Comparison of Dietary Oils with Different Polyunsaturated Fatty Acid n-3 and n-6 Content in the Rat Model of Cutaneous Wound Healing
Abstract
:1. Introduction
2. Results
2.1. EPA/DHA Content in the Mebolically Active Adipose Tissue Was Affected by Fish Oil and Schizochytrium Extract Diet
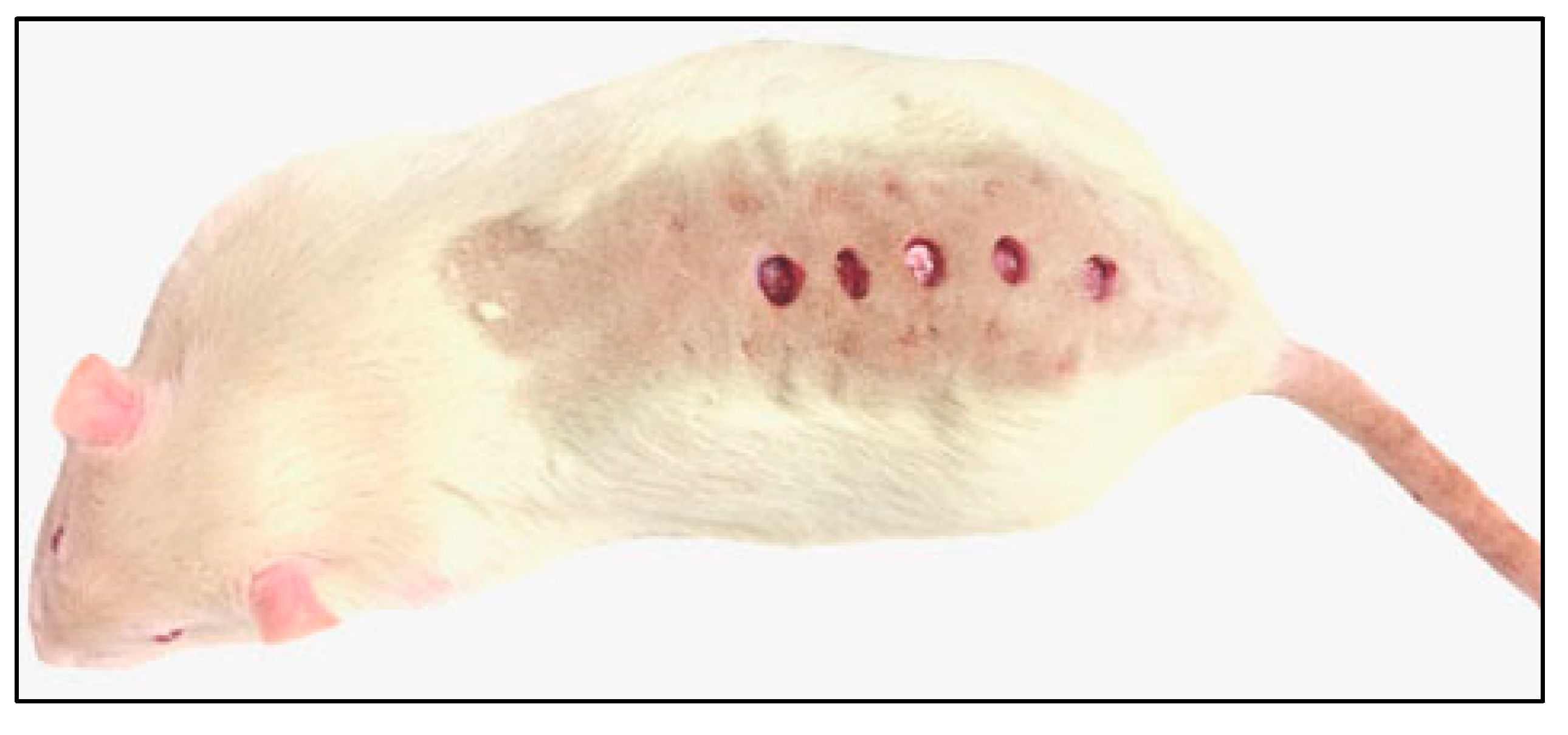
2.2. Wound Size Was Reduced in Schizochytrium Extract Treated Animals
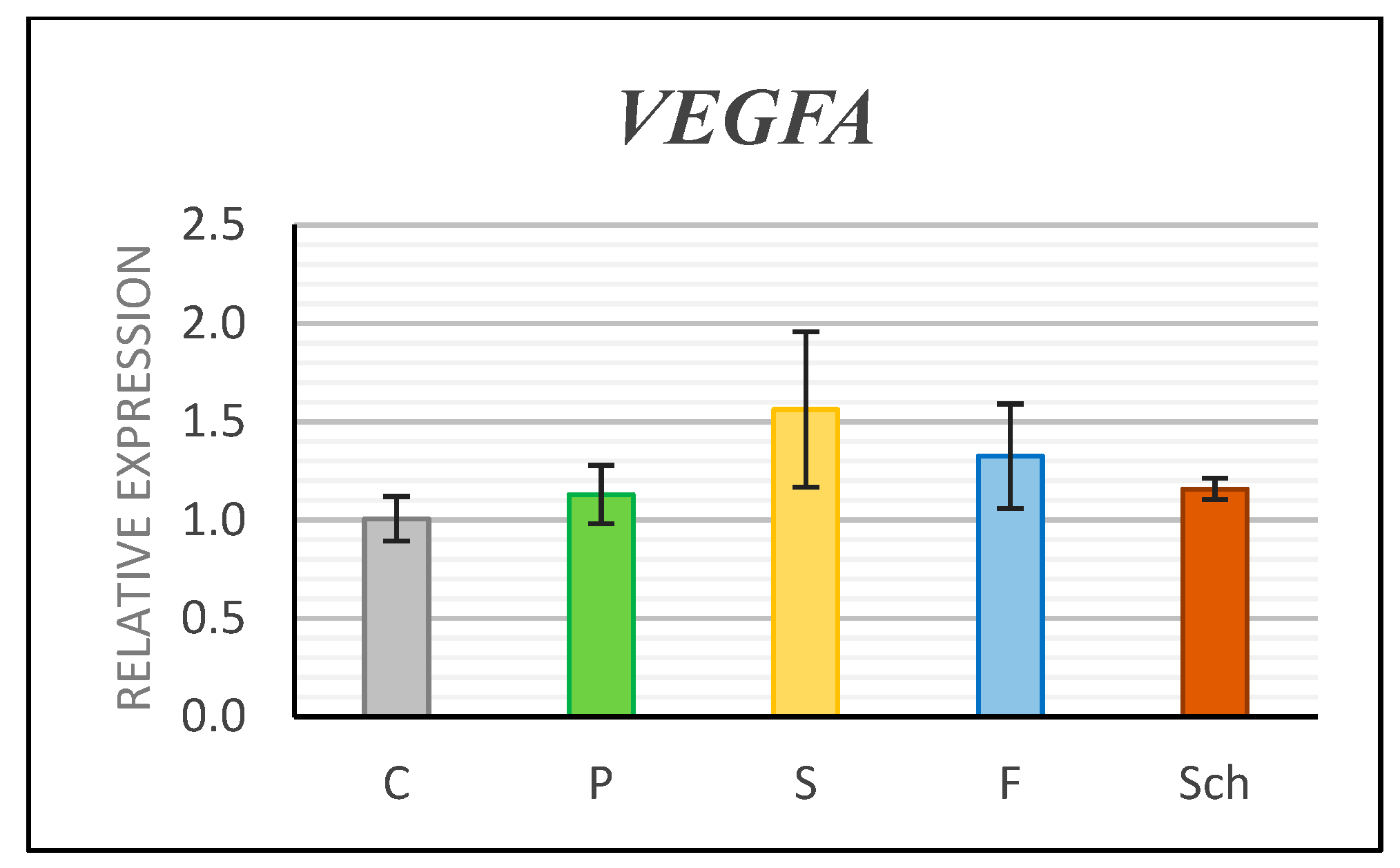
2.3. Angiogenesis Was Enhanced after Schizochytrium Extract Treatment
2.4. Fish Oil Increased Inflammatory Cells Infiltrate
2.5. Maturation of Collagenous Tissue in Dermis Following the Intake of Oil Supplemented Diet
2.6. MALDI-TOF Mass Spectrometry Analysis of Collagen III Fragment Revealed its Decrease in Fish Oil Supplementation
3. Discussion
3.1. Availability of Polyunsaturated Fatty Acids for Wound Healing
3.2. Effect of Polyunsaturated Fatty Acids n-3 on Wound Healing
3.3. Angiogenesis and Inflammation Are Affected by Oil Supplementation during Skin Healing
3.4. Dermis Healing and Collagenous Tissue Maturation
4. Materials and Methods
4.1. Chemicals
4.2. Animals, Dietary Interventions
4.3. Excisions, Sample Collection
4.4. Determination of Fatty Acids Content in Individual Diets
4.5. Quantification of Gene Expressions
4.6. Determination of Hydroxyproline Amount
4.7. Assessment of the Wound Extent
4.8. Immunohistochemistry of Immune Cells and Special Staining of Collagen Fibers
4.9. MALDI-TOF MSI
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Caetano, G.F.; Fronza, M.; Leite, M.N.; Gomes, A.; Frade, M.A. Comparison of collagen content in skin wounds evaluated by biochemical assay and by computer-aided histomorphometric analysis. Pharm. Biol. 2016, 54, 2555–2559. [Google Scholar] [CrossRef] [PubMed]
- Gerçek, A.; Yildirim, O.; Konya, D.; Bozkurt, S.U.; Ozgen, S.; Kiliç, T.; Sav, A.; Pamir, N.; Kılıç, T. Effects of Parenteral Fish-Oil Emulsion (Omegaven) on Cutaneous Wound Healing in Rats Treated With Dexamethasone. J. Parenter. Enter. Nutr. 2007, 31, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Wound repair and regeneration. Nat. Cell Biol. 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, G.; Ecker, J. The opposing effects of n−3 and n−6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef]
- Komprda, T. Eicosapentaenoic and docosahexaenoic acids as inflammation-modulating and lipid homeostasis influencing nutraceuticals: A review. J. Funct. Foods 2012, 4, 25–38. [Google Scholar] [CrossRef]
- Siriwardhana, N.; Kalupahana, N.S.; Cekanova, M.; Lemieux, M.; Greer, B.; Moustaid-Moussa, N. Modulation of adipose tissue inflammation by bioactive food compounds. J. Nutr. Biochem. 2013, 24, 613–623. [Google Scholar] [CrossRef]
- Flock, M.R.; Rogers, C.J.; Prabhu, K.S.; Kris-Etherton, P. Immunometabolic role of long-chain omega-3 fatty acids in obesity-induced inflammation. Diabetes/Metab. Res. Rev. 2013, 29, 431–445. [Google Scholar] [CrossRef]
- Oliver, E.; McGillicuddy, F.C.; Phillips, C.M.; Toomey, S.; Roche, H.M. The role of inflammation and macrophage accumulation in the development of obesity-induced type 2 diabetes mellitus and the possible therapeutic effects of long-chainn-3 PUFA. Proc. Nutr. Soc. 2010, 69, 232–243. [Google Scholar] [CrossRef] [Green Version]
- Serhan, C.N.; Dalli, J.; Colas, R.A.; Winkler, J.W.; Chiang, N. Protectins and maresins: New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2015, 1851, 397–413. [Google Scholar] [CrossRef] [Green Version]
- Serhan, C.N.; Savill, J. Resolution of inflammation: The beginning programs the end. Nat. Immunol. 2005, 6, 1191–1197. [Google Scholar] [CrossRef]
- Arita, M.; Yoshida, M.; Hong, S.; Tjonahen, E.; Glickman, J.N.; Petasis, N.A.; Blumberg, R.S.; Serhan, C.N. Resolvin E1, an endogenous lipid mediator derived from omega-3 eicosapentaenoic acid, protects against 2,4,6-trinitrobenzene sulfonic acid-induced colitis. Proc. Natl. Acad. Sci. USA 2005, 102, 7671–7676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cranmer-Byng, M.M.; Liddle, D.M.; De Boer, A.A.; Monk, J.M.; Robinson, L.E. Pro-inflammatory effects of arachidonic acid in a lipopolysaccharide-induced inflammatory microenvironment in 3T3-L1 adipocytes in vitro. Appl. Physiol. Nutr. Metab. 2015, 40, 142–154. [Google Scholar] [CrossRef] [PubMed]
- Romacho, T.; Glosse, P.; Richter, I.; Elsen, M.; Schoemaker, M.H.; Van Tol, E.A.; Eckel, J. Nutritional ingredients modulate adipokine secretion and inflammation in human primary adipocytes. Nutrients 2015, 7, 865–886. [Google Scholar] [CrossRef] [PubMed]
- De Boer, A.A.; Monk, J.M.; Robinson, L.E. Docosahexaenoic Acid Decreases Pro-Inflammatory Mediators in an In Vitro Murine Adipocyte Macrophage Co-Culture Model. PLoS ONE 2014, 9, e85037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McDaniel, J.C.; Belury, M.; Ahijevych, K.; Blakely, W. Omega-3 fatty acids effect on wound healing. Wound Repair Regen. 2008, 16, 337–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komprda, T. Effect of n-3 long-chain polyunsaturated fatty acids on wound healing using animal models—A review. Acta Vet. Brno 2018, 87, 309–320. [Google Scholar] [CrossRef] [Green Version]
- Otranto, M.; Msc, A.P.D.N.; Monte-Alto-Costa, A. Effects of supplementation with different edible oils on cutaneous wound healing. Wound Repair Regen. 2010, 18, 629–636. [Google Scholar] [CrossRef]
- Bsc, A.D.S.R.; Bsc, L.G.B.; Monte-Alto-Costa, A.; Romana-Souza, B. Supplementation with olive oil, but not fish oil, improves cutaneous wound healing in stressed mice. Wound Repair Regen. 2014, 22, 537–547. [Google Scholar] [CrossRef]
- Drzymała-Czyż, S.; Banasiewicz, T.; Tubacka, M.; Tarasiuk-Rusek, A.; Majewski, P.; Drews, M.; Walkowiak, J. Discrepancy between clinical and histological effects of DHA supplementation in a rat model of pouchitis. Folia Histochem. Cytobiol. 2012, 50, 125–129. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.-Y.; Lin, S.-Y.; Pan, H.-C.; Liao, S.-L.; Chuang, Y.-H.; Yen, Y.-J.; Lin, S.-Y.; Chen, C.-J. Beneficial effect of docosahexaenoic acid on cholestatic liver injury in rats. J. Nutr. Biochem. 2012, 23, 252–264. [Google Scholar] [CrossRef]
- Ekçi, B.; Karabicak, I.; Atukeren, P.; Altinlio, E.; Tomaoglu, K.; Tasci, I. The effect of omega-3 fatty acid and ascorbic acid on healing of ischemic colon anastomoses. Ann. Ital. Chir. 2012, 82, 475–479. [Google Scholar]
- Figueroa, J.D.; Cordero, K.; Baldeosingh, K.; Torrado, A.I.; Walker, R.L.; Miranda, J.D.; De Leon, M. Docosahexaenoic Acid Pretreatment Confers Protection and Functional Improvements after Acute Spinal Cord Injury in Adult Rats. J. Neurotrauma 2012, 29, 551–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olson, M.V.; Liu, Y.-C.; Dangi, B.; Zimmer, J.P.; Salem, N.; Nauroth, J.M. Docosahexaenoic acid reduces inflammation and joint destruction in mice with collagen-induced arthritis. Inflamm. Res. 2013, 62, 1003–1013. [Google Scholar] [CrossRef] [PubMed]
- Abbott, S.K.; Else, P.L.; Atkins, T.A.; Hulbert, A. Fatty acid composition of membrane bilayers: Importance of diet polyunsaturated fat balance. Biochim. Biophys. Acta (BBA)-Biomembr. 2012, 1818, 1309–1317. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.-C.; Yang, F.-L.; Subeq, Y.-M.; Tien, C.-C.; Chao, Y.-F.C.; Lee, R.-P. Lipid Emulsion Enriched in Omega-3 PUFA Accelerates Wound Healing: A Placebo-Controlled Animal Study. World J. Surg. 2017, 42, 1714–1720. [Google Scholar] [CrossRef]
- Kuda, O. Bioactive metabolites of docosahexaenoic acid. Biochimie 2017, 136, 12–20. [Google Scholar] [CrossRef]
- Busuioc, C.J.; Popescu, F.C.; Mogoşanu, G.D.; Lascăr, I.; Pirici, I.; Pop, O.T.; Mogoantă, L. Angiogenesis assessment in experimental third degree skin burns: A histological and immunohistochemical study. Rom. J. Morphol. Embryol. 2011, 52, 887–895. [Google Scholar]
- Finnson, K.W.; McLean, S.; Di Guglielmo, G.M.; Philip, A. Dynamics of Transforming Growth Factor Beta Signaling in Wound Healing and Scarring. Adv. Wound Care 2013, 2, 195–214. [Google Scholar] [CrossRef] [Green Version]
- Rezaii, M.; Oryan, S.; Javeri, A. Curcumin nanoparticles incorporated collagen-chitosan scaffold promotes cutaneous wound healing through regulation of TGF-β1/Smad7 gene expression. Mater. Sci. Eng. C 2019, 98, 347–357. [Google Scholar] [CrossRef]
- Watanabe, S.; Hiraoka, Y.; Endo, S.; Tanimoto, Y.; Tozawa, Y.; Watanabe, Y. An enzymatic method to estimate the content of L-hydroxyproline. J. Biotechnol. 2015, 199, 9–16. [Google Scholar] [CrossRef] [Green Version]
- Sasidharan, S.; Logeswaran, S.; Latha, L.Y. Wound Healing Activity of Elaeis guineensis Leaf Extract Ointment. Int. J. Mol. Sci. 2011, 13, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Lei, Z.-Y.; Chen, J.-J.; Cao, Z.-J.; Ao, M.; Yu, L. Efficacy of Aeschynomene indica L. leaves for wound healing and isolation of active constituent. J. Ethnopharmacol. 2019, 228, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Haukipuro, K.; Melkkocand, J.; Risteli, L.; Kairaluoma, M.I.; Risteli, J. Synthesis of Type I Collagen in Healing Wounds in Humans. Ann. Surg. 1991, 213, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Guran, R.; Zitka, O.; Rodrigo, M.A.M.; Adam, V.; Kizek, R. The maldi imaging for study of the physiological processes in tumors. Chem. Listy 2016, 110, 106–111. [Google Scholar]
- Guran, R.; Vaníčková, L.; Horak, V.; Krizkova, S.; Michalek, P.; Heger, Z.; Zitka, O.; Adam, V. MALDI MSI of MeLiM melanoma: Searching for differences in protein profiles. PLoS ONE 2017, 12, e0189305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewis, E.E.L.; Barrett, M.R.T.; Freeman-Parry, L.; Bojar, R.A.; Clench, M.R. Examination of the skin barrier repair/wound healing process using a living skin equivalent model and matrix-assisted laser desorption-ionization-mass spectrometry imaging. Int. J. Cosmet. Sci. 2018, 40, 148–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komprda, T.; Sládek, Z.; Švehlová, V.; Lacková, Z.; Guráň, R.; Do, T.; Wijacki, J.; Buchtová, M.; Neuwirthová, J.; Gál, B.; et al. The effect of different fatty acid sources on wound healing in rats assessed by matrix-assisted-laser-desorption-ionization mass-spectroscopy-imaging. Acta Vet. Brno 2019, 88, 443–449. [Google Scholar] [CrossRef]
- Pietrowska, M.; Gawin, M.; Polanska, J.; Widlak, P. Tissue fixed with formalin and processed without paraffin embedding is suitable for imaging of both peptides and lipids by MALDI-IMS. Proteomics 2016, 16, 1670–1677. [Google Scholar] [CrossRef]
- Komprda, T.; Rozíková, V.; Zamazalová, N.; Škultéty, O.; Vícenová, M.; Trčková, M.; Faldyna, M. Effect of dietary fish oil on fatty acid deposition and expression of cholesterol homeostasis controlling genes in the liver and plasma lipid profile: Comparison of two animal models. J. Anim. Physiol. Anim. Nutr. 2016, 101, 1093–1102. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- Rizzo, A.E.; Beckett, L.A.; Baier, B.S.; Isseroff, R.R. The linear excisional wound: An improved model for human ex vivo wound epithelialization studies. Ski. Res. Technol. 2011, 18, 125–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maia-Figueiró, T.L.; Odashiro, A.N.; De Menezes, G.P.; Coelho, L.R.; Breda, I.; De Souza, B.A.; Figueiró-Filho, E.A. Semi-Quantitative Histological Analysis of the Effect of Intense Pulsed Light (IPL) and Carbon Dioxide (CO2) Intradermic Injection on Fibroblast and Collagen Proliferation in the Skin of Wistar Rats. J. Cosmet. Dermatol. Sci. Appl. 2012, 2, 164–173. [Google Scholar] [CrossRef] [Green Version]
- Heger, Z.; Polanska, H.; Rodrigo, M.A.M.; Guran, R.; Kulich, P.; Kopel, P.; Masarik, M.; Eckschlager, T.; Stiborova, M.; Kizek, R.; et al. Prostate tumor attenuation in the nu/nu murine model due to anti-sarcosine antibodies in folate-targeted liposomes. Sci. Rep. 2016, 6, 33379. [Google Scholar] [CrossRef] [PubMed] [Green Version]












| Superficial Layer | Deep Layer | |||||||
|---|---|---|---|---|---|---|---|---|
| Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 1 | Sample 2 | Sample 3 | Sample 4 | |
| control | ++ | +++ | ++ | ++ | +++ | ++++ | ++ | ++ |
| palm oil | ++ | ++ | ++ | + | +++ | +++ | +++ | ++ |
| safflower oil | ++++ | +++ | ++++ | ++ | +++ | +++ | ++ | +++ |
| fish oil | ++ | ++++ | +++ | ++ | +++ | +++ | ++++ | ++ |
| Schizochytrium extract | ++ | ++ | +++ | +++ | +++ | +++ | +++ | ++ |
| Fatty Acid | Diet (% of the Sum of Total Fatty Acids) | ||||
|---|---|---|---|---|---|
| C | P | S | F | Sch | |
| 14:0 | 0.3 | 0.9 | 0.4 | 0.1 | 3.7 |
| 16:0 | 23.4 | 42.0 | 22.5 | 19.4 | 15.7 |
| 17:0 | 0.2 | 0.2 | 0.3 | 0.6 | 0.2 |
| 18:0 | 4.7 | 5.3 | 6.5 | 3.7 | 2.0 |
| 16:1n-7 | 0.5 | 0.2 | 0.5 | 8.3 | 0.3 |
| 18:1n-9 | 56.2 | 44.9 | 48.6 | 33.4 | 25.0 |
| 18:2n-6 | 9.4 | 2.2 | 17.3 | 2.0 | 2.3 |
| 18:3n-6 | 4.1 | 3.2 | 2.5 | 4.5 | 1.3 |
| 20:2n-6 | 0.1 | 0.0 | 0.1 | 0.4 | 0.1 |
| 20:3n-6 | 0.0 | 0.0 | 0.0 | 0.1 | 0.3 |
| 20:4n-6 | 0.1 | 0.1 | 0.2 | 0.3 | 0.5 |
| 22:4n-6 | 0.1 | 0.1 | 0.1 | 0.2 | 1.0 |
| 18:3n-3 | 0.1 | 0.1 | 0.0 | 0.3 | 3.3 |
| 20:5n-3 | 0.1 | 0.1 | 0.3 | 8.7 | 0.9 |
| 22:5n-3 | 0.5 | 0.2 | 0.5 | 1.3 | 7.4 |
| 22:6n-3 | 0.2 | 0.5 | 0.2 | 16.7 | 36.0 |
| Primary Antibody | Company | Catalog No. | Host Species | Dilution | Time/Temperature |
|---|---|---|---|---|---|
| alpha-SMA | Abcam, London, UK | ab5694 | rabbit | 1:100 | 60 min/RT |
| MPO | Abcam, London, UK | ab9535 | rabbit | 1:50 | 60 min/RT |
| CD68 | Invitrogen, Carlsbad, CA, USA | PA5-81594 | rabbit | 1:1000 | 75 min/RT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komprda, T.; Sladek, Z.; Sevcikova, Z.; Svehlova, V.; Wijacki, J.; Guran, R.; Do, T.; Lackova, Z.; Polanska, H.; Vrlikova, L.; et al. Comparison of Dietary Oils with Different Polyunsaturated Fatty Acid n-3 and n-6 Content in the Rat Model of Cutaneous Wound Healing. Int. J. Mol. Sci. 2020, 21, 7911. https://doi.org/10.3390/ijms21217911
Komprda T, Sladek Z, Sevcikova Z, Svehlova V, Wijacki J, Guran R, Do T, Lackova Z, Polanska H, Vrlikova L, et al. Comparison of Dietary Oils with Different Polyunsaturated Fatty Acid n-3 and n-6 Content in the Rat Model of Cutaneous Wound Healing. International Journal of Molecular Sciences. 2020; 21(21):7911. https://doi.org/10.3390/ijms21217911
Chicago/Turabian StyleKomprda, Tomas, Zbysek Sladek, Zuzana Sevcikova, Veronika Svehlova, Jan Wijacki, Roman Guran, Tomas Do, Zuzana Lackova, Hana Polanska, Lucie Vrlikova, and et al. 2020. "Comparison of Dietary Oils with Different Polyunsaturated Fatty Acid n-3 and n-6 Content in the Rat Model of Cutaneous Wound Healing" International Journal of Molecular Sciences 21, no. 21: 7911. https://doi.org/10.3390/ijms21217911
APA StyleKomprda, T., Sladek, Z., Sevcikova, Z., Svehlova, V., Wijacki, J., Guran, R., Do, T., Lackova, Z., Polanska, H., Vrlikova, L., Popelkova, V., Michalek, P., Zitka, O., & Buchtova, M. (2020). Comparison of Dietary Oils with Different Polyunsaturated Fatty Acid n-3 and n-6 Content in the Rat Model of Cutaneous Wound Healing. International Journal of Molecular Sciences, 21(21), 7911. https://doi.org/10.3390/ijms21217911







