EVs and Bioengineering: From Cellular Products to Engineered Nanomachines
Abstract
1. Introduction
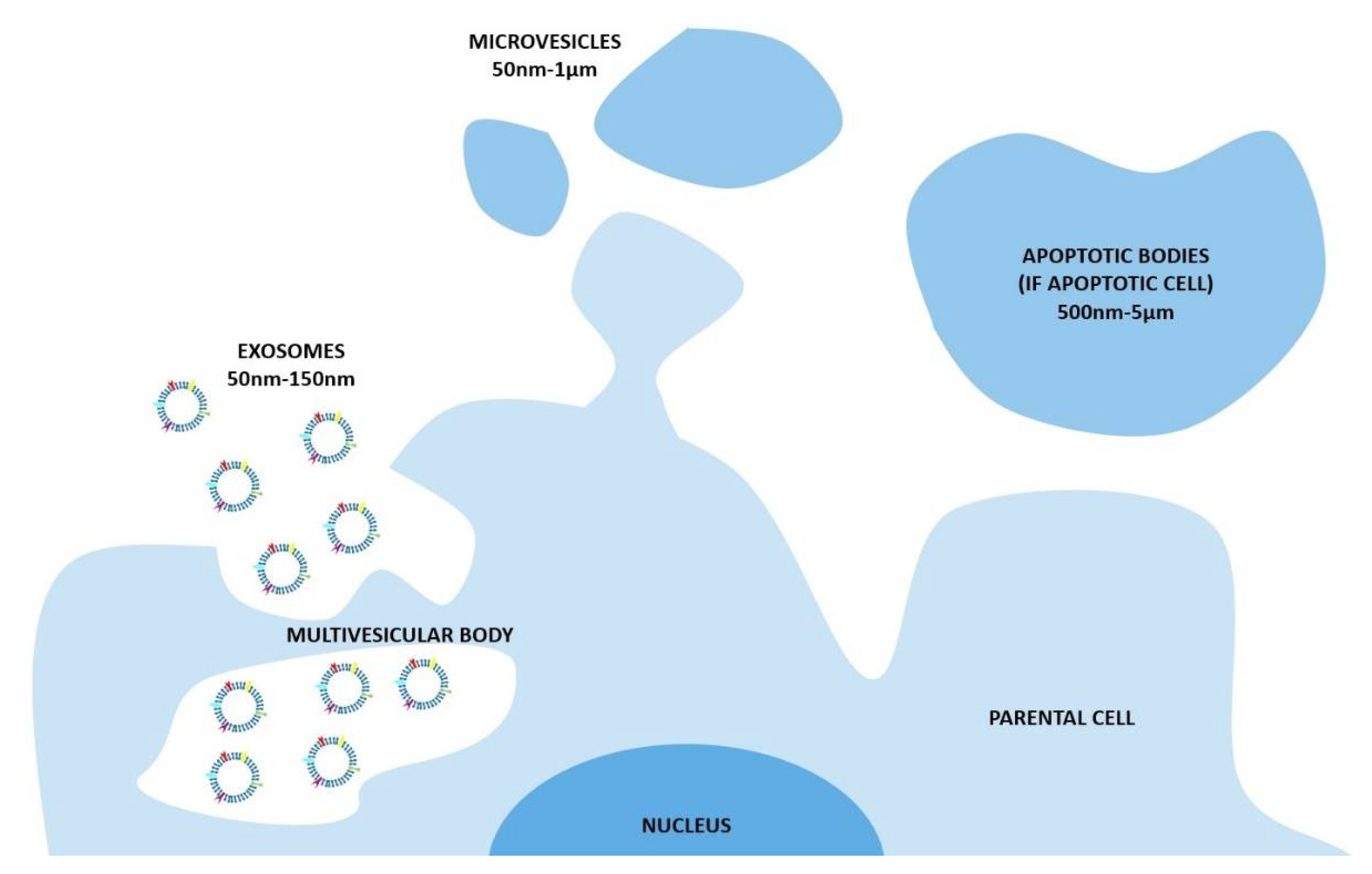
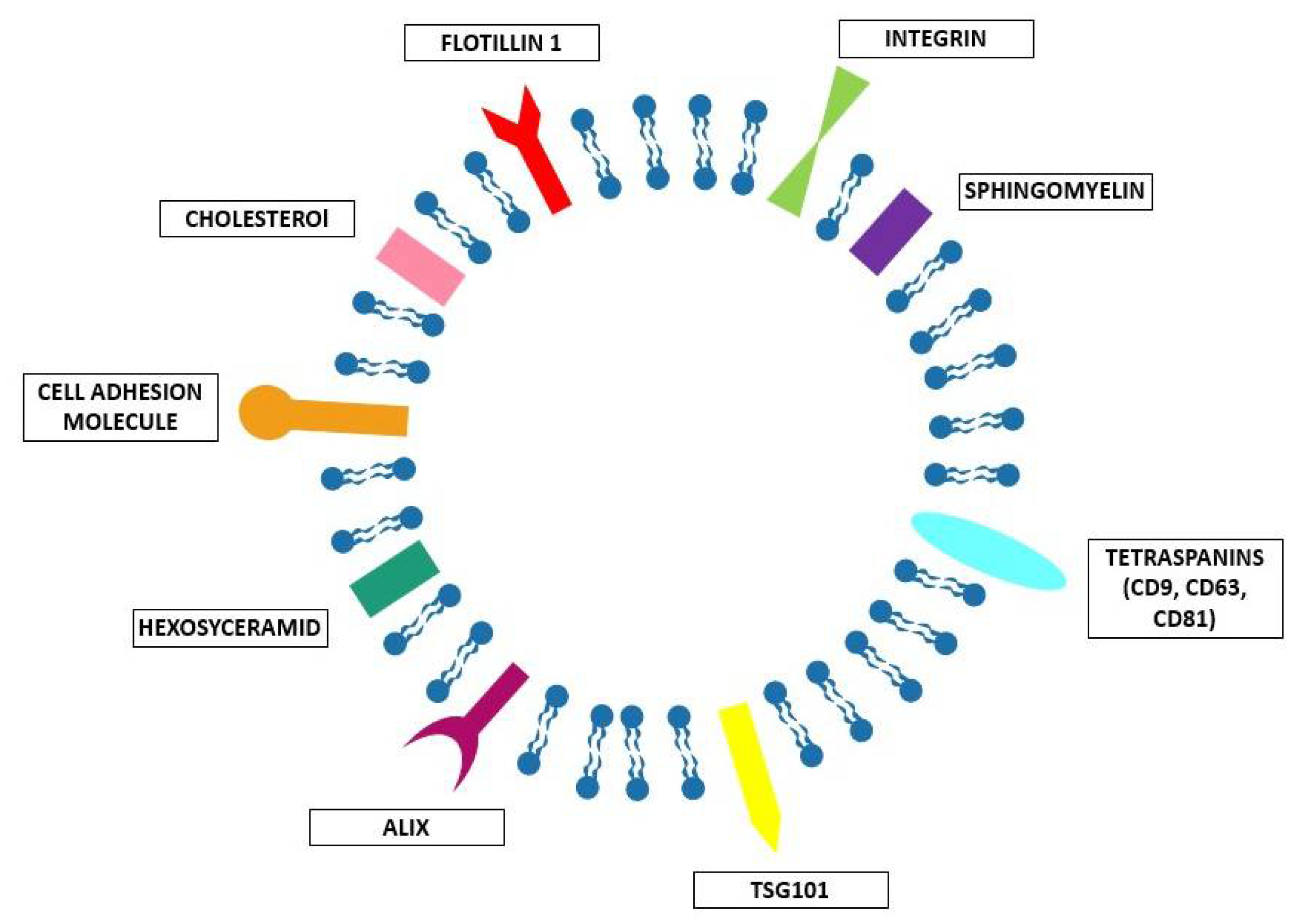
2. Natural EVs
2.1. EVs as Delivery Systems
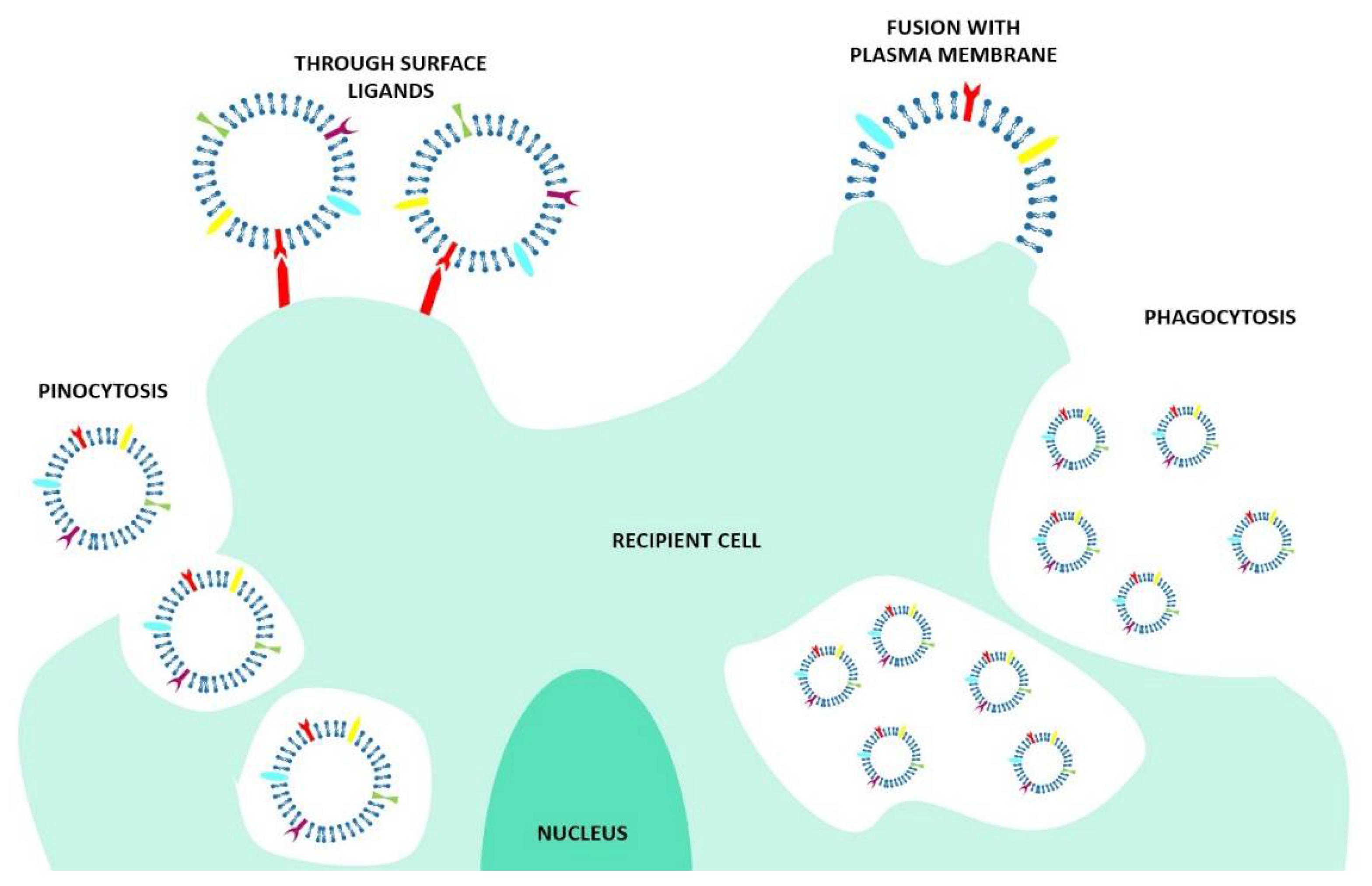
2.1.1. What Kind of EV–Cell Interactions Exist?
2.1.2. Which Kind of Cells Produce EVs?
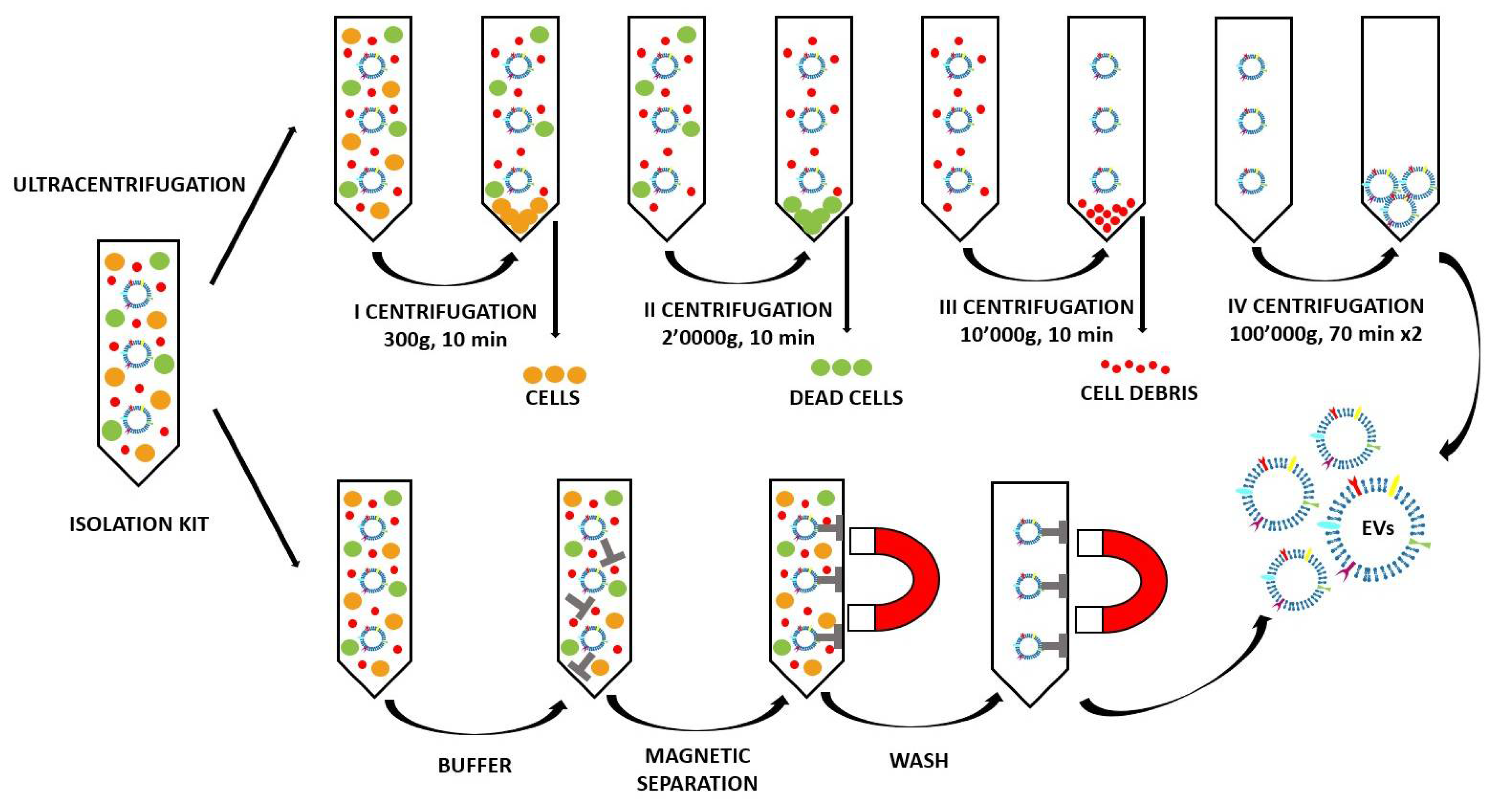
2.2. Method of EV Extraction
2.3. Cargo-Loaded EVs
- Hydrophilic components such as hydrophilic drugs, but also microRNA (miRNA), small interfering RNA (siRNA), DNA, and proteins. They can be encapsulated in the hydrophilic core of the EV [47].
- Hydrophobic drugs, which can be incorporated in the lipid bilayer [47].
- Macromolecules for imaging, tracking (as fluorophore-conjugate antibodies), and targeting purposes. They can be bound with surface modifications to the EV lipid bilayers or surface proteins [47].
2.3.1. Passive Loading Methods
Co-incubation
2.3.2. Active Loading Methods
Electroporation
Sonication
Extrusion
Freeze–Thaw
Chemical-Based Transfection
3. Engineered EVs
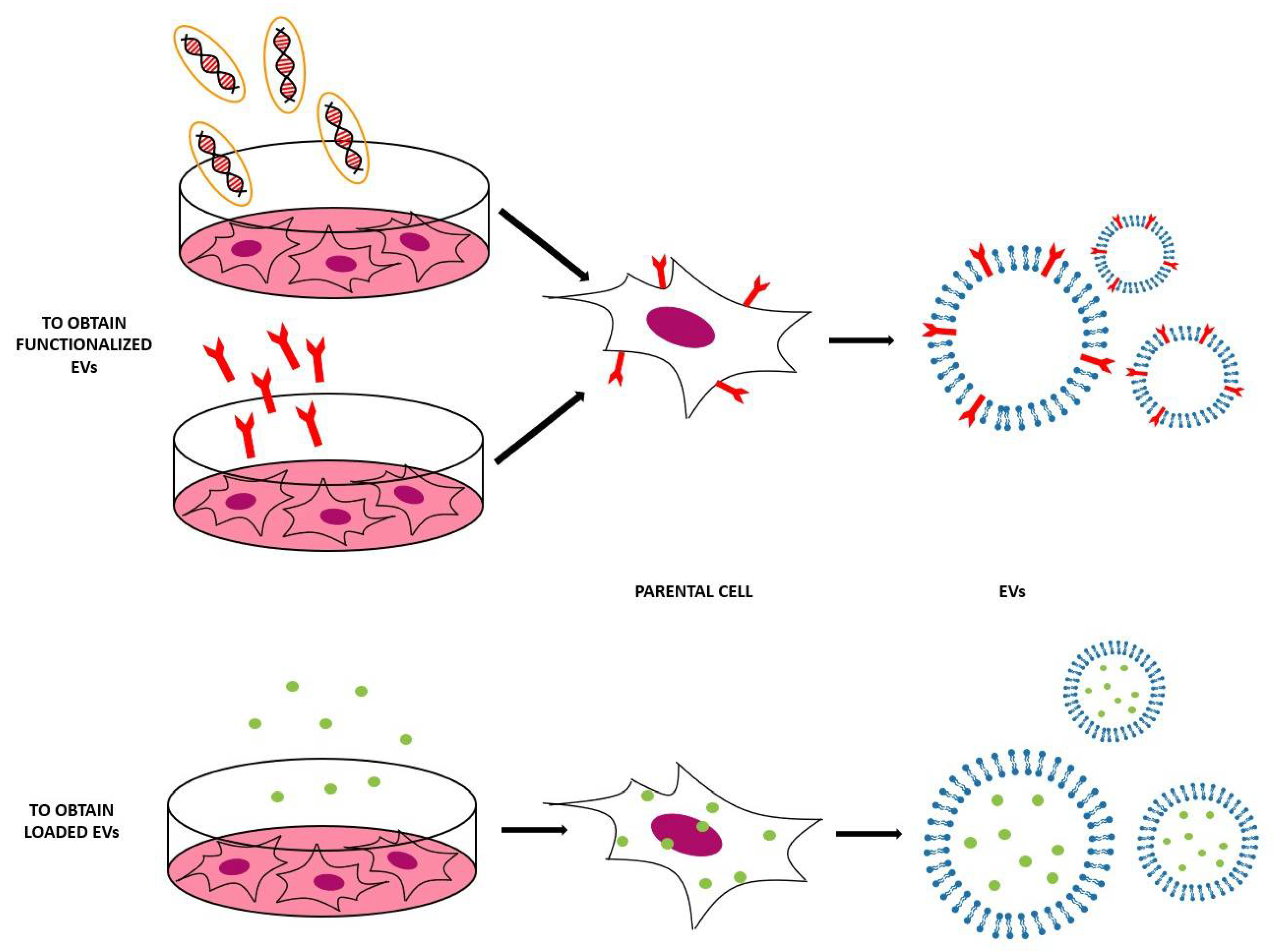
3.1. Indirect Methods
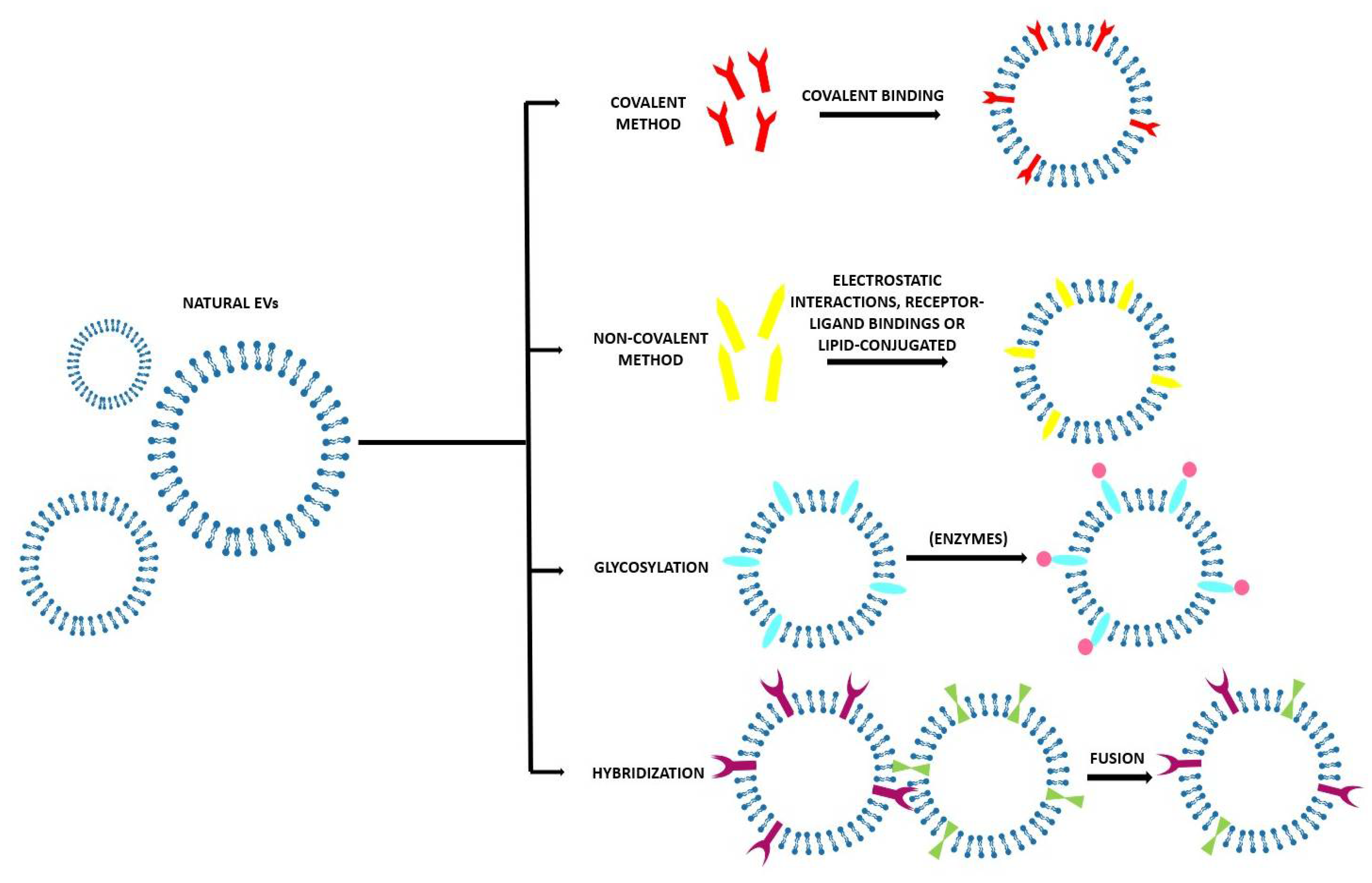
3.2. Direct Methods
3.2.1. Covalent Methods
3.2.2. Non-Covalent Methods
3.2.3. Glycosylation
3.2.4. Hybridization
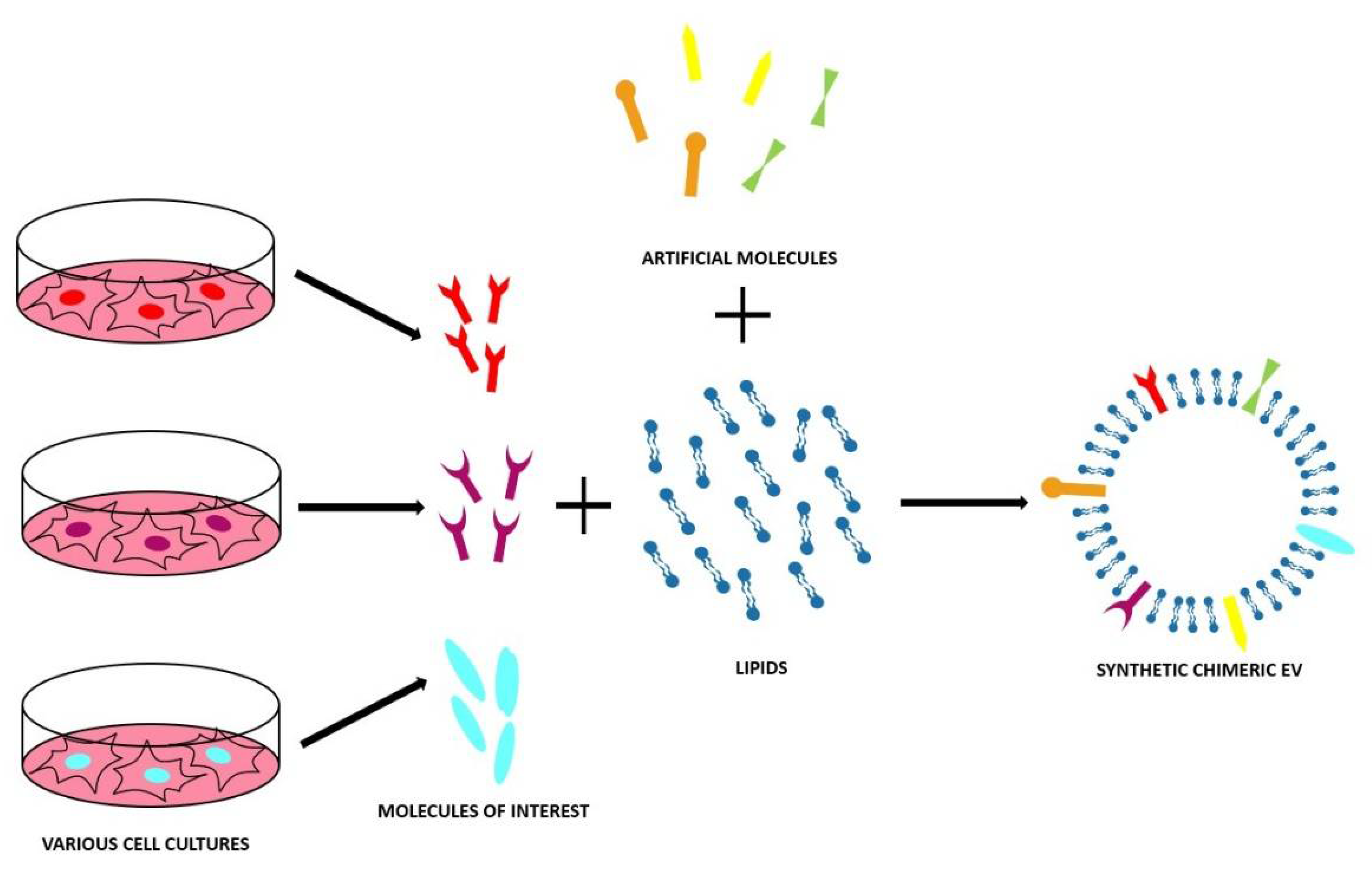
4. Synthetic and Chimeric EVs
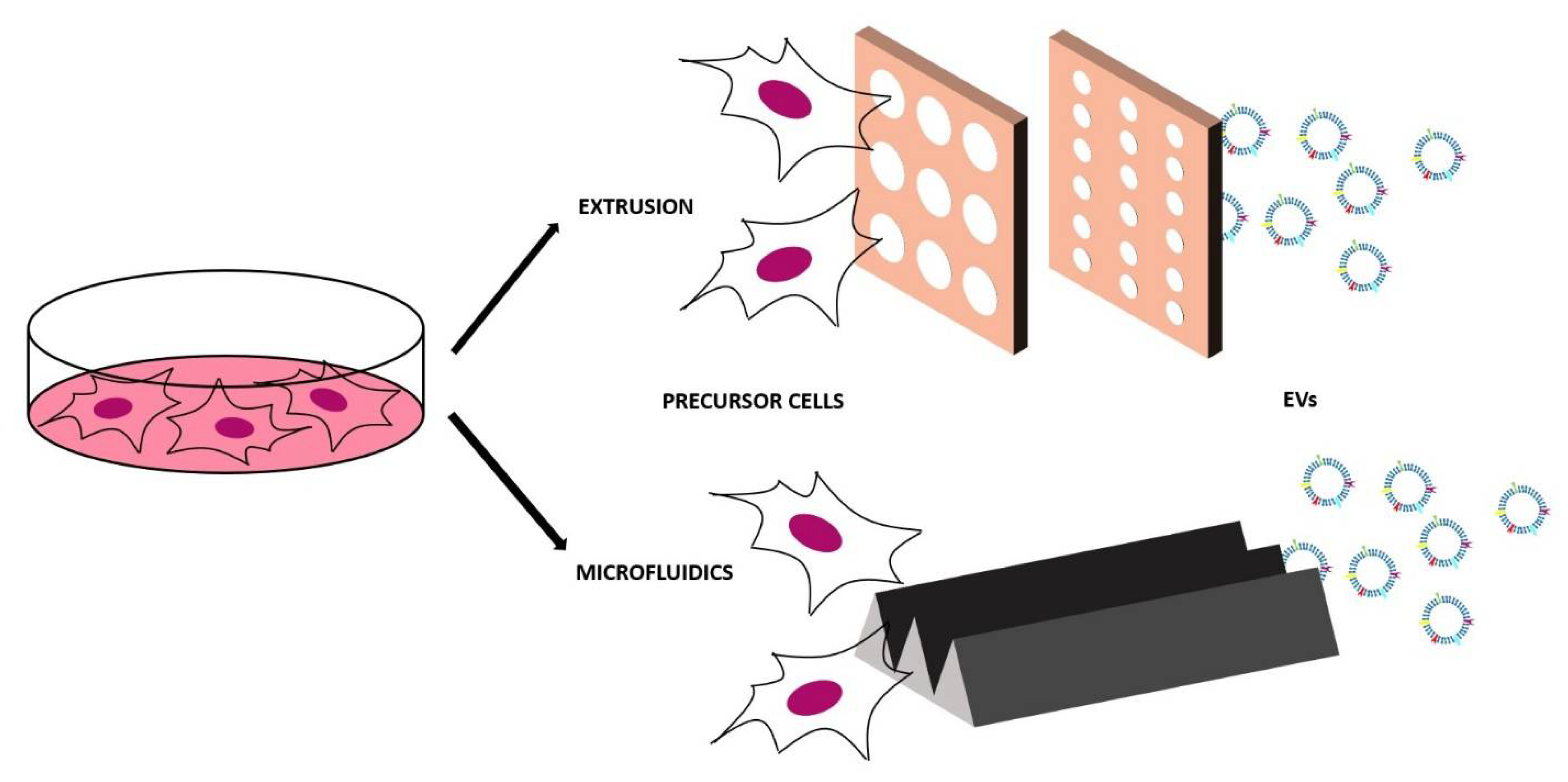
4.1. Top-Down Approaches
4.2. Bottom-Up Approach
5. Conclusions and Future Perspectives
6. Patents
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| i.v. | intra venous |
| i.n. | intra nasal |
| i.p. | intra peritoneal |
| i.d. | intra dermal |
| i.m. | intramuscular |
| i.t. | intra tumour |
| s.i. | subcutaneously injection |
| t.r. | transfecting reagent |
References
- Bunggulawa, E.J.; Wang, W.; Yin, T.; Wang, N.; Durkan, C.; Wang, Y.; Wang, G. Recent advancements in the use of exosomes as drug delivery systems. J. Nanobiotechnol. 2018, 16, 81. [Google Scholar] [CrossRef] [PubMed]
- De la Torre Gomez, C.; Goreham, R.V.; Bech Serra, J.J.; Nann, T.; Kussmann, M. “Exosomics”—A Review of Biophysics, Biology and Biochemistry of Exosomes With a Focus on Human Breast Milk. Front. Genet. 2018, 9, 92. [Google Scholar] [CrossRef] [PubMed]
- Zaborowski, M.P.; Balaj, L.; Breakefield, X.O.; Lai, C.P. Extracellular Vesicles: Composition, Biological Relevance, and Methods of Study. Bioscience 2015, 65, 783–797. [Google Scholar] [CrossRef]
- Kalra, H.; Drummen, G.P.; Mathivanan, S. Focus on Extracellular Vesicles: Introducing the Next Small Big Thing. Int. J. Mol. Sci. 2016, 17, 170. [Google Scholar] [CrossRef]
- Susa, F.; Limongi, T.; Dumontel, B.; Vighetto, V.; Cauda, V. Engineered Extracellular Vesicles as a Reliable Tool in Cancer Nanomedicine. Cancers 2019, 11, 1979. [Google Scholar] [CrossRef] [PubMed]
- Sutaria, D.S.; Badawi, M.; Phelps, M.A.; Schmittgen, T.D. Achieving the Promise of Therapeutic Extracellular Vesicles: The, Devil is in Details of Therapeutic Loading. Pharm. Res. 2017, 34, 1053–1066. [Google Scholar] [CrossRef] [PubMed]
- Gao, J. Exosomes as Novel Bio-Carriers for Gene and Drug Delivery. Int. J. Pharm. 2017, 521, 167–175. [Google Scholar]
- Thèry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, D.J.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atchin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Amreddy, N.; Pareek, V.; Chinnappan, M.; Ahmed, R.; Mehta, M.; Razaq, M.; Munshi, A.; Ramesh, R. Progress in extracellular vesicle biology and their application in cancer medicine. Wires Nanomed. Nanobi. 2020, 12, 4. [Google Scholar] [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Maas, S.L.N.; Breakefield, X.O.; Weaver, A.M. Extracellular Vesicles: Unique Intercellular Delivery Vehicles. Trends Cell Biol. 2017, 27, 172–188. [Google Scholar] [CrossRef] [PubMed]
- Lötvall, J.O.; Hill, A.F.; Hochberg, F.; Buzás, E.I.; Di Vizio, D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: A position statement from the International Society for Extracellular Vesicles. J. Extracell. Vesicles 2014, 3, 26913. [Google Scholar] [CrossRef] [PubMed]
- Caruso, S.; Poon, I.K.H. Apoptotic Cell-Derived Extracellular Vesicles: More Than Just Debris. Front. Immunol. 2018, 9, 1486. [Google Scholar] [CrossRef]
- Wolf, P. The Nature and Significance of Platelet Products in Human Plasma. Br. J. Haematol. 1967, 13, 3. [Google Scholar] [CrossRef] [PubMed]
- Gudbergsson, J.M.; Jønsson, K.; Simonsen, J.B.; Johnsen, K.B. Systematic review of targeted extracellular vesicles for drug delivery–Considerations on methodological and biological heterogeneity. J. Control. Release 2019, 306, 108–120. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.; Fatima, F. Extracellular Vesicles, Tunneling Nanotubes, and Cellular Interplay: Synergies and Missing Links. Front. Mol. Biosci. 2017, 4, 50. [Google Scholar] [CrossRef]
- Mathieu, M.; Martin-Jaular, L.; Lavieu, G.; Théry, C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat. Cell Biol. 2019, 21, 9–17. [Google Scholar] [CrossRef]
- Jabbari, N.; Akbariazar, E.; Feqhhi, M.; Rahbarghazi, R.; Rezaie, J. Breast cancer-derived exosomes: Tumor progression and therapeutic agents. J. Cell. Physiol. 2020. [Google Scholar] [CrossRef]
- Hoshino, A.; Costa-Silva, B.; Shen, T.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef]
- McKelvey, K.J.; Powell, K.L.; Ashton, A.W.; Morris, J.M.; McCracken, S.A. Exosomes: Mechanisms of Uptake. J. Circ. Biomark. 2015, 4, 7. [Google Scholar] [CrossRef]
- Buzás, E.I.; Tóth, E.Á.; Sódar, B.W.; Szabó-Taylor, K.É. Molecular interactions at the surface of extracellular vesicles. Semin. Immunopathol. 2018, 40, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Akuma, P.; Okagu, O.D.; Udenigwe, C.C. Naturally Occurring Exosome Vesicles as Potential Delivery Vehicle for Bioactive Compounds. Front. Sustain. Food Syst. 2019, 3, 23. [Google Scholar] [CrossRef]
- Sil, S.; Dagur, R.S.; Liao, K.; Peeples, E.S.; Hu, G.; Periyasamy, P.; Buch, S. 5 Strategies for the use of Extracellular Vesicles for the Delivery of Therapeutics. J. Neuroimmune Pharm. 2019. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Kosaka, N.; Ochiya, T. Latest advances in extracellular vesicles: From bench to bedside. Sci. Technol. Adv. Mater. 2020, 21, 420–421. [Google Scholar] [CrossRef] [PubMed]
- Batrakova, E.V.; Myung, S.K. Using exosomes, naturally-equipped nanocarriers, for drug delivery. J. Control Release 2015, 219, 396–405. [Google Scholar] [CrossRef]
- Antimisiaris, S.G.; Mourtas, S.; Marazioti, A. Exosomes and Exosome-Inspired Vesicles for Targeted Drug Delivery. Pharmaceutics 2018, 10, 218. [Google Scholar] [CrossRef]
- Chen, C.C.; Liu, L.; Ma, F.; Wong, C.W.; Guo, X.E.; Chacko, J.V.; Farhoodi, H.P.; Zhang, S.X.; Zimak, J.; Ségaliny, A.; et al. Elucidation of Exosome Migration across the Blood-Brain Barrier Model In Vitro. Cell. Mol. Bioeng. 2016, 9, 50. [Google Scholar] [CrossRef]
- Stahl, P.D.; Raposo, G. Extracellular Vesicles: Exosomes and Microvesicles, Integrators of Homeostasis. Physiology 2019, 34, 169–177. [Google Scholar] [CrossRef]
- Tai, Y.; Chu, P.; Lee, B.; Chen, K.; Yang, C.; Kuo, W.; Shen, T. Basics and applications of tumor-derived extracellular vesicles. J. Biomed. Sci. 2019, 26, 35. [Google Scholar] [CrossRef]
- Cappariello, A.; Rucci, N. Tumour-Derived Extracellular Vesicles (EVs): A Dangerous “Message in A Bottle” for Bone. Int. J. Mol. Sci. 2019, 20, 4805. [Google Scholar] [CrossRef]
- Chulpanova, D.S.; Kitaeva, K.V.; James, V.; Rizvanov, A.A.; Solovyeva, V.V. Therapeutic Prospects of Extracellular Vesicles in Cancer Treatment. Front. Immunol. 2018, 9, 1534. [Google Scholar]
- Wu, M.; Wang, G.; Hu, W.; Yao, Y.; Yu, X. Emerging roles and therapeutic value of exosomes in cancer metastasis. Mol. Cancer 2019, 18, 53. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, M.; Villa, A.; Crescenti, D.; Marzagalli, M.; Kuryk, L.; Limonta, P.; Mazzaferro, V.; Ciana, P. Heterologous and cross-species tropism of cancer-derived extracellular vesicles. Theranostics 2019, 9, 5681–5693. [Google Scholar] [CrossRef] [PubMed]
- Peinado, H.; Zhang, H.; Matei, I.; Costa-Silva, B.; Hoshino, A.; Rodrigues, G.; Psaila, B.; Kaplan, B.N.; Bromberg, J.F.; Kang, Y.; et al. Pre-metastatic niches: Organ-specific homes for metastases. Nat. Rev. Cancer 2017, 17, 302–317. [Google Scholar] [CrossRef]
- Villa, F.; Quarto, R.; Tasso, R. Extracellular Vesicles as Natural, Safe and Efficient Drug Delivery Systems. Pharmaceutics 2019, 11, 557. [Google Scholar] [CrossRef]
- Meng, W.; He, C.; Hao, Y.; Wang, L.; Li, L.; Zhu, G. Prospects and challenges of extracellular vesicle-based drug delivery system: Considering cell source. Drug Deliv. 2020, 27, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.M.; André, F.; Amigorena, S.; Soria, J.; Eggermont, A.; Kroemer, G.; Zitvogel, L. Dendritic cell-derived exosomes for cancer therapy. J. Clin. Investig. 2016, 126, 1224–1232. [Google Scholar] [CrossRef]
- Théry, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and Characterization of Exosomes from Cell Culture Supernatants and Biological Fluids. Curr. Protoc. Cell Biol. 2006, 30, 3.22.1–3.22.29. [Google Scholar]
- Li, P.; Kaslan, M.; Lee, S.H.; Yao, J.; Gao, Z. Progress in Exosome Isolation Techniques. Theranostics 2017, 7, 789–804. [Google Scholar] [CrossRef]
- Ramirez, M.I.; Amorim, M.G.; Gadelha, C.; Milic, I.; Welsh, J.A.; Freitas, V.M.; Nawaz, M.; Akbar, N.; Couch, Y.; Makin, L.; et al. Technical challenges of working with extracellular vesicles. Nanoscale 2018, 10, 881–906. [Google Scholar] [CrossRef]
- Ludwig, N.; Whiteside, T.L.; Reichert, T.E. Challenges in Exosome Isolation and Analysis in Health and Disease. Int. J. Mol. Sci. 2019, 20, 4684. [Google Scholar] [CrossRef] [PubMed]
- Konoshenko, M.Y.; Lekchnov, E.A.; Vlassov, A.V.; Laktionov, P.P. Isolation of Extracellular Vesicles: General Methodologies and Latest Trends. Biomed. Res. Int. 2018, 2018, 8545347. [Google Scholar] [CrossRef] [PubMed]
- Livshts, M.A.; Khomyakova, E.; Evtushenko, E.G.; Lazarev, V.N.; Kulemin, N.A.; Semina, S.E.; Generozov, E.V.; Govorun, V.M. Isolation of exosomes by differential centrifugation: Theoretical analysis of a commonly used protocol. Sci. Rep. 2015, 5, 17319. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Kang, M.H.; Jeyaraj, M.; Qasim, M.; Kim, J.H. Review of the Isolation, Characterization, Biological Function, and Multifarious Therapeutic Approaches of Exosomes. Cells 2019, 8, 307. [Google Scholar] [CrossRef]
- Szatanek, R.; Baran, J.; Siedlar, M.; Baj-Krzyworzeka, M. Isolation of extracellular vesicles: Determining the correct approach (Review). Int J. Mol. Med. 2015, 36, 11–17. [Google Scholar] [CrossRef]
- Coumans, F.A.W.; Brisson, A.R.; Buzas, E.I.; Dignat-George, F.; Drees, E.E.E.; El-Andaloussi, S.; Emanueli, C.; Gasecka, A.; Hendrix, A.; Hill, A.F.; et al. Methodological Guidelines to Study Extracellular Vesicles. Circ. Res. 2017, 120, 1632–1648. [Google Scholar] [CrossRef]
- Luan, X.; Sansanaphongpricha, K.; Myers, I.; Chen, H.; Yuan, H.; Sun, D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharm. Sin. 2017, 38, 754–763. [Google Scholar] [CrossRef]
- Srivastava, A.; Amreddy, N.; Babu, A.; Panneerselvam, J.; Mehta, M.; Muralidharan, R.; Chen, A.; Zhao, Y.D.; Razaq, M.; Riedinger, N.; et al. Nanosomes carrying doxorubicin exhibit potent anticancer activity against human lung cancer cells. Sci. Rep. 2016, 6, 38541. [Google Scholar] [CrossRef]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Mahajan, V.; Deygen, I.; Klyachko, N.L.; Inskoe, E.; Piroyan, A.; Sokolsky, M.; Okolie, O.; et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine 2016, 12, 655–664. [Google Scholar] [CrossRef]
- Dumontel, B.; Susa, F.; Limongi, T.; Canta, M.; Racca, L.; Chiodoni, A.; Garino, N.; Chiabotto, G.; Centomo, M.L.; Pignochino, Y.; et al. ZnO nanocrystals shuttled by extracellular vesicles as effective Trojan nano-horses against cancer cells. Nanomedicine 2019, 14, 2815–2833. [Google Scholar] [CrossRef]
- Haney, M.J.; Klyachko, N.L.; Zhao, Y.; Gupta, R.; Plotnikova, E.G.; He, Z.; Patel, T.; Piroyan, A.; Sokolsky, M.; Kabanov, A.V.; et al. Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J. Control. Release 2015, 207, 18–30. [Google Scholar] [CrossRef]
- Illes, B.; Hirschle, P.; Barnert, S.; Cauda, V. Engelke Hanna.Exosome-Coated Metal–Organic Framework Nanoparticles: An Efficient Drug Delivery Platform. Chem. Mater. 2017, 10, 24638–24647. [Google Scholar]
- Betzer, O.; Perets, N.; Angel, A.; Motiei, M.; Sadan, T.; Yadid, G.; Offen, D.; Popovtzer, R. In Vivo Neuroimaging of Exosomes Using Gold Nanoparticles. ACS Nano 2017, 11, 10883–10893. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Zhuang, X.; Xiang, X.; Liu, Y.; Zhang, S.; Li, C.; Barnes, S.; Grizzle, W.; Miller, D.; Zhang, H.G. A Novel Nanoparticle Drug Delivery System: The Anti-inflammatory Activity of Curcumin Is Enhanced When Encapsulated in Exosomes. Mol. Ther. 2010, 18, 1606–1614. [Google Scholar] [CrossRef] [PubMed]
- Fuhrmann, G.; Serio, A.; Mazo, M.; Nair, R.; Stevens, M.M. Active loading into extracellular vesicles significantly improves the cellular uptake and photodynamic effect of porphyrins. J. Control. Release 2015, 205, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Hood, J.L.; Scott, M.J.; Wickline, S.A. Maximizing Exosome Colloidal Stability Following Electroporation. Anal. Biochem. 2013, 448, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Wahlgren, J.; De, L.; Karlson, T.; Brisslert, M.; Vaziri Sani, F.; Telemo, E.; Sunnerhagen, P.; Valadi, H. Plasma exosomes can deliver exogenous short interfering RNA to monocytes and lymphocytes. Nucleic Acids Res. 2012, 40, e130. [Google Scholar] [CrossRef]
- Shtam, T.A.; Kovalev, R.A.; Varfolomeeva, E.Y.; Makarov, E.M.; Kil, Y.V.; Filatov, M.V. Exosomes are natural carriers of exogenous siRNA to human cells in vitro. Cell Commun. Signal. 2013, 11, 88. [Google Scholar] [CrossRef]
- Mentkowski, K.I.; Snitzer, J.D.; Rusnak, S.; Lang, J.K. Therapeutic Potential of Engineered Extracellular Vesicles. Aaps J. 2018, 20, 50. [Google Scholar] [CrossRef]
- Stickney, Z.; Losacco, J.; Mc Devitt, S.; Zhang, Z.; Lu, B. Development of exosome surface display technology in living human cells. Biochem. Biophys. Res. Commun. 2016, 472, 53–59. [Google Scholar] [CrossRef]
- Alvarez-Erviti, L.; Seow, Y.; Yin, H.; Betts, C.; Lakhal, S.; Wood, M.J. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011, 29, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Li, S.; Song, J.; Ji, T.; Zhu, M.; Anderson, G.J.; Wei, J.; Nie, G. A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy. Biomaterials 2014, 35, 2383–2390. [Google Scholar] [CrossRef] [PubMed]
- Kooijmans, S.A.; Aleza, C.G.; Roffler, S.R.; Van Solinge, W.W.; Vader, P.; Schiffelers, R.M. Display of GPI-anchored anti-EGFR nanobodies on extracellular vesicles promotes tumour cell targeting. J. Extracell. Vesicles. 2016, 5, 31053. [Google Scholar] [CrossRef] [PubMed]
- Ohno, S.; Takanashi, M.; Sudo, K.; Ueda, S.; Ishikawa, A.; Matsuyama, N.; Fujita, K.; Mizutani, T.; Ohgi, T.; Ochiya, T.; et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol. Ther. 2013, 21, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Hartman, Z.C.; Wei, J.; Glass, O.K.; Guo, H.; Lei, G.; Yang, X.Y.; Osada, T.; Hobeika, A.; Delcayre, A.; Le Pecq, J.B.; et al. Increasing vaccine potency through exosome antigen targeting. Vaccine 2011, 29, 9361–9367. [Google Scholar] [CrossRef]
- Rountree, R.B.; Mandl, S.J.; Nachtwey, J.M.; Dalpozzo, K.; Do, L.; Lombardo, J.R.; Schoonmaker, P.L.; Brinkmann, K.; Dirmeier, U.; Laus, R.; et al. Exosome targeting of tumor antigens expressed by cancer vaccines can improve antigen immunogenicity and therapeutic efficacy. Cancer Res. 2011, 71, 5235–5244. [Google Scholar] [CrossRef]
- Delcayre, A.; Estelles, A.; Sperinde, J.; Roulon, T.; Paz, P.; Aguilar, B.; Villanueva, J.; Khine, S.S.; Le Pecq, J.B. Exosome Display technology: Applications to the development of new diagnostics and therapeutics. Blood Cells Mol. Dis. 2005, 35, 158–168. [Google Scholar] [CrossRef]
- Wang, J.; Dong, Y.; Li, Y.; Li, W.; Cheng, K.; Qian, Y.; Xu, G.; Zhang, X.; Hu, L.; Chen, P.; et al. Designer Exosomes for Active Targeted Chemo-Photothermal Synergistic Tumor Therapy. Adv. Funct. Mater. 2018, 28, 1707360. [Google Scholar] [CrossRef]
- Zhang, W.; Yu, Z.L.; Wu, M.; Ren, J.G.; Xia, H.F.; Sa, G.L.; Zhu, J.Y. Magnetic and Folate Functionalization Enables Rapid Isolation and Enhanced Tumor-Targeting of Cell-Derived Microvesicles. Acs Nano 2017, 11, 277–290. [Google Scholar] [CrossRef]
- Dong, Z.; Zhao, Y.; Zhang, P. Folate-Engineered Microvesicles for Enhanced Target and Synergistic Therapy toward Breast Cancer. Acs Appl. Mater. Interfaces 2017, 9, 5100–5108. [Google Scholar]
- Chen, G.; Zhu, J.Y.; Zhang, Z.L.; Zhang, W.; Ren, J.G.; Wu, M.; Hong, Z.Y.; Lv, C.; Pang, D.W.; Zhao, Y.F. Transformation of Cell-Derived Microparticles into Quantum-Dot-Labeled Nanovectors for Antitumor siRNA Delivery. Angew. Chem. Int. Ed. Engl. 2014, 54, 1036–1040. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, W.; Zhang, L.; Ban, L.; Chen, P.; Du, W.; Feng, X.; Liu, B.F. Chemically edited exosomes with dual ligand purified by microfluidic device for active targeted drug delivery to tumor cells. Acs Appl. Mater. Interfaces. 2017, 9, 27441–27452. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.P.; Mardini, O.; Ericsson, M.; Prabhakar, S.; Maguire, C.A.; Chen, J.W.; Tannous, B.A.; Breakefield, X.O. Dynamic biodistribution of extracellular vesicles in vivo using a multimodal imaging reporter. Acs Nano 2014, 8, 483–494. [Google Scholar] [CrossRef] [PubMed]
- Morishita, M.; Takahashi, Y.; Nishikawa, M.; Ariizumi, R.; Takakura, Y. Enhanced class I tumor antigen presentation via cytosolic delivery of exosomal cargos by tumor cell-derived exosomes displaying a pH-sensitive fusogenic peptide. Mol. Pharm. 2017, 14, 4079–4086. [Google Scholar] [CrossRef] [PubMed]
- Pascucci, L.; Coccè, V.; Bonomi, A.; Ami, D.; Ceccarelli, P.; Ciusani, E.; Viganò, L.; Locatelli, A.; Sisto, F.; Doglia, S.M.; et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: A new approach for drug delivery. J. Control. Release 2014, 192, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Gao, H.; Lv, P.; Liu, J.; Liu, G. Extracellular vesicles as an efficient nanoplatform for the delivery of therapeutics. Hum. Vaccin. Immunother. 2017, 13, 2678–2687. [Google Scholar] [CrossRef]
- Tang, K.; Zhang, Y.; Zhang, H.; Xu, P.; Liu, J.; Ma, J.; Lv, M.; Li, D.; Katirai, F.; Shen, G.X.; et al. Delivery of chemotherapeutic drugs in tumour cell-derived microparticles. Nat. Commun. 2012, 3, 1282. [Google Scholar] [CrossRef]
- Mulens-Arias, V.; Nicolás-Boluda, A.; Silva, A.K.A.; Gazeau, F. Theranostic Iron Oxide Nanoparticle Cargo Defines Extracellular Vesicle-Dependent Modulation of Macrophage Activation and Migratory Behavior. Adv. Biosyst. 2018, 2, 1800079. [Google Scholar] [CrossRef]
- Silva, A.K.A.; Kolosnjaj-Tabi, J.; Bonneau, S.; Marangon, I.; Boggetto, N.; Aubertin, K.; Clément, O.; Bureau, M.F.; Luciani, N.; Gazeau, F.; et al. Magnetic and Photoresponsive Theranosomes: Translating Cell-Released Vesicles into Smart Nanovectors for Cancer Therapy. Acs Nano 2013, 7, 6. [Google Scholar] [CrossRef]
- Piffoux, M.; Silva, A.K.A.; Lugagne, J.B.; Hersen, P.; Wilhelm, C.; Gazeau, F. Extracellular Vesicle Production Loaded with Nanoparticles and Drugs in a Trade-off between Loading, Yield and Purity: Towards a Personalized Drug Delivery System. Adv. Biosys. 2017, 1, 1700044. [Google Scholar] [CrossRef]
- Lou, G.; Song, X.; Yang, F.; Wu, S.; Wang, J.; Chen, Z.; Liu, Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J. Hematol. Oncol. 2015, 8, 122. [Google Scholar] [CrossRef] [PubMed]
- Shimbo, K.; Miyaki, S.; Ishitobi, H.; Kato, Y.; Kubo, T.; Shimose, S.; Och, M. Exosome-formed synthetic microRNA-143 is transferred to osteosarcoma cells and inhibits their migration. Biochem. Biophys. Res. Commun. 2014, 445, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Katakowski, M.; Bullera, B.; Zheng, X.; Lu, Y.; Rogers, T.; Osobamiro, O.; Shu, W.; Jiang, F.; Chopp, M. Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett. 2013, 335, 201–204. [Google Scholar] [CrossRef]
- Maugeri, M.; Nawaz, M.; Papadimitriou, A.; Angerfors, A.; Camponeschi, A.; Na, M.; Hölttä, M.; Skantze, P.; Johansson, S.; Sundqvist, M.; et al. Linkage between endosomal escape of LNP-mRNA and loading into EVs for transport to other cells. Nat. Commun. 2019, 10, 4333. [Google Scholar] [CrossRef] [PubMed]
- Tian, T.; Wang, Y.; Wang, H.; Zhu, Z.; Xiao, Z. Visualizing of the cellular uptake and intracellular trafficking of exosomes by live-cell microscopy. J. Cell. Biochem. 2010, 111, 488–496. [Google Scholar] [CrossRef] [PubMed]
- Smyth, T.; Petrova, K.; Payton, N.M.; Persaud, I.; Redzic, J.S. Surface Functionalization of Exosomes Using Click Chemistry. Bioconjug. Chem. 2014, 25, 1777–1784. [Google Scholar] [CrossRef]
- Kooijmans, S.A.A.; Fliervoet, L.A.L.; Van Der Meel, R.; Fens, M.H.A.M.; Heijnen, H.F.G.; Van Bergen en Henegouwen, P.M.P.; Vader, P.; Schiffelers, R.M. PEGylated and targeted extracellular vesicles display enhanced cell specificity and circulation time. J. Control. Release. 2016, 224, 77–85. [Google Scholar] [CrossRef]
- Choi, E.S.; Song, J.; Kang, Y.Y.; Mok, H. Mannose-Modified Serum Exosomes for the Elevated Uptake to Murine Dendritic Cells and Lymphatic Accumulation. Macromol. Biosci. 2019, 19, e1900042. [Google Scholar] [CrossRef]
- Kim, M.S.; Haney, M.J.; Zhao, Y.; Yuana, D.; Deygen, I.; Batrakova, E.V. Engineering macrophage-derived exosomes for targeted paclitaxel delivery to pulmonary metastases: In vitro and in vivo evaluations. Nanomed. 2018, 14, 195–204. [Google Scholar] [CrossRef]
- Pi, F.; Binzel, D.W.; Lee, T.J.; Li, Z.; Sun, M.; Rychahou, P.; Li, H.; Haque, F.; Wang, S.; Croce, C.M.; et al. Nanoparticle orientation to control RNA loading and ligand display on extracellular vesicles for cancer regression. Nat. Nanotechnol. 2017, 13, 82–89. [Google Scholar] [CrossRef]
- Jia, G.; Han, Y.; An, Y.; Ding, Y.; He, C.; Wang, X.; Tang, Q. NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials 2018, 178, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Nakase, I.; Futaki, S. Combined treatment with a pH-sensitive fusogenic peptide and cationic lipids achieves enhanced cytosolic delivery of exosomes. Sci. Rep. 2015, 5, 10112. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Liu, C.; Long, L.; Ren, Y.; Zhang, S.; Chang, X.; Qian, X.; Jia, H.; Zhao, J.; Sun, J.; et al. Blood Exosomes Endowed with Magnetic and Targeting Properties for Cancer Therapy. ACS Nano 2016, 10, 3323–3333. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Ran, N.; Dong, X.; Zuo, B.; Yang, R.; Zhou, Q.; Moulton, H.M.; Seow, Y.; Yin, H. Anchor peptide captures, targets, and loads exosomes of diverse origins for diagnostics and therapy. Sci. Transl. Med. 2018, 10, 444. [Google Scholar] [CrossRef]
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.W.; Anchordoquy, T.J. Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J. Control. Release 2015, 199, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Royo, F.; Cossío, U.; de Angulo, A.R.; Llop, J.; Falcon-Perez, J.M. Modification of the glycosylation of extracellular vesicles alters their biodistribution in mice. Nanoscale 2019, 11, 1531–1537. [Google Scholar] [CrossRef]
- Dusoswa, S.A.; Horrevorts, S.K.; Ambrosini, M.; Kalay, H.; Paauw, N.J.; Nieuwland, R.; Pegtel, M.D.; Würdinger, T.; Van Kooyk, Y.; Garcia-Vallejo, J.J. Glycan modification of glioblastoma-derived extracellular vesicles enhances receptor-mediated targeting of dendritic cells. J. Extracell Vesicles 2019, 8, 1648995. [Google Scholar] [CrossRef]
- Hung, M.E.; Leonard, J.N. Stabilization of Exosome-targeting Peptides via Engineered Glycosylation. J. Biol. Chem. 2015, 290, 8166–8172. [Google Scholar] [CrossRef]
- Lin, Y.; Wu, J.; Gu, W.; Huang, Y.; Tong, Z.; Huang, L.; Tan, J. Exosome–Liposome Hybrid Nanoparticles Deliver CRISPR/Cas9 System in MSCs. Adv. Sci. 2018, 5, 1700611. [Google Scholar] [CrossRef]
- Sato, Y.T.; Umezaki, K.; Sawada, S.; Mukai, S.; Sasaki, Y.; Harada, N.; Shiku, H.; Akiyoshi, K. Engineering hybrid exosomes by membrane fusion with liposomes. Sci. Rep. 2016, 6, 21933. [Google Scholar] [CrossRef]
- Piffoux, M.; Silva, A.K.A.; Wilhelm, C.; Gazeau, F.; Tareste, D. Modification of Extracellular Vesicles by Fusion with Liposomes for the Design of Personalized Biogenic Drug Delivery Systems. Acs Nano 2018, 12, 6830–6842. [Google Scholar] [CrossRef] [PubMed]
- Rayamajhi, S.; Nguyen, T.D.T.; Marasini, R.; Aryal, S. Macrophage-derived exosome-mimetic hybrid vesicles for tumor targeted drug delivery. Acta Biomater. 2019, 94, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Ramasubramanian, L.; Kumar, P.; Wang, A. Engineering Extracellular Vesicles as Nanotherapeutics for Regenerative Medicine. Biomolecules 2019, 10, 48. [Google Scholar] [CrossRef] [PubMed]
- Presolski, S.I.; Hong, V.P.; Finn, M.G. Copper-Catalyzed Azide–Alkyne Click Chemistry for Bioconjugation. Curr. Protoc. Chem. Biol. 2011, 3, 153–162. [Google Scholar] [CrossRef]
- Jewett, J.C.; Bertozzi, C.R. Cu-free click cycloaddition reactions in chemical biology. Chem. Soc. Rev. 2010, 39, 1272–1279. [Google Scholar] [CrossRef]
- Cataldi, M.; Vigliotti, C.; Mosca, T.; Cammarota, M.R.; Capone, D. Emerging Role of the Spleen in the Pharmacokinetics of Monoclonal Antibodies, Nanoparticles and Exosomes. Int. J. Mol. Sci. 2017, 18, 1249. [Google Scholar] [CrossRef]
- Liang, Y.; Eng, W.S.; Colquhoun, D.R.; Dinglasan, R.R.; Graham, D.R.; Mahal, L.K. Complex N-Linked Glycans Serve as a Determinant for Exosome/Microvesicle Cargo Recruitment. J. Biol Chem. 2014, 289, 32526–32537. [Google Scholar] [CrossRef]
- Williams, C.; Pazos, R.; Royo, F.; González, E.; Roura-Ferrer, M.; Martinez, A.; Gamiz, J.; Reichardt, N.C.; Falcón-Pérez, J.M. Assessing the role of surface glycans of extracellular vesicles on cellular uptake. Sci. Rep. 2019, 9, 11920. [Google Scholar] [CrossRef]
- Cai, L.; Gu, Z.; Zhong, J.; Wen, D.; Chen, G.; He, L.; Wu, J.; Gu, Z. Advances in glycosylation-mediated cancer-targeted drug delivery. Drug Discov. Today 2018, 23, 1126–1138. [Google Scholar] [CrossRef]
- Mereiter, S.; Balmaña, M.; Campos, D.; Gomes, J.; Reis, C.A. Glycosylation in the Era of Cancer-Targeted Therapy: Where Are We Heading? Cancer Cell. 2019, 36, 6–16. [Google Scholar] [CrossRef]
- Lu, M.; Huang, Y. Bioinspired exosome-like therapeutics and delivery nanoplatforms. Biomaterials 2020, 242, 119925. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.C.; Kim, O.Y.; Yoon, C.M.; Choi, D.S.; Roh, T.Y.; Park, J.; Nilsson, J.; Lötvall, J.; Kim, Y.K.; Gho, Y.S. Bioinspired exosome-mimetic nanovesicles for targeted delivery of chemotherapeutics to malignant tumors. ACS Nano. 2013, 7, 7698–7710. [Google Scholar] [CrossRef] [PubMed]
- García-Manrique, P.; Gutiérrez, G.; Blanco-López, M.C. Fully Artificial Exosomes: Towards New Theranostic Biomaterials. Trends Biotechnol. 2018, 36, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Gangadaran, P.; Kalimuthu, S.; Oh, J.M.; Baek, S.H.; Jeong, S.Y.; Lee, S.W.; Lee, J.; Ahn, B.C. Novel alternatives to extracellular vesicle-based immunotherapy-exosome mimetics derived from natural killer cells. Artif. Cells Nanomed. Biotechnol. 2018, 46, S166–S179. [Google Scholar] [CrossRef]
- Wu, J.Y.; Ji, A.L.; Wang, Z.X.; Qiang, G.H.; Qu, Z.; Wu, J.H.; Jiang, C.P. Exosome-mimetic nanovesicles from hepatocytes promote hepatocyte proliferation in vitro and liver regeneration in vivo. Sci Rep. 2018, 8, 2471. [Google Scholar] [CrossRef]
- Hwang, D.W.; Choi, H.; Jang, S.C.; Yoo, M.Y.; Park, J.Y.; Choi, N.E.; Oh, H.J.; Ha, S.; Lee, Y.S.; Jeong, J.M.; et al. Noninvasive imaging of radiolabeled exosome-mimetic nanovesicle using 99mTc-HMPAO. Sci. Rep. 2015, 5, 15636. [Google Scholar] [CrossRef]
- Jo, W.; Kim, J.; Yoon, J.; Jeong, D.; Cho, S.; Jeong, H.; Yoon, Y.J.; Kim, S.C.; Ghod, Y.S.; Park, J. Large-scale generation of cell-derived nanovesicles. Nanoscale. 2014, 6, 12056–12064. [Google Scholar] [CrossRef]
- Jeong, D.; Jo, W.; Yoon, J.; Kim, J.; Gianchandani, S.; Gho, Y.S.; Park, J. Nanovesicles engineered from ES cells for enhanced cell proliferation. Biomaterials 2014, 35, 9302–9310. [Google Scholar] [CrossRef]
- Yang, Z.; Xie, J.; Zhu, J.; Kang, C.; Chiang, C.; Wang, X.; Wang, X.; Kuang, T.; Chen, F.; Chen, Z.; et al. Functional exosome-mimic for delivery of siRNA to cancer: In vitro and in vivo evaluation. J. Control. Release. 2016, 243, 160–171. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Prakash, G.; Lakshmi, R.R.; Liya, Z.; Min, O.J.; Won, L.H.; Arunnehru, G.; Hwan, B.S.; Young, J.S.; Sang-Woo, L.; et al. A New Approach for Loading Anticancer Drugs Into Mesenchymal Stem Cell-Derived Exosome Mimetics for Cancer Therapy. Front. Pharm. 2018, 9, 1116. [Google Scholar]
- Tao, S.C.; Rui, B.Y.; Wang, Q.Y.; Zhou, D.; Zhang, Y.; Guo, S.C. Extracellular vesicle-mimetic nanovesicles transport LncRNA-H19 as competing endogenous RNA for the treatment of diabetic wounds. Drug Deliv. 2018, 25, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.; Kim, S.R.; Kim, D.K.; Seo, M.W.; Lee, C.; Lee, H.M.; Oh, J.E.; Choi, E.Y.; Lee, D.S.; Gho, Y.S.; et al. In Vivo Differentiation of Therapeutic Insulin-Producing Cells from Bone Marrow Cells via Extracellular Vesicle-Mimetic Nanovesicles. Acs Nano 2015, 9, 11718–11727. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, J.Y.; Cho, R.; Shin, D.M.; Lee, S.W.; Oh, Y.M. Adipose stem cell-derived nanovesicles inhibit emphysema primarily via an FGF2-dependent pathway. Exp. Mol. Med. 2017, 49, e284. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Svennerholm, K.; Shelke, G.V.; Bandeira, E.; Lässer, C.; Jang, S.C.; Chandode, R.; Gribonika, I. Lötvall, J.; Mesenchymal stromal cell-derived nanovesicles ameliorate bacterial outer membrane vesicle-induced sepsis via IL-10. Stem Cell Res. Ther. 2019, 10, 231. [Google Scholar] [CrossRef]
- Choo, Y.W.; Kang, M.; Kim, H.Y.; Han, J.; Kang, S.; Lee, J.R.; Jeong, G.J.; Kwon, S.P.; Song, S.Y.; Go, S.; et al. M1 Macrophage-Derived Nanovesicles Potentiate the Anticancer Efficacy of Immune Checkpoint Inhibitors. Acs Nano 2018, 12, 8977–8993. [Google Scholar] [CrossRef]
- Yoona, J.; Joa, W.; Jeongb, D.; Kimb, J.; Jeonga, H.; Parkab, J. Generation of nanovesicles with sliced cellular membrane fragments for exogenous material delivery. Biomaterials 2015, 59, 12–20. [Google Scholar] [CrossRef]
- Jo, W.; Jeong, D.; Kim, J.; Cho, S.; Jang, S.C.; Han, C.; Kang, J.Y.; Gho, Y.S.; Park, J. Microfluidic fabrication of cell-derived nanovesicles as endogenous RNA carriers. Lab. Chip 2014, 14, 1261–1269. [Google Scholar] [CrossRef]
- De La Peña, H.; Madrigal, J.A.; Rusakiewicz, S.; Bencsik, M.; Cave, G.W.; Selman, A.; Rees, R.C.; Travers, P.J.; Dodi, I.A. Artificial exosomes as tools for basic and clinical immunology. J. Immunol. Methods. 2009, 344, 121–132. [Google Scholar] [CrossRef]
- Lu, M.; Zhao, X.; Xing, H.; Xun, Z.; Zhu, S.; Lang, L.; Yang, T.; Cai, C.; Wang, D.; Ding, P. Comparison of exosome-mimicking liposomes with conventional liposomes for intracellular delivery of siRNA. Int. J. Pharm. 2018, 550, 100–113. [Google Scholar] [CrossRef]
- Lu, M.; Zhao, X.; Xing, H.; Liu, H.; Lang, L.; Yang, T.; Xun, Z.; Wang, D.; Ding, P. Cell-free synthesis of connexin 43-integrated exosome-mimetic nanoparticles for siRNA delivery. Acta Biomater. 2019, 96, 517–536. [Google Scholar] [CrossRef]
- Vázquez-Ríos, A.J.; Molina-Crespo, Á.; Bouzo, B.L.; López-López, R.; Moreno-Bueno, G.; De La Fuente, M. Exosome-mimetic nanoplatforms for targeted cancer drug delivery. J. Nanobiotechnol. 2019, 17, 85. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lostao, L.; Garcí´a-Alvarez, F.; Basáñez, G.; Alegre-Aguarón, E.; Desportes, P.; Larrad, L.; Naval, J.; Martí´nez-Lorenzo, M.J.; Anel, A. Liposome-bound APO2L/TRAIL is an effective treatment in a rabbit model of rheumatoid arthritis. Arthritis Rheum. 2010, 62, 2272–2282. [Google Scholar] [CrossRef] [PubMed]
- De Miguel, D.; Basáñez, G.; Sánchez, D.; Malo, P.G.; Marzo, I.; Larrad, L.; Naval, J.; Pardo, J.; Anel, A.; Martinez-Lostao, L. Liposomes Decorated with Apo2L/TRAIL Overcome Chemoresistance of Human Hematologic Tumor Cells. Mol. Pharm. 2013, 10, 893–904. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Chang, S.; Wang, Z.; Zhao, X.; Chen, D. A novel micro-emulsion and micelle assembling method to prepare DEC205 monoclonal antibody coupled cationic nanoliposomes for simulating exosomes to target dendritic cells. Int. J. Pharm. 2015, 491, 105–112. [Google Scholar] [CrossRef]
- Zhang, K.L.; Wang, Y.J.; Sun, J.; Zhou, J.; Xing, C.; Huang, G.; Li, J.; Yang, H. Artificial chimeric exosomes for anti-phagocytosis and targeted cancer therapy. Chem Sci. 2019, 10, 1555–1561. [Google Scholar] [CrossRef]
- Molinaro, R.; Corbo, C.; Martinez, J.O.; Taraballi, F.; Evangelopoulos, M.; Minardi, S.; Yazdi, I.K.; Zhao, P.; De Rosa, E.; Sherman, M.; et al. Biomimetic proteolipid vesicles for targeting inflamed tissues. Nat. Mater. 2016, 15, 1037–1046. [Google Scholar] [CrossRef]
- Molinaro, R.; Evangelopoulos, M.; Hoffman, J.R.; Corbo, C.; Taraballi, F.; Martinez, J.O.; Hartman, K.A.; Cosco, D.; Costa, G.; Romeo, I.; et al. Design and Development of Biomimetic Nanovesicles Using a Microfluidic Approach. Adv. Mater. 2018, 30, e1702749. [Google Scholar] [CrossRef]

| Parental Cells | Cargo | Loading Conditions | Recipient Cells | Treatment Condition | Application | Reference |
|---|---|---|---|---|---|---|
| Co-Incubation | ||||||
| H1299 and YRC9 | Doxorubicin conjugated with gold NPs | Incubated at 37 °C with 250 rpm for 2 h | H1299, A549, MRC9, and Dox-sensitive HCASM | 1 × 105 cells per well and EVs with the equivalent of 5 μg Dox per well | Anticancer activity against human lung cancer cells | [48] |
| RAW 264.7 | Paclitaxel | Incubated at 37 °C for 1 hour with shaking | MDCKWT, MDCKMDR1, and 3LL-M27 IN VIVO: C57BL/6 mice | 5000 cells per well and exosomes IN VIVO: i.n. 107 particles/10 μL × 2 | Overcome multiple drug resistance in cancer cells | [49] |
| KB | ZnO nanocrystals | Various loading conditions | KB | 3 × 104 cells per well and EVs with the equivalent of 15 μg/mL of ZnO nanocrystals | Treatment of cancer cells | [50] |
| RAW 264.7 | Enzyme catalase | Incubated at RT for 18 hours | PC12 IN VIVO: C57BL/6 female mice | 50,000 cells per well and EVs 230 µg total protein/mL IN VIVO: i.n. or i.v. 2.4 × 1010 EVs | Parkinson’s disease therapy | [51] |
| HeLa | MOF loaded with calcein | Incubated at 37 °C for 1.5 h with shaking | HeLa | 1000 cells for each EV concentration (10−140 μg/mL) | Efficient drug delivery platform | [52] |
| MSCs | Glucose-coated gold NPs | Incubated for 3 h at 37 °C | IN VIVO: C57bl/6 male mice | IN VIVO: i.n. and i.v. 2.8 × 109 EVs | In vivo neuroimaging | [53] |
| EL-4, MDA-MB231, 4T-1 | Curcumin | Mixed at 22 °C, then sucrose gradient centrifugation | RAW 264.7 IN VIVO: 7- to 10-week female C57BL/6j mice | Exosomal curcumin 20 µmol/l, LPS 50 ng/mL IN VIVO: i.p. 4 mg/kg exosomal curcumin, 18.75 mg/kg LPS | Deliver anti-inflammatory agents to activated myeloid cells in vivo | [54] |
| MDAs, hUVECs, hMSCs and hESCs | Porphyrins of different hydrophobicities | Incubated at RT for 10 min | MDA-MB231 | 20,000 cells per well and EVs diluted 1:2 from the Stock solution (1.5 mg/mL of Por) | Improve the cellular uptake and photodynamic effect of porphyrins | [55] |
| Electroporation | ||||||
| RAW 264.7 | Paclitaxel | 1000 kV for 5 ms, then incubated at 37 °C for 30 min | MDCKWT, MDCKMDR1 and 3LL-M27 | 5000 cells per well and exosomes IN VIVO: i.n. 107 particles/10 μL × 2 | Overcome multiple drug resistance in cancer cells | [49] |
| MDAs, hUVECs, hMSCs and hESCs | Porphyrins of different hydrophobicities | 200 Ω, 500 μF, 200 mV, and pulse time of 20–30 ms | MDA-MB231 | 20,000 cells per well and EVs diluted 1:2 from the Stock solution (1.5 mg/mL of Por) | Improve the cellular uptake and photodynamic effect of porphyrins | [55] |
| B16-F10 | 5 nm SPIONs | High voltage setting | The formulation was not tested with cells or animals | The formulation was not tested with cells or animals | Maximizing exosome colloidal stability | [56] |
| HeLa, HTB-177, CD14+ monocytes and CD14− lymphocytes | siRNA | 0.150 kV/100 µF | HTB-177, CD14+ monocytes, and CD14− lymphocytes | 0.5 × 104 cells per well and 30 μL of exosomes with siRNA at 2 μmol/mL | Deliver exogenous siRNA to monocytes and lymphocytes | [57] |
| Sonication | ||||||
| RAW 264.7 | Paclitaxel | 20% amplitude, 6 cycles of 30 s on/off, 2 min pause, then incubated at 37 °C for 60 min | MDCKWT, MDCKMDR1 and 3LL-M27 | 5000 cells per well and exosomes IN VIVO: i.n. 107 particles/10 μL × 2 | Overcome multiple drug resistance in cancer cells | [49] |
| RAW 264.7 | Enzyme catalase | Sonicated twice at 500 v, 2 kHz, 20% power, 6 cycles by 4 s pulse/2 s pause | Neuronal PC12 IN VIVO: C57BL/6 female mice | 50,000 cells per well and EVs 230 µg total protein/mL IN VIVO: i.n. or i.v. 2.4 × 1010 EVs | Parkinson’s disease therapy | [51] |
| Extrusion | ||||||
| RAW 264.7 | Enzyme catalase | Extruded (x10 times) with 200 nm pores diameter | Neuronal PC12 IN VIVO: C57BL/6 female mice | 50,000 cells per well and EVs 230 µg total protein/mL IN VIVO: i.n. or i.v. 2.4 × 1010 EVs | Parkinson’s disease therapy | [51] |
| MDAs, hUVECs, hMSCs and hESCs | Porphyrins of different hydrophobicities | Extruded at 42 °C (31 times) with 400 nm pore diameter | MDA-MB231 | 20,000 cells per well and EVs diluted 1:2 from the Stock solution (1.5 mg/mL of Por) | Improve the cellular uptake and photodynamic effect of porphyrins | [55] |
| Freeze–Thaw | ||||||
| RAW 264.7 | Enzyme catalase | Incubated for 30 min, then −80° C, then RT (three times) | Neuronal PC12 IN VIVO: C57BL/6 female mice | 50,000 cells per well and EVs 230 µg total protein/mL IN VIVO: i.n. or i.v. 2.4 × 1010 EVs | Parkinson’s disease therapy | [51] |
| Chemical-Based Transfection | ||||||
| MDAs, hUVECs, hMSCs and hESCs | Porphyrins of different hydrophobicities | Addition of 0.1 mg/mL saponin at RT for 10 min | MDA-MB231 | 20,000 cells per well and EVs diluted 1:2 from the Stock solution (1.5 mg/mL of Por) | Improve the cellular uptake and photodynamic effect of porphyrins | [55] |
| HeLa, HTB-177, CD14+ monocytes and CD14− lymphocytes | siRNA | Addition of HiPerFect, then incubated for 10 min at RT | HTB-177, CD14+ monocytes, and CD14− lymphocytes | 0.5 × 104 cells per well and 30 μL of exosomes with siRNA at 2 μmol/mL | Deliver exogenous siRNA to monocytes and lymphocytes | [57] |
| RAW 264.7 | Enzyme catalase | Addition of 0.2% saponin, shaker for 20 min at RT, then incubated at RT for 18 hours | Neuronal PC12 IN VIVO: C57BL/6 female mice | 50,000 cells per well and EVs 230 µg total protein/mL IN VIVO: i.n. or i.v. 2.4 × 1010 EVs | Parkinson’s disease therapy | [51] |
| HeLa and HT1080 | siRNA | Addition of lipofectamine and incubated for 30 min at RT | HeLa and HT1080 | 0.5 × 106 cells per well and varying amounts of exosomes (0–460 µg) | Deliver siRNA to recipient cells in vitro | [58] |
| Parental Cells | Functionalization | Cell Engineering Conditions | Recipient Cells | Treatment Conditions | Application | Reference |
|---|---|---|---|---|---|---|
| HEK293 | Tetraspanins (CD63, CD9, CD81) | Transfected at 40~60% confluency using plasmid DNA (1–2 µg/well) for 48 h with PureFection Transfection Reagent or FuGENE6 t.r. | HEK293 | Cells at confluency of 80% and 50 µg of exosomes | Tracking, imaging and targeting drug delivery | [60] |
| GM-CSF | Lamp-2b fused to the neuron-specific RVG peptide | Transfected 4 days using 5 µg of pLamp2b and 5 µl of TransIT LT1 t.r. | C2C12 and Neuro2A IN VIVO: C57BL/6 mice |
Exosomes (12 µg proteins) and 400 nanomoles of siRNA IN VIVO: i.v. 150 µg of exosomes | Delivering of siRNA to the brain in mice with a reduced immunogenicity | [61] |
| Immaturedendritic cells (imDCs) | Lamp2b fused to CRGDKGPDC | Transfected with the vector expressing iRGD-Lamp2b fusion proteins using Lipofectamine 2000 t.r. | MDA-MB-231 IN VIVO: BALB/c nude mice | 2 mM Dox-loaded exosomes IN VIVO: i.v. EVs 3mg/kg Dox loaded exosomes | Targeted tumour therapy | [62] |
| Neuro2A | GPI | Transfected with pLNCX-DAF-R2 or pLNCX-DAF-EGa1 using TransIT 2020 t.r. | Neuro2A, HeLa, and A431 | 40,000 cells per well or cells at a confluency of 80–90% and EVs at 5 µg/mL | Promoting tumor cell targeting | [63] |
| HEK293 | GE11 or EGF | Transfected with pDisplay encoding GE11 or EGF using FuGENE HD t.r. | HCC70 HCC1954 MCF-7 IN VIVO: RAG2–/– mice | 1 × 105 breast cancer cells and 1 µg of exosomes IN VIVO: i.v. 1 µg of exosomes, once per week for 4 weeks | Delivering of antitumor microRNA to EGFR-expressing breast cancer cells | [64] |
| BT474, SKBR3, HER2+, JAWSII DCs, 4T1-HER2, and bmDCs | CEA and HER2 coupled to the C1C2 domain of lactadherin | Transfected with p6mLC1C2 containing either human CEA (nt 1-2025) or human HER2/neu (nt 1-1953) | IN VIVO: C57BL/6J and BALB/c mice, hCEA or HER2 transgenic mice | IN VIVO: 2.6 × 1010 or 5.2 × 109 or 1.05 × 109 viral particles | Increasing vaccine potency | [65] |
| HEK293-F, E6, and CT26 | PSA and PAP coupled to the C1C2 domain of lactadherin | Transfected with pPSA/Zeo, pPSA-C1C2/Zeo, pPAP/Hygro, or pPAP-C1C2/Hygro using Lipofectamine LTX reagent and PLUS Reagent | IN VIVO: Male BALB/c or C57BL/6 mice | IN VIVO: 5E7 TCID50 of the MVA-BN-PRO viral vectors once every 2 weeks for a total of three treatments | Targeting of tumor antigens to improve antigen immunogenicity and therapeutic efficacy | [66] |
| DCs | C1C2 domain of lactadherin | Transfected with modified p6mLC1C2 or pcDNA6-Myc/His using Fugene 6 t.r. | IN VIVO: Balb/C mice | IN VIVO: six inoculums of YAC exosomes with HLA-A2 or five inoculums of YAC/HLA-A2 exosomes with pMAGE-A3 | Usage of antibodies against tumor biomarkers to attach the drug target candidates | [67] |
| THP-1 | RGD- DSPE-PEG and/or DSPE-PEG-SH | Incubated with DSPE-PEG-SH and/or DSPE-PEG-RGD for 2 days | MCF-7 and HeLa IN VIVO: tumor-bearing mouse | 4 × 105 cells/mL and 100 µL per well of 50 µg/mL exosomes IN VIVO: i.v. 200 µL of exosomes at 5 mg/mL | Active targeted chemo-photothermal synergistic tumor therapy | [68] |
| THP-1 | DSPE-PEG-biotin and/or DSPE-PEG-FA | Incubated with DSPE-PEG-biotin and/or DSPE-PEG-folate for 2 days | HeLa IN VIVO: C57BL/6 mice | 40 μg/mL of EVs IN VIVO: i.v. EVs with a total of 1.16 mg iron | Rapid isolation and enhanced tumor targeting | [69] |
| Cal 27 cells | DSPE-PEG-biotin and DSPE-PEG-folate | Incubated with DSPE-PEG-biotin and DSPE-PEG-folate | MDA-MB-231 IN VIVO: BALB/C mice | Series of dose and concentration IN VIVO: 18–22 g of EVs via the tail vein | Enhanced target and synergistic therapy for breast cancer | [70] |
| HUVECs | DSPE-PEG-biotin (to then attach SA-QDs) | Cultured with DSPE-PEG-biotin for several days and then incubated with SA-QDs | EPCs IN VIVO: nude mice bearing A2058 xenografts | Short-term incubation IN VIVO: injection | Antitumor siRNA delivery | [71] |
| HUVECs | DSPE-PEG-biotin and SA-FITC | Incubated in modified medium containing 40 µg/mL DSPE-PEG-biotin for several days | HepG2 and 3T3 fibroblast IN VIVO: cervical cancer-bearing male BALB/c mice | 5 × 103 cells per well and 0, 10, 40, 80, 100, and 200 mg/mL of exosomes IN VIVO: exosomes at 5 mg/mL, 200µL per mice | Active targeted drug delivery to tumor cells | [72] |
| HEK 293T cells | GlucB with sshBirA to conjugate streptavidin–Alexa 680 | Transduced with lentivirus vectors, CSCW-Gluc-IRES-GFP or CSCW-GlucB-IRES-GFP, then infection with CSCW-sshBirA-IRES-mCherry lentiviruses | IN VIVO: athymic nude mice spiked with EV-GlucB | IN VIVO: injected with a bolus of 100 μg EV-GlucB via retro-orbital vein or via tail vein | Multimodal imaging in vivo, as well as monitoring of EV levels in the organs and biofluids | [73] |
| B16BL6 | Streptavidin–lactadherin and biotinylated GALA | 4 × 106 cells per dish transfected with the plasmid vector pCMVSAV−LA | MHC class I molecules of DCs | 5 × 104 cells per well and exosomes (1 μg of protein) diluted in 0.1 mL of Opti-MEM | Efficient cytosolic delivery of exosomal tumor antigens | [74] |
| Parental Cells | Functionalization | Functionalization Step | Recipient Cells | Treatment Conditions | Application | Reference |
|---|---|---|---|---|---|---|
| Covalent | ||||||
| PC12 cells | TAMRA-NHS | 200 µL of Exos added to 1 mL 0.1 M sodium bicarbonate with 100mg TAMRA-NHS | PC12 cells | 1 × 108 cells and 100 µL of exosome solutions | Visualization of cellular uptake and intracellular trafficking of exosomes | [85] |
| 4T1 cells | Alkyne groups conjugated with azide-fluor 545 | 80 μg of exosomes in PBS, Cu (II) sulfate pentahydrate, 1.44 M l-ascorbic acid, and bathophenanthrolinedisulfonic acid disodium salt trihydrate | 4T1 cells | Cells at a confluency of 75% and 5 μg of exosomes in 100 μL RPMI | Surface functionalization of exosomes | [86] |
| Neuro2A and platelets | EGFR conjugated to DMPE-PEG derivatives | Conjugation in a 8.6:1000 molar ratio of nanobody/DMPE-PEG-maleimide micelles and then mixed with EVs | A431 and Neuro2A IN VIVO: Crl:NU-Foxn1nu mice with human tumor xenografts | 3 × 104 cells per well and 8 µg/mL of EVs IN VIVO: i.v. of 2.5 µg of EVs in 100 µL PBS | Enhancing cell specificity and circulation time of EVs | [87] |
| Bovine serum | DSPE and chemical conjugation by NHS-PEG | Physical: DSPE-PEG-biotin mixed with the EXOs (500 µg in PBS) Chemical: NHS-PEG-biotin reacted with the primary amines (500 nmol) on the EXOs | RAW264.7, DC2.4, and NIH3T3 IN VIVO: mice | 6 × 105 or 4 × 105 cells per well and EXOs at an ICG concentration of 5.8 µg per well IN VIVO: s.i. at a DiI dose of 1.52 µg/kg | Efficient delivery of immune stimulators and antigens to the lymph nodes in vivo | [88] |
| RAW 264.7 cells and BMM from C57BL/6 mice | DSPE-PEG or DSPE-PEG-AA | Addition of DSPE-PEG or DSPE-PEG-AA at 50 μg/mL | IN VIVO: C57BL/6 with induced pulmonary metastases | IN VIVO: i.v. injected with the exos at 108 particles/100 μL, n = 4 per group | Targeted paclitaxel delivery to pulmonary metastases | [89] |
| HEK293T cells | FA, PSMA RNA aptamer, and EGFR RNA aptamer conjugated to 3WJ | Cholesterol-triethylene glycol was conjugated into the arrow-tail of the pRNA-3WJ to promote the anchorage of the 3WJ onto the EV membrane | MDA-MB-231, KB, LNCaP (PSMA+), PC3 (PSMA–) IN VIVO: KB xenograft mice model | Incubation with cells IN VIVO: 1 dose of equivalent 0.5 mg siRNA/kg every 3 days for a total of 6 doses | Control RNA loading and ligand display on EVs for cancer regression | [90] |
| RAW 264.7 | NRP-1-targeted peptide RGE | Surface modification with sulfo-NHS that can react with azide-modified RGE peptide, using salts and copper as catalyst | U251 and Bel-7404 IN VIVO: orthotopic glioma-bearing BALB/c nude mice | Cells and exos at the equivalent of 15 µg/mL of Cur/SPIONS IN VIVO: i.v. of Cur/SPIONS at 800 µg/200 µg Exos/200 µL PBS | Facilitate simultaneous imaging and therapy of glioma in vitro and in vivo | [91] |
| Non-Covalent | ||||||
| HeLa | Cationic lipid formulation, LTX, and GALA | 20 μL LTX added to a solution of exosomes and 20 μL GALA and incubated for 20 min at room temperature | HeLa and (CHO)-K1 | 2 mL with 2 × 105 cells and 20 μg/mL of exosomes | Enhancing cytosolic delivery of exosomes | [92] |
| RTCs | Superparamagnetic magnetite colloidal nanocrystal clusters | 1 mL of serum incubated with 200 µL of M-Tfs solution for 4 h at 4 °C | H22 cells IN VIVO: Kunming mice bearing a subcutaneous H22 cancer | 0.1 mg/mL of exos in a simulated blood circulation at 32.85 cm/s (artery), 14.60 cm/s (vein), and 0.05 cm/s (capillary) | Targeted drug delivery vehicle for cancer therapy with magnetic properties | [93] |
| Human serum and C2C12 | Rhodamine-labelled M12-CP05, FITC-labelled NP41-CP05 | CP05 (200 µg/mL) incubated with nickel beads, added into the precentrifuged serum (200 µL), and incubated for 30 min at 4 °C under rotation | IN VIVO: dystrophin-deficient and immunodeficient nude mice and C57BL/6 mice | IN VIVO: i.m.1 or 2 µg of EXOs, i.v. EXOs in 100 µL of saline solution | Enabling targeting, cargo loading, and capture of exosomes from diverse origins | [94] |
| 4T1, MCF-7, and PC3 | DiR labelling | 5µL of DIR, at a concentration of 220 µg/mL in ethanol, was mixed with 220 µg exosomes or liposomes in 100 µL PBS for 1 hour | IN VIVO: Balb/c, nude, and NOD.CB17- Prkdcscid/J mice with either 4T1 cells or PC3 cells | IN VIVO: i.v. 60 µg DIR-labeled exosomes in 200 µL PBS or i.t. 60 µg of DIR-labeled exosomes in 50 µL PBS | Biodistribution and delivery efficiency of unmodified tumor-derived exosomes | [95] |
| Glycosylation | ||||||
| MLP29 | Neuraminidase | Surface glycosylation of the EVs was manipulated by treatment with neuraminidase to remove the terminal residues of sialic acid | IN VIVO: wild-type mice | IN VIVO: i.v. of the EVs | Modification of the glycosylation of EVs to alter their biodistribution in vivo | [96] |
| U87 and GBM8 | Glycosylation and insertion of targeting ligand to DC-SIGN | Treated with a pan-sialic acid hydrolase Neuraminidase for 30 min at 37 °C and/or incubated with palmitoyl-LewisY while vortexing for 10 min | MoDCs | 500,000 cells incubated with EVs for 45 min on ice to allow receptor binding | Enhancing receptor-mediated targeting of dendritic cells | [97] |
| HEK293FT | Glycosylation of targeting-peptide-Lamp2b fusion proteins | 1.5 mL of 0.971 M sucrose was slowly pipetted underneath the 8.5 ml of exosome solution | HEK293FT and Neuro2A | Cells at 50% confluency and EVs for 2 h at 37 °C | Stabilization of exosome-targeting peptides | [98] |
| Hybridization | ||||||
| HEK293FT | CRISPR/CRISPR-associated protein 9 (Cas9) system | Addition of the plasmid–liposome complex to exosomes and incubated at 37 °C for 12 h in a volume ratio of 1:2 | MSCs | Incubation with cells at 90% of confluency | Efficiently encapsulate large plasmids and be endocytosed in MSCs | [99] |
| RAW 264.7, CMS7-wt, and CMS7-HE | DOPC, DOPS, DOTAP, and DOPS/PEG-DSPE | Exosomes (300 μg/mL, protein) mixed with 100 μM liposomes in a volume ratio of 1:1 and then several freeze–thaw cycles | HeLa cells | 4.5 μg protein in exosome incubated with 1 × 105 HeLa cells for 4 h at 37 °C | Control and modify the performance of exosomal nanocarriers | [100] |
| HUVECs and MSCs | Phosphatidylcholine, phosphatidylethanolaminein, and PEG | Liposomes and EVs were mixed at 40 °C in a total volume of 40−200 μL (2 × 1010 or 2 × 1011 objects); liposome/EV ratio of 1:1, 1:9, or 9:1 in PBS. PEG was added at 5−30% (w/v) | THP1-derived macrophages and CT26 | 100,000 cells per well and hybrid EVs containing 1 mol % of DiR, cells, and 400 μL of mTHPC-loaded hybrid EVs or (3D) 500 cells and mTHPC-loaded hybrid EVs | Design of personalized biogenic drug delivery systems | [101] |
| J774A.1 | L-a-phosphatidylcholine and cholesterol | EVs (200 µg protein) used to hydrate the dry 1000 µg of lipid film in a final volume of 1 mL; then, the solution was extruded through 400 and 200 nm polycarbonate membrane filter | K7M2, 4T1, and NIH/3T3 | 10,000–20,000 cells and 4 mL of 50 µg/mL of hybrid EVs at 37 °C for 3 h or 48 h | Tumor targeted drug delivery | [102] |
| Precursor Cells | Recipient Cells | Application | Reference |
|---|---|---|---|
| Extrusion | |||
| U937 and RAW 264.7 | TNF-α-treated HUVECs IN VIVO: colon adenocarcinoma-induced CT26 mouse | Targeted delivery of chemotherapeutic drugs | [112] |
| RAW 264.7 and HB1.F3 | IN VIVO: male BALB/c mice | Radiolabelling of EVs with 99mTc-HMPAO to understand in vivo distribution and behavior of exosomes | [116] |
| Murine mouse embryonic stem cell line D3 | NH-3T3 | Gene delivery of endogenous, precursor cell characteristic RNA (mOct ¾ and mNanog) | [117] |
| Murine mouse embryonic stem cell line D3 | Primary murine skin fibroblasts from BL6/C57 mice | Investigate the ability of these nanovesicles to improve proliferation by treating cells with the nanovesicles | [118] |
| Non-tumorigenic epithelial MCF-10A cells | MCF-7 IN VIVO: BALB/C nu/nu mice | Evaluation of the EV biosafety and uptake efficiency for the delivery of CDK4 siRNA | [119] |
| MSCs | MDA-MB-231 IN VIVO: nude BALB/c mice | Targeted delivery of paclitaxel for cancer treatment | [120] |
| H19-OE lentiviral vector-transfected HEK293 | HMEC-1 IN VIVO: diabetic rat model | Treatment of diabetic wounds through the delivery of LncRNA-H19 | [121] |
| MIN6 and NIH3T3 | IN VIVO: BALB/c and NSG mice | Facilitation of the differentiation of bone marrow cells to insulin-producing cells (β-cells) | [122] |
| Primary hepatocytes | Primary hepatocytes IN VIVO: two-thirds PH mouse model (C57Bl/6) | Promote hepatocyte proliferation and liver regeneration | [115] |
| ASCs | MLE-12 IN VIVO: C57BL/6 mice | Inhibition of emphysema trough increasing the proliferation rate of lung epithelial cells | [123] |
| MSCs | RAW 264.7 IN VIVO: wild-type mice C57BL/6 | Treatment of sepsis by down-regulating the cytokine storm induced by bacterial outer membrane vesicles (OMVs) in mice | [124] |
| M1 macrophages | CT26 and BMDMs IN VIVO: CT26-bearing mice | Repolarize M2 tumor-associated macrophages (TAMs) to M1 macrophages that release pro-inflammatory cytokines and induce antitumor immune responses | [125] |
| Natural killer (NK) cells NK92-MI | D54, MDA-MB-231, CAL-62, and HepG2 IN VIVO: female BALB/c nude mice | Immunotherapeutic agent for treatment of cancer | [114] |
| Microfluidics | |||
| Murine embryonic stem cells (ES-D3) | NIH 3T3 | Exogenous material delivery (polystyrene beads) | [126] |
| Murine embryonic stem cell line-D3 | NIH-3T3 fibroblasts | Gene delivery of RNAs, Oct ¾, and Nanog | [127] |
| Formulation | Recipient Cells | Application | Reference |
|---|---|---|---|
| PC:CHOL:DSPE-PEG:DSPE-PEG-MAL liposome coated with MHC Class I/ peptide complexes, anti-LFA1, anti-CD28, anti-CD27, anti-4-1BB, anti-CD40L, and T cell receptors in the form of Fab antibody regions | T cells IN VIVO: BALB/c mice | Targeted immunotherapy, inducing antigen-specific T cells responses | [128] |
| DOPC/SM/Chol/DOPS/DOPE at a molar ratio of 21/17.5/30/14/17.5 liposome with siRNA (siNC, FAM-siNC, and siVEGF) | A549 and HUVEC | Delivery of VEGF siRNA in a more efficient way and with less cytotoxicity | [129] |
| DOPC/SM/Chol/DOPS/DOPE at a molar ratio of 21/17.5/30/14/17.5 liposome integrated with connexin 43 (Cx43) | A549 and U87 MG | Delivery of siRNA | [130] |
| CH/PC/SM/Cer at a weight ratio of 0.9/1/0.4/0.03 functionalized with recombinant human integrin α6β4 protein, bovine serum albumin, and lysozyme | A549 IN VIVO: mice bearing lung cancers | Targeted delivery of therapeutic oligonucleotides to lung adenocarcinoma cells (microRNA-145 mimics) | [131] |
| Phosphatidylcholine, SM, ovine wool cholesterol, and DOGS-NTA in a weight ratio of 55:30:10:5 liposome bonded with histidine-tagged APO2L/TRAIL | IN VIVO: adult female NZW rabbits | Treatment of antigen-induced arthritis (AIA) | [132] |
| Phosphatidylcholine, sphingomyelin (SM), cholesterol, and DOGS-NTA-Ni liposome with rAPO2L/TRAIL | Jurkat clone E6.1, U937, U266, and MM.1S | Apoptosis-inducing ability of hematological tumors | [133] |
| Cremophor EL, PC, DOPE, and DC-Chol liposome conjugated with DEC205 monoclonal antibody | DCs | Development of antigen carriers for specific DC targeting | [134] |
| * Membrane proteins derived from RBCs (containing high CD47 levels to inhibit phagocytosis) and MCF-7 cancer cells (containing specific adhesion proteins) integrated into synthetic phospholipidic bilayers | MCF-7, HeLa, and RAW264.7 IN VIVO: MCF-7 tumor-bearing nude mice | Higher tumor accumulation, lower interception, and better antitumor therapeutic effect | [135] |
| * Proteins derived from the leukocytes’ plasmalemma trough extrusion integrated into a synthetic phospholipid bilayer (DPPC, DSPC, and DOPC and cholesterol) | IN VIVO: BALB/C mice | Selective and effective delivery of dexamethasone to inflamed tissues, and reduced phlogosis in a localized model of inflammation | [136] |
| * Membrane proteins derived from leukocytes from human blood and immortalized J774 murine macrophages within the lipid bilayer of liposome-like nanovesicles (DPPC, DOPC, and cholesterol in a molar ratio of 4/3/3) | HUVECs IN VIVO: Balb/c mice | Avoidance of macrophage uptake and promoting the adhesion to inflamed endothelium | [137] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villata, S.; Canta, M.; Cauda, V. EVs and Bioengineering: From Cellular Products to Engineered Nanomachines. Int. J. Mol. Sci. 2020, 21, 6048. https://doi.org/10.3390/ijms21176048
Villata S, Canta M, Cauda V. EVs and Bioengineering: From Cellular Products to Engineered Nanomachines. International Journal of Molecular Sciences. 2020; 21(17):6048. https://doi.org/10.3390/ijms21176048
Chicago/Turabian StyleVillata, Simona, Marta Canta, and Valentina Cauda. 2020. "EVs and Bioengineering: From Cellular Products to Engineered Nanomachines" International Journal of Molecular Sciences 21, no. 17: 6048. https://doi.org/10.3390/ijms21176048
APA StyleVillata, S., Canta, M., & Cauda, V. (2020). EVs and Bioengineering: From Cellular Products to Engineered Nanomachines. International Journal of Molecular Sciences, 21(17), 6048. https://doi.org/10.3390/ijms21176048






