Aging Induces Profound Changes in sncRNA in Rat Sperm and These Changes Are Modified by Perinatal Exposure to Environmental Flame Retardant
Abstract
1. Introduction
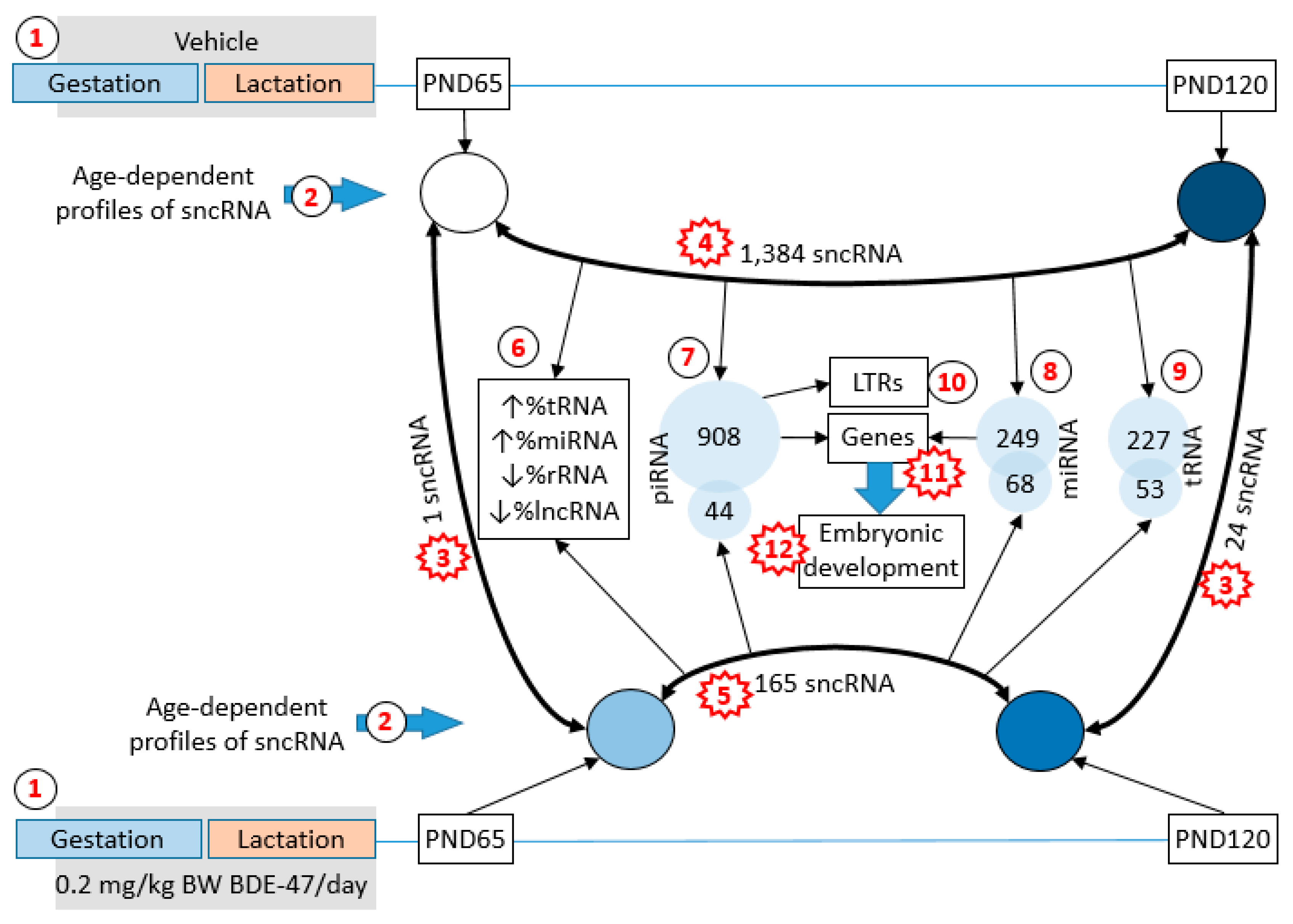
2. Results
2.1. Changes in the Profile of Small RNA Expression
2.2. Functional Analysis of Age-Dependent Changes in miRNA Expression
2.3. BDE-47 Exposure-Dependent Changes
2.4. Functional Analysis of Age-Dependent Changes in piRNA Expression
3. Discussion
3.1. sncRNA in Sperm
3.2. Justification of the Model
3.3. Age-Dependent Changes in sncRNA Fraction
3.4. BDE-47 Modifies Effect of Aging
3.5. Limitations and Future Directions
4. Materials and Methods
4.1. Animals and Treatment
4.2. Extraction of Sperm RNA
4.3. Preparation of sncRNA Libraries and Sequencing
4.4. Identification of Differentially Expressed Small RNA
4.5. Statistical Analysis
4.6. Functional Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Martin, J.A.; Hamilton, B.E.; Ventura, S.J.; Osterman, M.J.; Wilson, E.C.; Mathews, T.J. Births: Final data for 2010. Natl. Vital Stat. Rep. 2012, 61, 1–72. [Google Scholar] [PubMed]
- Khandwala, Y.S.; Zhang, C.A.; Lu, Y.; Eisenberg, M.L. The age of fathers in the USA is rising: An analysis of 168 867 480 births from 1972 to 2015. Hum. Reprod. 2017, 32, 2110–2116. [Google Scholar] [CrossRef] [PubMed]
- Nybo Andersen, A.M.; Hansen, K.D.; Andersen, P.K.; Davey Smith, G. Advanced paternal age and risk of fetal death: A cohort study. Am. J. Epidemiol. 2004, 160, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Alio, A.P.; Salihu, H.M.; McIntosh, C.; August, E.M.; Weldeselasse, H.; Sanchez, E.; Mbah, A.K. The effect of paternal age on fetal birth outcomes. Am. J. Mens. Health 2012, 6, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Urhoj, S.K.; Mortensen, L.H.; Nybo Andersen, A.M. Advanced Paternal Age and Risk of Musculoskeletal Congenital Anomalies in Offspring. Birth Defects Res. B Dev. Reprod. Toxicol. 2015, 104, 273–280. [Google Scholar] [CrossRef]
- Bille, C.; Skytthe, A.; Vach, W.; Knudsen, L.B.; Andersen, A.M.; Murray, J.C.; Christensen, K. Parent’s age and the risk of oral clefts. Epidemiology 2005, 16, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Sergentanis, T.N.; Thomopoulos, T.P.; Gialamas, S.P.; Karalexi, M.A.; Biniaris-Georgallis, S.I.; Kontogeorgi, E.; Papathoma, P.; Tsilimidos, G.; Skalkidou, A.; Iliadou, A.N.; et al. Risk for childhood leukemia associated with maternal and paternal age. Eur. J. Epidemiol. 2015, 30, 1229–1261. [Google Scholar] [CrossRef] [PubMed]
- Dockerty, J.D.; Draper, G.; Vincent, T.; Rowan, S.D.; Bunch, K.J. Case-control study of parental age, parity and socioeconomic level in relation to childhood cancers. Int. J. Epidemiol. 2001, 30, 1428–1437. [Google Scholar] [CrossRef][Green Version]
- Moll, A.C.; Imhof, S.M.; Kuik, D.J.; Bouter, L.M.; Den Otter, W.; Bezemer, P.D.; Koten, J.W.; Tan, K.E. High parental age is associated with sporadic hereditary retinoblastoma: The Dutch retinoblastoma register 1862–1994. Hum. Genet. 1996, 98, 109–112. [Google Scholar] [CrossRef]
- Heck, J.E.; Lombardi, C.A.; Meyers, T.J.; Cockburn, M.; Wilhelm, M.; Ritz, B. Perinatal characteristics and retinoblastoma. Cancer Causes Control 2012, 23, 1567–1575. [Google Scholar] [CrossRef]
- Nybo Andersen, A.M.; Urhoj, S.K. Is advanced paternal age a health risk for the offspring? Fertil. Steril. 2017, 107, 312–318. [Google Scholar] [CrossRef]
- Liebenberg, R.; van Heerden, B.; Ehlers, R.; Du Plessis, A.M.; Roos, J.L. Advancing paternal age at birth is associated with poorer social functioning earlier and later in life of schizophrenia patients in a founder population. Psychiatry Res. 2016, 243, 185–190. [Google Scholar] [CrossRef]
- Malaspina, D.; Harlap, S.; Fennig, S.; Heiman, D.; Nahon, D.; Feldman, D.; Susser, E.S. Advancing paternal age and the risk of schizophrenia. Arch. Gen. Psychiatry 2001, 58, 361–367. [Google Scholar] [CrossRef]
- Reichenberg, A.; Gross, R.; Weiser, M.; Bresnahan, M.; Silverman, J.; Harlap, S.; Rabinowitz, J.; Shulman, C.; Malaspina, D.; Lubin, G.; et al. Advancing paternal age and autism. Arch. Gen. Psychiatry 2006, 63, 1026–1032. [Google Scholar] [CrossRef]
- Hultman, C.M.; Sandin, S.; Levine, S.Z.; Lichtenstein, P.; Reichenberg, A. Advancing paternal age and risk of autism: New evidence from a population-based study and a meta-analysis of epidemiological studies. Mol. Psychiatry 2011, 16, 1203–1212. [Google Scholar] [CrossRef] [PubMed]
- Frans, E.M.; Sandin, S.; Reichenberg, A.; Lichtenstein, P.; Langstrom, N.; Hultman, C.M. Advancing paternal age and bipolar disorder. Arch. Gen. Psychiatry 2008, 65, 1034–1040. [Google Scholar] [CrossRef]
- D’Onofrio, B.M.; Rickert, M.E.; Frans, E.; Kuja-Halkola, R.; Almqvist, C.; Sjolander, A.; Larsson, H.; Lichtenstein, P. Paternal age at childbearing and offspring psychiatric and academic morbidity. JAMA Psychiatry 2014, 71, 432–438. [Google Scholar] [CrossRef]
- Wu, H.; Hauser, R.; Krawetz, S.A.; Pilsner, J.R. Environmental Susceptibility of the Sperm Epigenome During Windows of Male Germ Cell Development. Curr. Environ. Health Rep. 2015, 2, 356–366. [Google Scholar] [CrossRef]
- Marcho, C.; Oluwayiose, O.A.; Pilsner, J.R. The preconception environment and sperm epigenetics. Andrology 2020, 8, 924–942. [Google Scholar] [CrossRef]
- Sharma, R.; Agarwal, A.; Rohra, V.K.; Assidi, M.; Abu-Elmagd, M.; Turki, R.F. Effects of increased paternal age on sperm quality, reproductive outcome and associated epigenetic risks to offspring. Reprod. Biol. Endocrinol. 2015, 13, 35. [Google Scholar] [CrossRef]
- Perrin, M.C.; Brown, A.S.; Malaspina, D. Aberrant epigenetic regulation could explain the relationship of paternal age to schizophrenia. Schizophr. Bull. 2007, 33, 1270–1273. [Google Scholar] [CrossRef]
- Chen, Q.; Yan, W.; Duan, E. Epigenetic inheritance of acquired traits through sperm RNAs and sperm RNA modifications. Nat. Rev. Genet. 2016, 17, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Yan, W. Potential roles of noncoding RNAs in environmental epigenetic transgenerational inheritance. Mol. Cell. Endocrinol. 2014, 398, 24–30. [Google Scholar] [CrossRef]
- Sharma, U.; Conine, C.C.; Shea, J.M.; Boskovic, A.; Derr, A.G.; Bing, X.Y.; Belleannee, C.; Kucukural, A.; Serra, R.W.; Sun, F.; et al. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 2016, 351, 391–396. [Google Scholar] [CrossRef]
- McPherson, N.O.; Owens, J.A.; Fullston, T.; Lane, M. Preconception diet or exercise intervention in obese fathers normalizes sperm microRNA profile and metabolic syndrome in female offspring. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E805–E821. [Google Scholar] [CrossRef]
- Chen, Q.; Yan, M.; Cao, Z.; Li, X.; Zhang, Y.; Shi, J.; Feng, G.H.; Peng, H.; Zhang, X.; Zhang, Y.; et al. Sperm tsRNAs contribute to intergenerational inheritance of an acquired metabolic disorder. Science 2016, 351, 397–400. [Google Scholar] [CrossRef]
- Grandjean, V.; Fourre, S.; De Abreu, D.A.; Derieppe, M.A.; Remy, J.J.; Rassoulzadegan, M. RNA-mediated paternal heredity of diet-induced obesity and metabolic disorders. Sci. Rep. 2015, 5, 18193. [Google Scholar] [CrossRef]
- Fullston, T.; Ohlsson Teague, E.M.; Palmer, N.O.; DeBlasio, M.J.; Mitchell, M.; Corbett, M.; Print, C.G.; Owens, J.A.; Lane, M. Paternal obesity initiates metabolic disturbances in two generations of mice with incomplete penetrance to the F2 generation and alters the transcriptional profile of testis and sperm microRNA content. FASEB J. 2013, 27, 4226–4243. [Google Scholar] [CrossRef] [PubMed]
- de Castro Barbosa, T.; Ingerslev, L.R.; Alm, P.S.; Versteyhe, S.; Massart, J.; Rasmussen, M.; Donkin, I.; Sjogren, R.; Mudry, J.M.; Vetterli, L.; et al. High-fat diet reprograms the epigenome of rat spermatozoa and transgenerationally affects metabolism of the offspring. Mol. Metab. 2015, 5, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Donkin, I.; Versteyhe, S.; Ingerslev, L.R.; Qian, K.; Mechta, M.; Nordkap, L.; Mortensen, B.; Appel, E.V.; Jorgensen, N.; Kristiansen, V.B.; et al. Obesity and Bariatric Surgery Drive Epigenetic Variation of Spermatozoa in Humans. Cell. Metab. 2016, 23, 369–378. [Google Scholar] [CrossRef]
- Rompala, G.R.; Mounier, A.; Wolfe, C.M.; Lin, Q.; Lefterov, I.; Homanics, G.E. Heavy Chronic Intermittent Ethanol Exposure Alters Small Noncoding RNAs in Mouse Sperm and Epididymosomes. Front. Genet. 2018, 9, 32. [Google Scholar] [CrossRef]
- Vojtech, L.; Woo, S.; Hughes, S.; Levy, C.; Ballweber, L.; Sauteraud, R.P.; Strobl, J.; Westerberg, K.; Gottardo, R.; Tewari, M.; et al. Exosomes in human semen carry a distinctive repertoire of small non-coding RNAs with potential regulatory functions. Nucleic Acids Res. 2014, 42, 7290–7304. [Google Scholar] [CrossRef] [PubMed]
- Robb, G.W.; Amann, R.P.; Killian, G.J. Daily sperm production and epididymal sperm reserves of pubertal and adult rats. J. Reprod. Fertil. 1978, 54, 103–107. [Google Scholar] [CrossRef]
- Zanato, V.F.; Martins, M.P.; Anselmo-Franci, J.A.; Petenusci, S.O.; Lamano-Carvalho, T.L. Sexual development of male Wistar rats. Braz. J. Med. Biol. Res. 1994, 27, 1273–1280. [Google Scholar]
- Pilsner, J.; Shershebnev, A.; Wu, H.; Marcho, C.; Dribnokhodova, O.; Shtratnikova, V.; Sergeyev, O.; Suvorov, A. Aging-induced Changes in Sperm DNA Methylation are Modified by Low Dose of Perinatal Flame retardants. Preprints 2020. [Google Scholar] [CrossRef]
- Bourgon, R.; Gentleman, R.; Huber, W. Independent filtering increases detection power for high-throughput experiments. Proc. Natl. Acad. Sci. USA 2010, 107, 9546–9551. [Google Scholar] [CrossRef]
- Liu, W.; Wang, X. Prediction of functional microRNA targets by integrative modeling of microRNA binding and target expression data. Genome Biol. 2019, 20, 1–10. [Google Scholar] [CrossRef]
- Wong, N.; Wang, X. miRDB: An online resource for microRNA target prediction and functional annotations. Nucleic Acids Res. 2015, 43, D146–D152. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, P.; Lu, Y.; Li, Y.; Zheng, Y.; Kan, Y.; Chen, R.; He, S. piRBase: A comprehensive database of piRNA sequences. Nucleic Acids Res. 2019, 47, D175–D180. [Google Scholar] [CrossRef] [PubMed]
- Natt, D.; Ost, A. Male reproductive health and intergenerational metabolic responses from a small RNA perspective. J. Intern. Med. 2020, 288, 305–320. [Google Scholar] [CrossRef] [PubMed]
- Creasy, D.M. Evaluation of testicular toxicity in safety evaluation studies: The appropriate use of spermatogenic staging. Toxicol. Pathol. 1997, 25, 119–131. [Google Scholar] [CrossRef]
- Reik, W.; Dean, W.; Walter, J. Epigenetic reprogramming in mammalian development. Science 2001, 293, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Wang, P.J. Mammalian piRNAs: Biogenesis, function, and mysteries. Spermatogenesis 2014, 4, e27889. [Google Scholar] [CrossRef]
- Aravin, A.A.; Hannon, G.J.; Brennecke, J. The Piwi-piRNA pathway provides an adaptive defense in the transposon arms race. Science 2007, 318, 761–764. [Google Scholar] [CrossRef]
- Gou, L.T.; Dai, P.; Yang, J.H.; Xue, Y.; Hu, Y.P.; Zhou, Y.; Kang, J.Y.; Wang, X.; Li, H.; Hua, M.M.; et al. Pachytene piRNAs instruct massive mRNA elimination during late spermiogenesis. Cell Res. 2014, 24, 680–700. [Google Scholar] [CrossRef]
- Hua, M.; Liu, W.; Chen, Y.; Zhang, F.; Xu, B.; Liu, S.; Chen, G.; Shi, H.; Wu, L. Identification of small non-coding RNAs as sperm quality biomarkers for in vitro fertilization. Cell. Discov. 2019, 5, 1–5. [Google Scholar] [CrossRef]
- Trigg, N.A.; Eamens, A.L.; Nixon, B. The contribution of epididymosomes to the sperm small RNA profile. Reproduction 2019, 157, R209–R223. [Google Scholar] [CrossRef]
- Zijlstra, C.; Stoorvogel, W. Prostasomes as a source of diagnostic biomarkers for prostate cancer. J. Clin. Investig. 2016, 126, 1144–1151. [Google Scholar] [CrossRef]
- Sharma, U.; Sun, F.; Conine, C.C.; Reichholf, B.; Kukreja, S.; Herzog, V.A.; Ameres, S.L.; Rando, O.J. Small RNAs Are Trafficked from the Epididymis to Developing Mammalian Sperm. Dev. Cell 2018, 46, 481–494.e6. [Google Scholar] [CrossRef]
- Conine, C.C.; Sun, F.; Song, L.; Rivera-Perez, J.A.; Rando, O.J. Small RNAs Gained during Epididymal Transit of Sperm Are Essential for Embryonic Development in Mice. Dev. Cell 2018, 46, 470–480.e3. [Google Scholar] [CrossRef]
- Klastrup, L.K.; Bak, S.T.; Nielsen, A.L. The influence of paternal diet on sncRNA-mediated epigenetic inheritance. Mol. Genet. Genom. 2019, 294, 1–11. [Google Scholar] [CrossRef]
- Gabriel, S.M.; Roncancio, J.R.; Ruiz, N.S. Growth hormone pulsatility and the endocrine milieu during sexual maturation in male and female rats. Neuroendocrinology 1992, 56, 619–625. [Google Scholar] [CrossRef]
- Zemunik, T.; Peruzovic, M.; Capkun, V.; Zekan, L.J.; Milkovic, K. Pregnancy in adolescent rats, growth and neurodevelopment in their offspring. Arch. Physiol. Biochem. 2001, 109, 450–456. [Google Scholar] [CrossRef]
- Johnson-Restrepo, B.; Kannan, K.; Rapaport, D.P.; Rodan, B.D. Polybrominated diphenyl ethers and polychlorinated biphenyls in human adipose tissue from New York. Environ. Sci. Technol. 2005, 39, 5177–5182. [Google Scholar] [CrossRef]
- Suvorov, A.; Girard, S.; Lachapelle, S.; Abdelouahab, N.; Sebire, G.; Takser, L. Perinatal exposure to low-dose BDE-47, an emergent environmental contaminant, causes hyperactivity in rat offspring. Neonatology 2009, 95, 203–209. [Google Scholar] [CrossRef]
- Antignac, J.P.; Cariou, R.; Maume, D.; Marchand, P.; Monteau, F.; Zalko, D.; Berrebi, A.; Cravedi, J.P.; Andre, F.; Le Bizec, B. Exposure assessment of fetus and newborn to brominated flame retardants in France: Preliminary data. Mol. Nutr. Food Res. 2008, 52, 258–265. [Google Scholar] [CrossRef]
- Schecter, A.; Papke, O.; Harris, T.R.; Tung, K.C.; Musumba, A.; Olson, J.; Birnbaum, L. Polybrominated diphenyl ether (PBDE) levels in an expanded market basket survey of U.S. food and estimated PBDE dietary intake by age and sex. Environ. Health Perspect. 2006, 114, 1515–1520. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Jiao, Y.; Hu, Y.; Sun, Z.; Zhou, X.; Feng, J.; Li, J.; Wu, Y. Levels of tetrabromobisphenol A, hexabromocyclododecanes and polybrominated diphenyl ethers in human milk from the general population in Beijing, China. Sci. Total Environ. 2013, 452, 10–18. [Google Scholar] [CrossRef]
- Zhao, Y.; Ruan, X.; Li, Y.; Yan, M.; Qin, Z. Polybrominated Diphenyl Ethers (PBDEs) in Aborted Human Fetuses and Placental Transfer during the First Trimester of Pregnancy. Environ. Sci. Technol. 2013, 47, 5939–5946. [Google Scholar] [CrossRef] [PubMed]
- Herbstman, J.B.; Sjodin, A.; Apelberg, B.J.; Witter, F.R.; Patterson, D.G.; Halden, R.U.; Jones, R.S.; Park, A.; Zhang, Y.; Heidler, J.; et al. Determinants of prenatal exposure to polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs) in an urban population. Environ. Health Perspect. 2007, 115, 1794–1800. [Google Scholar] [CrossRef]
- Doucet, J.; Tague, B.; Arnold, D.L.; Cooke, G.M.; Hayward, S.; Goodyer, C.G. Persistent organic pollutant residues in human fetal liver and placenta from Greater Montreal, Quebec: A longitudinal study from 1998 through 2006. Environ. Health Perspect. 2009, 117, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Wilford, B.H.; Shoeib, M.; Harner, T.; Zhu, J.; Jones, K.C. Polybrominated diphenyl ethers in indoor dust in Ottawa, Canada: Implications for sources and exposure. Environ. Sci. Technol. 2005, 39, 7027–7035. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, S.A.; Kelsey, G. De novo DNA methylation: A germ cell perspective. Trends Genet. 2012, 28, 33–42. [Google Scholar] [CrossRef]
- Gkountela, S.; Li, Z.; Vincent, J.J.; Zhang, K.X.; Chen, A.; Pellegrini, M.; Clark, A.T. The ontogeny of cKIT+ human primordial germ cells proves to be a resource for human germ line reprogramming, imprint erasure and in vitro differentiation. Nat. Cell Biol. 2013, 15, 113–122. [Google Scholar] [CrossRef]
- Paoli, D.; Pecora, G.; Pallotti, F.; Faja, F.; Pelloni, M.; Lenzi, A.; Lombardo, F. Cytological and molecular aspects of the ageing sperm. Hum. Reprod. 2019, 34, 218–227. [Google Scholar] [CrossRef]
- Natt, D.; Kugelberg, U.; Casas, E.; Nedstrand, E.; Zalavary, S.; Henriksson, P.; Nijm, C.; Jaderquist, J.; Sandborg, J.; Flinke, E.; et al. Human sperm displays rapid responses to diet. PLoS Biol. 2019, 17, e3000559. [Google Scholar] [CrossRef]
- Garcia-Etxebarria, K.; Jugo, B.M. Genome-wide reexamination of endogenous retroviruses in Rattus norvegicus. Virology 2016, 494, 119–128. [Google Scholar] [CrossRef]
- Doolittle, W.F.; Sapienza, C. Selfish genes, the phenotype paradigm and genome evolution. Nature 1980, 284, 601–603. [Google Scholar] [CrossRef]
- Yoder, J.A.; Walsh, C.P.; Bestor, T.H. Cytosine methylation and the ecology of intragenomic parasites. Trends Genet. 1997, 13, 335–340. [Google Scholar] [CrossRef]
- Chuma, S.; Nakano, T. piRNA and spermatogenesis in mice. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2013, 368, 20110338. [Google Scholar] [CrossRef]
- Fu, B.; Ma, H.; Liu, D. Endogenous Retroviruses Function as Gene Expression Regulatory Elements During Mammalian Pre-implantation Embryo Development. Int. J. Mol. Sci. 2019, 20, 790. [Google Scholar] [CrossRef]
- Evsikov, A.V.; Marin de Evsikova, C. Friend or Foe: Epigenetic Regulation of Retrotransposons in Mammalian Oogenesis and Early Development. Yale J. Biol. Med. 2016, 89, 487–497. [Google Scholar] [PubMed]
- Burton, A.; Torres-Padilla, M.E. Chromatin dynamics in the regulation of cell fate allocation during early embryogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 723–734. [Google Scholar] [CrossRef]
- Schoorlemmer, J.; Perez-Palacios, R.; Climent, M.; Guallar, D.; Muniesa, P. Regulation of Mouse Retroelement MuERV-L/MERVL Expression by REX1 and Epigenetic Control of Stem Cell Potency. Front. Oncol. 2014, 4, 14. [Google Scholar] [CrossRef]
- Peaston, A.E.; Evsikov, A.V.; Graber, J.H.; de Vries, W.N.; Holbrook, A.E.; Solter, D.; Knowles, B.B. Retrotransposons regulate host genes in mouse oocytes and preimplantation embryos. Dev. Cell 2004, 7, 597–606. [Google Scholar] [CrossRef]
- Huang, Y.; Kim, J.K.; Do, D.V.; Lee, C.; Penfold, C.A.; Zylicz, J.J.; Marioni, J.C.; Hackett, J.A.; Surani, M.A. Stella modulates transcriptional and endogenous retrovirus programs during maternal-to-zygotic transition. Elife 2017, 6, e22345. [Google Scholar] [CrossRef]
- Hassan, M.A.; Killick, S.R. Effect of male age on fertility: Evidence for the decline in male fertility with increasing age. Fertil. Steril. 2003, 79, 1520–1527. [Google Scholar] [CrossRef]
- Frattarelli, J.L.; Miller, K.A.; Miller, B.T.; Elkind-Hirsch, K.; Scott, R.T., Jr. Male age negatively impacts embryo development and reproductive outcome in donor oocyte assisted reproductive technology cycles. Fertil. Steril. 2008, 90, 97–103. [Google Scholar] [CrossRef]
- Aboulghar, M.; Mansour, R.; Al-Inany, H.; Abou-Setta, A.M.; Aboulghar, M.; Mourad, L.; Serour, G. Paternal age and outcome of intracytoplasmic sperm injection. Reprod. Biomed. Online 2007, 14, 588–592. [Google Scholar] [CrossRef]
- Xie, Y.; Yao, L.; Yu, X.; Ruan, Y.; Li, Z.; Guo, J. Action mechanisms and research methods of tRNA-derived small RNAs. Signal. Transduct Target Ther. 2020, 5, 109. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Baker, M.A. The Role of Genetics and Oxidative Stress in the Etiology of Male Infertility-A Unifying Hypothesis? Front. Endocrinol. 2020, 11, 581838. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Welshons, W.V.; Vom Saal, F.S.; Toutain, P.L.; Myers, J.P. Should oral gavage be abandoned in toxicity testing of endocrine disruptors? Environ. Health 2014, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Suvorov, A.; Vandenberg, L.N. To Cull or not to Cull? Considerations for studies of endocrine disrupting chemicals. Endocrinology 2016, 157, 2586–2594. [Google Scholar] [CrossRef]
- Goodrich, R.J.; Anton, E.; Krawetz, S.A. Isolating mRNA and small noncoding RNAs from human sperm. Methods Mol. Biol. 2013, 927, 385–396. [Google Scholar]
- Andrews, S. FASTQC. A Quality Control Tool for High Throughput Sequence Data. Available online: https://www.bibsonomy.org/bibtex/f230a919c34360709aa298734d63dca3 (accessed on 2 April 2019).
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Rigden, D.J.; Fernandez-Suarez, X.M.; Galperin, M.Y. The 2016 database issue of Nucleic Acids Research and an updated molecular biology database collection. Nucleic Acids Res. 2016, 44, D1–D6. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.P.; Lowe, T.M. GtRNAdb 2.0: An expanded database of transfer RNA genes identified in complete and draft genomes. Nucleic Acids Res. 2016, 44, D184–D189. [Google Scholar] [CrossRef]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. 1000 Genome Project Data Processing Subgroup The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef]
- Quinlan, A.R.; Hall, I.M. BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 2010, 26, 841–842. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Anders, S.; Huber, W. Differential expression analysis for sequence count data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Tripathi, S.; Pohl, M.O.; Zhou, Y.; Rodriguez-Frandsen, A.; Wang, G.; Stein, D.A.; Moulton, H.M.; DeJesus, P.; Che, J.; Mulder, L.C.; et al. Meta- and Orthogonal Integration of Influenza “OMICs” Data Defines a Role for UBR4 in Virus Budding. Cell. Host Microbe 2015, 18, 723–735. [Google Scholar] [CrossRef]


| Effect of Age | Effect of Exposure | |||
|---|---|---|---|---|
| Control | BDE-47-Exposed | PND65 | PND120 | |
| miRNA * | 249 | 68 | 1 | 18 |
| piRNA | 908 | 44 | 0 | 0 |
| tRNA | 227 | 53 | 0 | 6 |
| Targets | Identified Non-Age-Dependent piRNA | Identified Age-Dependent piRNA | Enrichment, Fold Change | p-Value | Direction of Change in Age-Dependent Genes |
|---|---|---|---|---|---|
| All | 36,551 | 980 | -- | -- | 139 down 841 up |
| LINE | 1557 | 33 | −1.2 | 0.40 | 4 down 19 up |
| SINE | 1648 | 47 | 1.1 | 0.33 | 6 down 41 up |
| LTR | 521 | 80 | 5.3 | <0.00001 | 13 down 67 up |
| Satellite | 52 | 1 | −1.28 | 1 | 1 up |
| Protein-coding | 353 | 142 | 11.8 | <0.00001 | 24 down 118 up |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suvorov, A.; Pilsner, J.R.; Naumov, V.; Shtratnikova, V.; Zheludkevich, A.; Gerasimov, E.; Logacheva, M.; Sergeyev, O. Aging Induces Profound Changes in sncRNA in Rat Sperm and These Changes Are Modified by Perinatal Exposure to Environmental Flame Retardant. Int. J. Mol. Sci. 2020, 21, 8252. https://doi.org/10.3390/ijms21218252
Suvorov A, Pilsner JR, Naumov V, Shtratnikova V, Zheludkevich A, Gerasimov E, Logacheva M, Sergeyev O. Aging Induces Profound Changes in sncRNA in Rat Sperm and These Changes Are Modified by Perinatal Exposure to Environmental Flame Retardant. International Journal of Molecular Sciences. 2020; 21(21):8252. https://doi.org/10.3390/ijms21218252
Chicago/Turabian StyleSuvorov, Alexander, J. Richard Pilsner, Vladimir Naumov, Victoria Shtratnikova, Anna Zheludkevich, Evgeny Gerasimov, Maria Logacheva, and Oleg Sergeyev. 2020. "Aging Induces Profound Changes in sncRNA in Rat Sperm and These Changes Are Modified by Perinatal Exposure to Environmental Flame Retardant" International Journal of Molecular Sciences 21, no. 21: 8252. https://doi.org/10.3390/ijms21218252
APA StyleSuvorov, A., Pilsner, J. R., Naumov, V., Shtratnikova, V., Zheludkevich, A., Gerasimov, E., Logacheva, M., & Sergeyev, O. (2020). Aging Induces Profound Changes in sncRNA in Rat Sperm and These Changes Are Modified by Perinatal Exposure to Environmental Flame Retardant. International Journal of Molecular Sciences, 21(21), 8252. https://doi.org/10.3390/ijms21218252





