The Role of Cell Cycle Regulators in Cell Survival—Dual Functions of Cyclin-Dependent Kinase 20 and p21Cip1/Waf1
Abstract
1. Introduction
2. CDK20 and p21Cip1/Waf1 in Cell Cycle and Cell Growth
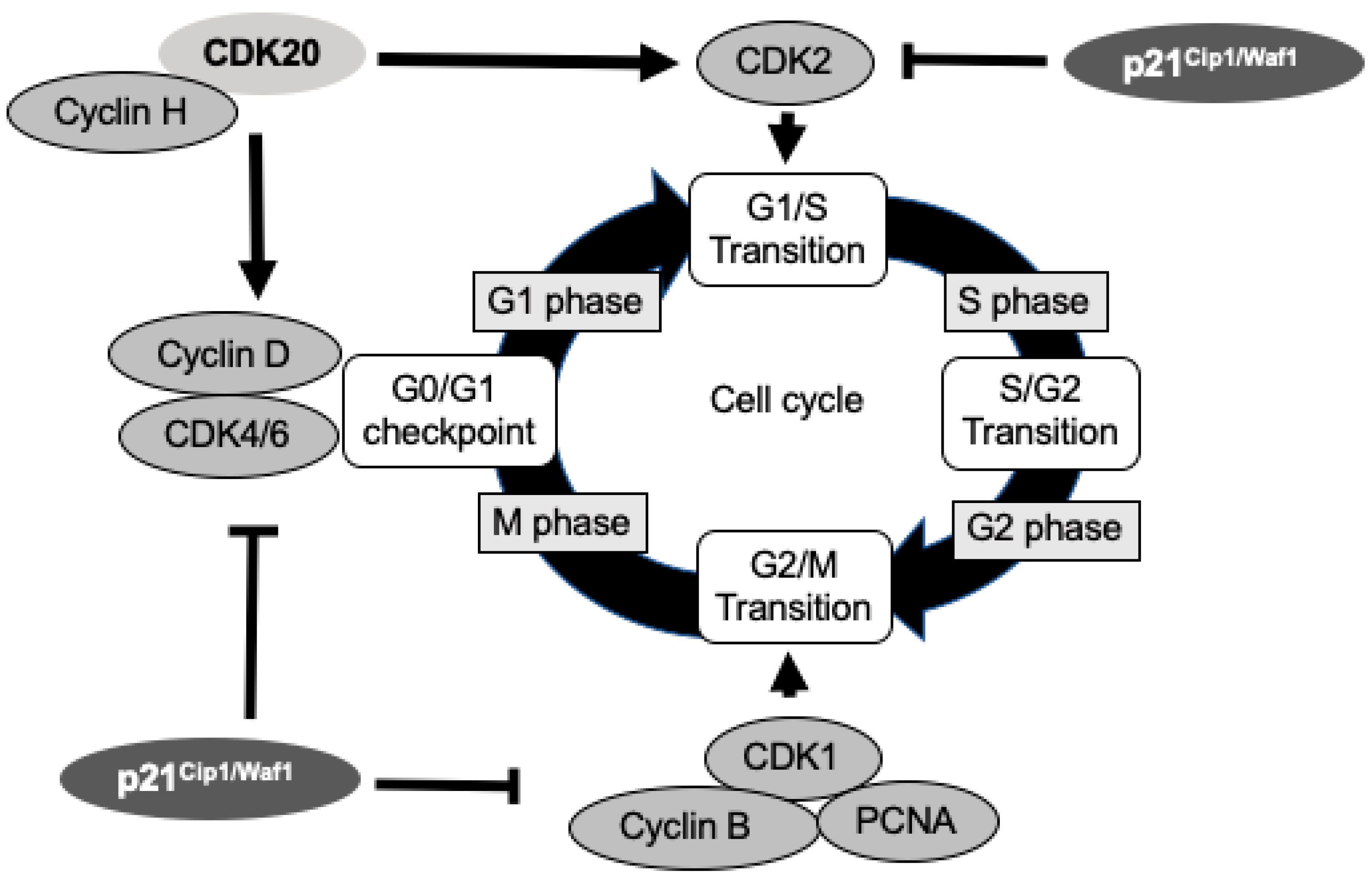
2.1. CDK20 and p21Cip1/Waf1 Regulate Cell Cycle via CDK2
2.2. The Role and Mechanisms of CDK20 and p21Cip1/Waf1 in Cell Growth
3. CDK20 and p21Cip1/Waf1 in Cell Death and Survival
3.1. CDK20 in the Regulation in Cell Survival and Its Variants
3.2. The Dual Roles of p21Cip1/Waf1 in Cell Death and Survival and Its Compartment-Specific Effects
4. The Relevance of CDK20 and p21Cip1/Waf1 in Cancers
5. Potential Role of CDK20 and p21Cip1/Waf1 in Cardio-Protection
6. Clinical Potential
7. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- Barnum, K.J.; O’Connell, M.J. Cell cycle regulation by checkpoints. Methods Mol. Biol. 2014, 1170, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Gordon, E.M.; Ravicz, J.R.; Liu, S.; Chawla, S.P.; Hall, F.L. Cell cycle checkpoint control: The cyclin G1/Mdm2/p53 axis emerges as a strategic target for broad-spectrum cancer gene therapy—A review of molecular mechanisms for oncologists. Mol. Clin. Oncol. 2018, 9, 115–134. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, G.K.; Shah, M.A. Targeting the cell cycle: A new approach to cancer therapy. J. Clin. Oncol. 2005, 23, 9408–9421. [Google Scholar] [CrossRef]
- Visconti, R.; Della Monica, R.; Grieco, D. Cell cycle checkpoint in cancer: A therapeutically targetable double-edged sword. J. Exp. Clin. Cancer Res. 2016, 35, 153. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M. Cyclin-dependent kinases. Genome Biol. 2014, 15, 122. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M.; Harlow, E.; Hunt, T.; Hunter, T.; Lahti, J.M.; Manning, G.; Morgan, D.O.; Tsai, L.H.; Wolgemuth, D.J. Cyclin-dependent kinases: A family portrait. Nat. Cell Biol. 2009, 11, 1275–1276. [Google Scholar] [CrossRef]
- Tian, Y.; Wan, H.; Tan, G. Cell cycle-related kinase in carcinogenesis. Oncol. Lett. 2012, 4, 601–606. [Google Scholar] [CrossRef]
- Kreis, N.N.; Louwen, F.; Yuan, J. The Multifaceted p21 (Cip1/Waf1/CDKN1A) in Cell Differentiation, Migration and Cancer Therapy. Cancers 2019, 11, 1220. [Google Scholar] [CrossRef]
- Li, J.M.; Brooks, G. Cell cycle regulatory molecules (cyclins, cyclin-dependent kinases and cyclin-dependent kinase inhibitors) and the cardiovascular system; potential targets for therapy? Eur. Heart J. 1999, 20, 406–420. [Google Scholar] [CrossRef]
- Xu, M.; Wan, C.X.; Huang, S.H.; Wang, H.B.; Fan, D.; Wu, H.M.; Wu, Q.Q.; Ma, Z.G.; Deng, W.; Tang, Q.Z. Oridonin protects against cardiac hypertrophy by promoting P21-related autophagy. Cell Death Dis. 2019, 10, 403. [Google Scholar] [CrossRef]
- Georgakilas, A.G.; Martin, O.A.; Bonner, W.M. p21: A Two-Faced Genome Guardian. Trends Mol. Med. 2017, 23, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, Y.; Abu El Maaty, M.A.; Chan, H.Y.; Wolker, J.; Ott, I.; Wolfl, S.; Cheng, X. p53-Dependent Anti-Proliferative and Pro-Apoptotic Effects of a Gold(I) N-Heterocyclic Carbene (NHC) Complex in Colorectal Cancer Cells. Front. Oncol. 2019, 9, 438. [Google Scholar] [CrossRef] [PubMed]
- Manu, K.A.; Cao, P.H.A.; Chai, T.F.; Casey, P.J.; Wang, M. p21cip1/waf1 Coordinate Autophagy, Proliferation and Apoptosis in Response to Metabolic Stress. Cancers 2019, 11, 1112. [Google Scholar] [CrossRef] [PubMed]
- Zohny, S.F.; Al-Malki, A.L.; Zamzami, M.A.; Choudhry, H. p21Waf1/Cip1: Its paradoxical effect in the regulation of breast cancer. Breast Cancer 2019, 26, 131–137. [Google Scholar] [CrossRef]
- Parveen, A.; Akash, M.S.; Rehman, K.; Kyunn, W.W. Dual Role of p21 in the Progression of Cancer and Its Treatment. Crit. Rev. Eukaryot Gene Expr. 2016, 26, 49–62. [Google Scholar] [CrossRef]
- Mok, M.T.; Zhou, J.; Tang, W.; Zeng, X.; Oliver, A.W.; Ward, S.E.; Cheng, A.S. CCRK is a novel signalling hub exploitable in cancer immunotherapy. Pharmacol. Ther. 2018, 186, 138–151. [Google Scholar] [CrossRef]
- Echalier, A.; Endicott, J.A.; Noble, M.E. Recent developments in cyclin-dependent kinase biochemical and structural studies. Biochim. Biophys. Acta 2010, 1804, 511–519. [Google Scholar] [CrossRef]
- Satyanarayana, A.; Kaldis, P. Mammalian cell-cycle regulation: Several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene 2009, 28, 2925–2939. [Google Scholar] [CrossRef]
- Su, T.T.; Stumpff, J. Promiscuity rules? The dispensability of cyclin E and Cdk2. Sci. STKE 2004, 2004, pe11. [Google Scholar] [CrossRef][Green Version]
- Berthet, C.; Aleem, E.; Coppola, V.; Tessarollo, L.; Kaldis, P. Cdk2 knockout mice are viable. Curr. Biol. 2003, 13, 1775–1785. [Google Scholar] [CrossRef]
- Tadesse, S.; Caldon, E.C.; Tilley, W.; Wang, S. Cyclin-Dependent Kinase 2 Inhibitors in Cancer Therapy: An Update. J. Med. Chem. 2019, 62, 4233–4251. [Google Scholar] [CrossRef]
- Kaldis, P.; Solomon, M.J. Analysis of CAK activities from human cells. Eur. J. Biochem. 2000, 267, 4213–4221. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, C.; Galaktionov, K. p42, a novel cyclin-dependent kinase-activating kinase in mammalian cells. J. Biol. Chem. 2004, 279, 4507–4514. [Google Scholar] [CrossRef] [PubMed]
- Kolupaeva, V.; Basilico, C. Overexpression of cyclin E/CDK2 complexes overcomes FGF-induced cell cycle arrest in the presence of hypophosphorylated Rb proteins. Cell Cycle 2012, 11, 2557–2566. [Google Scholar] [CrossRef] [PubMed]
- Abbas, T.; Dutta, A. p21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Saha, P.; Kornbluth, S.; Dynlacht, B.D.; Dutta, A. Cyclin-binding motifs are essential for the function of p21CIP1. Mol. Cell Biol. 1996, 16, 4673–4682. [Google Scholar] [CrossRef] [PubMed]
- Mansilla, S.F.; de la Vega, M.B.; Calzetta, N.L.; Siri, S.O.; Gottifredi, V. CDK-Independent and PCNA-Dependent Functions of p21 in DNA Replication. Genes 2020, 11, 593. [Google Scholar] [CrossRef] [PubMed]
- Al Bitar, S.; Gali-Muhtasib, H. The Role of the Cyclin Dependent Kinase Inhibitor p21cip1/waf1 in Targeting Cancer: Molecular Mechanisms and Novel Therapeutics. Cancers 2019, 11, 1475. [Google Scholar] [CrossRef]
- Otto, T.; Sicinski, P. Cell cycle proteins as promising targets in cancer therapy. Nat. Rev. Cancer 2017, 17, 93–115. [Google Scholar] [CrossRef]
- Shamloo, B.; Usluer, S. p21 in Cancer Research. Cancers 2019, 11, 1178. [Google Scholar] [CrossRef]
- Romanov, V.S.; Rudolph, K.L. p21 shapes cancer evolution. Nat. Cell Biol. 2016, 18, 722–724. [Google Scholar] [CrossRef] [PubMed]
- Snouffer, A.; Brown, D.; Lee, H.; Walsh, J.; Lupu, F.; Norman, R.; Lechtreck, K.; Ko, H.W.; Eggenschwiler, J. Cell Cycle-Related Kinase (CCRK) regulates ciliogenesis and Hedgehog signaling in mice. PLoS Genet 2017, 13, e1006912. [Google Scholar] [CrossRef] [PubMed]
- Maurya, A.K.; Rogers, T.; Sengupta, P. A CCRK and a MAK Kinase Modulate Cilia Branching and Length via Regulation of Axonemal Microtubule Dynamics in Caenorhabditis elegans. Curr. Biol. 2019, 29, 1286–1300 e1284. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Roine, N.; Makela, T.P. CCRK depletion inhibits glioblastoma cell proliferation in a cilium-dependent manner. EMBO Rep. 2013, 14, 741–747. [Google Scholar] [CrossRef] [PubMed]
- Gradilone, S.A.; Pisarello, M.J.L.; LaRusso, N.F. Primary Cilia in Tumor Biology: The Primary Cilium as a Therapeutic Target in Cholangiocarcinoma. Curr. Drug Targets 2017, 18, 958–963. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.S.; Cheung, Y.T.; An, X.M.; Chen, Y.C.; Li, M.; Li, G.H.; Cheung, W.; Sze, J.; Lai, L.; Peng, Y.; et al. Cell cycle-related kinase: A novel candidate oncogene in human glioblastoma. J. Natl. Cancer Inst. 2007, 99, 936–948. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xiong, Y.; Hannon, G.J.; Zhang, H.; Casso, D.; Kobayashi, R.; Beach, D. p21 is a universal inhibitor of cyclin kinases. Nature 1993, 366, 701–704. [Google Scholar] [CrossRef]
- LaBaer, J.; Garrett, M.D.; Stevenson, L.F.; Slingerland, J.M.; Sandhu, C.; Chou, H.S.; Fattaey, A.; Harlow, E. New functional activities for the p21 family of CDK inhibitors. Genes Dev. 1997, 11, 847–862. [Google Scholar] [CrossRef]
- Cheng, M.; Olivier, P.; Diehl, J.A.; Fero, M.; Roussel, M.F.; Roberts, J.M.; Sherr, C.J. The p21Cip1 and p27Kip1 CDK ‘inhibitors’ are essential activators of cyclin D-dependent kinases in murine fibroblasts. EMBO J. 1999, 18, 1571–1583. [Google Scholar] [CrossRef]
- Insinga, A.; Cicalese, A.; Faretta, M.; Gallo, B.; Albano, L.; Ronzoni, S.; Furia, L.; Viale, A.; Pelicci, P.G. DNA damage in stem cells activates p21, inhibits p53, and induces symmetric self-renewing divisions. Proc. Natl. Acad. Sci. USA 2013, 110, 3931–3936. [Google Scholar] [CrossRef]
- Coqueret, O. New roles for p21 and p27 cell-cycle inhibitors: A function for each cell compartment? Trends Cell Biol. 2003, 13, 65–70. [Google Scholar] [CrossRef]
- Kreis, N.N.; Louwen, F.; Yuan, J. Less understood issues: p21Cip1 in mitosis and its therapeutic potential. Oncogene 2015, 34, 1758–1767. [Google Scholar] [CrossRef] [PubMed]
- Monkawa, T.; Hiromura, K.; Wolf, G.; Shankland, S.J. The hypertrophic effect of transforming growth factor-beta is reduced in the absence of cyclin-dependent kinase-inhibitors p21 and p27. J. Am. Soc. Nephrol. 2002, 13, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.P.; Olson, E.N. Suppression of proliferation and cardiomyocyte hypertrophy by CHAMP, a cardiac-specific RNA helicase. Proc. Natl. Acad. Sci. USA 2002, 99, 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Zhang, P.; Harper, J.W.; Elledge, S.J.; Leder, P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 1995, 82, 675–684. [Google Scholar] [CrossRef]
- Campbell, G.J.; Hands, E.L.; Van de Pette, M. The Role of CDKs and CDKIs in Murine Development. Int. J. Mol. Sci. 2020, 21, 5343. [Google Scholar] [CrossRef]
- Qiu, H.; Dai, H.; Jain, K.; Shah, R.; Hong, C.; Pain, J.; Tian, B.; Vatner, D.E.; Vatner, S.F.; Depre, C. Characterization of a novel cardiac isoform of the cell cycle-related kinase that is regulated during heart failure. J. Biol. Chem. 2008, 283, 22157–22165. [Google Scholar] [CrossRef]
- Qiu, H.; Lai, H. Abstract 27: A Novel Variant Cell Cycle-related Kinase Provides Cardioprotection upon Pressure Overload by Promoting Autophagy. Circ. Res. 2014, 115, A27. [Google Scholar]
- CDK20 Cyclin Dependent Kinase 20 [Homo sapiens (Human)]. Available online: https://www.ncbi.nlm.nih.gov/gene/23552 (accessed on 15 July 2020).
- Child, E.S.; Mann, D.J. The intricacies of p21 phosphorylation: Protein/protein interactions, subcellular localization and stability. Cell Cycle 2006, 5, 1313–1319. [Google Scholar] [CrossRef]
- Janicke, R.U.; Sohn, D.; Essmann, F.; Schulze-Osthoff, K. The multiple battles fought by anti-apoptotic p21. Cell Cycle 2007, 6, 407–413. [Google Scholar] [CrossRef]
- Javelaud, D.; Besancon, F. Inactivation of p21WAF1 sensitizes cells to apoptosis via an increase of both p14ARF and p53 levels and an alteration of the Bax/Bcl-2 ratio. J. Biol. Chem. 2002, 277, 37949–37954. [Google Scholar] [CrossRef] [PubMed]
- Almond, J.B.; Cohen, G.M. The proteasome: A novel target for cancer chemotherapy. Leukemia 2002, 16, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, R.; Meuth, M. Chk1 and p21 cooperate to prevent apoptosis during DNA replication fork stress. Mol. Biol. Cell 2006, 17, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.H.; Jovanovic, A.; Serfas, M.S.; Tyner, A.L. The Cdk inhibitor p21 is required for necrosis, but it inhibits apoptosis following toxin-induced liver injury. J. Biol. Chem. 2003, 278, 30348–30355. [Google Scholar] [CrossRef]
- Ussat, S.; Werner, U.E.; Kruse, M.L.; Luschen, S.; Scherer, G.; Kabelitz, D.; Adam-Klages, S. Upregulation of p21WAF1/Cip1 precedes tumor necrosis factor-induced necrosis-like cell death. Biochem. Biophys. Res. Commun. 2002, 294, 672–679. [Google Scholar] [CrossRef]
- Capparelli, C.; Chiavarina, B.; Whitaker-Menezes, D.; Pestell, T.G.; Pestell, R.G.; Hulit, J.; Ando, S.; Howell, A.; Martinez-Outschoorn, U.E.; Sotgia, F.; et al. CDK inhibitors (p16/p19/p21) induce senescence and autophagy in cancer-associated fibroblasts, “fueling” tumor growth via paracrine interactions, without an increase in neo-angiogenesis. Cell Cycle 2012, 11, 3599–3610. [Google Scholar] [CrossRef]
- Fujiwara, K.; Daido, S.; Yamamoto, A.; Kobayashi, R.; Yokoyama, T.; Aoki, H.; Iwado, E.; Shinojima, N.; Kondo, Y.; Kondo, S. Pivotal role of the cyclin-dependent kinase inhibitor p21WAF1/CIP1 in apoptosis and autophagy. J. Biol. Chem. 2008, 283, 388–397. [Google Scholar] [CrossRef]
- Wu, G.Q.; Xie, D.; Yang, G.F.; Liao, Y.J.; Mai, S.J.; Deng, H.X.; Sze, J.; Guan, X.Y.; Zeng, Y.X.; Lin, M.C.; et al. Cell cycle-related kinase supports ovarian carcinoma cell proliferation via regulation of cyclin D1 and is a predictor of outcome in patients with ovarian carcinoma. Int. J. Cancer 2009, 125, 2631–2642. [Google Scholar] [CrossRef]
- Wohlbold, L.; Larochelle, S.; Liao, J.C.; Livshits, G.; Singer, J.; Shokat, K.M.; Fisher, R.P. The cyclin-dependent kinase (CDK) family member PNQALRE/CCRK supports cell proliferation but has no intrinsic CDK-activating kinase (CAK) activity. Cell Cycle 2006, 5, 546–554. [Google Scholar] [CrossRef]
- Ma, A.H.; Xia, L.; Desai, S.J.; Boucher, D.L.; Guan, Y.; Shih, H.M.; Shi, X.B.; deVere White, R.W.; Chen, H.W.; Tepper, C.G.; et al. Male germ cell-associated kinase, a male-specific kinase regulated by androgen, is a coactivator of androgen receptor in prostate cancer cells. Cancer Res. 2006, 66, 8439–8447. [Google Scholar] [CrossRef]
- Fu, Z.; Larson, K.A.; Chitta, R.K.; Parker, S.A.; Turk, B.E.; Lawrence, M.W.; Kaldis, P.; Galaktionov, K.; Cohn, S.M.; Shabanowitz, J.; et al. Identification of yin-yang regulators and a phosphorylation consensus for male germ cell-associated kinase (MAK)-related kinase. Mol. Cell Biol. 2006, 26, 8639–8654. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Cheng, A.S.; Tsang, D.P.; Li, M.S.; Go, M.Y.; Cheung, Y.S.; Zhao, G.J.; Ng, S.S.; Lin, M.C.; Yu, J.; et al. Cell cycle-related kinase is a direct androgen receptor-regulated gene that drives beta-catenin/T cell factor-dependent hepatocarcinogenesis. J. Clin. Investig. 2011, 121, 3159–3175. [Google Scholar] [CrossRef] [PubMed]
- Awuah, P.K.; Monga, S.P. Cell cycle-related kinase links androgen receptor and beta-catenin signaling in hepatocellular carcinoma: Why are men at a loss? Hepatology 2012, 55, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Schmider, A.; Gee, C.; Friedmann, W.; Lukas, J.J.; Press, M.F.; Lichtenegger, W.; Reles, A. p21WAF1/CIP1 protein expression is associated with prolonged survival but not with p53 expression in epithelial ovarian carcinoma. Gynecol. Oncol. 2000, 77, 237–242. [Google Scholar] [CrossRef]
- Besson, A.; Yong, V.W. Involvement of p21Waf1/Cip1 in protein kinase C alpha-induced cell cycle progression. Mol. Cell Biol. 2000, 20, 4580–4590. [Google Scholar] [CrossRef]
- van de Putte, G.; Holm, R.; Lie, A.K.; Trope, C.G.; Kristensen, G.B. Expression of p27, p21, and p16 protein in early squamous cervical cancer and its relation to prognosis. Gynecol. Oncol. 2003, 89, 140–147. [Google Scholar] [CrossRef]
- Fujii, R.; Osaka, E.; Sato, K.; Tokuhashi, Y. MiR-1 Suppresses Proliferation of Osteosarcoma Cells by Up-regulating p21 via PAX3. Cancer Genom. Proteom. 2019, 16, 71–79. [Google Scholar] [CrossRef]
- Pasz-Walczak, G.; Kordek, R.; Faflik, M. P21WAF1 expression in colorectal cancer: Correlation with P53 and cyclin D1 expression, clinicopathological parameters and prognosis. Pathol. Res. Pract. 2001, 197, 683–689. [Google Scholar] [CrossRef]
- Wang, Q.; Ma, J.; Lu, Y.; Zhang, S.; Huang, J.; Chen, J.; Bei, J.X.; Yang, K.; Wu, G.; Huang, K.; et al. CDK20 interacts with KEAP1 to activate NRF2 and promotes radiochemoresistance in lung cancer cells. Oncogene 2017, 36, 5321–5330. [Google Scholar] [CrossRef]
- Bae, K.M.; Wang, H.; Jiang, G.; Chen, M.G.; Lu, L.; Xiao, L. Protein kinase C epsilon is overexpressed in primary human non-small cell lung cancers and functionally required for proliferation of non-small cell lung cancer cells in a p21/Cip1-dependent manner. Cancer Res. 2007, 67, 6053–6063. [Google Scholar] [CrossRef]
- van Oijen, M.G.; Tilanus, M.G.; Medema, R.H.; Slootweg, P.J. Expression of p21Waf1/Cip1 in head and neck cancer in relation to proliferation, differentiation, p53 status and cyclin D1 expression. J. Oral Pathol. Med. 1998, 27, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.Y.; Tan, Q.X.; Zhu, X.; Qin, Q.H.; Zhu, F.B.; Mo, Q.G.; Yang, W.P. Expression of CDKN1A/p21 and TGFBR2 in breast cancer and their prognostic significance. Int. J. Clin. Exp. Pathol. 2015, 8, 14619–14629. [Google Scholar] [PubMed]
- Feng, H.; Yu, Z.; Tian, Y.; Lee, Y.Y.; Li, M.S.; Go, M.Y.; Cheung, Y.S.; Lai, P.B.; Chan, A.M.; To, K.F.; et al. A CCRK-EZH2 epigenetic circuitry drives hepatocarcinogenesis and associates with tumor recurrence and poor survival of patients. J. Hepatol. 2015, 62, 1100–1111. [Google Scholar] [CrossRef] [PubMed]
- Ehedego, H.; Boekschoten, M.V.; Hu, W.; Doler, C.; Haybaeck, J.; Gabetaler, N.; Muller, M.; Liedtke, C.; Trautwein, C. p21 ablation in liver enhances DNA damage, cholestasis, and carcinogenesis. Cancer Res. 2015, 75, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Mikhail, S.; Albanese, C.; Pishvaian, M.J. Cyclin-dependent kinase inhibitors and the treatment of gastrointestinal cancers. Am. J. Pathol. 2015, 185, 1185–1197. [Google Scholar] [CrossRef] [PubMed]
- Perri, F.; Pisconti, S.; Della Vittoria Scarpati, G. P53 mutations and cancer: A tight linkage. Ann. Transl. Med. 2016, 4, 522. [Google Scholar] [CrossRef] [PubMed]
- Martin-Caballero, J.; Flores, J.M.; Garcia-Palencia, P.; Serrano, M. Tumor susceptibility of p21Waf1/Cip1-deficient mice. Cancer Res. 2001, 61, 6234–6238. [Google Scholar] [PubMed]
- Mirzayans, R.; Andrais, B.; Scott, A.; Murray, D. New insights into p53 signaling and cancer cell response to DNA damage: Implications for cancer therapy. J. Biomed. Biotechnol. 2012, 2012, 170325. [Google Scholar] [CrossRef]
- Pang, L.Y.; Scott, M.; Hayward, R.L.; Mohammed, H.; Whitelaw, C.B.; Smith, G.C.; Hupp, T.R. p21WAF1 is component of a positive feedback loop that maintains the p53 transcriptional program. Cell Cycle 2011, 10, 932–950. [Google Scholar] [CrossRef]
- Van Jaarsveld, M.T.M.; Deng, D.; Wiemer, E.A.C.; Zi, Z. Tissue-Specific Chk1 Activation Determines Apoptosis by Regulating the Balance of p53 and p21. iScience 2019, 12, 27–40. [Google Scholar] [CrossRef]
- Di Stefano, V.; Giacca, M.; Capogrossi, M.C.; Crescenzi, M.; Martelli, F. Knockdown of cyclin-dependent kinase inhibitors induces cardiomyocyte re-entry in the cell cycle. J. Biol. Chem. 2011, 286, 8644–8654. [Google Scholar] [CrossRef] [PubMed]
- Kaija, H.M.; Sarkioja, T.; Kortelainen, M.L.; Vuoristo, J.T.; Huikuri, H.V.; Porvari, K.S. Stress-specific responses of p21 expression: Implication of transcript variant p21 alt-a in long-term hypoxia. J. Cell Biochem. 2012, 113, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Burton, P.B.; Yacoub, M.H.; Barton, P.J. Cyclin-dependent kinase inhibitor expression in human heart failure. A comparison with fetal development. Eur. Heart J. 1999, 20, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, I.; Minamino, T. Cellular senescence in cardiac diseases. J. Cardiol. 2019, 74, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lim, M.J.; Kim, M.H.; Yu, C.H.; Yun, Y.S.; Ahn, J.; Song, J.Y. An effective strategy for increasing the radiosensitivity of Human lung Cancer cells by blocking Nrf2-dependent antioxidant responses. Free Radic. Biol. Med. 2012, 53, 807–816. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wettersten, H.I.; Park, S.H.; Weiss, R.H. Small-molecule inhibitors of p21 as novel therapeutics for chemotherapy-resistant kidney cancer. Future Med. Chem. 2013, 5, 991–994. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, L.; Wu, F.; Qiu, L.; Luo, M.; Ke, Q.; Deng, X.; Luo, Z. Clinical and Prognostic Implications of P21WAF1/CIP1 Expression in Patients with Esophageal Cancer: A Systematic Review and Meta-Analysis. Dis. Mark. 2020, 2020, 6520259. [Google Scholar] [CrossRef]
- An, X.; Ng, S.S.; Xie, D.; Zeng, Y.X.; Sze, J.; Wang, J.; Chen, Y.C.; Chow, B.K.; Lu, G.; Poon, W.S.; et al. Functional characterisation of cell cycle-related kinase (CCRK) in colorectal cancer carcinogenesis. Eur. J. Cancer 2010, 46, 1752–1761. [Google Scholar] [CrossRef]


| Cancer Type | CDK20 | p21Cip1/Waf1 |
|---|---|---|
| Ovarian | cell proliferation [59] | cell survival [65] |
| Glioblastoma | cell proliferation [36] | cell proliferation [66] |
| Cervical carcinoma | cell proliferation [23] | High expression function unclear [67] |
| Osteosarcoma | cell proliferation [60] | Tumor suppressor [68] |
| Colorectal carcinoma | cell proliferation [60] | Tumor suppressor [69] |
| Lung cancer | Oncogene [70] | Tumor suppressor [71] |
| Head and neck cancer | N/A | Tumor suppressor [72] |
| Breast cancer | Oncogene [16] | Cell proliferation [73] |
| Liver cancer | Oncogene [74] | Tumor suppressor [75] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, L.; Shin, G.Y.; Qiu, H. The Role of Cell Cycle Regulators in Cell Survival—Dual Functions of Cyclin-Dependent Kinase 20 and p21Cip1/Waf1. Int. J. Mol. Sci. 2020, 21, 8504. https://doi.org/10.3390/ijms21228504
Lai L, Shin GY, Qiu H. The Role of Cell Cycle Regulators in Cell Survival—Dual Functions of Cyclin-Dependent Kinase 20 and p21Cip1/Waf1. International Journal of Molecular Sciences. 2020; 21(22):8504. https://doi.org/10.3390/ijms21228504
Chicago/Turabian StyleLai, Lo, Ga Yoon Shin, and Hongyu Qiu. 2020. "The Role of Cell Cycle Regulators in Cell Survival—Dual Functions of Cyclin-Dependent Kinase 20 and p21Cip1/Waf1" International Journal of Molecular Sciences 21, no. 22: 8504. https://doi.org/10.3390/ijms21228504
APA StyleLai, L., Shin, G. Y., & Qiu, H. (2020). The Role of Cell Cycle Regulators in Cell Survival—Dual Functions of Cyclin-Dependent Kinase 20 and p21Cip1/Waf1. International Journal of Molecular Sciences, 21(22), 8504. https://doi.org/10.3390/ijms21228504






