From Grafts to Human Bioengineered Vascularized Skin Substitutes
Abstract
:1. Introduction
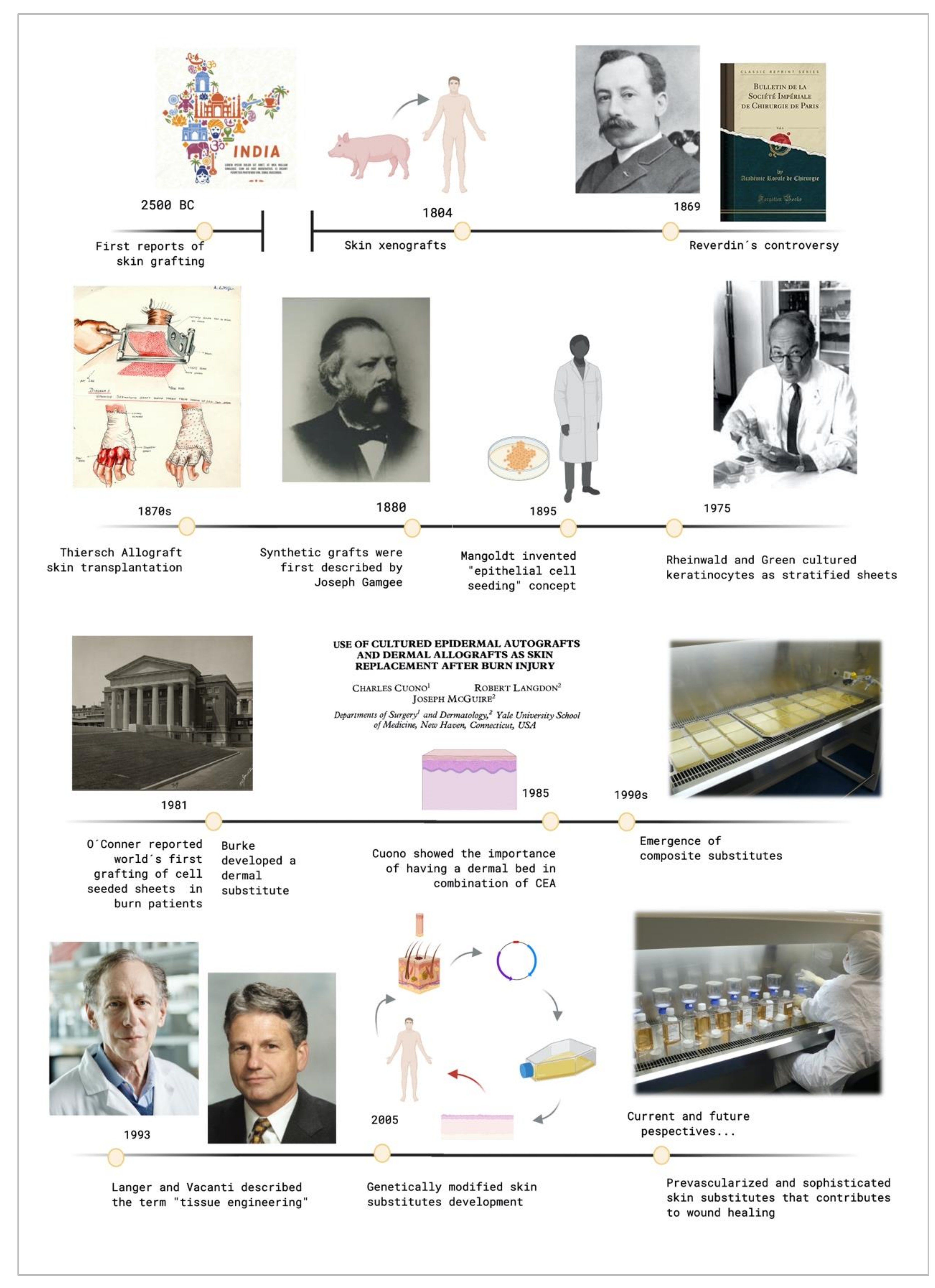
2. Chronological Review
3. Clinical Demands for Bioengineered Artificial Skin Substitutes (BASS)
3.1. Ethical and Legal Requirements
3.2. Protective Functions and Resistance to Infection
3.3. Biological Functions
3.4. Able to Prevent Water Loss and Water Accumulation
3.5. Adhesiveness
3.6. Low Antigenicity
3.7. Conform to Irregular Wound Surfaces
3.8. Withstand Shear Forces and Mechanical Tensions
3.9. Hypoxia Tolerant
3.10. Dermal and Epidermal Components
3.11. Easy to Prepare
3.12. Long Shelf Life and Easily Storage
3.13. Suitable Cost/Effectiveness
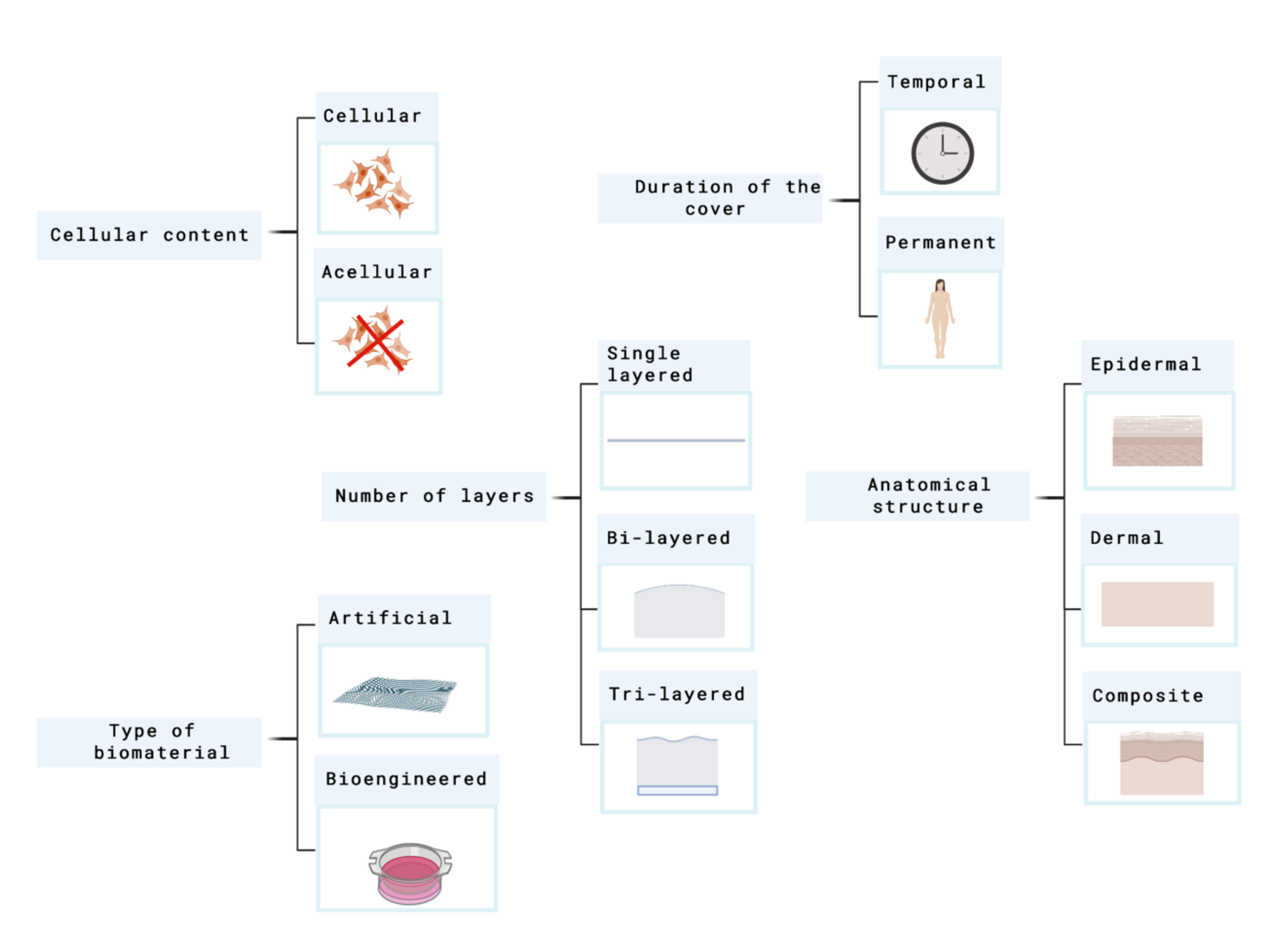
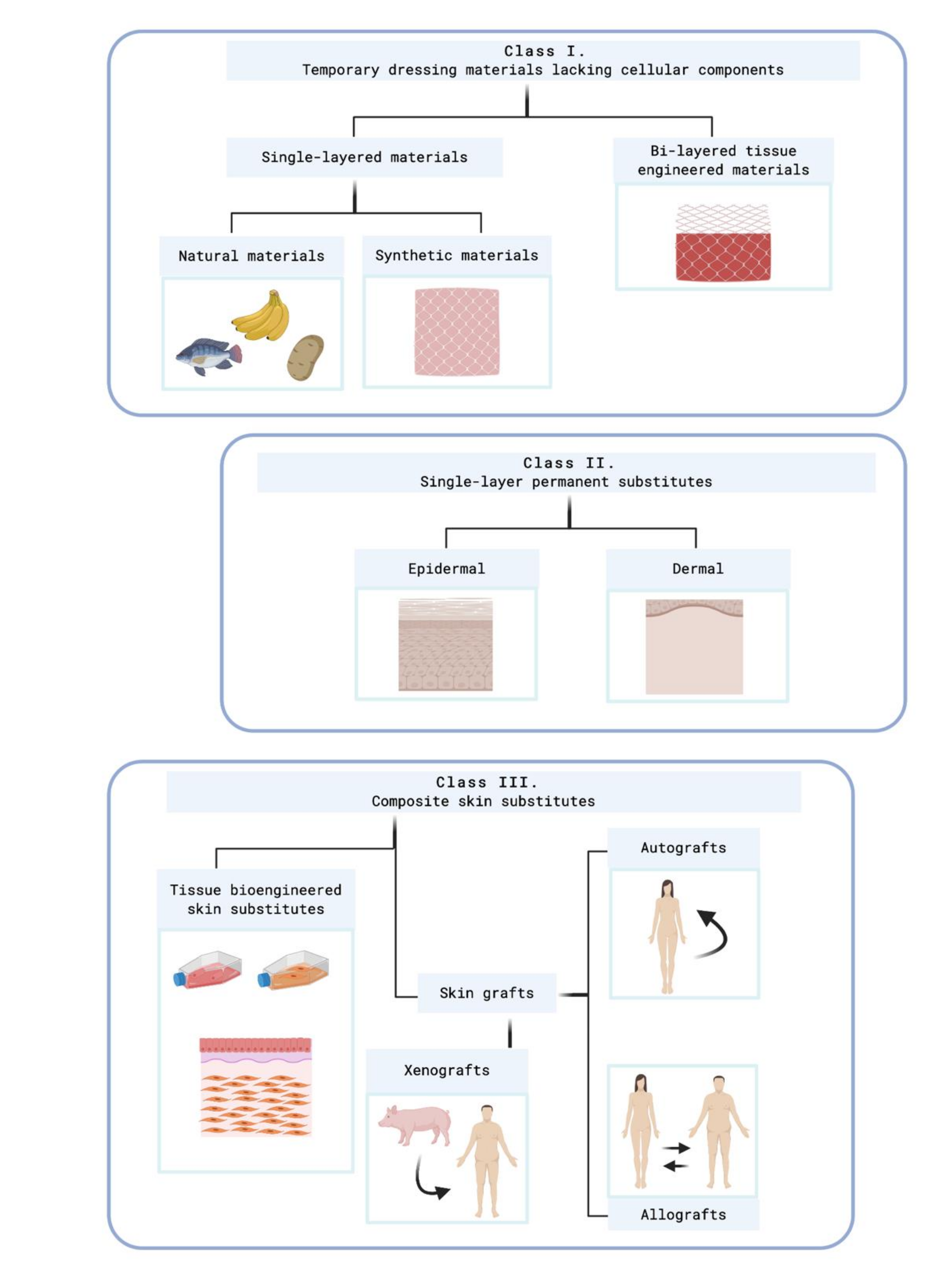
4. Skin Substitutes Classification
- I.
- Cultured epidermal substitutes
- II.
- Dermal components coming from skin or extracellular matrix (ECM) components
- III.
- Substitutes that include both dermal and epidermal components.
Temporary Skin Substitutes
5. Commercially Available Skin Substitutes
6. Trilayered Skin Substitutes
6.1. Importance of Vascularization in Wound Healing
6.2. Strategies for Vascularization
6.2.1. Angiogenic Strategies
6.2.2. Prevascularization Strategies
7. Future Perspectives
Funding
Conflicts of Interest
Abbreviations
| Ad-MVF | Adipose-tissue Derived Microvascular Fragments |
| AECs | Amniotic Epithelial Cells |
| AMCs | Amniotic Mesenchymal Cells |
| ASC | Adipose Stromal Cells |
| ATRA | All-trans Retinoic Acid |
| ATMPs | Advanced Therapy Medicinal Products |
| AV-loop | Arteriovenous Loop |
| BASS | Bioengineered Artificial Skin Substitutes |
| BM-MSC | Bone Marrow Mesenchymal Stem Cells |
| CEA | Cultured Epidermal Autograft |
| DCs | Dendritic Cells |
| DFUs | Diabetic Foot Ulcers |
| dsASCs | Discarded Skin Adipose Stromal Cells |
| GCP | Good Clinical Practice |
| GMP | Good Manufacturing Practice |
| EC | Endothelial Cells |
| ECM | Extracellular Matrix |
| EMA | European Medicines Agency |
| EPCs | Endothelial Progenitor Cells |
| FDA | Food and Drug Administration |
| FDCA | Food, Drug and Cosmetic Act |
| FGF | Fibroblasts Growth Factor |
| HAM | Human Amniotic Membrane |
| HBOEC | Human Blood Outgrowth Endothelial Cells |
| HDMEC | Human Dermal Microvascular Endothelial Cells |
| HUVEC | Human Umbilical Vein Endothelial Cells |
| IL | Interleukin |
| IRB | International Review Board |
| MHC | Major Histocompatibility Complex |
| NK | Natural Killer |
| PEG | Polyethyleneglycol |
| PHSA | Public Health Services Act |
| STSG | Split-Thickness Skin Grafts |
| SVF | Stromal Vascular Fraction |
| TBSA | Total Body Surface Area |
| TGF | Transformant Growth Factor |
| VEGF | Vascular Endothelial Growth Factor |
| VLUs | Venous Leg Ulcers |
References
- Peck, M.D. Epidemiology of burns throughout the world. Part I: Distribution and risk factors. Burns 2011, 37, 1087–1100. [Google Scholar] [CrossRef] [PubMed]
- Langer, R.; Vacanti, J.P. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langer, R.S.; Vacanti, J.P. Tissue engineering: The challenges ahead. Sci. Am. 1999, 280, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Rheinwald, J.G. Human epidermal keratinocyte cell culture and xenograft systems: Applications in the detection of potential chemical carcinogens and the study of epidermal transformation. Prog. Clin. Biol. Res. 1989, 298, 113. [Google Scholar] [PubMed]
- Berchialla, P.; Gangemi, E.N.; Foltran, F.; Haxhiaj, A.; Buja, A.; Lazzarato, F.; Stella, M.; Gregori, D. Predicting severity of pathological scarring due to burn injuries: A clinical decision making tool using Bayesian networks. Int. Wound J. 2014, 11, 246–252. [Google Scholar] [CrossRef] [Green Version]
- Cole-King, A.; Harding, K.G. Psychological factors and delayed healing in chronic wounds. Psychosom. Med. 2001, 63, 216–220. [Google Scholar] [CrossRef] [Green Version]
- Damanhuri, M.; Boyle, J.; Enoch, S. Advances in tissue-engineered skin substitutes. Wounds Int. 2011, 2, 27–34. [Google Scholar]
- Davis, J.S. The story of plastic surgery. Ann. Surg. 1941, 113, 641–656. [Google Scholar] [CrossRef]
- Leigh, I.; Lane, E.; Watt, F. The Keratinocyte Handbook; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Rheinwatd, J.G.; Green, H. Seria cultivation of strains of human epidemal keratinocytes: The formation keratinizin colonies from single cell is. Cell 1975, 6, 331–343. [Google Scholar] [CrossRef]
- O’Connor, N.E.; Mulliken, J.B.; Banks-Schlegel, S.; Kehinde, O.; Green, H. Grafting of burns with cultured epithelium prepared from autologous epidermal cells. Lancet 1981. [Google Scholar] [CrossRef]
- Green, H. The birth of therapy with cultured cells. BioEssays 2008, 30, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Gallico, G.G.; O’connor, N.E.; Compton, C.C.; Kehinde, O.; Green, H. Permanent Coverage of Large Burn Wounds with Autologous Cultured Human Epithelium. N. Engl. J. Med. 1984, 311, 448–451. [Google Scholar] [CrossRef]
- Burke, J.F.; Yannas, O.V.; Quinby, W.C.; Bondoc, C.C.; Jung, W.K. Successful use of a physiologically acceptable artificial skin in the treatment of extensive burn injury. Ann. Surg. 1981, 194, 413–427. [Google Scholar] [CrossRef] [PubMed]
- Cuono, C.; Langdon, R.; McGuire, J. Use of cultured epidermal autografts and dermal allografts as skin replacement after burn injury. Lancet 1986. [Google Scholar] [CrossRef]
- Nave, M. Wound bed preparation: Approaches to replacement of dermis. J. Burn Care Rehabil. 1992, 13, 147–153. [Google Scholar] [CrossRef]
- Hickerson, W.; Compton, C.; Fletchall, S.; Smith, L. Cultured epidermal autografts and allodermis combination for permanent burn wound coverage. Burns 1994, 20, S52–S56. [Google Scholar] [CrossRef]
- Abdel-Sayed, P.; Hirt-Burri, N.; De Buys Roessingh, A.; Raffoul, W.; Applegate, L.A. Evolution of Biological Bandages as First Cover for Burn Patients. Adv. Wound Care 2019, 8, 555–564. [Google Scholar] [CrossRef]
- European Parliament and the Council of the European Union. Regulation (ec) no 1394/2007 of the European Parliament and of the Council of 13 November 2007 on advanced therapy medicinal products and amending Directive 2001/83/EC and Regulation (EC) No 726/2004. Off. J. 2007, 324, 121–137. [Google Scholar]
- Iglesias-López, C.; Agustí, A.; Obach, M.; Vallano, A. Regulatory framework for advanced therapy medicinal products in Europe and United States. Front. Pharmacol. 2019, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.H.; Au, P.; Hyde, J.; Gacchina Johnson, C.; Heidaran, M.; Karandish, S.; Boxer, L.; Mendicino, M.; Yoon, D.; Tull, L.; et al. Translation of Regenerative Medicine Products Into the Clinic in the United States: FDA Perspective. Transl. Regen. Med. 2014, 49–74. [Google Scholar] [CrossRef]
- Van Schothorst, M.; Weeda, J.; Schiffers, K.; Oortwijn, W.; Hoekman, J.; Coppens, D.; De Bruin, M. Study on the Regulation of Advanced Therapies in Selected Jurisdictions; European Commission: Ispra, Italy, 2016; ISBN 9789292007317. [Google Scholar]
- Papadaki, M. Adaptation through collaboration: Developing novel platforms to advance the delivery of advanced therapies to patients. Front. Med. 2017, 4, 1–12. [Google Scholar] [CrossRef] [Green Version]
- D’Avignon, L.C.; Hogan, B.K.; Murray, C.K.; Loo, F.L.; Hospenthal, D.R.; Cancio, L.C.; Kim, S.H.; Renz, E.M.; Barillo, D.; Holcomb, J.B.; et al. Contribution of bacterial and viral infections to attributable mortality in patients with severe burns: An autopsy series. Burns 2010. [Google Scholar] [CrossRef]
- Mann, E.A.; Baun, M.M.; Meininger, J.C.; Wade, C.E. Comparison of mortality associated with sepsis in the Burn, trauma, and general intensive care unit patient: A systematic review of the literature. Shock 2012, 37, 4–16. [Google Scholar] [CrossRef] [Green Version]
- Nicholas, M.N.; Yeung, J. Current status and future of skin substitutes for chronic wound healing. J. Cutan. Med. Surg. 2017, 21, 23–30. [Google Scholar] [CrossRef]
- Van der Veen, V.C.; van der Wal, M.B.A.; van Leeuwen, M.C.E.; Ulrich, M.M.W.; Middelkoop, E. Biological background of dermal substitutes. Burns 2010, 36, 305–321. [Google Scholar] [CrossRef]
- Woodroof, E.A. The search for an ideal temporary skin substitute: AWBAT. Eplasty 2009, 9, e10. [Google Scholar]
- Namdar, T.; Stollwerck, P.L.; Stang, F.H.; Siemers, F.; Mailänder, P.; Lange, T. Transdermal fluid loss in severely burned patients. GMS Ger. Med. Sci. 2010. [Google Scholar] [CrossRef]
- MacNeil, S. Progress and opportunities for tissue-engineered skin. Nature 2007, 445, 874–880. [Google Scholar] [CrossRef]
- Biedermann, T.; Boettcher-Haberzeth, S.; Reichmann, E. Tissue engineering of skin for wound coverage. Eur. J. Pediatr. Surg. 2013, 23, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Egea-Guerrero, J.J.; Carmona, G.; Correa, E.; Mata, R.; Arias-Santiago, S.; Alaminos, M.; Gacto, P.; Cuende, N. Transplant of Tissue-Engineered Artificial Autologous Human Skin in Andalusia: An Example of Coordination and Institutional Collaboration. Transplant. Proc. 2019, 51, 3047–3050. [Google Scholar] [CrossRef]
- Carriel, V.; Garzón, I.; Jiménez, J.M.; Oliveira, A.C.X.; Arias-Santiago, S.; Campos, A.; Sánchez-Quevedo, M.C.; Alaminos, M. Epithelial and stromal developmental patterns in a novel substitute of the human skin generated with fibrin-agarose biomaterials. Cells Tissues Organs 2012, 196, 1–12. [Google Scholar] [CrossRef]
- Sierra-Sánchez, Á.; Fernández-González, A.; Lizana-Moreno, A.; Espinosa-Ibáñez, O.; Martinez-Lopez, A.; Guerrero-Calvo, J.; Fernández-Porcel, N.; Ruiz-García, A.; Ordóñez-Luque, A.; Carriel, V.; et al. Hyaluronic acid biomaterial for human tissue-engineered skin substitutes: Preclinical comparative in vivo study of wound healing. J. Eur. Acad. Dermatol. Venereol. 2020, 34, 2414–2427. [Google Scholar] [CrossRef]
- Yildirimer, L.; Thanh, N.T.K.; Seifalian, A.M. Skin regeneration scaffolds: A multimodal bottom-up approach. Trends Biotechnol. 2012, 30, 638–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balasubramani, M.; Kumar, T.R.; Babu, M. Skin substitutes: A review. Burns 2001, 27, 534–544. [Google Scholar] [CrossRef]
- Kumar, P. Classification of skin substitutes. Burns 2008, 34, 148–149. [Google Scholar] [CrossRef] [PubMed]
- Hermans, M.H.E. Porcine xenografts vs. (cryopreserved) allografts in the management of partial thickness burns: Is there a clinical difference? Burns 2014. [Google Scholar] [CrossRef]
- Borda, L.J.; Macquhae, F.E.; Kirsner, R.S. Wound Dressings: A Comprehensive Review. Curr. Dermatol. Rep. 2016, 5, 287–297. [Google Scholar] [CrossRef]
- Halim, A.S.; Khoo, T.L.; Shah, S.J. Biologic and synthetic skin substitutes: An overview. Indian J. Plast. Surg. 2010, 43, S23. [Google Scholar] [CrossRef]
- Wu, Q.; Tao, F.; Hongxin, L.; Chen, M.; Shi, P.; Xining, P.; Qi, G. Comparison of the proliferation, migration and angiogenic properties of human amniotic epithelial and mesenchymal stem cells and their effects on endothelial cells. Int. J. Mol. Med. 2017. [Google Scholar] [CrossRef] [Green Version]
- Litwiniuk, M.; Grzela, T. Amniotic membrane: New concepts for an old dressing. Wound Repair Regen. 2014, 22, 451–456. [Google Scholar] [CrossRef]
- Gore, M.A.; Akolekar, D. Evaluation of banana leaf dressing for partial thickness burn wounds. Burns 2003. [Google Scholar] [CrossRef]
- Hu, Z.; Yang, P.; Zhou, C.; Li, S.; Hong, P. Marine collagen peptides from the skin of Nile Tilapia (Oreochromis niloticus): Characterization and wound healing evaluation. Mar. Drugs 2017, 15, 102. [Google Scholar] [CrossRef] [PubMed]
- Zaulyanov, L.; Kirsner, R.S. A review of a bi-layered living cell treatment (Apligraf) in the treatment of venous leg ulcers and diabetic foot ulcers. Clin. Interv. Aging 2007, 2, 93–98. [Google Scholar] [CrossRef]
- Vanscheidt, W.; Ukat, A.; Horak, V.; Brüning, H.; Hunyadi, J.; Pavlicek, R.; Emter, M.; Hartmann, A.; Bende, J.; Zwingers, T.; et al. Treatment of recalcitrant venous leg ulcers with autologous keratinocytes in fibrin sealant: A multinational randomized controlled clinical trial. Wound Repair Regen. 2007. [Google Scholar] [CrossRef]
- Hilmi, A.B.M. Vital roles of stem cells and biomaterials in skin tissue engineering. World J. Stem Cells 2015. [Google Scholar] [CrossRef]
- Hart, C.E.; Loewen-Rodriguez, A.; Lessem, J. Dermagraft: Use in the Treatment of Chronic Wounds. Adv. Wound Care 2012. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naughton, G.; Mansbridge, J.; Gentzkow, G. A Metabolically Active Human Dermal Replacement for the Treatment of Diabetic Foot Ulcers. Artif. Organs 2008. [Google Scholar] [CrossRef]
- Tausche, A.K.; Skaria, M.; Böhlen, L.; Liebold, K.; Hafner, J.; Friedlein, H.; Meurer, M.; Goedkoop, R.J.; Wollina, U.; Salomon, D.; et al. An autologous epidermal equivalent tissue-engineered from follicular outer root sheath keratinocytes is as effective as split-thickness skin autograft in recalcitrant vascular leg ulcers. Wound Repair Regen. 2003. [Google Scholar] [CrossRef] [PubMed]
- Acher-Chenebaux, A.; Maillard, H.; Potier, A.; Nzeyimana, H.; Cazals, F.; Celerier, P. Cutaneous calciphylaxis treated by autologous keratinocytes graft and subtotal parathyroidectomy. Ann. Dermatol. Venereol. 2006. [Google Scholar] [CrossRef]
- Gravante, G.; Di Fede, M.C.; Araco, A.; Grimaldi, M.; De Angelis, B.; Arpino, A.; Cervelli, V.; Montone, A. A randomized trial comparing ReCell® system of epidermal cells delivery versus classic skin grafts for the treatment of deep partial thickness burns. Burns 2007. [Google Scholar] [CrossRef]
- Límová, M. Active Wound Coverings: Bioengineered Skin and Dermal Substitutes. Surg. Clin. N. Am. 2010, 90, 1237–1255. [Google Scholar] [CrossRef]
- Still, J.; Glat, P.; Silverstein, P.; Griswold, J.; Mozingo, D. The use of a collagen sponge/living cell composite material to treat donor sites in burn patients. Burns 2003, 29, 837–841. [Google Scholar] [CrossRef]
- Kumar, R.J.; Kimble, R.M.; Boots, R.; Pegg, S.P. Treatment of partial-thickness burns: A prospective, randomized trial using transcyteTM. ANZ J. Surg. 2004. [Google Scholar] [CrossRef]
- Taufique, Z.M.; Bhatt, N.; Zagzag, D.; Lebowitz, R.A.; Lieberman, S.M. Revascularization of AlloDerm Used during Endoscopic Skull Base Surgery. J. Neurol. Surg. Part B Skull Base 2018, 80, 46–50. [Google Scholar] [CrossRef]
- Whitaker, I.S.; Prowse, S.; Potokar, T.S. A critical evaluation of the use of biobrane as a biologic skin substitute A versatile tool for the plastic and reconstructive surgeon. Ann. Plast. Surg. 2008, 60, 333–337. [Google Scholar] [CrossRef]
- Fan, C.; Pek, C.H.; Por, Y.C.; Lim, G.J.S. Biobrane dressing for paediatric burns in singapore: A retrospective review. Singap. Med. J. 2018, 59, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Wood, F.M.; Stoner, M.L.; Fowler, B.V.; Fear, M.W. The use of a non-cultured autologous cell suspension and Integra® dermal regeneration template to repair full-thickness skin wounds in a porcine model: A one-step process. Burns 2007. [Google Scholar] [CrossRef]
- Min, J.H.; Yun, I.S.; Lew, D.H.; Roh, T.S.; Lee, W.J. The use of Matriderm and autologous skin graft in the treatment of full thickness skin defects. Arch. Plast. Surg. 2014. [Google Scholar] [CrossRef] [Green Version]
- Brown-Etris, M.; Milne, C.T.; Hodde, J.P. An extracellular matrix graft (Oasis ® wound matrix) for treating full-thickness pressure ulcers: A randomized clinical trial. J. Tissue Viability 2019. [Google Scholar] [CrossRef]
- Hodde, J. Extracellular matrix as a bioactive material for soft tissue reconstruction. ANZ J. Surg. 2006, 76, 1096–1100. [Google Scholar] [CrossRef]
- Marquez, S.; Mulier, K.; Nguyen, A.; Jackson, E.; Chipman, J.; Beilman, G.; Delaney, J. Comparision of PermacolTM and StratticeTM Biologic Mesh for the Repair of Abdominal Wall Defects. J. Surg. Res. 2010, 158, 353–354. [Google Scholar] [CrossRef]
- Mądry, R.; Struzyna, J.; Stachura-Kułach, A.; Drozdz, Ł.; Bugaj, M. Effectiveness of Suprathel® application in partial thickness burns, frostbites and Lyell syndrome treatment. Pol. J. Surg. 2011. [Google Scholar] [CrossRef] [PubMed]
- Highton, L.; Wallace, C.; Shah, M. Use of Suprathel® for partial thickness burns in children. Burns 2013. [Google Scholar] [CrossRef] [PubMed]
- Soejima, K.; Chen, X.; Nozaki, M.; Hori, K.; Sakurai, H.; Takeuchi, M. Novel application method of artificial dermis: One-step grafting procedure of artificial dermis and skin, rat experimental study. Burns 2006. [Google Scholar] [CrossRef] [PubMed]
- Yim, H.; Cho, Y.S.; Seo, C.H.; Lee, B.C.; Ko, J.H.; Kim, D.; Hur, J.; Chun, W.; Kim, J.H. The use of AlloDerm on major burn patients: AlloDerm prevents post-burn joint contracture. Burns 2010, 36, 322–328. [Google Scholar] [CrossRef]
- BIOBRANE|Smith & Nephew-Spain. Available online: https://www.smith-nephew.com/espana/productos/curacion-de-heridas/otros/epitelizacion/biobrane/ (accessed on 26 June 2020).
- Johnsen, S.; Ermuth, T.; Tanczos, E.; Bannasch, H.; Horch, R.E.; Zschocke, I.; Peschen, M.; Schöpf, E.; Vanscheidt, W.; Augustin, M. Treatment of therapy-refractive ulcera cruris of various origins with autologous keratinocytes in fibrin sealant. Vasa J. Vasc. Dis. 2005. [Google Scholar] [CrossRef]
- Carsin, H.; Ainaud, P.; Le Bever, H.; Rives, J.M.; Lakhel, A.; Stephanazzi, J.; Lambert, F.; Perrot, J. Cultured epithelial autografts in extensive burn coverage of severely traumatized patients: A five year single-center experience with 30 patients. Burns 2000. [Google Scholar] [CrossRef]
- Vacher, D. Autologous epidermal sheets production for skin cellular therapy. Ann. Pharm. Fr. 2003, 61, 203. [Google Scholar]
- Sharma, N.; El Refaiy, A.; Sibly, T.F. Short-term results of rotator cuff repair using GraftJacket as an interpositional tissue-matched thickness graft. J. Orthop. 2018. [Google Scholar] [CrossRef]
- Uccioli, L. A Clinical Investigation on the Characteristics and Outcomes of Treating Chronic Lower Extremity Wounds using the TissueTech Autograft System. Int. J. Low. Extrem. Wounds 2003. [Google Scholar] [CrossRef]
- Hodde, J.P.; Ernst, D.M.; Hiles, M.C. An investigation of the long-term bioactivity of endogenous growth factor in OASIS Wound Matrix. J. Wound Care 2005. [Google Scholar] [CrossRef] [PubMed]
- Van Dorp, A.G.; Verhoeven, M.C.; Koerten, H.K.; van Blitterswijk, C.A.; Ponec, M. Bilayered Biodegradable Poly(ethylene glycol)/poly(butylene Terephthalate) Copolymer (Polyactive) as Substrate for Human Fibroblasts and Keratinocytes. J. Biomed. Mater. Res. 1999, 47. [Google Scholar] [CrossRef]
- Holmes, J.H.; Molnar, J.A.; Shupp, J.W.; Hickerson, W.L.; King, B.T.; Foster, K.N.; Cairns, B.A.; Carter, J.E. Demonstration of the safety and effectiveness of the RECELL® System combined with split-thickness meshed autografts for the reduction of donor skin to treat mixed-depth burn injuries. Burns 2019, 45, 772–782. [Google Scholar] [CrossRef]
- Young, S.M.; Park, J.W.; Kim, Y.-D.; Woo, K.I. Use of Meshed Acellular Dermal Allograft as a Lining Material After Orbital Exenteration. Ophthalmic Plast. Reconstr. Surg. 2019. [Google Scholar] [CrossRef]
- Yamamoto, T.; Iwase, H.; King, T.W.; Hara, H.; Cooper, D.K.C. Skin xenotransplantation: Historical review and clinical potential. Burns 2018, 44, 1738–1749. [Google Scholar] [CrossRef] [PubMed]
- Richters, C.D.; Hoekstra, M.J.; Du Pont, J.S.; Kreis, R.W.; Kamperdijk, E.W.A. Immunology of skin transplantation. Clin. Dermatol. 2005, 23, 338–342. [Google Scholar] [CrossRef]
- Benichou, G.; Yamada, Y.; Yun, S.H.; Lin, C.; Fray, M.; Tocco, G. Immune recognition and rejection of allogeneic skin grafts. Immunotherapy 2011, 3, 757–770. [Google Scholar] [CrossRef] [Green Version]
- Niezgoda, J.A.; Van Gils, C.C.; Frykberg, R.G.; Hodde, J.P. Randomized clinical trial comparing OASIS Wound Matrix to Regranex Gel for diabetic ulcers. Adv. Ski. Wound Care 2005. [Google Scholar] [CrossRef]
- Lindberg, K.; Badylak, S.F. Porcine small intestinal submucosa (SIS): A bioscaffold supporting in vitro primary human epidermal cell differentiation and synthesis of basement membrane proteins. Burns 2001, 27, 254–266. [Google Scholar] [CrossRef]
- Haldar, S.; Sharma, A.; Gupta, S.; Chauhan, S.; Roy, P.; Lahiri, D. Bioengineered smart trilayer skin tissue substitute for efficient deep wound healing. Mater. Sci. Eng. C 2019. [Google Scholar] [CrossRef]
- Dhasmana, A.; Singh, S.; Kadian, S.; Singh, L. Skin Tissue Engineering: Principles and Advances. J. Dermatol. Ski. Care 2018, 1, 1–11. [Google Scholar]
- Cubo, N.; Garcia, M.; Del Cañizo, J.F.; Velasco, D.; Jorcano, J.L. 3D bioprinting of functional human skin: Production and in vivo analysis. Biofabrication 2017. [Google Scholar] [CrossRef] [Green Version]
- Monfort, A.; Soriano-Navarro, M.; García-Verdugo, J.M.; Izeta, A. Production of human tissue-engineered skin trilayer on a plasma-based hypodermis. J. Tissue Eng. Regen. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; He, Z.; Bian, Y.; Lei, Z.; Wang, H.; Long, Y.; Hu, M.; Li, J.; Xu, L.; Li, J.; et al. A Biomimetic Basement Membrane Substitute Based on Tri-Layered Nanofibrous Scaffold for Skin Reconstruction. J. Biomed. Nanotechnol. 2019. [Google Scholar] [CrossRef]
- Lin, H.Y.; Chen, S.H.; Chang, S.H.; Huang, S.T. Tri-layered chitosan scaffold as a potential skin substitute. J. Biomater. Sci. Polym. Ed. 2015. [Google Scholar] [CrossRef] [PubMed]
- Trottier, V.; Marceau-Fortier, G.; Germain, L.; Vincent, C.; Fradette, J. IFATS Collection: Using Human Adipose-Derived Stem/Stromal Cells for the Production of New Skin Substitutes. Stem Cells 2008. [Google Scholar] [CrossRef] [PubMed]
- Vermette, M.; Trottier, V.; Ménard, V.; Saint-Pierre, L.; Roy, A.; Fradette, J. Production of a new tissue-engineered adipose substitute from human adipose-derived stromal cells. Biomaterials 2007. [Google Scholar] [CrossRef]
- Sahota, P.S.; Burn, J.L.; Heaton, M.; Freedlander, E.; Suvarna, S.K.; Brown, N.J.; Mac Neil, S. Development of a reconstructed human skin model for angiogenesis. Wound Repair Regen. 2003. [Google Scholar] [CrossRef]
- Sorg, H.; Tilkorn, D.J.; Hager, S.; Hauser, J.; Mirastschijski, U. Skin Wound Healing: An Update on the Current Knowledge and Concepts. Eur. Surg. Res. 2017, 58, 81–94. [Google Scholar] [CrossRef]
- Lindley, L.E.; Stojadinovic, O.; Pastar, I.; Tomic-Canic, M. Biology and biomarkers for wound healing. Plast. Reconstr. Surg. 2016. [Google Scholar] [CrossRef]
- Sangkuhl, K.; Shuldiner, A.R.; Klein, T.E.; Altman, R.B. Platelet aggregation pathway. Pharmacogenet. Genom. 2011, 21, 516–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gauglitz, G.G.; Korting, H.C.; Pavicic, T.; Ruzicka, T.; Jeschke, M.G. Hypertrophic scarring and keloids: Pathomechanisms and current and emerging treatment strategies. Mol. Med. 2011. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Martin, P.; Tomic-Canic, M. Wound repair and regeneration: Mechanisms, signaling, and translation. Sci. Transl. Med. 2014, 6, 265sr6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berman, B.; Maderal, A.; Raphael, B. Keloids and hypertrophic scars: Pathophysiology, classification, and treatment. Dermatol. Surg. 2017, 43, S3–S18. [Google Scholar] [CrossRef]
- Chen, C.S.; Su, W.H.; Cheng, M.H.; Lee, W.L.; Tsou, T.S.; Chang, W.H.; Wang, P.H. Nonsteroidal anti-inflammatory drugs for wounds: Pain relief or excessive scar formation? Mediat. Inflamm. 2010, 2010, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Vig, K.; Chaudhari, A.; Tripathi, S.; Dixit, S.; Sahu, R.; Pillai, S.; Dennis, V.A.; Singh, S.R. Advances in skin regeneration using tissue engineering. Int. J. Mol. Sci. 2017, 18, 789. [Google Scholar] [CrossRef]
- Harvey, C. Wound healing. Orthop. Nurs. 2005, 24, 143–157. [Google Scholar] [CrossRef]
- Rhett, J.M.; Ghatnekar, G.S.; Palatinus, J.A.; O’Quinn, M.; Yost, M.J.; Gourdie, R.G. Novel therapies for scar reduction and regenerative healing of skin wounds. Trends Biotechnol. 2008, 26, 173–180. [Google Scholar] [CrossRef]
- Huang, S.P.; Hsu, C.C.; Chang, S.C.; Wang, C.H.; Deng, S.C.; Dai, N.T.; Chen, T.M.; Chan, J.Y.H.; Chen, S.G.; Huang, S.M. Adipose-derived stem cells seeded on acellular dermal matrix grafts enhance wound healing in a murine model of a full-thickness defect. Ann. Plast. Surg. 2012. [Google Scholar] [CrossRef]
- Du, P.; Suhaeri, M.; Ha, S.S.; Oh, S.J.; Kim, S.H.; Park, K. Human lung fibroblast-derived matrix facilitates vascular morphogenesis in 3D environment and enhances skin wound healing. Acta Biomater. 2017. [Google Scholar] [CrossRef]
- Ko, H.C.H.; Milthorpe, B.K.; McFarland, C.D. Engineering thick tissues-The vascularisation problem. Eur. Cells Mater. 2007, 14, 1–19. [Google Scholar] [CrossRef]
- Rivron, N.C.; Liu, J.; Rouwkema, J.; De Boer, J.; Van Blitterswijk, C.A. Engineering vascularised tissues in vitro. Eur. Cells Mater. 2008, 15, 27–40. [Google Scholar] [CrossRef]
- Scherberich, A.; Müller, A.M.; Schäfer, D.J.; Banfi, A.; Martin, I. Adipose tissue-derived progenitors for engineering osteogenic and vasculogenic grafts. J. Cell. Physiol. 2010, 225, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Papavasiliou, G.; Ming-Huei, C.; Brey, E.M. Strategies for vascularization of polymer scaffolds. J. Investig. Med. 2010. [Google Scholar] [CrossRef] [Green Version]
- Laschke, M.W.; Menger, M.D. Prevascularization in tissue engineering: Current concepts and future directions. Biotechnol. Adv. 2016, 34, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Lokmic, Z.; Stillaert, F.; Morrison, W.A.; Thompson, E.W.; Mitchell, G.M. An arteriovenous loop in a protected space generates a permanent, highly vascular, tissue-engineered construct. FASEB J. 2007. [Google Scholar] [CrossRef] [Green Version]
- Laschke, M.W.; Vollmar, B.; Menger, M.D. Inosculation: Connecting the life-sustaining Pipelines. Tissue Eng. Part B Rev. 2009, 15, 455–465. [Google Scholar] [CrossRef]
- Laschke, M.W.; Menger, M.D. Vascularization in tissue engineering: Angiogenesis versus inosculation. Eur. Surg. Res. 2012, 48, 85–92. [Google Scholar] [CrossRef]
- Kaully, T.; Kaufman-Francis, K.; Lesman, A.; Levenberg, S. Vascularization-the conduit to viable engineered tissues. Tissue Eng. Part B Rev. 2009, 15, 159–169. [Google Scholar] [CrossRef] [Green Version]
- Baranski, J.D.; Chaturvedi, R.R.; Stevens, K.R.; Eyckmans, J.; Carvalho, B.; Solorzano, R.D.; Yang, M.T.; Miller, J.S.; Bhatia, S.N.; Chen, C.S. Geometric control of vascular networks to enhance engineered tissue integration and function. Proc. Natl. Acad. Sci. USA 2013, 110, 7586–7591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frerich, B.; Winter, K.; Scheller, K.; Braumann, U.D. Comparison of Different Fabrication Techniques for Human Adipose Tissue Engineering in Severe Combined Immunodeficient Mice. Artif. Organs 2012. [Google Scholar] [CrossRef] [PubMed]
- West, J.; Moon, J. Vascularization of Engineered Tissues: Approaches to Promote Angiogenesis in Biomaterials. Curr. Top. Med. Chem. 2008, 8, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Klar, A.S.; Güven, S.; Biedermann, T.; Luginbühl, J.; Böttcher-Haberzeth, S.; Meuli-Simmen, C.; Meuli, M.; Martin, I.; Scherberich, A.; Reichmann, E. Tissue-engineered dermo-epidermal skin grafts prevascularized with adipose-derived cells. Biomaterials 2014, 35, 5065–5078. [Google Scholar] [CrossRef]
- Xue, L.; Greisler, H.P. Angiogenic effect of fibroblast growth factor-1 and vascular endothelial growth factor and their synergism in a novel in vitro quantitative fibrin-based 3-dimensional angiogenesis system. Surgery 2002, 132, 259–267. [Google Scholar] [CrossRef]
- Otrock, Z.K.; Mahfouz, R.A.R.; Makarem, J.A.; Shamseddine, A.I. Understanding the biology of angiogenesis: Review of the most important molecular mechanisms. Blood Cells Mol. Dis. 2007, 39, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Randi, A.M.; Smith, K.E.; Castaman, G. Von Willebrand factor regulation of blood vessel formation. Blood 2018, 132, 1–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heydarkhan-Hagvall, S.; Helenius, G.; Johansson, B.R.; Li, J.Y.; Mattsson, E.; Risberg, B. Co-culture of endothelial cells and smooth muscle cells affects gene expression of angiogenic factors. J. Cell. Biochem. 2003, 89, 1250–1259. [Google Scholar] [CrossRef]
- Jain, R.K. Molecular regulation of vessel maturation. Nat. Med. 2003, 9, 685–693. [Google Scholar] [CrossRef]
- Tremblay, P.L.; Hudon, V.; Berthod, F.; Germain, L.; Auger, F.A. Inosculation of tissue-engineered capillaries with the host’s vasculature in a reconstructed skin transplanted on mice. Am. J. Transplant. 2005. [Google Scholar] [CrossRef]
- Marino, D.; Luginbühl, J.; Scola, S.; Meuli, M.; Reichmann, E. Bioengineering: Bioengineering dermo-epidermal skin grafts with blood and lymphatic capillaries. Sci. Transl. Med. 2014. [Google Scholar] [CrossRef] [Green Version]
- Güven, S.; Mehrkens, A.; Saxer, F.; Schaefer, D.J.; Martinetti, R.; Martin, I.; Scherberich, A. Engineering of large osteogenic grafts with rapid engraftment capacity using mesenchymal and endothelial progenitors from human adipose tissue. Biomaterials 2011, 32, 5801–5809. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.J.; Koh, B.I.; Kim, H.; Joo, H.J.; Jin, H.K.; Jeon, J.; Choi, C.; Lee, D.H.; Chung, J.H.; Cho, C.H.; et al. Stromal vascular fraction from adipose tissue forms profound vascular network through the dynamic reassembly of blood endothelial cells. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1141–1150. [Google Scholar] [CrossRef] [Green Version]
- Müller, A.M.; Mehrkens, A.; Schäfer, D.J.; Jaquiery, C.; Güven, S.; Lehmicke, M.; Martinetti, R.; Farhadi, I.; Jakob, M.; Scherberich, A.; et al. Towards an intraoperative engineering of osteogenic and vasculogenic grafts from the stromal vascular fraction of human adipose tissue. Eur. Cells Mater. 2010, 19, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Boyd, N.L.; Nunes, S.S.; Krishnan, L.; Jokinen, J.D.; Ramakrishnan, V.M.; Bugg, A.R.; Hoying, J.B. Dissecting the role of human embryonic stem cell-derived mesenchymal cells in human umbilical vein endothelial cell network stabilization in three-dimensional environments. Tissue Eng. Part A 2013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koike, N.; Fukumura, D.; Gralla, O.; Au, P.; Schechner, J.; Jain, R. Creation of long-lasting blood vessels. Nature 2004, 428, 138–139. [Google Scholar] [CrossRef] [PubMed]
- Bergers, G.; Song, S. The role of pericytes in blood-vessel formation and maintenance. Neuro. Oncol. 2005. [Google Scholar] [CrossRef] [Green Version]
- Duttenhoefer, F.; Lara De Freitas, R.; Meury, T.; Loibl, M.; Benneker, L.M.; Richards, R.G.; Alini, M.; Verrier, S. 3D scaffolds co-seeded with human endothelial progenitor and mesenchymal stem cells: Evidence of prevascularisation within 7 days. Eur. Cells Mater. 2013, 26, 59–65. [Google Scholar] [CrossRef]
- Lin, Y.; Weisdorf, D.J.; Solovey, A.; Hebbel, R.P. Origins of circulating endothelial cells and endothelial outgrowth from blood. J. Clin. Investig. 2000. [Google Scholar] [CrossRef] [Green Version]
- Dai, N.T.; Huang, W.S.; Chang, F.W.; Wei, L.G.; Huang, T.C.; Li, J.K.; Fu, K.Y.; Dai, L.G.; Hsieh, P.S.; Huang, N.C.; et al. Development of a Novel Pre-Vascularized Three-Dimensional Skin Substitute Using Blood Plasma Gel. Cell Transplant. 2018, 27, 1535–1547. [Google Scholar] [CrossRef]
- Vitacolonna, M.; Belharazem, D.; Hohenberger, P.; Roessner, E.D. In-vivo quantification of the revascularization of a human acellular dermis seeded with EPCs and MSCs in co-culture with fibroblasts and pericytes in the dorsal chamber model in pre-irradiated tissue. Cell Tissue Bank. 2017. [Google Scholar] [CrossRef]
- Asahara, T. Endothelial progenitor cells for vascular medicine. Yakugaku Zasshi 2007, 127, 841–845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, R.K.; Zamora, D.O.; Wrice, N.L.; Baer, D.G.; Renz, E.M.; Christy, R.J.; Natesan, S. Development of a vascularized skin construct using adipose-derived stem cells from debrided burned skin. Stem Cells Int. 2012. [Google Scholar] [CrossRef]
- Abe, Y.; Ozaki, Y.; Kasuya, J.; Yamamoto, K.; Ando, J.; Sudo, R.; Ikeda, M.; Tanishita, K. Endothelial progenitor cells promote directional three-dimensional endothelial network formation by secreting vascular endothelial growth factor. PLoS ONE 2013. [Google Scholar] [CrossRef] [Green Version]
- Nicholas, M.N.; Jeschke, M.G.; Amini-Nik, S. Methodologies in creating skin substitutes. Cell. Mol. Life Sci. 2016, 73, 3453–3472. [Google Scholar] [CrossRef] [Green Version]
- Laschke, M.W.; Kleer, S.; Scheuer, C.; Schuler, S.; Garcia, P.; Eglin, D.; Alini, M.; Menger, M.D. Vascularisation of porous scaffolds is improved by incorporation of adipose tissue-derived microvascular fragments. Eur. Cells Mater. 2012. [Google Scholar] [CrossRef]
- Laschke, M.W.; Menger, M.D. Adipose tissue-derived microvascular fragments: Natural vascularization units for regenerative medicine. Trends Biotechnol. 2015, 33, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Frueh, F.S.; Später, T.; Körbel, C.; Scheuer, C.; Simson, A.C.; Lindenblatt, N.; Giovanoli, P.; Menger, M.D.; Laschke, M.W. Prevascularization of dermal substitutes with adipose tissue-derived microvascular fragments enhances early skin grafting. Sci. Rep. 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Frueh, F.S.; Später, T.; Lindenblatt, N.; Calcagni, M.; Giovanoli, P.; Scheuer, C.; Menger, M.D.; Laschke, M.W. Adipose Tissue-Derived Microvascular Fragments Improve Vascularization, Lymphangiogenesis, and Integration of Dermal Skin Substitutes. J. Investig. Dermatol. 2017. [Google Scholar] [CrossRef] [Green Version]
- Hasan, A.; Paul, A.; Vrana, N.E.; Zhao, X.; Memic, A.; Hwang, Y.S.; Dokmeci, M.R.; Khademhosseini, A. Microfluidic techniques for development of 3D vascularized tissue. Biomaterials 2014, 35, 7308–7325. [Google Scholar] [CrossRef] [Green Version]
- Schechner, J.S.; Nath, A.K.; Zheng, L.; Kluger, M.S.; Hughes, C.C.W.; Sierra-Honigmann, M.R.; Lorber, M.I.; Tellides, G.; Kashgarian, M.; Bothwell, A.L.M.; et al. In vivo formation of complex microvessels lined by human endothelial cells in an immunodeficient mouse. Proc. Natl. Acad. Sci. USA 2000. [Google Scholar] [CrossRef] [Green Version]
- Erber, R.; Thurnher, A.; Katsen, A.D.; Groth, G.; Kerger, H.; Hammes, H.P.; Menger, M.D.; Ullrich, A.; Vajkoczy, P. Combined inhibition of VEGF and PDGF signaling enforces tumor vessel regression by interfering with pericyte-mediated endothelial cell survival mechanisms. FASEB J. 2004. [Google Scholar] [CrossRef] [PubMed]
- Levenberg, S.; Rouwkema, J.; Macdonald, M.; Garfein, E.S.; Kohane, D.S.; Darland, D.C.; Marini, R.; Van Blitterswijk, C.A.; Mulligan, R.C.; D’Amore, P.A.; et al. Engineering vascularized skeletal muscle tissue. Nat. Biotechnol. 2005. [Google Scholar] [CrossRef]
- Gökçinar-Yagci, B.; Uçkan-Çetinkaya, D.; Çelebi-Saltik, B. Pericytes: Properties, functions and applications in tissue engineering. Stem Cell Rev. Rep. 2015, 11, 549–559. [Google Scholar] [CrossRef] [PubMed]


| Type of Temporary Cover | Example | Description |
|---|---|---|
| Xenogeneic decellularized skin | E-Z Derm® Mölnlycke | Porcine xenograft for skin loss injuries |
| Allogenic cadaveric human skin | Euro Skin Bank | Donated human skin allografts derived from cadavers |
| Human amnion | EpiBurn® Mimedx | Dehydrated human amnion allograft which acts as a protective barrier and promotes healing |
| Synthetic dressings | Gauzes or hydrocolloids | Covers made of synthetic materials |
| Alternative natural skin covers | Banana leaves Potato peel | Natural covers used specially on developing countries |
| Commercial Brand | Cell Content | Source | Conformation | Anatomical Structure | Type of Biomaterial | Description | Clinical Use |
|---|---|---|---|---|---|---|---|
| AlloDerm® [67] | Acellular | Allogeneic | Bi-layered | Dermal | Biological | Donated allograft human dermis decellularized and freeze-dried with a “dermal” side and a “basement membrane” side | Gingival augmentation, dental roots cover, burns |
| Apligraf® [45] | Cellular | Allogeneic | Bi-layered | Composite | Biological | Human foreskin neonatal keratinocytes and fibroblasts within a bovine type I collagen matrix | Licensed only for diabetic foot ulcers (DFUs) and venous leg ulcers (VLUs) |
| Biobrane® [58,68] | Acellular | Xenogeneic | Bi-layered | Dermal | Biosynthetic | Semipermeable silicone film partially imbedded in a 3D network of nylon functionalized with porcine collagen type I | Superficial partial thickness burns |
| Bioseed-S [69] | Cellular | Autologous | Single-layer | Epidermal | Biological | Autologous keratinocytes suspended on a fibrin sealant | Therapy-resistant chronic VLUs |
| CryoSkin [47] | Cellular | Allogeneic | Spray | Epidermal | Biological | A cell spray made of keratinocytes isolated from newborn foreskin cultured on silicone | Superficial wounds. |
| Dermagraft® [48] | Cellular | Allogeneic | Single-layer | Dermal | Biological | Foreskin fibroblast which secrete growth factors and ECM seeded on a bioabsorbable polyglactin mesh scaffold | Stalled diabetic foot ulcers (DFUs), other clinical indications |
| EPIBASE® [51,70] | Cellular | Autologous | Single-layered | Epidermal | Biological | Keratinocytes isolated from a small biopsy expanded originating CEA that is sprayed on the wound | Cutaneous calciphylaxis, burns |
| Epicel® [71] | Cellular | Autologous | Single-layered | Epidermal | Biological | Keratinocytes attached to a petrolatum gauze support | Deep dermal burns |
| EpidexTM® [50] | Cellular | Autologous | Single-layered | Epidermal | Biological | Expanded epidermal keratinocytes precursor cells derived from the follicular outer root sheath (ORS) by plucking hair armed on a sillicone membrane disc | Chronic leg ulcers |
| GraftJacket® [72] | Acellular | Allogeneic | Single-layered | Dermal | Biosynthetic | Human dermal collagen matrix with vascular channels | Rotator-cuff-tears |
| Hyalograft 3D® [73] | Cellular | Autologous | Single-layered | Dermal | Biological | Autologous fibroblasts seeded on a hyaluronic acid scaffold | Full-thickness and deep partial wound |
| Integra® [59] | Acellular | Xenogeneic | Bi-layered | Dermal | Biosynthetic | Matrix of bovine derived collagen fibers, chondroitin-6-sulphate and a silicone sheet that acts as a barrier | Burns or reconstructive surgery |
| Laserskin® [73] | Cellular | Autologous | Single-layered | Epidermal | Biosynthetic | Cultured keratinocytes on a hyaluronic acid microperforated membrane | Wound resurfacing |
| Matriderm® [60] | Acellular | Xenogeneic | Single-layered | Dermal | Biosynthetic | A decellularized dermal substitute of bovine origin with collagen matrix coated with -elastin hydrolysate | For split thickness skin grafting (STSG) |
| OASIS® [74] | Acellular | Xenogeneic | Single-layered | Composite | Biological | Matrix derived from porcine small intestinal submucosa | Wound closure, full-thickness ulcers |
| OrCel® [54] | Cellular | Allogeneic | Bi-layered | Composite | Biological | Epidermal keratinocytes and dermal fibroblasts co-cultured in separate layers, into a type I bovine collagen sponge matrix | Severely burned patients |
| Permacol™ surgical implant [63] | Acellular | Xenogeneic | Single-layered | Dermal | Biological | Decellularized dermal porcine containing collagen and elastin | Specially used for abdominal wall hernia and dermal reconstruction |
| PolyActive® [75] | Cellular | Autologous | Bi-layered | Composite | Biological | Soft polyethylene oxide terephthalate component and a hard polybutylene terephthalate component with a keratinocytes and fibroblasts | Not specified |
| Recell® [76] | Cellular | Autologous | Single-layered | Epidermal | Biological | Keratinocytes and melanocytes spray | Depth burns |
| Suprathel® [64] | Acellular | Cell-free | Single-layered | Epidermal | Synthetic | Porous membrane made of a co-polymer (terpolymer) of poly-dl-lactide, trimethylene carbonate and ε-caprolactone | Partial thickness burns and abrasions |
| SureDerm® [77] | Acellular | Allogeneic | Bi-layered | Composite | Biosynthetic | Decellularized human dermis coated with gelatin | Exposed orbit after exenteration |
| Terudermis® [66] | Acellular | Xenogeneic | Bi-layered | Dermal | Biological | Bovine lyophilized cross-linked collagen sponge made of collagen with silicone sheet. | Burns with muscle or bone exposition |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oualla-Bachiri, W.; Fernández-González, A.; Quiñones-Vico, M.I.; Arias-Santiago, S. From Grafts to Human Bioengineered Vascularized Skin Substitutes. Int. J. Mol. Sci. 2020, 21, 8197. https://doi.org/10.3390/ijms21218197
Oualla-Bachiri W, Fernández-González A, Quiñones-Vico MI, Arias-Santiago S. From Grafts to Human Bioengineered Vascularized Skin Substitutes. International Journal of Molecular Sciences. 2020; 21(21):8197. https://doi.org/10.3390/ijms21218197
Chicago/Turabian StyleOualla-Bachiri, Wasima, Ana Fernández-González, María I. Quiñones-Vico, and Salvador Arias-Santiago. 2020. "From Grafts to Human Bioengineered Vascularized Skin Substitutes" International Journal of Molecular Sciences 21, no. 21: 8197. https://doi.org/10.3390/ijms21218197







