Groundbreaking Anticancer Activity of Highly Diversified Oxadiazole Scaffolds
Abstract
1. Introduction
2. Enzyme Interactions
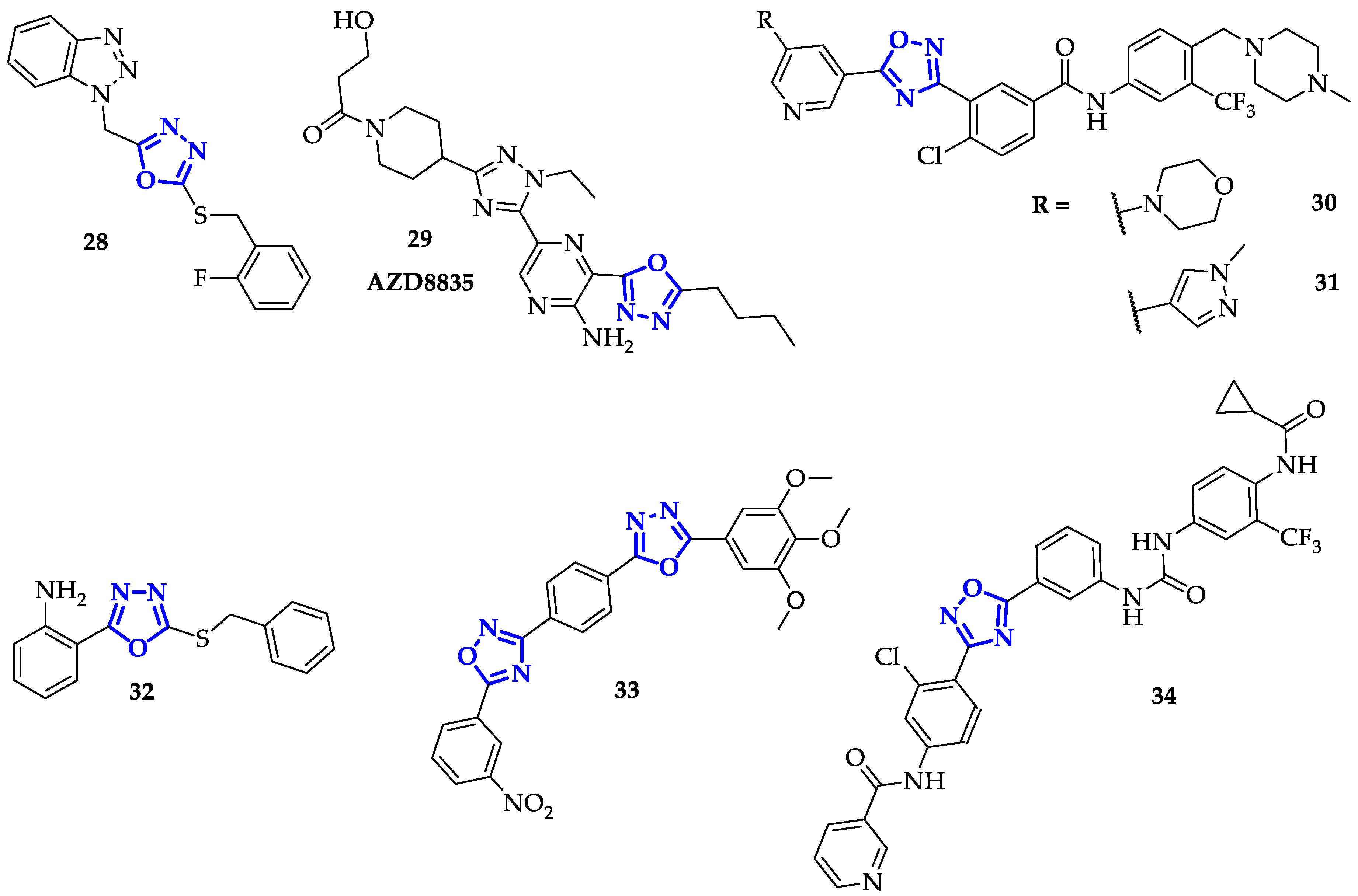
2.1. Specific Inhibitors of Telomerase
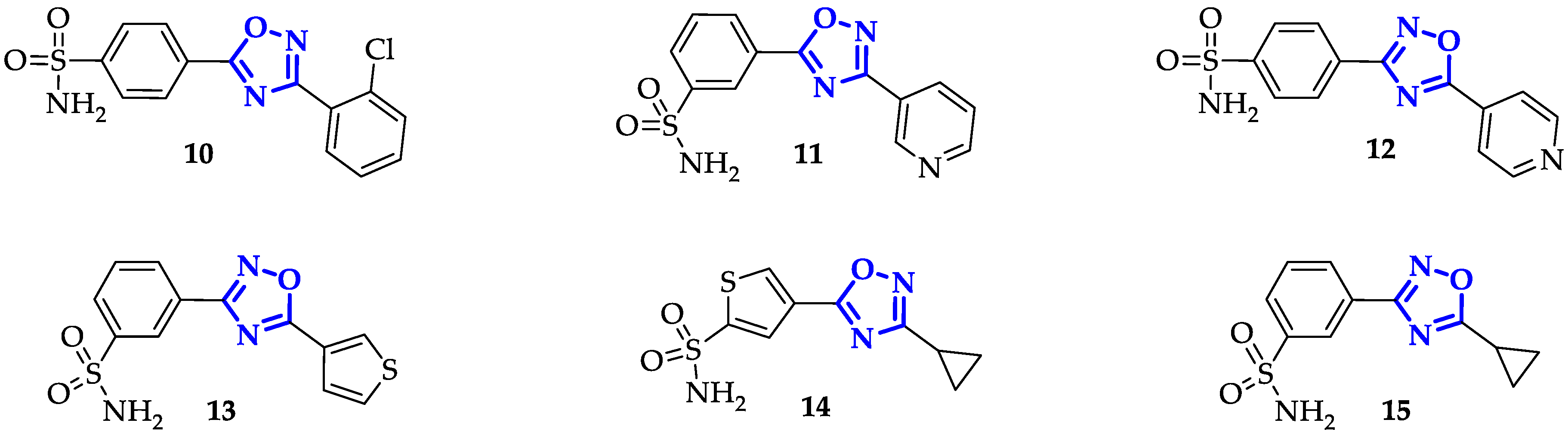
2.2. Selective Inhibition of Carbonic Anhydrase Isoforms
2.3. Specific Interactions with Histone Deacetylase Isoforms
2.4. Efficient Kinases Inhibitors
2.5. Other Targeted Enzymes
3. Specific Interaction with Globular Proteins: Tubulin-Binding Drugs
4. Nucleic Acid Structures as Oxadiazole Targets
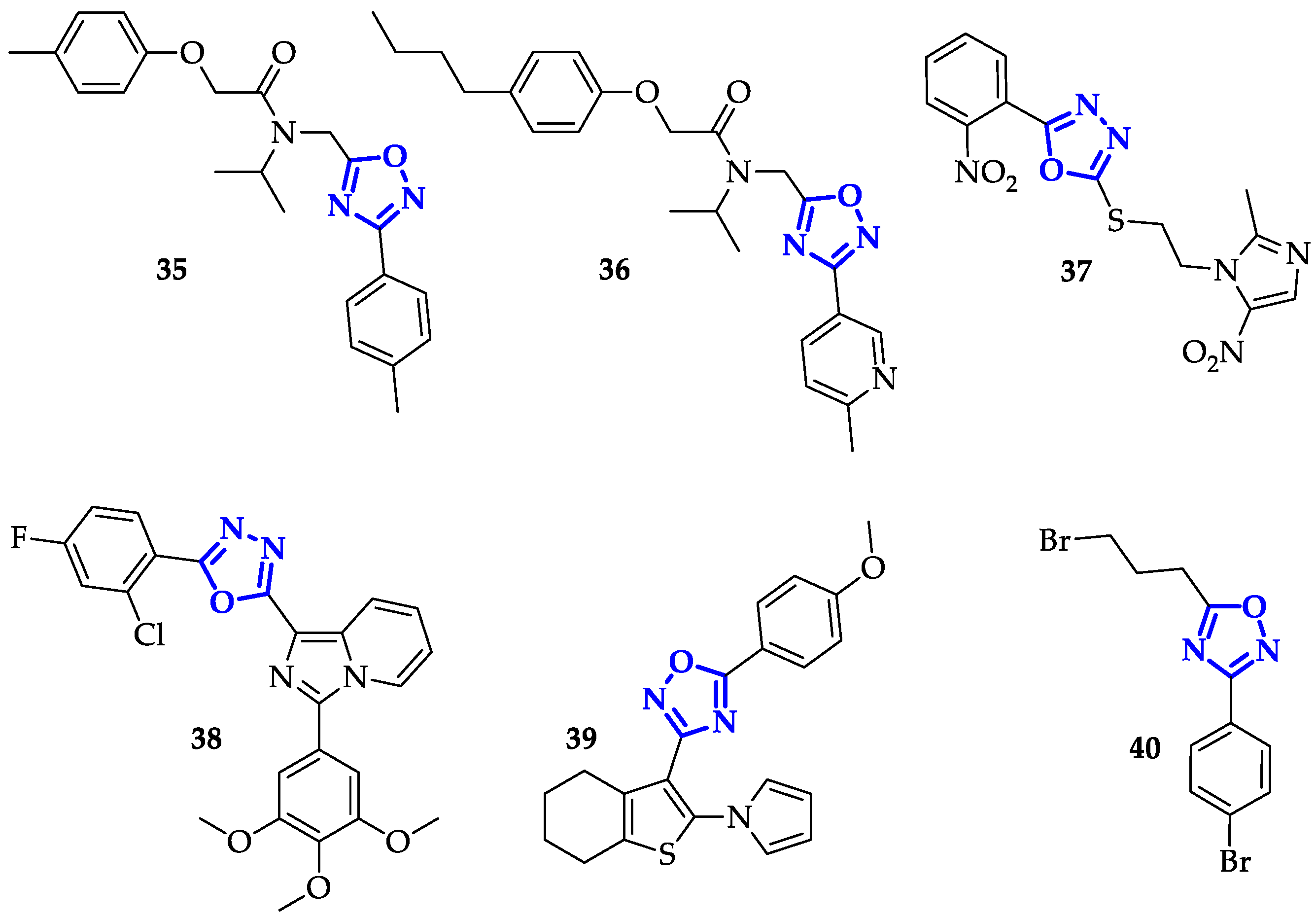
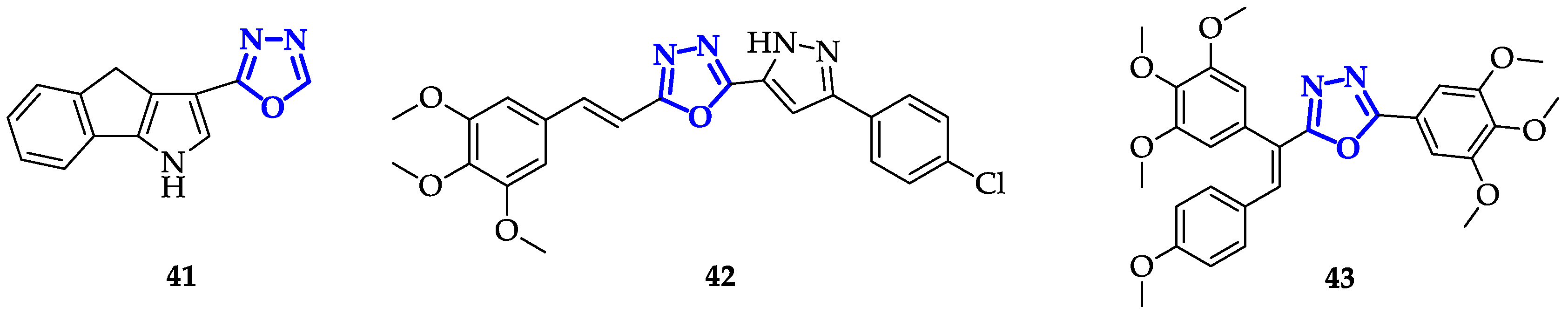
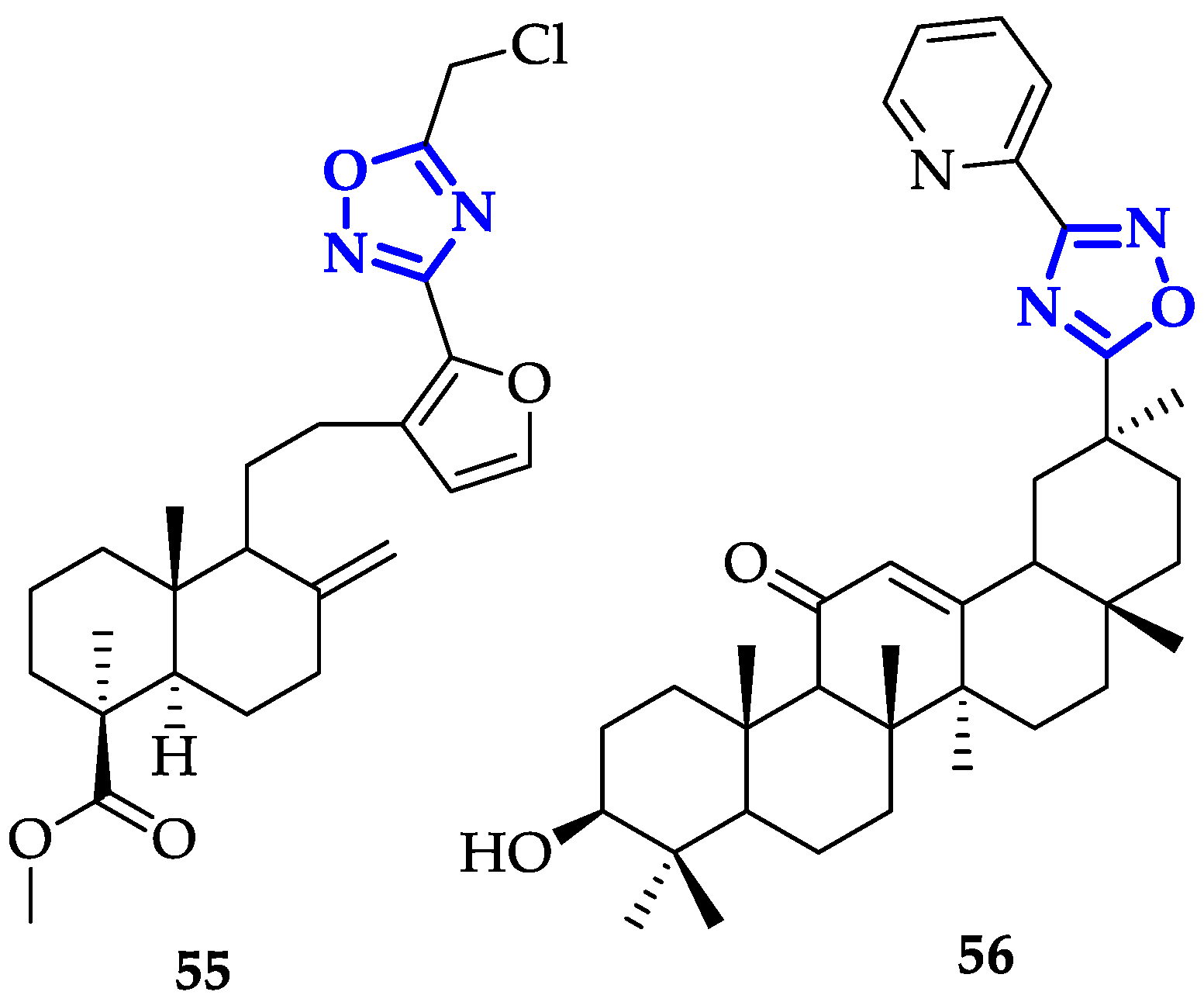
5. Oxadiazoles Derivatives as Anticancer Agents without a Specific Target Identified
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Jampilek, J. Heterocycles in Medicinal Chemistry. Molecules 2019, 24, 3839. [Google Scholar] [CrossRef]
- Berger, J.; Li, M.; Berger, S.; Meilak, M.; Rientjes, J.; Currie, P.D. Effect of Ataluren on dystrophin mutations. J. Cell. Mol. Med. 2020, 24, 6680–6689. [Google Scholar] [CrossRef]
- Caputo, F.; Corbetta, S.; Piccolo, O.; Vigo, D. Seeking for Selectivity and Efficiency: New Approaches in the Synthesis of Raltegravir. Org. Process Res. Dev. 2020, 24, 1149–1156. [Google Scholar] [CrossRef]
- Khallaf, A.; Wang, P.; Liu, H.; Zhuo, S.; Zhu, H. 1,2,4-Oxadiazole ring–containing pyridylpyrazole-4-carboxamides: Synthesis and evaluation as novel insecticides of the anthranilic diamide family. J. Heterocycl. Chem. 2020, 57, 1981–1992. [Google Scholar] [CrossRef]
- Fershtat, L.L.; Makhova, N.N. 1,2,5-Oxadiazole-Based High-Energy-Density Materials: Synthesis and Performance. ChemPlusChem 2020, 85, 13–42. [Google Scholar] [CrossRef]
- Salassa, G.; Terenzi, A. Metal Complexes of Oxadiazole Ligands: An Overview. Int. J. Mol. Sci. 2019, 20, 3483. [Google Scholar] [CrossRef] [PubMed]
- Boström, J.; Hogner, A.; Llinàs, A.; Wellner, E.; Plowright, A.T. Oxadiazoles in Medicinal Chemistry. J. Med. Chem. 2012, 55, 1817–1830. [Google Scholar] [CrossRef]
- Biernacki, K.; Daśko, M.; Ciupak, O.; Kubiński, K.; Rachon, J.; Demkowicz, S. Novel 1,2,4-Oxadiazole Derivatives in Drug Discovery. Pharmaceuticals 2020, 13, 111. [Google Scholar] [CrossRef] [PubMed]
- Tassinari, M.; Lena, A.; Butovskaya, E.; Pirota, V.; Nadai, M.; Freccero, M.; Doria, F.; Richter, S.N. A Fragment-Based Approach for the Development of G-Quadruplex Ligands: Role of the Amidoxime Moiety. Molecules 2018, 23, 1874. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Bao, X.; Ren, H.; Liao, P.; Yan, Y.; Wang, L.; Chen, Z. Design, synthesis, and pharmacological evaluation of 5-oxo-1,2,4-oxadiazole derivatives as AT1 antagonists with antihypertension activities. Clin. Exp. Hypertens. 2016, 38, 435–442. [Google Scholar] [CrossRef]
- Ibrahim, M.T.; Uzairu, A.; Shallangwa, G.A.; Ibrahim, A. In-silico studies of some oxadiazoles derivatives as anti-diabetic compounds. J. King Saud Univ.-Sci. 2020, 32, 423–432. [Google Scholar] [CrossRef]
- Chawla, G.; Naaz, B.; Siddiqui, A.A. Exploring 1,3,4-Oxadiazole Scaffold for Anti-inflammatory and Analgesic Activities: A Review of Literature From 2005-2016. Mini Rev. Med. Chem. 2018, 18, 216–233. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, S.; Asati, V.; Singh, J.; Roy, P.P. 1,3,4-Oxadiazoles: An emerging scaffold to target growth factors, enzymes and kinases as anticancer agents. Eur. J. Med. Chem. 2015, 97, 124–141. [Google Scholar] [CrossRef] [PubMed]
- Glomb, T.; Szymankiewicz, K.; Świątek, P. Anti-Cancer Activity of Derivatives of 1,3,4-Oxadiazole. Molecules 2018, 23, 3361. [Google Scholar] [CrossRef] [PubMed]
- Boström, J.; Hogner, A.; Schmitt, S. Do Structurally Similar Ligands Bind in a Similar Fashion? J. Med. Chem. 2006, 49, 6716–6725. [Google Scholar] [CrossRef]
- Ceyhan, B.B.; Karakurt, S. Effect of oxolamine on cough sensitivity in COPD patients. Respir. Med. 2002, 96, 61–63. [Google Scholar] [CrossRef]
- Landfeldt, E.; Sejersen, T.; Tulinius, M. A mini-review and implementation model for using ataluren to treat nonsense mutation Duchenne muscular dystrophy. Acta Paediatr. 2019, 108, 224–230. [Google Scholar] [CrossRef]
- Konstan, M.W.; VanDevanter, D.R.; Rowe, S.M.; Wilschanski, M.; Kerem, E.; Sermet-Gaudelus, I.; DiMango, E.; Melotti, P.; McIntosh, J.; De Boeck, K. Efficacy and safety of ataluren in patients with nonsense-mutation cystic fibrosis not receiving chronic inhaled aminoglycosides: The international, randomized, double-blind, placebo-controlled Ataluren Confirmatory Trial in Cystic Fibrosis (ACT CF). J. Cyst. Fibros. 2020, 19, 595–601. [Google Scholar] [CrossRef]
- Tully, W.R.; Gardner, C.R.; Gillespie, R.J.; Westwood, R. 2-(Oxadiazolyl)- and 2-(thiazolyl)imidazo[1,2-a]pyrimidines as agonists and inverse agonists at benzodiazepine receptors. J. Med. Chem. 1991, 34, 2060–2067. [Google Scholar] [CrossRef]
- Cecchini, D.M.; Martinez, M.G.; Morganti, L.M.; Rodriguez, C.G. Antiretroviral Therapy Containing Raltegravir to Prevent Mother-to-Child Transmission of HIV in Infected Pregnant Women. Infect. Dis. Rep. 2017, 9, 7017. [Google Scholar] [CrossRef]
- Toombs, J.M.; Van den Abbeele, K.; Democratis, J.; Merricks, R.; Mandal, A.K.J.; Missouris, C.G. COVID-19 in three people living with HIV in the United Kingdom. J. Med. Virol. 2020, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Calligari, P.; Bobone, S.; Ricci, G.; Bocedi, A. Molecular Investigation of SARS-CoV-2 Proteins and Their Interactions with Antiviral Drugs. Viruses 2020, 12, 445. [Google Scholar] [CrossRef]
- Lloyd, M.D. High-Throughput Screening for the Discovery of Enzyme Inhibitors. J. Med. Chem. 2020, 63, 10742–10772. [Google Scholar] [CrossRef]
- Yoo, Y.J.; Feng, Y.; Kim, Y.H.; Yagonia, C.F.J. Specificity of Enzymes. In Fundamentals of Enzyme Engineering; Springer: Dordrecht, The Netherlands, 2017; pp. 111–125. [Google Scholar]
- Baig, M.H.; Adil, M.; Khan, R.; Dhadi, S.; Ahmad, K.; Rabbani, G.; Bashir, T.; Imran, M.A.; Husain, F.M.; Lee, E.J.; et al. Enzyme targeting strategies for prevention and treatment of cancer: Implications for cancer therapy. Semin. Cancer Biol. 2019, 56, 1–11. [Google Scholar] [CrossRef]
- Eitsuka, T.; Nakagawa, K.; Kato, S.; Ito, J.; Otoki, Y.; Takasu, S.; Shimizu, N.; Takahashi, T.; Miyazawa, T. Modulation of Telomerase Activity in Cancer Cells by Dietary Compounds: A Review. Int. J. Mol. Sci. 2018, 19, 478. [Google Scholar] [CrossRef]
- Zheng, Q.-Z.; Zhang, X.-M.; Xu, Y.; Cheng, K.; Jiao, Q.-C.; Zhu, H.-L. Synthesis, biological evaluation, and molecular docking studies of 2-chloropyridine derivatives possessing 1,3,4-oxadiazole moiety as potential antitumor agents. Bioorganic Med. Chem. 2010, 18, 7836–7841. [Google Scholar] [CrossRef]
- Gillis, A.J.; Schuller, A.P.; Skordalakes, E. Structure of the Tribolium castaneum telomerase catalytic subunit TERT. Nature 2008, 455, 633–637. [Google Scholar] [CrossRef]
- Zhang, X.-M.; Qiu, M.; Sun, J.; Zhang, Y.-B.; Yang, Y.-S.; Wang, X.-L.; Tang, J.-F.; Zhu, H.-L. Synthesis, biological evaluation, and molecular docking studies of 1,3,4-oxadiazole derivatives possessing 1,4-benzodioxan moiety as potential anticancer agents. Bioorganic Med. Chem. 2011, 19, 6518–6524. [Google Scholar] [CrossRef]
- Zhang, Y.-B.; Wang, X.-L.; Liu, W.; Yang, Y.-S.; Tang, J.-F.; Zhu, H.-L. Design, synthesis and biological evaluation of heterocyclic azoles derivatives containing pyrazine moiety as potential telomerase inhibitors. Bioorganic Med. Chem. 2012, 20, 6356–6365. [Google Scholar] [CrossRef]
- Sun, J.; Zhu, H.; Yang, Z.-M.; Zhu, H.-L. Synthesis, molecular modeling and biological evaluation of 2-aminomethyl-5-(quinolin-2-yl)-1,3,4-oxadiazole-2(3H)-thione quinolone derivatives as novel anticancer agent. Eur. J. Med. Chem. 2013, 60, 23–28. [Google Scholar] [CrossRef]
- Mboge, M.Y.; Mahon, B.P.; McKenna, R.; Frost, S.C. Carbonic Anhydrases: Role in pH Control and Cancer. Metabolites 2018, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Krasavin, M.; Shetnev, A.; Sharonova, T.; Baykov, S.; Tuccinardi, T.; Kalinin, S.; Angeli, A.; Supuran, C.T. Heterocyclic periphery in the design of carbonic anhydrase inhibitors: 1,2,4-Oxadiazol-5-yl benzenesulfonamides as potent and selective inhibitors of cytosolic hCA II and membrane-bound hCA IX isoforms. Bioorganic Chem. 2018, 76, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Krasavin, M.; Shetnev, A.; Sharonova, T.; Baykov, S.; Kalinin, S.; Nocentini, A.; Sharoyko, V.; Poli, G.; Tuccinardi, T.; Presnukhina, S.; et al. Continued exploration of 1,2,4-oxadiazole periphery for carbonic anhydrase-targeting primary arene sulfonamides: Discovery of subnanomolar inhibitors of membrane-bound hCA IX isoform that selectively kill cancer cells in hypoxic environment. Eur. J. Med. Chem. 2019, 164, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Delcuve, G.P.; Khan, D.H.; Davie, J.R. Roles of histone deacetylases in epigenetic regulation: Emerging paradigms from studies with inhibitors. Clin. Epigenetics 2012, 4, 5. [Google Scholar] [CrossRef]
- Ruzzolini, J.; Laurenzana, A.; Andreucci, E.; Peppicelli, S.; Bianchini, F.; Carta, F.; Supuran, C.T.; Romanelli, M.N.; Nediani, C.; Calorini, L. A potentiated cooperation of carbonic anhydrase IX and histone deacetylase inhibitors against cancer. J. Enzym. Inhib. Med. Chem. 2020, 35, 391–397. [Google Scholar] [CrossRef]
- Verza, F.A.; Das, U.; Fachin, A.L.; Dimmock, J.R.; Marins, M. Roles of Histone Deacetylases and Inhibitors in Anticancer Therapy. Cancers 2020, 12, 1664. [Google Scholar] [CrossRef]
- Rajak, H.; Agarawal, A.; Parmar, P.; Thakur, B.S.; Veerasamy, R.; Sharma, P.C.; Kharya, M.D. 2,5-Disubstituted-1,3,4-oxadiazoles/thiadiazole as surface recognition moiety: Design and synthesis of novel hydroxamic acid based histone deacetylase inhibitors. Bioorganic Med. Chem. Lett. 2011, 21, 5735–5738. [Google Scholar] [CrossRef]
- Valente, S.; Trisciuoglio, D.; De Luca, T.; Nebbioso, A.; Labella, D.; Lenoci, A.; Bigogno, C.; Dondio, G.; Miceli, M.; Brosch, G.; et al. 1,3,4-Oxadiazole-Containing Histone Deacetylase Inhibitors: Anticancer Activities in Cancer Cells. J. Med. Chem. 2014, 57, 6259–6265. [Google Scholar] [CrossRef]
- Kavanaugh, S.A.; White, L.A.; Kolesar, J.M. Vorinostat: A novel therapy for the treatment of cutaneous T-cell lymphoma. Am. J. Health-Syst. Pharm. 2010, 67, 793–797. [Google Scholar] [CrossRef]
- Benson, E.K.; Mungamuri, S.K.; Attie, O.; Kracikova, M.; Sachidanandam, R.; Manfredi, J.J.; Aaronson, S.A. p53-dependent gene repression through p21 is mediated by recruitment of E2F4 repression complexes. Oncogene 2014, 33, 3959–3969. [Google Scholar] [CrossRef]
- Cai, J.; Wei, H.; Hong, K.H.; Wu, X.; Cao, M.; Zong, X.; Li, L.; Sun, C.; Chen, J.; Ji, M. Discovery and preliminary evaluation of 2-aminobenzamide and hydroxamate derivatives containing 1,2,4-oxadiazole moiety as potent histone deacetylase inhibitors. Eur. J. Med. Chem. 2015, 96, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pidugu, V.R.; Yarla, N.S.; Pedada, S.R.; Kalle, A.M.; Satya, A.K. Design and synthesis of novel HDAC8 inhibitory 2,5-disubstituted-1,3,4-oxadiazoles containing glycine and alanine hybrids with anti cancer activity. Bioorganic Med. Chem. 2016, 24, 5611–5617. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Shan, P.; Zhao, N.; Ge, D.; Zhu, K.; Jiang, C.-S.; Li, P.; Zhang, H. Development of hydroxamate-based histone deacetylase inhibitors containing 1,2,4-oxadiazole moiety core with antitumor activities. Bioorganic Med. Chem. Lett. 2019, 29, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Shen, M.; Tang, M.; Zhang, W.; Cui, X.; Zhang, Z.; Pei, H.; Li, Y.; Hu, M.; Bai, P.; et al. Discovery of 1,2,4-oxadiazole-Containing hydroxamic acid derivatives as histone deacetylase inhibitors potential application in cancer therapy. Eur. J. Med. Chem. 2019, 178, 116–130. [Google Scholar] [CrossRef]
- Bhullar, K.S.; Lagarón, N.O.; McGowan, E.M.; Parmar, I.; Jha, A.; Hubbard, B.P.; Rupasinghe, H.P.V. Kinase-targeted cancer therapies: Progress, challenges and future directions. Mol. Cancer 2018, 17, 48. [Google Scholar] [CrossRef]
- Zhang, S.; Luo, Y.; He, L.-Q.; Liu, Z.-J.; Jiang, A.-Q.; Yang, Y.-H.; Zhu, H.-L. Synthesis, biological evaluation, and molecular docking studies of novel 1,3,4-oxadiazole derivatives possessing benzotriazole moiety as FAK inhibitors with anticancer activity. Bioorganic Med. Chem. 2013, 21, 3723–3729. [Google Scholar] [CrossRef]
- Barlaam, B.; Cosulich, S.; Delouvrié, B.; Ellston, R.; Fitzek, M.; Germain, H.; Green, S.; Hancox, U.; Harris, C.S.; Hudson, K.; et al. Discovery of 1-(4-(5-(5-amino-6-(5-tert-butyl-1,3,4-oxadiazol-2-yl)pyrazin-2-yl)-1-ethyl-1,2,4-triazol-3-yl)piperidin-1-yl)-3-hydroxypropan-1-one (AZD8835): A potent and selective inhibitor of PI3Kα and PI3Kδ for the treatment of cancers. Bioorganic Med. Chem. Lett. 2015, 25, 5155–5162. [Google Scholar] [CrossRef]
- Han, M.; Li, S.; Ai, J.; Sheng, R.; Hu, Y.; Geng, M. Discovery of 4-chloro-3-(5-(pyridin-3-yl)-1,2,4-oxadiazole-3-yl)benzamides as novel RET kinase inhibitors. Bioorganic Med. Chem. Lett. 2016, 26, 5679–5684. [Google Scholar] [CrossRef]
- Liu, K.; Lu, X.; Zhang, H.-J.; Sun, J.; Zhu, H.-L. Synthesis, molecular modeling and biological evaluation of 2-(benzylthio)-5-aryloxadiazole derivatives as anti-tumor agents. Eur. J. Med. Chem. 2012, 47, 473–478. [Google Scholar] [CrossRef]
- Dokla, E.M.E.; Fang, C.S.; Abouzid, K.A.M.; Chen, C.S. 1,2,4-Oxadiazole derivatives targeting EGFR and c-Met degradation in TKI resistant NSCLC. Eur. J. Med. Chem. 2019, 182, 111607. [Google Scholar] [CrossRef]
- Zhang, X.; Gureasko, J.; Shen, K.; Cole, P.A.; Kuriyan, J. An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell 2006, 125, 1137–1149. [Google Scholar] [CrossRef] [PubMed]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging functions of the EGFR in cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Polothi, R.; Raolji, G.S.B.; Kuchibhotla, V.S.; Sheelam, K.; Tuniki, B.; Thodupunuri, P. Synthesis and biological evaluation of 1,2,4-oxadiazole linked 1,3,4-oxadiazole derivatives as tubulin binding agents. Synth. Commun. 2019, 49, 1603–1612. [Google Scholar] [CrossRef]
- Ozcan, S.; Kazi, A.; Marsilio, F.; Fang, B.; Guida, W.C.; Koomen, J.; Lawrence, H.R.; Sebti, S.M. Oxadiazole-isopropylamides as Potent and Noncovalent Proteasome Inhibitors. J. Med. Chem. 2013, 56, 3783–3805. [Google Scholar] [CrossRef]
- Zhang, N.; Yin, Y.; Xu, S.-J.; Chen, W.-S. 5-Fluorouracil: Mechanisms of resistance and reversal strategies. Molecules 2008, 13, 1551–1569. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.-R.; Li, D.-D.; Pi, Y.-Z.; Li, J.-R.; Sun, J.; Fang, F.; Zhong, W.-Q.; Gong, H.-B.; Zhu, H.-L. Novel 1,3,4-oxadiazole thioether derivatives targeting thymidylate synthase as dual anticancer/antimicrobial agents. Bioorganic Med. Chem. 2013, 21, 2286–2297. [Google Scholar] [CrossRef]
- Subba Rao, A.V.; Vishnu Vardhan, M.V.P.S.; Subba Reddy, N.V.; Srinivasa Reddy, T.; Shaik, S.P.; Bagul, C.; Kamal, A. Synthesis and biological evaluation of imidazopyridinyl-1,3,4-oxadiazole conjugates as apoptosis inducers and topoisomerase IIα inhibitors. Bioorganic Chem. 2016, 69, 7–19. [Google Scholar] [CrossRef]
- Abd El Hameid, M.K. Design, Synthesis, and Screening of 5-Aryl-3-(2-(pyrrolyl)thiophenyl)-1,2,4-oxadiazoles as Potential Antitumor Molecules on Breast Cancer MCF-7 Cell Line. Chem. Pharm. Bull. 2018, 66, 1181–1195. [Google Scholar] [CrossRef]
- Dai, H.; Sinclair, D.A.; Ellis, J.L.; Steegborn, C. Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacol. Ther. 2018, 188, 140–154. [Google Scholar] [CrossRef]
- Carafa, V.; Rotili, D.; Forgione, M.; Cuomo, F.; Serretiello, E.; Hailu, G.S.; Jarho, E.; Lahtela-Kakkonen, M.; Mai, A.; Altucci, L. Sirtuin functions and modulation: From chemistry to the clinic. Clin. Epigenetics 2016, 8, 61. [Google Scholar] [CrossRef]
- Schlicker, C.; Boanca, G.; Lakshminarasimhan, M.; Steegborn, C. Structure-based development of novel sirtuin inhibitors. Aging 2011, 3, 852–872. [Google Scholar] [CrossRef] [PubMed]
- Moniot, S.; Forgione, M.; Lucidi, A.; Hailu, G.S.; Nebbioso, A.; Carafa, V.; Baratta, F.; Altucci, L.; Giacché, N.; Passeri, D.; et al. Development of 1,2,4-Oxadiazoles as Potent and Selective Inhibitors of the Human Deacetylase Sirtuin 2: Structure–Activity Relationship, X-ray Crystal Structure, and Anticancer Activity. J. Med. Chem. 2017, 60, 2344–2360. [Google Scholar] [CrossRef] [PubMed]
- Janke, C.; Magiera, M.M. The tubulin code and its role in controlling microtubule properties and functions. Nat. Rev. Mol. Cell Biol. 2020, 21, 307–326. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, R.; Pilotto, S.; Peretti, U.; Caccese, M.; Kinspergher, S.; Carbognin, L.; Karachaliou, N.; Rosell, R.; Tortora, G.; Bria, E. Tubulin inhibitors in non-small cell lung cancer: Looking back and forward. Expert Opin Pharm. 2016, 17, 1113–1129. [Google Scholar] [CrossRef] [PubMed]
- Haider, K.; Rahaman, S.; Yar, M.S.; Kamal, A. Tubulin inhibitors as novel anticancer agents: An overview on patents (2013-2018). Expert Opin. Ther. Pat. 2019, 29, 623–641. [Google Scholar] [CrossRef]
- Martino, E.; Casamassima, G.; Castiglione, S.; Cellupica, E.; Pantalone, S.; Papagni, F.; Rui, M.; Siciliano, A.M.; Collina, S. Vinca alkaloids and analogues as anti-cancer agents: Looking back, peering ahead. Bioorganic Med. Chem. Lett. 2018, 28, 2816–2826. [Google Scholar] [CrossRef]
- Cummings, J.; Zweifel, M.; Smith, N.; Ross, P.; Peters, J.; Rustin, G.; Price, P.; Middleton, M.R.; Ward, T.; Dive, C. Evaluation of cell death mechanisms induced by the vascular disrupting agent OXi4503 during a phase I clinical trial. Br. J. Cancer 2012, 106, 1766–1771. [Google Scholar] [CrossRef]
- Ji, Y.T.; Liu, Y.N.; Liu, Z.P. Tubulin colchicine binding site inhibitors as vascular disrupting agents in clinical developments. Curr. Med. Chem. 2015, 22, 1348–1360. [Google Scholar] [CrossRef]
- Inglis, D.J.; Lavranos, T.C.; Beaumont, D.M.; Leske, A.F.; Brown, C.K.; Hall, A.J.; Kremmidiotis, G. The vascular disrupting agent BNC105 potentiates the efficacy of VEGF and mTOR inhibitors in renal and breast cancer. Cancer Biol. Ther. 2014, 15, 1552–1560. [Google Scholar] [CrossRef]
- Mita, M.M.; Spear, M.A.; Yee, L.K.; Mita, A.C.; Heath, E.I.; Papadopoulos, K.P.; Federico, K.C.; Reich, S.D.; Romero, O.; Malburg, L.; et al. Phase 1 First-in-Human Trial of the Vascular Disrupting Agent Plinabulin (NPI-2358) in Patients with Solid Tumors or Lymphomas. Clin. Cancer Res. 2010, 16, 5892. [Google Scholar] [CrossRef]
- Nieddu, V.; Pinna, G.; Marchesi, I.; Sanna, L.; Asproni, B.; Pinna, G.A.; Bagella, L.; Murineddu, G. Synthesis and Antineoplastic Evaluation of Novel Unsymmetrical 1,3,4-Oxadiazoles. J. Med. Chem. 2016, 59, 10451–10469. [Google Scholar] [CrossRef] [PubMed]
- Kamal, A.; Shaik, A.B.; Polepalli, S.; Santosh Reddy, V.; Bharath Kumar, G.; Gupta, S.; Rama Krishna, K.V.S.; Nagabhushana, A.; Mishra, R.K.; Jain, N. Pyrazole–oxadiazole conjugates: Synthesis, antiproliferative activity and inhibition of tubulin polymerization. Org. Biomol. Chem. 2014, 12, 7993–8007. [Google Scholar] [CrossRef] [PubMed]
- Kamal, A.; Srikanth, P.S.; Vishnuvardhan, M.V.P.S.; Kumar, G.B.; Suresh Babu, K.; Hussaini, S.M.A.; Kapure, J.S.; Alarifi, A. Combretastatin linked 1,3,4-oxadiazole conjugates as a Potent Tubulin Polymerization inhibitors. Bioorganic Chem. 2016, 65, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Terenzi, A.; Fanelli, M.; Ambrosi, G.; Amatori, S.; Fusi, V.; Giorgi, L.; Turco Liveri, V.; Barone, G. DNA binding and antiproliferative activity toward human carcinoma cells of copper(II) and zinc(II) complexes of a 2,5-diphenyl[1,3,4]oxadiazole derivative. Dalton Trans. 2012, 41, 4389–4395. [Google Scholar] [CrossRef]
- Terenzi, A.; Barone, G.; Piccionello, A.P.; Giorgi, G.; Guarcello, A.; Portanova, P.; Calvaruso, G.; Buscemi, S.; Vivona, N.; Pace, A. Synthesis, characterization, cellular uptake and interaction with native DNA of a bis(pyridyl)-1,2,4-oxadiazole copper(II) complex. Dalton Trans. 2010, 39, 9140–9145. [Google Scholar] [CrossRef]
- Jadala, C.; Sathish, M.; Reddy, T.S.; Reddy, V.G.; Tokala, R.; Bhargava, S.K.; Shankaraiah, N.; Nagesh, N.; Kamal, A. Synthesis and in vitro cytotoxicity evaluation of β-carboline-combretastatin carboxamides as apoptosis inducing agents: DNA intercalation and topoisomerase-II inhibition. Bioorganic Med. Chem. 2019, 27, 3285–3298. [Google Scholar] [CrossRef]
- Sathish, M.; Chetan Dushantrao, S.; Nekkanti, S.; Tokala, R.; Thatikonda, S.; Tangella, Y.; Srinivas, G.; Cherukommu, S.; Hari Krishna, N.; Shankaraiah, N.; et al. Synthesis of DNA interactive C3-trans-cinnamide linked β-carboline conjugates as potential cytotoxic and DNA topoisomerase I inhibitors. Bioorganic Med. Chem. 2018, 26, 4916–4929. [Google Scholar] [CrossRef]
- Formagio, A.S.N.; Tonin, L.T.D.; Foglio, M.A.; Madjarof, C.; de Carvalho, J.E.; da Costa, W.F.; Cardoso, F.P.; Sarragiotto, M.H. Synthesis and antitumoral activity of novel 3-(2-substituted-1,3,4-oxadiazol-5-yl) and 3-(5-substituted-1,2,4-triazol-3-yl) β-carboline derivatives. Bioorganic Med. Chem. 2008, 16, 9660–9667. [Google Scholar] [CrossRef]
- Savariz, F.C.; Foglio, M.A.; Goes Ruiz, A.L.T.; da Costa, W.F.; de Magalhães Silva, M.; Santos, J.C.C.; Figueiredo, I.M.; Meyer, E.; de Carvalho, J.E.; Sarragiotto, M.H. Synthesis and antitumor activity of novel 1-substituted phenyl 3-(2-oxo-1,3,4-oxadiazol-5-yl) β-carbolines and their Mannich bases. Bioorganic Med. Chem. 2014, 22, 6867–6875. [Google Scholar] [CrossRef]
- Petenzi, M.; Verga, D.; Largy, E.; Hamon, F.; Doria, F.; Teulade-Fichou, M.P.; Guédin, A.; Mergny, J.L.; Mella, M.; Freccero, M. Cationic pentaheteroaryls as selective G-quadruplex ligands by solvent-free microwave-assisted synthesis. Chemistry 2012, 18, 14487–14496. [Google Scholar] [CrossRef]
- Pirota, V.; Stasi, M.; Benassi, A.; Doria, F. Chapter Six-An overview of quadruplex ligands: Their common features and chemotype diversity. In Annual Reports in Medicinal Chemistry; Neidle, S., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 54, pp. 163–196. [Google Scholar]
- Doria, F.; Pirota, V.; Petenzi, M.; Teulade-Fichou, M.P.; Verga, D.; Freccero, M. Oxadiazole/Pyridine-Based Ligands: A Structural Tuning for Enhancing G-Quadruplex Binding. Molecules 2018, 23, 2162. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Liu, J.; Xiong, X.; Cheng, M.; Hu, X.; Narva, S.; Zhao, X.; Wu, Y.; Zhang, W. Design, synthesis of 4,5-diazafluorene derivatives and their anticancer activity via targeting telomeric DNA G-quadruplex. Eur. J. Med. Chem. 2019, 178, 484–499. [Google Scholar] [CrossRef] [PubMed]
- Dalzoppo, D.; Di Paolo, V.; Calderan, L.; Pasut, G.; Rosato, A.; Caccuri, A.M.; Quintieri, L. Thiol-Activated Anticancer Agents: The State of the Art. Anticancer Agents Med. Chem. 2017, 17, 4–20. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Rakesh, K.P.; Ravidar, L.; Fang, W.Y.; Qin, H.L. Pharmaceutical and medicinal significance of sulfur (S(VI))-Containing motifs for drug discovery: A critical review. Eur. J. Med. Chem. 2019, 162, 679–734. [Google Scholar] [CrossRef]
- Thasneem, C.K.; Biju, C.R.; Babu, G. Synthesis and anticancer study of chalcone linked 1,3,4-oxadiazole derivatives. Int. J. Pharm. Bio. Sci. 2014, 4, 20–28. [Google Scholar]
- Vazquez, M.R.; Kantarjian, H.M.; Borthakur, G.M.; O’Brien, S.; Verstovsek, S.; Ravandi, F.; Ferrajoli, A.; Cortes, J.E.; Kadia, T.M.; Wierda, W.G.; et al. A 20-Year Review of Imatinib in Chronic Phase Chronic Myeloid Leukemia Patients after Failure with Interferon Therapy. Blood 2019, 134, 2927. [Google Scholar] [CrossRef]
- Li, Y.-T.; Wang, J.-H.; Pan, C.-W.; Meng, F.-F.; Chu, X.-Q.; Ding, Y.-h.; Qu, W.-Z.; Li, H.-Y.; Yang, C.; Zhang, Q.; et al. Syntheses and biological evaluation of 1,2,3-triazole and 1,3,4-oxadiazole derivatives of imatinib. Bioorganic Med. Chem. Lett. 2016, 26, 1419–1427. [Google Scholar] [CrossRef]
- Chae, H.-S.; Chin, Y.-W. Anti-allergic effect of lambertianic acid from Thuja orientalis in mouse bone marrow-derived mast cells. Immunopharmacol. Immunotoxicol. 2012, 34, 250–255. [Google Scholar] [CrossRef]
- Mironov , M.E.; Pokrovsky, M.A.; Kharitonov , Y.V.; Shakirov, M.M.; Pokrovsky, A.G.; Shults , E.E. Furanolabdanoid–based 1,2,4-oxadiazoles: Synthesis and cytotoxic activity. ChemistrySelect 2016, 1, 417–424. [Google Scholar] [CrossRef]
- Markov, A.V.; Sen’kova, A.V.; Popadyuk, I.I.; Salomatina, O.V.; Logashenko, E.B.; Komarova, N.I.; Ilyina, A.A.; Salakhutdinov, N.F.; Zenkova, M.A. Novel 3’-Substituted-1’,2’,4’-Oxadiazole Derivatives of 18βH-Glycyrrhetinic Acid and Their O-Acylated Amidoximes: Synthesis and Evaluation of Antitumor and Anti-Inflammatory Potential In Vitro and In Vivo. Int. J. Mol. Sci. 2020, 21, 3511. [Google Scholar] [CrossRef]
- Hussain, H.; Green, I.R.; Shamraiz, U.; Saleem, M.; Badshah, A.; Abbas, G.; Rehman, N.U.; Irshad, M. Therapeutic potential of glycyrrhetinic acids: A patent review (2010-2017). Expert Opin. Ther. Pat. 2018, 28, 383–398. [Google Scholar] [CrossRef] [PubMed]
- Gamal El-Din, M.M.; El-Gamal, M.I.; Abdel-Maksoud, M.S.; Yoo, K.H.; Oh, C.-H. Synthesis and broad-spectrum antiproliferative activity of diarylamides and diarylureas possessing 1,3,4-oxadiazole derivatives. Bioorganic Med. Chem. Lett. 2015, 25, 1692–1699. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, M.J.; Sharma, J.; Singh, M.; Jadav, S.S.; Yasmin, S. Synthesis and Anticancer Activity of N-Aryl-5-substituted-1,3,4-oxadiazol-2-amine Analogues. Biomed Res. Int. 2014, 2014, 814984. [Google Scholar] [CrossRef][Green Version]
- Salahuddinm; Mazumder, A.; Shaharyar, M. Synthesis, Characterization, and In Vitro Anticancer Evaluation of Novel 2,5-Disubstituted 1,3,4-Oxadiazole Analogue. Biomed Res. Int. 2014, 2014, 491492. [Google Scholar] [CrossRef]
- Srinivas, M.; Satyaveni, S.; Ram, B. Synthesis and Anticancer Activity of 1,2,4-Oxadiazol Linked Benzimidazole Derivatives. Russ. J. Gen. Chem. 2018, 88, 2653–2657. [Google Scholar] [CrossRef]
- Gurupadaswamy, H.D.; Girish, V.; Kavitha, C.V.; Raghavan, S.C.; Khanum, S.A. Synthesis and evaluation of 2,5-di(4-aryloylaryloxymethyl)-1,3,4-oxadiazoles as anti-cancer agents. Eur. J. Med. Chem. 2013, 63, 536–543. [Google Scholar] [CrossRef]
- Gurupadaswamy, H.D.; Thirusangu, P.; Vijay Avin, B.R.; Vigneshwaran, V.; Prashanth Kumar, M.V.; Abhishek, T.S.; Lakshmi Ranganatha, V.; Khanum, S.A.; Prabhakar, B.T. DAO-9 (2,5-di(4-aryloylaryloxymethyl)-1,3,4-oxadiazole) exhibits p53 induced apoptogenesis through caspase-3 mediated endonuclease activity in murine carcinoma. Biomed. Pharmacother. 2014, 68, 791–797. [Google Scholar] [CrossRef]
- Tiwari, A.; Gopalan Kutty, N.; Kumar, N.; Chaudhary, A.; Vasanth Raj, P.; Shenoy, R.; Mallikarjuna Rao, C. Synthesis and evaluation of selected 1,3,4-oxadiazole derivatives for in vitro cytotoxicity and in vivo anti-tumor activity. Cytotechnology 2016, 68, 2553–2565. [Google Scholar] [CrossRef][Green Version]
- Mochona, B.; Qi, X.; Euynni, S.; Sikazwi, D.; Mateeva, N.; Soliman, K.F. Design and evaluation of novel oxadiazole derivatives as potential prostate cancer agents. Bioorganic Med. Chem. Lett. 2016, 26, 2847–2851. [Google Scholar] [CrossRef]
- Naresh Kumar, R.; Poornachandra, Y.; Nagender, P.; Santhosh Kumar, G.; Krishna Swaroop, D.; Ganesh Kumar, C.; Narsaiah, B. Synthesis of novel nicotinohydrazide and (1,3,4-oxadiazol-2-yl)-6-(trifluoromethyl)pyridine derivatives as potential anticancer agents. Bioorganic Med. Chem. Lett. 2016, 26, 4829–4831. [Google Scholar] [CrossRef]
- Cascioferro, S.; Attanzio, A.; Di Sarno, V.; Musella, S.; Tesoriere, L.; Cirrincione, G.; Diana, P.; Parrino, B. New 1,2,4-Oxadiazole Nortopsentin Derivatives with Cytotoxic Activity. Mar. Drugs 2019, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Maftei, C.V.; Fodor, E.; Jones, P.G.; Daniliuc, C.G.; Franz, M.H.; Kelter, G.; Fiebig, H.-H.; Tamm, M.; Neda, I. Novel 1,2,4-oxadiazoles and trifluoromethylpyridines related to natural products: Synthesis, structural analysis and investigation of their antitumor activity. Tetrahedron 2016, 72, 1185–1199. [Google Scholar] [CrossRef]
- Yadagiri, B.; Gurrala, S.; Bantu, R.; Nagarapu, L.; Polepalli, S.; Srujana, G.; Jain, N. Synthesis and evaluation of benzosuberone embedded with 1,3,4-oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole moieties as new potential anti proliferative agents. Bioorganic Med. Chem. Lett. 2015, 25, 2220–2224. [Google Scholar] [CrossRef] [PubMed]
- Tu, G.; Yan, Y.; Chen, X.; Lv, Q.; Wang, J.; Li, S. Synthesis and antiproliferative assay of 1,3,4-oxadiazole and 1,2,4-triazole derivatives in cancer cells. Drug Discov. Ther. 2013, 7, 58–65. [Google Scholar] [PubMed]
- Yonova, I.M.; Osborne, C.A.; Morrissette, N.S.; Jarvo, E.R. Diaryl and Heteroaryl Sulfides: Synthesis via Sulfenyl Chlorides and Evaluation as Selective Anti-Breast-Cancer Agents. J. Org. Chem. 2014, 79, 1947–1953. [Google Scholar] [CrossRef] [PubMed]
- Prasanna Kumar, B.N.; Mohana, K.N.; Mallesha, L.; Veeresh, B. Synthesis and in vitro antiproliferative activity of 2,5-disubstituted-1,3,4-oxadiazoles containing trifluoromethyl benzenesulfonamide moiety. Med. Chem. Res. 2014, 23, 3363–3373. [Google Scholar] [CrossRef]
- Ertl, P.; Altmann, E.; McKenna, J.M. The Most Common Functional Groups in Bioactive Molecules and How Their Popularity Has Evolved over Time. J. Med. Chem. 2020, 63, 8408–8418. [Google Scholar] [CrossRef]
- Khalil, N.A.; Kamal, A.M.; Emam, S.H. Design, Synthesis, and Antitumor Activity of Novel 5-Pyridyl-1,3,4-oxadiazole Derivatives against the Breast Cancer Cell Line MCF-7. Biol. Pharm. Bull. 2015, 38, 763–773. [Google Scholar] [CrossRef]
- Zhao, J.-J.; Wang, X.-F.; Li, B.-L.; Zhang, R.-L.; Li, B.; Liu, Y.-M.; Li, C.-W.; Liu, J.-B.; Chen, B.-Q. Synthesis and in vitro antiproliferative evaluation of novel nonsymmetrical disulfides bearing 1,3,4-oxadiazole moiety. Bioorganic Med. Chem. Lett. 2016, 26, 4414–4416. [Google Scholar] [CrossRef]
- Bacharaju, K.; Jambula, S.R.; Sivan, S.; JyostnaTangeda, S.; Manga, V. Design, synthesis, molecular docking and biological evaluation of new dithiocarbamates substituted benzimidazole and chalcones as possible chemotherapeutic agents. Bioorganic Med. Chem. Lett. 2012, 22, 3274–3277. [Google Scholar] [CrossRef]
- Li, Y.-B.; Yan, X.; Li, R.-D.; Liu, P.; Sun, S.-Q.; Wang, X.; Cui, J.-R.; Zhou, D.-M.; Ge, Z.-M.; Li, R.-T. Discovery of novel heteroarylmethylcarbamodithioates as potent anticancer agents: Synthesis, structure-activity relationship analysis and biological evaluation. Eur. J. Med. Chem. 2016, 112, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. How Taxol/paclitaxel kills cancer cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef] [PubMed]
- Avanzo, R.E.; Padrón, J.M.; D’Accorso, N.B.; Fascio, M.L. Synthesis and in vitro antiproliferative activities of (5-aryl-1,2,4-oxadiazole-3-yl) methyl d-ribofuranosides. Bioorganic Med. Chem. Lett. 2017, 27, 3674–3677. [Google Scholar] [CrossRef]
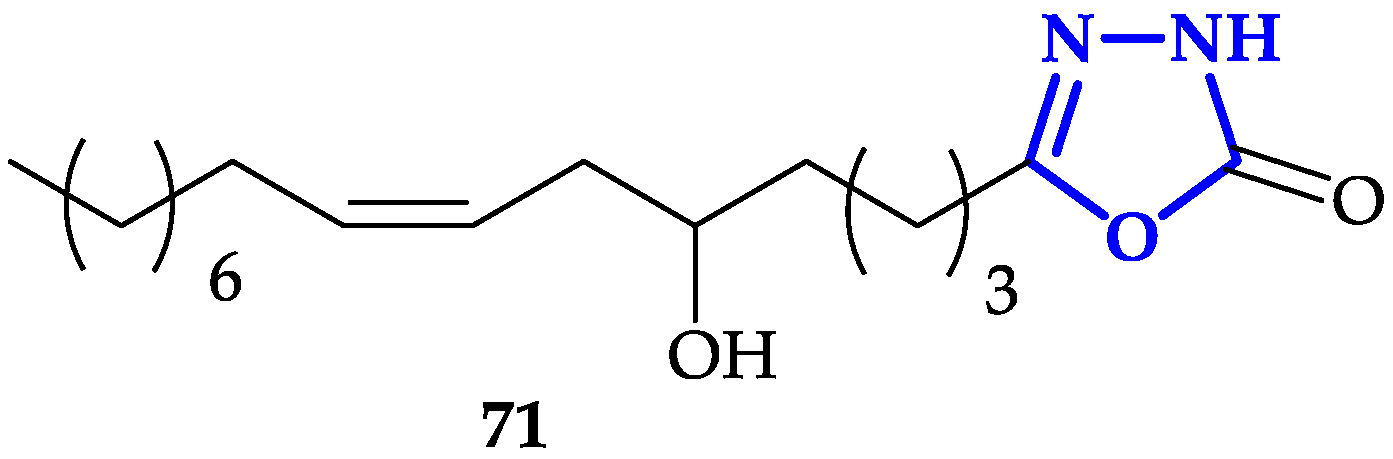
- Jóźwiak, M.; Filipowska, A.; Fiorino, F.; Struga, M. Anticancer activities of fatty acids and their heterocyclic derivatives. Eur. J. Pharmacol. 2020, 871, 172937. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.F.; Rauf, A.; Sherwani, A.; Owais, M. Synthesis and In Vitro Biological Evaluation of 1,3,4-Oxadiazol-2(3H)-one and Tetrahydropyridazine-3,6-dione Derivatives of Fatty Acids. Sci. Pharm. 2015, 83, 429–443. [Google Scholar] [CrossRef] [PubMed][Green Version]
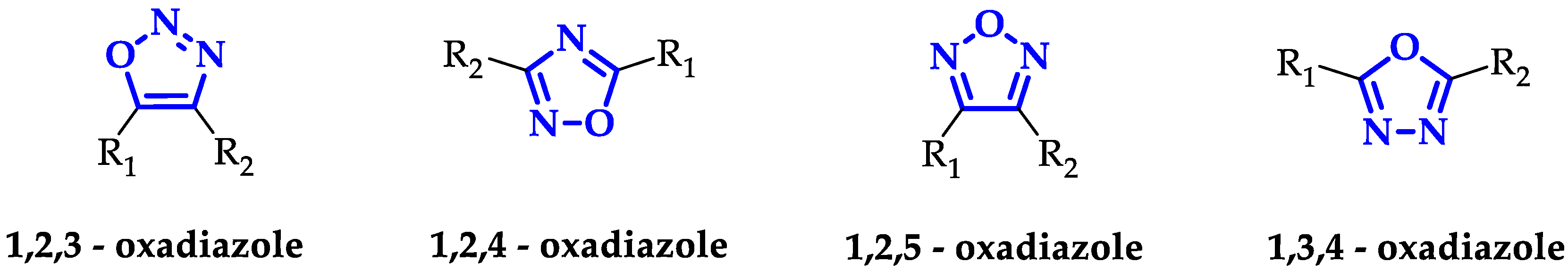






Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benassi, A.; Doria, F.; Pirota, V. Groundbreaking Anticancer Activity of Highly Diversified Oxadiazole Scaffolds. Int. J. Mol. Sci. 2020, 21, 8692. https://doi.org/10.3390/ijms21228692
Benassi A, Doria F, Pirota V. Groundbreaking Anticancer Activity of Highly Diversified Oxadiazole Scaffolds. International Journal of Molecular Sciences. 2020; 21(22):8692. https://doi.org/10.3390/ijms21228692
Chicago/Turabian StyleBenassi, Alessandra, Filippo Doria, and Valentina Pirota. 2020. "Groundbreaking Anticancer Activity of Highly Diversified Oxadiazole Scaffolds" International Journal of Molecular Sciences 21, no. 22: 8692. https://doi.org/10.3390/ijms21228692
APA StyleBenassi, A., Doria, F., & Pirota, V. (2020). Groundbreaking Anticancer Activity of Highly Diversified Oxadiazole Scaffolds. International Journal of Molecular Sciences, 21(22), 8692. https://doi.org/10.3390/ijms21228692






