Preventing Tumour Recurrence after Liver Transplantation: The Role of Machine Perfusion
Abstract
1. Introduction
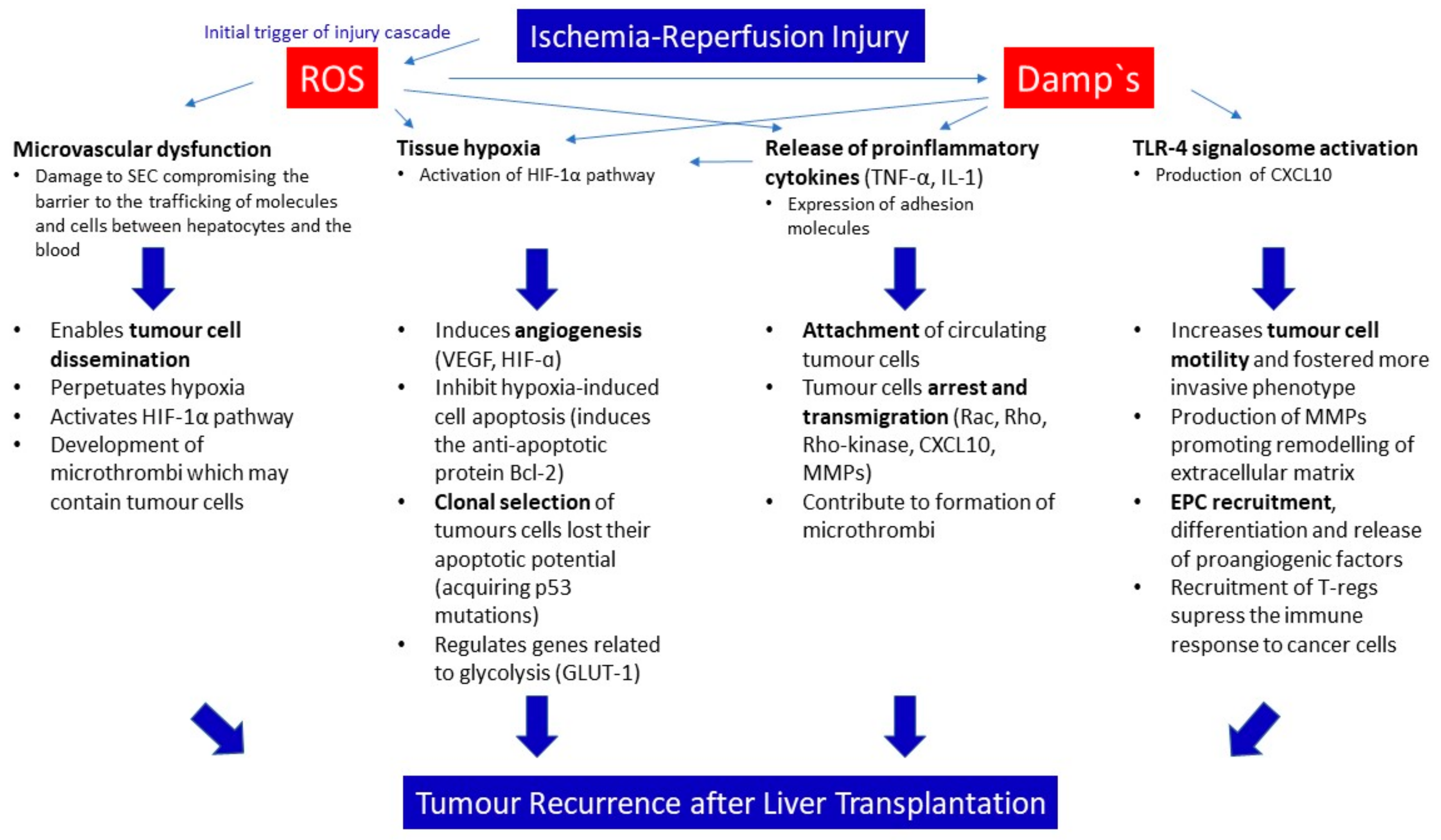
2. Mechanisms of Ischaemia-Reperfusion Injury
3. The Link between Ischaemia-Reperfusion Injury and Tumour Recurrence after Liver Transplantation
3.1. The Favourable Microenvironment
3.2. A Higher Aggressiveness of Tumour Cells
3.3. The Mobilisation of Progenitor Cells
4. The Clinical Evidence for the Link between Advanced IR Injury and Tumour Recurrence after Liver Transplantation
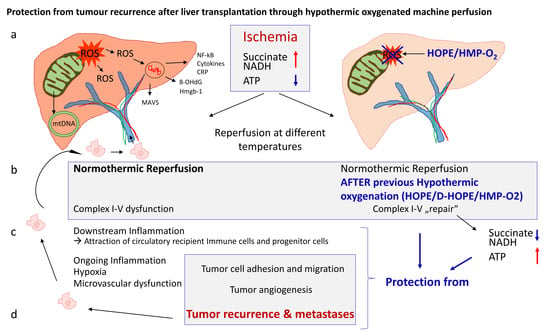
5. The Potential Role of Machine Perfusion of the Liver on Tumour Recurrence
5.1. Hypothermic Machine Perfusion of the Liver
5.2. Normothermic Machine Perfusion of the Liver (NMP)
6. Summary
Funding
Conflicts of Interest
Abbreviations
| AMP | Adenosine monophosphate |
| ADP | Adenosine diphosphate |
| ALT | Alanine aminotransferase |
| AS | Anastomotic strictures |
| AST | Aspartate-aminotransferase |
| ATP | Adenosine triphosphate |
| CD-68 | Cluster of differentiation 68 |
| CIT | Cold ischemia time |
| CS | Cold storage |
| XCL-10 | C-X-C motif ligand 10 |
| CXCR-3 | CXC-chemokine receptor-3 |
| DAMPs | Danger associated molecular patterns |
| DBD | Donation after brain death |
| DBQ | Decylubiquinone |
| DCD | Donation after circulatory death |
| D-HOPE | Dual hypothermic oxygenated perfusion |
| dWIT | Donor warm ischemia time |
| EAD | Early allograft dysfunction |
| ECD | Extended criteria donor |
| ECMO | Extracorporeal membrane oxygenation |
| FAD | Flavin adenine dinucleotide |
| FMN | Flavin mononucleotide |
| GSH | Glutathione |
| HA | Hepatic artery |
| HAR | Hexaammineruthenium |
| HAT | Hepatic artery thrombosis |
| HCC | Hepatocellular carcinoma |
| HIF-1α | Hypoxia-inducible factor-1α |
| HMGB-1 | High mobility group box-1 protein |
| HMP | Hypothermic machine perfusion |
| HOPE | Hypothermic oxygenated perfusion |
| H&E | Hematoxylin and Eosin |
| HR | Hazard ratio |
| IC | Ischemic cholangiopathy |
| ICAM-1 | Intercellular adhesion molecule-1; |
| ICU | Intensive care unit |
| IFOT | Ischaemia-free organ transplantation |
| IMP | Inosine monophosphate |
| IFOT | Ischaemia-free organ transplantation |
| KC‘s | Kupffer cells |
| LDH | Lactate dehydrogenase |
| LT | Liver transplantation |
| MELD | Model of end stage liver disease |
| MMP | Matrix metalloproteinases |
| MPS | Machine perfusion solution |
| MPT pore | Mitochondria permeability transition pore |
| NAD/NADH | Nicotine adenine dinucleotide (oxidized/reduced) |
| NADPH | Nicotinamide adenine dinucleotide phosphate hydrogen |
| NAS | Non-anastomotic strictures |
| NRP | Normothermic regional perfusion |
| NMP | Normothermic machine perfusion |
| OAA | Oxaloacetate |
| OLT | Orthotopic liver transplantation |
| PEG-1 | Prostaglandin E1 |
| PMH | Past medical history |
| PNF | Primary non function |
| PV | Portal vein |
| RET | Reverse electron flow |
| ROS | Reactive oxygen species |
| SDH | Succinate dehydrogenase |
| SEC | Sinusoidal endothelial cells |
| TLR-4 | Toll-like-receptor-4 |
| Tregs | Regulatory T cells |
| VCAM-1 | Vascular cell adhesion molecule 1 |
| VEGF | Vascular endothelial grow factor |
| 8-OHdG | 8-hydroxy-2-deoxy Guanosine |
References
- Yang, J.D.; Larson, J.J.; Watt, K.D.; Allen, A.M.; Wiesner, R.H.; Gores, G.J.; Roberts, L.R.; Heimbach, J.A.; Leise, M.D. Hepatocellular Carcinoma Is the Most Common Indication for Liver Transplantation and Placement on the Waitlist in the United States. Clin. Gastroenterol. Hepatol. 2017, 15, 767–775. [Google Scholar] [CrossRef] [PubMed]
- De’Angelis, N.; Landi, F.; Carra, M.C.; Azoulay, D. Managements of recurrent hepatocellular carcinoma after liver transplantation: A systematic review. World J. Gastroenterol. 2015, 21, 11185. [Google Scholar] [CrossRef] [PubMed]
- Bodzin, A.S.; Lunsford, K.E.; Markovic, D.; Harlander-Locke, M.P.; Busuttil, R.W.; Agopian, V.G. Predicting Mortality in Patients Developing Recurrent Hepatocellular Carcinoma after Liver Transplantation: Impact of Treatment Modality and Recurrence Characteristics. Ann. Surg. 2017, 266, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Duvoux, C.; Roudot-Thoraval, F.; Decaens, T.; Pessione, F.; Badran, H.; Piardi, T.; Francoz, C.; Compagnon, P.; Vanlemmens, C.; Dumortier, J.; et al. Liver transplantation for hepatocellular carcinoma: A model including α-fetoprotein improves the performance of milan criteria. Gastroenterology 2012, 143, 986–994. [Google Scholar] [CrossRef]
- Nagai, S.; Yoshida, A.; Facciuto, M.; Moonka, D.; Abouljoud, M.S.; Schwartz, M.E.; Florman, S.S. Ischemia time impacts recurrence of hepatocellular carcinoma after liver transplantation. Hepatology 2015, 61, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Dutkowski, P.; Oberkofler, C.E.; Slankamenac, K.; Puhan, M.A.; Schadde, E.; Mullhaupt, B.; Geier, A.; Clavien, P.A. Are there better guidelines for allocation in liver transplantation? A novel score targeting justice and utility in the model for end-stage liver disease era. Ann. Surg. 2011, 254, 745–753; discussion 753. [Google Scholar] [CrossRef] [PubMed]
- De Rougemont, O.; Dutkowski, P.; Clavien, P.-A. Clinical update on inflammatory disorders of the GI tract: Liver transplantation. Front. Gastrointest. Res. 2010, 26, 59–71. [Google Scholar]
- Ghinolfi, D.; Rreka, E.; Pezzati, D.; Filipponi, F.; De Simone, P. Perfusion machines and hepatocellular carcinoma: A good match between a marginal organ and an advanced disease? Transl. Gastroenterol. Hepatol. 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Grat, M.; Krawczyk, M.; Wronka, K.M.; Stypułkowski, J.; Lewandowski, Z.; Wasilewicz, M.; Krawczyk, P.; Grat, K.; Patkowski, W.; Zieniewicz, K. Ischemia-reperfusion injury and the risk of hepatocellular carcinoma recurrence after deceased donor liver transplantation. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef]
- Schlegel, A.; Muller, X.; Dutkowski, P. Machine perfusion strategies in liver transplantation. Hepatobiliary Surg. Nutr. 2019, 8, 490. [Google Scholar] [CrossRef]
- Boteon, Y.L.; Afford, S.C. Machine perfusion of the liver: Which is the best technique to mitigate ischaemia-reperfusion injury? World J. Transplant. 2019, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Petrowsky, H.; Hong, J.C.; Busuttil, R.W.; Kupiec-Weglinski, J.W. Ischaemia-reperfusion injury in liver transplantation—From bench to bedside. Nat. Rev. Gastroentrerol. Hepatol. 2013, 10, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.; Costa, A.; Gruszczyk, A.; Beach, T.; Allen, F.; Prag, H.; Hinchy, E.; Mahbubani, K.; Hamed, M.; Tronci, L.; et al. Succinate accumulation drives ischaemia-reperfusion injury during organ transplantation. Nat. Metab. 2019, 1, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Eltzschig, H.K.; Eckle, T. Ischemia and reperfusion-from mechanism to translation. Nat. Med. 2011, 17, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Chouchani, E.T.; Pell, V.R.; Gaude, E.; Aksentijević, D.; Sundier, S.Y.; Robb, E.L.; Logan, A.; Nadtochiy, S.M.; Ord, E.N.J.; Smith, A.C.; et al. Ischaemic accumulation of succinate controls reperfusion injury through mitochondrial ROS. Nature 2014, 515, 431–435. [Google Scholar] [CrossRef]
- Dröse, S.; Brandt, U.; Wittig, I. Mitochondrial respiratory chain complexes as sources and targets of thiol-based redox-regulation. Biochim. Biophys. Acta Proteins Proteom. 2014, 1844, 1344–1354. [Google Scholar] [CrossRef]
- Dröse, S. Differential effects of complex II on mitochondrial ROS production and their relation to cardioprotective pre- and postconditioning. Biochim. Biophys. Acta Bioenerg. 2013, 1827, 578–587. [Google Scholar] [CrossRef]
- Nakahira, K.; Hisata, S.; Choi, A.M.K. The Roles of Mitochondrial Damage-Associated Molecular Patterns in Diseases. Antioxid. Redox Signal. 2015, 23, 1329–1350. [Google Scholar] [CrossRef]
- Menger, M.D.; Richter, S.; Yamauchi, J.; Vollmar, B. Role of microcirculation in hepatic ischemia/reperfusion injury. Hepatogastroenterology 1999, 46, 1452–1457. [Google Scholar]
- Takeda, K.; Jin, M.B.; Fujita, M.; Fukai, M.; Sakurai, T.; Nakayama, M.; Taniguchi, M.; Suzuki, T.; Shimamura, T.; Furukawa, H.; et al. A novel inhibitor of Rho-associated protein kinase, Y-27632, ameliorates hepatic ischemia and reperfusion injury in rats. Surgery 2003, 133, 197–206. [Google Scholar] [CrossRef]
- Li, C.X.; Man, K.; Lo, C.M. The Impact of Liver Graft Injury on Cancer Recurrence Posttransplantation. Transplantation 2017, 101, 2665. [Google Scholar] [CrossRef]
- Li, C.X.; Ling, C.C.; Shao, Y.; Xu, A.; Li, X.C.; Ng, K.T.P.; Liu, X.B.; Ma, Y.Y.; Qi, X.; Liu, H.; et al. CXCL10/CXCR3 signaling mobilized-regulatory T cells promote liver tumor recurrence after transplantation. J. Hepatol. 2016, 65, 944–952. [Google Scholar] [CrossRef]
- Zhai, Y.; Da Shen, X.; Gao, F.; Zhao, A.; Freitas, M.C.; Lassman, C.; Luster, A.D.; Busuttil, R.W.; Kupiec-Weglinski, J.W. CXCL10 regulates liver innate immune response against ischemia and reperfusion injury. Hepatology 2008, 47, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Ling, C.C.; Ng, K.T.P.; Shao, Y.; Geng, W.; Xiao, J.W.; Liu, H.; Li, C.X.; Liu, X.B.; Ma, Y.Y.; Yeung, W.H.; et al. Post-transplant endothelial progenitor cell mobilization via CXCL10/CXCR3 signaling promotes liver tumor growth. J. Hepatol. 2014, 60, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Dar, W.A.; Sullivan, E.; Bynon, J.S.; Eltzschig, H.; Ju, C. Ischaemia reperfusion injury in liver transplantation: Cellular and molecular mechanisms. Liver Int. 2019, 39, 788–801. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, Z.; Xiao, Q.; Zeng, C.; Lai, C.H.; Fan, X.; Ye, Q.; Wang, Y.; Xiong, Y. Donor Treatment with a Hypoxia-Inducible Factor-1 Agonist Prevents Donation after Cardiac Death Liver Graft Injury in a Rat Isolated Perfusion Model. Artif. Organs 2018, 42, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Van Der Bilt, J.D.W.; Soeters, M.E.; Duyverman, A.M.M.J.; Nijkamp, M.W.; Witteveen, P.O.; Van Diest, P.J.; Kranenburg, O.; Rinkes, I.H.M.B. Perinecrotic hypoxia contributes to ischemia/reperfusion-accelerated outgrowth of colorectal micrometastases. Am. J. Pathol. 2007, 170, 1379–1388. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shannon, A.M.; Bouchier-Hayes, D.J.; Condron, C.M.; Toomey, D. Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Cancer Treat. Rev. 2003, 29, 297–307. [Google Scholar] [CrossRef]
- Ryan, H.E.; Lo, J.; Johnson, R.S. HIF-1α is required for solid tumor formation and embryonic vascularization. EMBO J. 1998, 17, 3005–3015. [Google Scholar] [CrossRef]
- Pidgeon, G.P.; Barr, M.P.; Harmey, J.H.; Foley, D.A.; Bouchier-Hayes, D.J. Vascular endothelial growth factor (VEGF) upregulates BCL-2 and inhibits apoptosis in human and murine mammary adenocarcinoma cells. Br. J. Cancer 2001, 85, 273–278. [Google Scholar] [CrossRef]
- Graeber, T.G.; Osmanian, C.; Jacks, T.; Housman, D.E.; Koch, C.J.; Lowe, S.W.; Giaccia, A.J. Hypoxia-mediated selection of cells with diminished apoptotic potential in solid tumours. Nature 1996, 379, 88–91. [Google Scholar] [CrossRef] [PubMed]
- Engelberg, H. Actions of heparin that may affect the malignant process. Cancer 1999, 85, 257–272. [Google Scholar] [CrossRef]
- Yonson, K.; Kusunoki, N.; Shiotani, M.; Maeda, I.; Iwasaki, T.; Tominaga, M.; Kitagawa, T.; Fukumoto, T.; Suzuki, Y.; Kuroda, Y. Stimulation of haematogenous liver metastases by ischaemia-reperfusion in rats. Eur. J. Surg. 1999, 165, 801–807. [Google Scholar] [CrossRef]
- Brodt, P.; Fallavollita, L.; Bresalier, R.S.; Meterissian, S.; Norton, C.R.; Wolitzky, B.A. Liver endothelial E-selectin mediates carcinoma cell adhesion and promotes liver metastasis. Int. J. Cancer 1997, 71, 612–619. [Google Scholar] [CrossRef]
- Higashiyama, A.; Watanabe, H.; Okumura, K.; Yagita, H. Involvement of tumor necrosis factor α and very late activation antigen 4/vascular cell adhesion molecule 1 interaction in surgical-stress-enhanced experimental metastasis. Cancer Immunol. Immunother. 1996, 181, 411–415. [Google Scholar] [CrossRef]
- Khatib, A.M.; Kontogiannea, M.; Fallavollita, L.; Jamison, B.; Meterissian, S.; Brodt, P. Rapid induction of cytokine and E-selectin expression in the liver in response to metastatic tumor cells. Cancer Res. 1999, 59, 1356–1361. [Google Scholar]
- Biancone, L.; Araki, M.; Araki, K.; Vassalli, P.; Stamenkovic, I. Redirection of tumor metastasis by expression of E-selectin in vivo. J. Exp. Med. 1996, 183, 581–587. [Google Scholar] [CrossRef]
- Huh, S.J.; Liang, S.; Sharma, A.; Dong, C.; Robertson, G.P. Transiently entrapped circulating tumor cells interact with neutrophils to facilitate lung metastasis development. Cancer Res. 2010, 70, 6071–6082. [Google Scholar] [CrossRef]
- Man, K.; Ng, K.T.; Lo, C.M.; Ho, J.W.; Sun, B.S.; Sun, C.K.; Lee, T.K.; Poon, R.T.P.; Fan, S.T. Ischemia-reperfusion of small liver remnant promotes liver tumor growth and metastases—Activation of cell invasion and migration pathways. Liver Transplant. 2007, 13, 1669–1677. [Google Scholar] [CrossRef]
- Ogawa, T.; Tashiro, H.; Miyata, Y.; Ushitora, Y.; Fudaba, Y.; Kobayashi, T.; Arihiro, K.; Okajima, M.; Asahara, T. Rho-associated kinase inhibitor reduces tumor recurrence after liver transplantation in a rat hepatoma model. Am. J. Transplant. 2007, 7, 347–355. [Google Scholar] [CrossRef]
- Man, K.; Co Shih, K.; Ng, K.T.P.; Xiao, J.W.; Guo, D.Y.; Sun, C.K.W.; Lim, Z.X.H.; Cheng, Q.; Liu, Y.; Fan, S.T.; et al. Molecular signature linked to acute phase injury and tumor invasiveness in small-for-size liver grafts. Ann. Surg. 2010, 251, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.F.; Leu, F.J.; Chen, H.I.; Wang, D. Oxygen radicals and matrix metalloproteinases mediate reperfusion liver injury. Transplant. Proc. 2005, 37, 4547–4549. [Google Scholar] [CrossRef] [PubMed]
- Khandoga, A. Matrix metalloproteinase-9 promotes neutrophil and T cell recruitment and migration in the postischemic liver. J. Leukoc. Biol. 2006, 79, 1295–1305. [Google Scholar] [CrossRef] [PubMed]
- Nicoud, I.B.; Jones, C.M.; Pierce, J.M.; Earl, T.M.; Matrisian, L.M.; Chari, R.S.; Gorden, D.L. Warm hepatic ischemia-reperfusion promotes growth of colorectal carcinoma micrometastases in mouse liver via matrix metalloproteinase-9 induction. Cancer Res. 2007, 67, 2720–2728. [Google Scholar] [CrossRef][Green Version]
- Ho, J.W.Y.; Pang, R.W.C.; Lau, C.; Sun, C.K.; Wan, C.Y.; Sheung, T.F.; Poon, R.T.P. Significance of circulating endothelial progenitor cells in hepatocellular carcinoma. Hepatology 2006, 44, 836–843. [Google Scholar] [CrossRef]
- Wolf, A.M.; Wolf, D.; Steurer, M.; Gastl, G.; Gunsilius, E.; Grubeck-Loebenstein, B. Increase of regulatory T cells in the peripheral blood of cancer patients. Clin. Cancer Res. 2003, 9, 606–612. [Google Scholar]
- Schlegel, A.; Kron, P.; Graf, R.; Clavien, P.-A.; Dutkowski, P. Hypothermic Oxygenated Perfusion (HOPE) downregulates the immune response in a rat model of liver transplantation. Ann. Surg. 2014, 260, 931–937; discussion 937–938. [Google Scholar] [CrossRef]
- Silverstein, J.; Roll, G.; Dodge, J.L.; Grab, J.D.; Yao, F.Y.; Mehta, N. Donation after Circulatory Death is Associated with Similar Post-Transplant Survival in All but the Highest Risk Hepatocellular Carcinoma Patients. Liver Transplant. 2020. [Google Scholar] [CrossRef]
- Martinez-Insfran, L.A.; Ramirez, P.; Cascales, P.; Alconchel, F.; Ferreras, D.; Febrero, B.; Martinez, M.; González, M.R.; Sanchez-Bueno, F.; Robles, R.; et al. Early Outcomes of Liver Transplantation Using Donors after Circulatory Death in Patients with Hepatocellular Carcinoma: A Comparative Study. Transplant. Proc. 2019, 2, 359–364. [Google Scholar] [CrossRef]
- Khorsandi, S.E.; Yip, V.S.; Cortes, M.; Jassem, W.; Quaglia, A.; O’Grady, J.; Heneghan, M.; Aluvihare, V.; Agarwal, K.; Menon, K.; et al. Does donation after cardiac death utilization adversely affect hepatocellular cancer survival? Transplantation 2016, 100, 1916–1924. [Google Scholar] [CrossRef]
- Orci, L.A.; Lacotte, S.; Delaune, V.; Slits, F.; Oldani, G.; Lazarevic, V.; Rossetti, C.; Rubbia-Brandt, L.; Morel, P.; Toso, C. Effects of the gut–liver axis on ischaemia-mediated hepatocellular carcinoma recurrence in the mouse liver. J. Hepatol. 2018, 68, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Kornberg, A.; Witt, U.; Kornberg, J.; Friess, H.; Thrum, K. Extended Ischemia Times Promote Risk of HCC Recurrence in Liver Transplant Patients. Dig. Dis. Sci. 2015, 60, 2832–2839. [Google Scholar] [CrossRef] [PubMed]
- Croome, K.P.; Lee, D.D.; Burns, J.M.; Musto, K.; Paz, D.; Nguyen, J.H.; Perry, D.K.; Harnois, D.M.; Taner, C.B. The Use of Donation after Cardiac Death Allografts Does Not Increase Recurrence of Hepatocellular Carcinoma. Am. J. Transplant. 2015, 15, 2704–2711. [Google Scholar] [CrossRef] [PubMed]
- Croome, K.P.; Wall, W.; Chandok, N.; Beck, G.; Marotta, P.; Hernandez-Alejandro, R. Inferior survival in liver transplant recipients with hepatocellular carcinoma receiving donation after cardiac death liver allografts. Liver Transplant. 2013, 19, 1214–1223. [Google Scholar] [CrossRef]
- Kornberg, A.; Witt, U.; Kornberg, J.; Friess, H.; Thrum, K. Treating ischaemia-reperfusion injury with prostaglandin E1 reduces the risk of early hepatocellular carcinoma recurrence following liver transplantation. Aliment. Pharmacol. Ther. 2015, 42, 1101–1110. [Google Scholar] [CrossRef]
- Lim, C.; Azoulay, D. Ischemia–Reperfusion Injury and Regrowth of Liver Cancers: To Clamp or Not to Clamp? Dig. Dis. Sci. 2014, 2030–2031. [Google Scholar] [CrossRef]
- Quintini, C.; Hashimoto, K.; Uso, T.D.; Miller, C. Is there an advantage of living over deceased donation in liver transplantation? Transpl. Int. 2013, 26, 11–19. [Google Scholar] [CrossRef]
- Fishbein, T.M.; Fiel, M.I.; Emre, S.; Cubukcu, O.; Guy, S.R.; Schwartz, M.E.; Miller, C.M.; Sheiner, P.A. Use of livers with microvesicular fat safely expands the donor pool. Transplantation 1997, 64, 248–251. [Google Scholar] [CrossRef]
- Feng, S.; Goodrich, N.P.; Bragg-Gresham, J.L.; Dykstra, D.M.; Punch, J.D.; DebRoy, M.A.; Greenstein, S.M.; Merion, R.M. Characteristics associated with liver graft failure: The concept of a donor risk index. Am. J. Transplant. 2006, 6, 783–790. [Google Scholar] [CrossRef]
- Schlegel, A.A.; Kalisvaart, M.; Muiesan, P. Machine perfusion in liver transplantation: An essential treatment or just an expensive toy? Minerva Anestesiol. 2018, 84, 236–245. [Google Scholar]
- Karangwa, S.; Panayotova, G.; Dutkowski, P.; Porte, R.J.; Guarrera, J.V.; Schlegel, A. Hypothermic Machine Perfusion in Liver Transplantation. Int. J. Surg. 2020, 21, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Graham, J.A.; Guarrera, J.V. “Resuscitation” of marginal liver allografts for transplantation with machine perfusion technology. J. Hepatol. 2014, 61, 418–431. [Google Scholar] [CrossRef] [PubMed]
- Mills, E.L.; Kelly, B.; Logan, A.; Costa, A.S.H.; Varma, M.; Bryant, C.E.; Tourlomousis, P.; Däbritz, J.H.M.; Gottlieb, E.; Latorre, I.; et al. Succinate Dehydrogenase Supports Metabolic Repurposing of Mitochondria to Drive Inflammatory Macrophages. Cell 2016, 167, 457–470.e13. [Google Scholar] [CrossRef] [PubMed]
- Chouchani, E.T.; Pell, V.R.; James, A.M.; Work, L.M.; Saeb-Parsy, K.; Frezza, C.; Krieg, T.; Murphy, M.P. A unifying mechanism for mitochondrial superoxide production during ischemia-reperfusion injury. Cell Metab. 2016, 23, 254–263. [Google Scholar] [CrossRef]
- Granger, D.N.; Kvietys, P.R. Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol. 2015, 6, 524–551. [Google Scholar] [CrossRef]
- Reiniers, M.J.; van Golen, R.F.; van Gulik, T.M.; Heger, M. Reactive oxygen and nitrogen species in steatotic hepatocytes: A molecular perspective on the pathophysiology of ischemia-reperfusion injury in the fatty liver. Antioxid. Redox Signal. 2014, 21, 1119–1142. [Google Scholar] [CrossRef]
- Muller, X.; Schlegel, A.; Kron, P.; Eshmuminov, D.; Würdinger, M.; Meierhofer, D.; Clavien, P.; Dutkowski, P. Novel real time prediction of liver graft function during hypothermic oxygenated machine perfusion prior to liver transplantation. Ann. Surg. 2019, 270, 783–790. [Google Scholar] [CrossRef]
- Gielisch, I.; Meierhofer, D. Metabolome and proteome profiling of complex i deficiency induced by rotenone. J. Proteome Res. 2015, 14, 224–235. [Google Scholar] [CrossRef]
- Stepanova, A.; Sosunov, S.; Niatsetskaya, Z.; Konrad, C.; Starkov, A.; Manfredi, G.; Wittig, I.; Ten, V.; Galkin, A. Redox-Dependent Loss of Flavin by Mitochondrial Complex I in Brain Ischemia/Reperfusion Injury. Antioxid. Redox Signal. 2019, 608–622. [Google Scholar] [CrossRef]
- Kahl, A.; Stepanova, A.; Konrad, C.; Anderson, C.; Manfredi, G.; Zhou, P.; Iadecola, C.; Galkin, A. Critical Role of Flavin and Glutathione in Complex I-Mediated Bioenergetic Failure in Brain Ischemia/Reperfusion Injury. Stroke 2018, 49, 1223–1231. [Google Scholar] [CrossRef]
- Stepanova, A.; Shurubor, Y.; Valsecchi, F.; Manfredi, G.; Galkin, A. Differential susceptibility of mitochondrial complex II to inhibition by oxaloacetate in brain and heart. Biochim. Biophys. Acta Bioenerg. 2016, 1857, 1561–1568. [Google Scholar] [CrossRef]
- Holt, P.J.; Efremov, R.G.; Nakamaru-Ogiso, E.; Sazanov, L.A. Reversible FMN dissociation from Escherichia coli respiratory complex I. Biochim. Biophys. Acta Bioenerg. 2016, 1857, 1777–1785. [Google Scholar] [CrossRef] [PubMed]
- Longatto Boteon, Y.; Schlegel, A.; Laing, R.; Attard, J.; Bhogal, R.; Wallace, L.; Reynolds, G.; Mirza, D.; Mergental, H.; Afford, S. Combination of hypothermic oxygenated machine perfusion followed by normothermic machine perfusion optimises the reconditioning of marginal human donor livers. HPB 2018, 20, S823. [Google Scholar] [CrossRef][Green Version]
- Boteon, Y.; Afford, S.; Mergental, H. Pushing the Limits: Machine Preservation of the Liver as a Tool to Recondition High-Risk Grafts. Curr. Transplant. Rep. 2018, 5, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, A.; Kron, P.; Graf, R.; Dutkowski, P.; Clavien, P.A. Warm vs. cold perfusion techniques to rescue rodent liver grafts. J. Hepatol. 2014, 61, 1267–1275. [Google Scholar] [CrossRef] [PubMed]
- Kron, P.; Schlegel, A.; Mancina, L.; Clavien, P.A.; Dutkowski, P. Hypothermic oxygenated perfusion (HOPE) for fatty liver grafts in rats and humans. J. Hepatol. 2018, 68, 82–91. [Google Scholar] [CrossRef]
- Hosgood, S.A.; Moore, T.; Kleverlaan, T.; Adams, T.; Nicholson, M.L. Haemoadsorption reduces the inflammatory response and improves blood flow during ex vivo renal perfusion in an experimental model. J. Transl. Med. 2017, 15, s12967-s017. [Google Scholar] [CrossRef]
- Nakajima, D.; Chen, F.; Yamada, T.; Sakamoto, J.; Osumi, A.; Fujinaga, T.; Shoji, T.; Sakai, H.; Bando, T.; Date, H. Hypothermic machine perfusion ameliorates ischemia-reperfusion injury in rat lungs from non-heart-beating donors. Transplantation 2011, 92, 858–863. [Google Scholar] [CrossRef]
- Michel, S.G.; La Muraglia, G.M.; Madariaga, M.L.L.; Titus, J.S.; Selig, M.K.; Farkash, E.A.; Allan, J.S.; Anderson, L.M.; Madsen, J.C. Twelve-hour hypothermic machine perfusion for donor heart preservation leads to improved ultrastructural characteristics compared to conventional cold storage. Ann. Transplant. 2015, 20, 461–468. [Google Scholar] [CrossRef]
- Jochmans, I.; Akhtar, M.Z.; Nasralla, D.; Kocabayoglu, P.; Boffa, C.; Kaisar, M.; Brat, A.; O’Callaghan, J.; Pengel, L.H.M.; Knight, S.; et al. Past, Present, and Future of Dynamic Kidney and Liver Preservation and Resuscitation. Am. J. Transplant. 2016, 16, 2545–2555. [Google Scholar] [CrossRef]
- Westerkamp, A.; Karimian, N.; Matton, A.; Mahboub, P.; van Rijn, R.; Wiersema-Buist, J.; de Boer, M.; Leuvenink, H.; Gouw, A.; Lisman, T.; et al. Oxygenated Hypothermic Machine Perfusion After Static Cold Storage Improves Hepatobiliary Function of Extended Criteria Donor Livers. Transplantation 2016, 100, 825–835. [Google Scholar] [CrossRef] [PubMed]
- De Vries, Y.; Matton, A.; Nijsten, M.; Werner, M.; van den Berg, A.; de Boer, M.; Buis, C.; Fujiyoshi, M.; de Kleine, R.; van Leeuwen, O.; et al. Pretransplant Sequential Hypo- and Normothermic Machine Perfusion of Suboptimal Livers Donated after Circulatory Death Using a Hemoglobin-based Oxygen Carrier Perfusion Solution. Am. J. Transplant. 2018, 19, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Guarrera, J.V.; Henry, S.D.; Samstein, B.; Reznik, E.; Musat, C.; Lukose, T.I.; Ratner, L.E.; Brown, R.S.; Kato, T.; Emond, J.C. Hypothermic machine preservation facilitates successful transplantation of “orphan” extended criteria donor livers. Am. J. Transplant. 2015, 15, 161–169. [Google Scholar] [CrossRef]
- Schlegel, A.A.; Muller, X.; Kalisvaart, M.; Muellhaupt, B.; Perera, M.; Isaac, J.; Clavien, P.; Muiesan, P.; Dutkowski, P. Outcomes of liver transplantations from donation after circulatory death (DCD) treated by hypothermic oxygenated perfusion (HOPE) before implantation. J. Hepatol. 2019, 50–57. [Google Scholar] [CrossRef]
- Schlegel, A.; De Rougemont, O.; Graf, R.; Clavien, P.A.; Dutkowski, P. Protective mechanisms of end-ischemic cold machine perfusion in DCD liver grafts. J. Hepatol. 2013, 58, 278–286. [Google Scholar] [CrossRef]
- Schlegel, A.; Muller, X.; Dutkowski, P. Hypothermic Machine Preservation of the Liver: State of the Art. Curr. Transplant. Rep. 2018, 5, 93–102. [Google Scholar] [CrossRef]
- Panayotova, G.; Cutler, Q.; Paterno, F.; McCarty, M.; Bailey, A.; Dikdan, G.; Rosado, J.; Schlegel, A.; Dutkowski, P.; Shah, S.; et al. A Novel Biomarker to Predict Ischemia/Reperfusion Injury after Hypothermic Oxygenated Machine Preservation in Human Liver Transplants. ASTS 2019, 2020-A-292, 2. [Google Scholar]
- Darius, T.; Vergauwen, M.; Smith, T.; Gerin, I.; Joris, V.; Mueller, M.; Aydin, S.; Muller, X.; Schlegel, A.; Nath, J.; et al. Brief O2 uploading during continuous hypothermic machine perfusion is simple yet effective oxygenation method to improve initial kidney function in a porcine autotransplant model. Am. J. Transplant. 2020. [Google Scholar] [CrossRef]
- Saelens, X.; Festjens, N.; Vande Walle, L.; van Gurp, M.; van Loo, G.; Vandenabeele, P. Toxic proteins released from mitochondria in cell death. Oncogene 2004, 23, 2861–2874. [Google Scholar] [CrossRef]
- Mills, E.L.; Kelly, B.; O’Neill, L.A.J. Mitochondria are the powerhouses of immunity. Nat. Immunol. 2017, 18, 488–498. [Google Scholar] [CrossRef]
- Dubois-Pot-Schneider, H.; Fekir, K.; Coulouarn, C.; Glaise, D.; Aninat, C.; Jarnouen, K.; Le Guével, R.; Kubo, T.; Ishida, S.; Morel, F.; et al. Inflammatory cytokines promote the retrodifferentiation of tumor-derived hepatocyte-like cells to progenitor cells. Hepatology 2014, 60, 2077–2090. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute. National Cancer Institute Milestones in Cancer Research and Discovery, February 2015; National Cancer Institute: Bethesda, MD, USA, 2015.
- Capece, D.; Fischietti, M.; Verzella, D.; Gaggiano, A.; Cicciarelli, G.; Tessitore, A.; Zazzeroni, F.; Alesse, E. The inflammatory microenvironment in hepatocellular carcinoma: A pivotal role for tumor-associated macrophages. BioMed Res. Int. 2013, 2013, 15. [Google Scholar] [CrossRef] [PubMed]
- Budhu, A.; Wang, X.W. The role of cytokines in hepatocellular carcinoma. J. Leukoc. Biol. 2006, 80, 1197–1213. [Google Scholar] [CrossRef]
- De Carlis, L.; De Carlis, R.; Lauterio, A.; Di Sandro, S.; Ferla, F.; Zanierato, M. Sequential Use of Normothermic Regional Perfusion and Hypothermic Machine Perfusion in Donation After Cardiac Death Liver Transplantation with Extended Warm Ischemia Time. Transplantation 2016, 100, e101–e102. [Google Scholar] [CrossRef]
- Patrono, D.; Surra, A.; Catalano, G.; Rizza, G.; Berchialla, P.; Martini, S.; Tandoi, F.; Lupo, F.; Mirabella, S.; Stratta, C.; et al. Hypothermic Oxygenated Machine Perfusion of Liver Grafts from Brain-Dead Donors. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Schlegel, A.; Dutkowski, P. Impact of Machine Perfusion on Biliary Complications after Liver Transplantation. Int. J. Mol. Sci. 2018, 19, 3567. [Google Scholar] [CrossRef]
- He, G.; Karin, M. NF-κB and STAT3- key players in liver inflammation and cancer. Cell Res. 2011, 21, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Karki, R.; Man, S.M.; Kanneganti, T.-D. Inflammasomes and Cancer. Cancer Immunol. Res. 2017, 5, 94–99. [Google Scholar] [CrossRef]
- Kron, P.; Schlegel, A.; de Rougemont, O.; Oberkofler, C.E.; Clavien, P.-A.; Dutkowski, P. Short, Cool, and Well Oxygenated—HOPE for Kidney Transplantation in a Rodent Model. Ann. Surg. 2016, 264, 815–822. [Google Scholar] [CrossRef]
- Kron, P.; Schlegel, A.; Muller, X.; Gaspert, A.; Clavien, P.A.; Dutkowski, P. Hypothermic Oxygenated Perfusion: A Simple and Effective Method to Modulate the Immune Response in Kidney Transplantation. Transplantation 2019, 103, e128–e136. [Google Scholar] [CrossRef]
- Mueller, M.; Kalisvaart, M.; O’Rourke, J.; Shetty, S.; Parente, A.; Muller, X.; Isaac, J.; Muellhaupt, B.; Muiesan, P.; Shah, T.; et al. Hypothermic oxygenated liver perfusion (HOPE) prevents tumor recurrence in liver transplantation from donation after circulatory death. Ann. Surg. 2020, 11. [Google Scholar]
- Watson, C.J.E.; Jochmans, I. From “Gut Feeling” to Objectivity: Machine Preservation of the Liver as a Tool to Assess Organ Viability. Curr. Transplant. Rep. 2018, 5, 72–81. [Google Scholar] [CrossRef]
- Matton, A.P.M.; de Vries, Y.; Burlage, L.C.; van Rijn, R.; Fujiyoshi, M.; de Meijer, V.E.; de Boer, M.T.; de Kleine, R.H.J.; Verkade, H.J.; Gouw, A.S.H.; et al. Biliary Bicarbonate, pH and Glucose Are Suitable Biomarkers of Biliary Viability during Ex Situ Normothermic Machine Perfusion of Human Donor Livers. Transplantation 2018, 103, 1405. [Google Scholar] [CrossRef] [PubMed]
- Mergental, H.; Stephenson, B.T.F.; Laing, R.W.; Kirkham, A.J.; Neil, D.A.H.; Wallace, L.L.; Boteon, Y.L.; Widmer, J.; Bhogal, R.H.; Perera, M.T.P.R.; et al. Development of Clinical Criteria for Functional Assessment to Predict Primary Nonfunction of High-Risk Livers Using Normothermic Machine Perfusion. Liver Transplant. 2018, 24, 1453–1469. [Google Scholar] [CrossRef] [PubMed]
- Eshmuminov, D.; Becker, D.; Bautista Borrego, L.; Hefti, M.; Schuler, M.J.; Hagedorn, C.; Muller, X.; Mueller, M.; Onder, C.; Graf, R.; et al. An integrated perfusion machine preserves injured human livers for 1 week. Nat. Biotechnol. 2020, 38, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Nasralla, D.; Coussios, C.C.; Mergental, H.; Akhtar, M.Z.; Butler, A.J.; Ceresa, C.D.L.; Chiocchia, V.; Dutton, S.J.; García-Valdecasas, J.C.; Heaton, N.; et al. A randomized trial of normothermic preservation in liver transplantation. Nature 2018, 557, 50. [Google Scholar] [CrossRef]
- Mergental, H.; Laing, R.W.; Kirkham, A.J.; Perera, M.T.P.R.; Boteon, Y.L.; Attard, J.; Barton, D.; Curbishley, S.; Wilkhu, M.; Neil, D.A.H.; et al. Transplantation of Discarded Livers Following Viability Testing with Normothermic Machine Perfusion. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef]
- Ceresa, C.D.L.; Nasralla, D.; Watson, C.J.E.; Butler, A.J.; Coussios, C.C.; Crick, K.; Hodson, L.; Imber, C.; Jassem, W.; Knight, S.R.; et al. Transient Cold Storage Prior to Normothermic Liver Perfusion May Facilitate Adoption of a Novel Technology. Liver Transplant. 2019, 25, 1503–1513. [Google Scholar] [CrossRef]
- Butler, A.J.; Kosmoliaptsis, V.; Richards, J.; Crick, K.; Fear, C.; Fedotovs, A.; Swift, L.; Robinson, S.; Gifford, D.; Watson, C.J.E.; et al. Liver transplantation outcomes for Maastricht category-III donors after circulatory death: Static cold storage vs. in situ normothermic regional perfusion vs. ex situ normothermic machine perfusion. J. Hepatol. 2020, 70, 658–665. [Google Scholar]
- He, X.; Guo, Z.; Zhao, Q.; Ju, W.; Wang, D.; Wu, L.; Yang, L.; Ji, F.; Tang, Y.; Zhang, Z.; et al. The first case of ischemia-free organ transplantation in humans: A proof of concept. Am. J. Transplant. 2018, 18, 737–744. [Google Scholar] [CrossRef]
- Imber, C.J.; St Peter, S.D.; Lopez de Cenarruzabeitia, I.; Pigott, D.; James, T.; Taylor, R.; McGuire, J.; Hughes, D.; Butler, A.; Rees, M.; et al. Advantages of normothermic perfusion over cold storage in liver preservation. Transplantation 2002, 73, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.B.; Gao, W.; Shi, Y.; Liu, L.; Ma, N.; Chen, J.; Zhu, Z.J. Protective role of normothermic machine perfusion during reduced-size liver transplantation in pigs. Liver Transplant. 2016, 22, 968–978. [Google Scholar] [CrossRef] [PubMed]
- Boteon, Y.L.; Attard, J.; Boteon, A.P.C.S.; Wallace, L.; Reynolds, G.; Hubscher, S.; Mirza, D.F.; Mergental, H.; Bhogal, R.H.; Afford, S.C. Manipulation of Lipid Metabolism During Normothermic Machine Perfusion: Effect of Defatting Therapies on Donor Liver Functional Recovery. Liver Transplant. 2019, 25, 1007–1022. [Google Scholar] [CrossRef] [PubMed]
- Thijssen, M.F.; Brüggenwirth, I.M.A.; Gillooly, A.; Khvorova, A.; Kowalik, T.F.; Martins, P.N. Gene silencing with si-RNA alleviates ischemia-reperfusion injury in the liver: Potential utilization during normothermic machine preservation. Transplantation 2018. [Google Scholar] [CrossRef]
- Goldaracena, N.; Echeverri, J.; Spetzler, V.N.; Kaths, J.M.; Barbas, A.S.; Louis, K.S.; Adeyi, O.A.; Grant, D.R.; Selzner, N.; Selzner, M. Anti-inflammatory signaling during ex vivo liver perfusion improves the preservation of pig liver grafts before transplantation. Liver Transplant. 2016, 22, 1573–1583. [Google Scholar] [CrossRef]
- Li, C.X.; Shao, Y.; Ng, K.T.P.; Liu, X.B.; Ling, C.C.; Ma, Y.Y.; Geng, W.; Fan, S.T.; Lo, C.M.; Man, K. FTY720 suppresses liver tumor metastasis by reducing the population of circulating endothelial progenitor cells. PLoS ONE 2012, 7, e32380. [Google Scholar] [CrossRef]
- Dutkowski, P.; Polak, W.G.; Muiesan, P.; Schlegel, A.; Verhoeven, C.J.; Scalera, I.; DeOliveira, M.L.; Kron, P.; Clavien, P.-A. First Comparison of Hypothermic Oxygenated PErfusion Versus Static Cold Storage of Human Donation After Cardiac Death Liver Transplants: An International-matched Case Analysis. Ann. Surg. 2015, 262, 764–771. [Google Scholar] [CrossRef]
- Boteon, Y.; Laing, R.; Schlegel, A.; Wallace, L.; Smith, A.; Attard, J.; RH, B.; Neil, D.A.; Hübscher, S.; Perera, M.T.P.R.; et al. Combined Hypothermic and Normothermic Machine Perfusion Improves Functional Recovery of Extended Criteria Donor Livers. Liver Transplant. 2018, 24, 1699–1715. [Google Scholar] [CrossRef]
- Van Leeuwen, O.B.; De Vries, Y.; Fujiyoshi, M.; Nijsten, M.W.N.; Ubbink, R.; Pelgrim, G.J.; Werner, M.J.M.; Reyntjens, K.M.E.M.; Van Den Berg, A.P.; De Boer, M.T.; et al. Transplantation of high-risk donor livers after ex situ resuscitation and assessment using combined hypo- A nd normothermic machine perfusion: A prospective clinical trial. Ann. Surg. 2019, 270, 906–914. [Google Scholar] [CrossRef]
- Oldani, G.; Peloso, A.; Slits, F.; Gex, Q.; Delaune, V.; Orci, L.A.; van de Looij, Y.; Colin, D.J.; Germain, S.; de Vito, C.; et al. The impact of short-term machine perfusion on the risk of cancer recurrence after rat liver transplantation with donors after circulatory death. PLoS ONE 2019, 14, e0224890. [Google Scholar] [CrossRef]
| First Author/Year Publication/Reference | Database/Centre | Donor Type | Number of Subjects | Preservation Method | Donor and Recipient Risk Factors Associated with Outcomes | Parameters Associated with IRI | Main Findings/Conclusion |
|---|---|---|---|---|---|---|---|
| Clinical studies with Machine Perfusion | |||||||
| Mueller M, et al., 2020, [47] | Two-centre comparison (UK, Switzerland) | DBD vs. DCD with HOPE | 70; 70 | Hypothermic oxygenated perfusion (HOPE) | Transplantation with non-perfused grafts (DBD), advanced HCC risk (outside Milan, UCSF, and Metroticket II) | ALT release, INR, CRP | 4-fold higher tumour recurrence rate in un-perfused DBD livers compared to HOPE-treated DCD cohort (25.7% vs. 5.7%, p = 0.002). 5-year tumour-free survival of 92% HOPE-treated DCD livers, despite high risk DCD donor livers and advanced HCC risk. DCD grafts can be used for recipients with higher risk HCC tumours (outside Milan, outside UCSF, and Metro-ticket II) if treated with hypothermic oxygenated perfusion (HOPE) prior to transplantation |
| Clinical studies with Standard Cold Storage | |||||||
| Silverstein J et al., 2020, [48] | UNOS | DBD vs. DCD | 6996; 567 | SCS | DCD organs (low cumulative risk, short cold ischemia time of 6 and 5.4 h; young median donor age with 45 and 33 years), donor age, DRI, MELD | – | Liver transplantation with DCD liver grafts was an independent predictor of mortality. Differences in survival were observed in subgroups with higher risk of recurrence, including RETREAT score >4, AFP > 100 ng/mL, and viable tumours on last imaging. Donor or graft quality and HCC parameters impact on outcomes |
| Martinez-Insfran A.L. et al., 2019, [49] | Single centre (Europe) | DBD vs. DCD | 18; 18 | SCS | Cold ischemia time (overall low risk DCD grafts, short donor warm and cold ischemia times) | AST release, ALT release, prothrombin time | DCD liver recipients have inferior patient survival, not significant with p = 0.266 and n = 18 in both groups; low risk DCD grafts can be used for standard HCC recipient |
| Grąt M, et al., 2018, [9] | Single centre (Europe) | DBD | 195 | SCS | Cold ischemia times, recipient WIT (implantation time) | AST release, LDH release, GGT, Peak Bilirubin, INR | AST ≥1896 U/L increases the risk of HCC recurrence after LT, already in patients within Milan criteria (p = 0.035) |
| Khorsandi SE et al., 2016, [50] | Single centre (UK) | DBD vs. DCD | 256; 91 | SCS | DCD vs. DBD grafts, donor warm and cold ischemia times, HCC risk factors | AST release, INR | DCD livers release more transaminases (AST) and have an impaired function (INR), recipients of good quality DCD livers have a similar risk of HCC recurrence compared to standard DBD donor liver recipients |
| Orci LA, et al., 2015, [51] | UNOS | DBD vs. DCD | 9724 | SCS | Donor WIT in DCD organs, donor age, donor BMI | – | Donor age >60 years and donor WIT was a risk factor for an increased HCC recurrence (p = 0.025) |
| Nagai et al., 2015, [5] | Two-centre analysis (USA) | DBD | 391 | SCS | Cold ischemia time and recipient WIT (implantation time), outside Milan, micro/macrovascular invasion, AFP >200 ng/mL; poor differentiation | AST release, ALT release | CIT >10 h and recipient WIT >50 min were independent risk factors for HCC recurrence after LT (p = 0.03; HR = 1.9; p = 0.003; HR = 2.84, respectively) |
| Kornberg A, et al., 2015, [52] | Single centre (Europe) | DBD | 106 | SCS | Cold ischemia time and recipient WIT, HCC risk factors | AST release, ALT release | Prolonged mean CIT (468.0 vs. 375.5 min; p = 0.001) and recipient WIT (58.4 vs. 45.7 min; p = 0.001) were associated with HCC recurrence after LT, protective effect of prostaglandin on recurrence free survival and HCC recurrence was more pronounced in recipients outside Milan criteria (p < 0.001) |
| Croome et al., 2015, [53] | Single centre (USA) | DBD vs. DCD | 340; 57 | SCS | Cold ischemia time and donor WIT, recipient AFP, recipient disease severity | – | Good DCD livers have a similar risk of HCC recurrence compared to standard DBD donor livers |
| Croome K et al., 2013, [54] | UNOS | DBD vs. DCD | 5638; 242 | SCS | Warm ischemia time, cold ischemia time, recipient age, lab MELD | – | DCD liver recipients have a higher risk of HCC recurrence compared to DBD graft recipients, recipients of livers with a warm ischemia time of >15 min, or a cold ischemia time of > 6 h 20 min had lower survival rates |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boteon, Y.; Flores Carvalho, M.A.; Panconesi, R.; Muiesan, P.; Schlegel, A. Preventing Tumour Recurrence after Liver Transplantation: The Role of Machine Perfusion. Int. J. Mol. Sci. 2020, 21, 5791. https://doi.org/10.3390/ijms21165791
Boteon Y, Flores Carvalho MA, Panconesi R, Muiesan P, Schlegel A. Preventing Tumour Recurrence after Liver Transplantation: The Role of Machine Perfusion. International Journal of Molecular Sciences. 2020; 21(16):5791. https://doi.org/10.3390/ijms21165791
Chicago/Turabian StyleBoteon, Yuri, Mauricio Alfredo Flores Carvalho, Rebecca Panconesi, Paolo Muiesan, and Andrea Schlegel. 2020. "Preventing Tumour Recurrence after Liver Transplantation: The Role of Machine Perfusion" International Journal of Molecular Sciences 21, no. 16: 5791. https://doi.org/10.3390/ijms21165791
APA StyleBoteon, Y., Flores Carvalho, M. A., Panconesi, R., Muiesan, P., & Schlegel, A. (2020). Preventing Tumour Recurrence after Liver Transplantation: The Role of Machine Perfusion. International Journal of Molecular Sciences, 21(16), 5791. https://doi.org/10.3390/ijms21165791