 | Gad2 |
| CISH | THRSP | Pou2f1 | Depdc7 | KLHDC8B | DENND3 | Olfr1436 | Gm17482 |
| BIRC3 | ADAMTS9 | Ssx9 | Adipoq | CDKAL1 | NEO1 | Vmn1r227 | Gm17428 |
| SCRIB | CAT | Olfr810 | B3gnt9 | TMEM170B | ARID1A | Olfr1323 | Pygl |
| CCDC54 | SYN2 | Gm14025 | Trim13 | FOXO1 | STPG2 | Olfr825 | Ubash3b |
| OR1B1 | CLHC1 | Gm3127 | Slx4 | SHISA4 | PODN | Ccdc152 | Rnase9 |
| PLSCR5 | LIPE | Olfr807 | Lrrc9 | DIRC3 | HPS1 | Iqcj | Fam19a1 |
| SPATA5 | AGBL2 | Lctl | Eif6 | PDE6A | CDH22 | Afp | Chd7 |
| SLC25A25 | HACD3 | Clec7a | Rcor2 | BRD9 | TTLL12 | Oas1a | Gm11111 |
| OR11H12 | GK | Ifna11 | Esp36 | FRY | RASSF3 | Gm20738 | Vpreb2 |
| LOC340074 | NAT2 | Atp8a1 | Arhgap33 | QRSL1 | MICAL2 | Gm10477 | Gm2745 |
| NDUFA8 | PDE11A | Sema3d | Ensa | FDXR | RGS7 | Olfr809 | Col16a1 |
| KSR1 | CST2 | 5330417C22Rik | Sh2b1 | C17orf64 | ATP11A | Gm5662 | Prlr |
| API5L1 | CD207 | Olfr392 | Pcdhb21 | KIRREL3 | LCTL | Gm19668 | Gm4399 |
| CCDC160 | SENP8 | Klra23 | Sema4g | LY6G6C | PITPNM2 | Hsfy2 | Gm4406 |
| OR2F1 | PDE1B | Gm10037 | D430020J02Rik | MUC3A | ADRBK1 | Rhbdl3 | Arhgef26 |
| LONRF1 | EPHA4 | Olfr133 | Saal1 | DEFB104A | MINK1 | Tmprss11c | Pnmal1 |
| TRIM49D2 | BCO1 | 4930402K13Rik | Olfr1076 | C9orf50 | LTBP2 | Lrit2 | Defa3 |
| POGZ | HPD | Pde9a | Sssca1 | TMEM129 | MAN2A2 | S100a14 | Defa17 |
| TACC3 | LAIR1 | Vmn1r4 | Fbxw17 | LAGE3 | SCUBE3 | Gm5538 | Vmn2r110 |
| PTK2 | SLC38A4 | Fpr-rs6 | Zadh2 | SLC2A7 | GSK3A | Mlana | Lrrtm3 |
| STK32A | ADIPOQ | 3425401B19Rik | Abi3bp | KRTAP1–5 | MKL | Sult2a2 | LOC102642717 |
| CCL20 | LIPA | Lama3 | Fam180a | RAB25 | RFESD | Olfr773 | Gm10436 |
| GMPR | SMOX | Pdk2 | Mtor | FLOT1 | CCDC181 | Car3 | Olfr1258 |
| C17orf47 | NME8 | Catsperd | Foxm1 | MYO1E | RGS21 | Elmod1 | Defb25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Sakharkar, M.K.; Wabitsch, M.; Yang, J. Effects of Sphingosine-1-Phosphate on Cell Viability, Differentiation, and Gene Expression of Adipocytes. Int. J. Mol. Sci. 2020, 21, 9284. https://doi.org/10.3390/ijms21239284
Wu X, Sakharkar MK, Wabitsch M, Yang J. Effects of Sphingosine-1-Phosphate on Cell Viability, Differentiation, and Gene Expression of Adipocytes. International Journal of Molecular Sciences. 2020; 21(23):9284. https://doi.org/10.3390/ijms21239284
Chicago/Turabian StyleWu, Xiyuan, Meena Kishore Sakharkar, Martin Wabitsch, and Jian Yang. 2020. "Effects of Sphingosine-1-Phosphate on Cell Viability, Differentiation, and Gene Expression of Adipocytes" International Journal of Molecular Sciences 21, no. 23: 9284. https://doi.org/10.3390/ijms21239284
APA StyleWu, X., Sakharkar, M. K., Wabitsch, M., & Yang, J. (2020). Effects of Sphingosine-1-Phosphate on Cell Viability, Differentiation, and Gene Expression of Adipocytes. International Journal of Molecular Sciences, 21(23), 9284. https://doi.org/10.3390/ijms21239284





