Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume
Abstract
:1. Introduction
2. Results
2.1. 3D Cell and Nucleus Volume
2.2. 2D Cell Shape and Area
2.3. 3D Cell Morphology
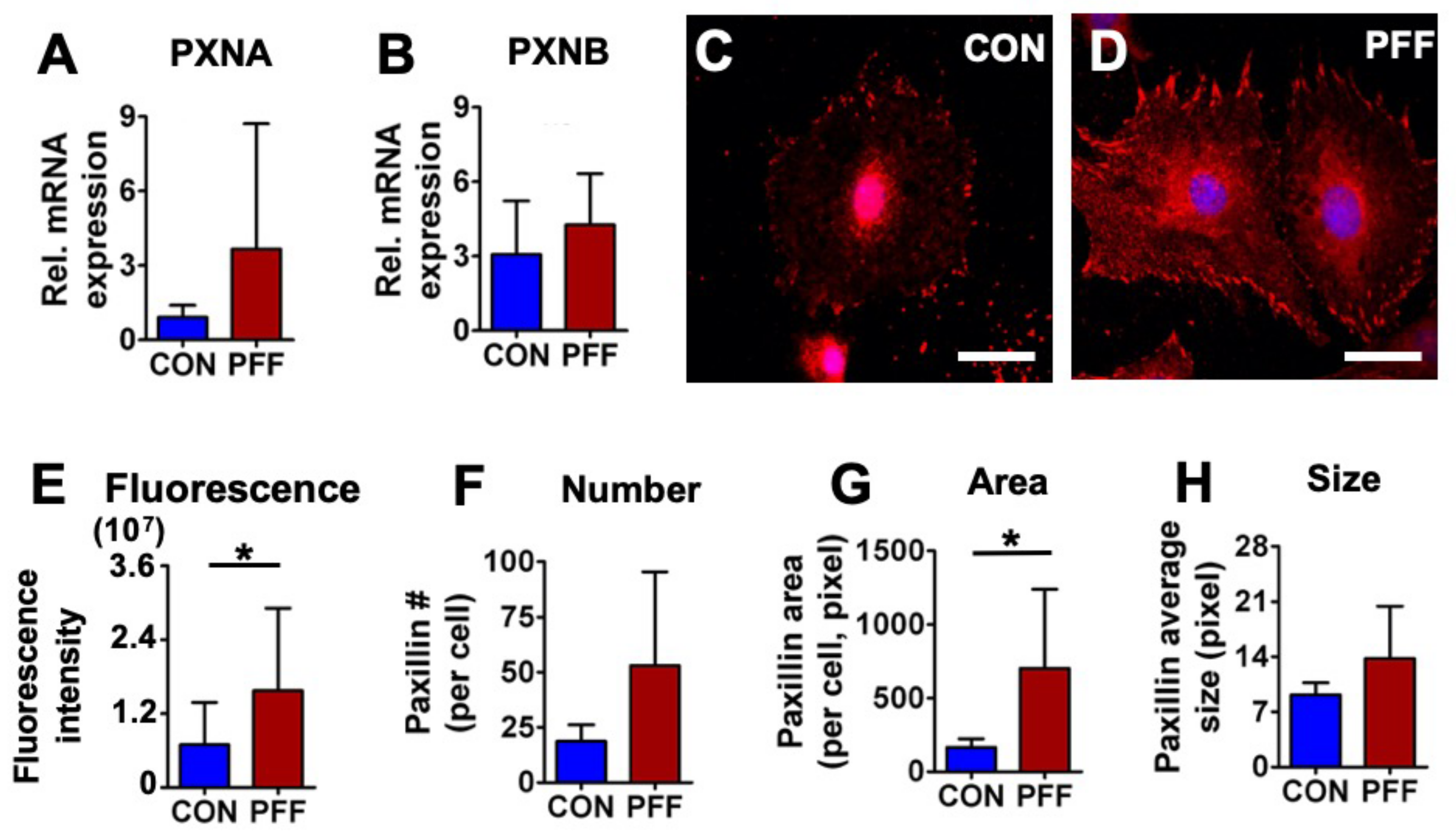
2.4. Expression of Paxillin
2.5. Expression of Integrin-α5
2.6. Expression of α-Tubulin and Nesprin 4
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Treatment with Pulsating Fluid Flow
4.3. Cell Morphology
4.4. Cell and Nucleus Volume
4.5. RNA Isolation and Quantitative Real Time Polymerase Chain Reaction (RT-PCR)
4.6. Protein Isolation and Western Blot
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALP | Alkaline phosphatase |
| APC | Anti-polyclonal |
| α-MEM | α-Minimal essential medium |
| BCA | Bicinchoninic acid |
| BSA | Bovine serum albumin |
| cDNA | Complementary DNA |
| CON | Control |
| CSC | China Scholarship Council |
| CTCF | Corrected total cell fluorescence |
| DAPI | 4′,6-Diamino−2-phenylindole |
| 2D | Two dimensional |
| 3D | Three dimensional |
| ECM | Extracellular matrix |
| EDTA | Ethylemediaminetetraacetic acid |
| FAK | Focal adhesion kinase |
| FBS | Fetal bovine serum |
| HRP | Horseradish peroxidase |
| IBM | International Business Machines Corporation |
| IGF-I | Insulin-like growth factor−1 |
| IgG | Immunoglobulin G |
| IPA | Isopropyl alcohol |
| ITGA51 | Integrin-α5 |
| KASH | Klarsicht, ANC-1, Syne homology |
| LDH | Lactate dehydrogenase |
| LINC | Linker of nucleoskeleton and cytoskeleton |
| LSCM | Laser scanning confocal microscope |
| MC3T3-E1 | Mouse calvaria 3T3-E1 |
| MITK | Medical imaging interaction toolkit |
| MSCs | Mesenchymal stem cells |
| NO | Nitric oxide |
| p-actin | Pan-actin |
| PBGD | Porphobilinogen deaminase |
| PFF | Pulsating fluid flow |
| PGE2 | Prostaglandin E2 |
| p-Paxillin | Phospho-paxillin |
| PXNA | Paxillin-a |
| PXNB | Paxillin-b |
| RhoA | Ras homolog family member A |
| RT-PCR | Real time polymerase chain reaction |
| SD | Standard deviation |
| SDS-PAGE | Sodium dodecyl sulfate-polyacrylamide gel electrophoresis |
| sHSPs | Small heat shock proteins |
| SiR | Spirochrome |
| SPSS | Statistical Product and Service Solutions |
| SUN | Sad1 and UNC-84 |
| TGF-β | Transforming growth factor-β |
| VCL | Vinculin |
Appendix A

Appendix A.1

Appendix A.2

Appendix A.3

References
- Shibamoto, A.; Ogawa, T.; Duyck, J.; Vandamme, K.; Naert, I.; Sasaki, K. Effect of high-frequency loading and parathyroid hormone administration on peri-implant bone healing and osseointegration. Int. J. Oral Sci. 2018, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Dutra, E.H.; Nanda, R.; Yadav, S. Bone response of loaded periodontal ligament. Curr. Osteoporos. Rep. 2016, 14, 280–283. [Google Scholar] [CrossRef]
- Tatsumi, S.; Ishii, K.; Amizuka, N.; Li, M.; Kobayashi, T.; Kohno, K.; Ito, M.; Takeshita, S.; Ikeda, K. Targeted ablation of osteocytes induces osteoporosis with defective mechanotransduction. Cell Metab. 2007, 5, 464–475. [Google Scholar] [CrossRef] [Green Version]
- Matsumoto, T.; Iimura, T.; Ogura, K.; Moriyama, K.; Yamaguchi, A. The role of osteocytes in bone resorption during orthodontic tooth movement. J. Dent. Res. 2013, 92, 340–345. [Google Scholar] [CrossRef]
- Inoue, M.; Ono, T.; Kameo, Y.; Sasaki, F.; Ono, T.; Adachi, T.; Nakashima, T. Forceful mastication activates osteocytes and builds a stout jawbone. Sci. Rep. 2019, 9, 4404. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Van der Plas, A.; Semeins, C.M.; Ajubi, N.E.; Frangos, J.A.; Nijweide, P.J.; Burger, E.H. Sensitivity of osteocytes to biomechanical stress in vitro. FASEB J. 1995, 9, 441–445. [Google Scholar] [CrossRef]
- Knothe Tate, M.L.; Steck, R.; Forwood, M.R.; Niederer, P. In vivo demonstration of load-induced fluid flow in the rat tibia and its potential implications for processes associated with functional adaptation. J. Exp. Biol. 2000, 203, 2737–2745. [Google Scholar]
- Ishijima, M.; Rittling, S.R.; Yamashita, T.; Tsuji, K.; Kurosawa, H.; Nifuji, A.; Denhardt, D.T.; Noda, M. Enhancement of osteoclastic bone resorption and suppression of osteoblastic bone formation in response to reduced mechanical stress do not occur in the absence of osteopontin. J. Exp. Med. 2002, 193, 399–404. [Google Scholar] [CrossRef] [Green Version]
- Fahlgren, A.; Bratengeier, C.; Semeins, C.M.; Klein-Nulend, J.; Bakker, A.D. Supraphysiological loading induces osteocyte-mediated osteoclastogenesis in a novel in vitro model for bone implant loosening. J. Orthop. Res. 2018, 36, 1425–1434. [Google Scholar] [CrossRef] [Green Version]
- Duncan, R.L.; Turner, C.H. Mechanotransduction and the functional response of bone to mechanical strain. Calcif. Tissue Int. 1995, 57, 344–358. [Google Scholar] [CrossRef]
- Siller-Jackson, A.; Burra, S.; Gu, S.; Harris, S.; Bonewald, L.; Sprague, E.; Jiang, J. The role of alpha 5 integrin as a mechanosensor in the regulation of connexin 43 hemichannel release of prostaglandin in response to mechanical stress. J. Bone Miner. Res. 2006, 3, S72. [Google Scholar]
- Bonewald, L.F. Mechanosensation and transduction in osteocytes. BoneKEy-Osteovision 2006, 3, 7–15. [Google Scholar] [CrossRef] [Green Version]
- Hamidouche, Z.; Fromigue, O.; Ringe, J.; Haupl, T.; Vaudin, P.; Pages, J.-C.; Srouji, S.; Livne, E.; Marie, P.J. Priming integrin 5 promotes human mesenchymal stromal cell osteoblast differentiation and osteogenesis. Proc. Natl. Acad. Sci. USA 2009, 106, 18587–18591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pavalko, F.M.; Otey, C.A.; Simon, K.O.; Burridge, K. α-Actinin: A direct link between actin and integrins. Biochem. Soc. Trans. 1991, 19, 1065–1069. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Zhou, C.; Wang, Q.; Cai, L.; Du, W.; Li, X.; Zhou, X.; Xie, J. Extracellular matrix elasticity regulates osteocyte gap junction elongation: Involvement of paxillin in intracellular signal transduction. Cell. Physiol. Biochem. 2018, 51, 1013–1026. [Google Scholar] [CrossRef]
- Guilluy, C.; Burridge, K. Nuclear mechanotransduction: Forcing the nucleus to respond. Nucleus 2015, 6, 19–22. [Google Scholar] [CrossRef] [Green Version]
- Guilak, F. Compression-induced changes in the shape and volume of the chondrocyte nucleus. J. Biomech. 1995, 28, 1529–1541. [Google Scholar] [CrossRef]
- Jorgens, D.M.; Inman, J.L.; Wojcik, M.; Robertson, C.; Palsdottir, H.; Tsai, W.-T.; Huang, H.; Bruni-Cardoso, A.; López, C.S.; Bissell, M.J.; et al. Deep nuclear invaginations are linked to cytoskeletal filaments—integrated bioimaging of epithelial cells in 3D culture. J. Cell Sci. 2016, 130, 177–189. [Google Scholar] [CrossRef] [Green Version]
- Duan, L.; Chen, H.; Gao, J. The cytoskeleton of the system. Lab. Cell Biol. Tech. 2018, 1, 17–22. [Google Scholar]
- Davies, P.F.; Robotewskyj, A.; Griem, M.L. Quantitative studies of endothelial cell adhesion. Directional remodeling of focal adhesion sites in response to flow forces. J. Clin. Investig. 1994, 93, 2031–2038. [Google Scholar] [CrossRef]
- Sun, J.; Chen, J.; Mohagheghian, E.; Wang, N. Force-induced gene up-regulation does not follow the weak power law but depends on H3K9 demethylation. Sci. Adv. 2020, 6, eaay9095. [Google Scholar] [CrossRef] [Green Version]
- Juffer, P.; Jaspers, R.T.; Lips, P.; Bakker, A.D.; Klein-Nulend, J. Expression of muscle anabolic and metabolic factors in mechanically loaded MLO-Y4 osteocytes. Am. J. Physiol. Metab. 2012, 302, E389–E395. [Google Scholar] [CrossRef] [PubMed]
- Schaffler, M.B.; Cheung, W.Y.; Majeska, R.; Kennedy, O. Osteocytes: Master orchestrators of bone. Calcif. Tissue Int. 2014, 94, 5–24. [Google Scholar] [CrossRef] [Green Version]
- Bacabac, R.G.; Mizuno, D.; Schmidt, C.F.; MacKintosh, F.C.; Van Loon, J.J.W.A.; Klein-Nulend, J.; Smit, T.H. Round versus flat: Bone cell morphology, elasticity, and mechanosensing. J. Biomech. 2008, 41, 1590–1598. [Google Scholar] [CrossRef]
- Palumbo, C. A three-dimensional ultrastructural study of osteoid-osteocytes in the tibia of chick embryos. Cell Tissue Res. 1986, 246, 125–131. [Google Scholar] [CrossRef]
- Finan, J.D.; Guilak, F. The effects of osmotic stress on the structure and function of the cell nucleus. J. Cell. Biochem. 2009, 109, 460–467. [Google Scholar] [CrossRef] [Green Version]
- McGarry, J.G.; Klein-Nulend, J.; Prendergast, P.J. The effect of cytoskeletal disruption on pulsatile fluid flow-induced nitric oxide and prostaglandin E2 release in osteocytes and osteoblasts. Biochem. Biophys. Res. Commun. 2005, 330, 341–348. [Google Scholar] [CrossRef]
- Gardinier, J.D.; Majumdar, S.; Duncan, R.L.; Wang, L. Cyclic hydraulic pressure and fluid flow differentially modulate cytoskeleton re-organization in MC3T3 osteoblasts. Cell. Mol. Bioeng. 2009, 2, 133–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakker, A.D.; Soejima, K.; Klein-Nulend, J.; Burger, E.H. The production of nitric oxide and prostaglandin E2 by primary bone cells is shear stress dependent. J. Biomech. 2001, 34, 671–677. [Google Scholar] [CrossRef]
- Tan, S.D.; De Vries, T.J.; Kuijpers-Jagtman, A.M.; Semeins, C.M.; Everts, V.; Klein-Nulend, J. Osteocytes subjected to fluid flow inhibit osteoclast formation and bone resorption. Bone 2007, 41, 745–751. [Google Scholar] [CrossRef]
- Starr, D.A.; Fridolfsson, H.N. Interactions between nuclei and the cytoskeleton are mediated by SUN-KASH nuclear-envelope bridges. Annu. Rev. Cell Dev. Biol. 2010, 26, 421–444. [Google Scholar] [CrossRef] [Green Version]
- Chancellor, T.J.; Lee, J.; Thodeti, C.K.; Lele, T. Actomyosin tension exerted on the nucleus through nesprin−1 connections influences endothelial cell adhesion, migration, and cyclic strain-induced reorientation. Biophys. J. 2010, 99, 115–123. [Google Scholar] [CrossRef] [Green Version]
- Anno, T.; Sakamoto, N.; Sato, M. Role of nesprin−1 in nuclear deformation in endothelial cells under static and uniaxial stretching conditions. Biochem. Biophys. Res. Commun. 2012, 424, 94–99. [Google Scholar] [CrossRef]
- Morgan, J.T.; Pfeiffer, E.R.; Thirkill, T.L.; Kumar, P.; Peng, G.; Fridolfsson, H.N.; Douglas, G.C.; Starr, D.A.; Barakat, A.I. Nesprin−3 regulates endothelial cell morphology, perinuclear cytoskeletal architecture, and flow-induced polarization. Mol. Biol. Cell 2011, 22, 4324–4334. [Google Scholar] [CrossRef] [PubMed]
- Chambliss, A.B.; Khatau, S.B.; Erdenberger, N.; Robinson, D.K.; Hodzic, D.; Longmore, G.D.; Wirtz, D. The LINC-anchored actin cap connects the extracellular milieu to the nucleus for ultrafast mechanotransduction. Sci. Rep. 2013, 3, 1087. [Google Scholar] [CrossRef] [Green Version]
- Jain, N.; Iyer, K.V.; Kumar, A.; Shivashankar, G.V. Cell geometric constraints induce modular gene-expression patterns via redistribution of HDAC3 regulated by actomyosin contractility. Proc. Natl. Acad. Sci. USA 2013, 110, 11349–11354. [Google Scholar] [CrossRef] [Green Version]
- Le, H.Q.; Ghatak, S.; Yeung, C.Y.C.; Tellkamp, F.; Günschmann, C.; Dieterich, C.; Yeroslaviz, A.; Habermann, B.; Pombo, A.; Niessen, C.M.; et al. Mechanical regulation of transcription controls Polycomb-mediated gene silencing during lineage commitment. Nat. Cell Biol. 2016, 18, 864–875. [Google Scholar] [CrossRef]
- Kim, D.H.; Hah, J.; Wirtz, D. Mechanics of the cell nucleus. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2018; Volume 1092, pp. 41–55. [Google Scholar]
- Bacabac, R.G.; Smit, T.H.; Mullender, M.G.; Dijcks, S.J.; Van Loon, J.J.W.; Klein-Nulend, J. Nitric oxide production by bone cells is fluid shear stress rate dependent. Biochem. Biophys. Res. Commun. 2004, 315, 823–829. [Google Scholar] [CrossRef]
- Bacabac, R.G.; Smit, T.H.; Cowin, S.C.; Van Loon, J.J.W.A.; Nieuwstadt, F.T.M.; Heethaar, R.; Klein-Nulend, J. Dynamic shear stress in parallel-plate flow chambers. J. Biomech. 2005, 38, 159–167. [Google Scholar] [CrossRef]
- Pavalko, F.M.; Chen, N.X.; Turner, C.H.; Burr, D.B.; Atkinson, S.; Hsieh, Y.F.; Qiu, J.; Duncan, R.L. Fluid shear-induced mechanical signaling in MC3T3-E1 osteoblasts requires cytoskeleton-integrin interactions. Am. J. Physiol. Physiol. 1998, 275, C1591–C1601. [Google Scholar] [CrossRef]
- Ingber, D.E. Tensegrity: The architectural basis of cellular mechanotransduction. Annu. Rev. Physiol. 2002, 59, 575–599. [Google Scholar] [CrossRef] [Green Version]
- Collier, M.P.; Benesch, J.L.P. Small heat-shock proteins and their role in mechanical stress. Cell Stress Chaperones 2020, 25, 601–613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hang, K.; Ye, C.; Chen, E.; Zhang, W.; Xue, D.; Pan, Z. Role of the heat shock protein family in bone metabolism. Cell Stress Chaperones 2018, 23, 1153–1164. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Roelofsen, J.; Sterck, J.G.H.; Semeins, C.M.; Burger, E.H. Mechanical loading stimulates the release of transforming growth factor-beta activity by cultured mouse calvariae and periosteal cells. J. Cell. Physiol. 1995, 163, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Klein-Nulend, J.; Burger, E.H.; Semeins, C.M.; Raisz, L.G.; Pilbeam, C.C. Pulsating fluid flow stimulates prostaglandin release and inducible prostaglandin G/H synthase mRNA expression in primary mouse bone cells. J. Bone Miner. Res. 1997, 12, 45–51. [Google Scholar] [CrossRef]
- Wettstein, G.; Bellaye, P.S.; Micheau, O.; Bonniaud, P. Small heat shock proteins and the cytoskeleton: An essential interplay for cell integrity? Int. J. Biochem. Cell Biol. 2012, 44, 1680–1686. [Google Scholar] [CrossRef]
- Jin, J.; Bakker, A.D.; Wu, G.; Klein-Nulend, J.; Jaspers, R.T. Physicochemical niche conditions and mechanosensing by osteocytes and myocytes. Curr. Osteoporos. Rep. 2019, 17, 235–249. [Google Scholar] [CrossRef] [Green Version]
- Florencio-Silva, R.; Sasso, G.; Sasso-Cerri, E.; Simões, M.; Cerri, P. Biology of bone tissue: Structure, function, and factors that influence bone cells. BioMed Res. Int. 2015, 2015, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Dráberová, E.; Stegurová, L.; Sulimenko, V.; Hájková, Z.; Dráber, P.; Dráber, P. Quantification of α-tubulin isotypes by sandwich ELISA with signal amplification through biotinyl-tyramide or immuno-PCR. J. Immunol. Methods 2013, 395, 63–70. [Google Scholar] [CrossRef]
- Klein-Nulend, J.; Bacabac, R.G.; Bakker, A.D. Mechanical loading and how it affects bone cells: The role of the osteocyte cytoskeleton in maintaining our skeleton. Eur. Cells Mater. 2012, 24, 278–291. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, D.A.; Mullins, R.D. Cell mechanics and the cytoskeleton. Nature 2010, 463, 485–492. [Google Scholar] [CrossRef] [Green Version]
- Hagel, M.; George, E.L.; Kim, A.; Tamimi, R.; Opitz, S.L.; Turner, C.E.; Imamoto, A.; Thomas, S.M. The adaptor protein paxillin is essential for normal development in the mouse and is a critical transducer of fibronectin signaling. Mol. Cell. Biol. 2009, 22, 901–915. [Google Scholar] [CrossRef] [Green Version]
- Young, S.R.L.; Gerard-O’Riley, R.; Kim, J.B.; Pavalko, F.M. Focal adhesion kinase is important for fluid shear stress-induced mechanotransduction in osteoblasts. J. Bone Miner. Res. 2009, 24, 411–424. [Google Scholar] [CrossRef]
- Mullen, C.A.; Haugh, M.G.; Schaffler, M.B.; Majeska, R.J.; McNamara, L.M. Osteocyte differentiation is regulated by extracellular matrix stiffness and intercellular separation. J. Mech. Behav. Biomed. Mater. 2013, 28, 183–194. [Google Scholar] [CrossRef] [Green Version]
- Engler, A.J.; Sen, S.; Sweeney, H.L.; Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 2006, 126, 677–689. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.S.; Tan, J.; Tien, J. Mechanotransduction at Cell-Matrix and Cell-Cell Contacts. Annu. Rev. Biomed. Eng. 2004, 6, 275–302. [Google Scholar] [CrossRef] [Green Version]
- Vatsa, A.; Semeins, C.M.; Smit, T.H.; Klein-Nulend, J. Paxillin localisation in osteocytes—Is it determined by the direction of loading? Biochem. Biophys. Res. Commun. 2008, 377, 1019–1024. [Google Scholar] [CrossRef]
- Mai, Z.; Peng, Z.; Wu, S.; Zhang, J.; Chen, L.; Liang, H.; Bai, D.; Yan, G.; Ai, H. Single bout short duration fluid shear stress induces osteogenic differentiation of MC3T3-E1 cells via integrin β1 and BMP2 signaling cross-talk. PLoS ONE 2013, 8, e61600. [Google Scholar] [CrossRef]
- Thomas, C.H.; Collier, J.H.; Sfeir, C.S.; Healy, K.E. Engineering gene expression and protein synthesis by modulation of nuclear shape. Proc. Natl. Acad. Sci. USA 2002, 99, 1972–1977. [Google Scholar] [CrossRef] [Green Version]
- Gupta, S.; Marcel, N.; Sarin, A.; Shivashankar, G.V. Role of actin dependent nuclear deformation in regulating early gene expression. PLoS ONE 2012, 7, e53031–e53039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruault, M.; Dubarry, M.; Taddei, A. Re-positioning genes to the nuclear envelope in mammalian cells: Impact on transcription. Trends Genet. 2008, 24, 574–581. [Google Scholar] [CrossRef]
- Reddy, K.L.; Zullo, J.M.; Bertolino, E.; Singh, H. Transcriptional repression mediated by repositioning of genes to the nuclear lamina. Nature 2008, 452, 243–247. [Google Scholar] [CrossRef]
- Santos, A.; Bakker, A.D.; Zandieh-Doulabi, B.; Semeins, C.M.; Klein-Nulend, J. Pulsating fluid flow modulates gene expression of proteins involved in Wnt signaling pathways in osteocytes. J. Orthop. Res. 2009, 27, 1280–1287. [Google Scholar] [CrossRef]
- Prodanov, L.; Semeins, C.M.; Van Loon, J.J.W.A.; Te Riet, J.; Jansen, J.A.; Klein-Nulend, J.; Walboomers, X.F. Influence of nanostructural environment and fluid flow on osteoblast-like cell behavior: A model for cell-mechanics studies. Acta Biomater. 2013, 9, 6653–6662. [Google Scholar] [CrossRef] [PubMed]
- Bakker, A.D.; Gakes, T.; Hogervorst, J.M.A.; De Wit, G.M.J.; Klein-Nulend, J.; Jaspers, R.T. Mechanical stimulation and IGF-1 enhance mRNA translation rate in osteoblasts via activation of the AKT-mTOR pathway. J. Cell. Physiol. 2016, 231, 1283–1290. [Google Scholar] [CrossRef]
- Etienne-Manneville, S.; Hall, A. Rho GTPases in cell biology. Nature 2002, 420, 629–635. [Google Scholar] [CrossRef]
- Hall, A. Rho GTPases and the actin cytoskeleton. Science 1998, 279, 509–514. [Google Scholar] [CrossRef] [Green Version]
- Juffer, P.; Bakker, A.D.; Klein-Nulend, J.; Jaspers, R.T. Mechanical loading by fluid shear stress of myotube glycocalyx stimulates growth factor expression and nitric oxide production. Cell Biochem. Biophys. 2014, 69, 411–419. [Google Scholar] [CrossRef] [PubMed]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, J.; Jaspers, R.T.; Wu, G.; Korfage, J.A.M.; Klein-Nulend, J.; Bakker, A.D. Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume. Int. J. Mol. Sci. 2020, 21, 8361. https://doi.org/10.3390/ijms21218361
Jin J, Jaspers RT, Wu G, Korfage JAM, Klein-Nulend J, Bakker AD. Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume. International Journal of Molecular Sciences. 2020; 21(21):8361. https://doi.org/10.3390/ijms21218361
Chicago/Turabian StyleJin, Jianfeng, Richard T. Jaspers, Gang Wu, Joannes A.M. Korfage, Jenneke Klein-Nulend, and Astrid D. Bakker. 2020. "Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume" International Journal of Molecular Sciences 21, no. 21: 8361. https://doi.org/10.3390/ijms21218361
APA StyleJin, J., Jaspers, R. T., Wu, G., Korfage, J. A. M., Klein-Nulend, J., & Bakker, A. D. (2020). Shear Stress Modulates Osteoblast Cell and Nucleus Morphology and Volume. International Journal of Molecular Sciences, 21(21), 8361. https://doi.org/10.3390/ijms21218361





