Saliva microRNA Biomarkers of Cumulative Concussion
Abstract
1. Introduction
2. Results
2.1. Participant Characteristics
2.2. Functional Measures of Balance, Neurocognition, and Olfaction
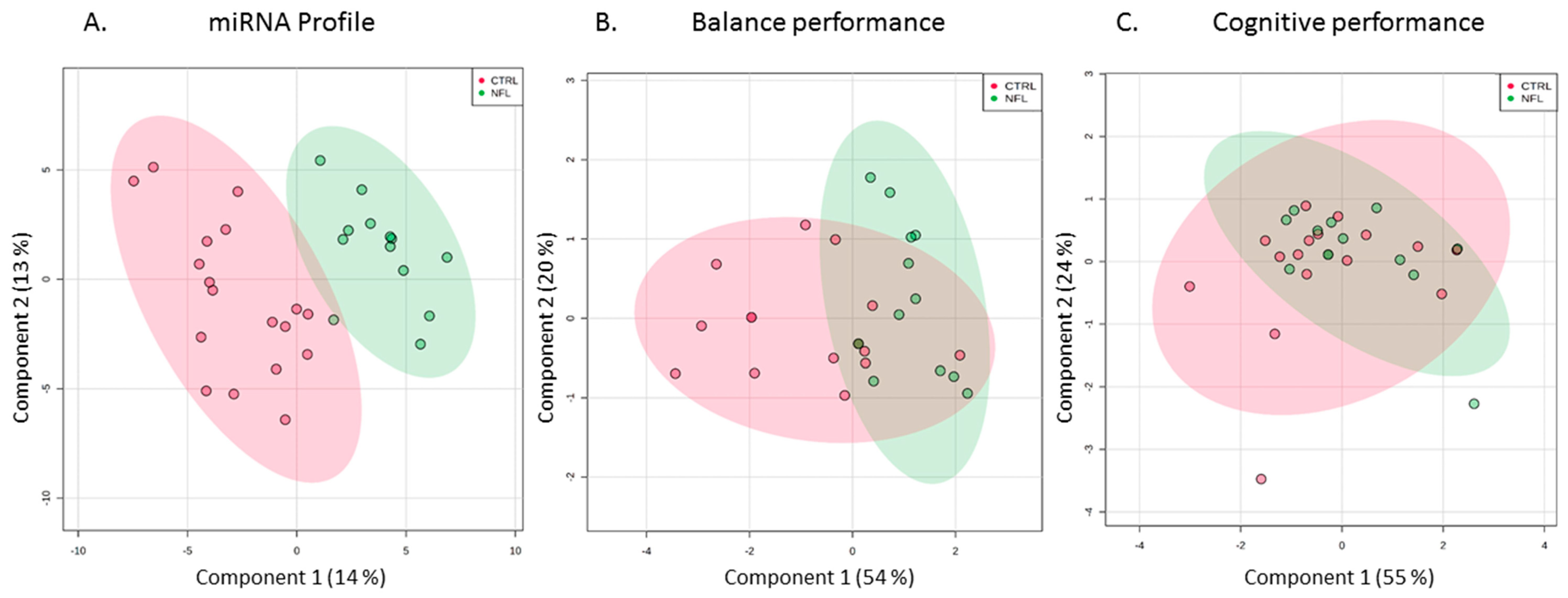
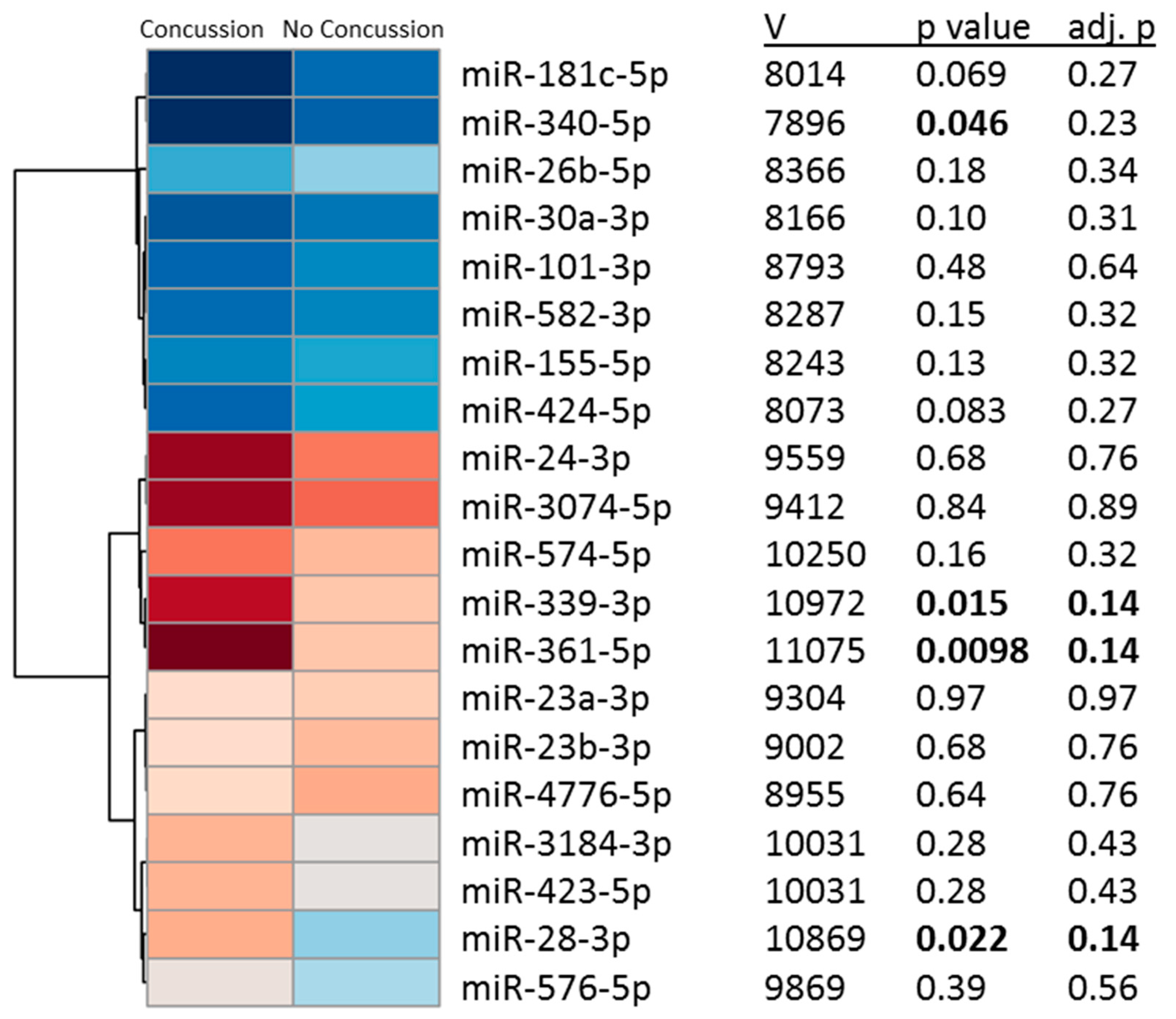
2.3. Salivary miRNAs Levels
2.4. Biologic Functions and Brain Relatedness of miRNA Candidates
3. Discussion
Conclusions
4. Materials and Methods
4.1. Participants
4.2. Measures
4.3. Saliva RNA
4.4. Statistics
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- McCrory, P.; Meeuwisse, W.; Dvorak, J.; Aubry, M.; Bailes, J.; Broglio, S.; Cantu, R.C.; Cassidy, D.; Echemendia, R.J.; Castellani, R.J. Consensus statement on concussion in sport—The 5th international conference on concussion in sport held in Berlin, October 2016. Br. J. Sports Med. 2017, 51, 838–847. [Google Scholar] [PubMed]
- Hootman, J.M.; Dick, R.; Agel, J. Epidemiology of collegiate injuries for 15 sports: Summary and recommendations for injury prevention initiatives. J. Athl. Train. 2007, 42, 311. [Google Scholar] [PubMed]
- Pellman, E.J.; Powell, J.W.; Viano, D.C.; Casson, I.R.; Tucker, A.M.; Feuer, H.; Lovell, M.; Waeckerle, J.F.; Robertson, D.W. Concussion in professional football: Epidemiological features of game injuries and review of the literature—Part 3. Neurosurgery 2004, 54, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Covassin, T.; Moran, R.; Wilhelm, K. Concussion symptoms and neurocognitive performance of high school and college athletes who incur multiple concussions. Am. J. Sports Med. 2013, 41, 2885–2889. [Google Scholar] [CrossRef] [PubMed]
- Castile, L.; Collins, C.L.; McIlvain, N.M.; Comstock, R.D. The epidemiology of new versus recurrent sports concussions among high school athletes, 2005–2010. Br. J. Sports Med. 2012, 46, 603–610. [Google Scholar] [CrossRef]
- Saunders, R.L.; Harbaugh, R.E. The second impact in catastrophic contact-sports head trauma. JAMA 1984, 252, 538–539. [Google Scholar] [CrossRef] [PubMed]
- Gronwall, D.; Wrightson, P. Cumulative effect of concussion. Lancet 1975, 306, 995–997. [Google Scholar] [CrossRef]
- Taylor, K.M.; Kioumourtzoglou, M.-A.; Clover, J.; Coull, B.A.; Dennerlein, J.T.; Bellinger, D.C.; Weisskopf, M.G. Concussion history and cognitive function in a large cohort of adolescent athletes. Am. J. Sports Med. 2018, 46, 3262–3270. [Google Scholar] [CrossRef]
- Barth, J.T.; Macciocchi, S.N.; Giordani, B.; Rimel, R.; Jane, J.A.; Boll, T.J. Neuropsychological sequelae of minor head injury. Neurosurgery 1983, 13, 529–533. [Google Scholar] [CrossRef]
- Dretsch, M.N.; Silverberg, N.D.; Iverson, G.L. Multiple past concussions are associated with ongoing post-concussive symptoms but not cognitive impairment in active-duty army soldiers. J. Neurotrauma 2015, 32, 1301–1306. [Google Scholar] [CrossRef]
- Guskiewicz, K.M.; Marshall, S.W.; Bailes, J.; McCrea, M.; Cantu, R.C.; Randolph, C.; Jordan, B.D. Association between recurrent concussion and late-life cognitive impairment in retired professional football players. Neurosurgery 2005, 57, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Guskiewicz, K.M.; Marshall, S.W.; Bailes, J.; McCrea, M.; Harding, H.P.; Matthews, A.; Mihalik, J.R.; Cantu, R.C. Recurrent concussion and risk of depression in retired professional football players. Med. Sci. Sports Exerc. 2007, 39, 903. [Google Scholar] [CrossRef] [PubMed]
- Kerr, Z.Y.; Marshall, S.W.; Harding, H.P., Jr.; Guskiewicz, K.M. Nine-year risk of depression diagnosis increases with increasing self-reported concussions in retired professional football players. Am. J. Sports Med. 2012, 40, 2206–2212. [Google Scholar] [CrossRef]
- Montenigro, P.H.; Alosco, M.L.; Martin, B.M.; Daneshvar, D.H.; Mez, J.; Chaisson, C.E.; Nowinski, C.J.; Au, R.; McKee, A.C.; Cantu, R.C. Cumulative head impact exposure predicts later-life depression, apathy, executive dysfunction, and cognitive impairment in former high school and college football players. J. Neurotrauma 2017, 34, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Guskiewicz, K.M.; McCrea, M.; Marshall, S.W.; Cantu, R.C.; Randolph, C.; Barr, W.; Onate, J.A.; Kelly, J.P. Cumulative effects associated with recurrent concussion in collegiate football players: The NCAA Concussion Study. JAMA 2003, 290, 2549–2555. [Google Scholar] [CrossRef] [PubMed]
- Collins, M.W.; Grindel, S.H.; Lovell, M.R.; Dede, D.E.; Moser, D.J.; Phalin, B.R.; Nogle, S.; Wasik, M.; Cordry, D.; Daugherty, M.K. Relationship between concussion and neuropsychological performance in college football players. JAMA 1999, 282, 964–970. [Google Scholar] [CrossRef]
- Broshek, D.K.; De Marco, A.P.; Freeman, J.R. A review of post-concussion syndrome and psychological factors associated with concussion. Brain Inj. 2015, 29, 228–237. [Google Scholar] [CrossRef]
- Kanaan, N.M.; Cox, K.; Alvarez, V.E.; Stein, T.D.; Poncil, S.; McKee, A.C. Characterization of early pathological tau conformations and phosphorylation in chronic traumatic encephalopathy. J. Neuropathol. Exp. Neurol. 2016, 75, 19–34. [Google Scholar] [CrossRef]
- Bieniek, K.F.; Ross, O.A.; Cormier, K.A.; Walton, R.L.; Soto-Ortolaza, A.; Johnston, A.E.; DeSaro, P.; Boylan, K.B.; Graff-Radford, N.R.; Wszolek, Z.K. Chronic traumatic encephalopathy pathology in a neurodegenerative disorders brain bank. Acta Neuropathol. 2015, 130, 877–889. [Google Scholar] [CrossRef]
- Cherry, J.D.; Tripodis, Y.; Alvarez, V.E.; Huber, B.; Kiernan, P.T.; Daneshvar, D.H.; Mez, J.; Montenigro, P.H.; Solomon, T.M.; Alosco, M.L. Microglial neuroinflammation contributes to tau accumulation in chronic traumatic encephalopathy. Acta Neuropathol. Commun. 2016, 4, 1–9. [Google Scholar] [CrossRef]
- Tagge, C.A.; Fisher, A.M.; Minaeva, O.V.; Gaudreau-Balderrama, A.; Moncaster, J.A.; Zhang, X.L.; Wojnarowicz, M.W.; Casey, N.; Lu, H.; Kokiko-Cochran, O.N.; et al. Concussion, microvascular injury, and early tauopathy in young athletes after impact head injury and an impact concussion mouse model. Brain 2018, 141, 422–458. [Google Scholar] [CrossRef] [PubMed]
- Giza, C.C.; Hovda, D.A. The neurometabolic cascade of concussion. J. Athl. Train. 2001, 36, 228. [Google Scholar] [CrossRef] [PubMed]
- Dretsch, M.N.; Daniel, T.A.; Goodman, A.M.; Katz, J.S.; Denney, T.; Deshpande, G.; Robinson, J.L. Differential neural activation when voluntarily regulating emotions in service members with chronic mild traumatic brain injury. Appl. Neuropsychol. Adult 2019, 26, 76–88. [Google Scholar] [CrossRef]
- Rangaprakash, D.; Dretsch, M.N.; Venkataraman, A.; Katz, J.S.; Denney, T.S., Jr.; Deshpande, G. Identifying disease foci from static and dynamic effective connectivity networks: Illustration in soldiers with trauma. Hum. Brain Mapp. 2018, 39, 264–287. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, S.; Black, I.; Salado, L.; Estrada, J.; Isla, K. The Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students. Appl. Sci. 2020, 10, 4131. [Google Scholar] [CrossRef]
- Im, H.-I.; Kenny, P.J. MicroRNAs in neuronal function and dysfunction. Trends Neurosci. 2012, 35, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Atif, H.; Hicks, S.D. A review of microRNA biomarkers in traumatic brain injury. J. Exp. Neurosci. 2019, 13, 1179069519832286. [Google Scholar] [CrossRef]
- Di Pietro, V.; Porto, E.; Ragusa, M.; Barbagallo, C.; Davies, D.; Forcione, M.; Logan, A.; Di Pietro, C.; Purrello, M.; Grey, M. Salivary MicroRNAs: Diagnostic markers of mild traumatic brain injury in contact-sport. Front. Mol. Neurosci. 2018, 11, 290. [Google Scholar] [CrossRef]
- Goldie, B.J.; Dun, M.D.; Lin, M.; Smith, N.D.; Verrills, N.M.; Dayas, C.V.; Cairns, M.J. Activity-associated miRNA are packaged in Map1b-enriched exosomes released from depolarized neurons. Nucleic Acids Res. 2014, 42, 9195–9208. [Google Scholar] [CrossRef]
- Devoto, C.; Lai, C.; Qu, B.-X.; Guedes, V.A.; Leete, J.; Wilde, E.A.; Walker, W.C.; Diaz-Arrastia, R.; Kenney, K.; Gill, J. Exosomal MicroRNAs in Military Persons with Mild Traumatic Brain Injury: Preliminary Results from a Chronic Effects of Neurotrauma Consortium (CENC) Biomarker Discovery Project. J. Neurotrauma 2020. [Google Scholar] [CrossRef]
- Hicks, S.D.; Johnson, J.; Carney, M.C.; Bramley, H.; Olympia, R.P.; Loeffert, A.C.; Thomas, N.J. Overlapping microRNA expression in saliva and cerebrospinal fluid accurately identifies pediatric traumatic brain injury. J. Neurotrauma 2018, 35, 64–72. [Google Scholar] [CrossRef] [PubMed]
- LaRocca, D.; Barns, S.; Hicks, S.D.; Brindle, A.; Williams, J.; Uhlig, R.; Johnson, P.; Neville, C.; Middleton, F.A. Comparison of serum and saliva miRNAs for identification and characterization of mTBI in adult mixed martial arts fighters. PLoS ONE 2019, 14, e0207785. [Google Scholar] [CrossRef]
- Johnson, J.J.; Loeffert, A.C.; Stokes, J.; Olympia, R.P.; Bramley, H.; Hicks, S.D. Association of salivary microRNA changes with prolonged concussion symptoms. JAMA Pediatr. 2018, 172, 65–73. [Google Scholar] [CrossRef]
- Schatz, P.; Robertshaw, S. Comparing post-concussive neurocognitive test data to normative data presents risks for under-classifying “above average” athletes. Arch. Clin. Neuropsychol. 2014, 29, 625–632. [Google Scholar] [CrossRef] [PubMed]
- Geary, E.K.; Kraus, M.F.; Pliskin, N.H.; Little, D.M. Verbal learning differences in chronic mild traumatic brain injury. J. Int. Neuropsychol. Soc. 2010, 16, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Hwang, C.K.; Zheng, H.; Wagley, Y.; Lin, H.Y.; Kim, D.K.; Law, P.Y.; Loh, H.H.; Wei, L.N. MicroRNA 339 down-regulates μ-opioid receptor at the post-transcriptional level in response to opioid treatment. FASEB J. 2013, 27, 522–535. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, S.; Sun, W. Expression of miR-28-3p in patients with Alzheimer’s disease before and after treatment and its clinical value. Exp. Ther. Med. 2020, 20, 2218–2226. [Google Scholar] [CrossRef]
- Taheri, S.; Tanriverdi, F.; Zararsiz, G.; Elbuken, G.; Ulutabanca, H.; Karaca, Z.; Selcuklu, A.; Unluhizarci, K.; Tanriverdi, K.; Kelestimur, F. Circulating microRNAs as potential biomarkers for traumatic brain injury-induced hypopituitarism. J. Neurotrauma 2016, 33, 1818–1825. [Google Scholar] [CrossRef]
- Papa, L.; Slobounov, S.M.; Breiter, H.C.; Walter, A.; Bream, T.; Seidenberg, P.; Bailes, J.E.; Bravo, S.; Johnson, B.; Kaufman, D.; et al. Elevations in microRNA biomarkers in serum are associated with measures of concussion, neurocognitive function, and Subconcussive trauma over a single national collegiate athletic association division I season in collegiate football players. J. Neurotrauma 2019, 36, 1343–1351. [Google Scholar] [CrossRef]
- Kerr, Z.Y.; Marshall, S.W.; Guskiewicz, K.M. Reliability of concussion history in former professional football players. Med. Sci. Sports Exerc. 2012, 44, 377–382. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. Embnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, I.S.; Zagganas, K.; Paraskevopoulou, M.D.; Georgakilas, G.; Karagkouni, D.; Vergoulis, T.; Dalamagas, T.; Hatzigeorgiou, A.G. DIANA-miRPath v3. 0: Deciphering microRNA function with experimental support. Nucleic Acids Res. 2015, 43, W460–W466. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, N.; Leidinger, P.; Becker, K.; Backes, C.; Fehlmann, T.; Pallasch, C.; Rheinheimer, S.; Meder, B.; Stähler, C.; Meese, E.; et al. Distribution of miRNA expression across human tissues. Nucleic Acids Res. 2016, 44, 3865–3877. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Wishart, D.S.; Xia, J. Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Curr. Protoc. Bioinform. 2019, 68, e86. [Google Scholar] [CrossRef]

| Group 1, Mean (Range) | Group 2, Mean (Range) | |||||
|---|---|---|---|---|---|---|
| All (n = 31) | Football Athletes (n = 13) | Controls (n = 18) | All (n = 310) | Concussion (n = 80) | No Concussion (n = 230) | |
| Demographics | ||||||
| Male sex, No. (%) | 31 (100) | 13 (100) | 18 (100) | 208 (67) | 61 (76) | 147 (64) * |
| Age (years) | 73 (46–89) | 73 (66–78) | 72 (46–89) | 20 (7–39) | 21 (10–35) | 19 (7–39) * |
| White race, No. (%) | 14 (74) | 9 (69) | 5 (83) | 224 (82) | 67 (83) | 187 (82) |
| Medical history | ||||||
| BMI (kg/m2) | 28 (20–38) | 30 (25–38) | 26 (20–34) * | 24 (13–40) | 25 (13–39) | 24 (13–40) |
| ADHD, No. (%) | 0 (0) | 0 (0) | 0 (0) | 24 (8) | 8 (10) | 16 (7) |
| Anxiety, No. (%) | 0 (0) | 0 (0) | 0 (0) | 18 (5) | 4 (5) | 14 (6) |
| Depression, No. (%) | 0 (0) | 0 (0) | 0 (0) | 10 (3) | 4 (5) | 6 (3) |
| Diagnosed concussions, No. (%) | 5 (16) | 2 (16) | 3 (16) | 80 (26) | 80 (100) | 0 (0) * |
| No. diagnosed concussions | 0.3 (1–5) | 0.4 (1–5) | 0.2 (0–1) | 0.4 (0–7) | 1.5 (1–7) | 0 (0) * |
| Undiagnosed concussions, No. (%) | NA | 13 (100) | NA | NA | NA | NA |
| No. undiagnosed concussion | NA | 5 (1–25) | NA | NA | NA | NA |
| Time since last concussion (years) | NA | 45 (38–56) | NA | NA | 1 (0–7) | NA |
| Professional football career | NA | 13 (2) | NA | NA | NA | NA |
| PCSS burden | NA | 10 (3–21) | NA | 2 (0–22) | 2 (0–22) | 2 (0–19) |
| PCSS severity | NA | 19 (3–78) | NA | 4 (0–91) | 5 (0–91) | 3 (0–42) |
| Sample collection time (24 h clock) | 12 (8–18) | 13 (10–18) | 11 (8–13) | 14 (7–19) | 14 (7–18) | 12 (7–19) * |
| Group 1, Mean (SD) | Group 2, Mean (SD) | |||||
|---|---|---|---|---|---|---|
| All (n = 28) | Football Athletes (n = 12) | Controls (n = 16) | All (n = 149) | Concussion (n = 37) | No Concussion (n = 112) | |
| Balance | ||||||
| TLEO | 72 (14) | 83 (5) | 63 (12) * | 85 (3) | 85 (3) | 85 (3) |
| TLEC | 69 (12) | 77 (9) | 63 (11) * | 84 (4) | 84 (4) | 84 (3) |
| TSEO | 65 (18) | 65 (22) | 64 (12) | 84 (4) | 83 (5) | 85 (4) * |
| TSEC | 48 (24) | 48 (26) | 48 (21) | 82 (8) | 79 (9) | 82 (7) * |
| TLEOFP | 61 (20) | 69 (18) | 54 (19) | 86 (7) | 85 (7) | 86 (6) |
| Neurocognition | ||||||
| SRT1 | 156 (40) | 159 (41) | 155 (39) | 191 (24) | 188 (22) | 192 (25) |
| SRT2 | 152 (36) | 148 (39) | 155 (32) | 186 (23) | 185 (22) | 186 (23) |
| PRT | 79 (13) | 76 (10) | 81 (14) | 100 (13) | 99 (12) | 100 (13) |
| GNG | 94 (15) | 91 (12) | 96 (17) | 121 (13) | 121 (13) | 121 (13) |
| Olfaction | ||||||
| BSIT | 69 (23) | 63 (23) | 82 (17) | NA | NA | NA |
| KEGG Pathway | p-Value | #Genes | #miRNAs |
|---|---|---|---|
| Adherens junction | 3.01 × 10−17 | 20 | 3 |
| ECM-receptor interaction | 2.5 × 10−07 | 9 | 3 |
| Bacterial invasion of epithelial cells | 4.3 × 10−06 | 16 | 3 |
| Hippo signaling pathway | 9.6 × 10−06 | 15 | 3 |
| Protein processing in endoplasmic reticulum | 6.2 × 10−05 | 25 | 3 |
| Proteoglycans in cancer | 4.2 × 10−4 | 24 | 3 |
| Lysine degradation | 9.3 × 10−4 | 7 | 2 |
| Cell cycle | 4.8 × 10−3 | 19 | 3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hicks, S.D.; Olympia, R.P.; Onks, C.; Kim, R.Y.; Zhen, K.J.; Fedorchak, G.; DeVita, S.; Rangnekar, A.; Heller, M.; Zwibel, H.; et al. Saliva microRNA Biomarkers of Cumulative Concussion. Int. J. Mol. Sci. 2020, 21, 7758. https://doi.org/10.3390/ijms21207758
Hicks SD, Olympia RP, Onks C, Kim RY, Zhen KJ, Fedorchak G, DeVita S, Rangnekar A, Heller M, Zwibel H, et al. Saliva microRNA Biomarkers of Cumulative Concussion. International Journal of Molecular Sciences. 2020; 21(20):7758. https://doi.org/10.3390/ijms21207758
Chicago/Turabian StyleHicks, Steven D., Robert P. Olympia, Cayce Onks, Raymond Y. Kim, Kevin J. Zhen, Gregory Fedorchak, Samantha DeVita, Aakanksha Rangnekar, Matthew Heller, Hallie Zwibel, and et al. 2020. "Saliva microRNA Biomarkers of Cumulative Concussion" International Journal of Molecular Sciences 21, no. 20: 7758. https://doi.org/10.3390/ijms21207758
APA StyleHicks, S. D., Olympia, R. P., Onks, C., Kim, R. Y., Zhen, K. J., Fedorchak, G., DeVita, S., Rangnekar, A., Heller, M., Zwibel, H., Monteith, C., Gagnon, Z., McLoughlin, C. D., Randall, J., Madeira, M., Campbell, T. R., Fengler, E., Dretsch, M. N., Neville, C., & Middleton, F. A. (2020). Saliva microRNA Biomarkers of Cumulative Concussion. International Journal of Molecular Sciences, 21(20), 7758. https://doi.org/10.3390/ijms21207758





