Comparison of Monovalent and Divalent Ions Removal from Aqueous Solutions Using Agricultural Waste Biochars Prepared at Different Temperatures—Experimental and Model Study
Abstract
1. Introduction
2. Results and Discussion
2.1. Effect of Pyrolysis Temperature on Biochar Surface Properties
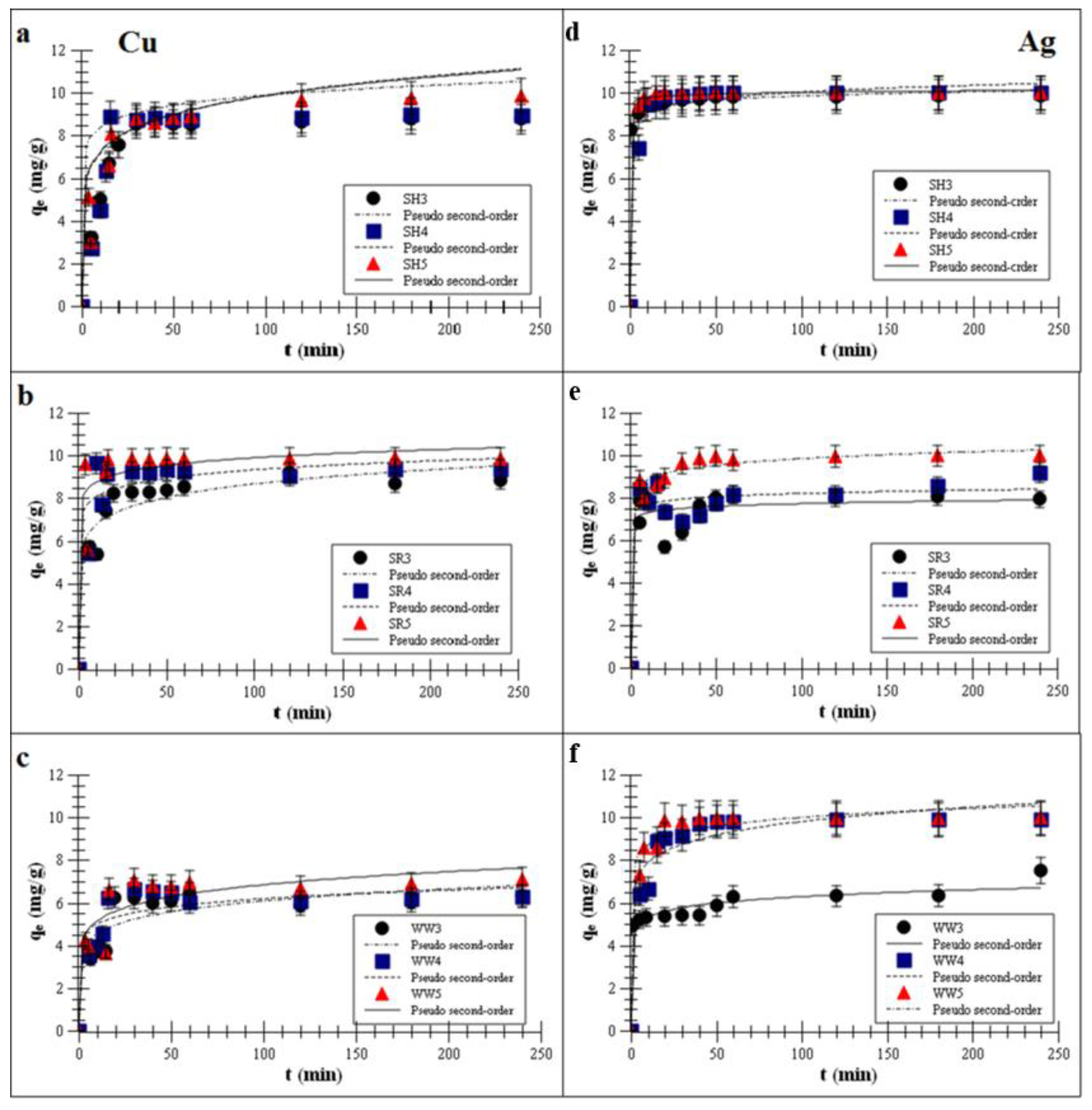
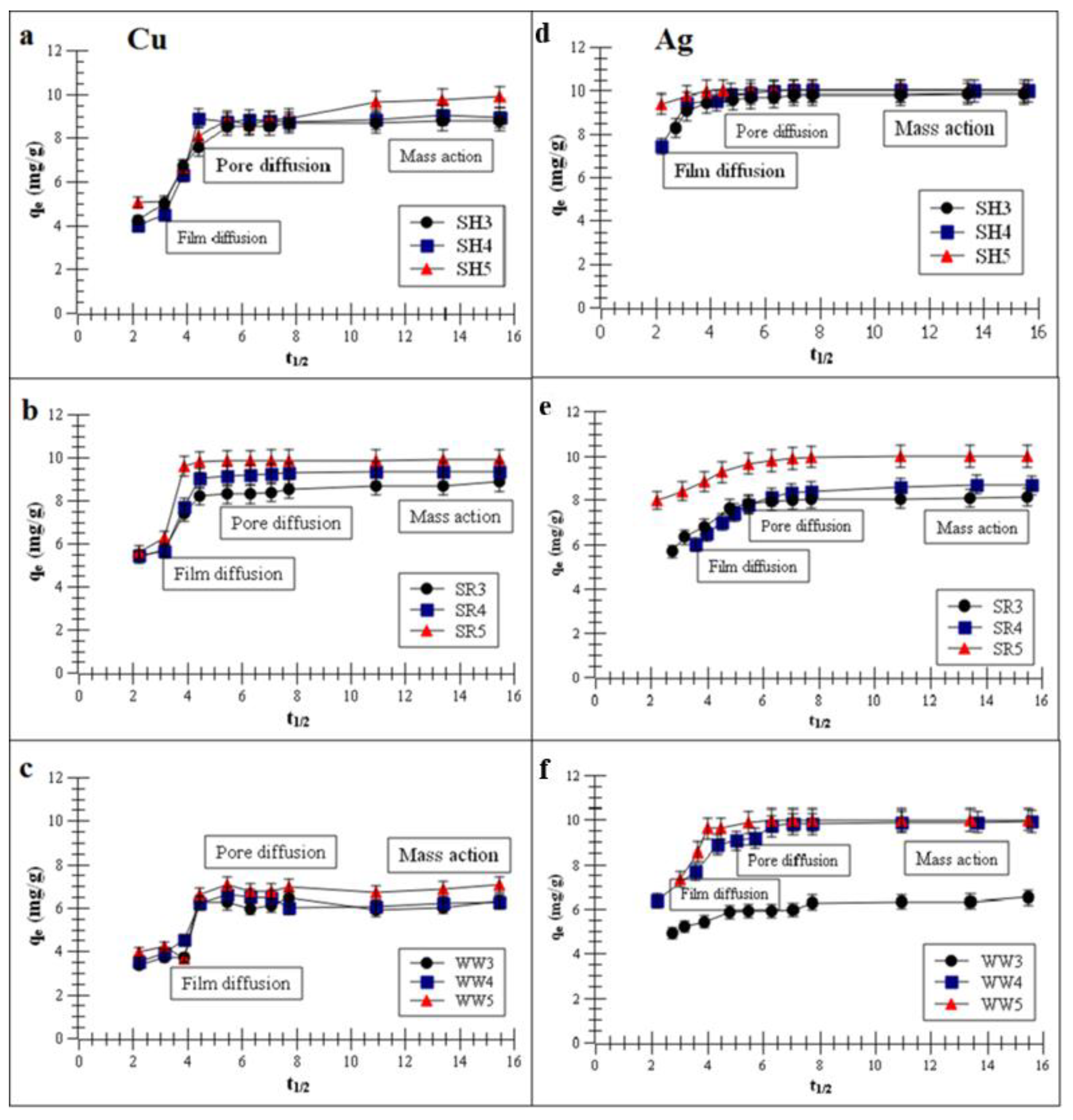
2.2. Adsorption of Monovalent and Divalent Metals on Biochar—Kinetics Studies
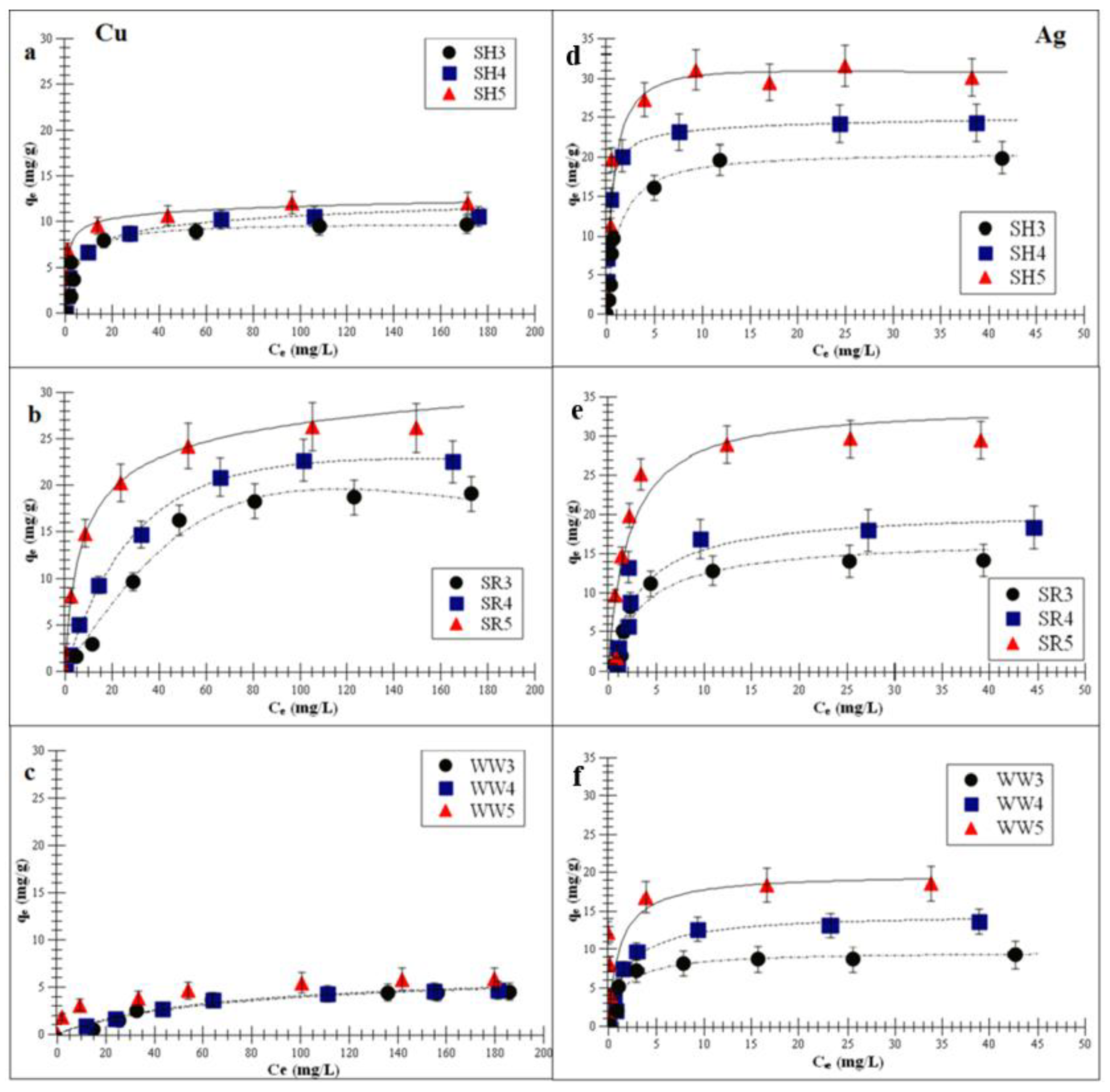
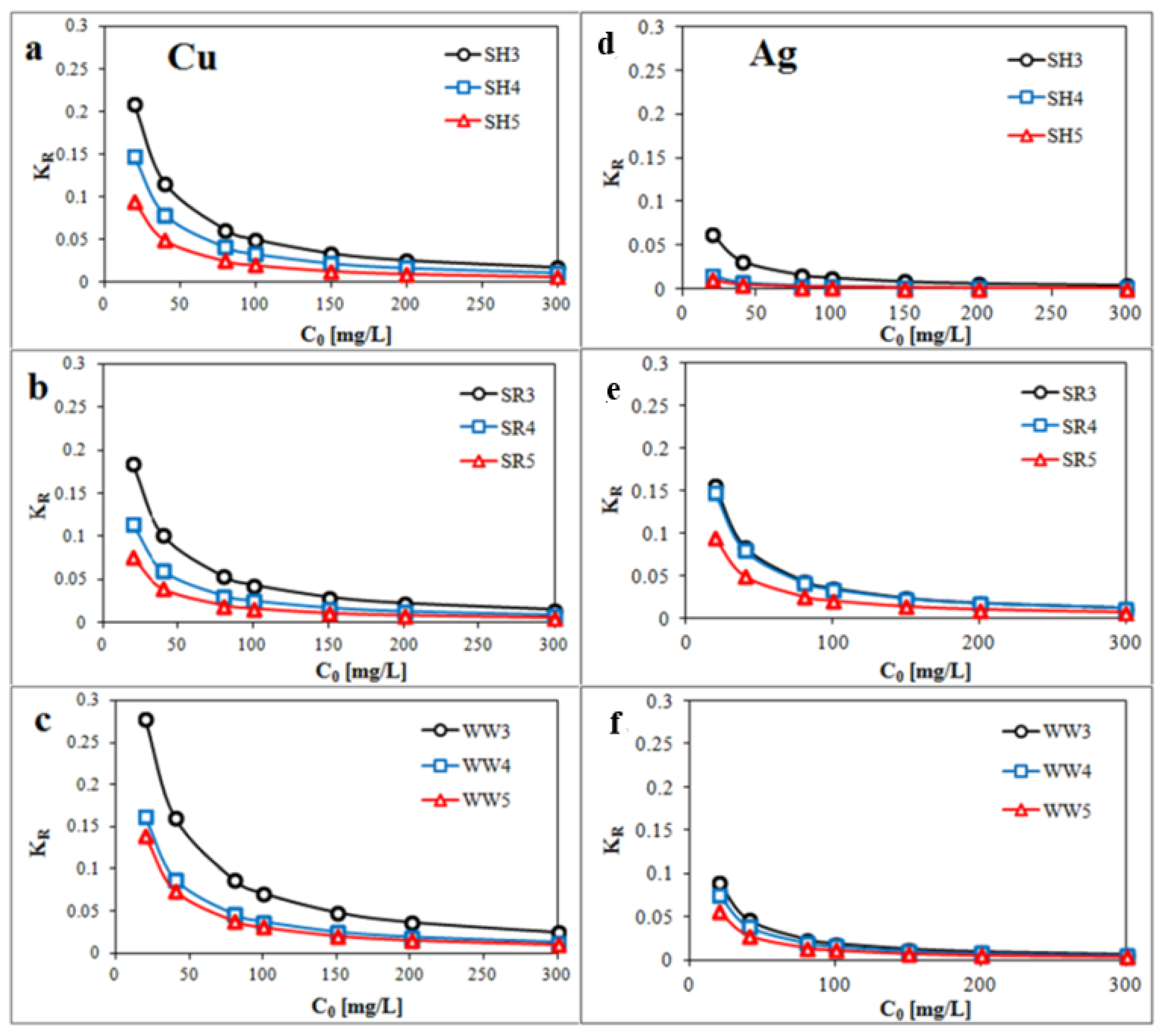
2.3. Adsorption of Monovalent and Divalent Metals on Biochar—Equilibrium Isotherm Studies
3. Materials and Methods
3.1. Biochar Production
3.2. Surface Characteristics of Biochars
3.3. Adsorption Study for Aqueous Systems with Monovalent and Divalent Metals
4. Conclusions
- Biochars obtained at 500 °C exhibited the highest specific surface areas but the lowest variable surface charges and the lowest contents of surface functional groups.
- Kinetics of the Cu and Ag sorption process were well-described by a pseudo-second-order model, and the IPD model revealed that Cu and Ag removals consisted of film diffusion, internal diffusion and mass action.
- Cu and Ag adsorptions on agricultural biochars were well-described by the Langmuir-Freundlich model.
- Agricultural biochar adsorption capacities of Cu and Ag increased with increasing the values of the pyrolysis temperature and with decreasing the valence of the metals.
- Monovalent Ag was better-removed than the divalent Cu ions from aqueous solutions. This was caused by the stronger attraction between the surface functional groups and Ag ions, which facilitated their penetration into biochar pores. This phenomenon was also dictated with the ionic radius of the adsorbates and electronegativity.
- Agricultural biochars produced at ≥500 °C demonstrate the highest removal efficiency of Cu and Ag and will have an important role as organic eco-adsorbents in the mono- and divalent heavy metal removals from aqueous media.
Author Contributions
Funding
Conflicts of Interest
References
- EPA. Secondary Drinking Water Regulations: Guidance for Nuisance Chemicals; EPA, 816-F-10-079; EPA: Washington, DC, USA, 2011. Available online: http://water.epa.gov/drink/contaminants/secondarystandards.cfm (accessed on 1 June 2011).
- Eckelman, M.J.; Graedel, T. Ag emissions and their environmental impacts: A multilevel assessment. Environ. Sci. Technol. 2007, 41, 6283–6289. [Google Scholar] [CrossRef]
- Sotiriou, G.A.; Pratsinis, S.E. Antibacterial activity of nanoAg ions and particles. Environ. Sci. Technol. 2010, 44, 5649–5654. [Google Scholar] [CrossRef]
- EPA. National Primary Drinking Water Regulation Table-Ground Water and Drinking Water; EPA 816-F-09-004; EPA: Washington, DC, USA, 2009. Available online: https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulation-table (accessed on 13 August 2020).
- Meng, J.; Feng, X.; Dai, Z.; Liu, X.; Wu, J.; Xu, J. Adsorption characteristics of Cu(II) from aqueous solution onto biochar derived from swine manure. Environ. Sci. Pollut. Res. Int. 2014, 21, 7035–7046. [Google Scholar] [CrossRef]
- Hubicki, Z.; Kołodyńska, D. Selective Removal of Heavy Metal Ions from Waters and Waste Waters Using Ion Exchange Methods; InTech Open Access Publisher: London, UK, 2012. [Google Scholar] [CrossRef]
- Abdel-Raouf, M.S.; Abdul-Raheim, A.R.M. Removal of Heavy Metals from Industrial Waste Water by Biomass-Based Materials: A Review. J. Pollut. Eff. Cont. 2017, 5, 180. [Google Scholar] [CrossRef]
- Ok, Y.S.; Uchimiya, S.M.; Chang, S.X.; Bolan, N. Biochars: Production, Characterization and Applications; CRC Press: New York, NY, USA, 2015. [Google Scholar]
- Awual, M.R. Novel ligand functionalized composite material for efficient copper(II) capturing from wastewater sample. Compos. Part B Eng. 2019, 172, 387–396. [Google Scholar] [CrossRef]
- Awual, M.R. Novel conjugated hybrid material for efficient lead(II) capturing from contaminated wastewater. Mater. Sci. Eng. C 2019, 101, 686–695. [Google Scholar] [CrossRef] [PubMed]
- Awual, M.R. Mesoporous composite material for efficient lead(II) detection and removal from aqueous media. J. Environ. Chem. Eng. 2019, 7, 103124. [Google Scholar] [CrossRef]
- Awual, M.R. Efficient phosphate removal from water for controlling eutrophication using novel composite adsorbent. J. Clean. Prod. 2019, 228, 1311–1319. [Google Scholar] [CrossRef]
- Awual, M.R. An efficient composite material for selective lead(II) monitoring and removal from wastewater. J. Environ. Chem. Eng. 2019, 7, 103087. [Google Scholar] [CrossRef]
- Awual, M.R. A facile composite material for enhanced cadmium(II) ion capturing from wastewater. J. Environ. Chem. Eng. 2019, 7, 103378. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M. A novel fine-tuning mesoporous adsorbent for simultaneous lead(II) detection and removal from wastewater. Sens. Actuators B Chem. 2014, 202, 395–403. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M. A ligand based innovative composite material for selective lead(II) capturing from wastewater. J. Mol. Liq. 2019, 294, 111679. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M.; Khaleque, M.A. Efficient selenium(IV) detection and removal from water by tailor-made novel conjugate adsorbent. Sens. Actuators B Chem. 2015, 209, 194–202. [Google Scholar] [CrossRef]
- Awual, M.R.; Hasan, M.M.; Znad, H. Organic–inorganic based nano-conjugate adsorbent for selective palladium(II) detection, separation and recovery. Chem. Eng. J. 2015, 259, 611–619. [Google Scholar] [CrossRef]
- Szewczuk-Karpisz, K.; Nowicki, P.; Sokołowska, Z.; Pietrzak, R. Hay-based activated biochars obtained using two different heating methods as effective low-cost sorbents: Solid surface characteristics, adsorptive properties and aggregation in the mixed Cu(II)/PAM system. Chemosphere 2020. [Google Scholar] [CrossRef]
- Malińska, K. Biowęgiel odpowiedzią na aktualne problem ochrony środowiska. Inżynieria Ochr. Środowiska 2012, 15, 387–403. (In Polish) [Google Scholar]
- Duan, S.; Wang, Y.; Liu, X.; Shao, D.; Hayat, T.; Alsaedi, A.; Li, J. Removal of U(VI) from Aqueous Solution by Amino Functionalized Flake Graphite Prepared by Plasma Treatment. ACS Sustain. Chem. Eng. 2017, 5, 4073–4085. [Google Scholar] [CrossRef]
- Tomczyk, A.; Sokołowska, Z.; Boguta, P. Biochar physicochemical properties: Pyrolysis temperature and feedstock kind effects. Rev. Environ. Sci. Biotechnol. 2020, 19, 191–215. [Google Scholar] [CrossRef]
- Wei, D.; Li, B.; Huang, H.; Luo, L.; Zhang, J.; Yang, Y.; Guo, J.; Tang, L.; Zang, G.; Zhou, Y. Biochar-based functional materials in the purification of agricultural wastewater: Fabrication, application and future research needs. Chemosphere 2018, 197, 165–180. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Cao, X.; Ma, L.; Gao, B.; Harris, W. Dairy-Manure Derived Biochar Effectively Sorbs Lead and Atrazine. Environ. Sci. Technol. 2009, 49, 3285–3291. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, S.; Yao, P.; Chen, Y.; Xu, X.; Li, T.; Gong, G. Removal of Pb(II) from aqueous solutions by Phytolacca americana L. biomass as a low cost biosorbent. Arab. J. Chem. 2018, 11, 99–110. [Google Scholar] [CrossRef]
- Chen, B.; Chen, Z. Sorption of naphthalene and 1-naphthol by biochars of orange peels with different pyrolytic temperatures. Chemosphere 2009, 76, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Gao, B.; Yao, Y.; Fang, J.; Zhang, M.; Zhou, Y.; Chen, H.; Yang, L. Effects of feedstock type, production method, and pyrolysis temperature on biochar and hydrochar properties. Chem. Eng. J. 2014, 240, 574–578. [Google Scholar] [CrossRef]
- Mia, S.; Singh, B.; Dijkstra, F.A. Aged biochar affects gross nitrogen mineralization and recovery; A 15N study in two contrasting soils. Glob. Chang. Biol. Bioenergy 2017, 9, 1196–1206. [Google Scholar] [CrossRef]
- Ghani, W.A.W.A.K.; Mohd, A.; de Silva, G.; Bachmann, R.T.; Taufiq-Yap, Y.H.; Rashid, U.; Al-Muhtaseb, A.H. Biochar production from waste rubber-wood-sawdust and its potential use in C sequestration: Chemical and physical characterization. Ind. Crop. Prod. 2013, 44, 18–24. [Google Scholar] [CrossRef]
- Jindo, K.; Mizumoto, H.; Sawada, Y.; Sanchez-Monedero, M.A.; Sonoki, T. Physical and chemical characterization of biochars derived from different agricultural residues. Biogeosciences 2014, 11, 6613–6621. [Google Scholar] [CrossRef]
- Schimmelpfennig, S.; Glaser, B. One step forward toward characterization: Some important material properties to distinguish biochars. J. Environ. Qual. 2012, 41, 1001–1013. [Google Scholar] [CrossRef]
- Zhao, S.-X.; Ta, N.; Wang, X.-D. Effect of temperature on the structural and physicochemical properties of biochar with apple tree branches as feedstock material. Energies 2017, 10, 1293. [Google Scholar] [CrossRef]
- Gai, X.; Wang, H.; Liu, J.; Zhai, L.; Liu, S.; Ren, T.; Liu, H. Effects of Feedstock and Pyrolysis Temperature on Biochar Adsorption of Ammonium and Nitrate. PLoS ONE 2014, 9, e113888. [Google Scholar] [CrossRef]
- Ho, Y.-S. Review of second-order models for adsorption systems. J. Hazard. Mater. 2006, 36, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.J.; Kwapiński, W.; Hayes, M.H.B.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Mohan, D., Jr.; Pittman, C.U.; Bricka, M.; Smith, F.; Yancey, B.; Mohammad, J.; Steele, P.H.; Alexandre-Franco, M.F.; Gómez-Serrano, V.; Gong, H. Sorption of arsenic, cadmium and lead by chars produced from fast pyrolysis of wood and bark during bio-oil production. J. Colloid Interface Sci. 2007, 310, 57–73. [Google Scholar] [CrossRef]
- Cibati, A.; Foereid, B.; Bissessur, A.; Hapca, S. Assessment of Miscanthus × giganteus derived biochar as copper and zinc adsorbent: Study of the effect of pyrolysis temperature, pH and hydrogen peroxide modification. J. Clean. Prod. 2017, 162, 1285–1296. [Google Scholar] [CrossRef]
- Wang, S.; Li, H.; Chen, X.; Yang, M.; Qi, Y. Selective adsorption of Ag ions from aqueous solution using polystyrene-supported trimercaptotriazine resin. J. Environ. Sci. 2012, 24, 2166–2172. [Google Scholar] [CrossRef]
- Jeon, C. Adsorption of Ag ions from industrial wastewater using waste coffee grounds. Korean J. Chem. Eng. 2017, 34, 384–391. [Google Scholar] [CrossRef]
- Antunes, E.; Jacob, M.V.; Brodie, G.; Schneider, P.A. Ag removal from aqueous solution by biochar produced from biosolids via microwave pyrolysis. J. Environ. Manag. 2017, 203, 264–272. [Google Scholar] [CrossRef]
- Trakal, L.; Šigut, R.; Šillerová, H.; Faturíková, D.; Komárek, M. Copper removal from aqueous solution using biochar: Effect of chemical activation. Arab. J. Chem. 2014, 7, 43–52. [Google Scholar] [CrossRef]
- Marsh, H.; Rodriguez-Reinoso, F. Chapter 4: Characterization of Activated Carbon. In Activated Carbon; Elsevier Science: Amsterdam, The Netherlands, 2016; pp. 143–242. [Google Scholar]
- Tomczyk, A.; Boguta, P.; Sokołowska, Z. Biochar efficiency in Cu removal from Haplic soils. Int. J. Environ. Sci. Technol. 2019, 16, 4899–4912. [Google Scholar] [CrossRef]
- Zhou, D.; Liu, D.; Gao, F.; Li, M.; Luo, X. Effects of biochar-derived sewage sludges on heavy metal adsorption and immobilization in soils. Int. J. Enivron. Res. Public Health 2017, 14, 681. [Google Scholar] [CrossRef]
- Abbas, T.; Rizwan, M.; Ali, S.; Adrees, M.; Rehman, M.Z.; Qayyum, M.F.; Ok, Y.S.; Murtaza, G. Effect of biochar on allevation of cadmium toxicity in wheat (Triticum aestivum L.) grown on Cd-contaminated saline soil. Environ. Sci. Pollut. Res. 2018, 25, 25668–25680. [Google Scholar] [CrossRef] [PubMed]
- Keiluweit, M.; Kleber, M. Molecular-level interactions in soils and sediments: The role of aromatic pi-systems. Environ. Sci. Technol. 2009, 43, 3421–3429. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Gao, B. Use of fruit and vegetable waste based biochar for removal of heavy metals from aqueous solution. Int. J. Agric. Biosyst. Eng. 2018, 12, 1–9. [Google Scholar]
- Qi, F.; Yan, Y.; Lamb, D.; Naidu, R.; Bolan, N.S.; Liu, Y.; Ok, Y.S.; Donne, S.W.; Semple, K.T. Thermal stability of biochar and its effects on cadmium sorption capacity. Bioresour. Technol. 2017, 246, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Jang, A.; Seo, Y.; Bishop, P.L. The removal of heavy metals in urban runoff by sorption mulch. Environ. Pollut. 2005, 133, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and non-conventional adsorbents for wastewater treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Pourret, O.; Houben, D. Characterization of metal binding sites onto biochar using rare earth elements as a fingerprint. Heliyon 2018, 4. [Google Scholar] [CrossRef]
- Uchimiya, M.; Wartelle, L.H.; Klasson, K.T.; Fortier, C.A.; Lima, I.M. Influence of pyrolysis temperature on biochar property and function as a heavy metal sorbent in soil. J. Agric. Food. Chem. 2011, 59, 2501–2510. [Google Scholar] [CrossRef]
- Komkiene, J.; Baltrenaite, E. Biochar as adsorbent for removal of heavy metal ions [Cadmium(II), Cu(II), Lead(II), Zinc(II)] from aqueous phase. Int. J. Environ. Sci. Technol. 2015, 13, 471–482. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of Cu and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, B.; Zimmerman, A.R.; Cao, X. Biochar-supported zerovalent iron reclaims Ag from aqueous solution to form antimicrobial nanocomposite. Chemosphere 2014, 117, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Bis, Z.; Kobyłecki, R.; Ścisłowska, M.; Zarzycki, R. Biochar—Potential tool to combat climate change and drought. Ecohydrol. Hydrobiol. 2018, 18, 441–453. [Google Scholar] [CrossRef]
- Kacprzak, A.; Kobyłecki, R.; Bis, Z. Clean energy from a carbon fuel cell. Arch. Thermodyn. 2011, 32, 145–157. [Google Scholar] [CrossRef]
- Kacprzak, A.; Kobyłecki, R.; Bis, Z. Influence of temperature and composition of NaOH-KOH and NaOH-LiOH electrolytes on the performance of a direct carbon fuel cell. J. Power Sour. 2013, 239, 409–414. [Google Scholar] [CrossRef]
- Kobyłecki, R.; Bis, Z. Autotermiczna termoliza jako efektywna technologia produkcji czystych i wysokoenergetycznych paliw. Arch. Spalania 2016, 6, 114–119. (In Polish) [Google Scholar]
- Kobyłecki, R.; Ścisłowska, M.; Bis, Z. Carbonization of biomass—An efficient tool to decrease the emission of CO2. Arch. Thermodyn. 2013, 34, 185–195. [Google Scholar] [CrossRef]
- Józefaciuk, G. Effect of acid and alkali treatments on surface-charge properties of selected minerals. Clay Clay Miner. 2002, 50, 647–656. [Google Scholar] [CrossRef]
- Schönherr, J.; Buchheim, J.R.; Sholz, P.; Adelhelm, P. Boehm Titration Revisited (Part I): Practical Aspects for Achieving a High Precision in Quantifying Oxygen-Containing Surface Groups on Carbon Materials. C J. Carbon Res. 2018, 4, 21. [Google Scholar] [CrossRef]
- Lee, Y.; Park, J.; Ryu, C.; Gang, K.S.; Yang, W.; Park, Y.-K.; Jung, J.; Hyun, S. Comparison of biochar properties from biomass residues produced by slow pyrolysis at 500 °C. Bioresour. Technol. 2013, 148, 196–201. [Google Scholar] [CrossRef]
- Polish Standard PN-Z-19010-1. Soil Quality. Determination of the Specific Surface Area of Soils by Water Sorption (BET); ALFA-WERO: Warsaw, Poland, 1997.
- Cao, Q.; Huang, Z.; Liu, S.; Wu, Y. Potential of Punica granatum biochar to adsorb Cu (II) in soil. Sci. Rep. 2019, 9, 11116. [Google Scholar] [CrossRef]
- Zhao, J.; Shen, X.; Domene, X.; Alcañiz, J.-M.; Liao, X.; Palet, C. Comparison of biochars derived from different types of feedstock and their potential for heavy metal removal in multiple-metal solutions. Sci. Rep. 2019, 9, 9869. [Google Scholar] [CrossRef] [PubMed]
- Vagliasindi, L.I.; Arena, G.; Bonomo, R.P.; Pappalardo, G.; Tabii, G. Cu complex species within a fragment of the N-terminal repeat region in opossum PrP protein. Dalton Trans. 2011, 40, 2441–2450. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; McKay, G. Pseudo-second-order model for sorption process. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Lagergren, S. Zur theorie der sogenannten adsorption gelöster stoffe. K. Sven. Vetensk. Handl. 1898, 24, 1–39. [Google Scholar]
- Weber, W.J.; Morris, J.C. Advances in water pollution research: Removal of biologically resistant pollutant from waste water by adsorption. In Proceedings of 1st International Conference on Water Pollution Symposium 2; Pergamon Press: Oxford, UK, 1962; pp. 231–266. [Google Scholar]
- Sips, R. On the structure of a Catalyst Surface. J. Chem. Phys. 1950, 18, 1024–1026. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Peereboom, L. Adsorption of Bio-Renewable Substrates on Supported Metal Catalyst in Water. Ph.D. Thesis, Department of Chemical Engineering, Michigan State University, Lansing, MI, USA, 2007. [Google Scholar]
- Ho, Y.-S.; Huan, C.T.; Huang, H.W. Equilibrium sorption isotherm for metal ions on tree fern. Process Biochem. 2002, 37, 1421–1430. [Google Scholar] [CrossRef]
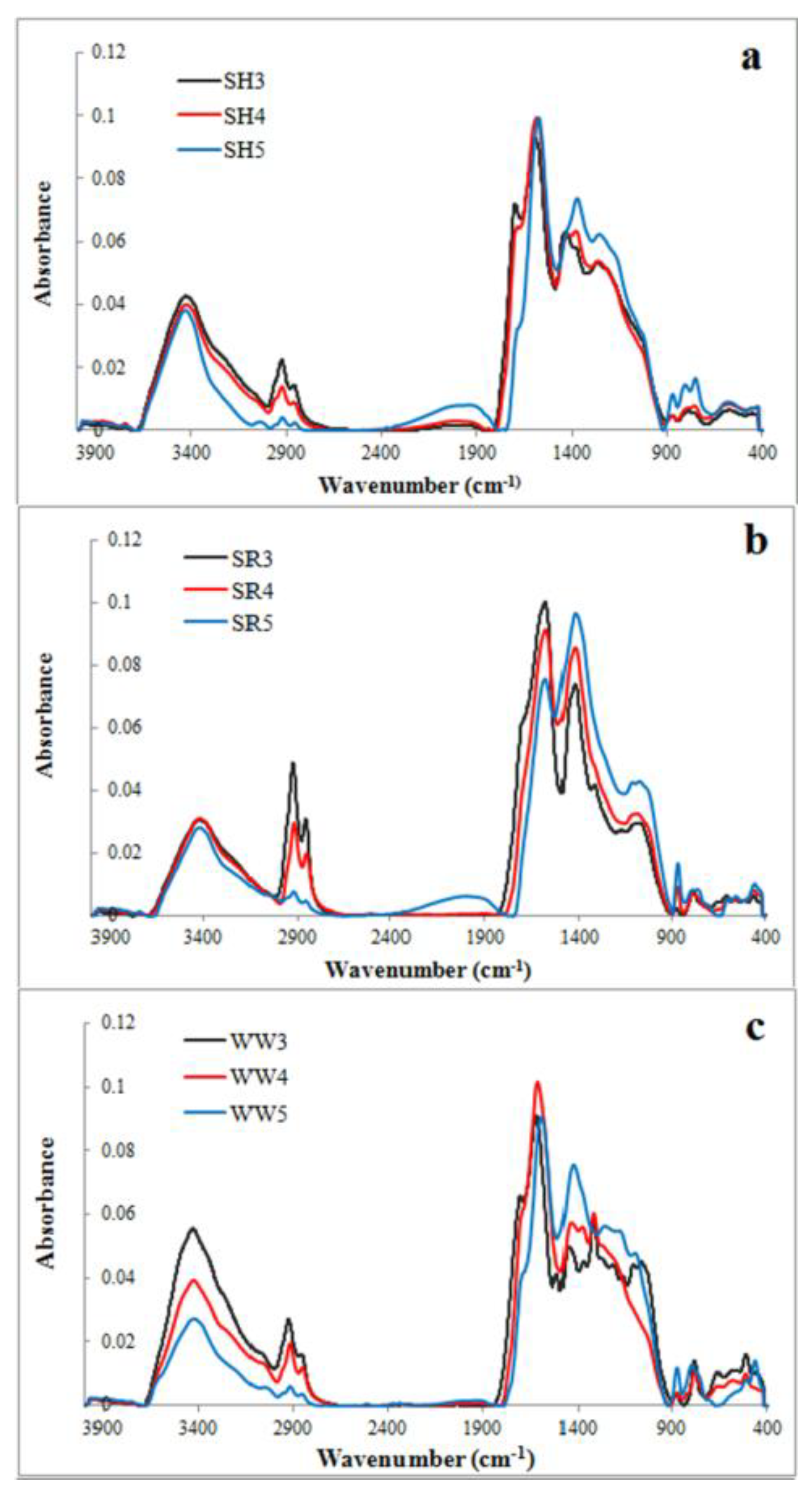
| Type of Biochar | pH | SBET (m2/g) | Q (cmol/kg) | Carboxylic Groups (cmol/kg) | Lactonic Groups (cmol/kg) | Phenolic Groups (cmol/kg) | H/C | O/C |
|---|---|---|---|---|---|---|---|---|
| SH3 | 9.9 ± 3.0 | 71.7 ± 21.3 | 141.2 ± 11.1 | 30 ± 11 | 110 ± 22 | 190 ± 21 | 0.9 ± 0.01 | 0.4 ± 0.02 |
| SH4 | 10.5 ± 2.0 | 78.8 ± 11.3 | 132.2 ± 10.6 | 30 ± 10 | 90 ± 11 | 190 ± 31 | 0.6 ± 0.02 | 0.2 ± 0.01 |
| SH5 | 11.1 ± 2.0 | 85.6 ± 28.4 | 108.8 ± 11.2 | 20 ± 11 | 100 ± 31 | 130 ± 72 | 0.4 ± 0.01 | 0.2 ±0.01 |
| SR3 | 10.1 ± 3.0 | 73.1 ± 23.4 | 281.6 ± 21.5 | 5 ± 2 | 185 ± 45 | 205 ± 52 | 1.0 ± 0.08 | 0.7 ± 0.04 |
| SR4 | 11.2 ± 5.0 | 74.1 ± 23.3 | 237.1 ± 11.4 | 5 ± 2 | 135 ± 50 | 125 ± 81 | 0.8 ± 0.06 | 0.3 ± 0.08 |
| SR5 | 11.3 ± 2.0 | 91.8 ± 18.4 | 203.6 ± 4.0 | 5 ± 1 | 105 ± 22 | 145 ± 75 | 0.4 ± 0.05 | 0.2 ± 0.01 |
| WW3 | 8.1 ± 1.1 | 53.1 ± 41.1 | 127.9 ± 81.8 | 40 ± 10 | 130 ± 11 | 140 ± 24 | 0.7 ± 0.04 | 0.3 ± 0.01 |
| WW4 | 9.5 ± 2.1 | 66.1 ± 3.4 | 87.2 ± 21.6 | 30 ± 11 | 140 ± 41 | 110 ± 52 | 0.6 ± 0.01 | 0.2 ± 0.02 |
| WW5 | 10.1 ± 1.1 | 70.3 ± 14.2 | 96.2 ± 31.6 | 30 ± 11 | 110 ± 11 | 140 ± 20 | 0.4 ± 0.03 | 0.2 ± 0.01 |
| Cu | Pseudo-First-Order (Lagergren) | Pseudo-Second-Order (Ho and Mckay) | Intra-Particle Diffusion Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| k1 × 10−2 (1/min) | qe (mg/g) | R2 | k2 × 10−2 (g/mg·min) | qe (mg/g) | R2 | kD × 10−2 (g/mg·min1/2) | qe (mg/g) | R2 | |
| SH3 | 0.01 ± 0.01 | 6.0 ± 2.2 | 0.9 ± 0.4 | 4.5 ± 1.3 | 9.0 ± 1.1 | 0.99 ± 0.1 | 6.6 ± 1.9 | 5.7 ± 1.7 | 0.98 ± 0.1 |
| SH4 | 0.09 ± 0.01 | 5.8 ± 2.2 | 0.8 ± 0.2 | 8.1 ± 1.8 | 9.2 ± 1.3 | 0.99 ± 0.1 | 6.6 ± 1.0 | 5.7 ± 1.7 | 0.98 ± 0.1 |
| SH5 | 0.15 ± 0.01 | 5.9 ± 2.8 | 0.9 ± 0.3 | 8.2 ± 1.7 | 9.6 ± 0.5 | 0.99 ± 0.1 | 7.4 ± 1.7 | 5.7 ± 1.6 | 0.91 ± 0.2 |
| SR3 | 0.11 ± 0.01 | 6.7 ± 2.7 | 0.9 ± 0.2 | 3.5 ± 1.0 | 9.0 ± 2.1 | 0.99 ± 0.1 | 8.2 ± 1.7 | 6.4 ± 1.2 | 0.94 ± 0.1 |
| SR4 | 0.19 ± 0.01 | 7.8 ± 2.8 | 0.8 ± 0.1 | 7.3 ± 2.0 | 9.4 ± 1.1 | 0.99 ± 0.1 | 12.7 ± 2.9 | 6.8 ± 1.8 | 0.91 ± 0.3 |
| SR5 | 0.35 ± 0.04 | 8.5 ± 2.6 | 0.9 ± 0.1 | 11.0 ± 1.7 | 9.6 ± 1.6 | 0.99 ± 0.1 | 13.5 ± 4.3 | 7.6 ± 1.5 | 0.93 ± 0.2 |
| WW3 | 0.09 ± 0.01 | 4.6 ± 1.2 | 0.9 ± 0.1 | 1.1 ± 0.5 | 6.3 ± 1.3 | 0.99 ± 0.1 | 7.3 ± 1.4 | 4.2 ± 1.4 | 0.94 ± 0.1 |
| WW4 | 0.12 ± 0.01 | 4.9 ± 1.0 | 0.8 ± 0.1 | 1.2 ± 2.7 | 6.3 ± 1.0 | 0.99 ± 0.1 | 8.8 ± 2.4 | 5.6 ± 1.4 | 0.93 ± 0.2 |
| WW5 | 0.12 ± 0.01 | 5.0 ± 1.7 | 0.9 ± 0.3 | 1.9 ± 1.6 | 7.2 ± 2.0 | 0.99 ± 0.1 | 9.2 ± 4.5 | 7.6 ± 1.0 | 0.93 ± 0.1 |
| Ag | Pseudo-First-Order (Lagergren) | Pseudo-Second-Order (Ho and Mckay) | Intra-Particle Diffusion Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| k1 × 10−2 (1/min) | qe (mg/g) | R2 | k2 × 10−2 (g/mg·min) | qe (mg/g) | R2 | kD × 10−2 (g/mg·min1/2) | qe (mg/g) | R2 | |
| SH3 | 0.02 ± 0.01 | 9.1 ± 2.3 | 0.9 ± 0.3 | 4.2 ± 1.1 | 9.9 ± 1.0 | 0.99 ± 0.1 | 2.4 ± 0.2 | 7.3 ± 1.4 | 0.92 ± 0.1 |
| SH4 | 0.02 ± 0.01 | 9.1 ± 3.1 | 0.9 ± 0.1 | 5.2 ± 1.5 | 10.0 ± 0.1 | 0.99 ± 0.1 | 4.6 ± 1.1 | 7.9 ± 1.4 | 0.96 ± 0.1 |
| SH5 | 0.08 ± 0.01 | 9.1 ± 3.8 | 0.9 ± 0.1 | 6.1 ± 1.0 | 10.0 ± 0.4 | 0.98 ± 0.1 | 8.3 ± 2.8 | 8.7 ± 0.2 | 0.92 ± 0.1 |
| SR3 | 0.04 ± 0.01 | 9.1 ± 3.7 | 0.9 ± 0.2 | 5.1 ± 0.9 | 9.1 ± 0.2 | 0.99 ± 0.1 | 10.1 ± 1.1 | 6.9 ± 3.7 | 0.97 ± 0.1 |
| SR4 | 0.06 ± 0.01 | 9.1 ± 2.4 | 0.8 ± 0.1 | 7.9 ± 3.4 | 9.1 ± 0.6 | 0.99 ± 0.1 | 12.9 ± 2.4 | 7.7 ± 1.7 | 0.93 ± 0.1 |
| SR5 | 0.14 ± 0.01 | 9.3 ± 1.0 | 0.9 ± 0.2 | 8.3 ± 2.8 | 10.1 ± 1.0 | 0.99 ± 0.1 | 14.7 ± 1.3 | 8.5 ± 1.7 | 0.93 ± 0.1 |
| WW3 | 0.05 ± 0.01 | 5.2 ± 0.9 | 0.8 ± 0.1 | 1.5 ± 0.3 | 9.2 ± 0.3 | 0.99 ± 0.1 | 6.7 ± 1.8 | 4.3 ± 2.0 | 0.93 ± 0.1 |
| WW4 | 0.05 ± 0.01 | 8.2 ± 2.1 | 0.9 ± 0.2 | 2.1 ± 0.2 | 10.0 ± 0.7 | 0.99 ± 0.1 | 8.4 ± 0.6 | 5.4 ± 1.9 | 0.94 ± 0.1 |
| WW5 | 0.08 ± 0.01 | 9.0 ± 2.1 | 0.8 ± 0.3 | 2.2 ± 0.6 | 10.0 ± 0.8 | 0.99 ± 0.1 | 12.0 ± 1.7 | 6.1 ± 0.4 | 0.89 ± 0.1 |
| Cu | Freundlich Isotherm | Langmuir Isotherm | Langmuir-Freundlich Isotherm | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| KF (mg/g(L/mg)1/n) | 1/n | R2 | KL (L/mg) | Qm (mg/g) | R2 | KLF (L/mg) | Am (mg/g) | m | R2 | |
| SH3 | 2.8 ± 0.5 | 0.3 ± 0.1 | 0.8 ± 0.5 | 0.2 ± 0.1 | 9.9 ± 2.1 | 0.92 ± 0.1 | 0.2 ± 0.1 | 6.4 ± 2.3 | 0.97 ± 0.2 | 0.99 ± 0.01 |
| SH4 | 2.7 ± 0.7 | 0.3 ± 0.1 | 0.8 ± 0.5 | 0.3 ± 0.1 | 11.5 ± 3.5 | 0.95 ± 0.1 | 0.4 ± 0.1 | 7.3 ± 2.3 | 0.94 ± 0.1 | 0.98 ± 0.07 |
| SH5 | 4.5 ± 1.5 | 0.2 ± 0.1 | 0.9 ± 0.1 | 0.5 ± 0.1 | 12.8 ± 5.2 | 0.94 ± 0.1 | 0.4 ± 0.1 | 9.3 ± 4.1 | 0.89 ± 0.2 | 0.99 ± 0.05 |
| SR3 | 0.9 ± 0.2 | 0.7 ± 0.1 | 0.8 ± 0.2 | 0.2 ± 0.1 | 14.3 ± 4.3 | 0.94 ± 0.1 | 0.5 ± 0.1 | 3.0 ± 1.1 | 0.98 ± 0.2 | 0.98 ± 0.04 |
| SR4 | 4.1 ± 1.1 | 0.4 ± 0.1 | 0.8 ± 0.1 | 0.4 ± 0.2 | 14.9 ± 4.0 | 0.95 ± 0.1 | 0.5 ± 0.1 | 10.6 ± 4.2 | 0.89 ± 0.1 | 0.99 ± 0.05 |
| SR5 | 5.4 ± 1.0 | 0.4 ± 0.1 | 0.9 ± 0.1 | 0.5 ± 0.1 | 16.2 ± 5.1 | 0.93 ± 0.1 | 1.3 ± 0.2 | 11.5 ± 6.7 | 0.88 ± 0.1 | 0.98 ± 0.01 |
| WW3 | 0.5 ± 0.1 | 0.4 ± 0.2 | 0.9 ± 0.1 | 0.1 ± 0.1 | 1.8 ± 2.0 | 0.95 ± 0.05 | 0.3 ± 0.1 | 0.6 ± 2.1 | 0.99 ± 0.1 | 0.99 ± 0.04 |
| WW4 | 0.5 ± 0.1 | 0.4 ± 0.3 | 0.9 ± 0.1 | 0.3 ± 0.1 | 3.5 ± 3.1 | 0.95 ± 0.07 | 0.5 ± 0.1 | 1.1 ± 2.9 | 0.95 ± 0.3 | 0.99 ± 0.03 |
| WW5 | 1.4 ± 0.2 | 0.3 ± 0.1 | 0.8 ± 0.2 | 0.3 ± 0.1 | 3.5 ± 2.5 | 0.94 ± 0.05 | 1.0 ± 0.1 | 1.8 ± 1.1 | 0.94 ± 0.2 | 0.99 ± 0.09 |
| Ag | Freundlich Isotherm | Langmuir Isotherm | Langmuir-Freundlich Isotherm | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| KF (mg/g(L/mg)1/n) | 1/n | R2 | KL (L/mg) | Qm (mg/g) | R2 | KLF (L/mg) | Am (mg/g) | m | R2 | |
| SH3 | 7.9 ± 1.2 | 0.6 ± 0.1 | 0.84 ± 0.3 | 0.2 ± 0.1 | 10.9 ± 2.9 | 0.94 ± 0.04 | 0.2 ± 0.1 | 14.5 ± 2.2 | 0.99 ± 0.1 | 0.99 ± 0.04 |
| SH4 | 8.1 ± 1.5 | 0.6 ± 0.1 | 0.84 ± 0.2 | 0.3 ± 0.1 | 14.1 ± 2.4 | 0.94 ± 0.03 | 0.8 ± 0.2 | 18.3 ± 2.2 | 0.97 ± 0.1 | 0.98 ± 0.03 |
| SH5 | 9.0 ± 1.6 | 0.4 ± 0.1 | 0.78 ± 0.2 | 0.4 ± 0.1 | 22.3 ± 2.4 | 0.91 ± 0.04 | 1.2 ± 0.6 | 21.7 ± 1.8 | 0.96 ± 0.1 | 0.99 ± 0.06 |
| SR3 | 8.4 ± 1.1 | 0.4 ± 0.1 | 0.77 ± 0.1 | 0.3 ± 0.1 | 6.9 ± 3.17 | 0.93 ± 0.04 | 0.8 ± 0.2 | 16.1 ± 4.5 | 0.88 ± 0.1 | 0.99 ± 0.05 |
| SR4 | 9.9 ± 1.7 | 0.3 ± 0.1 | 0.71 ± 0.1 | 0.3 ± 0.1 | 10.6 ± 2.8 | 0.94 ± 0.02 | 0.9 ± 0.1 | 27.9 ± 6.8 | 0.74 ± 0.1 | 0.99 ± 0.03 |
| SR5 | 11.0 ± 1.1 | 0.3 ± 0.01 | 0.76 ± 0.3 | 0.5 ± 0.1 | 14.0 ± 5.1 | 0.93 ± 0.04 | 1.6 ± 0.3 | 46.2 ± 2.3 | 0.62 ± 0.1 | 0.99 ± 0.04 |
| WW3 | 3.7 ± 1.4 | 0.5 ± 0.2 | 0.77 ± 0.1 | 0.5 ± 0.1 | 9.6 ± 1.2 | 0.92 ± 0.06 | 0.4 ± 0.1 | 12.1 ± 1.0 | 0.98 ± 0.1 | 0.98 ± 0.04 |
| WW4 | 5.3 ± 1.6 | 0.4 ± 0.1 | 0.84 ± 0.5 | 0.6 ± 0.2 | 14.6 ± 2.5 | 0.90 ± 0.01 | 0.3 ± 0.1 | 19.2 ± 3.5 | 0.83 ± 0.1 | 0.99 ± 0.05 |
| WW5 | 5.7 ± 1.0 | 0.4 ± 0.1 | 0.79 ± 0.7 | 0.7 ± 0.1 | 17.8 ± 2.2 | 0.90 ± 0.03 | 1.0 ± 0.2 | 26.8 ± 4.9 | 0.50 ± 0.1 | 0.99 ± 0.01 |
| SH3 | SH4 | SH5 | SR3 | SR4 | SR5 | WW3 | WW4 | WW5 | |
|---|---|---|---|---|---|---|---|---|---|
| Cu | 1.5 ± 0.3 | 1.7 ± 0.8 | 1.9 ± 0.2 | 1.2 ± 0.3 | 1.7 ± 0.4 | 2.4 ± 0.3 | 0.3 ± 0.1 | 0.6 ± 0.2 | 0.6 ± 0.1 |
| Ag | 2.7 ± 1.2 | 3.4 ± 0.5 | 4.2 ± 0.4 | 3.8 ± 1.1 | 4.6 ± 1.8 | 5.6 ± 1.8 | 2.2 ± 0.9 | 3.3 ± 1.1 | 4.0 ± 1.2 |
| SH3 | SH4 | SH5 | SR3 | SR4 | SR5 | WW3 | WW4 | WW5 | |
|---|---|---|---|---|---|---|---|---|---|
| Cu | 40.7 ± 12.3 | 43.3 ± 7.4 | 44.2 ± 21.4 | 69.2 ± 3.6 | 71.4 ± 12.2 | 71.5 ± 25.6 | 13.7 ± 14.7 | 19.1 ± 2.3 | 20.3 ± 7.8 |
| Ag | 93.6 ± 24.0 | 98.5 ± 9.9 | 99.2 ± 13.9 | 77.2 ± 18.7 | 95.6 ± 12.1 | 99.4 ± 17.9 | 43.4 ± 24.9 | 50.3 ± 25.9 | 65.4 ± 16.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomczyk, A.; Sokołowska, Z.; Boguta, P.; Szewczuk-Karpisz, K. Comparison of Monovalent and Divalent Ions Removal from Aqueous Solutions Using Agricultural Waste Biochars Prepared at Different Temperatures—Experimental and Model Study. Int. J. Mol. Sci. 2020, 21, 5851. https://doi.org/10.3390/ijms21165851
Tomczyk A, Sokołowska Z, Boguta P, Szewczuk-Karpisz K. Comparison of Monovalent and Divalent Ions Removal from Aqueous Solutions Using Agricultural Waste Biochars Prepared at Different Temperatures—Experimental and Model Study. International Journal of Molecular Sciences. 2020; 21(16):5851. https://doi.org/10.3390/ijms21165851
Chicago/Turabian StyleTomczyk, Agnieszka, Zofia Sokołowska, Patrycja Boguta, and Katarzyna Szewczuk-Karpisz. 2020. "Comparison of Monovalent and Divalent Ions Removal from Aqueous Solutions Using Agricultural Waste Biochars Prepared at Different Temperatures—Experimental and Model Study" International Journal of Molecular Sciences 21, no. 16: 5851. https://doi.org/10.3390/ijms21165851
APA StyleTomczyk, A., Sokołowska, Z., Boguta, P., & Szewczuk-Karpisz, K. (2020). Comparison of Monovalent and Divalent Ions Removal from Aqueous Solutions Using Agricultural Waste Biochars Prepared at Different Temperatures—Experimental and Model Study. International Journal of Molecular Sciences, 21(16), 5851. https://doi.org/10.3390/ijms21165851








