Terpenes and Terpenoids in Plants: Interactions with Environment and Insects
Abstract
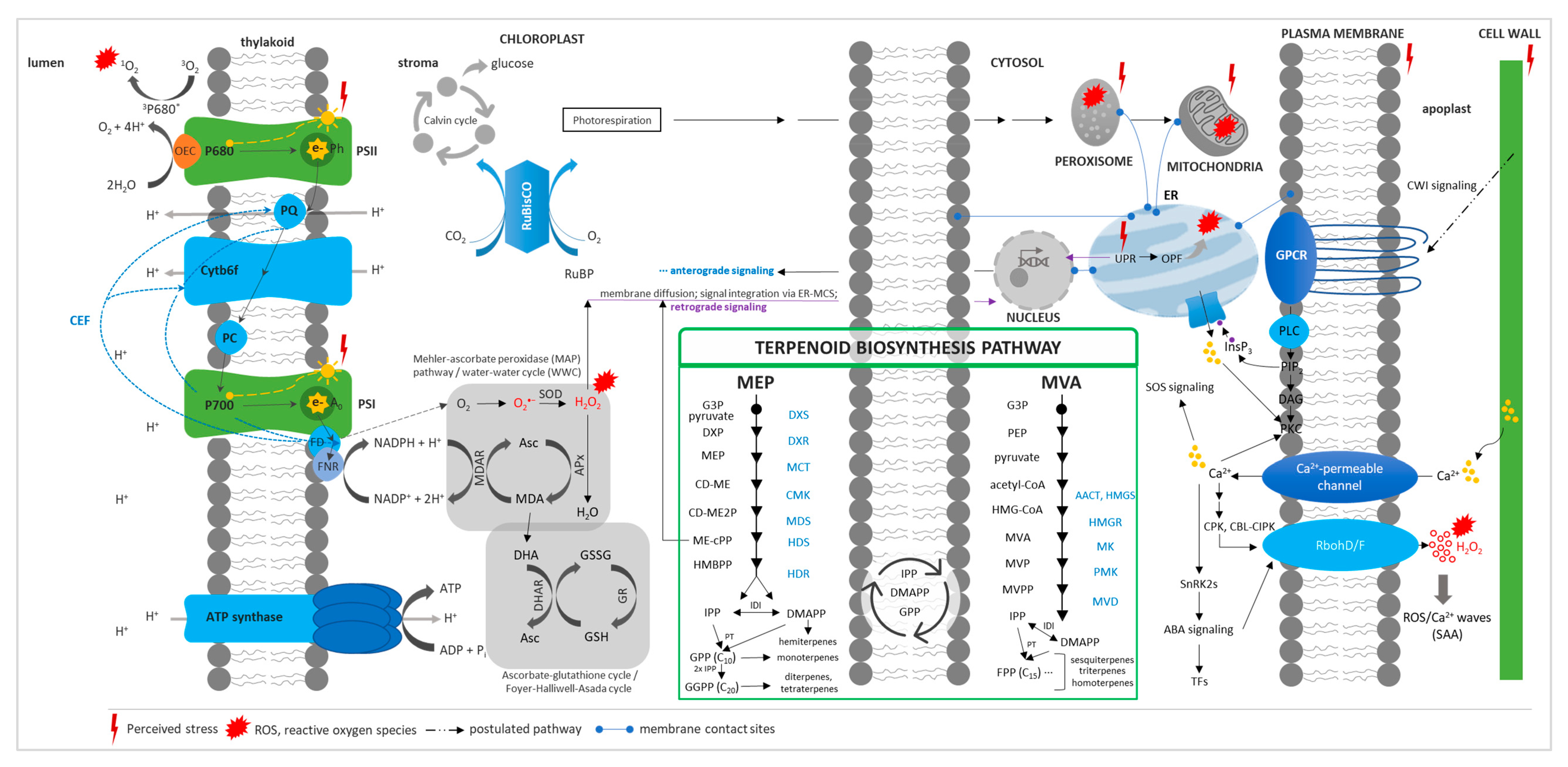
1. Plant Terpenes in Abiotic Stress
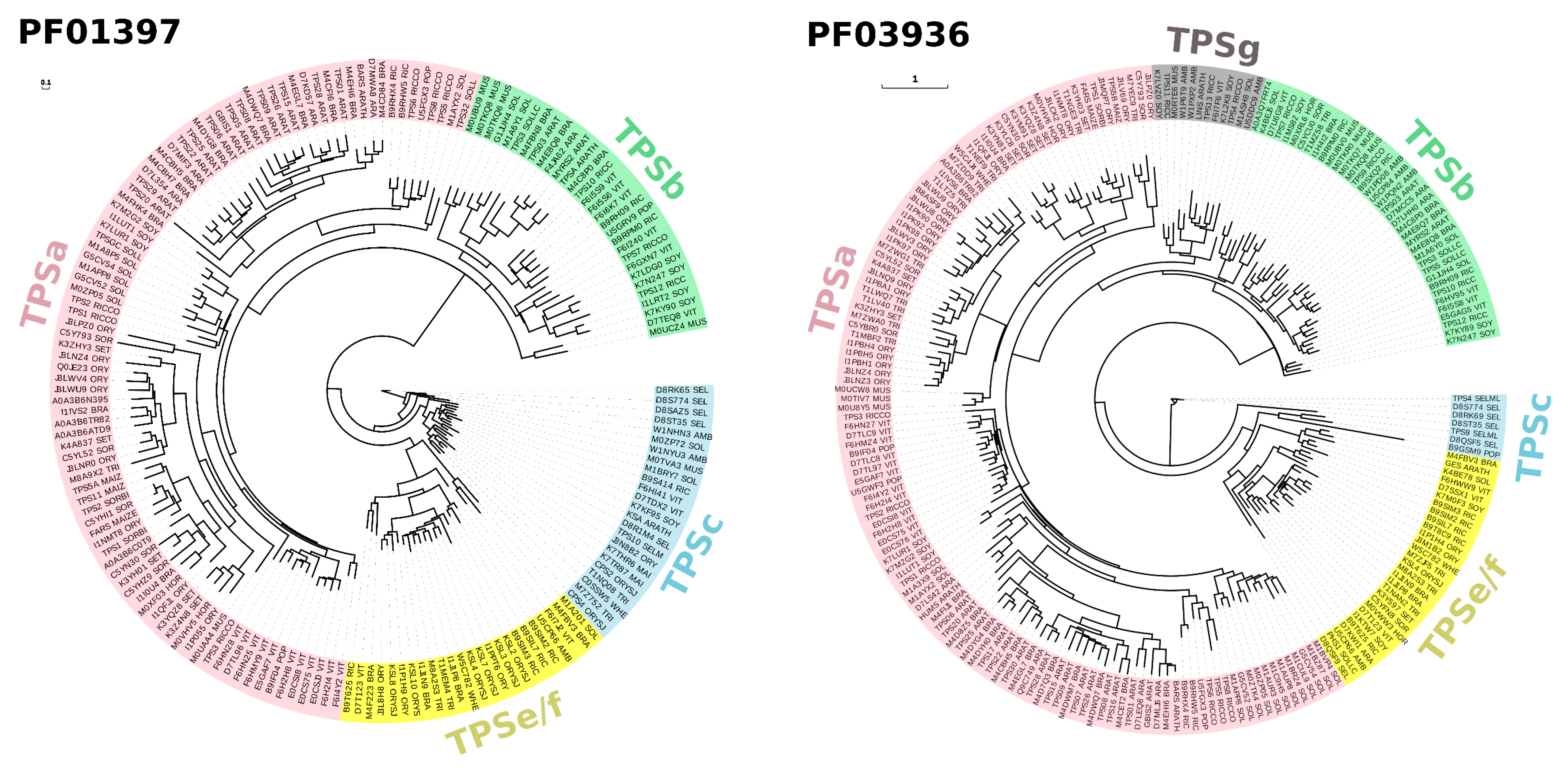
1.1. Diversity of Plant Terpenes
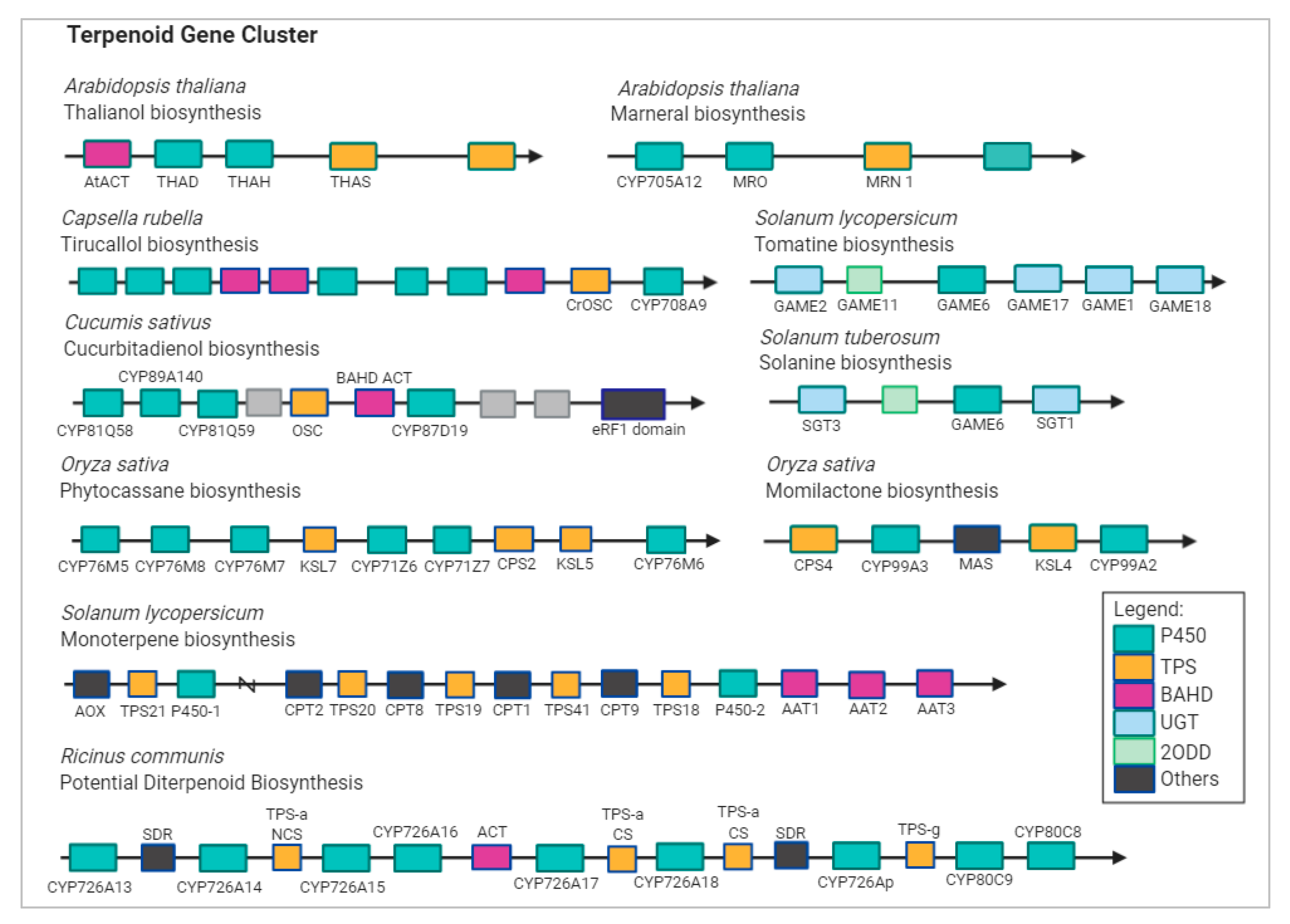
1.2. Stress Response and Terpenoid Biosynthetic Genes
1.3. Effects of Plant Terpenes on Abiotic Stress
2. Terpenoids in Plant–Insect Interactions
2.1. Terpenoids with Toxic and/or Repellent Effects on Insects
2.2. Terpenoids as Attractants to Predators or Parasitoids
2.3. Terpenoids as Attractants to Herbivores
2.4. Plant Terpenes with Beneficiaries
3. Potential Agricultural Biotechnology Applications
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007, 3, 408–414. [Google Scholar] [CrossRef]
- Nagel, R.; Schmidt, A.; Peters, R.J. Isoprenyl diphosphate synthases: The chain length determining step in terpene biosynthesis. Planta 2018, 249, 9–20. [Google Scholar] [CrossRef]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of Plant Volatiles1. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef]
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I.; Negre-Zakharov, F. Plant Volatiles: Recent Advances and Future Perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Yazaki, K.; Arimura, G.-I.; Ohnishi, T. ‘Hidden’ Terpenoids in Plants: Their Biosynthesis, Localization and Ecological Roles. Plant Cell Physiol. 2017, 58, 1615–1621. [Google Scholar] [CrossRef]
- Holopainen, J.K.; Himanen, S.J.; Yuan, J.S.; Chen, F.; Stewart, C.N. Ecological Functions of Terpenoids in Changing Climates. In Natural Products; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2013; pp. 2913–2940. [Google Scholar]
- Pazouki, L.; Niinemets, Ü. Multi-Substrate Terpene Synthases: Their Occurrence and Physiological Significance. Front. Plant Sci. 2016, 7, 1019. [Google Scholar] [CrossRef]
- Christianson, D.W. Structural and Chemical Biology of Terpenoid Cyclases. Chem. Rev. 2017, 117, 11570–11648. [Google Scholar] [CrossRef]
- Karunanithi, P.S.; Zerbe, P. Terpene Synthases as Metabolic Gatekeepers in the Evolution of Plant Terpenoid Chemical Diversity. Front. Plant Sci. 2019, 10, 1166. [Google Scholar] [CrossRef]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. BMC Plant Biol. 2011, 11, 163. [Google Scholar] [CrossRef]
- Zhu, J.-K. Abiotic Stress Signaling and Responses in Plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Hamann, T. Plant cell wall integrity maintenance as an essential component of biotic stress response mechanisms. Front. Plant Sci. 2012, 3, 77. [Google Scholar] [CrossRef]
- Tuteja, N.; Sopory, S.K. Plant signaling in stress. Plant Signal. Behav. 2008, 3, 79–86. [Google Scholar] [CrossRef]
- Muchlinski, A.; Chen, X.; Lovell, J.T.; Köllner, T.G.; Pelot, K.A.; Zerbe, P.; Ruggiero, M.; Callaway, L.I.; Laliberte, S.; Chen, F.; et al. Biosynthesis and Emission of Stress-Induced Volatile Terpenes in Roots and Leaves of Switchgrass (Panicum virgatum L.). Front. Plant Sci. 2019, 10, 1144. [Google Scholar] [CrossRef]
- Jia, Q.; Köllner, T.G.; Gershenzon, J.; Chen, F. MTPSLs: New Terpene Synthases in Nonseed Plants. Trends Plant Sci. 2018, 23, 121–128. [Google Scholar] [CrossRef]
- Alicandri, E.; Paolacci, A.R.; Osadolor, S.; Sorgonà, A.; Badiani, M.; Ciaffi, M. On the Evolution and Functional Diversity of Terpene Synthases in the Pinus Species: A Review. J. Mol. Evol. 2020, 88, 253–283. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.M.; Aubourg, S.P.; Schouwey, M.B.; Daviet, L.; Schalk, M.; Toub, O.; Lund, S.T.; Bohlmann, J. Functional Annotation, Genome Organization and Phylogeny of the Grapevine (Vitis vinifera) Terpene Synthase Gene Family Based on Genome Assembly, FLcDNA Cloning, and Enzyme Assays. BMC Plant Biol. 2010, 10, 226. [Google Scholar] [CrossRef]
- Falara, V.; Akhtar, T.A.; Nguyen, T.T.; Spyropoulou, E.A.; Bleeker, P.M.; Schauvinhold, I.; Matsuba, Y.; Bonini, M.E.; Schilmiller, A.L.; Last, R.L.; et al. The Tomato Terpene Synthase Gene Family. Plant Physiol. 2011, 157, 770–789. [Google Scholar] [CrossRef]
- Beran, F.; Köllner, T.G.; Gershenzon, J.; Tholl, D. Chemical convergence between plants and insects: Biosynthetic origins and functions of common secondary metabolites. New Phytol. 2019, 223, 52–67. [Google Scholar] [CrossRef]
- Jiang, S.-Y.; Jin, J.; Sarojam, R.; Ramachandran, S. A Comprehensive Survey on the Terpene Synthase Gene Family Provides New Insight into Its Evolutionary Patterns. Genome Biol. Evol. 2019, 11, 2078–2098. [Google Scholar] [CrossRef]
- Chen, F.; Tholl, D.; Bohlmann, J.; Pichersky, E. The family of terpene synthases in plants: A mid-size family of genes for specialized metabolism that is highly diversified throughout the kingdom. Plant J. 2011, 66, 212–229. [Google Scholar] [CrossRef]
- Chen, Y.-C.; Li, Z.; Zhao, Y.-X.; Gao, M.; Wang, J.-Y.; Liu, K.-W.; Wang, X.; Wu, L.-W.; Jiao, Y.-L.; Xu, Z.-L.; et al. The Litsea genome and the evolution of the laurel family. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Boutanaev, A.M.; Moses, T.; Zi, J.; Nelson, D.R.; Mugford, S.T.; Peters, R.J.; Osbourn, A. Investigation of terpene diversification across multiple sequenced plant genomes. Proc. Natl. Acad. Sci. USA 2014, 112, E81–E88. [Google Scholar] [CrossRef]
- Tohge, T.; Fernie, A.R. Co-regulation of Clustered and Neo-functionalized Genes in Plant-Specialized Metabolism. Plants 2020, 9, 622. [Google Scholar] [CrossRef]
- Seki, H.; Sawai, S.; Ohyama, K.; Mizutani, M.; Ohnishi, T.; Sudo, H.; Fukushima, E.O.; Akashi, T.; Aoki, T.; Saito, K.; et al. Triterpene Functional Genomics in Licorice for Identification of CYP72A154 Involved in the Biosynthesis of Glycyrrhizin. Plant Cell 2011, 23, 4112–4123. [Google Scholar] [CrossRef]
- Boachon, B.; Junker, R.R.; Miesch, L.; Bassard, J.-E.; Höfer, R.; Caillieaudeaux, R.; Seidel, D.; Lesot, A.; Heinrich, C.F.; Ginglinger, J.-F.; et al. CYP76C1 (Cytochrome P450)-Mediated Linalool Metabolism and the Formation of Volatile and Soluble Linalool Oxides in Arabidopsis Flowers: A Strategy for Defense against Floral Antagonists. Plant Cell 2015, 27, 2972–2990. [Google Scholar] [CrossRef] [PubMed]
- Gururani, M.A.; Venkatesh, J.; Tran, L.-S.P. Regulation of Photosynthesis during Abiotic Stress-Induced Photoinhibition. Mol. Plant 2015, 8, 1304–1320. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Quan, S.; Xiao, H. Towards efficient terpenoid biosynthesis: Manipulating IPP and DMAPP supply. Bioresour. Bioprocess. 2019, 6, 6. [Google Scholar] [CrossRef]
- Vickers, C.E.; Gershenzon, J.; Lerdau, M.; Loreto, F. A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat. Chem. Biol. 2009, 5, 283–291. [Google Scholar] [CrossRef]
- Finn, R.D.; Bateman, A.; Clements, J.; Coggill, P.; Eberhardt, R.Y.; Eddy, S.R.; Heger, A.; Hetherington, K.; Holm, L.; Mistry, J.; et al. Pfam: The protein families database. Nucleic Acids Res. 2013, 42, D222–D230. [Google Scholar] [CrossRef]
- Guindon, S.; Dufayard, J.-F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New Algorithms and Methods to Estimate Maximum-Likelihood Phylogenies: Assessing the Performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Lefort, V.; Longueville, J.-E.; Gascuel, O. SMS: Smart Model Selection in PhyML. Mol. Biol. Evol. 2017, 34, 2422–2424. [Google Scholar] [CrossRef]
- Priya, P.; Yadav, A.; Chand, J.; Yadav, G. Terzyme: A tool for identification and analysis of the plant terpenome. Plant Methods 2018, 14, 4. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, B.; Gao, S.; Lercher, M.J.; Hu, S.; Chen, W.-H. Evolview v3: A webserver for visualization, annotation, and management of phylogenetic trees. Nucleic Acids Res. 2019, 47, W270–W275. [Google Scholar] [CrossRef]
- Sewelam, N.; Kazan, K.; Schenk, P.M. Global Plant Stress Signaling: Reactive Oxygen Species at the Cross-Road. Front. Plant Sci. 2016, 7, 187. [Google Scholar] [CrossRef]
- Blande, J.D.; Holopainen, J.K.; Niinemets, Ü. Plant volatiles in polluted atmospheres: Stress responses and signal degradation. Plant Cell Environ. 2014, 37, 1892–1904. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Yeh, S. Isoprene emission from plants. Annu. Rev. Plant Biol. 2001, 52, 407–436. [Google Scholar] [CrossRef]
- Goh, C.-H.; Ko, S.-M.; Koh, S.; Kim, Y.-J.; Bae, H.-J. Photosynthesis and Environments: Photoinhibition and Repair Mechanisms in Plants. J. Plant Biol. 2011, 55, 93–101. [Google Scholar] [CrossRef]
- Havaux, M.; Eymery, F.; Porfirova, S.; Rey, P.; Dörmann, P. Vitamin E Protects against Photoinhibition and Photooxidative Stress in Arabidopsis thaliana. Plant Cell 2005, 17, 3451–3469. [Google Scholar] [CrossRef]
- Havaux, M.; Niyogi, K.K. The violaxanthin cycle protects plants from photooxidative damage by more than one mechanism. Proc. Natl. Acad. Sci. USA 1999, 96, 8762–8767. [Google Scholar] [CrossRef]
- Dall’Osto, L.; Fiore, A.; Cazzaniga, S.; Giuliano, G.; Bassi, R. Different Roles of - and -Branch Xanthophylls in Photosystem Assembly and Photoprotection. J. Biol. Chem. 2007, 282, 35056–35068. [Google Scholar] [CrossRef]
- Peng, L.; Ma, J.; Chi, W.; Guo, J.; Zhu, S.; Lu, Q.; Lu, C.; Zhang, L.-X. Low Psii Accumulation1 Is Involved in Efficient Assembly of Photosystem II in Arabidopsis thaliana. Plant Cell 2006, 18, 955–969. [Google Scholar] [CrossRef] [PubMed]
- Korankye, E.A.; Lada, R.R.; Asiedu, S.; Caldwell, C. Plant Senescence: The Role of Volatile Terpene Compounds (VTCs). Am. J. Plant Sci. 2017, 8, 3120–3139. [Google Scholar] [CrossRef]
- Pegadaraju, V.; Knepper, C.; Reese, J.; Shah, J. Premature Leaf Senescence Modulated by the Arabidopsis PHYTOALEXIN DEFICIENT4 Gene Is Associated with Defense against the Phloem-Feeding Green Peach Aphid. Plant Physiol. 2005, 139, 1927–1934. [Google Scholar] [CrossRef] [PubMed]
- Sade, N.; Rubio-Wilhelmi, M.D.M.; Umnajkitikorn, K.; Blumwald, E. Stress-induced senescence and plant tolerance to abiotic stress. J. Exp. Bot. 2017, 69, 845–853. [Google Scholar] [CrossRef]
- Romagni, J.G.; Allen, S.N.; Dayan, E.F. Allelopathic Effects of Volatile Cineoles on Two Weedy Plant Species. J. Chem. Ecol. 2000, 26, 303–313. [Google Scholar] [CrossRef]
- Chaimovitsh, D.; Abu-Abied, M.; Belausov, E.; Rubin, B.; Dudai, N.; Sadot, E. Microtubules are an intracellular target of the plant terpene citral. Plant J. 2010, 61, 399–408. [Google Scholar] [CrossRef]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B.; Rowshan, V.; Niazi, A.; Moghadam, A. Effect of salt stress on terpenoid biosynthesis in Salvia mirzayanii: From gene to metabolite. J. Hortic. Sci. Biotechnol. 2018, 94, 389–399. [Google Scholar] [CrossRef]
- Balazadeh, S.; Schildhauer, J.; Araújo, W.L.; Munné-Bosch, S.; Fernie, A.R.; Proost, S.; Humbeck, K.; Mueller-Roeber, B. Reversal of senescence by N resupply to N-starved Arabidopsis thaliana: Transcriptomic and metabolomic consequences. J. Exp. Bot. 2014, 65, 3975–3992. [Google Scholar] [CrossRef]
- Palmer-Young, E.C.; Veit, D.; Gershenzon, J.; Schuman, M.C. The Sesquiterpenes(E)-ß-Farnesene and (E)-α-Bergamotene Quench Ozone but Fail to Protect the Wild Tobacco Nicotiana attenuata from Ozone, UVB, and Drought Stresses. PLoS ONE 2015, 10, e0127296. [Google Scholar] [CrossRef]
- Loreto, F. Ozone Quenching Properties of Isoprene and Its Antioxidant Role in Leaves. Plant Physiol. 2001, 126, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Atkinson, R. Rate constants for the gas-phase reactions of O3 with a series of Terpenes and OH radical formation from the O3 reactions with Sesquiterpenes at 296 ± 2 K. Int. J. Chem. Kinet. 1994, 26, 1193–1205. [Google Scholar] [CrossRef]
- Lee, G.W.; Lee, S.; Chung, M.-S.; Jeong, Y.S.; Chung, B.Y. Rice terpene synthase 20 (OsTPS20) plays an important role in producing terpene volatiles in response to abiotic stresses. Protoplasma 2014, 252, 997–1007. [Google Scholar] [CrossRef]
- Bertamini, M.; Grando, M.S.; Zocca, P.; Pedrotti, M.; Lorenzi, S.; Cappellin, L. Linking monoterpenes and abiotic stress resistance in grapevines. BIO Web Conf. 2019, 13, 01003. [Google Scholar] [CrossRef]
- Kleiber, A.; Duan, Q.; Jansen, K.; Junker, L.V.; Kammerer, B.; Rennenberg, H.; Ensminger, I.; Gessler, A.; Kreuzwieser, J. Drought effects on root and needle terpenoid content of a coastal and an interior Douglas fir provenance. Tree Physiol. 2017, 37, 1648–1658. [Google Scholar] [CrossRef]
- Vaughan, M.M.; Christensen, S.; Schmelz, E.A.; Huffaker, A.; McAuslane, H.J.; Alborn, H.T.; Romero, M.; Allen, L.H.; Teal, P.E.A. Accumulation of terpenoid phytoalexins in maize roots is associated with drought tolerance. Plant Cell Environ. 2015, 38, 2195–2207. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Alegre, L. Drought-Induced Changes in the Redox State of α-Tocopherol, Ascorbate, and the Diterpene Carnosic Acid in Chloroplasts of Labiatae Species Differing in Carnosic Acid Contents1. Plant Physiol. 2003, 131, 1816–1825. [Google Scholar] [CrossRef]
- Plata-Rueda, A.; Campos, J.M.; Rolim, G.D.S.; Martínez, L.C.; Dos Santos, M.H.; Fernandes, F.L.; Serrão, J.E.; Zanuncio, J.C. Terpenoid constituents of cinnamon and clove essential oils cause toxic effects and behavior repellency response on granary weevil, Sitophilus granarius. Ecotoxicol. Environ. Saf. 2018, 156, 263–270. [Google Scholar] [CrossRef]
- Bustos-Segura, C.; Kulheim, C.; Foley, W.J. Effects of Terpene Chemotypes of Melaleuca alternifolia on Two Specialist Leaf Beetles and Susceptibility to Myrtle Rust. J. Chem. Ecol. 2015, 41, 937–947. [Google Scholar] [CrossRef]
- Ogah, E.O.; Smart, L.E.; Woodcock, C.M.; Caulfield, J.C.; Birkett, M.A.; Pickett, J.A.; Nwilene, F.E.; Bruce, T.J.A. Electrophysiological and behavioral responses of female African rice gall midge, Orseolia oryzivora Harris and Gagné, to host plant volatiles. J. Chem. Ecol. 2016, 43, 13–16. [Google Scholar] [CrossRef] [PubMed]
- Cascone, P.; Iodice, L.; Maffei, M.E.; Bossi, S.; Arimura, G.-I.; Guerrieri, E. Tobacco overexpressing β-ocimene induces direct and indirect responses against aphids in receiver tomato plants. J. Plant Physiol. 2015, 173, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Dodson, C.H.; Dressler, R.L.; Hills, H.G.; Adams, R.M.; Williams, N.H. Biologically Active Compounds in Orchid Fragrances. Science 1969, 164, 1243–1249. [Google Scholar] [CrossRef]
- Armbruster, W.S. Reproductive Interactions Between Sympatric Dalechampia Species: Are Natural Assemblages “Random” or Organized? Ecology 1986, 67, 522–533. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Herzig, A.L. Partitioning and Sharing of Pollinators by Four Sympatric Species of Dalechampia (Euphorbiaceae) in Panama. Ann. Mo. Bot. Gard. 1984, 71, 1. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Steiner, K.E. Pollination Ecology of Four Dalechampia Species (Euphorbiaceae) in Northern Natal, South Africa. Am. J. Bot. 1992, 79, 306. [Google Scholar] [CrossRef]
- Nieuwenhuizen, N.J.; Wang, M.Y.; Matich, A.J.; Green, S.A.; Chen, X.; Yauk, Y.-K.; Beuning, L.L.; Nagegowda, D.A.; Dudareva, N.; Atkinson, R.G. Two terpene synthases are responsible for the major sesquiterpenes emitted from the flowers of kiwifruit (Actinidia deliciosa). J. Exp. Bot. 2009, 60, 3203–3219. [Google Scholar] [CrossRef] [PubMed]
- Dötterl, S.; Jurgens, A.; Seifert, K.; Laube, T.; Weissbecker, B.; Schutz, S. Nursery pollination by a moth in Silene latifolia: The role of odours in eliciting antennal and behavioural responses. New Phytol. 2006, 169, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Terry, I.; Walter, G.H.; Moore, C.; Roemer, R.; Hull, C. Odor-Mediated Push-Pull Pollination in Cycads. Science 2007, 318, 70. [Google Scholar] [CrossRef]
- Chen, C.; Song, Q. Responses of the Pollinating Wasp Ceratosolen solmsi marchali to Odor Variation Between Two Floral Stages of Ficus hispida. J. Chem. Ecol. 2008, 34, 1536–1544. [Google Scholar] [CrossRef]
- Byers, K.J.R.P.; Bradshaw, H.D.; Riffell, J.A. Three floral volatiles contribute to differential pollinator attraction in monkeyflowers (Mimulus). J. Exp. Biol. 2013, 217, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Majetic, C.J.; Raguso, R.A.; Ashman, T.-L. The sweet smell of success: Floral scent affects pollinator attraction and seed fitness inHesperis matronalis. Funct. Ecol. 2009, 23, 480–487. [Google Scholar] [CrossRef]
- Dobson, H. Relationship between Floral Fragrance Composition and Type of Pollinator. In Biology of Floral Scent; Informa UK Limited: Colchester, UK, 2006; pp. 147–198. [Google Scholar]
- Nagnan, P.; Clement, J.-L.; Lange, C.; Peru, L.; Basselier, J.-J. Geranyllinalool (Diterpene Alcohol). J. Chem. Ecol. 1990, 16, 2067–2079. [Google Scholar] [CrossRef]
- Youngsteadt, E.; Baca, J.A.; Osborne, J.; Schal, C. Species-Specific Seed Dispersal in an Obligate Ant-Plant Mutualism. PLoS ONE 2009, 4, e4335. [Google Scholar] [CrossRef]
- Ahuja, I.; Rohloff, J.; Bones, A.M. Defence mechanisms of Brassicaceae: Implications for plant-insect interactions and potential for integrated pest management. A review. Agron. Sustain. Dev. 2010, 30, 311–348. [Google Scholar] [CrossRef]
- Dicke, M.; Van Loon, J.J.A.; De Jong, P.W. Ecogenomics Benefits Community Ecology. Science 2004, 305, 618–619. [Google Scholar] [CrossRef]
- Turlings, T.C.J.; Benrey, B. Effect of plant metabolites on the behavior and development of parasitic wasp. Ecoscience 1998, 5, 321–333. [Google Scholar]
- Wegener, R.; Schulz, S.; Meiners, T.; Hadwich, K.; Hilker, M. Analysis of volatiles induced by oviposition of elm leaf beetle Xanthogaleruca luteola on Ulmus minor. J. Chem. Ecol. 2001, 27, 499–515. [Google Scholar] [CrossRef] [PubMed]
- Wegener, R.; Schulz, S. Identification and synthesis of homoterpenoids emitted from elm leaves after elicitation by beetle eggs. Tetrahedron 2002, 58, 315–319. [Google Scholar] [CrossRef]
- Xu, X.; Cai, X.; Bian, L.; Luo, Z.; Xin, Z.; Chen, Z. Electrophysiological and Behavioral Responses of Chrysopa phyllochroma (Neuroptera: Chrysopidae) to Plant Volatiles. Environ. Èntomol. 2015, 44, 1425–1433. [Google Scholar] [CrossRef]
- Berkov, A.; Purzycki, K.L.; Meurer-Grimes, B. Do Lecythidaceae Specialists (Coleoptera, Cerambycidae) Shun Fetid Tree Species? Biotropica 2000, 32, 440–451. [Google Scholar] [CrossRef]
- Li, H.; Li, J.; Dong, Y.; Hao, H.; Ling, Z.; Bai, H.; Wang, H.; Cui, H.; Shi, L. Time-series transcriptome provides insights into the gene regulation network involved in the volatile terpenoid metabolism during the flower development of lavender. BMC Plant Biol. 2019, 19, 313. [Google Scholar] [CrossRef]
- Da Costa, C.P.; Jones, C.M.; Breedlove, J.R.; Trammell, G.T. Cucumber Beetle Resistance and Mite Susceptibility Controlled by the Bitter Gene in Cucumis sativus L. Science 1971, 172, 1145–1146. [Google Scholar] [CrossRef]
- Balkema-Boomstra, A.G.; Zijlstra, S.; Verstappen, F.W.A.; Inggamer, H.; Mercke, P.E.; Jongsma, M.A.; Bouwmeester, H. Role of Cucurbitacin C in Resistance to Spider Mite (Tetranychus urticae) in Cucumber (Cucumis sativus L.). J. Chem. Ecol. 2003, 29, 225–235. [Google Scholar] [CrossRef]
- Mitra, S.; Karmakar, A.; Mukherjee, A.; Barik, A. The Role of Leaf Volatiles of Ludwigia octovalvis (Jacq.) Raven in the Attraction of Altica cyanea (Weber) (Coleoptera: Chrysomelidae). J. Chem. Ecol. 2017, 43, 679–692. [Google Scholar] [CrossRef]
- Naidoo, S.; Christie, N.; Acosta, J.J.; Mphahlele, M.M.; Payn, K.G.; Myburg, A.A.; Kulheim, C. Terpenes associated with resistance against the gall wasp, Leptocybe invasa, in Eucalyptus grandis. Plant Cell Environ. 2018, 41, 1840–1851. [Google Scholar] [CrossRef]
- Oliveira, J.L.; Campos, E.V.R.; Pereira, A.D.E.S.; Nunes, L.E.S.; Da Silva, C.C.L.; Pasquoto, T.; De Lima, R.; Smaniotto, G.; Polanczyk, R.A.; Fraceto, L.F. Geraniol Encapsulated in Chitosan/Gum Arabic Nanoparticles: A Promising System for Pest Management in Sustainable Agriculture. J. Agric. Food Chem. 2018, 66, 5325–5334. [Google Scholar] [CrossRef]
- Aprotosoaie, A.C.; Luca, V.S.; Trifan, A.; Miron, A. Antigenotoxic Potential of Some Dietary Non-phenolic Phytochemicals. In Studies in Natural Products Chemistry; Elsevier BV: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Zimmermann, Y.; Roubik, D.W.; Eltz, T. Species-specific attraction to pheromonal analogues in orchid bees. Behav. Ecol. Sociobiol. 2006, 60, 833–843. [Google Scholar] [CrossRef]
- Armbruster, W.S. Evolution of Plant Pollination Systems: Hypotheses and Tests with the Neotropical Vinedalechampia. Evolytion 1993, 47, 1480–1505. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Howard, J.J.; Clausen, T.P.; Debevec, E.M.; Loquvam, J.C.; Matsuki, M.; Cerendolo, B.; Andel, F. Do Biochemical Exaptations Link Evolution of Plant Defense and Pollination Systems? Historical Hypotheses and Experimental Tests with Dalechampia Vines. Am. Nat. 1997, 149, 461–484. [Google Scholar] [CrossRef]
- Cheong, S.P.S.; Huang, J.; Bendena, W.G.; Tobe, S.S.; Hui, J.H. Evolution of Ecdysis and Metamorphosis in Arthropods: The Rise of Regulation of Juvenile Hormone. Integr. Comp. Biol. 2015, 55, 878–890. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Bendena, W.G.; Tobe, S.S.; Hui, J.H. Juvenile hormone and sesquiterpenoids in arthropods: Biosynthesis, signaling, and role of MicroRNA. J. Steroid Biochem. Mol. Biol. 2018, 184, 69–76. [Google Scholar] [CrossRef]
- Yu, F.; Utsumi, R. Diversity, regulation, and genetic manipulation of plant mono- and sesquiterpenoid biosynthesis. Cell. Mol. Life Sci. 2009, 66, 3043–3052. [Google Scholar] [CrossRef]
- Degenhardt, J.; Köllner, T.G.; Gershenzon, J. Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 2009, 70, 1621–1637. [Google Scholar] [CrossRef] [PubMed]
- Tsang, S.S.K.; Law, S.T.S.; Li, C.; Qu, Z.; Bendena, W.G.; Tobe, S.S.; Hui, J.H.L. Diversity of Insect Sesquiterpenoid Regulation. Front. Genet. 2020, 11, 1–13. [Google Scholar] [CrossRef]
- Marchal, E.; Zhang, J.; Badisco, L.; Everlinden, H.; Hult, E.F.; Van Wielendaele, P.; Yagi, K.J.; Tobe, S.S.; Broeck, J.V. Final steps in juvenile hormone biosynthesis in the desert locust, Schistocerca gregaria. Insect Biochem. Mol. Biol. 2011, 41, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Daimon, T.; Kozaki, T.; Niwa, R.; Kobayashi, I.; Furuta, K.; Namiki, T.; Uchino, K.; Banno, Y.; Katsuma, S.; Tamura, T.; et al. Precocious Metamorphosis in the Juvenile Hormone–Deficient Mutant of the Silkworm, Bombyx mori. PLoS Genet. 2012, 8, e1002486. [Google Scholar] [CrossRef]
- Le Trionnaire, G.; Jaubert-Possamai, S.; Bonhomme, J.; Gauthier, J.-P.; Guernec, G.; Le Cam, A.; Legeai, F.; Monfort, J.; Tagu, D. Transcriptomic profiling of the reproductive mode switch in the pea aphid in response to natural autumnal photoperiod. J. Insect Physiol. 2012, 58, 1517–1524. [Google Scholar] [CrossRef]
- Lücker, J.; Blaas, J.; Bouwmeester, H.; Schwab, W.; Van Der Plas, L.H.W.; Verhoeven, H.A. Expression of Clarkia S-linalool synthase in transgenic petunia plants results in the accumulation of S-linalyl-?-d-glucopyranoside. Plant J. 2001, 27, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Jongsma, M.A. Novel Genes for Control and Deterrence of Sucking Insect Pests. ISB News Rep. 2004, 1–4. Available online: www.isb.vt.edu/nov0401.htm (accessed on 17 September 2020).
- Aharoni, A.; Jongsma, M.A.; Kim, T.-Y.; Ri, M.-B.; Giri, A.P.; Verstappen, F.W.A.; Schwab, W.; Bouwmeester, H. Metabolic Engineering of Terpenoid Biosynthesis in Plants. Phytochem. Rev. 2006, 5, 49–58. [Google Scholar] [CrossRef]
- Aharoni, A.; Giri, A.P.; Deuerlein, S.; Griepink, F.; De Kogel, W.-J.; Verstappen, F.W.A.; Verhoeven, H.A.; Jongsma, M.A.; Schwab, W.; Bouwmeester, H. Terpenoid Metabolism in Wild-Type and Transgenic Arabidopsis Plants. Plant Cell 2003, 15, 2866–2884. [Google Scholar] [CrossRef]
- Bouwmeester, H.J.; Verstappen, F.W.; Posthumus, M.A.; Dicke, M. Spider Mite-Induced (3S)-(E)-Nerolidol Synthase Activity in Cucumber and Lima Bean. The First Dedicated Step in Acyclic C11-Homoterpene Biosynthesis. Plant Physiol. 1999, 121, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Schnee, C.; Köllner, T.G.; Held, M.; Turlings, T.C.J.; Gershenzon, J.; Degenhardt, J. The products of a single maize sesquiterpene synthase form a volatile defense signal that attracts natural enemies of maize herbivores. Proc. Natl. Acad. Sci. USA 2006, 103, 1129–1134. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, S.; De Cristofaro, A.; Guerrieri, E.; Bossi, S.; Cellini, F.; Di Leo, G.M.; Germinara, G.S.; Iodice, L.; Maffei, M.E.; Petrozza, A.; et al. Foraging activity of bumblebees (Bombus terrestris L.) on Bt-expressing eggplants. Arthropod Plant Interact. 2011, 5, 255–261. [Google Scholar] [CrossRef]
- Lewinsohn, E.; Schalechet, F.; Wilkinson, J.; Matsui, K.; Tadmor, Y.; Nam, K.-H.; Amar, O.; Lastochkin, E.; Larkov, O.; Ravid, U.; et al. Enhanced Levels of the Aroma and Flavor Compound S-Linalool by Metabolic Engineering of the Terpenoid Pathway in Tomato Fruits. Plant Physiol. 2001, 127, 1256–1265. [Google Scholar] [CrossRef]
- Lavy, M.; Zuker, A.; Lewinsohn, E.; Larkov, O.; Ravid, U.; Vainstein, A.; Weiss, D. Linalool and linalool oxide production in transgenic carnation flowers expressing the Clarkia breweri linalool synthase gene. Mol. Breed. 2002, 9, 103–111. [Google Scholar] [CrossRef]
- Lücker, J.; Schwab, W.; Franssen, M.C.R.; Van Der Plas, L.H.W.; Bouwmeester, H.; Verhoeven, H.A. Metabolic engineering of monoterpene biosynthesis: Two-step production of (+)-trans-isopiperitenol by tobacco. Plant J. 2004, 39, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Diemer, F.; Caissard, J.-C.; Moja, S.; Chalchat, J.-C.; Jullien, F. Altered monoterpene composition in transgenic mint following the introduction of 4S-limonene synthase. Plant Physiol. Biochem. 2001, 39, 603–614. [Google Scholar] [CrossRef]
- Weathers, P.J.; Arsenault, P.R.; Covello, P.S.; McMickle, A.; Teoh, K.H.; Reed, D. Artemisinin production in Artemisia annua: Studies in planta and results of a novel delivery method for treating malaria and other neglected diseases. Phytochem. Rev. 2010, 10, 173–183. [Google Scholar] [CrossRef]
- Yu, Z.-X.; Li, J.-X.; Yang, C.-Q.; Hu, W.-L.; Wang, L.-J.; Chen, X.-Y. The Jasmonate-Responsive AP2/ERF Transcription Factors AaERF1 and AaERF2 Positively Regulate Artemisinin Biosynthesis in Artemisia annua L. Mol. Plant 2012, 5, 353–365. [Google Scholar] [CrossRef]
- Abbas, F.; Ke, Y.; Yu, R.; Yue, Y.; Amanullah, S.; Jahangir, M.M.; Fan, Y. Volatile terpenoids: Multiple functions, biosynthesis, modulation and manipulation by genetic engineering. Planta 2017, 246, 803–816. [Google Scholar] [CrossRef]
- Aharoni, A.; Jongsma, M.A.; Bouwmeester, H. Volatile science? Metabolic engineering of terpenoids in plants. Trends Plant Sci. 2005, 10, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Wani, S.H.; Kumar, V.; Shriram, V.; Sah, S.K. Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop. J. 2016, 4, 162–176. [Google Scholar] [CrossRef]
| Plant Species | Terpenes | Abiotic Stress | References |
|---|---|---|---|
| Nicotianaattenuata | sesquiterpenes: (E)-β-Farnesene; (E)-α-Bergamotene | Tropospheric ozone, ROS | [50,51,52] |
| Oryzasativa | monoterpenes: limonene; sabinene, myrcene; α-terpinene; β-ocimene; γ-terpinene; α-terpinolene | High irradiance (UV-B, H2O2, and γ rays) | [53] |
| Vitis viniferacv. Chardonay | monoterpenes | Thermal stress | [54] |
| Pseudotsugamenziesii (Douglas fir) | monoterpenes: β-pinene; α-pinene; β-citronellol; 3-carene; camphene | Drought- and salt-induced stress | [55] |
| Zea mays | Sesquiterpenoid: zealexin; diterpenoid: kaulalexins together with ABA | [56] | |
| Salviaofficinalis, Salvia fruticose, Rosmarinus officinalis | diterpene: carnosic acid | [57] |
| Plant Species | Terpenes | Targets | Effects | References |
|---|---|---|---|---|
| Tropical orchids | monoterpene: 1, 8-cineole | Male euglossine bees | an attractant and reward to pollinator | [62] |
| Dalechampia (Euphorbiaceae) epiphytes (Clusia) | terpenoid resins (oxygenated triterpenes) | Female euglossine (Apidae), female anthidiine (Megachilidae), or worker meliponine (Apidae) bees | reward to pollinators for use in nest construction | [63,64,65] |
| Kiwifruit (Actinidia deliciosa) | Sesquiterpene: α-farnesene, germacrene D, Monoterpenes: (E)-β-ocimene, (Z,E)-α-farnesene | Mainly honeybees (Apidae) | attract a variety of pollinators | [66] |
| White Campion (Silene latifolia) | lilac aldehydes | Noctuid moths (Hadena bicruris) | scent cue for pollinators to locate their specific host | [67] |
| Pineapple zamia (Macrozamia lucida) | β-myrcene, (E)-β ocimene and allo-ocimene | Thrips (Cycadothrips chadwick) | repel or attract pollinators to complete the pollination from male to female | [68] |
| Fig (Ficus hispida) | Monoterpenes: linalool, limonene and β-pinene | Wasp (Ceratosolen solmsi marchali) | signals for pollinators to identify floral stages | [69] |
| Monkeyflower (Mimulus lewisii) | D-limonene, β-myrcene and E-β-ocimene. | Bumblebee (Bombus vosnesenskii) | attract specific pollinators | [70] |
| Sweet rocket (Hesperis Matronalis) | linalool, β-ocimene. | Mainly syrphid flies (Syrphidae) | attract a variety of pollinators | [71,72] |
| Radiator plant (Peperomia macrostachya) | geranyl linalool | Arboreal ants (Camponotus femoratus) | attract seed disperser to collect and plant their seeds | [73,74] |
| Cabbage plants (Brassica) | monoterpene: 1,8-cineole | Parasitic wasps (Cotesia glomerata) | attracts parasitoids that lay eggs in the caterpillars of specific herbivores | [75,76] |
| Maize (Zea mays) | terpene | Parasitic wasps (Cotesia marginventris) specialized parasitoid (Microplitis croceipes). | attracts the endoparasitoid that attacks larvae of a wide range of lepidopterous hosts | [77] |
| Elm (Ulmus minor) | homoterpenoids: (E)-2,6-dimethyl-6,8-nonadien-4-one, (E)-2,6-dimethyl-2,6,8-nonatrien-4-one, and (R,E)-2,3-epoxy-2,6-dimethyl-6,8-nonadiene | Parasitoid (Oomyzus gallerucae) | attracts egg parasitoids to attack the eggs of elm leaf beetles Xanthogaleruca luteola | [78,79] |
| Various plant species | (Z)-3-hexenyl acetate, (Z)-3-hexenol, (3E)-4,8-dimethyl-1,3,7-nonatriene, and linalool | Pest predator (Chrysopa phyllochroma) | specific concentrations of these terpenes are significantly attractive to this target | [80] |
| Various plant species | (Z)-3-hexenyl acetate, (3E)-4,8-dimethyl-1,3,7-nonatriene, and linalool | Pest predator (Chrysopa phyllochroma) | promote oviposition | [80] |
| Tomato and tobacco | β-ocimene | Parasitoid (Aphidius ervi) | attract parasitoids | [61] |
| Tomato and tobacco | β-ocimene | Pest (Macrosiphum euphorbiae) | defense against pest | [61] |
| Melalecua alternifolia | Terpinolene | Paropsisterna tigrina | pest adults cause less damage in the presence of a high level of terpinolene in the plant | [59] |
| Brazil nut family (Lecythidaceae) | S-methylmethionine, | Wood-boring longicorn beetles (Cerambycidae) | deterrent to specialist beetle seeking oviposition sites | [81] |
| Lavender (Lavandula angustifolia) | β-trans-ocimene, (+)-R-limonene | Aphids | deterrent to pest | [82] |
| Cucumber (Cucumis sativus) | Tetracyclic terpenes: Cucurbitacins | Spider mite (Tetranychus urticae) | antibiotic effect on spider mites but attractive to the pest cucumber beetle | [83,84] |
| Cinnamon and clove | Eugenol, caryophyllene oxide, α-pinene, α-humulene and α-phellandrene | Sitophilus granarius | toxic and repellent effects to adult pest | [58] |
| Water primrose (Ludwigia octovalvis) | α-pinene, linalool oxide, geraniol, and phytol | Weber (Altica cyanea) | attractive to pest females | [85] |
| Rice (Oryza sativa). | (S)-linalool, 4,8-dimethyl-1,3,7-nonatriene, (E)-caryophyllene, and (R/S)-(E)-nerolidol | African rice gall midge (Orseolia oryzivora) | attractive to mated female pest in intact rice, but repellent with different concentrations of the same volatiles in infested plant | [60] |
| Eucalyptus grandis | α-pinene, γ-terpinene | Leptocybe invasa | potentially attractive to pest | [86] |
| Various plant species | Geraniol | Bemisia tabaci | encapsulated geraniol shows attraction to B. tabaci | [87] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boncan, D.A.T.; Tsang, S.S.K.; Li, C.; Lee, I.H.T.; Lam, H.-M.; Chan, T.-F.; Hui, J.H.L. Terpenes and Terpenoids in Plants: Interactions with Environment and Insects. Int. J. Mol. Sci. 2020, 21, 7382. https://doi.org/10.3390/ijms21197382
Boncan DAT, Tsang SSK, Li C, Lee IHT, Lam H-M, Chan T-F, Hui JHL. Terpenes and Terpenoids in Plants: Interactions with Environment and Insects. International Journal of Molecular Sciences. 2020; 21(19):7382. https://doi.org/10.3390/ijms21197382
Chicago/Turabian StyleBoncan, Delbert Almerick T., Stacey S.K. Tsang, Chade Li, Ivy H.T. Lee, Hon-Ming Lam, Ting-Fung Chan, and Jerome H.L. Hui. 2020. "Terpenes and Terpenoids in Plants: Interactions with Environment and Insects" International Journal of Molecular Sciences 21, no. 19: 7382. https://doi.org/10.3390/ijms21197382
APA StyleBoncan, D. A. T., Tsang, S. S. K., Li, C., Lee, I. H. T., Lam, H.-M., Chan, T.-F., & Hui, J. H. L. (2020). Terpenes and Terpenoids in Plants: Interactions with Environment and Insects. International Journal of Molecular Sciences, 21(19), 7382. https://doi.org/10.3390/ijms21197382






