Site-Specific Phosphorylation of Histone H1.4 Is Associated with Transcription Activation
Abstract
1. Introduction
2. Results
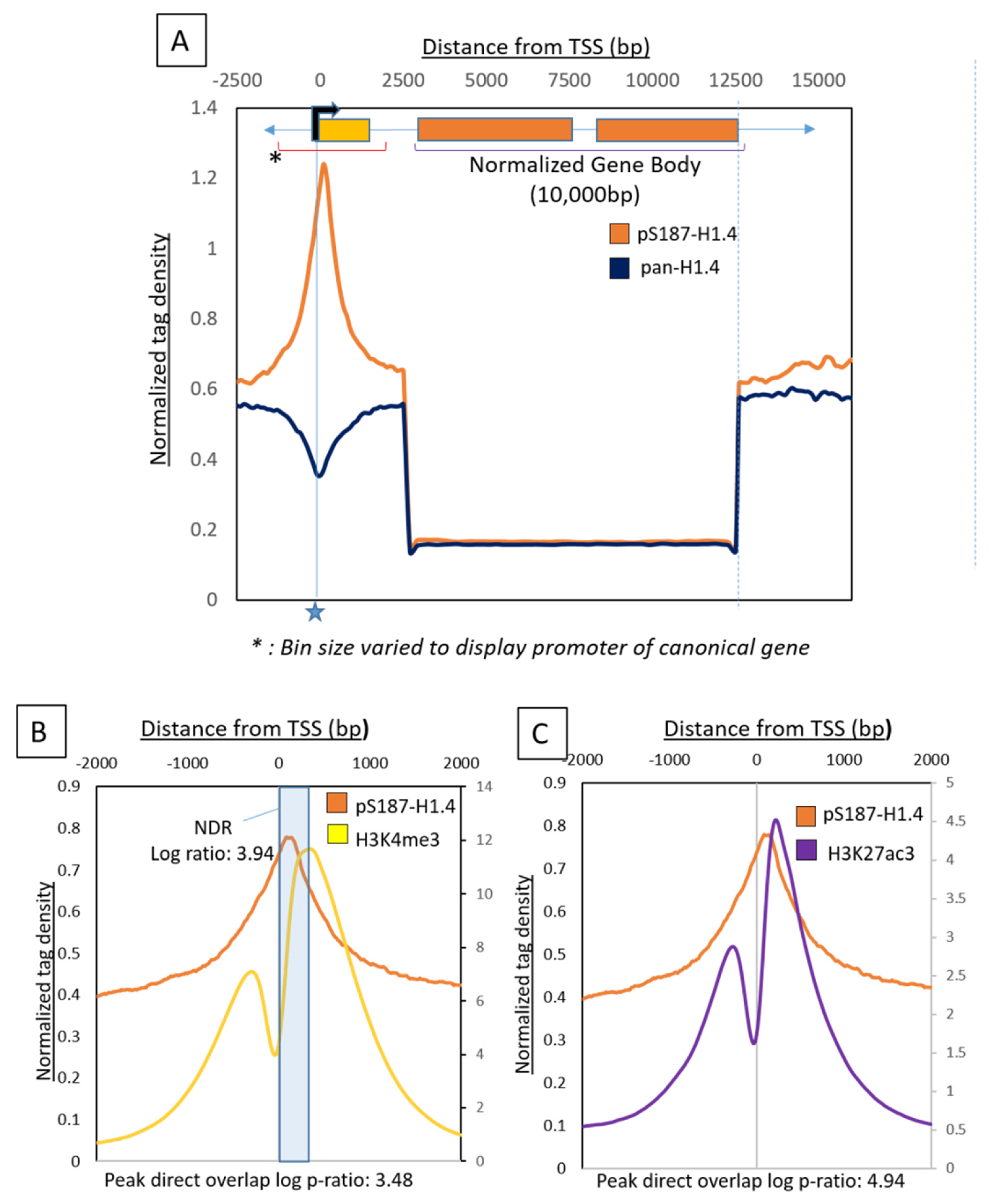
2.1. Genome-Wide Distribution of pS187-H1.4 Displays Distinct Patterns of Enrichment
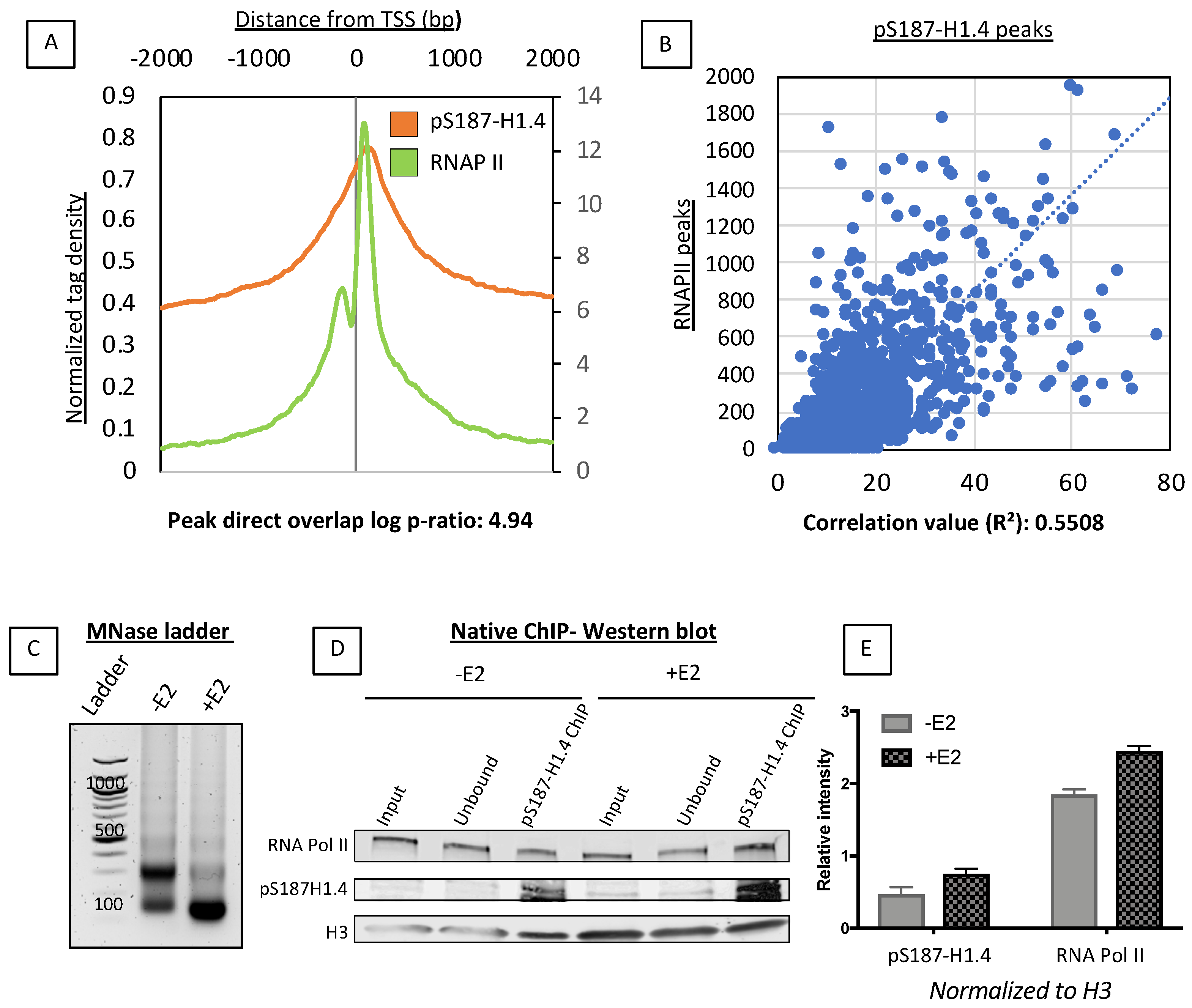
2.2. pS187-H1.4 Peaks Co-Localize with Promoter Associated RNAPII
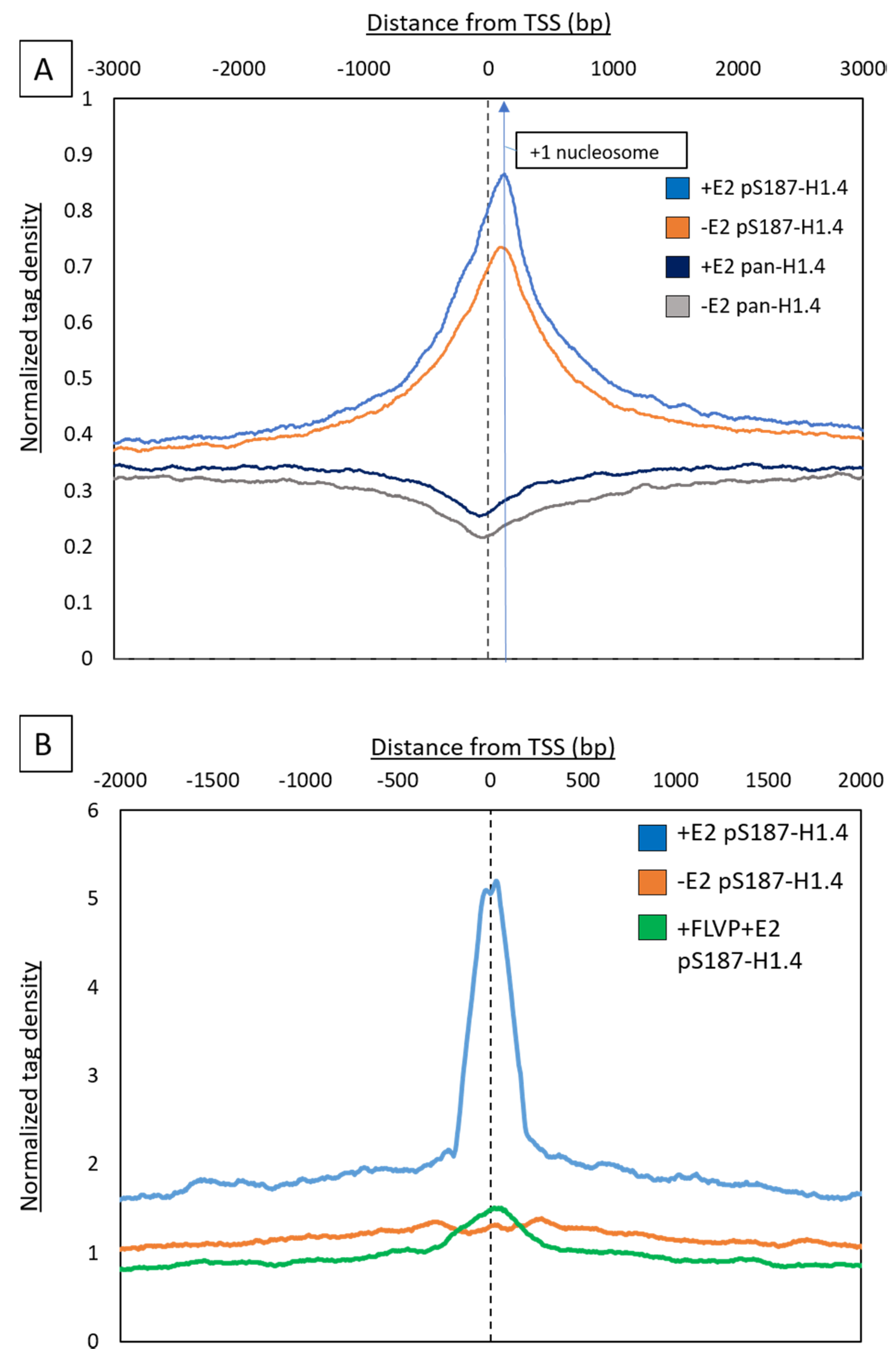
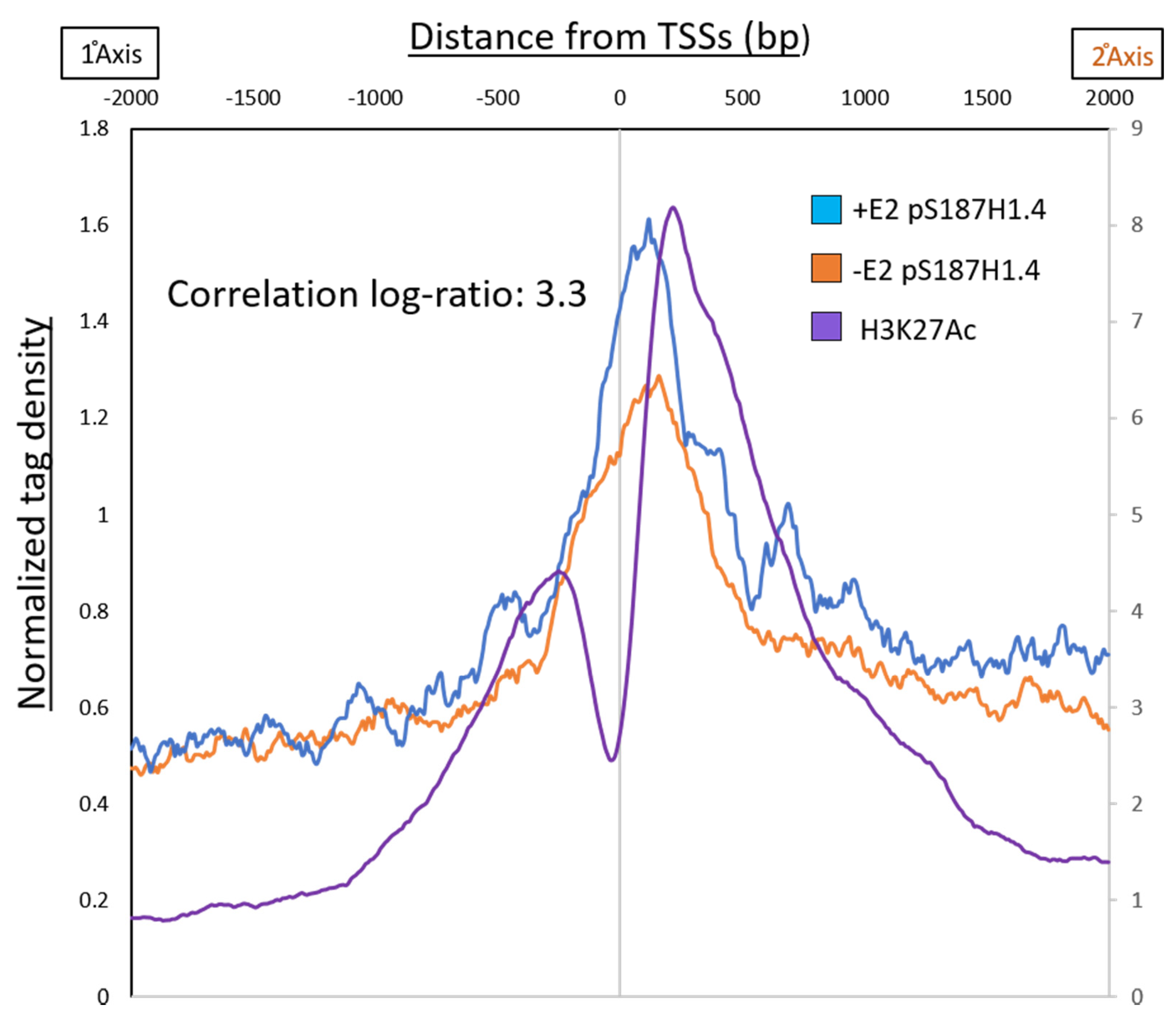
2.3. Estradiol (E2) Stimulation Resulted in pS187-H1.4 Peak Enrichment
2.4. X Differential pS187H1.4 Signals Enriched at Promoters and Can Be Quenched by CDK9 Inhibition
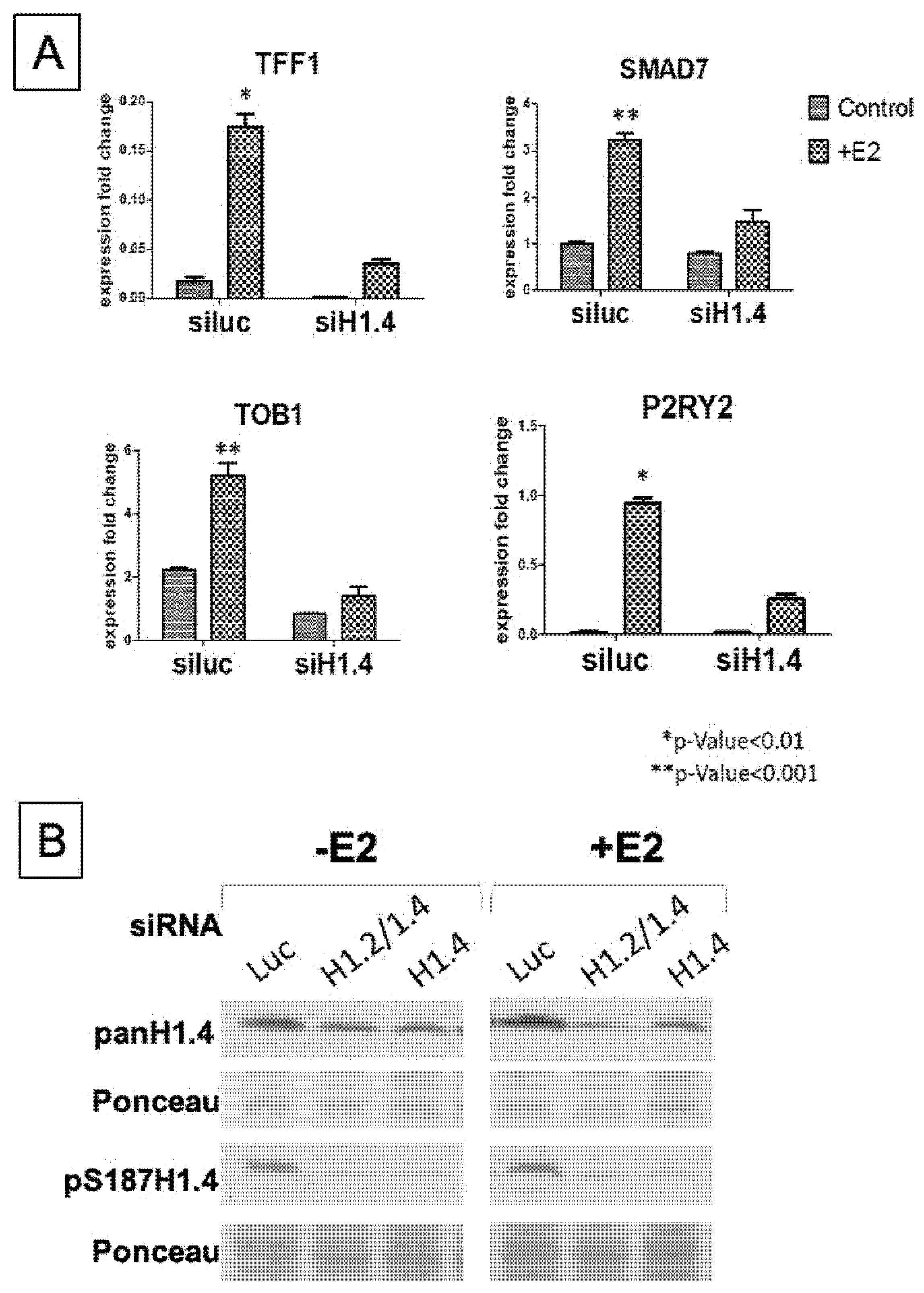
2.5. pS187-H1.4 Enrichment Is Associated with Previously Identified Estradiol Responsive Genes
3. Material and Methods
3.1. Cell Culture
3.2. siRNA Knockdown
3.3. Chromatin Immunoprecipitaiton
3.4. ChIP-Western Blot
3.5. Quantitative Real Time-PCR (qRT-PCR)
3.6. Sequencing and Bioinformatic Analysis
4. Discussion
4.1. pS187H1.4 Enrichment at Promoters Marks Active Genes
4.2. Genome-Wide Distribution of pS187-H1.4 Reveals a Possible Interaction with Multiple Transcriptional Regulatory Pathways
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Allan, J.S.; Hartman, P.G.; Cranerobinson, C.; Aviles, F.X. The structure of histone H1 and its location in chromatin. Nat. Cell Biol. 1980, 288, 675–679. [Google Scholar] [CrossRef]
- Bednar, J.; Horowitz, R.A.; Grigoryev, S.A.; Carruthers, L.M.; Hansen, J.C.; Koster, A.J.; Woodcock, C.L. Nucleosomes, linker DNA, and linker histone form a unique structural motif that directs the higher-order folding and compaction of chromatin. Proc. Natl. Acad. Sci. USA 1998, 95, 14173–14178. [Google Scholar] [CrossRef]
- Fan, Y.; Nikitina, T.; Zhao, J.; Fleury, T.J.; Bhattacharyya, R.; Bouhassira, E.E.; Stein, A.; Woodcock, C.L.; Skoultchi, A.I. Faculty Opinions recommendation of Histone H1 depletion in mammals alters global chromatin structure but causes specific changes in gene regulation. Cell 2006, 123, 1199–1212. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Gorovsky, M.A. Linker Histone H1 Regulates Specific Gene Expression but Not Global Transcription In Vivo. Cell 1996, 86, 475–483. [Google Scholar] [CrossRef]
- Lin, Q.; Inselman, A.; Han, X.; Xu, H.; Zhang, W.; Handel, M.A.; Skoultchi, A.I. Reductions in Linker Histone Levels Are Tolerated in Developing Spermatocytes but Cause Changes in Specific Gene Expression. J. Biol. Chem. 2004, 279, 23525–23535. [Google Scholar] [CrossRef] [PubMed]
- Sancho, M.; Diani, E.; Beato, M.; Jordan, A. Depletion of Human Histone H1 Variants Uncovers Specific Roles in Gene Expression and Cell Growth. PLoS Genet. 2008, 4, e1000227. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, S.K.; Knight, K.L.; Rana, T.M. Phosphorylation of histone H1 by P-TEFb is a necessary step in skeletal muscle differentiation. J. Cell. Physiol. 2012, 227, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Liao, R.; Mizzen, C.A. Interphase H1 phosphorylation: Regulation and functions in chromatin. Biochim. Biophys. Acta (BBA)-Bioenerg. 2016, 1859, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Happel, N.; Doenecke, D. Histone H1 and its isoforms: Contribution to chromatin structure and function. Gene 2009, 431, 1–12. [Google Scholar] [CrossRef]
- Stasevich, T.J.; Mueller, F.; Brown, D.T.; McNally, J.G. Dissecting the binding mechanism of the linker histone in live cells: an integrated FRAP analysis. EMBO J. 2010, 29, 1225–1234. [Google Scholar] [CrossRef]
- Sarg, B.; Helliger, W.; Talasz, H.; Förg, B.; Lindner, H.H. Histone H1 phosphorylation occurs site-specifically during interphase and mitosis: identification of a novel phosphorylation site on histone H1. J. Biol. Chem. 2006, 281, 6573–6580. [Google Scholar] [CrossRef] [PubMed]
- Hergeth, S.P.; Dundr, M.; Tropberger, P.; Zee, B.M.; Garcia, B.A.; Daujat, S.; Schneider, R. Isoform-specific phosphorylation of human linker histone H1.4 in mitosis by the kinase Aurora B. J. Cell Sci. 2011, 124, 1623–1628. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hergeth, S.P.; Schneider, R. The H1 linker histones: multifunctional proteins beyond the nucleosomal core particle. EMBO Rep. 2015, 16, 1439–1453. [Google Scholar] [CrossRef] [PubMed]
- Mizzen, C.A.; Dou, Y.; Liu, Y.; Cook, R.G.; Gorovsky, M.A.; Allis, C.D. Identification and mutation of phosphorylation sites in a linker histone. Phosphorylation of macronuclear H1 is not essential for viability in tetrahymena. J. Biol. Chem. 1999, 274, 14533–14536. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Cooke, M.; Panjwani, S.; Cao, K.; Krauth, B.; Ho, P.-Y.; Medrzycki, M.; Berhe, D.T.; Pan, C.; McDevitt, T.C.; et al. Histone H1 Depletion Impairs Embryonic Stem Cell Differentiation. PLoS Genet. 2012, 8, e1002691. [Google Scholar] [CrossRef]
- Dou, Y.; Gorovsky, M.A. Regulation of transcription by H1 phosphorylation in Tetrahymena is position independent and requires clustered sites. Proc. Natl. Acad. Sci. USA 2002, 99, 6142–6146. [Google Scholar] [CrossRef]
- Zheng, Y.; John, S.; Pesavento, J.J.; Schultz-Norton, J.R.; Schiltz, R.L.; Baek, S.; Nardulli, A.M.; Hager, G.L.; Kelleher, N.L.; Mizzen, C.A. Histone H1 phosphorylation is associated with transcription by RNA polymerases I and II. J. Cell Biol. 2010, 189, 407–415. [Google Scholar] [CrossRef]
- Talasz, H.; Sarg, B.; Lindner, H.H. Site-specifically phosphorylated forms of H1.5 and H1.2 localized at distinct regions of the nucleus are related to different processes during the cell cycle. Chromosoma 2009, 118, 693–709. [Google Scholar] [CrossRef]
- Liao, R.; Mizzen, C.A. Site-specific regulation of histone H1 phosphorylation in pluripotent cell differentiation. Epigenetics Chromatin 2017, 10, 29. [Google Scholar] [CrossRef]
- Hah, N.; Danko, C.G.; Core, L.; Waterfall, J.J.; Siepel, A.; Lis, J.T.; Kraus, W.L. A rapid, extensive, and transient transcriptional response to estrogen signaling in breast cancer cells. Cell 2011, 145, 622–634. [Google Scholar] [CrossRef]
- Hah, N.; Murakami, S.; Nagari, A.; Danko, C.G.; Kraus, W.L. Enhancer transcripts mark active estrogen receptor binding sites. Genome Res. 2013, 23, 1210–1223. [Google Scholar] [CrossRef] [PubMed]
- Stender, J.D.; Frasor, J.; Komm, B.; Chang, K.C.N.; Kraus, W.L.; Katzenellenbogen, B.S. Estrogen-Regulated Gene Networks in Human Breast Cancer Cells: Involvement of E2F1 in the Regulation of Cell Proliferation. Mol. Endocrinol. 2007, 21, 2112–2123. [Google Scholar] [CrossRef] [PubMed]
- Heinz, S.; Benner, C.; Spann, N.; Bertolino, E.; Lin, Y.C.; Laslo, P.; Cheng, J.X.; Murre, C.; Singh, H.; Glass, C.K. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 2010, 38, 576–589. [Google Scholar] [CrossRef] [PubMed]
- Millán-Ariño, L.; Islam, A.B.M.M.K.; Izquierdo-Bouldstridge, A.; Mayor, R.; Terme, J.-M.; Luque, N.; Sancho, M.; López-Bigas, N.; Jordan, A. Mapping of six somatic linker histone H1 variants in human breast cancer cells uncovers specific features of H1.2. Nucleic Acids Res. 2014, 42, 4474–4493. [Google Scholar] [CrossRef]
- Leadem, B.R.; Kagiampakis, I.; Wilson, C.; Cheung, T.K.; Arnott, D.; Trojer, P.; Classon, M.; Easwaran, H.; Baylin, S.B. A KDM5 Inhibitor Increases Global H3K4 Trimethylation Occupancy and Enhances the Biological Efficacy of 5-Aza-2′-Deoxycytidine. Cancer Res. 2018, 78, 1127–1139. [Google Scholar] [CrossRef]
- Grandy, R.A.; Whitfield, T.W.; Wu, H.; Fitzgerald, M.P.; VanOudenhove, J.J.; Zaidi, S.K.; Montecino, M.A.; Lian, J.B.; Van Wijnen, A.J.; Stein, J.L.; et al. Genome-Wide Studies Reveal that H3K4me3 Modification in Bivalent Genes Is Dynamically Regulated during the Pluripotent Cell Cycle and Stabilized upon Differentiation. Mol. Cell. Biol. 2015, 36, 615–627. [Google Scholar] [CrossRef]
- Howe, F.S.; Fischl, H.; Murray, S.C.; Mellor, J. Is H3K4me3 instructive for transcription activation? BioEssays 2017, 39, 1–12. [Google Scholar] [CrossRef]
- Voigt, P.; Tee, W.-W.; Reinberg, D. A double take on bivalent promoters. Genes Dev. 2013, 27, 1318–1338. [Google Scholar] [CrossRef]
- Baker, C.L.; Walker, M.; Kajita, S.; Petkov, P.M.; Paigen, K. PRDM9 binding organizes hotspot nucleosomes and limits Holliday junction migration. Genome Res. 2014, 24, 724–732. [Google Scholar] [CrossRef]
- Lee, C.K.; Shibata, Y.; Rao, B.; Strahl, B.D.; Lieb, J.D. Faculty Opinions recommendation of Evidence for nucleosome depletion at active regulatory regions genome-wide. Nat. Genet. 2004, 36, 900–905. [Google Scholar] [CrossRef]
- Lee, A.V.; Oesterreich, S.; Davidson, N.E. MCF-7 Cells--Changing the Course of Breast Cancer Research and Care for 45 Years. J. Natl. Cancer Inst. 2015, 107, djv073. [Google Scholar] [CrossRef] [PubMed]
- Frasor, J.; Danes, J.M.; Komm, B.; Chang, K.C.N.; Lyttle, C.R.; Katzenellenbogen, B.S. Profiling of Estrogen Up- and Down-Regulated Gene Expression in Human Breast Cancer Cells: Insights into Gene Networks and Pathways Underlying Estrogenic Control of Proliferation and Cell Phenotype. Endocrinology 2003, 144, 4562–4574. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, T.C.; Wijeratne, E.K.; Liang, J.Y.; Gunatilaka, A.L.; Wu, J.M. Differential control of growth, cell cycle progression, and expression of NF-kappaB in human breast cancer cells MCF-7, MCF-10A, and MDA-MB-231 by ponicidin and oridonin, diterpenoids from the chinese herb Rabdosia rubescens. Biochem. Biophys. Res. Commun. 2005, 337, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Dreijerink, K.M.; Groner, A.C.; Vos, E.; Font-Tello, A.; Gu, L.; Chi, D.; Reyes, J.; Cook, J.; Lim, E.; Lin, C.Y.; et al. Enhancer-Mediated Oncogenic Function of the Menin Tumor Suppressor in Breast Cancer. Cell Rep. 2017, 18, 2359–2372. [Google Scholar] [CrossRef] [PubMed]
- Danko, C.G.; Hah, N.; Luo, X.; Martins, A.L.; Core, L.; Lis, J.T.; Siepel, A.; Kraus, W.L. Signaling pathways differentially affect RNA polymerase II initiation, pausing, and elongation rate in cells. Mol. Cell 2013, 50, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, K.; Horie-Inoue, K.; Inoue, S. Identification of estrogen-responsive genes based on the DNA binding properties of estrogen receptors using high-throughput sequencing technology. Acta Pharmacol. Sin. 2015, 36, 24–31. [Google Scholar] [CrossRef]
- Kwak, H.; Fuda, N.J.; Core, L.J.; Lis, J.T. Precise Maps of RNA Polymerase Reveal How Promoters Direct Initiation and Pausing. Science 2013, 339, 950–953. [Google Scholar] [CrossRef]
- Adelman, K.; Lis, J.T. Promoter-proximal pausing of RNA polymerase II: emerging roles in metazoans. Nat. Rev. Genet. 2012, 13, 720–731. [Google Scholar] [CrossRef]
- Chao, S.H.; Fujinaga, K.; Marion, J.E.; Taube, R.; Sausville, E.A.; Senderowicz, A.M.; Peterlin, B.M. Price DHFlavopiridol inhibits P-TEFb and blocks HIV-1 replication. J. Biol. Chem. 2000, 275, 28345–28348. [Google Scholar] [CrossRef]
- Chao, S.H.; Price, D.H. Flavopiridol inactivates P-TEFb and blocks most RNA polymerase II transcription in vivo. J. Biol. Chem. 2001, 276, 31793–31799. [Google Scholar] [CrossRef]
- McLean, C.Y.; Bristor, D.; Hiller, M.; Clarke, S.L.; Schaar, B.T.; Lowe, C.B.; Wenger, A.M.; Bejerano, G. GREAT improves functional interpretation of cis-regulatory regions. Nat. Biotechnol. 2010, 28, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Kent, W.J.; Sugnet, C.W.; Furey, T.S.; Roskin, K.M.; Pringle, T.H.; Zahler, A.M.; Haussler, D. The human genome browser at UCSC. Genome Res. 2002, 12, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Acconcia, F.; Barnes, C.J.; Kumar, R. Estrogen and Tamoxifen Induce Cytoskeletal Remodeling and Migration in Endometrial Cancer Cells. Endocrinology 2006, 147, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Sacchi, N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Dennis, G., Jr.; Sherman, B.T.; Hosack, D.A.; Yang, J.; Gao, W.; Lane, H.C.; Lempicki, R.A. DAVID: Database for Annotation, Visualization, and Integrated Discovery. Genome Biol. 2003, 4, P3. [Google Scholar] [CrossRef]
- Balhorn, R.; Chalkley, R.; Granner, D. Lysine-rich histone phosphorylation. A positive correlation with cell replication. Biochemistry 1972, 11, 1094–1098. [Google Scholar] [CrossRef]
- Izzo, A.; Kamieniarz, K.; Schneider, R. The histone H1 family: specific members, specific functions? Biol. Chem. 2008, 389, 333–343. [Google Scholar] [CrossRef]
- Wiśniewski, J.R.; Zougman, A.; Krüger, S.; Mann, M. Mass Spectrometric Mapping of Linker Histone H1 Variants Reveals Multiple Acetylations, Methylations, and Phosphorylation as Well as Differences between Cell Culture and Tissue. Mol. Cell. Proteom. 2006, 6, 72–87. [Google Scholar] [CrossRef]
- Daujat, S.; Zeissler, U.; Waldmann, T.; Happel, N.; Schneider, R. HP1 binds specifically to Lys26-methylated histone H1.4, whereas simultaneous Ser27 phosphorylation blocks HP1 binding. J. Biol. Chem. 2005, 280, 38090–38095. [Google Scholar] [CrossRef]
- Kamieniarz-Gdula, K.; Izzo, A.; Dundr, M.; Tropberger, P.; Ozretić, L.; Kirfel, J.; Scheer, E.; Tropel, P.; Wiśniewski, J.R.; Tora, L.; et al. A dual role of linker histone H1.4 Lys 34 acetylation in transcriptional activation. Genes Dev. 2012, 26, 797–802. [Google Scholar] [CrossRef] [PubMed]
- DeRoo, B.J.; Korach, K.S. Estrogen receptors and human disease. J. Clin. Investig. 2006, 116, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, M.L.; Kraus, W.L. Transcriptional activation by nuclear receptors. Essays Biochem. 2004, 40, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Cheung, E.; Kraus, W.L. Genomic Analyses of Hormone Signaling and Gene Regulation. Annu. Rev. Physiol. 2010, 72, 191–218. [Google Scholar] [CrossRef]
- MacQuarrie, K.L.; Fong, A.P.; Morse, R.H.; Tapscott, S.J. Genome-wide transcription factor binding: Beyond direct target regulation. Trends Genet. 2011, 27, 141–148. [Google Scholar] [CrossRef]
- Sethi, I.; Sinha, S.; Buck, M.J. Role of chromatin and transcriptional co-regulators in mediating p63-genome interactions in keratinocytes. BMC Genom. 2014, 15, 1042. [Google Scholar] [CrossRef]
- Ping, Y.; Rana, T.M. DSIF and NELF Interact with RNA Polymerase II Elongation Complex and HIV-1 Tat Stimulates P-TEFb-mediated Phosphorylation of RNA Polymerase II and DSIF during Transcription Elongation. J. Biol. Chem. 2000, 276, 12951–12958. [Google Scholar] [CrossRef]
- Bartkowiak, B.; Greenleaf, A.L. Phosphorylation of RNAPII: To P-TEFb or not to P-TEFb? Transcription 2011, 2, 115–119. [Google Scholar] [CrossRef]
- D’Alessio, J.A.; Ng, R.; Willenbring, H.; Tjian, R. Faculty Opinions recommendation of Core promoter recognition complex changes accompany liver development. Proc. Natl. Acad. Sci. USA 2011, 108, 3906–3911. [Google Scholar] [CrossRef]
- Delaney, E.; Luse, D.S. Gdown1 Associates Efficiently with RNA Polymerase II after Promoter Clearance and Displaces TFIIF during Transcript Elongation. PLoS ONE 2016, 11, e0163649. [Google Scholar] [CrossRef]
- Kulaeva, O.I.; Hsieh, F.-K.; Chang, H.-W.; Luse, D.S.; Studitsky, V.M. Mechanism of transcription through a nucleosome by RNA polymerase II. Biochim. Biophys. Acta (BBA)-Bioenerg. 2013, 1829, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Deaton, A.M.; Bird, A. CpG islands and the regulation of transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Inoue, S.; Hiroi, H.; Orimo, A.; Kawashima, H.; Muramatsu, M. Isolation of Estrogen-Responsive Genes with a CpG Island Library. Mol. Cell. Biol. 1998, 18, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Bhan, A.; Mandal, S.S. Estradiol-Induced Transcriptional Regulation of Long Non-Coding RNA, HOTAIR. Methods Mol. Biol. 2016, 1366, 395–412. [Google Scholar] [CrossRef]

| Term Name | Binom Raw p-Value |
|---|---|
| Genes bound by ESR1 and up-regulated by estradiol in MCF-7 cells (breast cancer) expressing constitutevly active form of AKT1 | 7.64 × 10−126 |
| Genes bound by ESR1 and up-regulated by estradiol in MCF-7 cells (breast cancer). | 3.2717 × 10−107 |
| The ‘ER-alpha profile’: genes up-regulated in T47D cells upon activation of ESR1 by estradiol (E2) | 1.9154 × 10−92 |
| Genes upregulated in MCF7 cells (breast cancer) at 6 h of estradiol treatment. | 3.2198 × 10−65 |
| Genes upregulated in MCF7 cells (breast cancer) at 24 h of estradiol treatment. | 1.0162 × 10−59 |
| Myb-regulated genes in MCF7 (breast cancer) and lung epithelial cell lines overexpressing MYBL2, MYBL1 or MYB. | 4.9913 × 10−59 |
| Genes up-regulated in luminal-like breast cancer cell lines compared to the basal-like ones. | 2.038 × 10−50 |
| Genes whose expression negatively correlated with resistance of breast cancer cell lines to dasatinib. | 2.7928 × 10−48 |
| Genes regulated by ESR1 in MCF7 cells (breast cancer) | 3.5364 × 10−45 |
| Genes down-regulated in breast cancer tumor (formed by MCF7 xenografts) resistant to tamoxifen. | 5.1164 × 10−44 |
| Log p-Ratio | Annotation/Peaks |
|---|---|
| Significant association with pS187-H1.4 | |
| 3.64 | Cpg Island (h) * |
| 2.85 | Promoters (h) |
| 2.04 | Exons (h) |
| 2.04 | Protein-coding (h) |
| 4.94 | RNAPII (C) ** |
| 3.24 | H3K27Ac (C) |
| 3.48 | H3K4me3 (C) |
| Significant dissociation with pS187-H1.4 | |
| −9.92 | Centromeres (h) |
| −0.34 | Introns (h) |
| −1.64 | Intergenic (h) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saha, A.; Seward, C.H.; Stubbs, L.; Mizzen, C.A. Site-Specific Phosphorylation of Histone H1.4 Is Associated with Transcription Activation. Int. J. Mol. Sci. 2020, 21, 8861. https://doi.org/10.3390/ijms21228861
Saha A, Seward CH, Stubbs L, Mizzen CA. Site-Specific Phosphorylation of Histone H1.4 Is Associated with Transcription Activation. International Journal of Molecular Sciences. 2020; 21(22):8861. https://doi.org/10.3390/ijms21228861
Chicago/Turabian StyleSaha, Ankita, Christopher H. Seward, Lisa Stubbs, and Craig A. Mizzen. 2020. "Site-Specific Phosphorylation of Histone H1.4 Is Associated with Transcription Activation" International Journal of Molecular Sciences 21, no. 22: 8861. https://doi.org/10.3390/ijms21228861
APA StyleSaha, A., Seward, C. H., Stubbs, L., & Mizzen, C. A. (2020). Site-Specific Phosphorylation of Histone H1.4 Is Associated with Transcription Activation. International Journal of Molecular Sciences, 21(22), 8861. https://doi.org/10.3390/ijms21228861





