Egg Allergy: Diagnosis and Immunotherapy
Abstract
1. An Overview
2. Food Allergy
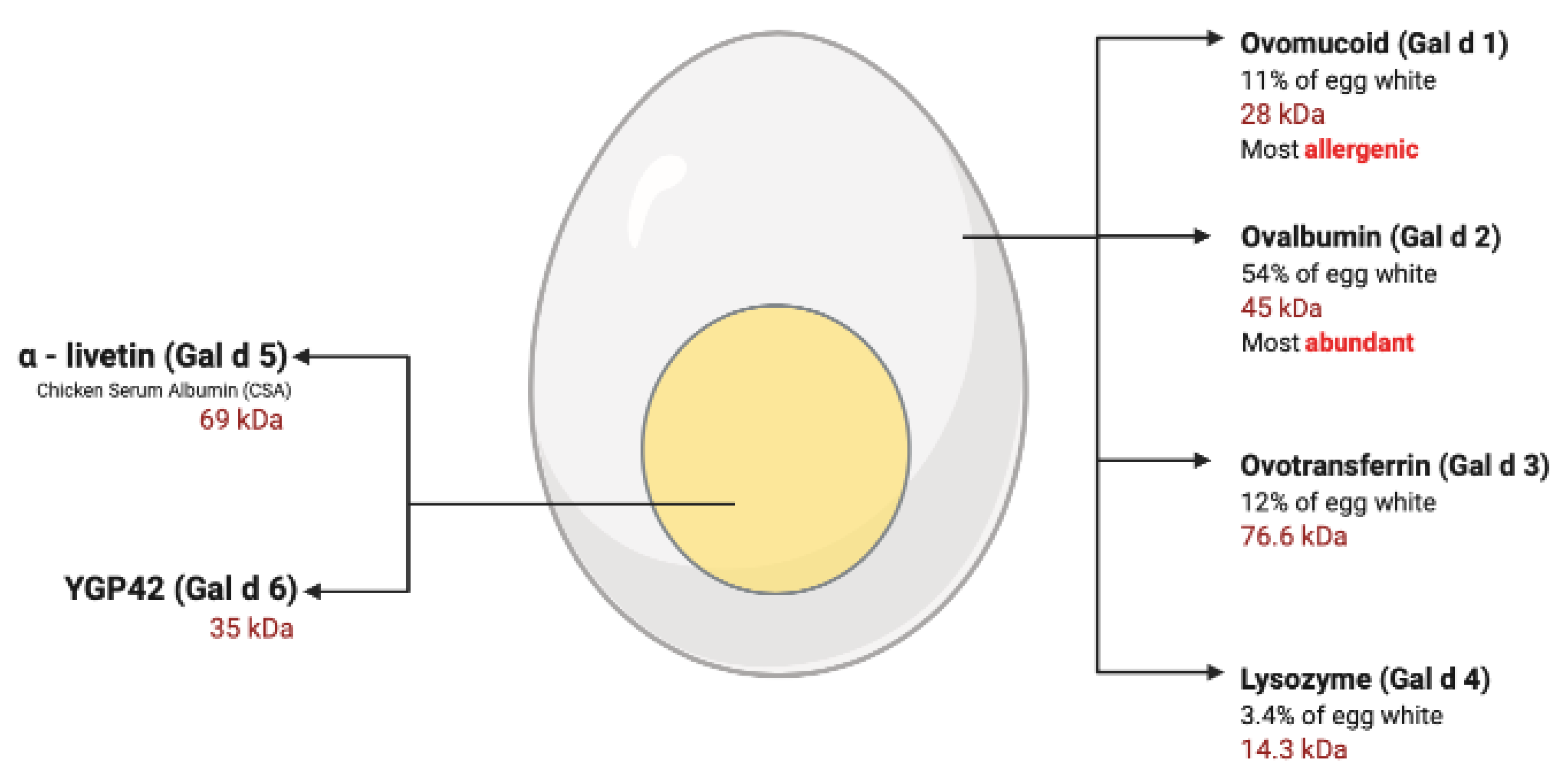
3. Egg Allergy
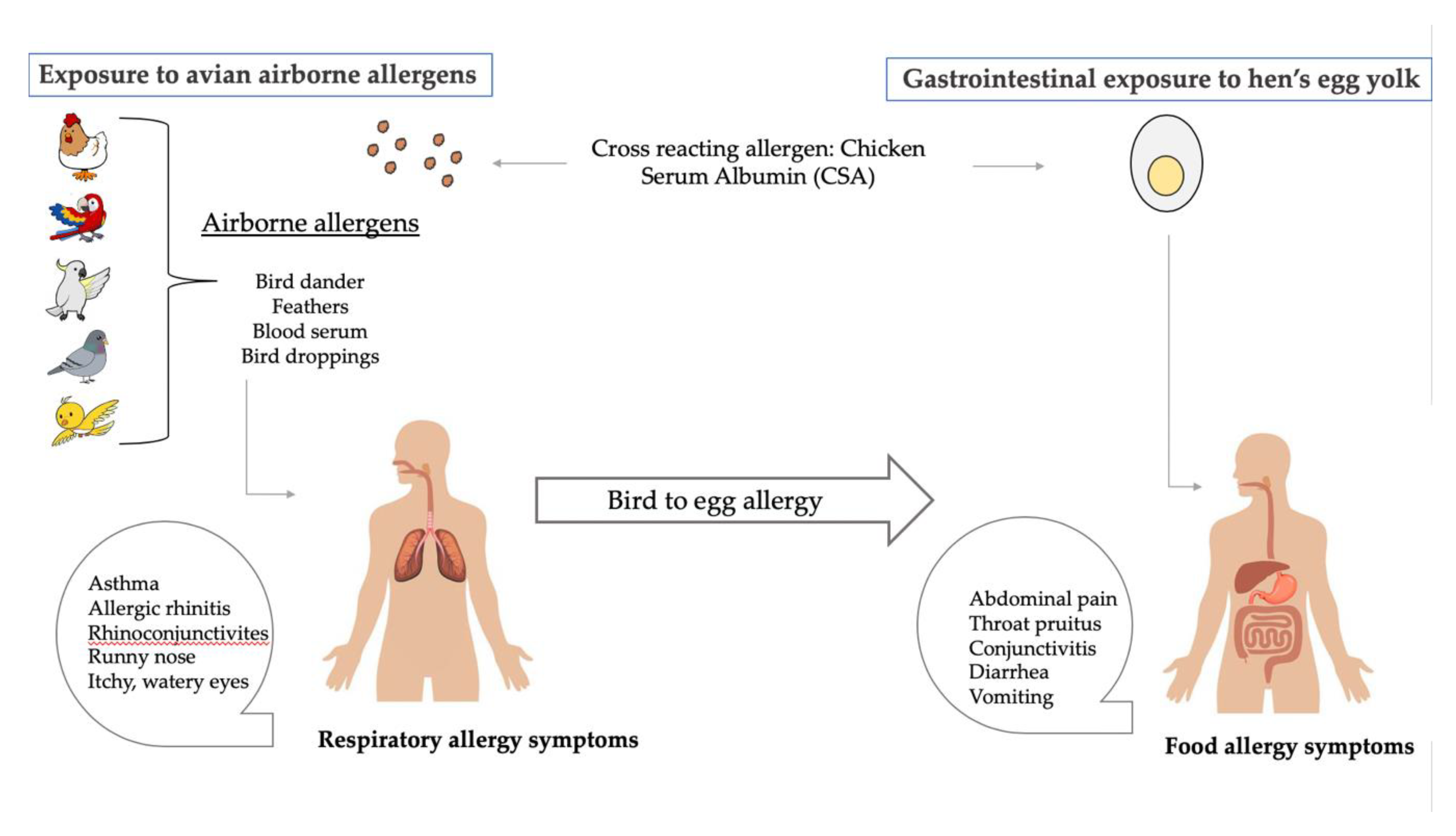
3.1. Egg Yolk Allergy
3.2. The Bird-Egg Syndrome
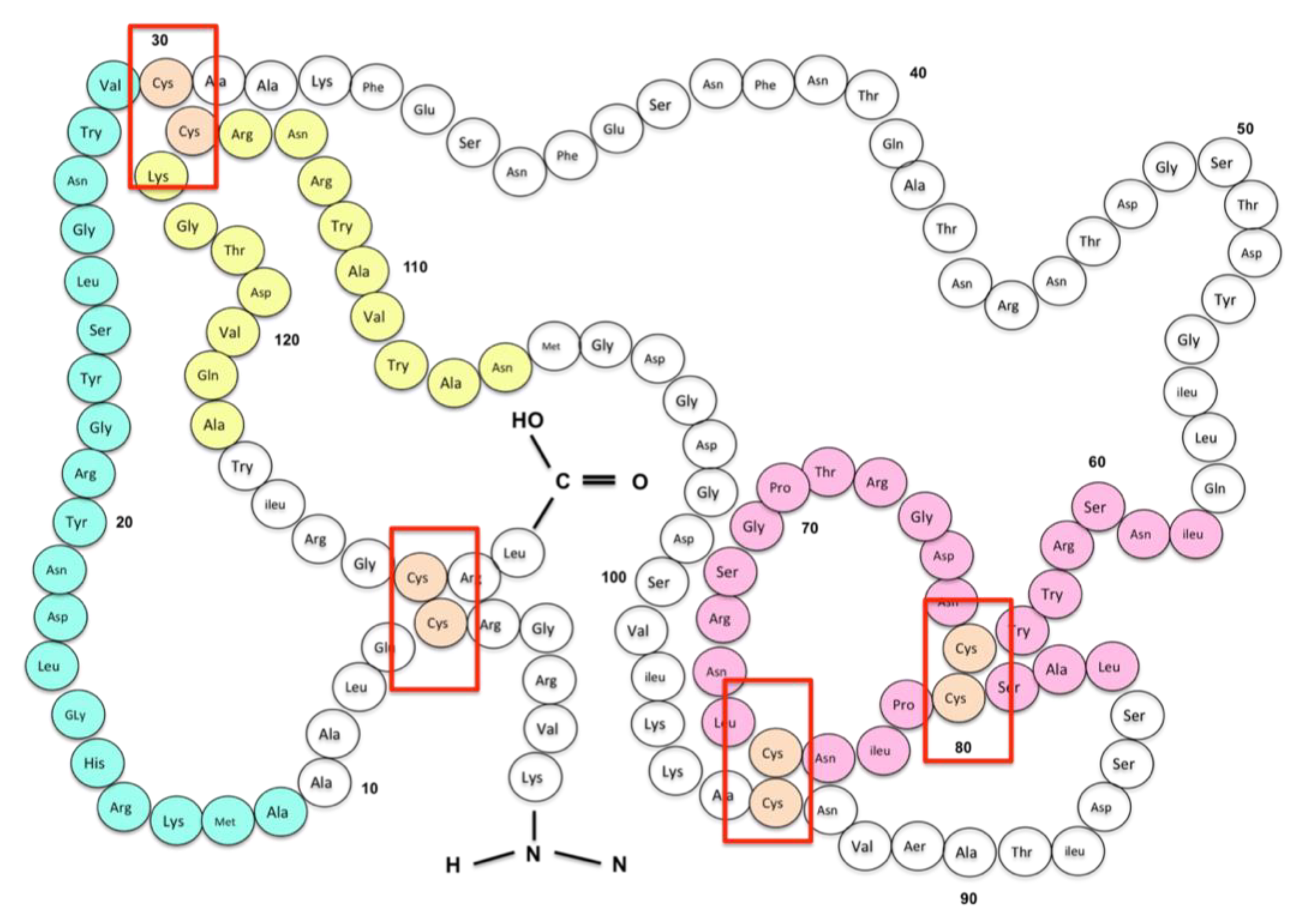
3.3. Egg White Allergy
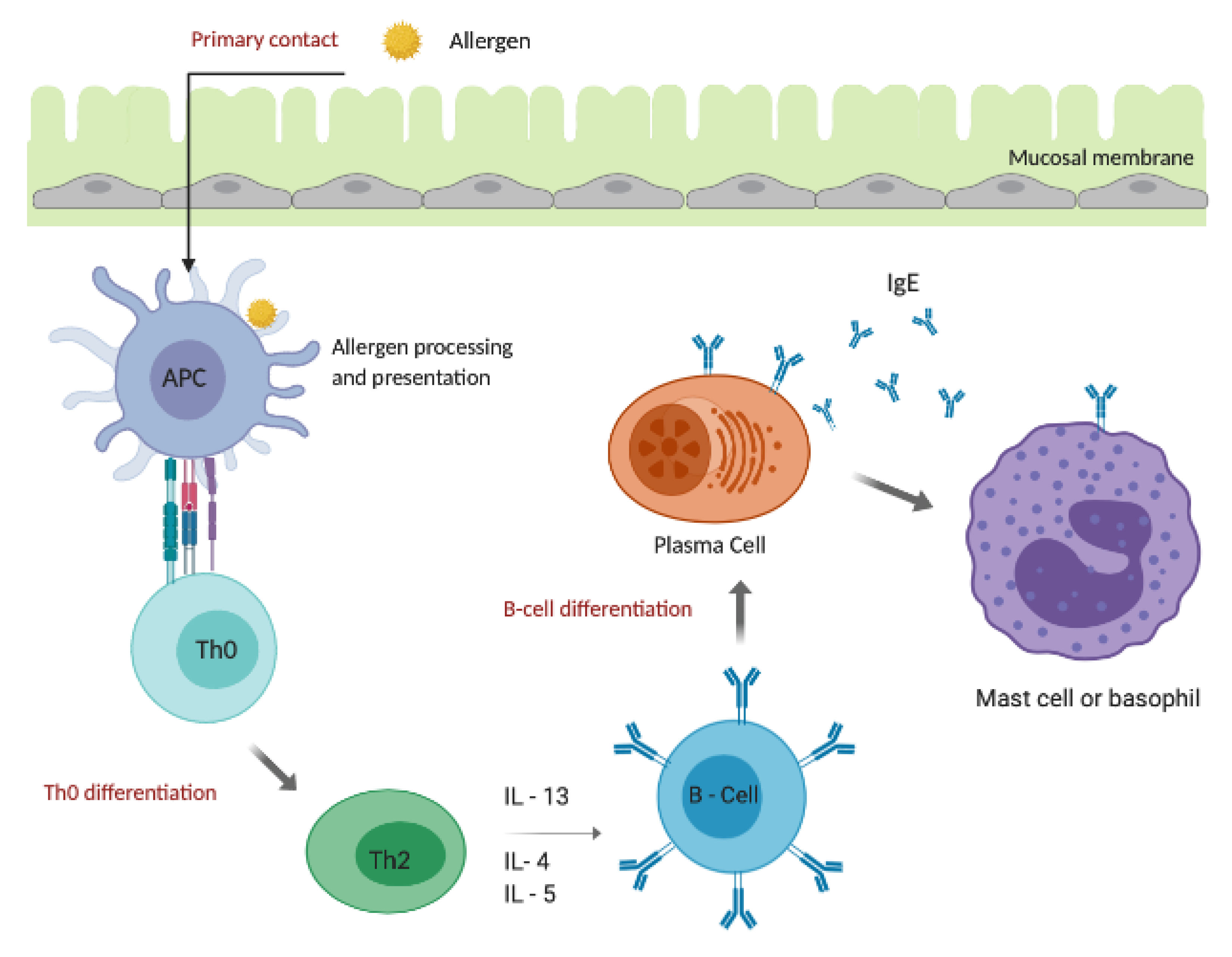
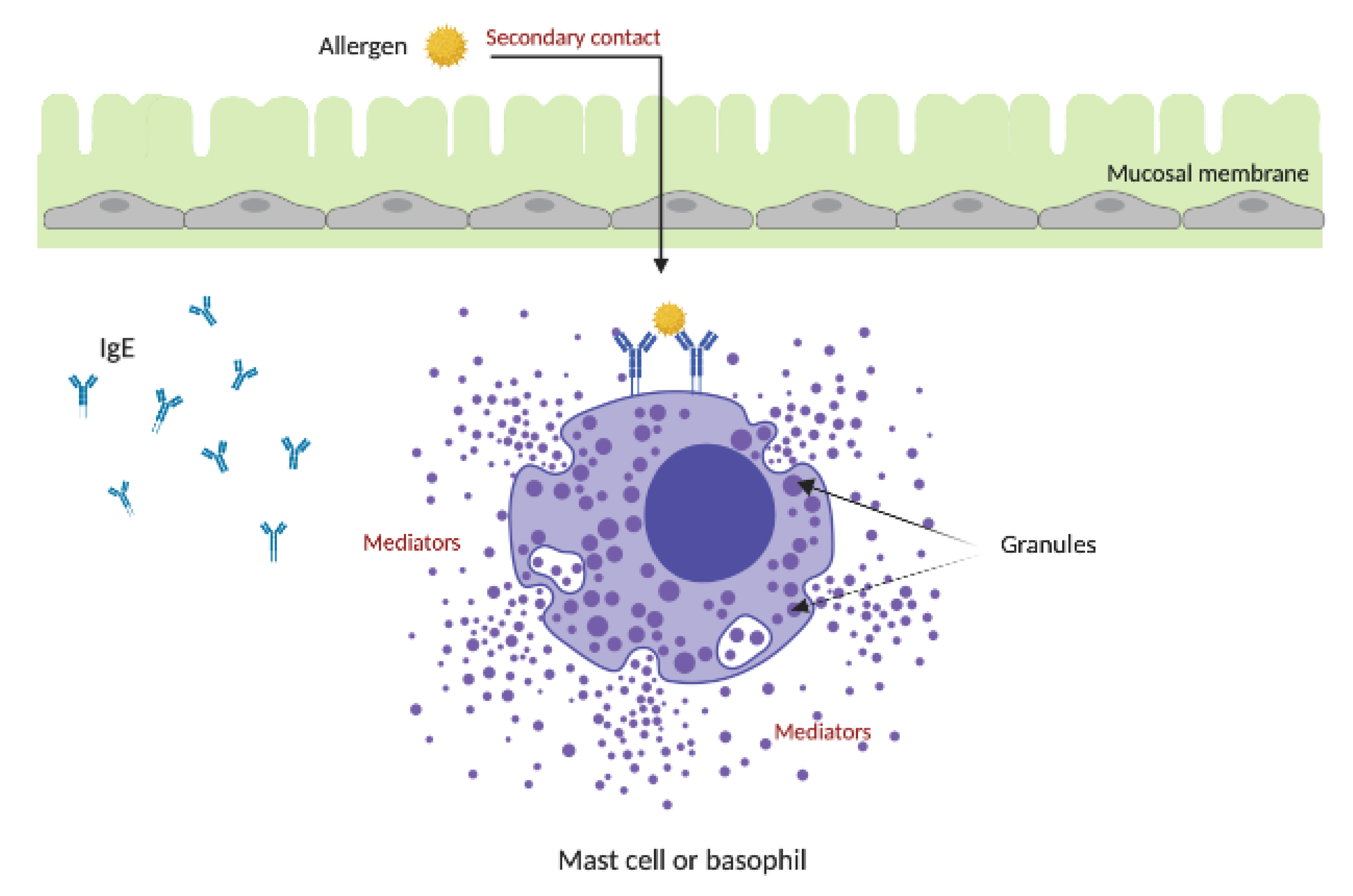
4. Allergic Response
5. Diagnosis of Allergy
Molecular-Based Allergy
6. Treatment
7. Production and Need for Recombinant and Hypoallergenic Egg Allergens
Recombinant Allergens in Animal Models
8. Current and Future Direction of Egg Allergy Research
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| IgE | Immunoglobulin E Antibody |
| APC | Antigen-Presenting Cells |
| Th0 | Naïve T-helper cells |
| Th2 | T-helper type 2 cells |
| IL-13 | Interleukin-13 |
| IL-4 | Interleukin-4 |
| IL-5 | Interleukin-5 |
| IL-10 | Interleukin-10 |
| IL-15 | Interleukin-15 |
| IL-25 | Interleukin-25 |
| IL-9 | Interleukin-9 |
| IL-14 | Interleukin-14 |
| IL-33 | Interleukin-33 |
| IL-18 | Interleukin-18 |
| IL-22 | Interleukin-21 |
| IgM | Immunoglobulin M Antibody |
| IgG | Immunoglobulin G Antibody |
| IgA | Immunoglobulin A Antibody |
| FcεRI | High-affinity IgE receptor |
| FDCs | Follicular dendritic cells |
| DCs | Dendritic cells |
| Tfh | T follicular helper |
| TSLP | Thymic stromal lymphopoietin |
| tIgE | Total serum IgE |
| SPT | Skin prick testing |
| IFNy | Interferon-gamma |
| EAACI | The European Academy of Allergy and Clinical Immunology |
| FEIA | Fluorescent enzyme immunoassay |
| SIT | Allergen-specific immunotherapy |
| AIT | Allergen immunotherapy |
| SLIT | Sublingual immunotherapy |
| DBPCFC | Double-blind, placebo-controlled food challenges |
| CDC | The US Centers for Disease Control and Prevention |
| OFC | Oral food challenges |
| FALCPA | Food Allergen Labelling and Consumer Protection Act |
| MCRI | Murdoch Children’s Research Institute |
| CSA | Chicken Serum Albumin |
| RAST | Radioallergosorbent test |
| Gal d 1 | Ovomucoid |
| Gal d 2 | Ovalbumin |
| Gal d 3 | Ovotransferrin |
| Gal d 4 | Lysozyme |
| Gal d 5 | α-livetin/CSA |
| Gal d 6 | Yolk glycoprotein 42/YGP42 |
| MA | Molecular based allergy |
| WHO | World Health Organization |
| IUIS | International Union of Immunological Species |
| ISAC | Immune-solid phase allergen chip |
| ISU-E | Standardized Units |
| kU/L | Kilo units per litre |
| BAT | The basophil activation test |
| ELISA | Enzyme-linked immunosorbent assay |
| NFA | Non-IgE-mediated food allergy |
| IFA | IgE-mediated food allergy |
| OIT | Oral immunotherapy |
| FDA | U.S Food and Drug Administration |
| sIgE | specific IgE |
| WAO | World Allergy Organization |
| FLG | Filaggrin gene |
| AD | Atopic dermatitis |
References
- Valenta, R.; Hochwallner, H.; Linhart, B.; Pahr, S. Food allergies: The basics. Gastroenterology 2015, 148, 1120–1131. [Google Scholar] [CrossRef] [PubMed]
- Quirce, S.; Maranon, F.; Umpierrez, A.; de las Heras, M.; Fernandez-Caldas, E.; Sastre, J. Chicken serum albumin (Gal d 5*) is a partially heat-labile inhalant and food allergen implicated in the bird-egg syndrome. Allergy 2001, 56, 754–762. [Google Scholar] [CrossRef] [PubMed]
- Anto, J.M.; Bousquet, J.; Akdis, M.; Auffray, C.; Keil, T.; Momas, I.; Postma, D.S.; Valenta, R.; Wickman, M.; Cambon-Thomsen, A.; et al. Mechanisms of the Development of Allergy (MeDALL): Introducing novel concepts in allergy phenotypes. J. Allergy Clin. Immunol. 2017, 139, 388–399. [Google Scholar] [CrossRef]
- Valenta, R.; Karaulov, A.; Niederberger, V.; Gattinger, P.; van Hage, M.; Flicker, S.; Linhart, B.; Campana, R.; Focke-Tejkl, M.; Curin, M.; et al. Chapter five—molecular aspects of allergens and allergy. In Advances in Immunology; Alt, F., Ed.; Academic Press: Cambridge, MA, USA, 2018; Volume 138, pp. 195–256. [Google Scholar]
- Pawankar, R. Allergic diseases and asthma: A global public health concern and a call to action. World Allergy Organ. J. 2014, 7, 1–3. [Google Scholar] [CrossRef]
- Kay, A.B. Allergy and Allergic Diseases, 2nd ed.; Blackwell Wiley InterScience: Hoboken, NJ, USA, 2008. [Google Scholar]
- Allergy. Available online: https://www.allergy.org.au/ascia-reports/allergy-and-immune-diseases-in-australia-2013 (accessed on 2 March 2020).
- Boyar, A.P. Richer, Alice, C. Food Allergies. CHOICE Curr. Rev. Acad. Libr. 2010, 5, 926. Available online: https://search.ebscohost.com/login.aspx?direct=true&db=edsgao&AN=edsgcl.251859486&authtype=sso&custid=deakin&site=eds-live&scope=site (accessed on 15 July 2020).
- Koplin, J.J.; Allen, K.J.; Gurrin, L.C.; Peters, R.L.; Lowe, A.J.; Tang, M.L.K.; Dharmage, S.C.; HealthNuts Study, T. The impact of family history of allergy on risk of food allergy: A population-based study of infants. Int. J. Environ. Res. Public Health 2013, 10, 5364–5377. [Google Scholar] [CrossRef]
- Sampson, H.A. Update on food allergy. J. Allergy Clin. Immunol. 2004, 113, 805–819. [Google Scholar] [CrossRef]
- Liu, A.H. Revisiting the hygiene hypothesis for allergy and asthma. J. Allergy Clin. Immunol. 2015, 136, 860–865. [Google Scholar] [CrossRef]
- von Mutius, E. Allergies, infections and the hygiene hypothesis—The epidemiological evidence. Immunobiology 2007, 212, 433–439. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Available online: http://www.cdc.gov/nchs/data/series/sr_10/sr10_258.pdf (accessed on 5 March 2020).
- Osborne, N.J.; Koplin, J.J.; Martin, P.E.; Gurrin, L.C.; Lowe, A.J.; Matheson, M.C.; Ponsonby, A.L.; Wake, M.; Tang, M.L.; Dharmage, S.C.; et al. Prevalence of challenge-proven IgE-mediated food allergy using population-based sampling and predetermined challenge criteria in infants. J. Allergy Clin. Immunol. 2011, 127, 668–676. [Google Scholar] [CrossRef]
- Loh, W.; Tang, M.L.K. The epidemiology of food allergy in the global context. Int. J. Environ. Res. Public Health 2018, 15, 2043. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L.; Pawankar, R.; Allen, K.J.; Campbell, D.E.; Sinn, J.K.; Fiocchi, A.; Ebisawa, M.; Sampson, H.A.; Beyer, K.; Lee, B.W. A global survey of changing patterns of food allergy burden in children. World Allergy Organ. J. 2013, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Pawankar, R.; Canonica, G.W.; Holgate, S.T.; Lockey, R.F.; Blaiss, M.S. World Allergy Organization (WAO) White Book on Allergy. Available online: https://www.worldallergy.org/UserFiles/file/WhiteBook2-2013-v8.pdf (accessed on 24 March 2020).
- Ring, J.; Krämer, U.; Schäfer, T.; Behrendt, H. Why are allergies increasing? Curr. Opin. Immunol. 2001, 13, 701–708. [Google Scholar] [CrossRef]
- Phelps, K.; Hassed, C. Allergies: General Practice: The Integrative Approach Series; Elsevier Health Sciences APAC: London, UK, 2012. [Google Scholar]
- Ko, J.; Lee, J.I.; Muñoz-Furlong, A.; Li, X.M.; Sicherer, S.H. Use of complementary and alternative medicine by food-allergic patients. Ann. Allergy Asthma Immunol. 2006, 97, 365–369. [Google Scholar] [CrossRef]
- Rona, R.J.; Keil, T.; Summers, C.; Gislason, D.; Zuidmeer, L.; Sodergren, E.; Sigurdardottir, S.T.; Lindner, T.; Goldhahn, K.; Dahlstrom, J.; et al. The prevalence of food allergy: A meta-analysis. J. Allergy Clin. Immunol. 2007, 120, 638–646. [Google Scholar] [CrossRef]
- Wei-Liang Tan, J.; Valerio, C.; Barnes, E.H.; Turner, P.J.; Van Asperen, P.A.; Kakakios, A.M.; Campbell, D.E. A randomized trial of egg introduction from 4 months of age in infants at risk for egg allergy. J. Allergy Clin. Immunol. 2017, 139, 1621–1628. [Google Scholar] [CrossRef]
- Savage, J.H.; Matsui, E.C.; Skripak, J.M.; Wood, R.A. The natural history of egg allergy. J. Allergy Clin. Immunol. 2007, 120, 1413–1417. [Google Scholar] [CrossRef]
- Dang, T.D.; Peters, R.L.; Koplin, J.J.; Dharmage, S.C.; Gurrin, L.C.; Ponsonby, A.L.; Martino, D.J.; Neeland, M.; Tang, M.L.K.; Allen, K.J. Egg allergen specific IgE diversity predicts resolution of egg allergy in the population cohort HealthNuts. Allergy 2019, 74, 318–326. [Google Scholar] [CrossRef]
- Peters, R.L.; Koplin, J.J.; Gurrin, L.C.; Dharmage, S.C.; Wake, M.; Ponsonby, A.L.; Tang, M.L.K.; Lowe, A.J.; Matheson, M.; Dwyer, T.; et al. The prevalence of food allergy and other allergic diseases in early childhood in a population-based study: HealthNuts age 4-year follow-up. J. Allergy Clin. Immunol. 2017, 140, 1451–1453. [Google Scholar] [CrossRef]
- Palmer, D.J.; Metcalfe, J.; Makrides, M.; Gold, M.S.; Quinn, P.; West, C.E.; Loh, R.; Prescott, S.L. Early regular egg exposure in infants with eczema: A randomized controlled trial. J. Allergy Clin. Immunol. 2013, 132, 3873–3892. [Google Scholar] [CrossRef]
- de Maat-Bleeker, F.; van Dijk, A.G.; Berrens, L. Allergy to egg yolk possibly induced by sensitization to bird serum antigens. Ann. Allergy 1985, 54, 245–248. [Google Scholar] [PubMed]
- Leduc, V.; Demeulemester, C.; Polack, B.; Guizard, C.; Le Guern, L.; Peltre, G. Immunochemical detection of egg-white antigens and allergens in meat products. Allergy 1999, 54, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Anet, J.; Back, J.F.; Baker, R.S.; Barnett, D.; Burley, R.W.; Howden, M.E.H. Allergens in the white and yolk of hen’s egg. Int. Arch. Allergy Immunol. 1985, 77, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Kay, A.B. Allergy and allergic diseases. First of two parts. N. Engl. J. Med. 2001, 344, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Iannotti, L.L.; Lutter, C.K.; Bunn, D.A.; Stewart, C.P. Eggs: The uncracked potential for improving maternal and young child nutrition among the world's poor. Nutr. Rev. 2014, 72, 355–368. [Google Scholar] [CrossRef]
- Peters, R.L.; Dharmage, S.C.; Gurrin, L.C.; Koplin, J.J.; Ponsonby, A.L.; Lowe, A.J.; Tang, M.L.; Tey, D.; Robinson, M.; Hill, D.; et al. The natural history and clinical predictors of egg allergy in the first 2 years of life: A prospective, population-based cohort study. J. Allergy Clin. Immunol. 2014, 133, 485–491. [Google Scholar] [CrossRef]
- Lemon-Mule, H.; Sampson, H.A.; Sicherer, S.H.; Shreffler, W.G.; Noone, S.; Nowak-Wegrzyn, A. Immunologic changes in children with egg allergy ingesting extensively heated egg. J. Allergy Clin. Immunol. 2008, 122, 977–983. [Google Scholar] [CrossRef]
- Dhanapala, P.; De Silva, C.; Doran, T.; Suphioglu, C. Cracking the egg: An insight into egg hypersensitivity. Mol. Immunol. 2015, 66, 375–383. [Google Scholar] [CrossRef]
- Caubet, J.C.; Wang, J. Current understanding of egg allergy. Pediatric Clin. N. Am. 2011, 58, 427–443. [Google Scholar] [CrossRef]
- Kovacs-Nolan, J.; Phillips, M.; Mine, Y. Advances in the value of eggs and egg components for human health. J. Agric. Food Chem. 2005, 53, 8421–8431. [Google Scholar] [CrossRef]
- De Silva, C.; Dhanapala, P.; King, S.; Doran, T.; Tang, M.; Suphioglu, C. Immunological comparison of native and recombinant hen's egg yolk allergen, chicken serum albumin (Gal d 5), produced in kluveromyces lactis. Nutrients 2018, 10, 757. [Google Scholar] [CrossRef] [PubMed]
- Inomata, N.; Kawano, K.; Aihara, M. Bird-egg syndrome induced by α-livetin sensitization in a budgerigar keeper: Successful induction of tolerance by avoiding exposure to avians. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Mandallaz, M.M.; de Weck, A.L.; Dahinden, C.A. Bird-egg syndrome. Cross-reactivity between bird antigens and egg-yolk livetins in IgE-mediated hypersensitivity. Int. Arch. Allergy Appl. Immunol. 1988, 87, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Fukutomi, Y. Occupational food allergy. Curr. Opin. Allergy Clin. Immunol. 2019, 19, 243–248. [Google Scholar] [CrossRef]
- Tsakok, T.; Marrs, T.; Mohsin, M.; Baron, S.; du Toit, G.; Till, S.; Flohr, C. Does atopic dermatitis cause food allergy? A systematic review. J. Allergy Clin. Immunol. 2016, 137, 1071–1078. [Google Scholar] [CrossRef]
- Brough, H.A.; Liu, A.H.; Sicherer, S.; Makinson, K.; Douiri, A.; Brown, S.J.; Stephens, A.C.; Irwin McLean, W.H.; Turcanu, V.; Wood, R.A.; et al. Atopic dermatitis increases the effect of exposure to peanut antigen in dust on peanut sensitization and likely peanut allergy. J. Allergy Clin. Immunol. 2015, 135, 1641–1670. [Google Scholar] [CrossRef]
- Werfel, T.; Breuer, K. Role of food allergy in atopic dermatitis. Curr. Opin. Allergy Clin. Immunol. 2004, 4, 379–385. [Google Scholar] [CrossRef]
- Berbegal, L.; DeLeon, F.J.; González, I.; Silvestre, J.F. Protein contact dermatitis caused by chicken meat in bird-egg syndrome. Contact Dermat. 2017, 77, 253–254. [Google Scholar] [CrossRef]
- Flohr, C.; England, K.; Radulovic, S.; McLean, W.H.I.; Campbell, L.E.; Barker, J.; Perkin, M.; Lack, G. Filaggrin loss-of-function mutations are associated with early-onset eczema, eczema severity and transepidermal water loss at 3 months of age. Br. J. Dermatol. 2010, 163, 1333–1336. [Google Scholar] [CrossRef]
- Akiyama, M. FLG mutations in ichthyosis vulgaris and atopic eczema: Spectrum of mutations and population genetics. Br. J. Dermatol. 2010, 162, 472–477. [Google Scholar] [CrossRef]
- Bartnikas, L.M.; Gurish, M.F.; Burton, O.T.; Leisten, S.; Janssen, E.; Oettgen, H.C.; Beaupré, J.; Lewis, C.N.; Austen, K.F.; Schulte, S.; et al. Epicutaneous sensitization results in IgE-dependent intestinal mast cell expansion and food-induced anaphylaxis. J. Allergy Clin. Immunol. 2013, 131, 4514–4560. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C.; Perkin, M.; Logan, K.; Marrs, T.; Radulovic, S.; Campbell, L.E.; MacCallum, S.F.; McLean, W.H.I.; Lack, G. Atopic dermatitis and disease severity are the main risk factors for food sensitization in exclusively breastfed infants. J. Investig. Dermatol. 2014, 134, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Ansotegui, I.J.; Melioli, G.; Canonica, G.W.; Gómez, R.M.; Jensen-Jarolim, E.; Ebisawa, M.; Luengo, O.; Caraballo, L.; Passalacqua, G.; Poulsen, L.K.; et al. A WAO—ARIA—GA2LEN consensus document on molecular-based allergy diagnosis (PAMD@): Update 2020. World Allergy Organ. J. 2020, 13. [Google Scholar] [CrossRef]
- Mann, K.; Mann, M. In-depth analysis of the chicken egg white proteome using an LTQ Orbitrap Velos. Proteome Sci. 2011, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Kay, A.B. Allergy and Allergic Diseases; Blackwell Science: Malden, MA, USA, 1997. [Google Scholar]
- Steinman, R.M. The dendritic cell system and its role in immunogenicity. Annu. Rev. Immunol. 1991, 9, 271–296. [Google Scholar] [CrossRef]
- Akdis, M.; Akdis, C.A. Mechanisms of allergen-specific immunotherapy. J. Allergy Clin. Immunol. 2007, 119, 780–791. [Google Scholar] [CrossRef]
- Johnson, J.G.; Jenkins, M.K. Co-stimulatory functions of antigen-presenting cells. J. Invest. Dermatol. 1992, 99, 62–65. [Google Scholar] [CrossRef]
- Jabs, F.; Plum, M.; Laursen, N.S.; Jensen, R.K.; Mølgaard, B.; Miehe, M.; Mandolesi, M.; Rauber, M.M.; Pfützner, W.; Jakob, T.; et al. Trapping IgE in a closed conformation by mimicking CD23 binding prevents and disrupts FcεRI interaction. Nat. Commun. 2018, 9, 7. [Google Scholar] [CrossRef]
- Lucas Moreno, J.M. Diarrhoea due to allergy to egg: Is there a role for specific IgG? Allergol. Immunopathol. 2014, 42, 177–179. [Google Scholar] [CrossRef]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef]
- Akdis, C.A.; Akdis, M. Mechanisms of allergen-specific immunotherapy. J. Allergy Clin. Immunol. 2011, 127, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Kakkar, R.; Lee, R.T. The IL-33/ST2 pathway: Therapeutic target and novel biomarker. Nat. Rev. Drug Discov. 2008, 7, 827–840. [Google Scholar] [CrossRef] [PubMed]
- Liew, F.Y.; Pitman, N.I.; McInnes, I.B. Disease-associated functions of IL-33: The new kid in the IL-1 family. Nat. Rev. Immunol. 2010, 10, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.H.; Meng, Q.; Liu, Z.; Paraschoudi, G. Generation of high-affinity CMV-specific T cells for adoptive immunotherapy using IL-2, IL-15, and IL-21. Clin. Immunol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kandikattu, H.K.; Upparahalli Venkateshaiah, S.; Mishra, A. Synergy of Interleukin (IL)-5 and IL-18 in eosinophil mediated pathogenesis of allergic diseases. Cytokine Growth Factor Rev. 2019, 47, 83–98. [Google Scholar] [CrossRef]
- Valenta, R.; Karaulov, A.; Niederberger, V.; Gattinger, P.; van Hage, M.; Flicker, S.; Linhart, B.; Campana, R.; Focke-Tejkl, M.; Curin, M.; et al. Molecular aspects of allergens and allergy. Adv. Immunol. 2018, 138, 195–256. [Google Scholar] [CrossRef]
- Helm, S.L.; Burton, G.F.; Szakal, A.K.; Tew, J.G. Follicular dendritic cells and the maintenance of IgE responses. Eur. J. Immunol. 1995, 25, 2362–2369. [Google Scholar] [CrossRef]
- Pattarini, L.; Trichot, C.; Bogiatzi, S.; Grandclaudon, M.; Meller, S.; Keuylian, Z.; Durand, M.; Volpe, E.; Madonna, S.; Cavani, A.; et al. TSLP-activated dendritic cells induce human T follicular helper cell differentiation through OX40-ligand. J. Exp. Med. 2017, 214, 1529–1546. [Google Scholar] [CrossRef]
- Ueno, H. Chapter 7—T follicular helper cells as a therapeutic target for autoimmune diseases. In Translational Immunology; Tan, S.L., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 185–204. [Google Scholar] [CrossRef]
- Calzada, D.; Baos, S.; Cremades-Jimeno, L.; Cárdaba, B. Immunological mechanisms in allergic diseases and allergen tolerance: The role of treg cells. J. Immunol. Res. 2018, 2018, 6012053. [Google Scholar] [CrossRef]
- Rodgers, P.M. Food Allergies: Symptoms, Diagnosis, and Treatment; Nova Science Publishers: Hauppauge, NY, USA, 2011. [Google Scholar]
- Eigenmann, P.A. Anaphylactic reactions to raw eggs after negative challenges with cooked eggs. J. Allergy Clin. Immunol. 2000, 105, 587–588. [Google Scholar] [CrossRef]
- Volcheck, G.W.; Skolnik, N.S. Clinical Allergy: Diagnosis and Management; Humana: Totowa, NJ, USA, 2009. [Google Scholar]
- Pasqui, F.; Poli, C.; Colecchia, A.; Marasco, G.; Festi, D. Adverse food reaction and functional gastrointestinal disorders: Role of the dietetic approach. J. Gastrointest. Liver Dis. 2015, 24, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Benedé, S.; Blázquez, A.B.; Chiang, D.; Tordesillas, L.; Berin, M.C. The rise of food allergy: Environmental factors and emerging treatments. EBioMedicine 2016, 7, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Jae Ho, L.; Noh, G.; Noh, J.; Lee, S.; Choi, W.S.; Kim, H.S.; Lee, K.; Choi, S.; Jin, H.; Cho, S.; et al. Clinical characteristics of oral tolerance induction of IgE-mediated and non-IgE-mediated food allergy using interferon gamma. Allergy Asthma Proc. 2010, 31, 39–47. [Google Scholar]
- Perez-Calderon, R.; Gonzalo-Garijo, M.A.; Lamilla-Yerga, A.; Mangas-Santos, R.; Moreno-Gaston, I. Recurrent angioedema due to lysozyme allergy. J. Investig. Allergol. Clin. Immunol. 2007, 17, 264–266. [Google Scholar]
- Pipet, A.; Botturi, K.; Pinot, D.; Vervloet, D.; Magnan, A. Allergen-specific immunotherapy in allergic rhinitis and asthma. Mechanisms and proof of efficacy. Respir. Med. 2009, 103, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Canonica, G.W.; Ansotegui, I.J.; Pawankar, R.; Schmid-Grendelmeier, P.; van Hage, M.; Baena-Cagnani, C.E.; Melioli, G.; Nunes, C.; Passalacqua, G.; Rosenwasser, L.; et al. A WAO—ARIA—GA²LEN consensus document on molecular-based allergy diagnostics. World Allergy Organ. J. 2013, 6, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Moser, M.; Crameri, R.; Brust, E.; Suter, M.; Menz, G. Diagnostic value of recombinant Aspergillus fumigatus allergen I/a for skin testing and serology. J. Allergy Clin. Immunol. 1994, 93, 1–11. [Google Scholar] [CrossRef]
- Valenta, R.; Karaulov, A.; Niederberger, V.; Zhernov, Y.; Elisyutina, O.; Campana, R.; Focke-Tejkl, M.; Curin, M.; Namazova-Baranova, L.; Wang, J.Y.; et al. Allergen extracts for in vivo diagnosis and treatment of allergy: Is there a future? J. Allergy Clin. Immunol. Pract. 2018, 6, 1845–1855. [Google Scholar] [CrossRef]
- Douglass, J.A.; O’Hehir, R.E. 1. Diagnosis, treatment and prevention of allergic disease: The basics. Med. J. Aust. 2006, 185, 228–233. [Google Scholar] [CrossRef]
- Soares-Weiser, K.; Takwoingi, Y.; Panesar, S.S.; Muraro, A.; Werfel, T.; Hoffmann-Sommergruber, K.; Roberts, G.; Halken, S.; Poulsen, L.; van Ree, R.; et al. The diagnosis of food allergy: A systematic review and meta-analysis. Allergy 2014, 69, 76–86. [Google Scholar] [CrossRef]
- Bock, S.A.; Sampson, H.A.; Atkins, F.M.; Zeiger, R.S.; Lehrer, S.; Sachs, M.; Bush, R.K.; Metcalfe, D.D. Double-blind, placebo-controlled food challenge (DBPCFC) as an office procedure: A manual. J. Allergy Clin. Immunol. 1988, 82, 986–997. [Google Scholar] [CrossRef]
- Treudler, R.; Simon, J.C. Overview of component resolved diagnostics. Curr. Allergy Asthma Rep. 2013, 13, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Dodig, S.; Čepelak, I. The potential of component-resolved diagnosis in laboratory diagnostics of allergy. Biochem. Med. 2018, 28, 020501. [Google Scholar] [CrossRef] [PubMed]
- Valenta, R.; Duchene, M.; Vrtala, S.; Birkner, T.; Ebner, C.; Hirschwehr, R.; Breitenbach, M.; Rumpold, H.; Scheiner, O.; Kraft, D. Recombinant allergens for immunoblot diagnosis of tree-pollen allergy. J. Allergy Clin. Immunol. 1991, 88, 889–894. [Google Scholar] [CrossRef]
- Sastre, J. Molecular diagnosis in allergy. Clin. Exp. allergy J. Br. Soc. Allergy Clin. Immunol. 2010, 40, 1442–1460. [Google Scholar] [CrossRef]
- Nowak-Węgrzyn, A.; Sampson, H.A. Future therapies for food allergies. J. Allergy Clin. Immunol. 2011, 127, 558–575. [Google Scholar] [CrossRef]
- Stapel, S.O.; Asero, R.; Ballmer-Weber, B.K.; Knol, E.F.; Strobel, S.; Vieths, S.; Kleine-Tebbe, J. Testing for IgG4 against foods is not recommended as a diagnostic tool: EAACI Task Force Report*. Allergy 2008, 63, 793–796. [Google Scholar] [CrossRef]
- Melioli, G.; Bonifazi, F.; Bonini, S.; Maggi, E.; Mussap, M.; Passalacqua, G.; Rossi, E.R.; Vacca, A.; Canonica, G.W. The ImmunoCAP ISAC molecular allergology approach in adult multi-sensitized Italian patients with respiratory symptoms. Clin. Biochem. 2011, 44, 1005–1011. [Google Scholar] [CrossRef]
- Westman, M.; Lupinek, C.; Bousquet, J.; Andersson, N.; Pahr, S.; Baar, A.; Bergström, A.; Holmström, M.; Stjärne, P.; Lødrup Carlsen, K.C.; et al. Early childhood IgE reactivity to pathogenesis-related class 10 proteins predicts allergic rhinitis in adolescence. J. Allergy Clin. Immunol. 2015, 135, 1199–1206. [Google Scholar] [CrossRef]
- Tripodi, S.; Frediani, T.; Lucarelli, S.; Macri, F.; Pingitore, G.; Di Rienzo Businco, A.; Dondi, A.; Pansa, P.; Ragusa, G.; Asero, R.; et al. Molecular profiles of IgE to Phleum pratense in children with grass pollen allergy: Implications for specific immunotherapy. J. Allergy Clin. Immunol. 2012, 129, 834–839. [Google Scholar] [CrossRef]
- Walsh, J.; O’Flynn, N. Diagnosis and assessment of food allergy in children and young people in primary care and community settings: NICE clinical guideline. Br. J. Gen. Pract.J. R. Coll. Gen. Pract. 2011, 61, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Flores Kim, J.; McCleary, N.; Nwaru, B.I.; Stoddart, A.; Sheikh, A. Diagnostic accuracy, risk assessment, and cost-effectiveness of component-resolved diagnostics for food allergy: A systematic review. Allergy 2018, 73, 1609–1621. [Google Scholar] [CrossRef] [PubMed]
- Calamelli, E.; Liotti, L.; Beghetti, I.; Piccinno, V.; Serra, L.; Bottau, P. Component-Resolved Diagnosis in Food Allergies. Medicina 2019, 55, 498. [Google Scholar] [CrossRef] [PubMed]
- Cianferoni, A.; Muraro, A. Food-induced anaphylaxis. Immunol. Allergy Clin. N. Am. 2012, 32, 165–195. [Google Scholar] [CrossRef]
- Wood, R.A. New horizons in allergen immunotherapy. J. Am. Med Assoc. 2016, 315, 1711–1712. [Google Scholar] [CrossRef]
- Matsui, E.C.; Eggleston, P.A. 23—Immunotherapy for Allergic Disease. In Pediatric Allergy: Principles and Practice, 3rd ed.; Leung, D.Y.M., Szefler, S.J., Bonilla, F.A., Akdis, C.A., Sampson, H.A., Eds.; Elsevier: London, UK, 2016; pp. 203–209. [Google Scholar] [CrossRef]
- Johnstone, D.E. Study of the role of antigen dosage in the treatment of pollenosis and pollen asthma. AMA J. Dis. Child. 1957, 94, 1–5. [Google Scholar] [CrossRef]
- Warner, J.O.; Price, J.F.; Soothill, J.F.; Hey, E.N. Controlled trial of hyposensitisation to Dermatophagoides pteronyssinus in children with asthma. Lancet 1978, 2, 912–915. [Google Scholar] [CrossRef]
- Epstein, T.G.; Liss, G.M.; Murphy-Berendts, K.; Bernstein, D.I. AAAAI/ACAAI surveillance study of subcutaneous immunotherapy, years 20082–012: An update on fatal and nonfatal systemic allergic reactions. J. Allergy Clin. Immunol. Pract. 2014, 2, 161–167. [Google Scholar] [CrossRef]
- Focke, M.; Swoboda, I.; Marth, K.; Valenta, R. Developments in allergen-specific immunotherapy: From allergen extracts to allergy vaccines bypassing allergen-specific immunoglobulin E and T cell reactivity. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2010, 40, 385–397. [Google Scholar] [CrossRef]
- Calderon, M.A.; Alves, B.; Jacobson, M.; Hurwitz, B.; Sheikh, A.; Durham, S. Allergen injection immunotherapy for seasonal allergic rhinitis. Cochrane Database Syst. Rev. 2007, 2007, Cd001936. [Google Scholar] [CrossRef]
- Ismail, I.H.; Tang, M.L. Oral immunotherapy for the treatment of food allergy. Isr. Med. Assoc. J. 2012, 14, 63–69. [Google Scholar] [PubMed]
- Bégin, P.; Chan, E.S.; Kim, H.; Wagner, M.; Cellier, M.S.; Favron-Godbout, C.; Abrams, E.M.; Ben-Shoshan, M.; Cameron, S.B.; Carr, S.; et al. CSACI guidelines for the ethical, evidence-based and patient-oriented clinical practice of oral immunotherapy in IgE-mediated food allergy. Allergy Asthma Clin. Immunol. 2020, 16, 1–45. [Google Scholar] [CrossRef]
- AAAAI. The Current State of Oral Immunotherapy (OIT) for the Treatment of Food Allergy. Available online: https://www.aaaai.org/conditions-and-treatments/library/allergy-library/oit (accessed on 1 April 2020).
- Buchanan, A.D.; Green, T.D.; Jones, S.M.; Scurlock, A.M.; Christie, L.; Althage, K.A.; Steele, P.H.; Pons, L.; Helm, R.M.; Lee, L.A.; et al. Egg oral immunotherapy in nonanaphylactic children with egg allergy. J. Allergy Clin. Immunol. 2007, 119, 199–205. [Google Scholar] [CrossRef]
- Vickery, B.P.; Pons, L.; Kulis, M.; Steele, P.; Jones, S.M.; Burks, A.W. Individualized IgE-based dosing of egg oral immunotherapy and the development of tolerance. Ann. Allergy Asthma Immunol. 2010, 105, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.L.K.; Martino, D.J. Oral immunotherapy and tolerance induction in childhood. Pediatric Allergy Immunol. 2013, 24, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.L.; Ponsonby, A.L.; Orsini, F.; Tey, D.; Robinson, M.; Su, E.L.; Licciardi, P.; Burks, W.; Donath, S. Administration of a probiotic with peanut oral immunotherapy: A randomized trial. J. Allergy Clin. Immunol. 2015, 135, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Caminiti, L.; Pajno, G.B.; Crisafulli, G.; Chiera, F.; Collura, M.; Panasci, G.; Ruggeri, P.; Guglielmo, F.; Passalacqua, G. Oral immunotherapy for egg allergy: A double-blind placebo-controlled study, with postdesensitization follow-up. J. Allergy Clin. Immunol. Pract. 2015, 3, 532–539. [Google Scholar] [CrossRef]
- Tang, M.L.K.; Mullins, R.J. Food allergy: Is prevalence increasing? Intern. Med. J. 2017, 47, 256–261. [Google Scholar] [CrossRef]
- Matricardi, P.M.; Kleine-Tebbe, J.; Hoffmann, H.J.; Valenta, R.; Hilger, C.; Hofmaier, S.; Aalberse, R.C.; Agache, I.; Asero, R.; Ballmer-Weber, B.; et al. EAACI molecular allergology user’s guide. Pediatric Allergy Immunol. 2016, 27, 1–250. [Google Scholar] [CrossRef]
- Curin, M.; Khaitov, M.; Karaulov, A.; Namazova-Baranova, L.; Campana, R.; Garib, V.; Valenta, R. Next-generation of allergen-specific immunotherapies: Molecular approaches. Curr. Allergy Asthma Rep. 2018, 18, 39. [Google Scholar] [CrossRef]
- Tscheppe, A.; Breiteneder, H. Recombinant allergens in structural biology, diagnosis, and immunotherapy. Int. Arch. Allergy Immunol. 2017, 172, 187–202. [Google Scholar] [CrossRef]
- Dhanapala, P.; Withanage-Dona, D.; Tang, M.L.; Doran, T.; Suphioglu, C. Hypoallergenic variant of the major egg white allergen Gal d 1 produced by disruption of cysteine bridges. Nutrients 2017, 9, 171. [Google Scholar] [CrossRef] [PubMed]
- Vrtala, S.; Hirtenlehner, K.; Vangelista, L.; Pastore, A.; Eichler, H.G.; Sperr, W.R.; Valent, P.; Ebner, C.; Kraft, D.; Valenta, R. Division of the major birch pollen allergen, Bet v 1, into two non-anaphylactic fragments. Int. Arch. Allergy Immunol. 1997, 113, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Vrtala, S.; Hirtenlehner, K.; Vangelista, L.; Pastore, A.; Eichler, H.G.; Sperr, W.R.; Valent, P.; Ebner, C.; Kraft, D.; Valenta, R. Conversion of the major birch pollen allergen, Bet v 1, into two nonanaphylactic T cell epitope-containing fragments: Candidates for a novel form of specific immunotherapy. J. Clin. Investig. 1997, 99, 1673–1681. [Google Scholar] [CrossRef]
- Zeiler, T.; Taivainen, A.; Rytkonen, M.; Rautiainen, J.; Karjalainen, H.; Mantyjarvi, R.; Tuomisto, L.; Virtanen, T. Recombinant allergen fragments as candidate preparations for allergen immunotherapy. J. Allergy Clin. Immunol. 1997, 100, 721–727. [Google Scholar] [CrossRef]
- Elfman, L.H.; Whitley, P.; Schmidt, M.; van Hage-Hamsten, M. IgE binding capacity of synthetic and recombinant peptides of the major storage mite (Lepidoglyphus destructor) allergen, Lep d 2. Int. Arch. Allergy Immunol. 1998, 117, 167–173. [Google Scholar] [CrossRef]
- Ball, T.; Vrtala, S.; Sperr, W.R.; Valent, P.; Susani, M.; Kraft, D.; Valenta, R. Isolation of an immunodominant IgE hapten from an epitope expression cDNA library. Dissection of the allergic effector reaction. J. Biol. Chem. 1994, 269, 28323–28328. [Google Scholar] [PubMed]
- Chen, K.W.; Fuchs, G.; Sonneck, K.; Gieras, A.; Swoboda, I.; Douladiris, N.; Linhart, B.; Jankovic, M.; Pavkov, T.; Keller, W.; et al. Reduction of the in vivo allergenicity of Der p 2, the major house-dust mite allergen, by genetic engineering. Mol. Immunol. 2008, 45, 2486–2498. [Google Scholar] [CrossRef] [PubMed]
- Vrtala, S.; Hirtenlehner, K.; Susani, M.; Akdis, M.; Kussebi, F.; Akdis, C.A.; Blaser, K.; Hufnagl, P.; Binder, B.R.; Politou, A.; et al. Genetic engineering of a hypoallergenic trimer of the major birch pollen allergen Bet v 1. FASEB J. 2001, 15, 2045–2047. [Google Scholar] [CrossRef]
- Campana, R.; Vrtala, S.; Maderegger, B.; Jertschin, P.; Stegfellner, G.; Swoboda, I.; Focke-Tejkl, M.; Blatt, K.; Gieras, A.; Zafred, D.; et al. Hypoallergenic derivatives of the major birch pollen allergen Bet v 1 obtained by rational sequence reassembly. J. Allergy Clin. Immunol. 2010, 126, 1024–1031. [Google Scholar] [CrossRef]
- Ball, T.; Linhart, B.; Sonneck, K.; Blatt, K.; Herrmann, H.; Valent, P.; Stoecklinger, A.; Lupinek, C.; Thalhamer, J.; Fedorov, A.A.; et al. Reducing allergenicity by altering allergen fold: A mosaic protein of Phl p 1 for allergy vaccination. Allergy 2009, 64, 569–580. [Google Scholar] [CrossRef]
- Mothes-Luksch, N.; Stumvoll, S.; Linhart, B.; Focke, M.; Krauth, M.T.; Hauswirth, A.; Valent, P.; Verdino, P.; Pavkov, T.; Keller, W.; et al. Disruption of allergenic activity of the major grass pollen allergen Phl p 2 by reassembly as a mosaic protein. J. Immunol. 2008, 181, 4864–4873. [Google Scholar] [CrossRef] [PubMed]
- Curin, M.; Weber, M.; Thalhamer, T.; Swoboda, I.; Focke-Tejkl, M.; Blatt, K.; Valent, P.; Marth, K.; Garmatiuk, T.; Gronlund, H.; et al. Hypoallergenic derivatives of Fel d 1 obtained by rational reassembly for allergy vaccination and tolerance induction. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2014, 44, 882–894. [Google Scholar] [CrossRef]
- Asturias, J.A.; Ibarrola, I.; Arilla, M.C.; Vidal, C.; Ferrer, A.; Gamboa, P.M.; Vinuela, J.E.; Sanz, M.L.; Andreu, C.; Martinez, A. Engineering of major house dust mite allergens Der p 1 and Der p 2 for allergen-specific immunotherapy. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2009, 39, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Blatt, K.; Thomas, W.R.; Swoboda, I.; Valent, P.; Valenta, R.; Vrtala, S. Hypoallergenic Der p 1/Der p 2 combination vaccines for immunotherapy of house dust mite allergy. J. Allergy Clin. Immunol. 2012, 130, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Linhart, B.; Focke-Tejkl, M.; Weber, M.; Narayanan, M.; Neubauer, A.; Mayrhofer, H.; Blatt, K.; Lupinek, C.; Valent, P.; Valenta, R. Molecular evolution of hypoallergenic hybrid proteins for vaccination against grass pollen allergy. J. Immunol. 2015, 194, 4008–4018. [Google Scholar] [CrossRef] [PubMed]
- Linhart, B.; Mothes-Luksch, N.; Vrtala, S.; Kneidinger, M.; Valent, P.; Valenta, R. A hypoallergenic hybrid molecule with increased immunogenicity consisting of derivatives of the major grass pollen allergens, Phl p 2 and Phl p 6. Biol. Chem. 2008, 389, 925–933. [Google Scholar] [CrossRef] [PubMed]
- King, T.P.; Jim, S.Y.; Monsalve, R.I.; Kagey-Sobotka, A.; Lichtenstein, L.M.; Spangfort, M.D. Recombinant allergens with reduced allergenicity but retaining immunogenicity of the natural allergens: Hybrids of yellow jacket and paper wasp venom allergen antigen 5s. J. Immunol. 2001, 166, 6057–6065. [Google Scholar] [CrossRef]
- Karamloo, F.; Schmid-Grendelmeier, P.; Kussebi, F.; Akdis, M.; Salagianni, M.; von Beust, B.R.; Reimers, A.; Zumkehr, J.; Soldatova, L.; Housley-Markovic, Z.; et al. Prevention of allergy by a recombinant multi-allergen vaccine with reduced IgE binding and preserved T cell epitopes. Eur. J. Immunol. 2005, 35, 3268–3276. [Google Scholar] [CrossRef]
- Fujimura, T.; Fujinami, K.; Ishikawa, R.; Tateno, M.; Tahara, Y.; Okumura, Y.; Ohta, H.; Miyazaki, H.; Taniguchi, M. Recombinant fusion allergens, Cry j 1 and Cry j 2 from Japanese cedar pollen, conjugated with polyethylene glycol potentiate the attenuation of Cry j 1-Specific IgE production in Cry j 1-Sensitized mice and Japanese cedar pollen allergen-sensitized monkeys. Int. Arch. Allergy Immunol. 2015, 168, 32–43. [Google Scholar] [CrossRef]
- Ferreira, F.; Ebner, C.; Kramer, B.; Casari, G.; Briza, P.; Kungl, A.J.; Grimm, R.; Jahn-Schmid, B.; Breiteneder, H.; Kraft, D.; et al. Modulation of IgE reactivity of allergens by site-directed mutagenesis: Potential use of hypoallergenic variants for immunotherapy. FASEB J. 1998, 12, 231–242. [Google Scholar] [CrossRef]
- Wallner, M.; Hauser, M.; Himly, M.; Zaborsky, N.; Mutschlechner, S.; Harrer, A.; Asam, C.; Pichler, U.; van Ree, R.; Briza, P.; et al. Reshaping the Bet v 1 fold modulates T(H) polarization. J. Allergy Clin. Immunol. 2011, 127, 1571–1578. [Google Scholar] [CrossRef] [PubMed]
- Engel, E.; Richter, K.; Obermeyer, G.; Briza, P.; Kungl, A.J.; Simon, B.; Auer, M.; Ebner, C.; Rheinberger, H.J.; Breitenbach, M.; et al. Immunological and biological properties of Bet v 4, a novel birch pollen allergen with two EF-hand calcium-binding domains. J. Biol. Chem. 1997, 272, 28630–28637. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Swoboda, I.; Bhalla, P.L.; Toriyama, K.; Singh, M.B. Engineering of hypoallergenic mutants of the Brassica pollen allergen, Bra r 1, for immunotherapy. FEBS Lett. 1998, 434, 255–260. [Google Scholar] [CrossRef]
- Swoboda, I.; Bugajska-Schretter, A.; Linhart, B.; Verdino, P.; Keller, W.; Schulmeister, U.; Sperr, W.R.; Valent, P.; Peltre, G.; Quirce, S.; et al. A recombinant hypoallergenic parvalbumin mutant for immunotherapy of IgE-mediated fish allergy. J. Immunol. 2007, 178, 6290–6296. [Google Scholar] [CrossRef] [PubMed]
- Bonura, A.; Amoroso, S.; Locorotondo, G.; Di Felice, G.; Tinghino, R.; Geraci, D.; Colombo, P. Hypoallergenic variants of the Parietaria judaica major allergen Par j 1: A member of the non-specific lipid transfer protein plant family. Int. Arch. Allergy Immunol. 2001, 126, 32–40. [Google Scholar] [CrossRef]
- Mine, Y.; Sasaki, E.; Zhang, J.W. Reduction of antigenicity and allergenicity of genetically modified egg white allergen, ovomucoid third domain. Biochem. Biophys. Res. Commun. 2003, 302, 133–137. [Google Scholar] [CrossRef]
- Drew, A.C.; Eusebius, N.P.; Kenins, L.; de Silva, H.D.; Suphioglu, C.; Rolland, J.M.; O’Hehir R, E. Hypoallergenic variants of the major latex allergen Hev b 6.01 retaining human T lymphocyte reactivity. J. Immunol. 2004, 173, 5872–5879. [Google Scholar] [CrossRef]
- Karisola, P.; Mikkola, J.; Kalkkinen, N.; Airenne, K.J.; Laitinen, O.H.; Repo, S.; Pentikainen, O.T.; Reunala, T.; Turjanmaa, K.; Johnson, M.S.; et al. Construction of hevein (Hev b 6.02) with reduced allergenicity for immunotherapy of latex allergy by comutation of six amino acid residues on the conformational IgE epitopes. J. Immunol. 2004, 172, 2621–2628. [Google Scholar] [CrossRef]
- Beezhold, D.H.; Hickey, V.L.; Sussman, G.L. Mutational analysis of the IgE epitopes in the latex allergen Hev b 5. J. Allergy Clin. Immunol. 2001, 107, 1069–1076. [Google Scholar] [CrossRef]
- Smith, A.M.; Chapman, M.D. Reduction of IgE antibody binding to rDer p 2 variants generated by site-directed mutagenesis. Adv. Exp. Med. Biol. 1996, 409, 391–394. [Google Scholar] [CrossRef] [PubMed]
- Bannon, G.A.; Cockrell, G.; Connaughton, C.; West, C.M.; Helm, R.; Stanley, J.S.; King, N.; Rabjohn, P.; Sampson, H.A.; Burks, A.W. Engineering, characterization and in vitro efficacy of the major peanut allergens for use in immunotherapy. Int. Arch. Allergy Immunol. 2001, 124, 70–72. [Google Scholar] [CrossRef] [PubMed]
- King, N.; Helm, R.; Stanley, J.S.; Vieths, S.; Luttkopf, D.; Hatahet, L.; Sampson, H.; Pons, L.; Burks, W.; Bannon, G.A. Allergenic characteristics of a modified peanut allergen. Mol. Nutr. Food Res. 2005, 49, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Rabjohn, P.; West, C.M.; Connaughton, C.; Sampson, H.A.; Helm, R.M.; Burks, A.W.; Bannon, G.A. Modification of peanut allergen Ara h 3: Effects on IgE binding and T cell stimulation. Int. Arch. Allergy Immunol. 2002, 128, 15–23. [Google Scholar] [CrossRef]
- Swoboda, I.; De Weerd, N.; Bhalla, P.L.; Niederberger, V.; Sperr, W.R.; Valent, P.; Kahlert, H.; Fiebig, H.; Verdino, P.; Keller, W.; et al. Mutants of the major ryegrass pollen allergen, Lol p 5, with reduced IgE-binding capacity: Candidates for grass pollen-specific immunotherapy. Eur. J. Immunol. 2002, 32, 270–280. [Google Scholar] [CrossRef]
- McGowan, E.C.; Saini, S. Update on the performance and application of basophil activation tests. Curr. Allergy Asthma Rep. 2013, 13, 101–109. [Google Scholar] [CrossRef]
- Dhanapala, P.; Doran, T.; Tang, M.L.; Suphioglu, C. Production and immunological analysis of IgE reactive recombinant egg white allergens expressed in Escherichia coli. Mol. Immunol. 2015, 65, 104–112. [Google Scholar] [CrossRef]
- Valenta, R.; Ferreira, F.; Focke-Tejkl, M.; Linhart, B.; Niederberger, V.; Swoboda, I.; Vrtala, S. From allergen genes to allergy vaccines. Annu. Rev. Immunol. 2010, 28, 211–241. [Google Scholar] [CrossRef]
- UniProt. UniProtKB—P00698 (LYSC_CHICK). Available online: https://www.uniprot.org/uniprot/P00698 (accessed on 25 April 2020).
- Matsuo, H.; Yokooji, T.; Taogoshi, T. Common food allergens and their IgE-binding epitopes. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2015, 64, 332–343. [Google Scholar] [CrossRef]
- Vrtala, S.; Akdis, C.A.; Budak, F.; Akdis, M.; Blaser, K.; Kraft, D.; Valenta, R. T cell epitope-containing hypoallergenic recombinant fragments of the major birch pollen allergen, Bet v 1, induce blocking antibodies. J. Immunol. 2000, 165, 6653–6659. [Google Scholar] [CrossRef]
- Linhart, B.; Valenta, R. Mechanisms underlying allergy vaccination with recombinant hypoallergenic allergen derivatives. Vaccine 2012, 30, 4328–4335. [Google Scholar] [CrossRef] [PubMed]
- Valenta, R.; Kraft, D. Recombinant allergen molecules: Tools to study effector cell activation. Immunol. Rev. 2001, 179, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Chatchatee, P.; Jarvinen, K.M.; Bardina, L.; Beyer, K.; Sampson, H.A. Identification of IgE- and IgG-binding epitopes on alpha(s1)-casein: Differences in patients with persistent and transient cow’s milk allergy. J. Allergy Clin. Immunol. 2001, 107, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Haeffner-Gormley, L.; Parente, L.; Wetlaufer, D.B. Use of proline-specific endopeptidase in the isolation of all four “native” disulfides of hen egg white lysozyme. Int. J. Pept. Protein Res. 1985, 26, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Kahlert, H.; Cromwell, O.; Fiebig, H. Measurement of basophil-activating capacity of grass pollen allergens, allergoids and hypoallergenic recombinant derivatives by flow cytometry using anti-CD203c. Clin. Exp. Allergy 2003, 33, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Marth, K.; Focke-Tejkl, M.; Lupinek, C.; Valenta, R.; Niederberger, V. Allergen peptides, recombinant allergens and hypoallergens for allergen-specific immunotherapy. Curr. Treat. Options Allergy 2014, 1, 91–106. [Google Scholar] [CrossRef]
- Jutel, M.; Jaeger, L.; Suck, R.; Meyer, H.; Fiebig, H.; Cromwell, O. Allergen-specific immunotherapy with recombinant grass pollen allergens. J. Allergy Clin. Immunol. 2005, 116, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Pauli, G.; Larsen, T.H.; Rak, S.; Horak, F.; Pastorello, E.; Valenta, R.; Purohit, A.; Arvidsson, M.; Kavina, A.; Schroeder, J.W.; et al. Efficacy of recombinant birch pollen vaccine for the treatment of birch-allergic rhinoconjunctivitis. J. Allergy Clin. Immunol 2008, 122, 951–960. [Google Scholar] [CrossRef]
- Tourdot, S.; Airouche, S.; Berjont, N.; Moussu, H.; Betbeder, D.; Nony, E.; Bordas-Le Floch, V.; Baron-Bodo, V.; Mascarell, L.; Moingeon, P. Efficacy of sublingual vectorized recombinant Bet v 1a in a mouse model of birch pollen allergic asthma. Vaccine 2013, 31, 2628–2637. [Google Scholar] [CrossRef]
- Jutel, M.; Solarewicz-Madejek, K.; Smolinska, S. Recombinant allergens: The present and the future. Hum. Vaccines Immunother. 2012, 8, 1534–1543. [Google Scholar] [CrossRef][Green Version]
- Winther, L.; Poulsen, L.K.; Robin, B.; Mélac, M.; Malling, H. Safety and tolerability of recombinant Bet v 1 (rBet v 1) Tablets in Sublingual Immunotherapy (SLIT). J. Allergy Clin. Immunol. 2009, 123, S215. [Google Scholar] [CrossRef]
- Purohit, A.; Niederberger, V.; Kronqvist, M.; Horak, F.; Gronneberg, R.; Suck, R.; Weber, B.; Fiebig, H.; van Hage, M.; Pauli, G.; et al. Clinical effects of immunotherapy with genetically modified recombinant birch pollen Bet v 1 derivatives. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2008, 38, 1514–1525. [Google Scholar] [CrossRef] [PubMed]
- Niederberger, V.; Horak, F.; Vrtala, S.; Spitzauer, S.; Krauth, M.T.; Valent, P.; Reisinger, J.; Pelzmann, M.; Hayek, B.; Kronqvist, M.; et al. Vaccination with genetically engineered allergens prevents progression of allergic disease. Proc. Natl. Acad. Sci. USA 2004, 101 (Suppl. 2), 14677–14682. [Google Scholar] [CrossRef]
- Reisinger, J.; Horak, F.; Pauli, G.; van Hage, M.; Cromwell, O.; Konig, F.; Valenta, R.; Niederberger, V. Allergen-specific nasal IgG antibodies induced by vaccination with genetically modified allergens are associated with reduced nasal allergen sensitivity. J. Allergy Clin. Immunol. 2005, 116, 347–354. [Google Scholar] [CrossRef]
- Pree, I.; Reisinger, J.; Focke, M.; Vrtala, S.; Pauli, G.; van Hage, M.; Cromwell, O.; Gadermaier, E.; Egger, C.; Reider, N.; et al. Analysis of epitope-specific immune responses induced by vaccination with structurally folded and unfolded recombinant Bet v 1 allergen derivatives in man. J. Immunol. 2007, 179, 5309–5316. [Google Scholar] [CrossRef]
- Meyer, W.; Narkus, A.; Salapatek, A.M.; Hafner, D. Double-blind, placebo-controlled, dose-ranging study of new recombinant hypoallergenic Bet v 1 in an environmental exposure chamber. Allergy 2013, 68, 724–731. [Google Scholar] [CrossRef]
- Nilsson, O.B.; Adedoyin, J.; Rhyner, C.; Neimert-Andersson, T.; Grundstrom, J.; Berndt, K.D.; Crameri, R.; Gronlund, H. In vitro evolution of allergy vaccine candidates, with maintained structure, but reduced B cell and T cell activation capacity. PLoS ONE 2011, 6, e24558. [Google Scholar] [CrossRef]
- Suzuki, K.; Kaminuma, O.; Yang, L.; Takai, T.; Mori, A.; Umezu-Goto, M.; Ohtomo, T.; Ohmachi, Y.; Noda, Y.; Hirose, S.; et al. Prevention of allergic asthma by vaccination with transgenic rice seed expressing mite allergen: Induction of allergen-specific oral tolerance without bystander suppression. Plant Biotechnol. J. 2011, 9, 982–990. [Google Scholar] [CrossRef]
- Takagi, H.; Hiroi, T.; Yang, L.; Tada, Y.; Yuki, Y.; Takamura, K.; Ishimitsu, R.; Kawauchi, H.; Kiyono, H.; Takaiwa, F. A rice-based edible vaccine expressing multiple T cell epitopes induces oral tolerance for inhibition of Th2-mediated IgE responses. Proc. Natl. Acad. Sci. USA 2005, 102, 17525–17530. [Google Scholar] [CrossRef]
- Hiroi, T.; Kaminuma, O.; Takaiwa, F. Vaccination with transgenic rice seed expressing mite allergen: A new option for asthma sufferers? Expert. Rev. Vaccines 2011, 10, 1249–1251. [Google Scholar] [CrossRef]
- Yang, L.; Hirose, S.; Suzuki, K.; Hiroi, T.; Takaiwa, F. Expression of hypoallergenic Der f 2 derivatives with altered intramolecular disulphide bonds induces the formation of novel ER-derived protein bodies in transgenic rice seeds. J. Exp. Bot. 2012, 63, 2947–2959. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Takahashi, H.; Kajiura, H.; Kawakatsu, T.; Fujiyama, K.; Takaiwa, F. Transgenic rice seeds accumulating recombinant hypoallergenic birch pollen allergen Bet v 1 generate giant protein bodies. Plant Cell Physiol. 2013, 54, 917–933. [Google Scholar] [CrossRef] [PubMed]
- Senti, G.; Crameri, R.; Kuster, D.; Johansen, P.; Martinez-Gomez, J.M.; Graf, N.; Steiner, M.; Hothorn, L.A.; Gronlund, H.; Tivig, C.; et al. Intralymphatic immunotherapy for cat allergy induces tolerance after only 3 injections. J. Allergy Clin. Immunol. 2012, 129, 1290–1296. [Google Scholar] [CrossRef]
- Wood, R.A.; Sicherer, S.H.; Burks, A.W.; Grishin, A.; Henning, A.K.; Lindblad, R.; Stablein, D.; Sampson, H.A. A phase 1 study of heat/phenol-killed, E. coli-encapsulated, recombinant modified peanut proteins Ara h 1, Ara h 2, and Ara h 3 (EMP-123) for the treatment of peanut allergy. Allergy 2013, 68, 803–808. [Google Scholar] [CrossRef]
- Gafvelin, G.; Thunberg, S.; Kronqvist, M.; Gronlund, H.; Gronneberg, R.; Troye-Blomberg, M.; Akdis, M.; Fiebig, H.; Purohit, A.; Horak, F.; et al. Cytokine and antibody responses in birch-pollen-allergic patients treated with genetically modified derivatives of the major birch pollen allergen Bet v 1. Int. Arch. Allergy Immunol. 2005, 138, 59–66. [Google Scholar] [CrossRef]
- Haselden, B.M.; Kay, A.B.; Larche, M. Immunoglobulin E-independent major histocompatibility complex-restricted T cell peptide epitope-induced late asthmatic reactions. J. Exp. Med. 1999, 189, 1885–1894. [Google Scholar] [CrossRef]
- Norman, P.S.; Ohman, J.L.J.; Long, A.A.; Creticos, P.S.; Gefter, M.A.; Shaked, Z.; Wood, R.A.; Eggleston, P.A.; Hafner, K.B.; Rao, P.; et al. Treatment of cat allergy with T-cell reactive peptides. Am. J. Respir. Crit. Care Med. 1996, 154, 1623–1628. [Google Scholar] [CrossRef]
- Worm, M.; Lee, H.H.; Kleine-Tebbe, J.; Hafner, R.P.; Laidler, P.; Healey, D.; Buhot, C.; Verhoef, A.; Maillere, B.; Kay, A.B.; et al. Development and preliminary clinical evaluation of a peptide immunotherapy vaccine for cat allergy. J. Allergy Clin. Immunol. 2011, 127, 89–97. [Google Scholar] [CrossRef]
- Patel, D.; Couroux, P.; Hickey, P.; Salapatek, A.M.; Laidler, P.; Larche, M.; Hafner, R.P. Fel d 1-derived peptide antigen desensitization shows a persistent treatment effect 1 year after the start of dosing: A randomized, placebo-controlled study. J. Allergy Clin. Immunol. 2013, 131, 103–109. [Google Scholar] [CrossRef]
- Muller, U.; Akdis, C.A.; Fricker, M.; Akdis, M.; Blesken, T.; Bettens, F.; Blaser, K. Successful immunotherapy with T-cell epitope peptides of bee venom phospholipase A2 induces specific T-cell anergy in patients allergic to bee venom. J. Allergy Clin. Immunol. 1998, 101, 747–754. [Google Scholar] [CrossRef]
- Fellrath, J.M.; Kettner, A.; Dufour, N.; Frigerio, C.; Schneeberger, D.; Leimgruber, A.; Corradin, G.; Spertini, F. Allergen-specific T-cell tolerance induction with allergen-derived long synthetic peptides: Results of a phase I trial. J. Allergy Clin. Immunol. 2003, 111, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Oldfield, W.L.; Larche, M.; Kay, A.B. Effect of T-cell peptides derived from Fel d 1 on allergic reactions and cytokine production in patients sensitive to cats: A randomised controlled trial. Lancet 2002, 360, 47–53. [Google Scholar] [CrossRef]
- Focke, M.; Mahler, V.; Ball, T.; Sperr, W.R.; Majlesi, Y.; Valent, P.; Kraft, D.; Valenta, R. Nonanaphylactic synthetic peptides derived from B cell epitopes of the major grass pollen allergen, Phl p 1, for allergy vaccination. FASEB J. 2001, 15, 2042–2044. [Google Scholar] [CrossRef] [PubMed]
- Focke, M.; Linhart, B.; Hartl, A.; Wiedermann, U.; Sperr, W.R.; Valent, P.; Thalhamer, J.; Kraft, D.; Valenta, R. Non-anaphylactic surface-exposed peptides of the major birch pollen allergen, Bet v 1, for preventive vaccination. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2004, 34, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Twaroch, T.E.; Focke, M.; Fleischmann, K.; Balic, N.; Lupinek, C.; Blatt, K.; Ferrara, R.; Mari, A.; Ebner, C.; Valent, P.; et al. Carrier-bound Alt a 1 peptides without allergenic activity for vaccination against Alternaria alternata allergy. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2012, 42, 966–975. [Google Scholar] [CrossRef]
- Twaroch, T.E.; Focke, M.; Civaj, V.; Weber, M.; Balic, N.; Mari, A.; Ferrara, R.; Quirce, S.; Spitzauer, S.; Swoboda, I.; et al. Carrier-bound, nonallergenic Ole e 1 peptides for vaccination against olive pollen allergy. J. Allergy Clin. Immunol. 2011, 128, 1781–1784. [Google Scholar] [CrossRef]
- Edlmayr, J.; Niespodziana, K.; Linhart, B.; Focke-Tejkl, M.; Westritschnig, K.; Scheiblhofer, S.; Stoecklinger, A.; Kneidinger, M.; Valent, P.; Campana, R.; et al. A combination vaccine for allergy and rhinovirus infections based on rhinovirus-derived surface protein VP1 and a nonallergenic peptide of the major timothy grass pollen allergen Phl p 1. J. Immunol. 2009, 182, 6298–6306. [Google Scholar] [CrossRef]
- Marth, K.; Breyer, I.; Focke-Tejkl, M.; Blatt, K.; Shamji, M.H.; Layhadi, J.; Gieras, A.; Swoboda, I.; Zafred, D.; Keller, W.; et al. A nonallergenic birch pollen allergy vaccine consisting of hepatitis PreS-fused Bet v 1 peptides focuses blocking IgG toward IgE epitopes and shifts immune responses to a tolerogenic and Th1 phenotype. J. Immunol. 2013, 190, 3068–3078. [Google Scholar] [CrossRef]
- Niespodziana, K.; Focke-Tejkl, M.; Linhart, B.; Civaj, V.; Blatt, K.; Valent, P.; van Hage, M.; Gronlund, H.; Valenta, R. A hypoallergenic cat vaccine based on Fel d 1-derived peptides fused to hepatitis B PreS. J. Allergy Clin. Immunol. 2011, 127, 1562–1570. [Google Scholar] [CrossRef]
- Edlmayr, J.; Niespodziana, K.; Popow-Kraupp, T.; Krzyzanek, V.; Focke-Tejkl, M.; Blaas, D.; Grote, M.; Valenta, R. Antibodies induced with recombinant VP1 from human rhinovirus exhibit cross-neutralisation. Eur. Respir. J. 2011, 37, 44–52. [Google Scholar] [CrossRef]
- Keet, C.A.; Wood, R.A. Emerging therapies for food allergy. J. Clin. Investig. 2014, 124, 1880–1886. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Valenta, R.; Ball, T.; Focke, M.; Linhart, B.; Mothes, N.; Niederberger, V.; Spitzauer, S.; Swoboda, I.; Vrtala, S.; Westritschnig, K.; et al. Immunotherapy of Allergic Disease. In Advances in Immunology; Academic Press: Cambridge, MA, USA, 2004; Volume 82, pp. 1051–1053. [Google Scholar]
- Attallah, N.A.; Sehon, A.H. Isolation of haptenic material from ragweed pollen. Immunochemistry 1969, 6, 609–619. [Google Scholar] [CrossRef]
- Gieni, R.S.; Hayglass, K.T. Regulation of murine IgE responses: Induction of long-lived inhibition of allergen-specific responses is genetically restricted. Cell. Immunol. 1991, 138, 64–78. [Google Scholar] [CrossRef]
- Valenta, R.; Niederberger, V. Recombinant allergens for immunotherapy. J. Allergy Clin. Immunol. 2007, 119, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Larché, M. Update on the current status of peptide immunotherapy. J. Allergy Clin. Immunol. 2007, 119, 906–909. [Google Scholar] [CrossRef]
- Oldfield, W.L.G.; Kay, A.B.; Larché, M. Allergen-derived T cell peptide-induced late asthmatic reactions precede the induction of antigen-specific hyporesponsiveness in atopic allergic asthmatic subjects. J. Immunol. 2001, 167, 1734–1739. [Google Scholar] [CrossRef]
- Reefer, A.J.; Carneiro, R.M.; Custis, N.J.; Platts-Mills, T.A.E.; Sung, S.S.J.; Hammer, J.; Woodfolk, J.A. A role for IL-10-mediated HLA-DR7-restricted T cell-dependent events in development of the modified Th2 response to cat allergen. J. Immunol. 2004, 172, 2763–2772. [Google Scholar] [CrossRef]
- Briner, T.J.; Kuo, M.C.; Keating, K.M.; Rogers, B.L.; Greenstein, J.L. Peripheral T-cell tolerance induced in naive and primed mice by subcutaneous injection of peptides from the major cat allergen Fel d I. Proc. Natl. Acad. Sci. USA 1993, 90, 7608–7612. [Google Scholar] [CrossRef]
- Riemer, A.; Scheiner, O.; Jensen-Jarolim, E. Allergen mimotopes. Methods 2004, 32, 321–327. [Google Scholar] [CrossRef]
- Ganglberger, E.; Grünberger, K.; Wiedermann, U.; Vermes, M.; Sponer, B.; Breiteneder, H.; Scheiner, O.; Boltz, G.; Jensen-Jarolim, E. IgE mimotopes of birch pollen allergen Bet v 1 induce blocking IgG in mice. Int. Arch. Allergy Immunol. 2001, 124, 395–397. [Google Scholar] [CrossRef]
- Szalai, K.; Fuhrmann, J.; Pavkov, T.; Scheidl, M.; Wallmann, J.; Brämswig, K.H.; Vrtala, S.; Scheiner, O.; Keller, W.; Saint-Remy, J.M.; et al. Mimotopes identify conformational B-cell epitopes on the two major house dust mite allergens Der p 1 and Der p 2. Mol. Immunol. 2008, 45, 1308–1317. [Google Scholar] [CrossRef] [PubMed]
- Larché, M.; Akdis, C.A.; Valenta, R. Immunological mechanisms of allergen-specific immunotherapy. Nat. Rev. Immunol. 2006, 6, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Slater, J.E.; Paupore, E.; Zhang, Y.T.; Colberg-Poley, A.M. The latex allergen Hev b 5 transcript is widely distributed after subcutaneous injection in BALB/c mice of its DNA vaccine. J. Allergy Clin. Immunol. 1998, 102, 469–475. [Google Scholar] [CrossRef]
- Jones, S.M.; Burks, A.W.; Dupont, C. State of the art on food allergen immunotherapy: Oral, sublingual, and epicutaneous. J. Allergy Clin. Immunol. 2014, 133, 318–323. [Google Scholar] [CrossRef]
- Fanta, C.; Bohle, B.; Hirt, W.; Siemann, U.; Horak, F.; Kraft, D.; Ebner, H.; Ebner, C. Systemic immunological changes induced by administration of grass pollen allergens via the oral mucosa during sublingual immunotherapy. Int. Arch. Allergy Immunol. 1999, 120, 218–224. [Google Scholar] [CrossRef]
- Akdis, C.A.; Barlan, I.B.; Bahceciler, N.; Akdis, M. Immunological mechanisms of sublingual immunotherapy. Allergy 2006, 61, 11–14. [Google Scholar] [CrossRef]
- Brożek, J.L.; Bousquet, J.; Baena-Cagnani, C.E.; Bonini, S.; Canonica, G.W.; Casale, T.B.; van Wijk, R.G.; Ohta, K.; Zuberbier, T.; Schünemann, H.J. Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines: 2010 Revision. J. Allergy Clin. Immunol. 2010, 126, 466–476. [Google Scholar] [CrossRef]
- Andri, L.; Senna, G.; Betteli, C.; Givanni, S.; Andri, G.; Falagiani, P. Local nasal immunotherapy for Dermatophagoides-induced rhinitis: Efficacy of a powder extract. J. Allergy Clin. Immunol. 1993, 91, 987–996. [Google Scholar] [CrossRef]
- Mistrello, G.; Brenna, O.; Roncarolo, D.; Zanoni, D.; Gentili, M.; Falagiani, P. Monomeric chemically modified allergens: Immunologic and Physicochemical characterigation. Allergy 1996, 51, 8–15. [Google Scholar] [CrossRef]
- Ngan, J.; Kind, L.S. Suppressor T cells for IgE and IgG in Peyer’s patches of mice made tolerant by the oral administration of ovalbumin. J. Immunol. 1978, 120, 861–865. [Google Scholar]
- Slütter, B.; Jiskoot, W. Dual role of CpG as immune modulator and physical crosslinker in ovalbumin loaded N-trimethyl chitosan (TMC) nanoparticles for nasal vaccination. J. Control. Release 2010, 148, 117–121. [Google Scholar] [CrossRef] [PubMed]
- Tandrup Schmidt, S.; Foged, C.; Smith Korsholm, K.; Rades, T.; Christensen, D. Liposome-based adjuvants for subunit vaccines: Formulation strategies for subunit antigens and immunostimulators. Pharmaceutics 2016, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Mao, H.Q.; Huang, S.K.; Leong, K.W. Oral gene delivery with chitosan–DNA nanoparticles generates immunologic protection in a murine model of peanut allergy. Nat. Med. 1999, 5, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Grönlund, H.; Vrtala, S.; Wiedermann, U.; Dekan, G.; Kraft, D.; Valenta, R.; Van Hage-Hamsten, M. Carbohydrate-based particles: A new adjuvant for allergen-specific immunotherapy. Immunology 2002, 107, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Valenta, R.; Kraft, D. From allergen structure to new forms of allergen-specific immunotherapy. Curr. Opin. Immunol. 2002, 14, 718–727. [Google Scholar] [CrossRef]
- Mohapatra, S.S.; Qazi, M.; Hellermann, G. Immunotherapy for allergies and asthma: Present and future. Curr. Opin. Pharmacol. 2010, 10, 276–288. [Google Scholar] [CrossRef]
- Vrtala, S.; Grote, M.; Ferreira, F.; Susani, M.; Stocker, B.; Kraft, D.; Valenta, R. Humoral immune responses to recombinant tree pollen allergens (Bet v I and Bet v II) in mice: Construction of a live oral allergy vaccine. Int. Arch. Allergy Immunol. 1995, 107, 290–294. [Google Scholar] [CrossRef]
- Bohle, B.; Breitwieser, A.; Zwölfer, B.; Jahn-Schmid, B.; Sára, M.; Sleytr, U.B.; Ebner, C. A novel approach to specific allergy treatment: The recombinant fusion protein of a bacterial cell surface (S-Layer) protein and the major birch pollen allergen Bet v 1 (rSbsC-Bet v 1) Combines reduced allergenicity with immunomodulating capacity. J. Immunol. 2004, 172, 6642–6648. [Google Scholar] [CrossRef]
- Sleytr, U.B.; Messner, P.; Pum, D.; Sára, M. Crystalline bacterial cell surface layers (S Layers): From supramolecular cell structure to biomimetics and nanotechnology. Angew. Chem. Int. Ed. 1999, 38, 1034–1054. [Google Scholar] [CrossRef]
- Itakura, K.; Hirose, T.; Crea, R.; Riggs, A.D.; Heyneker, H.L.; Bolivar, F.; Boyer, H.W. Expression in Escherichia coli of a chemically synthesized gene for the hormone somatostatin. Science 1977, 198, 1056–1063. [Google Scholar] [CrossRef]
- Rosano, G.L.; Ceccarelli, E.A. Recombinant protein expression in Escherichia coli: Advances and challenges. Front. Microbiol. 2014, 5, 172. [Google Scholar] [CrossRef] [PubMed]
- Frew, A.J.; Smith, H.E. Chapter 6—Allergen-specific Immunotherapy. In Middleton’s Allergy Essentials; O’Hehir, R.E., Holgate, S.T., Sheikh, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 133–150. [Google Scholar] [CrossRef]
- Mir, M.A. Chapter 4—Costimulation Immunotherapy in Allergies and Asthma. In Developing Costimulatory Molecules for Immunotherapy of Diseases; Mir, M.A., Ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 131–184. [Google Scholar] [CrossRef]
- Ilaria, P.; Francesca, L.S.; Paola, M. Chapter 5—Therapeutic approaches in allergic diseases. In Immune Rebalancing; Boraschi, D., Penton-Rol, G., Eds.; Academic Press: Cambridge, MA, USA, 2016; pp. 85–103. [Google Scholar] [CrossRef]
- Nowak-Wegrzyn, A.; Bloom, K.A.; Sicherer, S.H.; Shreffler, W.G.; Noone, S.; Wanich, N.; Sampson, H.A. Tolerance to extensively heated milk in children with cow’s milk allergy. J. Allergy Clin. Immunol. 2008, 122, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Canonica, G.W.; Bachert, C.; Hellings, P.; Ryan, D.; Valovirta, E.; Wickman, M.; De Beaumont, O.; Bousquet, J. Allergen Immunotherapy (AIT): A prototype of precision medicine. World Allergy Organ. J. 2015, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Berto, P.; Frati, F.; Incorvaia, C.; Cadario, G.; Contiguglia, R.; Di Gioacchino, M.; Puccinelli, P.; Senna, G.E.; Valle, C. Comparison of costs of sublingual immunotherapy and drug treatment in grass-pollen induced allergy: Results from the SIMAP database study. Curr. Med. Res. Opin. 2008, 24, 261–266. [Google Scholar] [CrossRef]
- Rolinck-Werninghaus, C.; Staden, U.; Mehl, A.; Hamelmann, E.; Beyer, K.; Niggemann, B. Specific oral tolerance induction with food in children: Transient or persistent effect on food allergy? Allergy 2005, 60, 1320–1322. [Google Scholar] [CrossRef]
| Avoid Foods Containing These Ingredients | Egg Proteins Can Be Found in |
|---|---|
|
|
| Allergen | Egg White (%) | Molecular Weight (SDS-PAGE) | Carbohydrate (%) | IgE-Binding Activity (Digestive) | Allergenic Activity | |
|---|---|---|---|---|---|---|
| Heat-Treated | Enzyme-Treated | |||||
| Gal d 1 | 11 | 28 kDa | 25 | Stable | +++ | |
| Gal d 2 | 54 | 45 kDa | 3 | Unstable | ++ | |
| Gal d 3 | 12 | 76.6 kDa | 2.6 | Unstable | + | |
| Gal d 4 | 3.4 | 14.3 kDa | 0 | Unstable | ++ | |
| Advantages | Disadvantages | |
|---|---|---|
| ISAC |
|
|
| ImmunoCAP |
|
|
| Skin Prick Test (SPT) |
|
|
| Strategy | Definition | Allergen Source | Molecule/s |
|---|---|---|---|
| Fragmentation | The cDNA coding for a specific allergen is fragmented into ≥ 2 parts; the fragments may overlap or express individually | Birch pollen | Bet v 1 [115,116] |
| Cow dander | Bos d 2 [117] | ||
| Storage mite (L. destructor) | Lep d 2 [118] | ||
| Timothy grass pollen | PhI p 1 [119] | ||
| House dust mite (D. pteronyssinus) | Der p 2 [120] | ||
| Oligomerization | ≥2 copies of the allergen-encoding cDNA are linked by oligonucleotide spacers with an open reading frame allowing the complete construct to express | Birch pollen | Bet v 1 [121] |
| Mosaics | The cDNA coding for a specific allergen is fragmented into several parts and the fragments are re-joined in an order different to the original sequence. If the sequence parts originate from >1 allergen the resulting protein is regarded as a hybrid mosaic | Birch pollen | Bet v 1 [122] |
| Timothy grass pollen | PhI p 1 [123] PhI p 2 [124] | ||
| Cat | Fel d 1 [125] | ||
| Chimeras/allergen hybrids | Chimeric/hybrid proteins are created by joining the genetic information of ≥2 different proteins such as constructs may contain parts or the complete original proteins | House dust mite (D. pteronyssinus) | Der p 1 [126] Der p 2 [127] |
| Timothy grass pollen | PhI p 1 [128] PhI p 2 [128,129] PhI p 5 [128] PhI p 6 [129] | ||
| Yellowjacket | Ves v 5 [130] | ||
| Paper wasp | Pol a 5 [130] | ||
| Honeybee | Api m 1 [131] Api m 2 [131] Api m 3 [131] | ||
| Japanese cedar | Cry j 1 [132] Cry j 2 [132] | ||
| Point mutations | One or more nucleotide triplets coding for a specific amino acid is/are altered to replace the original amino acid at its exact position by an amino acid with different physicochemical characteristics | Birch pollen | Bet v 1 [133,134] Bet v 4 [135] |
| Brassica rapa pollen | Bra r 1 [136] | ||
| Carp | Cyp c 1 [137] | ||
| P. Judaica pollen | Par j 1 [138] | ||
| Gallus gallus (Ovomucoid) | Gal d 1 [114,139] | ||
| Latex H. brasiliensis | Hev b 6.02 [140,141] Hev b 5 [142] | ||
| House dust mite (D. pteronyssinus) | Der p 2 [143] | ||
| Peanut | Ara h 1-3 [144,145,146] | ||
| Ryegrass pollen | Lol p 5 [147] |
| Recombinant Wildtype Allergens [129,160,161,162,163,164] | Derivatives of Recombinant Allergens [116,165,166,167,168,169,170,171,172,173,174,175,176,177,178] | T-cell Peptides [179,180,181,182,183,184,185] | Peptide Carrier Fusion Proteins [186,187,188,189,190,191,192,193] | |
|---|---|---|---|---|
| Immune response: | ||||
| IgE reactive | + | +/− | − | − |
| T-cell reactive | + | + | + | − |
| Induce protective antibodies | + | + | − | + |
| Possible side effects: | ||||
| IgE | + | − | − | − |
| T-cell mediated | + | + | + | − |
| Target | Type of Modification | Advantage |
|---|---|---|
| Antigen | Chemically modified allergen extracts (allergoids, haptens, PEG) | Allergenic activity is reduced, inducing tolerance and Th1 responses [163,166,196,197] |
| Recombinant allergens and recombinant hypoallergens | Allergen specificity; induces blocking of antibodies and increases safety Immunomodulation occurs with the induction of T-cell tolerance [160,198,199] | |
| T cell peptides | Induction of T-cell tolerance increases allergen specificity and increases safety [200,201,202] | |
| B cell peptides | Induction of blocking antibodies increases allergen specificity and increases safety [150,186] | |
| Mimotopes | Allergen specificity induces the blocking of antibodies in DNA vaccines [203,204,205] | |
| DNA Vaccines | Induction of Th1 response leads to allergen specificity [150,206,207] | |
| Route/mode of administration | Oral/sublingual administration | Safety. The induction of T-cell anergy makes the treatment easy to perform and convenient for patients [208,209,210] |
| Nasal administration | Safe and convenient for patients [211,212] | |
| Adjuvant | Al(OH)3 | Reduces anaphylactic side effects [213,214] |
| CpG, MPL liposomes | CpG reduces allergen activity Overall induces Th1 [215,216] | |
| Chitosan-nanoparticles | Induction of T-cell tolerance [215,217] | |
| Carbohydrate based particles | Ease of production and reduced tissue damage [206,218] | |
| Live vaccines | Induces Th1 response [219,220,221] | |
| Surface layers | Induces Th1 response [222,223] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dona, D.W.; Suphioglu, C. Egg Allergy: Diagnosis and Immunotherapy. Int. J. Mol. Sci. 2020, 21, 5010. https://doi.org/10.3390/ijms21145010
Dona DW, Suphioglu C. Egg Allergy: Diagnosis and Immunotherapy. International Journal of Molecular Sciences. 2020; 21(14):5010. https://doi.org/10.3390/ijms21145010
Chicago/Turabian StyleDona, Dulashi Withanage, and Cenk Suphioglu. 2020. "Egg Allergy: Diagnosis and Immunotherapy" International Journal of Molecular Sciences 21, no. 14: 5010. https://doi.org/10.3390/ijms21145010
APA StyleDona, D. W., & Suphioglu, C. (2020). Egg Allergy: Diagnosis and Immunotherapy. International Journal of Molecular Sciences, 21(14), 5010. https://doi.org/10.3390/ijms21145010






