Thermodynamic Insights by Microscale Thermophoresis into Translesion DNA Synthesis Catalyzed by DNA Polymerases Across a Lesion of Antitumor Platinum–Acridine Complex
Abstract
1. Introduction
2. Results and Discussion
2.1. Sequence Specificity of the Formation of an AMD Adduct Formed in DNA by Transcription Mapping
2.2. Replication Through DNA Adducts of AMD by Purified TLS Pols
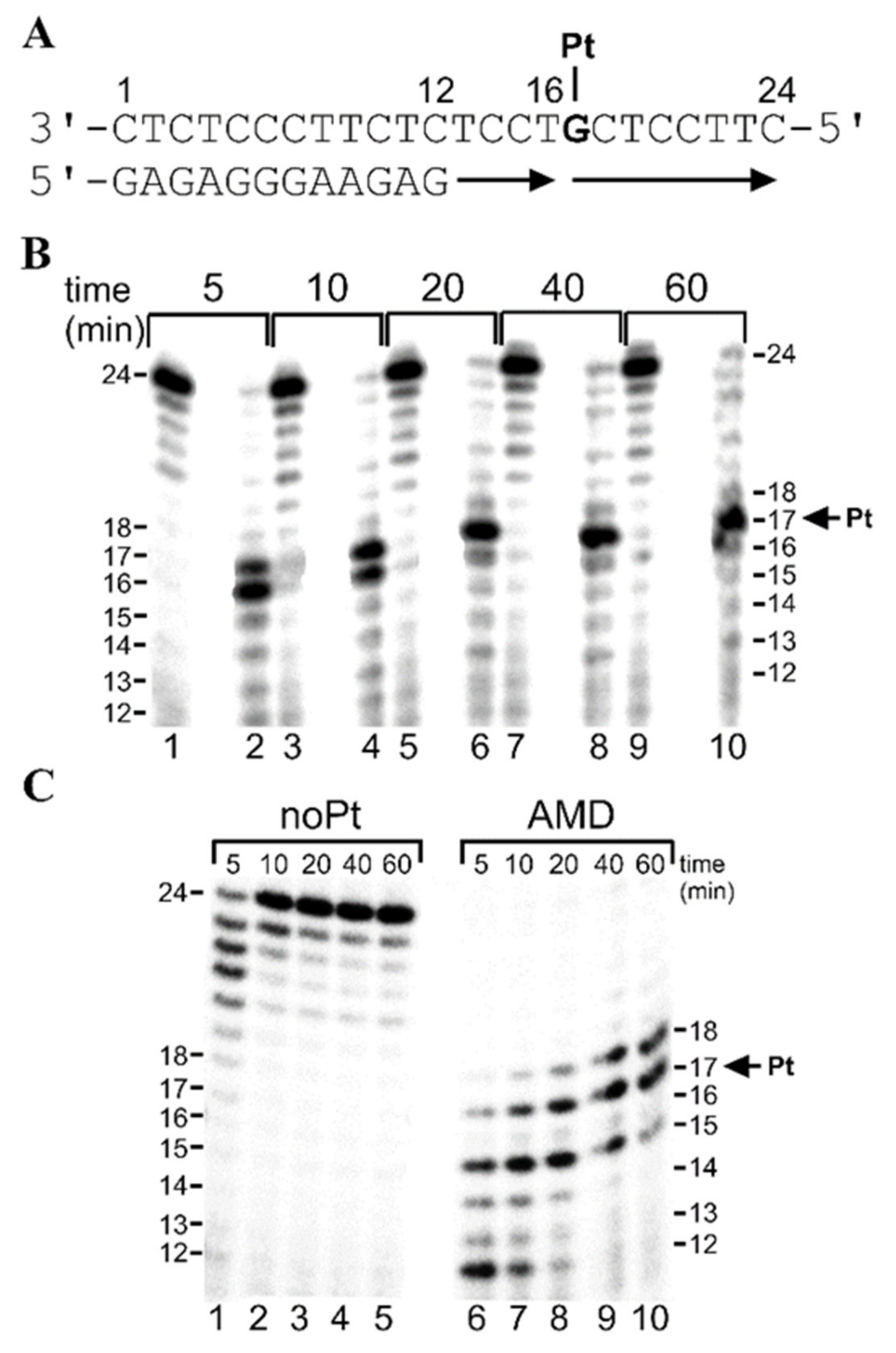
2.2.1. Running-Start Primer Extension Experiments in the Presence of All Four Natural dNTPs
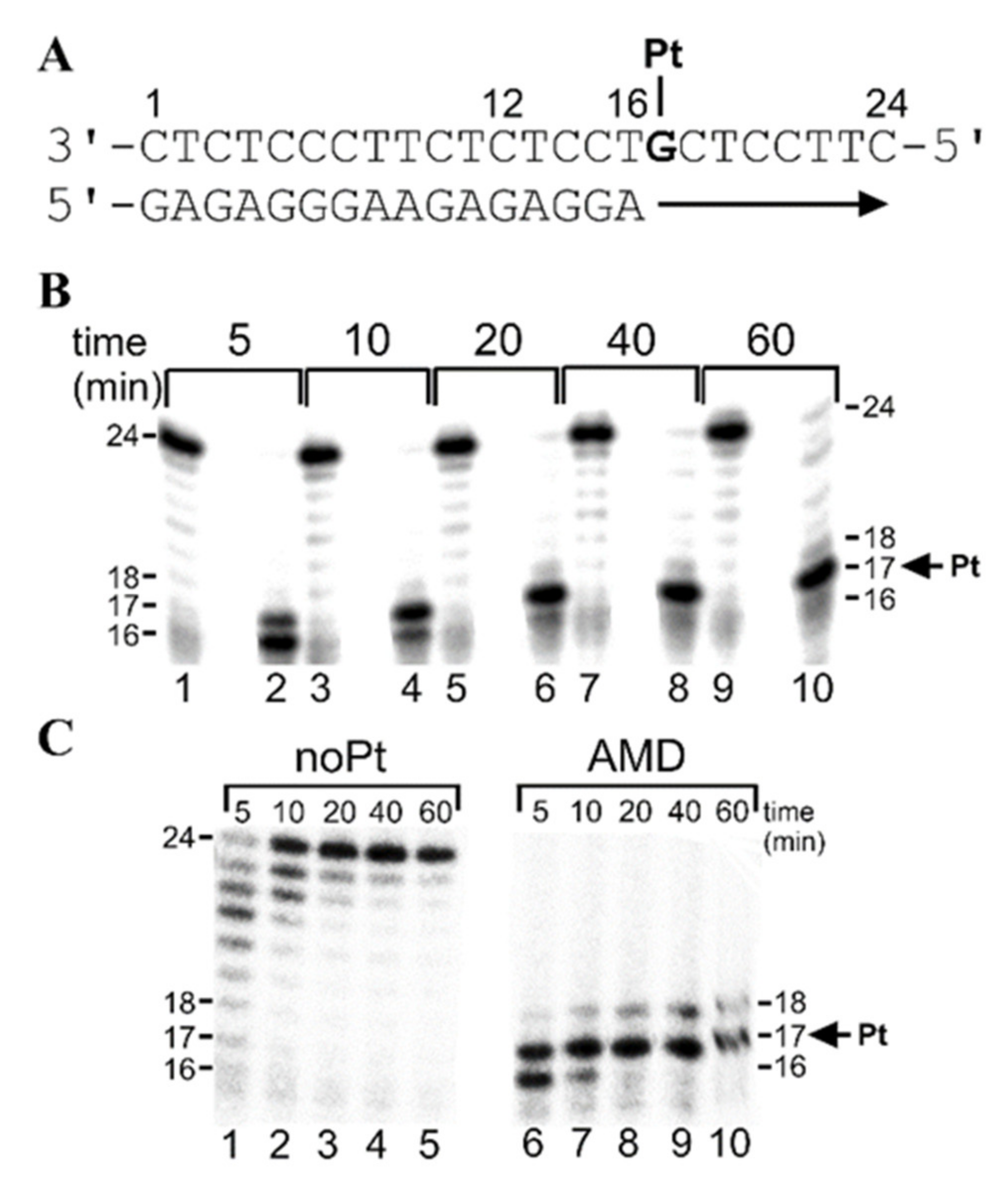
2.2.2. Standing-Start Primer Extension Experiments in the Presence of All Four Natural dNTPs
2.2.3. Steady-State Kinetics of dNTP Incorporation by KF− and Pol η Opposite G Unplatinated or Platinated by AMD
2.3. Probing the Thermodynamics of Translesion DNA Synthesis Across a Monofunctional Adduct of AMD by Microscale Thermophoresis (MST)
3. Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Transcription Mapping of DNA Platinum Adducts
4.3. Platination of Oligonucleotides
4.4. Translesion Synthesis Assays
4.5. Nucleotide Misinsertion by KF− and Human Polymerase η
4.6. Steady-State Kinetic Analysis for dNTP Incorporations by KF− and Pol η
4.7. Microscale Thermophoresis (MST)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACR | [PtCl(en)(L)](NO3)2 (en = ethane-1,2-diamine, L = 1-[2-(acridin-9-ylamino)ethyl]-1,3-dimethylthiourea) |
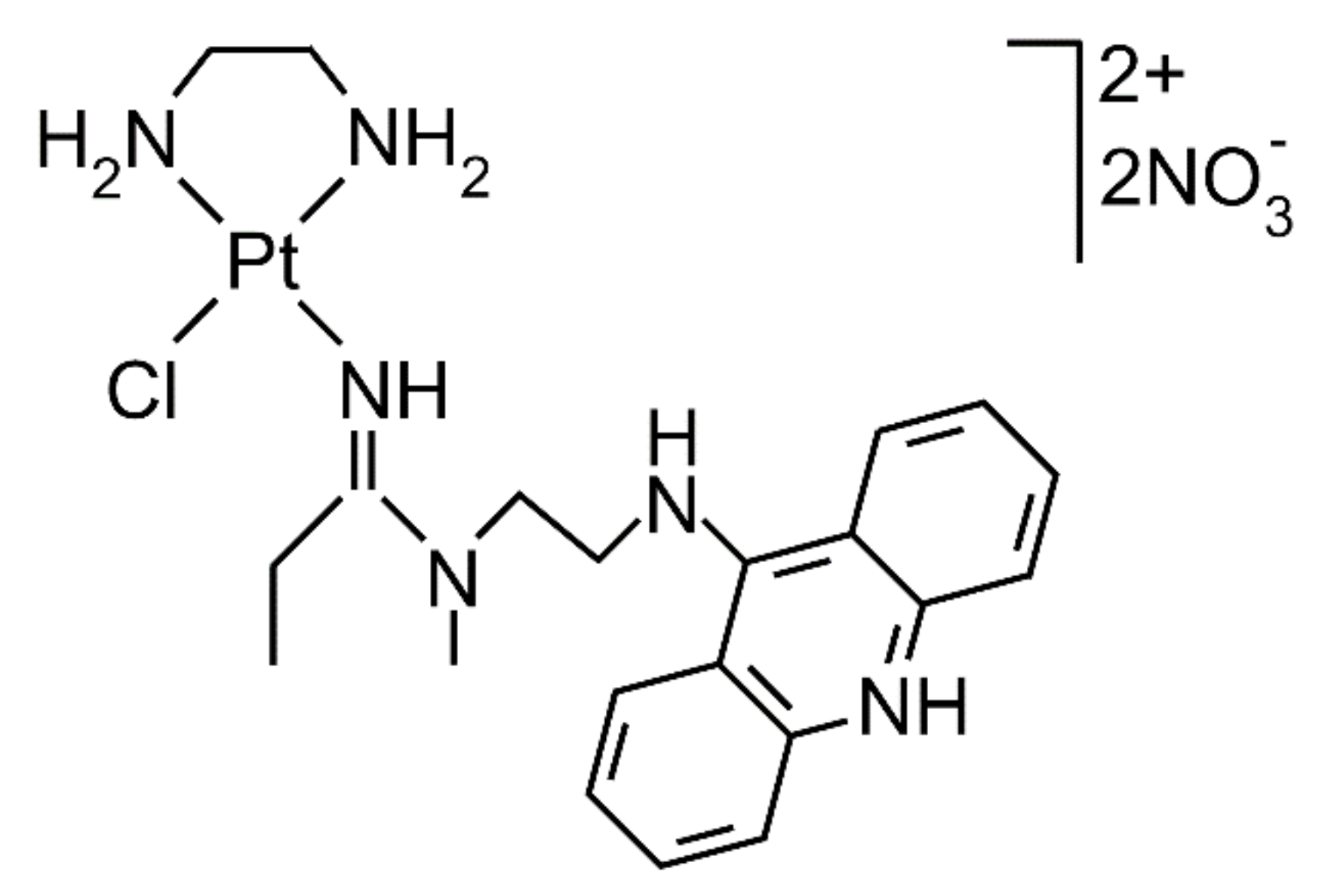
| AMD | [PtCl(en)(L)](NO3)2 (en = ethane-1,2-diamine, L = N-[2-(acridin-9-ylamino)ethyl]-N-methylpropionamidine) |
| TLS pol | translesion DNA polymerase |
| DNA pol | DNA polymerase |
| KF− | Klenow fragment of DNA polymerase I (the exonuclease deficient) |
| Pol η | DNA polymerase η |
| MST | microscale thermophoresis |
| TLS | translesion DNA synthesis |
| rb | number of molecules of the platinum complex bound per nucleotide residue |
| dien | diethylenetriamine (1,4,7-triazaheptane) |
| CT | calf thymus |
| dNTP | 2′-deoxyribonucleotide-5‘-triphosphate |
| PAA | polyacrylamide |
| Cy5 | far-red fluorescent label for protein or nucleic acid conjugates |
| Kd | equilibrium dissociation constant |
| RE | relative efficiency |
| DTT | dithiothreitol |
| BSA | bovine serum albumin |
References
- Johnstone, T.C.; Suntharalingam, K.; Lippard, S.J. The Next Generation of Platinum Drugs: Targeted Pt(II) Agents, Nanoparticle Delivery, and Pt(IV) Prodrugs. Chem. Rev. 2016, 116, 3436–3486. [Google Scholar] [CrossRef] [PubMed]
- Brabec, V.; Hrabina, O.; Kasparkova, J. Cytotoxic platinum coordination compounds. DNA binding agents. Co-Ord. Chem. Rev. 2017, 351, 2–31. [Google Scholar] [CrossRef]
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; Lippard, S.J. Direct cellular responses to platinum-induced DNA damage. Chem. Rev. 2007, 107, 1387–1407. [Google Scholar] [CrossRef]
- Wang, D.; Lippard, S.J. Cellular processing of platinum anticancer drugs. Nat. Rev. Drug Discov. 2005, 4, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Graham, L.A.; Wilson, G.M.; West, T.K.; Day, C.S.; Kucera, G.L.; Bierbach, U. Unusual Reactivity of a Potent Platinum–Acridine Hybrid Antitumor Agent. ACS Med. Chem. Lett. 2011, 2, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Pickard, A.J.; Liu, F.; Bartenstein, T.F.; Haines, L.G.; Levine, K.E.; Kucera, G.L.; Bierbach, U. Redesigning the DNA-Targeted Chromophore in Platinum-Acridine Anticancer Agents: A Structure-Activity Relationship Study. Chem. Eur. J. 2014, 20, 16174–16187. [Google Scholar] [CrossRef]
- Yao, X.; Tracy, C.M.; Bierbach, U. Cysteine-Directed Bioconjugation of a Platinum(II)–Acridine Anticancer Agent. Inorg. Chem. 2018, 58, 43–46. [Google Scholar] [CrossRef]
- Rose, P.K.; Watkins, N.H.; Yao, X.; Zhang, S.; Mancera-Ortiz, I.Y.; Sloop, J.T.; Donati, G.L.; Day, C.S.; Bierbach, U. Effect of the nonleaving groups on the cellular uptake and cytotoxicity of platinum-acridine anticancer agents. Inorg. Chim. Acta 2019, 492, 150–155. [Google Scholar] [CrossRef]
- Guddneppanavar, R.; Bierbach, U. Adenine-N3 in the DNA minor groove - an emerging target for platinum containing anticancer pharmacophores. Anti-Cancer Agents Med. Chem. 2007, 7, 125–138. [Google Scholar] [CrossRef]
- Ma, Z.; Choudhury, J.R.; Wright, M.W.; Day, C.S.; Saluta, G.; Kucera, G.L.; Bierbach, U. A Non-Cross-Linking Platinum−Acridine Agent with Potent Activity in Non-Small-Cell Lung Cancer. J. Med. Chem. 2008, 51, 7574–7580. [Google Scholar] [CrossRef] [PubMed]
- Cheung-Ong, K.; Song, K.T.; Ma, Z.; Shabtai, D.; Lee, A.Y.; Gallo, D.; Heisler, L.E.; Brown, G.W.; Bierbach, U.; Giaever, G.; et al. Comparative Chemogenomics To Examine the Mechanism of Action of DNA-Targeted Platinum-Acridine Anticancer Agents. ACS Chem. Biol. 2012, 7, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Barry, C.G.; Baruah, A.H.; Bierbach, U. Unprecedented Monofunctional Metalation of Adenine Nucleobase in Guanine- and Thymine-Containing Dinucleotide Sequences by a Cytotoxic Platinum−Acridine Hybrid Agent. J. Am. Chem. Soc. 2003, 125, 9629–9637. [Google Scholar] [CrossRef] [PubMed]
- Barry, C.G.; Day, C.S.; Bierbach, U. Duplex-Promoted Platination of Adenine-N3 in the Minor Groove of DNA: Challenging a Longstanding Bioinorganic Paradigm. J. Am. Chem. Soc. 2005, 127, 1160–1169. [Google Scholar] [CrossRef]
- Baruah, H.; Rector, C.L.; Monnier, S.M.; Bierbach, U. Mechanism of action of non-cisplatin type DNA-targeted platinum anticancer agents: DNA interactions of novel acridinylthioureas and their platinum conjugates. Biochem. Pharm. 2002, 64, 191–200. [Google Scholar] [CrossRef]
- Baruah, H.; Wright, M.W.; Bierbach, U. Solution Structural Study of a DNA Duplex Containing the Guanine-N7 Adduct Formed by a Cytotoxic Platinum−Acridine Hybrid Agent†,‡. Biochemistry 2005, 44, 6059–6070. [Google Scholar] [CrossRef]
- Budiman, M.E.; Alexander, R.W.; Bierbach, U. Unique Base-Step Recognition by a Platinum−Acridinylthiourea Conjugate Leads to a DNA Damage Profile Complementary to That of the Anticancer Drug Cisplatin†. Biochemistry 2004, 43, 8560–8567. [Google Scholar] [CrossRef]
- Martins, E.T.; Baruah, H.; Kramarczyk, J.; Saluta, G.; Day, C.S.; Kucera, G.L.; Bierbach, U. Design, Synthesis, and Biological Activity of a Novel Non-Cisplatin-type Platinum−Acridine Pharmacophore. J. Med. Chem. 2001, 44, 4492–4496. [Google Scholar] [CrossRef]
- Kostrhunova, H.; Malina, J.; Pickard, A.J.; Stepankova, J.; Vojtiskova, M.; Kasparkova, J.; Muchova, T.; Rohlfing, M.L.; Bierbach, U.; Brabec, V. Replacement of a thiourea with an amidine group in a monofunctional platinum-acridine antitumor agent. Effect on DNA interactions, DNA adduct recognition and repair. Mol. Pharm. 2011, 8, 1941–1954. [Google Scholar] [CrossRef]
- Guddneppanavar, R.; Choudhury, J.R.; Kheradi, A.R.; Steen, B.D.; Saluta, G.; Kucera, G.L.; Day, C.S.; Bierbach, U. Effect of the Diamine Nonleaving Group in Platinum−Acridinylthiourea Conjugates on DNA Damage and Cytotoxicity. J. Med. Chem. 2007, 50, 2259–2263. [Google Scholar] [CrossRef]
- Smyre, C.L.; Saluta, G.; Kute, T.E.; Kucera, G.L.; Bierbach, U. Inhibition of DNA Synthesis by a Platinum–Acridine Hybrid Agent Leads to Potent Cell Kill in Nonsmall Cell Lung Cancer. ACS Med. Chem. Lett. 2011, 2, 870–874. [Google Scholar] [CrossRef]
- Lehmann, A.R. Translesion synthesis in mammalian cells. Exp. Cell Res. 2006, 312, 2673–2676. [Google Scholar] [CrossRef][Green Version]
- Avkin, S.; Adar, S.; Blander, G.; Livneh, Z. Quantitative measurement of translesion replication in human cells: Evidence for bypass of abasic sites by a replicative DNA polymerase. Proc. Natl. Acad. Sci. USA 2002, 99, 3764–3769. [Google Scholar] [CrossRef]
- Hubscher, U.; Maga, G.; Spadari, S. Eukaryotic DNA polymerases. Annu. Rev. Biochem. 2002, 71, 133–163. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.; Johnson, R.E.; Prakash, L. Eukaryotic translesion synthesis dna polymerases: Specificity of Structure and Function. Annu. Rev. Biochem. 2005, 74, 317–353. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.; Weiss, S.J.; Fisher, P.A. Recognition and binding of template-primers containing defined abasic sites by Drosophila DNA polymerase alpha holoenzyme. J. Biol. Chem. 1989, 264, 13018–13023. [Google Scholar]
- Weiss, S.J.; A. Fisher, P. Interaction of Drosophila DNA polymerase alpha holoenzyme with synthetic template-primers containing mismatched primer bases or propanodeoxyguanosine adducts at various positions in template and primer regions. J. Biol. Chem. 1992, 267, 18520–18526. [Google Scholar] [PubMed]
- Lindsley, J.E.; Fuchs, R.P.P. Use of single-turnover kinetics to study bulky adduct bypass by T7 DNA polymerase. Biochemistry 1994, 33, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Miller, H.; Grollman, A.P. Kinetics of DNA Polymerase I (Klenow Fragment Exo-) Activity on Damaged DNA Templates: Effect of Proximal and Distal Template Damage on DNA Synthesis. Biochemistry 1997, 36, 15336–15342. [Google Scholar] [CrossRef]
- Minetti, C.A.; Remeta, D.P.; Miller, H.; Gelfand, C.A.; Plum, G.E.; Grollman, A.P.; Breslauer, K.J. The thermodynamics of template-directed DNA synthesis: Base insertion and extension enthalpies. Proc. Natl. Acad. Sci. USA 2003, 100, 14719–14724. [Google Scholar] [CrossRef]
- Liang, F.; Cho, B.P. Probing the Thermodynamics of Aminofluorene-Induced Translesion DNA Synthesis by Differential Scanning Calorimetry. J. Am. Chem. Soc. 2007, 129, 12108–12109. [Google Scholar] [CrossRef]
- Florian, J.; Brabec, V. Thermodynamics of Translesion Synthesis across a Major DNA Adduct of Antitumor Oxaliplatin: Differential Scanning Calorimetric Study. Chem. Eur. J. 2011, 18, 1634–1639. [Google Scholar] [CrossRef]
- Creighton, S.; Goodman, M.F. Gel Kinetic Analysis of DNA Polymerase Fidelity in the Presence of Proofreading Using Bacteriophage T4 DNA Polymerase. J. Biol. Chem. 1995, 270, 4759–4774. [Google Scholar] [CrossRef]
- Lam, W.-C.; Van Der Schans, E.J.C.; Sowers, L.C.; Millar, D.P. Interaction of DNA Polymerase I (Klenow Fragment) with DNA Substrates Containing Extrahelical Bases: Implications for Proofreading of Frameshift Errors during DNA Synthesis†. Biochemistry 1999, 38, 2661–2668. [Google Scholar] [CrossRef]
- Patel, P.H.; Suzuki, M.; Adman, E.; Shinkai, A.; Loeb, L.A. Prokaryotic DNA polymerase I: Evolution, structure, and “base flipping” mechanism for nucleotide selection. J. Mol. Biol. 2001, 308, 823–837. [Google Scholar] [CrossRef]
- Gali, V.K.; Balint, E.; Serbyn, N.; Frittmann, O.; Stutz, F.; Unk, I. Translesion synthesis DNA polymerase η exhibits a specific RNA extension activity and a transcription-associated function. Sci. Rep. 2017, 7, 13055. [Google Scholar] [CrossRef]
- Lemaire, M.A.; Schwartz, A.; Rahmouni, A.R.; Leng, M. Interstrand cross-links are preferentially formed at the d(GC) sites in the reaction between cis-diamminedichloroplatinum (II) and DNA. Proc. Natl. Acad. Sci. USA 1991, 88, 1982–1985. [Google Scholar] [CrossRef]
- Brabec, V.; Leng, M. DNA interstrand cross-links of trans-diamminedichloroplatinum(II) are preferentially formed between guanine and complementary cytosine residues. Proc. Natl. Acad. Sci. USA 1993, 90, 5345–5349. [Google Scholar] [CrossRef]
- Novakova, O.; Malina, J.; Kašpárková, J.; Halámiková, A.; Bernard, V.; Intini, F.P.; Natile, G.; Brabec, V. Energetics, Conformation, and Recognition of DNA Duplexes Modified by Methylated Analogues of [PtCl(dien)]+. Chem. Eur. J. 2009, 15, 6211–6221. [Google Scholar] [CrossRef]
- McGregor, T.D.; Hegmans, A.; Kašpárková, J.; Neplechová, K.; Nováková, O.; Peňazová, H.; Vrána, O.; Brabec, V.; Farrell, N. A comparison of DNA binding profiles of dinuclear platinum compounds with polyamine linkers and the trinuclear platinum phase II clinical agent BBR3464. JBIC J. Biol. Inorg. Chem. 2002, 7, 397–404. [Google Scholar] [CrossRef]
- Kasparkova, J.; Novakova, O.; Marini, V.; Najajreh, Y.; Gibson, D.; Perez, J.-M.; Brabec, V. Activation of Trans Geometry in Bifunctional Mononuclear Platinum Complexes by a Piperidine Ligand. J. Biol. Chem. 2003, 278, 47516–47525. [Google Scholar] [CrossRef] [PubMed]
- Comess, K.M.; Burstyn, J.N.; Essigmann, J.M.; Lippard, S.J. Replication inhibition and translesion synthesis on templates containing site- specifically placed cis-diamminedichloroplatinum(II) DNA adducts. Biochemistry 1992, 31, 3975–3990. [Google Scholar] [CrossRef] [PubMed]
- Suo, Z.; Johnson, K.A. DNA secondary structure effects on DNA synthesis catalyzed by HIV-1 reverse transcriptase. J. Biol. Chem. 1998, 273, 27259–27267. [Google Scholar] [CrossRef]
- Vaisman, A.; Warren, M.W.; Chaney, S.G. The effect of DNA structure on the catalytic efficiency and fidelity of human DNA polymerase beta on templates with platinum-DNA adducts. J. Biol. Chem. 2001, 276, 18999–19005. [Google Scholar] [CrossRef]
- Bassett, E.; Vaisman, A.; Havener, J.M.; Masutani, C.; Hanaoka, F.; Chaney, S.G. Efficiency of Extension of Mismatched Primer Termini across from Cisplatin and Oxaliplatin Adducts by Human DNA Polymerases β and η in Vitro†. Biochemistry 2003, 42, 14197–14206. [Google Scholar] [CrossRef]
- Moriarity, B.; Novakova, O.; Farrell, N.; Brabec, V.; Kasparkova, J. 1,2-GG intrastrand cross-link of antitumor dinuclear bifunctional platinum compound with spermidine linker inhibits DNA polymerization more effectively than the cross-link of conventional cisplatin. Arch. Biochem. Biophys. 2007, 459, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Alt, A.; Lammens, K.; Chiocchini, C.; Lammens, A.; Pieck, J.C.; Kuch, D.; Hopfner, K.-P.; Carell, T. Bypass of DNA Lesions Generated During Anticancer Treatment with Cisplatin by DNA Polymerase. Science 2007, 318, 967–970. [Google Scholar] [CrossRef] [PubMed]
- Creighton, S.; Bloom, L.B.; Goodman, M.F. [19] Gel fidelity assay measuring nucleotide misinsertion, exonucleolytic proofreading, and lesion bypass efficiencies. Methods Enzymol. 1995, 262, 232–256. [Google Scholar] [CrossRef]
- Minetti, C.; Remeta, D.P.; Johnson, F.; Iden, C.R.; Breslauer, K.J. Impact of alpha-hydroxy-propanodeoxyguanine adducts on DNA duplex energetics: Opposite base modulation and implications for mutagenicity and genotoxicity. Biopolymers 2010, 93, 370–382. [Google Scholar] [CrossRef]
- Hrabina, O.; Brabec, V.; Novakova, O. Translesion DNA Synthesis Across Lesions Induced by Oxidative Products of Pyrimidines: An Insight into the Mechanism by Microscale Thermophoresis. Int. J. Mol. Sci. 2019, 20, 5012. [Google Scholar] [CrossRef]
- Jerabek-Willemsen, M.; André, T.; Wanner, R.; Roth, H.M.; Duhr, S.; Baaske, P.; Breitsprecher, D. MicroScale Thermophoresis: Interaction analysis and beyond. J. Mol. Struct. 2014, 1077, 101–113. [Google Scholar] [CrossRef]
- Petruska, J.; Goodman, M.F.; Boosalis, M.S.; Sowers, L.C.; Cheong, C.I.; Tinoco, J. Comparison between DNA melting thermodynamics and DNA polymerase fidelity. Proc. Natl. Acad. Sci. USA 1988, 85, 6252–6256. [Google Scholar] [CrossRef]
- Guengerich, F.P. Interactions of carcinogen-bound DNA with individual DNA polymerases. Chem. Rev. 2006, 106, 420–452. [Google Scholar] [CrossRef]
- Kasparkova, J.; Mellish, K.J.; Qu, Y.; Brabec, V.; Farrell, N. Site-Specific d(GpG) Intrastrand Cross-Links Formed by Dinuclear Platinum Complexes. Bending and NMR Studies†. Biochemistry 1996, 35, 16705–16713. [Google Scholar] [CrossRef]
- Mendelman, L.V.; Petruska, J.; Goodman, M.F. Base mispair extension kinetics. Comparison of DNA polymerase alpha and reverse transcriptase. J. Biol. Chem. 1990, 265, 2338–2346. [Google Scholar]
- Goodman, M.F.; Creighton, S.; Bloom, L.B.; Petruska, J.; Kunkel, T.A. Biochemical Basis of DNA Replication Fidelity. Crit. Rev. Biochem. Mol. Biol. 1993, 28, 83–126. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.A.; Newmister, S.A.; Fiala, K.A.; Suo, Z. Mechanism of double-base lesion bypass catalyzed by a Y-family DNA polymerase. Nucleic Acids Res. 2008, 36, 3867–3878. [Google Scholar] [CrossRef] [PubMed]

| DNA Substrate | dNTP | Km (μM) | Vmax (%/min) | Vmax/Km | RE 1 |
|---|---|---|---|---|---|
| 5′----A 3′----TGCT---- | dCTP | 0.033 ± 0.003 | 9.88 ± 0.06 | 299.3 | |
| dATP | 2.4 ± 0.7 | 0.58 ± 0.05 | 0.242 | ||
| dGTP | 5 ± 1 | 10.5 ± 0.9 | 2.1 | ||
| dTTP | 5.4 ± 0.7 | 12.2 ± 0.6 | 2.259 | ||
| 5′----A 3′----TGCT---- • AMD | dCTP | 1.5 ± 0.8 | 8 ± 1 | 5.333 | 0.02 |
| dATP | 15 ± 2 | 0.4 ± 0.1 | 0.027 | 0.11 | |
| dGTP | 4 ± 1 | 0.47 ± 0.07 | 0.118 | 0.06 | |
| dTTP | 10 ± 2 | 0.33 ± 0.09 | 0.033 | 0.01 |
| DNA substrate | dNTP | Km (µM) | Vmax (%/min) | Vmax/Km | RE 1 |
|---|---|---|---|---|---|
| 5′----A 3′----TGCT---- | dCTP | 0.8 ± 0.2 | 9.9 ± 0.6 | 12.36 | |
| dATP | 450 ± 20 | 3.0 ± 0.8 | 0.007 | ||
| dGTP | 78 ± 4 | 2.8 ± 0.6 | 0.036 | ||
| dTTP | 93 ± 7 | 7.8 ± 0.8 | 0.084 | ||
| 5′----A 3′----TGCT---- • AMD | dCTP | 18 ± 4 | 7.4 ± 0.6 | 0.411 | 0.03 |
| dATP | 409 ± 24 | 1.1 ± 0.3 | 0.003 | 0.43 | |
| dGTP | 68 ± 21 | 2.8 ± 0.8 | 0.041 | 1.14 | |
| dTTP | 124 ± 12 | 5.7 ± 0.8 | 0.046 | 0.55 |
| 15-mer Template/ Primer n − 1 1 | ΔHb (kJmol−1) | ΔS2 (kJK−1mol−1) | ΔG0310 2 (kJmol−1) | Kd3 (nM) |
|---|---|---|---|---|
| noPt (control) | 187 | 0.511 | 28.5 | 15,846 |
| AMD adduct | 225 | 0.612 | 35.2 | 1179 |
| 15-mer template/ primer n (G∙C match)1 | ΔHb (kJmol−1) | ΔS2 (kJK−1mol−1) | ΔG0310 2 (kJmol−1) | Kd3 (nM) |
| noPt (control) | 295 (108) | 0.853 (0.342) | 30.4 (1.9) | 7585 |
| AMD adduct | 259 (34) | 0.720 (0.108) | 35.7 (0.5) | 971 |
| 15-mer template/ primer n + 1 (G∙C match)1 | ΔHb (kJmol−1) | ΔS2 (kJK−1mol−1) | ΔG0310 2 (kJmol−1) | Kd3 (nM) |
| noPt (control) | 327 (140) | 0.935 (0.424) | 37.0 (8.5) | 587 |
| AMD adduct | 293 (68) | 0.809 (0.197) | 42.1 (6.9) | 81 |
| 15-mer template/ primer n (G~G mismatch)1 | ΔHb (kJmol−1) | ΔS2b (kJK−1mol−1) | ΔG0310 2 (kJmol−1) | Kd3 (nM) |
| noPt (control) | 204 (17) | 0.573 (0.062) | 26.3 (−2.2) | 37,193 |
| AMD adduct | 250 (25) | 0.715(0.103) | 28.2 (−7.0) | 17,802 |
| 15-mer template/ primer n + 1 (G~G mismatch)1 | ΔHb (kJ mol−1) | ΔS2 (kJK−1mol−1) | ΔG0310 2 (kJmol−1) | Kd3 (nM) |
| noPt (control) | 245 (58) | 0.700 (0.189) | 27.9 (−0.6) | 19,998 |
| AMD adduct | 267 (42) | 0.766 (0.154) | 29.4 (−5.8) | 11,178 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hreusova, M.; Novakova, O.; Brabec, V. Thermodynamic Insights by Microscale Thermophoresis into Translesion DNA Synthesis Catalyzed by DNA Polymerases Across a Lesion of Antitumor Platinum–Acridine Complex. Int. J. Mol. Sci. 2020, 21, 7806. https://doi.org/10.3390/ijms21207806
Hreusova M, Novakova O, Brabec V. Thermodynamic Insights by Microscale Thermophoresis into Translesion DNA Synthesis Catalyzed by DNA Polymerases Across a Lesion of Antitumor Platinum–Acridine Complex. International Journal of Molecular Sciences. 2020; 21(20):7806. https://doi.org/10.3390/ijms21207806
Chicago/Turabian StyleHreusova, Monika, Olga Novakova, and Viktor Brabec. 2020. "Thermodynamic Insights by Microscale Thermophoresis into Translesion DNA Synthesis Catalyzed by DNA Polymerases Across a Lesion of Antitumor Platinum–Acridine Complex" International Journal of Molecular Sciences 21, no. 20: 7806. https://doi.org/10.3390/ijms21207806
APA StyleHreusova, M., Novakova, O., & Brabec, V. (2020). Thermodynamic Insights by Microscale Thermophoresis into Translesion DNA Synthesis Catalyzed by DNA Polymerases Across a Lesion of Antitumor Platinum–Acridine Complex. International Journal of Molecular Sciences, 21(20), 7806. https://doi.org/10.3390/ijms21207806







