Perspective: Quality Versus Quantity; Is It Important to Assess the Role of Enhancers in Complex Disease from an In Vivo Perspective?
Abstract
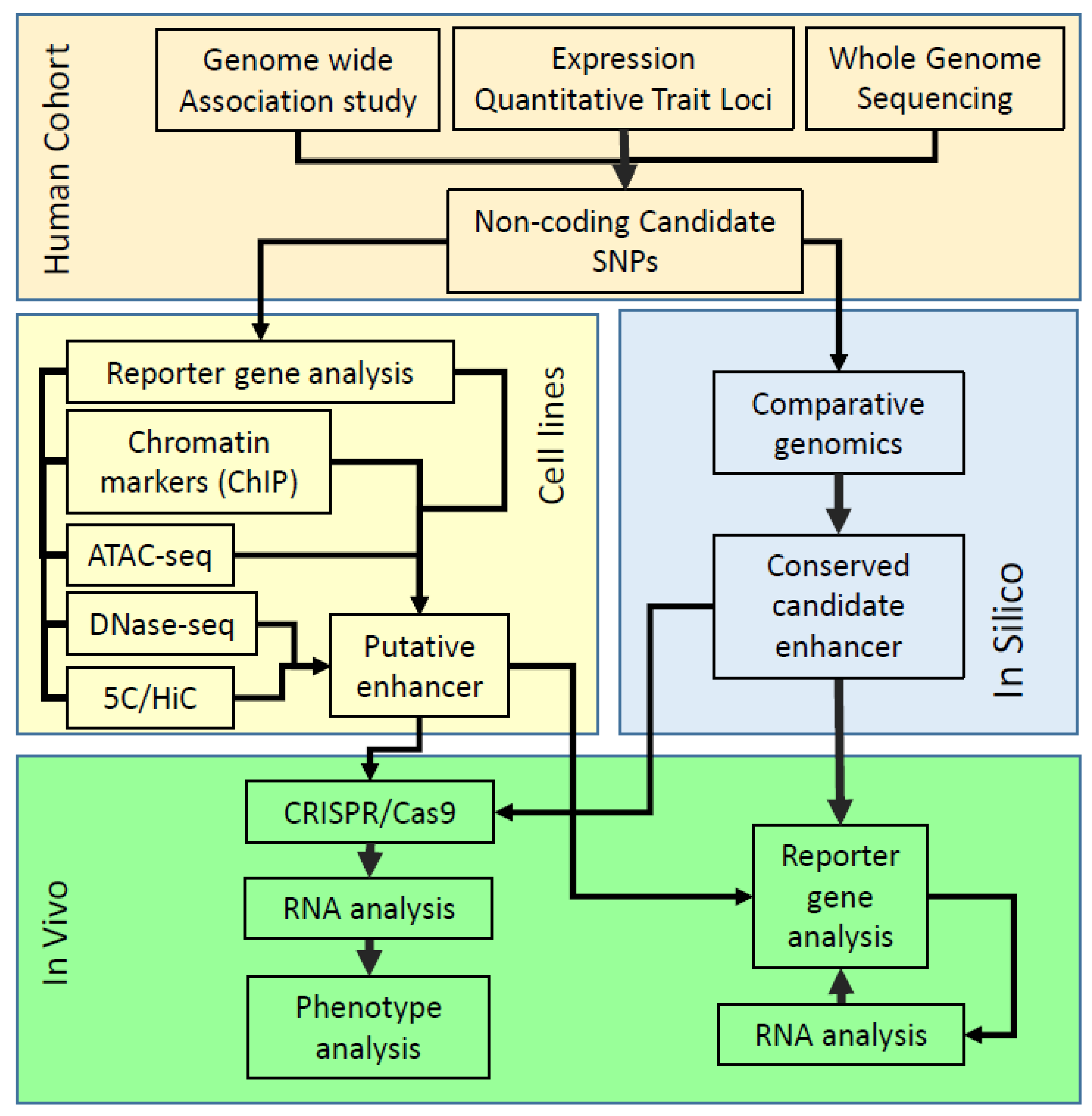
:1. Introduction
What Is the Regulatory Genome?
2. Identifying Enhancer Sequences; Lessons from Developmental Biology
3. Chromatin Markers
4. An Evolutionary Perspective
5. Conserved Enhancers in Adult Brain Activity
6. Gene Regulation and Pharmacogenomics?
7. Enhancers as Future Personalised Drug Targets?
8. Nature Versus Nurture: Epigenetics and the Functional Non-Coding Genome
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Uffelmann, E.; Posthuma, D. Emerging Methods and Resources for Biological Interrogation of Neuropsychiatric Polygenic-Signal. Biol. Psychiatry 2020. [Google Scholar] [CrossRef]
- Mallet, J.; le Strat, Y.; Dubertret, C.; Gorwood, P. Polygenic Risk Scores Shed Light on the Relationship between Schizophrenia and Cognitive Functioning: Review and Meta-Analysis. J. Clin. Med. 2020, 9, 341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hindorff, L.A.; Sethupathy, P.; Junkins, H.A.; Ramos, E.M.; Mehta, J.P.; Collins, F.S.; Manolio, T.A. Potential etiologic and functional implications of genome-wide association loci for human diseases and traits. Proc. Natl. Acad. Sci. USA 2009, 106, 9362–9367. [Google Scholar] [CrossRef] [Green Version]
- Boyle, E.A.; Li, Y.I.; Pritchard, J.K. An Expanded View of Complex Traits: From Polygenic to Omnigenic. Cell 2017, 169, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Danino, Y.M.; Even, D.; Ideses, D.; Juven-Gershon, T. The core promoter: At the heart of gene expression. Biochim. Biophys. Acta 2015, 1849, 1116–1131. [Google Scholar] [CrossRef]
- Juven-Gershon, T.; Kadonaga, J.T. Regulation of gene expression via the core promoter and the basal transcriptional machinery. Dev. Biol. 2009, 339, 225–229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davidson, S.; Macpherson, N.; Mitchell, J.A. Nuclear organization of RNA polymerase II transcription. Biochem. Cell Biol. 2013, 91, 22–30. [Google Scholar] [CrossRef]
- Orekhova, A.S.; Rubtsov, P.M. Bidirectional promoters in the transcription of mammalian genomes. Biochemistry 2013, 78, 335–341. [Google Scholar] [CrossRef]
- Carninci, P.; Sandelin, A.; Lenhard, B.; Katayama, S.; Shimokawa, K.; Ponjavic, J.; Semple, C.A.; Taylor, M.S.; Engstrom, P.G.; Frith, M.C.; et al. Genome-wide analysis of mammalian promoter architecture and evolution. Nat. Genet. 2006, 38, 626–635. [Google Scholar] [CrossRef]
- Natsume-Kitatani, Y.; Mamitsuka, H. Classification of Promoters Based on the Combination of Core Promoter Elements Exhibits Different Histone Modification Patterns. PLoS ONE 2016, 11, e0151917. [Google Scholar] [CrossRef] [Green Version]
- Du, X.; Han, L.; Guo, A.Y.; Zhao, Z. Features of methylation and gene expression in the promoter-associated CpG islands using human methylome data. Comp. Funct. Genom. 2012, 2012, 598987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahna, D.; Puri, S.; Sharma, S. DNA methylation signatures: Biomarkers of drug and alcohol abuse. Mutat. Res. 2018, 777, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Jeziorska, D.M.; Murray, R.J.S.; de Gobbi, M.; Gaentzsch, R.; Garrick, D.; Ayyub, H.; Chen, T.; Li, E.; Telenius, J.; Lynch, M.; et al. DNA methylation of intragenic CpG islands depends on their transcriptional activity during differentiation and disease. Proc. Natl. Acad. Sci. USA 2017, 114, E7526–E7535. [Google Scholar] [CrossRef] [Green Version]
- Chepelev, I.; Wei, G.; Wangsa, D.; Tang, Q.; Zhao, K. Characterization of genome-wide enhancer-promoter interactions reveals co-expression of interacting genes and modes of higher order chromatin organization. Cell Res. 2012, 22, 490–503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dunham, I.; Kundaje, A.; Aldred, S.F.; Collins, P.J.; Davis, C.A.; Doyle, F.; Epstein, C.B.; Frietze, S.; Harrow, J.; Kaul, R.; et al. An integrated encyclopedia of DNA elements in the human genome. Nature 2012, 489, 57–74. [Google Scholar]
- Benoist, C.; Chambon, P. In vivo sequence requirements of the SV40 early promotor region. Nature 1981, 290, 304–310. [Google Scholar] [CrossRef]
- Sebe-Pedros, A.; Ballare, C.; Parra-Acero, H.; Chiva, C.; Tena, J.J.; Sabido, E.; Gomez-Skarmeta, J.L.; di Croce, L.; Ruiz-Trillo, I. The Dynamic Regulatory Genome of Capsaspora and the Origin of Animal Multicellularity. Cell 2016, 165, 1224–1237. [Google Scholar] [CrossRef] [Green Version]
- Lettice, L.A.; Devenney, P.; de Angelis, C.; Hill, R.E. The Conserved Sonic Hedgehog Limb Enhancer Consists of Discrete Functional Elements that Regulate Precise Spatial Expression. Cell Rep. 2017, 20, 1396–1408. [Google Scholar] [CrossRef] [Green Version]
- Long, H.K.; Osterwalder, M.; Welsh, I.C.; Hansen, K.; Davies, J.O.J.; Liu, Y.E.; Koska, M.; Adams, A.T.; Aho, R.; Arora, N.; et al. Loss of Extreme Long-Range Enhancers in Human Neural Crest Drives a Craniofacial Disorder. Cell Stem Cell 2020. [Google Scholar] [CrossRef]
- Sikder, S.K.; Mitra, D.; Laurence, J. Identification of a novel cell-type and context specific enhancer within the negative regulatory element of the human immunodeficiency virus type 1 long terminal repeat. Arch. Virol. 1994, 137, 139–147. [Google Scholar] [CrossRef]
- MacKenzie, A.; Hing, B.; Davidson, S. Exploring the effects of polymorphisms on cis-regulatory signal transduction response. Trends Mol. Med. 2012, 19, 99–107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farley, E.K.; Olson, K.M.; Zhang, W.; Rokhsar, D.S.; Levine, M.S. Syntax compensates for poor binding sites to encode tissue specificity of developmental enhancers. Proc. Natl. Acad. Sci. USA 2016, 113, 6508–6513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farley, E.K.; Olson, K.M.; Zhang, W.; Brandt, A.J.; Rokhsar, D.S.; Levine, M.S. Suboptimization of developmental enhancers. Science 2015, 350, 325–328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farley, E.K.; Olson, K.M.; Levine, M.S. Regulatory Principles Governing Tissue Specificity of Developmental Enhancers. Cold Spring Harb. Symp. Quant. Biol. 2015, 80, 27–32. [Google Scholar] [CrossRef] [Green Version]
- Spurrell, C.H.; Dickel, D.E.; Visel, A. The Ties That Bind: Mapping the Dynamic Enhancer-Promoter Interactome. Cell 2016, 167, 1163–1166. [Google Scholar] [CrossRef]
- Spitz, F. Gene regulation at a distance: From remote enhancers to 3D regulatory ensembles. Semin. Cell Dev. Biol. 2016, 57, 57–67. [Google Scholar] [CrossRef]
- Rada-Iglesias, A.; Grosveld, F.G.; Papantonis, A. Forces driving the three-dimensional folding of eukaryotic genomes. Mol. Syst. Biol. 2018, 14, e8214. [Google Scholar] [CrossRef]
- Rowley, M.J.; Corces, V.G. Organizational principles of 3D genome architecture. Nat. Rev. Genet. 2018, 19, 789–800. [Google Scholar] [CrossRef]
- Williamson, I.; Kane, L.; Devenney, P.S.; Flyamer, I.M.; Anderson, E.; Kilanowski, F.; Hill, R.E.; Bickmore, W.A.; Lettice, L.A. Developmentally regulated Shh expression is robust to TAD perturbations. Development 2019, 146, dev179523. [Google Scholar] [CrossRef] [Green Version]
- Hnisz, D.; Weintraub, A.S.; Day, D.S.; Valton, A.L.; Bak, R.O.; Li, C.H.; Goldmann, J.; Lajoie, B.R.; Fan, Z.P.; Sigova, A.A.; et al. Activation of proto-oncogenes by disruption of chromosome neighborhoods. Science 2016, 351, 1454–1458. [Google Scholar] [CrossRef] [Green Version]
- Furlong, E.E.M.; Levine, M. Developmental enhancers and chromosome topology. Science 2018, 361, 1341–1345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, S.; Bohl, D.; Li, C.; Tuan, D. Transcription of the HS2 enhancer toward a cis-linked gene is independent of the orientation, position, and distance of the enhancer relative to the gene. Mol. Cell. Biol. 1997, 17, 3955–3965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murakawa, Y.; Yoshihara, M.; Kawaji, H.; Nishikawa, M.; Zayed, H.; Suzuki, H.; Fantom, C.; Hayashizaki, Y. Enhanced Identification of Transcriptional Enhancers Provides Mechanistic Insights into Diseases. Trends Genet. 2016, 32, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Creyghton, M.P.; Cheng, A.W.; Welstead, G.G.; Kooistra, T.; Carey, B.W.; Steine, E.J.; Hanna, J.; Lodato, M.A.; Frampton, G.M.; Sharp, P.A.; et al. Histone H3K27ac separates active from poised enhancers and predicts developmental state. Proc. Natl. Acad. Sci. USA 2010, 107, 21931–21936. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holmqvist, P.H.; Mannervik, M. Genomic occupancy of the transcriptional co-activators p300 and CBP. Transcription 2013, 4, 18–23. [Google Scholar] [CrossRef] [Green Version]
- Stergachis, A.B.; Neph, S.; Reynolds, A.; Humbert, R.; Miller, B.; Paige, S.L.; Vernot, B.; Cheng, J.B.; Thurman, R.E.; Sandstrom, R.; et al. Developmental fate and cellular maturity encoded in human regulatory DNA landscapes. Cell 2013, 154, 888–903. [Google Scholar] [CrossRef] [Green Version]
- Song, M.; Yang, X.; Ren, X.; Maliskova, L.; Li, B.; Jones, I.R.; Wang, C.; Jacob, F.; Wu, K.; Traglia, M.; et al. Mapping cis-regulatory chromatin contacts in neural cells links neuropsychiatric disorder risk variants to target genes. Nat. Genet. 2019, 51, 1252–1262. [Google Scholar] [CrossRef]
- Graur, D.; Zheng, Y.; Price, N.; Azevedo, R.B.; Zufall, R.A.; Elhaik, E. On the immortality of television sets: “function” in the human genome according to the evolution-free gospel of ENCODE. Genome Biol. Evol. 2013, 5, 578–590. [Google Scholar] [CrossRef] [Green Version]
- Villar, D.; Berthelot, C.; Aldridge, S.; Rayner, T.F.; Lukk, M.; Pignatelli, M.; Park, T.J.; Deaville, R.; Erichsen, J.T.; Jasinska, A.J.; et al. Enhancer evolution across 20 mammalian species. Cell 2015, 160, 554–566. [Google Scholar] [CrossRef] [Green Version]
- Blow, M.J.; McCulley, D.J.; Li, Z.; Zhang, T.; Akiyama, J.A.; Holt, A.; Plajzer-Frick, I.; Shoukry, M.; Wright, C.; Chen, F.; et al. ChIP-Seq identification of weakly conserved heart enhancers. Nat. Genet. 2010, 42, 806–810. [Google Scholar] [CrossRef] [Green Version]
- May, D.; Blow, M.J.; Kaplan, T.; McCulley, D.J.; Jensen, B.C.; Akiyama, J.A.; Holt, A.; Plajzer-Frick, I.; Shoukry, M.; Wright, C.; et al. Large-scale discovery of enhancers from human heart tissue. Nat. Genet. 2011, 44, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Oldridge, D.A.; Wood, A.C.; Weichert-Leahey, N.; Crimmins, I.; Sussman, R.; Winter, C.; McDaniel, L.D.; Diamond, M.; Hart, L.S.; Zhu, S.; et al. Genetic predisposition to neuroblastoma mediated by a LMO1 super-enhancer polymorphism. Nature 2015, 528, 418–421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baggs, J.E.; Hayes, K.R.; Hogenesch, J.B. Comparative genomics as a tool in the understanding of eukaryotic transcriptional regulation. Curr. Opin. Genet. Dev. 2005, 15, 634–639. [Google Scholar] [CrossRef]
- Visel, A.; Bristow, J.; Pennacchio, L.A. Enhancer identification through comparative genomics. Semin. Cell Dev. Biol. 2007, 18, 140–152. [Google Scholar] [CrossRef] [Green Version]
- MacKenzie, A.; Purdie, L.; Davidson, D.; Collinson, M.; Hill, R.E. Two enhancer domains control early aspects of the complex expression pattern of Msx1. Mech. Dev. 1997, 62, 29–40. [Google Scholar] [CrossRef]
- Miller, K.A.; Barrow, J.; Collinson, J.M.; Davidson, S.; Lear, M.; Hill, R.E.; Mackenzie, A. Ahighly conserved Wnt-dependent TCF4 binding site within the proximal enhancer of the anti-myogenic Msx1 gene supports expression within Pax3-expressing limb bud muscle precursor cells. Dev. Biol. 2007, 311, 665–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, K.A.; Davidson, S.; Liaros, A.; Barrow, J.; Lear, M.; Heine, D.; Hoppler, S.; MacKenzie, A. Prediction and characterisation of a highly conserved, remote and cAMP responsive enhancer that regulates Msx1 gene expression in cardiac neural crest and outflow tract. Dev. Biol. 2008, 317, 686–694. [Google Scholar] [CrossRef] [Green Version]
- Grice, J.; Noyvert, B.; Doglio, L.; Elgar, G. A Simple Predictive Enhancer Syntax for Hindbrain Patterning Is Conserved in Vertebrate Genomes. PLoS ONE 2015, 10, e0130413. [Google Scholar] [CrossRef] [Green Version]
- Smith, N.G.; Brandstrom, M.; Ellegren, H. Evidence for turnover of functional noncoding DNA in mammalian genome evolution. Genomics 2004, 84, 806–813. [Google Scholar] [CrossRef]
- Davidson, S.; Starkey, A.; MacKenzie, A. Evidence of uneven selective pressure on different subsets of the conserved human genome; implications for the significance of intronic and intergenic DNA. BMC Genom. 2009, 10, 614. [Google Scholar] [CrossRef] [Green Version]
- Hokfelt, T.; Barde, S.; Xu, Z.D.; Kuteeva, E.; Ruegg, J.; le Maitre, E.; Risling, M.; Kehr, J.; Ihnatko, R.; Theodorsson, E.; et al. Neuropeptide and Small Transmitter Coexistence: Fundamental Studies and Relevance to Mental Illness. Front. Neural. Circuits 2018, 12, 106. [Google Scholar] [CrossRef] [Green Version]
- Alpar, A.; Benevento, M.; Romanov, R.A.; Hokfelt, T.; Harkany, T. Hypothalamic cell diversity: Non-neuronal codes for long-distance volume transmission by neuropeptides. Curr. Opin. Neurobiol. 2019, 56, 16–23. [Google Scholar] [CrossRef]
- Ueda, H. Molecular mechanisms of neuropathic pain-phenotypic switch and initiation mechanisms. Pharmacol. Ther. 2006, 109, 57–77. [Google Scholar] [CrossRef]
- Ebner, K.; Singewald, N. The role of substance P in stress and anxiety responses. Amino Acids 2006, 31, 251–272. [Google Scholar] [CrossRef]
- Davidson, S.; Miller, K.A.; Dowell, A.; Gildea, A.; Mackenzie, A. A remote and highly conserved enhancer supports amygdala specific expression of the gene encoding the anxiogenic neuropeptide substance-P. Mol. Psychiatry 2006, 11, 410–421. [Google Scholar] [CrossRef] [Green Version]
- Shanley, L.; Lear, M.; Davidson, S.; Ross, R.; MacKenzie, A. Evidence for regulatory diversity and auto-regulation at the TAC1 locus in sensory neurones. J. Neuroinflammation. 2011, 8, 10. [Google Scholar] [CrossRef] [Green Version]
- Shanley, L.; Davidson, S.; Lear, M.; Thotakura, A.K.; McEwan, I.J.; Ross, R.A.; MacKenzie, A. Long-range regulatory synergy is required to allow control of the TAC1 locus by MEK/ERK signalling in sensory neurones. Neurosignals 2010, 18, 173–185. [Google Scholar] [CrossRef]
- Hay, C.W.; Shanley, L.; Davidson, S.; Cowie, P.; Lear, M.; McGuffin, P.; Riedel, G.; McEwan, I.J.; MacKenzie, A. Functional effects of polymorphisms on glucocorticoid receptor modulation of human anxiogenic substance-P gene promoter activity in primary amygdala neurones. Psychoneuroendocrinology 2014, 47, 43–55. [Google Scholar] [CrossRef] [Green Version]
- Barson, J.R.; Morganstern, I.; Leibowitz, S.F. Galanin and consummatory behavior: Special relationship with dietary fat, alcohol and circulating lipids. Exp. Suppl. 2010, 102, 87–111. [Google Scholar]
- Davidson, S.; Lear, M.; Shanley, L.; Hing, B.; Baizan-Edge, A.; Herwig, A.; Quinn, J.P.; Breen, G.; McGuffin, P.; Starkey, A.; et al. Differential activity by polymorphic variants of a remote enhancer that supports galanin expression in the hypothalamus and amygdala: Implications for obesity, depression and alcoholism. Neuropsychopharmacology 2011, 36, 2211–2221. [Google Scholar] [CrossRef]
- McEwan, A.R.; Davidson, C.; Hay, E.; Turnbull, Y.; Erickson, J.C.; Marini, P.; Wilson, D.; McIntosh, A.M.; Adams, M.J.; Murgatroyd, C.; et al. CRISPR disruption and UK Biobank analysis of a highly conserved polymorphic enhancer suggests a role in male anxiety and ethanol intake. Mol. Psychiatry 2020. [Google Scholar] [CrossRef] [PubMed]
- Low, B.E.; Christianson, G.J.; Lowell, E.; Qin, W.; Wiles, M.V. Functional humanization of immunoglobulin heavy constant gamma 1 Fc domain human FCGRT transgenic mice. mAbs 2020, 12, 1829334. [Google Scholar] [CrossRef] [PubMed]
- Pi, M.; Xu, F.; Ye, R.; Nishimoto, S.K.; Kesterson, R.A.; Williams, R.W.; Lu, L.; Quarles, L.D. Humanized GPRC6A(KGKY) is a gain-of-function polymorphism in mice. Sci. Rep. 2020, 10, 11143. [Google Scholar] [CrossRef] [PubMed]
- Gu, B.; Posfai, E.; Gertsenstein, M.; Rossant, J. Efficient Generation of Large-Fragment Knock-In Mouse Models Using 2-Cell (2C)-Homologous Recombination (HR)-CRISPR. Curr. Protoc. Mouse. Biol. 2020, 10, e67. [Google Scholar] [CrossRef]
- Cheah, S.S.; Behringer, R.R. Gene-targeting strategies. Methods Mol. Biol. 2000, 136, 455–463. [Google Scholar]
- Hodgson, K.; Mufti, S.J.; Uher, R.; McGuffin, P. Genome-wide approaches to antidepressant treatment: Working towards understanding and predicting response. Genome Med. 2012, 4, 52. [Google Scholar] [CrossRef] [Green Version]
- Budney, A.J.; Sofis, M.J.; Borodovsky, J.T. An update on cannabis use disorder with comment on the impact of policy related to therapeutic and recreational cannabis use. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 73–86. [Google Scholar] [CrossRef]
- Nicoll, G.; Davidson, S.; Shanley, L.; Hing, B.; Lear, M.; McGuffin, P.; Ross, R.; MacKenzie, A. Allele-specific differences in activity of a novel cannabinoid receptor 1 (CNR1) gene intronic enhancer in hypothalamus, dorsal root ganglia, and hippocampus. J. Biol. Chem. 2012, 287, 12828–12834. [Google Scholar] [CrossRef] [Green Version]
- Hay, E.A.; Cowie, P.; McEwan, A.R.; Ross, R.; Pertwee, R.G.; MacKenzie, A. Disease-associated polymorphisms within the conserved ECR1 enhancer differentially regulate the tissue-specific activity of the cannabinoid-1 receptor gene promoter; implications for cannabinoid pharmacogenetics. Hum. Mutat. 2020, 41, 291–298. [Google Scholar] [CrossRef]
- Hing, B.; Davidson, S.; Lear, M.; Breen, G.; Quinn, J.; McGuffin, P.; MacKenzie, A. A polymorphism associated with depressive disorders differentially regulates brain derived neurotrophic factor promoter IV activity. Biol. Psychiatry 2012, 71, 618–626. [Google Scholar] [CrossRef] [Green Version]
- Burton, M.A.; Lillycrop, K.A. Nutritional modulation of the epigenome and its implication for future health. Proc. Nutr. Soc. 2019, 78, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Doherty, T.S.; Roth, T.L. Epigenetic Landscapes of the Adversity-Exposed Brain. Prog. Mol. Biol. Transl. Sci. 2018, 157, 1–19. [Google Scholar] [PubMed]
- Kader, F.; Ghai, M.; Maharaj, L. The effects of DNA methylation on human psychology. Behav. Brain. Res. 2018, 346, 47–65. [Google Scholar] [CrossRef] [PubMed]
- Reul, J.M. Making memories of stressful events: A journey along epigenetic, gene transcription, and signaling pathways. Front. Psychiatry 2014, 5, 5. [Google Scholar] [CrossRef]
- Murgatroyd, C.; Wu, Y.; Bockmuhl, Y.; Spengler, D. The Janus face of DNA methylation in aging. Aging 2010, 2, 107–110. [Google Scholar] [CrossRef] [Green Version]
- Murgatroyd, C.; Wu, Y.; Bockmuhl, Y.; Spengler, D. Genes learn from stress: How infantile trauma programs us for depression. Epigenetics 2010, 5, 194–199. [Google Scholar] [CrossRef] [Green Version]
- Murgatroyd, C.; Patchev, A.V.; Wu, Y.; Micale, V.; Bockmuhl, Y.; Fischer, D.; Holsboer, F.; Wotjak, C.T.; Almeida, O.F.; Spengler, D. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat. Neurosci. 2009, 12, 1559–1566. [Google Scholar] [CrossRef]
- Hay, E.A.; McEwan, A.; Wilson, D.; Barrett, P.; D’Agostino, G.; Pertwee, R.G.; MacKenzie, A. Disruption of an enhancer associated with addictive behaviour within the cannabinoid receptor-1 gene suggests a possible role in alcohol intake, cannabinoid response and anxiety-related behaviour. Psychoneuroendocrinology 2019, 109, 104407. [Google Scholar] [CrossRef]
- Hay, E.H.; Cowie, P.; McEwan, A.J.; Wilson, D.; Ross, R.; Barrett, P.; Pertwee, R.G.; MacKenzie, A. Genetic and pharmacological influences modulating tissue specific regulation of the cannabinoid receptor-1 (CB1); implications for cannabinoid pharmacogenetics. BioRxiv 2019. [Google Scholar] [CrossRef]
- Visscher, P.M.; Wray, N.R.; Zhang, Q.; Sklar, P.; McCarthy, M.I.; Brown, M.A.; Yang, J. 10 Years of GWAS Discovery: Biology, Function, and Translation. Am. J. Hum. Genet. 2017, 101, 5–22. [Google Scholar] [CrossRef] [Green Version]
- King, C.R.; Nicolae, D.L. GWAS to Sequencing: Divergence in Study Design and Analysis. Genes 2014, 5, 460–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Consortium, G.T. Human genomics. The Genotype-Tissue Expression (GTEx) pilot analysis: Multitissue gene regulation in humans. Science 2015, 348, 648–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Brien, H.E.; Hannon, E.; Hill, M.J.; Toste, C.C.; Robertson, M.J.; Morgan, J.E.; McLaughlin, G.; Lewis, C.M.; Schalkwyk, L.C.; Hall, L.S.; et al. Expression quantitative trait loci in the developing human brain and their enrichment in neuropsychiatric disorders. Genome Biol. 2018, 19, 194. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McEwan, A.R.; MacKenzie, A. Perspective: Quality Versus Quantity; Is It Important to Assess the Role of Enhancers in Complex Disease from an In Vivo Perspective? Int. J. Mol. Sci. 2020, 21, 7856. https://doi.org/10.3390/ijms21217856
McEwan AR, MacKenzie A. Perspective: Quality Versus Quantity; Is It Important to Assess the Role of Enhancers in Complex Disease from an In Vivo Perspective? International Journal of Molecular Sciences. 2020; 21(21):7856. https://doi.org/10.3390/ijms21217856
Chicago/Turabian StyleMcEwan, Andrew R., and Alasdair MacKenzie. 2020. "Perspective: Quality Versus Quantity; Is It Important to Assess the Role of Enhancers in Complex Disease from an In Vivo Perspective?" International Journal of Molecular Sciences 21, no. 21: 7856. https://doi.org/10.3390/ijms21217856




