Search for Reliable Circulating Biomarkers to Predict Carotid Plaque Vulnerability
Abstract
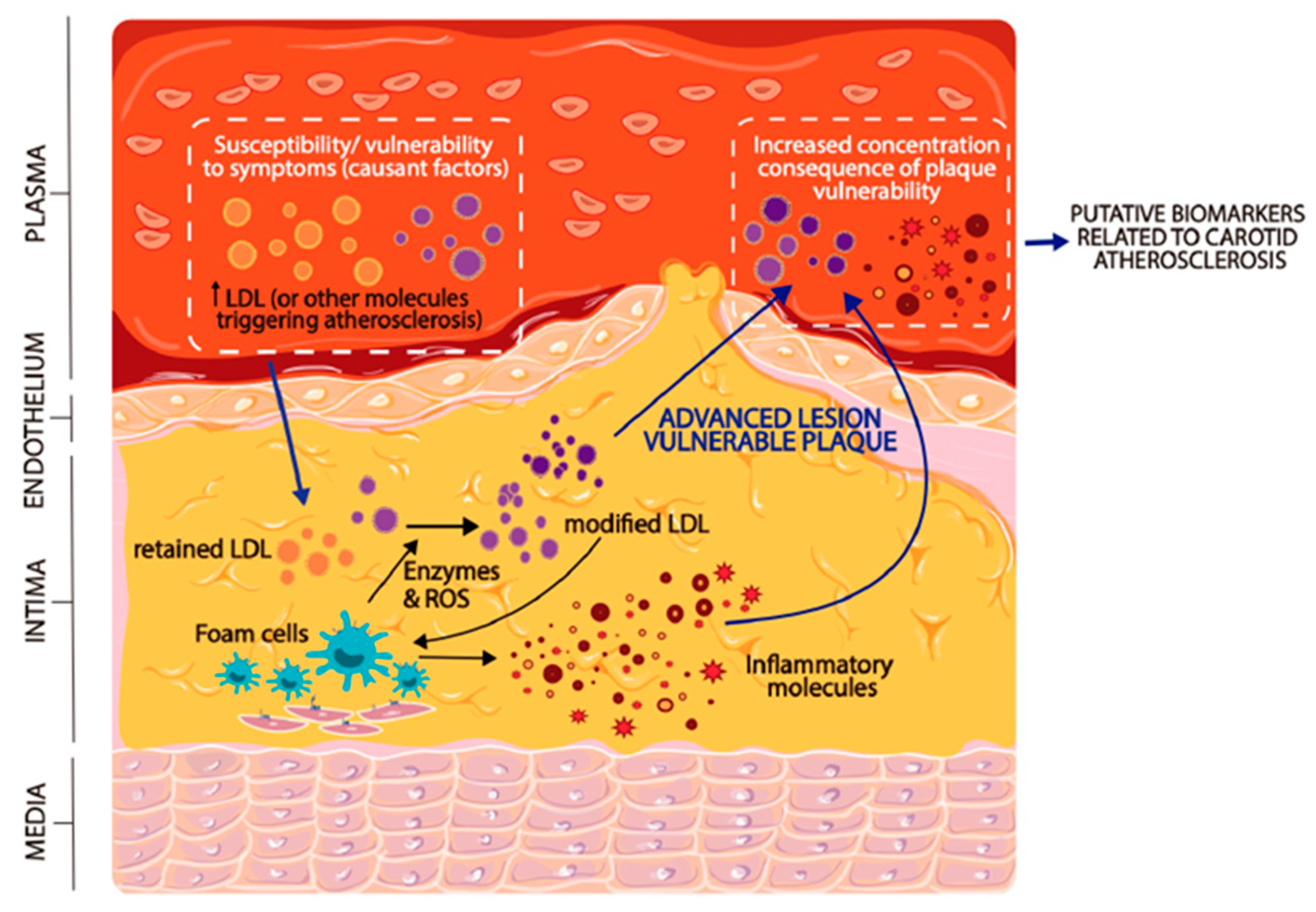
:1. Introduction
2. Atherosclerosis and Plaque Vulnerability
3. Carotid Plaque Vulnerability and Ischemic Stroke
4. Search for Circulating Biomarkers in Atherothrombotic Ischemic Stroke
5. Lipid-related and Lipoproteins Biomarkers
5.1. Lipids and Lipoproteins
5.2. Lipoprotein Components
5.3. Modified LDL
6. Inflammatory Biomarkers
6.1. Cytokines
6.2. Enzymes, Adhesion Molecules and Cell Receptors
6.3. Adipokines
7. Other Stroke Biomarkers
8. Novel Therapies
9. Plasma Biomarkers in the Clinical Practice and Future Directions
- (1)
- Testing the association between circulating biomarkers and clinical outcomes in big cohorts. Although little observational studies are useful to drive hypothesis, they have to be tested ultimately in big cohorts, in order to increase the sample size. In this regard, we would encourage basic and translational researchers to partnerships with clinicians in big clinical cohorts or trials. Biomarkers substudies within big cohort studies or trials are warranted to increase the evidence of the predictive value of circulating biomarkers.
- (2)
- Studying the association between circulating and imaging biomarkers. There are many validated imaging biomarkers that are currently used to predict carotid plaque vulnerability. Studying the association between these validated imaging biomarkers and circulating molecules can be a useful intermediate step for selecting good candidates before testing their predictive value in big cohorts.
- (3)
- Including circulating biomarkers in predictive scores. In the field of stroke, there are several examples that demonstrate that merging information from clinical, imaging, and laboratory variables increases the predictive power of some scales created to predict stroke recurrences. We believe that this approach should be taken into account in future research as it may increase the chances of a hypothetical implementation of circulating biomarkers in the clinical practice.
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saenger, A.K.; Christenson, R.H. Stroke biomarkers: Progress and challenges for diagnosis, prognosis, differentiation, and treatment. Clin. Chem. 2010, 56, 21–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranti, S.; Despres, J.P.; Fullerton, H.J.; Howard, V.J.; et al. Heart disease and stroke statistics—2015 update: A report from the american heart association. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petty, G.W.; Brown, R.D., Jr.; Whisnant, J.P.; Sicks, J.D.; O’Fallon, W.M.; Wiebers, D.O. Ischemic stroke subtypes: A population-based study of incidence and risk factors. Stroke 1999, 30, 2513–2516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaturvedi, S.; Bruno, A.; Feasby, T.; Holloway, R.; Benavente, O.; Cohen, S.N.; Cote, R.; Hess, D.; Saver, J.; Spence, J.D.; et al. Carotid endarterectomy—An evidence-based review: Report of the therapeutics and technology assessment subcommittee of the american academy of neurology. Neurology 2005, 65, 794–801. [Google Scholar] [CrossRef] [Green Version]
- Adams, H.P., Jr.; Bendixen, B.H.; Kappelle, L.J.; Biller, J.; Love, B.B.; Gordon, D.L.; Marsh, E.E., 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. Toast. Trial of org 10172 in acute stroke treatment. Stroke 1993, 24, 35–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bentzon, J.F.; Otsuka, F.; Virmani, R.; Falk, E. Mechanisms of plaque formation and rupture. Circ. Res. 2014, 114, 1852–1866. [Google Scholar] [CrossRef]
- Derdeyn, C.P. Cerebral hemodynamics in carotid occlusive disease. AJNR Am. J. Neuroradiol. 2003, 24, 1497–1499. [Google Scholar]
- Jickling, G.C.; Chaturvedi, S. Carotid plaque inflammation in stroke assessed by pet: A burning issue? Neurology 2014, 82, 1672–1673. [Google Scholar] [CrossRef] [Green Version]
- Rothwell, P.M.; Coull, A.J.; Giles, M.F.; Howard, S.C.; Silver, L.E.; Bull, L.M.; Gutnikov, S.A.; Edwards, P.; Mant, D.; Sackley, C.M.; et al. Change in stroke incidence, mortality, case-fatality, severity, and risk factors in oxfordshire, uk from 1981 to 2004 (oxford vascular study). Lancet 2004, 363, 1925–1933. [Google Scholar] [CrossRef]
- Rothwell, P.M.; Eliasziw, M.; Gutnikov, S.A.; Fox, A.J.; Taylor, D.W.; Mayberg, M.R.; Warlow, C.P.; Barnett, H.J. Analysis of pooled data from the randomised controlled trials of endarterectomy for symptomatic carotid stenosis. Lancet 2003, 361, 107–116. [Google Scholar] [CrossRef]
- Ballotta, E.; Da Giau, G.; Piccoli, A.; Baracchini, C. Durability of carotid endarterectomy for treatment of symptomatic and asymptomatic stenoses. J. Vasc. Surg. 2004, 40, 270–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spence, J.D.; Tamayo, A.; Lownie, S.P.; Ng, W.P.; Ferguson, G.G. Absence of microemboli on transcranial doppler identifies low-risk patients with asymptomatic carotid stenosis. Stroke 2005, 36, 2373–2378. [Google Scholar] [CrossRef] [Green Version]
- Martinez, E.; Martorell, J.; Riambau, V. Review of serum biomarkers in carotid atherosclerosis. J. Vasc. Surg. 2020, 71, 329–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skagen, K.; Skjelland, M.; Zamani, M.; Russell, D. Unstable carotid artery plaque: New insights and controversies in diagnostics and treatment. Croat. Med. J. 2016, 57, 311–320. [Google Scholar] [CrossRef]
- Spence, J.D.; Pilote, L. Importance of sex and gender in atherosclerosis and cardiovascular disease. Atherosclerosis 2015, 241, 208–210. [Google Scholar] [CrossRef]
- Ross, R. The pathogenesis of atherosclerosis: A perspective for the 1990s. Nature 1993, 362, 801–809. [Google Scholar] [CrossRef]
- Tabas, I. 2016 russell ross memorial lecture in vascular biology: Molecular-cellular mechanisms in the progression of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 183–189. [Google Scholar] [CrossRef] [Green Version]
- Karlof, E.; Seime, T.; Dias, N.; Lengquist, M.; Witasp, A.; Almqvist, H.; Kronqvist, M.; Gadin, J.R.; Odeberg, J.; Maegdefessel, L.; et al. Correlation of computed tomography with carotid plaque transcriptomes associates calcification with lesion-stabilization. Atherosclerosis 2019, 288, 175–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galis, Z.S.; Sukhova, G.K.; Lark, M.W.; Libby, P. Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J. Clin. Investig. 1994, 94, 2493–2503. [Google Scholar] [CrossRef] [Green Version]
- Pourcet, B.; Staels, B. Alternative macrophages in atherosclerosis: Not always protective! J. Clin. Investig. 2018, 128, 910–912. [Google Scholar] [CrossRef] [Green Version]
- Nuotio, K.; Ijas, P.; Heikkila, H.M.; Koskinen, S.M.; Saksi, J.; Vikatmaa, P.; Sorto, P.; Makitie, L.; Eriksson, H.; Kasari, S.; et al. Morphology and histology of silent and symptom-causing atherosclerotic carotid plaques—Rationale and design of the helsinki carotid endarterectomy study 2 (the heces2). Ann. Med. 2018, 50, 501–510. [Google Scholar] [CrossRef] [Green Version]
- Bartlett, E.S.; Walters, T.D.; Symons, S.P.; Fox, A.J. Quantification of carotid stenosis on ct angiography. AJNR Am. J. Neuroradiol. 2006, 27, 13–19. [Google Scholar]
- Camps-Renom, P.; Prats-Sanchez, L.; Casoni, F.; Gonzalez-de-Echavarri, J.M.; Marrero-Gonzalez, P.; Castrillon, I.; Marin, R.; Jimenez-Xarrie, E.; Delgado-Mederos, R.; Martinez-Domeno, A.; et al. Plaque neovascularization detected with contrast-enhanced ultrasound predicts ischaemic stroke recurrence in patients with carotid atherosclerosis. Eur. J. Neurol. 2020, 27, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Baradaran, H.; Schweitzer, A.D.; Kamel, H.; Pandya, A.; Delgado, D.; Dunning, A.; Mushlin, A.I.; Sanelli, P.C. Carotid plaque mri and stroke risk: A systematic review and meta-analysis. Stroke 2013, 44, 3071–3077. [Google Scholar] [CrossRef] [PubMed]
- Calogero, E.; Fabiani, I.; Pugliese, N.R.; Santini, V.; Ghiadoni, L.; Di Stefano, R.; Galetta, F.; Sartucci, F.; Penno, G.; Berchiolli, R.; et al. Three-dimensional echographic evaluation of carotid artery disease. J. Cardiovasc. Echogr. 2018, 28, 218–227. [Google Scholar]
- Saba, L.; Caddeo, G.; Sanfilippo, R.; Montisci, R.; Mallarini, G. Ct and ultrasound in the study of ulcerated carotid plaque compared with surgical results: Potentialities and advantages of multidetector row ct angiography. AJNR Am. J. Neuroradiol. 2007, 28, 1061–1066. [Google Scholar] [CrossRef] [Green Version]
- Kelly, P.J.; Camps-Renom, P.; Giannotti, N.; Marti-Fabregas, J.; Murphy, S.; McNulty, J.; Barry, M.; Barry, P.; Calvet, D.; Coutts, S.B.; et al. Carotid plaque inflammation imaged by (18)f-fluorodeoxyglucose positron emission tomography and risk of early recurrent stroke. Stroke 2019, 50, 1766–1773. [Google Scholar] [CrossRef] [PubMed]
- Skagen, K.; Johnsrud, K.; Evensen, K.; Scott, H.; Krohg-Sorensen, K.; Reier-Nilsen, F.; Revheim, M.E.; Fjeld, J.G.; Skjelland, M.; Russell, D. Carotid plaque inflammation assessed with (18)f-fdg pet/ct is higher in symptomatic compared with asymptomatic patients. Int. J. Stroke 2015, 10, 730–736. [Google Scholar] [CrossRef]
- Marnane, M.; Merwick, A.; Sheehan, O.C.; Hannon, N.; Foran, P.; Grant, T.; Dolan, E.; Moroney, J.; Murphy, S.; O’Rourke, K.; et al. Carotid plaque inflammation on 18f-fluorodeoxyglucose positron emission tomography predicts early stroke recurrence. Ann. Neurol. 2012, 71, 709–718. [Google Scholar] [CrossRef]
- Kelly, P.J.; Camps-Renom, P.; Giannotti, N.; Marti-Fabregas, J.; McNulty, J.P.; Baron, J.C.; Barry, M.; Coutts, S.B.; Cronin, S.; Delgado-Mederos, R.; et al. A risk score including carotid plaque inflammation and stenosis severity improves identification of recurrent stroke. Stroke 2020, 51, 838–845. [Google Scholar] [CrossRef]
- Whiteley, W.; Jackson, C.; Lewis, S.; Lowe, G.; Rumley, A.; Sandercock, P.; Wardlaw, J.; Dennis, M.; Sudlow, C. Inflammatory markers and poor outcome after stroke: A prospective cohort study and systematic review of interleukin-6. PLoS Med. 2009, 6, e1000145. [Google Scholar] [CrossRef]
- Ma, Z.; Yue, Y.; Luo, Y.; Wang, W.; Cao, Y.; Fang, Q. Clinical utility of the inflammatory factors combined with lipid markers in the diagnostic and prognostic assessment of ischemic stroke: Based on logistic regression models. J. Stroke Cerebrovasc. Dis. 2020, 29, 104653. [Google Scholar] [CrossRef]
- Bhosale, S.D.; Moulder, R.; Venalainen, M.S.; Koskinen, J.S.; Pitkanen, N.; Juonala, M.T.; Kahonen, M.A.P.; Lehtimaki, T.J.; Viikari, J.S.A.; Elo, L.L.; et al. Serum proteomic profiling to identify biomarkers of premature carotid atherosclerosis. Sci. Rep. 2018, 8, 9209. [Google Scholar] [CrossRef]
- Lepedda, A.J.; Nieddu, G.; Zinellu, E.; De Muro, P.; Piredda, F.; Guarino, A.; Spirito, R.; Carta, F.; Turrini, F.; Formato, M. Proteomic analysis of plasma-purified vldl, ldl, and hdl fractions from atherosclerotic patients undergoing carotid endarterectomy: Identification of serum amyloid a as a potential marker. Oxidative Med. Cell. Longev. 2013, 2013, 385214. [Google Scholar] [CrossRef] [PubMed]
- Rocchiccioli, S.; Pelosi, G.; Rosini, S.; Marconi, M.; Viglione, F.; Citti, L.; Ferrari, M.; Trivella, M.G.; Cecchettini, A. Secreted proteins from carotid endarterectomy: An untargeted approach to disclose molecular clues of plaque progression. J. Transl. Med. 2013, 11, 260. [Google Scholar] [CrossRef] [Green Version]
- Stamova, B.; Xu, H.; Jickling, G.; Bushnell, C.; Tian, Y.; Ander, B.P.; Zhan, X.; Liu, D.; Turner, R.; Adamczyk, P.; et al. Gene expression profiling of blood for the prediction of ischemic stroke. Stroke 2010, 41, 2171–2177. [Google Scholar] [CrossRef] [Green Version]
- Barr, T.L.; Conley, Y.; Ding, J.; Dillman, A.; Warach, S.; Singleton, A.; Matarin, M. Genomic biomarkers and cellular pathways of ischemic stroke by rna gene expression profiling. Neurology 2010, 75, 1009–1014. [Google Scholar] [CrossRef] [Green Version]
- Ijas, P.; Nuotio, K.; Saksi, J.; Soinne, L.; Saimanen, E.; Karjalainen-Lindsberg, M.L.; Salonen, O.; Sarna, S.; Tuimala, J.; Kovanen, P.T.; et al. Microarray analysis reveals overexpression of cd163 and ho-1 in symptomatic carotid plaques. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 154–160. [Google Scholar] [CrossRef] [Green Version]
- Saksi, J.; Ijas, P.; Nuotio, K.; Sonninen, R.; Soinne, L.; Salonen, O.; Saimanen, E.; Tuimala, J.; Lehtonen-Smeds, E.M.; Kaste, M.; et al. Gene expression differences between stroke-associated and asymptomatic carotid plaques. J. Mol. Med. 2011, 89, 1015–1026. [Google Scholar] [CrossRef] [Green Version]
- Salem, M.K.; Vijaynagar, B.; Sayers, R.D.; West, K.; Moore, D.; Robinson, T.G.; Naylor, A.R.; Bown, M.J. Histologically unstable asymptomatic carotid plaques have altered expression of genes involved in chemokine signalling leading to localised plaque inflammation and rupture. Eur. J. Vasc. Endovasc. Surg. 2013, 45, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Amarenco, P.; Labreuche, J.; Elbaz, A.; Touboul, P.J.; Driss, F.; Jaillard, A.; Bruckert, E. Blood lipids in brain infarction subtypes. Cerebrovasc. Dis. 2006, 22, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Yeh, P.S.; Yang, C.M.; Lin, S.H.; Wang, W.M.; Chen, P.S.; Chao, T.H.; Lin, H.J.; Lin, K.C.; Chang, C.Y.; Cheng, T.J.; et al. Low levels of high-density lipoprotein cholesterol in patients with atherosclerotic stroke: A prospective cohort study. Atherosclerosis 2013, 228, 472–477. [Google Scholar] [CrossRef]
- Ong, C.T.; Wong, Y.S.; Sung, S.F.; Wu, C.S.; Hsu, Y.C.; Su, Y.H.; Hung, L.C. Progression of mild to moderate stenosis in the internal carotid arteries of patients with ischemic stroke. Front. Neurol. 2018, 9, 1043. [Google Scholar] [CrossRef]
- Zeljkovic, A.; Vekic, J.; Spasojevic-Kalimanovska, V.; Jelic-Ivanovic, Z.; Bogavac-Stanojevic, N.; Gulan, B.; Spasic, S. Ldl and hdl subclasses in acute ischemic stroke: Prediction of risk and short-term mortality. Atherosclerosis 2010, 210, 548–554. [Google Scholar] [CrossRef]
- Chan, D.C.; Pang, J.; McQuillan, B.M.; Hung, J.; Beilby, J.P.; Barrett, P.H.; Watts, G.F. Plasma proprotein convertase subtilisin kexin type 9 as a predictor of carotid atherosclerosis in asymptomatic adults. Heart Lung Circ. 2016, 25, 520–525. [Google Scholar] [CrossRef] [Green Version]
- Dong, H.; Chen, W.; Wang, X.; Pi, F.; Wu, Y.; Pang, S.; Xie, Y.; Xia, F.; Zhang, Q. Apolipoprotein a1, b levels, and their ratio and the risk of a first stroke: A meta-analysis and case-control study. Metab. Brain Dis. 2015, 30, 1319–1330. [Google Scholar] [CrossRef]
- Patel, S.; Chung, S.H.; White, G.; Bao, S.; Celermajer, D.S. The “Atheroprotective” Mediators apolipoprotein a-i and foxp3 are over-abundant in unstable carotid plaques. Int. J. Cardiol. 2010, 145, 183–187. [Google Scholar] [CrossRef]
- Khan, T.A.; Shah, T.; Prieto, D.; Zhang, W.; Price, J.; Fowkes, G.R.; Cooper, J.; Talmud, P.J.; Humphries, S.E.; Sundstrom, J.; et al. Apolipoprotein e genotype, cardiovascular biomarkers and risk of stroke: Systematic review and meta-analysis of 14,015 stroke cases and pooled analysis of primary biomarker data from up to 60,883 individuals. Int. J. Epidemiol. 2013, 42, 475–492. [Google Scholar] [CrossRef] [Green Version]
- Aragones, G.; Auguet, T.; Guiu-Jurado, E.; Berlanga, A.; Curriu, M.; Martinez, S.; Alibalic, A.; Aguilar, C.; Hernandez, E.; Camara, M.L.; et al. Proteomic profile of unstable atheroma plaque: Increased neutrophil defensin 1, clusterin, and apolipoprotein e levels in carotid secretome. J. Proteome Res. 2016, 15, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, G.; Beiser, A.S.; Preis, S.R.; Courchesne, P.; Chouraki, V.; Levy, D.; Seshadri, S. Plasma clusterin levels and risk of dementia, alzheimer’s disease, and stroke. Alzheimers Dement. 2016, 3, 103–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yanni, A.E.; Agrogiannis, G.; Gkekas, C.; Perrea, D. Clusterin/apolipoprotein j immunolocalization on carotid artery is affected by tnf-alpha, cigarette smoking and anti-platelet treatment. Lipids Health Dis. 2014, 13, 70. [Google Scholar] [CrossRef] [Green Version]
- Elkind, M.S.; Tai, W.; Coates, K.; Paik, M.C.; Sacco, R.L. Lipoprotein-associated phospholipase a2 activity and risk of recurrent stroke. Cerebrovasc. Dis. 2009, 27, 42–50. [Google Scholar] [CrossRef]
- Oei, H.H.; van der Meer, I.M.; Hofman, A.; Koudstaal, P.J.; Stijnen, T.; Breteler, M.M.; Witteman, J.C. Lipoprotein-associated phospholipase a2 activity is associated with risk of coronary heart disease and ischemic stroke: The rotterdam study. Circulation 2005, 111, 570–575. [Google Scholar] [CrossRef] [Green Version]
- Katan, M.; Moon, Y.P.; Paik, M.C.; Wolfert, R.L.; Sacco, R.L.; Elkind, M.S. Lipoprotein-associated phospholipase a2 is associated with atherosclerotic stroke risk: The northern manhattan study. PLoS ONE 2014, 9, e83393. [Google Scholar] [CrossRef] [Green Version]
- Otsuka, F.; Zhao, X.; Trout, H.H.; Qiao, Y.; Wasserman, B.A.; Nakano, M.; Macphee, C.H.; Brandt, M.; Krug-Gourley, S.; Guo, L.; et al. Community-based statins and advanced carotid plaque: Role of cd163 positive macrophages in lipoprotein-associated phospholipase a2 activity in atherosclerotic plaque. Atherosclerosis 2017, 267, 78–89. [Google Scholar] [CrossRef]
- Wang, A.; Yang, Y.; Su, Z.; Yue, W.; Hao, H.; Ren, L.; Wang, Y.; Cao, Y. Association of oxidized low-density lipoprotein with prognosis of stroke and stroke subtypes. Stroke 2017, 48, 91–97. [Google Scholar] [CrossRef]
- Wang, A.; Dai, L.; Zhang, N.; Lin, J.; Chen, G.; Zuo, Y.; Li, H.; Wang, Y.; Meng, X. Oxidized low-density lipoprotein (ldl) and ldl cholesterol are associated with outcomes of minor stroke and tia. Atherosclerosis 2020, 297, 74–80. [Google Scholar] [CrossRef]
- Sigala, F.; Kotsinas, A.; Savari, P.; Filis, K.; Markantonis, S.; Iliodromitis, E.K.; Gorgoulis, V.G.; Andreadou, I. Oxidized ldl in human carotid plaques is related to symptomatic carotid disease and lesion instability. J. Vasc. Surg. 2010, 52, 704–713. [Google Scholar] [CrossRef] [Green Version]
- Lehti, S.; Nguyen, S.D.; Belevich, I.; Vihinen, H.; Heikkila, H.M.; Soliymani, R.; Kakela, R.; Saksi, J.; Jauhiainen, M.; Grabowski, G.A.; et al. Extracellular lipids accumulate in human carotid arteries as distinct three-dimensional structures and have proinflammatory properties. Am. J. Pathol. 2018, 188, 525–538. [Google Scholar] [CrossRef] [Green Version]
- Chaudhuri, J.R.; Mridula, K.R.; Umamahesh, M.; Swathi, A.; Balaraju, B.; Bandaru, V.C. High sensitivity c-reactive protein levels in acute ischemic stroke and subtypes: A study from a tertiary care center. Iran. J. Neurol. 2013, 12, 92–97. [Google Scholar] [PubMed]
- Yamagami, H.; Kitagawa, K.; Nagai, Y.; Hougaku, H.; Sakaguchi, M.; Kuwabara, K.; Kondo, K.; Masuyama, T.; Matsumoto, M.; Hori, M. Higher levels of interleukin-6 are associated with lower echogenicity of carotid artery plaques. Stroke 2004, 35, 677–681. [Google Scholar] [CrossRef] [Green Version]
- Yi, L.; Tang, J.; Shi, C.; Zhang, T.; Li, J.; Guo, F.; Zhang, W. Pentraxin 3, tnf-alpha, and ldl-c are associated with carotid artery stenosis in patients with ischemic stroke. Front. Neurol. 2019, 10, 1365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shindo, A.; Tanemura, H.; Yata, K.; Hamada, K.; Shibata, M.; Umeda, Y.; Asakura, F.; Toma, N.; Sakaida, H.; Fujisawa, T.; et al. Inflammatory biomarkers in atherosclerosis: Pentraxin 3 can become a novel marker of plaque vulnerability. PLoS ONE 2014, 9, e100045. [Google Scholar] [CrossRef]
- Ormstad, H.; Aass, H.C.; Lund-Sorensen, N.; Amthor, K.F.; Sandvik, L. Serum levels of cytokines and c-reactive protein in acute ischemic stroke patients, and their relationship to stroke lateralization, type, and infarct volume. J. Neurol. 2011, 258, 677–685. [Google Scholar] [CrossRef] [Green Version]
- Welsh, P.; Lowe, G.D.; Chalmers, J.; Campbell, D.J.; Rumley, A.; Neal, B.C.; MacMahon, S.W.; Woodward, M. Associations of proinflammatory cytokines with the risk of recurrent stroke. Stroke 2008, 39, 2226–2230. [Google Scholar] [CrossRef]
- Markstad, H.; Edsfeldt, A.; Yao Mattison, I.; Bengtsson, E.; Singh, P.; Cavalera, M.; Asciutto, G.; Bjorkbacka, H.; Fredrikson, G.N.; Dias, N.; et al. High levels of soluble lectinlike oxidized low-density lipoprotein receptor-1 are associated with carotid plaque inflammation and increased risk of ischemic stroke. J. Am. Heart Assoc. 2019, 8, e009874. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Xie, W.L.; Kong, W.W.; Chen, D.; Qu, P. Expression of the nlrp3 inflammasome in carotid atherosclerosis. J. Stroke Cerebrovasc. Dis. 2015, 24, 2455–2466. [Google Scholar] [CrossRef]
- Zaremba, J.; Losy, J. Interleukin-18 in acute ischaemic stroke patients. Neurol. Sci. 2003, 24, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Abbas, A.; Gregersen, I.; Holm, S.; Daissormont, I.; Bjerkeli, V.; Krohg-Sorensen, K.; Skagen, K.R.; Dahl, T.B.; Russell, D.; Almas, T.; et al. Interleukin 23 levels are increased in carotid atherosclerosis: Possible role for the interleukin 23/interleukin 17 axis. Stroke 2015, 46, 793–799. [Google Scholar] [CrossRef] [Green Version]
- Georgakis, M.K.; Malik, R.; Bjorkbacka, H.; Pana, T.A.; Demissie, S.; Ayers, C.; Elhadad, M.A.; Fornage, M.; Beiser, A.S.; Benjamin, E.J.; et al. Circulating monocyte chemoattractant protein-1 and risk of stroke: Meta-analysis of population-based studies involving 17 180 individuals. Circ. Res. 2019, 125, 773–782. [Google Scholar] [CrossRef] [Green Version]
- Lehmann, M.F.; Kallaur, A.P.; Oliveira, S.R.; Alfieri, D.F.; Delongui, F.; de Sousa Parreira, J.; de Araujo, M.C.; Rossato, C.; de Almeida, J.T.; Pelegrino, L.M.; et al. Inflammatory and metabolic markers and short-time outcome in patients with acute ischemic stroke in relation to toast subtypes. Metab. Brain Dis. 2015, 30, 1417–1428. [Google Scholar] [CrossRef]
- Mazzotta, G.; Sarchielli, P.; Caso, V.; Paciaroni, M.; Floridi, A.; Gallai, V. Different cytokine levels in thrombolysis patients as predictors for clinical outcome. Eur. J. Neurol. 2004, 11, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.B.; Chun, S.; Choi-Kwon, S.; Chi, H.S.; Nah, H.W.; Kwon, S.U.; Kim, W.K.; Kim, J.S. Biomarkers and location of atherosclerosis: Matrix metalloproteinase-2 may be related to intracranial atherosclerosis. Atherosclerosis 2012, 223, 442–447. [Google Scholar] [CrossRef]
- Iemolo, F.; Sanzaro, E.; Duro, G.; Giordano, A.; Paciaroni, M. The prognostic value of biomarkers in stroke. Immun. Ageing 2016, 13, 19. [Google Scholar] [CrossRef] [Green Version]
- Abbas, A.; Aukrust, P.; Russell, D.; Krohg-Sorensen, K.; Almas, T.; Bundgaard, D.; Bjerkeli, V.; Sagen, E.L.; Michelsen, A.E.; Dahl, T.B.; et al. Matrix metalloproteinase 7 is associated with symptomatic lesions and adverse events in patients with carotid atherosclerosis. PLoS ONE 2014, 9, e84935. [Google Scholar] [CrossRef] [Green Version]
- Palm, F.; Pussinen, P.J.; Safer, A.; Tervahartiala, T.; Sorsa, T.; Urbanek, C.; Becher, H.; Grau, A.J. Serum matrix metalloproteinase-8, tissue inhibitor of metalloproteinase and myeloperoxidase in ischemic stroke. Atherosclerosis 2018, 271, 9–14. [Google Scholar] [CrossRef] [Green Version]
- Zhong, C.; Yang, J.; Xu, T.; Peng, Y.; Wang, A.; Wang, J.; Peng, H.; Li, Q.; Ju, Z.; Geng, D.; et al. Serum matrix metalloproteinase-9 levels and prognosis of acute ischemic stroke. Neurology 2017, 89, 805–812. [Google Scholar] [CrossRef]
- Cojocarui, I.M.; Cojocaru, M.; Sapira, V.; Socoliuc, G.; Hertea, C.; Paveliu, S. Changes in plasma matrix metalloproteinase-9 levels in patients with acute ischemic stroke. Rom. J. Intern. Med. 2012, 50, 155–158. [Google Scholar]
- Guo, Z.Y.; Zhang, B.; Yan, Y.H.; Gao, S.S.; Liu, J.J.; Xu, L.; Hui, P.J. Specific matrix metalloproteinases and calcification factors are associated with the vulnerability of human carotid plaque. Exp. Ther. Med. 2018, 16, 2071–2079. [Google Scholar] [CrossRef] [Green Version]
- Langley, S.R.; Willeit, K.; Didangelos, A.; Matic, L.P.; Skroblin, P.; Barallobre-Barreiro, J.; Lengquist, M.; Rungger, G.; Kapustin, A.; Kedenko, L.; et al. Extracellular matrix proteomics identifies molecular signature of symptomatic carotid plaques. J. Clin. Investig. 2017, 127, 1546–1560. [Google Scholar] [CrossRef]
- Eilenberg, W.; Stojkovic, S.; Kaider, A.; Piechota-Polanczyk, A.; Nanobachvili, J.; Domenig, C.M.; Wojta, J.; Huk, I.; Demyanets, S.; Neumayer, C. Neutrophil gelatinase associated lipocalin (ngal) for identification of unstable plaques in patients with asymptomatic carotid stenosis. Eur. J. Vasc. Endovasc. Surg. 2019, 57, 768–777. [Google Scholar] [CrossRef]
- Orion, D.; von Landenberg, P.; Itsekson-Hayosh, Z.; Schwammenthal, Y.; Tsabari, R.; Merzeliak, O.; Chapman, J.; Tanne, D. Plasma myeloperoxidase levels in acute brain ischaemia and high grade carotid stenosis. Eur. J. Neurol. 2020, 27, 1604–1611. [Google Scholar] [CrossRef]
- Kozuka, K.; Kohriyama, T.; Nomura, E.; Ikeda, J.; Kajikawa, H.; Nakamura, S. Endothelial markers and adhesion molecules in acute ischemic stroke—Sequential change and differences in stroke subtype. Atherosclerosis 2002, 161, 161–168. [Google Scholar] [CrossRef]
- Wang, J.; Li, J.; Liu, Q. Association between platelet activation and fibrinolysis in acute stroke patients. Neurosci. Lett. 2005, 384, 305–309. [Google Scholar] [CrossRef]
- Hofmann, A.; Brunssen, C.; Wolk, S.; Reeps, C.; Morawietz, H. Soluble lox-1: A novel biomarker in patients with coronary artery disease, stroke, and acute aortic dissection? J. Am. Heart Assoc. 2020, 9, e013803. [Google Scholar] [CrossRef]
- Yokota, C.; Sawamura, T.; Watanabe, M.; Kokubo, Y.; Fujita, Y.; Kakino, A.; Nakai, M.; Toyoda, K.; Miyamoto, Y.; Minematsu, K. High levels of soluble lectin-like oxidized low-density lipoprotein receptor-1 in acute stroke: An age- and sex-matched cross-sectional study. J. Atheroscler. Thromb. 2016, 23, 1222–1226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, W.; Li, Q.; Chen, X.; Lin, Y.; Xue, J.; Cai, Z.; Zhang, W.; Wang, H.; Jin, K.; Shao, B. Soluble lectin-like oxidized low-density lipoprotein receptor-1 as a novel biomarker for large-artery atherosclerotic stroke. Int. J. Neurosci. 2017, 127, 881–886. [Google Scholar] [CrossRef]
- Skarpengland, T.; Skjelland, M.; Kong, X.Y.; Skagen, K.; Holm, S.; Otterdal, K.; Dahl, C.P.; Krohg-Sorensen, K.; Sagen, E.L.; Bjerkeli, V.; et al. Increased levels of lectin-like oxidized low-density lipoprotein receptor-1 in ischemic stroke and transient ischemic attack. J. Am. Heart Assoc. 2018, 7, e006479. [Google Scholar] [CrossRef] [Green Version]
- Isoviita, P.M.; Nuotio, K.; Saksi, J.; Turunen, R.; Ijas, P.; Pitkaniemi, J.; Soinne, L.; Kaste, M.; Kovanen, P.T.; Lindsberg, P.J. An imbalance between cd36 and abca1 protein expression favors lipid accumulation in stroke-prone ulcerated carotid plaques. Stroke 2010, 41, 389–393. [Google Scholar] [CrossRef]
- Handberg, A.; Skjelland, M.; Michelsen, A.E.; Sagen, E.L.; Krohg-Sorensen, K.; Russell, D.; Dahl, A.; Ueland, T.; Oie, E.; Aukrust, P.; et al. Soluble cd36 in plasma is increased in patients with symptomatic atherosclerotic carotid plaques and is related to plaque instability. Stroke 2008, 39, 3092–3095. [Google Scholar] [CrossRef] [Green Version]
- O’Connell, G.C.; Tennant, C.S.; Lucke-Wold, N.; Kabbani, Y.; Tarabishy, A.R.; Chantler, P.D.; Barr, T.L. Monocyte-lymphocyte cross-communication via soluble cd163 directly links innate immune system activation and adaptive immune system suppression following ischemic stroke. Sci. Rep. 2017, 7, 12940. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aristoteli, L.P.; Moller, H.J.; Bailey, B.; Moestrup, S.K.; Kritharides, L. The monocytic lineage specific soluble cd163 is a plasma marker of coronary atherosclerosis. Atherosclerosis 2006, 184, 342–347. [Google Scholar] [CrossRef]
- Lu, L.F.; Yang, S.S.; Wang, C.P.; Hung, W.C.; Yu, T.H.; Chiu, C.A.; Chung, F.M.; Shin, S.J.; Lee, Y.J. Elevated visfatin/pre-b-cell colony-enhancing factor plasma concentration in ischemic stroke. J. Stroke Cerebrovasc. Dis. 2009, 18, 354–359. [Google Scholar] [CrossRef]
- Kadoglou, N.P.; Fotiadis, G.; Lambadiari, V.; Maratou, E.; Dimitriadis, G.; Liapis, C.D. Serum levels of novel adipokines in patients with acute ischemic stroke: Potential contribution to diagnosis and prognosis. Peptides 2014, 57, 12–16. [Google Scholar] [CrossRef]
- Yue, J.; Chen, J.; Wu, Q.; Liu, X.; Li, M.; Li, Z.; Gao, Y. Serum levels of omentin-1 association with early diagnosis, lesion volume and severity of acute ischemic stroke. Cytokine 2018, 111, 518–522. [Google Scholar] [CrossRef]
- Xu, T.; Zuo, P.; Wang, Y.; Gao, Z.; Ke, K. Serum omentin-1 is a novel biomarker for predicting the functional outcome of acute ischemic stroke patients. Clin. Chem. Lab. Med. 2018, 56, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Efstathiou, S.P.; Tsiakou, A.G.; Tsioulos, D.I.; Panagiotou, T.N.; Pefanis, A.V.; Achimastos, A.D.; Mountokalakis, T.D. Prognostic significance of plasma resistin levels in patients with atherothrombotic ischemic stroke. Clin. Chim. Acta 2007, 378, 78–85. [Google Scholar] [CrossRef]
- Holm, S.; Ueland, T.; Dahl, T.B.; Michelsen, A.E.; Skjelland, M.; Russell, D.; Nymo, S.H.; Krohg-Sorensen, K.; Clausen, O.P.; Atar, D.; et al. Fatty acid binding protein 4 is associated with carotid atherosclerosis and outcome in patients with acute ischemic stroke. PLoS ONE 2011, 6, e28785. [Google Scholar] [CrossRef]
- Agardh, H.E.; Folkersen, L.; Ekstrand, J.; Marcus, D.; Swedenborg, J.; Hedin, U.; Gabrielsen, A.; Paulsson-Berne, G. Expression of fatty acid-binding protein 4/ap2 is correlated with plaque instability in carotid atherosclerosis. J. Intern. Med. 2011, 269, 200–210. [Google Scholar] [CrossRef] [Green Version]
- Tsai, P.C.; Liao, Y.C.; Wang, Y.S.; Lin, H.F.; Lin, R.T.; Juo, S.H. Serum microrna-21 and microrna-221 as potential biomarkers for cerebrovascular disease. J. Vasc. Res. 2013, 50, 346–354. [Google Scholar] [CrossRef]
- Eken, S.M.; Jin, H.; Chernogubova, E.; Li, Y.; Simon, N.; Sun, C.; Korzunowicz, G.; Busch, A.; Backlund, A.; Osterholm, C.; et al. Microrna-210 enhances fibrous cap stability in advanced atherosclerotic lesions. Circ. Res. 2017, 120, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Sepramaniam, S.; Tan, J.R.; Tan, K.S.; DeSilva, D.A.; Tavintharan, S.; Woon, F.P.; Wang, C.W.; Yong, F.L.; Karolina, D.S.; Kaur, P.; et al. Circulating micrornas as biomarkers of acute stroke. Int. J. Mol. Sci. 2014, 15, 1418–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, Y.; Liu, J.X.; Yan, Z.P.; Yao, X.H.; Liu, X.H. Potential microrna biomarkers for acute ischemic stroke. Int. J. Mol. Med. 2015, 36, 1639–1647. [Google Scholar] [CrossRef] [Green Version]
- Dolz, S.; Gorriz, D.; Tembl, J.I.; Sanchez, D.; Fortea, G.; Parkhutik, V.; Lago, A. Circulating micrornas as novel biomarkers of stenosis progression in asymptomatic carotid stenosis. Stroke 2017, 48, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Cipollone, F.; Felicioni, L.; Sarzani, R.; Ucchino, S.; Spigonardo, F.; Mandolini, C.; Malatesta, S.; Bucci, M.; Mammarella, C.; Santovito, D.; et al. A unique microrna signature associated with plaque instability in humans. Stroke 2011, 42, 2556–2563. [Google Scholar] [CrossRef] [Green Version]
- Maitrias, P.; Metzinger-Le Meuth, V.; Massy, Z.A.; M’Baya-Moutoula, E.; Reix, T.; Caus, T.; Metzinger, L. Microrna deregulation in symptomatic carotid plaque. J. Vasc. Surg. 2015, 62, 1245–1250.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, J.; Yang, S.; Wang, K.; Zhong, Q.; Ma, A.; Pan, X. Plasma mir-126 and mir-143 as potential novel biomarkers for cerebral atherosclerosis. J. Stroke Cerebrovasc. Dis. 2019, 28, 38–43. [Google Scholar] [CrossRef]
- An, T.H.; He, Q.W.; Xia, Y.P.; Chen, S.C.; Baral, S.; Mao, L.; Jin, H.J.; Li, Y.N.; Wang, M.D.; Chen, J.G.; et al. Mir-181b antagonizes atherosclerotic plaque vulnerability through modulating macrophage polarization by directly targeting notch1. Mol. Neurobiol. 2017, 54, 6329–6341. [Google Scholar] [CrossRef]
- Magenta, A.; Sileno, S.; D’Agostino, M.; Persiani, F.; Beji, S.; Paolini, A.; Camilli, D.; Platone, A.; Capogrossi, M.C.; Furgiuele, S. Atherosclerotic plaque instability in carotid arteries: Mir-200c as a promising biomarker. Clin. Sci. 2018, 132, 2423–2436. [Google Scholar] [CrossRef]
- Bazan, H.A.; Hatfield, S.A.; Brug, A.; Brooks, A.J.; Lightell, D.J., Jr.; Woods, T.C. Carotid plaque rupture is accompanied by an increase in the ratio of serum circr-284 to mir-221 levels. Circ. Cardiovasc. Genet. 2017, 10, e001720. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Qin, Y.; Zhu, G.; Li, Y.; Xue, J. Low serum mir-320b expression as a novel indicator of carotid atherosclerosis. J. Clin. Neurosci. 2016, 33, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Wezel, A.; Welten, S.M.; Razawy, W.; Lagraauw, H.M.; de Vries, M.R.; Goossens, E.A.; Boonstra, M.C.; Hamming, J.F.; Kandimalla, E.R.; Kuiper, J.; et al. Inhibition of microrna-494 reduces carotid artery atherosclerotic lesion development and increases plaque stability. Ann. Surg. 2015, 262, 841–848. [Google Scholar] [CrossRef]
- Abela, G.S. Cholesterol crystals piercing the arterial plaque and intima trigger local and systemic inflammation. J. Clin. Lipidol. 2010, 4, 156–164. [Google Scholar] [CrossRef]
- Edsfeldt, A.; Duner, P.; Stahlman, M.; Mollet, I.G.; Asciutto, G.; Grufman, H.; Nitulescu, M.; Persson, A.F.; Fisher, R.M.; Melander, O.; et al. Sphingolipids contribute to human atherosclerotic plaque inflammation. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1132–1140. [Google Scholar] [CrossRef] [Green Version]
- Gu, X.; Li, Y.; Chen, S.; Yang, X.; Liu, F.; Li, J.; Cao, J.; Liu, X.; Chen, J.; Shen, C.; et al. Association of lipids with ischemic and hemorrhagic stroke: A prospective cohort study among 267,500 chinese. Stroke 2019, 50, 3376–3384. [Google Scholar] [CrossRef]
- Laloux, P.; Galanti, L.; Jamart, J. Lipids in ischemic stroke subtypes. Acta Neurol. Belg. 2004, 104, 13–19. [Google Scholar] [PubMed]
- Hindy, G.; Engstrom, G.; Larsson, S.C.; Traylor, M.; Markus, H.S.; Melander, O.; Orho-Melander, M. Role of blood lipids in the development of ischemic stroke and its subtypes: A mendelian randomization study. Stroke 2018, 49, 820–827. [Google Scholar] [CrossRef]
- Amarenco, P.; Hobeanu, C.; Labreuche, J.; Charles, H.; Giroud, M.; Meseguer, E.; Lavallee, P.C.; Gabriel Steg, P.; Vicaut, E.; Bruckert, E.; et al. Carotid atherosclerosis evolution when targeting a low-density lipoprotein cholesterol concentration <70 mg/dL after an ischemic stroke of atherosclerotic origin. Circulation 2020, 142, 748–757. [Google Scholar] [CrossRef]
- Furberg, C.D.; Adams, H.P., Jr.; Applegate, W.B.; Byington, R.P.; Espeland, M.A.; Hartwell, T.; Hunninghake, D.B.; Lefkowitz, D.S.; Probstfield, J.; Riley, W.A.; et al. Effect of lovastatin on early carotid atherosclerosis and cardiovascular events. Asymptomatic carotid artery progression study (acaps) research group. Circulation 1994, 90, 1679–1687. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Liu, S.; Liu, Y.; Hua, Y.; Song, H.; Ren, Y.; Song, Y.; Liu, R.; Feng, W.; Ovbiagele, B.; et al. Achieving low density lipoprotein-cholesterol < 70mg/dL may be associated with a trend of reduced progression of carotid artery atherosclerosis in ischemic stroke patients. J. Neurol. Sci. 2017, 378, 26–29. [Google Scholar] [CrossRef] [Green Version]
- Xie, W.; Liu, J.; Wang, W.; Wang, M.; Qi, Y.; Zhao, F.; Sun, J.; Li, Y.; Zhao, D. Association between plasma pcsk9 levels and 10-year progression of carotid atherosclerosis beyond ldl-c: A cohort study. Int. J. Cardiol. 2016, 215, 293–298. [Google Scholar] [CrossRef]
- Hong, X.W.; Wu, D.M.; Lu, J.; Zheng, Y.L.; Tu, W.J.; Yan, J. Lipoprotein (a) as a predictor of early stroke recurrence in acute ischemic stroke. Mol. Neurobiol. 2018, 55, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Alam, R.; Yatsu, F.M.; Kasturi, R.; Bui, G. Low and high density lipoprotein metabolism in atherothrombotic brain infarction. Stroke 1992, 23, 1265–1270. [Google Scholar] [CrossRef] [Green Version]
- Tirschwell, D.L.; Smith, N.L.; Heckbert, S.R.; Lemaitre, R.N.; Longstreth, W.T., Jr.; Psaty, B.M. Association of cholesterol with stroke risk varies in stroke subtypes and patient subgroups. Neurology 2004, 63, 1868–1875. [Google Scholar] [CrossRef]
- Meilhac, O. High-density lipoproteins in stroke. Handb. Exp. Pharmacol. 2015, 224, 509–526. [Google Scholar] [PubMed] [Green Version]
- Tiozzo, E.; Gardener, H.; Hudson, B.I.; Dong, C.; Della-Morte, D.; Crisby, M.; Goldberg, R.B.; Elkind, M.S.; Cheung, Y.K.; Wright, C.B.; et al. Subfractions of high-density lipoprotein-cholesterol and carotid intima-media thickness: The northern manhattan study. Stroke 2016, 47, 1508–1513. [Google Scholar] [CrossRef] [Green Version]
- Chei, C.L.; Yamagishi, K.; Kitamura, A.; Kiyama, M.; Imano, H.; Ohira, T.; Cui, R.; Tanigawa, T.; Sankai, T.; Ishikawa, Y.; et al. High-density lipoprotein subclasses and risk of stroke and its subtypes in japanese population: The circulatory risk in communities study. Stroke 2013, 44, 327–333. [Google Scholar] [CrossRef] [Green Version]
- Jain, J.; Lathia, T.; Gupta, O.P.; Jain, V. Carotid intima-media thickness and apolipoproteins in patients of ischemic stroke in a rural hospital setting in central india: A cross-sectional study. J. Neurosci. Rural. Pract. 2012, 3, 21–27. [Google Scholar] [PubMed]
- Ye, F.; Liu, J.; Yang, S.; Guo, F.Q. Higher apolipoprotein b levels are associated with earlier onset of first-ever atherosclerotic stroke. Int. J. Neurosci. 2015, 125, 186–190. [Google Scholar] [CrossRef]
- Yue, Y.H.; Bai, X.D.; Li, Y.M.; Hu, L.; Liu, L.Y.; Mao, J.P.; Yang, X.Y.; Dila, N.M. The association of serum lipid level with ischemic stroke in the elderly of xinjiang. Neuroendocrinol. Lett. 2019, 39, 572–578. [Google Scholar]
- Charnay, Y.; Imhof, A.; Vallet, P.G.; Kovari, E.; Bouras, C.; Giannakopoulos, P. Clusterin in neurological disorders: Molecular perspectives and clinical relevance. Brain Res. Bull. 2012, 88, 434–443. [Google Scholar] [CrossRef]
- Song, H.; Zhou, H.; Qu, Z.; Hou, J.; Chen, W.; Cai, W.; Cheng, Q.; Chuang, D.Y.; Chen, S.; Li, S.; et al. From analysis of ischemic mouse brain proteome to identification of human serum clusterin as a potential biomarker for severity of acute ischemic stroke. Transl. Stroke Res. 2019, 10, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Karabina, S.A.; Liapikos, T.A.; Grekas, G.; Goudevenos, J.; Tselepis, A.D. Distribution of paf-acetylhydrolase activity in human plasma low-density lipoprotein subfractions. Biochim. Biophys. Acta 1994, 1213, 34–38. [Google Scholar] [CrossRef]
- Yang, Y.; Xue, T.; Zhu, J.; Xu, J.; Hu, X.; Wang, P.; Kong, T.; Yan, Y.; Yang, L.; Xue, S. Serum lipoprotein-associated phospholipase a2 predicts the formation of carotid artery plaque and its vulnerability in anterior circulation cerebral infarction. Clin. Neurol. Neurosurg. 2017, 160, 40–45. [Google Scholar] [CrossRef]
- Walsh, K.B.; Hart, K.; Roll, S.; Sperling, M.; Unruh, D.; Davidson, W.S.; Lindsell, C.J.; Adeoye, O. Apolipoprotein a-i and paraoxonase-1 are potential blood biomarkers for ischemic stroke diagnosis. J. Stroke Cerebrovasc. Dis. 2016, 25, 1360–1365. [Google Scholar] [CrossRef] [Green Version]
- Chawhan, S.S.; Mogarekar, M.R.; Wagh, R.V.; Das, R.R.; Pramanik, S.S.; Sonune, S.M.; Chawhan, S.M. Relation of paraoxonase1, arylesterase and lipid profile in ischemic stroke patients. J. Clin. Diagn. Res. 2015, 9, BC01–BC03. [Google Scholar] [CrossRef] [PubMed]
- Michalak, S.; Kazmierski, R.; Hellmann, A.; Wysocka, E.; Kocialkowska-Adamczewska, D.; Wencel-Warot, A.; Nowinski, W.L. Serum paraoxonase/arylesterase activity affects outcome in ischemic stroke patients. Cerebrovasc. Dis. 2011, 32, 124–132. [Google Scholar] [CrossRef]
- Ishigaki, Y.; Oka, Y.; Katagiri, H. Circulating oxidized ldl: A biomarker and a pathogenic factor. Curr. Opin. Lipidol. 2009, 20, 363–369. [Google Scholar] [CrossRef]
- Nishi, K.; Itabe, H.; Uno, M.; Kitazato, K.T.; Horiguchi, H.; Shinno, K.; Nagahiro, S. Oxidized ldl in carotid plaques and plasma associates with plaque instability. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1649–1654. [Google Scholar] [CrossRef] [Green Version]
- Yang, T.C.; Chang, P.Y.; Lu, S.C. L5-ldl from st-elevation myocardial infarction patients induces il-1beta production via lox-1 and nlrp3 inflammasome activation in macrophages. Am. J. Physiol. Heart Circ. Physiol. 2017, 312, H265–H274. [Google Scholar] [CrossRef] [Green Version]
- Chang, C.Y.; Chen, C.H.; Chen, Y.M.; Hsieh, T.Y.; Li, J.P.; Shen, M.Y.; Lan, J.L.; Chen, D.Y. Association between negatively charged low-density lipoprotein l5 and subclinical atherosclerosis in rheumatoid arthritis patients. J. Clin. Med. 2019, 8, 177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Estruch, M.; Sanchez-Quesada, J.L.; Ordonez Llanos, J.; Benitez, S. Electronegative ldl: A circulating modified ldl with a role in inflammation. Mediat. Inflamm. 2013, 2013, 181324. [Google Scholar] [CrossRef] [Green Version]
- Puig, N.; Montolio, L.; Camps-Renom, P.; Navarra, L.; Jimenez-Altayo, F.; Jimenez-Xarrie, E.; Sanchez-Quesada, J.L.; Benitez, S. Electronegative ldl promotes inflammation and triglyceride accumulation in macrophages. Cells 2020, 9, 583. [Google Scholar] [CrossRef] [Green Version]
- Shen, M.Y.; Chen, F.Y.; Hsu, J.F.; Fu, R.H.; Chang, C.M.; Chang, C.T.; Liu, C.H.; Wu, J.R.; Lee, A.S.; Chan, H.C.; et al. Plasma l5 levels are elevated in ischemic stroke patients and enhance platelet aggregation. Blood 2016, 127, 1336–1345. [Google Scholar] [CrossRef] [Green Version]
- Tuttolomondo, A.; Di Raimondo, D.; Pecoraro, R.; Arnao, V.; Pinto, A.; Licata, G. Inflammation in ischemic stroke subtypes. Curr. Pharm. Des. 2012, 18, 4289–4310. [Google Scholar] [CrossRef]
- Zhou, Y.; Han, W.; Gong, D.; Man, C.; Fan, Y. Hs-crp in stroke: A meta-analysis. Clin. Chim. Acta 2016, 453, 21–27. [Google Scholar] [CrossRef]
- Zheng, X.; Zeng, N.; Wang, A.; Zhu, Z.; Zhong, C.; Xu, T.; Peng, Y.; Peng, H.; Li, Q.; Ju, Z.; et al. Elevated c-reactive protein and depressed high-density lipoprotein cholesterol are associated with poor function outcome after ischemic stroke. Curr. Neurovasc. Res. 2018, 15, 226–233. [Google Scholar] [CrossRef]
- Zhang, Y.B.; Yin, Z.; Han, X.; Wang, Q.; Zhang, Z.; Geng, J. Association of circulating high-sensitivity c-reactive protein with late recurrence after ischemic stroke. Neuroreport 2017, 28, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Nayak, A.R.; Kashyap, R.S.; Purohit, H.J.; Kabra, D.; Taori, G.M.; Daginawala, H.F. Evaluation of the inflammatory response in sera from acute ischemic stroke patients by measurement of il-2 and il-10. Inflamm. Res. 2009, 58, 687–691. [Google Scholar] [CrossRef]
- Yuen, C.M.; Chiu, C.A.; Chang, L.T.; Liou, C.W.; Lu, C.H.; Youssef, A.A.; Yip, H.K. Level and value of interleukin-18 after acute ischemic stroke. Circ. J. 2007, 71, 1691–1696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vila, N.; Castillo, J.; Davalos, A.; Esteve, A.; Planas, A.M.; Chamorro, A. Levels of anti-inflammatory cytokines and neurological worsening in acute ischemic stroke. Stroke 2003, 34, 671–675. [Google Scholar] [CrossRef] [Green Version]
- Al-Bahrani, A.; Taha, S.; Shaath, H.; Bakhiet, M. Tnf-alpha and il-8 in acute stroke and the modulation of these cytokines by antiplatelet agents. Curr. Neurovasc. Res. 2007, 4, 31–37. [Google Scholar] [CrossRef]
- Krupinski, J.; Turu, M.M.; Font, M.A.; Ahmed, N.; Sullivan, M.; Rubio, F.; Badimon, L.; Slevin, M. Increased tissue factor, mmp-8, and d-dimer expression in diabetic patients with unstable advanced carotid atherosclerosis. Vasc. Health Risk Manag. 2007, 3, 405–412. [Google Scholar]
- Fassbender, K.; Mossner, R.; Motsch, L.; Kischka, U.; Grau, A.; Hennerici, M. Circulating selectin- and immunoglobulin-type adhesion molecules in acute ischemic stroke. Stroke 1995, 26, 1361–1364. [Google Scholar] [CrossRef]
- Frijns, C.J.; Kappelle, L.J. Inflammatory cell adhesion molecules in ischemic cerebrovascular disease. Stroke 2002, 33, 2115–2122. [Google Scholar] [CrossRef]
- Kobayashi, N.; Takano, M.; Hata, N.; Kume, N.; Yamamoto, M.; Yokoyama, S.; Shinada, T.; Tomita, K.; Shirakabe, A.; Otsuka, T.; et al. Soluble lectin-like oxidized ldl receptor-1 (slox-1) as a valuable diagnostic marker for rupture of thin-cap fibroatheroma: Verification by optical coherence tomography. Int. J. Cardiol. 2013, 168, 3217–3223. [Google Scholar] [CrossRef]
- Kume, N.; Mitsuoka, H.; Hayashida, K.; Tanaka, M.; Kita, T. Soluble lectin-like oxidized low-density lipoprotein receptor-1 predicts prognosis after acute coronary syndrome—A pilot study. Circ. J. 2010, 74, 1399–1404. [Google Scholar] [CrossRef] [Green Version]
- Olson, N.C.; Koh, I.; Reiner, A.P.; Judd, S.E.; Irvin, M.R.; Howard, G.; Zakai, N.A.; Cushman, M. Soluble cd14, ischemic stroke, and coronary heart disease risk in a prospective study: The regards cohort. J. Am. Heart Assoc. 2020, 9, e014241. [Google Scholar] [CrossRef]
- Zeller, J.A.; Tschoepe, D.; Kessler, C. Circulating platelets show increased activation in patients with acute cerebral ischemia. Thromb. Haemost. 1999, 81, 373–377. [Google Scholar]
- Gairolla, J.; Kler, R.; Modi, M.; Khurana, D. Leptin and adiponectin: Pathophysiological role and possible therapeutic target of inflammation in ischemic stroke. Rev. Neurosci. 2017, 28, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Efstathiou, S.P.; Tsioulos, D.I.; Tsiakou, A.G.; Gratsias, Y.E.; Pefanis, A.V.; Mountokalakis, T.D. Plasma adiponectin levels and five-year survival after first-ever ischemic stroke. Stroke 2005, 36, 1915–1919. [Google Scholar] [CrossRef]
- Kantorova, E.; Chomova, M.; Kurca, E.; Sivak, S.; Zelenak, K.; Kucera, P.; Galajda, P. Leptin, adiponectin and ghrelin, new potential mediators of ischemic stroke. Neuro. Endocrinol. Lett. 2011, 32, 716–721. [Google Scholar]
- Kim, B.J.; Lee, S.H.; Ryu, W.S.; Kim, C.K.; Yoon, B.W. Adipocytokines and ischemic stroke: Differential associations between stroke subtypes. J. Neurol. Sci. 2012, 312, 117–122. [Google Scholar] [CrossRef]
- Kochanowski, J.; Grudniak, M.; Baranowska-Bik, A.; Wolinska-Witort, E.; Kalisz, M.; Baranowska, B.; Bik, W. Resistin levels in women with ischemic stroke. Neuro. Endocrinol. Lett. 2012, 33, 603–607. [Google Scholar]
- Xu, T.; Zuo, P.; Cao, L.; Gao, Z.; Ke, K. Omentin-1 is associated with carotid plaque instability among ischemic stroke patients. J. Atheroscler. Thromb. 2018, 25, 505–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahl, T.B.; Yndestad, A.; Skjelland, M.; Oie, E.; Dahl, A.; Michelsen, A.; Damas, J.K.; Tunheim, S.H.; Ueland, T.; Smith, C.; et al. Increased expression of visfatin in macrophages of human unstable carotid and coronary atherosclerosis: Possible role in inflammation and plaque destabilization. Circulation 2007, 115, 972–980. [Google Scholar] [CrossRef]
- Yu, F.; Zhou, X.; Li, Z.; Feng, X.; Liao, D.; Liu, Z.; Huang, Q.; Li, X.; Yang, Q.; Xiao, B.; et al. Diagnostic significance of plasma levels of novel adipokines in patients with symptomatic intra- and extracranial atherosclerotic stenosis. Front. Neurol. 2019, 10, 1228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuttolomondo, A.; Di Raimondo, D.; Forte, G.I.; Casuccio, A.; Vaccarino, L.; Scola, L.; Pecoraro, R.; Serio, A.; Clemente, G.; Arnao, V.; et al. Single nucleotide polymorphisms (snps) of pro-inflammatory/anti-inflammatory and thrombotic/fibrinolytic genes in patients with acute ischemic stroke in relation to toast subtype. Cytokine 2012, 58, 398–405. [Google Scholar] [CrossRef]
- Biscetti, F.; Straface, G.; Bertoletti, G.; Vincenzoni, C.; Snider, F.; Arena, V.; Landolfi, R.; Flex, A. Identification of a potential proinflammatory genetic profile influencing carotid plaque vulnerability. J. Vasc. Surg. 2015, 61, 374–381. [Google Scholar] [CrossRef] [Green Version]
- Tso, A.R.; Merino, J.G.; Warach, S. Interleukin-6 174g/c polymorphism and ischemic stroke: A systematic review. Stroke 2007, 38, 3070–3075. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Li, Q.; Zhu, R.; He, Z. Association of il-10-1082a/g polymorphism with ischemic stroke: Evidence from a case-control study to an updated meta-analysis. Genet. Test. Mol. Biomark. 2017, 21, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Niu, F.; Wei, B.; Yan, M.; Li, J.; Ouyang, Y.; Jin, T. Matrix metalloproteinase-2 gene polymorphisms are associated with ischemic stroke in a hainan population. Medicine 2018, 97, e12302. [Google Scholar] [CrossRef]
- Wu, G.; Cai, H.; Li, G.; Meng, S.; Huang, J.; Xu, H.; Chen, M.; Hu, M.; Yang, W.; Wang, C.; et al. Influence of the matrix metalloproteinase 9 geners3918242 polymorphism on development of ischemic stroke: A meta-analysis. World Neurosurg. 2020, 133, e31–e61. [Google Scholar] [CrossRef]
- Misra, S.; Talwar, P.; Kumar, A.; Kumar, P.; Sagar, R.; Vibha, D.; Pandit, A.K.; Gulati, A.; Kushwaha, S.; Prasad, K. Association between matrix metalloproteinase family gene polymorphisms and risk of ischemic stroke: A systematic review and meta-analysis of 29 studies. Gene 2018, 672, 180–194. [Google Scholar] [CrossRef]
- Ding, G.; Wang, J.; Liu, K.; Huang, B.; Deng, W.; He, T. Association of e-selectin gene rs5361 polymorphism with ischemic stroke susceptibility: A systematic review and meta-analysis. Int. J. Neurosci. 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.S.; Lan, Y.; Meng, L.Q.; Nong, L.G. The association of l-selectin polymorphisms with l-selectin serum levels and risk of ischemic stroke. J. Thromb. Thrombolysis 2011, 32, 110–115. [Google Scholar] [CrossRef]
- Zhang, Y.; Zang, J.; Wang, B.; Li, B.; Yao, X.; Zhao, H.; Li, W. Cd36 genotype associated with ischemic stroke in chinese han. Int. J. Clin. Exp. Med. 2015, 8, 16149–16157. [Google Scholar]
- Ferreira, J.P.; Xhaard, C.; Lamiral, Z.; Borges-Canha, M.; Neves, J.S.; Dandine-Roulland, C.; LeFloch, E.; Deleuze, J.F.; Bacq-Daian, D.; Bozec, E.; et al. Pcsk9 protein and rs562556 polymorphism are associated with arterial plaques in healthy middle-aged population: The stanislas cohort. J. Am. Heart Assoc. 2020, 9, e014758. [Google Scholar] [CrossRef]
- Xiuju, C.; Zhen, W.; Yanchao, S. A meta-analysis of adiponectin gene rs22411766 t>g polymorphism and ischemic stroke susceptibility. Open Med. 2016, 11, 115–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, F.; Guo, T.; Zhou, L.; Zhou, Y.; Yu, D. Variants in the apob gene was associated with ischemic stroke susceptibility in chinese han male population. Oncotarget 2018, 9, 2249–2254. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y. Associations of platelet-activating factor acetylhydrolase gene polymorphisms with risk of ischemic stroke. Biomed. Rep. 2016, 4, 246–250. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahabreh, I.J.; Kitsios, G.D.; Kent, D.M.; Trikalinos, T.A. Paraoxonase 1 polymorphisms and ischemic stroke risk: A systematic review and meta-analysis. Genet. Med. 2010, 12, 606–615. [Google Scholar] [CrossRef] [Green Version]
- Glushakova, O.Y.; Glushakov, A.V.; Miller, E.R.; Valadka, A.B.; Hayes, R.L. Biomarkers for acute diagnosis and management of stroke in neurointensive care units. Brain Circ. 2016, 2, 28–47. [Google Scholar] [CrossRef]
- Raju, S.; Fish, J.E.; Howe, K.L. Micrornas as sentinels and protagonists of carotid artery thromboembolism. Clin. Sci. 2020, 134, 169–192. [Google Scholar] [CrossRef] [PubMed]
- Badacz, R.; Przewlocki, T.; Gacon, J.; Stepien, E.; Enguita, F.J.; Karch, I.; Zmudka, K.; Kablak-Ziembicka, A. Circulating mirna levels differ with respect to carotid plaque characteristics and symptom occurrence in patients with carotid artery stenosis and provide information on future cardiovascular events. Postepy Kardiol. Interwencyjnej 2018, 14, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Sun, Y.; Han, T.; Zhu, J.; Xie, Y.; Wang, S.; Wu, Y.; Fan, Y.; Sun, X.; Zhou, J.; et al. Upregulation of mir-330-5p is associated with carotid plaque’s stability by targeting talin-1 in symptomatic carotid stenosis patients. BMC Cardiovasc. Disord. 2019, 19, 149. [Google Scholar] [CrossRef]
- Kim, J.M.; Jung, K.H.; Chu, K.; Lee, S.T.; Ban, J.; Moon, J.; Kim, M.; Lee, S.K.; Roh, J.K. Atherosclerosis-related circulating micrornas as a predictor of stroke recurrence. Transl. Stroke Res. 2015, 6, 191–197. [Google Scholar] [CrossRef]
- Luque, A.; Farwati, A.; Krupinski, J.; Aran, J.M. Association between low levels of serum mir-638 and atherosclerotic plaque vulnerability in patients with high-grade carotid stenosis. J. Neurosurg. 2018, 131, 72–79. [Google Scholar] [CrossRef]
- Urra, X.; Cervera, A.; Obach, V.; Climent, N.; Planas, A.M.; Chamorro, A. Monocytes are major players in the prognosis and risk of infection after acute stroke. Stroke 2009, 40, 1262–1268. [Google Scholar] [CrossRef] [Green Version]
- del Zoppo, G.J. Acute anti-inflammatory approaches to ischemic stroke. Ann. N. Y. Acad. Sci. 2010, 1207, 143–148. [Google Scholar] [CrossRef] [Green Version]
- Ridker, P.M.; Everett, B.M.; Thuren, T.; MacFadyen, J.G.; Chang, W.H.; Ballantyne, C.; Fonseca, F.; Nicolau, J.; Koenig, W.; Anker, S.D.; et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 2017, 377, 1119–1131. [Google Scholar] [CrossRef]
- Liberale, L.; Diaz-Canestro, C.; Bonetti, N.R.; Paneni, F.; Akhmedov, A.; Beer, J.H.; Montecucco, F.; Luscher, T.F.; Camici, G.G. Post-ischaemic administration of the murine canakinumab-surrogate antibody improves outcome in experimental stroke. Eur. Heart J. 2018, 39, 3511–3517. [Google Scholar] [CrossRef]
- Smith, C.J.; Hulme, S.; Vail, A.; Heal, C.; Parry-Jones, A.R.; Scarth, S.; Hopkins, K.; Hoadley, M.; Allan, S.M.; Rothwell, N.J.; et al. Scil-stroke (subcutaneous interleukin-1 receptor antagonist in ischemic stroke): A randomized controlled phase 2 trial. Stroke 2018, 49, 1210–1216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuttolomondo, A.; Pecoraro, R.; Pinto, A. Studies of selective tnf inhibitors in the treatment of brain injury from stroke and trauma: A review of the evidence to date. Drug Des. Dev. Ther. 2014, 8, 2221–2238. [Google Scholar] [CrossRef] [Green Version]
- Arribas, J.; Esselens, C. Adam17 as a therapeutic target in multiple diseases. Curr. Pharm. Des. 2009, 15, 2319–2335. [Google Scholar] [CrossRef]
- Sander, D.; Winbeck, K.; Klingelhofer, J.; Etgen, T.; Conrad, B. Reduced progression of early carotid atherosclerosis after antibiotic treatment and chlamydia pneumoniae seropositivity. Circulation 2002, 106, 2428–2433. [Google Scholar] [CrossRef] [Green Version]
- Shingai, Y.; Kimura, N.; Doijiri, R.; Takahashi, K.; Yokosawa, M.; Kanoke, A.; Kikuchi, T.; Sugawara, T.; Tominaga, T. Effect of preoperative administration of proprotein convertase subtilisin/kexin type 9 inhibitor on carotid artery stenting. World Neurosurg. 2020, 135, e36–e42. [Google Scholar] [CrossRef]


| Basal State/Asymptomatic | ≤24 h | ≤14 d | ≥14 d | Plaque | References | ||
|---|---|---|---|---|---|---|---|
| Lipid-related and lipoproteins biomarkers | TC | P | P | [41,42] | |||
| LDLc | P | P | P | [41,42,43] | |||
| HDLc | P | P | P | [41,42] | |||
| HDL3 | P | [44] | |||||
| PCSK9 | P | Yes | [45] | ||||
| apoA-I | P, S | P, S | Yes | [46,47] | |||
| apoE | P | S | S | Yes | [48,49] | ||
| apoJ | P | S | S | Yes | [49,50,51] | ||
| PAF-AH | S | S | S | Yes | [52,53,54,55] | ||
| oxLDL | P | P | Yes | [56,57,58] | |||
| aggLDL | Yes | [59] | |||||
| Inflammatory biomarkers | hs-CRP | S | P, S | P, S | [60,61] | ||
| PTX3 | S | P | P | S | Yes | [62,63] | |
| IL-6 | P, S | P, S | P, S | Yes | [64,65,66] | ||
| IL-18 | S | S | Yes | [67,68] | |||
| IL-23 | S | Yes | [69] | ||||
| MCP1 | P, S | [70] | |||||
| TNF-α | P | P * | P * | Yes | [64,65,66,71,72] | ||
| MMP-2 | P | S | Yes | [66,73,74] | |||
| MMP-7 | P | Yes | [75] | ||||
| MMP-8 | S | [76] | |||||
| MMP-9 | P, S | Yes | [71,77,78,79,80] | ||||
| NGAL | S | [81] | |||||
| MPO | P, S | P | [76,82] | ||||
| E-selectin | P * | P * | Yes | [83] | |||
| P-selectin | P * | P * | P * | [83,84] | |||
| LOX-1 | P | S | P | P | Yes | [66,85,86,87,88] | |
| CD36 | P | Yes | [39,89,90] | ||||
| CD63 | P | [91] | |||||
| CD163 | S | Yes | [39,92] | ||||
| Ghrelin | P, S | [93,94] | |||||
| Omentin-1 | S | [95,96] | |||||
| Resistin | P | P | [97] | ||||
| Vaspin | P, S | [93,94] | |||||
| FABP4 | P | Yes | [98,99] | ||||
| miRNA | miR-21 | S | S | Yes | [100,101] | ||
| miR-126 | P | P | P | [102,103] | |||
| miR-130a | P | P | P | [102,104] | |||
| miR-133 | S | Yes | [105,106] | ||||
| miR-143/145 | P | P | Yes | [102,105,106,107] | |||
| miR-155 | P | Yes | [106] | ||||
| miR-181b | P | P | [108] | ||||
| miR-199b | P | [104] | |||||
| miR-200c | P | Yes | [109] | ||||
| miR-210 | P | P | Yes | [101] | |||
| miR-221 | S | Yes | [106,110] | ||||
| miR-320b | S | P | [102,111] | ||||
| miR-330 | S | Yes | [103,110] | ||||
| miR-242 | P | Yes | [102] | ||||
| miR-494 | P | [102,112] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puig, N.; Jiménez-Xarrié, E.; Camps-Renom, P.; Benitez, S. Search for Reliable Circulating Biomarkers to Predict Carotid Plaque Vulnerability. Int. J. Mol. Sci. 2020, 21, 8236. https://doi.org/10.3390/ijms21218236
Puig N, Jiménez-Xarrié E, Camps-Renom P, Benitez S. Search for Reliable Circulating Biomarkers to Predict Carotid Plaque Vulnerability. International Journal of Molecular Sciences. 2020; 21(21):8236. https://doi.org/10.3390/ijms21218236
Chicago/Turabian StylePuig, Núria, Elena Jiménez-Xarrié, Pol Camps-Renom, and Sonia Benitez. 2020. "Search for Reliable Circulating Biomarkers to Predict Carotid Plaque Vulnerability" International Journal of Molecular Sciences 21, no. 21: 8236. https://doi.org/10.3390/ijms21218236
APA StylePuig, N., Jiménez-Xarrié, E., Camps-Renom, P., & Benitez, S. (2020). Search for Reliable Circulating Biomarkers to Predict Carotid Plaque Vulnerability. International Journal of Molecular Sciences, 21(21), 8236. https://doi.org/10.3390/ijms21218236