Magnetite Nanoparticles and Essential Oils Systems for Advanced Antibacterial Therapies
Abstract
1. Introduction
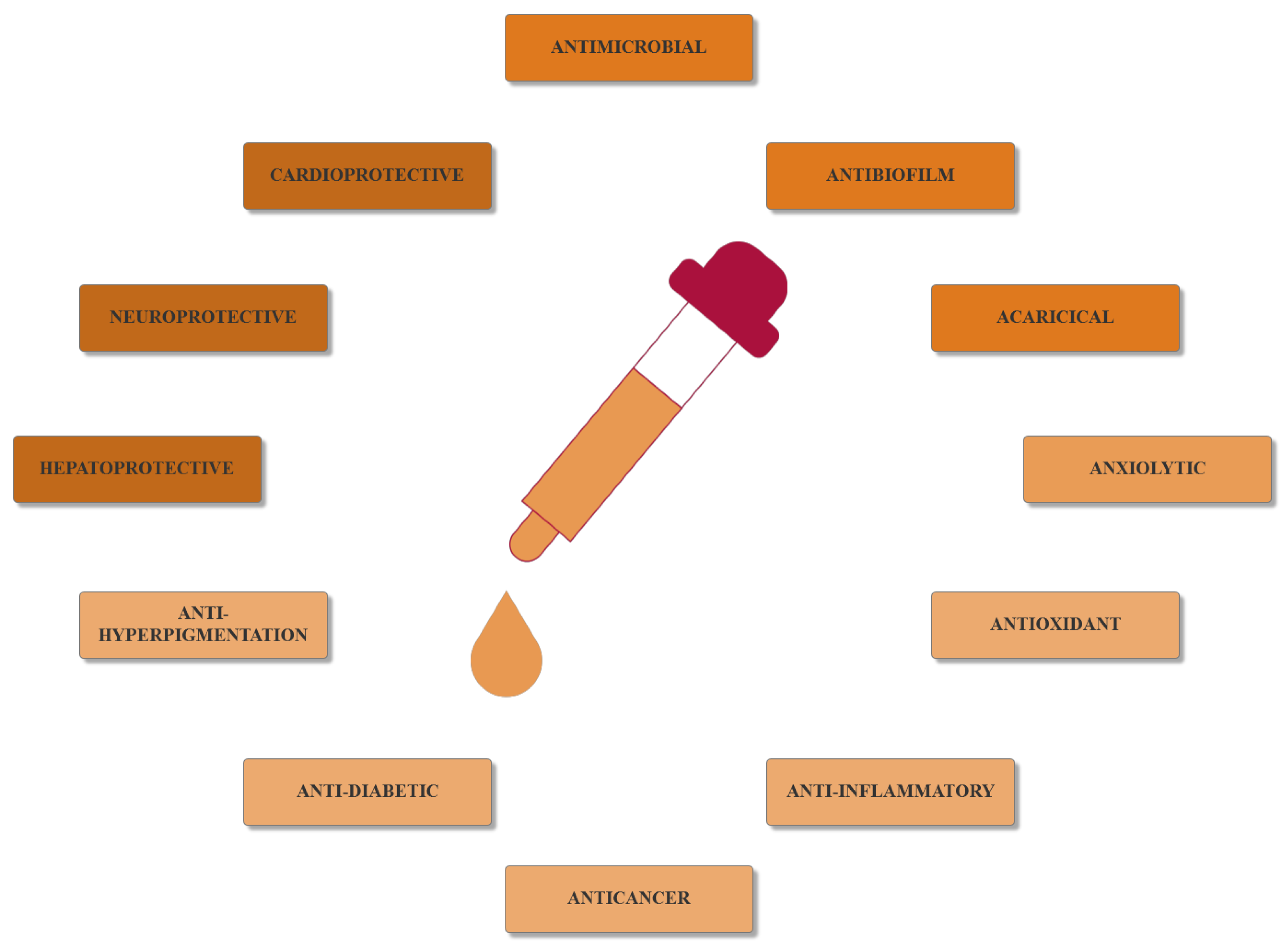
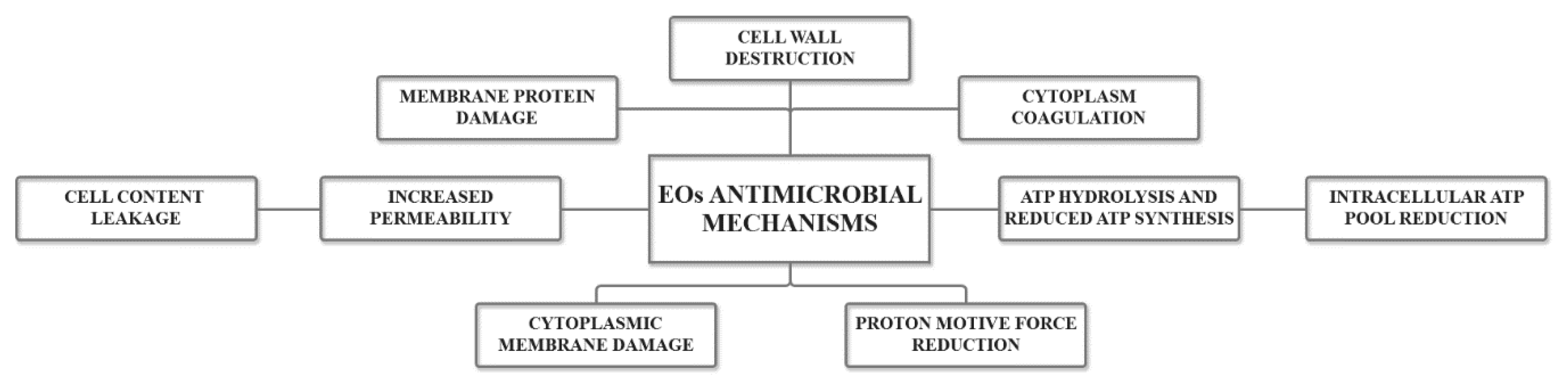
2. Essential Oils as Antimicrobial Agents
3. Magnetite Nanoparticles—Synthesis, Properties, and Functionalization
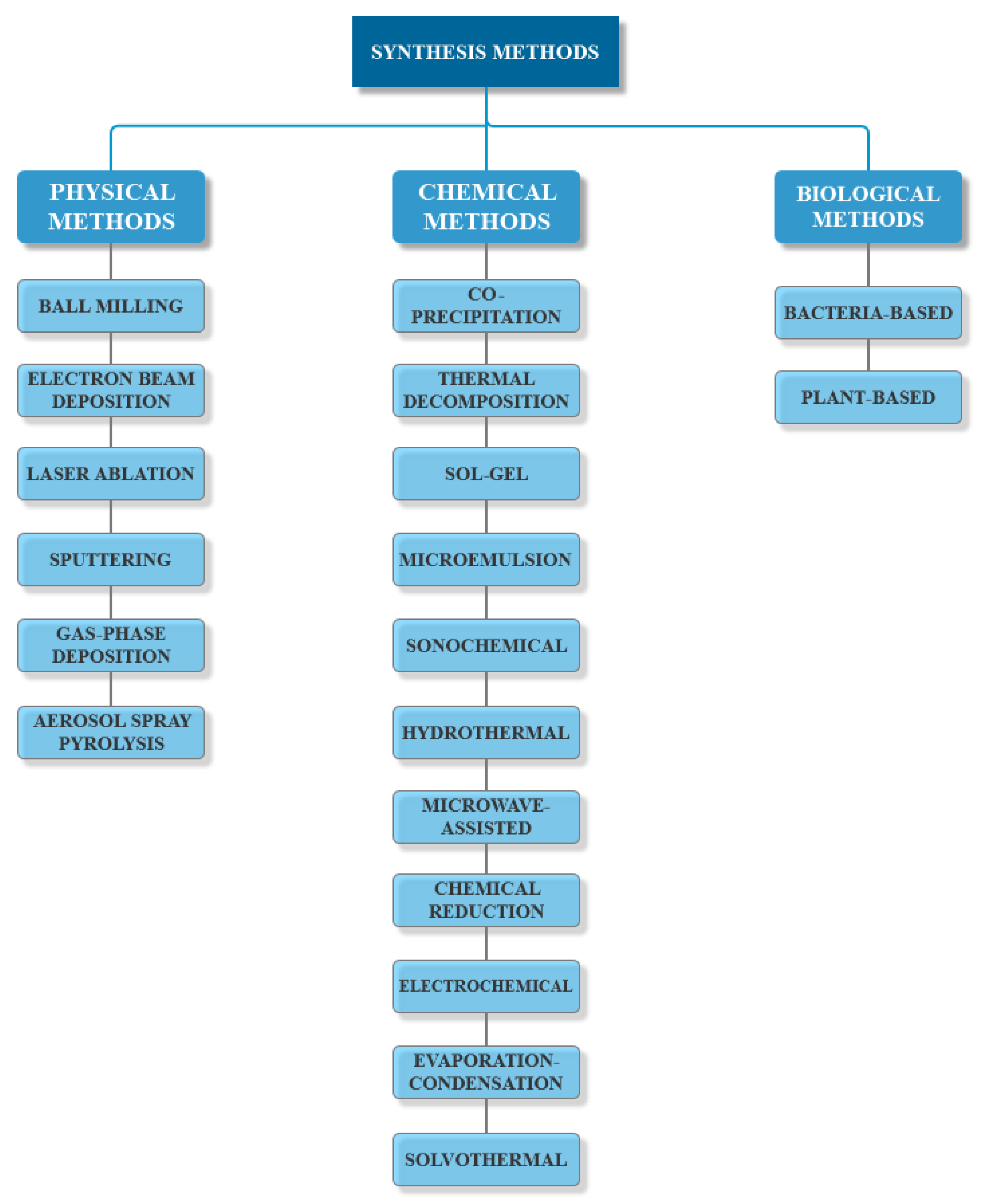
3.1. Synthesis Methods
3.1.1. Physical Methods
3.1.2. Chemical Methods
3.1.3. Biological Methods
3.2. Properties
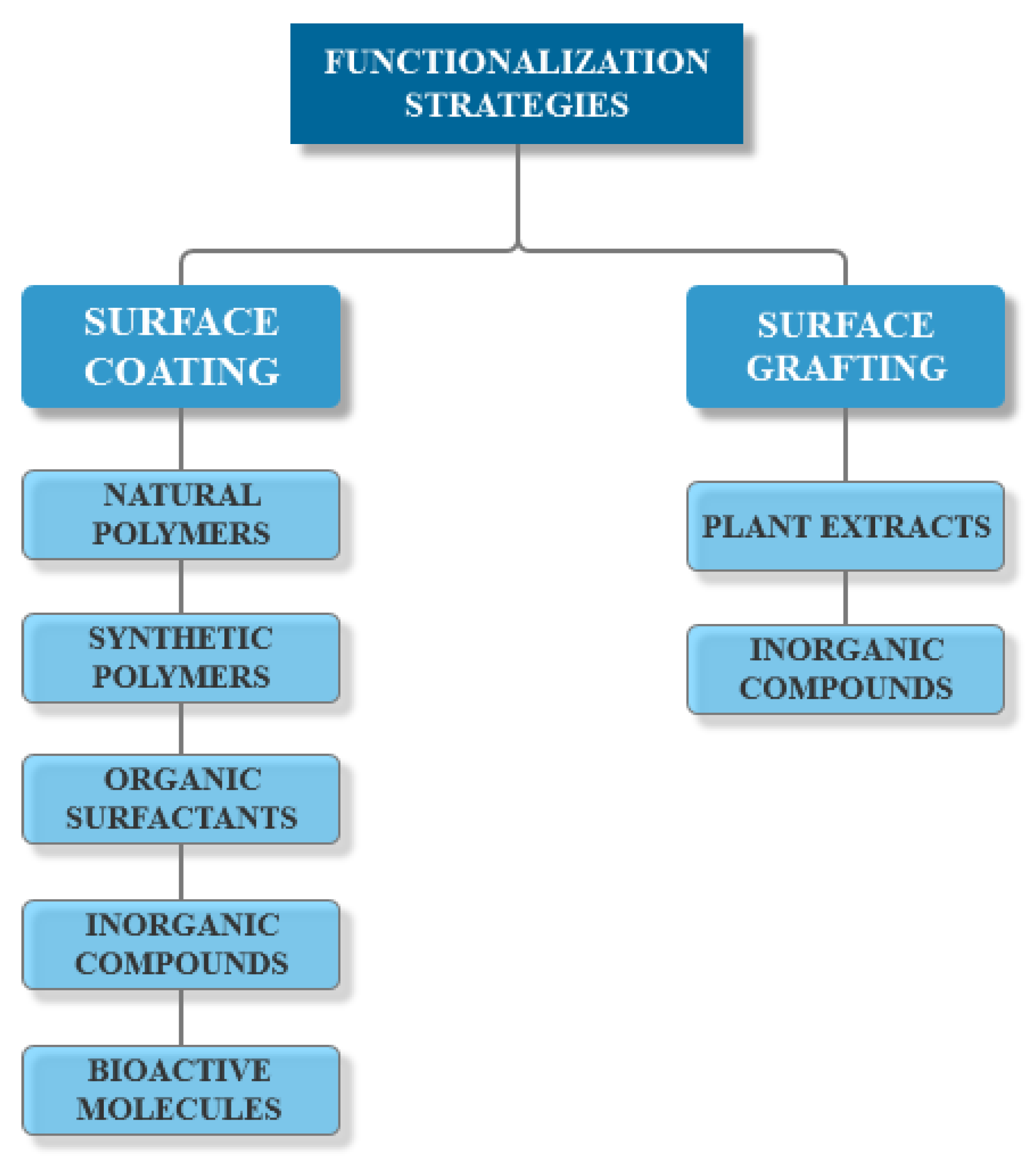
3.3. Functionalization
4. Antimicrobial Range of Essential Oils—Magnetite Nanosystems
5. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Pigłowski, M. Pathogenic and non-pathogenic microorganisms in the rapid alert system for food and feed. Int. J. Environ. Res. Public Health 2019, 16, 477. [Google Scholar] [CrossRef] [PubMed]
- Meena, M.; Swapnil, P.; Zehra, A.; Aamir, M.; Dubey, M.K.; Patel, C.B.; Upadhyay, R.S. Chapter 11—Virulence factors and their associated genes in microbes. In New and Future Developments in Microbial Biotechnology and Bioengineering; Singh, H.B., Gupta, V.K., Jogaiah, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 181–208. [Google Scholar] [CrossRef]
- Gutiérrez, T.J. Chapter 19—Antibiofilm enzymes as an emerging technology for food quality and safety. In Enzymes in Food Biotechnology; Kuddus, M., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 321–342. [Google Scholar] [CrossRef]
- Zalavras, C.G.; Patzakis, M.J. 129—Septic arthritis in anterior cruciate ligament surgery. In The Anterior Cruciate Ligament, 2nd ed.; Prodromos, C.C., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 513–517.e512. [Google Scholar] [CrossRef]
- Cook, P.P.; Siraj, D.S. Chapter 109—Bacterial arthritis. In Kelley and Firestein’s Textbook of Rheumatology, 10th ed.; Firestein, G.S., Budd, R.C., Gabriel, S.E., McInnes, I.B., O’Dell, J.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1876–1890. [Google Scholar] [CrossRef]
- Melton, C.N.; Anderson, G.G. Biofilms and Disease: A Persistent Threat. In Encyclopedia of Microbiology, 4th ed.; Schmidt, T.M., Ed.; Academic Press: Oxford, UK, 2019; pp. 510–519. [Google Scholar] [CrossRef]
- Stewart, P.S.; Bjarnsholt, T. Risk factors for chronic biofilm-related infection associated with implanted medical devices. Clin. Microbiol. Infect. 2020, 26, P1034–P1038. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, D.; Nouvong, A. Infection Protocols for Implants. Clin. Podiatr. Med. Surg. 2019, 36, 627–649. [Google Scholar] [CrossRef] [PubMed]
- Caplin, J.D.; García, A.J. Implantable antimicrobial biomaterials for local drug delivery in bone infection models. Acta Biomater. 2019, 93, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Tang, T. Surface treatment strategies to combat implant-related infection from the beginning. J. Orthop. Transl. 2019, 17, 42–54. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.A.; Baig, F.K.; Mehboob, R. Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pac. J. Trop. Biomed. 2017, 7, 478–482. [Google Scholar] [CrossRef]
- Edwardson, S.; Cairns, C. Nosocomial infections in the ICU. Anaesth. Intensive Care Med. 2019, 20, 14–18. [Google Scholar] [CrossRef]
- Agaba, P.; Tumukunde, J.; Tindimwebwa, J.V.B.; Kwizera, A. Nosocomial bacterial infections and their antimicrobial susceptibility patterns among patients in Ugandan intensive care units: A cross sectional study. BMC Res. Notes 2017, 10, 349. [Google Scholar] [CrossRef]
- Tolera, M.; Abate, D.; Dheresa, M.; Marami, D. Bacterial nosocomial infections and antimicrobial susceptibility pattern among patients admitted at Hiwot Fana Specialized University Hospital, Eastern Ethiopia. Adv. Med. 2018, 2018. [Google Scholar] [CrossRef]
- Chanda, W.; Manyepa, M.; Chikwanda, E.; Daka, V.; Chileshe, J.; Tembo, M.; Kasongo, J.; Chipipa, A.; Handema, R.; Mulemena, J.A. Evaluation of antibiotic susceptibility patterns of pathogens isolated from routine laboratory specimens at Ndola Teaching Hospital: A retrospective study. PLoS ONE 2019, 14, e0226676. [Google Scholar] [CrossRef] [PubMed]
- Kelly, S.A.; Rodgers, A.M.; O’Brien, S.C.; Donnelly, R.F.; Gilmore, B.F. Gut check time: Antibiotic delivery strategies to reduce antimicrobial resistance. Trends Biotechnol. 2019, 38, P447–P462. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Naseri, M.; He, Y.; Xu, C.; Walsh, L.J.; Ziora, Z.M. Non-antibiotic antimicrobial agents to combat biofilm-forming bacteria. J. Glob. Antimicrob. Resist. 2019, 21, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Niu, G.; Li, W. Next-generation drug discovery to combat antimicrobial resistance. Trends Biochem. Sci. 2019, 44, 961–972. [Google Scholar] [CrossRef] [PubMed]
- Trotter, A.J.; Aydin, A.; Strinden, M.J.; O’Grady, J. Recent and emerging technologies for the rapid diagnosis of infection and antimicrobial resistance. Curr. Opin. Microbiol. 2019, 51, 39–45. [Google Scholar] [CrossRef]
- Chatzopoulou, M.; Reynolds, L. Role of antimicrobial restrictions in bacterial resistance control: A systematic literature review. J. Hosp. Infect. 2020, 104, 125–136. [Google Scholar] [CrossRef]
- Sarmah, P.; Dan, M.; Adapa, D. A review on common pathogenic microorganisms and their impact on human health. Electron. J. Biol. 2018, 14, 50–58. [Google Scholar]
- Limmathurotsakul, D.; Dunachie, S.; Fukuda, K.; Feasey, N.A.; Okeke, I.N.; Holmes, A.H.; Moore, C.E.; Dolecek, C.; van Doorn, H.R.; Shetty, N.; et al. Improving the estimation of the global burden of antimicrobial resistant infections. Lancet Infect. Dis. 2019, 19, e392–e398. [Google Scholar] [CrossRef]
- Ogunsona, E.O.; Muthuraj, R.; Ojogbo, E.; Valerio, O.; Mekonnen, T.H. Engineered nanomaterials for antimicrobial applications: A review. Appl. Mater. Today 2020, 18, 100473. [Google Scholar] [CrossRef]
- Bhavaniramya, S.; Vishnupriya, S.; Al-Aboody, M.S.; Vijayakumar, R.; Baskaran, D. Role of essential oils in food safety: Antimicrobial and antioxidant applications. Grain Oil Sci. Technol. 2019, 2, 49–55. [Google Scholar] [CrossRef]
- Tariq, S.; Wani, S.; Rasool, W.; Shafi, K.; Bhat, M.A.; Prabhakar, A.; Shalla, A.H.; Rather, M.A. A comprehensive review of the antibacterial, antifungal and antiviral potential of essential oils and their chemical constituents against drug-resistant microbial pathogens. Microb. Pathog. 2019, 134, 103580. [Google Scholar] [CrossRef] [PubMed]
- Deyno, S.; Mtewa, A.G.; Abebe, A.; Hymete, A.; Makonnen, E.; Bazira, J.; Alele, P.E. Essential oils as topical anti-infective agents: A systematic review and meta-analysis. Complement. Ther. Med. 2019, 47, 102224. [Google Scholar] [CrossRef] [PubMed]
- Omonijo, F.A.; Ni, L.; Gong, J.; Wang, Q.; Lahaye, L.; Yang, C. Essential oils as alternatives to antibiotics in swine production. Anim. Nutr. 2018, 4, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Alphandéry, E. Iron oxide nanoparticles for therapeutic applications. Drug Discov. Today 2020, 25, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Sun, W.; Xiao, Y.; Shi, X. Ultrasmall iron oxide nanoparticles: Synthesis, surface modification, assembly, and biomedical applications. Drug Discov. Today 2019, 24, 835–844. [Google Scholar] [CrossRef] [PubMed]
- Aisida, S.O.; Akpa, P.A.; Ahmad, I.; Zhao, T.-k.; Maaza, M.; Ezema, F.I. Bio-inspired encapsulation and functionalization of iron oxide nanoparticles for biomedical applications. Eur. Polym. J. 2020, 122, 109371. [Google Scholar] [CrossRef]
- Afradi, N.; Foroughifar, N.; Qomi, M.; Pasdar, H. Folic acid-supported Fe3O4 magnetic nanoparticles as a new, highly effective heterogeneous biocatalyst for the synthesis of 3,4-dihydropyrimidine thiones and their in vitro investigation as antibacterial active agents. Biointerface Res. Appl. Chem. 2018, 8, 3661–3669. [Google Scholar]
- Bahrami, M.K.; Movafeghi, A.; Mahdavinia, G.R.; Hassanpouraghdam, M.B.; Gohari, G. Effects of bare and chitosan-coated Fe3O4 magnetic nanoparticles on seed germination and seedling growth of Capsicum annuum L. Biointerface Res. Appl. Chem. 2018, 8, 3552–3559. [Google Scholar]
- Naskar, A.; Bera, S.; Bhattacharya, R.; Roy, S.S.; Jana, S. Solution based PEG and PVP capped maghemite–reduced graphene oxide nanocomposites: Cell viability study. Biointerface Res. Appl. Chem. 2018, 8, 3751–3757. [Google Scholar]
- Aziz, Z.; Ahmad, A.; Mohd-Setapar, S.; Karakucuk, A.; Azim, M.; Lokhat, D.; Kamal, M.; Ashraf, G. Essential oils: Extraction techniques, pharmaceutical and therapeutic potential—A Review. Curr. Drug Metab. 2018, 19. [Google Scholar] [CrossRef]
- Jugreet, B.S.; Suroowan, S.; Rengasamy, R.R.K.; Mahomoodally, M.F. Chemistry, bioactivities, mode of action and industrial applications of essential oils. Trends Food Sci. Technol. 2020, 101, 89–105. [Google Scholar] [CrossRef]
- Podgoreanu, P.; Negrea, S.M.; Buia, R.; Delcaru, C.; Trusca, S.B.; Lazar, V.; Chifiriuc, M.C. Alternative strategies for fighting multidrug resistant bacterial infections. Biointerface Res. Appl. Chem. 2019, 9, 3834–3841. [Google Scholar] [CrossRef]
- Dias-Souza, M.V.; Dias, C.G.; Ferreira-Marçal, P.H. Interactions of natural products and antimicrobial drugs: Investigations of a dark matter in chemistry. Biointerface Res. Appl. Chem. 2018, 8, 3259–3264. [Google Scholar]
- De Matos, S.P.; Lucca, L.G.; Koester, L.S. Essential oils in nanostructured systems: Challenges in preparation and analytical methods. Talanta 2019, 195, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Keshvadi, M.; Karimi, F.; Valizadeh, S.; Valizadeh, A. Comparative study of antibacterial inhibitory effect of silver nanoparticles and garlic oil nanoemulsion with their combination. Biointerface Res. Appl. Chem. 2019, 9. [Google Scholar] [CrossRef]
- Khammee, T.; Phoonan, W.; Ninsuwan, U.; Jaratrungtawee, A.; Kuno, M. Volatile constituents, in vitro and in silico anti-hyaluronidase activity of the essential oil from gardenia carinata wall. ex roxb. flowers. Biointerface Res. Appl. Chem. 2019, 9, 4649–4654. [Google Scholar] [CrossRef]
- Prakash, B.; Kujur, A.; Yadav, A.; Kumar, A.; Singh, P.P.; Dubey, N.K. Nanoencapsulation: An efficient technology to boost the antimicrobial potential of plant essential oils in food system. Food Control 2018, 89, 1–11. [Google Scholar] [CrossRef]
- Ali, A.; Chua, B.L.; Chow, Y.H. An insight into the extraction and fractionation technologies of the essential oils and bioactive compounds in Rosmarinus officinalis L.: Past, present and future. Trends Anal. Chem. 2019, 118, 338–351. [Google Scholar] [CrossRef]
- Stratakos, A.C.; Koidis, A. Chapter 4—Methods for extracting essential oils. In Essential Oils in Food Preservation, Flavor and Safety; Preedy, V.R., Ed.; Academic Press: San Diego, CA, USA, 2016; pp. 31–38. [Google Scholar] [CrossRef]
- Mejri, J.; Aydi, A.; Abderrabba, M.; Mejri, M. Emerging extraction processes of essential oils: A review. Asian J. Green Chem. 2018, 2, 246–267. [Google Scholar] [CrossRef]
- Gandhi, G.R.; Vasconcelos, A.B.S.; Haran, G.H.; Calisto, V.K.d.S.; Jothi, G.; Quintans, J.d.S.S.; Cuevas, L.E.; Narain, N.; Júnior, L.J.Q.; Cipolotti, R.; et al. Essential oils and its bioactive compounds modulating cytokines: A systematic review on anti-asthmatic and immunomodulatory properties. Phytomedicine 2019, 152854. [Google Scholar] [CrossRef]
- Dagli, N.; Dagli, R.; Mahmoud, R.S.; Baroudi, K. Essential oils, their therapeutic properties, and implication in dentistry: A review. J. Int. Soc. Prev. Commun. Dent. 2015, 5, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Jurado, F.; Franco-Vega, A.; Ramírez-Corona, N.; Palou, E.; López-Malo, A. Essential oils: Antimicrobial activities, extraction methods, and their modeling. Food Eng. Rev. 2015, 7, 275–297. [Google Scholar] [CrossRef]
- Shojaee-Aliabadi, S.; Hosseini, S.M.; Mirmoghtadaie, L. Antimicrobial activity of essential oil. In Essential Oils in Food Processing; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2017; pp. 191–229. [Google Scholar] [CrossRef]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Antimicrobial properties of plant essential oils against human pathogens and their mode of action: An updated review. Evid. Based Complement. Altern. Med. 2016, 2016, 3012462. [Google Scholar] [CrossRef] [PubMed]
- Musielak, M.; Piotrowski, I.; Suchorska, W.M. Superparamagnetic iron oxide nanoparticles (SPIONs) as a multifunctional tool in various cancer therapies. Rep. Pract. Oncol. Radiother. 2019, 24, 307–314. [Google Scholar] [CrossRef]
- Paunovic, J.; Vucevic, D.; Radosavljevic, T.; Mandić-Rajčević, S.; Pantic, I. Iron-based nanoparticles and their potential toxicity: Focus on oxidative stress and apoptosis. Chem. Biol. Interact. 2020, 316, 108935. [Google Scholar] [CrossRef]
- Tong, S.; Zhu, H.; Bao, G. Magnetic iron oxide nanoparticles for disease detection and therapy. Mater. Today 2019, 31, 86–99. [Google Scholar] [CrossRef]
- Salviano, L.B.; Cardoso, T.M.d.S.; Silva, G.C.; Dantas, M.S.S.; Ferreira, A.d.M. Microstructural assessment of magnetite nanoparticles (Fe3O4) obtained by chemical precipitation under different synthesis conditions. Mater. Res. 2018, 21, e20170764. [Google Scholar] [CrossRef]
- Liu, S.; Yu, B.; Wang, S.; Shen, Y.; Cong, H. Preparation, surface functionalization and application of Fe3O4 magnetic nanoparticles. Adv. Colloid Interface Sci. 2020, 281, 102165. [Google Scholar] [CrossRef]
- Sangaiya, P.; Jayaprakash, R. A review on iron oxide nanoparticles and their biomedical applications. J. Supercond. Nov. Magn. 2018, 31, 3397–3413. [Google Scholar] [CrossRef]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knüchel, R.; Kiessling, F.; Lammers, T. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef]
- Wu, W.; Jiang, C.Z.; Roy, V.A.L. Designed synthesis and surface engineering strategies of magnetic iron oxide nanoparticles for biomedical applications. Nanoscale 2016, 8, 19421–19474. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.A.M.K.; Ficiarà, E.; Ruffinatti, F.A.; Stura, I.; Argenziano, M.; Abollino, O.; Cavalli, R.; Guiot, C.; D′Agata, F. Magnetic iron oxide nanoparticles: Synthesis, characterization and functionalization for biomedical applications in the central nervous system. Materials 2019, 12, 465. [Google Scholar] [CrossRef] [PubMed]
- Elazab, H.A.; El-Idreesy, T.T. Optimization of the catalytic performance of Pd/Fe3O4nanoparticles prepared via microwave-assisted synthesis for pharmaceutical and catalysis applications. Biointerface Res. Appl. Chem. 2019, 9, 3794–3799. [Google Scholar] [CrossRef]
- Elazab, H.A.; Gadalla, M.A.; Sadek, M.A.; El-Idreesy, T.T. Hydrothermal synthesis of graphene supported Pd/ Fe3O4nanoparticles as efficient magnetic catalysts for suzuki cross—Coupling. Biointerface Res. Appl. Chem. 2019, 9, 3906–3911. [Google Scholar] [CrossRef]
- Marcelo, G.A.; Lodeiro, C.; Capelo, J.L.; Lorenzo, J.; Oliveira, E. Magnetic, fluorescent and hybrid nanoparticles: From synthesis to application in biosystems. Mater. Sci. Eng. C 2020, 106, 110104. [Google Scholar] [CrossRef] [PubMed]
- Holban, A.M.; Grumezescu, A.M.; Andronescu, E. Chapter 10—Inorganic nanoarchitectonics designed for drug delivery and anti-infective surfaces. In Surface Chemistry of Nanobiomaterials; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 301–327. [Google Scholar] [CrossRef]
- Kumar, P.S.S.R.; Alexis, S.J. Chapter four—Synthesized carbon nanotubes and their applications. In Carbon-Based Nanofillers and Their Rubber Nanocomposites; Yaragalla, S., Mishra, R., Thomas, S., Kalarikkal, N., Maria, H.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 109–122. [Google Scholar] [CrossRef]
- Jamkhande, P.G.; Ghule, N.W.; Bamer, A.H.; Kalaskar, M.G. Metal nanoparticles synthesis: An overview on methods of preparation, advantages and disadvantages, and applications. J. Drug Deliv. Sci. Technol. 2019, 53, 101174. [Google Scholar] [CrossRef]
- Fu, X.; Cai, J.; Zhang, X.; Li, W.-D.; Ge, H.; Hu, Y. Top-down fabrication of shape-controlled, monodisperse nanoparticles for biomedical applications. Adv. Drug Deliv. Rev. 2018, 132, 169–187. [Google Scholar] [CrossRef]
- Iriarte-Mesa, C.; López, Y.C.; Matos-Peralta, Y.; de la Vega-Hernández, K.; Antuch, M. Gold, silver and iron oxide nanoparticles: Synthesis and bionanoconjugation strategies aimed at electrochemical applications. Top. Curr. Chem. 2020, 378, 12. [Google Scholar] [CrossRef]
- Magdalena, B.; Irena, K.; Lidia, D. The effect of magnetite nanoparticles synthesis conditions on their ability to separate heavy metal ions. Arch. Environ. Prot. 2017, 43, 3–9. [Google Scholar] [CrossRef]
- Yu, J.; Ju, Y.; Chen, F.; Che, S.; Zhao, L.; Sheng, F.; Hou, Y. Chemical synthesis and biomedical applications of iron oxide nanoparticles. In Nanobiomaterials; Wiley: Hoboken, NJ, USA, 2017; pp. 329–358. [Google Scholar] [CrossRef]
- Abd Elrahman, A.A.; Mansour, F.R. Targeted magnetic iron oxide nanoparticles: Preparation, functionalization and biomedical application. J. Drug Deliv. Sci. Technol. 2019, 52, 702–712. [Google Scholar] [CrossRef]
- Unni, M.; Uhl, A.M.; Savliwala, S.; Savitzky, B.H.; Dhavalikar, R.; Garraud, N.; Arnold, D.P.; Kourkoutis, L.F.; Andrew, J.S.; Rinaldi, C. Thermal decomposition synthesis of iron oxide nanoparticles with diminished magnetic dead layer by controlled addition of oxygen. ACS Nano 2017, 11, 2284–2303. [Google Scholar] [CrossRef] [PubMed]
- Cotin, G.; Kiefer, C.; Perton, F.; Ihiawakrim, D.; Blanco-Andujar, C.; Moldovan, S.; Lefevre, C.; Ersen, O.; Pichon, B.; Mertz, D.; et al. Unravelling the thermal decomposition parameters for the synthesis of anisotropic iron oxide nanoparticles. Nanomaterials 2018, 8, 881. [Google Scholar] [CrossRef] [PubMed]
- Bhateria, R.; Singh, R. A review on nanotechnological application of magnetic iron oxides for heavy metal removal. J. Water Process. Eng. 2019, 31, 100845. [Google Scholar] [CrossRef]
- Kammari, R.; Das, N.G.; Das, S.K. Chapter 6—Nanoparticulate systems for therapeutic and diagnostic applications. In Emerging Nanotechnologies for Diagnostics, Drug Delivery and Medical Devices; Mitra, A.K., Cholkar, K., Mandal, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 105–144. [Google Scholar] [CrossRef]
- Soleimani Zohr Shiri, M.; Henderson, W.; Mucalo, M.R. A review of the lesser-studied microemulsion-based synthesis methodologies used for preparing nanoparticle systems of the noble metals, Os, Re, Ir and Rh. Materials 2019, 12, 1896. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, N.N. Green synthesis of iron oxide nanoparticles (IONPs) and their nanotechnological applications. J. Bacteriol. Mycol. Open Access 2018, 6, 260–262. [Google Scholar] [CrossRef]
- Moaca, E.; Coricovac, E.D.; Soica, C.M.; Pinzaru, I.A.; Pacurariu, C.; Dehelean, C.A. Preclinical aspects on magnetic iron oxide nanoparticles and their interventions as anticancer agents: Enucleation, apoptosis and other mechanism. In Iron Ores and Iron Oxide Materials; Shatokha, V., Ed.; Intechopen: London, UK, 2018; pp. 229–254. [Google Scholar] [CrossRef]
- Pantapasis, K.; Anton, G.-C.; Bontas, D.-A.; Sarghiuta, D.; Grumezescu, A.M.; Holban, A.M. Chapter 2—Bioengineered nanomaterials for chemotherapy. In Nanostructures for Cancer Therapy; Ficai, A., Grumezescu, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 23–49. [Google Scholar] [CrossRef]
- Menon, P.K.; Sharma, A.; Lafuente, J.V.; Muresanu, D.F.; Aguilar, Z.P.; Wang, Y.A.; Patnaik, R.; Mössler, H.; Sharma, H.S. Chapter three—Intravenous administration of functionalized magnetic iron oxide nanoparticles does not induce CNS injury in the rat: Influence of spinal cord trauma and cerebrolysin treatment. In International Review of Neurobiology; Sharma, H.S., Sharma, A., Eds.; Academic Press: Cambridge, MA, USA, 2017; Volume 137, pp. 47–63. [Google Scholar]
- Muthuraman, A.; Kaur, J. Chapter 6—Antimicrobial nanostructures for neurodegenerative infections: Present and future perspectives. In Nanostructures for Antimicrobial Therapy; Ficai, A., Grumezescu, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 139–167. [Google Scholar] [CrossRef]
- Xie, W.; Guo, Z.; Gao, F.; Gao, Q.; Wang, D.; Liaw, B.-S.; Cai, Q.; Sun, X.; Wang, X.; Zhao, L. Shape-, size- and structure-controlled synthesis and biocompatibility of iron oxide nanoparticles for magnetic theranostics. Theranostics 2018, 8, 3284–3307. [Google Scholar] [CrossRef] [PubMed]
- Wallyn, J.; Anton, N.; Vandamme, T.F. Synthesis, principles, and properties of magnetite nanoparticles for in vivo imaging applications—A review. Pharmaceutics 2019, 11, 601. [Google Scholar] [CrossRef]
- Roca, A.G.; Gutiérrez, L.; Gavilán, H.; Fortes Brollo, M.E.; Veintemillas-Verdaguer, S.; Morales, M.d.P. Design strategies for shape-controlled magnetic iron oxide nanoparticles. Adv. Drug Deliv. Rev. 2019, 138, 68–104. [Google Scholar] [CrossRef]
- Kozlenko, D.P.; Dubrovinsky, L.S.; Kichanov, S.E.; Lukin, E.V.; Cerantola, V.; Chumakov, A.I.; Savenko, B.N. Magnetic and electronic properties of magnetite across the high pressure anomaly. Sci. Rep. 2019, 9, 4464. [Google Scholar] [CrossRef]
- Daoush, W. Co-precipitation and magnetic properties of magnetite nanoparticles for potential biomedical applications. J. Nanomed. Res. 2017, 5, 00118. [Google Scholar] [CrossRef]
- Sajid, M.; Płotka-Wasylka, J. Nanoparticles: Synthesis, characteristics, and applications in analytical and other sciences. Microchem. J. 2020, 154, 104623. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Z.; Li, X.; Zhang, Y.; Yin, M.; Li, J.; Song, H.; Shi, J.; Ling, D.; Wang, L.; et al. Deciphering active biocompatibility of iron oxide nanoparticles from their intrinsic antagonism. Nano Res. 2018, 11, 2746–2755. [Google Scholar] [CrossRef]
- Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; Lima, T.M.T.d.; Delbem, A.C.B.; Monteiro, D.R. Iron oxide nanoparticles for biomedical applications: A perspective on synthesis, drugs, antimicrobial activity, and toxicity. Antibiotics 2018, 7, 46. [Google Scholar] [CrossRef] [PubMed]
- Saqib, S.; Munis, M.F.H.; Zaman, W. Synthesis, characterization and use of iron oxide nano particles for antibacterial activity. Microsc. Res. Tech. 2019, 82, 415–420. [Google Scholar] [CrossRef]
- Cotin, G.; Piant, S.; Mertz, D.; Felder-Flesch, D.; Begin-Colin, S. Chapter 2—Iron oxide nanoparticles for biomedical applications: Synthesis, functionalization, and application. In Iron Oxide Nanoparticles for Biomedical Applications; Mahmoudi, M., Laurent, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 43–88. [Google Scholar] [CrossRef]
- Vallabani, N.V.S.; Singh, S. Recent advances and future prospects of iron oxide nanoparticles in biomedicine and diagnostics. 3 Biotech. 2018, 8, 279. [Google Scholar] [CrossRef]
- Zabel, R.A.; Morrell, J.J. Chapter Three—The characteristics and classification of fungi and bacteria. In Wood Microbiology, 2nd ed.; Zabel, R.A., Morrell, J.J., Eds.; Academic Press: San Diego, CA, USA, 2020; pp. 55–98. [Google Scholar] [CrossRef]
- Mai-Prochnow, A.; Clauson, M.; Hong, J.; Murphy, A.B. Gram positive and gram negative bacteria differ in their sensitivity to cold plasma. Sci. Rep. 2016, 6, 38610. [Google Scholar] [CrossRef]
- St. Geme, J.W.; Rempe, K.A. 114—Classification of bacteria. In Principles and Practice of Pediatric Infectious Diseases, 5th ed.; Long, S.S., Prober, C.G., Fischer, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 690–692.e691. [Google Scholar] [CrossRef]
- Berman, J.J. Chapter 3—Bacteria. In Taxonomic Guide to Infectious Diseases, 2nd ed.; Berman, J.J., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 39–119. [Google Scholar] [CrossRef]
- Waller, D.G.; Sampson, A.P. 51—Chemotherapy of infections. In Medical Pharmacology and Therapeutics, 5th ed.; Waller, D.G., Sampson, A.P., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 581–629. [Google Scholar] [CrossRef]
- Hughes, S.; Heard, K.; Moore, L. Antimicrobial therapies for Gram-positive infections. Lung Cancer 2018, 15. [Google Scholar]
- Brown, L.; Wolf, J.M.; Prados-Rosales, R.; Casadevall, A. Through the wall: Extracellular vesicles in Gram-positive bacteria, mycobacteria and fungi. Nat. Rev. Microbiol. 2015, 13, 620–630. [Google Scholar] [CrossRef]
- Raghavendra, P.; Pullaiah, T. Chapter 8—Future of cellular and molecular diagnostics: Bench to bedside. In Advances in Cell and Molecular Diagnostics; Raghavendra, P., Pullaiah, T., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 203–270. [Google Scholar] [CrossRef]
- Foster, T.J.; Geoghegan, J.A. Chapter 37—Staphylococcus aureus. In Molecular Medical Microbiology, 2nd ed.; Tang, Y.-W., Sussman, M., Liu, D., Poxton, I., Schwartzman, J., Eds.; Academic Press: Boston, NY, USA, 2015; pp. 655–674. [Google Scholar] [CrossRef]
- Anghel, I.; Grumezescu, A.M. Hybrid nanostructured coating for increased resistance of prosthetic devices to staphylococcal colonization. Nanoscale Res. Lett. 2013, 8, 6. [Google Scholar] [CrossRef]
- Rădulescu, M.; Andronescu, E.; Holban, A.M.; Vasile, B.S.; Iordache, F.; Mogoantă, L.; Mogoșanu, G.D.; Grumezescu, A.M.; Georgescu, M.; Chifiriuc, M.C. Antimicrobial nanostructured bioactive coating based on Fe3O4 and patchouli oil for wound dressing. Metals 2016, 6, 103. [Google Scholar] [CrossRef]
- Bilcu, M.; Grumezescu, A.M.; Oprea, A.E.; Popescu, R.C.; Mogoșanu, G.D.; Hristu, R.; Stanciu, G.A.; Mihailescu, D.F.; Lazar, V.; Bezirtzoglou, E.; et al. Efficiency of vanilla, patchouli and ylang ylang essential oils stabilized by iron oxide@C14 nanostructures against bacterial adherence and biofilms formed by Staphylococcus aureus and Klebsiella pneumoniae clinical strains. Molecules 2014, 19, 17943–17956. [Google Scholar] [CrossRef] [PubMed]
- Anghel, A.G.; Grumezescu, A.M.; Chirea, M.; Grumezescu, V.; Socol, G.; Iordache, F.; Oprea, A.E.; Anghel, I.; Holban, A.M. MAPLE Fabricated Fe3O4@Cinnamomum verum antimicrobial surfaces for improved gastrostomy tubes. Molecules 2014, 19, 8981–8994. [Google Scholar] [CrossRef] [PubMed]
- Negut, I.; Grumezescu, V.; Ficai, A.; Grumezescu, A.M.; Holban, A.M.; Popescu, R.C.; Savu, D.; Vasile, B.S.; Socol, G. MAPLE deposition of Nigella sativa functionalized Fe3O4 nanoparticles for antimicrobial coatings. Appl. Surf. Sci. 2018, 455, 513–521. [Google Scholar] [CrossRef]
- Mousavi, S.M.; Hashemi, S.A.; Ramakrishna, S.; Esmaeili, H.; Bahrani, S.; Koosha, M.; Babapoor, A. Green synthesis of supermagnetic Fe3O4–MgO nanoparticles via Nutmeg essential oil toward superior antibacterial and antifungal performance. J. Drug Deliv. Sci. Technol. 2019, 54, 101352. [Google Scholar] [CrossRef]
- Grumezescu, V.; Holban, A.M.; Iordache, F.; Socol, G.; Mogoşanu, G.D.; Grumezescu, A.M.; Ficai, A.; Vasile, B.Ş.; Truşcă, R.; Chifiriuc, M.C.; et al. MAPLE fabricated magnetite@eugenol and (3-hidroxybutyric acid-co-3-hidroxyvaleric acid)–polyvinyl alcohol microspheres coated surfaces with anti-microbial properties. Appl. Surf. Sci. 2014, 306, 16–22. [Google Scholar] [CrossRef]
- Anghel, I.; Holban, A.M.; Grumezescu, A.M.; Andronescu, E.; Ficai, A.; Anghel, A.G.; Maganu, M.; Lazăr, V.; Chifiriuc, M.C. Modified wound dressing with phyto-nanostructured coating to prevent staphylococcal and pseudomonal biofilm development. Nanoscale Res. Lett. 2012, 7, 690. [Google Scholar] [CrossRef]
- Srinivasan, L.; Evans, J.R. 40—Health care-associated infections. In Avery’s Diseases of the Newborn, 10th ed.; Gleason, C.A., Juul, S.E., Eds.; Content Repository Only!: Philadelphia, PA, USA, 2018; pp. 566–580.e566. [Google Scholar] [CrossRef]
- Anderson, A.C.; Jonas, D.; Huber, I.; Karygianni, L.; Wölber, J.; Hellwig, E.; Arweiler, N.; Vach, K.; Wittmer, A.; Al-Ahmad, A. Enterococcus faecalis from food, clinical specimens, and oral sites: Prevalence of virulence factors in association with biofilm formation. Front. Microbiol. 2016, 6, 1534. [Google Scholar] [CrossRef]
- Park, M.Y.; Eslick, G.D. Chapter 9—Infectious organisms associated with colorectal cancer. In Gastrointestinal Diseases and their Associated Infections; Eslick, G.D., Ed.; Elsevier: Philadelphia, PA, USA, 2019; pp. 113–124. [Google Scholar] [CrossRef]
- Skariyachan, S.; Garka, S. Chapter 1—Exploring the binding potential of carbon nanotubes and fullerene towards major drug targets of multidrug resistant bacterial pathogens and their utility as novel therapeutic agents. In Fullerens, Graphenes and Nanotubes; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2018; pp. 1–29. [Google Scholar] [CrossRef]
- Poolman, J.T. Escherichia coli. In International Encyclopedia of Public Health, 2nd ed.; Quah, S.R., Ed.; Academic Press: Oxford, UK, 2017; pp. 585–593. [Google Scholar] [CrossRef]
- Mathers, A.J.; Peirano, G.; Pitout, J.D.D. Chapter four—Escherichia coli ST131: The quintessential example of an international multiresistant high-risk clone. In Advances in Applied Microbiology; Sariaslani, S., Gadd, G.M., Eds.; Academic Press: Cambridge, MA, USA, 2015; Volume 90, pp. 109–154. [Google Scholar]
- Planet, P.J. 155—Pseudomonas aeruginosa. In Principles and Practice of Pediatric Infectious Diseases, 5th ed.; Long, S.S., Prober, C.G., Fischer, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 866–870.e861. [Google Scholar] [CrossRef]
- Toyofuku, M.; Yoon, S.-S. Chapter four—Nitric oxide, an old molecule with noble functions in pseudomonas aeruginosa biology. In Advances in Microbial Physiology; Poole, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2018; Volume 72, pp. 117–145. [Google Scholar]
- Wei, M.; Wang, P.G. Chapter two—Desialylation in physiological and pathological processes: New target for diagnostic and therapeutic development. In Progress in Molecular Biology and Translational Science; Zhang, L., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 162, pp. 25–57. [Google Scholar]
- Wu, M.; Li, X. Chapter 87—Klebsiella pneumoniae and Pseudomonas aeruginosa. In Molecular Medical Microbiology, 2nd ed.; Tang, Y.-W., Sussman, M., Liu, D., Poxton, I., Schwartzman, J., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 1547–1564. [Google Scholar] [CrossRef]
- Farver, C.F. 10—Bacterial diseases. In Pulmonary Pathology, 2nd ed.; Zander, D.S., Farver, C.F., Eds.; Content Repository Only!: Philadelphia, PA, USA, 2018; pp. 163–200. [Google Scholar] [CrossRef]
- Turner, S.A.; Butler, G. The Candida pathogenic species complex. Cold Spring Harb. Perspect Med. 2014, 4, a019778. [Google Scholar] [CrossRef]
- Yassin, M.T.; Mostafa, A.A.; Al-Askar, A.A.; Bdeer, R. In vitro antifungal resistance profile of Candida strains isolated from Saudi women suffering from vulvovaginitis. Eur. J. Med. Res. 2020, 25, 1. [Google Scholar] [CrossRef]
- Bernard, C.; Girardot, M.; Imbert, C. Candida albicans interaction with Gram-positive bacteria within interkingdom biofilms. J. De Mycol. Médic. 2020, 30, 100909. [Google Scholar] [CrossRef]
- Saviuc, C.; Grumezescu, A.; Chifiriuc, C.; Mihaiescu, D.; Hristu, R.; Stanciu, G.; Oprea, E.; Radulescu, V.; Lazar, V. Hybrid nanosystem for stabilizing essential oils in biomedical applications. Dig. J. Nanomater. Biostruct. 2011, 6, 1657–1666. [Google Scholar]
- Grumezescu, A.M.; Chifiriuc, M.C.; Saviuc, C.; Grumezescu, V.; Hristu, R.; Mihaiescu, D.E.; Stanciu, G.A.; Andronescu, E. hybrid nanomaterial for stabilizing the antibiofilm activity of Eugenia carryophyllata essential oil. IEEE Trans. Nanobiosci. 2012, 11, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Anghel, I.; Grumezescu, A.M.; Holban, A.M.; Ficai, A.; Anghel, A.G.; Chifiriuc, M.C. Biohybrid nanostructured iron oxide nanoparticles and satureja hortensis to prevent fungal biofilm development. Int. J. Mol. Sci. 2013, 14, 18110–18123. [Google Scholar] [CrossRef] [PubMed]
- Anghel, I.; Holban, A.M.; Andronescu, E.; Grumezescu, A.M.; Chifiriuc, M.C. Efficient surface functionalization of wound dressings by a phytoactive nanocoating refractory to Candida albicans biofilm development. Biointerphases 2013, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chifiriuc, C.; Grumezescu, V.; Grumezescu, A.M.; Saviuc, C.; Lazăr, V.; Andronescu, E. Hybrid magnetite nanoparticles/Rosmarinus officinalis essential oil nanobiosystem with antibiofilm activity. Nanoscale Res. Lett. 2012, 7, 209. [Google Scholar] [CrossRef] [PubMed]
| Extraction Method | Advantages | Disadvantages |
|---|---|---|
| Cold Pressing | Lower Temperatures Used During the Process | Low Yield; |
| Solvent Extraction | Lower Temperatures Used During the Process; Inexpensive; Fast | Trace Amounts Of Solvent; Low Yield; Loss of Volatile Compounds; Long Extraction Times |
| Enfleurage | Lower Temperatures Used During the Process | Time-Consuming; Labor-Intensive; Expensive; |
| Steam or Water Distillation | Straightforward Process | High Temperatures Which Can Cause Chemical Changes in the Compounds; Loss ff Volatile Compounds; Low Yield; Might Require Oil Rectification (Redistillation) |
| Supercritical Fluid Extraction | Low Temperatures Used During The Process; High Selectivity; Environmentally Friendly; Analytically and Industrially Scalable; Fast; Low Energy Costs | Lower EOs Quantities |
| Microwave-Assisted Extraction | Reasonable Costs; Good Performance under Atmospheric Conditions; Higher Extraction Yields; Shorter Extraction Times | Requires Higher Quantities of Organic Solvent; Less Environmentally Friendly |
| Ultrasound-Assisted Extraction | Minimum Effects on Extractable Compounds; Reduction/Avoidance of Organic Solvents; Shorter Extraction Times; High Yields | Expensive |
| Instantaneous Controlled Pressure Drop Process | Enhanced Global Diffusivity; Does Not Require the Use of Solvents; Higher Extraction Yields; Lower Energy Required; No Chemical Changes in the Compounds | - |
| Molecular Formula | Color | Density | Melting Temperature | Hardness | Type of Magnetism |
|---|---|---|---|---|---|
| Fe3O4 | black | 5.18 g/cm3 | 1583−1597 °C | 5.5 | Ferrimagnetic |
| Curie Temperature | Saturation Magnetization at 300 K | Standard Gibbs Free Energy of Formation | Crystallographic System | Structure Type | Lattice Parameter |
| 858 K | 92–100 A∙m2/kg | −1012.6 kJ/mol | cubic | inverse spinel | Å = 0.8396 nm |
| Microbial Species | Type of Nanosystem | EO Main Constituents | Nanosystem Dimension | MIC Value | Desired Application | Reference |
|---|---|---|---|---|---|---|
| Staphylococcus aureus | Core–Shell MNPs Functionalized with Mentha piperita EO | n.r. | 5 nm | n.r. | Catheter Coating | [101] |
| MNPs Functionalized with Patchouli EO | n.r. | 7.5 nm | n.r. | Wound Dressing Coating | [102] | |
| MNPs Functionalized with Patchouli, Vanilla, and Ylang Ylang EOs | n.r. | >20 nm | n.r. | Catheter Coating | [103] | |
| MNPs Functionalized with Cinnamomum verum EO | n.r. | 9–10 nm | n.r. | Gastrostomy Tube Coating | [104] | |
| MNPs Functionalized with Nigella sativa EO and MNPs Functionalized with Nigella Sativa EO Embedded into a Polymeric Matrix of Poly(lactic-co-glycolic acid) | n.r. | 14–21 nm | 0.62 mg/mL | Antimicrobial Coatings | [105] | |
| MNPs Functionalized with Nutmeg EO | Limonene, Terpineol, Carvacrol, and Thymol | 10–15 nm | 250 μg/mL and 125 μg/mL | Antimicrobial Coatings | [106] | |
| MNPs Functionalized with Eugenol | - | >10 nm | n.r. | Antimicrobial Coatings | [107] | |
| MNPs Functionalized with Eugenol And Limonene | - | 10 nm | n.r. | Wound Dressing Coating | [108] | |
| Enterococcus faecalis | MNPs Functionalized with Nutmeg EO | Limonene, Terpineol, Carvacrol, And Thymol | 10–15 nm | 250 μg/mL and 125 μg/mL | Antimicrobial Coatings | [106] |
| Escherichia coli | MNPs Functionalized with Cinnamomum Verum EO | n.r. | 9–10 nm | n.r. | Gastrostomy Tube Coating | [104] |
| MNPs Functionalized With Nigella Sativa EO And MNPs Functionalized With Nigella Sativa EO Mixed With A Polymeric Matrix Of Poly(Lactic-Co-Glycolic Acid) | n.r. | 14–21 nm | 1.25 mg/mL | Antimicrobial Coatings | [105] | |
| MNPs Functionalized with Nutmeg EO | Limonene, Terpineol, Carvacrol, and Thymol | 10–15 nm | 62.5 μg/mL and 31.25 μg/mL | Antimicrobial Coatings | [106] | |
| Pseudomonas aeruginosa | MNPs Functionalized with Nutmeg EO | Limonene, Terpineol, Carvacrol, and Thymol | 10–15 nm | 125 μg/mL and 62.5 μg/mL | Antimicrobial Coatings | [106] |
| MNPs Functionalized with Eugenol | - | >10 nm | n.r. | Antimicrobial Coatings | [107] | |
| MNPs Functionalized with Eugenol And Limonene | - | 10 nm | n.r. | Wound Dressing Coating | [108] | |
| Klebsiella pneumoniae | MNPs Functionalized with Patchouli, Vanilla, And Ylang-Ylang Eos | n.r. | >20 nm | n.r. | Catheter Coating | [103] |
| Candida albicans | Core-Shell MNPs Functionalized with Anethum Graveolens EO | Limonene, Carvone, and A-Phellandrene | n.r. | n.r. | Antimicrobial Coatings | [123] |
| Core-Shell MNPs Functionalized with Eugenia Carryophyllata EO | n.r. | >20 nm | n.r. | Catheter Coating | [124] | |
| MNPs Functionalized with Satureja Hortensis EO | Carvacrol, γ-Terpinene, p-Cymene, α-Terpinene, and Myrcene | 10 nm | n.r. | Wound Dressing Coating | [125] | |
| MNPs Functionalized with Anethum Graveolens And Salvia Officinalis Eos | cis-thujone, eucalyptol, α,β-pinene, and camphene | 13–17 nm | n.r. | Wound Dressing Coating | [126] | |
| MNPs Functionalized with Rosmarinus Officinalis EO | Eucalyptol, Camphor, Caryophyllene, and A-Pinene | >20 nm | n.r. | Catheter Coating | [127] | |
| MNPs Functionalized with Cinnamomum Verum EO | n.r. | 9–10 nm | n.r. | Gastrostomy Tube Coating | [104] | |
| MNPs Functionalized With Nigella Sativa EO And MNPs Functionalized With Nigella Sativa EO Mixed With A Polymeric Matrix Of Poly(Lactic-Co-Glycolic Acid) | n.r. | 14–21 nm | 0.62 mg/mL | Antimicrobial Coatings | [105] | |
| Candida tropicalis | Core-Shell MNPs Functionalized with Anethum Graveolens EO | limonene, carvone, and α-phellandrene | n.r. | n.r. | Antimicrobial Coatings | [123] |
| MNPs Functionalized with Rosmarinus Officinalis EO | eucalyptol, camphor, caryophyllene, and α-pinene | >20 nm | n.r. | Catheter Coating | [127] | |
| Candida famata | Core-Shell MNPs Functionalized with Anethum Graveolens EO | limonene, carvone, and α-phellandrene | n.r. | n.r. | Antimicrobial Coatings | [123] |
| Candida glabrata | Core-Shell MNPs Functionalized with Anethum Graveolens EO | limonene, carvone, and α-phellandrene | n.r. | n.r. | Antimicrobial Coatings | [123] |
| Core-Shell MNPs Functionalized with Eugenia Carryophyllata EO | n.r. | >20 nm | n.r. | Catheter Coating | [124] | |
| Candida krusei | Core-Shell MNPs Functionalized with Anethum Graveolens EO | limonene, carvone, and α-phellandrene | n.r. | n.r. | Antimicrobial Coatings | [123] |
| Core-Shell MNPs Functionalized with Eugenia Carryophyllata EO | n.r. | >20 nm | n.r. | Catheter Coating | [124] | |
| Candida parapsilosis | MNPs Functionalized with Cinnamomum Verum EO | n.r. | 9–10 nm | n.r. | Gastrostomy Tube Coating | [104] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mihai, A.D.; Chircov, C.; Grumezescu, A.M.; Holban, A.M. Magnetite Nanoparticles and Essential Oils Systems for Advanced Antibacterial Therapies. Int. J. Mol. Sci. 2020, 21, 7355. https://doi.org/10.3390/ijms21197355
Mihai AD, Chircov C, Grumezescu AM, Holban AM. Magnetite Nanoparticles and Essential Oils Systems for Advanced Antibacterial Therapies. International Journal of Molecular Sciences. 2020; 21(19):7355. https://doi.org/10.3390/ijms21197355
Chicago/Turabian StyleMihai, Antonio David, Cristina Chircov, Alexandru Mihai Grumezescu, and Alina Maria Holban. 2020. "Magnetite Nanoparticles and Essential Oils Systems for Advanced Antibacterial Therapies" International Journal of Molecular Sciences 21, no. 19: 7355. https://doi.org/10.3390/ijms21197355
APA StyleMihai, A. D., Chircov, C., Grumezescu, A. M., & Holban, A. M. (2020). Magnetite Nanoparticles and Essential Oils Systems for Advanced Antibacterial Therapies. International Journal of Molecular Sciences, 21(19), 7355. https://doi.org/10.3390/ijms21197355








