TETRALEC, Artificial Tetrameric Lectins: A Tool to Screen Ligand and Pathogen Interactions
Abstract
1. Introduction
2. Results
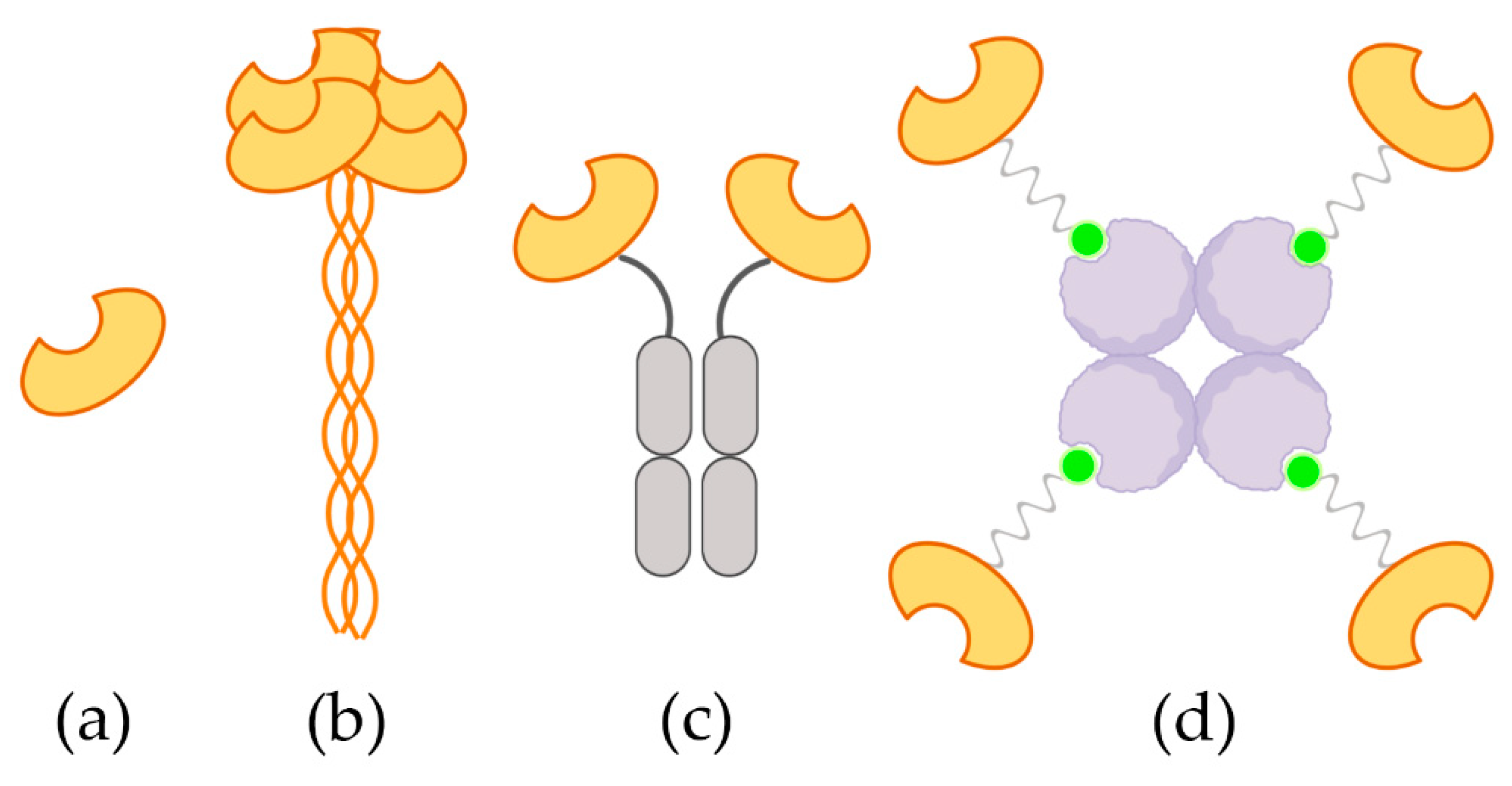
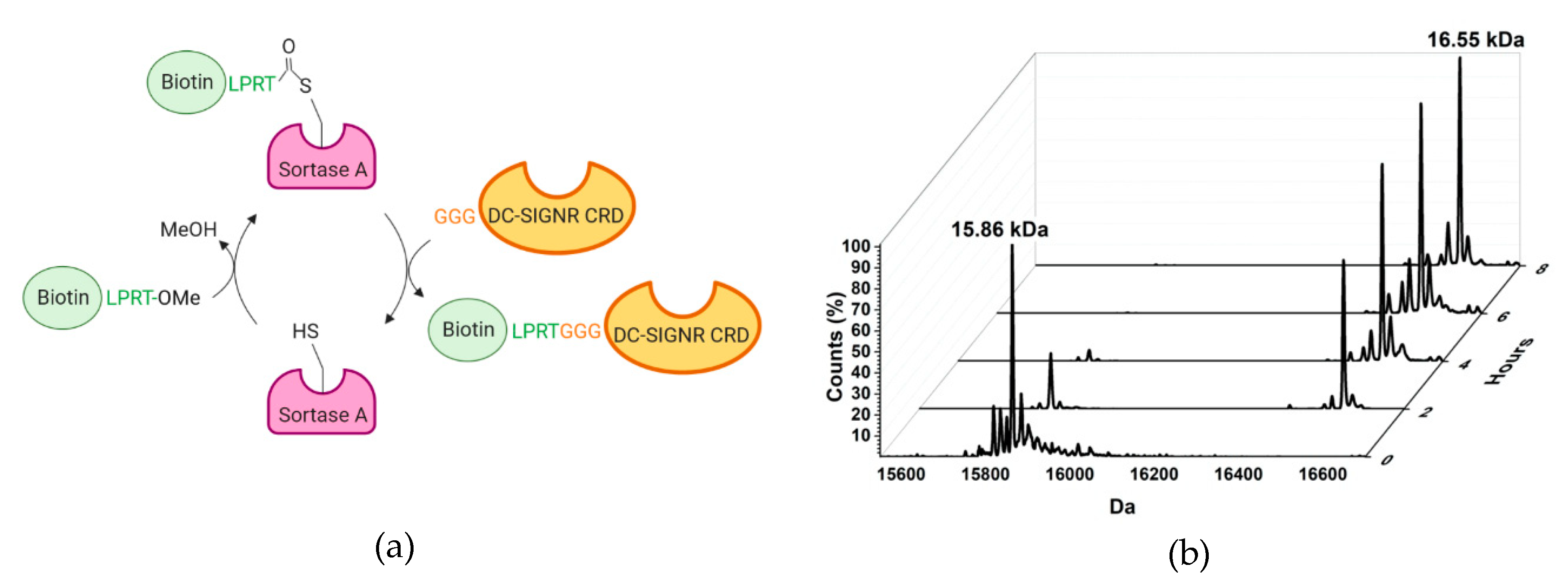
2.1. Design and Synthesis of the TETRALEC
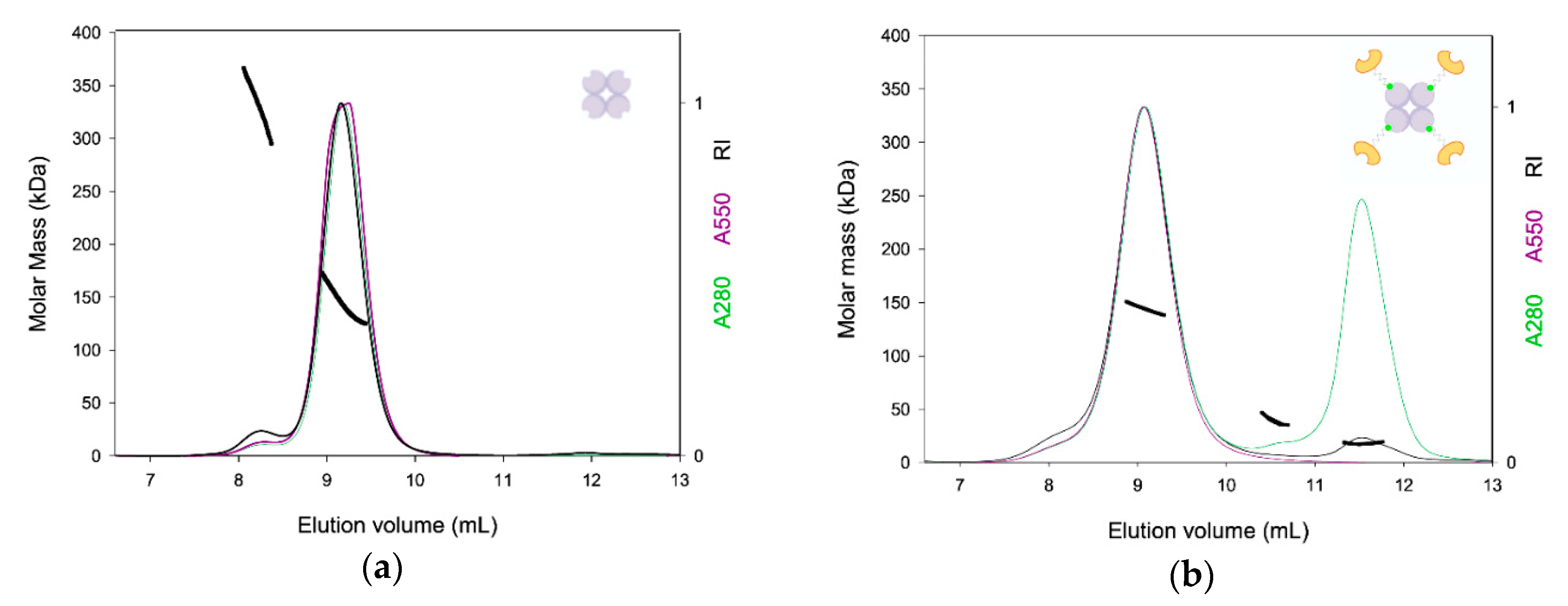
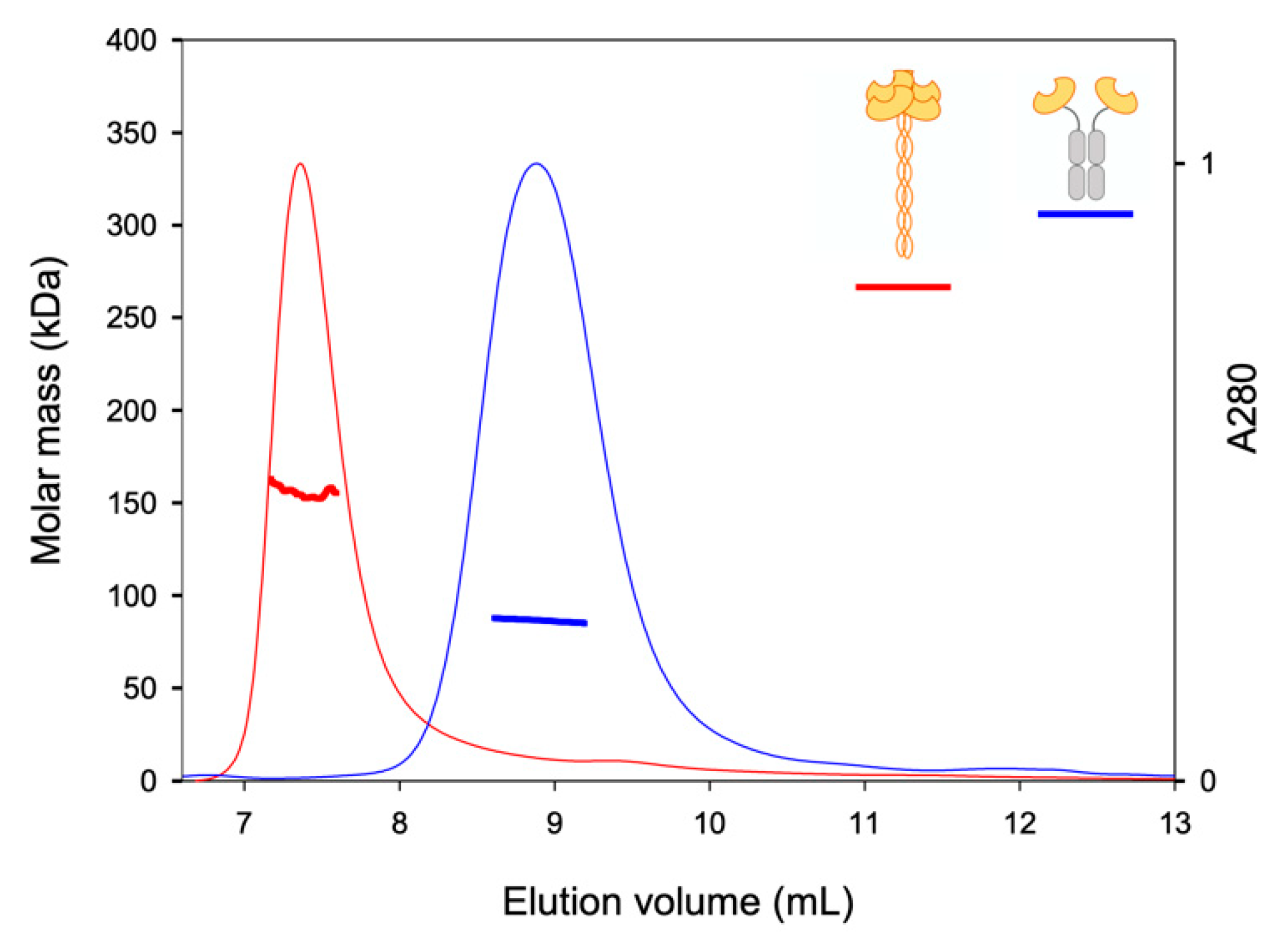
2.2. TETRALEC Structural Characterization
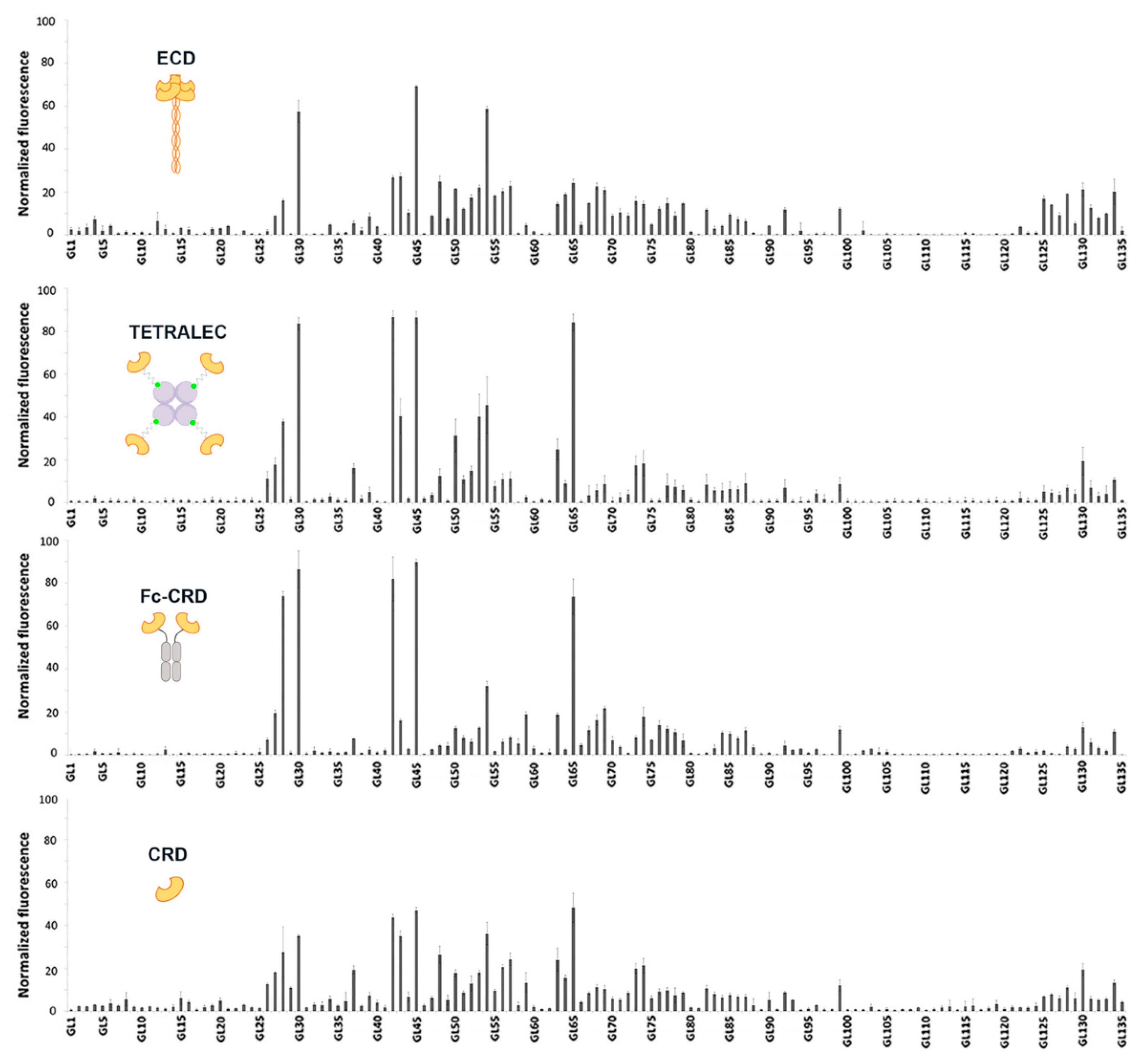
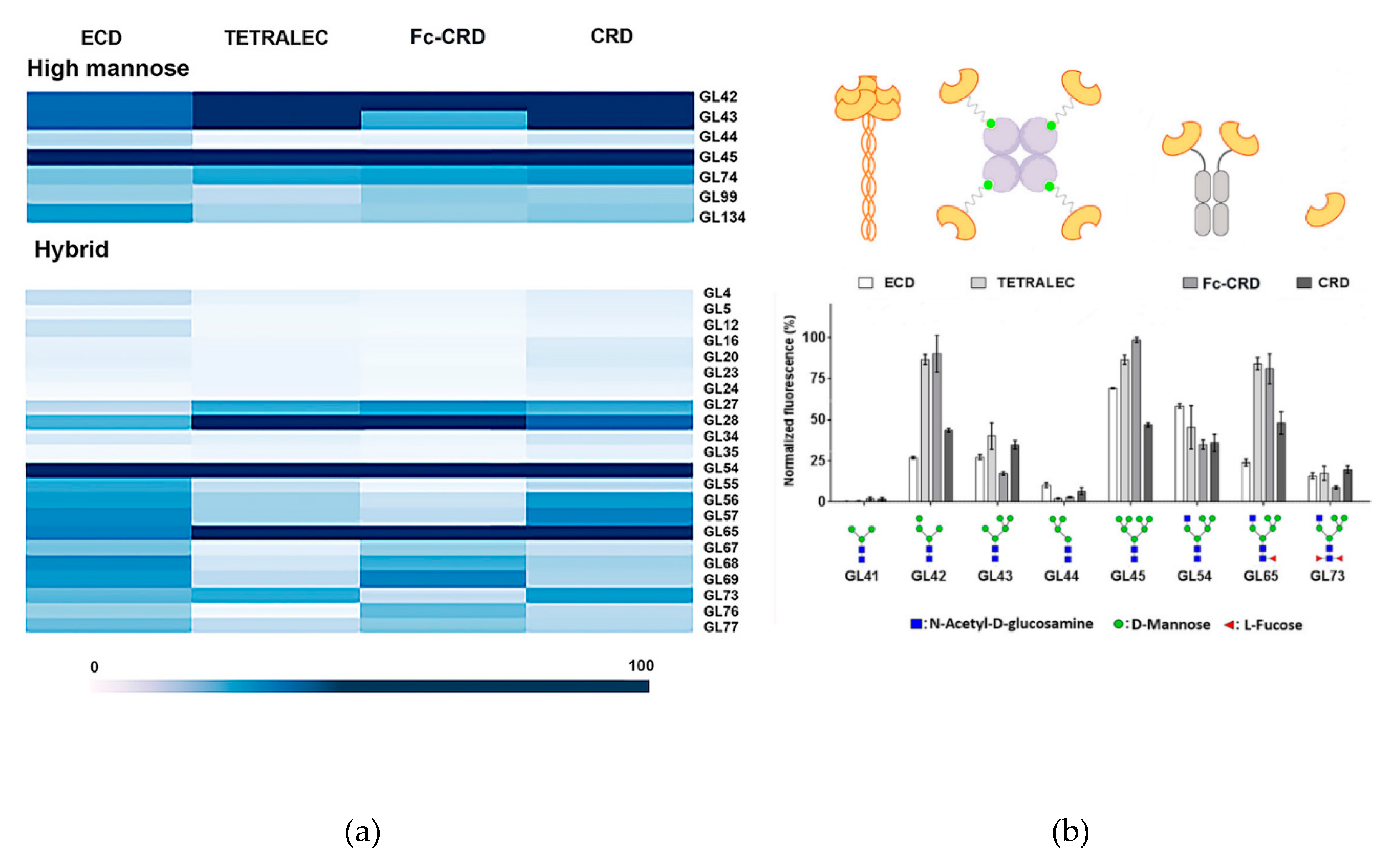
2.3. Validation on Glycan Array
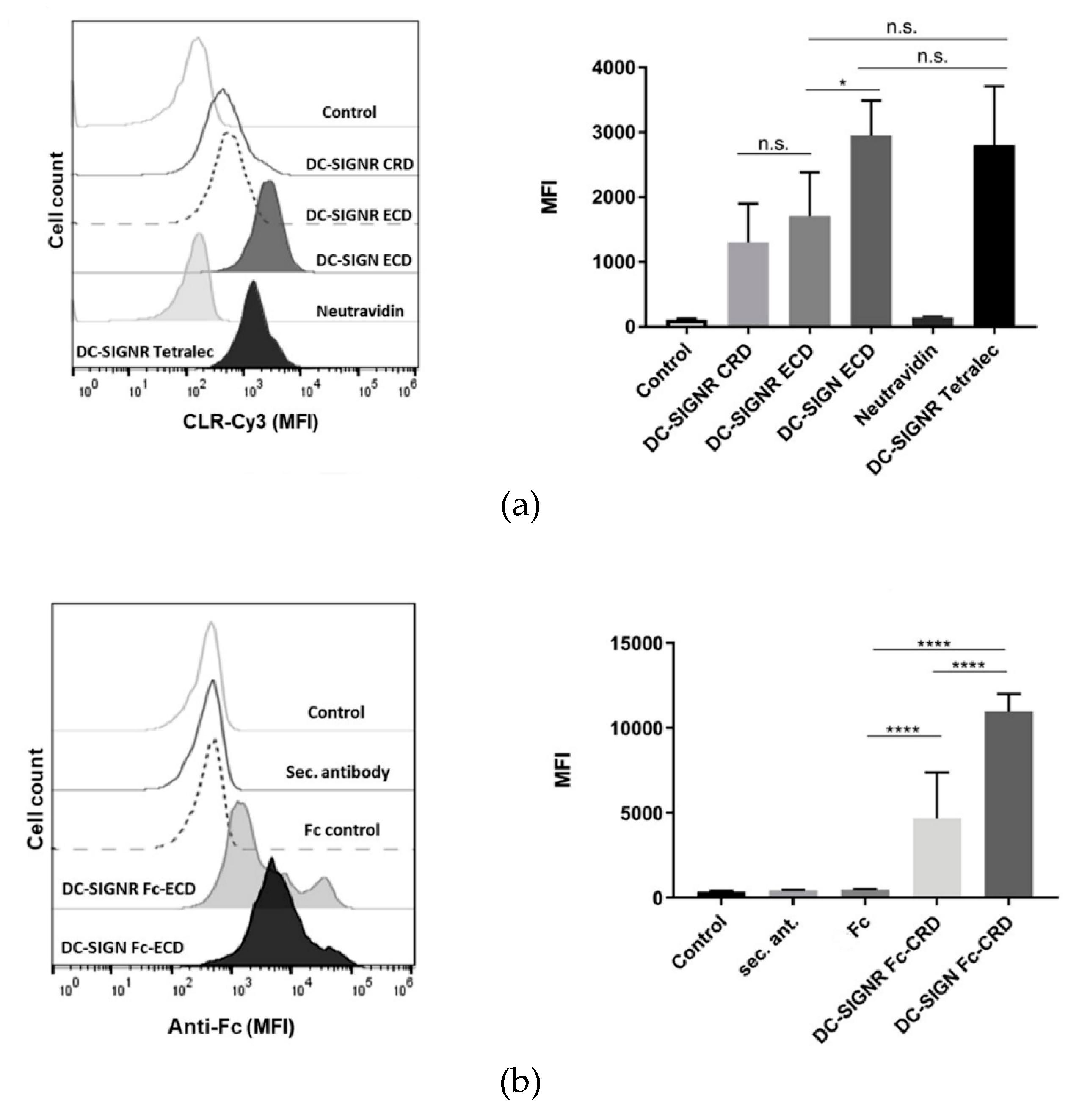
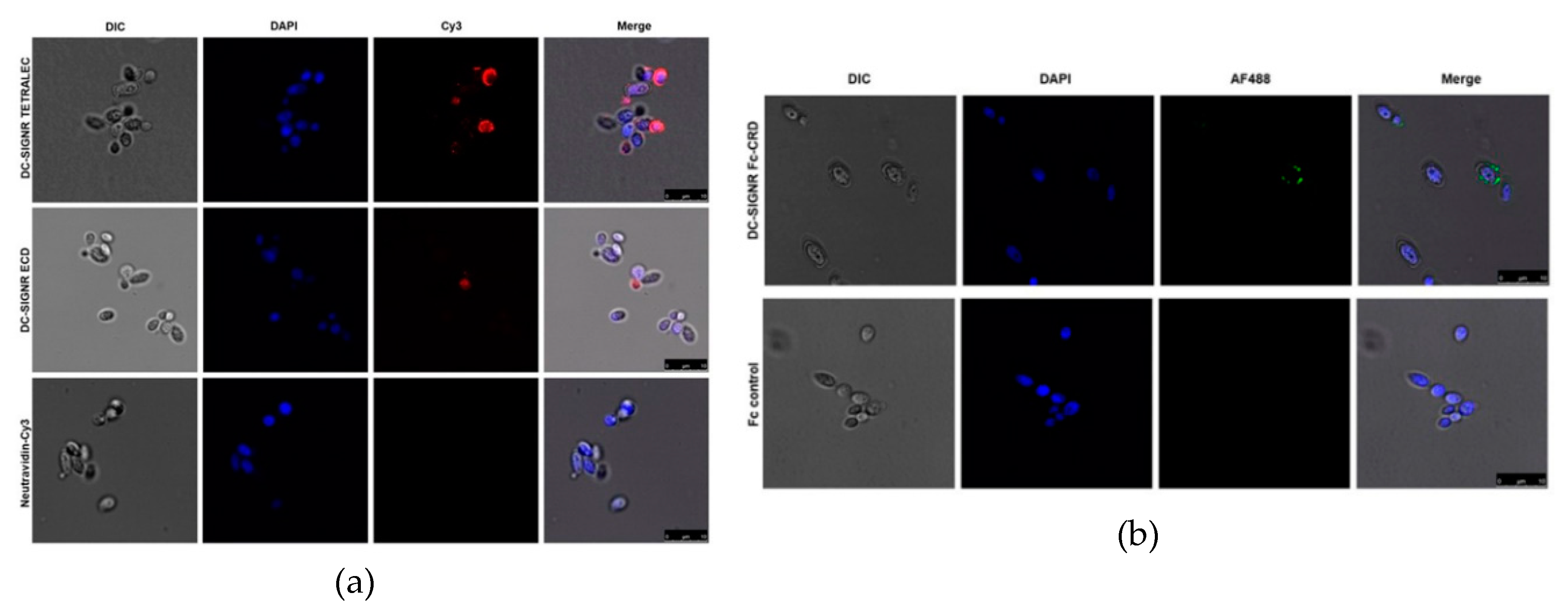
2.4. DC-SIGNR Recognizes Heat-Killed Candida albicans
3. Discussion
4. Materials and Methods
4.1. Cloning
4.2. Protein Expression and Purification
4.3. Labelling
4.4. TETRALEC Formation
4.5. SEC-LS Analysis
4.6. Glycan Array Analysis
4.7. Generation of Human DC-SIGNR-Fc Fusion Protein
4.8. Flow Cytometry-Based Binding to C. albicans
4.9. Confocal Fluorescence Microscopy
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CLR | C-type Lectin Receptor |
| CRD | Carbohydrate Recognition Domain |
| DC-SIGN | Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin |
| DC-SIGNR or L-SIGN | DC-SIGN related or Lymph node-specific intercellular adhesion molecule-3-grabbing integrin |
| DC | Dendritic Cell |
| DOL | Degree Of Labelling |
| ECD | Extracellular Domain |
| ESI-MS | Electrospray ionization mass spectrometry |
| GPI | GlycosylPhosphatidylInositol |
| HKCA | Heat-Killed C. albicans |
| HRP | Horseradish Peroxidase |
| IPTG | IsoproPyl 1-Thio-D-Galactopyranoside |
| LB | Lysogeny broth |
| PRR | Pattern Recognition Receptors |
| SEC | Size Exclusion Chromatography |
| SEC-LS | Size Exclusion Chromatography coupled to Light Scattering |
| SrtA | Sortase A |
References
- Schnaar, R.L. Glycobiology simplified: Diverse roles of glycan recognition in inflammation. J. Leukoc. Biol. 2016, 99, 825–838. [Google Scholar] [CrossRef]
- Levitz, S.M.; Golenbock, D.T. Beyond empiricism: Informing vaccine development through innate immunity research. Cell 2012, 148, 1284–1292. [Google Scholar] [CrossRef]
- Drickamer, K. Two distinct classes of carbohydrate-recognition domains in animal lectins. J. Biol. Chem. 1988, 263, 9557–9560. [Google Scholar] [PubMed]
- van Vliet, S.J.; García-Vallejo, J.J.; van Kooyk, Y. Dendritic cells and C-type lectin receptors: Coupling innate to adaptive immune responses. Immunol. Cell Biol. 2008, 86, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Sager, C.P.; Eriş, D.; Smieško, M.; Hevey, R.; Ernst, B. What contributes to an effective mannose recognition domain? Beilstein J. Org. Chem. 2017, 13, 2584–2595. [Google Scholar] [CrossRef] [PubMed]
- Varki, A.; Gagneux, P. Biological Functions of Glycans. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2015. [Google Scholar]
- East, L.; Isacke, C.M. The mannose receptor family. Biochim. Biophys. Acta 2002, 1572, 364–386. [Google Scholar] [CrossRef]
- Tabarani, G.; Thépaut, M.; Stroebel, D.; Ebel, C.; Vivès, C.; Vachette, P.; Durand, D.; Fieschi, F. DC-SIGN neck domain is a pH-sensor controlling oligomerization: SAXS and hydrodynamic studies of extracellular domain. J. Biol. Chem. 2009, 284, 21229–21240. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.E.; Drickamer, K.; Schnaar, R.L.; Etzler, M.E.; Varki, A. Discovery and Classification of Glycan-Binding Proteins. In Essentials of Glycobiology, 3rd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2017. [Google Scholar]
- de Bakker, B.I.; de Lange, F.; Cambi, A.; Korterik, J.P.; van Dijk, E.M.H.P.; van Hulst, N.F.; Figdor, C.G.; Garcia-Parajo, M.F. Nanoscale Organization of the Pathogen Receptor DC-SIGN Mapped by Single-Molecule High-Resolution Fluorescence Microscopy. ChemPhysChem 2007, 8, 1473–1480. [Google Scholar] [CrossRef]
- Scanlan, C.N.; Offer, J.; Zitzmann, N.; Dwek, R.A. Exploiting the defensive sugars of HIV-1 for drug and vaccine design. Nature 2007, 446, 1038. [Google Scholar] [CrossRef]
- Berzi, A.; Ordanini, S.; Joosten, B.; Trabattoni, D.; Cambi, A.; Bernardi, A.; Clerici, M. Pseudo-Mannosylated DC-SIGN Ligands as Immunomodulants. Sci. Rep. 2016, 6, 35373. [Google Scholar] [CrossRef]
- Kuno, A.; Uchiyama, N.; Koseki-Kuno, S.; Ebe, Y.; Takashima, S.; Yamada, M.; Hirabayashi, J. Evanescent-field fluorescence-assisted lectin microarray: A new strategy for glycan profiling. Nat. Methods 2005, 2, 851–856. [Google Scholar] [CrossRef]
- Pilobello, K.T.; Krishnamoorthy, L.; Slawek, D.; Mahal, L.K. Development of a Lectin Microarray for the Rapid Analysis of Protein Glycopatterns. ChemBioChem 2005, 6, 985–989. [Google Scholar] [CrossRef] [PubMed]
- Porkolab, V.; Pifferi, C.; Sutkeviciute, I.; Ordanini, S.; Taouai, M.; Thépaut, M.; Vivès, C.; Benazza, M.; Bernardi, A.; Renaudet, O.; et al. Development of C-type lectin-oriented surfaces for high avidity glycoconjugates: Towards mimicking multivalent interactions on the cell surface. Org. Biomol. Chem. 2020, 18, 4763–4772. [Google Scholar] [CrossRef] [PubMed]
- Czajkowsky, D.M.; Hu, J.; Shao, Z.; Pleass, R.J. Fc-fusion proteins: New developments and future perspectives. EMBO Mol. Med. 2012, 4, 1015–1028. [Google Scholar] [CrossRef]
- Chapman-Smith, A.; Cronan, J.E., Jr. The enzymatic biotinylation of proteins: A post-translational modification of exceptional specificity. Trends Biochem. Sci. 1999, 24, 359–363. [Google Scholar] [CrossRef]
- Antos, J.M.; Chew, G.-L.; Guimaraes, C.P.; Yoder, N.C.; Grotenbreg, G.M.; Popp, M.W.-L.; Ploegh, H.L. Site-Specific N- and C-Terminal Labeling of a Single Polypeptide Using Sortases of Different Specificity. J. Am. Chem. Soc. 2009, 131, 10800–10801. [Google Scholar] [CrossRef] [PubMed]
- Bashirova, A.A.; Geijtenbeek, T.B.H.; van Duijnhoven, G.C.F.; van Vliet, S.J.; Eilering, J.B.G.; Martin, M.P.; Wu, L.; Martin, T.D.; Viebig, N.; Knolle, P.A.; et al. A Dendritic Cell–Specific Intercellular Adhesion Molecule 3–Grabbing Nonintegrin (Dc-Sign)–Related Protein Is Highly Expressed on Human Liver Sinusoidal Endothelial Cells and Promotes HIV-1 Infection. J. Exp. Med. 2001, 193, 671–678. [Google Scholar] [CrossRef]
- Geijtenbeek, T.B.; Torensma, R.; van Vliet, S.J.; van Duijnhoven, G.C.; Adema, G.J.; van Kooyk, Y.; Figdor, C.G. Identification of DC-SIGN, a Novel Dendritic Cell–Specific ICAM-3 Receptor that Supports Primary Immune Responses. Cell 2000, 100, 575–585. [Google Scholar] [CrossRef]
- Zhang, F.; Ren, S.; Zuo, Y. DC-SIGN, DC-SIGNR and LSECtin: C-Type Lectins for Infection. Int. Rev. Immunol. 2014, 33, 54–66. [Google Scholar] [CrossRef]
- Khoo, U.-S.; Chan, K.Y.K.; Chan, V.S.F.; Lin, C.L.S. DC-SIGN and L-SIGN: The SIGNs for infection. J. Mol. Med. 2008, 86, 861–874. [Google Scholar] [CrossRef]
- Bradshaw, W.J.; Davies, A.H.; Chambers, C.J.; Roberts, A.K.; Shone, C.C.; Acharya, K.R. Molecular features of the sortase enzyme family. FEBS J. 2015, 282, 2097–2114. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Feinberg, H.; Conroy, E.; Mitchell, D.A.; Alvarez, R.; Blixt, O.; Taylor, M.E.; Weis, W.I.; Drickamer, K. Structural basis for distinct ligand-binding and targeting properties of the receptors DC-SIGN and DC-SIGNR. Nat. Struct. Amp Mol. Biol. 2004, 11, 591. [Google Scholar] [CrossRef] [PubMed]
- Brzezicka, K.; Echeverria, B.; Serna, S.; van Diepen, A.; Hokke, C.H.; Reichardt, N.-C. Synthesis and Microarray-Assisted Binding Studies of Core Xylose and Fucose Containing N-Glycans. ACS Chem. Biol. 2015, 10, 1290–1302. [Google Scholar] [CrossRef]
- Echeverria, B.; Serna, S.; Achilli, S.; Vivès, C.; Pham, J.; Thépaut, M.; Hokke, C.H.; Fieschi, F.; Reichardt, N.-C. Chemoenzymatic Synthesis of N-glycan Positional Isomers and Evidence for Branch Selective Binding by Monoclonal Antibodies and Human C-type Lectin Receptors. ACS Chem. Biol. 2018, 13, 2269–2279. [Google Scholar] [CrossRef] [PubMed]
- Quackenbush, J. Microarray data normalization and transformation. Nat. Genet. 2002, 32, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Pham, J.; Hernandez, A.; Cioce, A.; Achilli, S.; Goti, G.; Vivès, C.; Thépaut, M.; Bernardi, A.; Fieschi, F.; Reichardt, N. Chemo-enzymatic Synthesis of S. mansoni O-glycans and their Evaluation as Ligands for C-type Lectin Receptors MGL, DC-SIGN and DC-SIGNR. Chem. Eur. J. Accepted.
- Feinberg, H.; Castelli, R.; Drickamer, K.; Seeberger, P.H.; Weis, W.I. Multiple Modes of Binding Enhance the Affinity of DC-SIGN for High Mannose N-Linked Glycans Found on Viral Glycoproteins. J. Biol. Chem. 2007, 282, 4202–4209. [Google Scholar] [CrossRef]
- Feinberg, H. Structural Basis for Selective Recognition of Oligosaccharides by DC-SIGN and DC-SIGNR. Science 2001, 294, 2163–2166. [Google Scholar] [CrossRef]
- Feinberg, H.; Guo, Y.; Mitchell, D.A.; Drickamer, K.; Weis, W.I. Extended Neck Regions Stabilize Tetramers of the Receptors DC-SIGN and DC-SIGNR. J. Biol. Chem. 2005, 280, 1327–1335. [Google Scholar] [CrossRef]
- Feinberg, H.; Tso, C.K.W.; Taylor, M.E.; Drickamer, K.; Weis, W.I. Segmented Helical Structure of the Neck Region of the Glycan-Binding Receptor DC-SIGNR. J. Mol. Biol. 2009, 394, 613–620. [Google Scholar] [CrossRef]
- André, S.; Kožár, T.; Schuberth, R.; Unverzagt, C.; Kojima, S.; Gabius, H.-J. Substitutions in the N-Glycan Core as Regulators of Biorecognition: The Case of Core-Fucose and Bisecting GlcNAc Moieties. Biochemistry (Mosc.) 2007, 46, 6984–6995. [Google Scholar] [CrossRef]
- Harbison, A.; Fadda, E. An atomistic perspective on antibody-dependent cellular cytotoxicity quenching by core-fucosylation of IgG1 Fc N-glycans from enhanced sampling molecular dynamics. Glycobiology 2019. [Google Scholar] [CrossRef] [PubMed]
- Cambi, A.; Netea, M.G.; Mora-Montes, H.M.; Gow, N.A.R.; Hato, S.V.; Lowman, D.W.; Kullberg, B.-J.; Torensma, R.; Williams, D.L.; Figdor, C.G. Dendritic Cell Interaction with Candida albicans Critically Depends on N-Linked Mannan. J. Biol. Chem. 2008, 283, 20590–20599. [Google Scholar] [CrossRef] [PubMed]
- Takahara, K.; Arita, T.; Tokieda, S.; Shibata, N.; Okawa, Y.; Tateno, H.; Hirabayashi, J.; Inaba, K. Difference in Fine Specificity to Polysaccharides of Candida albicans Mannoprotein between Mouse SIGNR1 and Human DC-SIGN. Infect. Immun. 2012, 80, 1699–1706. [Google Scholar] [CrossRef] [PubMed]
- Cambi, A.; Gijzen, K.; de Vries, I.J.M.; Torensma, R.; Joosten, B.; Adema, G.J.; Netea, M.G.; Kullberg, B.-J.; Romani, L.; Figdor, C.G. The C-type lectin DC-SIGN (CD209) is an antigen-uptake receptor for Candida albicans on dendritic cells. Eur. J. Immunol. 2003, 33, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Hoving, J.C. Pneumocystis and interactions with host immune receptors. PLoS Pathog. 2018, 14, e1006807. [Google Scholar] [CrossRef] [PubMed]
- Rabes, A.; Zimmermann, S.; Reppe, K.; Lang, R.; Seeberger, P.H.; Suttorp, N.; Witzenrath, M.; Lepenies, B.; Opitz, B. The C-Type Lectin Receptor Mincle Binds to Streptococcus pneumoniae but Plays a Limited Role in the Anti-Pneumococcal Innate Immune Response. PLoS ONE 2015, 10, e0117022. [Google Scholar] [CrossRef]
- Lightfoot, Y.L.; Selle, K.; Yang, T.; Goh, Y.J.; Sahay, B.; Zadeh, M.; Owen, J.L.; Colliou, N.; Li, E.; Johannssen, T.; et al. SIGNR3-dependent immune regulation by Lactobacillus acidophilus surface layer protein A in colitis. EMBO J. 2015, 34, 881–895. [Google Scholar] [CrossRef]
- Schatz, P.J. Use of Peptide Libraries to Map the Substrate Specificity of a Peptide-Modifying Enzyme: A 13 Residue Consensus Peptide Specifies Biotinylation in Escherichia coli. Bio/Technology 1993, 11, 1138. [Google Scholar] [CrossRef]
- Powlesland, A.S.; Ward, E.M.; Sadhu, S.K.; Guo, Y.; Taylor, M.E.; Drickamer, K. Widely divergent biochemical properties of the complete set of mouse DC-SIGN-related proteins. J. Biol. Chem. 2006, 281, 20440–20449. [Google Scholar] [CrossRef]
- Stone, J.D.; Artyomov, M.N.; Chervin, A.S.; Chakraborty, A.K.; Eisen, H.N.; Kranz, D.M. Interaction of Streptavidin-Based Peptide-MHC Oligomers (Tetramers) with Cell-Surface T Cell Receptors. J. Immunol. Baltim. Md 1950 2011, 187, 6281–6290. [Google Scholar] [CrossRef]
- Theile, C.S.; Witte, M.D.; Blom, A.E.M.; Kundrat, L.; Ploegh, H.L.; Guimaraes, C.P. Site-specific N-terminal labeling of proteins using sortase-mediated reactions. Nat. Protoc. 2013, 8, 1800–1807. [Google Scholar] [CrossRef]
- Popp, M.W.; Antos, J.M.; Grotenbreg, G.M.; Spooner, E.; Ploegh, H.L. Sortagging: A versatile method for protein labeling. Nat. Chem. Biol. 2007, 3, 707. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Wang, Q.; Swarts, B.M.; Guo, Z. Sortase-Catalyzed Peptide−Glycosylphosphatidylinositol Analogue Ligation. J. Am. Chem. Soc. 2009, 131, 9878–9879. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Sly, K.L.; Conboy, J.C. Comparison of the Energetics of Avidin, Streptavidin, NeutrAvidin, and Anti-Biotin Antibody Binding to Biotinylated Lipid Bilayer Examined by Second-Harmonic Generation. Anal. Chem. 2012, 84, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, L.; Fant, J.; Lonn, H.; Brewer, C.F. Binding and precipitating activities of Lotus tetragonolobus isolectins with L-fucosyl oligosaccharides. Formation of unique homogeneous cross-linked lattices observed by electron microscopy. Biochemistry (Mosc.) 1990, 29, 7523–7530. [Google Scholar] [CrossRef]
- Bhattacharyya, L.; Brewer, C.F. Interactions of concanavalin A with asparagine-linked glycopeptides. Structure/activity relationships of the binding and precipitation of oligomannose and bisected hybrid-type glycopeptides with concanavalin A. Eur. J. Biochem. 1989, 178, 721–726. [Google Scholar] [CrossRef]
- Cutler, J.E. N-Glycosylation of yeast, with emphasis on Candida albicans. Med. Mycol. 2001, 39, 75–86. [Google Scholar] [CrossRef]
- dos Santos, Á.; Hadjivasiliou, A.; Ossa, F.; Lim, N.K.; Turgut, A.; Taylor, M.E.; Drickamer, K. Oligomerization domains in the glycan-binding receptors DC-SIGN and DC-SIGNR: Sequence variation and stability differences. Protein Sci. Publ. Protein Soc. 2017, 26, 306–316. [Google Scholar] [CrossRef]
- Maglinao, M.; Eriksson, M.; Schlegel, M.K.; Zimmermann, S.; Johannssen, T.; Götze, S.; Seeberger, P.H.; Lepenies, B. A platform to screen for C-type lectin receptor-binding carbohydrates and their potential for cell-specific targeting and immune modulation. J. Control. Release 2014, 175, 36–42. [Google Scholar] [CrossRef]
- Mayer, S.; Moeller, R.; Monteiro, J.T.; Ellrott, K.; Josenhans, C.; Lepenies, B. C-Type Lectin Receptor (CLR)–Fc Fusion Proteins As Tools to Screen for Novel CLR/Bacteria Interactions: An Exemplary Study on Preselected Campylobacter jejuni Isolates. Front. Immunol. 2018, 9, 213. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Achilli, S.; Monteiro, J.T.; Serna, S.; Mayer-Lambertz, S.; Thépaut, M.; Le Roy, A.; Ebel, C.; Reichardt, N.-C.; Lepenies, B.; Fieschi, F.; et al. TETRALEC, Artificial Tetrameric Lectins: A Tool to Screen Ligand and Pathogen Interactions. Int. J. Mol. Sci. 2020, 21, 5290. https://doi.org/10.3390/ijms21155290
Achilli S, Monteiro JT, Serna S, Mayer-Lambertz S, Thépaut M, Le Roy A, Ebel C, Reichardt N-C, Lepenies B, Fieschi F, et al. TETRALEC, Artificial Tetrameric Lectins: A Tool to Screen Ligand and Pathogen Interactions. International Journal of Molecular Sciences. 2020; 21(15):5290. https://doi.org/10.3390/ijms21155290
Chicago/Turabian StyleAchilli, Silvia, João T. Monteiro, Sonia Serna, Sabine Mayer-Lambertz, Michel Thépaut, Aline Le Roy, Christine Ebel, Niels-Christian Reichardt, Bernd Lepenies, Franck Fieschi, and et al. 2020. "TETRALEC, Artificial Tetrameric Lectins: A Tool to Screen Ligand and Pathogen Interactions" International Journal of Molecular Sciences 21, no. 15: 5290. https://doi.org/10.3390/ijms21155290
APA StyleAchilli, S., Monteiro, J. T., Serna, S., Mayer-Lambertz, S., Thépaut, M., Le Roy, A., Ebel, C., Reichardt, N.-C., Lepenies, B., Fieschi, F., & Vivès, C. (2020). TETRALEC, Artificial Tetrameric Lectins: A Tool to Screen Ligand and Pathogen Interactions. International Journal of Molecular Sciences, 21(15), 5290. https://doi.org/10.3390/ijms21155290




