The Roles of Host 5-Methylcytosine RNA Methyltransferases during Viral Infections
Abstract
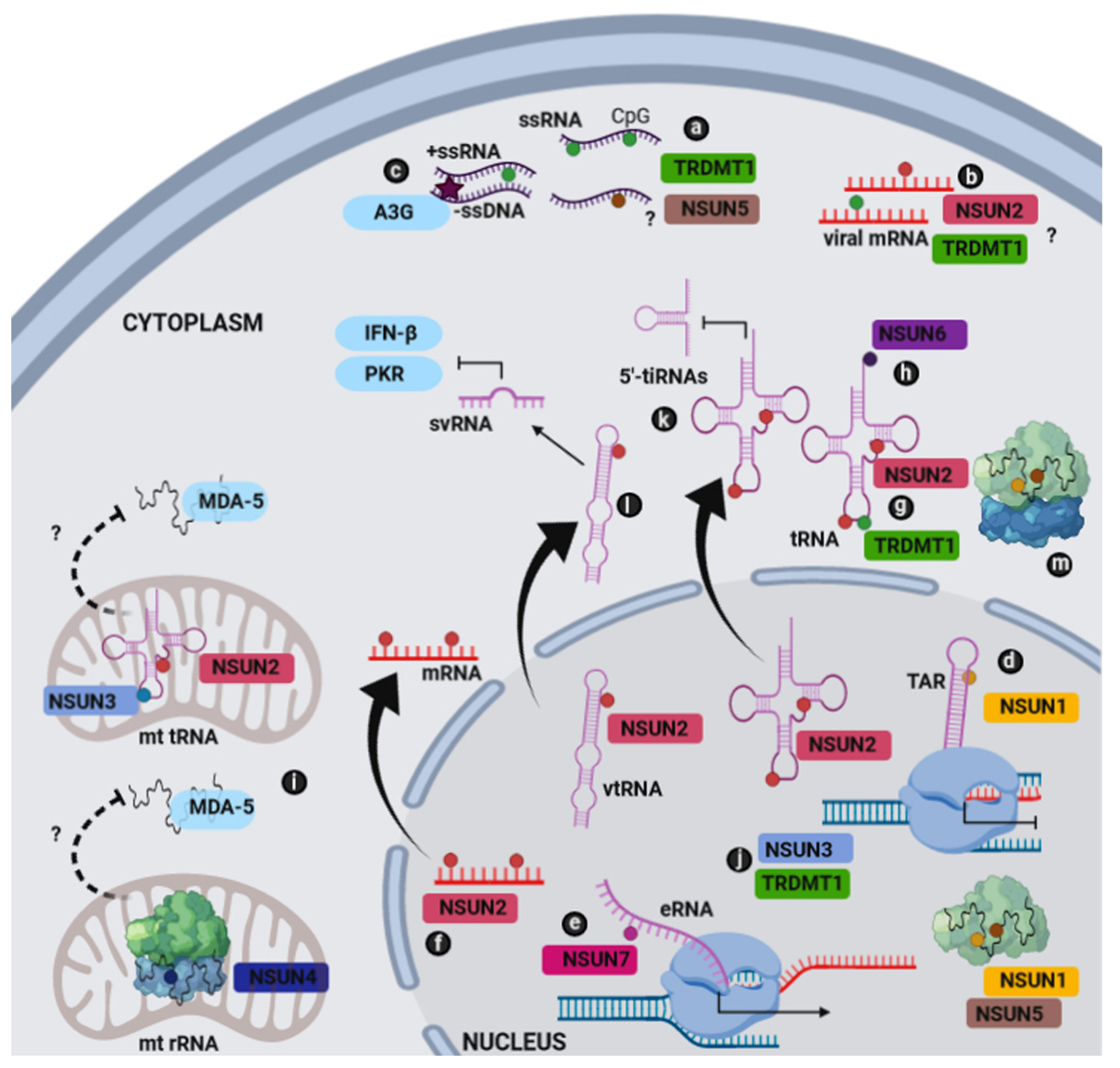
:1. Introduction
2. m5C RNA Methyltransferases and Their RNA Targets
3. Host-Based Methylation of the Fifth Position of Cytosine Residues in Viral RNA
4. The Modulation of Host Cellular Metabolism via Virus Hijacking of RNA Processing
5. Attenuation of Host Antiviral Response by Virus-Mediated Activation of NSUN2
6. Inhibitors of m5C RNA Methyltransferases as Antiviral Drugs
7. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| A/H5N1 | influenza A virus subtype H5N1 |
| A3G | apolipoprotein B mRNA editing enzyme catalytic subunit 3G (cytidine deaminase) |
| AAvV-1 | avian avulavirus 1 |
| ANG | angiogenin |
| APOBEC | apolipoprotein B editing complex |
| APOER2 | apolipoprotein E receptor 2 |
| CpG | 5′C—phosphate—G3′ |
| Cys | cysteine |
| DCV | Drosophila C virus |
| DENV | dengue virus |
| DNA | deoxyrybonucleic acid |
| DNMT2 | DNA methyltransferase 2 |
| dsRNA | double-stranded RNA |
| EBOV | Ebola virus |
| EBV | Epstein-Barr virus |
| EGCG | epigallocatechin-3-gallate |
| eRNA | enhancer RNA |
| FeHV-1 | feline herpesvirus type 1 |
| GEO | Gene Expression Omnibus |
| Glu | glutamic acid |
| Gly | glycine |
| HBV | hepatitis B virus |
| HCV | hepatitis C virus |
| hDNMT1 | human DNA methyltransferase 1 |
| hDNMT3Acat | human DNA methyltransferase 3A catalytic domain |
| HIV-1 | human immunodeficiency virus 1 |
| HMPV | human metapneumovirus |
| hnRNPK | heterogeneous nuclear ribonucleoprotein K |
| HPV | human papillomavirus |
| HSV-1 | herpes simplex virus 1 |
| HTLV | human T-lymphotropic virus |
| IAV | influenza A virus |
| IFN-β | interferon beta |
| KSHV | Kaposi’s sarcoma-associated herpesvirus |
| Leu | leucine |
| lncRNA | long non-coding RNA |
| m5C | 5-methylcytosine |
| m6A | N6-methyladenosine |
| MDA-5 | melanoma differentiation-associated protein 5 |
| Met | methionine |
| miRNA | microRNA |
| MLV | murine leukemia virus |
| mRNA | messenger RNA |
| mt tRNA | mitochondrial tRNA |
| NDV | Newcastle disease virus |
| Nop2 | nucleolar protein 2 |
| Nsp3 | non-structural protein 3 |
| NSUN | NOL1/NOP2/SUN domain |
| nt | nucleotide |
| PGC-1α | peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| PKR | RNA-dependent protein kinase |
| P-TEFb | positive transcription elongation factor b |
| Rcm1 | rRNA (cytosine-C5-)-methyltransferase |
| RNA | ribonucleic acid |
| rRNA | ribosomal RNA |
| RSV | respiratory syncytial virus |
| RVFV | Rift Valley fever virus |
| SAM | S-adenosylmethionine |
| SARS-Bat SRBD | SARS-CoV-like virus isolated from bats that contains the spike-protein receptor-binding domain from wild type SARS-CoV Urbani |
| SARS-CoV | severe acute respiratory syndrome coronavirus |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| snRNA | small nuclear RNA |
| +ssRNA | positive-sense single-stranded RNA |
| -ssRNA | negative-sense single-stranded RNA |
| ssRNA | single-stranded RNA |
| svRNA | small non-coding RNA fragments derived from vtRNA |
| svRNA | small viral RNA |
| TAR | transactivation response element |
| THOC4/ALYREF | THO complex subunit 4/Aly/REF export factor |
| Thr | threonine |
| tiRNA | tRNA-derived stress-induced RNA |
| TRDMT1 | tRNA methyltransferase 1 |
| TREX | transcription export complex |
| tRFs | tRNA-derived non-coding fragments |
| Trm4 | multisite-specific tRNA:m5C-methyltransferase |
| tRNA | transfer RNA |
| UHPLC | ultrahigh-performance liquid chromatography |
| UPLC-MS/MS | ultra-performance liquid chromatography and tandem mass spectrometry |
| VSV | vesicular stomatitis virus |
| vtRNA | small vault RNA |
| WNV | West Nile virus |
| ZIKV | Zika virus |
References
- Boccaletto, P.; A Machnicka, M.; Purta, E.; Piątkowski, P.; Bagiński, B.; Wirecki, T.K.; De Crécy-Lagard, V.; Ross, R.; A Limbach, P.; Kotter, A.; et al. MODOMICS: A database of RNA modification pathways. 2017 update. Nucleic Acids Res. 2018, 46, D303–D307. [Google Scholar] [CrossRef]
- Desrosiers, R.; Friderici, K.; Rottman, F. Identification of Methylated Nucleosides in Messenger RNA from Novikoff Hepatoma Cells. Proc. Natl. Acad. Sci. USA 1974, 71, 3971–3975. [Google Scholar] [CrossRef] [Green Version]
- Amort, T.; Soulière, M.F.; Wille, A.; Jia, X.-Y.; Fiegl, H.; Wörle, H.; Micura, R.; Lusser, A. Long non-coding RNAs as targets for cytosine methylation. RNA Biol. 2013, 10, 1002–1008. [Google Scholar] [CrossRef] [Green Version]
- Amort, T.; Rieder, D.; Wille, A.; Khokhlova-Cubberley, D.; Riml, C.; Trixl, L.; Jia, X.-Y.; Micura, R.; Lusser, A. Distinct 5-methylcytosine profiles in poly(A) RNA from mouse embryonic stem cells and brain. Genome Biol. 2017, 18, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Lewis, C.J.; Pan, T.; Kalsotra, A. RNA modifications and structures cooperate to guide RNA–protein interactions. Nat. Rev. Mol. Cell Biol. 2017, 18, 202–210. [Google Scholar] [CrossRef] [Green Version]
- Bohnsack, M.T.; Höbartner, C.; Bohnsack, M.T. Eukaryotic 5-methylcytosine (m5C) RNA Methyltransferases: Mechanisms, Cellular Functions, and Links to Disease. Genes 2019, 10, 102. [Google Scholar] [CrossRef] [Green Version]
- Schaefer, M.; Pollex, T.; Hanna, K.; Lyko, F. RNA cytosine methylation analysis by bisulfite sequencing. Nucleic Acids Res. 2008, 37, e12. [Google Scholar] [CrossRef] [Green Version]
- Edelheit, S.; Schwartz, S.; Mumbach, M.R.; Wurtzel, O.; Sorek, R. Transcriptome-Wide Mapping of 5-methylcytidine RNA Modifications in Bacteria, Archaea, and Yeast Reveals m5C within Archaeal mRNAs. PLoS Genet. 2013, 9, e1003602. [Google Scholar] [CrossRef] [Green Version]
- Khoddami, V.; Cairns, B.R. Identification of direct targets and modified bases of RNA cytosine methyltransferases. Nat. Biotechnol. 2013, 31, 458–464. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Aleksic, J.; Blanco, S.; Dietmann, S.; Frye, M. Characterizing 5-methylcytosine in the mammalian epitranscriptome. Genome Biol. 2013, 14, 215. [Google Scholar] [CrossRef] [Green Version]
- Trixl, L.; Lusser, A. The dynamic RNA modification 5-methylcytosine and its emerging role as an epitranscriptomic mark. Wiley Interdiscip. Rev. RNA 2018, 10, e1510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dang, W.; Xie, Y.; Cao, P.; Xin, S.; Wang, J.; Li, S.; Li, Y.; Lu, J. N6-Methyladenosine and Viral Infection. Front. Microbiol. 2019, 10, 417. [Google Scholar] [CrossRef] [Green Version]
- Manners, O.; Baquero-Perez, B.; Whitehouse, A. m6A: Widespread regulatory control in virus replication. Biochim. Biophys. Acta (BBA)-Bioenerg. 2019, 1862, 370–381. [Google Scholar] [CrossRef] [PubMed]
- Daffis, S.; Szretter, K.J.; Schriewer, J.; Li, J.; Youn, S.; Errett, J.; Lin, T.-Y.; Schneller, S.W.; Zust, R.; Dong, H.; et al. 2′-O methylation of the viral mRNA cap evades host restriction by IFIT family members. Nat. Cell Biol. 2010, 468, 452–456. [Google Scholar] [CrossRef]
- Pereira-Montecinos, C.; Valiente-Echeverría, F.; Soto-Rifo, R. Epitranscriptomic regulation of viral replication. Biochim. Biophys. Acta (BBA)-Bioenerg. 2017, 1860, 460–471. [Google Scholar] [CrossRef]
- Reid, R.; Greene, P.J.; Santi, D.V. Exposition of a family of RNA m5C methyltransferases from searching genomic and proteomic sequences. Nucleic Acids Res. 1999, 27, 3138–3145. [Google Scholar] [CrossRef]
- Liu, Y.; Santi, D.V. m5C RNA and m5C DNA methyl transferases use different cysteine residues as catalysts. Proc. Natl. Acad. Sci. USA 2000, 97, 8263–8265. [Google Scholar] [CrossRef] [Green Version]
- King, M.Y.; Redman, K.L. RNA Methyltransferases Utilize Two Cysteine Residues in the Formation of 5-Methylcytosine. Biochemistry 2002, 41, 11218–11225. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, G.; Ney, M.; Gaspar, I.; Aigueperse, C.; Schaefer, M.; Kellner, S.; Helm, M.; Motorin, Y. Eukaryotic rRNA Modification by Yeast 5-Methylcytosine-Methyltransferases and Human Proliferation-Associated Antigen p120. PLoS ONE 2015, 10, e0133321. [Google Scholar] [CrossRef] [Green Version]
- Gkatza, N.A.; Castro, C.; Harvey, R.F.; Heiß, M.; Popis, M.C.; Blanco, S.; Bornelöv, S.; Sajini, A.A.; Gleeson, J.G.; Griffin, J.L.; et al. Cytosine-5 RNA methylation links protein synthesis to cell metabolism. PLoS Biol. 2019, 17, e3000297. [Google Scholar] [CrossRef]
- Auxilien, S.; Guérineau, V.; Szweykowska-Kulinska, Z.; Golinelli-Pimpaneau, B. The human tRNA m5C methyltransferase Misu is multisite-specific. RNA Biol. 2012, 9, 1331–1338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Haute, L.; Lee, S.-Y.; McCann, B.J.; A Powell, C.; Bansal, D.; Vasiliauskaitė, L.; Garone, C.; Shin, S.; Kim, J.-S.; Frye, M.; et al. NSUN2 introduces 5-methylcytosines in mammalian mitochondrial tRNAs. Nucleic Acids Res. 2019, 47, 8720–8733. [Google Scholar] [CrossRef] [PubMed]
- Brzezicha, B.; Schmidt, M.; Makałowska, I.; Jarmołowski, A.; Pieńkowska, J.; Szweykowska-Kulińska, Z. Identification of human tRNA:m5C methyltransferase catalysing intron-dependent m5C formation in the first position of the anticodon of the. Nucleic Acids Res. 2006, 34, 6034–6043. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, A.; Sun, B.-F.; Yang, Y.; Han, Y.-N.; Yuan, X.; Chen, R.-X.; Wei, W.-S.; Liu, Y.; Gao, C.-C.; et al. 5-methylcytosine promotes pathogenesis of bladder cancer through stabilizing mRNAs. Nat. Cell Biol. 2019, 21, 978–990. [Google Scholar] [CrossRef]
- Haag, S.; Sloan, K.E.; Ranjan, N.; Warda, A.S.; Kretschmer, J.; Blessing, C.; Hubner, B.; Seikowski, J.; Dennerlein, S.; Rehling, P.; et al. NSUN3 and ABH1 modify the wobble position of mt-tRNAMet to expand codon recognition in mitochondrial translation. EMBO J. 2016, 35, 2104–2119. [Google Scholar] [CrossRef]
- Nakano, S.; Suzuki, T.; Kawarada, L.; Iwata, H.; Asano, K. NSUN3 methylase initiates 5-formylcytidine biogenesis in human mitochondrial tRNA(Met). Nat. Chem. Biol. 2016, 12, 546–551. [Google Scholar] [CrossRef]
- Metodiev, M.D.; Spåhr, H.; Polosa, P.L.; Meharg, C.; Becker, C.; Altmueller, J.; Habermann, B.; Larsson, N.-G.; Ruzzenente, B. NSUN4 Is a Dual Function Mitochondrial Protein Required for Both Methylation of 12S rRNA and Coordination of Mitoribosomal Assembly. PLoS Genet. 2014, 10, e1004110. [Google Scholar] [CrossRef] [Green Version]
- Sharma, S.; Yang, J.; Watzinger, P.; Kötter, P.; Entian, K.-D. Yeast Nop2 and Rcm1 methylate C2870 and C2278 of the 25S rRNA, respectively. Nucleic Acids Res. 2013, 41, 9062–9076. [Google Scholar] [CrossRef] [Green Version]
- Janin, M.; Ortiz-Barahona, V.; De Moura, M.C.; Martínez-Cardús, A.; Llinàs-Arias, P.; Soler, M.; Nachmani, D.; Pelletier, J.; Schumann, U.; Calleja-Cervantes, M.E.; et al. Epigenetic loss of RNA-methyltransferase NSUN5 in glioma targets ribosomes to drive a stress adaptive translational program. Acta Neuropathol. 2019, 138, 1053–1074. [Google Scholar] [CrossRef] [Green Version]
- Heissenberger, C.; Liendl, L.; Nagelreiter, F.; Gonskikh, Y.; Yang, G.; Stelzer, E.M.; Krammer, T.L.; Micutkova, L.; Vogt, S.; Kreil, D.P.; et al. Loss of the ribosomal RNA methyltransferase NSUN5 impairs global protein synthesis and normal growth. Nucleic Acids Res. 2019, 47, 11807–11825. [Google Scholar] [CrossRef] [Green Version]
- Haag, S.; Warda, A.S.; Kretschmer, J.; Günnigmann, M.A.; Höbartner, C.; Bohnsack, M.T. NSUN6 is a human RNA methyltransferase that catalyzes formation of m5C72 in specific tRNAs. RNA 2015, 21, 1532–1543. [Google Scholar] [CrossRef] [Green Version]
- Liu, R.-J.; Long, T.; Li, J.; Li, H.; Wang, E.-D. Structural basis for substrate binding and catalytic mechanism of a human RNA:m5C methyltransferase NSun6. Nucleic Acids Res. 2017, 45, 6684–6697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Long, T.; Li, J.; Li, H.; Zhou, M.; Zhou, X.-L.; Liu, R.-J.; Wang, E.-D. Sequence-specific and Shape-selective RNA Recognition by the Human RNA 5-Methylcytosine Methyltransferase NSun6. J. Biol. Chem. 2016, 291, 24293–24303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aguilo, F.; Li, S.; Balasubramaniyan, N.; Sancho, A.; Benko, S.; Zhang, F.; A Vashisht, A.; Rengasamy, M.; Andino, B.; Chen, C.-H.; et al. Deposition of 5-Methylcytosine on Enhancer RNAs Enables the Coactivator Function of PGC-1α. Cell Rep. 2016, 14, 479–492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goll, M.G.; Kirpekar, F.; Maggert, K.A.; Yoder, J.A.; Hsieh, C.-L.; Zhang, X.; Golic, K.G.; Jacobsen, S.E.; Bestor, T.H. Methylation of tRNAAsp by the DNA Methyltransferase Homolog Dnmt2. Science 2006, 311, 395–398. [Google Scholar] [CrossRef] [Green Version]
- Schaefer, M.; Lyko, F. Lack of evidence for DNA methylation of Invader4 retroelements in Drosophila and implications for Dnmt2-mediated epigenetic regulation. Nat. Genet. 2010, 42, 920–921. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Pizzollo, J.; Han, Z.; Marcho, C.; Zhang, K.; Mager, J. Nop2 is required for mammalian preimplantation development. Mol. Reprod. Dev. 2015, 83, 124–131. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Tuorto, F.; Menon, S.; Blanco, S.; Cox, C.; Flores, J.V.; Watt, S.; Kudo, N.R.; Lyko, F.; Frye, M. The Mouse Cytosine-5 RNA Methyltransferase NSun2 Is a Component of the Chromatoid Body and Required for Testis Differentiation. Mol. Cell. Biol. 2013, 33, 1561–1570. [Google Scholar] [CrossRef] [Green Version]
- Tuorto, F.; Liebers, R.; Musch, T.; Schaefer, M.; Hofmann, S.; Kellner, S.; Frye, M.; Helm, M.; Stoecklin, G.; Lyko, F. RNA cytosine methylation by Dnmt2 and NSun2 promotes tRNA stability and protein synthesis. Nat. Struct. Mol. Biol. 2012, 19, 900–905. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, P.; Li, W.; Sha, S.; Wang, Y.; Yuan, Z.; Shen, B.; Chen, L. Cognitive deficits in mice lacking Nsun5, a cytosine-5 RNA methyltransferase, with impairment of oligodendrocyte precursor cells. Glia 2019, 67, 688–702. [Google Scholar] [CrossRef]
- Selmi, T.; Hussain, S.; Dietmann, S.; Heiss, M.; Carter, J.-M.; Dennison, R.; Flad, S.; Huang, Y.-L.; Kellner, S.; Borneloev, S.; et al. Sequence- and structure-specific cytosine-5 mRNA methylation by NSUN6. bioRxiv 2020. [Google Scholar] [CrossRef]
- Harris, T.; Marquez, B.; Suarez, S.; Schimenti, J. Sperm Motility Defects and Infertility in Male Mice with a Mutation in Nsun7, a Member of the Sun Domain-Containing Family of Putative RNA Methyltransferases1. Biol. Reprod. 2007, 77, 376–382. [Google Scholar] [CrossRef] [Green Version]
- Dubin, D.T.; Stollar, V. Methylation of Sindbis virus “26S” messenger RNA. Biochem. Biophys. Res. Commun. 1975, 66, 1373–1379. [Google Scholar] [CrossRef]
- Dubin, D.T.; Stollar, V.; HsuChen, C.-C.; Timko, K.; Guild, G.M. Sindbis virus messenger RNA: The 5′-termini and methylated residues of 26 and 42 S RNA. Virology 1977, 77, 457–470. [Google Scholar] [CrossRef]
- Sommer, S.; Salditt-Georgieff, M.; Bachenheimer, S.; Darnell, J.; Furuichi, Y.; Morgan, M.; Shatkin, A. The methylation of adenovirus-specific nuclear and cytoplasmic RNA. Nucleic Acids Res. 1976, 3, 749–766. [Google Scholar] [CrossRef] [PubMed]
- Courtney, D.G.; Chalem, A.; Bogerd, H.P.; Law, B.A.; Kennedy, E.M.; Holley, C.L.; Cullen, B.R. Extensive Epitranscriptomic Methylation of A and C Residues on Murine Leukemia Virus Transcripts Enhances Viral Gene Expression. mBio 2019, 10, e01209-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Courtney, D.; Tsai, K.; Bogerd, H.P.; Kennedy, E.M.; Law, B.A.; Emery, A.; Swanstrom, R.; Holley, C.L.; Cullen, B.R. Epitranscriptomic Addition of m5C to HIV-1 Transcripts Regulates Viral Gene Expression. SSRN Electron. J. 2019, 26, 217. [Google Scholar] [CrossRef]
- Rima, B.K.; McFerran, N.V. Dinucleotide and stop codon frequencies in single-stranded RNA viruses. J. Gen. Virol. 1997, 78, 2859–2870. [Google Scholar] [CrossRef]
- Karlin, S.; Doerfler, W.; Cardon, L.R. Why is CpG suppressed in the genomes of virtually all small eukaryotic viruses but not in those of large eukaryotic viruses? J. Virol. 1994, 68, 2889–2897. [Google Scholar] [CrossRef] [Green Version]
- Hoelzer, K.; Shackelton, L.A.; Parrish, C.R. Presence and role of cytosine methylation in DNA viruses of animals. Nucleic Acids Res. 2008, 36, 2825–2837. [Google Scholar] [CrossRef]
- Cheng, X.; Virk, N.; Chen, W.; Ji, S.; Ji, S.; Sun, Y.; Wu, X. CpG Usage in RNA Viruses: Data and Hypotheses. PLoS ONE 2013, 8, e74109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matyasek, R.; Kovarik, A. Mutation patterns of human SARS-COV-2 and Bat RATG13 coronavirus genomes are strongly biased towards C>U transitions, indicating rapid evolution in their hosts. Genes 2020, 11, 761. [Google Scholar] [CrossRef] [PubMed]
- Viehweger, A.; Krautwurst, S.; Lamkiewicz, K.; Madhugiri, R.; Ziebuhr, J.; Hölzer, M.; Marz, M. Direct RNA nanopore sequencing of full-length coronavirus genomes provides novel insights into structural variants and enables modification analysis. Genome Res. 2019, 29, 1545–1554. [Google Scholar] [CrossRef] [Green Version]
- Chiang, C.-M.; D, K.; Jy, L.; Js, Y.; Jw, K.; Vn, K.; H, C. Faculty Opinions recommendation of The Architecture of SARS-CoV-2 Transcriptome. Fac. Opin. Post Publ. Peer Rev. Biomed. Lit. 2020, 181, 914. [Google Scholar] [CrossRef]
- Taiaroa, G.; Rawlinson, D.; Featherstone, L.; Pitt, M.; Caly, L.; Druce, J.; Purcell, D.; Harty, L.; Tran, T.; Roberts, J.; et al. Direct RNA sequencing and early evolution of SARS-CoV-2 2020. bioRxiv 2020, 2020, 976167. [Google Scholar]
- Sawicki, S.G.; Sawicki, D.L. Coronaviruses use Discontinuous Extension for Synthesis of Subgenome-Length Negative Strands. Adv. Exp. Med. Biol. 1995, 380, 499–506. [Google Scholar] [CrossRef] [Green Version]
- Zúñiga, S.; Sola, I.; Alonso, S.; Enjuanes, L. Sequence Motifs Involved in the Regulation of Discontinuous Coronavirus Subgenomic RNA Synthesis. J. Virol. 2004, 78, 980–994. [Google Scholar] [CrossRef] [Green Version]
- Sawicki, S.G.; Sawicki, D.L.; Siddell, S.G. A Contemporary View of Coronavirus Transcription. J. Virol. 2006, 81, 20–29. [Google Scholar] [CrossRef] [Green Version]
- Antzin-Anduetza, I.; Mahiet, C.; Granger, L.A.; Odendall, C.; Swanson, C.M. Increasing the CpG dinucleotide abundance in the HIV-1 genomic RNA inhibits viral replication. Retrovirology 2017, 14, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Wasson, M.K.; Borkakoti, J.; Kumar, A.; Biswas, B.; Vivekanandan, P. The CpG dinucleotide content of the HIV-1 envelope gene may predict disease progression. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Trus, I.; Udenze, D.; Berube, N.; Wheler, C.; Martel, M.-J.; Gerdts, V.; Karniychuk, U. CpG-Recoding in Zika Virus Genome Causes Host-Age-Dependent Attenuation of Infection With Protection Against Lethal Heterologous Challenge in Mice. Front. Immunol. 2020, 10, 3077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Durdevic, Z.; Schaefer, M. Dnmt2 methyltransferases and immunity: An ancient overlooked connection between nucleotide modification and host defense? BioEssays 2013, 35, 1044–1049. [Google Scholar] [CrossRef]
- Durdevic, Z.; Mobin, M.B.; Hanna, K.; Lyko, F.; Schaefer, M. The RNA Methyltransferase Dnmt2 Is Required for Efficient Dicer-2-Dependent siRNA Pathway Activity in Drosophila. Cell Rep. 2013, 4, 931–937. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.S.; Tabata, K.; Twu, W.-I.; Rahman, S.; Kim, H.S.; Yu, J.B.; Jee, M.H.; Bartenschlager, R.; Jang, S.K. RACK1 mediates rewiring of intracellular networks induced by hepatitis C virus infection. PLoS Pathog. 2019, 15, e1008021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stavrou, S.; Ross, S.R. APOBEC3 Proteins in Viral Immunity. J. Immunol. 2015, 195, 4565–4570. [Google Scholar] [CrossRef] [Green Version]
- Bishop, K.N.; Holmes, R.K.; Sheehy, A.M.; Davidson, N.O.; Cho, S.-J.; Malim, M.H. Cytidine Deamination of Retroviral DNA by Diverse APOBEC Proteins. Curr. Biol. 2004, 14, 1392–1396. [Google Scholar] [CrossRef] [Green Version]
- Beale, R.C.; Petersen-Mahrt, S.K.; Watt, I.N.; Harris, R.S.; Rada, C.; Neuberger, M.S. Comparison of the Differential Context-dependence of DNA Deamination by APOBEC Enzymes: Correlation with Mutation Spectra in vivo. J. Mol. Biol. 2004, 337, 585–596. [Google Scholar] [CrossRef]
- Kim, E.-Y.; Lorenzo-Redondo, R.; Little, S.J.; Chung, Y.-S.; Phalora, P.K.; Berry, I.M.; Archer, J.; Penugonda, S.; Fischer, W.; Richman, D.D.; et al. Human APOBEC3 Induced Mutation of Human Immunodeficiency Virus Type-1 Contributes to Adaptation and Evolution in Natural Infection. PLoS Pathog. 2014, 10, e1004281. [Google Scholar] [CrossRef]
- Wijesinghe, P.; Bhagwat, A.S. Efficient deamination of 5-methylcytosines in DNA by human APOBEC3A, but not by AID or APOBEC3G. Nucleic Acids Res. 2012, 40, 9206–9217. [Google Scholar] [CrossRef] [Green Version]
- McIntyre, W.; Netzband, R.; Bonenfant, G.; Biegel, J.M.; Miller, C.; Fuchs, G.; Henderson, E.; Arra, M.; Canki, M.; Fabris, D.; et al. Positive-sense RNA viruses reveal the complexity and dynamics of the cellular and viral epitranscriptomes during infection. Nucleic Acids Res. 2018, 46, 5776–5791. [Google Scholar] [CrossRef] [Green Version]
- Schwarz, K.B. Oxidative stress during viral infection: A review. Free Radic. Biol. Med. 1996, 21, 641–649. [Google Scholar] [CrossRef]
- Sloan, K.E.; Bohnsack, M.T.; Watkins, N.J. The 5S RNP Couples p53 Homeostasis to Ribosome Biogenesis and Nucleolar Stress. Cell Rep. 2013, 5, 237–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, W.; Biswas, A.; Zhou, D.; Fiches, G.; Fujinaga, K.; Santoso, N.; Zhu, J. Nucleolar protein Nop2/Nsun1 suppresses HIV-1 transcription and promotes viral latency by competing with TAT for TAR binding and methylation. PLoS Pathog. 2020, 16, e1008430. [Google Scholar] [CrossRef] [PubMed]
- Shlomai, A.; Rechtman, M.M.; Burdelova, E.O.; Zilberberg, A.; Hoffman, S.; Solar, I.; Fishman, S.; Halpern, Z.; Sklan, E.H. The metabolic regulator PGC-1α links hepatitis C virus infection to hepatic insulin resistance. J. Hepatol. 2012, 57, 867–873. [Google Scholar] [CrossRef]
- Thiagarajan, D.; Dev, R.R.; Khosla, S. The DNA methyltranferase Dnmt2 participates in RNA processing during cellular stress. Epigenetics 2011, 6, 103–113. [Google Scholar] [CrossRef]
- Dev, R.R.; Ganji, R.; Singh, S.P.; Mahalingam, S.; Banerjee, S.; Khosla, S. Cytosine methylation by DNMT2 facilitates stability and survival of HIV-1 RNA in the host cell during infection. Biochem. J. 2017, 474, 2009–2026. [Google Scholar] [CrossRef]
- Mitchell, H.D.; Eisfeld, A.J.; Sims, A.C.; McDermott, J.E.; Matzke, M.M.; Webb-Robertson, B.J.; Tilton, S.C.; Tchitchek, N.; Josset, L.; Li, C.; et al. A network integration approach to predict conserved regulators related to pathogenicity of influenza and SARS-COV respiratory viruses. PLoS ONE 2013, 8, e69374. [Google Scholar] [CrossRef] [Green Version]
- Linder, A.; Hornung, V. Mitochondrial dsRNA: A New DAMP for MDA5. Dev. Cell 2018, 46, 530–532. [Google Scholar] [CrossRef] [Green Version]
- Dhir, A.; Dhir, S.; Borowski, L.S.; Jimenez, L.; Teitell, M.; Rötig, A.; Crow, Y.J.; Rice, G.I.; Duffy, D.; Tamby, C.; et al. Mitochondrial double-stranded RNA triggers antiviral signalling in humans. Nat. Cell Biol. 2018, 560, 238–242. [Google Scholar] [CrossRef]
- Cheng, J.X.; Chen, L.; Li, Y.; Cloe, A.; Yue, M.; Wei, J.; Watanabe, K.A.; Shammo, J.M.; Anastasi, J.; Shen, Q.J.; et al. RNA cytosine methylation and methyltransferases mediate chromatin organization and 5-azacytidine response and resistance in leukaemia. Nat. Commun. 2018, 9, 1163. [Google Scholar] [CrossRef]
- Ivanov, P.; Emara, M.M.; Villen, J.; Gygi, S.P.; Anderson, P. Angiogenin-Induced tRNA Fragments Inhibit Translation Initiation. Mol. Cell 2011, 43, 613–623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodarzi, H.; Liu, X.; Nguyen, H.C.; Zhang, S.; Fish, L.; Tavazoie, S.F. Endogenous tRNA-Derived Fragments Suppress Breast Cancer Progression via YBX1 Displacement. Cell 2015, 161, 790–802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gebetsberger, J.V.; Wyss, L.; Mleczko, A.M.; Reuther, J.; Polacek, N. A tRNA-derived fragment competes with mRNA for ribosome binding and regulates translation during stress. RNA Biol. 2017, 14, 1364–1373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, J.; Ptashkin, R.N.; Chen, Y.; Cheng, Z.; Liu, G.; Phan, T.; Deng, X.; Zhou, J.; Lee, I.; Lee, Y.S.; et al. Respiratory Syncytial Virus Utilizes a tRNA Fragment to Suppress Antiviral Responses Through a Novel Targeting Mechanism. Mol. Ther. 2015, 23, 1622–1629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Yang, Y.; Sun, B.-F.; Chen, Y.-S.; Xu, J.-W.; Lai, W.-Y.; Li, A.; Wang, X.; Bhattarai, D.P.; Xiao, W.; et al. 5-methylcytosine promotes mRNA export — NSUN2 as the methyltransferase and ALYREF as an m5C reader. Cell Res. 2017, 27, 606–625. [Google Scholar] [CrossRef] [Green Version]
- Boyne, J.R.; Colgan, K.J.; Whitehouse, A. Recruitment of the complete HTREX complex is required for Kaposi’s sarcoma-associated herpesvirus intronless mRNA nuclear export and virus replication. PLoS Pathog. 2008, 4, e1000194. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Sajini, A.A.; Blanco, S.; Dietmann, S.; Lombard, P.; Sugimoto, Y.; Paramor, M.; Gleeson, J.G.; Odom, D.T.; Ule, J.; et al. NSun2-Mediated Cytosine-5 Methylation of Vault Noncoding RNA Determines Its Processing into Regulatory Small RNAs. Cell Rep. 2013, 4, 255–261. [Google Scholar] [CrossRef]
- Li, F.; Chen, Y.; Zhang, Z.; Ouyang, J.; Wang, Y.; Yan, R.; Huang, S.; Gao, G.F.; Guo, G.; Chen, J.-L. Robust expression of vault RNAs induced by influenza a virus plays a critical role in suppression of PKR-mediated innate immunity. Nucleic Acids Res. 2015, 43, 10321–10337. [Google Scholar] [CrossRef]
- McAllister, C.S.; Taghavi, N.; Samuel, C.E. Protein Kinase PKR Amplification of Interferon β Induction Occurs through Initiation Factor eIF-2α-mediated Translational Control. J. Biol. Chem. 2012, 287, 36384–36392. [Google Scholar] [CrossRef] [Green Version]
- Morales, L.; Oliveros, J.C.; Fernandez-Delgado, R.; Tenoever, B.R.; Enjuanes, L.; Sola, I. SARS-CoV-Encoded Small RNAs Contribute to Infection-Associated Lung Pathology. Cell Host Microbe 2017, 21, 344–355. [Google Scholar] [CrossRef] [Green Version]
- Ianevski, A.; Zusinaite, E.; Kuivanen, S.; Strand, M.; Lysvand, H.; Teppor, M.; Kakkola, L.; Paavilainen, H.; Laajala, M.; Kallio-Kokko, H.; et al. Novel activities of safe-in-human broad-spectrum antiviral agents. Antivir. Res. 2018, 154, 174–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Šorm, F.; Pískala, A.; Čihák, A.; Veselý, J. 5-Azacytidine, a new, highly effective cancerostatic. Cell. Mol. Life Sci. 1964, 20, 202–203. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, M.; Hagemann, S.; Hanna, K.; Lyko, F. Azacytidine Inhibits RNA Methylation at DNMT2 Target Sites in Human Cancer Cell Lines. Cancer Res. 2009, 69, 8127–8132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bouchard, J.; Walker, M.C.; Leclerc, J.M.; Lapointe, N.; Beaulieu, R.; Thibodeau, L. 5-azacytidine and 5-azadeoxycytidine inhibit human immunodeficiency virus type 1 replication in vitro. Antimicrob. Agents Chemother. 1990, 34, 206–209. [Google Scholar] [CrossRef] [Green Version]
- Dapp, M.J.; Clouser, C.L.; Patterson, S.; Mansky, L.M. 5-Azacytidine Can Induce Lethal Mutagenesis in Human Immunodeficiency Virus Type 1. J. Virol. 2009, 83, 11950–11958. [Google Scholar] [CrossRef] [Green Version]
- Diamantopoulos, P.T.; Michael, M.; Benopoulou, O.; Bazanis, E.; Tzeletas, G.; Meletis, J.; Vayopoulos, G.; Viniou, N.-A. Antiretroviral activity of 5-azacytidine during treatment of a HTLV-1 positive myelodysplastic syndrome with autoimmune manifestations. Virol. J. 2012, 9, 1. [Google Scholar] [CrossRef] [Green Version]
- Clouser, C.L.; Patterson, S.E.; Mansky, L.M. Exploiting Drug Repositioning for Discovery of a Novel HIV Combination Therapy. J. Virol. 2010, 84, 9301–9309. [Google Scholar] [CrossRef] [Green Version]
- Bösl, K.; Ianevski, A.; Than, T.T.; Andersen, P.I.; Kuivanen, S.; Teppor, M.; Zusinaite, E.; Dumpis, U.; Vitkauskiene, A.; Cox, R.J.; et al. Common Nodes of Virus–Host Interaction Revealed Through an Integrated Network Analysis. Front. Immunol. 2019, 10, 2186. [Google Scholar] [CrossRef]
- Huang, F.; Zhang, C.; Liu, Q.; Zhao, Y.; Zhang, Y.; Qin, Y.; Li, X.; Li, C.; Zhou, C.; Jin, N.; et al. Identification of amitriptyline HCl, flavin adenine dinucleotide, azacitidine and calcitriol as repurposing drugs for influenza A H5N1 virus-induced lung injury. PLoS Pathog. 2020, 16, e1008341. [Google Scholar] [CrossRef]
- Pauly, M.D.; Lauring, A.S. Effective Lethal Mutagenesis of Influenza Virus by Three Nucleoside Analogs. J. Virol. 2015, 89, 3584–3597. [Google Scholar] [CrossRef] [Green Version]
- Chan, A.T.C.; Tao, Q.; Robertson, K.D.; Flinn, I.W.; Mann, R.B.; Klencke, B.; Kwan, W.H.; Leung, T.W.-T.; Johnson, P.J.; Ambinder, R.F. Azacitidine Induces Demethylation of the Epstein-Barr Virus Genome in Tumors. J. Clin. Oncol. 2004, 22, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Takimoto, K. Reactivation and mutagenesis of herpes virus in 5-azacytidine-treated monkey kidney cells. Mutat. Res. Repair Rep. 1985, 146, 9–13. [Google Scholar] [CrossRef]
- Rao, S.P.; Rechsteiner, M.P.; Berger, C.; Sigrist, J.A.; Nadal, D.; Bernasconi, M. Zebularine reactivates silenced E-cadherin but unlike 5-azacytidine does not induce switching from latent to lytic Epstein-Barr virus infection in Burkitt’s lymphoma Akata cells. Mol. Cancer 2007, 6, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yebra, M.J.; Sánchez, J.; Martin, C.G.; Hardisson, C.; Barbes, C. The effect of sinefungin and synthetic analogues on RNA and DNA methyltransferases from Streptomyces. J. Antibiot. 1991, 44, 1141–1147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pugh, C.S.; Borchardt, R.T.; O Stone, H. Sinefungin, a potent inhibitor of virion mRNA(guanine-7-)-methyltransferase, mRNA(nucleoside-2′-)-methyltransferase, and viral multiplication. J. Biol. Chem. 1978, 253, 4075–4077. [Google Scholar]
- Kuroda, Y.; Yamagata, H.; Nemoto, M.; Inagaki, K.; Tamura, T.; Maeda, K. Antiviral effect of sinefungin on in vitro growth of feline herpesvirus type 1. J. Antibiot. 2019, 72, 981–985. [Google Scholar] [CrossRef]
- Li, J.; Chorba, J.S.; Whelan, S.P. Vesicular Stomatitis Viruses Resistant to the Methylase Inhibitor Sinefungin Upregulate RNA Synthesis and Reveal Mutations That Affect mRNA Cap Methylation. J. Virol. 2007, 81, 4104–4115. [Google Scholar] [CrossRef] [Green Version]
- Hercik, K.; Brynda, J.; Nencka, R.; Boura, E. Structural basis of Zika virus methyltransferase inhibition by sinefungin. Arch. Virol. 2017, 162, 2091–2096. [Google Scholar] [CrossRef]
- Lim, S.P.; Sonntag, L.S.; Noble, C.; Nilar, S.H.; Ng, R.H.; Zou, G.; Monaghan, P.; Chung, K.Y.; Dong, H.; Liu, B.; et al. Small Molecule Inhibitors That Selectively Block Dengue Virus Methyltransferase. J. Biol. Chem. 2010, 286, 6233–6240. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Liu, L.; Jones, S.A.; Banavali, N.; Kass, J.; Li, Z.; Zhang, J.; Kramer, L.D.; Ghosh, A.K.; Li, H. Selective inhibition of the West Nile virus methyltransferase by nucleoside analogs. Antivir. Res. 2013, 97, 232–239. [Google Scholar] [CrossRef] [Green Version]
- Krafcikova, P.; Silhan, J.; Nencka, R.; Boura, E. Structural analysis of the SARS-COV-2 methyltransferase complex involved in RNA cap creation bound to sinefungin. Nat. Commun. 2020, 11, 3717. [Google Scholar] [CrossRef] [PubMed]
- Mahalapbutr, P.; Kongtaworn, N.; Rungrotmongkol, T. Structural insight into the recognition of S-adenosyl-L-homocysteine and sinefungin in SARS-COV-2 NSP16/NSP10 RNA cap 2′-O-methyltransferase. Comput. Struct. Biotechnol. J. 2020, 18, 2757–2765. [Google Scholar] [CrossRef] [PubMed]
- Fang, M.Z.; Wang, Y.; Ai, N.; Hou, Z.; Sun, Y.; Lu, H.; Welsh, W.; Yang, C.S. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003, 63, 7563–7570. [Google Scholar] [PubMed]
- Halby, L.; Marechal, N.; Pechalrieu, D.; Cura, V.; Franchini, D.-M.; Faux, C.; Alby, F.; Troffer-Charlier, N.; Kudithipudi, S.; Jeltsch, A.; et al. Hijacking DNA methyltransferase transition state analogues to produce chemical scaffolds for PRMT inhibitors. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170072. [Google Scholar] [CrossRef] [Green Version]
- Kintzel, P.E. Anticancer Drug—Induced Kidney Disorders. Drug Saf. 2001, 24, 19–38. [Google Scholar] [CrossRef]
- Oakes, C.C.; Kelly, T.L.J.; Robaire, B.; Trasler, J.M. Adverse Effects of 5-Aza-2′-Deoxycytidine on Spermatogenesis Include Reduced Sperm Function and Selective Inhibition of de Novo DNA Methylation. J. Pharmacol. Exp. Ther. 2007, 322, 1171–1180. [Google Scholar] [CrossRef] [PubMed]
- Branch, S.; Francis, B.M.; Brownie, C.F.; Chernoff, N. Teratogenic effects of the demethylating agent 5-aza-2′-deoxycytidine in the Swiss Webster mouse. Toxicology 1996, 112, 37–43. [Google Scholar] [CrossRef]
- Valderrama, J.A.; González, M.F.; Colonelli, P.; Vásquez-Velásquez, D. Design and Synthesis of Angucyclinone 5-Aza Analogues. Synlett 2006, 2006, 2777–2780. [Google Scholar] [CrossRef]
- Meng, M.; Ducho, C. Oligonucleotide analogues with cationic backbone linkages. Beilstein J. Org. Chem. 2018, 14, 1293–1308. [Google Scholar] [CrossRef]
- Wang, J.; Ayano, E.; Maitani, Y.; Kanazawa, H. Enhanced cellular uptake and gene silencing activity of siRNA using temperature-responsive polymer-modified liposome. Int. J. Pharm. 2017, 523, 217–228. [Google Scholar] [CrossRef]
- Safinya, C.R.; Ewert, K.K.; Majzoub, R.N.; Leal, C. Cationic liposome–nucleic acid complexes for gene delivery and gene silencing. New J. Chem. 2014, 38, 5164–5172. [Google Scholar] [CrossRef] [Green Version]
- Ziemba, A.; Hayes, E.; Freeman, B.B.; Ye, T.; Pizzorno, G. Development of an Oral Form of Azacytidine: 2′3′5′Triacetyl-5-Azacytidine. Chemother. Res. Pract. 2011, 2011, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brueckner, B.; Rius, M.; Markelova, M.R.; Fichtner, I.; Hals, P.-A.; Sandvold, M.L.; Lyko, F. Delivery of 5-Azacytidine to Human Cancer Cells by Elaidic Acid Esterification Increases Therapeutic Drug Efficacy. Mol. Cancer Ther. 2010, 9, 1256–1264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, J.; Ding, Q.; Wang, J.; Deng, L.; Yang, L.; Tao, L.; Lei, H.; Lu, S. 5-Azacytidine delivered by mesoporous silica nanoparticles regulates the differentiation of P19 cells into cardiomyocytes. Nanoscale 2016, 8, 2011–2021. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wnuk, M.; Slipek, P.; Dziedzic, M.; Lewinska, A. The Roles of Host 5-Methylcytosine RNA Methyltransferases during Viral Infections. Int. J. Mol. Sci. 2020, 21, 8176. https://doi.org/10.3390/ijms21218176
Wnuk M, Slipek P, Dziedzic M, Lewinska A. The Roles of Host 5-Methylcytosine RNA Methyltransferases during Viral Infections. International Journal of Molecular Sciences. 2020; 21(21):8176. https://doi.org/10.3390/ijms21218176
Chicago/Turabian StyleWnuk, Maciej, Piotr Slipek, Mateusz Dziedzic, and Anna Lewinska. 2020. "The Roles of Host 5-Methylcytosine RNA Methyltransferases during Viral Infections" International Journal of Molecular Sciences 21, no. 21: 8176. https://doi.org/10.3390/ijms21218176
APA StyleWnuk, M., Slipek, P., Dziedzic, M., & Lewinska, A. (2020). The Roles of Host 5-Methylcytosine RNA Methyltransferases during Viral Infections. International Journal of Molecular Sciences, 21(21), 8176. https://doi.org/10.3390/ijms21218176





