FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy
Abstract
1. Introduction
2. The FLASH Effect
2.1. Normal Tissue Sparing
2.2. Tumor Control
3. Mechanisms Contributing to the FLASH Effect
3.1. Oxygen Depletion
3.2. ROS
3.3. Immune Response
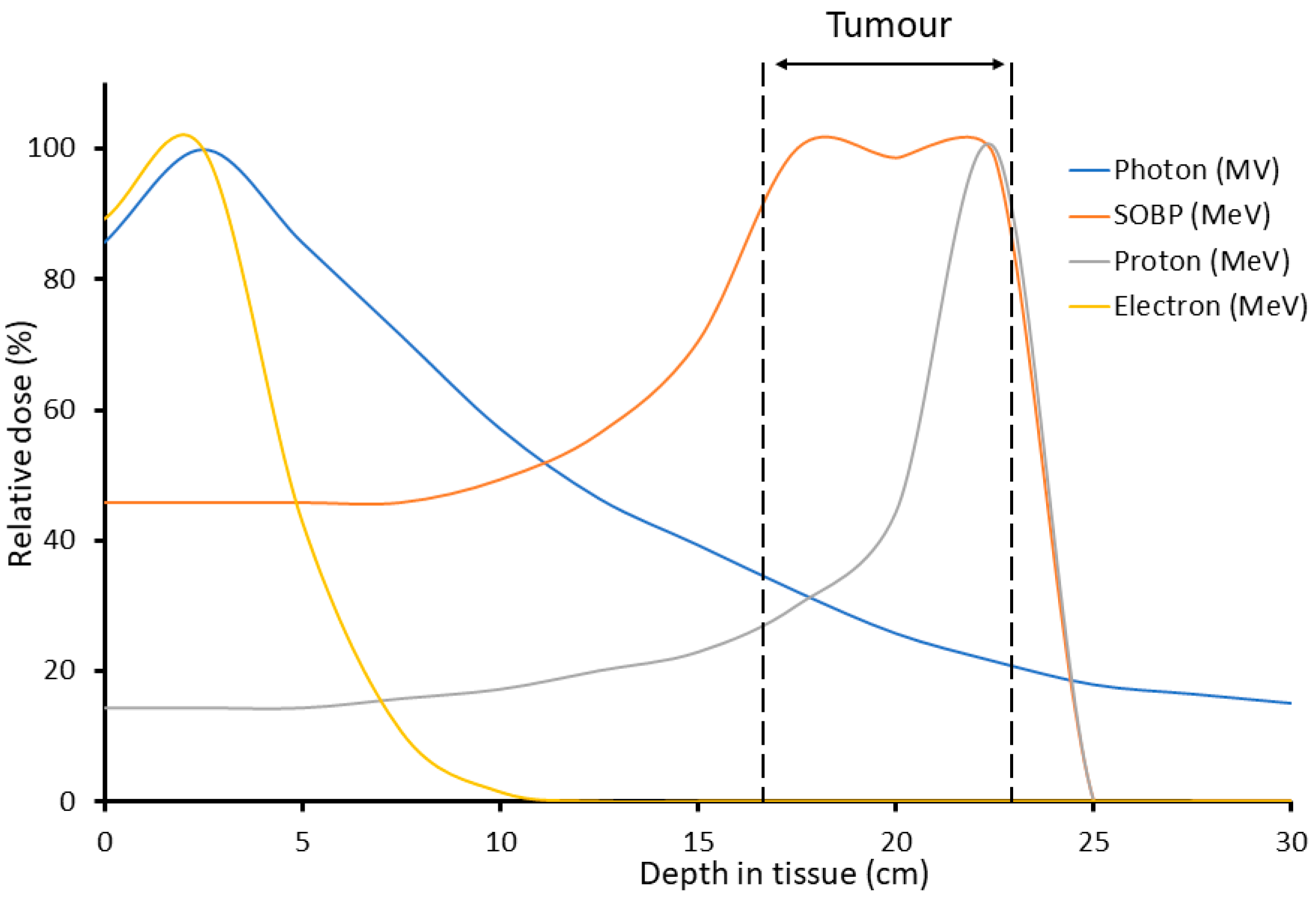
4. The Potential for FLASH Proton-Beam Therapy
Studies Investigating FLASH Protons
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| DSB | DNA double strand break |
| LET | Linear energy transfer |
| PBT | Proton-beam therapy |
| ROS | Reactive oxygen species |
| TGF-β | Transforming growth factor beta |
References
- Moding, E.J.; Kastan, M.B.; Kirsch, D.G. Strategies for optimizing the response of cancer and normal tissues to radiation. Nat. Rev. Drug. Discov. 2013, 12, 526–542. [Google Scholar] [CrossRef] [PubMed]
- Berkey, F.J. Managing the adverse effects of radiation therapy. Am. Fam. Physician 2010, 82, 381–388, 394. [Google Scholar]
- Siddiqui, F.; Movsas, B. Management of Radiation Toxicity in Head and Neck Cancers. Semin. Radiat. Oncol. 2017, 27, 340–349. [Google Scholar] [CrossRef]
- Kurup, A.; Pasternak, J.; Taylor, R.; Murgatroyd, L.; Ettlinger, O.; Shields, W.; Nevay, L.; Gruber, S.; Pozimski, J.; Lau, H.T.; et al. Simulation of a radiobiology facility for the Centre for the Clinical Application of Particles. Phys. Med. 2019, 65, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Kruger, S.; Ilmer, M.; Kobold, S.; Cadilha, B.L.; Endres, S.; Ormanns, S.; Schuebbe, G.; Renz, B.W.; D’Haese, J.G.; Schloesser, H.; et al. Advances in cancer immunotherapy 2019 - latest trends. J. Exp. Clin. Cancer Res. 2019, 38, 268. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A. IL-2: The first effective immunotherapy for human cancer. J. Immunol. 2014, 192, 5451–5458. [Google Scholar] [CrossRef]
- Al-Lazikani, B.; Banerji, U.; Workman, P. Combinatorial drug therapy for cancer in the post-genomic era. Nat. Biotechnol. 2012, 30, 679–692. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Meziani, L.; Yakkala, C.; Vozenin, M.C. Expanding the therapeutic index of radiation therapy by normal tissue protection. Br. J. Radiol. 2019, 92, 20180008. [Google Scholar] [CrossRef]
- Garibaldi, C.; Jereczek-Fossa, B.A.; Marvaso, G.; Dicuonzo, S.; Rojas, D.P.; Cattani, F.; Starzynska, A.; Ciardo, D.; Surgo, A.; Leonardi, M.C.; et al. Recent advances in radiation oncology. Ecancermedicalscience 2017, 11, 785. [Google Scholar] [CrossRef]
- Beaton, L.; Bandula, S.; Gaze, M.N.; Sharma, R.A. How rapid advances in imaging are defining the future of precision radiation oncology. Br. J. Cancer 2019, 120, 779–790. [Google Scholar] [CrossRef]
- Vitti, E.T.; Parsons, J.L. The Radiobiological Effects of Proton Beam Therapy: Impact on DNA Damage and Repair. Cancers 2019, 11, 946. [Google Scholar] [CrossRef] [PubMed]
- Favaudon, V.; Caplier, L.; Monceau, V.; Pouzoulet, F.; Sayarath, M.; Fouillade, C.; Poupon, M.F.; Brito, I.; Hupe, P.; Bourhis, J.; et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci. Transl. Med. 2014, 6, 245ra293. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Petersson, K.; Jaccard, M.; Boivin, G.; Germond, J.F.; Petit, B.; Doenlen, R.; Favaudon, V.; Bochud, F.; Bailat, C.; et al. Irradiation in a flash: Unique sparing of memory in mice after whole brain irradiation with dose rates above 100Gy/s. Radiother. Oncol. 2017, 124, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Vozenin, M.C.; De Fornel, P.; Petersson, K.; Favaudon, V.; Jaccard, M.; Germond, J.F.; Petit, B.; Burki, M.; Ferrand, G.; Patin, D.; et al. The Advantage of FLASH Radiotherapy Confirmed in Mini-pig and Cat-cancer Patients. Clin. Cancer Res. 2019, 25, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Bourhis, J.; Sozzi, W.J.; Jorge, P.G.; Gaide, O.; Bailat, C.; Duclos, F.; Patin, D.; Ozsahin, M.; Bochud, F.; Germond, J.F.; et al. Treatment of a first patient with FLASH-radiotherapy. Radiother. Oncol. 2019, 139, 18–22. [Google Scholar] [CrossRef]
- Berry, R.J. Effects of radiation dose-rate from protracted, continuous irradiation to ultra-high dose-rates from pulsed accelerators. Br. Med. Bull. 1973, 29, 44–47. [Google Scholar] [CrossRef]
- Hornsey, S.; Alper, T. Unexpected dose-rate effect in the killing of mice by radiation. Nature 1966, 210, 212–213. [Google Scholar] [CrossRef]
- Levy, K.; Natarajan, S.; Wang, J.; Chow, S.; Eggold, J.; Loo, P.; Manjappa, R.; Lartey, F.; Schüler, E.; Skinner, L.; et al. FLASH irradiation enhances the therapeutic index of abdominal radiotherapy in mice. bioRxiv [Preprint] 2020, 1–35. [Google Scholar] [CrossRef]
- Loo, B.W.; Schuler, E.; Lartey, F.M.; Rafat, M.; King, G.J.; Trovati, S.; Koong, A.C.; Maxim, P.G. (P003) Delivery of Ultra-Rapid Flash Radiation Therapy and Demonstration of Normal Tissue Sparing After Abdominal Irradiation of Mice. Int. J. Radiat. Oncol. Biol. Phys. 2017, 98. [Google Scholar] [CrossRef]
- Simmons, D.A.; Lartey, F.M.; Schuler, E.; Rafat, M.; King, G.; Kim, A.; Ko, R.; Semaan, S.; Gonzalez, S.; Jenkins, M.; et al. Reduced cognitive deficits after FLASH irradiation of whole mouse brain are associated with less hippocampal dendritic spine loss and neuroinflammation. Radiother. Oncol. 2019, 139, 4–10. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Bouchet, A.; Jaccard, M.; Patin, D.; Serduc, R.; Aim, W.; Petersson, K.; Petit, B.; Bailat, C.; Bourhis, J.; et al. X-rays can trigger the FLASH effect: Ultra-high dose-rate synchrotron light source prevents normal brain injury after whole brain irradiation in mice. Radiother. Oncol. 2018, 129, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Alaghband, Y.; Cheeks, S.N.; Allen, B.D.; Montay-Gruel, P.; Doan, N.L.; Petit, B.; Jorge, P.G.; Giedzinski, E.; Acharya, M.M.; Vozenin, M.C.; et al. Neuroprotection of Radiosensitive Juvenile Mice by Ultra-High Dose Rate FLASH Irradiation. Cancers 2020, 12, 1671. [Google Scholar] [CrossRef]
- Montay-Gruel, P.; Acharya, M.M.; Petersson, K.; Alikhani, L.; Yakkala, C.; Allen, B.D.; Ollivier, J.; Petit, B.; Jorge, P.G.; Syage, A.R.; et al. Long-term neurocognitive benefits of FLASH radiotherapy driven by reduced reactive oxygen species. Proc. Natl. Acad. Sci. USA 2019, 116, 10943–10951. [Google Scholar] [CrossRef] [PubMed]
- Abel, E.; Girdhani, S.; Jackson, I.; Eley, J.; Katsis, A.; Marshall, A.; Rodriguez, A.; Senapati, S.; Bentzen, S.M.; Vujaskovic, Z.; et al. Characterization of Radiation-Induced Lung Fibrosis and Mode of Cell Death Using Single and Multi-Pulsed Proton Flash Irradiation. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, E652–E653. [Google Scholar] [CrossRef]
- Girdhani, S.; Abel, E.; Katsis, A.; Rodriquez, A.; Senapati, S.; KuVillanueva, A.; Jackson, I.; Eley, J.; Vujaskovic, Z.; Parry, R. Abstract LB-280: FLASH: A novel paradigm changing tumor irradiation platform that enhances therapeutic ratio by reducing normal tissue toxicity and activating immune pathways. Cancer Res. 2019, 79, LB-280. [Google Scholar] [CrossRef]
- Fouillade, C.; Curras-Alonso, S.; Giuranno, L.; Quelennec, E.; Heinrich, S.; Bonnet-Boissinot, S.; Beddok, A.; Leboucher, S.; Karakurt, H.U.; Bohec, M.; et al. FLASH Irradiation Spares Lung Progenitor Cells and Limits the Incidence of Radio-induced Senescence. Clin. Cancer Res. 2020, 26, 1497–1506. [Google Scholar] [CrossRef] [PubMed]
- Diffenderfer, E.S.; Verginadis, I.I.; Kim, M.M.; Shoniyozov, K.; Velalopoulou, A.; Goia, D.; Putt, M.; Hagan, S.; Avery, S.; Teo, K.; et al. Design, Implementation, and in Vivo Validation of a Novel Proton FLASH Radiation Therapy System. Int J. Radiat. Oncol. Biol. Phys. 2020, 106, 440–448. [Google Scholar] [CrossRef]
- Vozenin, M.C.; Hendry, J.H.; Limoli, C.L. Biological Benefits of Ultra-high Dose Rate FLASH Radiotherapy: Sleeping Beauty Awoken. Clin. Oncol. 2019, 31, 407–415. [Google Scholar] [CrossRef]
- Rama, N.; Saha, T.; Shukla, S.; Goda, C.; Milewski, D.; Mascia, A.E.; Vatner, R.E.; Sengupta, D.; Katsis, A.; Abel, E.; et al. Improved Tumor Control Through T-cell Infiltration Modulated by Ultra-High Dose Rate Proton FLASH Using a Clinical Pencil Beam Scanning Proton System. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, S164–S165. [Google Scholar] [CrossRef]
- Wilson, J.D.; Hammond, E.M.; Higgins, G.S.; Petersson, K. Ultra-High Dose Rate (FLASH) Radiotherapy: Silver Bullet or Fool’s Gold? Front. Oncol. 2019, 9, 1563. [Google Scholar] [CrossRef]
- Morgan, W.F.; Sowa, M.B. Effects of ionizing radiation in nonirradiated cells. Proc. Natl. Acad. Sci. USA 2005, 102, 14127–14128. [Google Scholar] [CrossRef] [PubMed]
- Santivasi, W.L.; Xia, F. Ionizing radiation-induced DNA damage, response, and repair. Antioxid. Redox. Signal. 2014, 21, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Spitz, D.R.; Buettner, G.R.; Petronek, M.S.; St-Aubin, J.J.; Flynn, R.T.; Waldron, T.J.; Limoli, C.L. An integrated physico-chemical approach for explaining the differential impact of FLASH versus conventional dose rate irradiation on cancer and normal tissue responses. Radiother. Oncol. 2019, 139, 23–27. [Google Scholar] [CrossRef] [PubMed]
- Grimes, D.R.; Partridge, M. A mechanistic investigation of the oxygen fixation hypothesis and oxygen enhancement ratio. Biomed. Phys. Eng. Express 2015, 1, 045209. [Google Scholar] [CrossRef]
- Antonovic, L.; Lindblom, E.; Dasu, A.; Bassler, N.; Furusawa, Y.; Toma-Dasu, I. Clinical oxygen enhancement ratio of tumors in carbon ion radiotherapy: The influence of local oxygenation changes. J. Radiat. Res. 2014, 55, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Bristow, R.G.; Hill, R.P. Hypoxia and metabolism. Hypoxia, DNA repair and genetic instability. Nat. Rev. Cancer 2008, 8, 180–192. [Google Scholar] [CrossRef]
- Bourhis, J.; Montay-Gruel, P.; Goncalves Jorge, P.; Bailat, C.; Petit, B.; Ollivier, J.; Jeanneret-Sozzi, W.; Ozsahin, M.; Bochud, F.; Moeckli, R.; et al. Clinical translation of FLASH radiotherapy: Why and how? Radiother. Oncol. 2019, 139, 11–17. [Google Scholar] [CrossRef]
- Pratx, G.; Kapp, D.S. A computational model of radiolytic oxygen depletion during FLASH irradiation and its effect on the oxygen enhancement ratio. Phys. Med. Biol. 2019, 64, 185005. [Google Scholar] [CrossRef]
- Dewey, D.L.; Boag, J.W. Modification of the oxygen effect when bacteria are given large pulses of radiation. Nature 1959, 183, 1450–1451. [Google Scholar] [CrossRef]
- Dewey, D.L. An oxygen-dependent X-ray dose-rate effect in Serratia marcescens. Radiat. Res. 1969, 38, 467–474. [Google Scholar] [CrossRef]
- Epp, E.R.; Weiss, H.; Santomasso, A. The oxygen effect in bacterial cells irradiated with high-intensity pulsed electrons. Radiat. Res. 1968, 34, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Phillips, T.L.; Worsnop, B.R. Ultra-high dose-rate effects in radiosensitive bacteria. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1969, 14, 573–575. [Google Scholar] [CrossRef] [PubMed]
- Hendry, J.H.; Moore, J.V.; Hodgson, B.W.; Keene, J.P. The constant low oxygen concentration in all the target cells for mouse tail radionecrosis. Radiat. Res. 1982, 92, 172–181. [Google Scholar] [CrossRef]
- Epp, E.R.; Weiss, H.; Djordjevic, B.; Santomasso, A. The radiosensitivity of cultured mammalian cells exposed to single high intensity pulses of electrons in various concentrations of oxygen. Radiat. Res. 1972, 52, 324–332. [Google Scholar] [CrossRef]
- Adrian, G.; Konradsson, E.; Lempart, M.; Back, S.; Ceberg, C.; Petersson, K. The FLASH effect depends on oxygen concentration. Br. J. Radiol. 2020, 93, 20190702. [Google Scholar] [CrossRef] [PubMed]
- Cygler, J.; Klassen, N.V.; Ross, C.K.; Bichay, T.J.; Raaphorst, G.P. The survival of aerobic and anoxic human glioma and melanoma cells after irradiation at ultrahigh and clinical dose rates. Radiat. Res. 1994, 140, 79–84. [Google Scholar] [CrossRef]
- Buonanno, M.; Grilj, V.; Brenner, D.J. Biological effects in normal cells exposed to FLASH dose rate protons. Radiother. Oncol. 2019, 139, 51–55. [Google Scholar] [CrossRef]
- Town, C.D. Radiobiology. Effect of high dose rates on survival of mammalian cells. Nature 1967, 215, 847–848. [Google Scholar] [CrossRef]
- McKeown, S.R. Defining normoxia, physoxia and hypoxia in tumours-implications for treatment response. Br. J. Radiol. 2014, 87, 20130676. [Google Scholar] [CrossRef]
- Petersson, K.; Adrian, G.; Butterworth, K.; McMahon, S.J. A Quantitative Analysis of the Role of Oxygen Tension in FLASH Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2020, 107, 539–547. [Google Scholar] [CrossRef]
- Vanpouille-Box, C.; Diamond, J.M.; Pilones, K.A.; Zavadil, J.; Babb, J.S.; Formenti, S.C.; Barcellos-Hoff, M.H.; Demaria, S. TGFbeta Is a Master Regulator of Radiation Therapy-Induced Antitumor Immunity. Cancer Res. 2015, 75, 2232–2242. [Google Scholar] [CrossRef] [PubMed]
- Arina, A.; Beckett, M.; Fernandez, C.; Zheng, W.; Pitroda, S.; Chmura, S.J.; Luke, J.J.; Forde, M.; Hou, Y.; Burnette, B.; et al. Tumor-reprogrammed resident T cells resist radiation to control tumors. Nat. Commun. 2019, 10, 3959. [Google Scholar] [CrossRef] [PubMed]
- Holmgaard, R.B.; Schaer, D.A.; Li, Y.; Castaneda, S.P.; Murphy, M.Y.; Xu, X.; Inigo, I.; Dobkin, J.; Manro, J.R.; Iversen, P.W.; et al. Targeting the TGFbeta pathway with galunisertib, a TGFbetaRI small molecule inhibitor, promotes anti-tumor immunity leading to durable, complete responses, as monotherapy and in combination with checkpoint blockade. J. Immunother. Cancer 2018, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Durante, M.; Brauer-Krisch, E.; Hill, M. Faster and safer? FLASH ultra-high dose rate in radiotherapy. Br. J. Radiol. 2018, 91, 20170628. [Google Scholar] [CrossRef]
- Schuler, E.; Trovati, S.; King, G.; Lartey, F.; Rafat, M.; Villegas, M.; Praxel, A.J.; Loo, B.W., Jr.; Maxim, P.G. Experimental Platform for Ultra-high Dose Rate FLASH Irradiation of Small Animals Using a Clinical Linear Accelerator. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 195–203. [Google Scholar] [CrossRef]
- Lempart, M.; Blad, B.; Adrian, G.; Back, S.; Knoos, T.; Ceberg, C.; Petersson, K. Modifying a clinical linear accelerator for delivery of ultra-high dose rate irradiation. Radiother. Oncol. 2019, 139, 40–45. [Google Scholar] [CrossRef]
- Van Marlen, P.; Dahele, M.; Folkerts, M.; Abel, E.; Slotman, B.J.; Verbakel, W. Bringing FLASH to the Clinic: Treatment Planning Considerations for Ultrahigh Dose-Rate Proton Beams. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 621–629. [Google Scholar] [CrossRef]
- Colangelo, N.W.; Azzam, E.I. The Importance and Clinical Implications of FLASH Ultra-High Dose-Rate Studies for Proton and Heavy Ion Radiotherapy. Radiat. Res. 2020, 193, 1–4. [Google Scholar] [CrossRef]
- Carter, R.J.; Nickson, C.M.; Thompson, J.M.; Kacperek, A.; Hill, M.A.; Parsons, J.L. Complex DNA Damage Induced by High Linear Energy Transfer Alpha-Particles and Protons Triggers a Specific Cellular DNA Damage Response. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 776–784. [Google Scholar] [CrossRef]
- Carter, R.J.; Nickson, C.M.; Thompson, J.M.; Kacperek, A.; Hill, M.A.; Parsons, J.L. Characterisation of Deubiquitylating Enzymes in the Cellular Response to High-LET Ionizing Radiation and Complex DNA Damage. Int. J. Radiat. Oncol. Biol. Phys. 2019, 104, 656–665. [Google Scholar] [CrossRef]
- Beyreuther, E.; Brand, M.; Hans, S.; Hideghety, K.; Karsch, L.; Lessmann, E.; Schurer, M.; Szabo, E.R.; Pawelke, J. Feasibility of proton FLASH effect tested by zebrafish embryo irradiation. Radiother. Oncol. 2019, 139, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Zlobinskaya, O.; Siebenwirth, C.; Greubel, C.; Hable, V.; Hertenberger, R.; Humble, N.; Reinhardt, S.; Michalski, D.; Roper, B.; Multhoff, G.; et al. The effects of ultra-high dose rate proton irradiation on growth delay in the treatment of human tumor xenografts in nude mice. Radiat. Res. 2014, 181, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Van de Water, S.; Safai, S.; Schippers, J.M.; Weber, D.C.; Lomax, A.J. Towards FLASH proton therapy: The impact of treatment planning and machine characteristics on achievable dose rates. Acta Oncol. 2019, 58, 1463–1469. [Google Scholar] [CrossRef] [PubMed]
- Patriarca, A.; Fouillade, C.; Auger, M.; Martin, F.; Pouzoulet, F.; Nauraye, C.; Heinrich, S.; Favaudon, V.; Meyroneinc, S.; Dendale, R.; et al. Experimental Set-up for FLASH Proton Irradiation of Small Animals Using a Clinical System. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 619–626. [Google Scholar] [CrossRef]
| Model (Site of Irradiation) | Assay/Endpoint | Dose (Gy) | Dose Rate (Gy/s) | Radiation Source | Reference |
|---|---|---|---|---|---|
| Mice (WBI) 1 | Memory tests, neurogenesis | 10 | >100 | Electron | [13] |
| Mice (WBI) 1 | Neurocognitive tests, mature/immature neurons, growth hormone levels | 8 | 4.4 × 106 | Electron | [22] |
| Mice (WBI) 1 | Neurocognitive tests, dendritic spine density, microglial activation, inflammation | 30 | 200/300 | Electron | [20] |
| Mice (WBI) 1 | Neurocognitive tests, neuroinflammation, neuronal morphology | 10 | >100 | Electron | [23] |
| Mice (WBI) 1 | Neurocognitive tests, hippocampal cell division, astrogliosis | 10 | 37 | X-ray | [21] |
| Mice (thorax) | Survival, dermatitis, breathing function, lung pathology | 15/17.5/20 | 40 | Proton | [24] |
| Mice (thorax) | Lung fibrosis, skin dermatitis, survival | 15/17.5/20 | 40 | Proton | [25] |
| Mice (thorax) | Lung fibrosis, TGF-β signaling, apoptosis | 17 | 40–60 | Electron | [12] |
| Mice (thorax) | Cellular proliferation, pro-inflammatory gene expression, DNA damage (53BP1/γH2AX foci), senescence | 17 | 40–60 | Electron | [26] |
| Mice (abdomen) | Survival | 10–22 | 70–210 | Electron | [19] |
| Mice (abdomen) | Survival, stool production, crypt cell regeneration, apoptosis, DNA damage | 12–16 | 216 | Electron | [18] |
| Mice (abdomen) | Intestinal crypt cell proliferation | 15 Gy | 78 | Proton | [27] |
| Mice (local intestinal) | Fibrosis | 18 Gy | 78 | Proton | [27] |
| Mini-pig (skin) | Skin toxicity/injury | 22–34 | 300 | Electron | [14] |
| Zebrafish Embryo | Morphology | 8 | >100 | Electron | [23] |
| Model | Assay/Endpoint | Dose (Gy) | Dose Rate (Gy/s) | Radiation Source | Reference |
|---|---|---|---|---|---|
| Mice, HBCx-12A, and Hep-2 human xenografts (local) | Tumor growth | 17–25 | 60 | Electron | [12] |
| Mice, orthotopic engrafted lung carcinoma luciferase+ TC-1 cells (thorax) | Tumor growth | 15–28 | 60 | Electron | [12] |
| Mice, ID8 syngeneic ovarian cancer (thorax) | Tumor number/weight | 14 | 216 | Electron | [18] |
| Mice, orthotopic engrafted Lewis lung carcinoma (thorax) | Tumor size | 18 | 40 | Proton | [29] |
| Mice, pancreatic MH641905 flank tumor | Tumor growth | 12/15 | 78 | Proton | [27] |
| Cat, nasal planum SCC (local) | Tumor growth | 25–41 | 130–390 | Electron | [14] |
| Human, CD30+ T-cell cutaneous lymphoma | Tumor response | 15 | 167 | Electron | [15] |
| Model | Dose (Gy) | FLASH Dose-Rate (Gy/s) | Outcome | Reference |
|---|---|---|---|---|
| Zebrafish embryo | 0–43 | 100 | No survival difference | [61] |
| Mice (thorax) | 15/17.5/20 | 40 | Normal tissue protection with FLASH | [24] |
| Mice (thorax) | 15/17.5/20 | 40 | Normal tissue protection with FLASH | [25] |
| Mice (abdomen) | 15 | 78 | Normal tissue protection with FLASH | [27] |
| Mice (local intestinal) | 18 | 78 | Normal tissue protection with FLASH | [27] |
| Mice, orthotopic engrafted Lewis lung carcinoma (thorax) | 18 | 40 | Improved tumor control with FLASH, increased T-lymphocyte tumor infiltration | [29] |
| Mice, pancreatic MH641905 flank tumor | 12/15 | 78 | No difference in tumor control | [27] |
| Mice, FaDu head, and neck squamous cell carcinoma transplantation | 17.4 | >109 | No difference in tumor control | [62] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hughes, J.R.; Parsons, J.L. FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy. Int. J. Mol. Sci. 2020, 21, 6492. https://doi.org/10.3390/ijms21186492
Hughes JR, Parsons JL. FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy. International Journal of Molecular Sciences. 2020; 21(18):6492. https://doi.org/10.3390/ijms21186492
Chicago/Turabian StyleHughes, Jonathan R., and Jason L. Parsons. 2020. "FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy" International Journal of Molecular Sciences 21, no. 18: 6492. https://doi.org/10.3390/ijms21186492
APA StyleHughes, J. R., & Parsons, J. L. (2020). FLASH Radiotherapy: Current Knowledge and Future Insights Using Proton-Beam Therapy. International Journal of Molecular Sciences, 21(18), 6492. https://doi.org/10.3390/ijms21186492






