The scale-up biotransformation products were separated using 1000 µm preparative TLC silica gel plates (Anatech, Gehrden, Germany). The mobile phase contained a mixture of chloroform and methanol in a 9:1 (v/v) ratio. Separation products were scraped out and extracted twice using ethyl acetate.
3.5.2. NMR Spectroscopy
The NMR analysis was performed with a DRX 600 MHz Bruker spectrometer (Bruker, Billerica, MA, USA) with an UltraShield Plus magnet and measured in DMSO-d6 or Acetone-d6.
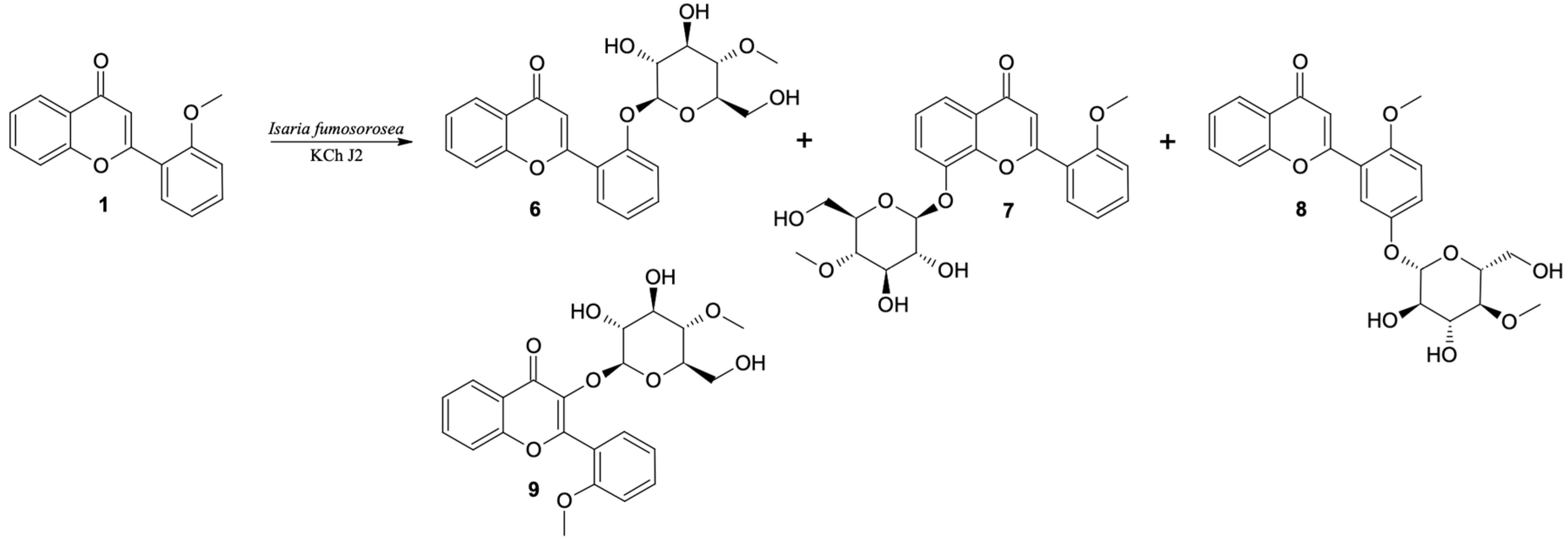
2′-O-β-D-(4″-O-Methylglucopyranosyl)-Flavone (6)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.07 (dd, 1H, J = 9.5, 9.1 Hz, H-4″), 3.29-3.35 (m, 1H, H-2″), 3.41-3.54 (m, 3H, H-3″, H-5″and one of H-6″), 3.46 (s, 3H, C-4″-OCH3), 3.64 (ddd, 1H, J = 11.6, 4.9, 1.6 Hz, one of H-6″), 4.72 (dd, 1H, J = 6.2, 5.0 Hz, C-6″-OH), 5.16 (d, 1H, J = 7.8 Hz, H-1″), 5.27 (d, 1H, J = 5.8 Hz, 3″-OH), 5.39 (d, 1H, J = 5.5 Hz, C-2″-OH), 7.08 (s, 1H, H-3), 7.22 (ddd, 1H, J = 7.3, 6.9, 1.0 Hz, H-5′), 7.35 (dd, 1H, J = 8.6, 0.8 Hz, H-3′), 7.50 (ddd, 1H, J = 8.1, 7.1, 1.1 Hz, H-6), 7.50 (ddd, 1H, J = 8.1, 7.1, 1.1 Hz, H-4′), 7.74 (dd, 1H, J = 8.5, 0.7 Hz, H-8), 7.83 (ddd, 1H, J = 8.7, 7.1, 1.7 Hz, H-7), 7.93 (dd, 1H, J = 7.9, 1.7 Hz, H-6′), 8.06 (ddd, 1H, J = 7.9, 1.6, 0.4 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 59.74 (C-4″-OCH3), 60.15 (C-6″), 73.50 (C-2″), 75.70 (C-5″), 76.55 (C-3″), 78.89 (C-4″), 99.73 (C-1″), 112.17 (C-3), 115.37 (C-3′), 118.58 (C-8), 120.72 (C-1′), 122.00 (C-5′), 123.20 (C-4a), 124.75 (C-5), 125.37 (C-6), 129.26 (C-6′), 132.70 (C-4′), 134.22 (C-7), 155.30 (C-2′), 156.01 (C-8a), 160.37 (C-2), 177.27 (C-4).
8-O-β-D-(4″-O-Methylglucopyranosyl)-2′-Methoxyflavone (7)
1H NMR (600 MHz) (Acetone-d6) δ (ppm): 3.27 (dd, 1H, J = 9.7, 8.9 Hz, H-4″), 3.53 (ddd, 1H, J = 9.7, 4.7, 2.1 Hz, H-5″), 3.61 (dd, 1H, J = 12.5, 4.4 Hz, H-2″), 3.64-3.74 (m, 2H, H-3″ and one of H-6″), 3.85 (ddd, 1H, J = 10.9, 4.8, 2.0 Hz, one of H-6″), 4.03 (s, 3H, C-4″-OCH3), 5.18 (d, 1H, J = 7.5 Hz, H-1″), 7.10 (s, 1H, H-3), 7.16 (ddd, 1H, J = 7.9, 7.4, 1.1 Hz, H-5′), 7.25 (dd, 1H, J = 8.4, 0.9 Hz, H-3′), 7.35 (t, 1H, J = 8.0 Hz, H-6), 7.56 (ddd, 1H, J = 8.4, 7.4, 1.8 Hz, H-4′), 7.61 (dd, 1H, J = 8.1, 1.5 Hz, H-7), 7.74 (dd, 1H, J = 8.0, 1.5 Hz, H-5), 8.22 (dd, 1H, J = 7.9, 1.7 Hz, H-6′). 13C NMR (151 MHz, Acetone-d6) δ = 56.36 (C-4″-OCH3), 60.58 (C-4″-OCH3), 61.99 (C-6″), 75.02 (C-2″), 77.21 (C-5″), 78.13 (C-3″), 80.01 (C-4″), 102.24 (C-1″), 112.68 (C-3), 113.06 (C-3′), 118.59 (C-5), 120.84 (C-7), 121.26 (C-1′), 121.83 (C-5′), 125.43 (C-6), 125.76 (C-4a), 130.59 (C-6′), 133.51 (C-4′), 147.70 (C-8a), 148.02 (C-8), 159.23 (C-2′), 160.88 (C-2), 178.08 (C-4).
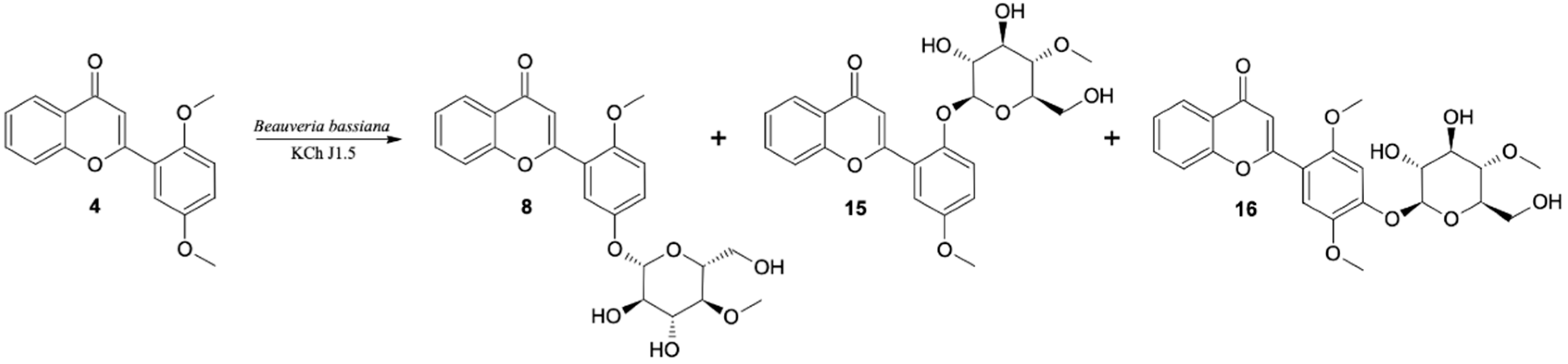
5′-O-β-D-(4″-O-Methylglucopyranosyl)-2′-Methoxyflavone (8)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.02 (t, 1H, J = 9.4 Hz, H-4″), 3.26 (td, 1H, J = 9.0, 4.0 Hz, H-2″), 3.40-3.45 (m, 2H, H-3″, H-5″), 3.46 (s, 3H, C-4″-OCH3), 3.53 (ddd, 1H, J = 11.6, 6.2, 5.8 Hz, one of H-6″), 3.67 (ddd, 1H, J = 11.4, 4.7, 1.4 Hz, one of H-6″), 3.90 (s, 3H, C-2″-OCH3), 4.77 (t, 1H, J = 5.7 Hz, C-6″-OH), 4.87 (d, 1H, J = 7.8 Hz, H-1″), 5.29 (d, 1H, J = 5.4 Hz, C-3″-OH), 5.43 (d, 1H, J = 4.5 Hz, C-2″-OH), 6.99 (s, 1H, H-3), 7.20 (d, 1H, J = 9.2 Hz, H-3′), 7.27 (dd, 1H, J = 9.1, 3.1 Hz, H-4′), 7.50 (ddd, 1H, J = 8.0, 7.0, 1.0 Hz, H-6), 7.63 (dd, 1H, J = 3.0, Hz, H-6′), 7.76 (dd, 1H, J = 7.9, 0.6 Hz, H-8), 7.83 (ddd, 1H, J = 8.6, 7.0, 1.6 Hz, H-7), 8.05 (dd, 1H, J = 7.9, 1.6 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 56.39 (C-2′-OCH3), 59.71 (C-4″-OCH3), 60.39 (C-6″), 73.48 (C-2″), 75.73 (C-5″), 76.28 (C-3″), 79.20 (C-4″), 101.11 (C-1″), 111.71 (C-3), 113.68 (C-3′), 116.93 (C-6′), 118.61 (C-8), 120.14 (C-1′), 120.88 (C-4′), 123.10 (C-4a), 124.71 (C-5), 125.45 (C-6), 134.33 (C-7), 151.17 (C-5′), 152.88 (C-2′), 155.91 (C-8a), 160.05 (C-2), 177.18 (C-4).
3-O-β-D-(4″-O-Methylglucopyranosyl)-2′-Methoxyflavone (9)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.07 (t, 1H, J = 9.3 Hz, H-4″), 3.27-3.37 (m, 2H, H-2″ and H-5″), 3.40 (s, 3H, C-4″-OCH3), 3.41-3.55 (m, 2H, H-6″), 3.79 (s, 3H, C-3-OCH3), 4.45 (t, 1H, J = 5.3 Hz, C-6″-OH), 5.14 (d, 1H, J = 5.5 Hz, 3″-OH), 5.27 (d, 1H, J = 7.8 Hz, H-1″), 5.31 (d, 1H, J = 4.7 Hz, C-2″-OH), 7.06 (td, 1H, J = 7.4, 0.8 Hz, H-5′), 7.16 (d, 1H, J = 8.3 Hz, H-3′), 7.50-7.55 (m, 2H, H-4′ and H-6), 7.66 (dd, 1H, J = 8.4, 0.4 Hz, H-8), 7.73 (dd, 1H, J = 7.6, 1.7 Hz, H-6′), 7.82 (ddd, 1H, J = 8.6, 7.1, 1.7 Hz, H-7), 8.14 (dd, 1H, J = 8.0, 1.4 Hz, H-5).
2′-Hydroxyflavone (10)
1H NMR (600 MHz) (Acetone-d6) δ (ppm): 7.07 (ddd, 1H, J = 8.0, 7.3, 1.1 Hz, H-5′), 7.13 (dd, 1H, J = 8.2, 1.0 Hz, H-3′), 7.19 (s, 1H, H-3), 7.42 (ddd, 1H, J = 8.2, 7.3, 1.7 Hz, H-4′), 7.48 (ddd, 1H, J = 8.1, 7.1, 1.1 Hz, H-6), 7.70 (ddd, 1H, J = 8.4, 1.1, 0.5 Hz, H-8), 7.80 (dd, 1H, J = 8.8, 6.9, 1.6 Hz, H-7), 7.99 (dd, 1H, J = 7.9, 1.7 Hz, H-5), 10.03 (s, C-2′-OH). 13C NMR (151 MHz, Acetone-d6) δ = 112.54 (C-3), 117.93 (C-3′), 119.18 (C-8), 119.58 (C-1′), 120.91 (C-5′), 124.71 (C-4a), 125.85 (C-6), 125.89 (C-5), 129.73 (C-6′), 133.25 (C-4′), 134.62 (C-7), 157.16 (C-2′), 157.32 (C-8a), 161.76 (C-2), 178.25 (C-4).
3′-Hydroxyflavone (11)
1H NMR (600 MHz) (DMSO) δ (ppm): 6.94 (s, 1H, H-3), 7.01 (ddd, 1H, J = 8.1, 2.5, 0.9 Hz, H-4′), 7.38 (t, 1H, J = 7.9 Hz, H-5′), 7.44 (t, 1H, J = 1.9 Hz, H-2′), 7.51 (ddd, 1H, J = 8.0, 6.9, 1.1 Hz, H-6), 7.53 (ddd, 1H, J = 7.8, 1.8, 1.0 Hz, H-6′), 7.77 (ddd, 1H, J = 8.4, 1.2, 0.5 Hz, H-8), 7.86 (ddd, 1H, J = 8.5, 6.9, 1.6 Hz, H-7), 8.06 (ddd, 1H, J = 7.9, 1.7, 0.5 Hz, H-5), 9.90 (s, C-3′-OH). 13C NMR (151 MHz, DMSO) δ = 106.93 (C-3), 112.85 (C-2′), 117.23 (C-6′), 118.53 (C-8), 118.88 (C-4′), 123.36 (C-4a), 124.82 (C-5), 125.57 (C-6), 130.29 (C-5′), 132.45 (C-1′), 134.38 (C-7), 155.68 (C-8a), 157.92 (C-3′) 162.73 (C-2), 177.10 (C-4).
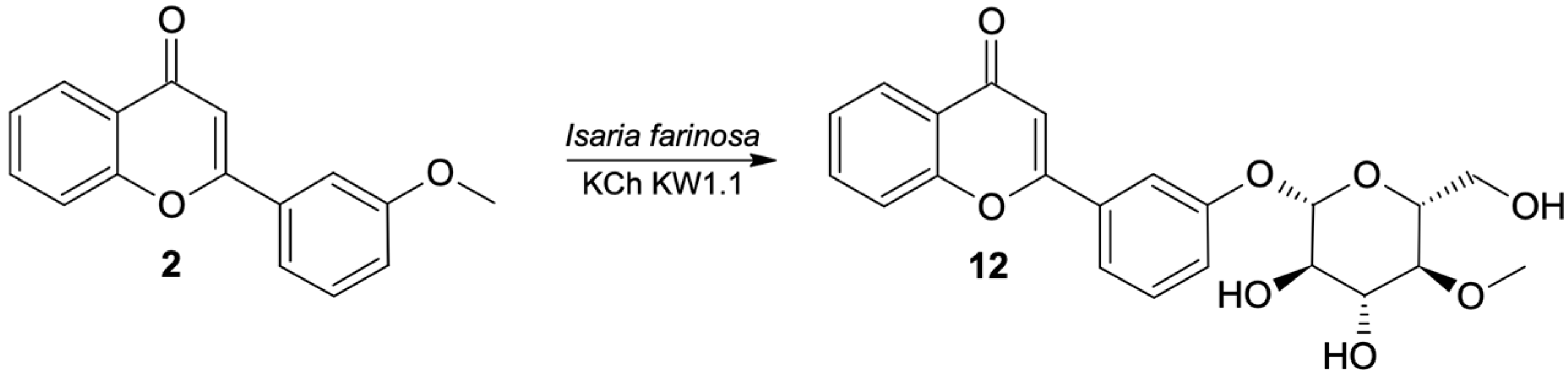
3′-O-β-D-(4″-O-Methylglucopyranosyl)-Flavone (12)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.05 (t, 1H, J = 9.2 Hz, H-4″), 3.25-3.34 (m, 1H, H-2″), 3.42-3.57 (m, 3H, H-3″, H-5″and one of H-6″), 3.47 (s, 3H, C-4″-OCH3), 3.68 (dd, 1H, J = 10.0, 4.9 Hz, one of H-6″), 4.80 (t, 1H, J = 5.2 Hz, C-6″-OH), 5.07 (d, 1H, J = 7.8 Hz, H-1″), 5.31 (d, 1H, J = 5.5 Hz, C-3″-OH), 5.46 (d, 1H, J = 5.2 Hz, C-2″-OH), 7.07 (s, 1H, H-3), 7.26 (dd, 1H, J = 8.2, 2.4 Hz, H-4′), 7.47-7.55 (m, 2H, H-5′ and H-6), 7.73 (d, 1H, J = 2.0 Hz, H-2′), 7.76 (d, 1H, J = 7.8 Hz, H-8), 7.79-7.87 (m, 2H, H-6′ and H-7), 8.06 (d, 1H, J = 7.9 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 59.76 (C-4″-OCH3), 60.35 (C-6″), 73.48 (C-2″), 75.75 (C-5″), 76.34 (C-3″), 79.19 (C-4″), 100.02 (C-1″), 107.24 (C-3), 113.84 (C-2′), 118.69 (C-6′), 119.79 (C-4′), 120.00 (C-8), 123.36 (C-4a), 124.81 (C-5), 125.63 (C-6), 130.31 (C-5′), 132.52 (C-1′), 134.43 (C-7), 155.72 (C-8a), 157.81 (C-3′), 162.25 (C-2), 177.23 (C-4).
4′-Hydroxyflavone (13)
1H NMR (600 MHz) (DMSO) δ (ppm): 6.88 (s, 1H, H-3), 6.92-6.96 (m, 2H, H-3′, H-5′), 7.48 (ddd, 1H, J = 8.0, 7.0, 1.2 Hz, 1H, H-6), 7.75 (ddd, 1H, J = 8.4, 1.2, 0.4 Hz, H-8), 7.81 (ddd, 1H, J = 8.5, 6.9, 1.7 Hz, H-7), 7.95-7.99 (m, 2H, H-2′, H-6′), 8.03 (dd, 1H, J = 7.9, 1.7, 0.4 Hz, H-5), 10.32 (s, C-4′-OH). 13C NMR (151 MHz, DMSO) δ = 104.83 (C-3), 115.97 (C-3′ and C-5′), 118.39 (C-8), 121.60 (C-1′), 123.34 (C-4a), 124.75 (C-5), 125.35 (C-6), 128.39 (C-2′ and C-6′), 134.06 (C-7), 155.61 (C-8a), 161.00 (C-4′), 163.09 (C-2), 176.91 (C-4).
4′-O-β-D- (4″-O-Methylglucopyranosyl)-Flavone (14)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.06 (t, 1H, J = 9.3 Hz, H-4″), 3.29 (ddd, 1H, J = 9.1, 7.8, 5.2 Hz, H-2″), 3.42-3.49 (m, 2H, H-3″, H-5″), 3.47 (s, 3H, C-4″-OCH3), 3.52 (ddd, 1H, J = 11.5, 6.4, 5.1 Hz, one of H-6″), 3.65 (ddd, J = 11.4, 5.0, 1.5 Hz, one of H-6″), 4.74 (dd, 1H, J = 6.2, 5.1 Hz, C-6″-OH), 5.07 (d, 1H, J = 7.8 Hz, H-1″), 5.30 (d, 1H, J = 5.5 Hz, C-3″-OH), 5.48 (d, 1H, J = 5.2 Hz, C-2″-OH), 6.99 (s, 1H, H-3), 7.18-7.22 (m, 2H, H-3′, H-5′), 7.50 (ddd, 1H, J = 8.1, 6.8, 1.3 Hz, H-6), 7.78 (ddd, 1H, J = 8.5, 1.3, 0.4 Hz, H-8), 7.83 (ddd, 1H, J = 8.5, 6.8, 1.7 Hz, H-7), 8.04 (ddd, 1H, J = 7.9, 1.6, 0.5 Hz, H-5), 8.06-8.10 (m, 2H, H-2′, H-6′). 13C NMR (151 MHz, DMSO) δ = 59.74 (C-4″-OCH3), 60.18 (C-6″), 73.37 (C-2″), 75.69 (C-5″), 76.25 (C-3″), 78.94 (C-4″), 99.50 (C-1″), 105.76 (C-3), 116.54 (C-3′ and C-5′), 118.50 (C-8), 123.33 (C-4a), 124.41 (C-5), 124.78 (C-6), 125.46 (C-1′), 128.11 (C-2′ and C-6′), 134.20 (C-7), 155.65 (C-8a), 160.05 (C-4′), 162.46 (C-2), 177.02 (C-4).
2′-O-β-D-(4″-O-Methylglucopyranosyl)-5′-Methoxyflavone (15)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.05 (t, 1H, J = 9.4 Hz, H-4″), 3.25-3.29 (m, 1H, H-2″), 3.40-3.45 (m, 2H, H-3″, H-5″), 3.45 (s, 3H, C-4″-OCH3), 3.51-3.55 (m, 1H, one of H-6″), 3.63 (ddd, 1H, J = 12.1, 4.8, 1.7 Hz, one of H-6″), 3.81 (s, 3H, C-6″-OCH3), 4.69 (d, 1H, J = 5.5 Hz, C-6″-OH), 5.03 (d, 1H, J = 7.8 Hz, H-1″), 5.24 (d, 1H, J = 5.8 Hz, C-3″-OH), 5.34 (d, J = 5.6 Hz, C-2″-OH), 7.13 (dd, 1H, J = 9.2, 3.2 Hz, H-4′), 7.30 (d, 1H, J = 9.2 Hz, H-3′), 7.44 (d, 1H, J = 3.1 Hz, H-6′), 7.50 (ddd, 1H, J = 8.1, 7.0, 1.1 Hz, H-6), 7.77 (dd, 1H, J = 8.3, 0.7 Hz, H-8), 7.83 (ddd, 1H, J = 8.6, 7.1, 1.8 Hz, H-7), 8.05 (dd, 1H, J = 7.9, 1.8 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 55.73 (C-2′-OCH3), 59.72 (C-4″-OCH3), 60.21 (C-6″), 73.55 (C-2″), 75.70 (C-5″), 76.56 (C-3″), 78.96 (C-4″), 100.56 (C-1″), 112.36 (C-3), 113.87 (C-6′), 117.13 (C-3′), 117.99 (C-4′), 118.71 (C-8), 121.58 (C-1′), 123.20 (C-4a), 124.71 (C-5), 125.37 (C-6), 134.18 (C-7), 149.34 (C-5′), 153.94 (C-2′), 155.99 (C-8a), 160.14 (C-2), 177.27 (C-4).
4′-O-β-D-(4″-O-Methylglucopyranosyl)-2′,5′-Dimethoxyflavone (16)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.00 (t, 1H, J = 9.2 Hz, H-4″), 3.32-3.37 (m, 1H, H-2″), 3.39-3.43 (m, 1H, H-5″), 3.46 (s, 3H, C-4″-OCH3), 3.47-3.54 (m, 2H, H-3″ and one of H-6″), 3.64-3.69 (m, 1H, one of H-6″), 3.84 (s, 3H, C-5″-OCH3), 3.90 (s, 3H, C-2″-OCH3), 4.79 (t, 1H, J = 5.1 Hz, C-6″-OH), 5.14 (d, 1H, J = 7.9 Hz, H-1″), 5.32 (d, 1H, J = 5.6 Hz, C-3″-OH), 5.49 (d, 1H, J = 5.6 Hz, C-2″-OH), 6.98 (s, 1H, H-3), 7.00 (s, 1H, H-3′), 7.48 (ddd, 1H, J = 8.0, 7.1, 1.3 Hz, H-6), 7.56 (s, 1H, H-6′), 7.79 (dd, 1H, J = 8.4, 1.3 Hz, H-8), 7.81 (ddd, 1H, J = 8.5, 7.1, 1.5 Hz, H-7), 8.03 (dd, 1H, J = 7.9, 1.5 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 56.41 (C-2′-OCH3), 56.69 (C-6′-OCH3), 59.75 (C-4″-OCH3), 60.39 (C-6″), 73.27 (C-2″), 75.96 (C-5″), 76.67 (C-3″), 79.36 (C-4″), 99.48 (C-1″), 101.40 (C-3′), 110.56 (C-3), 111.82 (C-1′), 113.06 (C-6′), 119.60 (C-8), 123.11 (C-4a), 124.66 (C-5), 125.29 (C-6), 134.33 (C-7), 142.81 (C-4′), 150.23 (C-5′), 153.23 (C-2′), 155.78 (C-8a), 160.31 (C-2), 177.12 (C-4).
5′-Hydroxy-2′-Methoxyflavone (17)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.84 (s, 3H, C-2′-OCH3), 6.94 (s, 1H, H-3), 6.97 (dd, 1H, J = 8.9, 3.0 Hz, H-4′), 7.09 (d, 1H, J = 9.0 Hz, H-3′), 7.32 (d, 1H,J = 3.0 Hz, H-6′), 7.50 (ddd, J = 8.0, 7.1, 0.8 Hz, H-6), 7.71 (d, 1H, J = 8.2 Hz, H-8), 7.83 (ddd, 1H, J = 8.6, 7.2, 1.7 Hz, H-7), 8.05 (dd, 1H, J = 7.9, 1.4 Hz, H-5), 9.41 (s, C-5′-OH). 13C NMR (151 MHz, DMSO) δ = 56.33 (C-2′-OCH3), 111.57 (C-3), 114.07 (C-3′), 115.01 (C-6′), 118.43 (C-8), 119.36 (C-4′), 120.29 (C-1′), 123.13 (C-4a), 124.77 (C-5), 125.42 (C-6), 134.33 (C-7), 150.80 (C-2′), 151.12 (C-5′), 155.89 (C-8a), 160.53 (C-2), 177.16 (C-4).
4′-Hydroxy-2′,5′-Dimethoxyflavone (18)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.84 (s, 3H, C-5′-OCH3), 3.85 (s, 3H, C-2′-OCH3), 6.70 (s, 1H, H-3′), 6.95 (s, 1H, H-3), 7.47 (ddd, 1H, J = 8.1, 6.4, 1.9 Hz, H-6), 7.53 (s, 1H, H-6′), 7.78 (ddd, 1H, J = 8.6, 7.2, 1.7 Hz, H-7), 7.81 (d, 1H, J = 8.2 Hz, H-8), 8.02 (dd, 1H, J = 7.9, 1.5 Hz, H-5), 10.07 (s, C-4′-OH). 13C NMR (151 MHz, DMSO) δ = 56.17 (C-5′-OCH3), 56.64 (C-2′-OCH3), 101.11 (C-3′), 109.36 (C-1′), 109.90 (C-3), 112.87 (C-6′), 118.51 (C-8), 123.11 (C-4a), 124.63 (C-5), 125.16 (C-6), 133.89 (C-7), 141.87 (C-4′), 151.41 (C-5′), 153.86 (C-2′), 155.74 (C-8a), 160.67 (C-2), 177.08 (C-4).
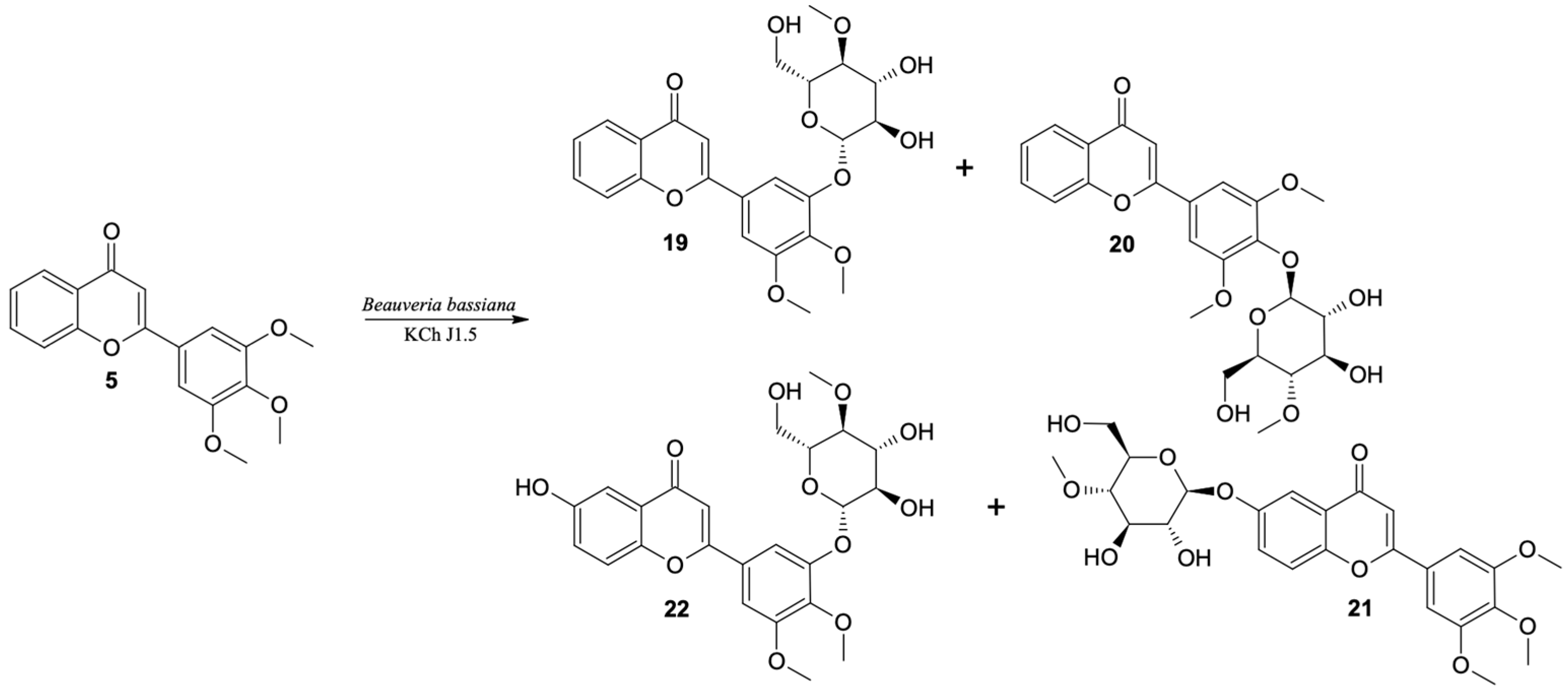
3′-O-β-D-(4″-O-Methylglucopyranosyl)-4′,5′-Dimethoxyflavone (19)
3.04 (t, 1H,
J = 9.1 Hz, H-4″), 3.31-3.36 (m, 1H, H-2″), 3.48 (s, 3H, C-4″-OC
H3), 3.46-3.58 (m, 3H, H-3″, H-5″ and one of H-6″), 3.70 (dd, 1H,
J = 9.9, 5.0 Hz, one of H-6″), 3.81 (s, 3H, C-4′-OC
H3), 3.91 (s, 3H, C-5′-OC
H3), 4.84 (t, 1H,
J = 5.5 Hz, C-6″-O
H), 5.07 (d, 1H,
J = 7.8 Hz, H-1″), 5.32 (d, 1H,
J = 5.6 Hz, C-3″-O
H), 5.46 (d, 1H,
J = 5.7 Hz, C-2″-O
H), 7.11 (s, 1H, H-3), 7.41 (d, 1H,
J = 2.0 Hz, H-6′), 7.49 (ddd, 1H,
J = 8.1, 5.3, 2.9 Hz, H-6), 7.54 (d, 1H,
J = 2.0 Hz, H-2′), 7.81-7.84 (m, 2H, H-7 and H-8), 8.04 (ddd, 1H,
J = 8.0, 1.5, 0.6 Hz, H-5).
13C NMR (151 MHz, DMSO) is presented in
Table 7.
4′-O-β-D-(4″-O-Methylglucopyranosyl)-3′,5′-Dimethoxyflavone (20)
3.03 (t, 1H, J = 9.1 Hz, H-4″), 3.31-3.36 (m, 1H, H-2″), 3.44 (s, 3H, C-4″-OCH3), 3.46-3.58 (m, 3H, H-3″, H-5″ and one of H-6″), 3.69 (dd, 1H, J = 9.9, 5.0 Hz, one of H-6″), 3.90 (s, 6H, C-3′-OCH3 and C-5′-OCH3), 4.52 (dd, 1H, J = 5.9, 4.9 Hz, C-6″-OH), 5.14 (d, 1H, J = 7.6 Hz, H-1″), 5.19 (d, 1H, J = 5.5 Hz, C-3″-OH), 5.23 (d, 1H, J = 4.9 Hz, C-2″-OH), 7.14 (s, 1H, H-3), 7.39 (s, 2H, H-2′ and H-6′), 7.49 (m, 1H, H-6), 7.81-7.84 (m, 2H, H-7 and H-8), 8.04 (ddd, 1H, J = 8.0, 1.5, 0.6 Hz, H-5). 13C NMR (151 MHz, DMSO) δ = 56.76 (C-3′-OCH3 and C-5′-OCH3), 59.66 (C-4″-OCH3), 60.34 (C-6″), 74.40 (C-2″), 76.08 (C-5″), 76.42 (C-3″), 79.18 (C-4″), 101.90 (C-1″), 104.89 (C-2′, and C-6′), 106.78 (C-3), 118.73 (C-8), 123.33 (C-4a), 124.77 (C-5), 125.53 (C-6), 126.27 (C-1′), 134.21 (C-7), 137.35 (C-4′), 152.93 (C-3′ and C-5′), 155.71 (C-8a), 162.44 (C-2), 177.22 (C-4).
6-O-β-D-(4″-O-Methylglucopyranosyl)-3′,4′,5′-Trimethoxyflavone (21)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.08 (t, 1H,
J = 9.3 Hz, H-4″), 3.28 (ddd, 1H,
J = 9.1, 7.3, 4.7 Hz, H-2″), 3.36-3.39 (m, 1H, H-5″), 3.41-3.46 (m, 2H, H-3″ and one of H-6″), 3.47 (s, 3H, C-4″-OC
H3), 3.62-3.67 (m, 1H, one of H-6″), 3.75 (s, 3H, C-4′-OC
H3), 3.91 (s, 6H, C-3′-OC
H3 and C-5′-OC
H3), 4.74 (dd, 1H,
J = 6.4, 5.0 Hz, C-6″-O
H), 5.01 (d, 1H,
J = 7.8 Hz, H-1″), 5.28 (d, 1H,
J = 5.5 Hz, C-3″-O
H), 5.46 (d, 1H,
J = 5.2 Hz, C-2″-O
H), 7.14 (s, 1H, H-3), 7.39 (s, 2H, H-2′ and H-6′), 7.51 (dd, 1H,
J = 9.1, 3.1 Hz, H-7), 7.58 (d, 1H,
J = 3.1 Hz, H-5), 7.83 (d, 1H,
J = 9.2 Hz, H-8).
13C NMR (151 MHz, DMSO) is presented in
Table 7.
3′-O-β-D-(4″-O-Methylglucopyranosyl)-6-Hydroxy-4′,5′-Dimethoxyflavone (22)
1H NMR (600 MHz) (DMSO) δ (ppm): 3.03 (t, 1H,
J = 9.1 Hz, H-4″), 3.30-3.35 (m, 1H, H-2″), 3.47 (s, 3H, C-4″-OC
H3), 3.43-3.57 (m, 3H, H-3″, H-5″ and one of H-6″), 3.68-372 (m, 1H, one of H-6″), 3.80 (s, 3H, C-4′-OC
H3), 3.90 (s, 3H, C-5′-OC
H3), 4.81 (t, 1H,
J = 5.4 Hz, C-6″-O
H), 5.05 (d, 1H,
J = 7.9 Hz, H-1″), 5.29 (d, 1H,
J = 5.7 Hz, C-3″-O
H), 5.43 (d, 1H,
J = 5.7 Hz, C-2″-O
H), 7.02 (s, 1H, H-3), 7.25 (dd, 1H,
J = 9.0, 3.0 Hz, H-6), 7.31 (d, 1H,
J = 3.0 Hz, H-8), 7.38 (d, 1H,
J = 2.0 Hz, H-6′), 7.50 (d, 1H,
J = 2.0 Hz, H-2′), 7.68 (d, 1H,
J = 9.0 Hz, H-5), 10.02 (s, 1H, -O
H).
13C NMR (151 MHz, DMSO) is presented in
Table 7.