Salivary Biomarkers and Their Application in the Diagnosis and Monitoring of the Most Common Oral Pathologies
Abstract
1. Introduction
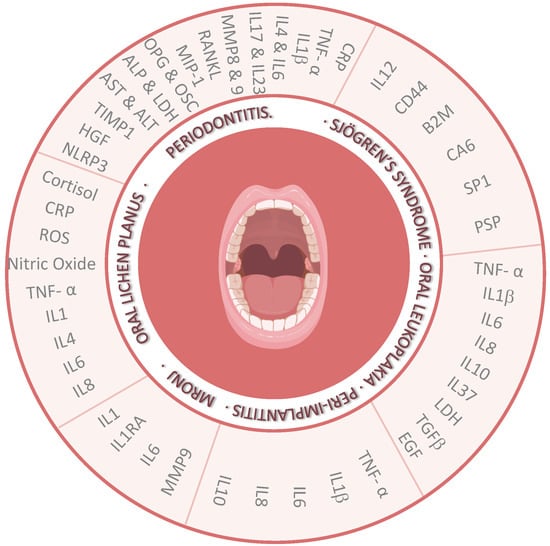
2. Biomarkers in Saliva in Different Oral Diseases
2.1. Oral Lichen Planus
2.2. Periodontitis
2.3. Primary Sjögren’s Syndrome
2.4. Oral Leukoplakia
2.5. Peri-Implantitis
2.6. Medication-Related Osteonecrosis of the Jaw
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| OSCC | oral squamous cell carcinoma |
| ILs | interleukins |
| CRP | C-reactive protein |
| OLP | oral lichen planus |
| PD | periodontitis |
| pSS | primary Sjögren’s syndrome |
| MRONJ | medication-related osteonecrosis of the jaw |
| NO | nitric oxide |
| ROS | reactive oxygen species |
| TNF-α | tumor necrosis factor α |
| Th | T-helper |
| IL-2R | IL-2 receptor |
| TRIM21 | tripartite motif-containing 21 |
| RANKL | receptor activator of nuclear factor κB ligand |
| PGE2 | prostaglandin E2 |
| OPG | osteoprotegerin |
| OSC | osteocalcin |
| ALP | alkaline phosphatase |
| LDH | lactate dehydrogenase |
| AST | aspartate aminotransferase |
| ALT | alanine aminotransferase |
| MMPs | matrix metalloproteases |
| TIMP-1 | tissue inhibitor metalloproteinase-1 |
| HGF | hepatocyte growth factor |
| NLRP3 | nod-like receptor family pyrin domain containing protein 3 |
| SP-1 | salivary protein-1 |
| PSP | parotid secretory protein |
| CA-6 | carbonic anhydrase VI |
| TGFβ | transforming growth factor β |
| EGF | epidermal growth factor |
| BPs | bisphosphonates |
| IL-1RA | interleukin-1 receptor antagonist |
References
- Wu, J.Y.; Yi, C.; Chung, H.R.; Wang, D.J.; Chang, W.C.; Lee, S.Y.; Lin, C.T.; Yang, Y.C.; Yang, W.C.V. Potential biomarkers in saliva for oral squamous cell carcinoma. Oral Oncol. 2010, 46, 226–231. [Google Scholar] [CrossRef]
- Gaba, F.I.; Sheth, C.C.; Veses, V. Salivary biomarkers and their efficacies as diagnostic tools for Oral Squamous Cell Carcinoma: Systematic review and meta-analysis. J. Oral Pathol. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA A Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Sankhla, B.; Sundaragiri, K.S.; Bhargava, A. A Review of Salivary Biomarker: A Tool for Early Oral Cancer Diagnosis. Adv. Biomed. Res. 2017, 6, 90. [Google Scholar] [CrossRef]
- Prasad, G.; McCullough, M. Chemokines and Cytokines as Salivary Biomarkers for the Early Diagnosis of Oral Cancer. Int. J. Dent. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-Z.; Cheng, X.Q.; Li, J.-Y.; Zhang, P.; Yi, P.; Xu, X.; Zhou, X.-D. Saliva in the diagnosis of diseases. Int. J. Oral Sci. 2016, 8, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Martínez, P.M. La saliva como fluido diagnóstico. Ed. Cont. Lab. Clín. 2013, 16, 93–108. [Google Scholar]
- Berga-Hidalgo, M.C. Marcadores Salivales en Lesiones Potencialmente Malignas de la Cavidad oral y en Carcinoma oral de Células Escamosas; Ed Cont Lab Clín Universidad de Zaragoza: Zaragoza, Spain, 2014. [Google Scholar]
- Lee, Y.-H.; Wong, D.T. Saliva: An emerging biofluid for early detection of diseases. Am. J. Dent. 2009, 22, 241–248. [Google Scholar]
- Pan, X.; Wang, Z.; Wu, X.; Wen, S.W.; Liu, A. Salivary cortisol in post-traumatic stress disorder: A systematic review and meta-analysis. BMC Psychiatry 2018, 18, 324. [Google Scholar] [CrossRef]
- Santos, S.; Santos, E.; Gaztambide, S.; Salvador, J. Diagnóstico y diagnóstico diferencial del síndrome de Cushing. Endocrinol. Nutr. 2009, 56, 71–84. [Google Scholar] [CrossRef]
- Gohel, V.; Jones, J.; Wehler, C. Salivary biomarkers and cardiovascular disease: A systematic review. Clin. Chem. Lab. Med. 2018, 56, 1432–1442. [Google Scholar] [CrossRef] [PubMed]
- Parisi, M.R.; Soldini, L.; Vidoni, G.; Mabellini, C.; Belloni, T.; Brignolo, L.; Negri, S.; Schlusnus, K.; Dorigatti, F.; Lazzarin, A. Point-of-care testing for HCV infection: Recent advances and implications for alternative screening. New Microbiol. 2014, 37, 449–457. [Google Scholar] [PubMed]
- Nefzi, F.; Ben Salem, N.A.; Khelif, A.; Feki, S.; Aouni, M.; Gautheret-Dejean, A. Quantitative analysis of human herpesvirus-6 and human cytomegalovirus in blood and saliva from patients with acute leukemia. J. Med. Virol. 2014, 87, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Aitken, J.P.; Ortiz, C.; Morales-Bozo, I.; Rojas-Alcayaga, G.; Baeza, M.; Beltran, C.; Escobar, A. α-2-Macroglobulin in Saliva Is Associated with Glycemic Control in Patients with Type 2 Diabetes Mellitus. Dis. Markers 2015, 2015, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Rathnayake, N.; Åkerman, S.; Klinge, B.; Lundegren, N.; Jansson, H.; Tryselius, Y.; Sorsa, T.; Gustafsson, A. Salivary Biomarkers for Detection of Systemic Diseases. PLoS ONE 2013, 8, e61356. [Google Scholar] [CrossRef]
- López-Jornet, P.; Zavattaro, E.; Mozaffari, H.R.; Ramezani, M.; Sadeghi, M. Evaluation of the Salivary Level of Cortisol in Patients with Oral Lichen Planus: A Meta-Analysis. Medicine 2019, 55, 213. [Google Scholar] [CrossRef]
- Lopez-Jornet, P.; Cayuela, C.A.; Tvarijonaviciute, A.; Escribano, D.; Cerón, J.; Parra-Perez, F. Oral lichen planus: Salival biomarkers cortisol, immunoglobulin A, adiponectin. J. Oral Pathol. Med. 2015, 45, 211–217. [Google Scholar] [CrossRef]
- Humberto, J.S.M.; Pavanin, J.V.; Da Rocha, M.J.A.; Motta, A.C.F. Cytokines, cortisol, and nitric oxide as salivary biomarkers in oral lichen planus: A systematic review. Braz. Oral Res. 2018, 32. [Google Scholar] [CrossRef]
- Shah, B.; Ashok, L.; Sujatha, G. Evaluation of salivary cortisol and psychological factors in patients with oral lichen planus. Indian J. Dent. Res. 2009, 20, 288. [Google Scholar] [CrossRef]
- Karthikeyan, P.; Aswath, N. Stress as an etiologic co-factor in recurrent aphthous ulcers and oral lichen planus. J. Oral Sci. 2016, 58, 237–240. [Google Scholar] [CrossRef]
- Tvarijonaviciute, A.; Aznar-Cayuela, C.; Rubio, C.P.; Ceron, J.J.; López-Jornet, P.; Asta, T.; Cristina, A.C.; Camila, P.R.; Joaquin, C.J. Evaluation of salivary oxidate stress biomarkers, nitric oxide and C-reactive protein in patients with oral lichen planus and burning mouth syndrome. J. Oral Pathol. Med. 2016, 46, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, M.; Iwase, M.; Nagumo, M. Elevated production of salivary nitric oxide in oral mucosal diseases. J. Oral Pathol. Med. 1999, 28, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Darczuk, D.; Krzysciak, W.; Vyhouskaya, P.; Kesek, B.; Galecka-Wanatowicz, D.; Lipska, W.; Kaczmarzyk, T.; Gluch-Lutwin, M.; Mordyl, B.; Chomyszyn-Gajewska, M. Salivary oxidative status in patients with oral lichen planus. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2016, 67, 885–894. [Google Scholar]
- Shahidi, M.; Jafari, S.; Barati, M.; Mahdipour, M.; Gholami, M.S. Predictive value of salivary microRNA-320a, vascular endothelial growth factor receptor 2, CRP and IL-6 in Oral lichen planus progression. Inflammopharmacology 2017, 25, 577–583. [Google Scholar] [CrossRef]
- Shiva, A.; Arab, S.; Mousavi, S.J.; Zamanian, A.; Maboudi, A. Serum and Salivary Level of Nitric Oxide (NOx) and CRP in Oral Lichen Planus (OLP) Patients. J. Dent. Shiraz 2020, 21, 6–11. [Google Scholar]
- Ramseier, C.A.; Kinney, J.S.; Herr, A.E.; Braun, T.; Sugai, J.V.; Shelburne, C.A.; Rayburn, L.A.; Tran, H.M.; Singh, A.K.; Giannobile, W.V. Identification of Pathogen and Host-Response Markers Correlated with Periodontal Disease. J. Periodontol. 2009, 80, 436–446. [Google Scholar] [CrossRef]
- Ehrchen, J.M.; Sunderkötter, C.; Foell, D.; Vogl, T.; Roth, J. The endogenous Toll-like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J. Leukoc. Boil. 2009, 86, 557–566. [Google Scholar] [CrossRef]
- Aurer, A.; Aurer-Kozelj, J.; Stavljenić-Rukavina, A.; Kalenić, S.; Ivić-Kardum, M.; Haban, V. Inflammatory mediators in saliva of patients with rapidly progressive periodontitis during war stress induced incidence increase. Coll. Antropol. 1999, 23, 117–124. [Google Scholar]
- Aurer, A.; Jorgić-Srdjak, K.; Plancak, D.; Stavljenić-Rukavina, A.; Aurer-Kozelj, J. Proinflammatory factors in saliva as possible markers for periodontal disease. Coll. Antropol. 2005, 29, 435–439. [Google Scholar]
- Mozaffari, H.R.; Ramezani, M.; Mahmoudiahmadabadi, M.; Omidpanah, N.; Sadeghi, M. Salivary and serum levels of tumor necrosis factor-alpha in oral lichen planus: A systematic review and meta-analysis study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2017, 124, e183–e189. [Google Scholar] [CrossRef]
- Lu, R.; Zhang, J.; Sun, W.; Du, G.; Zhou, G. Inflammation-related cytokines in oral lichen planus: An overview. J. Oral Pathol. Med. 2013, 44, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Thanthoni, M.; Sathasivasubramanian, S. Quantitative Analysis of Salivary TNF-αin Oral Lichen Planus Patients. Int. J. Dent. 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Frodge, B.D.; Ebersole, J.L.; Kryscio, R.J.; Thomas, M.V.; Miller, C.S. Bone Remodeling Biomarkers of Periodontal Disease in Saliva. J. Periodontol. 2008, 79, 1913–1919. [Google Scholar] [CrossRef]
- Wu, Y.-C.; Ning, L.; Tu, Y.; Huang, C.-P.; Huang, N.-T.; Chen, Y.-F.; Chang, P.-C. Salivary biomarker combination prediction model for the diagnosis of periodontitis in a Taiwanese population. J. Formos. Med. Assoc. 2018, 117, 841–848. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J. Cytokine regulation of immune responses to Porphyromonas gingivalis. Periodontology 2000 2010, 54, 160–194. [Google Scholar] [CrossRef]
- Preshaw, P.; Taylor, J. How has research into cytokine interactions and their role in driving immune responses impacted our understanding of periodontitis? J. Clin. Periodontol. 2011, 38, 60–84. [Google Scholar] [CrossRef]
- Ebersole, J.; Schuster, J.L.; Stevens, J.; Dawson, D.; Kryscio, R.J.; Lin, Y.; Thomas, M.V.; Miller, C.S. Patterns of Salivary Analytes Provide Diagnostic Capacity for Distinguishing Chronic Adult Periodontitis from Health. J. Clin. Immunol. 2012, 33, 271–279. [Google Scholar] [CrossRef]
- Rathnayake, N.; Åkerman, S.; Klinge, B.; Lundegren, N.; Jansson, H.; Tryselius, Y.; Sorsa, T.; Gustafsson, A. Salivary biomarkers of oral health—A cross-sectional study. J. Clin. Periodontol. 2012, 40, 140–147. [Google Scholar] [CrossRef]
- Deepthi, G.; Nandan, S.R.K.; Kulkarni, P.G. Salivary Tumour Necrosis Factor-α as a Biomarker in Oral Leukoplakia and Oral Squamous Cell Carcinoma. Asian Pac. J. Cancer Prev. 2019, 20, 2087–2093. [Google Scholar] [CrossRef]
- Kaur, J.; Jacobs, R. Proinflammatory cytokine levels in oral lichen planus, oral leukoplakia, and oral submucous fibrosis. J. Korean Assoc. Oral Maxillofac. Surg. 2015, 41, 171–175. [Google Scholar] [CrossRef]
- Hsu, H.J.; Yang, Y.H.; Shieh, T.Y.; Chen, C.H.; Kao, Y.-H.; Yang, C.F.; Ko, E. Role of cytokine gene (interferon-γ, transforming growth factor-β1, tumor necrosis factor-α, interleukin-6, and interleukin-10) polymorphisms in the risk of oral precancerous lesions in Taiwanese. Kaohsiung J. Med. Sci. 2014, 30, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Brailo, V.; Vucicevic-Boras, V.; Lukac, J.; Biocina-Lukenda, D.; Alajbeg, I.; Milenovic, A.; Balija, M. Salivary and serum interleukin 1 beta, interleukin 6 and tumor necrosis factor alpha in patients with leukoplakia and oral cancer. Med. Oral Patol. Oral Cir. Bucal 2011, 17, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- Wenghoefer, M.; Pantelis, A.; Najafi, T.; Deschner, J.; Allam, J.; Novak, N.; Reich, R.; Martini, M.; Berge, S.; Fischer, H.; et al. Gene expression of oncogenes, antimicrobial peptides, and cytokines in the development of oral leukoplakia. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2010, 110, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Abduljabbar, T.; Vohra, F.; Ullah, A.; Alhamoudi, N.; Khan, J.; Javed, F. Relationship between self-rated pain and peri-implant clinical, radiographic and whole salivary inflammatory markers among patients with and without peri-implantitis. Clin. Implant. Dent. Relat. Res. 2019, 21, 1218–1224. [Google Scholar] [CrossRef] [PubMed]
- Arias-Bujanda, N.; Regueira-Iglesias, A.; Blanco-Pintos, T.; Alonso-Sampedro, M.; Relvas, M.; González-Peteiro, M.M.; Balsa-Castro, C.; Tomás, I.; Sampedro-Alonso, M. Diagnostic accuracy of IL1β in saliva: The development of predictive models for estimating the probability of the occurrence of periodontitis in non-smokers and smokers. J. Clin. Periodontol. 2020, 47, 702–714. [Google Scholar] [CrossRef]
- Kaushik, R.; Yeltiwar, R.K.; Pushpanshu, K. Salivary Interleukin-1β Levels in Patients with Chronic Periodontitis before and after Periodontal Phase I Therapy and Healthy Controls: A Case-Control Study. J. Periodontol. 2011, 82, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Mirrielees, J.; Crofford, L.J.; Lin, Y.; Kryscio, R.J.; Dawson, L.R.; Ebersole, J.L.; Miller, C.S. Rheumatoid arthritis and salivary biomarkers of periodontal disease. J. Clin. Periodontol. 2010, 37, 1068–1074. [Google Scholar] [CrossRef]
- Gürsoy, U.K.; Könönen, E.; Uitto, V.-J.; Pussinen, P.; Hyvärinen, K.; Knuuttila, M.; Suominen-Taipale, L. Salivary interleukin-1βconcentration and the presence of multiple pathogens in periodontitis. J. Clin. Periodontol. 2009, 36, 922–927. [Google Scholar] [CrossRef]
- Tobón-Arroyave, S.I.; Jaramillo-González, P.; Isaza-Guzman, D.M. Correlation between salivary IL-1β levels and periodontal clinical status. Arch. Oral Boil. 2008, 53, 346–352. [Google Scholar] [CrossRef]
- Assuma, R.; Oates, T.; Cochran, D.; Amar, S.; Graves, D.T. IL-1 and TNF antagonists inhibit the inflammatory response and bone loss in experimental periodontitis. J. Immunol. 1998, 160, 403–409. [Google Scholar]
- Barksby, H.E.; Lea, S.R.; Preshaw, P.M.; Taylor, J. The expanding family of interleukin-1 cytokines and their role in destructive inflammatory disorders. Clin. Exp. Immunol. 2007, 149, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Ng, P.Y.B.; Donley, M.; Hausmann, E.; Hutson, A.D.; Rossomando, E.F.; Scannapieco, F. Candidate salivary biomarkers associated with alveolar bone loss: Cross-sectional and in vitro studies. FEMS Immunol. Med. Microbiol. 2007, 49, 252–260. [Google Scholar] [CrossRef]
- Liukkonen, J.; Gursoy, U.K.; Pussinen, P.J.; Suominen, A.L.; Könönen, E. Salivary Concentrations of Interleukin (IL)-1β, IL-17A, and IL-23 Vary in Relation to Periodontal Status. J. Periodontol. 2016, 87, 1484–1491. [Google Scholar] [CrossRef] [PubMed]
- Isaza-Guzman, D.M.; Medina-Piedrahíta, V.M.; Gutiérrez-Henao, C.; Tobón-Arroyave, S.I. Salivary Levels of NLRP3 Inflammasome-Related Proteins as Potential Biomarkers of Periodontal Clinical Status. J. Periodontol. 2017, 88, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Rocha, F.S.; Jesus, R.N.R.; Rocha, F.M.S.; Moura, C.C.G.; Zanetta-Barbosa, D. Saliva Versus Peri-implant Inflammation: Quantification of IL-1β in Partially and Totally Edentulous Patients. J. Oral Implant. 2014, 40, 169–173. [Google Scholar] [CrossRef]
- Sims, J.; Smith, D.E. The IL-1 family: Regulators of immunity. Nat. Rev. Immunol. 2010, 10, 89–102. [Google Scholar] [CrossRef]
- Gabay, C.; Lamacchia, C.; Palmer, G. IL-1 pathways in inflammation and human diseases. Nat. Rev. Rheumatol. 2010, 6, 232–241. [Google Scholar] [CrossRef]
- Bagan, J.; Sheth, C.C.; Soria, J.M.; Margaix, M.; Bagan, L. Bisphosphonates-related osteonecrosis of the jaws: A preliminary study of salivary interleukins. J. Oral Pathol. Med. 2012, 42, 405–408. [Google Scholar] [CrossRef]
- Bagan, J.; Sáez, G.; Tormos, M.; Hens, E.; Terol, M.; Bagan, L.; Diaz-Fernandez, J.; Lluch, A.; Camps, C. Interleukin-6 concentration changes in plasma and saliva in bisphosphonate-related osteonecrosis of the jaws. Oral Dis. 2013, 20, 446–452. [Google Scholar] [CrossRef]
- Mozaffari, H.R.; Zavattaro, E.; Saeedi, M.; Lopez-Jornet, P.; Sadeghi, M.; Safaei, M.; Imani, M.M.; Nourbakhsh, R.; Moradpoor, H.; Golshah, A.; et al. Serum and salivary interleukin-4 levels in patients with oral lichen planus: A systematic review and meta-analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 128, 123–131. [Google Scholar] [CrossRef]
- Mozaffari, H.R.; Molavi, M.; López-Jornet, P.; Sadeghi, M.; Safaei, M.; Imani, M.; Sharifi, R.; Moradpoor, H.; Golshah, A.; Jamshidy, L. Salivary and Serum Interferon-Gamma/Interleukin-4 Ratio in Oral Lichen Planus Patients: A Systematic Review and Meta-Analysis. Medicine 2019, 55, 257. [Google Scholar] [CrossRef] [PubMed]
- Prakasam, S.; Srinivasan, M. Evaluation of salivary biomarker profiles following non-surgical management of chronic periodontitis. Oral Dis. 2013, 20, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Man Gu, G.; Martin, M.D.; Darveau, R.P.; Truelove, E.; Epstein, J. Oral and serum IL-6 levels in oral lichen planus patients. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 98, 673–678. [Google Scholar] [CrossRef]
- Liu, J.; Shi, Q.; Yang, S.; Wang, Q.; Xu, J.; Guo, B. The relationship between levels of salivary and serum interleukin-6 and oral lichen planus. J. Am. Dent. Assoc. 2017, 148, 743–749.e9. [Google Scholar] [CrossRef]
- Cheng, Y.-S.L.; Jordan, L.; Gorugantula, L.M.; Schneiderman, E.; Chen, H.-S.; Rees, T. Salivary Interleukin-6 and -8 in Patients with Oral Cancer and Patients with Chronic Oral Inflammatory Diseases. J. Periodontol. 2014, 85, 956–965. [Google Scholar] [CrossRef]
- Costa, P.P.; Trevisan, G.L.; Macedo, G.O.; Palioto, D.B.; De Souza, S.L.S.; Grisi, M.F.; Novaes, A.B.; Taba, M.; Taba, M., Jr. Salivary Interleukin-6, Matrix Metalloproteinase-8, and Osteoprotegerin in Patients with Periodontitis and Diabetes. J. Periodontol. 2010, 81, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Teles, R.; Likhari, V.; Socransky, S.S.; Haffajee, A.D. Salivary cytokine levels in subjects with chronic periodontitis and in periodontally healthy individuals: A cross-sectional study. J. Periodontal Res. 2009, 44, 411–417. [Google Scholar] [CrossRef]
- Irwin, C.R.; Myrillas, T.T. The role of IL-6 in the pathogenesis of periodontal disease. Oral Dis. 2008, 4, 43–47. [Google Scholar] [CrossRef]
- Bartold, P.M.; Narayanan, A.S. Molecular and cell biology of healthy and diseased periodontal tissues. Periodontology 2000 2006, 40, 29–49. [Google Scholar] [CrossRef]
- Juretić, M.; Cerović, R.; Belušić-Gobić, M.; Pršo, I.B.; Kqiku, L.; Špalj, S.; Pezelj-Ribarić, S. Salivary levels of TNF-? and IL-6 in patients with oral premalignant and malignant lesions. Folia Boil. 2013, 59, 99–102. [Google Scholar]
- Selvam, N.P.; Sadaksharam, J. Salivary interleukin-6 in the detection of oral cancer and precancer. Asia-Pac. J. Clin. Oncol. 2015, 11, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Punyani, S.R.; Sathawane, R.S. Salivary level of interleukin-8 in oral precancer and oral squamous cell carcinoma. Clin. Oral Investig. 2012, 17, 517–524. [Google Scholar] [CrossRef]
- Chang, K.P.; Kao, H.K.; Wu, C.C.; Fang, K.H.; Chang, Y.L.; Huang, Y.C.; Liu, S.C.; Cheng, M.H. Pretreatment Interleukin-6 Serum Levels Are Associated with Patient Survival for Oral Cavity Squamous Cell Carcinoma. Otolaryngol. Neck Surg. 2013, 148, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Liskmann, S.; Vihalemm, T.; Salum, O.; Zilmer, K.; Fischer, K.; Zilmer, M. Correlations between clinical parameters and interleukin-6 and interleukin-10 levels in saliva from totally edentulous patients with peri-implant disease. Int. J. Oral Maxillofac. Implant. 2006, 21, 543–550. [Google Scholar]
- Mozaffari, H.R.; Sharifi, R.; Mirbahari, S.; Montazerian, S.; Sadeghi, M.; Rostami, S. A systematic review and meta-analysis study of salivary and serum interleukin-8 levels in oral lichen planus. Adv. Dermatol. Allergol. 2018, 35, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Ghoreishian, F.S.; Tavangar, A.; Ghalayani, P.; Boroujeni, M.A. Salivary levels of interleukin-8 in oral lichen planus and diabetic patients: A biochemical study. Dent. Res. J. 2017, 14, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.; Wang, J.; Chia, J.-S.; Chiang, C.-P. Serum interleukin-8 level is a more sensitive marker than serum interleukin-6 level in monitoring the disease activity of oral lichen planus. Br. J. Dermatol. 2005, 152, 1187–1192. [Google Scholar] [CrossRef]
- Fonseca, F.J.P.O.; Junior, M.M.; Lourenço, E.J.V.; Teles, D.D.M.; Figueredo, C.M.S. Cytokines expression in saliva and peri-implant crevicular fluid of patients with peri-implant disease. Clin. Oral Implant. Res. 2012, 25, 68–72. [Google Scholar] [CrossRef]
- Gonçalves, A.S.; Mosconi, C.; Jaeger, F.; Wastowski, I.; Aguiar, M.C.F.; Silva, T.A.; Ribeiro-Rotta, R.; Costa, N.L.; Batista, A.C. Overexpression of immunomodulatory mediators in oral precancerous lesions. Hum. Immunol. 2017, 78, 752–757. [Google Scholar] [CrossRef]
- Cecchettini, A.; Finamore, F.; Puxeddu, I.; Ferro, F.; Baldini, C. Salivary extracellular vesicles versus whole saliva: New perspectives for the identification of proteomic biomarkers in Sjögren’s syndrome. Clin. Exp. Rheumatol. 2019, 37 (Suppl. 118), 240–248. [Google Scholar]
- Lin, L.; Wang, J.; Liu, N.; Liu, S.; Xu, H.; Ji, N.; Zhou, M.; Zeng, X.; Zhang, D.; Li, J.; et al. Interleukin-37 expression and its potential role in oral leukoplakia and oral squamous cell carcinoma. Sci. Rep. 2016, 6, 26757. [Google Scholar] [CrossRef]
- Tobón-Arroyave, S.I.; Isaza-Guzman, D.M.; Restrepo-Cadavid, E.M.; Zapata-Molina, S.M.; Martínez-Pabón, M.C. Association of salivary levels of the bone remodelling regulators sRANKL and OPG with periodontal clinical status. J. Clin. Periodontol. 2012, 39, 1132–1140. [Google Scholar] [CrossRef] [PubMed]
- Buduneli, N.; Kinane, D.F. Host-derived diagnostic markers related to soft tissue destruction and bone degradation in periodontitis. J. Clin. Periodontol. 2011, 38, 85–105. [Google Scholar] [CrossRef]
- Al-Sabbagh, M.; Alladah, A.; Lin, Y.; Kryscio, R.J.; Thomas, M.V.; Ebersole, J.L.; Miller, C.S. Bone remodeling-associated salivary biomarker MIP-1α distinguishes periodontal disease from health. J. Periodontal Res. 2011, 47, 389–395. [Google Scholar] [CrossRef]
- Fine, D.H.; Markowitz, K.; Furgang, D.; Fairlie, K.; Ferrandiz, J.; Nasri, C.; McKiernan, M.; Donnelly, R.; Gunsolley, J. Macrophage Inflammatory Protein-1α: A Salivary Biomarker of Bone Loss in a Longitudinal Cohort Study of Children at Risk for Aggressive Periodontal Disease? J. Periodontol. 2009, 80, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Miricescu, D.; Totan, A.; Calenic, B.; Mocanu, B.; Didilescu, A.; Mohora, M.; Spinu, T.; Greabu, M. Salivary biomarkers: Relationship between oxidative stress and alveolar bone loss in chronic periodontitis. Acta Odontol. Scand. 2013, 72, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Zappacosta, B.; Manni, A.; Persichilli, S.; Boari, A.; Scribano, D.; Minucci, A.; Raffaelli, L.; Giardina, B.; De Sole, P. Salivary thiols and enzyme markers of cell damage in periodontal disease. Clin. Biochem. 2007, 40, 661–665. [Google Scholar] [CrossRef]
- Kugahara, T.; Shosenji, Y.; Ohashi, K. Screening for periodontitis in pregnant women with salivary enzymes. J. Obstet. Gynaecol. Res. 2007, 34, 40–46. [Google Scholar] [CrossRef]
- Luke, R.; Khan, S.N.; Iqbal, P.S.; Soman, R.R.; Chakkarayan, J.; Krishnan, V. Estimation of Specific Salivary Enzymatic Biomarkers in Individuals with Gingivitis and Chronic Periodontitis: A Clinical and Biochemical Study. J. Int. Oral Health 2015, 7, 54–57. [Google Scholar]
- Dabra, S.; China, K.; Kaushik, A. Salivary enzymes as diagnostic markers for detection of gingival/periodontal disease and their correlation with the severity of the disease. J. Indian Soc. Periodontol. 2012, 16, 358–364. [Google Scholar] [CrossRef]
- Nomura, Y.; Tamaki, Y.; Tanaka, T.; Arakawa, H.; Tsurumoto, A.; Kirimura, K.; Sato, T.; Hanada, N.; Kamoi, K. Screening of periodontitis with salivary enzyme tests. J. Oral Sci. 2006, 48, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Gürsoy, U.K.; Könönen, E.; Pradhan-Palikhe, P.; Tervahartiala, T.; Pussinen, P.; Suominen-Taipale, L.; Sorsa, T. Salivary MMP-8, TIMP-1, and ICTP as markers of advanced periodontitis. J. Clin. Periodontol. 2010, 37, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Lira-Junior, R.; Öztürk, V.Ö.; Emingil, G.; Bostanci, N.; Boström, E.A. Salivary and Serum Markers Related to Innate Immunity in Generalized Aggressive Periodontitis. J. Periodontol. 2017, 88, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Thumbigere-Math, V.; Michalowicz, B.S.; De Jong, E.P.; Griffin, T.J.; Basi, D.L.; Hughes, P.J.; Tsai, M.L.; Swenson, K.K.; Rockwell, L.; Gopalakrishnan, R. Salivary proteomics in bisphosphonate-related osteonecrosis of the jaw. Oral Dis. 2013, 21, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Thumbigere-Math, V.; Michalowicz, B.S.; Hughes, P.J.; Basi, D.L.; Tsai, M.L.; Swenson, K.K.; Rockwell, L.; Gopalakrishnan, R. Serum Markers of Bone Turnover and Angiogenesis in Patients with Bisphosphonate-Related Osteonecrosis of the Jaw after Discontinuation of Long-Term Intravenous Bisphosphonate Therapy. J. Oral Maxillofac. Surg. 2015, 74, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Isaza-Guzman, D.M.; Arias-Osorio, C.; Martínez-Pabón, M.C.; Tobón-Arroyave, S.I. Salivary levels of matrix metalloproteinase (MMP)-9 and tissue inhibitor of matrix metalloproteinase (TIMP)-1: A pilot study about the relationship with periodontal status and MMP-9−1562C/T gene promoter polymorphism. Arch. Oral Boil. 2011, 56, 401–411. [Google Scholar] [CrossRef] [PubMed]
- Rudrakshi, C.; Srinivas, N.; Mehta, D.S. A comparative evaluation of hepatocyte growth factor levels in gingival crevicular fluid and saliva and its correlation with clinical parameters in patients with and without chronic periodontitis: A clinico-biochemical study. J. Indian Soc. Periodontol. 2011, 15, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Wilczyńska-Borawska, M.; Borawski, J.; Baginska, J.; Małyszko, J.; Myśliwiec, M. Hepatocyte Growth Factor in Saliva of Patients with Renal Failure and Periodontal Disease. Ren. Fail. 2012, 34, 942–951. [Google Scholar] [CrossRef]
- Wilczyńska-Borawska, M.; Borawski, J.; Kovalchuk, O.; Chyczewski, L.; Stokowska, W. Hepatocyte growth factor in saliva is a potential marker of symptomatic periodontal disease. J. Oral Sci. 2006, 48, 47–50. [Google Scholar] [CrossRef][Green Version]
- Aqrawi, L.A.; Galtung, H.K.; Guerreiro, E.M.; Øvstebø, R.; Thiede, B.; Utheim, T.P.; Chen, X.; Utheim Øygunn, A.; Palm, Ø.; Skarstein, K.; et al. Proteomic and histopathological characterisation of sicca subjects and primary Sjögren’s syndrome patients reveals promising tear, saliva and extracellular vesicle disease biomarkers. Arthritis Res. Ther. 2019, 21, 181. [Google Scholar] [CrossRef]
- Garza-García, F.; Delgado-García, G.; Garza-Elizondo, M.; Ceceñas-Falcón, L.Á.; Galarza-Delgado, D.; Riega-Torres, J. Salivary β2-microglobulin positively correlates with ESSPRI in patients with primary Sjögren’s syndrome. Rev. Bras. Reum. Engl. Ed. 2017, 57, 182–184. [Google Scholar] [CrossRef]
- Shen, X.; Xi, G.; Maile, L.A.; Wai, C.; Rosen, C.J.; Clemmons, D.R. Insulin-Like Growth Factor (IGF) Binding Protein 2 Functions Coordinately with Receptor Protein Tyrosine Phosphatase β and the IGF-I Receptor to Regulate IGF-I-Stimulated Signaling. Mol. Cell. Boil. 2012, 32, 4116–4130. [Google Scholar] [CrossRef]
- Suresh, L.; Malyavantham, K.S.; Shen, L.; Ambrus, J.L. Investigation of novel autoantibodies in Sjogren’s syndrome utilizing Sera from the Sjogren’s international collaborative clinical alliance cohort. BMC Ophthalmol. 2015, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Metgud, R. Estimation of salivary lactate dehydrogenase in oral leukoplakia and oral squamous cell carcinoma: A biochemical study. J. Cancer Res. Ther. 2015, 11, 119. [Google Scholar] [CrossRef] [PubMed]
- Shetty, S.R.; Chadha, R.; Babu, S.; Kumari, S.; Bhat, S.; Achalli, S. Salivary lactate dehydrogenase levels in oral leukoplakia and oral squamous cell carcinoma: A biochemical and clinicopathological study. J. Cancer Res. Ther. 2012, 8, 123. [Google Scholar]
- Jaeger, F.; Assunção, A.C.; Caldeira, P.C.; Queiroz-Junior, C.M.; Bernardes, V.F.; Aguiar, M.C.F. Is salivary epidermal growth factor a biomarker for oral leukoplakia? A preliminary study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 451–458. [Google Scholar] [CrossRef][Green Version]
- Hoffmann, R.R.; Yurgel, L.S.; Campos, M.M. Evaluation of salivary endothelin-1 levels in oral squamous cell carcinoma and oral leukoplakia. Regul. Pept. 2011, 166, 55–58. [Google Scholar] [CrossRef]
- González-Moles, M.Á.; Warnakulasuriya, S.; González-Ruiz, I.; González-Ruiz, L.; Ayén, Á.; Lenouvel, D.; Ruiz-Ávila, I.; Ramos-García, P. Worldwide prevalence of oral lichen planus: A systematic review and meta-analysis. Oral Dis. 2020. [Google Scholar] [CrossRef]
- González-Moles, M.Á.; Ruiz-Ávila, I.; González-Ruíz, L.; Ayén, Á.; Gil-Montoya, J.A.; Ramos-García, P. Malignant transformation risk of oral lichen planus: A systematic review and comprehensive meta-analysis. Oral Oncol. 2019, 96, 121–130. [Google Scholar] [CrossRef]
- Kurago, Z. Etiology and pathogenesis of oral lichen planus: An overview. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, 72–80. [Google Scholar] [CrossRef]
- Montero-López, E.; Santos-Ruiz, A.; Gonzalez, R.; Navarrete-Navarrete, N.; Ortego-Centeno, N.; Martínez-Augustín, O.; Rodríguez-Blázquez, M.; Peralta-Ramírez, M.I. Analyses of hair and salivary cortisol for evaluating hypothalamic–pituitary–adrenal axis activation in patients with autoimmune disease. Stress 2017, 20, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.B.; Carnelio, S.; Shenoy, R.; Gyawali, P.; Mukherjee, M. Oxidative stress and antioxidant defense in oral lichen planus and oral lichenoid reaction. Scand. J. Clin. Lab. Investig. 2010, 70, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Sezer, E.; Ozugurlu, F.; Ozyurt, H.; Sahin, S.; Etikan, I. Lipid peroxidation and antioxidant status in lichen planus. Clin. Exp. Dermatol. 2007, 32, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Sugermann, P.B.; Savage, N.W.; Seymour, G.; Walsh, L.J. Is there a role for tumor necrosis factor-alpha (TNF-alpha) in oral lichen planus? J. Oral Pathol. Med. 1996, 25, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, T. IL-6: From its discovery to clinical applications. Int. Immunol. 2010, 22, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Wang, Y.; Sun, Q.; Jiang, C.; Zhu, M.; Song, C.; Li, C.; Du, G.; Deng, Y.; Nie, H.; et al. Enhanced T-cell proliferation and IL-6 secretion mediated by overexpression of TRIM21 in oral lesions of patients with oral lichen planus. J. Oral Pathol. Med. 2019, 49, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Slots, J. Periodontitis: Facts, fallacies and the future. Periodontology 2000 2017, 75, 7–23. [Google Scholar] [CrossRef]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Page, R.C. The role of inflammatory mediators in the pathogenesis of periodontal disease. J. Periodontal Res. 1991, 26, 230–242. [Google Scholar] [CrossRef]
- Birkedal-Hansen, H. Role of cytokines and inflammatory mediators in tissue destruction. J. Periodontal Res. 1993, 28, 500–510. [Google Scholar] [CrossRef]
- Di Benedetto, A.; Gigante, I.; Colucci, S.; Grano, M. Periodontal Disease: Linking the Primary Inflammation to Bone Loss. Clin. Dev. Immunol. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Fox, I.R.; Kang, I. Pathogenesis of Sjögren’s syndrome. Rheum. Dis. Clin. N. Am. 1992, 18, 517–538. [Google Scholar]
- Tincani, A.; Andreoli, L.; Cavazzana, I.; Doria, A.; Favero, M.; Fenini, M.-G.; Franceschini, F.; Lojacono, A.; Nascimbeni, G.; Santoro, A.; et al. Novel aspects of Sjögren’s syndrome in 2012. BMC Med. 2013, 11, 93. [Google Scholar] [CrossRef]
- Venables, P.J. Sjögren’s syndrome. Best Pr. Res. Clin. Rheumatol. 2004, 18, 313–329. [Google Scholar] [CrossRef] [PubMed]
- Mavragani, C.P. Mechanisms and New Strategies for Primary Sjögren’s Syndrome. Annu. Rev. Med. 2017, 68, 331–343. [Google Scholar] [CrossRef]
- Márton, K.D.; Boros, I.; Varga, G.; Zelles, T.; Fejérdy, P.; Zeher, M.; Nagy, G. Evaluation of palatal saliva flow rate and oral manifestations in patients with Sjogren’s syndrome. Oral Dis. 2006, 12, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Vitali, C.; Bombardieri, S.; Jonsson, R.; Moutsopoulos, H.M.; Alexander, E.L.E.; Carsons, S.E.; Daniels, T.; Fox, P.C.I.; Fox, R.; Kassan, S.S.; et al. Classification criteria for Sjogren’s syndrome: A revised version of the European criteria proposed by the American-European Consensus Group. Ann. Rheum. Dis. 2002, 61, 554–558. [Google Scholar] [CrossRef]
- Shiboski, S.C.; Shiboski, C.H.; Criswell, L.A.; Baer, A.N.; Challacombe, S.; Lanfranchi, H.; Schiodt, M.; Umehara, H.; Vivino, F.; Zhao, Y.; et al. American College of Rheumatology classification criteria for Sjögren’s syndrome: A data-driven, expert consensus approach in the Sjögren’s International Collaborative Clinical Alliance cohort. Arthritis Rheum. 2012, 64, 475–487. [Google Scholar] [CrossRef]
- Shiboski, C.H.; Shiboski, S.C.; Seror, R.A.; Criswell, L.; Labetoulle, M.; Lietman, T.M.; Rasmussen, A.; Scofield, H.; Vitali, C.; Bowman, S.J.; et al. 2016 American College of Rheumatology/European League against Rheumatism classification criteria for primary Sjögren’s syndrome. Ann. Rheum. Dis. 2016, 76, 9–16. [Google Scholar] [CrossRef]
- Naushin, T.; Khan, M.M.; Ahmed, S.; Hassan, M.-U.; Iqbal, F.; Bashir, N.; Khan, A.S. Determination of Ki-67 expression in oral leukoplakia in snuff users and non-users in Khyber Pakhtunkhwa province of Pakistan. Prof. Med. J. 2020, 27, 682–687. [Google Scholar] [CrossRef]
- Mehta, T.; Shah, S.; Dave, B.; Shah, R.; Dave, R. Socioeconomic and cultural impact of tobacco in India. J. Fam. Med. Prim. Care 2018, 7, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Sujatha, D.; Hebbar, P.B.; Pai, A. Prevalence and correlation of oral lesions among tobacco smokers, tobacco chewers, areca nut and alcohol users. Asian Pac. J. Cancer Prev. 2012, 13, 1633–1637. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Abhisheik, K.; Balasundari, S.; Devendra, N.K.; Shadab, K.; Anupama, M. Identification of Candida albicans using different culture media and its association in leukoplakia and oral squamous cell carcinoma. J. Oral Maxillofac. Pathol. 2019, 23, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Yamaura, C.; Ohara-Nemoto, Y.; Tajika, S.; Kodama, Y.; Ohya, T.; Harada, R.; Kimura, S. Streptococcus anginosus infection in oral cancer and its infection route. Oral Dis. 2005, 11, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Kazanowska-Dygdała, M.; Duś, I.; Radwan-Oczko, M. The presence of Helicobacter pylori in oral cavities of patients with leukoplakia and oral lichen planus. J. Appl. Oral Sci. 2016, 24, 18–23. [Google Scholar] [CrossRef] [PubMed]
- De La Cour, C.D.; Sperling, C.D.; Belmonte, F.; Syrjänen, S.; Kjaer, S.K. Human papillomavirus prevalence in oral potentially malignant disorders: Systematic review and meta-analysis. Oral Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Guidry, J.T.; Birdwell, C.E.; Scott, R.S. Epstein-Barr virus in the pathogenesis of oral cancers. Oral Dis. 2017, 24, 497–508. [Google Scholar] [CrossRef]
- Kushlinskiĭ, E.; Nagibin, A.A.; Laptev, I.P. Determination of the sensitivity of tumorous and pretumorous processes in the oral mucosa to steroid hormones. Stomatology 1988, 67, 32–33. [Google Scholar]
- Sridharan, G.; Ramani, P.; Patankar, S.; Vijayaraghavan, R. Analysis of estrogen metabolites in oral Leukoplakia and oral squamous cell carcinoma. Int. J. Pharm. Bio Sci. 2017, 8. [Google Scholar] [CrossRef]
- Mello, F.W.; Miguel, A.F.P.; Dutra-Horstmann, K.L.; Porporatti, A.L.; Warnakulasuriya, S.; Guerra, E.N.S.; Rivero, E.R.C. Prevalence of oral potentially malignant disorders: A systematic review and meta-analysis. J. Oral Pathol. Med. 2018, 47, 633–640. [Google Scholar] [CrossRef]
- Warnakulasuriya, S.; Ariyawardana, A. Malignant transformation of oral leukoplakia: A systematic review of observational studies. J. Oral Pathol. Med. 2015, 45, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Van Der Waal, I. Oral leukoplakia: A diagnostic challenge for clinicians and pathologists. Oral Dis. 2018, 25, 348–349. [Google Scholar] [CrossRef] [PubMed]
- Jung, R.E.; Zembic, A.; Pjetursson, B.E.; Zwahlen, M.; Thoma, D.S. Systematic review of the survival rate and the incidence of biological, technical, and aesthetic complications of single crowns on implants reported in longitudinal studies with a mean follow-up of 5 years. Clin. Oral Implant. Res. 2012, 23, 2–21. [Google Scholar] [CrossRef] [PubMed]
- Khammissa, R.A.G.; Feller, L.; Meyerov, R.; Lemmer, J. Peri-implant mucositis and peri-implantitis: Clinical and histopathological characteristics and treatment. SADJ J. S. Afr. Dent. Assoc. = Tydskr. Suid-Afrik. Tandheelkd. Ver. 2012, 67, 124–126. [Google Scholar]
- Wilson, V. An Insight into Peri-Implantitis: A Systematic Literature Review. Prim. Dent. J. 2013, 2, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Smeets, R.; Henningsen, A.; Jung, O.; Heiland, M.; Hammächer, C.; Stein, J.M. Definition, etiology, prevention and treatment of peri-implantitis—A review. Head Face Med. 2014, 10, 34. [Google Scholar] [CrossRef]
- Gomes, A.M.; Douglas-De-Oliveira, D.W.; Costa, F.O. Could the biomarker levels in saliva help distinguish between healthy implants and implants with peri-implant disease? A systematic review. Arch. Oral Boil. 2018, 96, 216–222. [Google Scholar] [CrossRef]
- Kolokythas, A.; Karras, M.; Collins, E.; Flick, W.; Miloro, M.; Adami, G. Salivary Biomarkers Associated with Bone Deterioration in Patients with Medication-Related Osteonecrosis of the Jaws. J. Oral Maxillofac. Surg. 2015, 73, 1741–1747. [Google Scholar] [CrossRef]
- Marx, R.E. Pamidronate (Aredia) and zoledronate (Zometa) induced avascular necrosis of the jaws: A growing epidemic. J. Oral Maxillofac. Surg. 2003, 61, 1115–1117. [Google Scholar] [CrossRef]
- Ruggiero, S.L.; Dodson, T.B.; Fantasia, J.; Goodday, R.; Aghaloo, T.; Mehrotra, B.; O’Ryan, F. American Association of Oral and Maxillofacial Surgeons Position Paper on Medication-Related Osteonecrosis of the Jaw—2014 Update. J. Oral Maxillofac. Surg. 2014, 72, 1938–1956. [Google Scholar] [CrossRef]
- Manzano-Moreno, F.J.; Ramos-Torrecillas, J.; De Luna-Bertos, E.; Reyes-Botella, C.; Ruiz, C.; García-Martínez, O. Nitrogen-containing bisphosphonates modulate the antigenic profile and inhibit the maturation and biomineralization potential of osteoblast-like cells. Clin. Oral Investig. 2014, 19, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Manzano-Moreno, F.J.; Ramos-Torrecillas, J.; De Luna-Bertos, E.; Ruiz, C.; García-Martínez, O. High doses of bisphosphonates reduce osteoblast-like cell proliferation by arresting the cell cycle and inducing apoptosis. J. Cranio-Maxillofac. Surg. 2015, 43, 396–401. [Google Scholar] [CrossRef]
- Mashiba, T.; Mori, S.; Komatsubara, S.; Cao, Y.; Manabe, T.; Norimatsu, H.; Burr, D.B. The effects of suppressed bone remodeling by bisphosphonates on microdamage accumulation and degree of mineralization in the cortical bone of dog rib. J. Bone Miner. Metab. 2005, 23, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Landesberg, R.; Cozin, M.; Cremers, S.; Woo, V.; Kousteni, S.; Sinha, S.; Garrett-Sinha, L.A.; Raghavan, S. Inhibition of Oral Mucosal Cell Wound Healing by Bisphosphonates. J. Oral Maxillofac. Surg. 2008, 66, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Fedele, S.; Porter, S.; D’Aiuto, F.; Aljohani, S.; Vescovi, P.; Manfredi, M.; Arduino, P.G.; Broccoletti, R.; Musciotto, A.; Di Fede, O.; et al. Nonexposed Variant of Bisphosphonate-associated Osteonecrosis of the Jaw: A Case Series. Am. J. Med. 2010, 123, 1060–1064. [Google Scholar] [CrossRef]
- O’Ryan, F.; Khoury, S.; Liao, W.; Han, M.M.; Hui, R.L.; Baer, D.; Martin, D.; Donald, L.; Lo, J. Intravenous Bisphosphonate-Related Osteonecrosis of the Jaw: Bone Scintigraphy as an Early Indicator. J. Oral Maxillofac. Surg. 2009, 67, 1363–1372. [Google Scholar] [CrossRef]
- Yatsuoka, W.; Ueno, T.; Miyano, K.; Uezono, Y.; Enomoto, A.; Kaneko, M.; Ota, S.; Soga, T.; Sugimoto, M.; Ushijima, T. Metabolomic profiling reveals salivary hypotaurine as a potential early detection marker for medication-related osteonecrosis of the jaw. PLoS ONE 2019, 14, e0220712. [Google Scholar] [CrossRef]
- Bagan, J.; Jiménez-Soriano, Y.; Gomez, D.; Sirera, R.; Poveda, R.; Scully, C. Collagen telopeptide (serum CTX) and its relationship with the size and number of lesions in osteonecrosis of the jaws in cancer patients on intravenous bisphosphonates. Oral Oncol. 2008, 44, 1088–1089. [Google Scholar] [CrossRef]
- Prá, K.J.D.; Lemos, C.; Okamoto, R.; Soubhia, A.; Pellizzer, E. Efficacy of the C-terminal telopeptide test in predicting the development of bisphosphonate-related osteonecrosis of the jaw: A systematic review. Int. J. Oral Maxillofac. Surg. 2017, 46, 151–156. [Google Scholar] [CrossRef]
- Kim, J.W.; Kong, K.A.; Kim, S.J.; Choi, S.K.; Cha, I.H.; Kim, M.-R. Prospective biomarker evaluation in patients with osteonecrosis of the jaw who received bisphosphonates. Bone 2013, 57, 201–205. [Google Scholar] [CrossRef][Green Version]
| Biomarker | Oral Pathology | Salivary Levels in Diagnosed Patients | Clinical Relevance | |
|---|---|---|---|---|
| Cortisol | OLP | Increased levels [17,18,19] | Diagnosis and recurrence of the pathology [20,21] | |
| Nitric Oxide | OLP | Increased levels [19] | Prognosis and presence of ulcers [22,23] | |
| ROS | OLP | Unaltered levels [22] | Cellular oxidative stress [22,24] | |
| CRP | OLP | Increased levels [22,25,26] | OLP progression [26] | |
| PD | Increased levels [27,28,29,30] | PD prognosis (modulation of the inflammation) [27,28,29,30] | ||
| TNF- α | OLP | Increased levels [19,31,32,33] | OLP diagnosis, commencement and progression [19,31] | |
| PD | Increased levels [34] Decreased levels [35] | Uncertain diagnosis, and prognosis role [36,37,38,39] | ||
| OL | Increased levels [40,41,42] Unaltered levels [43,44] | OL prognosis (malignant transformation, pre-oral cancer, and precancerous marker) [40,41,42] | ||
| PI | Increased levels [45] | Diagnosis of the pathology [45] | ||
| IL1 | IL1β | PD | Increased levels [35,38,39,46,47,48,49,50] | Diagnosis and progression (inflammatory modulation, severity-bone resorption, generalized PD and PD severity) [51,52,53,54,55] |
| OL | Unaltered levels [44] | - | ||
| PI | Increased levels [45,56] | Diagnosis of the pathology [45,56] | ||
| IL1α & IL1β | OLP | Increased levels [19,32] | Immune and inflammatory response modulator [57,58] | |
| MRONJ | Increased levels [59,60] | MRONJ diagnosis [59,60] | ||
| IL1RA | MRONJ | Increased levels [59,60] | MRONJ diagnosis [59,60] | |
| IL4 | OLP | Increased levels [19,61] | IL4 is not a good salivary marker for OLP prognosis [32,62] | |
| PD | Increased levels [63] | - | ||
| IL6 | OLP | Increased levels [19,32,64,65] | OLP prognosis (severity and wound marker). IL6 salivary marker is a good option for monitoring the treatment response [32,66] | |
| PD | Increased levels [38,63,67] Unaltered levels [27,39,49,68] | PD prognosis (inflammatory modulator) [37,69,70] | ||
| OL | Increased levels [41,71,72,73] Unaltered levels [44] | OL prognosis (tumor growth and higher blood vessel density) [74] | ||
| PI | Increased levels [45,75] | Early diagnosis and prognostic value [45,75] | ||
| MRONJ | Increased levels [59,60] | MRONJ diagnosis [59,60] | ||
| IL8 | OLP | Increased Levels [19,76,77] | IL8 is a solid salivary biomarker for OLP severity [32,66,78] | |
| OL | Increased levels [41,71,72,73] Unaltered levels [44] | OL prognosis (tumor growth and higher blood vessel density) [74] | ||
| PI | Increased Levels [79] | PI diagnosis [79] | ||
| IL10 | OL | Increased Levels [42,80] Unaltered levels [44] | Uncertain association with premalignant oral lesions [42,80] | |
| PI | Increased levels [45,75] | Early diagnosis and prognostic value [45,75] | ||
| IL12 | pSS | Increased Levels [81] | Diagnostic and prognostic value [81] | |
| IL17 | PD | Increased levels [54,63] | Localized periodontitis [54] | |
| IL23 | PD | Increased levels [54] | Localized periodontitis [54] | |
| IL37 | OL | Increased Levels [82] | ||
| RANKL | PD | Increased levels [83] Unaltered levels [34,84] | Uncertain prognosis value (bone loss) [34,83,84] | |
| MIP-1 | PD | Increased levels [85,86] | Diagnosis [85,86] | |
| OPG | PD | Decreased levels [83] Unaltered levels [34,84] | Uncertain prognosis value (bone loss) [34,83,84] | |
| OSC | PD | Decreased levels [83] Unaltered levels [34,84] | Uncertain prognosis value (bone loss) [34,83,84] | |
| ALP | PD | Increased levels [67,87,88,89,90,91,92] | Diagnosis of the pathology [67,87,88,89,90,91,92] | |
| LDH | PD | Increased levels [67,87,88,89,90,91,92] | Diagnosis of the pathology [67,87,88,89,90,91,92] | |
| AST | PD | Increased levels [67,87,88,89,90,91,92] | Diagnosis of the pathology [67,87,88,89,90,91,92] | |
| ALT | PD | Increased levels [67,87,88,89,90,91,92] | Diagnosis of the pathology [67,87,88,89,90,91,92] | |
| MMP8 | PD | Increased levels [27,39,47,48,67,87,93] | Very useful salivary biomarker for the diagnosis of PD [27,39,47,48,67,87,93] and PD severity [94] | |
| MMP9 | PD | Increased levels [27,35] | Diagnosis [27,35] | |
| MRONJ | Increased levels [95,96] | MRONJ diagnosis [95,96] | ||
| TIMP1 | PD | Decreased levels [93,97] | PD prognosis (advanced PD) [93] | |
| HGF | PD | Increased levels [98,99] | Prognosis of the pathology [98,99,100] | |
| NLRP3 | PD | Increased levels [55] | PD severity and chronicity. Also useful as a salivary biomarker for preventive or therapeutic purposes [55] | |
| CD44 | pSS | Increased levels [101] | Diagnostic and prognostic value [101] | |
| B2M | pSS | Increased levels [102] | Diagnostic and prognostic value [102] | |
| SP1 | pSS | Increased levels [103,104] | Early diagnosis and prognostic value [103,104] | |
| PSP | pSS | Increased levels [103,104] | Early diagnosis and prognostic value [103,104] | |
| CA6 | pSS | Increased levels [103,104] | Early diagnosis and prognostic value [103,104] | |
| LDH | OL | Increased levels [105,106] | Risk of malignant transformation of OL [105,106] | |
| TGFβ | OL | Unaltered levels [80,107,108] | Uncertain diagnosis and prognosis value [80,107,108] | |
| EGF | OL | Unaltered levels [80,107,108] | Uncertain diagnosis and prognosis value [80,107,108] | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melguizo-Rodríguez, L.; Costela-Ruiz, V.J.; Manzano-Moreno, F.J.; Ruiz, C.; Illescas-Montes, R. Salivary Biomarkers and Their Application in the Diagnosis and Monitoring of the Most Common Oral Pathologies. Int. J. Mol. Sci. 2020, 21, 5173. https://doi.org/10.3390/ijms21145173
Melguizo-Rodríguez L, Costela-Ruiz VJ, Manzano-Moreno FJ, Ruiz C, Illescas-Montes R. Salivary Biomarkers and Their Application in the Diagnosis and Monitoring of the Most Common Oral Pathologies. International Journal of Molecular Sciences. 2020; 21(14):5173. https://doi.org/10.3390/ijms21145173
Chicago/Turabian StyleMelguizo-Rodríguez, Lucía, Victor J. Costela-Ruiz, Francisco Javier Manzano-Moreno, Concepción Ruiz, and Rebeca Illescas-Montes. 2020. "Salivary Biomarkers and Their Application in the Diagnosis and Monitoring of the Most Common Oral Pathologies" International Journal of Molecular Sciences 21, no. 14: 5173. https://doi.org/10.3390/ijms21145173
APA StyleMelguizo-Rodríguez, L., Costela-Ruiz, V. J., Manzano-Moreno, F. J., Ruiz, C., & Illescas-Montes, R. (2020). Salivary Biomarkers and Their Application in the Diagnosis and Monitoring of the Most Common Oral Pathologies. International Journal of Molecular Sciences, 21(14), 5173. https://doi.org/10.3390/ijms21145173