Effects of Detergent on α-Synuclein Structure: A Native MS-Ion Mobility Study
Abstract
:1. Introduction
2. Results
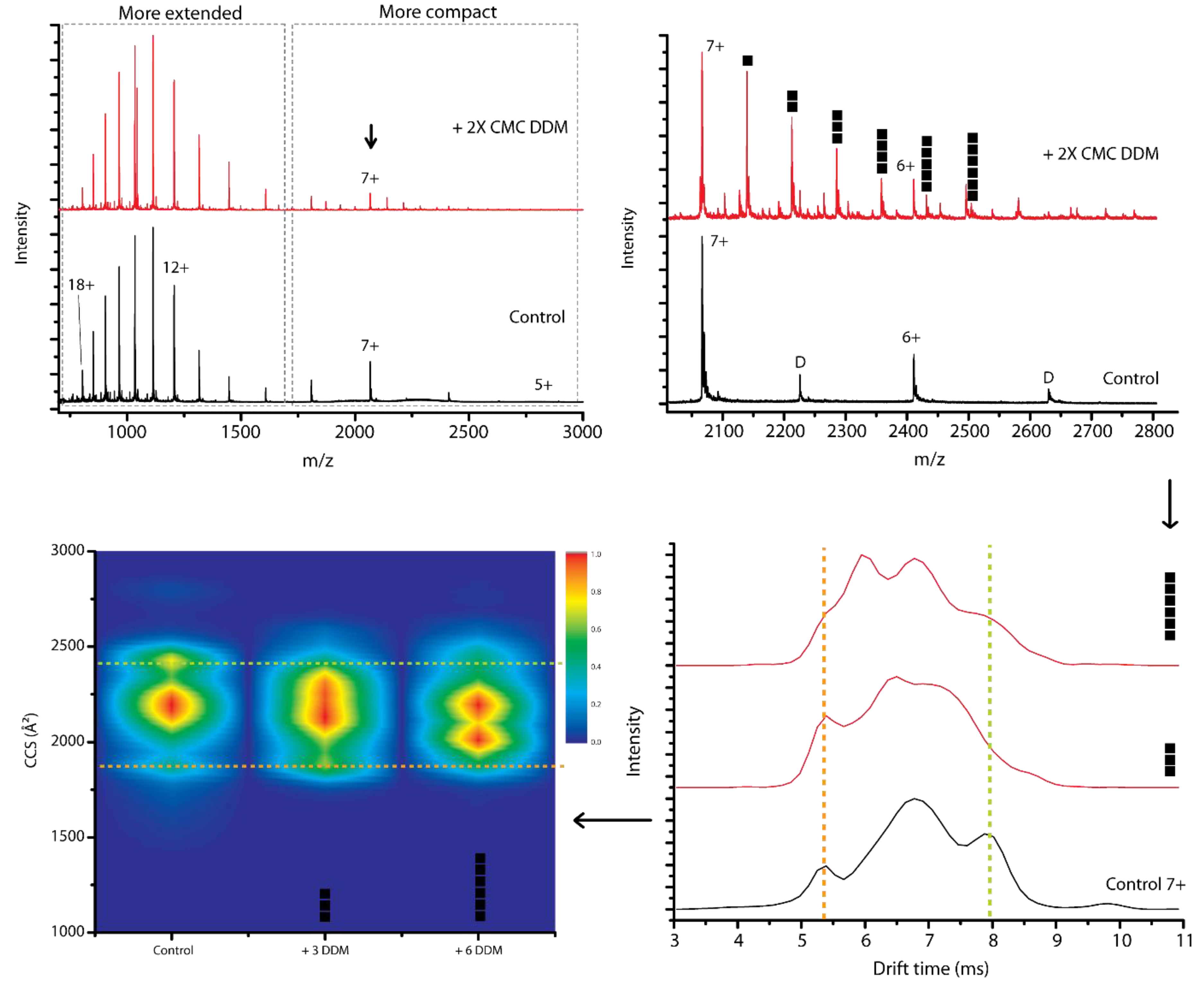
2.1. Interaction of Detergents with α-Synuclein
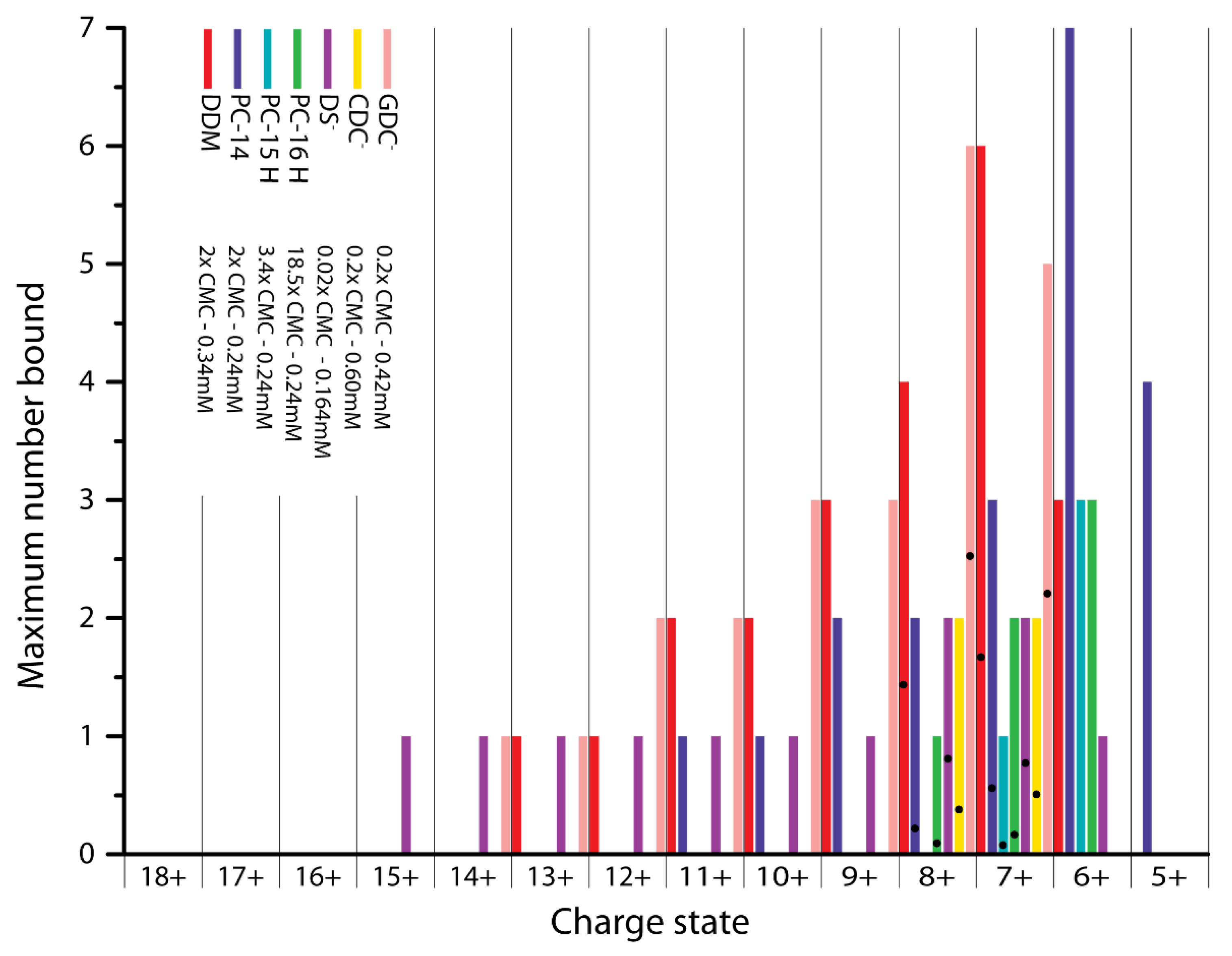
2.2. Detergent Binding Capacity and Stoichiometry
2.3. Conformational Selectivity of Detergent Binding
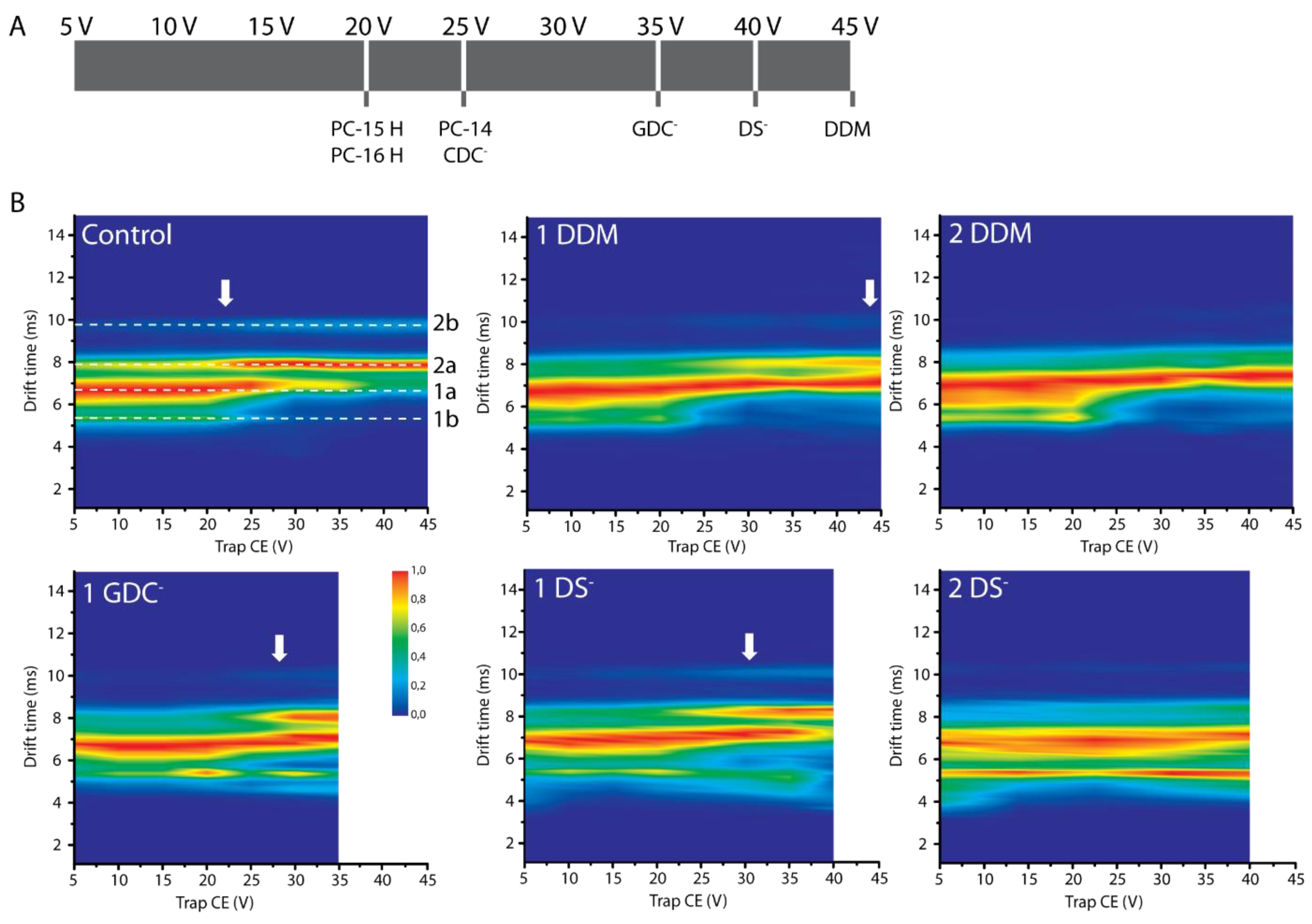
2.4. Conformational Effects of Detergent Binding
2.5. Probing Conformational Transitions
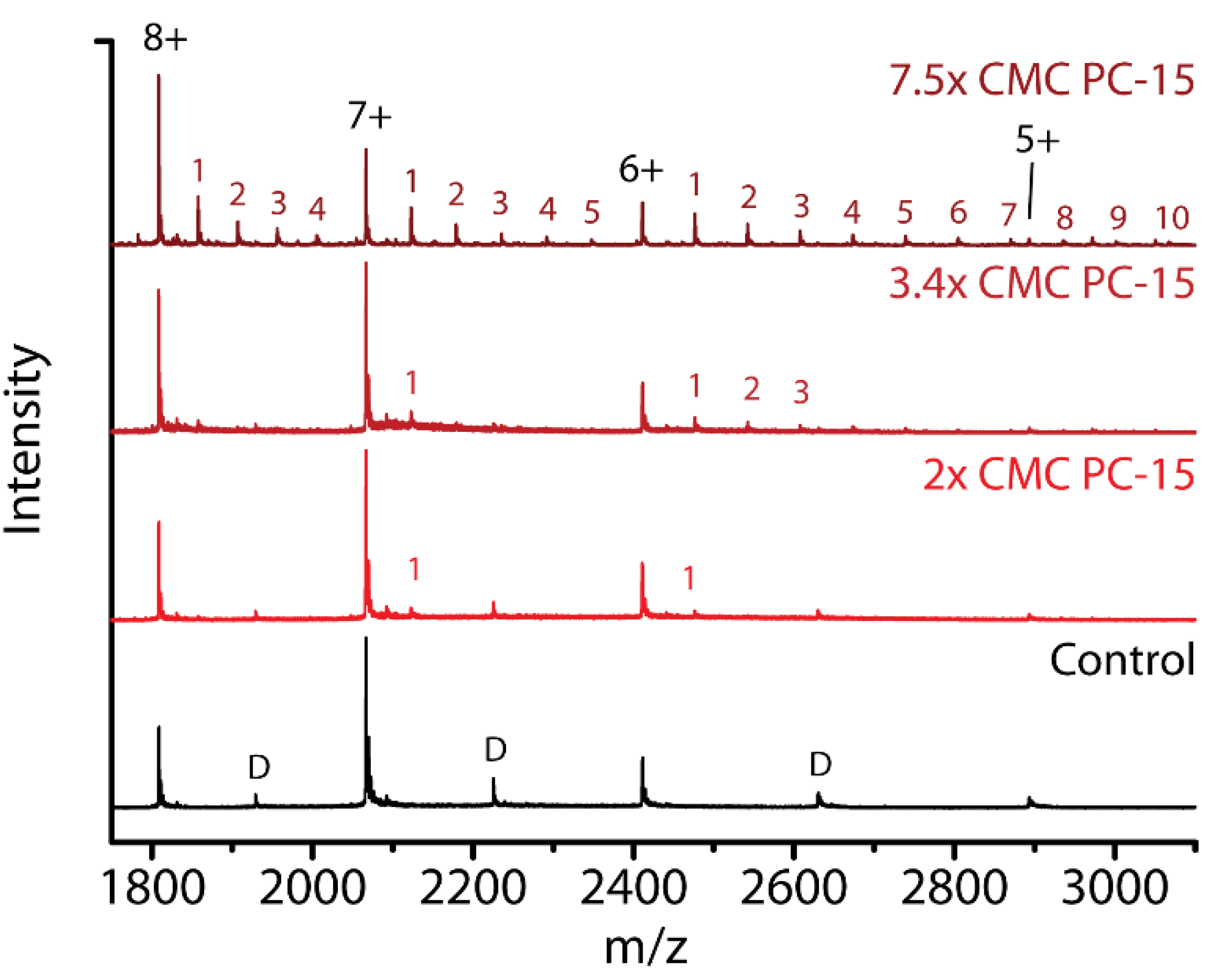
2.6. Detergent Concentration Effects
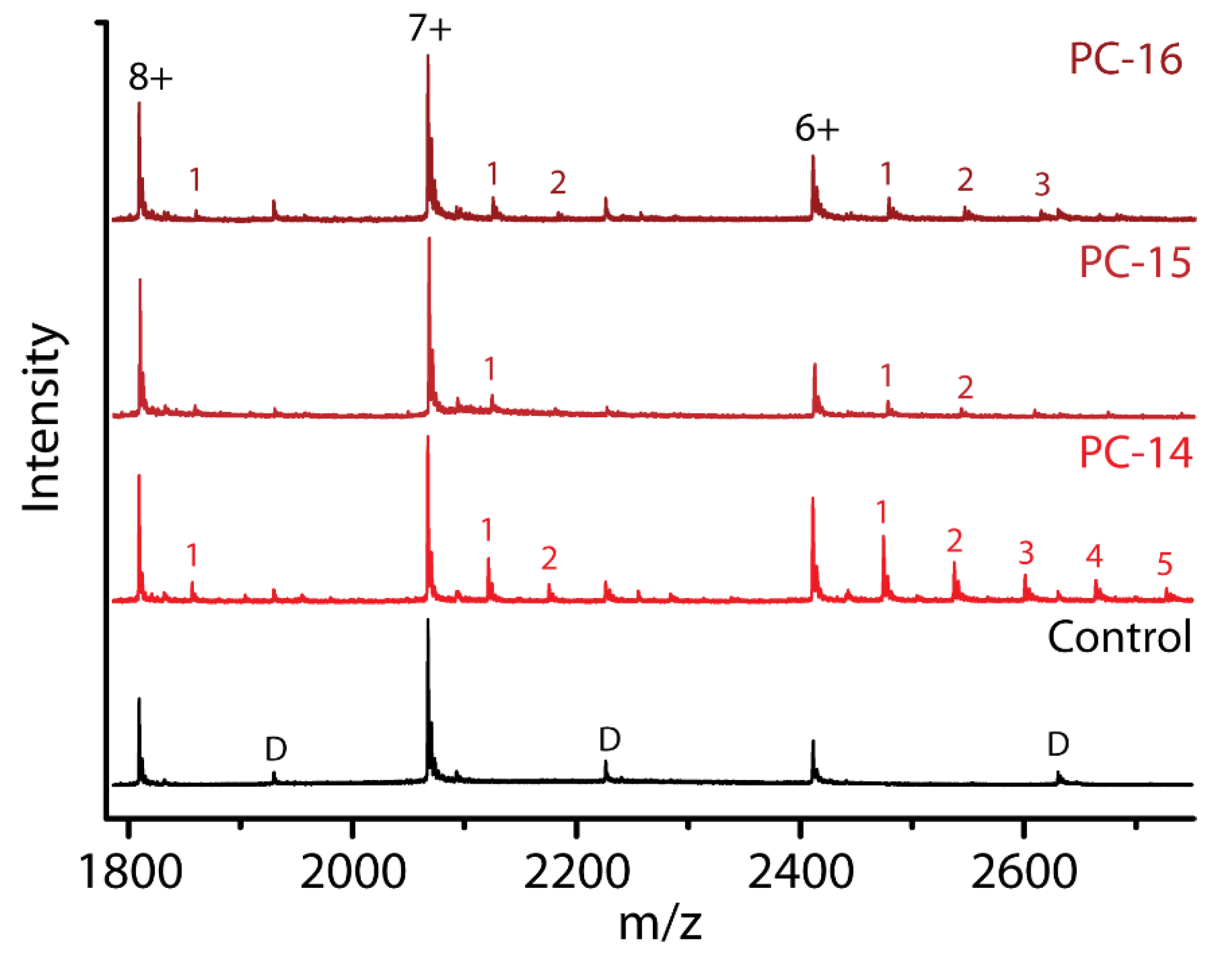
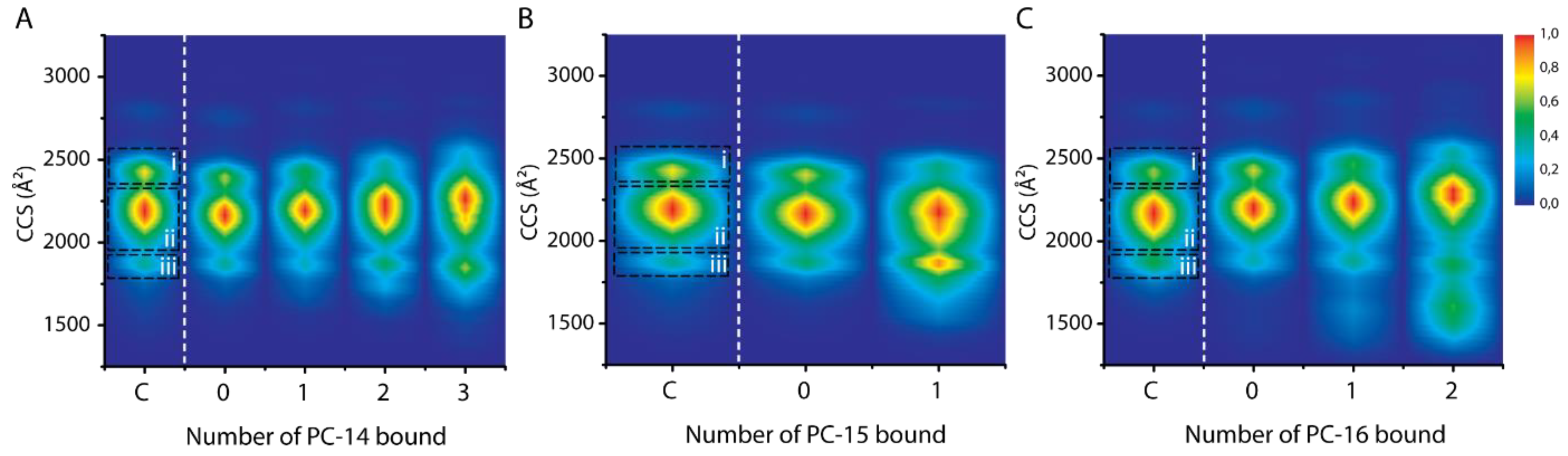
2.7. Effect of Chain Length within the Same Detergent Class
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| α-syn | α-synuclein |
| aa | amino acid |
| PD | Parkinson’s disease |
| NAC | Non-Amyloid-β Component |
| PTM | Post translational modification |
| SMALP | Styrene maleic acid lipid particle |
| SUV | Small unilamellar vesicles |
| CMC | Critical micelle concentration |
| nESI-IM-MS | Nano-electrospray ionisation ion mobility-mass spectrometry |
| CCS | Collision cross section |
| CIU | Collision induced unfolding |
| β-lac | Β-lactoglobulin |
| DDM | n-Dodecyl β-D-maltoside |
| PC-14 | n-tetradecylphosphocholine |
| PC-15 | n-pentadecylphosphocholine |
| PC-16 | n-hexadecylphosphocholine |
| SDS | sodium dodecyl sulfate |
| SCDC | sodium chenodeoxycholate |
| SGDC | sodium glycodeoxycholate |
| CTAB | Cetrimonium bromide |
| SASA | Solvent accessible surface area |
| MD | Molecular dynamics |
| HD | High definition |
References
- Iwai, A.; Masliah, E.; Yoshimoto, M.; Ge, N.; Flanagan, L.; De Silva, H.R.; Kittei, A.; Saitoh, T. The precursor protein of non-A beta component of Alzheimer’s disease amyloid is a presynaptic protein of the central nervous system. Neuron 1995, 14, 467–475. [Google Scholar] [CrossRef] [Green Version]
- Guerrero-Ferreira, R.; Taylor, N.M.; Mona, D.; Ringler, P.; Lauer, M.E.; Riek, R.; Britschgi, M.; Stahlberg, H. Cryo-EM structure of alpha-synuclein fibrils. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Spillantini, M.G.; Schmidt, M.L.; Lee, V.M.; Trojanowski, J.Q.; Jakes, R.; Goedert, M. Alpha-synuclein in Lewy bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef] [PubMed]
- Weinreb, P.H.; Zhen, W.; Poon, A.W.; Conway, K.A.; Lansbury, P.T., Jr. NACP, a protein implicated in Alzheimer’s disease and learning, is natively unfolded. Biochemistry 1996, 35, 13709–13715. [Google Scholar] [CrossRef] [PubMed]
- Mensch, C.; Konijnenberg, A.; Van Elzen, R.; Lambeir, A.M.; Sobott, F.; Johannessen, C. Raman optical activity of human alpha-synuclein in intrinsically disordered, micelle-bound alpha-helical, molten globule and oligomeric beta-sheet state. J. Raman Spectrosc. 2017, 48, 910–918. [Google Scholar] [CrossRef]
- Ulmer, T.S.; Bax, A.; Cole, N.B.; Nussbaum, R.L. Structure and dynamics of micelle-bound human alpha-synuclein. J. Biol. Chem. 2005, 280, 9595–9603. [Google Scholar] [CrossRef] [Green Version]
- Khalaf, O.; Fauvet, B.; Oueslati, A.; Dikiy, I.; Mahul-Mellier, A.L.; Ruggeri, F.S.; Mbefo, M.K.; Vercruysse, F.; Dietler, G.; Lee, S.J.; et al. The H50Q mutation enhances alpha-synuclein aggregation, secretion, and toxicity. J. Biol. Chem. 2014, 289, 21856–21876. [Google Scholar] [CrossRef] [Green Version]
- Kruger, R.; Kuhn, W.; Muller, T.; Woitalla, D.; Graeber, M.; Kosel, S.; Przuntek, H.; Epplen, J.T.; Schols, L.; Riess, O. Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease. Nat. Genet. 1998, 18, 106–108. [Google Scholar] [CrossRef]
- Lesage, S.; Anheim, M.; Letournel, F.; Bousset, L.; Honore, A.; Rozas, N.; Pieri, L.; Madiona, K.; Durr, A.; Melki, R.; et al. G51D alpha-synuclein mutation causes a novel parkinsonian-pyramidal syndrome. Ann. Neurol. 2013, 73, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Zarranz, J.J.; Alegre, J.; Gomez-Esteban, J.C.; Lezcano, E.; Ros, R.; Ampuero, I.; Vidal, L.; Hoenicka, J.; Rodriguez, O.; Atares, B.; et al. The new mutation, E46K, of alpha-synuclein causes Parkinson and Lewy body dementia. Ann. Neurol. 2004, 55, 164–173. [Google Scholar] [CrossRef]
- Mbefo, M.K.; Fares, M.B.; Paleologou, K.; Oueslati, A.; Yin, G.; Tenreiro, S.; Pinto, M.; Outeiro, T.; Zweckstetter, M.; Masliah, E.; et al. Parkinson disease mutant E46K enhances alpha-synuclein phosphorylation in mammalian cell lines, in yeast, and in vivo. J. Biol. Chem. 2015, 290, 9412–9427. [Google Scholar] [CrossRef] [Green Version]
- Oueslati, A. Implication of Alpha-Synuclein Phosphorylation at S129 in Synucleinopathies: What Have We Learned in the Last Decade? J. Parkinsons Dis. 2016, 6, 39–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fusco, G.; Chen, S.W.; Williamson, P.T.F.; Cascella, R.; Perni, M.; Jarvis, J.A.; Cecchi, C.; Vendruscolo, M.; Chiti, F.; Cremades, N.; et al. Structural basis of membrane disruption and cellular toxicity by alpha-synuclein oligomers. Science 2017, 358, 1440–1443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fusco, G.; Sanz-Hernandez, M.; De Simone, A. Order and disorder in the physiological membrane binding of alpha-synuclein. Curr. Opin. Struct. Biol. 2018, 48, 49–57. [Google Scholar] [CrossRef]
- Meade, R.M.; Fairlie, D.P.; Mason, J.M. Alpha-synuclein structure and Parkinson‘s disease-lessons and emerging principles. Mol. Neurodegener. 2019, 14, 29. [Google Scholar] [CrossRef] [Green Version]
- Giannakis, E.; Pacifico, J.; Smith, D.P.; Hung, L.W.; Masters, C.L.; Cappai, R.; Wade, J.D.; Barnham, K.J. Dimeric structures of alpha-synuclein bind preferentially to lipid membranes. Biochim. Biophys. Acta 2008, 1778, 1112–1119. [Google Scholar] [CrossRef] [Green Version]
- Van Rooijen, B.D.; Claessens, M.M.; Subramaniam, V. Lipid bilayer disruption by oligomeric alpha-synuclein depends on bilayer charge and accessibility of the hydrophobic core. Biochim. Biophys. Acta 2009, 1788, 1271–1278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartels, T.; Ahlstrom, L.S.; Leftin, A.; Kamp, F.; Haass, C.; Brown, M.F.; Beyer, K. The N-terminus of the intrinsically disordered protein alpha-synuclein triggers membrane binding and helix folding. Biophys. J. 2010, 99, 2116–2124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.J.; Lee, J.W.; Choi, T.S.; Jin, K.S.; Lee, S.; Ban, C.; Kim, H.I. Probing conformational change of intrinsically disordered alpha-synuclein to helical structures by distinctive regional interactions with lipid membranes. Anal. Chem. 2014, 86, 1909–1916. [Google Scholar] [CrossRef]
- Fusco, G.; De Simone, A.; Arosio, P.; Vendruscolo, M.; Veglia, G.; Dobson, C.M. Structural Ensembles of Membrane-bound alpha-Synuclein Reveal the Molecular Determinants of Synaptic Vesicle Affinity. Sci. Rep. 2016, 6, 27125. [Google Scholar] [CrossRef] [Green Version]
- Lautenschlager, J.; Stephens, A.D.; Fusco, G.; Strohl, F.; Curry, N.; Zacharopoulou, M.; Michel, C.H.; Laine, R.; Nespovitaya, N.; Fantham, M.; et al. C-terminal calcium binding of alpha-synuclein modulates synaptic vesicle interaction. Nat. Commun. 2018, 9, 712. [Google Scholar] [CrossRef] [Green Version]
- Ruf, V.C.; Nubling, G.S.; Willikens, S.; Shi, S.; Schmidt, F.; Levin, J.; Botzel, K.; Kamp, F.; Giese, A. Different Effects of alpha-Synuclein Mutants on Lipid Binding and Aggregation Detected by Single Molecule Fluorescence Spectroscopy and ThT Fluorescence-Based Measurements. ACS Chem. Neurosci. 2019, 10, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- Denisov, I.G.; Sligar, S.G. Nanodiscs for structural and functional studies of membrane proteins. Nat. Struct. Mol. Biol. 2016, 23, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, I.L.; Kemmer, G.C.; Pomorski, T.G. Membrane protein reconstitution into giant unilamellar vesicles: A review on current techniques. Eur. Biophys. J. 2017, 46, 103–119. [Google Scholar] [CrossRef]
- Catoire, L.J.; Warnet, X.L.; Warschawski, D.E. Micelles, Bicelles, Amphipols, Nanodiscs, Liposomes, or Intact Cells: The Hitchhiker’s Guide to the Study of Membrane Proteins by NMR. In Membrane Proteins Production for Structural Analysis; Springer: New York, NY, USA, 2014; pp. 315–345. [Google Scholar]
- Postis, V.; Rawson, S.; Mitchell, J.K.; Lee, S.C.; Parslow, R.A.; Dafforn, T.R.; Baldwin, S.A.; Muench, S.P. The use of SMALPs as a novel membrane protein scaffold for structure study by negative stain electron microscopy. Biochim. Biophys. Acta 2015, 1848, 496–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ehrnhoefer, D.E.; Bieschke, J.; Boeddrich, A.; Herbst, M.; Masino, L.; Lurz, R.; Engemann, S.; Pastore, A.; Wanker, E.E. EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat. Struct. Mol. Biol. 2008, 15, 558–566. [Google Scholar] [CrossRef] [PubMed]
- Calderon, R.O.; Attema, B.; DeVries, G.H. Lipid composition of neuronal cell bodies and neurites from cultured dorsal root ganglia. J. Neurochem. 1995, 64, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Casares, D.; Escriba, P.V.; Rossello, C.A. Membrane Lipid Composition: Effect on Membrane and Organelle Structure, Function and Compartmentalization and Therapeutic Avenues. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [Green Version]
- Lim, L.; Wenk, M.R. Handbook of Neurochemistry and Molecular Neurobiology; Abel Lajtha, G.T., Goracci, G., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; Volume 3, pp. 223–238. [Google Scholar]
- Rosen, M.J. Surfactants and Interfacial Phenomena, 3rd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004; p. 455. [Google Scholar]
- Le Maire, M.; Champeil, P.; Moller, J.V. Interaction of membrane proteins and lipids with solubilizing detergents. Biochim. Biophys. Acta 2000, 1508, 86–111. [Google Scholar] [CrossRef] [Green Version]
- Carey, M.C.; Small, D.M. Micelle formation by bile salts. Physical-chemical and thermodynamic considerations. Arch. Intern. Med. 1972, 130, 506–527. [Google Scholar] [CrossRef]
- Bhuyan, A.K. On the mechanism of SDS-induced protein denaturation. Biopolymers 2010, 93, 186–199. [Google Scholar] [CrossRef]
- Johnson, M. Detergents: Triton X-100, Tween-20, and More. Mater. Methods 2013, 3. [Google Scholar] [CrossRef]
- Bussell, R., Jr.; Ramlall, T.F.; Eliezer, D. Helix periodicity, topology, and dynamics of membrane-associated alpha-synuclein. Protein Sci. 2005, 14, 862–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Necula, M.; Chirita, C.N.; Kuret, J. Rapid anionic micelle-mediated alpha-synuclein fibrillization in vitro. J. Biol. Chem. 2003, 278, 46674–46680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anatrace. Detergents and their uses in membrane protein science. In Detergents and Their Uses in Membrane Protein Science; Anatrace: Maumee, OH, USA; 12p, Available online: http://wolfson.huji.ac.il/purification/PDF/detergents/ANATRACE_Detergent_Book.pdf (accessed on 23 October 2020).
- Wolfe, A.J.; Si, W.; Zhang, Z.; Blanden, A.R.; Hsueh, Y.C.; Gugel, J.F.; Pham, B.; Chen, M.; Loh, S.N.; Rozovsky, S.; et al. Quantification of Membrane Protein-Detergent Complex Interactions. J. Phys. Chem. B 2017, 121, 10228–10241. [Google Scholar] [CrossRef]
- Konijnenberg, A.; Yilmaz, D.; Ingolfsson, H.I.; Dimitrova, A.; Marrink, S.J.; Li, Z.; Venien-Bryan, C.; Sobott, F.; Kocer, A. Global structural changes of an ion channel during its gating are followed by ion mobility mass spectrometry. Proc. Natl. Acad. Sci. USA 2014, 111, 17170–17175. [Google Scholar] [CrossRef] [Green Version]
- Nozaki, Y.; Reynolds, J.A.; Tanford, C. The interaction of a cationic detergent with bovine serum albumin and other proteins. J. Biol. Chem. 1974, 249, 4452–4459. [Google Scholar]
- Borysik, A.J.; Hewitt, D.J.; Robinson, C.V. Detergent release prolongs the lifetime of native-like membrane protein conformations in the gas-phase. J. Am. Chem. Soc. 2013, 135, 6078–6083. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.; Clarke, M.W.; Warnica, J.M.; Boddington, K.F.; Graether, S.P. Structure of an Intrinsically Disordered Stress Protein Alone and Bound to a Membrane Surface. Biophys. J. 2016, 111, 480–491. [Google Scholar] [CrossRef] [Green Version]
- Seddon, A.M.; Curnow, P.; Booth, P.J. Membrane proteins, lipids and detergents: Not just a soap opera. Biochim. Biophys. Acta 2004, 1666, 105–117. [Google Scholar] [CrossRef] [Green Version]
- Moons, R.; Konijnenberg, A.; Mensch, C.; Van Elzen, R.; Johannessen, C.; Maudsley, S.; Lambeir, A.M.; Sobott, F. Metal ions shape α-synuclein. Sci. Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Stephens, A.D.; Zacharopoulou, M.; Moons, R.; Fusco, G.; Seetaloo, N.; Chiki, A.; Woodhams, P.J.; Mela, I.; Lashuel, H.A.; Phillips, J.J.; et al. Extent of N-terminus exposure of monomeric alpha-synuclein determines its aggregation propensity. Nat. Commun. 2020, 11, 2820. [Google Scholar] [CrossRef] [PubMed]
- Dixit, S.M.; Polasky, D.A.; Ruotolo, B.T. Collision induced unfolding of isolated proteins in the gas phase: Past, present, and future. Curr. Opin. Chem. Biol. 2018, 42, 93–100. [Google Scholar] [CrossRef]
- Anatrace Website. Available online: https://www.anatrace.com/Products/Lipids/FOS-CHOLINE (accessed on 22 September 2020).
- Oliver, R.C.; Lipfert, J.; Fox, D.A.; Lo, R.H.; Doniach, S.; Columbus, L. Dependence of micelle size and shape on detergent alkyl chain length and head group. PLoS ONE 2013, 8, e62488. [Google Scholar] [CrossRef]
- Owen, S.C.; Chan, D.P.Y.; Shoichet, M.S. Polymeric micelle stability. Nano Today 2012, 7, 53–65. [Google Scholar] [CrossRef]
- Hassink, G.C.; Raiss, C.C.; Segers-Nolten, I.M.J.; van Wezel, R.J.A.; Subramaniam, V.; le Feber, J.; Claessens, M. Exogenous alpha-synuclein hinders synaptic communication in cultured cortical primary rat neurons. PLoS ONE 2018, 13, e0193763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scollo, F.; Tempra, C.; Lolicato, F.; Sciacca, M.F.M.; Raudino, A.; Milardi, D.; La Rosa, C. Phospholipids Critical Micellar Concentrations Trigger Different Mechanisms of Intrinsically Disordered Proteins Interaction with Model Membranes. J. Phys. Chem. Lett. 2018, 9, 5125–5129. [Google Scholar] [CrossRef]
- Jao, C.C.; Hegde, B.G.; Chen, J.; Haworth, I.S.; Langen, R. Structure of membrane-bound alpha-synuclein from site-directed spin labeling and computational refinement. Proc. Natl. Acad. Sci. USA 2008, 105, 19666–19671. [Google Scholar] [CrossRef] [Green Version]
- Das, T.; Eliezer, D. Membrane interactions of intrinsically disordered proteins: The example of alpha-synuclein. Biochim. Biophys. Acta Proteins Proteom. 2019, 1867, 879–889. [Google Scholar] [CrossRef]
- Van Raaij, M.E.; Segers-Nolten, I.M.; Subramaniam, V. Quantitative morphological analysis reveals ultrastructural diversity of amyloid fibrils from alpha-synuclein mutants. Biophys. J. 2006, 91, L96–L98. [Google Scholar] [CrossRef] [Green Version]
- Bush, M.F.; Hall, Z.; Giles, K.; Hoyes, J.; Robinson, C.V.; Ruotolo, B.T. Collision cross sections of proteins and their complexes: A calibration framework and database for gas-phase structural biology. Anal. Chem. 2010, 82, 9557–9565. [Google Scholar] [CrossRef] [PubMed]
- Haynes, S.E.; Polasky, D.A.; Dixit, S.M.; Majmudar, J.D.; Neeson, K.; Ruotolo, B.T.; Martin, B.R. Variable-Velocity Traveling-Wave Ion Mobility Separation Enhancing Peak Capacity for Data-Independent Acquisition Proteomics. Anal. Chem. 2017, 89, 5669–5672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polasky, D.A.; Dixit, S.M.; Fantin, S.M.; Ruotolo, B.T. CIUSuite 2: Next-Generation Software for the Analysis of Gas-Phase Protein Unfolding Data. Anal. Chem. 2019, 91, 3147–3155. [Google Scholar] [CrossRef] [PubMed]
- Galvagnion, C. The Role of Lipids Interacting with alpha-Synuclein in the Pathogenesis of Parkinson’s Disease. J. Parkinsons Dis. 2017, 7, 433–450. [Google Scholar] [CrossRef] [Green Version]



| Name | CMC (mM) | Standard Concentration (mM) | Standard Concentration (× CMC) | Equal Concentration (mM) | Equal Concentration (× CMC) |
|---|---|---|---|---|---|
| DDM | 0.17 | 0.34 | 2.0 | 0.2 | 1.18 |
| PC-14 | 0.12 | 0.24 | 2.0 | 0.2 | 1.67 |
| PC-15 L | 0.07 | 0.14 | 2.0 | 0.2 | |
| PC-15 H | 0.07 | 0.24 | 3.4 | 0.2 | 2.86 |
| PC-16 L | 0.013 | 0.026 | 2.0 | 0.2 | |
| PC-16 H | 0.013 | 0.24 | 18.5 | 0.2 | 15.38 |
| SDS | 8.2 | 0.164 | 0.02 | 0.2 | 0.024 |
| SCDC | 3.0 | 0.6 | 0.2 | 0.2 | 0.067 |
| SGDC | 2.1 | 0.42 | 0.2 | 0.2 | 0.095 |
| Structure | Name | Type | CMC (mM) | x CMC Used | Molecular Mass (Da) |
|---|---|---|---|---|---|
 | DDM | Non-ionic | 0.170 | 2 | 510.6 |
 | Triton X-100 | Non-ionic | 0.230 | 2 | 650 * |
 | PC-14 | Zwitterionic | 0.120 | 2 | 379.5 |
 | PC-15 | Zwitterionic | 0.070 | 2 & 3.4 | 393.5 |
 | PC-16 | Zwitterionic | 0.013 | 2 & 18.5 | 407.5 |
 | SCDC | Anionic | 3.000 | 0.2 | 414.6 |
 | SGDC | Anionic | 2.100 | 0.2 | 471.6 |
 | SDS | Anionic | 8.200 | 0.02 | 288.4 |
 | CTAB | Cationic | 0.920 | 0.2 | 364.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moons, R.; van der Wekken-de Bruijne, R.; Maudsley, S.; Lemière, F.; Lambeir, A.-M.; Sobott, F. Effects of Detergent on α-Synuclein Structure: A Native MS-Ion Mobility Study. Int. J. Mol. Sci. 2020, 21, 7884. https://doi.org/10.3390/ijms21217884
Moons R, van der Wekken-de Bruijne R, Maudsley S, Lemière F, Lambeir A-M, Sobott F. Effects of Detergent on α-Synuclein Structure: A Native MS-Ion Mobility Study. International Journal of Molecular Sciences. 2020; 21(21):7884. https://doi.org/10.3390/ijms21217884
Chicago/Turabian StyleMoons, Rani, Renate van der Wekken-de Bruijne, Stuart Maudsley, Filip Lemière, Anne-Marie Lambeir, and Frank Sobott. 2020. "Effects of Detergent on α-Synuclein Structure: A Native MS-Ion Mobility Study" International Journal of Molecular Sciences 21, no. 21: 7884. https://doi.org/10.3390/ijms21217884
APA StyleMoons, R., van der Wekken-de Bruijne, R., Maudsley, S., Lemière, F., Lambeir, A.-M., & Sobott, F. (2020). Effects of Detergent on α-Synuclein Structure: A Native MS-Ion Mobility Study. International Journal of Molecular Sciences, 21(21), 7884. https://doi.org/10.3390/ijms21217884





