Poly(3-Hydroxybutyrate)-Based Nanoparticles for Sorafenib and Doxorubicin Anticancer Drug Delivery
Abstract
1. Introduction
2. Results and Discussion
2.1. Polymer Synthesis and Preliminary Nanoparticle Preparation
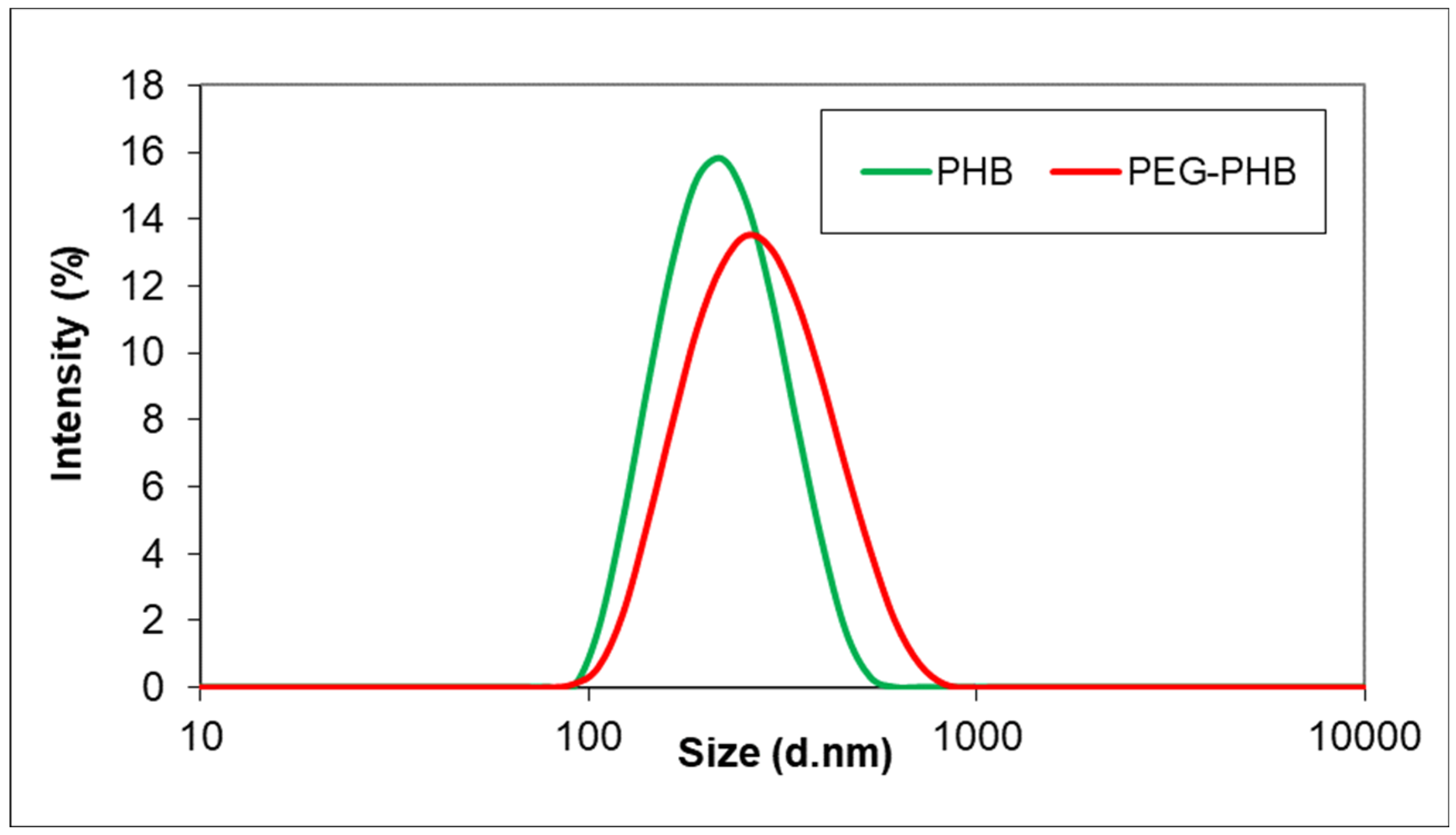
2.2. Morphology and Size
2.3. Yield, Drug Loading and Encapsulation Efficiency
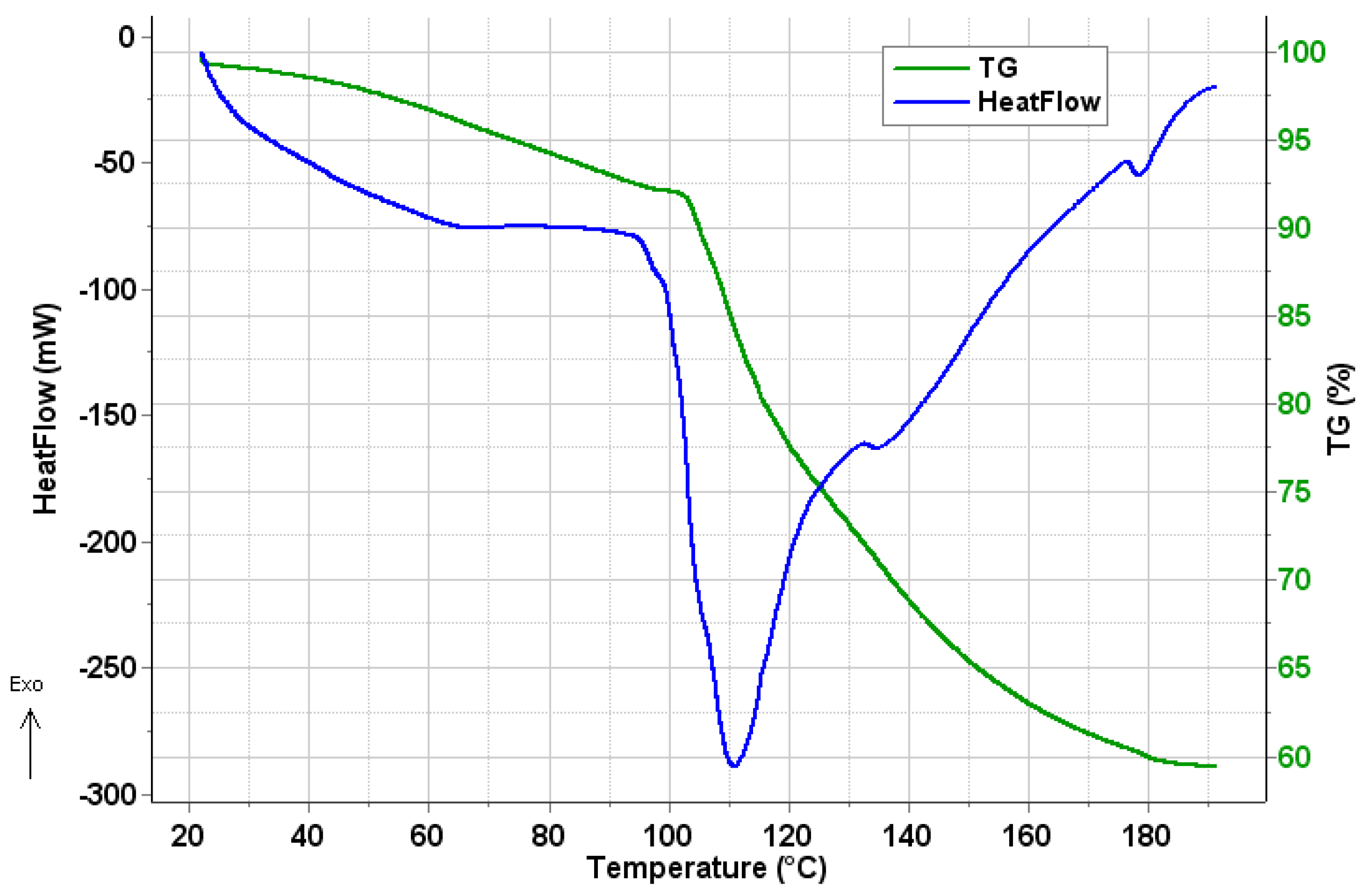
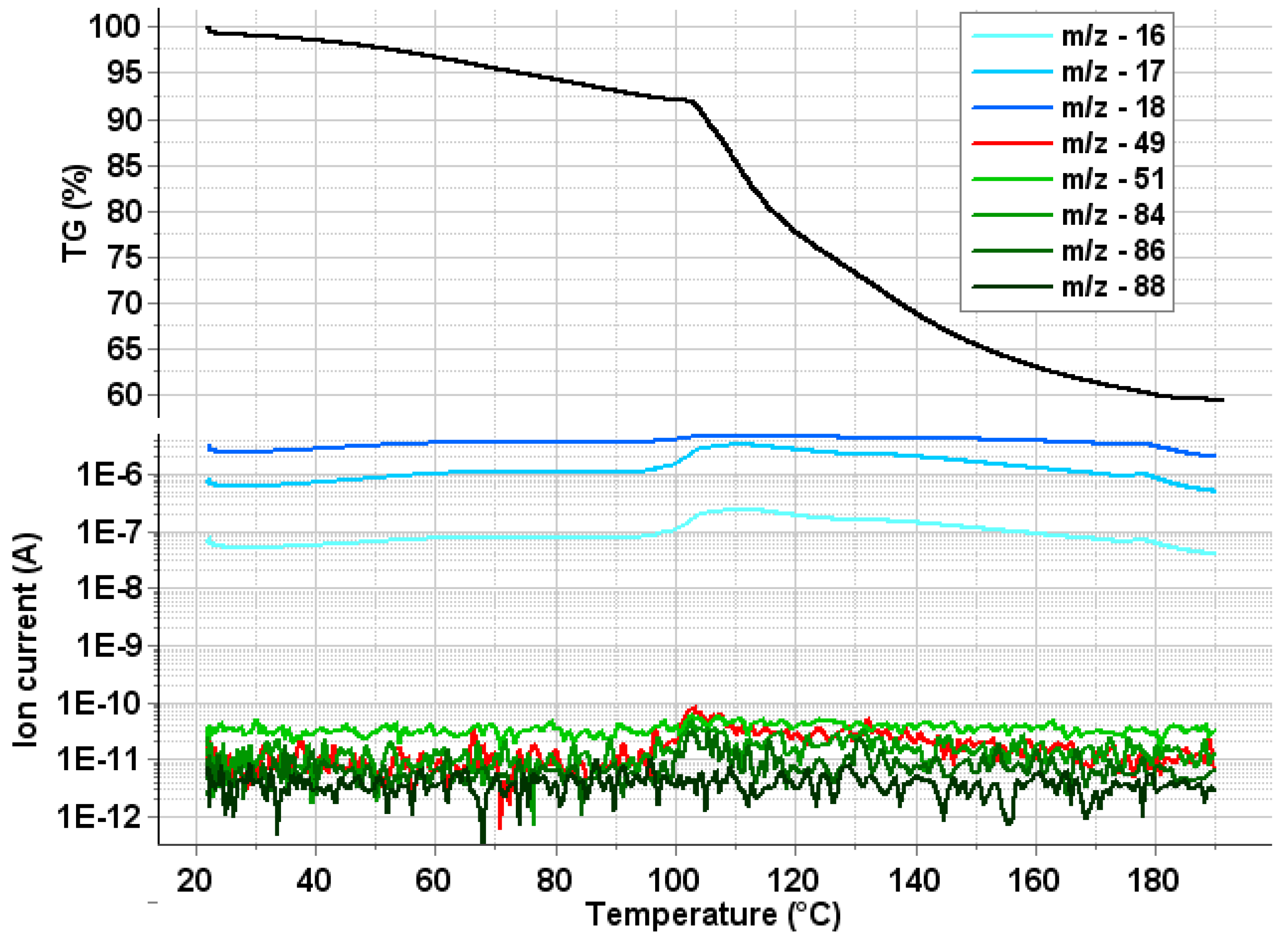
2.4. Thermal Analysis
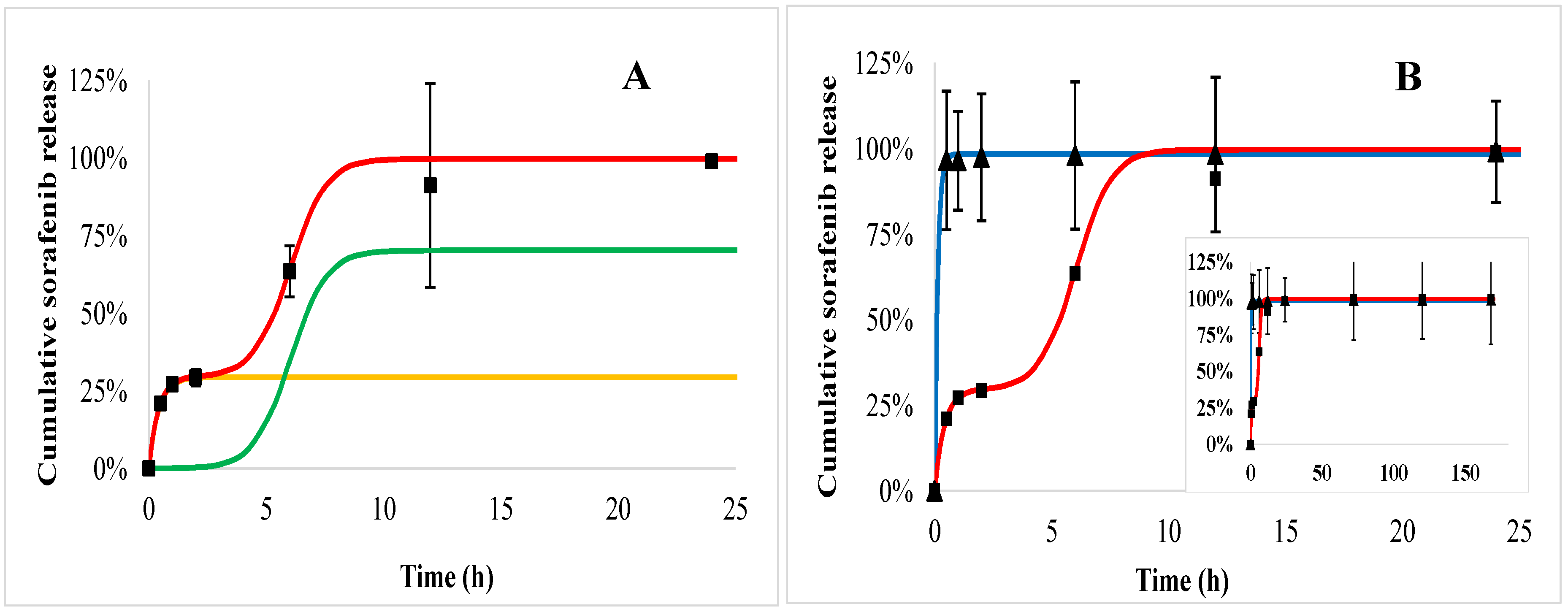
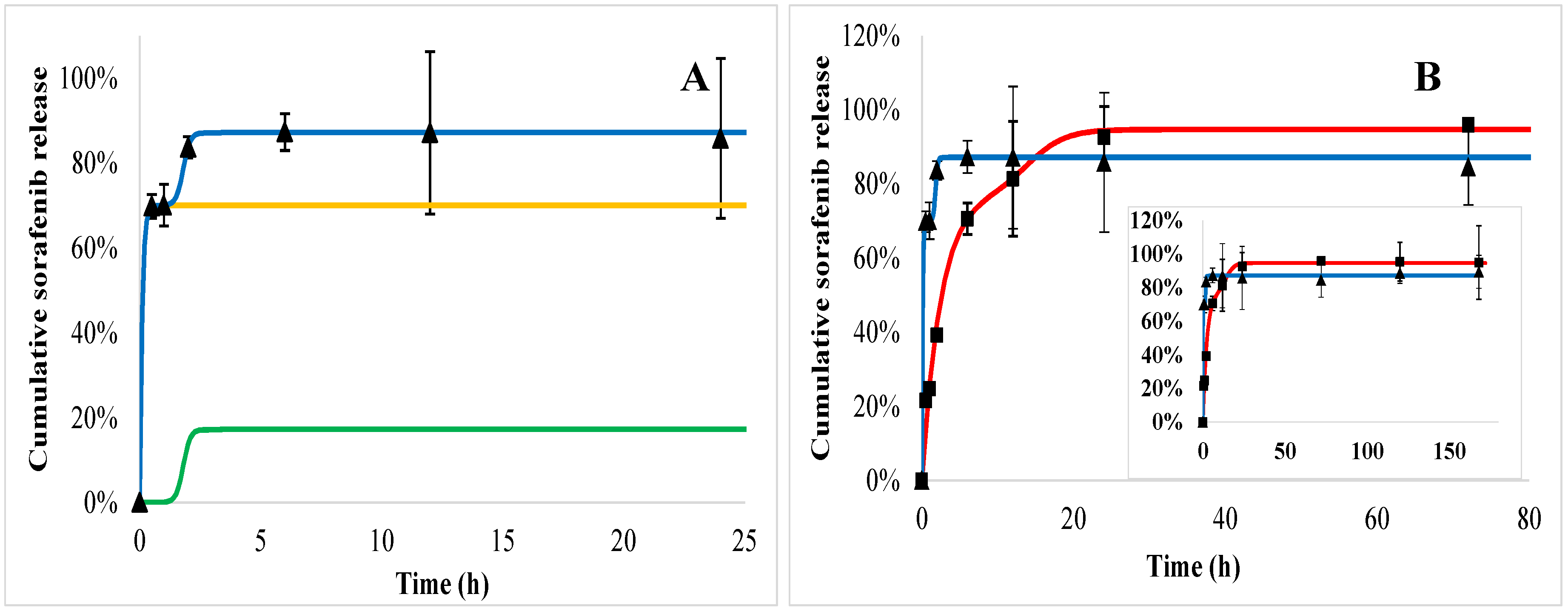
2.5. In Vitro Drug Release Kinetics
2.6. In Vitro Drug Release Kinetics
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Encapsulating Polymers
3.3. Preparation of Nanoparticles by Nanoprecipitation
3.4. Preparation of Nanoparticles by Emulsion Solvent Evaporation
3.5. Investigation of Nanoparticles
3.5.1. Morphology and Particle Size
3.5.2. Yield, Drug Loading and Encapsulation Efficiency
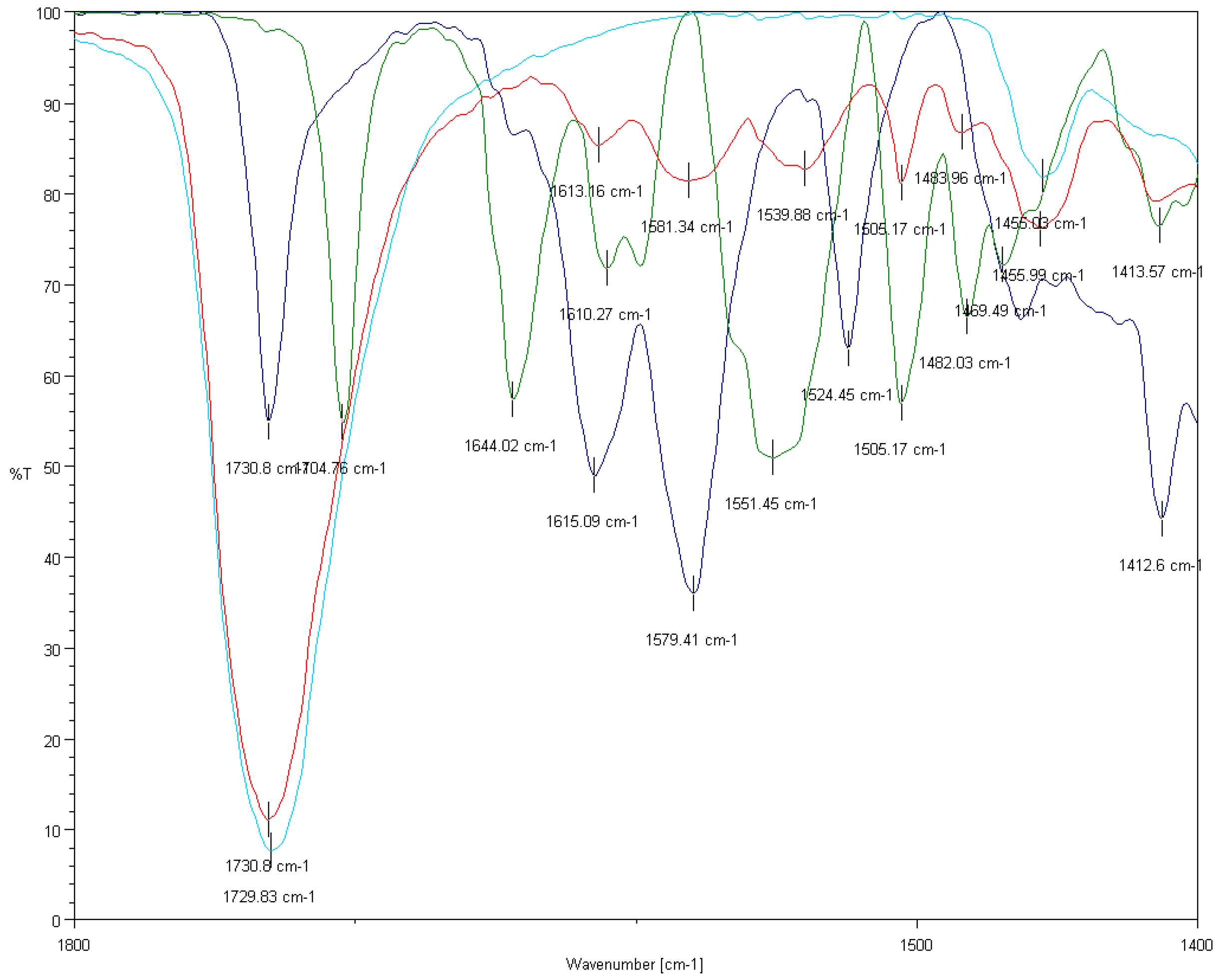
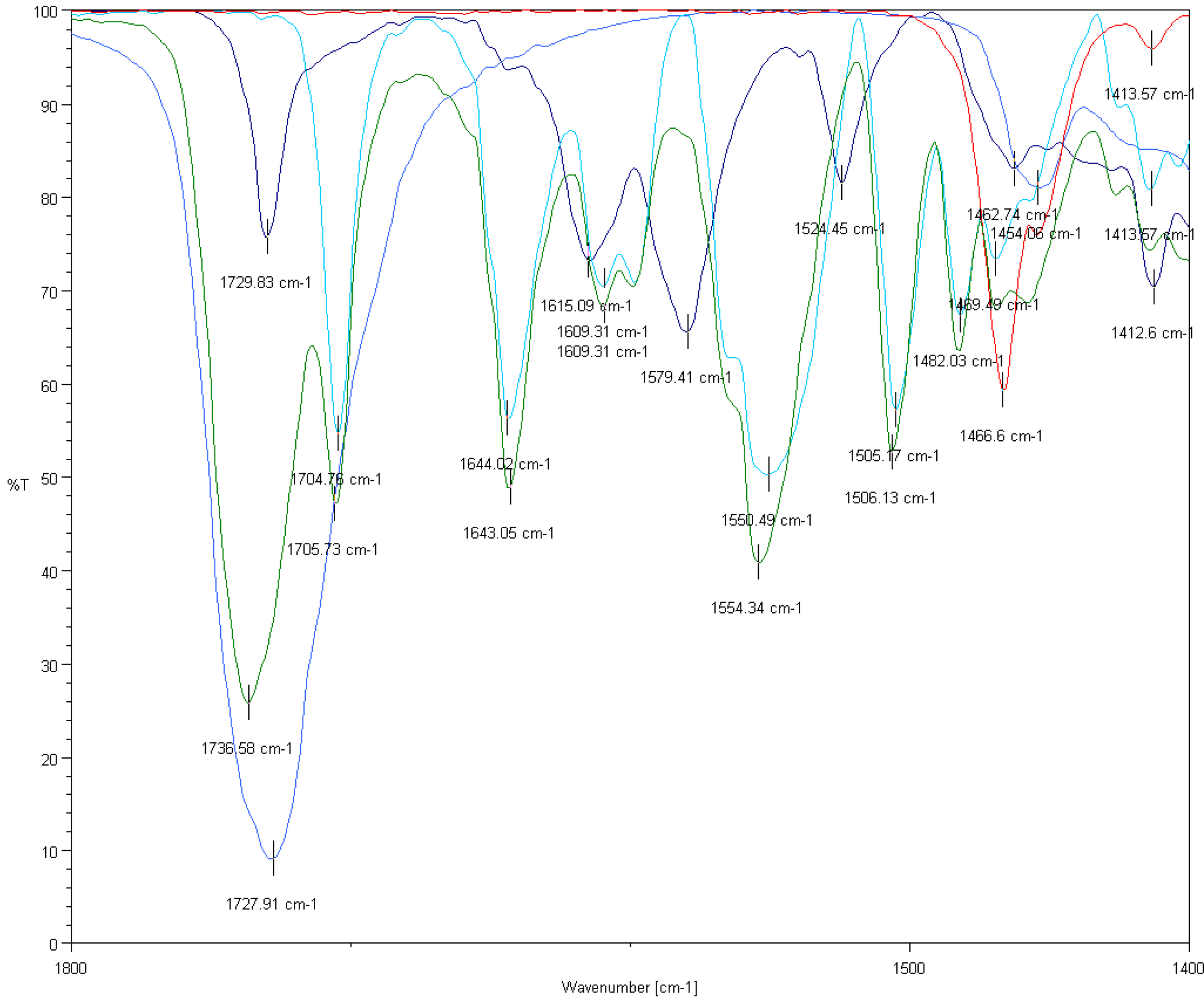
3.5.3. FTIR Analysis
3.5.4. TG–DSC–MSEGA Analysis
3.5.5. PEGylation of Nanoparticles
3.5.6. In Vitro Drug Release Study
3.5.7. Release Kinetics
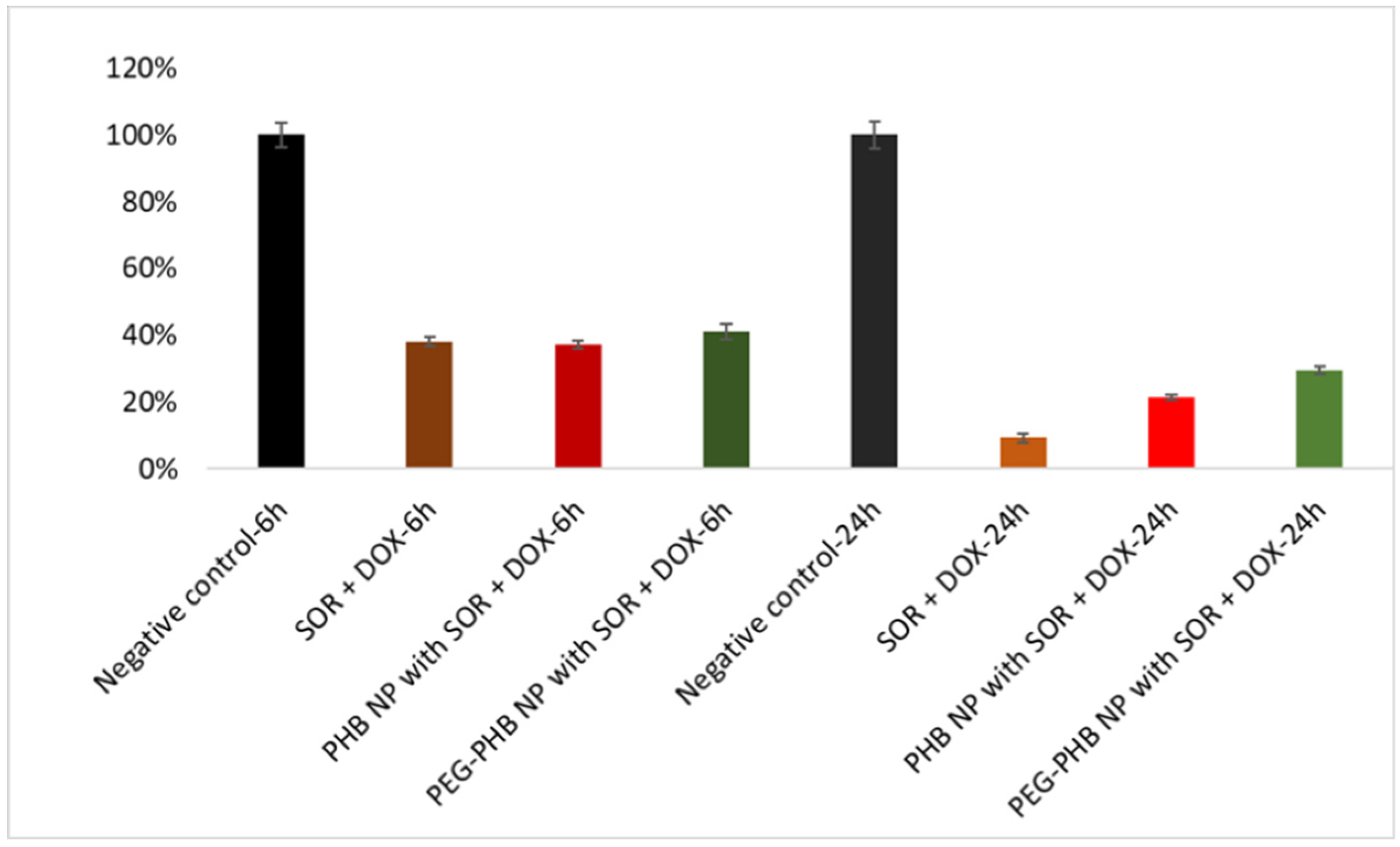
2.5.8. Cytotoxicity Assay
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, Z.; Suo, C.; Mao, X.; Jiang, Y.; Jin, L.; Zhang, T.; Chen, X. Global incidence trends in primary liver cancer by age at diagnosis, sex, region, and etiology, 1990–2017. Cancer 2020, 126, 2267–2278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Sun, T.; Liang, L.; Wu, T.; Wang, Q. Drug promiscuity of P-glycoprotein and its mechanism of interaction with paclitaxel and doxorubicin. Soft Matter 2014, 10, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.-C.; Hsieh, M.-L.; Chuang, P.-H.; Hsieh, V.C.-R. Cost-Effectiveness of Sorafenib Monotherapy and Selected Combination Therapy with Sorafenib in Patients with Advanced Hepatocellular Carcinoma. Value Health Reg. Issues 2018, 15, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Ockenfels, H.M.; Brockmeyer, N.H.; Hengge, U.; Goos, M. Cutaneous angiosarcoma: A novel therapy with liposomal doxorubicin? J. Eur. Acad. Dermatol. Venereol. 1996, 6, 71–75. [Google Scholar] [CrossRef]
- Chawla, S.P.; Bruckner, H.; Morse, M.A.; Assudani, N.; Hall, F.L.; Gordon, E.M. A Phase I-II Study Using Rexin-G Tumor-Targeted Retrovector Encoding a Dominant-Negative Cyclin G1 Inhibitor for Advanced Pancreatic Cancer. Mol. Ther.-Oncolytics 2019, 12, 56–67. [Google Scholar] [CrossRef]
- Miele, E.; Spinelli, G.P.; Miele, E.; Tomao, F.; Tomao, S. Albumin-bound formulation of paclitaxel (Abraxane ABI-007) in the treatment of breast cancer. Int. J. Nanomed. 2009, 4, 99–105. [Google Scholar]
- Berges, R. Eligard (R): Pharmacokinetics, Effect on Testosterone and PSA Levels and Tolerability. Eur. Urol. Suppl. 2005, 4, 20–25. [Google Scholar] [CrossRef]
- Calzoni, E.; Cesaretti, A.; Polchi, A.; Di Michele, A.; Tancini, B.; Emiliani, C. Biocompatible Polymer Nanoparticles for Drug Delivery Applications in Cancer and Neurodegenerative Disorder Therapies. J. Funct. Biomater. 2019, 10, 4. [Google Scholar] [CrossRef]
- Feczkó, T.; Piiper, A.; Pleli, T.; Schmithals, C.; Denk, D.; Hehlgans, S.; Rödel, F.; Vogl, T.J.; Wacker, M.G. Theranostic Sorafenib-Loaded Polymeric Nanocarriers Manufactured by Enhanced Gadolinium Conjugation Techniques. Pharmaceutics 2019, 11, 489. [Google Scholar] [CrossRef]
- Espinoza, S.; Patil, I.; San Martín-Martínez, E.; Martinez, M.; Casañas Pimentel, R.; Pradum, P.; Ige, P.; Pimentel, R.; Ige, P. Poly-e-caprolactone (PCL), a promising polymer for pharmaceutical and biomedical applications: Focus on nanomedicine in cancer. Int. J. Polym. Mater. Polym. Biomater. 2019, 69, 85–126. [Google Scholar] [CrossRef]
- Babos, G.; Biró, E.; Meiczinger, M.; Feczkó, T. Dual Drug Delivery of Sorafenib and Doxorubicin from PLGA and PEG-PLGA Polymeric Nanoparticles. Polymers 2018, 10, 895. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Guo, J.; Feng, Z.; Liu, X.; Wang, C.; Huang, P.; Zhang, J.; Deng, L.; Wang, W.; Dong, A.; et al. An injectable thermosensitive hydrogel self-supported by nanoparticles of PEGylated amino-modified PCL for enhanced local tumor chemotherapy. Soft Matter 2020, 16, 5750–5758. [Google Scholar] [CrossRef] [PubMed]
- Dozier, J.K.; Distefano, M.D. Site-specific pegylation of therapeutic proteins. Int. J. Mol. Sci. 2015, 16, 25831–25864. [Google Scholar] [CrossRef] [PubMed]
- Cherpinski, A.; Gozutok, M.; Sasmazel, H.T.; Torres-Giner, S.; Lagaron, J.M. Electrospun Oxygen Scavenging Films of Poly(3-hydroxybutyrate) Containing Palladium Nanoparticles for Active Packaging Applications. Nanomaterials 2018, 8, 469. [Google Scholar] [CrossRef]
- Lin, X.; Yin, M.; Liu, Y.; Li, L.; Ren, X.; Sun, Y.; Huang, T. Biodegradable polyhydroxybutyrate/poly-ɛ-caprolactone fibrous membranes modified by silica composite hydrol for super hydrophobic and outstanding antibacterial application. J. Ind. Eng. Chem. 2018, 63, 303–311. [Google Scholar] [CrossRef]
- Panaitescu, D.M.; Ionita, E.R.; Nicolae, C.A.; Gabor, A.R.; Ionita, M.D.; Trusca, R.; Lixandru, B.E.; Codita, I.; Dinescu, G. Poly(3-hydroxybutyrate) modified by nanocellulose and plasma treatment for packaging applications. Polymers 2018, 10, 1249. [Google Scholar] [CrossRef]
- Kai, D.; Chong, H.M.; Chow, L.P.; Jiang, L.; Lin, Q.; Zhang, K.; Zhang, H.; Zhang, Z.; Loh, X.J. Strong and biocompatible lignin/poly (3-hydroxybutyrate) composite nanofibers. Compos. Sci. Technol. 2018, 158, 26–33. [Google Scholar] [CrossRef]
- Degli Esposti, M.; Chiellini, F.; Bondioli, F.; Morselli, D.; Fabbri, P. Highly porous PHB-based bioactive scaffolds for bone tissue engineering by in situ synthesis of hydroxyapatite. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 100, 286–296. [Google Scholar] [CrossRef]
- Krucińska, I.; Żywicka, B.; Komisarczyk, A.; Szymonowicz, M.; Kowalska, S.; Zaczyńska, E.; Struszczyk, M.; Czarny, A.; Jadczyk, P.; Umińska-Wasiluk, B.; et al. Biological properties of low-toxicity PLGA and PLGA/PHB fibrous nanocomposite implants for osseous tissue regeneration. Part I: Evaluation of potential biotoxicity. Molecules 2017, 22, 2092. [Google Scholar] [CrossRef]
- Zhao, J.; Weng, G.; Li, J.; Zhu, J.; Zhao, J. Polyester-based nanoparticles for nucleic acid delivery. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 92, 983–994. [Google Scholar]
- Abdelwahab, M.; Salahuddin, N.; Gaber, M.; Mousa, M. Poly(3-hydroxybutyrate)/polyethylene glycol-NiO nanocomposite for NOR delivery: Antibacterial activity and cytotoxic effect against cancer cell lines. Int. J. Biol. Macromol. 2018, 114, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Barouti, G.; Jaffredo, C.G.; Guillaume, S.M. Advances in drug delivery systems based on synthetic poly(hydroxybutyrate) (co)polymers. Prog. Polym. Sci. 2017, 73, 1–31. [Google Scholar] [CrossRef]
- Zawidlak-Wegrzyńska, B.; Kawalec, M.; Bosek, I.; Łuczyk-Juzwa, M.; Adamus, G.; Rusin, A.; Filipczak, P.; Głowala-Kosińska, M.; Wolańska, K.; Krawczyk, Z.; et al. Synthesis and antiproliferative properties of ibuprofen-oligo(3-hydroxybutyrate) conjugates. Eur. J. Med. Chem. 2010, 45, 1833–1842. [Google Scholar] [CrossRef] [PubMed]
- Kök, F.; Hasirci, V. Polyhydroxybutyrate and its copolymers: Applications in the medical field. In Tissue Engineering and Novel Delivery Systems, 1st ed.; Arcel Dekker: New York, NY, USA, 2004; pp. 543–561. [Google Scholar]
- Loh, X.J.; Ong, S.J.; Tung, Y.T.; Choo, H.T. Incorporation of Poly[(R)-3-hydroxybutyrate] into Cationic Copolymers Based on Poly(2-(dimethylamino)ethyl methacrylate) to Improve Gene Delivery. Macromol. Biosci. 2013, 13, 1092–1099. [Google Scholar] [CrossRef]
- Silva, T.G.D.; Gobbi, V.G.; Teixeira, B.N.; Mendonça, T.D.S.; Cubiça, T.B.; Aquino, L.F.; Silva, J.A.d.N.; Thiré, R.M.d.S.M.; Mendonça, R.H. Mass Variation Rate, in Acidic Environment, of Polyhydroxybutyrate and Chitosan matrices with Potential Application as Controlled Drug Delivery System. Mater. Res. 2019, 22. [Google Scholar] [CrossRef]
- Radu, I.C.; Hudita, A.; Zaharia, C.; Stanescu, P.O.; Vasile, E.; Iovu, H.; Stan, M.; Ginghina, O.; Galateanu, B.; Costache, M.; et al. Poly(HydroxyButyrate-co-HydroxyValerate) (PHBHV) nanocarriers for silymarin release as adjuvant therapy in colo-rectal cancer. Front. Pharmacol. 2017, 8, 508. [Google Scholar] [CrossRef]
- Radu, I.C.; Hudita, A.; Zaharia, C.; Galateanu, B.; Iovu, H.; Tanasa, E.; Nitu, S.G.; Ginghina, O.; Negrei, C.; Tsatsakis, A.; et al. Poly(3-hydroxybutyrate-CO-3-hydroxyvalerate) PHBHV biocompatible nanocarriers for 5-FU delivery targeting colorectal cancer. Drug Deliv. 2019, 26, 318–327. [Google Scholar] [CrossRef]
- Kundrat, V.; Cernekova, N.; Kovalcik, A.; Enev, V.; Marova, I. Drug release kinetics of electrospun PHB meshes. Materials 2019, 12, 1924. [Google Scholar] [CrossRef]
- Perveen, K.; Masood, F.; Hameed, A. Preparation, characterization and evaluation of antibacterial properties of epirubicin loaded PHB and PHBV nanoparticles. Int. J. Biol. Macromol. 2020, 144, 259–266. [Google Scholar] [CrossRef]
- Bokrova, J.; Marova, I.; Matouskova, P.; Pavelkova, R. Fabrication of novel PHB-liposome nanoparticles and study of their toxicity in vitro. J. Nanoparticle Res. 2019, 21, 49. [Google Scholar] [CrossRef]
- Xu, Y.; Kim, C.-S.; Saylor, D.M.; Koo, D. Polymer degradation and drug delivery in PLGA-based drug-polymer applications: A review of experiments and theories. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 1692–1716. [Google Scholar] [CrossRef] [PubMed]
- Wacker, M. Nanomedicines—A Scientific Toy or an Emerging Market? In Polymer Nanoparticles for Nanomedicines; Springer: Basel, Switzerland, 2016; pp. 591–614. ISBN 978-3-319-41419-5. [Google Scholar]
- Das, G.; Nicastri, A.; Coluccio, M.L.; Gentile, F.; Candeloro, P.; Cojoc, G.; Liberale, C.; De Angelis, F.; Di Fabrizio, E. FT-IR, Raman, RRS measurements and DFT calculation for doxorubicin. Microsc. Res. Tech. 2010, 73, 991–995. [Google Scholar] [CrossRef] [PubMed]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies, Tables and Charts, 3rd ed.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2001; ISBN 0-471-85298-8. [Google Scholar]
- Elsayed, M.M.; Mostafa, M.E.; Alaaeldin, E.; Sarhan, H.A.; Shaykoon, M.S.; Allam, S.; Ahmed, A.R.; Elsadek, B.E. Design And Characterisation Of Novel Sorafenib-Loaded Carbon Nanotubes With Distinct Tumour-Suppressive Activity In Hepatocellular Carcinoma. Int. J. Nanomed. 2019, 14, 8445–8467. [Google Scholar] [CrossRef] [PubMed]
- Jakić, M.; Stipanelov Vrandečić, N.; Erceg, M. Thermal degradation of poly(3-hydroxybutyrate)/poly(ethylene oxide) blends: Thermogravimetric and kinetic analysis. Eur. Polym. J. 2016, 81, 376–385. [Google Scholar] [CrossRef]
- Vahabi, H.; Michely, L.; Moradkhani, G.; Akbari, V.; Cochez, M.; Vagner, C.; Renard, E.; Saeb, M.R.; Langlois, V. Thermal Stability and Flammability Behavior of Poly(3-hydroxybutyrate) (PHB) Based Composites. Materials 2019, 12, 2239. [Google Scholar] [CrossRef]
- Vogel, C.; Morita, S.; Sato, H.; Noda, I.; Ozaki, Y.; Siesler, H.W. Thermal Degradation of Poly(3-hydroxybutyrate) and Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) in Nitrogen and Oxygen Studied by Thermogravimetric--Fourier Transform Infrared Spectroscopy. Appl. Spectrosc. 2007, 61, 755–764. [Google Scholar] [CrossRef]
- Soares, P.I.P.; Sousa, A.I.; Silva, J.C.; Ferreira, I.M.M.; Novo, C.M.M.; Borges, J.P. Chitosan-based nanoparticles as drug delivery systems for doxorubicin: Optimization and modelling. Carbohydr. Polym. 2016, 147, 304–312. [Google Scholar] [CrossRef]
- Tod, M.; Mir, O.; Bancelin, N.; Coriat, R.; Thomas-Schoemann, A.; Taieb, F.; Boudou-Rouquette, P.; Ropert, S.; Michels, J.; Abbas, H.; et al. Functional and clinical evidence of the influence of sorafenib binding to albumin on sorafenib disposition in adult cancer patients. Pharm. Res. 2011, 28, 3199–3207. [Google Scholar] [CrossRef]
- Lucero-Acuña, A.; Guzmán, R. Nanoparticle encapsulation and controlled release of a hydrophobic kinase inhibitor: Three stage mathematical modeling and parametric analysis. Int. J. Pharm. 2015, 494, 249–257. [Google Scholar] [CrossRef]
- Kawalec, M.; Coulembier, O.; Gerbaux, P.; Sobota, M.; De Winter, J.; Dubois, P.; Kowalczuk, M.; Kurcok, P. Traces do matter—Purity of 4-methyl-2-oxetanone and its effect on anionic ring-opening polymerization as evidenced by phosphazene superbase catalysis. React. Funct. Polym. 2012, 72, 509–520. [Google Scholar] [CrossRef]
- Lucero-Acuña, A.; Gutiérrez-Valenzuela, C.; Esquivel, R.; Guzmán-Zamudio, R. Mathematical modeling and parametrical analysis of the temperature dependency of control drug release from biodegradable nanoparticles. RSC Adv. 2019, 9, 8728–8739. [Google Scholar] [CrossRef]
- Korsmeyer, R.W.; Gurny, R.; Doelker, E.; Buri, P.; Peppas, N.A. Mechanisms of solute release from porous hydrophilic polymers. Int. J. Pharm. 1983, 15, 25–35. [Google Scholar] [CrossRef]
- Ribeiro, A.R.; Leite, P.E.; Falagan-Lotsch, P.; Benetti, F.; Micheletti, C.; Budtz, H.C.; Jacobsen, N.R.; Lisboa-Filho, P.N.; Rocha, L.A.; Kühnel, D.; et al. Challenges on the toxicological predictions of engineered nanoparticles. NanoImpact 2017, 8, 59–72. [Google Scholar] [CrossRef]



| PHB | PEGylated PHB | |
|---|---|---|
| Yield (% w/w) | 66.9 ± 1.3 | 55.4 ± 1.4 |
| EE (DOX) (%, w/w) | 77 ± 3.7 | 64 ± 4.1 |
| EE (SOR) (%, w/w) | 84 ± 2.5 | 70 ± 1.2 |
| DOX loading (%, w/w) | 2.6 | 2.6 |
| SOR loading (%, w/w) | 8.4 | 7.7 |
| Ѳb | kb (1/h) | Ѳr | kr (1/h) | tmax (h) | kd | n | |
|---|---|---|---|---|---|---|---|
| DOX_PHB in acid | 0.22 | 1.1 | 0.48 | 2.8 | 5.4 | 0.132 | 0.17 |
| DOX_PHB in plasma | 0.38 | 4.36 | 0.16 | 4.5 | 1.7 | 0.022 | 0.38 |
| DOX_PEG_PHB in acid | 0.26 | 9.6 | 0.35 | 4.4 | 2.1 | 0.137 | 0.19 |
| DOX PEG_PHB in plasma | 0.40 | 5.5 | 0.16 | >5.0 | 1.4 | 0.019 | 0.33 |
| SOR_PHB in acid | 0.29 | 2.5 | 0.70 | >1.3 | 6.0 | - | - |
| SOR_PHB in plasma | 0.98 | 7.9 | - | - | - | - | - |
| SOR_PEG_PHB in acid | 0.76 | 0.4 | 0.18 | 0.4 | 14.0 | - | - |
| SOR_PEG_PHB in plasma | 0.70 | >10.0 | 0.17 | 6.8 | 1.8 | - | - |
| Time (min) | Tetrafluoroacetic Acid in H2O (%) | Methanol (%) |
|---|---|---|
| 0.0 | 70.0 | 30.0 |
| 1.0 | 70.0 | 30.0 |
| 5.00 | 0.0 | 100.0 |
| 6.00 | 0.0 | 100.0 |
| 10.00 | 70.0 | 30.0 |
| 2.00 | 70.0 | 30.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babos, G.; Rydz, J.; Kawalec, M.; Klim, M.; Fodor-Kardos, A.; Trif, L.; Feczkó, T. Poly(3-Hydroxybutyrate)-Based Nanoparticles for Sorafenib and Doxorubicin Anticancer Drug Delivery. Int. J. Mol. Sci. 2020, 21, 7312. https://doi.org/10.3390/ijms21197312
Babos G, Rydz J, Kawalec M, Klim M, Fodor-Kardos A, Trif L, Feczkó T. Poly(3-Hydroxybutyrate)-Based Nanoparticles for Sorafenib and Doxorubicin Anticancer Drug Delivery. International Journal of Molecular Sciences. 2020; 21(19):7312. https://doi.org/10.3390/ijms21197312
Chicago/Turabian StyleBabos, György, Joanna Rydz, Michal Kawalec, Magdalena Klim, Andrea Fodor-Kardos, László Trif, and Tivadar Feczkó. 2020. "Poly(3-Hydroxybutyrate)-Based Nanoparticles for Sorafenib and Doxorubicin Anticancer Drug Delivery" International Journal of Molecular Sciences 21, no. 19: 7312. https://doi.org/10.3390/ijms21197312
APA StyleBabos, G., Rydz, J., Kawalec, M., Klim, M., Fodor-Kardos, A., Trif, L., & Feczkó, T. (2020). Poly(3-Hydroxybutyrate)-Based Nanoparticles for Sorafenib and Doxorubicin Anticancer Drug Delivery. International Journal of Molecular Sciences, 21(19), 7312. https://doi.org/10.3390/ijms21197312








