Implications of Altered Endosome and Lysosome Biology in Space Environments
Abstract
:1. Introduction
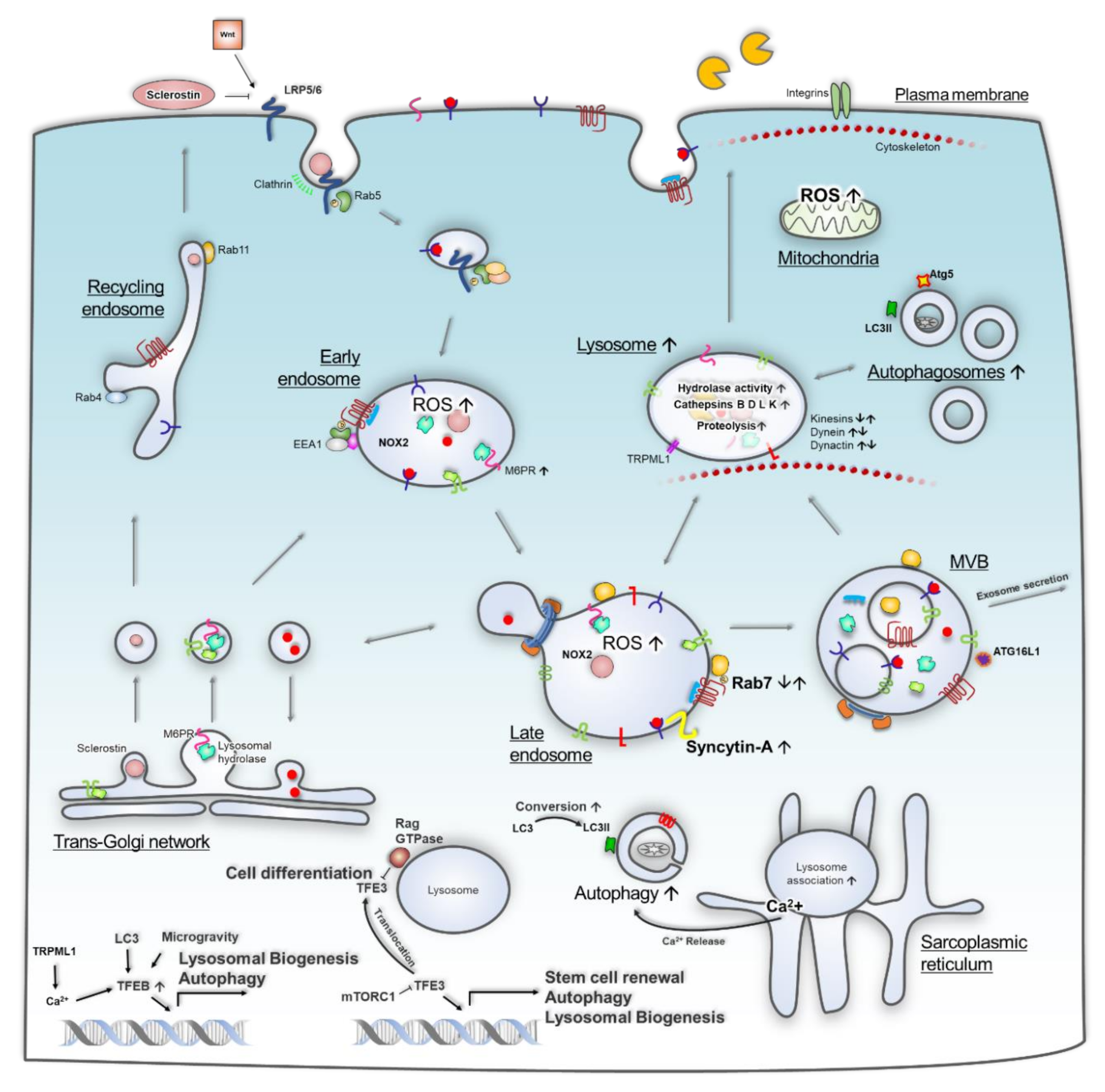
2. The Endosome-Lysosome System and Autophagy in Microgravity-Induced Pathophysiology
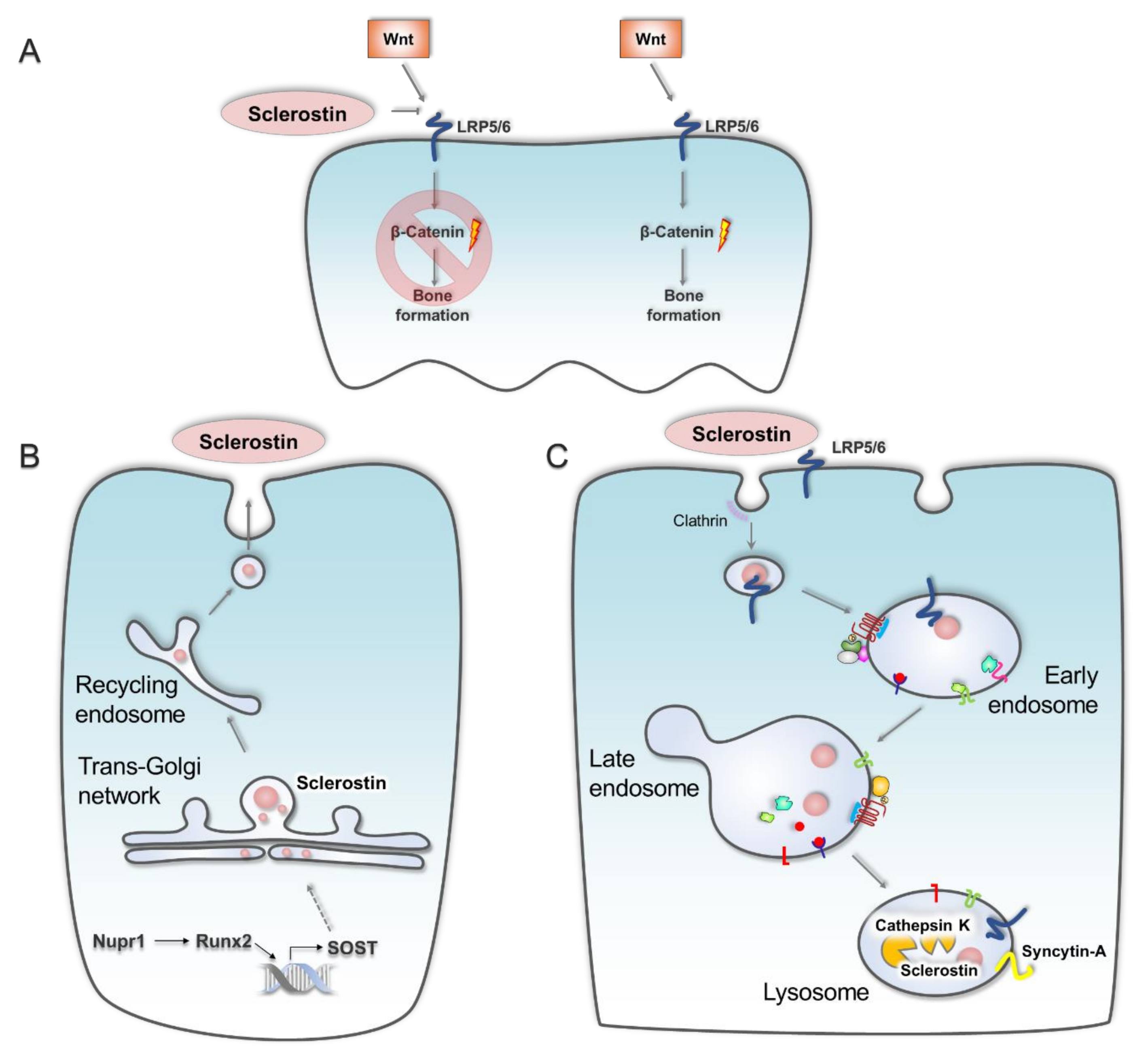
2.1. Bone Loss
2.2. Muscle Atrophy
2.3. Cell Differentiations
2.4. Oxidative Metabolism
2.5. The Lysosomal System as a Monitor for Physiological Stress
3. Discussion and Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| µg | Microgravity |
| ROS | Reactive oxygen species |
| TFEB | Transcription factor EB |
| RPM | Random positioning machine |
| RWV | Rotating wall vessel |
| SR | Sarcoplasmic reticulum |
| EEA1 | Early Endosome Antigen 1 |
| M6PR | Mannose-6-phosphate receptor |
| TRPML1 | Transient receptor potential cation channel 1 |
References
- Ballabio, A.; Bonifacino, J.S. Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat. Rev. Mol. Cell Biol. 2020, 21, 101–118. [Google Scholar] [CrossRef]
- Luzio, J.P.; Hackmann, Y.; Dieckmann, N.M.; Griffiths, G.M. The biogenesis of lysosomes and lysosome-related organelles. Cold Spring Harbor. Perspect.Biol. 2014, 6, a016840. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandri, M. Protein breakdown in muscle wasting: Role of autophagy-lysosome and ubiquitin-proteasome. Int. J. Biochem. Cell Biol. 2013, 45, 2121–2129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sambandam, Y.; Townsend, M.T.; Pierce, J.J.; Lipman, C.M.; Haque, A.; Bateman, T.A.; Reddy, S.V. Microgravity Control of Autophagy Modulates Osteoclastogenesis. Bone 2014, 61, 125–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ethiraj, P.; Link, J.R.; Sinkway, J.M.; Brown, G.D.; Parler, W.A.; Reddy, S.V. Microgravity Modulation of Syncytin-a Expression Enhance Osteoclast Formation. J. Cell Biochem. 2018, 119, 5696–5703. [Google Scholar] [CrossRef] [PubMed]
- Hessvik, N.P.; Llorente, A. Current Knowledge on Exosome Biogenesis and Release. Cell Mol Life Sci. 2018, 75, 193–208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, I.R.; Parkinson-Lawrence, E.J.; Shandala, T.; Weigert, R.; Butler, L.M.; Brooks, D.A. Altered Endosome Biogenesis in Prostate Cancer Has Biomarker Potential. Mol. Cancer Res. 2014, 12, 1851–1862. [Google Scholar] [CrossRef] [Green Version]
- Fennelly, C.; Amaravadi, R.K. Lysosomal Biology in Cancer. Methods Mol. Biol. 2017, 1594, 293–308. [Google Scholar] [PubMed] [Green Version]
- Zhang, L.; Sheng, R.; Qin, Z. The Lysosome and Neurodegenerative Diseases. Acta. Biochim. Biophys. Sin. 2009, 41, 437–445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carmona-Gutierrez, D.; Hughes, A.L.; Madeo, F.; Ruckenstuhl, C. The Crucial Impact of Lysosomes in Aging and Longevity. Ageing Res. Rev. 2016, 32, 2–12. [Google Scholar] [CrossRef]
- Vernikos, J.; Schneider, V.S. Space, Gravity and the Physiology of Aging: Parallel or Convergent Disciplines? A Mini-Review. Gerontology 2010, 56, 157–166. [Google Scholar] [CrossRef]
- Demontis, G.; Germani, M.M.; Caiani, E.; Barravecchia, I.; Passino, C.; Angeloni, D. Human Pathophysiological Adaptations to the Space Environment. Front. Physiol. 2017, 8, 547. [Google Scholar] [CrossRef]
- Kourtidou-Papadeli, C.; Papadelis, C.L.; Vernikos, J.; Bamidis, P.D.; Hitoglou-Antoniadou, M.; Perantoni, E.; Vlachogiannis, E. The Therapeutic Benefits of Gravity in Space and on Earth. Hippokratia 2008, 12, 28–31. [Google Scholar] [PubMed]
- Orwoll, E.S.; Adler, R.A.; Amin, S.; Binkley, N.; Lewiecki, E.M.; Petak, S.M.; Shapses, S.A.; Sinaki, M.; Watts, N.B.; Sibonga, J.D. Skeletal Health in Long-Duration Astronauts: Nature, Assessment, and Management Recommendations from the Nasa Bone Summit. J. Bone Miner. Res. 2013, 28, 1243–1255. [Google Scholar] [CrossRef]
- Strollo, F.; Gentile, S.; Strollo, G.; Mambro, A.; Vernikos, J. Recent Progress in Space Physiology and Aging. Front. Physiol. 2018, 9, 1551. [Google Scholar] [CrossRef] [Green Version]
- Grimm, D.; Egli, M.; Krüger, M.; Riwaldt, S.; Corydon, T.J.; Kopp, S.; Wehland, M.; Wise, P.; Infanger, M.; Mann, V.; et al. Tissue Engineering under Microgravity Conditions–Use of Stem Cells and Specialized Cells. Stem Cells Dev. 2018, 27, 787–804. [Google Scholar] [CrossRef]
- Steller, J.G.; Alberts, J.R.; Ronca, A.E. Oxidative Stress as Cause, Consequence, or Biomarker of Altered Female Reproduction and Development in the Space Environment. Int. J. Mol. Sci. 2018, 19, 3729. [Google Scholar] [CrossRef] [Green Version]
- Gunga, H.-C. Human Physiology in Extreme Environments; Academic Press: Boston, MA, USA, 2014; pp. 273–311. [Google Scholar]
- Elizabeth, A.B.; Michael, J.P.; Karen, R.J. Spaceflight Activates Autophagy Programs and the Proteasome in Mouse Liver. Int. J. Mol. Sci. 2017, 18, 2062. [Google Scholar]
- Markolefa, I.; Lambrou, G.I. The Role of Autophagy During Osteoclastogenesis under Microgravity Conditions. Int. J. Astrobiol. 2019, 18, 384–390. [Google Scholar] [CrossRef]
- Pajevic, P.D.; Spatz, J.M.; Garr, J.; Adamson, C.; Misener, L. Osteocyte Biology and Space Flight. Curr. Biotechnol. 2013, 2, 179–183. [Google Scholar] [CrossRef] [Green Version]
- Rokhlenko, K.D.; Mul’diiarov, P. Myocardial Ultrastructure of Rats Exposed Aboard Biosatellite “Cosmos-936”. Kosm. Biol. Aviakosm. Med. 1981, 15, 77–82. [Google Scholar] [PubMed]
- Mednieks, M.I.; Hand, A.R. Salivary Gland Ultrastructure and Cyclic Amp-Dependent Reactions in Spacelab 3 Rats. Am. J. Physiol. 1987, 252, R233–R239. [Google Scholar] [CrossRef] [PubMed]
- Gambara, G.; Salanova, M.; Ciciliot, S.; Furlan, S.; Gutsmann, M.; Schiffl, G.; Ungethuem, U.; Volpe, P.; Gunga, H.C.; Blottner, D. Microgravity-Induced Transcriptome Adaptation in Mouse Paraspinal Longissimus Dorsi Muscle Highlights Insulin Resistance-Linked Genes. Front. Physiol. 2017, 8, 279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bradamante, S.; Rivero, D.; Barenghi, L.; Balsamo, M.; Minardi, S.P.; Vitali, F.; Cavalieri, D. Scd—Stem Cell Differentiation toward Osteoblast Onboard the International Space Station. Microgravity Sci. Technol. 2018, 30, 713–729. [Google Scholar] [CrossRef] [Green Version]
- Nogues, C.; Pfister, A.; Kaiser, R. Comparative Study of Lesions in the Brain of Rats Exposed to Heavy Ions: Accelerator and Stratospheric Flight Research. Life Sci. Space Res. 1978, 16, 119–125. [Google Scholar]
- Doty, S.B.; Morey-Holton, E. Alterations in Bone Forming Cells Due to Reduced Weight Bearing. Physiologist 1984, 27, S81–S82. [Google Scholar]
- Dagdeviren, D.; Beallias, J.; Khan, I.; Mednieks, M.I.; Hand, A.R. Response of the Mouse Sublingual Gland to Spaceflight. Eur. J. Oral. Sci. 2018, 126, 373–381. [Google Scholar] [CrossRef]
- Riley, D.A.; Ellis, S.; Slocum, G.R.; Satyanarayana, T.; Bain, J.L.; Sedlak, F.R. Hypogravity-Induced Atrophy of Rat Soleus and Extensor Digitorum Longus Muscles. Muscle Nerve 1987, 10, 560–568. [Google Scholar] [CrossRef]
- Sandonà, D.; Desaphy, J.F.; Camerino, G.M.; Bianchini, E.; Ciciliot, S.; Danieli-Betto, D.; Dobrowolny, G.; Furlan, S.; Germinario, E.; Goto, K.; et al. Adaptation of Mouse Skeletal Muscle to Long-Term Microgravity in the Mds Mission. PLoS ONE 2012, 7, e33232. [Google Scholar] [CrossRef] [Green Version]
- García-Ovejero, D.; Trejo, J.L.; Ciriza, I.; Walton, K.D.; García-Segura, L.M. Space Flight Affects Magnocellular Supraoptic Neurons of Young Prepuberal Rats: Transient and Permanent Effects. Dev. Brain Res. 2001, 130, 191–205. [Google Scholar] [CrossRef]
- Rodionova, N.V.; Oganov, V.S.; Zolotova, N.V. Ultrastructural Changes in Osteocytes in Microgravity Conditions. Adv. Space Res. 2002, 30, 765–770. [Google Scholar] [CrossRef]
- Roffino, S.; Carnino, A.; Chopard, A.; Mutin, M.; Marini, J.F. Structural Remodeling of Unweighted Soleus Myotendinous Junction in Monkey. C. R. Biol. 2006, 329, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Onishi, Y.; Hirasaka, K.; Ishihara, I.; Oarada, M.; Goto, J.; Ogawa, T.; Suzue, N.; Nakano, S.; Furochi, H.; Ishidoh, K.; et al. Identification of Mono-Ubiquitinated Ldh-a in Skeletal Muscle Cells Exposed to Oxidative Stress. Biochem. Biophys. Res. Commun. 2005, 336, 799–806. [Google Scholar] [CrossRef]
- Tischler, M.E.; Kirby, C.; Rosenberg, S.; Tome, M.; Chase, P. Mechanisms of Accelerated Proteolysis in Rat Soleus Muscle Atrophy Induced by Unweighting or Denervation. Physiologist 1991, 34, S177–S178. [Google Scholar]
- Taillandier, D.; Aurousseau, E.; Meynial-Denis, D.; Bechet, D.; Ferrara, M.; Cottin, P.; Ducastaing, A.; Bigard, X.; Guezennec, C.Y.; Schmid, H.P.; et al. Coordinate Activation of Lysosomal, Ca 2+-Activated and Atp-Ubiquitin-Dependent Proteinases in the Unweighted Rat Soleus Muscle. Biochem. J. 1996, 316 (Pt 1), 65–72. [Google Scholar] [CrossRef] [Green Version]
- Dupont-Versteegden, E.E.; Fluckey, J.D.; Knox, M.; Gaddy, D.; Peterson, C.A. Effect of Flywheel-Based Resistance Exercise on Processes Contributing to Muscle Atrophy During Unloading in Adult Rats. J. Appl. Physiol. 2006, 101, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.; Vitorino, R.; Neuparth, M.J.; Appell, H.-J.; Amado, F.; Duarte, J.A. Cellular Patterns of the Atrophic Response in Murine Soleus and Gastrocnemius Muscles Submitted to Simulated Weightlessness. Eur. J. Appl. Physiol. 2007, 101, 331–340. [Google Scholar] [CrossRef]
- Ferreira, R.; Neuparth, M.J.; Vitorino, R.; Appell, H.J.; Amado, F.; Duarte, J.A. Evidences of Apoptosis During the Early Phases of Soleus Muscle Atrophy in Hindlimb Suspended Mice. Physiol. Res. 2008, 57, 601–611. [Google Scholar]
- Liu, H.; Xie, Q.; Xin, B.-M.; Liu, J.-L.; Liu, Y.; Li, Y.-Z.; Wang, J.-P. Inhibition of Autophagy Recovers Cardiac Dysfunction and Atrophy in Response to Tail-Suspension. Life Sci. 2015, 121, 1–9. [Google Scholar] [CrossRef]
- Sambandam, Y.; Blanchard, J.J.; Daughtridge, G.; Kolb, R.J.; Shanmugarajan, S.; Pandruvada, S.N.M.; Bateman, T.A.; Reddy, S.V. Microarray Profile of Gene Expression During Osteoclast Differentiation in Modelled Microgravity. J. Cell. Biochem. 2010, 111, 1179–1187. [Google Scholar] [CrossRef]
- Li, C.-F.; Sun, J.-X.; Gao, Y.; Shi, F.; Pan, Y.-K.; Wang, Y.-C.; Sun, X.-Q. Clinorotation-Induced Autophagy Via Hdm2-P53-Mtor Pathway Enhances Cell Migration in Vascular Endothelial Cells. Cell Death Dis. 2018, 9, 147. [Google Scholar] [CrossRef] [PubMed]
- Ryu, H.-W.; Choi, S.-H.; Namkoong, S.; Jang, I.-S.; Seo, D.H.; Choi, I.; Kim, H.-S.; Park, J. Simulated Microgravity Contributes to Autophagy Induction by Regulating Amp-Activated Protein Kinase. DNA Cell Biol. 2014, 33, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Yoo, Y.-M.; Han, T.-Y.; Kim, H.S. Melatonin Suppresses Autophagy Induced by Clinostat in Preosteoblast Mc3t3-E1 Cells. Int. J. Mol. Sci. 2016, 17, 526. [Google Scholar] [CrossRef] [Green Version]
- Arun, R.; Sivanesan, D.; Vidyasekar, P.; Verma, R. Pten/Foxo3/Akt Pathway Regulates Cell Death and Mediates Morphogenetic Differentiation of Colorectal Cancer Cells under Simulated Microgravity. Sci. Rep. 2017, 7, 5952. [Google Scholar] [CrossRef]
- Jeong, A.J.; Kim, Y.J.; Lim, M.H.; Lee, H.; Noh, K.; Kim, B.H.; Chung, J.W.; Cho, C.H.; Kim, S.; Ye, S.K. Microgravity Induces Autophagy Via Mitochondrial Dysfunction in Human Hodgkin’s Lymphoma Cells. Sci. Rep. 2018, 8, 14646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukazawa, T.; Tanimoto, K.; Shrestha, L.; Imura, T.; Takahashi, S.; Sueda, T.; Hirohashi, N.; Hiyama, E.; Yuge, L. Simulated Microgravity Enhances Cddp-Induced Apoptosis Signal Via P53-Independent Mechanisms in Cancer Cells. PLoS ONE 2019, 14, e0219363. [Google Scholar] [CrossRef] [Green Version]
- Ferranti, F.; Caruso, M.; Cammarota, M.; Masiello, M.G.; Scheri, K.C.; Fabrizi, C.; Fumagalli, L.; Schiraldi, C.; Cucina, A.; Catizone, A.; et al. Cytoskeleton Modifications and Autophagy Induction in Tcam-2 Seminoma Cells Exposed to Simulated Microgravity. BioMed Res. Int. 2014, 2014, 904396. [Google Scholar] [CrossRef]
- Morabito, C.; Guarnieri, S.; Catizone, A.; Schiraldi, C.; Ricci, G.; Mariggiò, M. Transient Increases in Intracellular Calcium and Reactive Oxygen Species Levels in Tcam-2 Cells Exposed to Microgravity. Sci. Rep. 2017, 7, 15648. [Google Scholar] [CrossRef] [Green Version]
- Indo, H.P.; Tomiyoshi, T.; Suenaga, S.; Tomita, K.; Suzuki, H.; Masuda, D.; Terada, M.; Ishioka, N.; Gusev, O.; Cornette, R.; et al. Mnsod Downregulation Induced by Extremely Low 0.1 Mgy Single and Fractionated X-Rays and Microgravity Treatment in Human Neuroblastoma Cell Line, Nb-1. J. Clin. Biochem. Nutr. 2015, 57, 98–104. [Google Scholar] [CrossRef] [Green Version]
- Cho, H.-J.; Baek, M.-O.; Khaliq, S.A.; Chon, S.J.; Son, K.H.; Lee, S.H.; Yoon, M.-S. Microgravity inhibits Decidualization Via Decreasing Akt Activity and Foxo3a Expression in Human Endometrial Stromal Cells. Sci. Rep. 2019, 9, 12094. [Google Scholar] [CrossRef]
- Nabavi, N.; Khandani, A.; Camirand, A.; Harrison, R.E. Effects of Microgravity on Osteoclast Bone Resorption and Osteoblast Cytoskeletal Organization and Adhesion. Bone 2011, 49, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Boya, P.; Codogno, P.; Rodriguez-Muela, N. Autophagy in Stem Cells: Repair, Remodelling and Metabolic Reprogramming. Development 2018, 145, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carmeliet, G.; Bouillon, R. The Effect of Microgravity on Morphology and Gene Expression of Osteoblasts in Vitro. FASEB J. 1999, 13, 129–134. [Google Scholar] [CrossRef] [Green Version]
- Sibonga, J. Spaceflight-Induced Bone Loss: Is There an Osteoporosis Risk? Curr. Osteoporos. Rep. 2013, 11, 92–98. [Google Scholar] [CrossRef]
- Garber, M.A.; McDowell, D.L.; Hutton, W.C. Bone Loss During Simulated Weightlessness: A Biomechanical and Mineralization Study in the Rat Model. Aviat. Space Environ. Med. 2000, 71, 586–592. [Google Scholar]
- Lang, T.F.; Leblanc, A.D.; Evans, H.J.; Lu, Y. Adaptation of the Proximal Femur to Skeletal Reloading after Long-Duration Spaceflight. J. Bone Miner. Res. 2006, 21, 1224–1230. [Google Scholar] [CrossRef]
- Clément, G. The Musculo-Skeletal System in Space. In Fundamentals of Space Medicine; Springer: New York, NY, USA, 2011; Volume 23, pp. 181–216. [Google Scholar]
- Laurence, V.; Alan, H. Skeletal Changes During and after Spaceflight. Nat. Rev. Rheumatol. 2018, 14, 229–245. [Google Scholar]
- Lotinun, S.; Kiviranta, R.; Matsubara, T.; Alzate, J.A.; Neff, L.; Lüth, A.; Koskivirta, I.; Kleuser, B.; Vacher, J.; Vuorio, E.; et al. Osteoclast-Specific Cathepsin K Deletion Stimulates S1p-Dependent Bone Formation. J. Clin. Invesig. 2013, 123, 666–681. [Google Scholar] [CrossRef] [PubMed]
- Ducy, P.; Schinke, T.; Karsenty, G. The Osteoblast: A Sophisticated Fibroblast under Central Surveillance. Science 2000, 289, 1501–1504. [Google Scholar] [CrossRef]
- Teitelbaum, S.L. Bone Resorption by Osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef]
- Karsenty, G.; Kronenberg, H.M.; Settembre, C. Genetic Control of Bone Formation. Ann. Rev. Cell Dev. Biol. 2009, 25, 629–648. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Zaidi, M. Molecular Regulation of Mechanotransduction. Biochem. Biophys. Res. Commun. 2005, 328, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Spatz, J.M.; Fields, E.E.; Yu, E.W.; Pajevic, P.D.; Bouxsein, M.L.; Sibonga, J.D.; Zwart, S.R.; Smith, S.M. Serum Sclerostin Increases in Healthy Adult Men During Bed Rest. J. Clin. Endocrinol. Metab. 2012, 97, E1736–E1740. [Google Scholar] [CrossRef] [PubMed]
- Frings-Meuthen, P.; Boehme, G.; Liphardt, A.M.; Baecker, N.; Heer, M.; Rittweger, J. Sclerostin and Dkk1 Levels During 14 and 21 Days of Bed Rest in Healthy Young Men. J. Musculoskelet Neuronal. Interact. 2013, 13, 45–52. [Google Scholar]
- Li, X.; Zhang, Y.; Kang, H.; Liu, W.; Liu, P.; Zhang, J.; Harris, S.E.; Wu, D. Sclerostin Binds to Lrp5/6 and Antagonizes Canonical Wnt Signaling. J. Biol. Chem. 2005, 280, 19883–19887. [Google Scholar] [CrossRef] [Green Version]
- Spatz, J.M.; Wein, M.N.; Gooi, J.H.; Qu, Y.; Garr, J.L.; Liu, S.; Barry, K.J.; Uda, Y.; Lai, F.; Dedic, C.; et al. The Wnt Inhibitor Sclerostin Is up-Regulated by Mechanical Unloading in Osteocytes in Vitro. J. Biol. Chem. 2015, 290, 16744–16758. [Google Scholar] [CrossRef] [Green Version]
- Suen, P.K.; Qin, L. Sclerostin, an Emerging Therapeutic Target for Treating Osteoporosis and Osteoporotic Fracture: A General Review. J. Orthop. Translat. 2016, 4, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Whitty, C.; Wardale, R.J.; Henson, F.M.D. The Regulation of Sclerostin by Cathepsin K in Periodontal Ligament Cells. Biochem. Biophys. Res. Commun. 2018, 503, 550–555. [Google Scholar] [CrossRef]
- Shiraki, M.; Xu, X.; Iovanna, J.L.; Kukita, T.; Hirata, H.; Kamohara, A.; Kubota, Y.; Miyamoto, H.; Mawatari, M.; Kukita, A. Deficiency of Stress-Associated Gene Nupr1 Increases Bone Volume by Attenuating Differentiation of Osteoclasts and Enhancing Differentiation of Osteoblasts. FASEB J. 2019, 33, 8836–8852. [Google Scholar] [CrossRef] [Green Version]
- Gould, G.W.; Lippincott-Schwartz, J. New Roles for Endosomes: From Vesicular Carriers to Multi-Purpose Platforms. Nat. Rev. Mol. Cell Biol. 2009, 10, 287–292. [Google Scholar] [CrossRef] [Green Version]
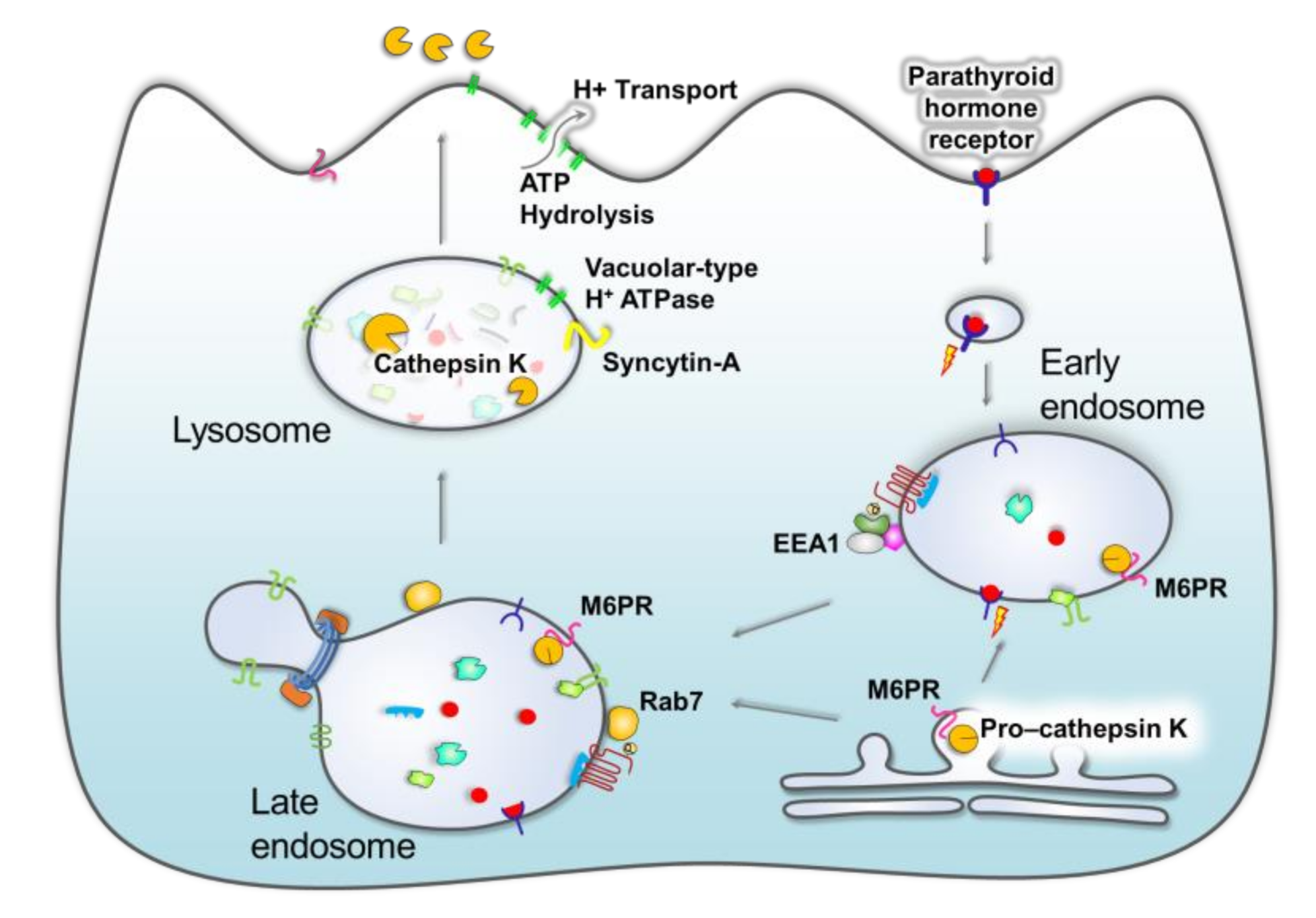
- Zhao, H. Membrane Trafficking in Osteoblasts and Osteoclasts: New Avenues for Understanding and Treating Skeletal Diseases. Traffic 2012, 13, 1307–1314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keusch, I.; Traebert, M.; Lotscher, M.; Kaissling, B.; Murer, H.; Biber, J. Parathyroid Hormone and Dietary Phosphate Provoke a Lysosomal Routing of the Proximal Tubular Na/Pi-Cotransporter Type Ii. Kidney Int. 1998, 54, 1224–1232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, W.C.; Samuel, H.G.; Jean-Pierre, V.; Thomas, J.G. Pth Receptor-1 Signalling—Mechanistic Insights and Therapeutic Prospects. Nat. Rev. Endocrinol. 2015, 11, 712–724. [Google Scholar]
- Smith, S.M.; McCoy, T.; Gazda, D.; Morgan, J.L.L.; Heer, M.; Zwart, S.R. Space Flight Calcium: Implications for Astronaut Health, Spacecraft Operations, and Earth. Nutrients 2012, 4, 2047–2068. [Google Scholar] [CrossRef] [Green Version]
- Iwase, S.; Nishimura, N.; Tanaka, K.; Mano, T. Effects of Microgravity on Human Physiology. In Beyond LEO—Human Health Issues for Deep Space Exploration; Reynolds, R.J., Ed.; IntechOpen: London, UK, 2020; pp. 1–22. [Google Scholar]
- Florencio-Silva, R.; Sasso, G.; Simoes, M.D.; Simoes, R.; Baracat, M.; Sasso-Cerri, E.; Cerri, P.S. Osteoporosis and Autophagy: What Is the Relationship? Rev. Assoc. Med. Bras. 2017, 63, 173–179. [Google Scholar] [CrossRef] [Green Version]
- Mulari, M.T.K.; Zhao, H.; Lakkakorpi, P.T.; Väänänen, H.K. Osteoclast Ruffled Border Has Distinct Subdomains for Secretion and Degraded Matrix Uptake. Traffic 2003, 4, 113–125. [Google Scholar] [CrossRef]
- Palokangas, H.; Mulari, M.; Väänänen, H.K. Endocytic Pathway from the Basal Plasma Membrane to the Ruffled Border Membrane in Bone-Resorbing Osteoclasts. J. Cell Sci. 1997, 110, 1767–1780. [Google Scholar]
- Van Loon, J.J.; Bervoets, D.J.; Burger, E.H.; Dieudonné, S.C.; Hagen, J.W.; Semeins, C.M.; Doulabi, B.Z.; Veldhuijzen, J.P. Decreased Mineralization and Increased Calcium Release in Isolated Fetal Mouse Long Bones under near Weightlessness. J. Bone Miner. Res. 1995, 10, 550–557. [Google Scholar] [CrossRef]
- Hughes-Fulford, M.; Lewis, M.L. Effects of Microgravity on Osteoblast Growth Activation. Exp. Cell Res. 1996, 224, 103–109. [Google Scholar] [CrossRef] [Green Version]
- Rucci, N.; Migliaccio, S.; Zani, B.M.; Taranta, A.; Teti, A. Characterization of the Osteoblast-Like Cell Phenotype under Microgravity Conditions in the Nasa-Approved Rotating Wall Vessel Bioreactor (Rwv). J. Cell. Biochem. 2002, 85, 167–179. [Google Scholar] [CrossRef]
- Lacombe, J.; Karsenty, G.; Ferron, M. Regulation of Lysosome Biogenesis and Functions in Osteoclasts. Cell Cycle 2013, 12, 2744–2752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blaber, E.; Sato, K.; Almeida, E.A.C. Stem Cell Health and Tissue Regeneration in Microgravity. Stem Cells Dev. 2014, 23 (Suppl 1), 73–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, H.; Laitala-Leinonen, T.; Parikka, V.; Vaananen, H.K. Downregulation of Small Gtpase Rab7 Impairs Osteoclast Polarization and Bone Resorption. J. Biol. Chem. 2001, 276, 39295–39302. [Google Scholar] [CrossRef] [Green Version]
- Clement, J. Gene Expression Microarrays in Microgravity Research: Toward the Identification of Major Space Genes. In Innovations of Biotechnology; Agbo, E.C., Ed.; IntechOpen: London, UK, 2012; pp. 319–348. [Google Scholar]
- Ethiraj, P.; Ottinger, A.M.; Singh, T.; Singh, A.; Haire, K.M.; Reddy, S.V. Proteasome Inhibition Suppress Microgravity Elevated Rank Signaling During Osteoclast Differentiation. Cytokine 2020, 125, 154821. [Google Scholar] [CrossRef]
- Nakamura, S.; Shigeyama, S.; Minami, S.; Shima, T.; Akayama, S.; Matsuda, T.; Esposito, A.; Napolitano, G.; Kuma, A.; Namba-Hamano, T.; et al. Lc3 Lipidation Is Essential for Tfeb Activation During the Lysosomal Damage Response to Kidney Injury. Nat. Cell Biol. 2020, 22, 1252–1263. [Google Scholar] [CrossRef] [PubMed]
- Sardiello, M.; Palmieri, M.; di Ronza, A.; Medina, D.L.; Valenza, M.; Gennarino, V.A.; Di Malta, C.; Donaudy, F.; Embrione, V.; Polishchuk, R.S.; et al. A Gene Network Regulating Lysosomal Biogenesis and Function. Science 2009, 325, 473–477. [Google Scholar] [CrossRef] [Green Version]
- Gao, M.; Gao, W.; Papadimitriou, J.M.; Zhang, C.; Gao, J.; Zheng, M. Exosomes-the Enigmatic Regulators of Bone Homeostasis. Bone Res. 2018, 6, 36. [Google Scholar] [CrossRef]
- Guo, H.; Chitiprolu, M.; Roncevic, L.; Javalet, C.; Hemming, F.J.; Trung, M.T.; Meng, L.; Latreille, E.; Tanese de Souza, C.; McCulloch, D.; et al. Atg5 Disassociates the V1v0-Atpase to Promote Exosome Production and Tumor Metastasis Independent of Canonical Macroautophagy. Dev. Cell 2017, 43, 716–730. [Google Scholar] [CrossRef] [Green Version]
- Gudbergsson, J.M.; Johnsen, K.B. Exosomes and Autophagy: Rekindling the Vesicular Waste Hypothesis. J. Cell. Commun. Signal. 2019, 13, 443–450. [Google Scholar] [CrossRef]
- Ralston, E.; Lu, Z.; Ploug, T. The Organization of the Golgi Complex and Microtubules in Skeletal Muscle Is Fiber Type-Dependent. J. Neurosci. 1999, 19, 10694–10705. [Google Scholar] [CrossRef] [Green Version]
- Grumati, P.; Coletto, L.; Sabatelli, P.; Cescon, M.; Angelin, A.; Bertaggia, E.; Blaauw, B.; Urciuolo, A.; Tiepolo, T.; Merlini, L.; et al. Autophagy Is Defective in Collagen Vi Muscular Dystrophies, and Its Reactivation Rescues Myofiber Degeneration. Nat. Med. 2010, 16, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Penna, F.; Costamagna, D.; Pin, F.; Camperi, A.; Fanzani, A.; Chiarpotto, E.M.; Cavallini, G.; Bonelli, G.; Baccino, F.M.; Costelli, P. Autophagic Degradation Contributes to Muscle Wasting in Cancer Cachexia. Am. J. Pathol. 2013, 182, 1367–1378. [Google Scholar] [CrossRef]
- Brocca, L.; Cannavino, J.; Coletto, L.; Biolo, G.; Sandri, M.; Bottinelli, R.; Pellegrino, M.A. The Time Course of the Adaptations of Human Muscle Proteome to Bed Rest and the Underlying Mechanisms. J. Physiol. 2012, 590, 5211–5230. [Google Scholar] [CrossRef] [PubMed]
- Hargens, A.R.; Bhattacharya, R.; Schneider, S.M. Space Physiology Vi: Exercise, Artificial Gravity, and Countermeasure Development for Prolonged Space Flight. Eur. J. Appl. Physiol. 2013, 113, 2183–2192. [Google Scholar] [CrossRef]
- Wang, X.; Blagden, C.; Fan, J.; Nowak, S.; Taniuchi, I.; Littman, D.; Burden, S. Runx1 Prevents Wasting, Myofibrillar Disorganization, and Autophagy of Skeletal Muscle. Genes Dev. 2005, 19, 1715–1722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stein, T.P.; Schluter, M.D. Excretion of Amino Acids by Humans During Space Flight. Acta Astronaut. 1998, 42, 205–214. [Google Scholar] [CrossRef]
- Fitts, R.H.; Trappe, S.W.; Costill, D.L.; Gallagher, P.M.; Creer, A.C.; Colloton, P.A.; Peters, J.R.; Romatowski, J.G.; Bain, J.L.; Riley, D.A. Prolonged Space Flight-Induced Alterations in the Structure and Function of Human Skeletal Muscle Fibres. J. Physiol. 2010, 588, 3567–3592. [Google Scholar] [CrossRef]
- Choi, S.Y.; Saravia-Butler, A.; Shirazi-Fard, Y.; Leveson-Gower, D.; Stodieck, L.S.; Cadena, S.M.; Beegle, J.; Solis, S.; Ronca, A.; Globus, R.K. Validation of a New Rodent Experimental System to Investigate Consequences of Long Duration Space Habitation. Sci. Rep. 2020, 10, 2336. [Google Scholar] [CrossRef] [Green Version]
- Hanna, R.A.; Quinsay, M.N.; Orogo, A.M.; Giang, K.; Rikka, S.; Gustafsson, A.B. Microtubule-Associated Protein 1 Light Chain 3 (Lc3) Interacts with Bnip3 Protein to Selectively Remove Endoplasmic Reticulum and Mitochondria Via Autophagy. J. Biol. Chem. 2012, 287, 19094–19104. [Google Scholar] [CrossRef] [Green Version]
- Romanello, V.; Sandri, M. Mitochondrial Biogenesis and Fragmentation as Regulators of Muscle Protein Degradation. Curr. Hypertens. Rep. 2010, 12, 433–439. [Google Scholar] [CrossRef]
- Zhao, T.; Tang, X.; Umeshappa, C.S.; Ma, H.; Gao, H.; Deng, Y.; Freywald, A.; Xiang, J. Simulated Microgravity Promotes Cell Apoptosis through Suppressing Uev1a/Ticam/Traf/Nf-Kappab-Regulated Anti-Apoptosis and P53/Pcna- and Atm/Atr-Chk1/2-Controlled DNA-Damage Response Pathways. J. Cell Biochem. 2016, 117, 2138–2148. [Google Scholar] [CrossRef]
- Ma, X.; Godar, R.J.; Liu, H.; Diwan, A. Enhancing Lysosome Biogenesis Attenuates Bnip3-Induced Cardiomyocyte Death. Autophagy 2012, 8, 297–309. [Google Scholar] [CrossRef] [Green Version]
- Mansueto, G.; Armani, A.; Viscomi, C.; D’Orsi, L.; De Cegli, R.; Polishchuk, E.V.; Lamperti, C.; Di Meo, I.; Romanello, V.; Marchet, S.; et al. Transcription Factor Eb Controls Metabolic Flexibility During Exercise. Cell Metab. 2017, 25, 182–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.; Chen, Y.; Li, X.; Zhang, W.; Liu, Z.; Wu, M.; Pan, Q.; Liu, H. Emerging Role of Transcription Factor Eb in Mitochondrial Quality Control. Biomed. Pharmacother. 2020, 128, 110272. [Google Scholar] [CrossRef] [PubMed]
- Milan, G.; Romanello, V.; Pescatore, F.; Armani, A.; Paik, J.H.; Frasson, L.; Seydel, A.; Zhao, J.; Abraham, R.; Goldberg, A.L.; et al. Regulation of Autophagy and the Ubiquitin-Proteasome System by the Foxo Transcriptional Network During Muscle Atrophy. Nat. Commun. 2015, 6, 6670. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Brault, J.J.; Schild, A.; Cao, P.; Sandri, M.; Schiaffino, S.; Lecker, S.H.; Goldberg, A.L. Foxo3 Coordinately Activates Protein Degradation by the Autophagic/Lysosomal and Proteasomal Pathways in Atrophying Muscle Cells. Cell Metab. 2007, 6, 472–483. [Google Scholar] [CrossRef] [Green Version]
- Ehmsen, J.T.; Hoke, A. Cellular and Molecular Features of Neurogenic Skeletal Muscle Atrophy. Exp. Neurol. 2020, 331, 113379. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Liu, H.; Li, W.; Wang, D. Intestine-Specific Activity of Insulin Signaling Pathway in Response to Microgravity Stress in Caenorhabditis Elegans. Biochem. Biophys. Res. Commun. 2019, 517, 278–284. [Google Scholar] [CrossRef]
- Sun, Y.; Li, M.; Zhao, D.; Li, X.; Yang, C.; Wang, X. Lysosome Activity Is Modulated by Multiple Longevity Pathways and Is Important for Lifespan Extension in C. Elegans. Elife 2020, 9, e55745. [Google Scholar] [CrossRef]
- Tharyan, R.G.; Annibal, A.; Schiffer, I.; Laboy, R.; Atanassov, I.; Weber, A.L.; Gerisch, B.; Antebi, A. Nfyb-1 Regulates Mitochondrial Function and Longevity Via Lysosomal Prosaposin. Nat. Metab. 2020, 2, 387–396. [Google Scholar] [CrossRef]
- Gustafsson, T.; Osterlund, T.; Flanagan, J.N.; von Walden, F.; Trappe, T.A.; Linnehan, R.M.; Tesch, P.A. Effects of 3 Days Unloading on Molecular Regulators of Muscle Size in Humans. J. Appl. Physiol. 2010, 109, 721–727. [Google Scholar] [CrossRef] [PubMed]
- Aston, D.; Capel, R.A.; Ford, K.L.; Christian, H.C.; Mirams, G.R.; Rog-Zielinska, E.A.; Kohl, P.; Galione, A.; Burton, R.A.; Terrar, D.A. High Resolution Structural Evidence Suggests the Sarcoplasmic Reticulum Forms Microdomains with Acidic Stores (Lysosomes) in the Heart. Sci. Rep. 2017, 7, 40620. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kinnear, N.P.; Boittin, F.X.; Thomas, J.M.; Galione, A.; Evans, A.M. Lysosome-Sarcoplasmic Reticulum Junctions. A Trigger Zone for Calcium Signaling by Nicotinic Acid Adenine Dinucleotide Phosphate and Endothelin-1. J. Biol. Chem. 2004, 279, 54319–54326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bootman, M.D.; Chehab, T.; Bultynck, G.; Parys, J.B.; Rietdorf, K. The Regulation of Autophagy by Calcium Signals: Do We Have a Consensus? Cell Calcium. 2018, 70, 32–46. [Google Scholar] [CrossRef]
- Ho, T.T.; Warr, M.R.; Adelman, E.R.; Lansinger, O.M.; Flach, J.; Verovskaya, E.V.; Figueroa, M.E.; Passegue, E. Autophagy Maintains the Metabolism and Function of Young and Old Stem Cells. Nature 2017, 543, 205–210. [Google Scholar] [CrossRef] [Green Version]
- Yamaguchi, Y.; Sakai, E.; Okamoto, K.; Kajiya, H.; Okabe, K.; Naito, M.; Kadowaki, T.; Tsukuba, T. Rab44, a Novel Large Rab Gtpase, Negatively Regulates Osteoclast Differentiation by Modulating Intracellular Calcium Levels Followed by Nfatc1 Activation. Cell Mol. Life Sci. 2018, 75, 33–48. [Google Scholar] [CrossRef]
- Erkhembaatar, M.; Gu, D.R.; Lee, S.H.; Yang, Y.M.; Park, S.; Muallem, S.; Shin, D.M.; Kim, M.S. Lysosomal Ca2+ Signaling Is Essential for Osteoclastogenesis and Bone Remodeling. J. Bone Miner. Res. 2017, 32, 385–396. [Google Scholar] [CrossRef]
- Villegas, F.; Lehalle, D.; Mayer, D.; Rittirsch, M.; Stadler, M.B.; Zinner, M.; Olivieri, D.; Vabres, P.; Duplomb-Jego, L.; De Bont, E.; et al. Lysosomal Signaling Licenses Embryonic Stem Cell Differentiation Via Inactivation of Tfe3. Cell Stem Cell. 2019, 24, 257–270. [Google Scholar] [CrossRef] [Green Version]
- Chang, N.C. Autophagy and Stem Cells: Self-Eating for Self-Renewal. Front. Cell Dev. Biol. 2020, 8, 138. [Google Scholar] [CrossRef] [Green Version]
- Zakrzewski, W.; Dobrzyński, M.; Szymonowicz, M.; Rybak, Z. Stem Cells: Past, Present, and Future. Stem Cell Res. Ther. 2019, 10, 68. [Google Scholar] [CrossRef]
- Grimm, D.; Wehland, M.; Corydon, T.J.; Richter, P.; Prasad, B.; Bauer, J.; Egli, M.; Kopp, S.; Lebert, M.; Krüger, M. The Effects of Microgravity on Differentiation and Cell Growth in Stem Cells and Cancer Stem Cells. Stem Cells Transl. Med. 2020, 9, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Kopp, S.; Warnke, E.; Wehland, M.; Aleshcheva, G.; Magnusson, N.E.; Hemmersbach, R.; Corydon, T.J.; Bauer, J.; Infanger, M.; Grimm, D. Mechanisms of Three-Dimensional Growth of Thyroid Cells During Long-Term Simulated Microgravity. Sci. Rep. 2015, 5, 16691. [Google Scholar] [CrossRef] [Green Version]
- White, E. Deconvoluting the Context-Dependent Role for Autophagy in Cancer. Nat. Rev. Cancer 2012, 12, 401–410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hynds, R.E.; Vladimirou, E.; Janes, S.M. The Secret Lives of Cancer Cell Lines. Dis. Model Mech. 2018, 11, dmm037366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pavlakou, P.; Dounousi, E.; Roumeliotis, S.; Eleftheriadis, T.; Liakopoulos, V. Oxidative Stress and the Kidney in the Space Environment. Int. J. Mol. Sci. 2018, 19, 3176. [Google Scholar] [CrossRef] [Green Version]
- Haigis, M.C.; Yankner, B.A. The Aging Stress Response. Mol. Cell 2010, 40, 333–344. [Google Scholar] [CrossRef]
- Limoli, C.L.; Giedzinski, E.; Rola, R.; Otsuka, S.; Palmer, T.D.; Fike, J.R. Radiation Response of Neural Precursor Cells: Linking Cellular Sensitivity to Cell Cycle Checkpoints, Apoptosis and Oxidative Stress. Radiat. Res. 2004, 161, 17–27. [Google Scholar] [CrossRef]
- Mavragani, I.V.; Nikitaki, Z.; Souli, M.P.; Aziz, A.; Nowsheen, S.; Aziz, K.; Rogakou, E.; Georgakilas, A.G. Complex DNA Damage: A Route to Radiation-Induced Genomic Instability and Carcinogenesis. Cancers 2017, 9, 91. [Google Scholar] [CrossRef]
- Spitz, D.R.; Hauer-Jensen, M. Ionizing Radiation-Induced Responses: Where Free Radical Chemistry Meets Redox Biology and Medicine. Antioxid. Redox. Signal. 2014, 20, 1407–1409. [Google Scholar] [CrossRef]
- Ishikawa, K.; Takenaga, K.; Akimoto, M.; Koshikawa, N.; Yamaguchi, A.; Imanishi, H.; Nakada, K.; Honma, Y.; Hayashi, J. Ros-Generating Mitochondrial DNA Mutations Can Regulate Tumor Cell Metastasis. Science 2008, 320, 661–664. [Google Scholar] [CrossRef] [Green Version]
- Stein, T.P. Space Flight and Oxidative Stress. Nutrition 2002, 18, 867–871. [Google Scholar] [CrossRef]
- Yamamori, T.; Yasui, H.; Yamazumi, M.; Wada, Y.; Nakamura, Y.; Nakamura, H.; Inanami, O. Ionizing Radiation Induces Mitochondrial Reactive Oxygen Species Production Accompanied by Upregulation of Mitochondrial Electron Transport Chain Function and Mitochondrial Content under Control of the Cell Cycle Checkpoint. Free Radic. Biol. Med. 2012, 53, 260–270. [Google Scholar] [CrossRef] [Green Version]
- Versari, S.; Longinotti, G.; Barenghi, L.; Maier, J.A.; Bradamante, S. The Challenging Environment on Board the International Space Station Affects Endothelial Cell Function by Triggering Oxidative Stress through Thioredoxin Interacting Protein Overexpression: The Esa-Sphinx Experiment. FASEB J. 2013, 27, 4466–4475. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.; An, L.; Fan, Y.; Hang, H.; Wang, S. Simulated Microgravity Potentiates Generation of Reactive Oxygen Species in Cells. Biophy. Rep. 2016, 2, 100–105. [Google Scholar] [CrossRef] [Green Version]
- Yun, H.R.; Jo, Y.H.; Kim, J.; Shin, Y.; Kim, S.S.; Choi, T.G. Roles of Autophagy in Oxidative Stress. Int. J. Mol. Sci. 2020, 21, 3289. [Google Scholar] [CrossRef]
- Cao, Q.; Yang, Y.; Zhong, X.Z.; Dong, X.-P. The Lysosomal Ca Release Channel Trpml1 Regulates Lysosome Size by Activating Calmodulin. J. Biol. Chem. 2017, 292, 8424–8435. [Google Scholar] [CrossRef] [Green Version]
- Todkar, K.; Ilamathi, H.S.; Germain, M. Mitochondria and Lysosomes: Discovering Bonds. Front.Cell Dev. Biol. 2017, 5, 106. [Google Scholar] [CrossRef] [Green Version]
- Ushio-Fukai, M. Localizing Nadph Oxidase-Derived Ros. Sci. STKE 2006, 2006, re8. [Google Scholar] [CrossRef] [PubMed]
- Davis Volk, A.P.; Moreland, J.G. Ros-Containing Endosomal Compartments: Implications for Signaling. Methods Enzymol. 2014, 535, 201–224. [Google Scholar]
- Li, L.; Tan, J.; Miao, Y.; Lei, P.; Zhang, Q. Ros and Autophagy: Interactions and Molecular Regulatory Mechanisms. Cell Mol. Neurobiol. 2015, 35, 615–621. [Google Scholar] [CrossRef]
- Harrison, I.P.; Vinh, A.; Johnson, I.R.D.; Luong, R.; Drummond, G.R.; Sobey, C.G.; Tiganis, T.; Williams, E.D.; O’Leary, J.J.; Brooks, D.A.; et al. Nox2 Oxidase Expressed in Endosomes Promotes Cell Proliferation and Prostate Tumour Development. Oncotarget 2018, 9, 35378–35393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsubata, T. Involvement of Reactive Oxygen Species (Ros) in Bcr Signaling as a Second Messenger. Adv. Exp. Med. Biol. 2020, 1254, 37–46. [Google Scholar]
- Karunakaran, U.; Elumalai, S.; Moon, J.S.; Won, K.C. Cd36 Dependent Redoxosomes Promotes Ceramide-Mediated Pancreatic Beta-Cell Failure Via P66shc Activation. Free Radic. Biol. Med. 2019, 134, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Stein, T.P.; Schulter, M.D.; Boden, G. Development of Insulin Resistance by Astronauts During Spaceflight. Aviat. Space Environ. Med. 1994, 65, 1091–1096. [Google Scholar] [PubMed]
- Hughson, R.L.; Robertson, A.D.; Arbeille, P.; Shoemaker, J.K.; Rush, J.W.; Fraser, K.S.; Greaves, D.K. Increased Postflight Carotid Artery Stiffness and Inflight Insulin Resistance Resulting from 6-Mo Spaceflight in Male and Female Astronauts. Am. J. Physiol. Heart Circ. Physiol. 2016, 310, H628–H638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tiganis, T. Reactive Oxygen Species and Insulin Resistance: The Good, the Bad and the Ugly. Trends Pharmacol. Sci. 2011, 32, 82–89. [Google Scholar] [CrossRef]
- Mann, V.; Sundaresan, A.; Mehta, S.K.; Crucian, B.; Doursout, M.F.; Devakottai, S. Effects of Microgravity and Other Space Stressors in Immunosuppression and Viral Reactivation with Potential Nervous System Involvement. Neurol. India 2019, 67, 198–203. [Google Scholar]
- Blaber, E.A.; Dvorochkin, N.; Torres, M.L.; Yousuf, R.; Burns, B.P.; Globus, R.K.; Almeida, E.A.C. Mechanical Unloading of Bone in Microgravity Reduces Mesenchymal and Hematopoietic Stem Cell-Mediated Tissue Regeneration. Stem Cell Res. 2014, 13, 181–201. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Kim, H.R.; Quinley, C.; Kim, J.; Gonzalez-Navajas, J.; Xavier, R.; Raz, E. Autophagy Suppresses Interleukin-1β (Il-1β) Signaling by Activation of P62 Degradation Via Lysosomal and Proteasomal Pathways. J. Biol. Chem. 2012, 287, 4033–4040. [Google Scholar] [CrossRef] [Green Version]
- Nedergaard, A.; Karsdal, M.A.; Sun, S.; Henriksen, K. Serological Muscle Loss Biomarkers: An Overview of Current Concepts and Future Possibilities. J. Cachexia Sarcopenia Muscle 2013, 4, 1–17. [Google Scholar] [CrossRef]
- Diebold, B.A.; Smith, S.M.E.; Li, Y.; Lambeth, J.D. Nox2 as a Target for Drug Development: Indications, Possible Complications, and Progress. Antioxid. Redox Signal. 2015, 23, 375–405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mao, X.W.; Nishiyama, N.C.; Pecaut, M.J.; Campbell-Beachler, M.; Gifford, P.; Haynes, K.E.; Becronis, C.; Gridley, D.S. Simulated Microgravity and Low-Dose/Low-Dose-Rate Radiation Induces Oxidative Damage in the Mouse Brain. Radiat. Res. 2016, 185, 647–657. [Google Scholar] [CrossRef]
- Corydon, T.J.; Kopp, S.; Wehland, M.; Braun, M.; Schutte, A.; Mayer, T.; Hulsing, T.; Oltmann, H.; Schmitz, B.; Hemmersbach, R.; et al. Alterations of the Cytoskeleton in Human Cells in Space Proved by Life-Cell Imaging. Sci. Rep. 2016, 6, 20043. [Google Scholar] [CrossRef] [Green Version]
- Thiel, C.S.; Tauber, S.; Lauber, B.; Polzer, J.; Seebacher, C.; Uhl, R.; Neelam, S.; Zhang, Y.; Levine, H.; Ullrich, O. Rapid Morphological and Cytoskeletal Response to Microgravity in Human Primary Macrophages. Int. J. Mol. Sci. 2019, 20, 2402. [Google Scholar] [CrossRef] [Green Version]
- Thiel, C.S.; Tauber, S.; Seebacher, C.; Schropp, M.; Uhl, R.; Lauber, B.; Polzer, J.; Neelam, S.; Zhang, Y.; Ullrich, O. Real-Time 3d High-Resolution Microscopy of Human Cells on the International Space Station. Int. J. Mol. Sci. 2019, 20, 2033. [Google Scholar] [CrossRef] [Green Version]
| Physiological, Protein, Gene or Organelle Change | Effect | Organism/Model System | Radiation or Simulated/Real µg | Ref. |
|---|---|---|---|---|
| Lysosomes, phagosomes | Increased | Purkinje cells, Rat | Radiation (291 Mev/nucleon, 100–300/mm2) | [26] |
| Autophagosomes | Increased | Myocardia, Rat | Microgravity (Real) | [22] |
| Lysosomal activity | Elevated | Bone | Microgravity (Real) | [27] |
| Autophagic vacuoles, endocytic vesicles, lysosomes | Increased number, size | Salivary gland, Rat & Mouse | Microgravity (Real) | [23,28] |
| Lysosomal tripeptidyl peptidase | Altered distribution | Skeletal muscle, Rat | Microgravity (Real) | [29] |
| Sarcoplasmic reticulum (SR) | Localise to myofibril disruptions – may release Ca2+, may induce autophagy | |||
| Glutathione | Decreased | Liver, mouse | Microgravity (Real) | [19] |
| Lipid membrane metabolism, endocytosis, inflammatory pathways | Enriched | |||
| MuRF-1, MAFbx | Increased expression | Soleus and EDL muscle, mice | Microgravity (Real) | [30] |
| LC3β, CTSL | Unchanged | |||
| IGF-1 and IL-6 | Increased in EDL, Decreased in Soleus | |||
| c-Fos | Upregulated | Supraoptic neurons, Rat | Microgravity (Real) | [31] |
| Vasopressin | Downregulated | |||
| Autophagosomes, lysosome-like bodies | Increased | Osteocytes and Soleus myotendinous junction, Rhesus monkey | Microgravity (Real) | [32,33] |
| Lactate dehydrogenase type A | Mono-ubiquitinated | Myoblastic L6 cells, Rat | Microgravity (Simulated, tail suspension) | [34] |
| Ubiquitinated protein & MAFbx, Murf-1, Nedd4, XIAP | Upregulated expression | Soleus Muscle, Rat | Microgravity (Simulated, hindlimb suspension) | [35,36,37] |
| Glucose metabolism | Increased response upon muscle atrophy | |||
| Proteolysis | Increased (Ca2+ regulated from SR?) | |||
| Lysosomal and Ca2+-dependent proteolysis | Enhanced activity | |||
| Cathepsin B, D, L | Upregulated expression | |||
| N-acetylglucosaminidase (lysosomal) | Increased activity | Gastrocnemius & soleus muscle, CD1 mice | Microgravity (Simulated, hindlimb suspension) | [38,39] |
| Bcl-2, Bax | Increased expression ratio | |||
| Vps34, Beclin-1, Cathepsin D | Upregulated | Cardiac tissue, rat | Microgravity (Simulated, tail suspension) | [40] |
| p62 | Downregulated | |||
| mTOR | Decreased phosphorylation (s2448) | |||
| Ca2+ | Oscillates | HEK293, Myoblast C2C12, Mouse bone marrow, RAW 264.7 osteoclast progenitor, MC3T3-E1, HepG2, colorectal, HUVEC, L-540, HDLM-2 cell lines | Microgravity (Simulated, clinostat & RWV) | [4,5,41,42,43,44,45,46,47] |
| LC3 +ve autophagosomes | Increased puncta (72 h) | |||
| LC3-II | Increased conversion from LC3-I | |||
| AMPK, T172, ULK1, ATF4, Beclin-1 | Increased phosphorylation | |||
| AMPK S485 | Reduced phosphorylation at 24 h only | |||
| ATG12, ATG5, ATP1A1, ATP5A1, Bcl-2, MnSOD, Cu/ZnSOD, Mcl-1, mTOR, Rab7, GPCR, P58 | Downregulated | |||
| ATG14, ATG16L1, ATG4B, ATG7, ATG5, Bax, Beclin-1, Ire1α, LC3, MnSOD, p62, Syncytin-A Tnfsf10, ULK1, MITF, s100a8, CREB, CXCL4 | Upregulated | |||
| P62 | Reduced at 72h | |||
| ROS | Elevated | |||
| Cytoskeleton | Temporary collapse of microvilli at 24 h & altered microtubule/actin remodelling | TCam-2 seminoma cell line | Microgravity (Simulated, RPM) | [48,49] |
| LC3 +ve autophagosomes | Increased | |||
| LC3-II | Increased conversion from LC3-I | |||
| Ca2+, ROS | Elevated temporarily at 24 h | |||
| MnSOD, Casp9, BECN1 | Downregulated | NB-1 neuroblastoma cell line | X-ray, Microgravity (Simulated, clinostat) | [50] |
| VDAC2 | Upregulated | |||
| Vps15, Atg14L, Beclin1, p62, FOXO3A | Downregulated | Endometrial stromal cells | Microgravity (Simulated, RPM) | [51] |
| LC3BII | Decreased |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, I.R.D.; Nguyen, C.T.; Wise, P.; Grimm, D. Implications of Altered Endosome and Lysosome Biology in Space Environments. Int. J. Mol. Sci. 2020, 21, 8205. https://doi.org/10.3390/ijms21218205
Johnson IRD, Nguyen CT, Wise P, Grimm D. Implications of Altered Endosome and Lysosome Biology in Space Environments. International Journal of Molecular Sciences. 2020; 21(21):8205. https://doi.org/10.3390/ijms21218205
Chicago/Turabian StyleJohnson, Ian R. D., Catherine T. Nguyen, Petra Wise, and Daniela Grimm. 2020. "Implications of Altered Endosome and Lysosome Biology in Space Environments" International Journal of Molecular Sciences 21, no. 21: 8205. https://doi.org/10.3390/ijms21218205







