Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer
Abstract
1. Introduction
2. Results
2.1. The Effects and the Safety of FMT on Colorectal Cancer-Implanted Mice Challenged with FOLFOX
2.2. The Effects of FMT on FOLFOX-Induced Intestinal Mucosal Damage in Colorectal Carcinoma-Implanted Mice
2.3. The Effects of FMT on the Epithelial Barrier of Villi in Colorectal Cancer-Bearing Mice after FOLFOX Treatment
2.4. The Effects of FMT on Proliferation, Regeneration, and Apoptosis of Crypts in Colorectal Cancer-Bearing Mice after FOLFOX Treatment
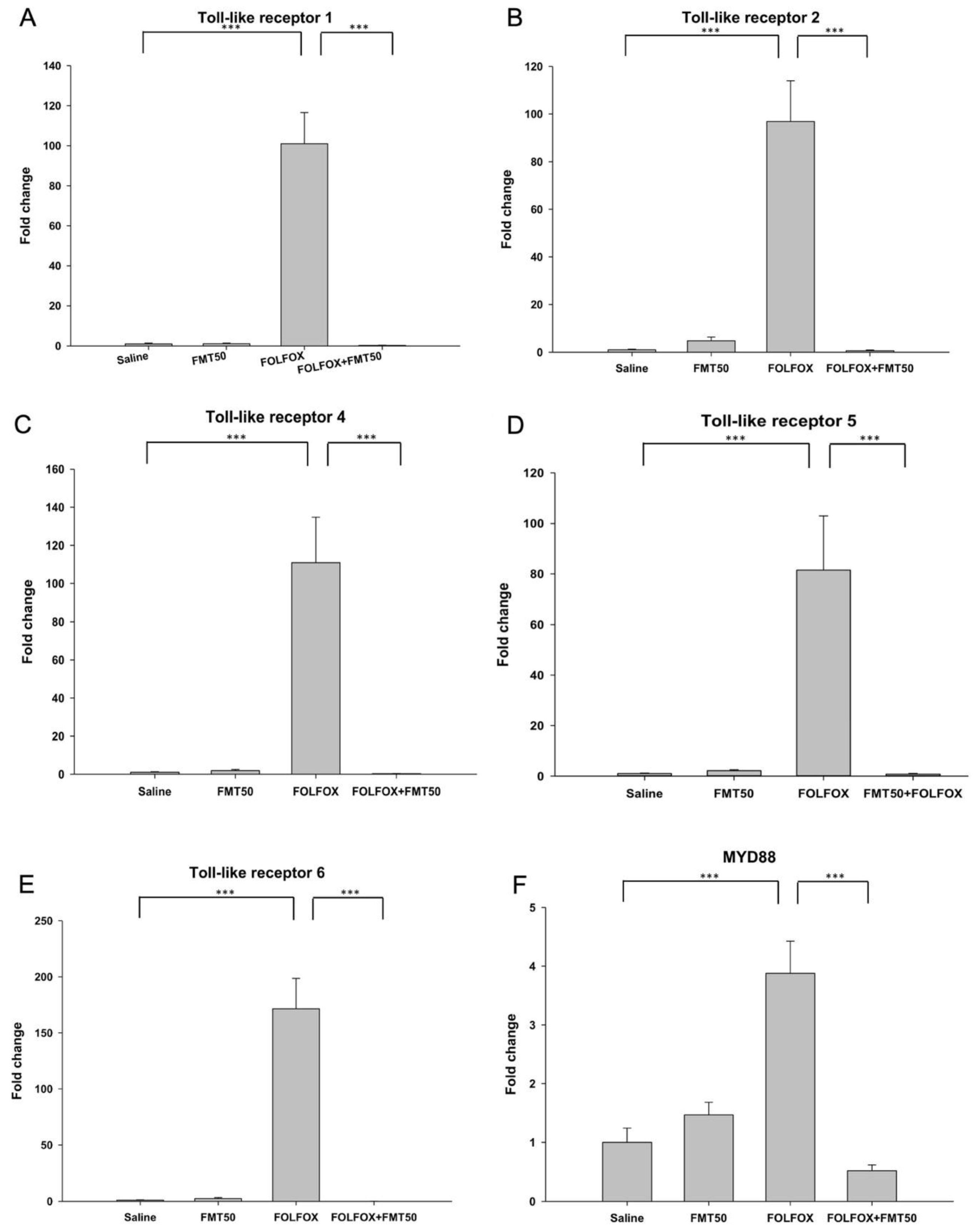
2.5. The Effects of FMT on mRNA Expression Levels of Tight Junction Proteins and Toll-Like Receptors in the Jejunum of Colorectal Cancer-Bearing Mice Challenged with FOLFOX
2.6. The Effects of FMT on the Levels of Serum Inflammatory Cytokines in Colorectal Cancer-Bearing Mice Challenged with FOLFOX
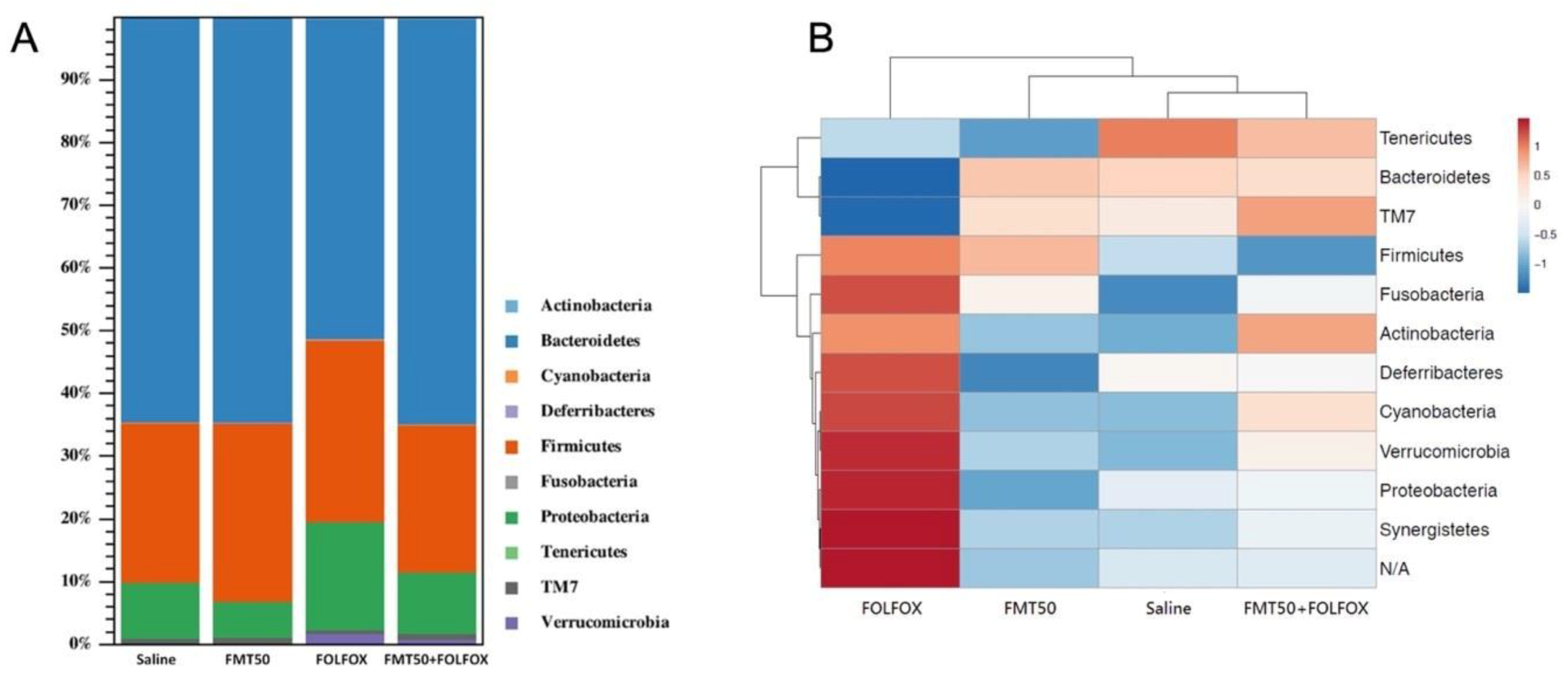
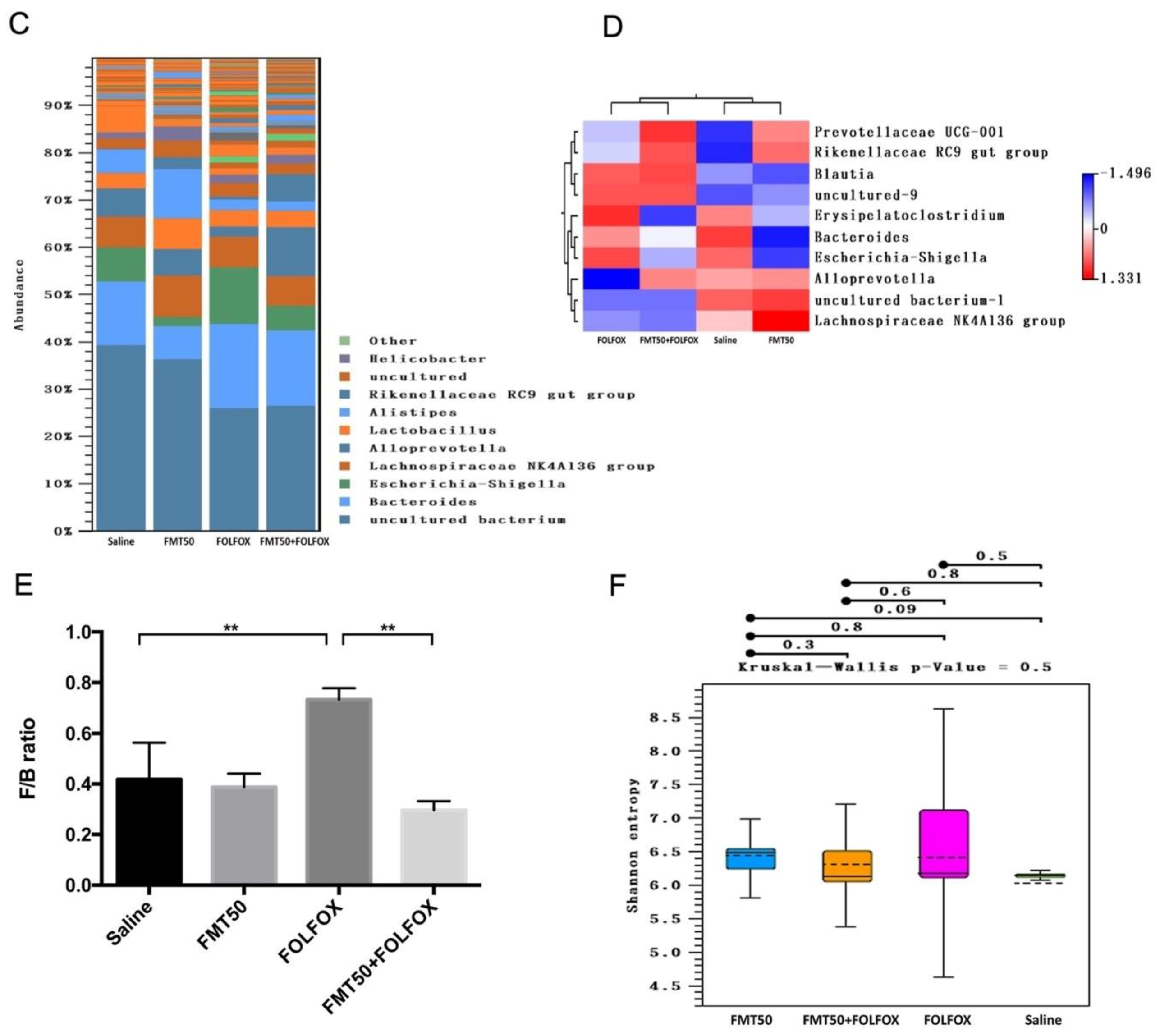
2.7. The Effects of FMT on the Fecal Gut Microbiota Composition of FOLFOX-Challenged, Colorectal Cancer-Bearing Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Cell Culture
4.3. Tumor Implantation
4.4. Transplantation of Fecal Microbiota
4.5. Chemotherapy Regimen
4.6. Disease Severity Evaluation
4.7. Safety of FMT: Translocation of Gut Microbiota
4.8. Histological Analyses
4.9. Immunohistochemical Staining
4.10. Inflammatory Cytokine Analysis
4.11. Relative Expression of mRNA Using Real-Time Quantitative (q)PCR
4.12. Gut Microbiota Analysis
4.12.1. Sample Collection and DNA Extraction
4.12.2. Sequencing and Analysis of 16S rRNA
4.13. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CLDNs | Claudin(s) |
| EDTA | Ethylenediaminetetraacetic acid |
| FMT | Fecal microbiota transplant |
| FOLFOX | 5-FU, leucovorin, and oxaliplatin |
| 5-FU | 5-fluorouracil |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase |
| IL | Interleukin |
| I.P. | Intraperitoneally |
| IRF1 | IFN-regulatory factor 1 |
| JAM-A | Junctional adhesion molecule-A |
| MAPKs | Mitogen activated protein kinases |
| MRS | de Man, Rogosa and Sharpe Broth |
| MyD88 | Myeloid differentiation primary-response gene 88 |
| NF-κB | Nuclear factor-κB |
| OTUs | Operational taxonomic unit (s) |
| PAS/AB | Periodic acid–Schiff /alcian blue |
| RPMI | Roswell Park Memorial Institute |
| TBST | Tris-buffered saline, 0.1% Tween 20 |
| TEG | Triethylene glycol |
| TIR | Toll/ interleukin-1 receptor |
| TIRAP/MAL | MyD88-adaptor-like protein |
| TJ | Tight junction |
| TLR | Toll-like receptor |
| TNF-α | Tumor necrosis factor alpha |
| TRAM | TRIF-related adaptor molecule |
| TRIF | TIR-domain-containing adaptor protein inducing interferon-β |
| TUNEL | Terminal deoxynucleotidyl transferase dUTP nick-end labeling |
| ZO-1 | Zonula occludens-1 |
References
- Aarnoutse, R.; Ziemons, J.; Penders, J.; Rensen, S.S.; de Vos-Geelen, J.; Smidt, M.L. The Clinical Link between Human Intestinal Microbiota and Systemic Cancer Therapy. Int. J. Mol. Sci. 2019, 20, 4145. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.L.; Wilson, I.D.; Teare, J.; Marchesi, J.R.; Nicholson, J.K.; Kinross, J.M. Gut microbiota modulation of chemotherapy efficacy and toxicity. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Villeger, R.; Lopes, A.; Carrier, G.; Veziant, J.; Billard, E.; Barnich, N.; Gagniere, J.; Vazeille, E.; Bonnet, M. Intestinal Microbiota: A Novel Target to Improve Anti-Tumor Treatment? Int. J. Mol. Sci. 2019, 20, 4584. [Google Scholar] [CrossRef]
- Stringer, A.M.; Gibson, R.J.; Bowen, J.M.; Keefe, D.M. Chemotherapy-induced modifications to gastrointestinal microflora: Evidence and implications of change. Curr. Drug Metab. 2009, 10, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Stringer, A.M.; Gibson, R.J.; Logan, R.M.; Bowen, J.M.; Yeoh, A.S.; Hamilton, J.; Keefe, D.M. Gastrointestinal microflora and mucins may play a critical role in the development of 5-Fluorouracil-induced gastrointestinal mucositis. Exp. Biol. Med. 2009, 234, 430–441. [Google Scholar] [CrossRef]
- Touchefeu, Y.; Montassier, E.; Nieman, K.; Gastinne, T.; Potel, G.; Bruley des Varannes, S.; Le Vacon, F.; de La Cochetiere, M.F. Systematic review: The role of the gut microbiota in chemotherapy- or radiation-induced gastrointestinal mucositis—Current evidence and potential clinical applications. Aliment. Pharmacol. Ther. 2014, 40, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Ryan, E.J.; Doherty, G.A. Gastro-intestinal toxicity of chemotherapeutics in colorectal cancer: The role of inflammation. World J. Gastroenterol. 2014, 20, 3751–3761. [Google Scholar] [CrossRef]
- Sharma, R.; Tobin, P.; Clarke, S.J. Management of chemotherapy-induced nausea, vomiting, oral mucositis, and diarrhoea. Lancet Oncol. 2005, 6, 93–102. [Google Scholar] [CrossRef]
- Logan, R.M.; Stringer, A.M.; Bowen, J.M.; Yeoh, A.S.; Gibson, R.J.; Sonis, S.T.; Keefe, D.M. The role of pro-inflammatory cytokines in cancer treatment-induced alimentary tract mucositis: Pathobiology, animal models and cytotoxic drugs. Cancer Treat. Rev. 2007, 33, 448–460. [Google Scholar] [CrossRef]
- van Nood, E.; Vrieze, A.; Nieuwdorp, M.; Fuentes, S.; Zoetendal, E.G.; de Vos, W.M.; Visser, C.E.; Kuijper, E.J.; Bartelsman, J.F.; Tijssen, J.G.; et al. Duodenal infusion of donor feces for recurrent Clostridium difficile. N. Engl. J. Med. 2013, 368, 407–415. [Google Scholar] [CrossRef]
- Damman, C.J.; Miller, S.I.; Surawicz, C.M.; Zisman, T.L. The microbiome and inflammatory bowel disease: Is there a therapeutic role for fecal microbiota transplantation? Am. J. Gastroenterol. 2012, 107, 1452–1459. [Google Scholar] [CrossRef]
- Smits, L.P.; Bouter, K.E.; de Vos, W.M.; Borody, T.J.; Nieuwdorp, M. Therapeutic potential of fecal microbiota transplantation. Gastroenterology 2013, 145, 946–953. [Google Scholar] [CrossRef]
- Ooijevaar, R.E.; Terveer, E.M.; Verspaget, H.W.; Kuijper, E.J.; Keller, J.J. Clinical Application and Potential of Fecal Microbiota Transplantation. Annu. Rev. Med. 2019, 70, 335–351. [Google Scholar] [CrossRef]
- Khoruts, A.; Sadowsky, M.J. Understanding the mechanisms of faecal microbiota transplantation. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 508–516. [Google Scholar] [CrossRef]
- Wang, S.; Xu, M.; Wang, W.; Cao, X.; Piao, M.; Khan, S.; Yan, F.; Cao, H.; Wang, B. Systematic Review: Adverse Events of Fecal Microbiota Transplantation. PLoS ONE 2016, 11, e0161174. [Google Scholar] [CrossRef]
- Cammarota, G.; Ianiro, G.; Tilg, H.; Rajilic-Stojanovic, M.; Kump, P.; Satokari, R.; Sokol, H.; Arkkila, P.; Pintus, C.; Hart, A.; et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut 2017, 66, 569–580. [Google Scholar] [CrossRef]
- Xu, M.Q.; Cao, H.L.; Wang, W.Q.; Wang, S.; Cao, X.C.; Yan, F.; Wang, B.M. Fecal microbiota transplantation broadening its application beyond intestinal disorders. World J. Gastroenterol. 2015, 21, 102–111. [Google Scholar] [CrossRef]
- Hefazi, M.; Patnaik, M.M.; Hogan, W.J.; Litzow, M.R.; Pardi, D.S.; Khanna, S. Safety and Efficacy of Fecal Microbiota Transplant for Recurrent Clostridium difficile Infection in Patients With Cancer Treated With Cytotoxic Chemotherapy: A Single-Institution Retrospective Case Series. Mayo Clin. Proc. 2017, 92, 1617–1624. [Google Scholar] [CrossRef]
- Moss, E.L.; Falconer, S.B.; Tkachenko, E.; Wang, M.; Systrom, H.; Mahabamunuge, J.; Relman, D.A.; Hohmann, E.L.; Bhatt, A.S. Long-term taxonomic and functional divergence from donor bacterial strains following fecal microbiota transplantation in immunocompromised patients. PLoS ONE 2017, 12, e0182585. [Google Scholar] [CrossRef]
- Kelly, C.R.; Ihunnah, C.; Fischer, M.; Khoruts, A.; Surawicz, C.; Afzali, A.; Aroniadis, O.; Barto, A.; Borody, T.; Giovanelli, A.; et al. Fecal microbiota transplant for treatment of Clostridium difficile infection in immunocompromised patients. Am. J. Gastroenterol. 2014, 109, 1065–1071. [Google Scholar] [CrossRef]
- Mohelnikova-Duchonova, B.; Melichar, B.; Soucek, P. FOLFOX/FOLFIRI pharmacogenetics: The call for a personalized approach in colorectal cancer therapy. World J. Gastroenterol. 2014, 20, 10316–10330. [Google Scholar] [CrossRef]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Bano, N.; Najam, R.; Qazi, F.; Mateen, A. Gastrointestinal adverse effects in advanced colorectal carcinoma patients treated with different schedules of FOLFOX. Asian Pac. J. Cancer Prev. 2014, 15, 8089–8093. [Google Scholar] [CrossRef]
- Chang, C.W.; Liu, C.Y.; Lee, H.C.; Huang, Y.H.; Li, L.H.; Chiau, J.C.; Wang, T.E.; Chu, C.H.; Shih, S.C.; Tsai, T.H.; et al. Lactobacillus casei Variety rhamnosus Probiotic Preventively Attenuates 5-Fluorouracil/Oxaliplatin-Induced Intestinal Injury in a Syngeneic Colorectal Cancer Model. Front. Microbiol. 2018, 9, 983. [Google Scholar] [CrossRef]
- Wells, J.M. Immunomodulatory mechanisms of lactobacilli. Microb. Cell Factories 2011, 10 (Suppl. 1), S17. [Google Scholar] [CrossRef]
- Cario, E.; Gerken, G.; Podolsky, D.K. Toll-like receptor 2 enhances ZO-1-associated intestinal epithelial barrier integrity via protein kinase C. Gastroenterology 2004, 127, 224–238. [Google Scholar] [CrossRef]
- Tian, Z.; Liu, J.; Liao, M.; Li, W.; Zou, J.; Han, X.; Kuang, M.; Shen, W.; Li, H. Beneficial Effects of Fecal Microbiota Transplantation on Ulcerative Colitis in Mice. Dig. Dis. Sci. 2016, 61, 2262–2271. [Google Scholar] [CrossRef]
- Li, X.; Li, X.; Shang, Q.; Gao, Z.; Hao, F.; Guo, H.; Guo, C. Fecal microbiota transplantation (FMT) could reverse the severity of experimental necrotizing enterocolitis (NEC) via oxidative stress modulation. Free Radic. Biol. Med. 2017, 108, 32–43. [Google Scholar] [CrossRef]
- Cui, M.; Xiao, H.; Li, Y.; Zhou, L.; Zhao, S.; Luo, D.; Zheng, Q.; Dong, J.; Zhao, Y.; Zhang, X.; et al. Faecal microbiota transplantation protects against radiation-induced toxicity. EMBO Mol. Med. 2017, 9, 448–461. [Google Scholar] [CrossRef]
- Li, H.L.; Lu, L.; Wang, X.S.; Qin, L.Y.; Wang, P.; Qiu, S.P.; Wu, H.; Huang, F.; Zhang, B.B.; Shi, H.L.; et al. Alteration of Gut Microbiota and Inflammatory Cytokine/Chemokine Profiles in 5-Fluorouracil Induced Intestinal Mucositis. Front. Cell. Infect. Microbiol. 2017, 7, 455. [Google Scholar] [CrossRef]
- Wardill, H.R.; Bowen, J.M. Chemotherapy-induced mucosal barrier dysfunction: An updated review on the role of intestinal tight junctions. Curr. Opin. Support. Palliat. Care 2013, 7, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Capaldo, C.T.; Powell, D.N.; Kalman, D. Layered defense: How mucus and tight junctions seal the intestinal barrier. J. Mol. Med. 2017, 95, 927–934. [Google Scholar] [CrossRef] [PubMed]
- France, M.M.; Turner, J.R. The mucosal barrier at a glance. J. Cell Sci. 2017, 130, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Miwa, H. Gastrointestinal mucosal barrier function and diseases. J. Gastroenterol. 2016, 51, 768–778. [Google Scholar] [CrossRef]
- Volynets, V.; Rings, A.; Bardos, G.; Ostaff, M.J.; Wehkamp, J.; Bischoff, S.C. Intestinal barrier analysis by assessment of mucins, tight junctions, and alpha-defensins in healthy C57BL/6J and BALB/cJ mice. Tissue Barriers 2016, 4, e1208468. [Google Scholar] [CrossRef]
- Song, M.K.; Park, M.Y.; Sung, M.K. 5-Fluorouracil-induced changes of intestinal integrity biomarkers in BALB/c mice. J. Cancer Prev. 2013, 18, 322–329. [Google Scholar] [CrossRef]
- de Barros, P.A.V.; Rabelo Andrade, M.E.; de Vasconcelos Generoso, S.; Mendes Miranda, S.E.; Dos Reis, D.C.; Lacerda Leocadio, P.C.; de Sales, E.S.E.L.; Dos Santos Martins, F.; da Gama, M.A.S.; Cassali, G.D.; et al. Conjugated linoleic acid prevents damage caused by intestinal mucositis induced by 5-fluorouracil in an experimental model. Biomed. Pharmacother. 2018, 103, 1567–1576. [Google Scholar] [CrossRef]
- Danai, P.A.; Moss, M.; Mannino, D.M.; Martin, G.S. The epidemiology of sepsis in patients with malignancy. Chest 2006, 129, 1432–1440. [Google Scholar] [CrossRef]
- Pagano, L.; Tacconelli, E.; Tumbarello, M.; Laurenti, L.; Ortu-La Barbera, E.; Antinori, A.; Caponera, S.; Cauda, R.; Leone, G. Bacteremia in patients with hematological malignancies. Analysis of risk factors, etiological agents and prognostic indicators. Haematologica 1997, 82, 415–419. [Google Scholar]
- Papanicolas, L.E.; Gordon, D.L.; Wesselingh, S.L.; Rogers, G.B. Not Just Antibiotics: Is Cancer Chemotherapy Driving Antimicrobial Resistance? Trends Microbiol. 2017. [Google Scholar] [CrossRef]
- Goldberg, R.M. Therapy for metastatic colorectal cancer. Oncologist 2006, 11, 981–987. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rakoff-Nahoum, S.; Paglino, J.; Eslami-Varzaneh, F.; Edberg, S.; Medzhitov, R. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis. Cell 2004, 118, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.W.; Chang, W.J.; Chen, P.H.; Hsu, C.M. Commensal microflora induce host defense and decrease bacterial translocation in burn mice through toll-like receptor 4. J. Biomed. Sci. 2010, 17, 48. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 2015, 16, 343–353. [Google Scholar] [CrossRef] [PubMed]
- Frosali, S.; Pagliari, D.; Gambassi, G.; Landolfi, R.; Pandolfi, F.; Cianci, R. How the Intricate Interaction among Toll-Like Receptors, Microbiota, and Intestinal Immunity Can Influence Gastrointestinal Pathology. J. Immunol. Res. 2015, 2015, 489821. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Kawai, T. Toll-like receptor signaling pathways. Front. Immunol. 2014, 5, 461. [Google Scholar] [CrossRef]
- Thorpe, D.W.; Stringer, A.M.; Gibson, R.J. Chemotherapy-induced mucositis: The role of the gastrointestinal microbiome and toll-like receptors. Exp. Biol. Med. 2013, 238, 1–6. [Google Scholar] [CrossRef]
- O′Neill, L.A.; Golenbock, D.; Bowie, A.G. The history of Toll-like receptors—Redefining innate immunity. Nat. Rev. Immunol. 2013, 13, 453–460. [Google Scholar] [CrossRef]
- Abreu, M.T. Toll-like receptor signalling in the intestinal epithelium: How bacterial recognition shapes intestinal function. Nat. Rev. Immunol. 2010, 10, 131–144. [Google Scholar] [CrossRef]
- Pekkala, S.; Keskitalo, A.; Kettunen, E.; Lensu, S.; Nykanen, N.; Kuopio, T.; Ritvos, O.; Hentila, J.; Nissinen, T.A.; Hulmi, J.J. Blocking Activin Receptor Ligands Is Not Sufficient to Rescue Cancer-Associated Gut Microbiota-A Role for Gut Microbial Flagellin in Colorectal Cancer and Cachexia? Cancers 2019, 11, 1799. [Google Scholar] [CrossRef]
- Chang, C.T.; Ho, T.Y.; Lin, H.; Liang, J.A.; Huang, H.C.; Li, C.C.; Lo, H.Y.; Wu, S.L.; Huang, Y.F.; Hsiang, C.Y. 5-Fluorouracil induced intestinal mucositis via nuclear factor-kappaB activation by transcriptomic analysis and in vivo bioluminescence imaging. PLoS ONE 2012, 7, e31808. [Google Scholar]
- Suzuki, T.; Yoshinaga, N.; Tanabe, S. Interleukin-6 (IL-6) regulates claudin-2 expression and tight junction permeability in intestinal epithelium. J. Biol. Chem. 2011, 286, 31263–31271. [Google Scholar] [CrossRef] [PubMed]
- Al-Dasooqi, N.; Sonis, S.T.; Bowen, J.M.; Bateman, E.; Blijlevens, N.; Gibson, R.J.; Logan, R.M.; Nair, R.G.; Stringer, A.M.; Yazbeck, R.; et al. Emerging evidence on the pathobiology of mucositis. Support. Care Cancer 2013, 21, 3233–3241. [Google Scholar] [CrossRef] [PubMed]
- van Vliet, M.J.; Harmsen, H.J.; de Bont, E.S.; Tissing, W.J. The role of intestinal microbiota in the development and severity of chemotherapy-induced mucositis. PLoS Pathog. 2010, 6, e1000879. [Google Scholar] [CrossRef]
- Boulange, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016, 8, 42. [Google Scholar] [CrossRef]
- Sougiannis, A.T.; VanderVeen, B.N.; Enos, R.T.; Velazquez, K.T.; Bader, J.E.; Carson, M.; Chatzistamou, I.; Walla, M.; Pena, M.M.; Kubinak, J.L.; et al. Impact of 5 Fluorouracil Chemotherapy on Gut Inflammation, Functional Parameters, and Gut Microbiota. Brain Behav. Immun. 2019. [Google Scholar] [CrossRef]
- Eckburg, P.B.; Bik, E.M.; Bernstein, C.N.; Purdom, E.; Dethlefsen, L.; Sargent, M.; Gill, S.R.; Nelson, K.E.; Relman, D.A. Diversity of the human intestinal microbial flora. Science 2005, 308, 1635–1638. [Google Scholar] [CrossRef]
- Wang, A.; Ling, Z.; Yang, Z.; Kiela, P.R.; Wang, T.; Wang, C.; Cao, L.; Geng, F.; Shen, M.; Ran, X.; et al. Gut microbial dysbiosis may predict diarrhea and fatigue in patients undergoing pelvic cancer radiotherapy: A pilot study. PLoS ONE 2015, 10, e0126312. [Google Scholar] [CrossRef]
- Le Bastard, Q.; Ward, T.; Sidiropoulos, D.; Hillmann, B.M.; Chun, C.L.; Sadowsky, M.J.; Knights, D.; Montassier, E. Fecal microbiota transplantation reverses antibiotic and chemotherapy-induced gut dysbiosis in mice. Sci. Rep. 2018, 8, 6219. [Google Scholar] [CrossRef]
- Montonye, D.R.; Ericsson, A.C.; Busi, S.B.; Lutz, C.; Wardwell, K.; Franklin, C.L. Acclimation and Institutionalization of the Mouse Microbiota Following Transportation. Front. Microbiol. 2018, 9, 1085. [Google Scholar] [CrossRef]
- Laukens, D.; Brinkman, B.M.; Raes, J.; De Vos, M.; Vandenabeele, P. Heterogeneity of the gut microbiome in mice: Guidelines for optimizing experimental design. FEMS Microbiol. Rev. 2016, 40, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Yeung, C.Y.; Chan, W.T.; Jiang, C.B.; Cheng, M.L.; Liu, C.Y.; Chang, S.W.; Chiang Chiau, J.S.; Lee, H.C. Amelioration of Chemotherapy-Induced Intestinal Mucositis by Orally Administered Probiotics in a Mouse Model. PLoS ONE 2015, 10, e0138746. [Google Scholar] [CrossRef] [PubMed]
- Brattain, M.G.; Strobel-Stevens, J.; Fine, D.; Webb, M.; Sarrif, A.M. Establishment of mouse colonic carcinoma cell lines with different metastatic properties. Cancer Res. 1980, 40, 2142–2146. [Google Scholar] [PubMed]
- Li, M.; Liang, P.; Li, Z.; Wang, Y.; Zhang, G.; Gao, H.; Wen, S.; Tang, L. Fecal microbiota transplantation and bacterial consortium transplantation have comparable effects on the re-establishment of mucosal barrier function in mice with intestinal dysbiosis. Front. Microbiol. 2015, 6, 692. [Google Scholar] [CrossRef] [PubMed]
- El-Salhy, M.; Hilding, L.; Royson, H.; Tjomsland, V. Comparison between triple therapy with octreotide, galanin and serotonin vs. irinotecan or oxaliplatin in combination with 5-fluorouracil/leukovorin in human colon cancer. Int. J. Oncol. 2005, 27, 687–691. [Google Scholar]
- Wagner, M.; Roh, V.; Strehlen, M.; Laemmle, A.; Stroka, D.; Egger, B.; Trochsler, M.; Hunt, K.K.; Candinas, D.; Vorburger, S.A. Effective treatment of advanced colorectal cancer by rapamycin and 5-FU/oxaliplatin monitored by TIMP-1. J. Gastrointest. Surg. 2009, 13, 1781–1790. [Google Scholar] [CrossRef]
- Liu, C.Y.; Liao, H.F.; Wang, T.E.; Lin, S.C.; Shih, S.C.; Chang, W.H.; Yang, Y.C.; Lin, C.C.; Chen, Y.J. Etoposide sensitizes CT26 colorectal adenocarcinoma to radiation therapy in BALB/c mice. World J. Gastroenterol. 2005, 11, 4895–4898. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.M.; Stringer, A.M.; Gibson, R.J.; Yeoh, A.S.; Hannam, S.; Keefe, D.M. VSL#3 probiotic treatment reduces chemotherapy-induced diarrhea and weight loss. Cancer Biol. Ther. 2007, 6, 1449–1454. [Google Scholar]
- Carasi, P.; Diaz, M.; Racedo, S.M.; De Antoni, G.; Urdaci, M.C.; Serradell Mde, L. Safety characterization and antimicrobial properties of kefir-isolated Lactobacillus kefiri. Biomed. Res. Int. 2014, 2014, 208974. [Google Scholar] [CrossRef]
- Chen, Y.X.; Lai, L.N.; Zhang, H.Y.; Bi, Y.H.; Meng, L.; Li, X.J.; Tian, X.X.; Wang, L.M.; Fan, Y.M.; Zhao, Z.F.; et al. Effect of artesunate supplementation on bacterial translocation and dysbiosis of gut microbiota in rats with liver cirrhosis. World J. Gastroenterol. 2016, 22, 2949–2959. [Google Scholar] [CrossRef]
- Haisan, A.; Rogojanu, R.; Croitoru, C.; Jitaru, D.; Tarniceriu, C.; Danciu, M.; Carasevici, E. Digital microscopy assessment of angiogenesis in different breast cancer compartments. Biomed. Res. Int. 2013, 2013, 286902. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glockner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
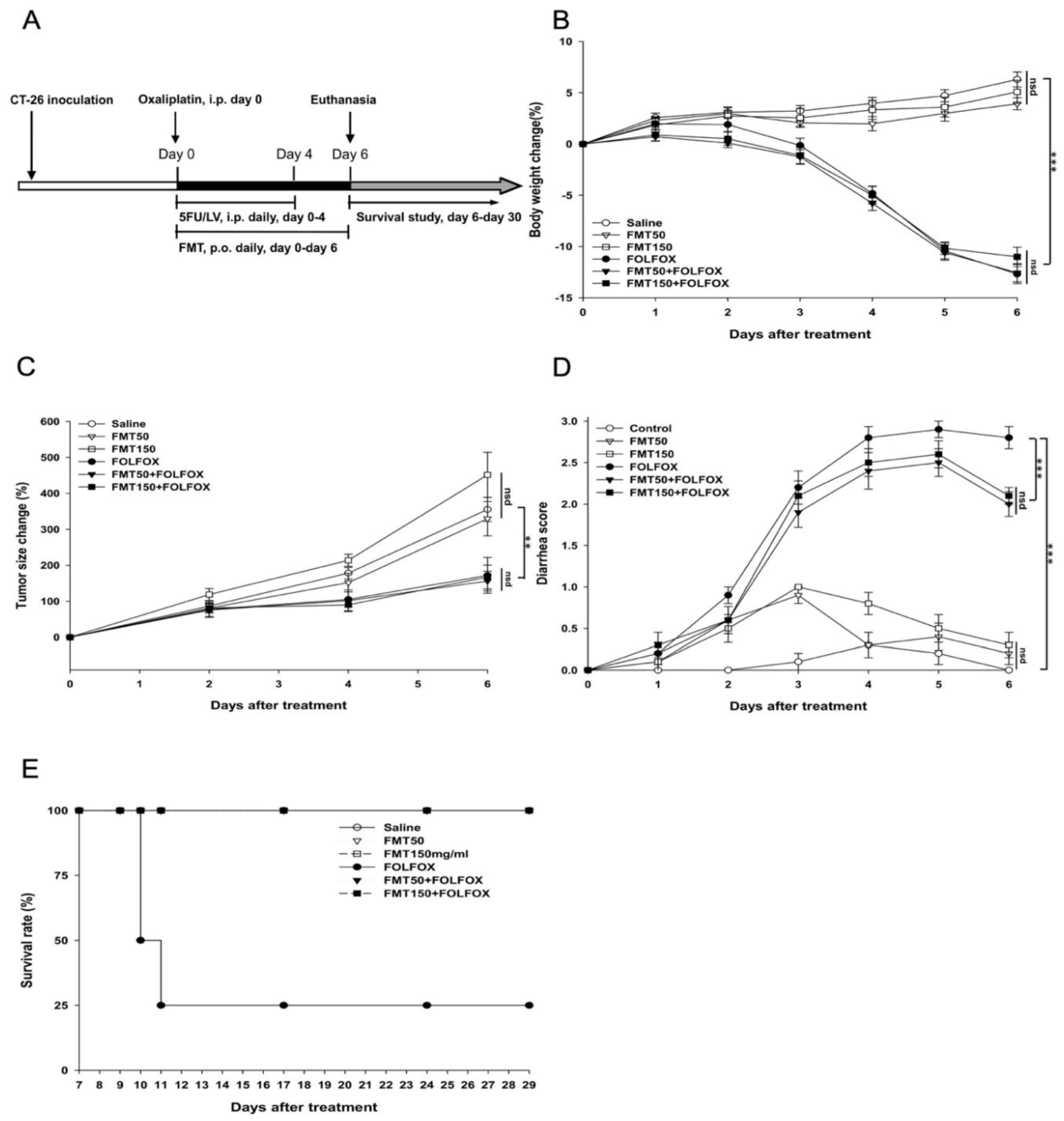
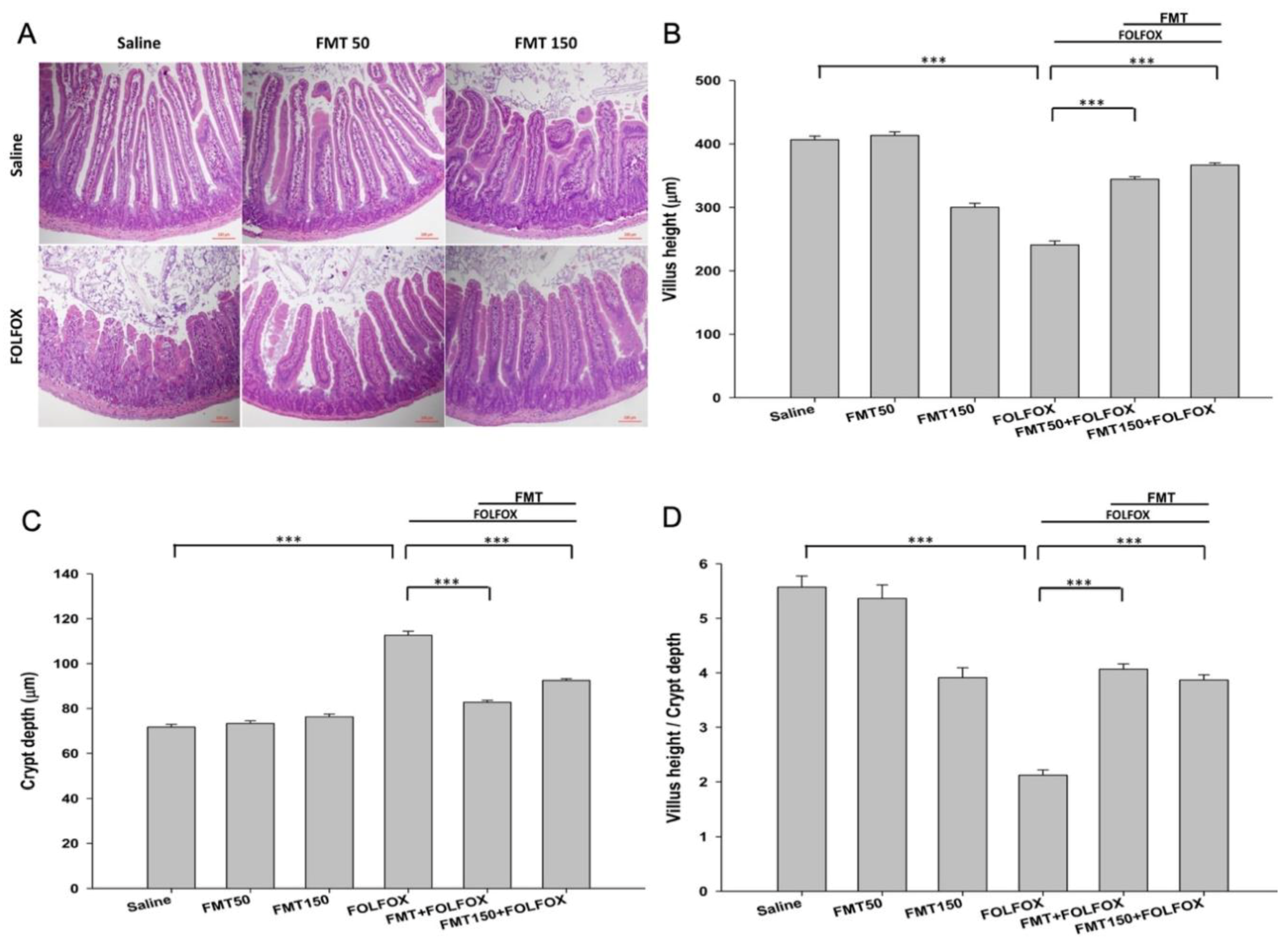
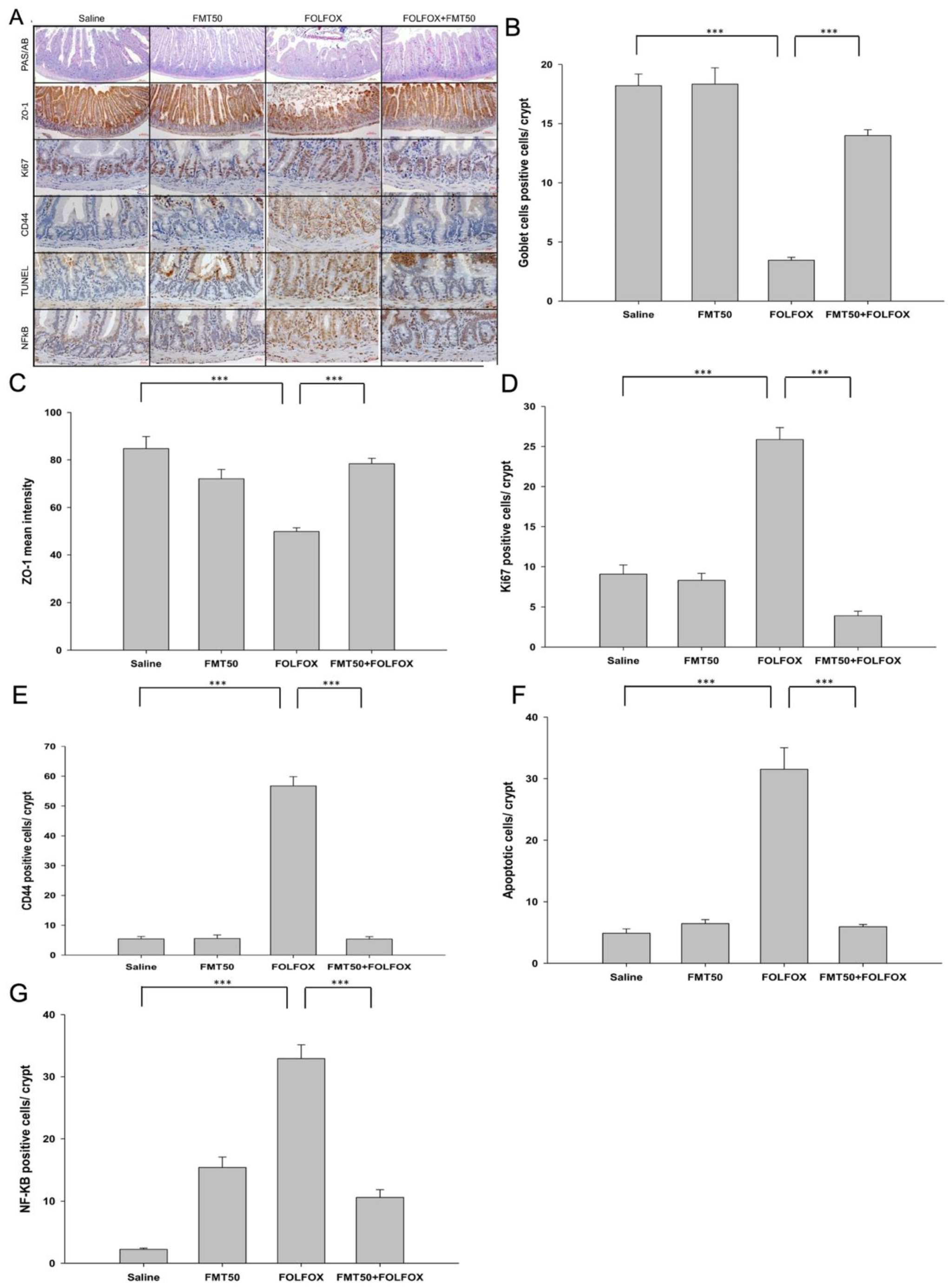
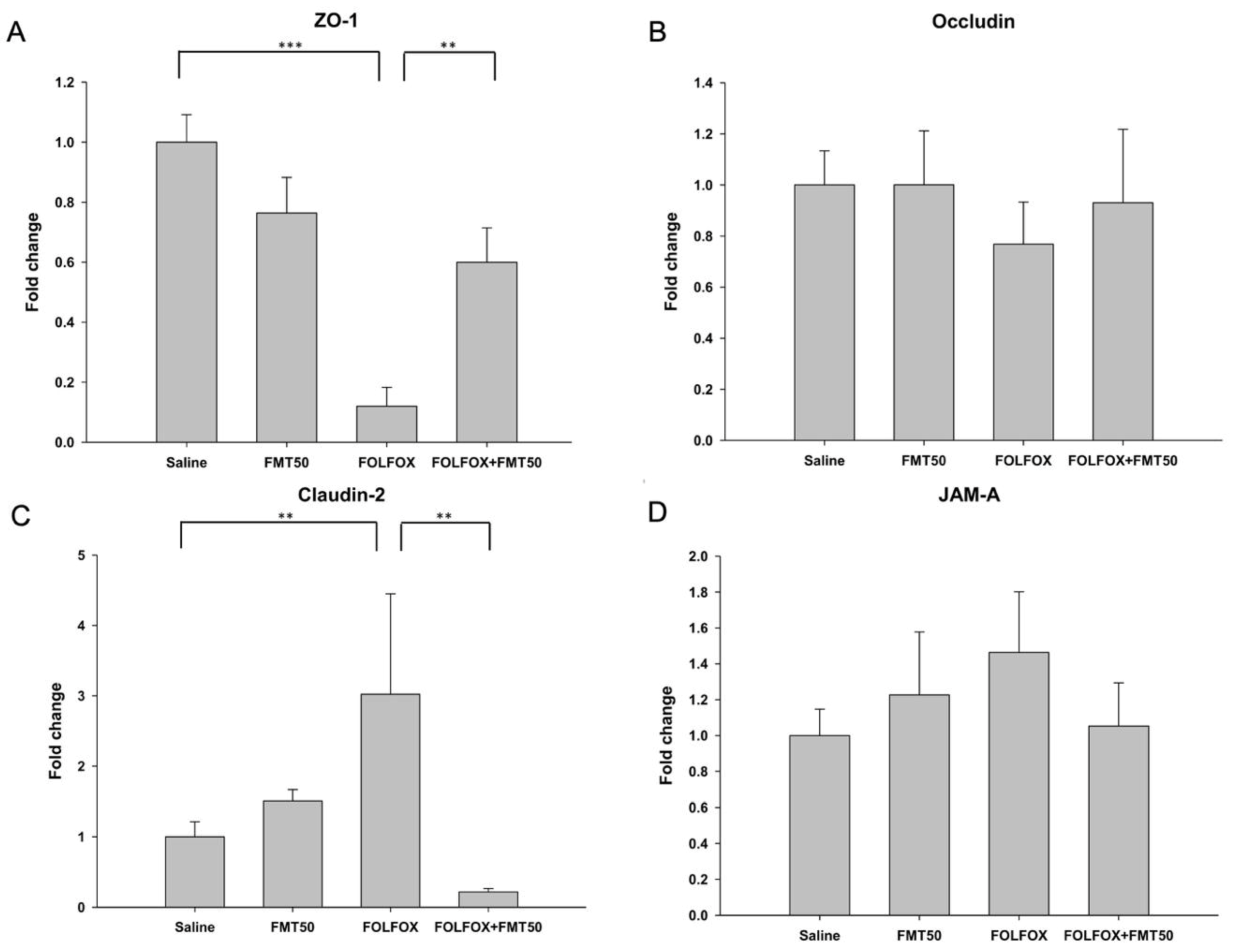

| Groups | Regimen |
|---|---|
| 1 | Saline (untreated control) |
| 2 | FMT50 |
| 3 | FMT150 |
| 4 | FOLFOX |
| 5 | FOLFOX+FMT50 |
| 6 | FOLFOX+FMT150 |
| Group | MLNs | Spleen | Liver | Blood | TR |
|---|---|---|---|---|---|
| Saline | 0/6 (0.0%) | 0/6 (0.0%) | 0/6 (0.0%) | 0/6 (0.0%) | 0/24(0.0%) |
| FMT50 | 1/6 (16.7%) | 0/6 (0.0%) | 0/6 (0.0%) | 0/6 (0.0%) | 1/24(4.2%) |
| FMT150 | 2/6 (33.3%) | 1/6 (16.7%) | 1/6 (16.7%) | 0/6 (0.0%) | 4/24(16.7%) * |
| FOLFOX | 2/6 (33.3%) | 1/6 (16.7%) | 2/6 (33.3%) | 0/6 (0.0%) | 5/24(20.8%) * |
| FOLFOX+FMT50 | 1/6 (16.7%) | 2/6 (33.3%) | 0/6 (0.0%) | 0/6 (0.0%) | 3/24(12.5%) |
| FOLFOX+FMT150 | 1/6 (16.7%) | 2/6 (33.3%) | 1/6 (16.7%) | 0/6 (0.0%) | 4/24(16.7%) * |
| Bray-Curtis | Jaccard | ||||||
|---|---|---|---|---|---|---|---|
| F | p | FDR Corrected p Values | F | p | FDR Corrected p Values | ||
| Saline | FMT50 | 2.15145 | 0.00794 | 0.009528 | 1.88344 | 0.00794 | 0.011910 |
| Saline | FOLFOX | 2.94918 | 0.03968 | 0.039680 | 2.37435 | 0.03968 | 0.039680 |
| FMT50 | FOLFOX | 4.16423 | 0.00794 | 0.009528 | 2.94634 | 0.00794 | 0.011910 |
| Saline | FOLFOX+FMT50 | 8.49609 | 0.00794 | 0.009528 | 5.64205 | 0.00794 | 0.011910 |
| FMT50 | FOLFOX+FMT50 | 10.77795 | 0.00794 | 0.009528 | 6.44849 | 0.00794 | 0.011910 |
| FOLFOX | FOLFOX+FMT50 | 2.73125 | 0.00794 | 0.009528 | 2.35479 | 0.01587 | 0.019044 |
| Gene | Forward Primer (5′→3′) | Reverse Primer (5′→3′) |
|---|---|---|
| ZO-1 | ACCCGAAACTGATGCTGTGGATAG | GTGGTTGTCACCAGCATCAG |
| Occludin | ATGTCCGGCCGATGCTCTC | TTTGGCTGCTCTTGGGTCTGTAT |
| Claudin 2 | ATGCCTTCTTGAGCCTGCTT | AAGGCCTAGGATGTAGCCCA |
| JAM-A | CTGATCTTTGACCCCGTGAC | ACCAGACGCCAAAAATCAAG |
| TLR1 | TCAAGCATTTGGACCTCTCCT | TTCTTTGCATATAGGCAGGGC |
| TLR2 | ACAATAGAGGGAGACGCCTTT | AGTGTCTGGTAAGGATTTCCCAT |
| TLR4 | ATGGCATGGCTTACACCACC | GAGGCCATTTTTGTCTCCACA |
| TLR5 | AGC CTC CGC CTC CAT TCT TC | TCA CGC CTC TGA AGG GGT TC |
| TLR6 | TGAGCCAAGACAGAAAACCCA | GGGACATGAGTAAGGTTCCTGTT |
| MYD88 | ATCGCTGTTCTTGAACCCTCG | CTCACGGTCTAACAAGGCCAG |
| GAPDH | TGAACGGGAAGCTCACTGG | TCCACCACCCTGTTGCTGTA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-W.; Lee, H.-C.; Li, L.-H.; Chiang Chiau, J.-S.; Wang, T.-E.; Chuang, W.-H.; Chen, M.-J.; Wang, H.-Y.; Shih, S.-C.; Liu, C.-Y.; et al. Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer. Int. J. Mol. Sci. 2020, 21, 386. https://doi.org/10.3390/ijms21020386
Chang C-W, Lee H-C, Li L-H, Chiang Chiau J-S, Wang T-E, Chuang W-H, Chen M-J, Wang H-Y, Shih S-C, Liu C-Y, et al. Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer. International Journal of Molecular Sciences. 2020; 21(2):386. https://doi.org/10.3390/ijms21020386
Chicago/Turabian StyleChang, Ching-Wei, Hung-Chang Lee, Li-Hui Li, Jen-Shiu Chiang Chiau, Tsang-En Wang, Wei-Hung Chuang, Ming-Jen Chen, Horng-Yuan Wang, Shou-Chuan Shih, Chia-Yuan Liu, and et al. 2020. "Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer" International Journal of Molecular Sciences 21, no. 2: 386. https://doi.org/10.3390/ijms21020386
APA StyleChang, C.-W., Lee, H.-C., Li, L.-H., Chiang Chiau, J.-S., Wang, T.-E., Chuang, W.-H., Chen, M.-J., Wang, H.-Y., Shih, S.-C., Liu, C.-Y., Tsai, T.-H., & Chen, Y.-J. (2020). Fecal Microbiota Transplantation Prevents Intestinal Injury, Upregulation of Toll-Like Receptors, and 5-Fluorouracil/Oxaliplatin-Induced Toxicity in Colorectal Cancer. International Journal of Molecular Sciences, 21(2), 386. https://doi.org/10.3390/ijms21020386






